Abstract
Schistosomiasis is a debilitating disease affecting approximately 600 million people in 74 developing countries, with 800 million, mostly children at risk. To circumvent the threat of having praziquantel (PZQ) as the only drug used for treatment, several PZQ derivatives were synthesized, and drugs destined for other parasites were used with success. A plethora of plant-derived oils and extracts were found to effectively kill juvenile and adult schistosomes, yet none was progressed to pre- and clinical studies except an oleo-gum resin extracted from the stem of Commiphora molmol, myrrh, which action was challenged in several trials. We have proposed an essential fatty acid, a component of our diet and cells, the polyunsaturated fatty acid arachidonic acid (ARA) as a remedy for schistosomiasis, due to its ability to activate the parasite tegument-bound neutral sphingomyelinase, with subsequent hydrolysis of the apical lipid bilayer sphingomyelin molecules, allowing access of specific antibody molecules, and eventual worm attrition. This concept was convincingly supported using larval and adult Schistosoma mansoni and Schistosoma haematobium worms in in vitro experiments, and in vivo studies in inbred mice and outbred hamsters. Even if ARA proves to be an entirely effective and safe therapy for schistosomiasis, it will not prevent reinfection, and accordingly, the need for developing an effective vaccine remains an urgent priority. Our studies have supported the status of S. mansoni calpain, glutathione-S-transferase, aldolase, triose phosphate isomerase, glyceraldehyde 3-phosphate dehydrogenase, enolase, and 2-cys peroxiredoxin as vaccine candidates, as they are larval excreted-secreted products and, contrary to the surface membrane molecules, are entirely accessible to the host immune system effector elements. We have proposed that the use of these molecules, in conjunction with Th2 cytokines-inducing adjuvants for recruiting and activating eosinophils and basophils, will likely lead to development and implementation of a sterilizing vaccine in a near future.
Keywords: Schistosoma mansoni, Schistosoma haematobium, Schistosomicides, Arachidonic acid, Excretory–secretory products, Type 2 adjuvants
Schistosomiasis
Schistosomiasis, a debilitating disease endemic in 74 countries of the developing world, does not affect 207 million people as reported in 2006 [1], as recent articles document infection of 391–597 million people, with 800 million, mostly children, at risk of the infection [2–4]. Additionally, schistosomiasis does not cause the annual loss of between 1.7 and 4.5 [1], but between 24 and 56 [2], and up to 70 [3,4] million disability adjusted life years. Egyptian health authorities and public media continuously advocate the near elimination of schistosomiasis from Egypt, statement that was not denied by the patients in rural populations because of the stigma of poverty and social status inferiority linked to this infection. Yet, several articles document that Egypt has more than 10 million persons infected with schistosomes [2,5,6]. An article published in 2012 reports that Egypt is the MENA (Middle East North African) country with the highest prevalence (7.2 out of 80.4 million people, about 9%) of schistosomiasis [7]. This controversy must be laid at rest via generation or publication of objective, and scientifically valid data regarding the present prevalence of schistosomiasis in Egypt.
Schistosomiasis is caused by the platyhelminth worms of the genus Schistosoma, trematodes that live in the bloodstream of humans and animals. Three species (Schistosoma mansoni, Schistosoma haematobium and Schistosoma japonicum) account for the majority of human infections. Cercariae break out of the snail tissues into the water, swimming actively till dying or penetrating the unbroken skin of humans or animals, the definitive host, where they change into schistosomula. During the first 24 h after infection, nearly 90% of S. mansoni and S. haematobium schistosomula are present only in the blood-free, lymph-free epidermis. Majority of schistosomula are found in the dermis only after 48 h, and they appear to reach the dermal vessels around 72 h after infection [8,9]. Once in the blood capillaries, the schistosomula are carried passively by the blood flow till reaching the right heart and then the lungs. Depending on the species, schistosomula stay inside the pulmonary capillaries from 3 to 16 days, where they change into much longer and slender organisms, such a shape that enables them to traverse the thin pulmonary capillaries to the left heart and the systemic circulation [10]. Following this period, the larvae make their way to the liver via the splanchnic vasculature. Upon reaching the liver, schistosomula start feeding and growing by active cell division. Once they reach maturity the worms start pairing, between 28 and 35 days post-infection. The paired adults migrate out of the liver, with the male carrying the female, to where they will finally reside in the mesenteric veins (S. mansoni), or in the pelvic venous plexus and veins around the bladder (S. haematobium) [11,12]. Massive numbers of daily deposited eggs must traverse the walls of the blood venules to enter the lumen of the intestine or bladder, to be excreted with the feces or urine. The morbidity associated with schistosomiasis results from the immunologic reactions to egg-derived antigens, beside the mechanical and toxic irritation caused by eggs trapped in the wall of blood vessels [11–14]. Some of the most common pathological changes seen in chronic schistosomiasis infections include bleeding into the intestine or urinary system, liver and spleen enlargement, and periportal fibrosis [11–14]. A connection between the chronic urinary form of schistosomiasis and bladder cancer is suspected [12,14].
In addition to the severe sequelae of schistosomiasis, the infection, even light, predisposes the host to, and accelerates the course and increases the severity of, infection with human immunodeficiency [15,16], and hepatitis B and C (HCV) viruses. For example, Egypt has the highest prevalence of HCV being >15% in rural areas where schistosomiasis is endemic, whereby co-infection with schistosomiasis causes more severe liver disease than infection with HCV alone [17,18]. Infection with schistosomes renders the host particularly susceptible to mycobacteria, and protozoa, especially of the genus Plasmodium, Leishmania, Toxoplasma, and Trypanosoma [reviewed in 19].
Schistosomicides
Antimonial compounds, usually used for cure of leishmaniasis, have been the cornerstone of schistosome chemotherapy for about 50 years. Their mode of action is believed to be strong inhibition of the schistosome phosphofructokinase (the enzyme catalyzing the conversion of fructose-6-phosphate to fructose-1,6-diphosphate) at concentrations 65–80 times lower than those effective against the human enzyme [20]. This wide difference indicates that inhibition of host phosphofructokinase cannot be the only cause for the antimonial drugs’ excessive toxicity and severe side effects, which have rendered them now obsolete.
The organophosphorus insecticide 2,2,2-trichloro-1-hydroxyethyl dimethyl phosphonate was slightly modified to give rise to metrifonate (O,O-dimethyl-2,2,2-trichloro-1-hydroxyethylphosphonate), which has showed constant activity against S. haematobium. Metrifonate is administered orally, and is metabolized leading to several byproducts, among which dichlorvos is the most potent, inducing schistosome acetylcholinesterase inhibition with subsequent reversible paralysis of the worm [21,22]. Paralyzed schistosomes lose their hold on the inner wall of blood vessels and are swept away with the blood stream, S. mansoni to the liver and S. haematobium to the lungs via the vena cava. Upon drug concentration decrease, S. mansoni are capable of regaining their original location in the mesenteric veins, whereas S. haematobium remain trapped in the lungs [23]. This might explain that metrifonate mediates killing of S. haematobium but not S. mansoni. It is of note that the drug also depresses host plasma and erythrocyte cholinesterase, explaining the severe side effects: fatigue, muscular weakness, tremor, sweating, fainting, diarrhea, nausea, vomiting, bronchospasm, consequences of cholinergic stimulation. Besides, three oral doses are absolutely required to reach a cure rate of 90%. This might explain why metrifonate has been taken out of the World Health Organization (WHO) list of essential drugs [24], leaving oxamniquine and praziquantel the only available drugs for treating schistosomiasis.
Oxamniquine, 1,2,3,4-tetrahydro-2-(isopropylamino)methyl(-)7-nitro-6-nitro-quinoline methanol, when administered orally is effective against S. mansoni, male worms being more affected than females, while it has no effect on S. haematobium. A single, two or three oral doses of 20 mg/kg each, are needed for a cure rate of 80–90%, depending on the geographic region. After a curative dose in healthy volunteers, peak plasma concentrations of 1–4 mg/L are reached in 1–4 h and the plasma half-life is of the order of 1.5–2 h. Oxamniquine concentrations of 40 μg/mL are also lethal to adult S. mansoni in vitro [25,26]. It is now believed that oxamniquine undergoes esterification by a sulfotransferase uniquely present in sensitive schistosomes. The ester spontaneously dissociates, yielding an electrophilic reactant capable of alkylating schistosome DNA, with subsequent inhibition of DNA and RNA synthesis; the absence of this enzyme in mammals, including humans, explains the low toxicity of oxamniquine [27]. Resistance to oxamniquine has emerged in various foci, perhaps due to a mutation in the schistosome gene encoding the critical bioactivating (esterifying) enzyme [27,28]. Oxamniquine is used on a large scale only in Brazil. For the rest of the world, a single drug is now in use for treatment of schistosomiasis: praziquantel.
Praziquantel (PZQ), a pyrazino-isoquinoline derivative is practically insoluble in water, sparingly soluble in ethanol, but very soluble in chloroform and dimethylsulfoxide. It is the drug of choice for treatment of all five species of human schistosomes, leading to cure rates of 60–90% in different epidemiological settings [29,30]. Adult schistosomes exposed in vitro to 1–3 μM PZQ undergo almost immediate spastic paralysis. In parallel, vacuolization of parts of the tegument and surface blebbing occur, especially in male worms (Table 1). All worms die thereafter. These reproducible findings indicate that adult schistosomes bind PZQ, and that PZQ per se is schistosomicidal [31]. In vivo, plasma concentrations of 1–10 μM are readily reached after a standard curative dose. In vivo, PZQ-induced muscle contraction and tegumental lesions produce a loss of attachment to the endothelial veins and dislodgment to the liver. Host cells of the defense system attach to the tegumental vacuoles and start to penetrate to the interior of the parasite early after treatment [28,32–34].
Table 1.
Schistosomicides target and mechanism of action.
| Drug | Mode of action | References |
|---|---|---|
| Antimonials | Inhibition of schistosome phosphofructokinase | [20] |
| Metrifonate | Schistosome acetylcholinesterase inhibition and paralysis | [22] |
| Oxamniquine | Mediates inhibition of parasite DNA and RNA synthesis | [27] |
| Praziquantel | Still elusive; binding to parasite calcium channels? | [35–38] |
| Tegument disruption? | [40–42] | |
| Binding and polymerization of actin? | [56–60] | |
| Exposure of surface membrane antigens | [33,34] | |
| Artemether | Interacts with haemin | [71–74] |
| Mefloquine | Inhibits hemozoin formation | [76] |
| Acyclic nucleotide analogues | Inhibit parasite DNA polymerase? | [79,80] |
| Oxadiazoles | Inhibit parasite thioredoxin-glutathione reductase | [81,82] |
| Trioxaquines | Alkylation of, and stacking with, heme | [83] |
| Myrrh | Worm uncoupling and extravasation | [98] |
Despite that the initial effects of the drug included a rapid influx of calcium into the worm and calcium-dependent muscle contraction and paralysis, the exact mode of action of PZQ is not known as yet [35–38], and the PZQ receptor on schistosomes remains elusive [38,39]. One hypothesis to explain PZQ mode of action is that this drug inserts itself in the membrane producing lipid phase transition [40] and subsequent destabilizing effects on the membrane [41]. It is certainly documented that PZQ interacts with the surface tegument and severs it dramatically [42].
Control initiatives in numerous countries, including Egypt, rely on large-scale administration of PZQ [43–47]. Therefore, reports of low drug efficacy [48–50] and schistosome resistance to the drug [39,51–55] have raised intense alarm. The likely possibility that PZQ binds and polymerizes schistosome and mammalian actin [56–60] should preclude its use in children, pregnant women, and individuals with vascular diseases [61].
Novel therapeutic approaches
PZQ derivatives and combination chemotherapy
Intense efforts are now directed at circumventing the flaw of having a single drug for such a dreadful infection via synthesizing derivatives of PZQ [39,62] and benzodiazepine schistosomicides [63], and examining their schistosomicidal activity in vitro and in vivo. Additionally, attempts were conducted to use PZQ in combination with other potential schistosomicides such as artemether [64–68], or hepatoprotective remedies [69,70] in an aim to reduce the PZQ dose, potentiate its schistosomicidal action, and alleviate side effects in experimental hosts.
Novel drugs
Several drugs targeting other parasites were tested for potential schistosomicidal activity (Table 1). The anti malarial drug artemether, a methoxy derivative of artemisinin, has been shown to be active against S. japonica, S. mansoni, and S. haematobium in experimentally infected animals [71]. A single oral injection of 400 mg/kg artemether to mice infected with approximately 80 cercariae of S. mansoni 21 (pre-patent) or 49 (patent period) days earlier led to 71–81% reduction in total worm burden [72–75]. Mefloquine, another anti-malarial drug, was also found to have significant anti-schistosome activity in vitro, and in vivo as well, as a single dose (200 or 400 mg/kg), administered orally to mice infected with adult S. mansoni, resulted in high worm burden reductions of 72.3–100% [76,77]. It has been shown that artemether interacts with haemin to exert a toxic effect on schistosomes, while mefloquine is believed to inhibit hemozoin formation. Yet, there are objections regarding a possible interference with the primary use of artemether and mefloquine as antimalarials [71–77]. Trioxolanes, which were considered for their potent anti-malarial activity, showed significant schistosomicidal activity in vitro and in vivo against S. mansoni and S. japonicum [78]. Acyclic nucleotide analogues, such as (S)-9-[3-hydroxy-2-(phosphonomethoxy) propyl]adenine [(S)-HPMPA] and their derivatives, possess effective broad-spectrum activity against many viruses via inhibition of DNA polymerase and/or reverse transcriptase, and were found to be actively schistosomicidal in vivo and in vitro [79,80]. The anti-microbial and anti-cancer oxadiazoles, which are five-membered heteroaromatic rings containing two carbons, two nitrogens, and one oxygen atom, have been found to possess inhibitory activity against S. mansoni and S. japonicum redox protein thioredoxin-glutathione reductase [81,82]. Trioxaquines, hybrid drugs containing a 1,2,4-trioxane and a 4-aminoquinoline, initially developed against malaria, exhibit a dual mode of action: alkylation of heme with the trioxane entity, and stacking with heme due to the aminoquinoline moiety, leading to inhibition of hemozoin formation in vitro, thus explaining their potent anti-S. mansoni activity in vitro and in vivo [83]. Imidazolidines, pentagonal heterocyclic compounds with broad biological anti-microbial and anti-fungal activities, were considered for treatment of schistosomiasis because of their potent in vitro schistosomicidal effects [84]. Of great interest is the class of compounds targeting schistosome histone modifying enzymes, namely histone acetyltransferases and histone deacetylases, and leading to parasite apoptosis and death in in vitro cultures [85].
Plant-derived schistosomicidal compounds
A different approach to therapy of schistosomiasis relied on plants known for medicinal effects. Extracts and oils of several medicinal plants were tested for potential therapeutic activity against schistosome infection [86,87], and were exhaustively compiled in excellent reviews [88–91]. Curcumin, the major constituent in the rhizome of Curcuma longa has been shown to display potent schistosomicidal activities in vivo and in vitro against adult S. mansoni worms [92–96]. The most successful product was myrrh, an oleo-gum resin extracted from the stem of Commiphora molmol [97], believed to affect schistosome musculature, leading to uncoupling of male and female couples and their extravasation to the liver tissue [98]. The product has been licensed for human use by the Egyptian Ministry of Health, and introduced in the Egyptian market in the form of gelatinous capsules by the pharmaceutical company, Pharco (Alexandria, Egypt) under the name of Mirazid. However, conflicting reports on its efficacy shed doubts upon the usefulness of its use as a novel therapy for schistosomiasis [98–103].
Arachidonic acid, a novel remedy for schistosomiasis
Mode of action
Our approach to the therapy of schistosomiasis is entirely novel and is herein clarified. Developing and adult schistosomes are covered by the tegument, a 2–4 μm thick syncytium. The innermost membrane of the tegument is a conventional membrane (the basal membrane), whereas the outer membrane on the syncytium is trilaminate in cercariae. By about 3 h after penetration, the trilaminate outer membrane of the cercariae is gradually replaced by a unique heptalaminate membrane, widely believed to cause protection against elements of the host immune system. Notably, the surface membrane at the parasite/host interface consists of two tightly apposed bilayers, the inner and outer bilayers, each of which is composed of inner and outer leaflets [104]. As for conventional plasma membranes, proteins are embedded in schistosome outer and inner lipid bilayers among phosphoglycerides, cholesterol and sphingomyelin molecules [105]. Conversely from conventional plasma membranes, proteins in the schistosome outer lipid bilayer are concealed, hidden, and totally inaccessible to host antibodies. We have recently shown that this major immune evasion phenomenon is due to the sphingomyelin of the outer lipid bilayer, likely via concealment of surface membrane proteins behind a tight barrier of hydrogen bonds sphingomyelin readily forms with water molecules [106–110].
Sphingomyelin hydrolysis is catalyzed by a group of enzymes called sphingomyelinases, yielding ceramide and phosphorylcholine. Many sphingomyelinase activities have been identified and may be classified into two main groups. The acid forms function at an optimal pH of 4.5, and are found in the cell acidic compartments. The neutral forms function at a neutral pH and are found in different locations within the cell, especially the outer membrane [111]. Tegument-bound neutral sphingomyelinase (nSMase) activity is optimal at pH > 7.4, exhibits an absolute dependence on magnesium or manganese (2 and 10 mM for Mg2+ and up to 2.5 mM for Mn2+), and is readily inhibited by the compound GW4869 [112]. Like mammalian nSMase [111–113], S. mansoni tegument-associated nSMase activity is increased several folds, in vitro and in vivo, by polyunsaturated fatty acids, especially arachidonic acid (ARA) [114–116]. We have documented that in vitro treatment of lung-stage schistosomula and adult worms with unsaturated fatty acids, especially ARA, leads to exposure of otherwise hidden surface membrane antigens to specific antibody binding [106,109,110,117]. More importantly, feeding S. mansoni or S. haematobium-infected mice with unsaturated fatty acids-enriched food led to significant (P < 0.02) decrease in worm burden [109]. Accordingly, we proposed that ARA may represent a novel remedy for schistosomiasis.
Arachidonic acid properties
Arachidonic acid (ARA) is an omega-6 fatty acid: 20:4(ω-6), with a 20-carbon chain and four cis double bonds; the first double bond located at the sixth carbon from the omega end (Fig. 1). It is present in the phospholipids of membranes of the body’s cells, and is abundant in the brain. The ARA four cis double bonds (Fig. 1) are the source of its flexibility, keeping the pure fatty acid liquid, even at subzero temperatures, and helping to give mammalian cell membranes their correct fluidity at physiological temperatures. The double bonds are also the key to the propensity of ARA to react with molecular oxygen. This can happen nonenzymatically, contributing to oxidative stress, or through the actions of three types of oxygenase: cyclooxygenase (COX), lipoxygenase (LOX), and cytochrome P450 [118,119].
Fig. 1.
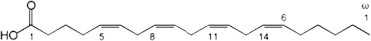
Arachidonic acid structure.
Proposing intake of exogenous ARA could be met with awe, as ARA may be metabolized to end products that produce pain and inflammation, namely the prostaglandins (COX pathway), which are mediators of the vascular phases of inflammation, are potent vasodilators, and increase vascular permeability; prostacycline (vasodilator and reduces platelet aggregation); thromboxane (increases vasoconstricton and platelet aggregation); and leukotrienes (LOX pathway) that are important mediators of inflammation. Of great concern too, is the fact that exogenous ARA is acted upon by COX and LOX enzymes, despite that many of which are located on the endoplasmic reticulum or nuclear membrane [118,119].
We have embarked on use of ARA for schistosomiasis therapy sure of the fact that ARA is an essential constituent of membrane lipids, and is the base material used by the body to synthesize a key series of hormones referred to collectively as dienolic prostaglandins (PG) that include the prostaglandins PGE2 and PGF2a. Proper development of the brain, retina and other body tissues depends upon provision of ARA either directly in the diet or through synthesis from linoleic acid (LA). Additionally, ARA is an essential fatty acid, which is consumed in our regular diets. It is found mainly in lean meat, egg yolks and some fish oils. Tissue ARA pools originate from the diet, and from hepatic and extrahepatic desaturation–elongation of dietary LA; in humans who ingest 0.2–0.3 g of ARA and 10–20 g of LA per day on a Western diet the formation of ARA from LA exceeds the dietary supply of ARA [120]. Indeed, most evidence supports that ingestion of moderate amounts of ARA is not disadvantageous. In a series of studies, humans ingested a diet containing 1.7 g of ARA per day for 50 days, and were then extensively studied. Dietary ARA at these levels nearly doubled ARA levels in the plasma phospholipids and cholesteryl ester, whereby ARA mainly replaced LA, which was reduced by 20%. Some increases in platelets, red blood cells, and tissue lipids were also found, but no significant increase in ARA was seen in adipose tissue triglycerides and phospholipids. The plasma free fatty acids showed a small but statistically significant rise in the ARA level. No harmful effects could be proven during the studies, although an increased production of eicosanoids was observed [121].
It is of note that ingestion of ω-3 essential fatty acids, namely EPA (eicosapentaenoic acid) entirely protects from any inflammatory effect ARA might elicit, by displacement, competitive inhibition, and direct counteraction. Docosahexaenoic acid (DHA) was reported to have an effect similar to EPA [122,123]. DHA is an omega-3 essential fatty acid, 22:6(ω-3), with a 22-carbon chain and six cis double bonds; the first double bond located at the third carbon from the omega end. Fortunately, Martek Biosciences Corporation recently produced infant formulas and capsules containing ARA (ARASCOR) and DHA. Using controlled fermentation conditions, Martek Biosciences obtains ARA from the soil fungus Mortierella alpina, and DHA from the non-photosynthetic marine micro-algae Crypthecodinium cohnii, products of proven entire safety [124]. Molecular Nutrition Company developed X-Factor, Arachidonic Acid as a food supplement during exercise. X FactorTM Arachidonic Acid is provided in the form of Softgels containing each 250 mg ARA, whereby 1 to 5 Softgels are recommended per day. In addition, ARA (ARASCOR, Martek Biosciences Corporation) was recently allowed to be added to infant formulas.
Schistosomicidal effects in vitro and in vivo
In our studies, pure ARA from Sigma was used for in vitro and preliminary in vivo studies, while ARA from Martek was used for in vivo studies [125,126]. We have demonstrated that 5 mM pure ARA (Sigma) leads to irreversible killing of ex vivo 1, 3, 4, 5, and 6 week-old S. mansoni and 9, 10 and 12 week-old S. haematobium worms, within 3–4 h, depending on the parasite age, even when worms were maintained in up to 50% fetal calf serum. ARA-mediated worm attrition was prevented by nSMase inhibitors such as CaCl2 and GW4869. Scanning and transmission electron microscopy revealed that ARA-mediated worm killing was associated with spine destruction, membrane blebbing, and disorganization of the apical membrane structure [125]. ARA-mediated S. mansoni and S. haematobium worm attrition was reproduced in vivo whereby a series of 20 independent experiments using BALB/c or C57BL/6 mice, or hamsters indicated that ARA in a pure form (Sigma), included in infant formula (Nestle), or capsules (X Factor, Molecular Nutrition) consistently led to between 50% and 80% decrease in S. mansoni or S. haematobium worm burden [125,126]. ARA-mediated attrition in vivo appeared to be associated with high titers of serum antibodies to tegumental antigens. In support, serum antibodies from patently-infected and ARA-administered hamsters readily bound to the surface membrane of ARA-treated, but not untreated, adult worms, as judged by indirect membrane immunofluorescence. More importantly, addition of serum antibodies and peripheral blood mononuclear cells significantly enhanced ARA-mediated adult worm attrition in vitro [126]. Immune responses to adult worm tegumental antigens are certainly more powerful in adults and children with patent infection than in hamsters, and ARA is already marketed for human use in USA and Canada for proper development of newborns, and muscle growth of athletes. Accordingly, it is recommended to start pre-clinical and clinical studies in human volunteers for development of ARA as a safe and cost-effective remedy to schistosomiasis, especially that no ARA-related adverse effects were seen in any experiment in mice or hamsters. Indeed, no changes were observed between untreated and ARA-treated experimental hosts in serum levels of triglycerides or cholesterol, number of erythrocytes or platelets, thrombin or prothrombin values in accord with the numerous reports documenting the entire safety of ARA ingestion [125,126].
Vaccine candidates and trials
The possibility of developing an effective vaccine for schistosomiasis stemmed from the highly significant protection obtained in experimental hosts using irradiated cercariae [reviewed in 127] and from the identification of humans resistant to schistosomiasis. Individuals classed as ‘‘endemic normals’’ are repeatedly exposed to infective cercariae and yet have no record of previous or current infection, and appear uninfected as judged by repeated stool examination. ‘‘Resistant’’ subjects are those whose stool and urine samples remain negative for eggs or show very low levels of reinfection following drug treatment of previous S. mansoni or S. haematobium infection despite unequivocal evidence of continued and high contact with schistosome-contaminated watersources [128–135]. The first subunit vaccine candidate antigens discovery was based on these models, whereby antibodies from irradiated cercariae (RA) vaccine-immunized mice and resistant individuals were used to screen schistosome cDNA libraries. These studies led to identification of S. mansoni irradiation associated vaccine antigen, IrV 5 [136,137], glutathione-S-transferase, GST [138,139], triose phosphate isomerase, TPI [140–142], paramyosin [131,143,144], fatty acid binding protein, Sm 14, [145], the surface membrane antigen Sm23 [146–148], the calcium-dependent cysteine protease, calpain [4,149], and glyceraldehyde 3-phosphate dehydrogenase [130,150,151]. We have challenged the unique use of serum antibodies for selection of schistosome candidate vaccine antigens, because of the extensive cross-reactivity of B cell epitopes and the need to ascertain that the selected antigen is able to induce T cell help. Accordingly, we have used the T cell western technique to examine the proliferative and cytokine production activity of mouse spleen cells (SC) and human peripheral blood mononuclear cells (PBMC) in response to electroseparated and electroblotted soluble adult worm antigens (SAWA). Extensive analyses of the T and B cell reactivity of gamma or ultraviolet-irradiated cercariae-immunized inbred and outbred mice to separated SAWA antigens [152–154] led to identification of S. mansoni enolase [152,155] and calreticulin [156] as the antigens preferentially recognized by RA vaccinated mice. Studies using PBMC and sera obtained from endemic resistant humans or individuals resistant to reinfection after PZQ treatment ended into rediscovery of glyceraldehyde 3-phosphate dehydrogenase (SG3PDH) as potential candidate vaccine antigen [134,135]. The cDNA encoding the full length molecule was cloned and expressed by Charles Shoemaker, and SG3PDH joined the group of candidate vaccine antigens, IrV5, paramyosin, GST, TPI, Sm14, Sm23, and calpain selected by the World Health Organization (WHO) for in vitro and in vivo trials. The in vitro studies using PBMC and sera of Egyptian [157] and Brazilian [158] donors led to inconclusive answers, while the in vivo trials in inbred mice indicated that none of the vaccine candidate antigens induced higher than 40% reduction in challenge worm burden, the benchmark set by the WHO for progression of schistosome vaccine antigens into clinical trials [159].
The S. mansoni fatty acid-binding protein, Sm14 antigen stands out, due to its steady progress towards field trials. Work has now progressed to the scale-up level and an industrial production process has successfully been put in place [160]. S. mansoni calpain in recombinant or DNA constructs appeared as successful, inducing protection levels higher than 50% in C57BL/6 mice and baboons vaccinated with the immunogen emulsified in complete Freund’s adjuvant, alum, or CpG [4,161,162]. Numerous protection trials were subsequently performed in outbred and inbred mice using SG3PDH, in a recombinant or peptidic form. Peptides with lowest homology to the human counterpart synthesized as linear, monopeptidic or dipeptidic multiple antigen peptide (MAP) constructs, or full length recombinant SG3PDH were used in conjunction with complete (CFA) or incomplete (IFA) Freund’s adjuvant, alum, or TiterMax adjuvant, leading to protection levels between 10 and 35% [163–166 and references therein].
The failure with SG3PDH was ascribed to the fact that the potential candidate vaccine antigens must be located at the host-parasite interface to be accessible to antibody-dependent cell-mediated cytotoxicity (ADCC) the once thought plausible mechanism of schistosome elimination. We have used as immunogen the S. mansoni glucose transporter, SGTP4, cloned and sequenced by Patrick Skelly and Charles Schoemaker [167]. We have circumvented the poor immunogenicity and strong hydrophobicity of the molecule that possesses 12 transmembrane domains, by using the large extracellular loop in a recombinant form, and the other extracellular stretches as synthetic linear peptides. Extensive studies in C57BL/6, BALB/c, and outbred CD1 mice immunized with SGTP4 in a recombinant or peptidic form in conjunction with CFA and IFA resulted into lack of protection against challenge S. mansoni infection, despite considerable specific humoral and cellular immune responses [168]. More recently, the S. mansoni integral surface membrane proteins tetraspanins (TSP)-1 and TSP-2 encoding cDNA were cloned, sequenced, and expressed and shown to elicit protection levels of 57% and 64% (TSP-2) and 34% and 52% (TSP-1) for mean adult worm burdens and liver egg burdens, respectively, over two independent trials in CBA/CaH mice [169]. The protection levels were not reproduced using S. japonicum counterpart, whereby mice immunized with the recombinant protein of a single TSP-2 subclass showed no protection, while immunization with a mixture of seven recombinant TSP-2 subclasses provided a moderate protection [170]. The failure in inducing consistent protection was ascribed to polymorphism in the extracellular site used for vaccination [170–172]. A recent study used S. mansoni TSP-2 extracellular loop 2 region in conjunction with alum and CpG as adjuvants and reported extremely variable protection levels against challenge infection within cohorts of highly inbred C57BL/6 mice [173].
Novel prevention approaches
Up to today, there is no available vaccine for schistosomiasis. One major reason for this failure resides in selecting unsuitable vaccine candidates. Thus, it is not clear how the host immune effector elements would eliminate parasites via interacting with internal and cytosolic proteins such as paramyosin, fatty acid binding protein, GST, TPI, or SG3PDH [164,174]. Additionally, surface membrane antigens are hidden in the developing larvae and adult worms and, hence, are entirely inaccessible to the host immune system unless the parasite is suffering excessive loss of tegument integrity and poised to die [106–110,175–177]. Conversely, viable lung schistosomula excretory–secretory products (ESP) are ideal potential vaccines. ESP can readily play a central role in induction of local primary and memory immune responses, and are available targets to the effector immune elements, mediating generation of immune effectors, toxic radicals, and inflammatory cytokines and cells that would directly target, surround, and pursue the larvae while painstakingly negotiating the tight lung capillaries [178].
Accordingly, schistosomula were separated from BALB/c lung 6 days after infection with cercariae of S. mansoni, isolated free of host erythrocytes and lung cells, and cultured for 48 h in protein-free medium under conditions that entirely preserved the viability of larvae and any remaining cells. The released ESP proteins were minute in quantity, corroborating the results obtained with in vitro-grown lung larvae [179,180], yet were highly immunogenic in BALB/c mice, inducing production of specific antibodies, and expression of mRNA for interleukin (IL)-12p40, interferon-gamma (IFN-γ) and tumor necrosis factor-alpha (TNF-α) [178]. Proteomic analysis of the ESP molecules of 25, 28 and 40 kDa revealed sequences belonging to innumerable rodent host-derived and a few S. mansoni-specific proteins. S. mansoni fructose 1,6 biphosphate aldolase (aldolase) and calpain were previously identified in products released from in vitro-cultured S. mansoni lung schistosomula [179]. Additionally, ex vivo lung stage schistosomula were found to release TPI, GST, SG3PDH, and 2-cys-peroxyredoxin (thioredoxin peroxidase, TPX) together with cytosolic molecules such as 14-3-3 protein homologue 1, actin and histone. It is of note that calpain, aldolase, TPI, GST, SG3PDH, TPX, and 14-3-3 protein homolog1 were detected among soluble proteins of in vitro-grown 8 day S. mansoni schistosomula [181], and secretions of S. mansoni cercariae [182,183]. The status of many of these molecules as vaccine candidates was made understood, as they are readily excreted-secreted by intact larvae and are, hence, entirely accessible to the host immune system effector elements [178].
In the vasculature, larval ESP and endothelial cells-associated co-stimulatory molecules [184] induce T helper (Th) lymphocytes to release cytokines, which activate monocytes, basophils, eosinophils, and neutrophils to target and eliminate the migrating larvae. ESP interaction with specific antibodies may also trigger myeloid cells towards participating in the killing of migrating larvae. It is documented that larval antigens induce Th1 immune responses [180,185,186]. Additionally, MAP construct containing S. mansoni TPI-derived T- and B-cell epitopes was able to trigger naïve donor immune responses towards a type-1 cytokine profile, consisting mainly of IFN-γ [187]. We have repeatedly observed that 14-3-3-like antigen and TPX stimulate strong IFN-γ and interleukin (IL)-17-driven responses in SC obtained from naïve mice and mice 7 days after S. mansoni infection, while rSG3PDH and TPX-derived peptide induced slightly lower levels of these cytokines and a small amount of IL-4 [188].
Th1 inflammatory cytokines such as IFN-γ and TNF-α trigger monocytes and endothelial cells to release the potentially larvacidal superoxide anions and oxygen and nitrogen radicals, and the pro-inflammatory cytokines, IL-1, IL-6, and IL-23 [189,190]. Monocytes activation generates high levels of hydrogen peroxide and nitric oxide, which might be lethal to many parasites [191,192], but may induce others to tighten their protective outer double lipid membrane, via inactivation of tegument-associated neutral sphingomyelinase [110]. Additionally, activated neutrophils recruited to the site by ESP- induced IL-17 [188,193] can release a range of toxic species and web-like structures termed neutrophil extracellular traps that entrap the larvae within the vasculature [189,190,194]. Eosinophils and basophils, activated via binding ESP-antibody complexes or type 2 cytokines, may release larvacidal molecules and pro-inflammatory mediators in the vicinity of the migrating larvae, and produce factors that alter the blood capillary permeability via disrupting endothelial integrity [194]. Larval ESP, however, are known to induce Th1 and Th17 cytokine-driven responses, thus hindering the recruitment, and activation of eosinophils and basophils.
To engage eosinophils and basophils in the hunt of migrating larvae, outbred, akin to humans, mice were immunized with 10 μg recombinant SG3PDH and 15 μg TPX-derived peptide in conjunction with 10 μg of the cysteine protease papain, or 200 ng of the type 2 cytokines, thymic stromal lymphopoietin, IL-25, or IL-33 as adjuvant [195–200]. Two weeks later, untreated, adjuvant controls, and immunized mice were challenged with 100 or 125 cercariae. Results of 8 separate experiments indicated that these formulations elicited IgM, IgG1, and IgA specific antibodies, and increase in ex vivo SC release of IL-4 and IL-5, correlated with highly significant (up to P < 0.0001) reduction of 62–78% in challenge worm burden [200].
Improvement of ESP selection, singly or in a combination, and immunization regimen, namely ESP and type-2 cytokine dose and injection site and schedule, will likely lead to a sterilizing schistosomiasis vaccine in a foreseeable future.
Acknowledgments
Experiments related to novel approaches to therapy and vaccinations were supported, in part, by the Science and Technology Development Fund, Egypt, Grants No. 144 and 2073 to R. El Ridi.
Footnotes
Peer review under responsibility of Cairo University.

References
- 1.Steinmann P., Keiser J., Bos R., Tanner M., Utzinger J. Schistosomiasis and water resources development: systematic review, meta-analysis, and estimates of people at risk. Lancet Infect Dis. 2006;6:411–425. doi: 10.1016/S1473-3099(06)70521-7. [DOI] [PubMed] [Google Scholar]
- 2.King C.H. Parasites and poverty: the case of schistosomiasis. Acta Trop. 2010;113:95–104. doi: 10.1016/j.actatropica.2009.11.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Gray D.J., McManus D.P., Li Y., Williams G.M., Bergquist R., Ross A.G. Schistosomiasis elimination: lessons from the past guide the future. Lancet Infect Dis. 2010;10:733–736. doi: 10.1016/S1473-3099(10)70099-2. [DOI] [PubMed] [Google Scholar]
- 4.Siddiqui A.A., Siddiqui B.A., Ganley-Leal L. Schistosomiasis vaccines. Hum Vaccin. 2011;7:1192–1197. doi: 10.4161/hv.7.11.17017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Utzinger J., Raso G., Brooker S., De Savigny D., Tanner M., Ornbjerg N. Schistosomiasis and neglected tropical diseases: towards integrated and sustainable control and a word of caution. Parasitology. 2009;136:1859–1874. doi: 10.1017/S0031182009991600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hotez P.J. The neglected tropical diseases and their devastating health and economic impact on the member nations of the organisation of the Islamic conference. PLoS Negl Trop Dis. 2009;3:e539. doi: 10.1371/journal.pntd.0000539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hotez PJ, Savioli L, Fenwick A. Neglected tropical diseases of the middle east and north Africa: review of their prevalence, distribution, and opportunities for control. PLoS Negl Trop Dis 201;6:e1475. [DOI] [PMC free article] [PubMed]
- 8.Curwen R.S., Wilson R.A. Invasion of skin by schistosome cercariae: some neglected facts. Trends Parasitol. 2003;19:63–66. doi: 10.1016/s1471-4922(02)00019-3. [DOI] [PubMed] [Google Scholar]
- 9.He Y.X., Salafsky B., Ramaswamy K. Comparison of skin invasion among three major species of Schistosoma. Trends Parasitol. 2005;21:201–203. doi: 10.1016/j.pt.2005.03.003. [DOI] [PubMed] [Google Scholar]
- 10.Rheinberg C.E., Moné H., Caffrey C.R., Imbert-Establet D., Jourdane J., Ruppel A. Schistosoma haematobium, S. intercalatum, S. japonicum, S. mansoni, S. rodhaini in mice: relationship between patterns of lung migration by schistosomula and perfusion recovery of adult worms. Parasitol Res. 1998;84:338–342. doi: 10.1007/s004360050407. [DOI] [PubMed] [Google Scholar]
- 11.Sturrock RF. The parasites and their life cycle. In: Jordan P, Webbe G, Sturrock RF, editors. Human schistosomiasis. Wallingford (UK): CAB International; 1993. p. 1–32.
- 12.Farid Z. Schistosomes with terminal-spined eggs: pathological and clinical aspects. In: Jordan P, Webbe G, Sturrock RF, editors. Human schistosomiasis. Wallingford (UK): CAB International; 1993. p. 159–93.
- 13.Andersson K.L., Chung R.T. Hepatic schistosomiasis. Curr Treat Options Gastroenterol. 2007;10:504–512. doi: 10.1007/s11938-007-0050-y. [DOI] [PubMed] [Google Scholar]
- 14.Burke M.L., Jones M.K., Gobert G.N., Li Y.S., Ellis M.K., McManus D.P. Immunopatho-genesis of human schistosomiasis. Parasite Immunol. 2009;3:163–176. doi: 10.1111/j.1365-3024.2009.01098.x. [DOI] [PubMed] [Google Scholar]
- 15.Mbabazi P.S., Andan O., Fitzgerald D.W., Chitsulo L., Engels D., Downs J.A. Examining the relationship between urogenital schistosomiasis and HIV infection. PLoS Negl Trop Dis. 2011;5:e1396. doi: 10.1371/journal.pntd.0001396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Secor W.E. The effects of schistosomiasis on HIV/AIDS infection, progression and transmission. Curr Opin HIV AIDS. 2012;7:254–259. doi: 10.1097/COH.0b013e328351b9e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Strickland G.T. Liver disease in Egypt: hepatitis C superseded schistosomiasis as a result of iatrogenic and biological factors. Hepatology. 2006;43:915–922. doi: 10.1002/hep.21173. [DOI] [PubMed] [Google Scholar]
- 18.El-Zayadi A.R., Hepatitis C. Comorbidities affecting the course and response to therapy. World J Gastroenterol. 2009;15:4993–4999. doi: 10.3748/wjg.15.4993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Abruzzi A., Fried B. Coinfection of Schistosoma (Trematoda) with bacteria, protozoa and helminths. Adv Parasitol. 2011;77:1–85. doi: 10.1016/B978-0-12-391429-3.00005-8. [DOI] [PubMed] [Google Scholar]
- 20.Bueding E., Fisher J. Factors affecting the inhibition of phosphofructokinase activity of Schistosoma mansoni by trivalent organic antimonials. Biochem Pharmacol. 1966;115:1197–1211. doi: 10.1016/0006-2952(66)90285-1. [DOI] [PubMed] [Google Scholar]
- 21.Denham D.A., Holdsworth R.J. The effect of metrifonate in vitro on Schistosoma haematobium and Schistosoma mansoni adults. Trans Roy Soc Trop Med Hyg. 1971;65:969. doi: 10.1016/0035-9203(71)90070-8. [DOI] [PubMed] [Google Scholar]
- 22.Holmstedt B., Nordgren I., Sandoz M., Sundwall A. Metrifonate. Summary of toxicological and pharmacological information available. Arch Toxicol. 1978;41:3–29. doi: 10.1007/BF00351766. [DOI] [PubMed] [Google Scholar]
- 23.Forsyth D.M. Treatment of urinary schistosomiasis: practice and theory. Lancet. 1965;2:354–358. doi: 10.1016/s0140-6736(65)90338-7. [DOI] [PubMed] [Google Scholar]
- 24.Bergquist R. Strategies for control of infection and disease: current practice and future potential. In: Mahmoud A.A.F., editor. Schistosomiasis. Imperial College Press; London: 2001. pp. 413–467. [Google Scholar]
- 25.Foster R., Cheetham B.L. Studies with the schistosomicide oxamniquine (UK-4271). II. Activity in primates. Trans Roy Soc Trop Med Hyg. 1973;67:685–693. doi: 10.1016/0035-9203(73)90039-4. [DOI] [PubMed] [Google Scholar]
- 26.Foster R. A review of clinical experience with oxamniquine. Trans Roy Soc Trop Med Hyg. 1987;81:55–59. doi: 10.1016/0035-9203(87)90282-3. [DOI] [PubMed] [Google Scholar]
- 27.Pica-Mattoccia L., Cioli D. Studies on the mode of action of oxamniquine and related schistosomicidal drugs. Am J Trop Med Hyg. 1985;34:112–118. doi: 10.4269/ajtmh.1985.34.112. [DOI] [PubMed] [Google Scholar]
- 28.Cioli D., Pica-Mattoccia L., Archer S. Antischistosomal drugs: past, present … and future? Pharmac Ther. 1995;68:35–85. doi: 10.1016/0163-7258(95)00026-7. [DOI] [PubMed] [Google Scholar]
- 29.Davis A. Antischistosomal drugs and clinical practice. In: Jordan P, Webbe G, Sturrock RF, editors. Human schistosomiasis. Wallingford: CAB International; 1993. p. 367–404.
- 30.Kumar V., Gryseels B. Use of praziquantel against schistosomiasis: a review of current status. Int J Antimicrob Agents. 1994;4:313–320. doi: 10.1016/0924-8579(94)90032-9. [DOI] [PubMed] [Google Scholar]
- 31.Xiao S.H., Catto B.A., Webster L.T. Effects of praziquantel on different developmental stages of Schistosoma mansoni in vitro and in vivo. J Infect Dis. 1985;151:1130–1137. doi: 10.1093/infdis/151.6.1130. [DOI] [PubMed] [Google Scholar]
- 32.Mehlhorn H., Becker B., Andrews P., Thomas H., Frenkel J.K. In vivo and in vitro experiments on the effects of praziquantel on Schistosoma mansoni. A light and electron microscopic study. Arzneim Forsch/Drug Res. 1981;31:544–554. [PubMed] [Google Scholar]
- 33.Andrews P. Praziquantel: mechanisms of anti-schistosomal activity. Pharmac Ther. 1985;29:129–156. doi: 10.1016/0163-7258(85)90020-8. [DOI] [PubMed] [Google Scholar]
- 34.Day T.A., Bennett J.L., Pax R.A. Praziquantel: the enigmatic antiparasitic. Parasitol Today. 1992;8:342–344. doi: 10.1016/0169-4758(92)90070-i. [DOI] [PubMed] [Google Scholar]
- 35.Kohn A.B., Anderson P.A.V., Roberts-Misterly J., Greenberg R.M. Schistosome calcium channel β subunits unusual modulatory effects and potential role in the action of the antischistosomal drug praziquantel. J Biol Chem. 2001;276:36873–36876. doi: 10.1074/jbc.C100273200. [DOI] [PubMed] [Google Scholar]
- 36.Greenberg R.M. Ca2+ signalling, voltage-gated Ca2+ channels and praziquantel in flatworm neuromusculature. Parasitology. 2005;131(Suppl):S97–S108. doi: 10.1017/S0031182005008346. [DOI] [PubMed] [Google Scholar]
- 37.Jeziorski M.C., Greenberg R.M. Voltage-gated calcium channel subunits from platyhelminths: potential role in praziquantel action. Int J Parasitol. 2006;31(36):625–632. doi: 10.1016/j.ijpara.2006.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Salvador-Recatalà V., Greenberg R.M. Calcium channels of schistosomes: unresolved questions and unexpected answers. Wiley Interdiscip Rev Membr Transp Signal. 2012;1:85–93. doi: 10.1002/wmts.19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Doenhoff M.J., Cioli D., Utzinger J. Praziquantel: mechanisms of action, resistance and new derivatives for schistosomiasis. Curr Opin Infect Dis. 2008;21:659–667. doi: 10.1097/QCO.0b013e328318978f. [DOI] [PubMed] [Google Scholar]
- 40.Harder A., Goossens J., Andrews P. Influence of praziquantel and Ca2+ on the bilayer-isotropic-hexagonal transition of model membranes. Mol Biochem Parasitol. 1988;29:55–60. doi: 10.1016/0166-6851(88)90119-3. [DOI] [PubMed] [Google Scholar]
- 41.Schepers H., Brasseur R., Goormaghtigh E., Duquenoy P., Ruysschaert J.M. Mode of insertion of praziquantel and derivatives into lipid membranes. Biochem Pharmacol. 1988;37:1615–1623. doi: 10.1016/0006-2952(88)90026-3. [DOI] [PubMed] [Google Scholar]
- 42.Lima S.F., Vieira L.Q., Harder A., Kusel J.R. Altered behaviour of carbohydrate-bound molecules and lipids in areas of the tegument of adult Schistosoma mansoni worms damaged by praziquantel. Parasitology. 1994;100:469–477. doi: 10.1017/s0031182000080720. [DOI] [PubMed] [Google Scholar]
- 43.Fenwick A., Savioli L., Engels D., Bergquist N.R., Todd M.H. Drugs for the control of parasitic diseases: current status and development in schistosomiasis. Trends Parasitol. 2003;19:509–515. doi: 10.1016/j.pt.2003.09.005. [DOI] [PubMed] [Google Scholar]
- 44.Doenhoff M.J., Hagan P., Cioli D., Southgate V., Pica-Mattoccia L., Botros S. Praziquantel: its use in control of schistosomiasis in sub-Saharan Africa and current research needs. Parasitology. 2009;136:1825–1835. doi: 10.1017/S0031182009000493. [DOI] [PubMed] [Google Scholar]
- 45.Stothard J.R., Sousa-Figueiredo J.C., Betson M., Green H.K., Seto E.Y., Garba A. Closing the praziquantel treatment gap: new steps in epidemiological monitoring and control of schistosomiasis in African infants and preschool-aged children. Parasitology. 2011;138:1593–1606. doi: 10.1017/S0031182011001235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Oshish A., AlKohlani A., Hamed A., Kamel N., AlSoofi A., Farouk H. Towards nationwide control of schistosomiasis in Yemen: a pilot project to expand treatment to the whole community. Trans Roy Soc Trop Med Hyg. 2011;105:617–627. doi: 10.1016/j.trstmh.2011.07.013. [DOI] [PubMed] [Google Scholar]
- 47.Leslie J., Garba A., Oliva E.B., Barkire A., Tinni A.A., Djibo A. Schistosomiais and soil-transmitted helminth control in Niger: cost effectiveness of school based and community distributed mass drug administration. PLoS Negl Trop Dis. 2011;5:e1326. doi: 10.1371/journal.pntd.0001326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Gryseels B., Mbaye A., De Vlas S.J., Stelma F.F., Guisse F., Van Lieshout L. Are poor responses to praziquantel for the treatment of Schistosoma mansoni infections in Senegal due to resistance? An overview of the evidence. Trop Med Intl Hlth. 2001;6:864–873. doi: 10.1046/j.1365-3156.2001.00811.x. [DOI] [PubMed] [Google Scholar]
- 49.Raso G., N’Goran E.K., Toty A., Luginbuhl A., Adjoua C.A., Tian-Bi N.T. Efficacy and side effects of praziquantel against Schistosoma mansoni in a community of western Cote d’Ivoire. Trans Roy Soc Trop Med Hyg. 2004;98:18–27. doi: 10.1016/s0035-9203(03)00003-8. [DOI] [PubMed] [Google Scholar]
- 50.Barakat R., El Morshedy H. Efficacy of two praziquantel treatments among primary school children in an area of high Schistosoma mansoni endemicity, Nile Delta, Egypt. Parasitology. 2011;138:440–446. doi: 10.1017/S003118201000154X. [DOI] [PubMed] [Google Scholar]
- 51.Fallon P.G., Tao L.F., Ismail M.M., Bennett J.L. Schistosome resistance to praziquantel: fact or artifact? Parasitol Today. 1996;12:316–320. doi: 10.1016/0169-4758(96)10029-6. [DOI] [PubMed] [Google Scholar]
- 52.Bennett J.L., Day T., Feng-Tao L., Ismail M., Farghaly A. The development of resistance to anthelmintics: a perspective with an emphasis on the antischistosomal drug praziquantel. Exp Parasitol. 1997;87:260–267. doi: 10.1006/expr.1997.4229. [DOI] [PubMed] [Google Scholar]
- 53.Kusel J., Hagan P. Praziquantel – its use, cost and possible development of resistance. Parasitol Today. 1999;15:352–354. doi: 10.1016/s0169-4758(99)01510-0. [DOI] [PubMed] [Google Scholar]
- 54.Ismail M., Botros S., Metwally A., William S., Farghally A., Tao L.F. Resistance to praziquantel: direct evidence from Schistosoma mansoni isolated from Egyptian villagers. Am J Trop Med Hyg. 1999;60:932–935. doi: 10.4269/ajtmh.1999.60.932. [DOI] [PubMed] [Google Scholar]
- 55.Sabra A.N., Botros S.S. Response of Schistosoma mansoni isolates having different drug sensitivity to praziquantel over several life cycle passages with and without therapeutic pressure. J Parasitol. 2008;94:537–541. doi: 10.1645/GE-1297.1. [DOI] [PubMed] [Google Scholar]
- 56.Linder E., Thors C. Schistosoma mansoni: praziquantel-induced tegumental lesion exposes actin of surface spines and allows binding of actin depolymerizing factor, gelsolin. Parasitology. 1992;105:71–79. doi: 10.1017/s0031182000073704. [DOI] [PubMed] [Google Scholar]
- 57.Tallima H., El Ridi R. Praziquantel binds Schistosoma mansoni adult worm actin. Int J Antimicrob Agents. 2007;29:570–575. doi: 10.1016/j.ijantimicag.2006.12.018. [DOI] [PubMed] [Google Scholar]
- 58.Tallima H., El Ridi R. Re: is actin the praziquantel receptor? Int J Antimicrob Agents. 2007;30:566–567. doi: 10.1016/j.ijantimicag.2007.08.002. [DOI] [PubMed] [Google Scholar]
- 59.Pica-Mattoccia L., Valle C., Basso A., Troiani A.R., Vigorosi F., Liberti P. Cytochalasin D abolishes the schistosomicidal activity of praziquantel. Exp Parasitol. 2007;115:344–351. doi: 10.1016/j.exppara.2006.09.017. [DOI] [PubMed] [Google Scholar]
- 60.Gnanasekar M., Salunkhe A.M., Mallia A.K., He Y.X., Kalyanasundaram R. Praziquantel affects the regulatory myosin light chain of Schistosoma mansoni. Antimicrob Agents Chemother. 2009;53:1054–1060. doi: 10.1128/AAC.01222-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Richard-Lenoble D., Chandenier J., Duong T.H. Antiparasitic treatments in pregnant women and in children in 2003. Med Trop (Mars) 2003;63:491–497. [PubMed] [Google Scholar]
- 62.Liu H., William S., Herdtweck E., Botros S., Dömling A. MCR synthesis of praziquantel derivatives. Chem Biol Drug Des. 2012;79:470–477. doi: 10.1111/j.1747-0285.2011.01288.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Menezes C.M., Rivera G., Alves M.A., Do Amaral D.N., Thibaut J.P., Noël F. Synthesis, biological evaluation and structure-activity relationship of clonazepam, meclonazepam and 1,4-benzodiazepine compounds with schistosomicidal activity. Chem Biol Drug Des. 2012;79:943–949. doi: 10.1111/j.1747-0285.2012.01354.x. [DOI] [PubMed] [Google Scholar]
- 64.N’Goran E.K., Utzinger J., Gnaka H.N., Yapi A., N’Guessan N.A., Kigbafori S.D. Randomized, double-blind, placebo-controlled trial of oral artemether for the prevention of patent Schistosoma haematobium infections. Am J Trop Med Hyg. 2003;68:24–32. [PubMed] [Google Scholar]
- 65.Utzinger J., Chollet J., You J., Mei J., Tanner M., Xiao S. Effect of combined treatment with praziquantel and artemether on Schistosoma japonicum and Schistosoma mansoni in experimentally infected animals. Acta Trop. 2001;80:9–18. doi: 10.1016/s0001-706x(01)00138-3. [DOI] [PubMed] [Google Scholar]
- 66.Utzinger J., Keiser J., Shuhua X., Tanner M., Singer B.H. Combination chemotherapy of schistosomiasis in laboratory studies and clinical trials. Antimicrob Agents Chemother. 2003;47:1487–1495. doi: 10.1128/AAC.47.5.1487-1495.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Mahmoud M.R., Botros S.S. Artemether as adjuvant therapy to praziquantel in murine Egyptian Schistosomiasis mansoni. J Parasitol. 2005;91:175–178. doi: 10.1645/GE-322R. [DOI] [PubMed] [Google Scholar]
- 68.Botros S.S., Hammam O., Mahmoud M., Bergquist R. Praziquantel efficacy in mice infected with PZQ non-susceptible S. mansoni isolate treated with artemether: parasitological, biochemical and immunohistochemical assessment. APMIS. 2010;118:692–702. doi: 10.1111/j.1600-0463.2010.02645.x. [DOI] [PubMed] [Google Scholar]
- 69.El-Lakkany N.M., Hammam O.A., El-Maadawy W.H., Badawy A.A., Ain-Shoka A.A., Ebeid F.A. Anti-inflammatory/anti-fibrotic effects of the hepatoprotective silymarin and the schistosomicide praziquantel against Schistosoma mansoni-induced liver fibrosis. Parasit Vectors. 2012;5:9. doi: 10.1186/1756-3305-5-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Chen B.L., Zhang G.Y., Wang S.P., Li Q., Xu M.H., Shen Y.M. The combined treatment of praziquantel with osteopontin immunoneutralization reduces liver damage in Schistosoma japonicum-infected mice. Parasitology. 2012;139:522–529. doi: 10.1017/S0031182011002241. [DOI] [PubMed] [Google Scholar]
- 71.Xiao S., Tanner M., N’Goran E.K., Utzinger J., Chollet J., Bergquist R. Recent investigations of artemether, a novel agent for the prevention of schistosomiasis japonica, mansoni and haematobia. Acta Trop. 2002;82:175–181. doi: 10.1016/s0001-706x(02)00009-8. [DOI] [PubMed] [Google Scholar]
- 72.Xiao S.H., Catto B.A. In vitro and in vivo studies of the effect of artemether on Schistosoma mansoni. Antimicrob Agents Chemother. 1989;33:1557–1562. doi: 10.1128/aac.33.9.1557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Xiao S., Utzinger J., Chollet J., Endriss Y., N’Goran E.K., Tanner M. Effect of artemether against Schistosoma haematobium in experimentally infected hamsters. Int J Parasitol. 2000;30:1001–1006. doi: 10.1016/s0020-7519(00)00091-6. [DOI] [PubMed] [Google Scholar]
- 74.Utzinger J., Xiao S.H., Tanner M., Keiser J. Artemisinins for schistosomiasis and beyond. Curr Opin Investig Drugs. 2007;8:105–116. [PubMed] [Google Scholar]
- 75.Abdul-Ghani R., Loutfi N., Sheta M., Hassan A. Artemether shows promising female schistosomicidal and ovicidal effects on the Egyptian strain of Schistosoma mansoni after maturity of infection. Parasitol Res. 2011;108:1199–1205. doi: 10.1007/s00436-010-2163-9. [DOI] [PubMed] [Google Scholar]
- 76.Keiser J., Chollet J., Xiao S.H., Mei J.Y., Jiao P.Y., Utzinger J. Mefloquine – an aminoalcohol with promising antischistosomal properties in mice. PLoS Negl Trop Dis. 2009;3:e350. doi: 10.1371/journal.pntd.0000350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Keiser J.M., Vargas M., Doenhoff M.J. Activity of artemether and mefloquine against juvenile and adult Schistosoma mansoni in athymic and immunocompetent NMRI mice. Am J Trop Med Hyg. 2010;82:112–114. doi: 10.4269/ajtmh.2010.09-0461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Xiao S.H., Keiser J., Chollet J., Utzinger J., Dong Y., Endriss Y. In vitro and in vivo activities of synthetic trioxolanes against major human schistosome species. Antimicrob Agents Chemother. 2007;51:1440–1445. doi: 10.1128/AAC.01537-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Botros S., William S., Hammam O., Zídek Z., Holý A. Activity of 9-(S)-[3-hydroxy-2- (phosphonomethoxy)propyl]adenine against Schistosomiasis mansoni in mice. Antimicrob Agents Chemother. 2003;47:3853–3858. doi: 10.1128/AAC.47.12.3853-3858.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Botros S.S., William S., Beadle J.R., Valiaeva N., Hostetler K.Y. Antischistosomal activity of hexadecyloxypropyl cyclic 9-(S)-[3-hydroxy-2-(phosphonomethoxy)propyl] adenine and other alkoxyalkyl esters of acyclic nucleoside phosphonates assessed by schistosome worm killing in vitro. Antimicrob Agents Chemother. 2009;53:5284–5287. doi: 10.1128/AAC.00840-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Sayed A.A., Simeonov A., Thomas C.J., Inglese J., Austin C.P., Williams D.L. Identification of oxadiazoles as new drug leads for the control of schistosomiasis. Nat Med. 2008;14:407–412. doi: 10.1038/nm1737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Prast-Nielsen S., Huang H.H., Williams D,L. Thioredoxin glutathione reductase: its role in redox biology and potential as a target for drugs against neglected diseases. Biochim Biophys Acta. 2011;1810:1262–1271. doi: 10.1016/j.bbagen.2011.06.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Portela J., Boissier J., Gourbal B., Pradines V., Collière V., Coslédan F. Antischistosomal activity of trioxaquines: in vivo efficacy and mechanism of action on Schistosoma mansoni. PLoS Negl Trop Dis. 2012;6(2):e1474. doi: 10.1371/journal.pntd.0001474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Neves J.K., de Lima Mdo C., Pereira V.R., de Melo C.M., Peixoto C.A., Pitta Ida R. Antischistosomal action of thioxo-imidazolidine compounds: an ultrastructural and cytotoxicity study. Exp Parasitol. 2011;128:82–90. doi: 10.1016/j.exppara.2011.01.021. [DOI] [PubMed] [Google Scholar]
- 85.Pierce RJ, Dubois-Abdesselem F, Caby S, Trolet J, Lancelot J, Oger F, et al. Chromatin regulation in schistosomes and histone modifying enzymes as drug targets. Mem Inst Oswaldo Cruz 201;106:794–801. [DOI] [PubMed]
- 86.Moraes J., Nascimento C., Lopes P.O., Nakano E., Yamaguchi L.F., Kato M.J. Schistosoma mansoni: in vitro schistosomicidal activity of piplartine. Exp Parasitol. 2011;127:357–364. doi: 10.1016/j.exppara.2010.08.021. [DOI] [PubMed] [Google Scholar]
- 87.de Melo NI, Magalhaes LG, de Carvalho CE, Wakabayashi KA, de P Aguiar G, Ramos RC, et al. Schistosomicidal activity of the essential oil of Ageratum conyzoides L. (Asteraceae) against adult Schistosoma mansoni worms. Molecules 2011;16:762–73. [DOI] [PMC free article] [PubMed]
- 88.Tagboto S., Townson S. Antiparasitic properties of medicinal plants and other naturally-occurring products. Adv Parasitol. 2001;50:199–295. doi: 10.1016/s0065-308x(01)50032-9. [DOI] [PubMed] [Google Scholar]
- 89.Yousif F, Hifnawy MS, Soliman G, Boulos L, Labib T, Mahmoud S, et al. Large-scale in vitro screening of Egyptian native and cultivated plants for schistosomicidal activity. Pharmaceut Biol 2007;45:501–10.
- 90.Allegretti SM, Nascimento C, de Oliveira F, de Oliveira RN, Frezza TF, Garcia Rehder VL. The use of Brazilian medicinal plants to combat Schistosoma mansoni. In: Rokni MB, editor. Schistosomiasis. InTechOpen; 2012. p. 27–70. <http://www.intechopen.com/books/schistosomiasis/the-use-of-brazilian-medicinal-plants-to-combat-schistosoma-mansoni> [chapter 3].
- 91.De Moraes J. Antischistosomal natural compounds: present challenges for new drug screens. In: Rodriguez-Morales A, editor. Current topics in tropical medicine. InTech Open; 2012. p. 323–57. <http://www.intechopen.com/books/current-topics-in-tropical-medicine/antischistosomal-natural-compounds-present-challenges-for-new-drug-screens> [chapter 20].
- 92.El-Ansary A.K., Ahmed S.A., Aly S.A. Antischistosomal and liver protective effects of Curcuma longa extract in Schistosoma mansoni infected mice. Indian J Exp Biol. 2007;45:791–801. [PubMed] [Google Scholar]
- 93.El-Banhawey M.A., Ashry M.A., El-Ansary A.K., Aly S.A. Effect of curcuma longa or parziquantel on Schistosoma mansoni infected mice liver-histological and histochemical study. Indian J Exp Biol. 2007;45:877–889. [PubMed] [Google Scholar]
- 94.Magalhães L.G., Machado C.B., Morais E.R., Moreira E.B., Soares CS., da Silva S.H. In vitro schistosomicidal activity of curcumin against Schistosoma mansoni adult worms. Parasitol Res. 2009;104:1197–1201. doi: 10.1007/s00436-008-1311-y. [DOI] [PubMed] [Google Scholar]
- 95.Allam G. Immunomodulatory effects of curcumin treatment on murine Schistosomiasis mansoni. Immunobiology. 2009;214:712–727. doi: 10.1016/j.imbio.2008.11.017. [DOI] [PubMed] [Google Scholar]
- 96.Luz P.P., Magalhães L.G., Pereira A.C., Cunha W.R., Rodrigues V., Andrade E. Curcumin-loaded into PLGA nanoparticles: preparation and in vitro schistosomicidal activity. Parasitol Res. 2012;110:593–598. doi: 10.1007/s00436-011-2527-9. [DOI] [PubMed] [Google Scholar]
- 97.Tonkal A.M., Morsy T.A. An update review on Commiphora molmol and related species. J Egypt Soc Parasitol. 2008;38:763–796. [PubMed] [Google Scholar]
- 98.Badria F., Abou-Mohamad G., El-Mowafi A., Masoud A., Salama O. Mirazid: a new schistosomicidal drug. Pharma Biol. 2001;39:127–131. [Google Scholar]
- 99.Botros S., William S., Ebeid F., Cioli D., Katz N., Day T.A. Lack of evidence for an antischistosomal activity of myrrh in experimental animals. Am J Trop Med Hyg. 2004;71:206–210. [PubMed] [Google Scholar]
- 100.Botros S., Sayed H., El-Dusoki H., Sabry H., Rabie I., El-Ghannam M. Efficacy of Mirazid in comparison with praziquantel in Egyptian Schistosoma mansoni-infected school children and households. Am J Trop Med Hyg. 2005;72:119–123. [PubMed] [Google Scholar]
- 101.Barakat R., Elmorshedy H., Fenwick A. Efficacy of myrrh in the treatment of human Schistosomiasis mansoni. Am J Trop Med Hyg. 2005;73:365–367. [PubMed] [Google Scholar]
- 102.Yakoot M. A short review of the anthelmintic role of Mirazid. Arq Gastroenterol. 2010;47:393–394. doi: 10.1590/s0004-28032010000400014. [DOI] [PubMed] [Google Scholar]
- 103.Osman M.M., El-Taweel H.A., Shehab A.Y., Farag H.F. Ineffectiveness of myrrh- derivative Mirazid against schistosomiasis and fascioliasis in humans. Eastern Med Health J. 2010;16:932–936. [PubMed] [Google Scholar]
- 104.Hockley D.J. Ultrastructure of the tegument of Schistosoma. Adv Parasitol. 1973;11:233–305. doi: 10.1016/s0065-308x(08)60188-8. [DOI] [PubMed] [Google Scholar]
- 105.Singer S.J., Nicolson G.L. The fluid mosaic model of the structure of cell membranes. Science. 1972;175:720–731. doi: 10.1126/science.175.4023.720. [DOI] [PubMed] [Google Scholar]
- 106.El Ridi R., Mohamed S.H., Tallima H. Incubation of Schistosoma mansoni lung-stage schistosomula in corn oil exposes their surface membrane antigenic specificities. J Parasitol. 2003;89:1064–1067. doi: 10.1645/GE-3122RN. [DOI] [PubMed] [Google Scholar]
- 107.El Ridi R., Tallima H., Mohamed S.H., Montash M. Depletion of Schistosoma mansoni lung-stage schistosomula cholesterol by methyl-β-cyclodextrin dramatically increases specific antibody binding to surface membrane antigens. J Parasitol. 2004;90:727–732. doi: 10.1645/GE-3334. [DOI] [PubMed] [Google Scholar]
- 108.Tallima H., El Ridi R. Methyl-β-cyclodextrin treatment and filipin staining reveal therole of cholesterol in surface membrane antigen sequestration of Schistosoma mansoni and S. haematobium lung-stage larvae. J Parasitol. 2005;91:720–725. doi: 10.1645/GE-439R. [DOI] [PubMed] [Google Scholar]
- 109.Tallima H., Salah M., El Ridi R. In vitro and in vivo effects of unsaturated fatty acids on Schistosoma mansoni and S. haematobium lung-stage larvae. J Parasitol. 2005;91:1094–1102. doi: 10.1645/GE-514R.1. [DOI] [PubMed] [Google Scholar]
- 110.El Ridi R., Tallima H. Equilibrium in lung schistosomula sphingomyelin breakdown and biosynthesis allows very small molecules, but not antibody, to access proteins at the host-parasite interface. J Parasitol. 2006;92:730–737. doi: 10.1645/GE-745R1.1. [DOI] [PubMed] [Google Scholar]
- 111.Huwiler A., Kolter T., Pfeilschifter J., Sandhoff K. Review. Physiology and pathophysiology of sphingolipid metabolism and signaling. Biochim Biophys Acta. 2000;1485:63–99. doi: 10.1016/s1388-1981(00)00042-1. [DOI] [PubMed] [Google Scholar]
- 112.Luberto C., Hassler D.F., Signorelli P., Okamoto Y., Sawai H., Boros E. Inhibition of tumor necrosis factor-induced cell death in MCF7 by a novel inhibitor of neutral sphingomyelinase. J Biol Chem. 2002;277:41128–41139. doi: 10.1074/jbc.M206747200. [DOI] [PubMed] [Google Scholar]
- 113.Hofmann K., Tomiuk S., Wolff G., Stoffel W. Cloning and characterization of the mammalian brain-specific, Mg2+-dependent neutral sphingomyelinase. Proc Natl Acad Sci USA. 2000;97:5895–5900. doi: 10.1073/pnas.97.11.5895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Robinson B.S., Hii C.S., Poulos A., Ferrante A. Activation of neutral sphingomyelinase in human neutrophils by polyunsaturated fatty acids. Immunology. 1997;91:274–280. doi: 10.1046/j.1365-2567.1997.d01-2227.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Tallima, H. Biochemical exposure of schistosoma surface membrane antigens. Ph.D thesis. Egypt: Faculty of Science, Cairo University; 2006.
- 116.Tallima H., Al-Halbosiy M.F., El Ridi R. Enzymatic activity and immunolocalization of Schistosoma mansoni and Schistosoma haematobium neutral sphingomyelinase. Mol Biochem Parasitol. 2011;178:23–28. doi: 10.1016/j.molbiopara.2011.04.003. [DOI] [PubMed] [Google Scholar]
- 117.Tallima H., Hamada M., El Ridi R. Evaluation of cholesterol content and impact on antigen exposure in the outer lipid bilayer of adult schistosomes. Parasitology. 1997;134:1775–1783. doi: 10.1017/S0031182007003174. [DOI] [PubMed] [Google Scholar]
- 118.Brash A.R. Arachidonic acid as a bioactive molecule. J Clin Invest. 2001;107:1339–1345. doi: 10.1172/JCI13210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Zhou L., Nilsson A. Sources of eicosanoid precursor fatty acid pools in tissues. J Lipid Res. 2001;42:1521–1542. [PubMed] [Google Scholar]
- 120.Nelson G.J., Kelley D.S., Emken E.A., Phinney S.D., Kyle D., Ferretti A. A human dietary arachidonic acid supplementation study conducted in a metabolic research unit: rationale and design. Lipids. 1997;32:415–420. doi: 10.1007/s11745-997-0054-8. [DOI] [PubMed] [Google Scholar]
- 121.Roberts MD, Iosia M, Kerksick CM, Taylor LW, Campbell B, Wilborn CD, et al. Effects of arachidonic acid supplementation on training adaptations in resistance-trained males. J Int Soc Sports Nutr 2007;28(4):21–34. [DOI] [PMC free article] [PubMed]
- 122.Simopoulos A. Evolutionary aspects of diet and essential fatty acids. World Rev Nutr Diet. 2001;88:18–27. doi: 10.1159/000059742. [DOI] [PubMed] [Google Scholar]
- 123.Simopoulos A. Evolutionary aspects of diet, essential fatty acids and cardiovascular disease. Eur Heart J Suppl 2001;3(Suppl D):D8–21.
- 124.Hempenius R.A., Van Delft J.M.H., Prinsen M., Lina B.A.R. Preliminary safety assessment of an arachidonic acid-enriched oil derived from Mortierella alpina: summary of toxicological data. Food Chem Toxicol. 1997;35:573–581. doi: 10.1016/s0278-6915(97)00025-2. [DOI] [PubMed] [Google Scholar]
- 125.El Ridi R., Aboueldahab M., Tallima H., Salah M., Mahana N., Fawzi S. In vitro and in vivo activities of arachidonic acid against Schistosoma mansoni and Schistosoma haematobium. Antimicrob Agents Chemother. 2010;4:3383–3389. doi: 10.1128/AAC.00173-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.El Ridi R., Tallima H., Salah M., Aboueldahab M., Fahmy O.M., Farhan Al-Halbosiy M. Arachidonic acid efficacy and mechanism of action in treatment of hamsters infected with Schistosoma mansoni or S. haematobium. Int J Antimicrobial Agents. 2012;39:232–239. doi: 10.1016/j.ijantimicag.2011.08.019. [DOI] [PubMed] [Google Scholar]
- 127.Dean D.A. A review. Schistosoma and related genera: Acquired resistance in mice. Exp Parasitol. 1983;55:1–104. doi: 10.1016/0014-4894(83)90002-4. [DOI] [PubMed] [Google Scholar]
- 128.Butterworth A.E., Capron M., Cordingley J.S., Dalton P.R., Dunne D.W., Kariuki H.C. Immunity after treatment of human schistosomiasis mansoni. II. Identification of resistant individuals, and analysis of their immune responses. Trans Roy Soc Trop Med Hyg. 1985;79:393–408. doi: 10.1016/0035-9203(85)90391-8. [DOI] [PubMed] [Google Scholar]
- 129.Hagan P., Blumenthal U.J., Chaudri M., Greenwood B.M., Hayes R.J., Hodgson I. Resistance to reinfection with Schistosoma haematobium in Gambian children: analysis of their immune responses. Trans Roy Soc Trop Med Hyg. 1987;81:938–946. doi: 10.1016/0035-9203(87)90359-2. [DOI] [PubMed] [Google Scholar]
- 130.Dessein A.J., Begley M., Demeure C., Caillol D., Fueri J., dos Reis M.G. Human resistance to Schistosoma mansoni is associated with IgG reactivity to a 37-kDa larval surface antigen. J Immunol. 1988;140:2727–2736. [PubMed] [Google Scholar]
- 131.Correa-Oliveira R., Pearce E.J., Oliveira G.C., Golgher D.B., Katz N., Bahia L.G. The human immune response to defined immunogens of Schistosoma mansoni: elevated antibody levels to paramyosin in stool-negative individuals from two endemic areas in Brazil. Trans Roy Soc Trop Med Hyg. 1989;83:798–804. doi: 10.1016/0035-9203(89)90334-9. [DOI] [PubMed] [Google Scholar]
- 132.Hagan P., Blumenthal U.J., Dunn D., Simpson A.J.G., Wilkins H.A. Human IgE, IgG4 and resistance to reinfection with Schistosoma haematobium. Nature. 1991;349:243–245. doi: 10.1038/349243a0. [DOI] [PubMed] [Google Scholar]
- 133.Viana I.R., Sher A., Carvalho O.S., Massara C.L., Eloi-Santos S.M., Pearce E.J. Interferon-gamma production by peripheral blood mononuclear cells from residents of an area endemic for Schistosoma mansoni. Trans Roy Soc Trop Med Hyg. 1994;88:466–470. doi: 10.1016/0035-9203(94)90436-7. [DOI] [PubMed] [Google Scholar]
- 134.El Ridi R., Farouk F., Sherif M., Al-Sherbiny M., Osman A., El Gengehi N. T and B cell reactivity to a 42-kDa protein is associated with human resistance to both Schistosomiasis mansoni and haematobium. J Infect Dis. 1998;177:1364–1372. doi: 10.1086/515274. [DOI] [PubMed] [Google Scholar]
- 135.El Ridi R, Shoemaker CB, Farouk F, El Sherif NH, Afifi A. Human T- and B-cell responses to Schistosoma mansoni recombinant glyceraldehyde 3-phosphate dehydrogenase correlate with resistance to reinfection with S. mansoni or Schistosoma haematobium after chemotherapy. Infect Immun 2001;69:237–44. [DOI] [PMC free article] [PubMed]
- 136.Soisson L.M., Masterson C.P., Tom T.D., McNally M.T., Lowell G.H., Strand M. Induction of protective immunity in mice using a 62-kDa recombinant fragment of a Schistosoma mansoni surface antigen. J Immunol. 1992;149:3612–3620. [PubMed] [Google Scholar]
- 137.Soisson L.A., Reid G.D.F., Farah I.O., Nyindo M., Strand M. Protective immunity in baboons vaccinated with a recombinant antigen or radiation-attenuated cercariae of Schistosoma mansoni is antibody-dependent. J Immunol. 1993;151:4782–4789. [PubMed] [Google Scholar]
- 138.Balloul J.M., Sondermeyer P., Dreyer D., Capron M., Grzych J.M., Pierce R.J. Molecular cloning of a protective antigen of schistosomes. Nature. 1987;326:149–153. doi: 10.1038/326149a0. [DOI] [PubMed] [Google Scholar]
- 139.Boulanger D., Reid G.D., Sturrock R.F., Wolowczuk I., Balloul J.M., Grezel D. Immunization of mice and baboons with the recombinant Sm28GST affects both worm viability and fecundity after experimental infection with Schistosoma mansoni. Parasite Immunol. 1991;13:473–490. doi: 10.1111/j.1365-3024.1991.tb00545.x. [DOI] [PubMed] [Google Scholar]
- 140.Harn D.A., Mitsuyama M., Huguenel E.D., Oligino L., David J.R. Identification by monoclonal antibodies of a major (28 kDa) surface membrane antigen of Schistosoma mansoni. Mol Biochem Parasitol. 1985;16:345–354. doi: 10.1016/0166-6851(85)90075-1. [DOI] [PubMed] [Google Scholar]
- 141.Harn D.A., Wei G., Oligino L.D., Mitsuyama M., Gebremichael A., Richter D. A protective monoclonal antibody specifically recognizes and alters the catalytic activity of schistosome triose-phosphate isomerase. J Immunol. 1992;148:562–567. [PubMed] [Google Scholar]
- 142.Shoemaker C., Gross A., Gebremichael A., Harn D. CDNA cloning and functional expression of the Schistosoma mansoni protective antigen triose-phosphate isomerase. Proc Natl Acad Sci USA. 1992;89:1842–1846. doi: 10.1073/pnas.89.5.1842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Lanar D.E., Pearce E.J., James S.L., Sher A. Identification of paramyosin as schistosome antigen recognized by intradermally vaccinated mice. Science. 1986;234:593–596. doi: 10.1126/science.3094144. [DOI] [PubMed] [Google Scholar]
- 144.Fonseca C.T., Cunha-Neto E., Goldberg A.C., Kalil J., de Jesus A.R., Carvalho E.M. Identification of paramyosin T cell epitopes associated with human resistance to Schistosoma mansoni reinfection. Clin Exp Immunol. 2005;142:539–547. doi: 10.1111/j.1365-2249.2005.02941.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Moser D., Tendler M., Griffiths G., Klinkert M.Q. A 14-kDa Schistosoma mansoni polypeptide is homologous to a gene family of fatty acid binding proteins. J Biol Chem. 1991;266:8447–8454. [PubMed] [Google Scholar]
- 146.Harn D.A., Mitsuyama M., Huguenel E.D., David J.R. Schistosoma mansoni: detection by monoclonal antibody of a 22,000-dalton surface membrane antigen which may be blocked by host molecules on lung stage parasites. J Immunol. 1985;135:2115–2120. [PubMed] [Google Scholar]
- 147.Reynolds S.R., Shoemaker C.B., Harn D.A. T and B cell epitope mapping of Sm23, an integral membrane protein of Schistosoma mansoni. J Immunol. 1992;149:3995–4001. [PubMed] [Google Scholar]
- 148.Koster B., Hall M.R.T., Strand M. Schistosoma mansoni: immuno-reactivity of human sera with the surface antigen Sm23. Exp Parasitol. 1993;77:282–294. doi: 10.1006/expr.1993.1086. [DOI] [PubMed] [Google Scholar]
- 149.Karcz S.R., Podesta R.B., Siddiqui A.A., Dekaban G.A., Strejan G.H., Clarke M.W. Molecular cloning and sequence analysis of a calcium-activated neutral protease (calpain) from Schistosoma mansoni. Mol Biochem Parasitol. 1991;49:333–336. doi: 10.1016/0166-6851(91)90078-k. [DOI] [PubMed] [Google Scholar]
- 150.Goudot-Crozel V., Caillol D., Djabali M., Dessein A.J. The major parasite surface antigen associated with human resistance to schistosomiasis is a 37-kD glyceraldehyde-3P-dehydrogenase. J Exp Med. 1989;170:2065–2080. doi: 10.1084/jem.170.6.2065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Charrier-Ferrara S., Caillol D., Goudot-Crozel V. Complete sequence of the Schistosoma mansoni glyceraldehyde-3-phosphate dehydrogenase gene encoding a major surface antigen. Mol Biochem Parasitol. 1992;56:339–343. doi: 10.1016/0166-6851(92)90184-l. [DOI] [PubMed] [Google Scholar]
- 152.El Ridi R., Abdel Tawab N., Guirguis N. Schistosoma mansoni: identification and protective immunity of adult worm antigens recognized by T lymphocytes of outbred Swiss mice immunized with irradiated cercariae. Exp Parasitol. 1993;76:265–277. doi: 10.1006/expr.1993.1032. [DOI] [PubMed] [Google Scholar]
- 153.Osman A., El Ridi R., Guirguis N., Dean D.A. Identification of Schistosoma mansoni antigens recognized by T cells of C57BL/6 mice immunized with gamma-irradiated cercariae. J Parasitol. 1994;80:421–431. [PubMed] [Google Scholar]
- 154.Osman A, El Ridi R, Guirguis N, Dean DA. Identification of Schistosoma mansoni antigens recognized by spleen cells of C57B1/6 mice immunized with ultraviolet-irradiated cercariae. Int J Parasitol 194;24:943–50. [DOI] [PubMed]
- 155.Abdel Tawab N. Identification and molecular characterization of protective antigens against murine Schistosomiasis mansoni. Ph.D thesis. Faculty of Science, Cairo University; 1994. p. 280.
- 156.El Gengehi N., El Ridi R., Tawab N.A., El Demellawy M., Mangold B.L. A Schistosoma mansoni 62-kDa band is identified as an irradiated vaccine T-cell antigen and characterized as calreticulin. J Parasitol. 2000;86:993–1000. doi: 10.1645/0022-3395(2000)086[0993:ASMKBI]2.0.CO;2. [DOI] [PubMed] [Google Scholar]
- 157.Al-Sherbiny M., Osman A., Barakat R., El Morshedy H., Bergquist R., Olds R. In vitro cellular and humoral responses to Schistosoma mansoni vaccine candidate antigens. Acta Trop. 2003;88:117–130. doi: 10.1016/s0001-706x(03)00195-5. [DOI] [PubMed] [Google Scholar]
- 158.Ribeiro de Jesus A., Araújo I., Bacellar O., Magalhães A., Pearce E., Harn D. Human immune responses to Schistosoma mansoni vaccine candidate antigens. Infect Immun. 2000;68:2797–2803. doi: 10.1128/iai.68.5.2797-2803.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Todd C.W., Colley D.G. Practical and ethical issues in the development of a vaccine against Schistosomiasis mansoni. Am J Trop Med Hyg. 2002;66:348–358. doi: 10.4269/ajtmh.2002.66.348. [DOI] [PubMed] [Google Scholar]
- 160.Tendler M., Simpson A.J. The biotechnology-value chain: development of Sm14 as a schistosomiasis vaccine. Acta Trop. 2008;108:263–266. doi: 10.1016/j.actatropica.2008.09.002. [DOI] [PubMed] [Google Scholar]
- 161.Siddiqui A.A., Ahmad G., Damian R.T., Kennedy R.C. Experimental vaccines in animal models for schistosomiasis. Parasitol Res. 2008;102:825–833. doi: 10.1007/s00436-008-0887-6. [DOI] [PubMed] [Google Scholar]
- 162.Ahmad G., Zhang W., Torben W., Ahrorov A., Damian R.T., Wolf R.F. Preclinical prophylactic efficacy testing of Sm-p80-based vaccine in a nonhuman primate model of Schistosoma mansoni infection and immunoglobulin G and E responses to Sm-p80 in human serum samples from an area where schistosomiasis is endemic. J Infect Dis. 2011;204:1437–1449. doi: 10.1093/infdis/jir545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.El Ridi R., Mahrous A., Afifi A., Montash M., Velek J., Jezek J. Human and murine humoral immune recognition of multiple peptides from Schistosoma mansoni glyceraldehyde 3-P dehydrogenase is associated with resistance to Schistosomiasis. Scand J Immunol. 2001;54:477–485. doi: 10.1046/j.1365-3083.2001.00992.x. [DOI] [PubMed] [Google Scholar]
- 164.Tallima H., Montash M., Veprek P., Velek J., Jezek J., El Ridi R. Differences in immunogenicity and vaccine potential of peptides from Schistosoma mansoni glyceraldehyde 3- phosphate dehydrogenase. Vaccine. 2003;21:3290–3300. doi: 10.1016/s0264-410x(03)00180-4. [DOI] [PubMed] [Google Scholar]
- 165.El Ridi R., Montash M., Tallima H. Immunogenicity and vaccine potential of di- peptidic multiple antigen peptides from Schistosoma mansoni glyceraldehyde 3-phosphate dehydrogenase. Scand J Immunol. 2004;60:392–402. doi: 10.1111/j.0300-9475.2004.01497.x. [DOI] [PubMed] [Google Scholar]
- 166.Veprek P., Jezek J., Velek J., Tallima H., Montash M., El Ridi R. Peptides and multiple antigen peptides from Schistosoma mansoni glyceraldehyde 3-phosphate dehydrogenase: preparation, immunogenicity and immunoprotective capacity in C57BL/6 mice. J Pept Sci. 2004;10:350–362. doi: 10.1002/psc.550. [DOI] [PubMed] [Google Scholar]
- 167.Skelly P.J., Tielens A.G.M., Shoemaker C.B. Glucose transport and metabolism in mammalian-stage schistosomes. Parasitol Today. 1998;14:402–406. doi: 10.1016/s0169-4758(98)01319-2. [DOI] [PubMed] [Google Scholar]
- 168.Mahana NA. Human and murine immune responses to the Schistosoma mansoni glucose transporter. Ph.D thesis. Faculty of Science, Cairo University. p. 240.
- 169.Tran M.H., Pearson M.S., Bethony J.M., Smyth D.J., Jones M.K., Duke M. Tetraspanins on the surface of Schistosoma mansoni are protective antigens against schistosomiasis. Nat Med. 2006;12:835–840. doi: 10.1038/nm1430. [DOI] [PubMed] [Google Scholar]
- 170.Cai P., Bu L., Wang J., Wang Z., Zhong X., Wang H. Molecular characterization of Schistosoma japonicum tegument protein tetraspanin-2: sequence variation and possible implications for immune evasion. Biochem Biophys Res Commun. 2008;372:197–202. doi: 10.1016/j.bbrc.2008.05.042. [DOI] [PubMed] [Google Scholar]
- 171.Zhang W., Li J., Duke M., Jones M.K., Kuang L., Zhang J. Inconsistent protective efficacy and marked polymorphism limits the value of Schistosoma japonicum tetraspanin-2 as a vaccine target. PLoS Negl Trop Dis. 2011;5:e1166. doi: 10.1371/journal.pntd.0001166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Cupit PM, Steinauer ML, Tonnessen BW, Eric Agola L, Kinuthia JM, Mwangi IN, et al. Polymorphism associated with the Schistosoma mansoni tetraspanin-2 gene. Int J Parasitol 2011; 41:1249–52. [DOI] [PMC free article] [PubMed]
- 173.Pearson M.S., Pickering D.A., McSorley H.J., Bethony J.M., Tribolet L., Dougall A.M. Enhanced protective efficacy of a chimeric form of the schistosomiasis vaccine antigen Sm-TSP-2. PLoS Negl Trop Dis. 2012;6:e1564. doi: 10.1371/journal.pntd.0001564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.McManus D.P., Loukas A. Current status of vaccines for schistosomiasis. Clin Microbiol Rev. 2008;21:225–242. doi: 10.1128/CMR.00046-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Keating J.H., Wilson R.A., Skelly P.J. No overt cellular inflammation around intravascular schistosomes in vivo. J Parasitol. 2006;92:1365–1369. doi: 10.1645/GE-864R.1. [DOI] [PubMed] [Google Scholar]
- 176.Loukas A., Tran M., Pearson M.S. Schistosome membrane proteins as vaccines. Int J Parasitol. 2007;37:257–263. doi: 10.1016/j.ijpara.2006.12.001. [DOI] [PubMed] [Google Scholar]
- 177.Tallima H., El Ridi R. Schistosoma mansoni glyceraldehyde 3-phosphate dehydrogenase is a lung-stage schistosomula surface membrane antigen. Folia Parasitol (Praha) 2008;55:180–186. doi: 10.14411/fp.2008.025. [DOI] [PubMed] [Google Scholar]
- 178.El Ridi R., Tallima H. Schistosoma mansoni ex vivo lung-stage larvae excretory–secretory antigens as vaccine candidates against schistosomiasis. Vaccine. 2009;27:666–673. doi: 10.1016/j.vaccine.2008.11.039. [DOI] [PubMed] [Google Scholar]
- 179.Harrop R., Wilson R.A. Protein synthesis and release by cultured schistosomula of Schistosoma mansoni. Parasitology. 1993;107:265–274. doi: 10.1017/s0031182000079245. [DOI] [PubMed] [Google Scholar]
- 180.Harrop R., Coulson P.S., Wilson R.A. Characterization, cloning and immunogenicity of antigens released by lung-stage larvae of Schistosoma mansoni. Parasitology. 1999;118:583–594. doi: 10.1017/s003118209900431x. [DOI] [PubMed] [Google Scholar]
- 181.Curwen R.S., Ashton P.D., Johnston D.A., Wilson R.A. The Schistosoma mansoni soluble proteome: a comparison across four life-cycle stages. Mol Biochem Parasitol. 2004;138:57–66. doi: 10.1016/j.molbiopara.2004.06.016. [DOI] [PubMed] [Google Scholar]
- 182.Knudsen G.M., Medzihradszky K.F., Lim K.C., Hansell E., McKerrow J.H. Proteomic analysis of Schistosoma mansoni cercarial secretions. Mol Cell Proteomics. 2005;4:1862–1875. doi: 10.1074/mcp.M500097-MCP200. [DOI] [PubMed] [Google Scholar]
- 183.Curwen R.S., Ashton P.D., Sundaralingam S., Wilson R.A. Identification of novel proteases and immunomodulators in the secretions of schistosome cercariae that facilitate host entry. Mol Cell Proteomics. 2006;5:835–844. doi: 10.1074/mcp.M500313-MCP200. [DOI] [PubMed] [Google Scholar]
- 184.Kroczek R.A., Mages H.W., Hutloff A. Emerging paradigms of T-cell co-stimulation. Curr Opin Immunol. 2004;16:321–327. doi: 10.1016/j.coi.2004.03.002. [DOI] [PubMed] [Google Scholar]
- 185.Pearce E.J., Caspar P., Grzych J.M., Lewis F.A., Sher A. Downregulation of Th1 cytokine production accompanies induction of Th2 responses by a parasitic helminth, Schistosoma mansoni. J Exp Med. 1991;173:159–166. doi: 10.1084/jem.173.1.159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Coulson P.S. The radiation-attenuated vaccine against schistosomes in animal models: paradigm for a human vaccine? Adv Parasitol. 1997;39:271–336. doi: 10.1016/s0065-308x(08)60048-2. [DOI] [PubMed] [Google Scholar]
- 187.Reis E.A., Mauadi Carmo T.A., Athanazio R., Reis M.G., Harn D.A., Jr. Schistosoma mansoni triose phosphate isomerase peptide MAP4 is able to trigger naïve donor immune response towards a type-1 cytokine profile. Scand J Immunol. 2008;68:169–176. doi: 10.1111/j.1365-3083.2008.02131.x. [DOI] [PubMed] [Google Scholar]
- 188.El Ridi R., Tallima H., Mahana N., Dalton J.P. Innate immunogenicity and in vitro protective potential of Schistosoma mansoni lung schistosomula excretory–secretory candidate vaccine antigens. Microbes Infect. 2010;12:700–709. doi: 10.1016/j.micinf.2010.04.012. [DOI] [PubMed] [Google Scholar]
- 189.Dale D.C., Boxer L., Liles W.C. The phagocytes: neutrophils and monocytes. Blood. 2008;112:935–945. doi: 10.1182/blood-2007-12-077917. [DOI] [PubMed] [Google Scholar]
- 190.Hickey M., Kubes P. Intravascular immunity: the host-pathogen encounter in blood vessels. Nat Rev Immunol. 2009;9:364–375. doi: 10.1038/nri2532. [DOI] [PubMed] [Google Scholar]
- 191.Wynn T.A., Oswald I.P., Eltoum I.A., Caspar P., Lowenstein C.J., Lewis F.A. Elevated expression of Th1 cytokines and nitric oxide synthase in the lungs of vaccinated mice after challenge infection with Schistosoma mansoni. J Immunol. 1994;153:5200–5209. [PubMed] [Google Scholar]
- 192.Ahmed S.F., Oswald I.P., Caspar P., Hieny S., Keefer L., Sher A. Developmental differences determine larval susceptibility to nitric oxide-mediated killing in a murine model of vaccination against Schistosoma mansoni. Infect Immun. 1997;65:219–226. doi: 10.1128/iai.65.1.219-226.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Tallima H., Salah M., Guirguis F.R., El Ridi R. Transforming growth factor-β and Th17 responses in innate resistance to murine Schistosomiasis mansoni. Cytokine. 2009;48:239–245. doi: 10.1016/j.cyto.2009.07.581. [DOI] [PubMed] [Google Scholar]
- 194.Saadi S., Wrenshall L.E., Platt J.L. Regional manifestations and control of the immune system. FASEB J. 2002;16:849–856. doi: 10.1096/fj.01-0690hyp. [DOI] [PubMed] [Google Scholar]
- 195.Humphreys N.E., Xu D., Hepworth M.R., Liew F.Y., Grencis R.K. IL-33, a potent inducer of adaptive immunity to intestinal nematodes. J Immunol. 2008;180:2443–2449. doi: 10.4049/jimmunol.180.4.2443. [DOI] [PubMed] [Google Scholar]
- 196.Saenz S.A., Taylor B.C., Artis D. Welcome to the neighborhood: epithelial cell-derived cytokines license innate and adaptive immune responses at mucosal sites. Immunol Rev. 2008;226:172–190. doi: 10.1111/j.1600-065X.2008.00713.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Tang H., Cao W., Kasturi S.P., Ravindran R., Nakaya H.I., Kundu K. The T helper type 2 response to cysteine proteases requires dendritic cell-basophil cooperation via ROS-mediated signaling. Nat Immunol. 2010;11:608–617. doi: 10.1038/ni.1883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Siracusa M.C., Saenz S.A., Hill D.A., Kim B.S., Headley M.B., Doering T.A. TSLP promotes interleukin-3-independent basophil haematopoiesis and type 2 inflammation. Nature. 2011;477:229–233. doi: 10.1038/nature10329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Neill D.R., McKenzie A.N. Nuocytes and beyond: new insights into helminth expulsion. Trends Parasitol. 2011;27:214–221. doi: 10.1016/j.pt.2011.01.001. [DOI] [PubMed] [Google Scholar]
- 200.El Ridi R, Tallima H. Vaccine-induced protection against murine Schistosomiasis mansoni with larval excretory–secretory antigens and papain or type-2 cytokines. J Parasitol 2013;99:194–202. [DOI] [PubMed]


