Abstract
Background:
Endoscopic ultrasound-guided transbronchial fine-needle aspiration (EBUS-TFNA) is a minimally invasive technique for diagnosis of mediastinal masses/lesions. Although most studies have reported the utility of EBUS-TFNA in malignancy, its use has been extended to the benign conditions as well.
Objective:
The present study focused on utility of EBUS in contributing to reach the final diagnosis of sarcoidosis.
Design:
From May 2010 to December 2013, 19 of 80 patients who underwent EBUS-TFNA at one center for radiologically suspicious lesions for sarcoidosis, and with no definite histological diagnosis, were included in this retrospective study.
Results:
When the histological diagnosis was taken as the gold standard, the sensitivity of EBUS-TFNA was 84.2% and specificity 100% with the positive predictive value of 100. The combined diagnostic sensitivity of EBUS-TFNA and transbronchial lung biopsy was 100%.
Conclusion:
EBUS is a valuable, minimally invasive diagnostic modality to support the diagnosis of sarcoidosis in appropriate clinical setting, after conventional work-up – particularly if patients have suspicious radiological findings. This minimally invasive procedure helps in providing a final diagnosis without exposing the patient to the risk of complications from more invasive procedures.
Keywords: Endoscopic ultrasound-guided transbronchial fine-needle cytology, mediastinal lymph nodes, sarcoidosis
INTRODUCTION
Sarcoidosis is multi-organ granulomatous disease of unknown etiology which affects mostly young males and females. The pulmonary and the mediastinal lymph node involvement by sarcoidosis is seen in approximately 90% and 85% cases, respectively.[1] An essential part of the diagnosis of sarcoidosis is the exclusion of alternative possibilities. Granulomas can be seen in a range of diseases including infection (Mycobacterium tuberculosis, Mycobacteria avium intracellulare, fungi), reaction to metals/minerals as berylliosis, aluminum and talc, hypersensitivity pneumonitis Wegener granulomatosis. Differentiation of sarcoidosis from tuberculosis is challenging especially in those regions where incidence of tuberculosis is high. Occasionally, the clinico-radiologic features of the two are overlapping.[2] Several tests are recommended in the initial evaluation of sarcoidosis as a routine procedure for all patients [Table 1].[3] There is no specific confirmatory test for the diagnosis of sarcoidosis but it can be greatly substantiated by combination of compatible clinical and radiological picture together with the pathologic evidence of typical noncaseating epithelioid cell granulomas in the absence of organisms.[4]
Table 1.
Recommended tests for initial evaluation of sarcoidosis

The diagnostic approach to pulmonary sarcoidosis has traditionally been transbronchial lung biopsy (TBLB)[5] with a diagnostic yield highly variable and operator dependent ranging from 32% to 100% for stage I (bilateral hilar lymphadenopathy without lung infiltrate)[6] and 63–100% in stage II (hilar lymphadenopathy with lung infiltrates on a chest radiograph).[7] However, TBLB can be associated with complications such as pneumothorax and bleeding. If this procedure is unsuccessful in obtaining a tissue diagnosis, more invasive surgical techniques have to be used, including mediastinoscopy, video assisted thoracoscopy surgery, and open lung biopsy.
These procedures have a diagnostic yield approaching 90%, but the drawback it requires general anesthesia, inpatient hospital stay, and associated with morbidity and mortality.[8,9]
Endoscopic ultrasound-guided transbronchial fine-needle aspiration (EBUS-TFNA) is a minimally invasive technique for the diagnosis of mediastinal lymphadenopathy. Although most studies have reported the utility of EBUS-TFNA in malignancy, its use has been extended to the benign conditions as well. Use of the on-site evaluation of the adequacy of the specimens has improved the sensitivity of this procedure by increasing the rate of adequate specimens.[10,11] Shidham's method can be applied to the remaining cytology specimen to improve the quality of diagnostic material in cell block sections.[12] Recently, it has been used as a diagnostic tool in pulmonary sarcoidosis with yields similar to those of TBLB.[13] This technique has substantially changed the clinical approach to patients with suspected sarcoidosis. It has emerged as an accurate, minimally invasive, safe technique, and sensitive method with the diagnostic yield > 82% in detecting noncaseating granulomas in the cytological smears of patients with a suspicion of pulmonary sarcoidosis.[14,15]
We undertook this present study through a retrospective review of EBUS-TFNA cytology samples obtained from the patients with suspected pulmonary sarcoidosis, to assess and evaluate the diagnostic yield and utility of this procedure for the detection and demonstration of noncaseating granulomas.
PATIENTS AND METHODS
Study design
At King Khalid University Hospital, Riyadh, EBUS-TFNA is often used as the diagnostic test of choice to confirm noncaseating granulomas in patients with suspected sarcoidosis. In the present study, 19 patients who underwent EBUS-TFNA between May 2010 and December 2013, for suspected sarcoidosis (with enlarged [>1 cm] hilar or mediastinal lymph nodes on chest computed tomography [CT]), were included in this study and were clinically and radiologically followed.
Method
Endoscopic ultrasound-guided transbronchial fine-needle aspiration was conducted by one experienced pulmonologist in the bronchoscopy suite on all the suspected sarcoidosis patients with mediastinal lymphadenopathy based on chest CT. These patients understood the procedure required to establish the diagnosis, and written informed consent was obtained from all the participants. Local anesthesia with mild conscious sedation was used for the procedure.
Endobronchial ultrasonography was performed using fiberoptic ultrasound bronchoscope with a linear scanning probe (BF-UC260FW, Olympus Medical System, Tokyo, Japan). A dedicated ultrasound scanner (ALOKA SSD-Alpha 10, ALOKA Ultrasound System, Japan) was used for image processing. Targeted lymph node was sequentially sampled with a dedicated 21-gauge needle (NA-201SX-4021, Olympus Medical System, Tokyo, Japan).
Smears were made at the bedside in the bronchoscopy suite.
Preparation of smears
The aspirated material from the needle was expelled onto glass slides by the operating bronchoscopist and smeared by a cytotechnologist. About 2–4 slides were prepared from each pass, taking care that any clotted material is preserved for cell block. Air-dried (for Diff quick staining), and fixed smears (fixed immediately in 95% ethyl alcohol for subsequent Papanicolaou staining) were prepared in an almost equal ratio with more emphasis on fixed smears. Pass number (indicating a specific site of collection) was marked on each slide and site of collection of each pass was noted on a separate paper.
One or two representative of the air-dried smears from each pass were immediately stained with rapid Romanowsky (Diff Quick stain from Shandon) and examined under microscope for specimen adequacy assessment, preliminary diagnostic interpretation if necessary and to suggest additional studies if indicated.
Additional material was collected for ancillary studies polymerase chain reaction (PCR) and was preserved in normal saline (for microbiology). Material collected for cell block was grossly examined before the end of the procedure, and if the collected material was still not enough to make a good cell block, an additional pass was dedicated to cell block only.
Cytology request form was completed by the consultant bronchoscopist, including the details of the procedure and site of specimen for each pass.
Number of fixed and air-dried slides and description of any other material being sent to the laboratory was noted on the request form. Sample was transported to the laboratory as early as possible and processed for staining of remaining slides, preparation of the cell block.
Preparation of the cell block
Remaining sediment including any clotted material was fixed immediately in cell block fixative (10% alcohol formalin), centrifuged and material transferred to a histology embedding the cassette, and processed for routine histological examination using standard techniques.
Microscopy
All smears and cell block sections were reviewed by experienced cytopathologist (Dr. ER). The aspirates were considered to be representative if noncaseating granulomas or nodal lymphoid tissue were found, or if the aspirate contained other cellular material that resulted in a specific diagnosis.
Granulomas are defined by loose collections of epithelioid or spindle cell histiocytes with lymphocytes, necrotic material and neutrophils. In sarcoidosis, numerous granulomas can be seen in cytological samples without evidence of necrosis [Figures 1 and 2]. It is important to exclude local sarcoid-like reactions and infective causes.
Figure 1.
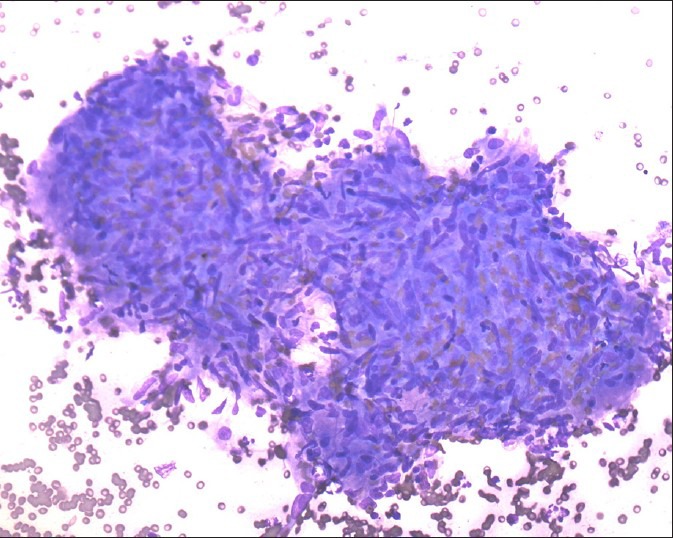
Noncaseating granuloma, smear shows well-defined and compact collection of epithelioid histiocytes with no evidence of necrosis (Diff Quick stain, ×200)
Figure 2.
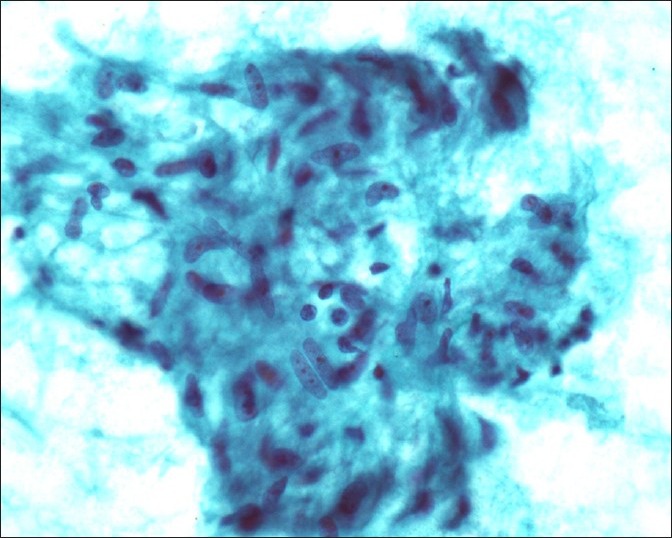
Granuloma, high power view, smear, well-delineated aggregate of epithelioid histiocytes reveal ill-defined cell borders, vesicular and elongated nuclei with few lymphocytes (Papanicolau stain, ×500)
In cases with subsequent trans-bronchial core biopsy, tissues were fixed in 10% formalin and thin histologic sections were cut from paraffin-embedded tissue blocks followed by H and E staining. A review of all available histologic slides was performed and correlated with the respective cytologic diagnoses.
Data analysis
In all the patients, the diagnosis of sarcoidosis was suspected based on the clinical and radiological findings. The final diagnosis was established based on lymph node aspirate by EBUS-TFNA cytology reveling noncaseating epithelioid cell granulomas, clinical and radiological follow-up, and histology in those cases where mediastinoscopy was performed. The sensitivity of EBUS-TFNA for the assessment of non-caseating granulomas was calculated as the number of patients in whom EBUS demonstrated granulomas as a proportion of the total of patients with a final diagnosis of sarcoidosis.
RESULTS
Data from 19 consecutive patients with suspected sarcoidosis who underwent EBUS-TFNA were analyzed. Eleven patients were male, and eight were female and the median age for all patients was 50 years (range 31–78 years).
In total, 23 lymph nodes were sampled from 19 patients by EBUS-TFNA. The subcarinal lymph node was most frequently sampled (11/23, 48%) followed by para-tracheal lymph nodes (6/23, 26%). About 13 out of 19 cases (68.4%) had simultaneous bronchial lavage; all of them were reported as inflammatory smears and were negative for malignancy.
Cell block was available for 12 samples, whereas cytology slides were available for all the samples. Out of these 19 patients, tuberculosis culture was done in 15 cases (83%). The sample was sent for PCR and fungal culture in 12 cases (63%) each, the results of which were negative in all the cases. By EBUS-TFNA cytology, granulomas were identified in 16 out of 19 patients (84.2%).
Sixteen cases (84.2%) of 19 cases had corresponding tissue biopsy. Eleven patient underwent trans bronchial biopsy, two cases had both transbronchial and endobronchial biopsy, two cases had both transbronchial and mediastinal trucut lymph node biopsy, one case had only mediastinal lymph node trucut biopsy, and one case had only endobronchial biopsy. The results are summarized in Table 2.
Table 2.
Comparison of diagnostic accuracy between procedures
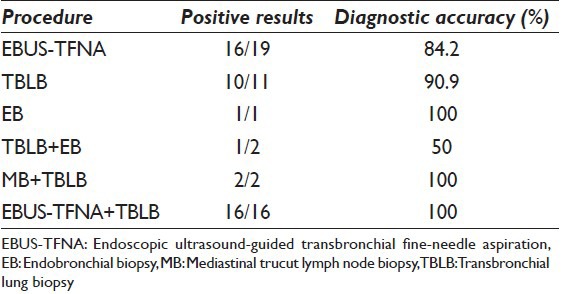
On histology, granulomas were identified in 13 cases (87%), of these; 9 cases (69%) were noncaseating granulomas. When the histological diagnosis was taken as the gold standard, the sensitivity of EBUS-TFNA was 84.2% and specificity 100% with the positive predictive value (PPV) of 100 and negative predictive value (NPV) of 25%. The combined diagnostic sensitivity of EBUS-TFNA and TBLB was 100%.
DISCUSSION
Until now; TBLB and bronchoscopic transbronchial needle aspiration (TBNA) have been the primary procedures to diagnose sarcoidosis with yields of 76–91%[16] and 62–87%[17] respectively. However, both procedures are performed blindly because the targeted lymph node or lung lesion are not visualized during biopsy or fine-needle aspiration.[18] Instead, aspiration is guided only by knowledge from prior CT imaging and consequently, the technique is considered difficult.[19]
If the lesion still remains undiagnosed, patients are often referred for more invasive procedures such as mediastinoscopy, thoracoscopy or even thoracotomy which carry a small but definite risk to the patient, and they are costly.[18]
Endoscopic ultrasound is a relatively new diagnostic modality which is less invasive, with real-time ultrasound imaging of the target which allows for a quick and reproducible diagnostic examination which is also easy to perform. It is less expensive than more invasive procedures because it allows investigation in an outpatient setting. In addition, it is associated with <1% significant bleeding or pneumothorax.[20,21]
Endoscopic ultrasound-guided transbronchial fine-needle aspiration was introduced in recent years to the Pulmonology department in our hospital as a new modality to diagnose and stage lung cancer and to evaluate mediastinal diseases. In a previous study from our institution, which included a total of 80 EBUS-TFNA specimens, we demonstrated that in approximately 78.75% (63 out of 80) patients with mediastinal mass/lesions, valid diagnosis in general can be reached in a minimally invasive manner by EBUS-TFNA.[22] The present study demonstrate that in patients with a high pretest probability of sarcoidosis, this procedure was able to establish the diagnosis in 84.2% of patients. Although, the sample size of our study was relatively small, the diagnostic yield was 83–91% in agreement with authors who had used EBUS among patients with suspected sarcoidosis.[23,24,25,26,27]
Data from our study confirm the results of the preliminary study by Annema et al.[14] (n = 51), where EBUS-TFNA demonstrated noncaseating granulomas in 82% of the patients with a suspected sarcoidosis stage I or II. Furthermore, the study published by Plit et al.[28] concluded that in patients with a high pretest probability of sarcoidosis, EBUS-TBNA had a high diagnostic accuracy of 84% for all stages, 80% for stage I and 86% for stage 2 disease which appeared similar to TBLB.
In a study by Fritscher-Ravens et al. [29] (n = 19 patients), they found a sensitivity and specificity of EBUS-TFNA in diagnosing sarcoidosis of 100% and 94%, respectively. Costabel et al.[30] reported EBUS-TBNA as a safe and useful tool for diagnosing the sarcoidosis stage I and II with a sensitivity of 83–93% and a specificity of 100%. The somewhat lower yield found in the current study cohort might be due to large number of patients with sarcoidosis stage II who tend to have more fibrotic LNs compared with stage I. In these particular cases with extensive fibrosis, EUS-FNA did not provide representative material.
Our TFNA diagnostic yield is lower than that reported by Nakajima et al.[7] who recently reported a diagnostic yield for EBUS-TFNA for all stages of 91.4%.[9] This can be partially explained by their cytological diagnostic criteria for sarcoidosis from aspirated lymph nodes which included the presence of epithelioid cells without granuloma formation which was reported in more than half their cases.
Agarwal et al.[31] did the meta-analysis of 15 studies that were published across the globe (553 patients of sarcoidosis) and concluded that the diagnostic yield of EBUS-TBNA ranged from 54% to 93% with the pooled diagnostic accuracy being 79% by the random effects model. The yield was not statistically different in studies employing on-site cytological evaluation (80.1%) versus those without (81.3%). However, the diagnostic yield was significantly higher in prospective studies (83.9%) as compared to the retrospective studies (74.3%).
CONCLUSION
Endoscopic ultrasound-guided transbronchial fine-needle aspiration is the minimally invasive, cost-effective, accurate, safe, and promising technique in identifying the granulomas and thus helps in supporting the diagnosis of mediastinal mass/lesions. It is, usually, performed under local anesthesia, using conscious sedation in an outpatient setting. Its sensitivity for identifying granulomas, hence supporting the diagnosis of sarcoidosis in our study was 84.2% and specificity 100% with the PPV of 100 and NPV of 25%, thus having a high diagnostic yield. However, the optimum use of EBUS-TFNA for reaching the confirmed diagnosis of sarcoidosis depends on the effective collaboration between the cytotechnologist, pathologist, and the bronchoscopist.
COMPETING INTERESTS STATEMENT BY ALL AUTHORS
The authors declare that they have no competing interests.
AUTHORSHIP STATEMENT BY ALL AUTHORS
All the authors of this article declare that we qualify for authorship as defined by ICMJE. Each author has participated sufficiently in the work and takes public responsibility for appropriate portions of the content of this article. Both authors read and approved the final manuscript. Each author acknowledges that this final version was read and approved.
ETHICS STATEMENT BY ALL AUTHORS
This study was conducted with the approval from Institutional Review Board (IRB) of King Khalid University Hospital, King Saud University, Riyadh, Saudi Arabia.
Author takes responsibility to maintain relevant documentation in this respect.
EDITORIAL/PEER-REVIEW STATEMENT
To ensure the integrity and highest quality of CytoJournal publications, the review process of this manuscript was conducted under a double blind model (authors are blinded for reviewers and vice versa) through automatic online system.
Contributor Information
Emad Raddaoui, Email: eraddaoui@yahoo.com.
Esam H. Alhamad, Email: esamalhamad@yahoo.com.
Shaesta Naseem Zaidi, Email: snz24@yahoo.com.
Maha Arafah, Email: marafah@hotmail.com.
Fatmah Fahad AlHabeeb, Email: dr.falhabeeb@hotmail.com.
REFERENCES
- 1.Von Bartheld MB, Veseliç-Charvat M, Rabe KF, Annema JT. Endoscopic ultrasound-guided fine-needle aspiration for the diagnosis of sarcoidosis. Endoscopy. 2010;42:213–7. doi: 10.1055/s-0029-1243890. [DOI] [PubMed] [Google Scholar]
- 2.Litinsky I, Elkayam O, Flusser G, Segal R, Yaron M, Caspi D. Sarcoidosis: TB or not TB? Ann Rheum Dis. 2002;61:385–6. doi: 10.1136/ard.61.5.385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Costabel U, Hunninghake GW. ATS/ERS/WASOG statement on sarcoidosis. Sarcoidosis Statement Committee. American Thoracic Society. European Respiratory Society. World association for sarcoidosis and other granulomatous disorders. Eur Respir J. 1999;14:735–7. doi: 10.1034/j.1399-3003.1999.14d02.x. [DOI] [PubMed] [Google Scholar]
- 4.Davis GS. The role of endobronchial ultrasound-guided transbronchial needle aspiration (EBUS TBNA) in the diagnosis of sarcoidosis. Cancer Cytopathol. 2014;122:239–40. doi: 10.1002/cncy.21380. [DOI] [PubMed] [Google Scholar]
- 5.Hunninghake GW, Costabel U, Ando M, Baughman R, Cordier JF, du Bois R, et al. ATS/ERS/WASOG statement on sarcoidosis. American Thoracic Society/European Respiratory Society/World Association of Sarcoidosis and other Granulomatous Disorders. Sarcoidosis Vasc Diffuse Lung Dis. 1999;16:149–73. [PubMed] [Google Scholar]
- 6.Shorr AF, Torrington KG, Hnatiuk OW. Endobronchial biopsy for sarcoidosis: A prospective study. Chest. 2001;120:109–14. doi: 10.1378/chest.120.1.109. [DOI] [PubMed] [Google Scholar]
- 7.Nakajima T, Yasufuku K, Kurosu K, Takiguchi Y, Fujiwara T, Chiyo M, et al. The role of EBUS-TBNA for the diagnosis of sarcoidosis – comparisons with other bronchoscopic diagnostic modalities. Respir Med. 2009;103:1796–800. doi: 10.1016/j.rmed.2009.07.013. [DOI] [PubMed] [Google Scholar]
- 8.Trisolini R, Lazzari Agli L, Cancellieri A, Poletti V, Candoli P, Paioli D, et al. Transbronchial needle aspiration improves the diagnostic yield of bronchoscopy in sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis. 2004;21:147–51. [PubMed] [Google Scholar]
- 9.Statement on sarcoidosis. Joint Statement of the American Thoracic Society (ATS), the European Respiratory Society (ERS) and the World Association of Sarcoidosis and Other Granulomatous Disorders (WASOG) adopted by the ATS Board of Directors and by the ERS Executive Committee, February 1999. Am J Respir Crit Care Med. 1999;160:736–55. doi: 10.1164/ajrccm.160.2.ats4-99. [DOI] [PubMed] [Google Scholar]
- 10.Griffin AC, Schwartz LE, Baloch ZW. Utility of on-site evaluation of endobronchial ultrasound-guided transbronchial needle aspiration specimens. Cytojournal. 2011;8:20. doi: 10.4103/1742-6413.90081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Monaco SE, Schuchert MJ, Khalbuss WE. Diagnostic difficulties and pitfalls in rapid on-site evaluation of endobronchial ultrasound guided fine needle aspiration. Cytojournal. 2010;7:9. doi: 10.4103/1742-6413.64385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Varsegi GM, Shidham V. Cell block preparation from cytology specimen with predominance of individually scattered cells. J Vis Exp. 2009 doi: 10.3791/1316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Reich JM, Brouns MC, O’Connor EA, Edwards MJ. Mediastinoscopy in patients with presumptive stage I sarcoidosis: A risk/benefit, cost/benefit analysis. Chest. 1998;113:147–53. doi: 10.1378/chest.113.1.147. [DOI] [PubMed] [Google Scholar]
- 14.Annema JT, Veseliç M, Rabe KF. Endoscopic ultrasound-guided fine-needle aspiration for the diagnosis of sarcoidosis. Eur Respir J. 2005;25:405–9. doi: 10.1183/09031936.05.00098404. [DOI] [PubMed] [Google Scholar]
- 15.Michael H, Ho S, Pollack B, Gupta M, Gress F. Diagnosis of intra-abdominal and mediastinal sarcoidosis with EUS-guided FNA. Gastrointest Endosc. 2008;67:28–34. doi: 10.1016/j.gie.2007.07.049. [DOI] [PubMed] [Google Scholar]
- 16.Trisolini R, Tinelli C, Cancellieri A, Paioli D, Alifano M, Boaron M, et al. Transbronchial needle aspiration in sarcoidosis: Yield and predictors of a positive aspirate. J Thorac Cardiovasc Surg. 2008;135:837–42. doi: 10.1016/j.jtcvs.2007.11.011. [DOI] [PubMed] [Google Scholar]
- 17.Cetinkaya E, Yildiz P, Altin S, Yilmaz V. Diagnostic value of transbronchial needle aspiration by Wang 22-gauge cytology needle in intrathoracic lymphadenopathy. Chest. 2004;125:527–31. doi: 10.1378/chest.125.2.527. [DOI] [PubMed] [Google Scholar]
- 18.Eckardt J, Olsen KE, Jørgensen OD, Licht PB. Minimally invasive diagnosis of sarcoidosis by EBUS when conventional diagnostics fail. Sarcoidosis Vasc Diffuse Lung Dis. 2010;27:43–8. [PubMed] [Google Scholar]
- 19.Haponik EF, Cappellari JO, Chin R, Adair NE, Lykens M, Alford PT, et al. Education and experience improve transbronchial needle aspiration performance. Am J Respir Crit Care Med. 1995;151:1998–2002. doi: 10.1164/ajrccm.151.6.7767550. [DOI] [PubMed] [Google Scholar]
- 20.Eapen GA, Shah AM, Lei X, Jimenez CA, Morice RC, Yarmus L, et al. Complications, consequences, and practice patterns of endobronchial ultrasound-guided transbronchial needle aspiration: Results of the AQuIRE registry. Chest. 2013;143:1044–53. doi: 10.1378/chest.12-0350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Von Bartheld MB, Dekkers OM, Szlubowski A, Eberhardt R, Herth FJ, in ’t Veen JC, et al. Endosonography vs conventional bronchoscopy for the diagnosis of sarcoidosis: The granuloma randomized clinical trial. JAMA. 2013;309:2457–64. doi: 10.1001/jama.2013.5823. [DOI] [PubMed] [Google Scholar]
- 22.Raddaoui E, Alhamad EH, Zaidi SN, Al-Habeeb FF, Arafah M. Utility and diagnostic accuracy of endobronchial ultrasound-guided transbronchial fine-needle aspiration cytology of mediastinal lesions: Saudi Arabian experience. Cytojournal. 2014;11:19. doi: 10.4103/1742-6413.137760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Tremblay A, Stather DR, Maceachern P, Khalil M, Field SK. A randomized controlled trial of standard vs endobronchial ultrasonography-guided transbronchial needle aspiration in patients with suspected sarcoidosis. Chest. 2009;136:340–6. doi: 10.1378/chest.08-2768. [DOI] [PubMed] [Google Scholar]
- 24.Tournoy KG, Bolly A, Aerts JG, Pierard P, De Pauw R, Leduc D, et al. The value of endoscopic ultrasound after bronchoscopy to diagnose thoracic sarcoidosis. Eur Respir J. 2010;35:1329–35. doi: 10.1183/09031936.00111509. [DOI] [PubMed] [Google Scholar]
- 25.Kitamura A, Takiguchi Y, Kurosu K, Takigawa N, Saegusa F, Hiroshima K, et al. Feasibility of cytological diagnosis of sarcoidosis with endobronchial US-guided transbronchial aspiration. Sarcoidosis Vasc Diffuse Lung Dis. 2012;29:82–9. [PubMed] [Google Scholar]
- 26.Oki M, Saka H, Kitagawa C, Kogure Y, Murata N, Ichihara S, et al. Prospective study of endobronchial ultrasound-guided transbronchial needle aspiration of lymph nodes versus transbronchial lung biopsy of lung tissue for diagnosis of sarcoidosis. J Thorac Cardiovasc Surg. 2012;143:1324–9. doi: 10.1016/j.jtcvs.2012.01.040. [DOI] [PubMed] [Google Scholar]
- 27.Gupta D, Dadhwal DS, Agarwal R, Gupta N, Bal A, Aggarwal AN. Endobronchial ultrasound-guided transbronchial needle aspiration vs conventional transbronchial needle aspiration in the diagnosis of sarcoidosis. Chest. 2014;146:547–56. doi: 10.1378/chest.13-2339. [DOI] [PubMed] [Google Scholar]
- 28.Plit M, Pearson R, Havryk A, Da Costa J, Chang C, Glanville AR. Diagnostic utility of endobronchial ultrasound-guided transbronchial needle aspiration compared with transbronchial and endobronchial biopsy for suspected sarcoidosis. Intern Med J. 2012;42:434–8. doi: 10.1111/j.1445-5994.2011.02446.x. [DOI] [PubMed] [Google Scholar]
- 29.Fritscher-Ravens A, Sriram PV, Topalidis T, Hauber HP, Meyer A, Soehendra N, et al. Diagnosing sarcoidosis using endosonography-guided fine-needle aspiration. Chest. 2000;118:928–35. doi: 10.1378/chest.118.4.928. [DOI] [PubMed] [Google Scholar]
- 30.Costabel U, Bonella F, Ohshimo S, Guzman J. Diagnostic modalities in sarcoidosis: BAL, EBUS, and PET. Semin Respir Crit Care Med. 2010;31:404–8. doi: 10.1055/s-0030-1262207. [DOI] [PubMed] [Google Scholar]
- 31.Agarwal R, Srinivasan A, Aggarwal AN, Gupta D. Efficacy and safety of convex probe EBUS-TBNA in sarcoidosis: A systematic review and meta-analysis. Respir Med. 2012;106:883–92. doi: 10.1016/j.rmed.2012.02.014. [DOI] [PubMed] [Google Scholar]


