Significance
Circadian clocks regulate gene expression levels to allow an organism to anticipate environmental conditions. These clocks reside in all the major branches of life and confer a competitive advantage to the organisms that maintain them. The clock in the fungus Neurospora crassa is an excellent model for basic understanding of core circadian architecture as well as for filamentous fungi. Here, we identify genes whose expression is clock regulated; indeed, as much as 40% of the transcriptome may be clock regulated, broadly directing daytime catabolism and nighttime growth. Both transcriptional control and posttranscriptional regulation play major roles in control of cycling transcripts such that DNA binding of transcription factors alone appears insufficient to set the phase of circadian transcription.
Keywords: circadian, RNA-Seq, transcription, Neurospora, clock-controlled genes
Abstract
Neurospora crassa has been for decades a principal model for filamentous fungal genetics and physiology as well as for understanding the mechanism of circadian clocks. Eukaryotic fungal and animal clocks comprise transcription-translation–based feedback loops that control rhythmic transcription of a substantial fraction of these transcriptomes, yielding the changes in protein abundance that mediate circadian regulation of physiology and metabolism: Understanding circadian control of gene expression is key to understanding eukaryotic, including fungal, physiology. Indeed, the isolation of clock-controlled genes (ccgs) was pioneered in Neurospora where circadian output begins with binding of the core circadian transcription factor WCC to a subset of ccg promoters, including those of many transcription factors. High temporal resolution (2-h) sampling over 48 h using RNA sequencing (RNA-Seq) identified circadianly expressed genes in Neurospora, revealing that from ∼10% to as much 40% of the transcriptome can be expressed under circadian control. Functional classifications of these genes revealed strong enrichment in pathways involving metabolism, protein synthesis, and stress responses; in broad terms, daytime metabolic potential favors catabolism, energy production, and precursor assembly, whereas night activities favor biosynthesis of cellular components and growth. Discriminative regular expression motif elicitation (DREME) identified key promoter motifs highly correlated with the temporal regulation of ccgs. Correlations between ccg abundance from RNA-Seq, the degree of ccg-promoter activation as reported by ccg-promoter–luciferase fusions, and binding of WCC as measured by ChIP-Seq, are not strong. Therefore, although circadian activation is critical to ccg rhythmicity, posttranscriptional regulation plays a major role in determining rhythmicity at the mRNA level.
A well-recognized model for fungal genetics, photobiology, and circadian systems, Neurospora is the established model for nearly all aspects of growth and metabolism among the filamentous fungi. The core clock of Neurospora crassa has been well studied; its oscillator comprises a transcription-translation feedback loop involving a complex of five core proteins, White Collar 1 (WC-1), White Collar 2 (WC-2), Frequency (FRQ), Frequency Interacting RNA Helicase (FRH), and Casein Kinase 1 (CK1), as well as several ancillary factors. Transcription factors (TFs) WC-1 and WC-2 form the white collar complex (WCC), which drives the rhythmic expression of FRQ. FRQ binds to FRH to form the FRQ/FRH complex (FFC), which then acts with CK1 on the WCC to inhibit its activity, thus closing the loop. Throughout the circadian cycle, FRQ interacts with many partners that affect the number and location of its posttranslational modifications as well as its stability; it is these posttranslational modifications of FRQ that set the length of the circadian period (1–3).
Although historically much interest has focused on clock mechanism, it is through circadian control of transcription that the clock exerts behavioral, physiological, and metabolic control over an organism. A substantial fraction of expressed genes are regulated by the circadian clock in a variety of organisms (refs. 4–7; reviewed in ref. 8), and previous work using ESTs or microarrays in N. crassa has reported ∼5–15% of genes to be rhythmic clock-controlled genes (ccgs) (9–12). However, prior work is flawed in that there is serious concern in calling a gene rhythmic from expression data based on a single circadian day or even sampled over multiple days with a resolution less than 2 h; indeed, sampling only every 4 h may miss up to 90% of clock-regulated genes (13). No extended time series has been sampled at high temporal resolution in any fungus. High-throughput sequencing techniques such as RNA sequencing (RNA-Seq), being more quantitative and having greater dynamic range than microarrays (14, 15), have been used in mammalian and fly systems to improve the qualitative and the quantitative nature of the identification of rhythmic genes (4, 6, 16). Although these studies provided novel insights, they were either completed under conditions of diurnal entrainment by light–dark cycles (which will, as noted by ref. 16, confound acute responses to light with endogenous circadian control) or were sampled at temporal resolution >2 h.
To profit from conceptual and technical advances and to avoid recognized problems, we have used RNA-Seq to identify rhythmic transcripts (mRNA) in N. crassa in triplicate time courses, each spanning 48 h with a 2-h resolution, all collected in constant-dark conditions. We identified 872 genes as being consistently rhythmic, constituting roughly 10% of the N. crassa genome, and a larger set of 4,112 genes, most of which are likely rhythmic. We report that rhythmic genes are enriched for specific pathways at different times of day and furthermore have identified promoter motifs that may be important for the temporal control of the activation of these genes. Analyses of the correlation between clock-regulated promoter activation and mRNA levels reveal that mRNA levels are controlled by the clock both via rhythmic gene activation at the promoter and via posttranscriptional controls that can separately govern rhythmic steady-state transcript levels. To discern the mechanism through which the WCC controls ccg expression, we performed ChIP on WC-2 over a circadian time course and show that it displays a single peak of DNA binding during the day, regardless of the time of the peak of the ccg it activates. These data are consistent with the action of the WCC as the initial rhythm generating TFs in a hierarchical cascade (17).
Results
Genome-Wide Sequencing of Neurospora Circadian Transcription.
To identify mRNAs that cycle in a circadian manner, we performed three circadian time courses, in each case extracting RNA from tissue every 2 h over a period of 48 h (Fig. S1A). These RNA samples were then analyzed for frq mRNA levels by real-time PCR over circadian time to ensure that the samples were oscillating as expected. frq mRNA levels oscillated with an amplitude of twofold to threefold over the circadian day (Fig. S1B), confirming that the extracted RNA samples constituted a rhythmic RNA set. These samples were then used to prepare Illumina libraries using standard protocols and subjected to Illumina high-throughput sequencing. The raw data were then normalized to reads per kilobase of exon model per million mapped reads (RPKM) values and underwent further normalization and smoothing (Materials and Methods).
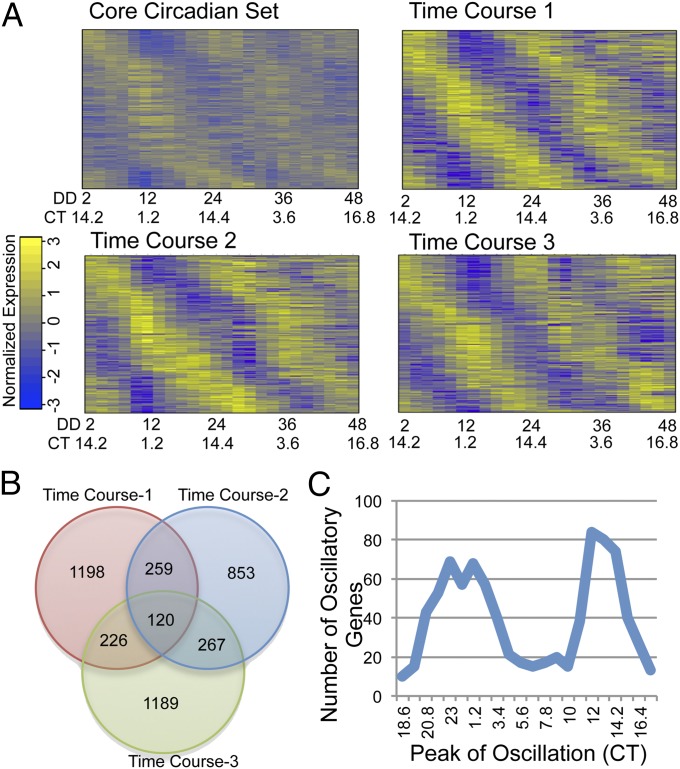
To determine how many of the genes were rhythmic at the transcript level, we analyzed the normalized data using JTK cycle, a nonparametric algorithm for detecting rhythmic components in genome-scale datasets (18). For each dataset, we identified roughly 1,600 genes (roughly 17% of the genome) that were cycling according to JTK cycle with a q value (the minimum false discovery rate) of <0.05 as a conservative estimate (Fig. 1A and Dataset S1). In this study, we considered mRNA that was identified as rhythmic in two of the three datasets as consistently rhythmic, and using this cutoff we identified 872 consistently cycling genes (Fig. 1B and Dataset S1). In all, 4,112 genes were found to be rhythmic in at least one dataset.
Fig. 1.
Neurospora circadian mRNA-Seq. (A) Heat maps of the core 872 genes that are rhythmic in at least two of the three datasets using information from the average of the three datasets (core circadian set) as well as the three individual time courses (time courses 1, 2, and 3). CT, circadian time; DD, time after light to dark transfer. The color bar shows log2 of the normalized level of gene expression. (B) A Venn diagram of the genes found to be rhythmic in the three individual time courses and the overlap between the three sets. (C) A graph depicting the number of genes that are rhythmic at a given CT.
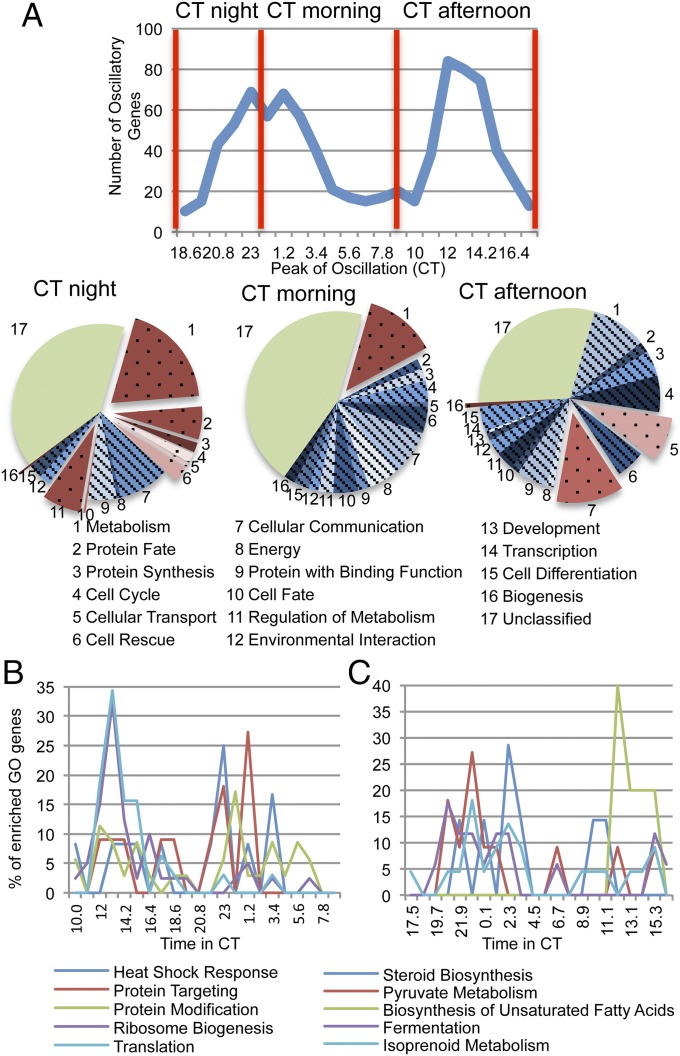
When the peaks of expression of mRNA levels for the significantly rhythmic genes were analyzed by assessing their expression levels on a heat map, it was apparent that peak times of mRNA expression were not evenly spaced but biphasic, peaking during the late night to early circadian day [circadian time (CT) 20–5 h], as previously seen in Neurospora and as would be expected from the activation due to the WCC (Fig. 1 A and C) (1, 19); this is also seen in insect and mammalian models (6, 16). Roughly 35% of the genes identified as rhythmic peaked between CT 11–15 h, which would be assigned to the evening gene category (Fig. 1C).
Following Gene Activation with Luciferase Reporters Affords High-Resolution Tracking of Circadian Promoter Activation.
To correlate rhythmic promoter activation of circadian genes with rhythmic expression of mRNA, we tracked promoter activation of 296 genes known to be bound by WC-2 based on ChIP or known to be decidedly rhythmic based on other work (20, 21). We generated DNA constructs that replaced the coding sequence of the gene with an N. crassa codon optimized luciferase gene (22) for each of the 296 genes investigated (Fig. S2A), preserving the promoter and 500 bp of 3′-UTR. Promoter sizes varied between 1,500 and 10,000 bp and were based on the location of adjacent genes from the RNA-Seq data (www.broadinstitute.org/annotation/genome/neurospora/MultiHome.html).
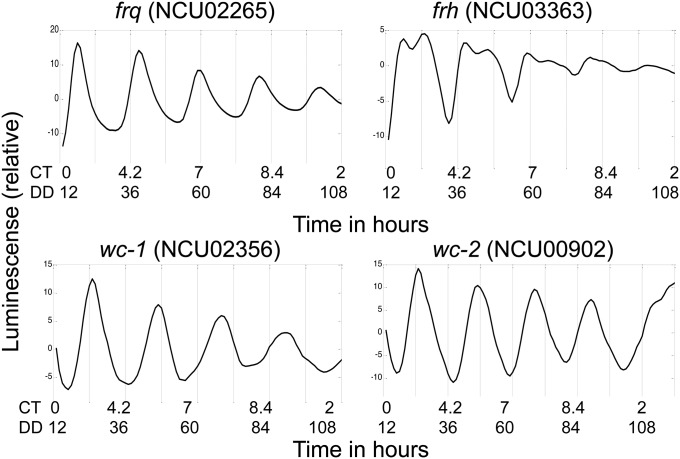
These cassettes were then transformed into the cyclosporin-resistance 1 (csr-1) locus of wild type (FGSC2489) as previously described (23). For each cassette, at least three transformants were subjected to 12:12 dark:light (D:L) entrainment for 48 h before being released to DD and imaged by a CCD camera (Fig. S2B). Genes encoding each of the four core clock proteins were used as functional controls for the assay. As expected, frq was highly rhythmic under this analysis (Fig. 2) (10, 22, 24–26). Although not predicted from protein levels to be strongly rhythmic at the mRNA level (27), wc-2, wc-1, and frh showed promoter rhythms under our experimental conditions, with the rhythmic activation at the frh promoter less defined than the wc-1 and wc-2 promoters (10) (Fig. 2 and Fig. S3A).
Fig. 2.
Luciferase traces track rhythmic genes in the core clock. Luciferase traces acquired using the described format for the four core clock genes after detrending and rescaling.
In total, of the 296 genes tested in various media, 230 showed rhythms in luciferase reporter activity for at least the first several days in at least one medium (Dataset S2). Known ccgs (ccg-1, -2, -4, -6, -7, -8, -9, -12, -13, -14, and -15) displayed varying levels of rhythmicity (Fig. S3B), with some displaying extended and robust rhythms and others showing little to no persistence of rhythms beyond the first peak (compare ccg-4 to ccg-13) (Fig. S3B). This suggested that rhythmic expression of even bona fide ccgs might not correlate with promoter activation rhythms and that posttranscriptional mechanisms may be at play. This analysis also identified rhythms in 75 low-abundance transcripts for which rhythmicity was not detected by RNA-Seq, a finding discussed below.
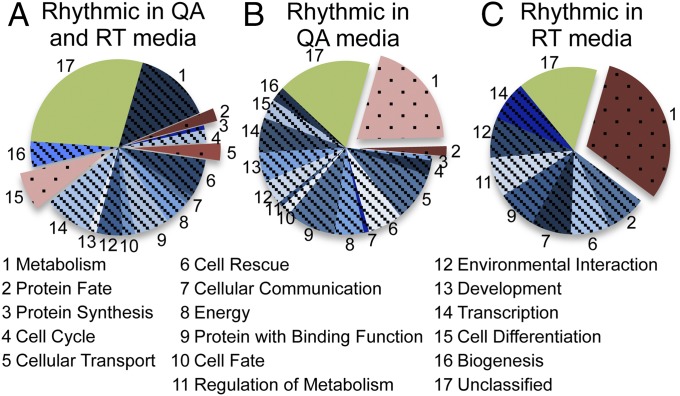
To a limited extent, we also examined the effect of growth medium on rhythmicity. While carrying out controls for other experiments, we noted that the amplitude of ccg-luciferase rhythms were often larger when 0.01 M quinic acid was added to a low-arginine, low-glucose media (QA media) (Dataset S3). To investigate this more systematically, 157 genes were investigated on QA media and race tube (RT) media. Fifty percent of the genes were rhythmic in both RT and QA media, whereas 35% were rhythmic only on QA, 5% were rhythmic only on RT, and 10% were rhythmic in neither. The lists of distinct gene sets from each independent category were subjected to Functional Category (FunCat) term analysis using FungiFun to determine whether the genes were enriched in a particular pathway with a value of P < 0.05 as compared with the entire Neurospora genome (28) (Fig. 3). FunCat classifies genes based on the pathway in which the gene acts instead of the chemical mechanism (29). Genes that were rhythmic in both media were enriched in core cellular functions including cell cycle, cell transport, and protein fate (Fig. 3A). However, genes that were only rhythmic in either QA or RT media were enriched in the metabolic FunCat term category (Fig. 3 B and C). This interesting observation suggests that the subset of genes circadianly regulated by the core clock depends on environmental conditions (in this case growth medium). Although circadian control over core biological functions (e.g., cell cycle, cell transport, etc.) is required irrespective of the environment, circadian regulation is plastic enough to control gene expression hierarchy to best suit the need of the organism in a particular environment.
Fig. 3.
Media changes affect which types of genes are regulated by the clock. FunCat term analysis of the genes found to be rhythmic at the activation level in (A) both QA and RT media; (B) QA media only; and (C) RT media only. In this and all subsequent figures displaying FunCat analysis, categories considered significant (values of P < 0.05) are pushed away from the pie chart and highlighted in red, blue categories have a value of P > 0.05, and green represents genes that are unclassified. The number next to each slice corresponds to the FunCat category.
These data are consistent with a study of the effect of different nitrogen sources on the gene expression patterns in Saccharomyces cerevisiae, which revealed that close to 500 (∼7.5%) genes are regulated differentially by different sources of nitrogen (30). Our observation in N. crassa suggests that the core clock exerts substantial control on at least a subset of these differentially regulated genes and hence forms a conduit for environmental sensing and circadian gene expression.
Genes Enriched in Circadian Transcription.
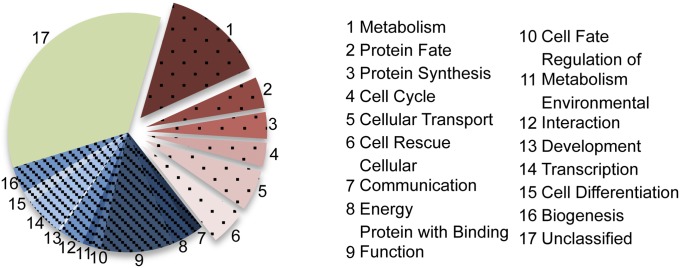
To identify pathways that are enriched in circadian transcription, we subjected the 1,050 genes identified as rhythmic by RNA-Seq analysis as well as luciferase monitoring to FunCat categorization using FungiFun (28) (Dataset S4). The genes that were functionally annotated according to FunCat and the Neurospora crassa Genome Project were subjected to an enrichment analysis that identified overrepresented modulated categories compared with the entire Neurospora genome (28) (Datasets S5 and S6). Some of these categories, such as those representing metabolism, protein fate, protein synthesis, DNA processing, cellular transport, and the cell stress response, were found to be highly enriched in the FunCat analysis of the circadianly regulated genes (Fig. 4).
Fig. 4.
FunCat term analysis highlights rhythmic gene enrichment in specific cellular processes. Breakdown of FunCat terms from all genes designated rhythmic in this dataset with a heat map of the specific genes.
In the metabolic gene subset, genes that are involved in the regulation of carbohydrate, fatty acid, and vitamin metabolism are particularly enriched and are mostly up-regulated during the morning-to-day phase of the clock (CT 0–8). This is also the case for some of the other FunCat categories, including the stress response and cellular transport. It is likely that genes in these categories are up-regulated in anticipation of stress from heat as well as light that fungi face on a daily basis (31, 32) (Fig. S4 A–D) (Dataset S7). Interestingly, categories such as protein synthesis and DNA processing reach their peak expression levels during the late circadian day and evening (CT 10–20) (Fig. S4 E and F) (Dataset S7), suggesting that Neurospora builds up basic cell building material in the day to produce the more complicated resources at night.
To confirm this finding, we then subjected the data to FunCat term analysis in a time-specific manner. Rhythmic genes were divided into three categories by peak of expression (circadian night, circadian morning, and circadian afternoon/evening), and each of these gene lists was sorted by FunCat (Dataset S8). Metabolism was highly enriched in the late night and circadian morning, whereas protein synthesis was highly enriched in the circadian afternoon and early night (Fig. 5A). One of the more interesting findings in the FunCat terms relates to the genes involved in the production and processing of proteins. Although genes involved in ribosome biogenesis and translation peak in the circadian dusk to early evening, genes involved in downstream functions involving proteins, like those involved in protein folding, targeting, and modification, peak in the early circadian morning (Fig. 5B). Not surprisingly, genes like methionine synthase and deoxyhypusine hydroxylase (required for eIF5A) peak during the early circadian evening. We also evaluated our rhythmic gene set using the Kyoto Encyclopedia of Genes and Genomes or KEGG (33). Using this method, we found several enriched pathways. When we graphed the peaks of each of these pathways, we found that, similar to the protein pathway, the peaks of the pathways correlated well with the accumulation of biological materials (Fig. 5C and Fig. S5). For example, early in the circadian day, genes involved in isoprenoid metabolism peak, leading to the development of protective materials including carotenoids (Fig. 5C). Genes involved in pyruvate metabolism (which develops precursors for fatty acid biosynthesis as well as providing energy) and fermentation peak first, followed by those involved in steroid biosynthesis (Fig. 5C and Fig. S5 A and B). Finally, late in the circadian day/early evening, there is a peak in fatty acid biosynthesis, using the building blocks that were created earlier in the circadian day (Fig. 5C and Fig. S5C). The intricate phase networking of the circadianly regulated genes shows the level of fine-tuning the clock provides the biological system.
Fig. 5.
FunCat and KEGG analysis highlight time-of-day–specific gene enrichment. (A) FunCat analysis of rhythmic genes peaking at specific circadian times. (B) The peak in expression of the genes in specific FunCat terms that are involved in protein production. (C) The peak in expression of genes in specific FunCat and KEGG analysis that are involved in metabolic function within the cell.
Discriminative Regular Expression Motif Elicitation Analysis Identifies Motifs Correlated with Circadian Regulation.
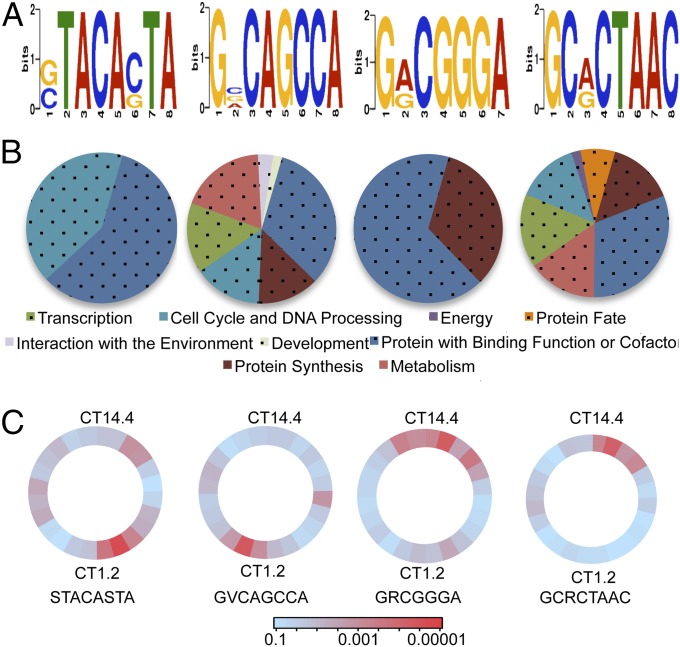
Using the 872 genes identified as being rhythmic based on RNA-Seq analyses, we used discriminative regular expression motif elicitation (DREME) to identify motifs that are significantly enriched among rhythmic genes (34). We analyzed 1,000 bp of the promoter of these 872 rhythmic genes and identified a total of 36 short (up to 8 bp in length) DNA motifs that were enriched in these sequences. To obtain a confident list of circadian-related motifs, we further examined the enrichment of these motifs in the promoters of rhythmic genes that displayed peak expression at different time windows of the circadian clock compared with all genes in the genome that contained these motifs. For example, for the motif STACASTA, we used the Find Individual Motif Occurrences (FIMO) algorithm to examine the motif’s existence in the promoters of all genes. We found that the promoters of 2,823 genes out of the 9,729 gene promoters in the Neurospora genome (29%) contained this motif. We then looked at the number of genes from the rhythmic dataset that peaked in each specific time window; in the CT 20.8–1.2 window, there were 292 genes from the 872 gene rhythmic dataset with a peak of expression, and 113 of these 292 (39%) have the motif STACASTA. We found this to be a significantly enriched motif at this window based on Fisher’s exact test: the probability that 113 genes contain this motif from among the 292 peaking at this time is significantly (P value of 0.0002) greater than the probability that 2,823 promoters from the genome contain this motif. Using this method, motifs were identified as circadian-related if they were significantly enriched (P < 0.001) in at least one specific time window.
Out of the 36 motifs, 4 were found to be significantly enriched in a circadian-related manner: STACASTA, GRCGGGA, GCRCTAAC, and GVCAGCCA (Fig. 6A). We performed FunCat analysis on the genes identified from the 872 rhythmic genes that possessed these motifs in their promoters (Dataset S9). We found that two of the motifs were enriched in the same FunCat terms (metabolism, protein fate, protein synthesis, and cell cycle) that were generally associated with rhythmic gene expression (compare GRCGGGA and GVCAGCCA from Fig. 6B to Fig. 4). However, the other two motifs (STACASTA and GCRCTAAC) were preferentially enriched primarily in the area of “Proteins with binding function or cofactor,” suggesting that these motifs specifically affected this pathway.
Fig. 6.
DREME analysis highlights time-of-day–specific promoter elements. (A) The four significantly enriched promoter motifs identified from the rhythmic gene set. (B) FunCat term analysis of the genes in the rhythmic dataset containing the above sequence in their promoters. (C) Position weight motif plots of motifs from A, showing the P value of the significance of enrichment of a specific motif identified from the rhythmic genes as compared the same to motif in promoters over entire genome, in a 4-h sliding scale over 24 h. Thus, dark red corresponds to a value of P < 0.00001.
By investigating the motif enrichment across different circadian time windows, this method not only identified with high confidence several circadian-related motifs but also provided the particular time at which they might play regulatory functions. As shown in Fig. 6C, the motifs STACASTA and GRCGGGA are most significantly enriched in promoters of rhythmic genes with peak expression in the late night to early circadian morning. For motifs GCRCTAAC and GVCAGCCA, the opposite was true—most of the rhythmic genes associated with the motifs peaked in the circadian evening (Fig. 6C). This suggests that the active phases for these motifs are during these windows and the role that the motifs play in the transcription of the genes associated with these motifs occurs during this time.
Differences in Peak Times Between Genes Showing Rhythmic Promoter Activation and Rhythmic Steady-State mRNA Levels Suggests Posttranscriptional Regulation of Clock-Controlled mRNAs.
The genes chosen for analysis by luciferase were selected independently of the 872 rhythmic genes. Therefore, the overlap of rhythmic genes between the two sets affords an estimate of how often rhythmic transcription (as assessed by luciferase) leads to rhythmic RNA levels (as assessed by RNA-Seq). As noted above, a variety of different media were used for luciferase activity rhythm measurements; in all, 187 genes were examined by luciferase using a medium comparable to that used for the RNA-Seq experiments. Of these, 96 were rhythmic, 24 of which (25%) were rhythmic both by luciferase and by RNA-Seq, whereas 72 (75%) were rhythmic only by luciferase (Dataset S10). Although some of these are low-abundance transcripts for which the limits to consensus sequencing did not produce statistically different RPKM read numbers at different times of day, many probably reflect genes whose transcripts may be too stable to cycle on a circadian timescale. Conversely, an additional 11 genes that were identified as rhythmic by RNA-Seq failed to show rhythms by luciferase, again indicating that rhythmicity can be conferred on transcripts by regulation occurring after a gene is transcribed.
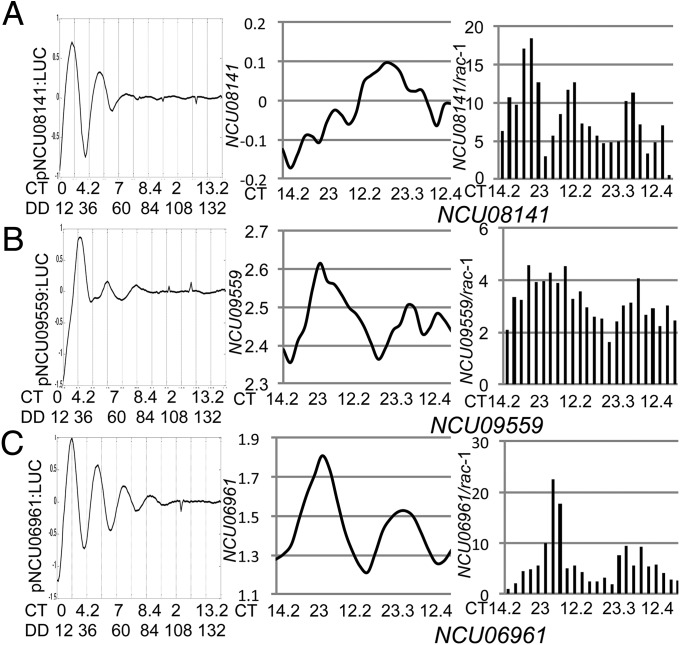
These conclusions were supported by independent analyses of individual genes by RT-PCR using the RNA samples used for the RNA-Seq analyses. The correlation between rhythmicity as detected by RT-PCR and RNA-Seq was very high: of the 29 candidate rhythmic genes (from RNA-Seq) analyzed by RT-PCR, rhythmicity was confirmed for 27 (95%) (Fig. S6). In many cases where rhythmicity was detected by luciferase but not by RNA-Seq or by RT-PCR, the transcripts were of low abundance. For instance, Fig. 7A shows a gene (NCU08141) that is plainly rhythmic by luciferase but appears to show ultradian cycling by RNA-Seq and by RT-PCR; however, the transcript abundance arising from this gene is quite low. For other genes, however, transcripts are quite abundant, and rhythmicity is plain in the RNA-Seq and RT-PCR analysis but is plainly muted by luciferase reporter assays (Fig. 7B). Such genes are strong candidates for regulation at levels following transcription. The existence of such regulation is further supported by differences in phase between luciferase assays that report promoter activity and sequencing/RT-PCR that report transcript abundance. As an example, Fig. 7C shows NCU06961 that is robustly rhythmic by both assays but displays a significant phase lag in abundance compared with transcription.
Fig. 7.
RNA-Seq and real-time PCR correlate weakly with promoter activation. A comparison between luciferase traces, RNA-Seq data and RT-PCR analysis for three genes that exemplify cases where a gene is rhythmic by Luc only (A), by RNA-Seq only (B), and by both methods (C). (A) NCU08141 showed rhythms by luciferase but not by RNA-Seq or RT-PCR analysis, most likely due to low transcript abundance. (B) NCU09559 demonstrated rhythms by RNA-Seq and RT-PCR but not by luciferase, most likely due to posttranscriptional regulation. (C) NCU06961 exhibited rhythms with all methods but with a lag between peak of activation and mRNA levels, potentially from posttranscriptional mechanisms. Luciferase data are detrended and rescaled and RPKM values are shown in log scale.
The WCC Acts as the Initiating TF for Gene Rhythmicity.
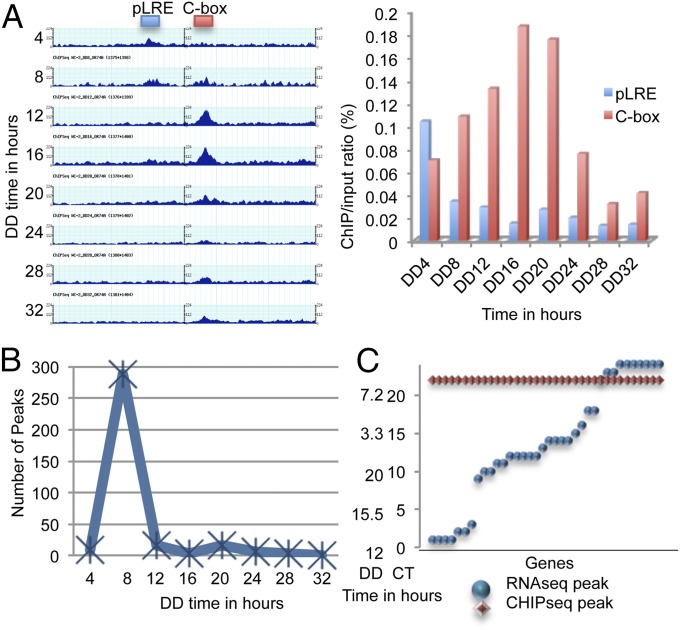
Circadian regulation of WCC activity in the core clock is understood as providing the principal means of circadian output from the clock, when the WCC regulates expression of genes whose products do not impact the core clock (1, 35). Thus, examining the spectrum of gene promoters bound by WC-2 (reflecting the WCC) and the time at which peak binding occurs affords a glimpse at the first step of output. To identify the circadian targets of WC-2, we performed a ChIP sequencing assay (ChIP-Seq) at regular intervals on cultures that were kept in the dark (after transfer from constant light) using antibody against WC-2. We then performed a RT-PCR using probes for the proximal light-regulated element (pLRE) and Clock box (C-box) (36, 37) motifs upstream of the frq gene to test whether our ChIP worked as expected. We found that occupation at the pLRE decreased following the light-to-dark transition and remained low for the remainder of the time points; however, there was still appreciable binding to the pLRE after even 8 h in darkness, establishing that the effects of the previous light cycle on gene expression can be long-lasting (Fig. 8A). This serves as a caution indicating that analyses of “circadian” gene expression must not rely solely on data from the first day in darkness. Also, as expected, occupation of the C-box changed rhythmically over the circadian day, peaking at about 16 h after light-to-dark transition [Fig. 8A (38)]. MACS software was used to identify regions in the genome where WC-2 binding was enriched compared with a WC-1 knockout strain in which the WCC does not bind DNA. A peak was identified at both the pLRE and C-box of frq (Dataset S11). We then compared these results from the ChIP-Seq data to the ChIP–RT-PCR and found that the reported peaks matched the RT-PCR data (Fig. 8A). When the WC-2 ChIP-Seq data were examined all together by MACS, WC-2 was found to bind at nearly 300 sites in the genome with a peak in the late circadian night similar to that found for the C-box (Fig. 8B). Comparing the genes that were bound by WC-2 in the ChIP experiment to a list of predicted TFs in Neurospora, we found that 8% of predicted TFs were bound by WC-2.
Fig. 8.
In WC-2 target genes, the delay between WC-2 binding and mRNA levels suggests that the WCC acts as a pioneer TF. (A) Left panel shows results of ChIP-Seq of WC-2 revealing sites bound within the frq promoter. Here, frq is transcribed right to left with the pLRE close to the transcription start site and the C-box more distal. Plots for ChIP-Seq data from gbrowse (60) are shown for each time in darkness from DD4 to DD32. All are plotted as FPKM on the same vertical scale. Right panel shows the RT-PCR of DNA used for ChIP-Seq at both the pLRE and C-box of frq demonstrating peak binding of the C-box at DD16 as in the left panel. (B) The number of WC-2 peaks identified by MACS plotted against circadian time shows the highest number of peaks identified as bound by WC-2 is at DD8. (C) Distribution of the peak phase of WC-2 DNA binding (in red) and the peak phase for mRNA levels of the same gene (in blue) for the direct targets of WC-2.
To determine whether the WCC is the initiator of rhythmicity but not necessarily responsible for setting the phase of ccgs, we compared the genes that have WC-2 peaks to genes that are rhythmic in the RNA-Seq dataset. Of the 872 genes that were rhythmic from the RNA-Seq, 36 of them had major binding sites for WC-2. We then mapped the peak of binding by WC-2 to the peak of mRNA levels for each gene. It was evident that, although the peak binding time was in all cases near to CT0 for WC-2, the peak mRNA levels were delayed to phases spanning the circadian cycle (Fig. 8C). We compared the ChIP-Seq data to the RNA-Seq data for a few of the genes and noted that this trend was confirmed by visual observation of specific genes (Fig. S7 A–D).
Discussion
Circadian regulation of gene expression is the point at which the circadian oscillator begins to impact the biology of the organism. For this reason, a detailed description of the circadian transcriptome is key to understanding the physiology of Neurospora, and indeed elucidation of the circadian regulation of the transcriptome has thus been a topic of interest in all of the key circadian model organisms (4, 6, 10, 11, 16, 39–43). Temporal control over the expression of key metabolic/functional genes has always been a prediction for the role of an “anticipatory” core clock in any organism, and ccgs have been identified at the genome-wide level through the use of ESTs, microarrays, and RNA-Seq. In fungi and animals, the clock generates rhythmic activation of a heterodimeric TF joined by interacting PAS domains, and the first step in output is the action of this transcription factor to activate genes whose products do not directly impact the oscillator. Studies in animal systems have used various techniques, beginning with ChIP-Seq, to report binding of the PAS heterodimer that initiates transcription and Nascent-Seq (e.g., refs. 6 and 16) to distinguish transcript rhythmicity conferred by transcription from rhythmicity later conferred through posttranscriptional regulation (reviewed in ref. 44). Recent years have also seen increased sophistication in data analysis software and in realization of the importance of extended time courses and dense (2 h or less) sampling resolution for the identification of ccgs (e.g., refs. 13 and 18). We aimed here for a definitive dataset, and the data provided from our studies are unique in using both true circadian (nonentrained) conditions and high temporal resolution sampling over two cycles.
mRNAs displaying rhythmic abundance were identified using three independent 48-h time courses with a 2-h resolution using RNA-Seq, the most thorough analysis of a rhythmic transcriptome in any organism to date. Because Nascent-Seq requires the isolation of nuclei, difficult across a circadian time course at this resolution in N. crassa, we have used a high-throughput promoter-luc strategy to correlate promoter activation with rhythmicity at the mRNA level. Although this approach lacks the depth of transcriptome analysis afforded by Nascent-Seq, its advantages include much finer detail of the transcriptional dynamics of individual genes, better estimates of phase, estimates of rhythm damping and persistence not afforded by Nascent-Seq except at inordinate expense, and creation of reporters that can be reused for subsequent dissection of the regulatory network.
We find that there is a significant discordance between the promoter activation of a gene and rhythmicity of its transcript at the mRNA level; i.e., we have identified a number of cases where promoters are rhythmically activated but the corresponding mRNAs do not display rhythmic abundance and vice versa (Fig. 7) (see also ref. 16). This discordance is also strikingly similar to that found in a similar analysis completed in mouse liver (45). Our analysis of the individual genes using RT-PCR showed good correlation with the RNA-Seq data (95% of the genes rhythmic by RNA-Seq are rhythmic by RT-PCR), suggesting that there is indeed a level of posttranscriptional regulation between gene activation and rhythmic abundance of mRNA (Fig. 7 A and B). Moreover, in some cases, we find significant phase differences between the peak of gene activation and rhythmic mRNA levels, which suggests that accumulation of mRNA is not always solely governed by the activation of the corresponding gene.
Several possible mechanisms may underlie this discordance outside of artifactual issues, including specific RNA stability and/or differential RNA processing. One can also envision a situation where RNA from a nonrhythmically driven gene could have different stability at different times of the day owing to its binding to stabilizing/destabilizing RNA-binding proteins (RBPs). This time-specific binding might induce robust rhythms at the mRNA level. In fact, the strategy of time-of-day–dependent differential transcript stability exists in N. crassa to generate robust frq rhythms (46). Similarly, CCR-LIKE (CCL) and SENESCENCE ASSOCIATED GENE 1 transcripts in Arabidopsis have time-of-day–specific changes in stability, and the changes in CCL mRNA stability are under bona fide clock control, i.e., they persist under free-running conditions (47). Other examples include RBPs like HuR that binds to and stabilizes cyclin D1 transcript as opposed to AUF1 that binds to and destabilizes cyclin D1 transcript—both processes required for a highly coordinated and regulated cell cycle progression (48).
Our RNA-Seq data also allowed us to correlate the phase of WCC binding (WC-2 ChIP) with the phase of expression of its target genes. It is quite obvious from our analysis that, although WCC binding is very discrete, the phase of expression for its target genes is heterogeneous. A similar scenario was observed when the phase of BMAL1-CLOCK binding and the phase of expression of its target genes were compared in mouse livers (16). However, this contrasts with the observation in flies where both binding of CLK:CYC to its target genes and the expression of its targets were found to be in the same phase (49). The absence of a fixed phase of target gene expression despite a discrete phase of WCC binding suggests that, for genes that show delayed expression, there is a requirement for additional factors (TFs, chromatin remodelers) to set the phase of expression for these genes. More recently, it has been shown that BMAL1-CLOCK behaves like a pioneer-like TF by modulating rhythmic opening of chromatin through chromatin modification, which allows other TFs to bind adjacent to BMAL1-CLOCK (50) and modulate gene expression. Recent research from our group has shown that WCC recruits the SWI–SNF complex (51) and modulates rhythmic opening of chromatin at the frq locus. It is thus possible that WCC similarly binds to various targets in the genome as a pioneer-like TF and promotes remodeling of the chromatin so as to allow other TFs to bind. This could potentially explain the heterogeneity in the phase of peak expression of its target genes.
Setting aside the exact means of circadian control, a major outcome of this study is the picture it affords of the circadian control of physiology in Neurospora. Concomitant functional and pathway categorization of our rhythmic genes using FunCat and KEGG analysis, respectively, revealed broad categories (e.g., metabolism, protein fate, DNA processing, etc.) as well as subcategories/pathways that were enriched and could be studied for temporal relevance in the circadian biology of the organism. For example, genes involved in ribosome biogenesis and protein assembly peak late in the circadian day compared with genes involved in protein folding, targeting, and modification (Fig. 5B), similar to what is seen in mouse liver (52). Logically, this makes sense as one can envision the peak for genes required for translational activity and ribosome assembly to precede the peak for protein folding/targeting/modification as the latter events can be “phase-concentrated” after a burst of protein translation, thereby making the process more efficient (Fig. 5B). Indeed, most major categories of cellular metabolism display some rhythmic control (Fig. 4). When querying specific pathways for rhythmicity, catabolic pathways such as pyruvate metabolism peak earlier in the day than those involved in anabolic pathways like steroid biosynthesis and fatty acid biosynthesis (Fig. 5C and Fig. S5). The enrichment of metabolic genes is not altogether surprising as many metabolic genes have been shown to be circadianly controlled in several other studies (e.g., ref. 53). As a broad generalization, the transcriptome data suggest that daytime activities involve catabolism, energy production, and assembly of precursors, whereas nighttime activities favor biosynthesis of cellular components and growth.
Moreover, it is now highly regarded that the clock is not just a static master controller that sets up anticipatory metabolic gene expression but can also serve as a conduit between the environment and the transcriptome. In other words, the subset of genes that is circadianly expressed can be altered depending on the environment and the clock can coordinate this alteration. Although it is tough to test this from RNA-Seq data (all biological sets grown in one media), use of high-throughput promoter-luc assays clearly shows that subsets of the genes were rhythmically activated in specific media (Fig. 3). These subsets were again enriched in metabolic FunCat categories suggesting the core-clock indeed coordinates, undoubtedly with the help of other factors (TFs, metabolite-binding proteins, etc.), the gene expression landscape as a function of the environment. More interestingly, rhythmicity of genes involved in core cellular functions (cell cycle, cell transport, etc.) appears not to be dependent on the media (environment), suggesting a more stringent and environmentally refractory circadian control over these genes.
The DREME analysis of rhythmic genes (872) in our RNA-Seq dataset also allowed us to identify four motifs that were significantly enriched (STACASTA, GRCGGGA, GCRCTAAC, and GVCAGCCA) in the promoters of rhythmic genes compared with the rest of the genome. For two of the motifs (STACASTA and GRCGGGA), we find that time of day (CTs) at which there is significant enrichment in the promoters of rhythmic genes is coincident with the time of day (morning) where a large fraction of rhythmic genes shows peak mRNA expression in the rhythmic dataset. This appears not to be the case with the other two motifs (GCRCTAAC and GVCAGCCA) where the time of significant enrichment is in the early evening and does not coincide with the time where most genes in the rhythmic dataset show peak mRNA levels. This analysis does not allow us to conclude what role a particular motif is playing during its active period, whether it is an activator or repressor, only that the impact on gene expression for each motif is likely during that circadian time window.
Approximately 10% of the N. crassa transcriptome is reproducibly rhythmic at the mRNA level under normal growth, and potentially the clock may influence 40% of the genome. Undeniably, circadian control has a major impact on the biology of Neurospora: Much of metabolism is clock-controlled, and in broad outline, daytime activities involve catabolism, energy production, and assembly of precursors, whereas nighttime activities favor biosynthesis of cellular components and growth. Through use of a high-throughput assay using promoter-luc fusions, we show that rhythmic gene activation in and of itself is insufficient to dictate the transcript cycling at the mRNA level. This indicates that posttranscriptional regulation plays a major role in control of cycling transcripts and recommends further analysis of how such posttranscriptional regulators involved are themselves regulated (circadian expression, circadian activity, circadian binding, etc.). Finally, WCC binding alone is insufficient to set the phase of circadian transcription in N. crassa, indicating that additional factors are needed to determine the phase at which promoters of cycling genes are activated.
Materials and Methods
Strains.
A protocol similar to that described (54) was used in all transformants. The Neurospora strain FGSC2489 was used for all analyses. mRNA was extracted for the circadian time course as previously described, with minor exceptions (21). RT-PCR was performed on the same mRNA extracted for use in the library construction as previously described, with minor changes (21) using primers (Dataset S12). Camera recordings and data analysis were as described (22, 25), with minor modifications (SI Text).
Library Construction and Mapping.
Libraries were prepared as per the manufacturers instructions and libraries were analyzed using Illumina/Solexa sequencing (SI Text). The resulting raw and processed data was deposited to National Center for Biotechnology Information (NCBI) Short Read Archive (SRA) under accession number SRP046458.
Data and FunCat Analysis.
RPKM values were subjected to further normalization and each individual time course was subjected to JTK cycle analysis (18). Any gene with a false-discovery rate of Q < 0.05 was selected as a rhythmic gene. DREME analysis (34) was used to identify significantly enriched motifs (SI Text). Functional annotation of the differently expressed genes was performed according to Functional Categories (55) and N. crassa Genome Database (56). FunCat term and KEGG enrichment was performed using the FungiFun (28) and a value of P < 0.05 was used as a cutoff for significantly enriched terms.
ChIP-Seq.
Neurospora tissue was cross-linked with 1% paraformaldehyde for 15 min and quenched with 0.125 M glycine for an additional 5 min for samples ranging from DD4 to DD32 . ChIP was performed as described previously (38, 57) on 500 mg of tissue with WC-2 antibody with some modifications (58). Indexed sequencing libraries were prepared as described (59) (SI Text). Raw and processed data were submitted to the NCBI SRA under accession number SRP045821.
Supplementary Material
Acknowledgments
We thank the Fungal Genetics Stock Center at the University of Missouri for Neurospora strains. This work is supported by NIH-National Institute of General Medical Sciences Grants GM083336 (to J.J.L.), GM34985 and GM068087 (to J.C.D.), and DE-AC02-05CH11231 (US Department of Energy).
Footnotes
The authors declare no conflict of interest.
Data deposition: The data reported in this paper have been deposited in the National Center for Biotechnology Information Short Read Archive (SRA) (accession nos. SRP045821 and SRP046458).
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1418963111/-/DCSupplemental.
References
- 1.Baker CL, Loros JJ, Dunlap JC. The circadian clock of Neurospora crassa. FEMS Microbiol Rev. 2012;36(1):95–110. doi: 10.1111/j.1574-6976.2011.00288.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Liu Y, Bell-Pedersen D. Circadian rhythms in Neurospora crassa and other filamentous fungi. Eukaryot Cell. 2006;5(8):1184–1193. doi: 10.1128/EC.00133-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Diernfellner AC, Schafmeier T. Phosphorylations: Making the Neurospora crassa circadian clock tick. FEBS Lett. 2011;585(10):1461–1466. doi: 10.1016/j.febslet.2011.03.049. [DOI] [PubMed] [Google Scholar]
- 4.Hughes ME, Grant GR, Paquin C, Qian J, Nitabach MN. Deep sequencing the circadian and diurnal transcriptome of Drosophila brain. Genome Res. 2012;22(7):1266–1281. doi: 10.1101/gr.128876.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lowrey PL, Takahashi JS. Mammalian circadian biology: Elucidating genome-wide levels of temporal organization. Annu Rev Genomics Hum Genet. 2004;5:407–441. doi: 10.1146/annurev.genom.5.061903.175925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rodriguez J, et al. Nascent-Seq analysis of Drosophila cycling gene expression. Proc Natl Acad Sci USA. 2013;110(4):E275–E284. doi: 10.1073/pnas.1219969110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Vollmers C, et al. Time of feeding and the intrinsic circadian clock drive rhythms in hepatic gene expression. Proc Natl Acad Sci USA. 2009;106(50):21453–21458. doi: 10.1073/pnas.0909591106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Masri S, Sassone-Corsi P. The circadian clock: A framework linking metabolism, epigenetics and neuronal function. Nat Rev Neurosci. 2013;14(1):69–75. doi: 10.1038/nrn3393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Nowrousian M, Duffield GE, Loros JJ, Dunlap JC. The frequency gene is required for temperature-dependent regulation of many clock-controlled genes in Neurospora crassa. Genetics. 2003;164(3):923–933. doi: 10.1093/genetics/164.3.923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Dong W, et al. Systems biology of the clock in Neurospora crassa. PLoS One. 2008;3(8):e3105. doi: 10.1371/journal.pone.0003105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zhu H, et al. Analysis of expressed sequence tags from two starvation, time-of-day-specific libraries of Neurospora crassa reveals novel clock-controlled genes. Genetics. 2001;157(3):1057–1065. doi: 10.1093/genetics/157.3.1057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Correa A, et al. Multiple oscillators regulate circadian gene expression in Neurospora. Proc Natl Acad Sci USA. 2003;100(23):13597–13602. doi: 10.1073/pnas.2233734100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hughes ME, et al. Harmonics of circadian gene transcription in mammals. PLoS Genet. 2009;5(4):e1000442. doi: 10.1371/journal.pgen.1000442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zhao S, Fung-Leung WP, Bittner A, Ngo K, Liu X. Comparison of RNA-Seq and microarray in transcriptome profiling of activated T cells. PLoS One. 2014;9(1):e78644. doi: 10.1371/journal.pone.0078644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wang Z, Gerstein M, Snyder M. RNA-Seq: A revolutionary tool for transcriptomics. Nat Rev Genet. 2009;10(1):57–63. doi: 10.1038/nrg2484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Menet JS, Rodriguez J, Abruzzi KC, Rosbash M. Nascent-Seq reveals novel features of mouse circadian transcriptional regulation. eLife. 2012;1:e00011. doi: 10.7554/eLife.00011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Dunlap JC, Loros JJ. The Neurospora circadian system. J Biol Rhythms. 2004;19(5):414–424. doi: 10.1177/0748730404269116. [DOI] [PubMed] [Google Scholar]
- 18.Hughes ME, Hogenesch JB, Kornacker K. JTK_CYCLE: An efficient nonparametric algorithm for detecting rhythmic components in genome-scale data sets. J Biol Rhythms. 2010;25(5):372–380. doi: 10.1177/0748730410379711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bell-Pedersen D, Shinohara ML, Loros JJ, Dunlap JC. Circadian clock-controlled genes isolated from Neurospora crassa are late night- to early morning-specific. Proc Natl Acad Sci USA. 1996;93(23):13096–13101. doi: 10.1073/pnas.93.23.13096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Smith KM, et al. Transcription factors in light and circadian clock signaling networks revealed by genomewide mapping of direct targets for Neurospora white collar complex. Eukaryot Cell. 2010;9(10):1549–1556. doi: 10.1128/EC.00154-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Chen CH, Ringelberg CS, Gross RH, Dunlap JC, Loros JJ. Genome-wide analysis of light-inducible responses reveals hierarchical light signalling in Neurospora. EMBO J. 2009;28(8):1029–1042. doi: 10.1038/emboj.2009.54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gooch VD, et al. Fully codon-optimized luciferase uncovers novel temperature characteristics of the Neurospora clock. Eukaryot Cell. 2008;7(1):28–37. doi: 10.1128/EC.00257-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bardiya N, Shiu PK. Cyclosporin A-resistance based gene placement system for Neurospora crassa. Fungal Genet Biol. 2007;44(5):307–314. doi: 10.1016/j.fgb.2006.12.011. [DOI] [PubMed] [Google Scholar]
- 24.Morgan LW, Greene AV, Bell-Pedersen D. Circadian and light-induced expression of luciferase in Neurospora crassa. Fungal Genet Biol. 2003;38(3):327–332. doi: 10.1016/s1087-1845(02)00562-5. [DOI] [PubMed] [Google Scholar]
- 25.Larrondo LF, Loros JJ, Dunlap JC. High-resolution spatiotemporal analysis of gene expression in real time: In vivo analysis of circadian rhythms in Neurospora crassa using a FREQUENCY-luciferase translational reporter. Fungal Genet Biol. 2012;49(9):681–683. doi: 10.1016/j.fgb.2012.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gooch VD, et al. A kinetic study of the effects of light on circadian rhythmicity of the frq promoter of Neurospora crassa. J Biol Rhythms. 2014;29(1):38–48. doi: 10.1177/0748730413517981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lee K, Loros JJ, Dunlap JC. Interconnected feedback loops in the Neurospora circadian system. Science. 2000;289(5476):107–110. doi: 10.1126/science.289.5476.107. [DOI] [PubMed] [Google Scholar]
- 28.Priebe S, Linde J, Albrecht D, Guthke R, Brakhage AA. FungiFun: A web-based application for functional categorization of fungal genes and proteins. Fungal Genet Biol. 2011;48(4):353–358. doi: 10.1016/j.fgb.2010.11.001. [DOI] [PubMed] [Google Scholar]
- 29.Ruepp A, et al. The FunCat, a functional annotation scheme for systematic classification of proteins from whole genomes. Nucleic Acids Res. 2004;32(18):5539–5545. doi: 10.1093/nar/gkh894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Godard P, et al. Effect of 21 different nitrogen sources on global gene expression in the yeast Saccharomyces cerevisiae. Mol Cell Biol. 2007;27(8):3065–3086. doi: 10.1128/MCB.01084-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Shinohara ML, Correa A, Bell-Pedersen D, Dunlap JC, Loros JJ. Neurospora clock-controlled gene 9 (ccg-9) encodes trehalose synthase: Circadian regulation of stress responses and development. Eukaryot Cell. 2002;1(1):33–43. doi: 10.1128/EC.1.1.33-43.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Bahn YS, et al. Sensing the environment: Lessons from fungi. Nat Rev Microbiol. 2007;5(1):57–69. doi: 10.1038/nrmicro1578. [DOI] [PubMed] [Google Scholar]
- 33.Kanehisa M, Goto S. KEGG: Kyoto Encyclopedia of Genes and Genomes. Nucleic Acids Res. 2000;28(1):27–30. doi: 10.1093/nar/28.1.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bailey TL. DREME: Motif discovery in transcription factor ChIP-seq data. Bioinformatics. 2011;27(12):1653–1659. doi: 10.1093/bioinformatics/btr261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Fuller KK, Hurley JM, Loros J, Dunlap J. Photobiology and circadian clocks in Neurospora. In: Nowrousian M, editor. The Mycota. Vol XIII. Springer; Heidelberg: 2014. pp. 121–148. [Google Scholar]
- 36.Froehlich AC, Liu Y, Loros JJ, Dunlap JC. White Collar-1, a circadian blue light photoreceptor, binding to the frequency promoter. Science. 2002;297(5582):815–819. doi: 10.1126/science.1073681. [DOI] [PubMed] [Google Scholar]
- 37.Froehlich AC, Loros JJ, Dunlap JC. Rhythmic binding of a WHITE COLLAR-containing complex to the frequency promoter is inhibited by FREQUENCY. Proc Natl Acad Sci USA. 2003;100(10):5914–5919. doi: 10.1073/pnas.1030057100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Belden WJ, Lewis ZA, Selker EU, Loros JJ, Dunlap JC. CHD1 remodels chromatin and influences transient DNA methylation at the clock gene frequency. PLoS Genet. 2011;7(7):e1002166. doi: 10.1371/journal.pgen.1002166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Akhtar RA, et al. Circadian cycling of the mouse liver transcriptome, as revealed by cDNA microarray, is driven by the suprachiasmatic nucleus. Curr Biol. 2002;12(7):540–550. doi: 10.1016/s0960-9822(02)00759-5. [DOI] [PubMed] [Google Scholar]
- 40.Duffield GE, et al. Circadian programs of transcriptional activation, signaling, and protein turnover revealed by microarray analysis of mammalian cells. Curr Biol. 2002;12(7):551–557. doi: 10.1016/s0960-9822(02)00765-0. [DOI] [PubMed] [Google Scholar]
- 41.Harmer SL, et al. Orchestrated transcription of key pathways in Arabidopsis by the circadian clock. Science. 2000;290(5499):2110–2113. doi: 10.1126/science.290.5499.2110. [DOI] [PubMed] [Google Scholar]
- 42.Rodriguez J, Menet JS, Rosbash M. Nascent-seq indicates widespread cotranscriptional RNA editing in Drosophila. Mol Cell. 2012;47(1):27–37. doi: 10.1016/j.molcel.2012.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Storch KF, et al. Extensive and divergent circadian gene expression in liver and heart. Nature. 2002;417(6884):78–83. doi: 10.1038/nature744. [DOI] [PubMed] [Google Scholar]
- 44.Beckwith EJ, Yanovsky MJ. Circadian regulation of gene expression: At the crossroads of transcriptional and post-transcriptional regulatory networks. Curr Opin Genet Dev. 2014;27:35–42. doi: 10.1016/j.gde.2014.03.007. [DOI] [PubMed] [Google Scholar]
- 45.Koike N, et al. Transcriptional architecture and chromatin landscape of the core circadian clock in mammals. Science. 2012;338(6105):349–354. doi: 10.1126/science.1226339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Guo J, Cheng P, Yuan H, Liu Y. The exosome regulates circadian gene expression in a posttranscriptional negative feedback loop. Cell. 2009;138(6):1236–1246. doi: 10.1016/j.cell.2009.06.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Lidder P, Gutiérrez RA, Salomé PA, McClung CR, Green PJ. Circadian control of messenger RNA stability. Association with a sequence-specific messenger RNA decay pathway. Plant Physiol. 2005;138(4):2374–2385. doi: 10.1104/pp.105.060368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Lal A, et al. Concurrent versus individual binding of HuR and AUF1 to common labile target mRNAs. EMBO J. 2004;23(15):3092–3102. doi: 10.1038/sj.emboj.7600305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Abruzzi KC, et al. Drosophila CLOCK target gene characterization: Implications for circadian tissue-specific gene expression. Genes Dev. 2011;25(22):2374–2386. doi: 10.1101/gad.178079.111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Menet JS, Pescatore S, Rosbash M. CLOCK:BMAL1 is a pioneer-like transcription factor. Genes Dev. 2014;28(1):8–13. doi: 10.1101/gad.228536.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Wang B, Kettenbach AN, Gerber SA, Loros JJ, Dunlap JC. Neurospora WC-1 recruits SWI/SNF to remodel frequency and initiate a circadian cycle. PLoS Genet. 2014;10(9):e1004599. doi: 10.1371/journal.pgen.1004599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Jouffe C, et al. The circadian clock coordinates ribosome biogenesis. PLoS Biol. 2013;11(1):e1001455. doi: 10.1371/journal.pbio.1001455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Sancar G, Sancar C, Brunner M. Metabolic compensation of the Neurospora clock by a glucose-dependent feedback of the circadian repressor CSP1 on the core oscillator. Genes Dev. 2012;26(21):2435–2442. doi: 10.1101/gad.199547.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Colot HV, et al. A high-throughput gene knockout procedure for Neurospora reveals functions for multiple transcription factors. Proc Natl Acad Sci USA. 2006;103(27):10352–10357. doi: 10.1073/pnas.0601456103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Ashburner M, et al. The Gene Ontology Consortium Gene Ontology: Tool for the unification of biology. Nat Genet. 2000;25(1):25–29. doi: 10.1038/75556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Galagan JE, et al. The genome sequence of the filamentous fungus Neurospora crassa. Nature. 2003;422(6934):859–868. doi: 10.1038/nature01554. [DOI] [PubMed] [Google Scholar]
- 57.Johnson L, Cao X, Jacobsen S. Interplay between two epigenetic marks. DNA methylation and histone H3 lysine 9 methylation. Curr Biol. 2002;12(16):1360–1367. doi: 10.1016/s0960-9822(02)00976-4. [DOI] [PubMed] [Google Scholar]
- 58.Denault DL, Loros JJ, Dunlap JC. WC-2 mediates WC-1-FRQ interaction within the PAS protein-linked circadian feedback loop of Neurospora. EMBO J. 2001;20(1-2):109–117. doi: 10.1093/emboj/20.1.109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Connolly LR, Smith KM, Freitag M. The Fusarium graminearum histone H3 K27 methyltransferase KMT6 regulates development and expression of secondary metabolite gene clusters. PLoS Genet. 2013;9(10):e1003916. doi: 10.1371/journal.pgen.1003916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Stein LD, et al. The generic genome browser: A building block for a model organism system database. Genome Res. 2002;12(10):1599–1610. doi: 10.1101/gr.403602. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.