Abstract
Pre-conditioning by ischemia, hyperthermia, hypothermia, hyperbaric oxygen (and numerous other modalities) is a rapidly growing area of investigation that is used in pathological conditions where tissue damage may be expected. The damage caused by surgery, heart attack, or stroke can be mitigated by pre-treating the local or distant tissue with low levels of a stress-inducing stimulus, that can induce a protective response against subsequent major damage. Low-level laser (light) therapy (LLLT) has been used for nearly 50 years to enhance tissue healing and to relieve pain, inflammation and swelling. The photons are absorbed in cytochrome(c) oxidase (unit four in the mitochondrial respiratory chain), and this enzyme activation increases electron transport, respiration, oxygen consumption and ATP production. A complex signaling cascade is initiated leading to activation of transcription factors and up- and down-regulation of numerous genes. Recently it has become apparent that LLLT can also be effective if delivered to normal cells or tissue before the actual insult or trauma, in a pre-conditioning mode. Muscles are protected, nerves feel less pain, and LLLT can protect against a subsequent heart attack. These examples point the way to wider use of LLLT as a pre-conditioning modality to prevent pain and increase healing after surgical/medical procedures and possibly to increase athletic performance.
Keywords: Pre-conditioning, Remote Ischemic Pre-conditioning, LLLT, Photobiomodulation, Mitochondria, Reactive Oxygen Species
INTRODUCTION
Many diseases and traumatic events involving tissue damage are injurious because of ischemia, a sudden or more gradual process characterized by deprivation of the tissues of life-sustaining oxygen. Ischemic heart disease and ischemic stroke account for the largest burden of mortality and morbidity in modern life. The term pre-conditioning (PC) was first applied to a regimen in which repetitive short episodes of ischemia and reperfusion could lead to the development of resistance of the myocardium to a subsequent ischemic insult (heart attack) (Das and Das, 2008). A novel treatment strategy to counter ischemic cardiac disease and stroke developed which was termed ischemic pre-conditioning IPC) (Koch et al, 2014). IPC rests on the basic premise that brief durations of ischemia induce intrinsic cellular defense systems, and lead to the tolerance of vital organs to subsequent more severe ischemia. In direct pre-conditioning, for example, brief occlusions of a coronary or carotid artery protects the heart or the brain from longer durations of ischemia and reduces the resulting infarct volumes after heart attack or stroke. In remote IPC, brief ischemia is deliberately induced in one organ, typically an easily accessible tissue such as a limb, and subsequently protects a distant organ (typically the brain or the heart) from more severe ischemia. This concept was originally discovered using a rabbit heart attack model, when an initial episode of mild ischemia followed by reperfusion made the heart more resistant to a subsequent lethal ischemic insult (Murry et al, 1986). Since then IPC has become a powerful experimental technique to combat ischemic insults in most organs, and has been extensively studied in many laboratories around the world (Narayanan et al, 2013).
In the brain, IPC responses are found to occur in specific time windows consisting of an early and a late phase. The early phase, if the duration between the initial pre-conditioning and the final insult is around one hour, has the maximum protective effect (Narayanan et al, 2013). In the second (late) phase the combination of released factors and activated pathways results in delayed pre-conditioning. If this late phase is extended to several days after the preconditioning insult, it may provide more robust and longer lasting neuroprotection than the early phase.
MECHANISMS OF IPC
Previously it was believed that the mechanism of IPC was mediated by increased flow through the coronary collateral circulation. However, this theory was abandoned after a study by Murray et al (1986) in which they measured tissue radioactivity after injecting radiolabeled microspheres into the left atrial chamber of the dog heart and found no difference in collateral blood flow between preconditioned dogs and controls. IPC represents a complex molecular process that is rapidly generated and multi-factorial in nature, that is then transduced into an intracellular message and amplified to produce the final effector mechanism (Hawaleshka and Jacobsohn, 1998). In the response to a brief duration of sublethal ischemic insults, the receptors of the triggering factors may activate signaling pathways which consist of a well-organized series of events. These delayed pre-conditioning-activated signaling pathways govern gene expression resulting in cells expressing a phenotype that is highly resistant to ischemic insults. Locally released agonists such as adenosine, bradykinin, catecholamines and opioids activate the protective response through various G- protein coupled receptors which, when stimulated, increase activity of phospholipases C and D (Yang et al, 2010b). Kinases such as protein kinase C tyrosine kinase p38MAPKinase contribute in the signaling pathway. Ytrehus et al (1994) demonstrated inhibition of the protection obtained after IPC by administration of PKC inhibitors in the rabbit heart model. It has also been shown that among the PKC isozymes, the PKC-ε isoenzyme specifically provides cardioprotection against ischemia/ reperfusion damage (Dorn et al, 1999; Ping et al, 2002). Consistently, in all the animal models tested including mice, rats, rabbits and pigs the PKC-ε-selective activating peptide protected hearts, and inhibitors of PKC-ε inhibited ischemic/hypoxic or pharmacologic preconditioning (Yang et al, 2010b). On the other hand, deletion of cardiac PKC-ε resulted in failure to decrease infarct size in mice, suggesting a major role of PKC-ε activation in IPC (Inagaki et al, 2005). Another hypothesis for the underlying mechanism of IPC is by production of an inducible 70 kD stress or heat shock protein (HSP70i) (Currie et al, 1987). A variety of stimuli (including haemodynamic overload, myocyte stretch, hypertension, ischaemia, exercise, and oxidative stress) lead to an increase in the synthesis of HSP70i. After 24 hours of IPC, it has been shown that the levels of HSP70i were increased, and notably over-expression of the HSP70i gene in transgenic mice decreased the infarct size and improved myocardial recovery (Marber et al, 1995; Yellon et al, 1992). The mechanism of cardioprotection in IPC by HSP70i could be due to identification of damaged proteins and synthesis of new proteins under myocardial stress (Richard et al, 1996).
Another primary mediator in IPC could be activation of hypoxia inducible factor 1α (HIF1α) which is the means by which tissue and cells can sense hypoxia. An autonomous adaptive response by the cell to chronic hypoxia regulated by HIF-1 is decreased mitochondrial mass and/ or metabolism. Fibroblasts deficient in HIF-1 when exposed to chronic hypoxia generate high levels of reactive oxygen species (ROS), leading to cell death (Semenza et al, 2011). The HIF-1α gene has been shown to produce cardioprotective effects during preconditioning by stimulating the secretion of the hormone erythropoietin (EPO) in the kidney (Wang and Semenza, 1995). HIF-1α knock-out (KO) mice exposed to alternating cycles of ambient hypoxia and re-oxygenation failed to develop the protective cardiac preconditioning as compared to the wild type mice (Cai et al, 2003). Direct administration of EPO to HIF-1α KO mice into perfused hearts resulted in acute protection against ischemia-reperfusion injury (Cai and Semenza, 2004). The levels of adenosine increased in the hearts subjected to ischemic pre-conditioning, and blocking the adenosine receptor inhibited the protective effects of ischemic preconditioning, whereas adenosine receptor agonists protected the heart (Marber et al, 1993; Thornton et al, 1992). The HIF-1 gene induced CD73, the enzyme that produces adenosine thus indirectly helping in ischemic preconditioning. On the other hand, HIF-1-regulates mitochondrial metabolism thus contributing to the protective effects of ischemic preconditioning (Thornton et al, 1992). Increased plasma IL-10 levels and decreased myocardial infarct size are observed in remote ischemic preconditioned wild type mice, but could not be demonstrated in HIF-1α KO mice. Injecting the active form of HIF-1α into the mouse hind limb muscle significantly increased plasma IL-10 levels and decreasedd myocardial infarct size. In conclusion, HIF-1 plays a major role in IPC thus protecting the myocardium (Cai et al, 2013).
OTHER MODALITIES FOR PRE-CONDITIONING
In addition to ischemic preconditioning, other effective preconditioning stimuli and regimens have been reported. These are both numerous and diverse (Stetler et al, 2014), including hypothermia, hyperthermia, physical exercise, hyperbaric oxygen, exposure to neurotoxins and pharmacological agents such as resveratrol, volatile anesthetics and xenon, and even the mild inflammation produced by endotoxin.
In aged animals, there is decrease in ischemic tolerance leading to the impairment of the cardioprotective effect of IPC. Heat preconditioning (HP) protects myocardial injury induced by 8 weeks of overload training. HP enables adjustment of the activities of creatine kinase (CK), catalase (CAT) and superoxide dismutase (SOD), regulates the expressions of apoptosis-related genes (Bcl-2, Bax, caspase-3, caspase-9),and adhesion molecules (ICAM-1, VCAM-1, and PECAM-1) resulting in enhanced capacity against overload training induced injury and reduced damage to the myocardium (Yang et al, 2013). Hypothermic preconditioning is induced by rapid cycles of cooling and warming which produces sub-lethal stress to the brain’s microenvironment leading to the protection of the CNS from successive insults such as cerebral ischemia (Katz et al, 2009). Exercise preconditioning lessens the harmful effect of ischemia/ reperfusion injury by increasing the expression of neurotrophic factors, the extracellular matrix (ECM) proteins, integrins, angiogenic factors, heat shock proteins (Hsp-70) and decreasing expression of matrix metalloproteinase (MMP-9) and Toll-like receptor-4. Chronic exercise pre-conditioning increases cerebral metabolism following periods of hypoxia by increasing the neuronal response by increasing ATP production (Ding et al, 2005; Dornbos and Ding, 2012).
Pre-conditioning with hyperbaric oxygen reduces the number of apoptotic neural cells and promotes the nerve functional recovery in rats after spinal cord injury (Lu et al, 2013). Additionally, pre-conditioning coronary artery disease patients with hyperbaric oxygen prior to on-pump cardiopulmonary bypass or coronary artery bypass graft (CABG) surgery was capable of improving left ventricular function, and resulted in reduced myocardial injury, less intraoperative blood loss, shorter length of stay in ICU, fewer postoperative complications, and saved on patient costs, post-CABG (Yogaratnam et al, 2010). Lifelong caloric restriction has been found to modify the physiological and pathophysiological changes induced by aging and also increases life expectancy. In aged rats short term caloric restriction was found to provide cardioprotection by improving ischemic tolerance (Shinmura et al, 2005). Pre-treatment with perivascular or intramuscular neurotoxins such as botulinum toxin A (Btx-A) has a potential role in ischemic preconditioning of muscle flaps which is believed to be achieved through the release of substance P, calcitonin gene-related peptide (CGRP), and vascular endothelial growth factor (VEGF) (Akcal et al, 2013). Preconditioning with resveratrol has been found to induce brain tolerance against ischemia by suppression of the inflammatory response via regulation of NF-κB, COX-2 and iNOS (Simao et al, 2012). The administration of volatile anesthetics before a prolonged ischemic episode is known as anesthetic preconditioning, which has been developed as a cardioprotective modality in both animal and humans coronary disease models (Muntean et al, 2013). Moreover, pre-conditioning with anesthetics such as xenon gas can also exert neuroprotective and cardioprotective effects in different models suggesting the use of xenon preconditioning as an alternative strategy for the prevention of various neurological and cardiovascular diseases (Liu et al, 2013). Studies have shown that lipopolysaccharide (LPS) from Gram-negative bacteria used in pre-conditioning provides neuroprotection against successive cerebral ischemic injury. LPS activates Toll-like receptors and causes systemic inflammation. After stroke, mice pre-conditioned with LPS had a significant decrease in the levels of TNF-alpha, neuronal TNF-receptor 1 (TNFR1), and TNFR-associated death domain (TRADD). Additionally, to establish LPS-induced tolerance before ischemia, it was necessary to show upregulation of TNF-alpha, whereas if LPS was given during ischemia then suppression of TNF-alpha signaling was associated with neuroprotection after LPS preconditioning (Rosenzweig et al, 2007).
LOW-LEVEL LASER (LIGHT) THERAPY (LLLT)
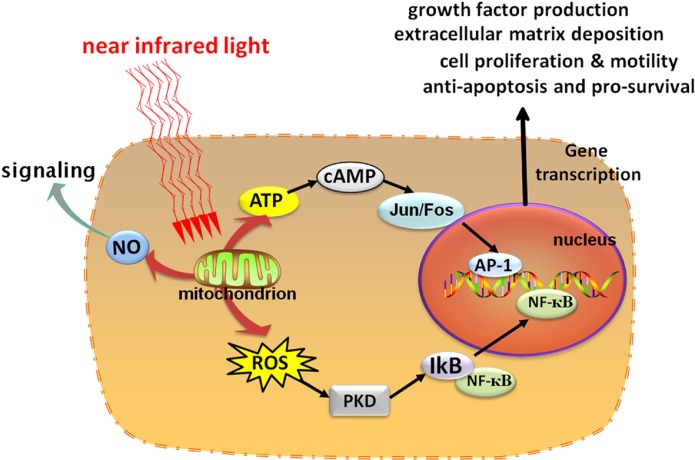
Low-level laser (light) therapy or photobiomodulation has been known for almost 50 years since shortly after the discovery of lasers in 1960. For many years it was thought that there was something special about laser light, but it is now known that many wavelengths of visible light (especially in the red or near-infrared regions) produced from LEDs or other light sources are equally effective. Longer red and near-infrared wavelengths are much better at penetrating tissue than shorter blue/ green wavelengths and are therefore preferred clinically. Although the mechanism of action of LLLT is still not fully understood, much information is now known concerning the primary photoacceptor molecules, the immediate effectors, the signaling pathways, transcription factor activation and short and long-term effects of LLLT on cells and tissues. The most important photoacceptor is accepted to be cytochrome c oxidase (unit IV in the mitochondrial respiratory chain). Increase in mitochondrial membrane potential, oxygen consumption and ATP production are rapidly observed. Nitric oxide and ROS are transiently produced. Cyclic AMP is involved in signaling pathways. Transcription factors such as NF-kB are activated. Anti-apoptotic proteins, heat shock proteins, anti-oxidant defense pathways and anti-inflammatory cytokines are increased. In the long-term parameters involved in healing such as cell migration, cell adhesion, protein synthesis, and DNA synthesis are stimulated (Fig. 1) (Huang et al, 2011).
FIG. 1.
Schematic depiction of the cellular signaling pathways triggered by LLLT. After photons are absorbed by chromophores in the mitochondria, respiration and ATP is increased but in addition signaling molecules such as reactive oxygen species (ROS) and nitric oxide (NO) are also produced (with permission from Huang et al, 2011).
SIMILARITIES BETWEEN IPC AND LLLT
Part of the aim of this review is to draw comparisons between the hypothesized mechanisms of IPC and those of LLLT when used in a pre-conditioning or post-injury mode. It is in fact quite remarkable how many similarities that can be found between the two modalities, although of course, there are also some differences (Table 1). One of the unifying mechanisms is hypothesized to be at the level of mitochondria (Dirnagl and Meisel, 2008).
TABLE 1.
Comparison of cellular and molecular mechanisms that have been proposed for ischemic preconditioning and for LLLT.
| Pathway or mediator | IPC | LLLT |
|---|---|---|
| Inflammation | Reduction in vascular permeability and neutrophil migration in rat paw (Souza Filho et al, 2009) | Reduction of COX-2 mRNA in rat paw and brain (Prianti et al, 2014) |
| Cytokines (IL10) | Upregulation of IL10 in plasma and heart (Cai et al, 2012) | Reduced IL6 and TNFa and increased IL10 in muscle of rats with heart failure (Hentschke et al, 2013) |
| Nitric oxide | Increases circulating nitrites in mice (Rassaf et al, 2014) | Increases venous nitric oxide levels (Mitchell and Mack, 2013) |
| Oxidative stress | Reduction of oxidative stress and downregulation of COX-2 expression in rat kidney (Sedaghat et al, 2013) | Reduction of oxidative and nitrative stress in injured tibialis anterior muscle in rat (Assis et al, 2012) |
| Heat shock proteins | Over-expression of the HSP70i gene in transgenic mice limits infarct size and improves post-ischaemic contractile recovery (Marber et al, 1995) | Decrease in the HSP27 phosphorylation thereby decreasing NF-κB p65 translocation human gingival fibroblast cells. (Lim et al, 2013) |
| MAP tyrosine kinase | Phosphorylation of p38 MAPK via mitochondria translocation of Bcl-xL in mice brain (Zhao et al, 2013) | Increase in MAP kinase activity leading to increase in VEGF levels in human granulosa cells. (Kawano et al, 2012) |
| Kinin receptors | Down-regulates kinin receptor expression in neutrophils of patients undergoing heart surgery. (Saxena et al, 2013) | Downregulates kinin receptors mRNA expression in the subplantar muscle of rat paw (Bortone et al, 2008) |
| Ion channels | K(+) inward rectifier subunits of the 6.2 isotype (Kir6.2) required for cardioprotection (Wojtovich et al, 2013) | Increased Ca2+ in oral tissue cells via TRPC1 ion channels (Chellini et al, 2010) |
| Cyclic AMP/CREB | CREB phosphorylation contributes to triggering preconditioning in rat heart (Marais et al, 2008) | Promotes the proliferation and osteogenic differentiation of human periodontal ligament cells via cyclic AMP (Wu et al, 2013) |
| VEGF | Overexpression of VEGF protects intrahepatic cholangiocytes after liver transplantation in rats (Zhou et al, 2010) | Increases angiogenesis in a model of ischemic skin flap in rats mediated by VEGF, HIF-1α and MMP-2 (Cury et al, 2013) |
| Hypoxia-inducible factor 1 | HIF-1 activates IL10 gene transcription and is required for remote ischemic preconditioning in mice heart (Cai et al, 2013; Kalakech et al, 2013) | Reduces the overexpressions of HIF-1α, TNF-α, and IL-1β, and increases the amounts of VEGF, NGF, and S100 proteins in rats with chronic constriction injury (Hsieh et al, 2012) |
| TFG-beta | Downregulated TGFβ1 but upregulated smad7 in cardiac infarcts (Wang et al, 2013) | Increase in proliferation of vascular endothelial cells and decrease in VEGF and TGF-beta secretion. (Szymanska et al, 2013) |
| Vasodilation and endothelial function | Prevents reduction in brachial artery flow-mediated dilation after strenuous exercise in humans. (Bailey et al, 2012) | Rapid increase of human skin microcirculation at the local and systemic levels(Samoilova et al, 2008) |
| Hemostasis | Prevents systemic platelet activation in humans (Pedersen et al, 2011) | Speeds wound healing in hemophilia by enhancing platelet procoagulant activity in humans (Hoffman and Monroe, 2012) |
| Stem cell activation | Increases the intestinal stem cell activities in the intestinal crypts in mice (Chen et al, 2014) | Induces autologous bone-marrow-derived mesenchymal stem cells in infarcted heart of rats (Tuby et al, 2011) |
SKELETAL MUSCLE PRE-CONDITIONING THROUGH LIGHT
In acute conditions, skeletal muscle fatigue impairs the strength of muscle, its motor coordination and decreases the capacity of the muscle to perform work (Allen et al, 2008). Overall fatigue decreases the muscle function, which is believed to be a result of metabolic changes such as depletion of ATP and glycogen, oxidative stress, tissue hypoxia and blood acidification. Phototherapy has been shown to improve muscle fatigue and exhaustion (Leal Junior et al, 2010a). Studies have shown that pre-treatment with specific doses of phototherapy decreases inflammatory biomarkers and lactate levels in blood after strenuous upper and lower extremity exercise (Lopes-Martins et al, 2006b). On the basis of these studies we can infer that pre-conditioning the muscle with phototherapy increases the contractile function, decreases exercise induced muscle fatigue and improvement in post exercise recovery. We have tabulated the experimental studies on animals in Table 2, and the clinical studies in humans in Table 3.
TABLE 2.
LLLT preconditioning and exercise: experimental animal models (adapted from Ferraresi et al 2012).
| Ref | Light source/Wavelength | Spot area (cm2) | Performance characteristics | Muscle(s) | Exercise |
|---|---|---|---|---|---|
| Lopes-Martins et al, 2006a | Laser 655 nm | 0.08 |
|
Tibialis anterior | Neuromuscular electric stimulation |
| Leal Junior et al, 2009c | Laser 904 nm | 0.2 |
|
Tibialis anterior | Neuromuscular electric stimulation |
| de Almeida et al, 2011 | Laser 904 nm | 0.2 |
|
Tibialis anterior | Neuromuscular electric stimulation |
TABLE 3.
LLLT preconditioning and exercise: clinical studies in humans (adapted from Ferraresi et al 2012).
| Ref | Light source/Wavelength | Spot area (cm2) | Performance characteristics | Muscle(s) | Exercise |
|---|---|---|---|---|---|
| de Almeida et al, 2012 | Laser 660nm versus 830nm | 0.0028 |
|
Biceps brachii | MVC per 60 s in Scott bench |
| Gorgey et al, 2008 | Cluster with 4 diodes laser 808 nm |
|
Femoral quadriceps | Isokinetic dynamometer | |
| Leal Junior et al, 2008 | Laser 655 nm | 0.01 |
|
Biceps brachii | Scott bench |
| Leal Junior et al, 2009e | Laser 830 nm | 0.0028 |
|
Biceps brachii | Scott bench |
| Leal Junior et al, 2010b | Cluster with 5 diodes laser 810 nm | 0.0364 |
|
Biceps brachii | Scott bench |
| Baroni et al, 2010a | Cluster with 5 diodes laser 810 nm | 0.029 |
|
Femoral quadriceps | Isokinetic dynamometer |
| De Marchi et al, 2012 | Cluster with 5 laser diodes 810 nm | 0.0364 |
|
Femoral quadriceps, hamstrings, gastrocnemius | Running on treadmill until exhaustion |
| Leal Junior et al, 2009a | Laser 810 nm versus Cluster with 69 LEDs 660/850 nm |
0.036 versus 0.2 |
versus
|
Rectus femoris | Wingate test |
| Leal Junior et al, 2009d | Cluster with 69 LEDs 660/850 nm | 0.2 |
|
Biceps brachii | Scott bench |
| Baroni et al, 2010b | Cluster with 69 LEDs 660/850 nm | 0.2 cm2 |
|
Femoral quadriceps | Isokinetic dynamometer |
There are two different phases of exercise-induced damage; one is primary and the other is secondary. The primary phase of muscle damage results from direct exercise-induced mechanical stress, and secondary muscle damage is due to the inflammatory response that follows after mechanical stress (Merrick et al, 2002). Pre-conditioning with phototherapy protects the muscle both from primary as well as secondary damage, while phototherapy when administered after injury protects cells from secondary damage only. In vivo studies have shown that pre-conditioning phototherapy to injured muscles produces anti-inflammatory and antioxidant effects, thus protecting the muscle from secondary damage (Avni et al, 2005).
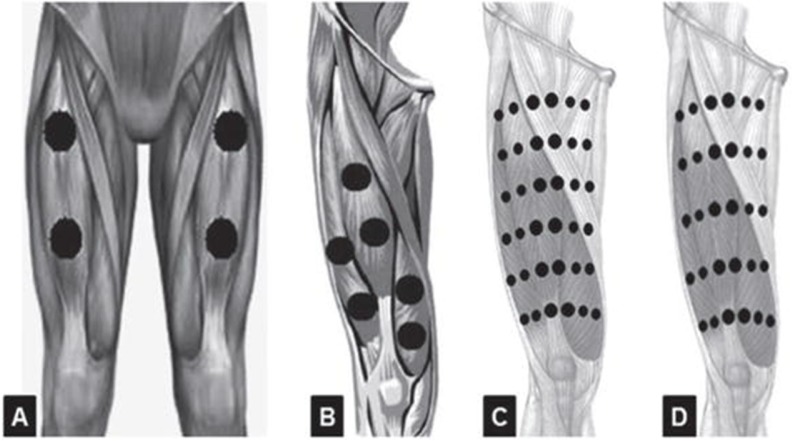
An important parameter for the muscle to receive the optimum irradiation is that the irradiation points should be designed to cover the largest area and to better distribute the energy applied over the muscles (Ferraresi et al, 2012). Figures 2 and 3 demonstrate some examples of the number and distribution of the irradiation points on femoral quadriceps using LLLT or LEDT (Ferraresi et al, 2012).
FIG. 2.
Number of LLLT or LEDT radiation points applied on femoral quadriceps muscle (with permission from Ferraresi et al, 2012).
FIG. 3.
LEDT array on a patient to improve muscle function (with permission from Ferraresi and Parizotto, 2014).
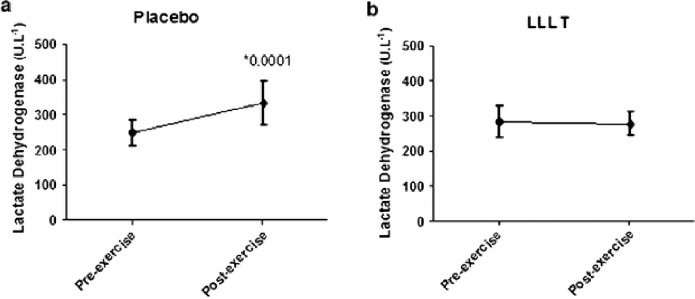
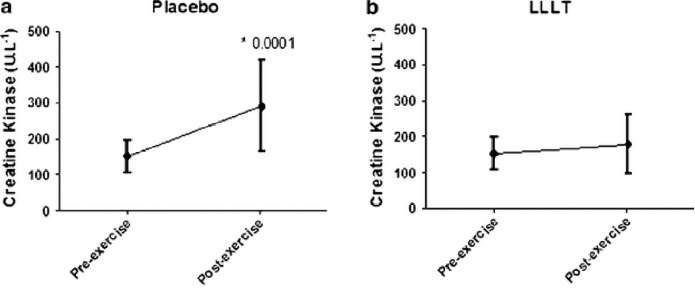
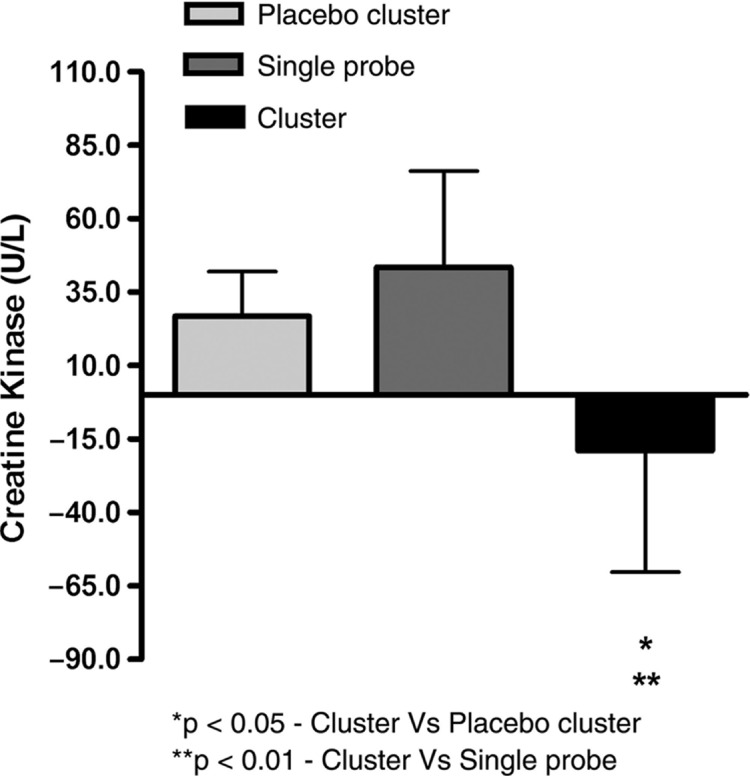
Studies support that hypothesis that the ergogenic effects of phototherapy on muscles are due to increases in intramuscular microcirculation (Baroni et al, 2010b), decreases in the lactic acid production (Leal Junior et al, 2009a; Leal Junior et al, 2009b; Leal Junior et al, 2009d), improved mitochondrial function, improved antioxidant ability of the exercising muscles, enhanced contractile function, prevention of exercise-induced cell damage, and improved post exercise recovery of strength and function (Borsa et al, 2013; Leal Junior et al, 2011; Leal Junior et al, 2010b). Borsa et al (2013) critically evaluated these studies that address the ability of phototherapeutic devices, such as lasers and light-emitting diodes (LEDs), to improve skeletal muscle contractile function, decrease exercise-induced muscle fatigue, and enable post exercise recovery. The main outcome measures included total lapsed time to fatigue, number of repetitions to fatigue, overall work performed, maximal voluntary isometric contraction (strength), electromyographic activity, and post exercise biomarker levels. Ten of the randomized controlled trials met the inclusion criteria including 32 data sets. In all the studies Borsa et. al (2013) consistently found that phototherapy administered before resistance exercise provided ergogenic and prophylactic benefits to skeletal muscle. A randomized double-blind study using 22 untrained male volunteers was performed by De Marchi et al (2011). The subjects were pre-conditioned with LLLT for 30 seconds on a lower-limb before a standardized exercise protocol on a treadmill until they were exhausted. Figures 4 and 5 show pre- and post-exercise activity of markers of muscle damage-lactate dehydrogenase (LDH) and creatine kinase (CK). De Marchi et al (2011) observed that preconditioning with LLLT before exercise also decreased the post-exercise improvement in the activities of CK and LDH compared to the placebo group, demonstrating that pre-treatment with LLLT can protect skeletal muscle against exercise-induced damage in long-duration exercises (De Marchi et al, 2012). In a similar study, male volleyball players were preconditioned with: 1) an active LEDT cluster-probe (660nm=10mW, 850nm=30mW); (2) a placebo cluster-probe with no output; and (3) a single-diode 810-nm 200-mW laser on the rectus femoris muscle and then asked to perform three Wingate cycle tests. It was observed that post exercise CK levels were significantly lower in active LEDT cluster group compared to the placebo cluster or single diode group (Leal Junior et al, 2009a). (Fig. 6).
FIG. 4.
Pre and post-exercise activity of lactate dehydrogenase (a) in placebo (b) and in active LLLT group. * Statistical significance from pre to post-exercise levels using the software SPSS 18.0 for Windows (with permission from De Marchi et al, 2012).
FIG. 5.
Pre and post-exercise activity of creatine kinase (a) in placebo (b) and in active LLLT group. * Statistical significance from pre to post-exercise levels using the software SPSS 18.0 for Windows (with permission from De Marchi et al, 2012).
FIG. 6.
Changes in creatine kinase levels post-exercise. (with permission from Leal Junior et al, 2009a).
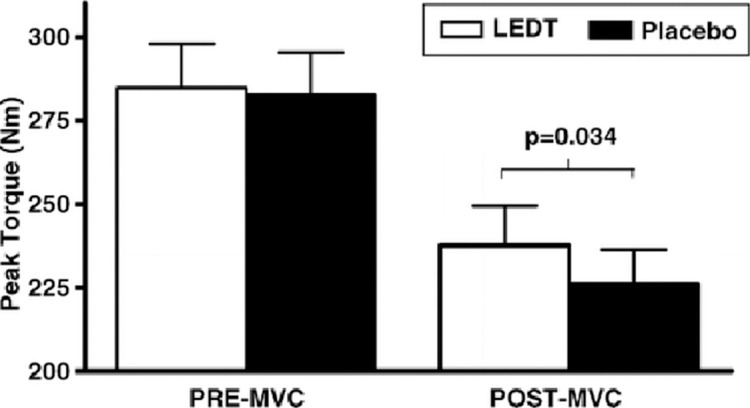
Conversely, preconditioning with LEDT in healthy male volunteers did not improve maximal knee extensor muscle isokinetic voluntary contractions (preMVC) performed before the isokinetic fatigue test as compared to the placebo group. However, higher torque values were observed in post-MVC subjects pre-conditioned with LEDT compared to the placebo group (Fig. 7). Pretreatment with LEDT increased the exercise performance by producing higher isometric torque after high-intensity concentric isokinetic exercise (Baroni et al, 2010b).
FIG. 7.
PRE-MVC and POST-MVC knee-extensor maximal isometric torques for LEDT and placebo treatments (mean SEM). The bracket indicates differences in POST-MVC between LEDT and placebo (p value above the bracket) (with permission from Baroni et al, 2010b).
Leal-Junior et al. (2013) performed a meta-analysis to explore the effects of phototherapy applied before, during and after exercises. Data from thirteen randomized control trial was assessed based on number of repetitions and time until exhaustion for muscle performance, and creatinine kinase (CK) activity to assess risk for exercise-induced muscle damage. The meta-analysis concluded that preconditioning skeletal muscles with phototherapy (lasers and LEDs) improves muscular performance and accelerates recovery.
PRECONDITIONING WITH LIGHT IMPROVES INFLAMMATION AND ANALGESIC EFFECT:
In temporomandibular joint disorders pain is usually a very common finding. Barretto et al (2013) studied the effect of LLLT of pain and inflammation on temporomandibular joint (TMJ) of rats. For the assessment of nociceptive response, formalin and carrageenan was injected to induce inflammation into the rat TMJ. Nociceptive responses were quantified by assessing behavioral responses categorized by rubbing the orofacial region and flinching the head quickly. Barretto et al observed that rats pretreated with diclofenac sodium- (10mg/kg i.p.) and LLLT infrared (LST group, 780nm, 70mW, 30s, 2.1J, 52.5J/cm2 GaAlAs), significantly reduced the formalin nociceptive response. The inflammatory response was assessed by the presence of infiltrate rich in neutrophils, presence of liquefactive necrosis and interstitial edema, hemorrhagic areas, or enlargement of the joint space on the region on histomorphological analysis. The rats pretreated with LLLT showed significantly less inflammatory response and exuberant granulation tissue with higher vascularization, and formation of new collagen fibers. The study established that the pre-conditioning LLLT had an anti-nociceptive and anti-inflammatory effect on the inflammation induced in the TMJ of rodents (Barretto et al, 2013).
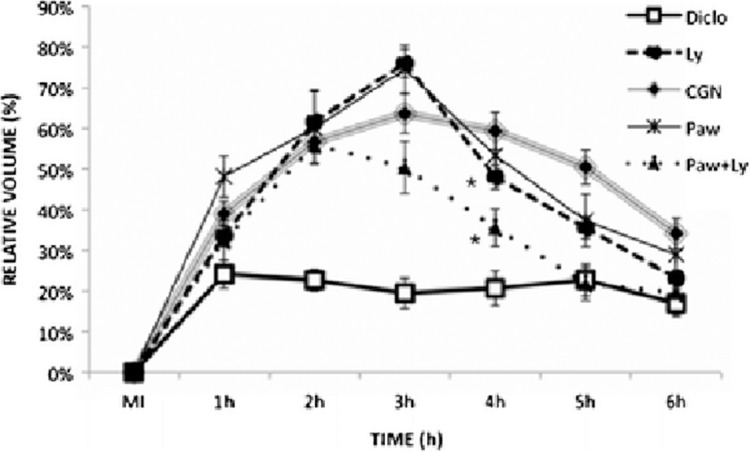
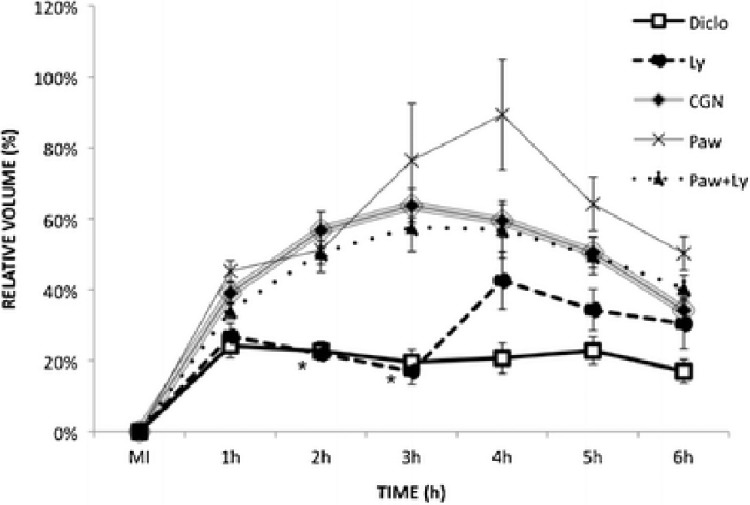
A similar study, revealing the effect of infrared LLLT in modulating the inflammatory processes and immunological responses was done on the mouse hind paw. Carrageenan inoculation (CGN) was used to induce edema into the plantar surface of left hind paw in male mice. Animals were distributed into five groups: CGN (control), no treatment; Diclo: sodium diclofenac; Paw: LLLT on the paw; Ly: LLLT on the inguinal lymph nodes; and Paw+Ly: LLLT on both paw and lymph nodes. They were also subdivided by time of irradiation: A: 1 h and 2 h before CGN, B: 1 h and immediately before CGN, C: 1 and 2 h after CGN, and D: 3.5 and 4.5 h after CGN. Figure 8 shows the average of the volume of edema at time A. The mice were preconditioned with irradiation 2 and 1 h before edema induction. The study showed that on the second hour there was a steady decrease in the edema of Paw+Ly group and was lower than all irradiated groups on the fourth hour, suggesting that Paw+Ly group was the best irradiation option when preconditioned 2 and 1 h before edema induction. Figure 9 shows the average of the volume of edema at time B where preconditioning was performed 1 h and immediately before the edema induction. The Ly group was the best group to prevent edema 1 h and immediately before the induction and the edema reduction was 44.9 % compared with CGN group. These results suggest that LLLT preconditioning produced both anti-inflammatory and pro-inflammatory effects depending on to the site and timing of irradiation (Meneguzzo et al, 2013).
FIG. 8.
Average of the volume of edema (%) ± SEM of moment A.. The irradiation was carried out 2 and 1 h before the injection of carrageenan (MI); N=27 for CGN group, N=13 for Diclo group, and N=5 for the other groups. Statistically significant differences compared to CGN are indicated by an asterisk (p<0.05) (with permission from Meneguzzo et al, 2013).
FIG. 9.
Average of the volume of edema (%) ± SEM of moment B. The irradiation was carried 1 h and immediately before the injection of carrageenan at the initial moment (MI); N=27 for CGN group, N=13 for Diclo group, and N=5 for the other groups. Statistically significant differences compared to CGN are indicated by an asterisk (p<0.05) (with permission from Meneguzzo et al, 2013).
LLLT has been implicated in reducing neuropathic pain by releasing local neurotransmitters such as serotonin (Walker, 1983), stimulating release of endorphins and reduce inflammatory cells (Albertini et al, 2007; Hagiwara et al, 2008; Lopes-Martins et al, 2005). As a mode of peripheral endogenous opioid analgesia the opioid-containing immune cells migrate to inflamed sites and release endorphins to inhibit pain. Pre-conditioning of blood by LLLT on peripheral endogenous opioid analgesia in a rat model of inflamed paw tissue induces analgesia that was transiently antagonized by naloxone. An upregulation in the expression of peripheral opioid precursor including endorphin precursors, pro-opiomelanocortin and corticotrophin releasing factor in blood cells, suggesting a direct induction by LLLT to mediate analgesia (Hagiwara et al, 2008).
Another study, exploring the effect of pre-conditioning with light to reduce pain in rats was done by Yang et al (2010) They pre-treated with 650 nm laser and moxibustion in a visceral traction pain model of rats. The differences in the pain score, systolic pressure, activity of acetylcholinesterase (AChE), leuenkephaline (LEK) and the positive index of c-Fos protein and glial fibrillary acidic protein (GFAP) were assessed. The study determined that the pre-conditioned group that received both laser at 650nm and moxibustion could inhibit visceral traction pain by a mechanism that might be due to decreased activity of AChE and increasing the activity of LEK thereby decreasing the expression of c-Fos protein and GFAP.
The above studies suggest that preconditioning with phototherapy might reduce pain and inflammation. Although the mechanism needs to be further explored, this gives us a hope in clinical practice for various pain syndromes.
PRE-CONDITIONING WITH LLLT REDUCES DAMAGE AFTER HEART ATTACK
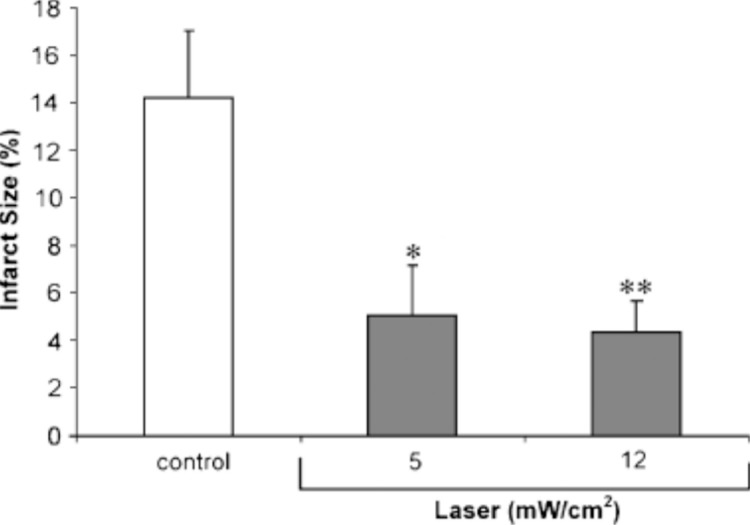
The beneficial effect of LLLT has been revealed in the infarcted heart model of rat and dogs, which showed reduction of 50–70% of infarct size in 4–6 weeks post-infarction (Oron et al, 2001a; Oron et al, 2001b; Yaakobi et al, 2001). A study by Tuby et al. (2006) irradiated the heart with LLLT in both intact and post infarction rats. Myocardial infarction (MI) was induced by occlusion of left anterior descending artery. The myocardium was irradiated for 2 minutes at an energy density of 5 and 12 mW/cm2. The myocardium was preconditioned with LLLT and after 7 days myocardial infarction was induced and rats were euthanized 21 days post- infarction. Figure 10 shows the effect of preconditioning with LLLT before MI, on the development of infarct post-MI. Tuby et al. (2006) observed that laser irradiated pre-conditioned hearts with a power density of 5 mW/cm2 had 64% less infarct size, and with a power density of 12 mW/cm2 had 69% less infarct size compared to those that were not pre-conditioned with laser irradiation prior to MI-induction. The expression of genes (iNOS and vascular endothelial growth factor (VEGF)) responsible for angiogenesis and cardioprotection was analyzed. Pre-conditioning the heart with laser treatment before infarction induction resulted in increased levels of VEGF and iNOS in the rats heart and a significant reduction in the infarct size Another study by Zhang et al. (2010) studied the effect of pre-conditioning the infarcted myocardium with LLLT prior to cell transplantation. Cell transplantation has emerged as a promising method to regenerate the injured myocardium. Myocardium infarction was induced by ligating the left anterior descending artery of rats. Pre-conditioning to the myocardium was performed by irradiating with a 635 nm, 5mW diode laser and energy density of 0.96 J/cm2 for 150 seconds. After three weeks bone marrow mesenchymal stem cells (BMSCs) were injected to rats with or without LLLT pretreatment. After LLLT pre-conditioning, there was an increased protein and mRNA expression of VEGF, glucose-regulated protein 78 (GRP78) as well as increased activity of superoxide dismutase (SOD) activity and reduced malondialdehyde (MDA) in the infarcted myocardium. LLLT pre-conditioning increased cell survival rate, decreased apoptotic percentage of implanted BMSCs and increased angiogenesis. The study established that LLLT pre-conditioning is a unique non-invasive approach for cell transplantation given intraoperatively to augment cell survival and therapeutic prospective (Zhang et al, 2010).
FIG. 10.
Effect of LLLT, 7 days before myocardial infarction, on the reduction of infarct size. Infarct size was measured as the relation between the infarct size area to the left ventricle area. Each column represents meanSEM of 7–9 rats; *P<0.05 or **P<0.01. (with permission from Tuby et al, 2006).
PRECONDITIONING WITH LLLT PROTECTS CELLS FROM TOXINS
In mammalian tissues, the major photoacceptor molecules that absorb light in NIR range are hemoglobin, myoglobin, and cytochrome c oxidase (Liu et al, 2001). Among these, only cytochrome c oxidase (EC 1.9.3.1) has been implicated in the production of energy. Cytochrome c oxidase, a photoacceptor in the NIR range plays an important role in therapeutic photobiomodulation (Liu et al, 2001). In primary neuronal cultures, light-emitting diode (LED) 670 nm in NIR range was able to reverse the effect of tetrodotoxin (TTX) (voltage-dependent sodium channel blocker), that impedes neuronal impulse activity, decreases ATP demand, and down-regulates cytochrome c oxidase activity. The treatment with LED 670 nm not only brought the cytochrome c oxidase back to control levels but it also up-regulated enzyme activity of normal neurons above control levels (Liu et al, 2001). LED treatments improved the retinal function in rats intoxicated with methanol, by removing the inhibition by formic acid on cytochrome c oxidase (Eells et al, 2003) and thus plays a significant role in the therapeutic process of photobiomodulation. Based on these findings, Wong-Riley et al. (2005) studied the effect of the cytochrome c oxidase inhibitor, potassium cyanide (KCN) on primary cultured neurons, and whether KCN could reduce the beneficial effect of LED treatment. They observed that LED treatment partially restored cytochrome c oxidase activity which had been blocked by KCN (10–100 microM) and significantly reduced neuronal cell death (induced by KCN at 300 microM) from 83.6 to 43.5%. Pre-conditioning of the primary neuronal cells enhanced the protective action of LED during KCN exposure (10–100 microM). LED pre-treatment for 10 min using a total energy density of 30 J/cm2, effectively reduced the number of neurons exhibiting cell death after 300 μM KCN by 48%. Thus, pre-conditioning of neuronal cells by photobiomodulation upregulatd cytochrome c oxidase that led to increased energy metabolism in neurons and promoted wound healing and reversed intoxication by various toxins that inactivate cytochrome c oxidase in vitro (Wong-Riley et al, 2005).
Liang et al (2006) investigated the effect of NIR-LED preconditioning on primary neuronal cultures to test if it could inhibit apoptotic cell death induced by KCN. The primary neuronal cells were cultured from postnatal rat visual cortex and were pre-treated with LED for 10 min at a total energy density of 30 J/cm2 and were then exposed to potassium cyanide (100–300 μM) for 28 h. The neural cell death via apoptosis was confirmed by electron microscopy Hoechst 33258, single-stranded DNA, Bax, and active caspase-3. LED pre-conditioning applied to the primary neuronal cells significantly reduced apoptosis representing a 50.3% (100μM KCN) and 32.8% (300μM KCN) reduction. LED pretreatment significantly decreased the expression of KCN-induced caspase-3, reversed the increased expression of Bax and decreased expression of Bcl-2 to control levels. The study concluded that preconditioning with light partly protected the neuronal cells from cyanide by a mechanism most likely involving decrease of ROS production, down-regulation of pro-apoptotic proteins, activation of anti-apoptotic proteins and by increasing energy metabolism in the neurons (Liang et al, 2006).
PRECONDITIONING WITH LLLT IN WOUND HEALING:
The penetration of NIR is deeper than UV or visible light and is harmless to living tissue. Therapeutic devices using NIR have been successfully used for the treatment of various injuries chiefly infected, ischemic, and hypoxic wounds (Conlan et al, 1996; Sommer et al, 2001; Whelan et al, 2001). Studies have shown that NIR promotes wound healing but the mechanisms are poorly understood.
Wound healing is a complex process consisting of multiple physiologic events which are controlled by an infinite array of signaling mechanisms (Barolet and Boucher, 2010). Any dysregulation in the process of wound healing leads to abnormal scarring and might lead to the development of keloids and hypertrophic cells. Treatment of these abnormal scars is really challenging to the clinicians and is associated with low self-esteem and impaired quality of life in affected individuals. Recent studies have implicated a major role of transforming growth factor beta-I (TGF-β1) in the formation of hypertrophic scars and keloids. A higher expression or dysregulation of TGF-β1 is associated with fibroblastic proliferation and excess collagen deposition (Bouzari et al, 2007; Wolfram et al, 2009). LLLT with red light and with NIR has been shown to promote wound healing including modulation of TGF-β1 making it useful in scar treatment. A study by Barolet and Boucher (2010) used LED pre-conditioning with NIR irradiation (805nm at 30mW/cm2) for 30 days in three patients prone to development of hypertrophic scars or keloids after surgery. They found a significant improvement on the NIR-treated areas compared to the control scars. The enhancement of wound healing process might be due to modulation of proteins involved in regulation of wound repair such as TGF-β1 platelet- derived growth factor (PDGF), interleukins (IL-6, 13, 15) and matrix metalloproteinases (MMPs) by LLLT. However large patient samples are needed for evaluation of this promising technique.
PRECONDITIONING WITH LLLT IN THE CENTRAL NERVOUS SYSTEM
The function of brain is highly dependent on cerebral bloodflow (CBF) (Balestreri et al, 2006; Ling and Neal, 2005). In diseases with reduced CBF such as traumatic brain injury, degenerative diseases such as Parkinson’s disease and Alzheimer’s disease, improving the CBF has shown to not only prevent these diseases but also increases the response to treatment of these diseases (Uozumi et al, 2010; Wolfson et al, 1985). Photobiostimulation effects of near-infrared (NIR) laser irradiation is known for a long time. It has also been shown that NIR laser irradiation is very efficient for reducing cerebral ischemia both in vivo and clinically. The NIR laser irradiation produces vasodilatory effects which might be mediated by nitric oxide (NO) (Hamblin, 2008). Uozumi et al. (2010) studied the effect of 808 nm laser irradiation at three different power densities (0.8, 1.6 and 3.2 W/cm2) on CBF in mice and measured directly NO in brain tissue during NIR laser irradiation using an amperometric NO-selective electrode. They also observed the influence of NO and a neurotransmitter, glutamate, to the regulation of CBF by using a nitric oxide synthase (NOS) inhibitor, nitro-L-arginine methyl ester hydrochloride (L-NAME), and an N-methyl-D-aspartate (NMDA) receptor blocker, MK-801, respectively. The protective effect of NIR laser irradiation on transient cerebral ischemia using a model of transient bilateral common carotid artery occlusion (BCCAO) in mice was also investigated. As compared to the control mice, the NIR laser irradiation (1.6 W/cm2 for 15–45 minutes) significantly increased local CBF and cerebral NO concentration. Pre-conditioning the brain with NIR laser irradiation increased the residual CBF by 30% and significantly decreased the numbers of apoptotic cells in the hippocampus in mice subjected to BCCAO and neuronal damage. These results can be partly explained by the fact that pre-conditioning with NIR laser irradiation led to the alleviation of NO surge after the reperfusion.
Low-energy laser irradiation in the far red to NIR range not only has vasodilatory effects but can also modulate many biological processes (Karu, 1999) by increasing mitochondrial respiration, or ATP synthesis (Karu et al, 1995; Passarella et al, 1984) and promoting cell survival (Shefer et al, 2002). NIR is believed to accelerate the electron transfer in the respiratory chain and activation of photoacceptors, including cytochrome c oxidase in the mitochondria (Beauvoit et al, 1994). Cyanide toxicity has been implicated in the cell death by inhibition of cytochrome oxidase (Bhattacharya and Lakshmana Rao, 2001) and compromising energy status (Wong-Riley et al, 2005). Moreover, depending on the concentration (Prabhakaran et al, 2004) or the susceptibility of brain (Mills et al, 1999) cyanide produces neuronal cell deathy either by apoptosis or necrosis. Exposure to cyanide produces apoptosis in cortical neurons but necrosis in mesencephalic cells (Prabhakaran et al, 2002).
PRECONDITIONING WITH LLLT PROTECTS SKIN FROM ULTRAVIOLET DAMAGE
Ultraviolet radiation (UVA, 290–320nm; UVB 320–400 nm) exposure, damages skin cells which leads to photoaging and cancer of the skin (Calles et al, 2010; Sinha and Hader, 2002). The mechanisms responsible for the UV damage to the skin could be due to the breakdown of collagen fibers, free radical formation, DNA damage and inhibition of the immune system (Schroeder et al, 2010). The effects of UV radiation can be minimized by avoiding sun exposure or the use of sunscreens. Conversely, the exposure to sun cannot be avoided in certain occupations and the use of sunscreen has many limitations such as compliance, allergy and possible harmful effects of nanoparticles that are contained in most sunscreens (Kimura et al, 2012). Recent studies have shown protective effects of infrared (IR) exposure against harmful effects of UV-irradiation to the skin (Avci et al, 2013).
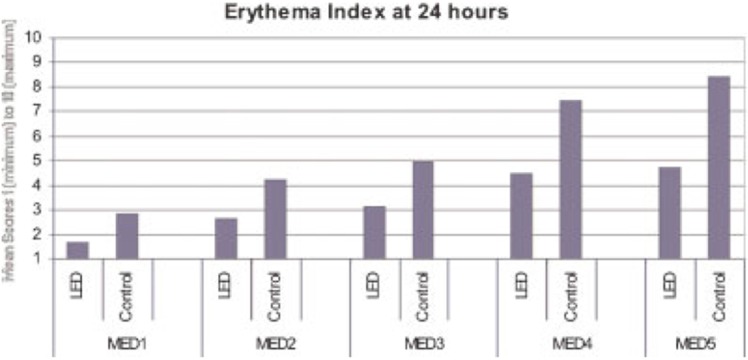
The light in the early morning is predominantly composed of visible and NIR solar wavelengths and at noon contains substantial amounts of UVB and UVA radiation, suggesting a natural defense mechanism for mammals that involves pre-conditioning the skin with red/NIR light in the morning to protect against impending damaging UV radiation later in the day (Barolet and Boucher, 2008). Patients with polymorphous light eruption (PLE) and healthy controls were exposed to LED treatments (660 nm) on experimental anterior thigh region. Baseline minimal erythema doses (MED) were then determined. UV radiation was thereafter performed on both experimental and control areas and digital pictures of the MED TEST areas were taken prior to UV exposure and immediately, 24 hours, 7 and 14 days after. Figures 11 and 12 show Erythema Index at 24 hours as a function of MEDs for the LED pre-treated and control sides. There was a significant reduction in UV-B induced erythema reaction in both control and patients suffering from PLE preconditioned with LED. Overall, less redness was observed for each tested MED on the preconditioned experimental areas compared to control areas. These results suggest that pre-conditioning with LED prior to UV exposure provided significant protection against UV-B induced erythema (Barolet and Boucher, 2008).
FIG. 11.
Mean scores for the Erythema Index at 24 hours as a function of MEDs for the LED pre-treated and CONTROL sides. (with permission from Barolet and Boucher, 2008).
FIG. 12.
MED responses for the LED and CONTROL side at 24 hours for MED 1–MED 5 for a subject from Group II. (with permission from Barolet and Boucher, 2008).
Illumination of the skin with non-coherent NIR (700– 2000 nm) generated a strong cellular defense mechanism against solar UV cytotoxicity and induced cellular mitosis without increasing skin temperature. This protection against UV light was long lasting (at least 24 hours) and cumulative effect (Menezes et al, 1998). Furthermore, another study proposed that pre-conditioning with IR radiation protects human fibroblasts cells by inducing resistance to the harmful effects of UV light by affecting the genes of mitochondrial apoptotic pathway. IR preconditioning of human fibroblasts inhibited UVB activation of caspase-9 and -3 and partial release of cytochrome c and Smac/Diablo, decrease proapoptotic proteins (ie, Bax), and increase antiapoptotic proteins (ie, Bcl-2 or Bcl-xL), contributing the cell to resist UVB-triggered apoptosis. This effect could be mediated most likely by the induction of heat shock protein Hsp27 by IR, which prevents apoptosome assembly (Frank et al, 2004).
CONCLUSION
Pre-conditioning regimens are increasingly receiving more attention in the biomedical arena. The concept that low levels of stress can induce a protective response against all manner of subsequent insults is inherently attractive. One of the problems preventing wider acceptance is that of striking a balance between the degree of preconditioning stress needed, the severity of the possible adverse consequence and its likelihood of happening. For instance being subjected to hypoxia, caloric restriction, hypothermia or hyperthermia might be considered too much of a price to pay, to protect oneself against a slim chance of relatively mild extra damage in a heart bypass operation. The same cannot be said about light therapy. LLLT has many advantages in that it is painless, inexpensive if LEDs are employed, and free of potential side effects, making it a “no-brainer” before surgery or potentially hazardous adventures. Moreover the multiple demonstrations of LLLT effectiveness for preconditioning of muscles before exercise, suggests that it will soon be taken up by the general public, and may in fact have to be regulated by the athletic authorities as a concern that could be called “light-doping”.
One topic that is of great interest to all investigators working in the pre-conditioning field, is the similarities and differences between the molecular and cellular mechanisms that have been proposed to explain the effects of each different modality. It is quite clear that there is not going to be a single “unified field theory” that will explain every pre-conditioning modality that has been reported. However, it is equally clear that there an intriguing number of overlaps between the various modalities. Mitochondria, reactive oxygen species, HIF1α, HSF have all been implicated in more than one modality of preconditioning and there may yet be more common pathways left to discover. As the progression of preconditioning therapy into the mainstream of clinical medicine continues we expect that much interesting science will be reported.
Acknowledgments
Research in the Hamblin laboratory is supported by US NIH grant R01AI050875.
REFERENCES
- Akcal A, Sevim KZ, Yesilada A, Kiyak V, Sucu DO, Tatlidede HS, Sakiz D, Kaya H. Comparison of perivascular and intramuscular applied botulinum toxin a pretreatment on muscle flap ischemia-reperfusion injury and chemical delay. J Craniofac Surg. 2013;24:278–83. doi: 10.1097/SCS.0b013e318270fd5b. [DOI] [PubMed] [Google Scholar]
- Albertini R, Villaverde AB, Aimbire F, Salgado MA, Bjordal JM, Alves LP, Munin E, Costa MS. Anti-inflammatory effects of low-level laser therapy (LLLT) with two different red wavelengths (660 nm and 684 nm) in carrageenan-induced rat paw edema. J Photochem Photobiol B. 2007;89:50–5. doi: 10.1016/j.jphotobiol.2007.08.005. [DOI] [PubMed] [Google Scholar]
- Allen DG, Lamb GD, Westerblad H. Skeletal muscle fatigue: cellular mechanisms. Physiol Rev. 2008;88:287–332. doi: 10.1152/physrev.00015.2007. [DOI] [PubMed] [Google Scholar]
- Assis L, Moretti AI, Abrahao TB, Cury V, Souza HP, Hamblin MR, Parizotto NA. Low-level laser therapy (808 nm) reduces inflammatory response and oxidative stress in rat tibialis anterior muscle after cryolesion. Lasers Surg Med. 2012;44:726–35. doi: 10.1002/lsm.22077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avci P, Gupta A, Sadasivam M, Vecchio D, Pam Z, Pam N, Hamblin MR. Low-level laser (light) therapy (LLLT) in skin: stimulating, healing, restoring. Semin Cutan Med Surg. 2013;32:41–52. [PMC free article] [PubMed] [Google Scholar]
- Avni D, Levkovitz S, Maltz L, Oron U. Protection of skeletal muscles from ischemic injury: low-level laser therapy increases antioxidant activity. Photomed Laser Surg. 2005;23:273–7. doi: 10.1089/pho.2005.23.273. [DOI] [PubMed] [Google Scholar]
- Bailey TG, Birk GK, Cable NT, Atkinson G, Green DJ, Jones H, Thijssen DH. Remote ischemic preconditioning prevents reduction in brachial artery flow-mediated dilation after strenuous exercise. Am J Physiol Heart Circ Physiol. 2012;303:H533–8. doi: 10.1152/ajpheart.00272.2012. [DOI] [PubMed] [Google Scholar]
- Balestreri M, Czosnyka M, Hutchinson P, Steiner LA, Hiler M, Smielewski P, Pickard JD. Impact of intracranial pressure and cerebral perfusion pressure on severe disability and mortality after head injury. Neurocrit Care. 2006;4:8–13. doi: 10.1385/NCC:4:1:008. [DOI] [PubMed] [Google Scholar]
- Barolet D, Boucher A. LED photoprevention: reduced MED response following multiple LED exposures. Lasers Surg Med. 2008;40:106–12. doi: 10.1002/lsm.20615. [DOI] [PubMed] [Google Scholar]
- Barolet D, Boucher A. Prophylactic low-level light therapy for the treatment of hypertrophic scars and keloids: a case series. Lasers Surg Med. 2010;42:597–601. doi: 10.1002/lsm.20952. [DOI] [PubMed] [Google Scholar]
- Baroni BM, Leal Junior EC, De Marchi T, Lopes AL, Salvador M, Vaz MA. Low level laser therapy before eccentric exercise reduces muscle damage markers in humans. Eur J Appl Physiol. 2010a;110:789–96. doi: 10.1007/s00421-010-1562-z. [DOI] [PubMed] [Google Scholar]
- Baroni BM, Leal Junior EC, Geremia JM, Diefenthaeler F, Vaz MA. Effect of light-emitting diodes therapy (LEDT) on knee extensor muscle fatigue. Photomed Laser Surg. 2010b;28:653–8. doi: 10.1089/pho.2009.2688. [DOI] [PubMed] [Google Scholar]
- Barretto SR, de Melo GC, dos Santos JC, de Oliveira MG, Pereira-Filho RN, Alves AV, Ribeiro MA, Lima-Verde IB, Quintans Junior LJ, de Albuquerque-Junior RL, Bonjardim LR. Evaluation of anti-nociceptive and anti-inflammatory activity of low-level laser therapy on temporomandibular joint inflammation in rodents. J Photochem Photobiol B. 2013;129:135–42. doi: 10.1016/j.jphotobiol.2013.10.002. [DOI] [PubMed] [Google Scholar]
- Beauvoit B, Kitai T, Chance B. Contribution of the mitochondrial compartment to the optical properties of the rat liver: a theoretical and practical approach. Biophys J. 1994;67:2501–10. doi: 10.1016/S0006-3495(94)80740-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhattacharya R, Lakshmana Rao PV. Pharmacological interventions of cyanide-induced cytotoxicity and DNA damage in isolated rat thymocytes and their protective efficacy in vivo. Toxicol Lett. 2001;119:59–70. doi: 10.1016/s0378-4274(00)00309-x. [DOI] [PubMed] [Google Scholar]
- Borsa PA, Larkin KA, True JM. Does phototherapy enhance skeletal muscle contractile function and postexercise recovery? A systematic review. J Athl Train. 2013;48:57–67. doi: 10.4085/1062-6050-48.1.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bortone F, Santos HA, Albertini R, Pesquero JB, Costa MS, Silva JA., Jr Low level laser therapy modulates kinin receptors mRNA expression in the subplantar muscle of rat paw subjected to carrageenan-induced inflammation. Int Immunopharmacol. 2008;8:206–10. doi: 10.1016/j.intimp.2007.09.004. [DOI] [PubMed] [Google Scholar]
- Bouzari N, Davis SC, Nouri K. Laser treatment of keloids and hypertrophic scars. Int J Dermatol. 2007;46:80–8. doi: 10.1111/j.1365-4632.2007.03104.x. [DOI] [PubMed] [Google Scholar]
- Cai Z, Luo W, Zhan H, Semenza GL. Hypoxia-inducible factor 1 is required for remote ischemic preconditioning of the heart. Proc Natl Acad Sci U S A. 2013;110:17462–7. doi: 10.1073/pnas.1317158110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cai Z, Manalo DJ, Wei G, Rodriguez ER, Fox-Talbot K, Lu H, Zweier JL, Semenza GL. Hearts from rodents exposed to intermittent hypoxia or erythropoietin are protected against ischemia-reperfusion injury. Circulation. 2003;108:79–85. doi: 10.1161/01.CIR.0000078635.89229.8A. [DOI] [PubMed] [Google Scholar]
- Cai Z, Semenza GL. Phosphatidylinositol-3-kinase signaling is required for erythropoietin-mediated acute protection against myocardial ischemia/reperfusion injury. Circulation. 2004;109:2050–3. doi: 10.1161/01.CIR.0000127954.98131.23. [DOI] [PubMed] [Google Scholar]
- Cai ZP, Parajuli N, Zheng X, Becker L. Remote ischemic preconditioning confers late protection against myocardial ischemia-reperfusion injury in mice by upregulating interleukin-10. Basic Res Cardiol. 2012;107:277. doi: 10.1007/s00395-012-0277-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calles C, Schneider M, Macaluso F, Benesova T, Krutmann J, Schroeder P. Infrared A radiation influences the skin fibroblast transcriptome: mechanisms and consequences. J Invest Dermatol. 2010;130:1524–36. doi: 10.1038/jid.2010.9. [DOI] [PubMed] [Google Scholar]
- Chellini F, Sassoli C, Nosi D, Deledda C, Tonelli P, Zecchi-Orlandini S, Formigli L, Giannelli M. Low pulse energy Nd:YAG laser irradiation exerts a biostimulative effect on different cells of the oral microenvironment: “an in vitro study”. Lasers Surg Med. 2010;42:527–39. doi: 10.1002/lsm.20861. [DOI] [PubMed] [Google Scholar]
- Chen Y, Lee SH, Tsai YH, Tseng SH. Ischemic preconditioning increased the intestinal stem cell activities in the intestinal crypts in mice. J Surg Res. 2014;187:85–93. doi: 10.1016/j.jss.2013.10.001. [DOI] [PubMed] [Google Scholar]
- Conlan MJ, Rapley JW, Cobb CM. Biostimulation of wound healing by low-energy laser irradiation. A review. J Clin Periodontol. 1996;23:492–6. doi: 10.1111/j.1600-051x.1996.tb00580.x. [DOI] [PubMed] [Google Scholar]
- Currie RW. Effects of ischemia and perfusion temperature on the synthesis of stress-induced (heat shock) proteins in isolated and perfused rat hearts. J Mol Cell Cardiol. 1987;19:795–808. doi: 10.1016/s0022-2828(87)80390-5. [DOI] [PubMed] [Google Scholar]
- Cury V, Moretti AI, Assis L, Bossini P, Crusca Jde S, Neto CB, Fangel R, de Souza HP, Hamblin MR, Parizotto NA. Low level laser therapy increases angiogenesis in a model of ischemic skin flap in rats mediated by VEGF, HIF-1alpha and MMP-2. J Photochem Photobiol B. 2013;125:164–70. doi: 10.1016/j.jphotobiol.2013.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Das M, Das DK. Molecular mechanism of preconditioning. IUBMB Life. 2008;60:199–203. doi: 10.1002/iub.31. [DOI] [PubMed] [Google Scholar]
- de Almeida P, Lopes-Martins RA, De Marchi T, Tomazoni SS, Albertini R, Correa JC, Rossi RP, Machado GP, da Silva DP, Bjordal JM, Leal Junior EC. Red (660 nm) and infrared (830 nm) low-level laser therapy in skeletal muscle fatigue in humans: what is better? Lasers Med Sci. 2012;27:453–8. doi: 10.1007/s10103-011-0957-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Almeida P, Lopes-Martins RA, Tomazoni SS, Junior JA, de Tarso Camillo de Carvalho P, Bjordal JM, Leal Junior EC. Low-level laser therapy improves skeletal muscle performance, decreases skeletal muscle damage and modulates mRNA expression of COX-1 and COX-2 in a dose-dependent manner. Photochem Photobiol. 2011;87:1159–63. doi: 10.1111/j.1751-1097.2011.00968.x. [DOI] [PubMed] [Google Scholar]
- De Marchi T, Leal Junior EC, Bortoli C, Tomazoni SS, Lopes-Martins RA, Salvador M. Low-level laser therapy (LLLT) in human progressive-intensity running: effects on exercise performance, skeletal muscle status, and oxidative stress. Lasers Med Sci. 2012;27:231–6. doi: 10.1007/s10103-011-0955-5. [DOI] [PubMed] [Google Scholar]
- Ding YH, Young CN, Luan X, Li J, Rafols JA, Clark JC, McAllister JP, 2nd, Ding Y. Exercise preconditioning ameliorates inflammatory injury in ischemic rats during reperfusion. Acta Neuropathol. 2005;109:237–46. doi: 10.1007/s00401-004-0943-y. [DOI] [PubMed] [Google Scholar]
- Dirnagl U, Meisel A. Endogenous neuroprotection: mitochondria as gateways to cerebral preconditioning? Neuropharmacology. 2008;55:334–44. doi: 10.1016/j.neuropharm.2008.02.017. [DOI] [PubMed] [Google Scholar]
- Dorn GW, 2nd, Souroujon MC, Liron T, Chen CH, Gray MO, Zhou HZ, Csukai M, Wu G, Lorenz JN, Mochly-Rosen D. Sustained in vivo cardiac protection by a rationally designed peptide that causes epsilon protein kinase C translocation. Proc Natl Acad Sci U S A. 1999;96:12798–803. doi: 10.1073/pnas.96.22.12798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dornbos D, 3rd, Ding Y. Mechanisms of neuronal damage and neuroprotection underlying ischemia/reperfusion injury after physical exercise. Curr Drug Targets. 2012;13:247–62. doi: 10.2174/138945012799201658. [DOI] [PubMed] [Google Scholar]
- Eells JT, Henry MM, Summerfelt P, Wong-Riley MT, Buchmann EV, Kane M, Whelan NT, Whelan HT. Therapeutic photobiomodulation for methanol-induced retinal toxicity. Proc Natl Acad Sci U S A. 2003;100:3439–44. doi: 10.1073/pnas.0534746100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferraresi C, Hamblin MR, Parizotto NA. Low-level laser (light) therapy (LLLT) on muscle tissue: performance, fatigue and repair benefited by the power of light. Photonics Lasers Med. 2012;1:267–286. doi: 10.1515/plm-2012-0032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferraresi C, Parizotto NA. Chapter 53 in Handbook of Photomedicine. Taylor and Francis; LLC, Boca Raton FL: 2014. [Google Scholar]
- Frank S, Oliver L, Lebreton-De Coster C, Moreau C, Lecabellec MT, Michel L, Vallette FM, Dubertret L, Coulomb B. Infrared radiation affects the mitochondrial pathway of apoptosis in human fibroblasts. J Invest Dermatol. 2004;123:823–31. doi: 10.1111/j.0022-202X.2004.23472.x. [DOI] [PubMed] [Google Scholar]
- Gorgey AS, Wadee AN, Sobhi NN. The effect of low-level laser therapy on electrically induced muscle fatigue: a pilot study. Photomed Laser Surg. 2008;26:501–6. doi: 10.1089/pho.2007.2161. [DOI] [PubMed] [Google Scholar]
- Hagiwara S, Iwasaka H, Hasegawa A, Noguchi T. Pre-Irradiation of blood by gallium aluminum arsenide (830 nm) low-level laser enhances peripheral endogenous opioid analgesia in rats. Anesth Analg. 2008;107:1058–63. doi: 10.1213/ane.0b013e31817ee43e. [DOI] [PubMed] [Google Scholar]
- Hamblin MR. The role of nitric oxide in low level light therapy. Proc SPIE. 2008;6846:1–14. [Google Scholar]
- Hawaleshka A, Jacobsohn E. Ischaemic preconditioning: mechanisms and potential clinical applications. Can J Anaesth. 1998;45:670–82. doi: 10.1007/BF03012100. [DOI] [PubMed] [Google Scholar]
- Hentschke VS, Jaenisch RB, Schmeing LA, Cavinato PR, Xavier LL, Dal Lago P. Low-level laser therapy improves the inflammatory profile of rats with heart failure. Lasers Med Sci. 2013;28:1007–16. doi: 10.1007/s10103-012-1190-4. [DOI] [PubMed] [Google Scholar]
- Hoffman M, Monroe DM. Low intensity laser therapy speeds wound healing in hemophilia by enhancing platelet procoagulant activity. Wound Repair Regen. 2012;20:770–7. doi: 10.1111/j.1524-475X.2012.00828.x. [DOI] [PubMed] [Google Scholar]
- Hsieh YL, Chou LW, Chang PL, Yang CC, Kao MJ, Hong CZ. Low-level laser therapy alleviates neuropathic pain and promotes function recovery in rats with chronic constriction injury: possible involvements in hypoxia-inducible factor 1alpha (HIF-1alpha) J Comp Neurol. 2012;520:2903–16. doi: 10.1002/cne.23072. [DOI] [PubMed] [Google Scholar]
- Huang YY, Sharma SK, Carroll J, Hamblin MR. Biphasic dose response in low level light therapy - an update. Dose Response. 2011;9:602–18. doi: 10.2203/dose-response.11-009.Hamblin. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Inagaki K, Begley R, Ikeno F, Mochly-Rosen D. Cardioprotection by epsilon-protein kinase C activation from ischemia: continuous delivery and antiarrhythmic effect of an epsilon-protein kinase C-activating peptide. Circulation. 2005;111:44–50. doi: 10.1161/01.CIR.0000151614.22282.F1. [DOI] [PubMed] [Google Scholar]
- Kalakech H, Tamareille S, Pons S, Godin-Ribuot D, Carmeliet P, Furber A, Martin V, Berdeaux A, Ghaleh B, Prunier F. Role of hypoxia inducible factor-1alpha in remote limb ischemic preconditioning. J Mol Cell Cardiol. 2013;65:98–104. doi: 10.1016/j.yjmcc.2013.10.001. [DOI] [PubMed] [Google Scholar]
- Karu T. Primary and secondary mechanisms of action of visible to near-IR radiation on cells. J Photochem Photobiol B. 1999;49:1–17. doi: 10.1016/S1011-1344(98)00219-X. [DOI] [PubMed] [Google Scholar]
- Karu T, Pyatibrat L, Kalendo G. Irradiation with He-Ne laser increases ATP level in cells cultivated in vitro. J Photochem Photobiol B. 1995;27:219–23. doi: 10.1016/1011-1344(94)07078-3. [DOI] [PubMed] [Google Scholar]
- Katz LM, Frank JE, Dvorak A, Finch A, Szymanowski A, Gordon CJ. Independence of brain and trunk temperature during hypothermic preconditioning in rats. J Neurosci Methods. 2009;179:179–83. doi: 10.1016/j.jneumeth.2009.01.025. [DOI] [PubMed] [Google Scholar]
- Kawano Y, Utsunomiya-Kai Y, Kai K, Miyakawa I, Ohshiro T, Narahara H. The production of VEGF involving MAP kinase activation by low level laser therapy in human granulosa cells. Laser Ther. 2012;21:269–74. doi: 10.5978/islsm.12-OR-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kimura E, Kawano Y, Todo H, Ikarashi Y, Sugibayashi K. Measurement of skin permeation/penetration of nanoparticles for their safety evaluation. Biol Pharm Bull. 2012;35:1476–86. doi: 10.1248/bpb.b12-00103. [DOI] [PubMed] [Google Scholar]
- Koch S, Della-Morte D, Dave KR, Sacco RL, Perez-Pinzon MA. Biomarkers for ischemic preconditioning: finding the responders. J Cereb Blood Flow Metab. 2014 doi: 10.1038/jcbfm.2014.42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leal Junior EC, de Godoi V, Mancalossi JL, Rossi RP, De Marchi T, Parente M, Grosselli D, Generosi RA, Basso M, Frigo L, Tomazoni SS, Bjordal JM, Lopes-Martins RA. Comparison between cold water immersion therapy (CWIT) and light emitting diode therapy (LEDT) in short-term skeletal muscle recovery after high-intensity exercise in athletes—preliminary results. Lasers Med Sci. 2011;26:493–501. doi: 10.1007/s10103-010-0866-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leal Junior EC, Lopes-Martins RA, Baroni BM, De Marchi T, Rossi RP, Grosselli D, Generosi RA, de Godoi V, Basso M, Mancalossi JL, Bjordal JM. Comparison between single-diode low-level laser therapy (LLLT) and LED multi-diode (cluster) therapy (LEDT) applications before high-intensity exercise. Photomed Laser Surg. 2009a;27:617–23. doi: 10.1089/pho.2008.2350. [DOI] [PubMed] [Google Scholar]
- Leal Junior EC, Lopes-Martins RA, Baroni BM, De Marchi T, Taufer D, Manfro DS, Rech M, Danna V, Grosselli D, Generosi RA, Marcos RL, Ramos L, Bjordal JM. Effect of 830 nm low-level laser therapy applied before high-intensity exercises on skeletal muscle recovery in athletes. Lasers Med Sci. 2009b;24:857–63. doi: 10.1007/s10103-008-0633-4. [DOI] [PubMed] [Google Scholar]
- Leal Junior EC, Lopes-Martins RA, Dalan F, Ferrari M, Sbabo FM, Generosi RA, Baroni BM, Penna SC, Iversen VV, Bjordal JM. Effect of 655-nm low-level laser therapy on exercise-induced skeletal muscle fatigue in humans. Photomed Laser Surg. 2008;26:419–24. doi: 10.1089/pho.2007.2160. [DOI] [PubMed] [Google Scholar]
- Leal Junior EC, Lopes-Martins RA, de Almeida P, Ramos L, Iversen VV, Bjordal JM. Effect of low-level laser therapy (GaAs 904 nm) in skeletal muscle fatigue and biochemical markers of muscle damage in rats. Eur J Appl Physiol. 2009c;108:1083–8. doi: 10.1007/s00421-009-1321-1. [DOI] [PubMed] [Google Scholar]
- Leal Junior EC, Lopes-Martins RA, de Almeida P, Ramos L, Iversen VV, Bjordal JM. Effect of low-level laser therapy (GaAs 904 nm) in skeletal muscle fatigue and biochemical markers of muscle damage in rats. Eur J Appl Physiol. 2010a;108:1083–8. doi: 10.1007/s00421-009-1321-1. [DOI] [PubMed] [Google Scholar]
- Leal Junior EC, Lopes-Martins RA, Frigo L, De Marchi T, Rossi RP, de Godoi V, Tomazoni SS, Silva DP, Basso M, Filho PL, de Valls Corsetti F, Iversen VV, Bjordal JM. Effects of low-level laser therapy (LLLT) in the development of exercise-induced skeletal muscle fatigue and changes in biochemical markers related to postexercise recovery. J Orthop Sports Phys Ther. 2010b;40:524–32. doi: 10.2519/jospt.2010.3294. [DOI] [PubMed] [Google Scholar]
- Leal Junior EC, Lopes-Martins RA, Rossi RP, De Marchi T, Baroni BM, de Godoi V, Marcos RL, Ramos L, Bjordal JM. Effect of cluster multi-diode light emitting diode therapy (LEDT) on exercise-induced skeletal muscle fatigue and skeletal muscle recovery in humans. Lasers Surg Med. 2009d;41:572–7. doi: 10.1002/lsm.20810. [DOI] [PubMed] [Google Scholar]
- Leal Junior EC, Lopes-Martins RA, Vanin AA, Baroni BM, Grosselli D, De Marchi T, Iversen VV, Bjordal JM. Effect of 830 nm low-level laser therapy in exercise-induced skeletal muscle fatigue in humans. Lasers Med Sci. 2009e;24:425–31. doi: 10.1007/s10103-008-0592-9. [DOI] [PubMed] [Google Scholar]
- Leal-Junior EC, Vanin AA, Miranda EF, de Carvalho PD, Dal Corso S, Bjordal JM. Effect of phototherapy (low-level laser therapy and light-emitting diode therapy) on exercise performance and markers of exercise recovery: a systematic review with meta-analysis. Lasers Med Sci. 2013 doi: 10.1007/s10103-013-1465-4. [DOI] [PubMed] [Google Scholar]
- Liang HL, Whelan HT, Eells JT, Meng H, Buchmann E, Lerch-Gaggl A, Wong-Riley M. Photobiomodulation partially rescues visual cortical neurons from cyanide-induced apoptosis. Neuroscience. 2006;139:639–49. doi: 10.1016/j.neuroscience.2005.12.047. [DOI] [PubMed] [Google Scholar]
- Lim W, Kim J, Kim S, Karna S, Won J, Jeon SM, Kim SY, Choi Y, Choi H, Kim O. Modulation of lipopolysaccharide-induced NF-kappaB signaling pathway by 635 nm irradiation via heat shock protein 27 in human gingival fibroblast cells. Photochem Photobiol. 2013;89:199–207. doi: 10.1111/j.1751-1097.2012.01225.x. [DOI] [PubMed] [Google Scholar]
- Ling GS, Neal CJ. Maintaining cerebral perfusion pressure is a worthy clinical goal. Neurocrit Care. 2005;2:75–81. doi: 10.1385/NCC:2:1:075. [DOI] [PubMed] [Google Scholar]
- Liu W, Liu Y, Chen H, Liu K, Tao H, Sun X. Xenon preconditioning: molecular mechanisms and biological effects. Med Gas Res. 2013;3:3. doi: 10.1186/2045-9912-3-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu YY, Wong-Riley MT, Liu HL, Jia Y, Jiao XY, Wang CT, You SW, Ju G. Increase in cytochrome oxidase activity in regenerating nerve fibers of hemitransected spinal cord in the rat. Neuroreport. 2001;12:3239–42. doi: 10.1097/00001756-200110290-00019. [DOI] [PubMed] [Google Scholar]
- Lopes-Martins RA, Albertini R, Martins PS, Bjordal JM, Faria Neto HC. Spontaneous effects of low-level laser therapy (650 nm) in acute inflammatory mouse pleurisy induced by carrageenan. Photomed Laser Surg. 2005;23:377–81. doi: 10.1089/pho.2005.23.377. [DOI] [PubMed] [Google Scholar]
- Lopes-Martins RA, Marcos RL, Leonardo PS, Prianti AC, Jr, Muscara MN, Aimbire F, Frigo L, Iversen VV, Bjordal JM. Effect of low-level laser (Ga-Al-As 655 nm) on skeletal muscle fatigue induced by electrical stimulation in rats. J Appl Physiol. 2006a;101:283–8. doi: 10.1152/japplphysiol.01318.2005. [DOI] [PubMed] [Google Scholar]
- Lopes-Martins RA, Marcos RL, Leonardo PS, Prianti AC, Jr, Muscara MN, Aimbire F, Frigo L, Iversen VV, Bjordal JM. Effect of low-level laser (Ga-Al-As 655 nm) on skeletal muscle fatigue induced by electrical stimulation in rats. J Appl Physiol (1985) 2006b;101:283–8. doi: 10.1152/japplphysiol.01318.2005. [DOI] [PubMed] [Google Scholar]
- Lu PG, Feng H, Yuan SJ, Zhang RW, Li M, Hu R, Liu ZS, Yin J. Effect of preconditioning with hyperbaric oxygen on neural cell apoptosis after spinal cord injury in rats. J Neurosurg Sci. 2013;57:253–8. [PubMed] [Google Scholar]
- Marais E, Genade S, Lochner A. CREB activation and ischaemic preconditioning. Cardiovasc Drugs Ther. 2008;22:3–17. doi: 10.1007/s10557-007-6078-3. [DOI] [PubMed] [Google Scholar]
- Marber MS, Latchman DS, Walker JM, Yellon DM. Cardiac stress protein elevation 24 hours after brief ischemia or heat stress is associated with resistance to myocardial infarction. Circulation. 1993;88:1264–72. doi: 10.1161/01.cir.88.3.1264. [DOI] [PubMed] [Google Scholar]
- Marber MS, Mestril R, Chi SH, Sayen MR, Yellon DM, Dillmann WH. Overexpression of the rat inducible 70-kD heat stress protein in a transgenic mouse increases the resistance of the heart to ischemic injury. J Clin Invest. 1995;95:1446–56. doi: 10.1172/JCI117815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meneguzzo DT, Lopes LA, Pallota R, Soares-Ferreira L, Lopes-Martins RA, Ribeiro MS. Prevention and treatment of mice paw edema by near-infrared low-level laser therapy on lymph nodes. Lasers Med Sci. 2013;28:973–80. doi: 10.1007/s10103-012-1163-7. [DOI] [PubMed] [Google Scholar]
- Menezes S, Coulomb B, Lebreton C, Dubertret L. Non-coherent near infrared radiation protects normal human dermal fibroblasts from solar ultraviolet toxicity. J Invest Dermatol. 1998;111:629–33. doi: 10.1046/j.1523-1747.1998.00338.x. [DOI] [PubMed] [Google Scholar]
- Merrick MA. Secondary injury after musculoskeletal trauma: a review and update. J Athl Train. 2002;37:209–17. [PMC free article] [PubMed] [Google Scholar]
- Mills EM, Gunasekar PG, Li L, Borowitz JL, Isom GE. Differential susceptibility of brain areas to cyanide involves different modes of cell death. Toxicol Appl Pharmacol. 1999;156:6–16. doi: 10.1006/taap.1999.8630. [DOI] [PubMed] [Google Scholar]
- Mitchell UH, Mack GL. Low-level laser treatment with near-infrared light increases venous nitric oxide levels acutely: a single-blind, randomized clinical trial of efficacy. Am J Phys Med Rehabil. 2013;92:151–6. doi: 10.1097/PHM.0b013e318269d70a. [DOI] [PubMed] [Google Scholar]
- Muntean DM, Ordodi V, Ferrera R, Angoulvant D. Volatile anaesthetics and cardioprotection: lessons from animal studies. Fundam Clin Pharmacol. 2013;27:21–34. doi: 10.1111/j.1472-8206.2012.01055.x. [DOI] [PubMed] [Google Scholar]
- Murry CE, Jennings RB, Reimer KA. Preconditioning with ischemia: a delay of lethal cell injury in ischemic myocardium. Circulation. 1986;74:1124–36. doi: 10.1161/01.cir.74.5.1124. [DOI] [PubMed] [Google Scholar]
- Narayanan SV, Dave KR, Perez-Pinzon MA. Ischemic preconditioning and clinical scenarios. Curr Opin Neurol. 2013;26:1–7. doi: 10.1097/WCO.0b013e32835bf200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oron U, Yaakobi T, Oron A, Hayam G, Gepstein L, Rubin O, Wolf T, Ben Haim S. Attenuation of infarct size in rats and dogs after myocardial infarction by low-energy laser irradiation. Lasers Surg Med. 2001a;28:204–11. doi: 10.1002/lsm.1039. [DOI] [PubMed] [Google Scholar]
- Oron U, Yaakobi T, Oron A, Mordechovitz D, Shofti R, Hayam G, Dror U, Gepstein L, Wolf T, Haudenschild C, Haim SB. Low-energy laser irradiation reduces formation of scar tissue after myocardial infarction in rats and dogs. Circulation. 2001b;103:296–301. doi: 10.1161/01.cir.103.2.296. [DOI] [PubMed] [Google Scholar]
- Passarella S, Casamassima E, Molinari S, Pastore D, Quagliariello E, Catalano IM, Cingolani A. Increase of proton electrochemical potential and ATP synthesis in rat liver mitochondria irradiated in vitro by helium-neon laser. FEBS Lett. 1984;175:95–9. doi: 10.1016/0014-5793(84)80577-3. [DOI] [PubMed] [Google Scholar]
- Pedersen CM, Cruden NL, Schmidt MR, Lau C, Botker HE, Kharbanda RK, Newby DE. Remote ischemic preconditioning prevents systemic platelet activation associated with ischemia-reperfusion injury in humans. J Thromb Haemost. 2011;9:404–7. doi: 10.1111/j.1538-7836.2010.04142.x. [DOI] [PubMed] [Google Scholar]
- Ping P, Song C, Zhang J, Guo Y, Cao X, Li RC, Wu W, Vondriska TM, Pass JM, Tang XL, Pierce WM, Bolli R. Formation of protein kinase C(epsilon)-Lck signaling modules confers cardioprotection. J Clin Invest. 2002;109:499–507. doi: 10.1172/JCI13200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prabhakaran K, Li L, Borowitz JL, Isom GE. Cyanide induces different modes of death in cortical and mesencephalon cells. J Pharmacol Exp Ther. 2002;303:510–9. doi: 10.1124/jpet.102.039453. [DOI] [PubMed] [Google Scholar]
- Prabhakaran K, Li L, Borowitz JL, Isom GE. Caspase inhibition switches the mode of cell death induced by cyanide by enhancing reactive oxygen species generation and PARP-1 activation. Toxicol Appl Pharmacol. 2004;195:194–202. doi: 10.1016/j.taap.2003.11.012. [DOI] [PubMed] [Google Scholar]
- Prianti AC, Jr, Silva JA, Jr, Dos Santos RF, Rosseti IB, Costa MS. Low-level laser therapy (LLLT) reduces the COX-2 mRNA expression in both subplantar and total brain tissues in the model of peripheral inflammation induced by administration of carrageenan. Lasers Med Sci. 2014 doi: 10.1007/s10103-014-1543-2. [DOI] [PubMed] [Google Scholar]
- Rassaf T, Totzeck M, Hendgen-Cotta UB, Shiva S, Heusch G, Kelm M. Circulating Nitrite Contributes to Cardioprotection by Remote Ischemic Preconditioning. Circ Res. 2014 doi: 10.1161/CIRCRESAHA.114.303822. [DOI] [PubMed] [Google Scholar]
- Richard V, Kaeffer N, Thuillez C. Delayed protection of the ischemic heart—from pathophysiology to therapeutic applications. Fundam Clin Pharmacol. 1996;10:409–15. doi: 10.1111/j.1472-8206.1996.tb00595.x. [DOI] [PubMed] [Google Scholar]
- Rosenzweig HL, Minami M, Lessov NS, Coste SC, Stevens SL, Henshall DC, Meller R, Simon RP, Stenzel-, Poore MP. Endotoxin preconditioning protects against the cytotoxic effects of TNFalpha after stroke: a novel role for TNFalpha in LPS-ischemic tolerance. J Cereb Blood Flow Metab. 2007;27:1663–74. doi: 10.1038/sj.jcbfm.9600464. [DOI] [PubMed] [Google Scholar]
- Samoilova KA, Zhevago NA, Petrishchev NN, Zimin AA. Role of nitric oxide in the visible light-induced rapid increase of human skin microcirculation at the local and systemic levels: II. healthy volunteers. Photomed Laser Surg. 2008;26:443–9. doi: 10.1089/pho.2007.2205. [DOI] [PubMed] [Google Scholar]
- Saxena P, Aggarwal S, Misso NL, Passage J, Newman MA, Thompson PJ, d’Udekem Y, Praporski S, Konstantinov IE. Remote ischaemic preconditioning down-regulates kinin receptor expression in neutrophils of patients undergoing heart surgery. Interact Cardiovasc Thorac Surg. 2013;17:653–8. doi: 10.1093/icvts/ivt279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schroeder P, Calles C, Benesova T, Macaluso F, Krutmann J. Photoprotection beyond ultraviolet radiation—effective sun protection has to include protection against infrared A radiation-induced skin damage. Skin Pharmacol Physiol. 2010;23:15–7. doi: 10.1159/000257259. [DOI] [PubMed] [Google Scholar]
- Sedaghat Z, Kadkhodaee M, Seifi B, Salehi E, Najafi A, Dargahi L. Remote preconditioning reduces oxidative stress, downregulates cyclo-oxygenase-2 expression and attenuates ischaemia-reperfusion-induced acute kidney injury. Clin Exp Pharmacol Physiol. 2013;40:97–103. doi: 10.1111/1440-1681.12044. [DOI] [PubMed] [Google Scholar]
- Semenza GL. Hypoxia-inducible factor 1: regulator of mitochondrial metabolism and mediator of ischemic preconditioning. Biochim Biophys Acta. 2011;1813:1263–8. doi: 10.1016/j.bbamcr.2010.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shefer G, Partridge TA, Heslop L, Gross JG, Oron U, Halevy O. Low-energy laser irradiation promotes the survival and cell cycle entry of skeletal muscle satellite cells. J Cell Sci. 2002;115:1461–9. doi: 10.1242/jcs.115.7.1461. [DOI] [PubMed] [Google Scholar]
- Shinmura K, Tamaki K, Bolli R. Short-term caloric restriction improves ischemic tolerance independent of opening of ATP-sensitive K+ channels in both young and aged hearts. J Mol Cell Cardiol. 2005;39:285–96. doi: 10.1016/j.yjmcc.2005.03.010. [DOI] [PubMed] [Google Scholar]
- Simao F, Matte A, Pagnussat AS, Netto CA, Salbego CG. Resveratrol preconditioning modulates inflammatory response in the rat hippocampus following global cerebral ischemia. Neurochem Int. 2012;61:659–65. doi: 10.1016/j.neuint.2012.06.009. [DOI] [PubMed] [Google Scholar]
- Sinha RP, Hader DP. UV-induced DNA damage and repair: a review. Photochem Photobiol Sci. 2002;1:225–36. doi: 10.1039/b201230h. [DOI] [PubMed] [Google Scholar]
- Sommer AP, Pinheiro AL, Mester AR, Franke RP, Whelan HT. Biostimulatory windows in low-intensity laser activation: lasers, scanners, and NASA’s light-emitting diode array system. J Clin Laser Med Surg. 2001;19:29–33. doi: 10.1089/104454701750066910. [DOI] [PubMed] [Google Scholar]
- Souza Filho MV, Loiola RT, Rocha EL, Simao AF, Gomes AS, Souza MH, Ribeiro RA. Hind limb ischemic preconditioning induces an anti-inflammatory response by remote organs in rats. Braz J Med Biol Res. 2009;42:921–9. doi: 10.1590/s0100-879x2009005000025. [DOI] [PubMed] [Google Scholar]
- Stetler RA, Leak RK, Gan Y, Li P, Zhang F, Hu X, Jing Z, Chen J, Zigmond MJ, Gao Y. Preconditioning provides neuroprotection in models of CNS disease: Paradigms and clinical significance. Prog Neurobiol. 2014;114C:58–83. doi: 10.1016/j.pneurobio.2013.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szymanska J, Goralczyk K, Klawe JJ, Lukowicz M, Michalska M, Goralczyk B, Zalewski P, Newton JL, Gryko L, Zajac A, Rosc D. Phototherapy with low-level laser influences the proliferation of endothelial cells and vascular endothelial growth factor and transforming growth factor-beta secretion. J Physiol Pharmacol. 2013;64:387–91. [PubMed] [Google Scholar]
- Thornton JD, Liu GS, Olsson RA, Downey JM. Intravenous pretreatment with A1-selective adenosine analogues protects the heart against infarction. Circulation. 1992;85:659–65. doi: 10.1161/01.cir.85.2.659. [DOI] [PubMed] [Google Scholar]
- Tuby H, Maltz L, Oron U. Modulations of VEGF and iNOS in the rat heart by low level laser therapy are associated with cardioprotection and enhanced angiogenesis. Lasers Surg Med. 2006;38:682–8. doi: 10.1002/lsm.20377. [DOI] [PubMed] [Google Scholar]
- Tuby H, Maltz L, Oron U. Induction of autologous mesenchymal stem cells in the bone marrow by low-level laser therapy has profound beneficial effects on the infarcted rat heart. Lasers Surg Med. 2011;43:401–9. doi: 10.1002/lsm.21063. [DOI] [PubMed] [Google Scholar]
- Uozumi Y, Nawashiro H, Sato S, Kawauchi S, Shima K, Kikuchi M. Targeted increase in cerebral blood flow by transcranial near-infrared laser irradiation. Lasers Surg Med. 2010;42:566–76. doi: 10.1002/lsm.20938. [DOI] [PubMed] [Google Scholar]
- Walker J. Relief from chronic pain by low power laser irradiation. Neurosci Lett. 1983;43:339–44. doi: 10.1016/0304-3940(83)90211-2. [DOI] [PubMed] [Google Scholar]
- Wang GL, Semenza GL. Purification and characterization of hypoxia-inducible factor 1. J Biol Chem. 1995;270:1230–7. doi: 10.1074/jbc.270.3.1230. [DOI] [PubMed] [Google Scholar]
- Wang ZF, Wang NP, Harmouche S, Philip T, Pang XF, Bai F, Zhao ZQ. Postconditioning promotes the cardiac repair through balancing collagen degradation and synthesis after myocardial infarction in rats. Basic Res Cardiol. 2013;108:318. doi: 10.1007/s00395-012-0318-9. [DOI] [PubMed] [Google Scholar]
- Whelan HT, Smits RL, Jr, Buchman EV, Whelan NT, Turner SG, Margolis DA, Cevenini V, Stinson H, Ignatius R, Martin T, Cwiklinski J, Philippi AF, Graf WR, Hodgson B, Gould L, Kane M, Chen G, Caviness J. Effect of NASA light-emitting diode irradiation on wound healing. J Clin Laser Med Surg. 2001;19:305–14. doi: 10.1089/104454701753342758. [DOI] [PubMed] [Google Scholar]
- Wojtovich AP, Urciuoli WR, Chatterjee S, Fisher AB, Nehrke K, Brookes PS. Kir6.2 is not the mitochondrial KATP channel but is required for cardioprotection by ischemic preconditioning. Am J Physiol Heart Circ Physiol. 2013;304:H1439–45. doi: 10.1152/ajpheart.00972.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolfram D, Tzankov A, Pulzl P, Piza-Katzer H. Hypertrophic scars and keloids—a review of their pathophysiology, risk factors, and therapeutic management. Dermatol Surg. 2009;35:171–81. doi: 10.1111/j.1524-4725.2008.34406.x. [DOI] [PubMed] [Google Scholar]
- Wolfson LI, Leenders KL, Brown LL, Jones T. Alterations of regional cerebral blood flow and oxygen metabolism in Parkinson’s disease. Neurology. 1985;35:1399–405. doi: 10.1212/wnl.35.10.1399. [DOI] [PubMed] [Google Scholar]
- Wong-Riley MT, Liang HL, Eells JT, Chance B, Henry MM, Buchmann E, Kane M, Whelan HT. Photobiomodulation directly benefits primary neurons functionally inactivated by toxins: role of cytochrome c oxidase. J Biol Chem. 2005;280:4761–71. doi: 10.1074/jbc.M409650200. [DOI] [PubMed] [Google Scholar]
- Wu JY, Chen CH, Yeh LY, Yeh ML, Ting CC, Wang YH. Low-power laser irradiation promotes the proliferation and osteogenic differentiation of human periodontal ligament cells via cyclic adenosine monophosphate. Int J Oral Sci. 2013;5:85–91. doi: 10.1038/ijos.2013.38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yaakobi T, Shoshany Y, Levkovitz S, Rubin O, Ben Haim SA, Oron U. Long-term effect of low energy laser irradiation on infarction and reperfusion injury in the rat heart. J Appl Physiol (1985) 2001;90:2411–9. doi: 10.1152/jappl.2001.90.6.2411. [DOI] [PubMed] [Google Scholar]
- Yang HY, Guo TT, Ma YN, Liu TY, Gao M. [Effects of 650 nm laser and moxibustion pre-treatment on enteric nervous system and medullary visceral zone in rats with visceral traction pain] Zhongguo Zhen Jiu. 2010a;30:745–51. [PubMed] [Google Scholar]
- Yang X, Cohen MV, Downey JM. Mechanism of cardioprotection by early ischemic preconditioning. Cardiovasc Drugs Ther. 2010b;24:225–34. doi: 10.1007/s10557-010-6236-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang Y, Li Z, Liu H, Shi W, Zhang J. Effects of heat preconditioning on overload training induced myocardial injury. J Sports Med Phys Fitness. 2013;53:93–100. [PubMed] [Google Scholar]
- Yellon DM, Pasini E, Cargnoni A, Marber MS, Latchman DS, Ferrari R. The protective role of heat stress in the ischaemic and reperfused rabbit myocardium. J Mol Cell Cardiol. 1992;24:895–907. doi: 10.1016/0022-2828(92)91102-b. [DOI] [PubMed] [Google Scholar]
- Yogaratnam JZ, Laden G, Guvendik L, Cowen M, Cale A, Griffin S. Hyperbaric oxygen preconditioning improves myocardial function, reduces length of intensive care stay, and limits complications post coronary artery bypass graft surgery. Cardiovasc Revasc Med. 2010;11:8–19. doi: 10.1016/j.carrev.2009.03.004. [DOI] [PubMed] [Google Scholar]
- Ytrehus K, Liu Y, Downey JM. Preconditioning protects ischemic rabbit heart by protein kinase C activation. Am J Physiol. 1994;266:H1145–52. doi: 10.1152/ajpheart.1994.266.3.H1145. [DOI] [PubMed] [Google Scholar]
- Zhang H, Hou JF, Shen Y, Wang W, Wei YJ, Hu S. Low level laser irradiation precondition to create friendly milieu of infarcted myocardium and enhance early survival of transplanted bone marrow cells. J Cell Mol Med. 2010;14:1975–87. doi: 10.1111/j.1582-4934.2009.00886.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao L, Liu X, Liang J, Han S, Wang Y, Yin Y, Luo Y, Li J. Phosphorylation of p38 MAPK mediates hypoxic preconditioning-induced neuroprotection against cerebral ischemic injury via mitochondria translocation of Bcl-xL in mice. Brain Res. 2013;1503:78–88. doi: 10.1016/j.brainres.2013.01.051. [DOI] [PubMed] [Google Scholar]
- Zhou B, Zhang PJ, Tian T, Jin C, Li Y, Feng M, Liu XY, Jie L, Tao LD. Role of vascular endothelial growth factor in protection of intrahepatic cholangiocytes mediated by hypoxic preconditioning after liver transplantation in rats. Transplant Proc. 2010;42:2457–62. doi: 10.1016/j.transproceed.2010.04.028. [DOI] [PubMed] [Google Scholar]