Abstract
Objective:
The authors aimed to develop a mechanical model for delivering controlled impact injuries to an articular cartilage surface, with the ability to control peak contact pressure while maintaining a constant rate of loading across all impact levels.
Methods:
A pendulum-style impactor with a direct contact interface was fabricated, based on previously published successful models. This impactor was tested in 2 separate experiments: first with a single analog specimen and next with 15 cadaveric rabbit knee specimens. In both experiments, impacts were performed at 3 pressure levels with multiple repetitions. Peak pressure, rate of pressure application, impulse, and time to peak were evaluated.
Results:
There was a significant difference between each of the pressure level groups for both the analog and cadaveric experiments. There was no significant difference between the rates of pressure application for the analog specimen and a statistically significant difference only between the highest and lowest pressure level groups for the cadaveric specimen.
Conclusions:
Previous studies have shown that peak pressure and rate of pressure application are both significant contributors to cartilage injury. Previous models have controlled only peak contact pressure, allowing rate of loading to change as a function of peak contact pressure. This model is the first to control both variables, with a rate of pressure application that is typical of an athletic or traumatic injury in humans. Use of this model will enable researchers to investigate the isolated effects of peak pressure on progressive cartilage injuries.
Keywords: animal models, biomechanics, articular cartilage, posttraumatic arthritis, knee
Introduction
Osteoarthritis affects an estimated 12.1% of the population in the United States between the ages of 25 and 75, making it one of the leading causes of disability and impairment in this age group.1 Many surgical treatments address the end stages of cartilage injury and osteoarthritis, but few treatments are available to prevent the development of the disease. Recent research has indicated that blunt-impact injuries play a role in the development of osteoarthritis.2 Because a single injury may lead to osteoarthritis, it may be possible to prevent or forestall the onset of the disease. This fact has prompted research focusing on evaluating possible disease-modifying pharmacologic agents, including oral medications and intra-articular injections. Oral glucosamine and chondroitin sulfate have demonstrated promise in slowing the progression of established osteoarthritis in randomized clinical trials, as evidenced by improved function and decreased radiographic joint space narrowing when compared to placebo.3-5 Several intra-articular hyaluronic acid preparations have demonstrated effectiveness in relieving the symptoms of osteoarthritis, and some data suggest that these agents have disease-modifying potential as well.6
Development of a reliable model for the early stages of cartilage injury and osteoarthritis would enable the testing of novel treatments that could slow or even reverse the progression of this debilitating disease. Several animal models for initiating osteoarthritis have been proposed by applying blunt impact to articular cartilage but with inconsistent results.7-15 Both impact force magnitude and rate of pressure application have been shown to be important factors in blunt cartilage injury.10 Although previous models have been successful in varying the impact force magnitude, none have done so while maintaining constant rate of loading analogous to the rate found during athletic or traumatic injury.12 The closest approximation of the rate of pressure application available in literature is the time to peak force. The time to peak force is thought to occur at between 20 and 50 ms in an automobile accident.10 A model capable of controlling both peak pressure and the rate of pressure application would allow investigators to examine each factor independently.
Direct impact to cartilage has been shown to be the best technique available to allow precise control of both location and force application and thus was chosen for use in our model.7,8 Direct-impact techniques have typically used an artificial impactor to directly impact the posterior aspect of the medial femoral condoyle. The major sources of variation among direct impactors are the impact interface and the method of energy production. Both conforming7,8 and nonconforming12 impactor interfaces have been used; the conforming type delivers a more even and controllable pressure distribution with less stress concentration than a nonconforming interface.15 The energy required for these impacts has generally been created by a hydraulic apparatus,12 drop tower,8,15 or pendulum.7 Hydraulic devices, although able to precisely control variable impacts, have not reproduced the rates of pressure application thought to be typical of those seen in human articular cartilage injury. Drop tower–type impactors have inconsistencies related to friction resulting from the use of linear bearings. Therefore, we chose to use a pendulum-style impactor for energy production, which consistently produces variable forces at variable rates, and a conforming impactor interface, which produces a more uniform and consistent pressure distribution.
The current study’s aim was to develop a system that could simultaneously control factors of peak pressure and rate of pressure application. The New Zealand white rabbit was chosen as a model because of its size and well-documented use as a model for cartilage injury and osteoarthritis.7,12 We hypothesize that our blunt-impact delivery device will create a variable impact magnitude while maintaining a constant rate of pressure application that is analogous to that of an impact typically seen in an athletic or traumatic injury.
Methods
A pendulum-style impactor device was manufactured based on the impactor design of Borrelli et al.7 (Fig. 1) to create the energy necessary for impact. The pendulum arm was constructed out of specialty aluminum tubing designed to be rigid yet lightweight (80/20 Inc., Columbia City, IN). The 0.9-m pendulum arm was attached to a supporting ladder-like structure using low-friction bearings. The pendulum arm swing was initiated by the release of an electromagnet. Weights were attached to the bottom of the pendulum arm to adjust the amount of energy created from the swing. The arm struck a foam-padded impactor assembly riding on a unidirectional rail. A load cell (Sensotec, Columbus, OH) was mounted between the impactor head and the remainder of this assembly. A specimen was transfixed to a mounting assembly via 2 bicortical 2-mm Kirchner wires. An XY table on this assembly along with controlled rail position allowed precise positioning of the specimen in 3 dimensions. When the specimen was correctly positioned, the table was locked in place.
Figure 1.
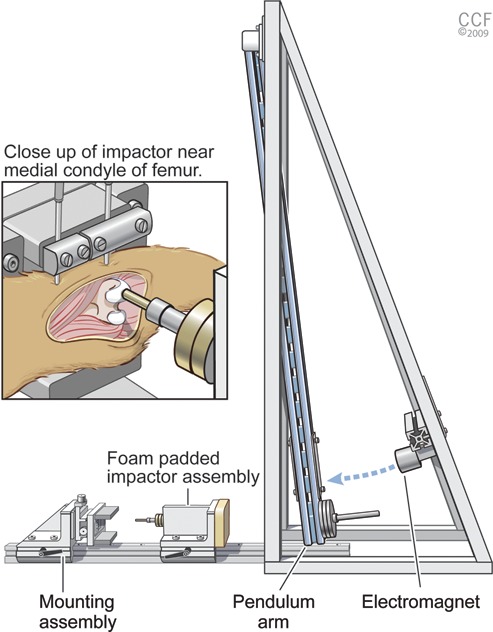
Impact apparatus with close-up of impact interface. The main components are a pendulum arm, electromagnet, foam-padded impactor assembly, and mounting assembly. Reprinted with permission, Cleveland Clinic Center for Medical Art & Photography © 2009-2010. All rights reserved.
The energy created from the pendulum was transferred to the cartilage surface via a stainless steel conforming impactor head with a 7-mm concave spherical radius. This shape was modeled after the indenter head used by Borrelli et al.,7 who found the curvature characteristics of the rabbit medial condyle to be a sagittal radius of 7.14 mm and a coronal radius of 5.56 mm. We approximated this curvature to be 7 mm in both directions because of the difficulties associated with manufacturing this complex shape and properly aligning the specimen to the impactor head. To align the medial condyle and the impactor head, Pressurex Micro supersensitive pressure film (Sensor Products, East Hanover, NJ) was placed between the contact surfaces. The contact area between the impactor and cartilage surface was uniform in each case, validating the use of a single radius curvature for the impactor. Pressure film was not used during actual impaction; rather, it was used only to verify proper alignment prior to impact. Data collection on actual conveyance of pressure during the impaction was collected via a single-axis load cell.
Force data were acquired from the load cell at 5000 Hz. These data were used to calculate time to peak, peak pressure, rate of pressure application, and impulse. Figure 2 shows a response curve from a typical impact. Time to peak was calculated as the time between first contact and the peak pressure. Peak pressure was calculated as the maximum pressure and was always found on the second peak. Rate of pressure application was calculated as the slope of the curve with the peak pressure. To calculate rate of pressure application, the maximum rate along the peak curve was identified. Points 10% on either side of the increasing side of the curve were then calculated, and the slope was found between these points.16 Finally, impulse or the energy created from the impact is a representation of the area under the curve.
Figure 2.
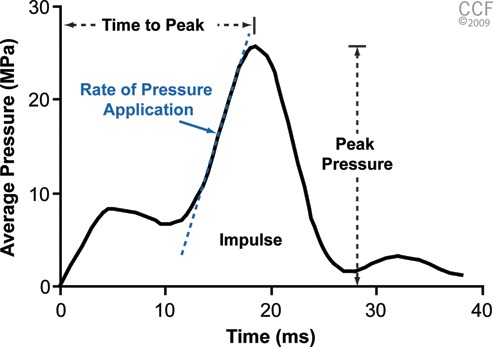
Typical profile showing the impact of a 25-MPa impact specimen. Values obtained from the curve were time to peak, peak pressure, rate of pressure application, and impulse (area under the curve). Reprinted with permission, Cleveland Clinic Center for Medical Art & Photography © 2009-2010. All rights reserved.
Two series of tests were performed to establish the ability to control rate while adjusting pressure magnitude in first an analog model and then a cadaveric model. The analog model used in the first testing series consisting of a wood block with a 1.0-mm-thick piece of viscoelastic foam. This design provided a loading profile similar to that of the medial femoral condyle. Testing was performed on an analog rather than cadaveric specimen to remove specimen-to-specimen variation. Testing consisted of impacting the same specimen with 10 repetitions at each of 3 pressure levels between 15 and 45 MPa. Pendulum weights and foam were adjusted to reach a rate of pressure application of approximately 3900 MPa/s, which resulted in a time to peak of between 20 and 50 ms for all trials.
The second series of tests was performed on cadaveric specimens from adult rabbits. New Zealand White rabbits (n = 15) were used. Immediately following euthanasia, cadaveric femurs were dissected, placed in a buffered saline solution with protease inhibitors (Calbiochem, San Diego, CA), and stored on ice until testing. The purpose of this second experiment was to establish the ability of the impactor to create variable pressure-level impacts while maintaining a constant rate of pressure application in a cadaveric specimen. For this series of tests, 3 peak pressure goals were identified: 20, 25, and 30 MPa. Five cadaveric femurs were tested at each of these pressure levels, resulting in a total of 15 specimens.
Data for each of the 2 experiments were analyzed using a one-way analysis of variance (ANOVA) of pressure group for peak pressure magnitude, rate of pressure application, impulse, and time to peak. Significance between groups was analyzed using a Tukey post hoc analysis. Significance levels for both the ANOVA and the post hoc analysis were set at α = 0.05.
Results
Peak Pressure
Table 1 shows the results for the analog and cadaveric experiments. Peak pressure, as shown in Figure 2, is the maximum pressure conveyed to the specimen during impact. The analog experiment showed significant differences between each of the pressure levels (P < 0.001), demonstrating that the low-, medium-, and high-pressure groups resulted in 3 statistically independent peak pressure levels. Likewise, the cadaveric experiment showed significant differences between each of the pressure levels (P < 0.001). The peak pressure results are shown in Figure 3.
Table 1.
Results for Analog and Cadaveric Experiments (Mean ± 95% Confidence Interval)
| Impact Level | Peak Pressure, MPa | Pressure Rate, MPa/s | Impulse, N-s | Time to Peak, ms |
|---|---|---|---|---|
| Analog experiment | ||||
| Low | 18.37 ± 0.56 | 3900 ± 135 | 1.26 ± 0.01 | 21.5 ± 0.53 |
| Medium | 26.87 ± 0.80 | 3928 ± 311 | 2.32 ± 0.01 | 32.5 ± 1.02 |
| High | 41.82 ± 1.08 | 3868 ± 128 | 5.14 ± 0.01 | 45.5 ± 0.39 |
| Cadaveric experiment | ||||
| Low | 21.69 ± 1.02 | 3727 ± 292 | 1.82 ± 0.01 | 18.0 ± 0.25 |
| Medium | 26.64 ± 0.54 | 4272 ± 364 | 2.40 ± 0.01 | 23.3 ± 1.20 |
| High | 31.55 ± 1.01 | 4587 ± 211 | 3.04 ± 0.02 | 24.2 ± 0.26 |
Figure 3.
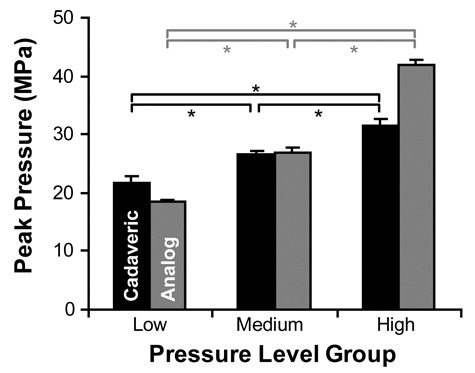
Peak pressure magnitudes (mean ± 95% confidence interval) for low-, medium-, and high-pressure groups. In both the cadaveric and analog experiments, the 3 pressure-level groups were significantly different from one another (*P < 0.001).
Rate of Pressure Application
Pressure rate, shown in Figure 2, was defined as the rate during the peak loading curve. The analog experiment revealed no significant change in rate of pressure application between each of the pressure-level groups, demonstrating that changes in pressure magnitude did not significantly affect the rate of pressure application. The cadaveric experiment found no difference between the medium-/low-pressure and medium-/high-pressure groups, but there was a significant change between the low- and high-pressure groups (P < 0.05). The results for rate of pressure application are shown in Figure 4.
Figure 4.
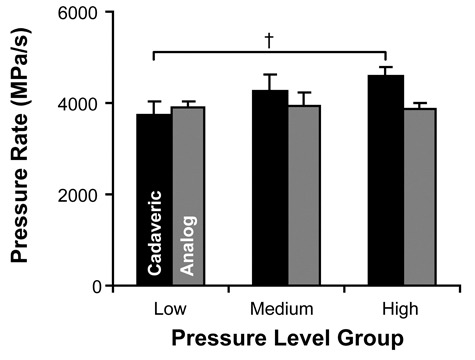
Rate of pressure application (mean ± 95% confidence interval) for low-, medium-, and high-pressure groups. There were no statistically significant changes to the pressure rate except between the low- and high-pressure level groups of the cadaveric experiment (†P < 0.05).
Time to Peak
Time to peak, shown in Figure 2, is the time between the point of first contact and the point of peak pressure. The analog experiment showed significant increased time to peak for increasing pressure levels (P < 0.001). Likewise, the cadaveric experiment showed a significant correlation between increasing pressure levels and increasing time to peak (P < 0.001).
Impulse
Impulse, shown in Figure 2, is the area under the force-time curve. Impulse is a representation of the level of energy transferred from the impactor to the specimen during impact. The analog experiment showed a significant correlation between pressure level and impulse (P < 0.001). As the pressure-level group increased, the impulse also increased. Likewise, the cadaveric experiment showed a similar significant relationship between the pressure-level group and impulse (P < 0.001).
Discussion
Osteoarthritis affects approximately 12.1% of the US population. It is a serious and sometimes debilitating condition that is one of the leading causes of disability.1 Still, little is known about the natural progression of the disease, and few treatments are available to limit this progression. Previous authors have used models similar to the one presented here to investigate the progression and treatment of osteoarthritis in animal models.7,8,12,15 However, none maintained a rate of pressure application similar to the rate exhibited in an athletic or traumatic injury in humans while simultaneously examining various pressure levels. This is an important distinction, because, in 2002, Ewers et al.10 showed that the rate of pressure application plays a critical role in the development of an injury following impact. Therefore, our study was undertaken to develop the first model of a mechanical impact to rabbit articular cartilage that allows for control of both the peak pressure and the rate of pressure application, at a clinically relevant rate. Development of such a model will enable further research looking at the progression of osteoarthritis following impact injury as well as testing of novel treatments to combat this progression.
Peak pressure was evaluated at 3 levels for both the analog and cadaveric experiments. In the analog experiment, our goal was to achieve 3 distinct pressure groups between 15 and 45 MPa, based on our estimate from the literature on the extremes of when a progressive injury would develop for a rate of pressure of approximately 3900 MPa/s. In addition to showing that we were able to achieve 3 statistically distinct pressure levels, we also demonstrated that we were able to encompass our estimate of the extremes of pressure that initiate a progressive osteoarthritic injury (15-45 MPa). In the cadaveric experiment, we began testing with the high peak pressure impact level and demonstrated the ability to precisely control the peak pressure by accurately hitting targets 10 and 5 MPa below this level (see Table 1). The cadaveric experiment showed that we were able to again reach 3 statistically distinct levels in an environment more similar to the surgical setup to be used in future testing.
For rate of pressure application, our goal was to maintain a constant rate of loading throughout each experiment. We found no direct description in the literature of the rate at which force is transmitted to femoral articular cartilage during an athletic or traumatic injury. The closest approximation of this rate was performed by Ewers et al.,10 who reported that the peak force occurs between 20 and 50 ms after impact in an automobile accident. We chose our rate of pressure application by trying to fit this window in the analog experiment. This strategy gave us a rate of approximately 3900 MPa/s as a rate that would allow all of our pressure levels to fall within the 20- to 50-ms window, as shown in Table 1. In the analog experiment, there was no significant difference between the pressure groups for this rate (P < 0.001), and the means of each group were close to 3900 MPa/s. For the cadaveric experiment, the first group evaluated was the high-pressure level group. After testing at this level, we observed the rate of pressure application was too high. In the next pressure group evaluated, the low-pressure level, we overcompensated for this and undershot the goal rate of pressure application. The final group, the medium-pressure level, was centered between the 2 groups but was still slightly higher than the pressure-level goal. As a result, there was a statistically significant difference between the low- and high-pressure groups (P < 0.05) but not between any of the others (Fig. 4).
The design of our impactor can be subdivided into 2 components: a method for energy creation and a method of impact interface. For energy creation, we used a pendulum-style apparatus that uses gravity to create the energy needed. This design has previously been shown in the literature to be a consistent method for producing energy, and we found the same. For impact interface, we used a direct-contact interface. This method requires surgically opening the joint to get direct access to the surface of interest, as opposed to transmitting the energy through multiple layers of tissue from an exterior impact. This technique has been shown in literature to be the best way of controlling the impact.
As with any study, we acknowledge that there are limitations to our approach. One limitation was the use of cadaveric rather than living specimens. However, we do not believe that there are significant differences in the material properties between the two types that would justify increasing the number of live animals used in the study. Another potential limitation is the calculation of contact pressure. Because we measure force going to the rigid impactor head and not the direct-contact pressure, we are actually calculating an average contact pressure over the entire surface of the impactor head. We have attempted to make this contact as uniform as possible by contouring the impactor head to match the curvature of the condyle, but we cannot be sure that we have perfect alignment and that the contact stays uniform throughout the deformation caused by the impact.
In conclusion, we have developed a mechanical method for creating an impact that can control both the peak pressure and the rate of pressure application in an animal model of knee cartilage injury. Ours is the first model to investigate multiple pressure levels while maintaining a constant rate of pressure that mimics that of an athletic or traumatic injury in humans. This model will enable future research to investigate the isolated effects of peak pressure and pressure rate on progressive cartilage injuries.
Acknowledgments
The authors thank Sam Butler of Cleveland Clinic’s Department of Quantitative Health Sciences for statistical analysis.
Footnotes
Declaration of Conflicting Interests: Stephen Fening, Morgan Jones, and Anthony Miniaci have received unrestricted research grants from Arthrex, Donjoy, BREG, and Stryker. Anthony Miniaci has received royalties from Zimmer and Tenet, has received nonincome support from Arthrosurface, and is a consultant or employee for Arthrosurface and Stryker.
Funding: The authors received the following financial support for the research and/or authorship of this article: Internal Research Programs Committee grant from the Cleveland Clinic (PI: Jones) and Medical Grant from NFL Charities (PI: Jones).
References
- 1. Lawrence RC, Helmick CG, Arnett FC, Deyo RA, Felson DT, Giannini EH, et al. Estimates of the prevalence of arthritis and selected musculoskeletal disorders in the United States. Arthritis Rheum. 1998;41:778-99. [DOI] [PubMed] [Google Scholar]
- 2. Carter DR, Beaupre GS, Wong M, Smith RL, Andriacchi TP, Schurman DJ. The mechanobiology of articular cartilage development and degeneration. Clin Orthop Relat Res. 2004;427(suppl):S69-77. [DOI] [PubMed] [Google Scholar]
- 3. McAlindon TE, LaValley MP, Gulin JP, Felson DT. Glucosamine and chondroitin for treatment of osteoarthritis: a systematic quality assessment and meta-analysis. JAMA. 2000;283:1469-75. [DOI] [PubMed] [Google Scholar]
- 4. Pavelka K, Gatterova J, Olejarova M, Machacek S, Giacovelli G, Rovati LC. Glucosamine sulfate use and delay of progression of knee osteoarthritis: a 3-year, randomized, placebo-controlled, double-blind study. Arch Intern Med. 2002;162:2113-23. [DOI] [PubMed] [Google Scholar]
- 5. Reginster JY, Deroisy R, Rovati LC, Lee RL, Lejeune E, Bruyere O, et al. Long-term effects of glucosamine sulphate on osteoarthritis progression: a randomised, placebo-controlled clinical trial. Lancet. 2001;357:251-6. [DOI] [PubMed] [Google Scholar]
- 6. Goldberg VM, Buckwalter JA. Hyaluronans in the treatment of osteoarthritis of the knee: evidence for disease-modifying activity. Osteoarthritis Cartilage. 2005;13:216-24. [DOI] [PubMed] [Google Scholar]
- 7. Borrelli J, Jr, Burns ME, Ricci WM, Silva MJ. A method for delivering variable impact stresses to the articular cartilage of rabbit knees. J Orthop Trauma. 2002;16:182-8. [DOI] [PubMed] [Google Scholar]
- 8. Vrahas MS, Smith GA, Rosler DM, Baratta RV. Method to impact in vivo rabbit femoral cartilage with blows of quantifiable stress. J Orthop Res. 1997;15:314-7. [DOI] [PubMed] [Google Scholar]
- 9. Atkinson PJ, Ewers BJ, Haut RC. Blunt injuries to the patellofemoral joint resulting from transarticular loading are influenced by impactor energy and mass. J Biomech Eng. 2001;123:293-5. [DOI] [PubMed] [Google Scholar]
- 10. Ewers BJ, Jayaraman VM, Banglmaier RF, Haut RC. Rate of blunt impact loading affects changes in retropatellar cartilage and underlying bone in the rabbit patella. J Biomech. 2002;35:747-55. [DOI] [PubMed] [Google Scholar]
- 11. Haut RC, Ide TM, De Camp CE. Mechanical responses of the rabbit patello-femoral joint to blunt impact. J Biomech Eng. 1995;117:402-8. [DOI] [PubMed] [Google Scholar]
- 12. Milentijevic D, Rubel IF, Liew AS, Helfet DL, Torzilli PA. An in vivo rabbit model for cartilage trauma: a preliminary study of the influence of impact stress magnitude on chondrocyte death and matrix damage. J Orthop Trauma. 2005;19:466-73. [DOI] [PubMed] [Google Scholar]
- 13. Repo RU, Finlay JB. Survival of articular cartilage after controlled impact. J Bone Joint Surg Am. 1977;59:1068-76. [PubMed] [Google Scholar]
- 14. Newberry WN, Garcia JJ, Mackenzie CD, Decamp CE, Haut RC. Analysis of acute mechanical insult in an animal model of post-traumatic osteoarthrosis. J Biomech Eng. 1998;120:704-9. [DOI] [PubMed] [Google Scholar]
- 15. Zhang H, Vrahas MS, Baratta RV, Rosler DM. Damage to rabbit femoral articular cartilage following direct impacts of uniform stresses: an in vitro study. Clin Biomech (Bristol, Avon). 1999;14:543-8. [DOI] [PubMed] [Google Scholar]
- 16. Flynn JM, Holmes JD, Andrews DM. The effect of localized leg muscle fatigue on tibial impact acceleration. Clin Biomech (Bristol, Avon). 2004;19:726-32. [DOI] [PubMed] [Google Scholar]


