Abstract
Objective
Assess the frequency and potential clinical impact of periodic leg movements during sleep, with or without arousals, as recorded incidentally from children before and after adenotonsillectomy.
Methods
Children scheduled for adenotonsillectomy for any clinical indications who participated in the Washtenaw County Adenotonsillectomy Cohort II were studied at enrollment and again 6 months thereafter. Assessments included laboratory-based polysomnography, a Multiple Sleep Latency Test (MSLT), parent-completed behavioral rating scales, neuropsychological testing, and psychiatric evaluation.
Results
Participants included 144 children (81 boys) aged 3–12 years. Children generally showed mild to moderate obstructive sleep apnea (median respiratory disturbance index 4.5 [Q1=2.0, Q3=9.5]) at baseline, and 15 subjects (10%) had at least 5 periodic leg movements per hour of sleep (PLMI≥5). After surgery, 21 (15%) of n=137 subjects who had follow-up studies showed PLMI≥5 (p=.0067). Improvements were noted after surgery in the respiratory disturbance index; insomnia symptoms; sleepiness symptoms; mean sleep latencies; hyperactive behavior; memory, learning, attention, and executive functioning on NEPSY assessments; and frequency of Attention-Deficit/Hyperactivity Disorder (DSM-IV criteria). However, PLMI≥5 failed to show associations with worse morbidity in these domains at baseline or follow-up. New appearance of PLMI≥5 after surgery failed to predict worsening of these morbidities (all p>.05), with only one exception (NEPSY) where the magnitude of association was nonetheless negligible. Similar findings emerged for periodic leg movements with arousals (PLMAI≥1).
Conclusion
Periodic leg movements during sleep, with and without arousals, become more common after adenotonsillectomy in children. However, results in this setting did not suggest substantial clinical impact.
Keywords: periodic leg movements, periodic limb movement disorder, sleep apnea, obstructive, polysomnography, child, arousals, cognition, sleepiness
INTRODUCTION
A periodic leg movement index (PLMI, number per hour of sleep) along with a periodic leg movement arousal index (PLMAI) are required data on sleep study reports.1 Yet, the meaning and clinical implications of periodic leg movements during sleep (PLMS) and PLMS with arousals, usually found incidentally during studies ordered for other indications, remain poorly understood and at risk for misinterpretation. In no age group is this lack of existing knowledge more notable than among children.
Although PLMS commonly occur in both adults and children with restless leg syndrome, or Willis Ekbom disease, and in fact can help to confirm this diagnosis, PLMS are most frequently encountered in the sleep studies of patients evaluated for suspected obstructive sleep apnea. Among adults who undergo laboratory-based polysomnography for this indication, the presence of PLMS does not predict increased risk for excessive daytime sleepiness, and in fact -- especially when the PLMS are associated with arousals -- they actually predict slightly less sleepiness than seen in adults without PLMS.2,3 Among children, data on PLMS and sleepiness are not available, but PLMS are reported to be especially common among children with Attention-Deficit/Hyperactivity Disorder (ADHD),4,5 and a high proportion of children with PLMS are in turn reported to have ADHD.6–8 The PLMS in children, perhaps because of associated arousals, have been hypothesized to contribute to ADHD and hyperactive behavior more generally.
If PLMS are associated with hyperactive behavior, then some understanding of the role that PLMS may play in pediatric obstructive sleep apnea would be useful, as would an understanding of how PLMS evolve after adenotonsillectomy for obstructive sleep apnea. Among adults, treatment of obstructive sleep apnea, usually during an initial titration of positive airway pressure, has been reported to increase PLMS,9 decrease PLMS with arousals,10 have no overall effect,11 or have variable effects depending on apnea severity.12 A decrease in PLMS might be hypothesized to occur if PLMS arise in part as a reaction to disrupted sleep, subtle hypopneas, or some other feature provoked by untreated obstructive sleep apnea; conversely, an increase in PLMS conceivably could be understood as a release phenomenon whereby untreated apneic events mask or temporarily relieve an underlying drive for expression of PLMS.12 In children, one study of 44 snoring children awaiting adenotonsillectomy, at baseline and 6 months later, showed only a non-significant increase in PLMI, from a median of 1.7 to 2.3.13 Another study of 25 children with obstructive sleep apnea, before and after treatment (mainly with positive airway pressure), suggested that PLMS diminish after sleep apnea is treated.14 Whether the children were tested during their first titration night, on positive airway pressure, is not clear.
Amidst this uncertainty, we took advantage of data collected in the Washtenaw County Adenotonsillectomy Cohort II15,16 to study the frequency, persistence, and clinical impact of PLMS and PLMS with arousals among children, before and after adenotonsillectomy performed for clinical indications. Although these subjects cannot shed light on the much smaller numbers of patients referred specifically for restless leg syndrome or periodic limb movement disorder, the sample is well-suited for testing several hypotheses. We sought to confirm and quantify the frequency of PLMS among these children at high risk for sleep apnea; assess whether PLMS increase after adenotonsillectomy; and examine whether PLMS or PLMS with arousals may contribute to insomnia, sleepiness, inattention, hyperactive behavior, or ADHD.
METHODS
Overview
Subjects were recruited from the 2 largest otolaryngology groups in Washtenaw County for this institutional review board (IRBMED) approved study. Clinical staff helped to identify families with children, aged 3.0 to 12.9 years, who were scheduled for adenotonsillectomy for any clinical purposes, but as is most often the case17 were not judged to require sleep studies prior to the procedure. Exclusion criteria have been detailed previously.15 They included medical, mental, or physical conditions that might hamper interpretation of EEG or neurobehavioral findings; otolaryngologists’ need for pre-operative polysomnographic data; current or past treatment for obstructive sleep apnea; medical conditions or syndromes that carry high risk for obstructive sleep apnea or daytime sleepiness; or imminent expectation of further surgery or family relocation. For the current analyses, data were included from any Cohort participant who underwent adenotonsillectomy and had complete baseline polysomnography, with a total sleep time > 6 hours, and next-day Multiple Sleep Latency Tests.
A parent signed a written informed consent, and each child assented to participate. Sleep and neurobehavioral assessments were then completed up to three days before the adenotonsillectomy, and again 6 months after surgery. A child psychiatrist, child psychologist, or behavioral developmental pediatrician conducted a structured interview with each family. A full laboratory-based polysomnogram was followed on the next day by a Multiple Sleep Latency Test. Between naps, children underwent neuropsychological testing. A parent completed behavioral rating scales and a standard socioeconomic survey.18 After each of the two major testing visits, children received a $25 gift certificate to a local toy store, and parents received $125 for their time and effort.
Pediatric polysomnography for this study conformed to standard recommendations,19 published after this study began, except that piezoelectric strain gauges rather than inductance plethysmography were used to monitor thoracic and abdominal excursion.15 Esophageal pressure was monitored through a water-filled, 6-French pediatric feeding tube.20,21 Leg movements were monitored in each leg independently, using two surface electromyography leads, one over each anterior tibialis muscle. Multiple Sleep Latency Tests followed standard recommendations,22 with two exceptions to better accommodate these young volunteer research subjects: 4 naps were performed instead of 5, and nap opportunities were lengthened from the adult standard (20 minutes) to 30 minutes.23,24
Scoring
All sleep studies were scored, or in a minority of instances, thoroughly rescored, by a single sleep and electroencephalography-registered technologist with extensive experience in pediatric polysomnography. To prevent bias and minimize any effect of scoring drift with time, all scoring was performed in batches that each contained the pre- and post-surgical studies of several subjects, all de-identified, and without access to other study measures. Sleep staging followed recommended criteria.19 Periodic leg movements and arousals were scored according to standard criteria as well, and were considered to be associated with each other when < 0.5 seconds separated the end of one event and the onset of the other.19 The PLMI and PLMAI were defined as the number of PLMS and PLMS with arousals, respectively, per hour of recorded sleep. Obstructive apneas of 2 or more respiratory cycles in duration, hypopneas, respiratory effort-related arousals (RERAs), and central apneas were scored according to criteria recommended for children by the American Academy of Sleep Medicine (AASM) in 2007.19 The respiratory disturbance index was calculated as the number of the above events per hour of sleep. In Multiple Sleep Latency Tests, the mean sleep latency across all nap opportunities provided an objective measure of daytime sleepiness.25
Subjective Insomnia and Daytime Sleepiness
The Pediatric Sleep Questionnaire was administered to parents before and after adenotonsillectomy. This commonly used instrument contains 3 items that ask about “difficulty falling asleep at night”, “waking up more than twice a night on average”, and “trouble falling back asleep if he or she wakes up at night”. These items have face validity as assessments for difficulties with sleep initiation or maintenance that define the core features of insomnia26 or sleeplessness in children,27 and have proven useful in previous research.28,29 The Pediatric Sleep Questionnaire also contains a well-validated 22-item Sleep-Related Breathing Disorder Scale,30,31 with a 4-item sleepiness subscale that itself has been validated as predictive of both obstructive sleep apnea and Multiple Sleep Latency Test results in children.30,32 The 3-item insomnia subscale and 4-item sleepiness subscale were used as subjective assessments in the current study. The parent, with help from the child when necessary, answered each item as “Yes”, “No”, or “Don’t Know”. One or more endorsed symptom was used to identify children who, in comparison to their peers, were more likely to have subjective insomnia or sleepiness.
Neurobehavioral Outcomes
Standardized and well-validated instruments were employed to identify DSM-IV diagnoses, behavioral problems, and cognitive deficits thought to reflect the most important morbidity in childhood obstructive sleep apnea.33–39 Psychiatric assessments included administration of the Computerized Diagnostic Interview Schedule for Children -- Parent,40–42 and the Children’s Psychiatric Rating Scale.43–45 However, the final categorical diagnostic outcome variable was presence or absence of DSM-IV-defined Attention-Deficit/Hyperactivity Disorder, as determined by the interviewing clinician. This individual was a child psychiatrist, behavioral/developmental pediatrician, or child psychologist and in almost all cases, baseline and follow-up evaluations were performed by the same person.
A composite Behavioral Hyperactivity Index46 (mean 50; standard deviation [s.d] 10) was created from the average of the attention-deficit/hyperactivity disorder T-scores generated by each of two validated parental rating scales: the Conners’ Parent Rating Scales47 and the Child Symptom Inventory-448 (or the Early Childhood Inventory-449 for children between 3 and 5 years). Higher Behavioral Hyperactivity Indices indicated more significant symptoms. Cognitive testing lasted about 2 hours and included the NEPSY,50 a developmental neuropsychological test battery designed for children aged 3–12 years. From the NEPSY, the Memory and Learning Score and the Attention/Executive Functions Score were averaged to generate a composite Cognitive Index (mean 100, SD 15; with higher scores indicating better performance). Children also completed the Continuous Performance Test-Second Edition (CPT-II or Kiddie CPT for children aged 3 or 4 years). The average of the omissions t score, commissions t score, and variability t score, on which higher scores are less desirable and normal means are 50, comprised the Continuous Performance Test outcome variable.
Analyses
Data were summarized as means ± s.d. when they showed normal distributions, and otherwise as medians with first and third quartiles. The primary explanatory variables were PLMI≥5 and PLMAI≥1, as the continuous variables PLMI and PLMAI where highly skewed. The former threshold (PLMI≥5) is thought to have clinical meaning as a cut-point above which leg movements can have clinical impact in children, and periodic limb movement disorder can be diagnosed.26 Although no PLMAI threshold for abnormality is widely accepted, we used PLMAI≥1 to identify children who had more of these events than did their peers. This cut point corresponds to the recommended one-event-per-hour threshold for diagnosis of obstructive sleep apnea in children.26
Outcome measures were provided by the assessments of insomnia, sleepiness, cognition, behavior, and mental health. Changes in each outcome, defined as the postoperative score minus the preoperative score, were tested for significance by paired T tests, Wilcoxon signed rank tests (for non-parametric continuous variables), or McNemar’s tests. To assess the extent to which PLMI or PLMAI were associated with each concurrent neurobehavioral morbidity at baseline and again (separately) at follow-up, logistic regression models were used, except when outcome measures were normally distributed, in which case general linear models were used. A similar approach was used to compare changes in PLMI or PLMAI to changes in neurobehavioral outcomes. All regression models were adjusted for the following potential confounds, or changes in these variables where relevant: age, gender, body mass index z-score (BMI), socioeconomic level, and respiratory disturbance index. The level of significance was set at p < .05. Results were not adjusted for multiple comparisons, to maintain sensitivity in this novel effort to detect any possible associations between PLMS and neurobehavioral outcomes within a cohort not referred for specific concern about restless legs, periodic leg movements, or mental health problems. Although PLMS are described at ages as young as 15 months,51 and periodic limb movement disorder is recognized as a useful diagnostic alternative to restless legs syndrome in children under 6 years old,52 fewer data are available to associate PLMS with neurobehavioral outcomes among these young children. Therefore, regression models were repeated after excluding children less than 6 years-old from the analyses. All analyses were performed with SAS 9.2 (SAS Institute, Cary, NC).
RESULTS
Subjects and measures at baseline and follow-up
A total of 899 families with children scheduled for adenotonsillectomy were approached about this research during a visit to their otolaryngologist’s office. Among these, 147 families consented to participate and were evaluated before adenotonsillectomy. Reasons that families cited when they declined to participate were most often lack of time, desire to avoid any additional stress around the time of surgery, or lack of interest in research. Among the 147 participating children, 144 underwent nocturnal polysomnography successfully with at least 6 hours of recorded sleep, and a Multiple Sleep Latency Test on the next day. These children provided the data for the analyses reported below. Among the 144, 137 (95%) were re-assessed about 6 months after surgery, 7.2 ± 0.9 months after the initial testing.
Demographic and polysomnographic variables at baseline and follow-up are shown in Table 1. In general, the sample reflected mild to moderate pediatric obstructive sleep apnea: 126 (88%) received adenotonsillectomy for clinical indications that included suspected sleep-disordered breathing, and the baseline respiratory disturbance index was at least 1.0 for 132 (92%). After adenotonsillectomy, obstructive sleep apnea improved significantly, and body mass index z scores increased as anticipated.53 The sleep efficiency, arousal index, and sleep staging showed statistically significant improvements in this sizeable sample, but the magnitudes of the changes were generally small and of questionable clinical significance.
Table 1.
Demographic and polysomnographic data for participants
| Pre-Adenotonsillectomy N=144 No. (%), Median [Q1, Q3], or Mean ± SD |
Post-Adenotonsillectomy N=137 No. (%), Median [Q1, Q3], or Mean ± SD |
P * | |
|---|---|---|---|
| Age (yrs) | 7.1 ± 2.5 | 7.8 ± 2.5 | NA |
| Male gender | 81 (56%) | 76 (55%) | NA |
| Body Mass Index z-score | 0.63 ±1.31 | 0.84 ±1.05 | <.0001 a |
| Socioeconomic class (at baseline only) | 2.0 [2.0, 3.0] | NA | NA |
| Total Recording Time (TRT, min) | 601 ± 38 | 607 ± 42 | .17 a |
| Total Sleep Time (TST, min) | 526 ± 51 | 540 ± 54 | .0061 a |
| Sleep Efficiency (TST*100/TRT) | 88 ± 7 | 89 ± 7 | .025 a |
| Arousal index (events/hr of sleep) | 11.1 [8.5, 14.4] | 9.5 [7.8, 11.2] | <.0001 b |
| % Stage N1 | 12 ± 8 | 10 ± 3 | .0001 a |
| % Stage N2 | 44 ± 7 | 47 ± 6 | <.0001 a |
| % Stage N3 | 25 ± 5 | 23 ± 6 | <.0001 a |
| % Stage R | 19 ± 5 | 21 ± 4 | .0028 a |
| Respiratory disturbance index (events/hr of sleep) | 4.5 [2.0, 9.5] | 1.5 [0.8, 2.6] | <.0001 b |
| Minimum oxygen saturation (%) | 92 [89, 94] | 93 [91, 94] | <.0001 b |
| Periodic Leg Movement Index (PLMI) | 0.0 [0.0, 1.4] | 0.4 [0.0, 2.0] | .010 b |
| Periodic Leg Movement Arousal Index (PLMAI) | 0.0 [0.0, 0.1] | 0.0 [0.0, 0.4] | .0014 b |
| Periodic Leg Movement Index ≥5 | 15 (10%) | 21 (15%) | .0067 c |
| Periodic Leg Movement Arousal Index ≥1 | 10 (7%) | 18 (13%) | .0075 c |
for change after adenotonsillectomy, among those subjects who had follow-up assessments
paired T test
Wilcoxon signed rank test
McNemar’s Test
In contrast to obstructive sleep apnea, PLMS became somewhat more common at follow up. After surgery, the proportion of subjects who had a PLMI≥5 increased by one-half, from 10% to 15% of the sample (p = .0067). Among 12 subjects who had PLMI≥5 before AT, 11 (92%) still met this criterion after AT. Among 125 subjects without PLMI≥5 before AT, 10 (8%) newly met this criterion for the first time after AT (calculation limited to those subjects with follow-up assessments). Among 8 subjects who had PLMAI≥1 before AT, 6 (75%) still met this criterion after AT. Among 129 subjects without PLMAI≥1 before AT, 12 children (9%) met this criterion for the first time after AT. Some number of PLMS were scored (i.e., PLMI > 0) on the baseline studies of 57 (40%) of the 144 subjects, and some number of PLMS with arousals were scored for 45 (31%). After adenotonsillectomy, PLMS were noted on the baseline studies of 72 (53%) of the 137 subjects, and some number of PLMS with arousals were scored for 56 (41%). Overall, the PLMI and PLMAI before AT showed correlations of moderate strength with the PLMI and PLMAI after AT (rho = .43, p <.0001, and rho = .33, p <.0001 respectively).
Outcome measures at baseline and follow-up are shown in Table 2. Missing data points, where noted, were mainly attributable to inability of the youngest children to perform the Continuous Performance Test. As with the sleep measures, outcome measures including those for insomnia symptoms, subjective sleepiness, objective sleepiness, hyperactive behavior, executive dysfunction, inattention, and diagnosis of Attention-Deficit/Hyperactivity Disorder all improved after adenotonsillectomy, with the sole exception of inattention as reflected by the computerized continuous performance task. The magnitude of these changes ranged from small, for example on the Multiple Sleep Latency Test, to robust, as reflected by subjective sleepiness, insomnia symptoms, executive dysfunction, and frequency of Attention-Deficit/Hyperactivity Disorder.
Table 2.
Outcome measures – insomnia symptoms, subjective sleepiness, objective sleepiness, hyperactive behavior, executive dysfunction, inattention, and diagnosis of Attention-Deficit/Hyperactivity Disorder – before and after adenotonsillectomy.
| Pre-Adenotonsillectomy | Post-Adenotonsillectomy | P * | |||
|---|---|---|---|---|---|
| N | No. Positive (%), Median [Q1, Q3], or Mean ± SD | N | No. Positive (%), Median [Q1, Q3], or Mean ± SD | ||
|
| |||||
| Insomnia Scale (one or more of 3 symptoms) | 144 | 73 (51%) | 137 | 34 (25%) | <.0001 c |
|
| |||||
| Sleepiness Scale (Proportion with one or more of 4 symptoms) | 144 | 117 (81%) | 137 | 53 (39%) | <.0001 c |
|
| |||||
| Mean Sleep Latency on Multiple Sleep Latency Test (min) | 144 | 25.7 [22.2, 29.5] | 137 | 26.5 [23.8, 29.9] | .022 b |
|
| |||||
| Behavioral Hyperactivity Index (BHI) | 137 | 55.0 [48.0, 66.5] | 135 | 50.0 [46.0, 59.5] | <.0001 b |
|
| |||||
| NEPSY Cognitive Index (CI) | 137 | 101.4 ± 13.0 | 135 | 109.9 ± 12.1 | <.0001 a |
|
| |||||
| Continuous Performance Test | 124 | 56.6 ± 8.6 | 112 | 55.5 ± 9.2 | .12 a |
|
| |||||
| Attention-Deficit/Hyperactivity Disorder (Proportion with diagnosis) | 144 | 58 (40%) | 137 | 31 (23%) | <.0001 c |
for change after adenotonsillectomy, among those subjects who had follow-up assessments
paired T test
Wilcoxon signed rank test
McNemar’s Test
Are PLMS or PLMS with arousals associated with adverse concurrent morbidity?
Fully adjusted regression models showed that at baseline, PLMS (PLMI≥5) were associated (inversely) with one or more sleepiness symptoms (OR = .29, 95% CI [.09, .98]), but not with any of several other comorbidities: presence of one or more insomnia symptom (OR = .47 [.14, 1.51]), low mean sleep latency (bottom quartile, ≤ 22 minutes, OR = 1.29 [.36, 4.66]), high Behavioral Hyperactivity Index (≥ 65, OR =.92 [.25, 3.41]), lower scores on the NEPSY Cognitive Index (beta = .19, se = 3.67, t = .05, p = .96), worse scores on the Continuous Performance Test (beta = −2.01, se = 2.96, t = −.68, p = .50), or presence of ADHD (OR = .95 [.29, 3.08]). For an unadjusted example, 7 among 58 children with ADHD (12%) had PLMI≥5, but so did 8 among 86 children (9%) without ADHD. Similarly, in fully adjusted regression models PLMS with arousals (PLMAI≥1) were associated (inversely) with one or more sleepiness symptoms (OR = .12 [.03, .51]), but not with one or more insomnia symptoms (OR = .35, [.08, 1.51]), low mean sleep latency (bottom quartile, ≤22 minutes, OR = 1.50 [.34, 6.63]), high Behavioral Hyperactivity Index (≥65, OR = .44 [.08, 2.39]), lower NEPSY Cognitive Index (beta = −.90, se = 4.21, t = −.21, p = .83), poor scores on the Continuous Performance Test (beta = −4.15, se = 3.88, t = −1.07, p = .29), or presence of ADHD (OR = .53 [.13, 2.20]). Associations were similarly nonsignificant, except for those with sleepiness symptoms, when PLMI and PLMAI were tested as continuous variables, instead of their dichotomized analogues, or when the analyses were confined to those subjects (n=90) aged 6 or more years.
After AT, neither PLMI≥5 nor PLMAI≥1 were associated with any of the above measures of concurrent morbidity (all p > .10). Similarly, after AT, neither PLMI nor PLMAI (as continuous variables) were associated with any concurrent morbidity measure (all p > .10). Results were no different when analyses were confined to subjects aged 6 or more years.
Are changes in PLMS or PLMS with arousals associated with changes in comorbidities after AT?
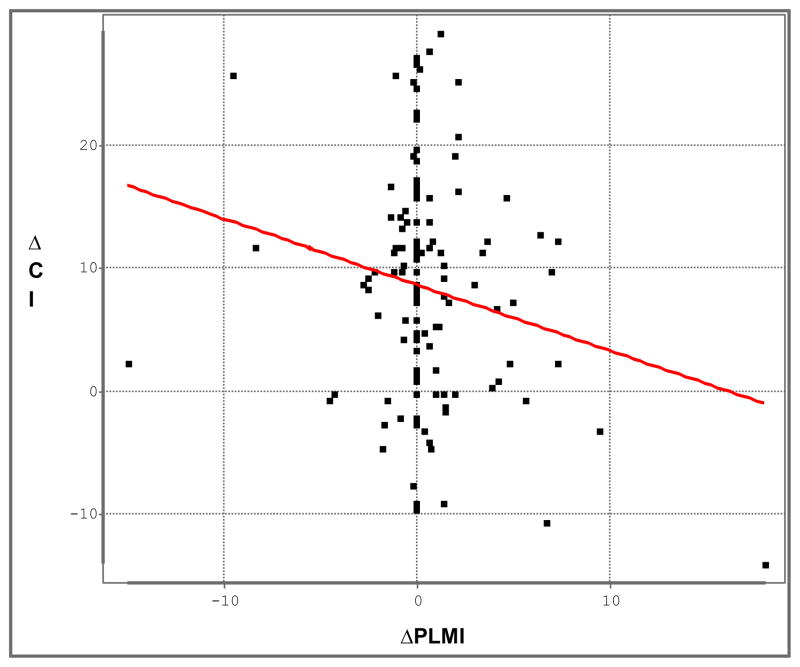
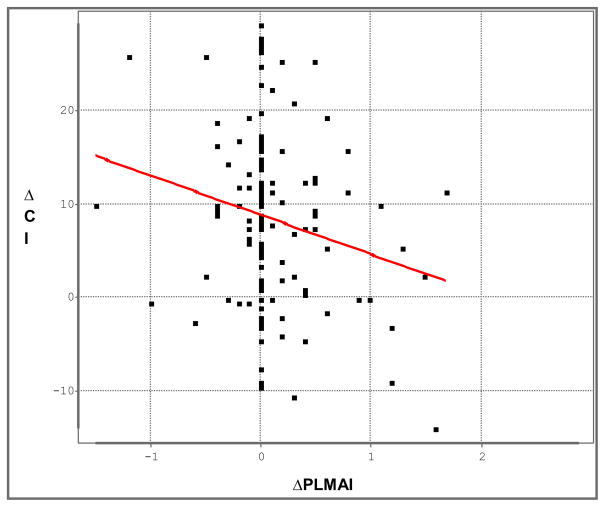
In fully adjusted regression models, children who had PLMI < 5 before AT but PLMI≥5 after AT (n=10) in comparison to the other children studied before and after AT (n=127) showed no significantly greater worsening of insomnia symptoms, sleepiness symptoms, mean sleep latency on MSLTs, Behavioral Hyperactivity Index, Continuous Performance Test scores, or tendency to resolve an initial diagnosis of ADHD (Table 3). Results were similar when the analysis was confined to n=5 vs. n=81 such subjects aged 6 years or older, though some of the models may not have been reliable with such small numbers of subjects in the first group. Children who had PLMI < 5 before AT but PLMI≥5 after AT, in comparison to other children, did experience less improvement on the NEPSY Cognitive Index. Parallel results emerged when PLMAI≥1, newly detected for n=12 children after surgery, was tested in place of PLMI≥5 (Table 4). Results were similar when pre-to-post AT changes in PLMI and PLMAI were used as continuous rather than dichotomous variables in regression models (data not shown). Figures 1 and 2 do show that the changes in NEPSY scores were associated with PLMI and PLMAI, respectively, but in both cases a few outliers with extreme changes in PLMI or PLMAI appeared likely to drive the weak associations that nonetheless achieved significance.
Table 3.
Regression of changes in sleep and neurobehavioral outcome measures, after adenotonsillectomy, on new emergence of periodic leg movements (PLMI ≥ 5) *
| Outcome Variable | Overall Model | Emergence of PLMI ≥ 5 (within context of overall model) | ||||
|---|---|---|---|---|---|---|
| R2 | p | Beta | S.E. | T or Wald Chi- Square | p | |
| Insomnia Scale (one or more of 3 symptoms) | .058 | .243 | −.19 | .10 | −1.92 | .057 |
| Sleepiness Scale (Proportion with one or more of 4 symptoms) | .052 | .313 | .19 | .11 | 1.71 | .090 |
| Mean Sleep Latency on Multiple Sleep Latency Test (min) | .083 | .076 | −.80 | 1.51 | −.53 | .598 |
| Behavioral Hyperactivity Index (BHI) | .070 | .180 | −1.20 | 2.86 | −.42 | .676 |
| NEPSY Cognitive Index (CI) | .074 | .145 | −7.64 | 3.36 | −2.27 | .025 |
| Continuous Performance Test | .033 | .749 | 2.04 | 2.89 | .71 | .482 |
| Attention-Deficit/Hyperactivity Disorder (Proportion with diagnosis) | .022 | .820 | .62 | .77 | .66 | .417 |
Results of general linear models, or a logistic regression model (for Attention-Deficit/Hyperactivity Disorder), after adjustment for age, gender, change in body mass index z-score, socioeconomic status, and change in respiratory disturbance index.
Table 4.
Regression of changes in sleep and neurobehavioral outcome measures, after adenotonsillectomy, on new emergence of periodic leg movements with arousals (PLMAI ≥ 1) *
| Outcome Variable | Overall Model | Emergence of PLMAI ≥ 1 (within context of overall model) | ||||
|---|---|---|---|---|---|---|
| R2 | p | Beta | S.E. | T or Wald Chi- Square | p | |
| Insomnia Scale (one or more of 3 symptoms) | .035 | .591 | −.05 | .09 | −.61 | .541 |
| Sleepiness Scale (Proportion with one or more of 4 symptoms) | .039 | .519 | .10 | .10 | 1.01 | .313 |
| Mean Sleep Latency on Multiple Sleep Latency Test (min) | .082 | .081 | −.40 | 1.36 | −.30 | .768 |
| Behavioral Hyperactivity Index (BHI) | .070 | .179 | −1.13 | 2.64 | −.43 | .669 |
| NEPSY Cognitive Index (CI) | .093 | .059 | −8.70 | 3.10 | −2.80 | .006 |
| Continuous Performance Test | .031 | .775 | −1.41 | 2.56 | −.55 | .582 |
| Attention-Deficit/Hyperactivity Disorder (Proportion with diagnosis) | .029 | .681 | .91 | .67 | 1.83 | .176 |
Results of general linear models, or a logistic regression model (for Attention-Deficit/Hyperactivity Disorder), after adjustment for age, gender, change in body mass index z-score, socioeconomic status, and change in respiratory disturbance index.
Figure 1.
The change in the NEPSY Cognitive Index (ΔCI) after adenotonsillectomy is plotted against the change in the periodic leg movement index (ΔPLMI). R2 = .037, p = .030. The few outliers with more extreme changes in PLMI were likely to be influential, as a non-parametric Spearman correlation coefficient did not confirm a significant association (rho = −.11, p = .23).
Figure 2.
The change in the NEPSY Cognitive Index (ΔCI) after adenotonsillectomy is plotted against the change in the periodic leg movement with arousal index (ΔPLMAI). R2 = .044, p = .017. The few outliers with more extreme changes in PLMAI were likely to be influential, as a non-parametric Spearman correlation coefficient did not confirm a significant association (rho = −.14, p = .11).
DISCUSSION
This study of 144 children scheduled for adenotonsillectomy combined gold-standard sleep laboratory assessments with intensive evaluations of neurobehavioral morbidity to show that periodic leg movements during sleep, with or without arousal, are fairly common at baseline and more common after surgery, but appear unlikely at either time point to have a strong clinical impact. Among the 137 children studied both before and after adenotonsillectomy, the frequency of PLMS sufficient to meet polysomnographic criteria for periodic limb movement disorder increased by 50%. However, neither PLMS nor PLMS with arousals predicted worse concurrent morbidity, with respect to insomnia symptoms, sleepiness symptoms, objective sleepiness on an MSLT, parental ratings for hyperactivity, an objective test of attention, or qualification for diagnosis of ADHD. Analyses of changes in PLMS or PLMS with arousals, versus changes in several sleep complaints and neurobehavioral measures, showed no statistically significant associations (with only one possible exception) that might support biological cause-and-effect relationships between the leg movements and the outcomes studied. These findings are especially interesting in the context of previous literature on PLMS among patients with sleep disordered breathing, and patients studied before and after treatment for sleep apnea, in the context of ADHD, and in childhood in particular.
We previously reported, among 1,124 referred adults with suspected or confirmed obstructive sleep apnea, that on full polysomnography 24% had PLMI≥5.3 Although referred children are thought to have PLMS less commonly, figures have ranged widely, including for example 6% of 591 children,54 10% of 101 children,55 and 23% of 252 children.51 Children with sickle cell disease, narcolepsy, and some other specific disorders may have PLMS more frequently than the snoring children commonly referred to sleep disorders centers.56,57 Our finding of a 10% frequency among children scheduled for adenotonsillectomy, if generalizable to the 500,000 adenotonsillectomies performed annually in the US,58 suggest that about 50,000 of these children would demonstrate PLMI≥5 preoperatively, and 7 months later 75,000 may have PLMS at a frequency thought to have potential impact.
Therefore, a key question is whether PLMI≥5 is in fact clinically significant. Pediatric obstructive sleep apnea is associated with a wide range of adverse outcomes.59 Although adenotonsillectomy almost always improves sleep apnea, baseline morbidity does not always resolve.46 Investigators have hypothesized that to the extent PLMS may be more common after sleep apnea is treated, they could explain persistent neurobehavioral morbidity.60 However, our findings now suggest that PLMS in this setting are unlikely to have these consequences. At baseline, no outcome measures were worse in association with PLMS or PLMS with arousals. Conversely, the PLMS and especially PLMS with arousals did predict a lower rather than higher likelihood of sleepiness, in parallel with previous findings from adults.2,3 When PLMS occur, less sleepy individuals may be more easily aroused. Arousals more often precede than follow PLMS,61 and can occur exactly when expected within a series of PLMS even when a leg movement fails to materialize.62 Some have argued that the clinical significance of PLMS outside the context of specific neurological conditions associated with PLMS – mainly those conditions that involve abnormal dopaminergic transmission -- may be negligible.63,64
Our findings on follow-up after adenotonsillectomy further support this conclusion. Although the frequency of children with PLMI≥5 increased by 50%, still no association with concurrent neurobehavioral morbidity or sleepiness could be identified. Absence of relationships between changes in PLMS or PLMS with arousals and changes in neurobehavioral morbidity failed to support a significant biological cause-and-effect relationship between the leg movements and the putative outcomes. Tables 3 and 4 show that newly developed PLMS or PLMS with arousals were associated with only a few scattered changes in outcome measures after surgery, in each case to an extent that was clinically negligible in magnitude.
Previous studies that have focused on children referred with ADHD, or periodic limb movement disorder (PLMI≥5 with relevant sleep symptoms), have emphasized close connections between these conditions.4–8 However, findings may have been influenced by referral bias, as patients with both sleep and behavioral problems may be referred to specialists known for expertise at their intersection. Current findings also contrast with our own previous data, for example from surveys administered in general pediatric waiting rooms,65 or from a large sleep laboratory where referrals are not linked to clinicians with any specific interest.66 The present study, with more in-depth, gold standard assessments of neurobehavioral outcomes, and could not confirm similar associations between PLMS and behavior.
Our current data appear to be the first to show an increase in PLMS, well after adenostonsillectomy. Previous studies of PLMS among adults treated for OSA have shown variable results and, as noted above, were usually performed during an initial night of continuous positive airway pressure (CPAP) titration, before habituation to nightly therapy or more permanent treatment by surgery. One study of 14 adults with PLMI≥5 on repeat polysomnography, several months after home use of CPAP, did show that the PLMI had increased from baseline.9
Exactly why PLMS would increase after treatment for obstructive sleep apnea remains uncertain. Some authors have speculated that treatment of sleep apnea might unmask occult PLMS.12 A leg movement that could qualify as a PLM, especially when associated with an arousal, is often followed by transient depression in respiration that could qualify as an hypopnea; in such cases, scoring rules require that the hypopnea, rather than the putative PLM, be scored.19 Thus elimination of hypopneas could lead to recognition that underlying PLMS are present. However, we were unable to replicate the previous observation12 that the baseline respiratory disturbance index correlated with the PLMI at follow-up (data not shown). Others have speculated that CPAP may induce PLMS in children.60 Although this remains possible, it does not explain why our subjects would have increased PLMS 6 months after adenotonsillectomy.
Strengths of the current study include its novel focus on PLMS in the setting in which they are most often discovered, among children at risk for obstructive sleep apnea. This study capitalized on gold-standard sleep laboratory measures; longitudinal if not randomized data; a high rate of successful follow-up at 6 months; well-validated, rigorous neurobehavioral assessments; recording of PLMS in individual legs; a stable recording rather than a CPAP titration with changing conditions; and follow-up well after treatment for obstructive sleep apnea has been established. The sample size was sizeable. Although only a minority of children had PLMI≥5 or PLMAI≥1, the sample was sufficient to replicate previous suggestions that PLMS can be weakly associated with fewer sleepiness complaints.2,3 Numbers of subjects with new, postoperative PLMS or PLMS with arousals were sufficient to detect an association, with NEPSY changes, that explained <10% of their variance. Other associations that might have reached significance in an even larger sample might similarly have failed to explain a clinically meaningful proportion of the relevant outcomes.
Limitations of this study do include the absence of more rigorous, recently suggested approaches to PLMS scoring67 that might have helped to distinguish PLMS related to restless leg syndrome from PLMS arising for other reasons. We recorded PLMS for only one night, before and after surgery. Although the one-night protocol replicated data that can be obtained in clinical practice, night-to-night variability in PLMS could have limited our ability to identify correlates with neurobehavioral outcomes. Use of esophageal pressure monitoring conceivably could have disrupted sleep and fostered PLMS. However, previously published data from our laboratory, on 290 adults and 20 children, suggested no clinically significant effects on scored sleep and no statistically significant effects on PLMS.68 In the present study, we did not assess outcomes other than cognition, behavior, sleepiness, and symptoms. Periodic leg movements can be associated with high blood pressure69 and perhaps other outcomes that were not assessed. An observational study design cannot prove that adenotonsillectomy actually causes increased PLMS. Finally, only 16% of families approached about this study agreed to participate. Although this level of participation resembles that found in many clinical studies that demand significant time and effort from subjects and their families, findings from this study, especially where they concern observed frequencies rather than associations or intra-subject changes, may not be generalizable to all children who undergo adenotonsillectomy.
Nonetheless, current results do suggest several implications for clinical practice. The frequency of PLMS before and especially after treatment for obstructive sleep apnea raises the question of whether laboratory-based sleep studies to detect PLMS should be considered more often than they are now. However, at the same time, this study provides little if any evidence that for children in this setting, PLMS are likely to have detrimental impact on cognition, behavior, sleep, or sleepiness. Clinicians who see patients back with persistent sleep-related concerns after adenotonsillectomy should consider PLMS. However, further research will be needed to clarify exactly what adverse consequences, if any, arise from these polysomnographic findings.
Supplementary Material
Highlights.
Periodic leg movements during sleep (PLMS) are common in children who have T&A.
PLMS among such children become more common after adenotonsillectomy.
These PLMS are unlikely to affect sleepiness, insomnia, cognition, or behavior.
Acknowledgments
Support: National Institutes of Health (HL080941, HL105999)
The investigators are grateful to the patients and families who volunteered considerable time and effort, and to the surgeons and staff who identified these participants at the clinics of the University of Michigan Division of Pediatric Otolaryngology and the Michigan Otolaryngology Surgery Associates. Surgeons who assisted at the University of Michigan included Drs. Charles Koopmann, Marci Lesperance, Glenn Green, Marc Thorne, and Susan Garetz. Surgeons who assisted at the Michigan Otolaryngology Surgery Associates included Drs. Thomas Weimert, Ronald Bogdasarian, Paul Hoff, and Laurence Ho.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Berry RB, Brooks R, Gamaldo CE, et al. The AASM Manual for the Scoring of Sleep and Associated Events: Rules, Terminology and TEchnical Specifications. Darien, Illinois: American Academy of Sleep Medicine; 2012. [Google Scholar]
- 2.Mendelson WB. Are periodic leg movements associated with clinical sleep disturbance? Sleep. 1996;19:219–23. doi: 10.1093/sleep/19.3.219. [DOI] [PubMed] [Google Scholar]
- 3.Chervin RD. Periodic leg movements and sleepiness in patients evaluated for sleep-disordered breathing. American Journal of Respiratory & Critical Care Medicine. 2001;164:1454–8. doi: 10.1164/ajrccm.164.8.2011062. [DOI] [PubMed] [Google Scholar]
- 4.Picchietti DL, England SJ, Walters AS, Willis K, Verrico T. Periodic limb movement disorder and restless legs syndrome in children with attention-deficit-hyperactivity disorder. Journal of Child Neurology. 1998;13:588–94. doi: 10.1177/088307389801301202. [DOI] [PubMed] [Google Scholar]
- 5.Picchietti DL, Underwood DJ, Farris WA, et al. Further studies on periodic limb movement disorder and restless legs syndrome in children with attention-deficit hyperactivity disorder. Movement Disorders. 1999;14:1000–7. doi: 10.1002/1531-8257(199911)14:6<1000::aid-mds1014>3.0.co;2-p. [DOI] [PubMed] [Google Scholar]
- 6.Picchietti DL, Walters AS. Restless legs syndrome and periodic limb movement disorder in children and adolescents: comorbidity with attention-deficit hyperactivity disorder. Child and Adolescent Psychiatry Clinics of North America. 1996;5:729–40. [Google Scholar]
- 7.Picchietti DL, Walters AS. Severe periodic limb movement disorder in childhood and adolescence. Sleep Research. 1996;25:333. doi: 10.1093/sleep/22.3.297. [DOI] [PubMed] [Google Scholar]
- 8.Picchietti DL, Walters AS. Moderate to severe periodic limb movement disorder in childhood and adolescence. Sleep. 1999;22:297–300. doi: 10.1093/sleep/22.3.297. [DOI] [PubMed] [Google Scholar]
- 9.Fry JM, DiPhillipo MA, Pressman MR. Periodic leg movements in sleep following treatment of obstructive sleep apnea with nasal continuous positive airway pressure. Chest. 1989;96:89–91. doi: 10.1378/chest.96.1.89. [DOI] [PubMed] [Google Scholar]
- 10.Yamashiro Y, Kryger MH. Acute effect of nasal CPAP on periodic limb movements associated with breathing disorders during sleep. Sleep. 1994;17:172–5. doi: 10.1093/sleep/17.2.172. [DOI] [PubMed] [Google Scholar]
- 11.Hedli LC, Christos P, Krieger AC. Unmasking of periodic limb movements with the resolution of obstructive sleep apnea during continuous positive airway pressure application. J Clin Neurophysiol. 2012;29:339–44. doi: 10.1097/WNP.0b013e3182624567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Baran AS, Richert AC, Douglass AB, May W, Ansarin K. Change in periodic limb movement index during treatment of obstructive sleep apnea with continuous positive airway pressure. Sleep. 2003;26:717–20. doi: 10.1093/sleep/26.6.717. [DOI] [PubMed] [Google Scholar]
- 13.Kohler MJ, Lushington K, van den Heuvel CJ, Martin J, Pamula Y, Kennedy D. Adenotonsillectomy and neurocognitive deficits in children with Sleep Disordered Breathing. PLoS One. 2009;4:e7343. doi: 10.1371/journal.pone.0007343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Scholle S, Scholle HC, Zwacka G. Periodic leg movements and sleep-disordered breathing in children. Somnologie. 2001;5:153–8. [Google Scholar]
- 15.Chervin RD, Ruzicka DL, Hoban TF, et al. Esophageal Pressures, Polysomnography, and Neurobehavioral Outcomes of Adenotonsillectomy in Children. Chest. 2012 doi: 10.1378/chest.11-2456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Chervin RD, Garetz SL, Ruzicka DL, et al. Do respiratory cycle-related EEG changes or arousals from sleep predict neurobehavioral deficits and response to adenotonsillectomy in children? Journal of Clinical Sleep Medicine. doi: 10.5664/jcsm.3968. (in press) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Weatherly RA, Mai EF, Ruzicka DL, Chervin RD. Identification and evaluation of obstructive sleep apnea prior to adenotonsillectomy in children: a survey of practice patterns. Sleep Medicine. 2003;4:297–307. doi: 10.1016/s1389-9457(03)00100-x. [DOI] [PubMed] [Google Scholar]
- 18.Hollingshead AB. Two Factor Index of Social Position. New Haven: Yale Press; 1965. [Google Scholar]
- 19.Iber C, Ancoli-Israel S, Chesson A, Quan SF for the American Academy of Sleep M. The AASM Manual for the Scoring of Sleep and Associated Events: Rules, Terminology and Technical Specifications. Westchester, Illinois: American Academy of Sleep Medicine; 2007. [Google Scholar]
- 20.Kushida CA, Giacomini A, Lee MK, Guilleminault C, Dement WC. Technical protocol for the use of esophageal manometry in the diagnosis of sleep-related breathing disorders. Sleep Medicine. 2002;3:163–73. doi: 10.1016/s1389-9457(01)00143-5. [DOI] [PubMed] [Google Scholar]
- 21.Chervin RD, Ruzicka DL, Wiebelhaus JL, et al. Tolerance of esophageal pressure monitoring during polysomnography in children. Sleep. 2003;26:1022–6. doi: 10.1093/sleep/26.8.1022. [DOI] [PubMed] [Google Scholar]
- 22.Littner MR, Kushida C, Wise M, et al. Practice Parameters for clinical use of the multiple sleep latency test and the maintenance of wakefulness test - An American Academy of Sleep Medicine Report - Standards of practice committee of the American Academy of Sleep Medicine. Sleep. 2005;28:113–21. doi: 10.1093/sleep/28.1.113. [DOI] [PubMed] [Google Scholar]
- 23.Gozal D, Wang M, Pope DW., Jr Objective sleepiness measures in pediatric obstructive sleep apnea. Pediatrics. 2001;108:693–7. doi: 10.1542/peds.108.3.693. [DOI] [PubMed] [Google Scholar]
- 24.Golan N, Suraya S, Pillar G. Daytime sleepiness in children with attention deficit hyperactive disorder. Sleep. 2003;26 (Abstract Supplement):A127-A. [Google Scholar]
- 25.Hoban TF, Chervin RD. Assessment of sleepiness in children. Seminars in Pediatric Neurology. 2001;8:216–28. doi: 10.1053/spen.2001.29043. [DOI] [PubMed] [Google Scholar]
- 26.American Academy of Sleep M. Diagnostic and Coding Manual. 2. Westchester, Illinois: American Academy of Sleep Medicine; 2005. International Classification of Sleep Disorders. [Google Scholar]
- 27.Sheldon SH. Disorders of initiating and maintaining sleep. In: Sheldon SH, Ferber R, Kryger MH, editors. Principles and Practice of Pediatric Sleep Medicine. Elsevier Saunders; 2005. pp. 127–60. [Google Scholar]
- 28.Archbold KH, Pituch KJ, Panahi P, Chervin RD. Symptoms of sleep disturbances among children at two general pediatric clinics. Journal of Pediatrics. 2002;140:97–102. doi: 10.1067/mpd.2002.119990. [DOI] [PubMed] [Google Scholar]
- 29.Chervin RD, Archbold KH, Panahi P, Pituch KJ. Sleep problems seldom addressed at two general pediatric clinics. Pediatrics. 2001;107:1375–80. doi: 10.1542/peds.107.6.1375. [DOI] [PubMed] [Google Scholar]
- 30.Chervin RD, Hedger KM, Dillon JE, Pituch KJ. Pediatric Sleep Questionnaire (PSQ): validity and reliability of scales for sleep-disordered breathing, snoring, sleepiness, and behavioral problems. Sleep Medicine. 2000;1:21–32. doi: 10.1016/s1389-9457(99)00009-x. [DOI] [PubMed] [Google Scholar]
- 31.Chervin RD, Weatherly RA, Garetz SL, et al. Pediatric sleep questionnaire: Prediction of sleep apnea and outcomes. Archives of Otolaryngology-Head & Neck Surgery. 2007;133:216–22. doi: 10.1001/archotol.133.3.216. [DOI] [PubMed] [Google Scholar]
- 32.Chervin RD, Weatherly RA, Ruzicka DL, et al. Subjective sleepiness and polysomnographic correlates in children scheduled for adenotonsillectomy vs. other surgical care. Sleep. 2006;29:495–503. [PMC free article] [PubMed] [Google Scholar]
- 33.Guilleminault C, Korobkin R, Winkle R. A review of 50 children with obstructive sleep apnea syndrome. Lung. 1981;159:275–87. doi: 10.1007/BF02713925. [DOI] [PubMed] [Google Scholar]
- 34.Guilleminault C, Winkle R, Korobkin R, Simmons B. Children and nocturnal snoring-- evaluation of the effects of sleep related respiratory resistive load and daytime functioning. European Journal of Pediatrics. 1982;139:165–71. doi: 10.1007/BF01377349. [DOI] [PubMed] [Google Scholar]
- 35.Ali NJ, Pitson DJ, Stradling JR. Snoring, sleep disturbance, and behaviour in 4–5 year olds. Archives of Disease in Childhood. 1993;68:360–6. doi: 10.1136/adc.68.3.360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hansen DE, Vandenberg B. Neuropsychological features and differential diagnosis of sleep apnea syndrome in children. Journal of Clinical Child Psychology. 1997;26:304–10. doi: 10.1207/s15374424jccp2603_9. [DOI] [PubMed] [Google Scholar]
- 37.Ali NJ, Pitson D, Stradling JR. Sleep disordered breathing: effects of adenotonsillectomy on behaviour and psychological functioning. European Journal of Pediatrics. 1996;155:56–62. doi: 10.1007/BF02115629. [DOI] [PubMed] [Google Scholar]
- 38.Weissbluth M, Davis AT, Poncher J, Reiff J. Signs of airway obstruction during sleep and behavioral, developmental, and academic problems. Developmental and Behavioral Pediatrics. 1983;1983:119–21. doi: 10.1097/00004703-198306000-00008. [DOI] [PubMed] [Google Scholar]
- 39.Rhodes SK, Shimoda KC, Wald LR, et al. Neurocognitive deficits in morbidly obese children with obstructive sleep apnea. Journal of Pediatrics. 1995;127:741–4. doi: 10.1016/s0022-3476(95)70164-8. [DOI] [PubMed] [Google Scholar]
- 40.Shaffer D, Fisher P, Dulcan MK, et al. The NIMH Diagnostic Interview Schedule for Children Version 2. 3: Description, acceptability, prevalence rates, and performance in the MECA study. Journal of the American Academy of Child and Adolescent Psychiatry. 1996;35:865–77. doi: 10.1097/00004583-199607000-00012. [DOI] [PubMed] [Google Scholar]
- 41.Halperin JM, Newcorn JH, Kopstein I, et al. Serotonin, aggression, and parental psychopathology in children with attention-deficit hyperactivity disorder. Journal of the American Academy of Child and Adolescent Psychiatry. 1997;36:1391–8. doi: 10.1097/00004583-199710000-00021. [DOI] [PubMed] [Google Scholar]
- 42.Swanson JM, Wigal S, Greenhill LL. Analog classroom assessment of Adderall in children with ADHD. Journal of the American Academy of Child and Adolescent Psychiatry. 1998;37:519–26. [PubMed] [Google Scholar]
- 43.Spencer EK, Alpert M, Pouget ER. Scales for the assessment of neuroleptic response in schizophrenic children: specific measures derived from the CPRS. Psychopharmacology Bulletin. 1994;30:199–202. [PubMed] [Google Scholar]
- 44.Overall JE, Campbell M. Behavioral assessment of psychopathology in children: infantile autism. Journal of Clinical Psychology. 1988;44:708–16. doi: 10.1002/1097-4679(198809)44:5<708::aid-jclp2270440507>3.0.co;2-t. [DOI] [PubMed] [Google Scholar]
- 45.National Institute of Mental H. Children’s Psychiatric Rating Scale -- NIMH. Psychopharmacology Bulletin. 1985;21:770. [Google Scholar]
- 46.Chervin RD, Ruzicka DL, Giordani BJ, et al. Sleep-disordered breathing, behavior, and cognition in children before and after adenotonsillectomy. Pediatrics. 2006;117:e769–e78. doi: 10.1542/peds.2005-1837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Conners CK. Conners’ Rating Scales - Revised. North Tonawanda, NY: Multi-Health Systems Publishing; 1997. [Google Scholar]
- 48.Gadow KD, Sprafkin J. Child Symptom Inventory-4. Stony Brook, N.Y: Checkmate Plus; 1994. [Google Scholar]
- 49.Sprafkin J, Gadow KD. Early Childhood Symptom Inventory Manual. Stony Brook, N.Y: Checkmate Plus; 1996. [Google Scholar]
- 50.Korkman M, Kirk U, Kemp S. A Developmental Neuropsychological Assessment. San Antonio: Harcourt Brace Jovanovich; 1998. [Google Scholar]
- 51.Martinez S, Guilleminault C. Periodic leg movements in prepubertal children with sleep disturbance. Dev Med Child Neurol. 2004;46:765–70. doi: 10.1017/s0012162204001318. [DOI] [PubMed] [Google Scholar]
- 52.Picchietti DL, Picchietti MA. Restless Legs Syndrome. In: Kothare SV, Kotagal S, editors. Sleep in Childhood Neurological Disorders. New York: Demos Medical; 2011. pp. 57–70. [Google Scholar]
- 53.Amin R, Anthony L, Somers V, et al. Growth velocity predicts recurrence of sleep-disordered breathing 1 year after adenotonsillectomy. American Journal of Respiratory and Critical Care Medicine. 2008;177:654–9. doi: 10.1164/rccm.200710-1610OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Kirk VG, Bohn S. Periodic limb movements in children: prevalence in a referred population. Sleep. 2004;27:313–5. doi: 10.1093/sleep/27.2.313. [DOI] [PubMed] [Google Scholar]
- 55.Martin BT, Williamson BD, Edwards N, Teng AY. Parental symptom report and periodic limb movements of sleep in children. J Clin Sleep Med. 2008;4:57–61. [PMC free article] [PubMed] [Google Scholar]
- 56.Rogers VE, Marcus CL, Jawad AF, et al. Periodic limb movements and disrupted sleep in children with sickle cell disease. Sleep. 2011;34:899–908. doi: 10.5665/SLEEP.1124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Jambhekar SK, Com G, Jones E, et al. Periodic limb movements during sleep in children with narcolepsy. J Clin Sleep Med. 2011;7:597–601. doi: 10.5664/jcsm.1458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Bhattacharyya N, Lin HW. Changes and consistencies in the epidemiology of pediatric adenotonsillar surgery, 1996–2006. Otolaryngol Head Neck Surg. 2010;143:680–4. doi: 10.1016/j.otohns.2010.06.918. [DOI] [PubMed] [Google Scholar]
- 59.Marcus CL, Brooks LJ, Draper KA, et al. Diagnosis and management of childhood obstructive sleep apnea syndrome. Pediatrics. 2012;130:576–84. doi: 10.1542/peds.2012-1671. [DOI] [PubMed] [Google Scholar]
- 60.Pai V, Khatwa U, Ramgopal S, Singh K, Fitzgerald R, Kothare SV. Prevalence of pediatric periodic leg movements of sleep after initiation of PAP therapy. Pediatr Pulmonol. 2013 doi: 10.1002/ppul.22802. [DOI] [PubMed] [Google Scholar]
- 61.Karadeniz D, Ondze B, Besset A, Billiard M. EEG arousals and awakenings in relation with periodic leg movements during sleep. Journal of Sleep Research. 2000;9:273–7. doi: 10.1046/j.1365-2869.2000.00202.x. [DOI] [PubMed] [Google Scholar]
- 62.El-Ad B, Chervin RD. The case of a missing PLM. Sleep. 2000;23:450–1. [PubMed] [Google Scholar]
- 63.Nicolas A, Lesperance P, Montplaisir J. Is excessive daytime sleepiness with periodic leg movements during sleep a specific diagnostic category? European Neurology. 1998;40:22–6. doi: 10.1159/000007951. [DOI] [PubMed] [Google Scholar]
- 64.Montplaisir J, Michaud M, Denesle R, Gosselin A. Periodic leg movements are not more prevalent in insomnia or hypersomnia but are specifically associated with sleep disorders involving a dopaminergic impairment. Sleep Medicine. 2000;1:163–7. doi: 10.1016/s1389-9457(00)00014-9. [DOI] [PubMed] [Google Scholar]
- 65.Chervin RD, Archbold KH, Dillon JE, et al. Associations between symptoms of inattention, hyperactivity, restless legs, and periodic leg movements. Sleep. 2002;25:213–8. [PubMed] [Google Scholar]
- 66.Chervin RD, Archbold KH. Hyperactivity and polysomnographic findings in children evaluated for sleep-disordered breathing. Sleep. 2001;24:313–20. doi: 10.1093/sleep/24.3.313. [DOI] [PubMed] [Google Scholar]
- 67.Ferri R, Bruni O, Novelli L, Picchietti MA, Picchietti DL. Time structure of leg movement activity during sleep in attention-deficit/hyperactivity disorder and effects of levodopa. Sleep Medicine. 2013;14:359–66. doi: 10.1016/j.sleep.2012.12.012. [DOI] [PubMed] [Google Scholar]
- 68.Chervin RD, Aldrich MS. Effects of esophageal pressure monitoring on sleep architecture. American Journal of Respiratory & Critical Care Medicine. 1997;156:881–5. doi: 10.1164/ajrccm.156.3.9701021. [DOI] [PubMed] [Google Scholar]
- 69.Wing YK, Zhang J, Ho CK, Au CT, Li AM. Periodic limb movement during sleep is associated with nocturnal hypertension in children. Sleep. 2010;33:759–65. doi: 10.1093/sleep/33.6.759. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.