Abstract
Connexin26 (Cx26, GJB2) mutations account for >50% of nonsyndromic hearing loss. The deafness is not always congenital. A large group of these patients (~30%) demonstrate a late-onset hearing loss, starting in childhood. They have normal hearing early in life and are therefore good candidates for applying protective and therapeutic interventions. However, the underlying deafness mechanism is unclear. In this study, we used a time-controlled, inducible gene knockout technique to knockout Cx26 expression in the cochlea after birth. We found that deletion of Cx26 after postnatal day 5 (P5) in mice could lead to late-onset hearing loss. Similar to clinical observations, the mice demonstrated progressive, mild to moderate hearing loss. The hearing loss initiated at high frequencies and then extended to the middle- and low-frequency range. The cochlea showed normal development and had no apparent hair cell loss. However, distortion product otoacoustic emission (DPOAE) was reduced. The reduction was also progressive and large at high-frequencies. Consistent with DPOAE reduction, we found that outer hair cell electromotility associated nonlinear capacitance was shifted to the right and the slope of voltage dependence was reduced. The endocochlear potential was reduced in Cx26 conditional knockout (cKO) mice but the reduction was not associated with progressive hearing loss. These data suggest that Cx26 deficiency may impair active cochlear amplification leading to late-onset hearing loss. Our study also helps develop newer protective and therapeutic interventions to this common nonsyndromic hearing loss.
Keywords: gap junction, connexin, Cx26, deafness, inner ear, active cochlear amplification
Introduction
Connexin26 (Cx26, GJB2) mutations are a common genetic cause for nonsyndromic hearing loss, responsible for >50% of nonsyndromic hearing loss in children. Clinically, the majority of Cx26 deafness mutations are recessive but a few are dominant, resulting in DFNB1 (recessive) and DFNA3 (dominant) nonsyndromic hearing loss (Zhao et al. 2006; Castillo and Castillo, 2011; Chan and Chang, 2014). The hearing loss, especially caused by p.M34T and p.V37I mutations, is not always congenital. A significant minority of these patients (~30%) demonstrate a progressive, late-onset hearing loss. The hearing loss starts or occurs in childhood, is mild to moderate, and usually appears severe at high-frequencies (Cohn et al., 1999; Murgia et al., 1999; Wilcox et al., 2000; Kenneson et al., 2002; Engel-Yeger et al., 2003; Pollak et al., 2007; Orzan and Murgia, 2007; Gopalarao et al., 2008; Kenna et al., 2010; Chan et al., 2010). These individuals have normal hearing early in life and are good candidates for applying protective and therapeutic interventions. However, little is known about the underlying deafness mechanism.
Previous studies show that knockout (KO) of Cx26 in the cochlea can cause congenital deafness with cochlear development disorders, cell degeneration, and endocochlear potential (EP) reduction (Cohen-Salmon et al., 2002; Sun et al., 2009; Wang et al., 2009; Liang et al., 2012; Chen et al., 2014). We further found that hair cell degeneration and EP reduction are not primary causes for congenital deafness; the congenital deafness is associated with cochlear development disorders (Liang et al., 2012; Chen et al., 2014). These studies provide invaluable information about Cx26 mutation induced congenital hearing loss but not about Cx26 deficiency induced late-onset hearing loss.
Recently, we also found that Cx26 deficiency can reduce active cochlear amplification (Zhu et al., 2013), even though there is no gap junction and connexin expression in hair cells (Kikuchi et al., 1995; Zhao and Santos-Sacchi, 1999; Zhao, 2000; Zhao and Yu, 2006; Yu and Zhao 2009). Active cochlear amplification is required for normal mammalian hearing to amplify acoustic stimulation increasing hearing sensitivity and frequency selectivity (Dallos 2008; Hudspeth 2008). In this study, we used a time-controlled, inducible gene-knockout technique to delete Cx26 expression in the cochlea after birth. We found that Cx26 deficiency could reduce active cochlear amplification leading to late-onset, progressive hearing loss.
Materials and Methods
Cx26 conditional KO mice and genotyping
As we previously reported (Chen et al. 2014), Cx26loxP/loxP transgenic mice (European Mouse Mutant Archive, E00245) were crossed with the ROSA26Cre/Esr1 mouse line (Jackson Lab, stock No. 004847), which has a tamoxifen-inducible Cre-mediated recombination system driven by Gt(ROSA)26Sor promoter and can be excised by tamoxifen. The ROSA26CreEsr1 transgene and the Cx26 floxed allele were detected on tail genomic DNA by PCR amplification using the following primers: CreF: 5′-GCG GTC TGG CAG TAA AAA CTA TC-3′ and CreR: 5′-GTG AAA CAG CAT TGC TGT CAC TT-3′ for the CreEsr1 transgene; Cx26F: 5′-CTT TCC AAT GCT GGT GGAGTG-3′ and Cx26R: 5′-ACA GAA ATG TGT TGG TGA TGG-3′ for the Cx26 floxed allele (Cohen-Salmon et al., 2002). Cx26loxP/loxP and WT mice generated 400 and 300 bps bands, respectively. The bands of ROSA26-Esr1/Cre positive and WT mice were located at 102 and 400 bps, respectively.
Mice were intraperitoneally injected with 4-Hydroxytamoxifen (4-HTMX, H7904, Sigma-Aldrich, St. Louis, MO) with 0.5 mg/10g per day for three days to activate Cre expression to delete Cx26. The injection was performed at postnatal day 10 (P10), unless otherwise indicated in text. This injection procedure ensured that Cx26 expression in the cochlea could be deleted but the mice have no congenital hearing loss (Chen et al., 2014). All experimental procedures were conducted in accordance with the policies of University of the Kentucky Animal Care & Use Committee.
Auditory brainstem response and distortion product otoacoustic emission measurements
Auditory brainstem response (ABR) and distortion product otoacoustic emission (DPOAE) were recorded in a double-wall sound isolated chamber by use of a Tucker-Davis ABR workstation (Tucker-Davis Tech. Alachua, FL). As we previously reported (Liang et al., 2012; Zhu et al., 2013; Chen et al., 2014), mice were anesthetized by intraperitoneal injection with a mixture of Ketamine and Xylazine (a stock solution: 8.5 ml saline+1 ml Ketamine+0.55 ml Xylazine) given at a dose of 0.1 ml/10 g body weight. Body temperature was maintained at 37–38°C by placing anesthetized mice on an isothermal pad (Deltaphase, model 39dp, Braintree Scientific Inc., Massachusetts). Subdermal needle electrodes were inserted at the vertex (an active electrode) and ventrolaterally to the right ear (a reference electrode) and to the left ear (a ground electrode). ABR was measured by both clicks and series of tone bursts (4 – 40 kHz, 10–80 dB SPL, a 5dB-step). The ABR threshold was determined by the lowest level at which an ABR can be recognized. If mice had severe hearing loss, the ABR test at the intensity range of 70 – 110 dB SPL was used.
DPOAE was recorded as described by our previous reports (Zhu et al., 2013). Two plastic tubes were inserted into the external ear canal and sealed with an earplug. Two pure tones (f1 and f2) were simultaneously delivered into the ear. The ratio of f2 versus f1 (f2/f1) was 1.22. The test frequencies were presented by a geometric mean of f1 and f2 [f0 = (f1 × f2)1/2] from f0=4 to 20 kHz. The intensity of f1 (L1) was set at 5 dB SPL higher than that of f2 (L2). The distortion product was recorded from the L1/L2 level of 25/20 – 65/60 dB SPL. One hundred fifty responses were averaged. A cubic distortion component of 2f1−f2 in DPOAEs was measured.
Immunofluorescent staining
The cochlear tissue preparation and immunofluorescent staining were performed as we previously reported (Zhao and Yu, 2006, Liu and Zhao, 2008). The cochlear cross-section or isolated sensory epithelia were fixed with 4% paraformaldehyde in 0.1 M PBS (pH 7.4) for 30 min. After being washed with PBS (0.1 M) 3 times, the tissue was incubated in a blocking solution (10% goat serum and 1% BSA in the PBS) with 0.1% Triton X-100 for 30 min at room temperature. The tissue then was incubated with primary antibody in the blocking solution at 4°C overnight. Monoclonal mouse anti-Cx26 (Cat# 33–5800), and polyclonal rabbit anti-Cx30 (Cat#71-2200, Invitrogen Corp, Carlsbad, CA) were used. After completely washing out the primary antibodies with PBS, the reaction to a 1:600 dilution of secondary Alexa Fluor® 488 or 568 conjugated antibodies (Molecular Probes) in the blocking solution followed at room temperature for 1 h. After completely washing out, the section was mounted with a fluorescence mounting medium (H-1000, Vector Lab, CA) and observed under a Leica confocal microscope (Leica TCS SP2).
For quantitative analysis of Cx30 expression in the cochlea, immunofluorescent staining of the cochlear sections of WT mice and Cx26 cKO mice were performed in parallel at the same time and observed under the same confocal settings as we previously reported (Zhao and Yu, 2006). Serial sections of the confocal image were taken along the Z-axis. The staining intensity of Cx30 was measured by use of ImageJ software (NIH, Bethesda, USA) (Zhao and Yu, 2006; Yu et al., 2008).
Patch-clamp recording and nonlinear capacitance measurement
Outer hair cells (OHCs) were freshly isolated from the cochlea (Yu and Zhao, 2008; Zhu et al., 2013). The classical patch clamp recording was performed under the whole-cell configuration by using an Axopatch 200B patch clamp amplifier (Molecular Devices, CA) with jClamp (Scisft, New Haven, CT) (Yu and Zhao, 2008). The patch pipette was filled with an intracellular ionic blocking solution (140 CsCl, 10 EGTA, 2 MgCl2, 10 HEPES in mM; 310 mOsm and pH 7.2) and had initial resistance of 2.5–3.5 MΩ in bath solution (142 NaCl, 5.37 KCl, 1.47 MgCl2, 2 CaCl2, 10 HEPES in mM, 310 mOsm and pH 7.2). The OHC electromotility associated nonlinear capacitance (NLC) was measured by a two-sinusoidal method (Santos-Sacchi et al., 1998; Yu and Zhao, 2008). The signal was filtered by a 4-pole low-pass Bessel filter with a cut-off frequency of 10 kHz and digitized utilizing a Digidata 1322A (Molecular Devices, CA). The capacitance was calculated by admittance analysis of the current response. All experimental procedures were performed at room temperature (23 °C).
The voltage-dependent NLC was fitted to the first derivative of a two-state Boltzmann function with jClamp and MATLAB (Yu and Zhao, 2008, 2009):
| (1) |
where NLC is the nonlinear capacitance component associated with OHC electromotility, Qmax is the maximum charge transferred, Vpk is the voltage at the peak of NLC, and α is the slope of voltage dependence. Membrane potential (Vm) was corrected for electrode access resistance (Rs).
Data processing and statistical analysis
Data were plotted by SigmaPlot and statistically analyzed by SPSS v18.0 (SPSS Inc. Chicago, IL). Error bars represent s.e.m. and data were expressed as mean ± s.e.m. other than indicated in text. Data were plotted by SigmaPlot software (SPSS Inc., Chicago).
Results
Late-onset, progressive hearing loss in Cx26 conditional KO mice
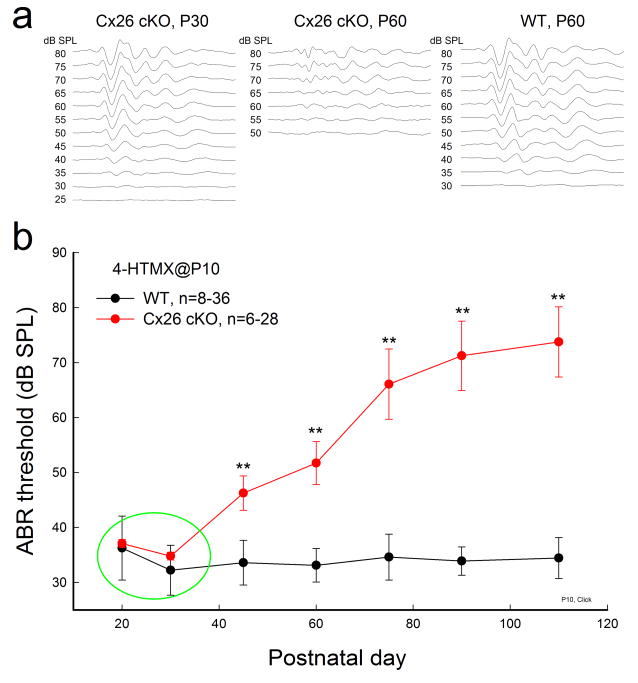
Fig. 1 shows that deletion of Cx26 expression in the cochlea at P10 could cause late-onset, progressive hearing loss. ABR recording shows that Cx26 conditional KO (cKO) mice had normal hearing after maturation. The ABR threshold was at the same level as WT mice until P30 (Fig. 1a and Fig. 1b indicated by a green circle). Then, the ABR threshold in Cx26 cKO mice showed a progressive increase (Fig. 1b). At P45–60, the ABR threshold significantly increased to 45–50 dB SPL (P<0.001, one-way ANOVA with a Bonferroni correction). At P90–110, the ABR threshold increased to > 70 dB SPL. In comparison with WT mice, the ABR thresholds in Cx26 cKO mice at P90–110 were significantly increased by 30–40 dB SPL (P<0.001, one-way ANOVA with a Bonferroni correction). However, the ABR threshold of WT littermates in the whole recording period was not substantially increased (Fig. 1b).
Fig. 1.
Late-onset, progressive hearing loss in Cx26 cKO mice. The mice were injected with 4-HTMX at P10. ABR was evoked by click stimulations. WT littermates with 4-HTMX injection served as controls. a: ABR waveforms recorded from Cx26 cKO and WT mice. The Cx26 cKO mice retain normal hearing sensitivity at P30 (left panel) but have hearing loss at P60 (middle panel), demonstrating a late-onset hearing loss. Right panel: ABR in WT mice at the same age. b: Hearing loss in Cx26 cKO mice is late-onset and progressive. A green circle indicates that ABR thresholds at P20 and P30 in Cx26 cKO mice are not significantly different from WT mice. Data are expressed as mean ± S.D; **: P<0.001 as determined by one-way ANOVA with a Bonferroni correction.
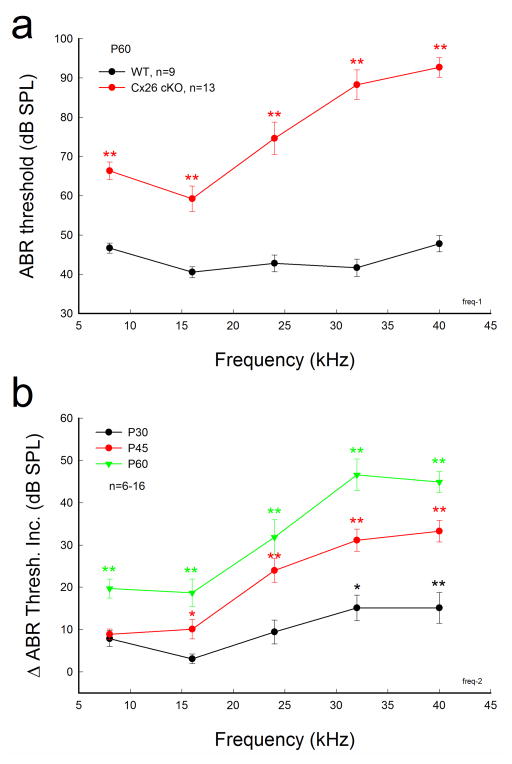
Hearing loss in Cx26 cKO mice appeared severe at high-frequencies (Fig. 2). At P60, the ABR thresholds in Cx26 cKO mice at 8, 16, 24, 32, and 40 kHz were 66.3±2.25, 59.2±3.25, 74.6±4.14, 88.3±3.75, and 92.7±2.47 dB SPL, respectively (Fig. 2a). The ABR threshold demonstrated a large increase at high-frequencies. The hearing loss also extended to middle and low frequencies (Fig. 2b). At P45, the ABR threshold at 40 kHz was increased by 33.3±2.55 dB SPL and progressively increased by 44.9±2.47 dB at P60 (Fig. 2b). However, the ABR threshold at 8 kHz at P45 had no significant increase (Fig. 2b). At P60, the ABR thresholds at 8 and 16 kHz were increased by 19.7±2.25 and 18.7±3.25 dB SPL, respectively. The increments were significant (P<0.001, one-way ANOVA with a Bonferroni correction).
Fig. 2.
Severe hearing loss in high-frequency range in Cx26 cKO mice. The mice were injected with 4-HTMX at P10. a: Frequency tone burst-evoked ABR thresholds at P60. The ABR thresholds were further elevated at high frequencies. b: Progressive increase in ABR threshold in Cx26 cKO mice. The increments were obtained from comparison with ABR thresholds of WT littermate mice at the same time point. The increments are apparent at high-frequencies. Data are expressed as mean ± S.D; **: P<0.001 as determined by one-way ANOVA with a Bonferroni correction.
Deletion of Cx26 in the cochlea in Cx26 cKO mice
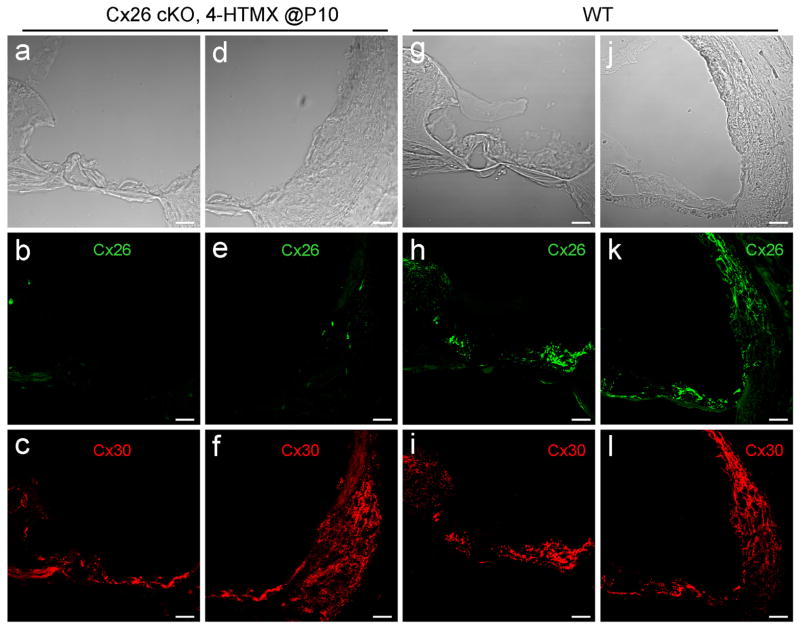
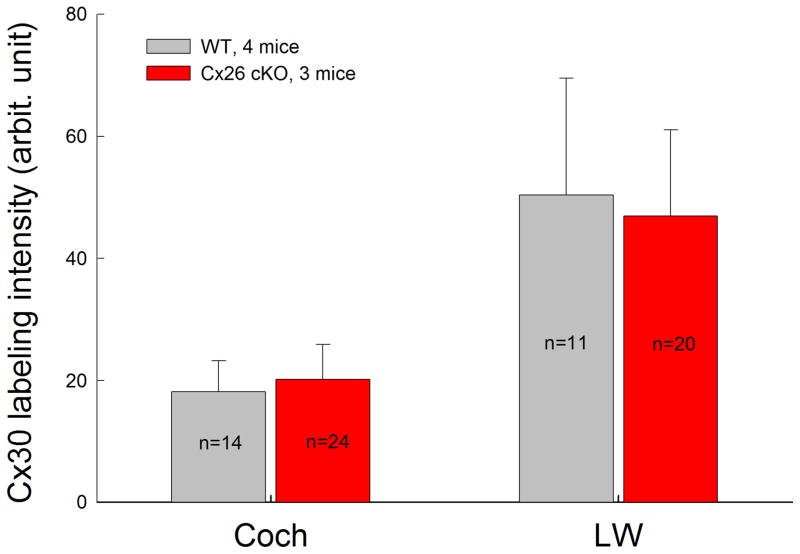
We examined Cx26 deletion in the cochlea in Cx26 cKO mice. Fig. 3 shows that Cx26 labeling was almost completely absent in the cochlea in Cx26 cKO mice following injection of 4-HTMX at P10 (Fig. 3). However, there were no detectable developmental disorders in Cx26 cKO mice. The cochlear tunnel in Cx26 cKO mice was open (Fig. 3a). In addition, co-expressed Cx30 in Cx26 cKO mice appeared normal in immunofluorescent staining (Fig. 3c&f). Quantitative analyses showed that there was no significant difference in Cx30 staining between WT and Cx26 cKO mice (Fig. 4).
Fig. 3.
Deletion of Cx26 in the cochlea in Cx26 cKO mice. a–f: Immunofluorescent staining of the cochlea for Cx26 (green) and Cx30 (red) in Cx26 cKO mice. The mouse was P30 old and 4-HTMX was injected at P10 for Cx26 deletion. g–l: Immunofluorescent staining of the cochlear cross-section for Cx26 and Cx30 in WT mice. Scale bars: 25 μm.
Fig. 4.
Quantitative analysis of Cx30 expression in the cochlea and lateral wall (LW) in the Cx26 cKO mice. The section was serially scanned along the Z-axis, and then the intensity of Cx30 labeling was measured. n represents measurement times. The data were collected from 4 WT mice and 3 Cx26 cKO mice, and then averaged. Error bars represent SD. There is no significant difference in Cx30 expression between WT mice and Cx26 cKO mice (P=0.14, Cochlea; P=0.31, LW, t-test).
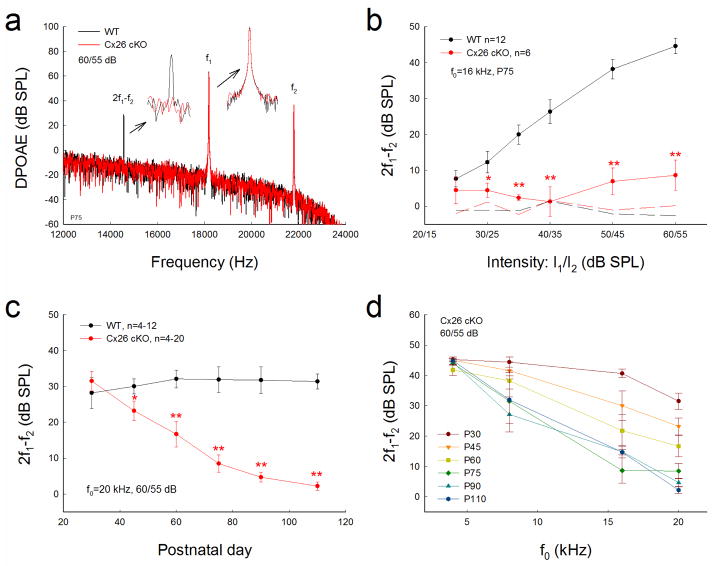
Reduction of DPOAE in Cx26 cKO mice
Acoustic emission reflects the activation of active amplification in the cochlea. Fig. 5 shows that Cx26 cKO mice had a significant reduction in DPOAE (Fig. 5a&b). The reduction was large at high intensity and could be up to 40 dB (Fig. 5b). The reduction of DPOAE in Cx26 cKO mice also appeared as late-onset and was progressive. Fig. 5c shows that the DPOAE in the Cx26 cKO mice was normal after maturation. At P30, there was no significant difference in DPOAE measurements between Cx26 cKO mice and WT mice. Then, the DPOAE in the Cx26 cKO mice was reduced. At P90–110, the DPOAE in Cx26 cKO mice was reduced to noise level and was almost undetectable. The reduction of DPOAE also demonstrated a frequency-dependence and first occurred at higher frequencies (Fig. 5d). The decrease was large at higher frequencies and the reduction was progressively increased. DPOAEs at 16 kHz and 20 kHz in Cx26 cKO mice were 21.8±5.29 and 16.7±3.56 dB SPL, respectively, at P60, and 14.9±2.09 and 4.71±1.39 dB SPL, respectively, at P90 (Fig. 5d). After P90, the DPOAE was almost reduced to zero (Fig. 5c&d). The DPOAE in Cx26 cKO mice at P110 was 2.22±1.17 dB SPL at 20 kHz.
Fig. 5.
Progressive reduction of DPOAE in Cx26 cKO mice. The mice were injected with 4-HTMX at P10. WT littermates served as controls. a: Spectrum of acoustic emission recorded from Cx26 cKO mice and WT mice. Insets: Large scale plotting of 2f1-f2 and f1 peaks. The peak of DPOAE (2f1-f2) in Cx26 cKO mice was reduced but f1 and f2 peaks remained the same as those in WT mice. f0=20 kHz. b: Reduction of DPOAE in Cx26 cKO mice in I/O plot. Dashed lines represent the noise levels of recording. c: Progressive reduction of DPOAE in Cx26 cKO mice. d: Large reduction of DPOAE at high-frequencies in Cx26 cKO mice. *: P<0.05, **: P < 0.001 as determined by one-way ANOVA with a Bonferroni correction.
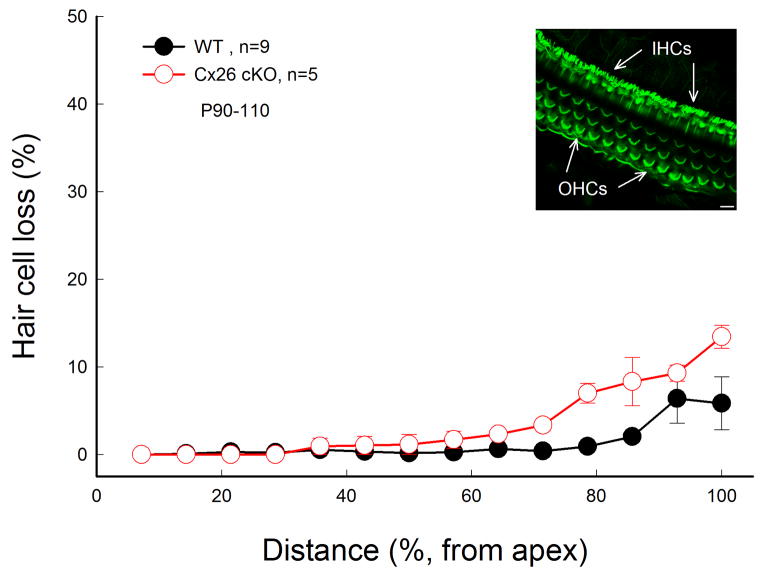
No substantial hair cell loss in Cx26 cKO mice
However, there was no substantial hair cell loss in Cx26 cKO mice (Fig. 6). Hair cell loss at P90 in the Cx26 cKO mice was similar to that in WT mice. Only at the very high frequency region in the basal turn, hair cell loss in both WT and Cx26 cKO mice was visible but less than 10% (Fig. 6). Hair cells in Cx26 cKO mice also displayed normal shape; no apparent destruction of stereocilia is visible (Inset in Fig. 6).
Fig. 6.
No substantial hair cell loss in Cx26 cKO mice. Mice were injected with 4-HTMX at P10 and were P90–110 old. WT littermates with 4-HTMX injection served as control. Insets: Stereocilia of inner hair cells (IHCs) and outer hair cells (OHCs) in Cx26 cKO mice were stained with phalloidin-Alexa 488 in whole-mount preparation and show normal shape. Scale bar: 10 μm.
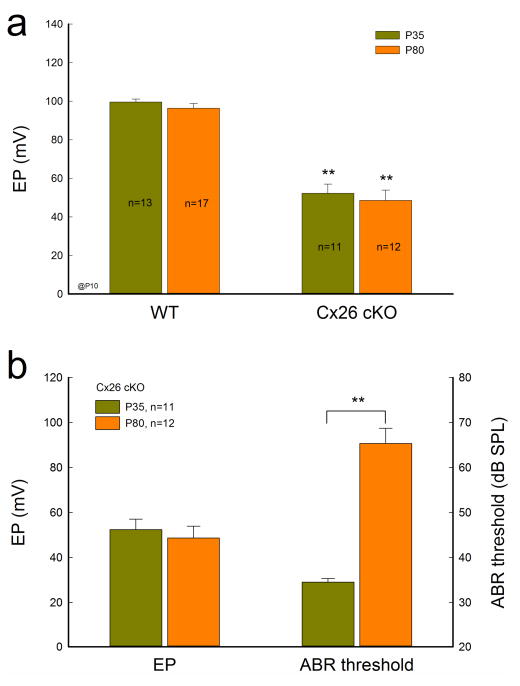
EP reduction in Cx26 cKO mice
EP is a driving force required for hair cells generating auditory receptor current and potential. EP reduction can induce hearing loss (Chen and Zhao, 2014). Fig. 7 shows that EP in Cx26 cKO mice was reduced but the reduction was not associated with progressive hearing loss. The recorded EP in WT and Cx26 cKO mice at P35 was 99.6±1.57 and 52.3±4.77 mV, respectively. The EP in Cx26 cKO mice was significantly reduced by 47 mV (P <0.001, t-test). At P80, the EP in Cx26 cKO mice was 48.6±5.27 mV, similar to that at P35 (Fig. 7a). There was no significant difference in EP reduction between P35 and P80 (P=0.31, t-test). However, in contrast to the non-significant changes in EP, ABR threshold was significantly increased from 34.8±0.69 dB SPL at P35 to 66.1±6.40 dB SPL at P80 (P<0.001, t-test, Fig. 7b).
Fig. 7.
EP reduced but not associated progressive hearing loss in Cx26 cKO mice. Mice were injected with 4-HTMX at P10 for deletion of Cx26. a: The EP has significant reduction in Cx26 KO mice. b: The EP reduction is not associated with progressive hearing loss in Cx26 cKO mice. The ABR thresholds were measured by click stimulations. EP has no significant changes in Cx26 cKO mice between P35 and P80. However, the ABR threshold at P80 has significant increase in comparison with that at P35. **: P<0.001 (t-test).
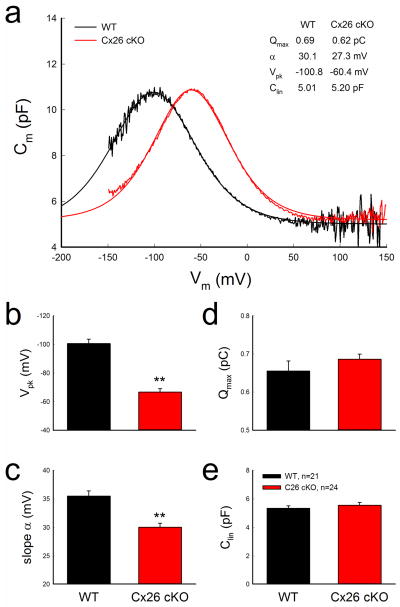
Shift of outer hair cell NLC in Cx26 cKO mice
We further examined OHC electromotility, which serves as an active cochlear amplifier (Brownell et al., 1985; Ashmore, 2008), by NLC measurement. In Cx26 cKO mice, NLC retained a normal bell-shape but was shifted to the right in the depolarization direction (Fig. 8a). The voltage of peak capacitance (Vpk) was significantly shifted from −100.5±3.11 mV in WT mice to −66.7±2.28 mV in Cx26 cKO mice (P<0.001, t-test) (Fig. 8b). The slope (α) of voltage dependence was also significantly reduced from 35.5±0.9 mV in WT mice to 30.1±0.7 mV in Cx26 cKO mice (Fig. 8c, P<0.001, t-test). However, there was no reduction in the maximum charge (Qmax), which reflects the functional expression of prestin on the plasma membrane (Ashmore 2008), in Cx26 cKO mice (Fig. 8d). Qmax in WT and Cx26 cKO mice was 0.655±0.026 and 0.686±0.013 pC, respectively (P=0.15, t-test). Moreover, there was no significant difference in linear capacitance (Clin) of recorded OHCs between WT and Cx26 cKO groups (Fig. 8e). The recorded Clin in WT and Cx26 cKO mice was 5.34±0.17 and 5.55±0.19 pF, respectively (P=0.21, t-test), indicating that the recorded OHCs in both WT and Cx26 cKO groups had similar cell length.
Fig. 8.
Shift of OHC electromotility associated NLC in Cx26 cKO mice. Mice were injected with 4-HTMX at P10 and were recorded at P45–90. a: NLC recorded from Cx26 cKO and WT mice. Smooth lines represent fitting by the first derivative of Boltzmann equation. b–e: Parameters of NLC fitting. WT littermates served as a control group. **: P < 0.001 (t-test).
Hearing loss following deletion of Cx26 at different postnatal developmental stages
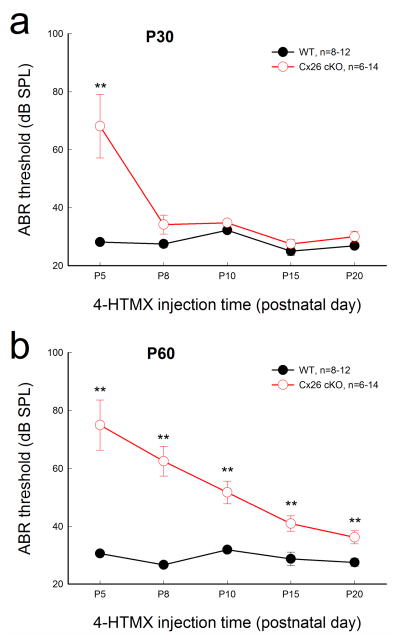
We further tested the effect of Cx26 deletion time on hearing function (Fig. 9). As we previously reported (Chen et al., 2014), mice showed normal hearing at young ages following deletion of Cx26 after P5 (Fig. 9a). At P30, ABR thresholds were 68.1±10.9, 34.2±3.27, 34.8±0.69, 27.5±1.44, and 30.0±1.89 dB SPL following deletion of Cx26 at P5, P8, P10, P15, and P20, respectively. In comparison with WT mice, there were no significant increments in ABR thresholds except that at P5. However, at P60, the ABR thresholds were 75.0±8.66, 51.7±3.90, and 36.3±2.26 dB SPL with deletion of Cx26 at P5, 10, and 20, respectively (Fig. 9b). Hearing loss was apparent in Cx26 cKO mice, with earlier deletion of Cx26 in the cochlea producing more severe hearing loss.
Fig. 9.
Effect of Cx26 deletion at different postnatal times on hearing. Mice were injected with 4-HTMX to delete Cx26 at different postnatal times. ABR was evoked by click stimulations. a: The ABR thresholds were measured at P30. Mice with deletion of Cx26 after P5 show normal ABR thresholds. b: The ABR thresholds were measured at P60. Data are expressed as mean ± S.D; **: P<0.001 as determined by one-way ANOVA with a Bonferroni correction.
Discussion
In this study, we used an inducible gene knockout technique to delete Cx26 expression in the cochlea after birth. In mouse postnatal development, the cochlea shows normal morphology and the cochlear tunnel starts to open at P5. By P10, the EP and K+ concentration in the endolymph arise to high levels (Ehret, 1976; Kraus and Aulbach-Kraus, 1981). Hearing starts around P14 and matures at P19–20 (Liang et al., 2012). We found that the mice retained normal hearing before P30 following deletion of Cx26 after P5 (Figs. 1, 2, 9). Then, hearing loss became apparent, severe at high-frequencies (Figs. 1, 2). This progression of hearing loss is similar to late-onset hearing loss observed in Cx26 mutation-induced DFNA3 and DFNB1 nonsyndromic deafness patients, who have normal hearing in early life; the hearing loss starts in childhood (Pollak et al., 2007; Orzan and Murgia, 2007; Gopalarao et al., 2008). Moreover, consistent with DPOAE reduction observed in patients (Engel-Yeger et al., 2002, 2003; Santarelli et al., 2007), the mouse model also demonstrated a progressive DPOAE reduction, severe at high frequencies (Fig. 5). These data indicate that late-onset hearing loss induced by Cx26 deficiency may result from impairment in active cochlear amplification.
Normal mammalian hearing relies upon active cochlear amplification to amplify acoustic stimulation increasing hearing sensitivity and frequency selectivity (Dallos, 2008; Hudspeth, 2008). Defects of active cochlear amplification can cause up to 40 dB SPL hearing loss (Ashmore 2008; Dallos, 2008), which we found in the experiments (Figs. 1, 2, 9). The hearing loss in Cx26 cKO mice was mild to moderate. The ABR threshold was increased up to 40 dB (Figs. 1, 2, 9). Moreover, we found that OHC NLC in Cx26 cKO mice was shifted to the right in the depolarization direction and the slope of voltage dependence was reduced (Fig. 8). OHC electromotility serves as a major basis of active cochlear amplifier in mammals (Brownell et al., 1985; Zheng et al., 2000; Ashmore 2008). Our previous studies demonstrated that Cx26 deficiency can affect OHC electromotility to reduce active amplification in the cochlea leading to hearing loss (Yu and Zhao, 2009; Zhu et al., 2013), even though hair cells have neither gap junctional coupling nor connexin expression (Kikuchi et al., 1995; Zhao and Santos-Sacchi, 1999; Zhao, 2000; Zhao and Yu, 2006; Yu and Zhao 2009). As we previously mentioned (Yu and Zhao, 2009; Zhu et al., 2013), deletion of Cx26 may alter cytoskeleton formation and tension in OHC supporting cells to change OHC loading influencing OHC electromotility. This may also change the phase difference between OHC electromotility and active force generation (Gummer et al., 1996; Fridberger et al., 1998; Zha et al., 2012) and hair bundle-based cochlear amplification (Chan and Hudspeth, 2005; Kennedy et al., 2005).
In the experiment, we found that deletion of Cx26 in the cochlea after birth shifted OHC NLC to the right (Fig. 8) and reduced DPOAE (Fig. 5). This is consistent with a previous report in which right-shift of OHC electromotility could reduce active cochlear amplification (Dallos et al., 2008). However, we previously reported that targeted-deletion of Cx26 in Deiters cells and outer pillar cells shifted OHC electromotility to the left (Zhu et al., 2013). There are several differences between these two mouse models. First, they have different genetic backgrounds. In both studies, we used WT littermates as control groups. The Vpk values in both WT control groups are different. In the current study, Vpk in WT mice was −100.5±3.11 mV (Fig. 8b) whereas the Vpk in WT mice in our previous study was −73.8±2.14 mV (Zhu et al., 2013). Second, EP was reduced in the current Cx26 cKO mice (Fig. 7) whereas EP was normal with targeted-deletion of Cx26 in DCs and OPCs (Zhu et al., 2013). Finally, time of deletion of Cx26 is different. In the current study, we deleted Cx26 at P10, when the cochlear postnatal development is almost completed. In our previous study, we deleted Cx26 at P0, when the postnatal development is just starting. It has been reported that both left and right shift of OHC electromotility can reduce OHC electromotility amplification and active force generation (Fridberger et al., 1998; Rajagopalan et al., 2007). Indeed, both mouse models had DPOAE reduction (Fig. 5 and also see Fig. 3 in Zhu et al., 2013).
We also found that EP was significantly reduced in Cx26 cKO mice (Fig. 7). However, the EP reduction was not associated with progressive hearing loss. The EP in Cx26 cKO mice at P35 and P80 was 52.3±4.77 and 48.6±5.27 mV, respectively. There was no significant change in EP between P35 and P80 (Fig. 7a). However, the ABR threshold significantly increased from 34.8±0.69 dB SPL at P35 to 66.1±6.40 dB SPL at P80, showing apparent hearing loss (Fig. 7b). Thus, the EP reduction is not a determining factor in Cx26 deficiency induced late-onset hearing loss. In addition, the Cx26 cKO mice had normal cochlear development and had no substantial hair cell loss (Figs. 3, 6), indicating that late-onset hearing loss is not a result of cell degeneration and developmental disorders.
Cx26 mutations can induce a high incidence of nonsyndromic hearing loss (Zhao et al. 2006; Castillo and Castillo, 2011). However, the underlying deafness mechanism(s) is still unclear. Several deafness mechanisms, such as K+-recycling and Ca++-wave propagation hypotheses, have been proposed (Kikuchi et al., 1995; Zhao et al. 2006; Beltramello et al., 2005). It has been hypothesized that Cx26 mutations may impair K+-recycling from hair cell’s extracellular space to endolymph leading to K+ accumulation, thereby damaging hair cells and causing hearing loss (Kikuchi et al., 1995; Zhao et al. 2006). It also has been hypothesized that Cx26 deficiency may impair Ca++-wave propagation inducing hearing loss (Beltramello et al., 2005), because Cx26 deafness mutant p.R75W can impair the spread of Ca++-waves between cells (Beltramello et al., 2005; Yum et al., 2010; Zhang et al., 2011; Kamiya et al., 2014). However, so far, these hypotheses still lack direct evidence as to whether Cx26 deficiency impairs K+-recycling in the cochlea, and how such Ca++-wave propagation is related to hearing. In this study, we found that after deletion of Cx26 in the cochlea (Fig. 3), mice still retained normal hearing at young ages (Figs. 1, 2, 9). These data imply that Cx26 deficiency may not impair K+-recycling and Ca++-wave propagation in the cochlea (which could result from possible compensations), or that impaired K+-recycling and Ca++-wave propagation do not lead to hearing loss (if Cx26 deficiency can in fact impair K+-recycling and Ca++-wave propagation in the cochlea). In any case, both K+-recycling and Ca++-wave impairments cannot be the principle deafness mechanisms underlying Cx26 deficiency induced hearing loss.
Cx26 mutations can induce congenital deafness and late-onset hearing loss (Castillo and Castillo, 2011; Chan and Chang, 2014). We recently found that congenital deafness induced by Cx26 deficiency is associated with cochlear developmental disorders rather than hair cell loss and EP reduction (Liang et al., 2012; Chen et al., 2014). In this study, we further found that that Cx26 deficiency induced late-onset hearing loss may result from the reduction of active cochlear amplification rather than possible impairment in K+-recycling and Ca++-wave propagation (Figs. 1, 2, 5, 8). Thus, these studies provide a full image for deafness mechanisms underlying Cx26 mutation induced hearing loss and also provide important information for developing new protective and therapeutic strategies for this common deafness, which clinically accounts for more than 50% of nonsyndromic hearing loss.
Highlights.
Cx26 deletion after birth can lead to late-onset, progressive hearing loss
The deletion leads to hearing loss by the reduction of active cochlear amplification
The deletion shifts outer hair cell NLC to reduce active amplification
This study provides a new deafness mechanism due to Cx26 deficiency
Acknowledgments
This work was supported by NIH R01 DC 05989.
Footnotes
Conflict interest: The authors declare no competing financial interests.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Ashmore J. Cochlear outer hair cell motility. Physiol Rev. 2008;88:173–210. doi: 10.1152/physrev.00044.2006. [DOI] [PubMed] [Google Scholar]
- Beltramello M, Piazza V, Bukauskas FF, Pozzan T, Mammano F. Impaired permeability to Ins(1,4,5)P3 in a mutant connexin underlies recessive hereditary deafness. Nat Cell Biol. 2005;7:63–69. doi: 10.1038/ncb1205. [DOI] [PubMed] [Google Scholar]
- Brownell WE, Bader CR, Bertrand D, Ribaupierre Y. Evoked mechanical responses of isolated cochlear outer hair cells. Science. 1985;227:194–196. doi: 10.1126/science.3966153. [DOI] [PubMed] [Google Scholar]
- Castillo FJ, Castillo I. The DFNB1 subtype of autosomal recessive non-syndromic hearing impairment. Front Biosci. 2011;17:3252–3274. doi: 10.2741/3910. [DOI] [PubMed] [Google Scholar]
- Chan DK, Chang KW. GJB2-associated hearing loss: systematic review of worldwide prevalence, genotype, and auditory phenotype. Laryngoscope. 2014;124:E34–53. doi: 10.1002/lary.24332. [DOI] [PubMed] [Google Scholar]
- Chan DK, Schrijver I, Chang KW. Connexin-26-associated deafness: Phenotypic variability and progression of hearing loss. Genet Med. 2010;12:174–181. doi: 10.1097/GIM.0b013e3181d0d42b. [DOI] [PubMed] [Google Scholar]
- Chan DK, Hudspeth AJ. Ca2+ current-driven nonlinear amplification by the mammalian cochlea in vitro. Nat Neurosci. 2005;8:149–155. doi: 10.1038/nn1385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen J, Zhao HB. The role of an inwardly rectifying K+ channel (Kir4.1) in the inner ear and hearing loss. Neuroscience. 2014;265:137–146. doi: 10.1016/j.neuroscience.2014.01.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen J, Chen J, Zhu Y, Liang C, Zhao HB. Deafness induced by Connexin26 (GJB2) deficiency is not determined by endocochlear potential (EP) reduction but is associated with cochlear developmental disorders. Biochem Biophys Res Commun. 2014;448:28–32. doi: 10.1016/j.bbrc.2014.04.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohn ES, Kelley PM, Fowler TW, Gorga MP, Lefkowitz DM, Kuehn HJ, Schaefer GB, Gobar LS, Hahn FJ, Harris DJ, Kimberling WJ. Clinical studies of families with hearing loss attributable to mutations in the connexin 26 gene (GJB2/DFNB1) Pediatrics. 1999;103:546–550. doi: 10.1542/peds.103.3.546. [DOI] [PubMed] [Google Scholar]
- Cohen-Salmon M, Ott T, Michel V, Hardelin JP, Perfettini I, Eybalin M, Wu T, Marcus DC, Wangemann P, Willecke K, Petit C. Targeted ablation of connexin26 in the inner ear epithelial gap junction network causes hearing impairment and cell death. Curr Biol. 2002;12:1106–1111. doi: 10.1016/s0960-9822(02)00904-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dallos P. Cochlear amplification, outer hair cells and prestin. Curr Opin Neurobiol. 2008;18:370–376. doi: 10.1016/j.conb.2008.08.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dallos P, Wu X, Cheatham MA, Gao J, Zheng J, Anderson CT, Jia S, Wang X, Cheng WH, Sengupta S, He DZ, Zuo J. Prestin-based outer hair cell motility is necessary for mammalian cochlear amplification. Neuron. 2008;58:333–339. doi: 10.1016/j.neuron.2008.02.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ehret G. Development of absolute auditory thresholds in the house mouse (Mus musculus) J Am Audiol Soc. 1976;1:179–184. [PubMed] [Google Scholar]
- Engel-Yeger B, Zaaroura S, Zlotogora J, Shalev S, Hujeirat Y, Carrasquillo M, Barges S, Pratt H. The effects of a connexin 26 mutation--35delG--on oto-acoustic emissions and brainstem evoked potentials: homozygotes and carriers. Hear Res. 2002;163:93–100. doi: 10.1016/s0378-5955(01)00386-0. [DOI] [PubMed] [Google Scholar]
- Engel-Yeger B, Zaaroura S, Zlotogora J, Shalev S, Hujeirat Y, Carrasquillo M, Saleh B, Pratt H. Otoacoustic emissions and brainstem evoked potentials in compound carriers of connexin 26 mutations. Hear Res. 2003;175:140–151. doi: 10.1016/s0378-5955(02)00719-0. [DOI] [PubMed] [Google Scholar]
- Fridberger A, Flock A, Ulfendahl M, Flock B. Acoustic overstimulation increases outer hair cell Ca2+ concentrations and causes dynamic contractions of the hearing organ. Proc Natl Acad Sci USA. 1998;95:7127–7132. doi: 10.1073/pnas.95.12.7127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gopalarao D, Kimberling WJ, Jesteadt W, Kelley PM, Beauchaine KL, Cohn ES. Is hearing loss due to mutations in the Connexin 26 gene progressive? Int J Audiol. 2008;47:11–20. doi: 10.1080/14992020701602087. [DOI] [PubMed] [Google Scholar]
- Gummer AW, Hemmert W, Zenner HP. Resonant tectorial membrane motion in the inner ear: its crucial role in frequency tuning. Proc Natl Acad Sci USA. 1996;93:8727–8732. doi: 10.1073/pnas.93.16.8727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hudspeth AJ. Making an effort to listen: mechanical amplification in the ear. Neuron. 2008;59:530–545. doi: 10.1016/j.neuron.2008.07.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamiya K, Yum SW, Kurebayashi N, Muraki M, Ogawa K, Karasawa K, Miwa A, Guo X, Gotoh S, Sugitani Y, Yamanaka H, Ito-Kawashima S, Iizuka T, Sakurai T, Noda T, Minowa O, Ikeda K. Assembly of the cochlear gap junction macromolecular complex requires connexin 26. J Clin Invest. 2014;124:1598–1607. doi: 10.1172/JCI67621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kenna MA, Feldman HA, Neault MW, Frangulov A, Wu BL, Fligor B, Rehm HL. Audiologic phenotype and progression in GJB2 (Connexin 26) hearing loss. Arch Otolaryngol Head Neck Surg. 2010;136:81–87. doi: 10.1001/archoto.2009.202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kennedy HJ, Crawford AC, Fettiplace R. Force generation by mammalian hair bundles supports a role in cochlear amplification. Nature. 2005;433:880–883. doi: 10.1038/nature03367. [DOI] [PubMed] [Google Scholar]
- Kenneson A, Van Naarden Braun K, Boyle C. GJB2 (connexin 26) variants and nonsyndromic sensorineural hearing loss: a HuGE review. Genet Med. 2002;4:258–274. doi: 10.1097/00125817-200207000-00004. [DOI] [PubMed] [Google Scholar]
- Kikuchi T, Kimura RS, Paul DL, Adams JC. Gap junctions in the rat cochlea: immunohistochemical and ultrastructural analysis. Anat Embryol. 1995;191:101–118. doi: 10.1007/BF00186783. [DOI] [PubMed] [Google Scholar]
- Kraus HJ, Aulbach-Kraus K. Morphological changes in the cochlea of the mouse after the onset of hearing. Hear Res. 1981;4:89–102. doi: 10.1016/0378-5955(81)90038-1. [DOI] [PubMed] [Google Scholar]
- Liang C, Zhu Y, Zong L, Lu GJ, Zhao HB. Cell degeneration is not a primary causer for Connexin26 (GJB2) deficiency associated hearing loss. Neurosci Lett. 2012;528:36–41. doi: 10.1016/j.neulet.2012.08.085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu YP, Zhao HB. Cellular characterization of Connexin26 and Connnexin30 expression in the cochlear lateral wall. Cell Tissue Res. 2008;333:395–403. doi: 10.1007/s00441-008-0641-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murgia A, Orzan E, Polli R, Martella M, Vinanzi C, Leonardi E, Arslan E, Zacchello F. Cx26 deafness: mutation analysis and clinical variability. J Med Genet. 1999;36:829–832. [PMC free article] [PubMed] [Google Scholar]
- Orzan E, Murgia A. Connexin 26 deafness is not always congenital. INT J Pediatro Otorhi. 2007;71:501–507. doi: 10.1016/j.ijporl.2006.12.002. [DOI] [PubMed] [Google Scholar]
- Pollak A, Skórka A, Mueller-Malesińska M, Kostrzewa G, Kisiel B, Waligóra J, Krajewski P, Ołdak M, Korniszewski L, Skarzyński H, Ploski R. M34T and V37I mutations in GJB2 associated hearing impairment: evidence for pathogenicity and reduced penetrance. Am J Med Genet A. 2007;143A:2534–2543. doi: 10.1002/ajmg.a.31982. [DOI] [PubMed] [Google Scholar]
- Rajagopalan L, Greeson JN, Xia A, Liu H, Sturm A, Raphael RM, Davidson AL, Oghalai JS, Pereira FA, Brownell WE. Tuning of the outer hair cell motor by membrane cholesterol. J Biol Chem. 2007;282:36659–36670. doi: 10.1074/jbc.M705078200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santarelli R, Scimemi P, Dal Monte E, Genovese E, Arslan E. Auditory neuropathy in systemic sclerosis: A speech perception and evoked potential study before and after cochlear implantation. Eur Arch Otorhinolaryngol. 2007;263:809–815. doi: 10.1007/s00405-006-0075-1. [DOI] [PubMed] [Google Scholar]
- Santos-Sacchi J, Kakehata S, Takahashi S. Effects of membrane potential on the voltage dependence of motility-related charge in outer hair cells of the guinea-pig. J Physiol. 1998;510:225–235. doi: 10.1111/j.1469-7793.1998.225bz.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun Y, Tang W, Chang Q, Wang Y, Kong W, Lin X. Connexin30 null and conditional connexin26 null mice display distinct pattern and time course of cellular degeneration in the cochlea. J Comp Neurol. 2009;516:569–579. doi: 10.1002/cne.22117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y, Chang Q, Tang W, Sun Y, Zhou B, Li H, Lin X. Targeted connexin26 ablation arrests postnatal development of the organ of Corti. Biochem Biophys Res Commun. 2009;385:33–37. doi: 10.1016/j.bbrc.2009.05.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilcox SA, Saunders K, Osborn AH, Arnold A, Wunderlich J, Kelly T, Collins V, Wilcox LJ, McKinlay Gardner RJ, Kamarinos M, Cone-Wesson B, Williamson R, Dahl HH. High frequency hearing loss correlated with mutations in the GJB2 gene. Hum Genet. 2000;106:399–405. doi: 10.1007/s004390000273. [DOI] [PubMed] [Google Scholar]
- Yu N, Zhao HB. ATP activates P2x receptors and requires extracellular Ca2+ participation to modify outer hair cell nonlinear capacitance. Pflugers Arch. 2008;457:453–461. doi: 10.1007/s00424-008-0522-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu N, Zhu ML, Johnson B, Liu YP, Jones RO, Zhao HB. Prestin up-regulation in chronic salicylate (aspirin) administration: An implication of functional dependence of prestin expression. Cell Mol Life Sci. 2008;65:2407–2418. doi: 10.1007/s00018-008-8195-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu N, Zhao HB. Modulation of outer hair cell electromotility by cochlear supporting cells and gap junctions. PLoS One. 2009;4(11):e7923. doi: 10.1371/journal.pone.0007923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yum SW, Zhang J, Scherer SS. Dominant connexin26 mutants associated with human hearing loss have trans-dominant effects on connexin30. Neurobiol Dis. 2010;38:226–236. doi: 10.1016/j.nbd.2010.01.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zha D, Chen F, Ramamoorthy S, Fridberger A, Choudhury N, Jacques SL, Wang RK, Nuttall AL. In vivo outer hair cell length changes expose the active process in the cochlea. PLoS One. 2012;7:e32757. doi: 10.1371/journal.pone.0032757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang J, Scherer SS, Yum SW. Dominant Cx26 mutants associated with hearing loss have dominant-negative effects on wild type Cx26. Mol Cell Neurosci. 2011;47:71–78. doi: 10.1016/j.mcn.2010.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao HB. Directional rectification of gap junctional voltage gating between Deiters cells in the inner ear of guinea pig. Neurosci Lett. 2000;296:105–108. doi: 10.1016/s0304-3940(00)01626-8. [DOI] [PubMed] [Google Scholar]
- Zhao HB, Kikuchi T, Ngezahayo A, White TW. Gap junctions and cochlear homeostasis. J Memb Biol. 2006;209:177–186. doi: 10.1007/s00232-005-0832-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao HB, Santos-Sacchi J. Auditory collusion and a coupled couple of outer hair cells. Nature. 1999;399:359–362. doi: 10.1038/20686. [DOI] [PubMed] [Google Scholar]
- Zhao HB, Yu N. Distinct and gradient distributions of connexin26 and connexin30 in the cochlear sensory epithelium of guinea pigs. J Comp Neurol. 2006;499:506–518. doi: 10.1002/cne.21113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng J, Shen W, He DZ, Long KB, Madison LD, Dallos P. Prestin is the motor protein of cochlear outer hair cells. Nature. 2000;405:149–155. doi: 10.1038/35012009. [DOI] [PubMed] [Google Scholar]
- Zhu Y, Liang C, Chen J, Zong L, Chen GD, Zhao HB. Active cochlear amplification is dependent on supporting cell gap junctions. Nat Commun. 2013;4:1786. doi: 10.1038/ncomms2806. [DOI] [PMC free article] [PubMed] [Google Scholar]