Abstract
Despite a strong historical association between frontal lobe lesions and executive dysfunction, questions remain regarding the neuropsychological characterization of specific regions within the prefrontal cortex, as well as the utility of some executive function measures. The Delis-Kaplan Executive Function System (D-KEFS; Delis, Kaplan, & Kramer, 2001) has gained popularity as a collection of executive function tests, but its sensitivity and specificity for detecting focal frontal lobe dysfunction remain uncertain. The current study investigated performances of patients with focal ventromedial prefrontal (vmPFC, n=13), dorsolateral prefrontal (dlPFC, n=14), and non-frontal (NF, n=18) lesions on the entire D-KEFS battery. We also obtained IQ data from conventional measures (the WAIS-III/IV). On six D-KEFS indices, patients with dlPFC lesions performed lower than patients with vmPFC and NF lesions. On three other indices, the performances of the dlPFC group were lower than the NF group, but did not differ from the vmPFC group. However, none of these between-group differences were statistically significant after Full Scale IQ and processing speed were controlled for, suggesting that the observed weaknesses in patients with dlPFC lesions were not specific to executive functions. Patients with vmPFC and NF lesions could not be differentiated based on their performances on any D-KEFS measures. While some D-KEFS measures show promise in differentiating patients with dlPFC lesions, the clinical advantage of using the D-KEFS over more traditional measures is not empirically compelling.
Keywords: Prefrontal lesions, D-KEFS, Executive function, Frontal lobe, vmPFC
INTRODUCTION
Executive functions is a term referring to a variety of higher-order cognitive processes that have been historically associated with frontal lobe function. While the relationship between frontal lobe damage and executive dysfunction has been well established, this relationship is complex and there is no one-to-one correspondence between neuroanatomical damage and functional outcome. Furthermore, there is evidence of significant functional complexity and variability within the subregions of the frontal lobes (e.g., Damasio, Anderson, & Tranel, 2012).
One way to subdivide the prefrontal cortex is to consider the ventromedial (vmPFC) and dorsolateral (dlPFC) regions. Executive functions associated with the vmPFC and dlPFC have been described as “hot” and “cold”, respectively (Chan, Shum, Toulopoulou, & Chen, 2008). “Cold” functions are more logically-based and include skills that are typically considered as “executive,” including attention regulation, inhibition, problem-solving, and working memory. These skills contribute to what has been described as “cognitive control” necessary for planning and executing behavior (Gläscher et al., 2012). “Hot” executive functions, on the other hand, consist of more emotionally- and socially-based skills, such as judgment, emotional regulation, and decision-making that relies on personal preferences and desires. They have been referred to as the “valuation” system to denote functions that motivate behavior (Gläscher et al., 2012). Gläscher et al. (2012) used voxel-based lesion-symptom method (VLSM) to demonstrate that “largely non-overlapping sectors of the prefrontal cortex subserve cognitive control and valuation” (p. 14683) even when controlling for general verbal and visual abilities and memory. Cognitive control was assessed using common neuropsychological measures of executive function (Controlled Oral Word Association Test, Wisconsin Card Sorting Test, Stroop Color-Word Test, and Trail-Making Test) and lower performances on these measures were associated with specific sectors of the prefrontal cortex, including primarily the dorsal areas of the medial prefrontal cortex, as well as dorsal and ventral portions of the lateral prefrontal cortex. Valuation was measured with the Iowa Gambling Task (IGT), which was the only task associated with lesions in the ventral and polar regions of the prefrontal cortex, although it was also associated with other areas distributed in the prefrontal cortex. This study supported a high degree of functional specificity within the prefrontal cortex and the notion that commonly used neuropsychological measures of executive function are better suited to detect difficulties associated with dlPFC dysfunction.
However, there is another variable to consider when discussing the degree of specificity of the prefrontal cortices to different executive tasks. Barbey, Colom, and Grafman (2012) examined whether dlPFC damage resulted in deficits in specific executive skills or more overarching cognitive processes by accounting for the general intelligence factor (g), which was derived from performances on the Wechsler Adult Intelligence Scale – III (WAIS-III). In this study, patients with dlPFC lesions performed worse than comparison participants on several D-KEFS tests but the differences disappeared once the results were controlled for g. The authors concluded that dlPFC lesions were associated with reductions in g and that the dlPFC “is not functionally dedicated to support specific executive processes but may instead support higher-level mechanisms for general intelligence” (p.5). The Gläscher et al. (2012) study and the Barbey, Colom, and Grafman (2012) study used different measures and methods, and questions remain regarding the sensitivity and specificity of the D-KEFS and more traditional executive function measures to focal damage in different sectors of the frontal lobes. As a final note, while the IGT has been found to be associated with vmPFC, it is not yet widely used in clinical settings. Thus, the quantitative characterization of vmPFC dysfunction during a clinical evaluation remains elusive (Zald & Andreotti, 2010).
A wide range of measures exists for the assessment of executive functions in a clinical situation. Most of these measures have a long history and have accumulated a large scientific literature. Some are relatively new and require further investigation. The D-KEFS is in the latter category. The battery is comprised of nine tests mostly derived from existing measures with some modifications. Its’ advantages over single measures include the incorporation of the process approach to analyze various components of performance, as well as a uniform normative sample for a more direct comparison between the tests. The examiner could use the D-KEFS as a full battery or choose to administer specific tests or even parts of tests. While the D-KEFS has been accumulating scientific literature, most lesion studies are performed by the same group of researchers and none report on the whole battery. The purpose of this study was to further investigate the D-KEFS, using a neuropsychological approach that featured groups of patients with focal lesions in the vmPFC, dlPFC, and NF regions. Since all subtests of the D-KEFS were administered to the same group of participants, this allowed us to evaluate the relative sensitivity and specificity of the measures to damage in the dlPFC, vmPFC, and NF regions. Establishing the clinical utility of the D-KEFS is important, as it is becoming more widely used in clinical settings to make diagnostic decisions. However, concerns have been raised about whether the D-KEFS provides superior measures of executive function compared to already existing and often simpler and shorter instruments (Strauss, Sherman, & Spreen, 2006; Lezak, Howieson, Bigler, & Tranel, 2012). There have also been concerns raised regarding the psychometric properties of many of the D-KEFS measures, particularly with regard to reliability (Crawford, Sutherland, & Garthwaite, 2008; Lezak et al., 2012).
A detailed review of each of the nine D-KEFS subtests is beyond the scope of this article. Briefly, we will review relevant lesion studies using the D-KEFS. Yochim, Baldo, Nelson, and Delis (2007) found that patients with lateral prefrontal lesions (N = 12) performed significantly slower compared to normal controls on the Number-Letter Switching condition of the D-KEFS Trail Making Test. Baldo, Shimamura, and Delis (2001) reported that patients with focal frontal lobe lesions (N = 11) performed significantly worse than normal controls on the D-KEFS Verbal Fluency test. Participants with left frontal lesions performed worse than participants with right frontal lesions. In the same sample, patients with frontal lobe lesions performed significantly worse than normal controls on the Design Fluency subtest (Baldo et al., 2001). Baldo, Delis, Wilkins, and Shimamura (2004) showed that patients with frontal lobe lesions (N = 12) asked significantly more concrete and inefficient questions to arrive at the target items on the 20 Questions Test compared to normal controls. Keil, Baldo, Kaplan, Kramer, and Delis (2005) found that patients with prefrontal cortex lesions (N = 12) had greater difficulty inferring the meaning of made-up words on the Word Context test compared to normal controls. Patients with lateral prefrontal lesions (N = 12) obtained a significantly lower overall achievement score on the D-KEFS Tower test and had higher time-per-move ratio and more rule violations compared to normal controls (Yochim, Baldo, Kane, & Delis, 2008).
A case study of a man with vmPFC damage revealed a speed/accuracy trade-off on the D-KEFS Trails Letter-Number Switching condition, where his performance was fast but inaccurate (Cato, Delis, Abildskov, & Bigler, 2004). The same patient performed in the average range on all conditions of the D-KEFS Verbal Fluency and Design Fluency tests but the speed/accuracy trade-off was again observed on the Switching component of Design Fluency. He performed normally on the Free Sorting and Sort Recognition conditions of the D-KEFS Sorting test. However, he had a greater than normal number of incorrect sort descriptions on the Sort Recognition condition. On the D-KEFS Color-Word Interference test, the patient did well on the first three conditions, but committed multiple errors on the Switching condition, which combines inhibition and set shifting. His performance on the D-KEFS 20-Questions Test was normal.
In summary, most studies suggest that participants with frontal lobe lesions perform worse on multiple D-KEFS measures compared to healthy comparison participants. The majority of the lesion patients represented in this literature had lateral lesions, which could be considered closest to dlPFC, while patients with vmPFC lesions were underrepresented, making it difficult to draw conclusions about their performances on the D-KEFS and how they compare to patients with dlPFC and non-frontal lesions. Also, most of the above-mentioned investigations did not include the full D-KEFS battery or compare D-KEFS performances of patients with frontal lobe lesions to patients with non-frontal lesions. Based on reviewing the literature pertaining to the D-KEFS and tests it was derived from, we predicted that patients with dlPFC lesions would show greater impairment on the majority of the D-KEFS tests than patients with vmPFC and non-frontal (NF) lesions. To sharpen the brain-behavior relationship results in regard to the D-KEFS, we also explored the role of general intelligence in D-KEFS performances—general intelligence is well known to play a role in many of the functions tapped by D-KEFS tests, but it has often been unaddressed in previous studies.
METHOD
Participants
Participants were identified through the Patient Registry of the Division of Behavioral Neurology and Cognitive Neuroscience at the University of Iowa Hospitals and Clinics. All patients in the Patient Registry have stable focal lesions, well characterized through structural imaging using Brainvox (Damasio & Frank, 1992; Frank, Damasio, & Grabowski, 1997). Patients with a history of mental retardation, learning disability, psychiatric disorder, including active clinical depression, substance abuse, aphasia, or a neurological condition other than the one resulting in the lesion, were excluded. Patients whose lesions resulted from a lobectomy for seizure control and patients with lesions affecting subcortical structures involved in emotional regulation and memory (amygdala and the hippocampus) were also excluded. All participants were in the chronic stage of recovery and were neuropsychologically stable. While chronic stage of recovery was defined as three months or more post lesion onset, in the current sample, the shortest time since lesion onset was three years in the vmPFC group, two years in the dlPFC group, and one year in the NF group. Participants were assigned to groups based on a neuroanatomical analysis, which involved lesion mapping using MAP-3. The final study included 45 participants. Thirteen participants had lesions in the vmPFC (5 bilateral; 3 right; 5 left), and fourteen had lesions in the dlPFC (2 bilateral; 7 right; 5 left). Several lesions in the dlPFC group extended into the superior temporal/parietal regions. MAP-3 lesion overlap maps were available for all 13 participants with vmPFC lesions and 11 participants with dlPFC lesions. These maps are presented in Figures 1 and 2.
Figure 1.
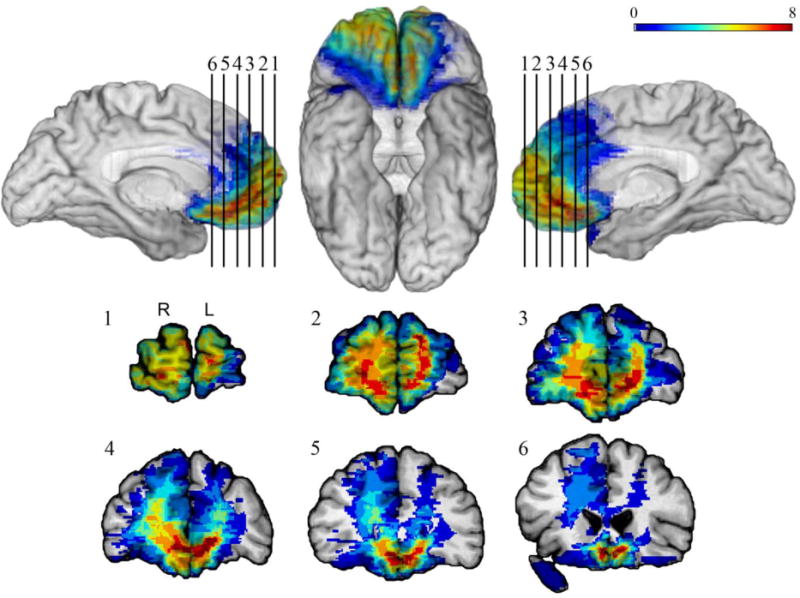
MAP-3 lesion overlap map for the vmPFC lesion group. The color bar shows the extent of overlap, with warmer colors indicating higher numbers of patients with lesions overlapping at a given location. Overlap is mapped on mesial left hemisphere (top left), ventral (top middle), and mesial right hemisphere (top right) views, and the coronal sections (1–6) show the extent of overlap in subcortical areas.
Figure 2.
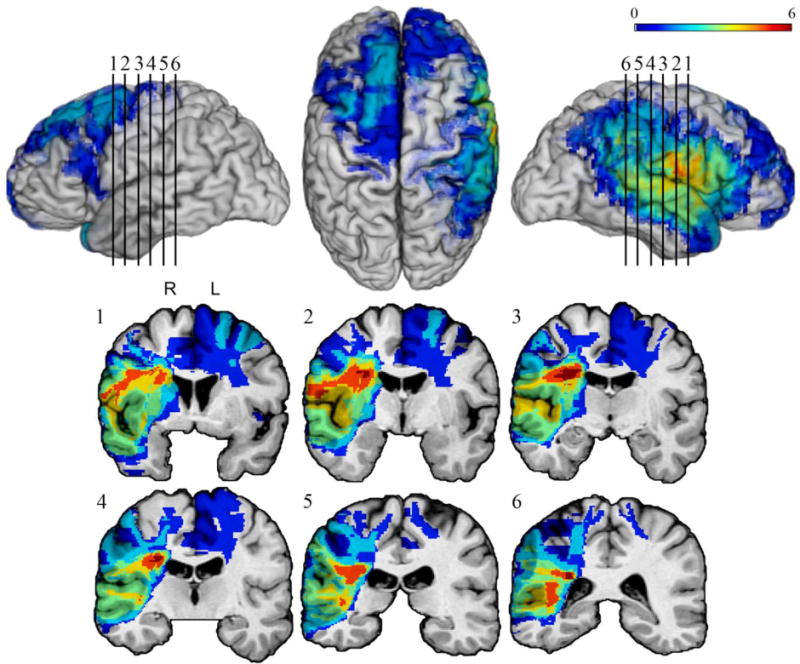
MAP-3 lesion overlap map for the dlPFC lesion group. The color bar shows the extent of overlap, with warmer colors indicating higher numbers of patients with lesions overlapping at a given location. Overlap is mapped on lateral left hemisphere (top left), dorsal (top middle), and lateral right hemisphere (top right) views, and the coronal sections (1–6) show the extent of overlap in subcortical areas.
The NF lesion group consisted of 18 participants (1 bilateral; 6 right; 11 left). Lesion etiologies were varied (Table 1), but in all cases, patients had stable, focal lesions that could be clearly demarcated from neuroimaging and that were static and unchanging over time.
Table 1.
Lesion etiologies
| VMPC (n=13) |
DLPC (n=14) |
Non-Frontal (n=18) |
|
|---|---|---|---|
| CVA | 1 | 8 | 14 |
| Aneurysm | 7 | 2 | 0 |
| Meningioma | 3 | 3 | 3 |
| TBI | 1 | 0 | 1 |
| AVM | 1 | 1 | 0 |
Note: CVA = Cerebrovascular accident (ischemic or hemorrhagic); Meningioma = Resection of benign meningioma tumor; TBI = Traumatic brain injury with focal neuroimaging findings; AVM = Arteriovenous malformation
Instruments
Each subtest of the D-KEFS provides a variety of outcome measures. We decided to limit our investigation to only the primary measures from each subtest, with the exception of ones that were not designed to measure executive functioning (e.g., Motor Speed subtest from the Trail Making Test). The following variables were included: Trail Making Test (Number-Letter Switching), Verbal Fluency Test (Letter Fluency, Category Fluency, and Category Switching Total Correct Responses), Design Fluency Test (Filled Dots, Switching, and Composite), Color-Word Interference Test (Inhibition and Inhibition/Switching), Sorting Test (Free Sorting Confirmed Correct Sorts, Free Sorting Description, and Sort Recognition), Twenty Questions Test (Initial Abstraction and Total Questions Asked), Word Context Test (Total Consecutively Correct), Tower Test (Total Achievement Score), Proverb Test (Total Achievement Score: Free Inquiry). The D-KEFS was administered in one testing session by the first author of this article or a trained research assistant, both of whom were blind to participants’ lesion locations at the time of administration and scoring.
The Patient Registry includes regularly updated data on participants’ intellectual and psychological status. ANOVAs were initially used to explore group differences in test performances. Subsequently, we considered the effects of general intellectual level [Full Scale IQ (FSIQ)] derived from the WAIS-III or the WAIS-IV to increase the specificity of the findings. As a general rule, FSIQ was obtained within two years (prior or post) of participation in the current project (and all of the IQ data were obtained in the chronic post-lesion epoch, 3 months or more after lesion onset). In addition to FSIQ, specific WAIS Index scores were considered in order to evaluate their impact on D-KEFS performances. Groups were also compared on WRAT-3 or 4 Word Reading scores as an estimate of premorbid intellectual function. All analyses were performed using SPSS for Windows (17th edition) using alpha of .05. We note that for the 17 tests reported in Tables 3 and 4, a Bonferroni-corrected alpha is .003. We are using the .05 level for a preliminary survey of the results, simply as a point of reference. Since all of the group contrasts are non-significant with correction for IQ (see below), the issue of alpha correction becomes more or less moot.
Table 3.
Group performances on the D-KEFS using Scaled Scores
| vmPFC (n=13) |
dlPFC (n=14) |
NF (n=18) |
F 2,42 |
P | ηp2 | Obs. Pwr |
|
|---|---|---|---|---|---|---|---|
| Trails | 10.62(3.33) | 8.50(3.6) | 10.00(3.03) | 1.50 | .24 | .07 | .30 |
| 95% CI | 8.60–12.63 | 6.42–10.58 | 8.49–11.51 | ||||
| Letter Fluency | 10.69(4.27) | 7.71(2.6) | 9.22(3.93) | 2.20 | .12 | .10 | .43 |
| 8.11–13.27 | 6.20–9.22 | 7.27–11.18 | |||||
| Cat. Fluency | 10.77(4.53) | 7.00(2.88) | 9.83(2.68) | 4.71 | .01* | .18 | .76 |
| 8.03–13.51 | 5.34–8.66 | 8.50–11.70 | |||||
| Cat. Fluency | 9.92(4.15) | 6.50(3.25) | 9.44(4.03) | 3.29 | .05* | .14 | .59 |
| Switching | 7.41–12.43 | 4.62–8.38 | 7.44–11.45 | ||||
| Des Fluency | 11.54(3.13) | 9.29(2.20) | 11.94(3.12) | 3.71 | .03* | .15 | .65 |
| Composite | 9.65–13.43 | 8.02–10.56 | 10.40–13.49 | ||||
| Des Fluency | 10.85(3.63) | 9.00(2.42) | 11.44(3.05) | 2.63 | .08 | .11 | .50 |
| Filled Dots | 8.66–13.04 | 7.60–10.40 | 9.93–12.96 | ||||
| Design Fluency | 11.08(2.36) | 8.79(2.16) | 10.94(3.19) | 3.32 | .05* | .14 | .60 |
| Switching | 9.65–12.50 | 7.54–10.03 | 9.36–12.53 | ||||
| C-W Inhibition | 10.23(3.32) | 7.43(3.63) | 10.39(2.20) | 4.41 | .02* | .17 | .73 |
| 8.22–12.24 | 5.33–9.53 | 9.29–11.48 | |||||
| C-W Inhibition | 10.00(3.34) | 7.79(3.85) | 10.78(2.80) | 3.34 | .05* | .14 | .60 |
| Switching | 7.98–12.02 | 5.56–10.01 | 9.39–12.17 | ||||
| Free Sort Conf. | 10.38(3.21) | 9.00(1.84) | 11.61(2.83) | 3.70 | .03* | .15 | .65 |
| Correct Sorts | 8.43–12.34 | 7.94–10.06 | 10.20–13.02 | ||||
| Free Sorting | 9.62(3.07) | 8.50(2.41) | 11.06(3.00) | 3.24 | .05* | .13 | .59 |
| Description | 7.76–11.47 | 7.11–9.89 | 9.57–12.54 | ||||
| Sort | 10.00(4.38) | 7.15(2.34) | 10.22(2.96) | 3.75 | .03* | .16 | .65 |
| Recognition | 7.35–12.65 | 5.74–8.57 | 8.75–11.70 | ||||
| 20 Questions | 10.92(3.38) | 9.64(1.55) | 11.67(4.22) | 1.45 | .25 | .07 | .29 |
| Initial Abstract. | 8.88–12.96 | 8.75–10.54 | 9.57–13.76 | ||||
| 20 Questions | 10.69(2.59) | 10.43(2.59) | 11.11(3.16) | .24 | .79 | .01 | .09 |
| Total Questions | 9.12–12.26 | 8.93–11.93 | 9.54–12.68 | ||||
| Word Context | 10.92(3.52) | 9.14(3.35) | 10.06(2.18) | 1.20 | .31 | .05 | .25 |
| Total Correct | 8.79–13.05 | 7.21–11.08 | 8.97–11.14 | ||||
| Tower Total | 13.00(2.83) | 10.43(3.25) | 10.89(2.70) | 2.99 | .06 | .13 | .55 |
| Achievement | 11.29–14.71 | 8.55–12.31 | 9.54–12.24 | ||||
| Proverb Free | 11.77(2.46) | 10.07(3.87) | 11.50(2.75) | 1.25 | .30 | .06 | .26 |
| Inquiry Ach. | 10.29–13.25 | 7.84–12.31 | 10.13–12.87 |
significant at .05 level
Table 4.
D-KEFS FSIQ-adjusted means and group differences with FSIQ as a covariate
| vmPFC (n=13) |
dlPFC (n=14) |
NF (n=18) |
F 2,42 |
P | ηp2 | Obs. Power |
|
|---|---|---|---|---|---|---|---|
| Trails | 10.02(.83) | 9.33(.83) | 9.78(.70) | .17 | .85 | .01 | .07 |
| 95% CI | 8.33–11.71 | 7.67–11.00 | 8.37–11.19 | ||||
| Letter Fluency | 10.10(.96) | 8.55(.95) | 9.00(.80) | .67 | .51 | .03 | .16 |
| 8.16–12.03 | 6.64–10.46 | 7.39–10.62 | |||||
| Cat. Fluency | 10.37(.92) | 7.56(.91) | 9.69(.77) | 2.47 | .10 | .11 | .47 |
| 8.51–12.22 | 5.73–9.39 | 8.14–11.24 | |||||
| Cat. Fluency | 9.39(1.03) | 7.25(1.01) | 9.25(.86) | 1.37 | .27 | .06 | .28 |
| Switching | 7.31–11.47 | 5.20–9.29 | 7.52–10.98 | ||||
| Des Fluency | 10.95(.69) | 10.11(.68) | 11.73(.58) | 1.61 | .21 | .07 | .32 |
| Composite | 9.55–12.35 | 8.73–11.49 | 10.56–12.90 | ||||
| Des Fluency | 10.23(.75) | 9.86(.74) | 11.22(.62) | 1.11 | .34 | .05 | .23 |
| Filled Dots | 8.73–11.74 | 8.38–11.35 | 9.96–12.48 | ||||
| Design Fluency | 10.68(.71) | 9.35(.70) | 10.80(.59) | 1.36 | .27 | .06 | .28 |
| Switching | 9.25–12.10 | 7.94–10.75 | 9.61–11.99 | ||||
| C-W Inhibition | 9.79(.81) | 8.04(.80) | 10.23(.67) | 2.23 | .12 | .10 | .43 |
| 8.17–11.42 | 6.44–9.65 | 8.87–11.59 | |||||
| C-W Inhibition | 9.51(.87) | 8.48(.86) | 10.60(.73) | 1.76 | .19 | .08 | .35 |
| Switching | 7.74–11.27 | 6.74–10.22 | 9.12–12.07 | ||||
| Free Sort Conf. | 9.93(.69) | 9.64(.69) | 11.45(.58) | 2.49 | .10 | .11 | .47 |
| Correct Sorts | 8.53–11.33 | 8.25–11.02 | 10.27–12.62 | ||||
| Free Sorting | 9.11(.72) | 9.21(.71) | 10.87(.60) | 2.41 | .10 | .11 | .46 |
| Description | 7.65–10.57 | 7.77–10.65 | 9.65–12.09 | ||||
| Sort | 9.47(.84) | 7.92(.86) | 10.05(.70) | 1.84 | .17 | .08 | .36 |
| Recognition | 7.77–11.16 | 6.20–9.65 | 8.64–11.47 | ||||
| 20 Questions | 10.14(.76) | 10.74(.75) | 11.38(.63) | .82 | .45 | .04 | .18 |
| Initial Abstract | 8.61–11.67 | 9.23–12.25 | 10.10–12.66 | ||||
| 20 Questions | 10.41(.79) | 10.82(.78) | 11.01(.66) | .18 | .84 | .01 | .08 |
| Total Questions | 8.82–12.00 | 9.26–12.39 | 9.68–12.33 | ||||
| Word Context | 10.15(.63) | 10.23(.62) | 9.77(.53) | .19 | .83 | .01 | .08 |
| Total Correct | 8.88–11.42 | 8.97–11.48 | 8.71–10.84 | ||||
| Tower Total | 12.74(.82) | 10.79(.81) | 10.80(.68) | 2.00 | .15 | .09 | .39 |
| Achievement | 11.09–14.39 | 9.16–12.42 | 9.42–12.17 | ||||
| Proverb Free | 11.08(.71) | 11.03(.70) | 11.25(.60) | .03 | .97 | .00 | .05 |
| Inquiry Ach. | 9.64–12.52 | 9.61–12.46 | 10.05–12.45 |
RESULTS
Participants’ demographic information, lesion chronicity, FSIQ scores, IQ Index Scores, and WRAT Word Reading Scores are presented in Table 2. All participants were Caucasian.
Table 2.
Demographic characteristics, lesion chronicity, FSIQ, and BDI-II scores
| VMPC (n=13) |
DLPC (n=14) |
Non-Frontal (n=18) |
|
|---|---|---|---|
| Sex | 8 Male 5 Female |
11 Male 3 Female |
7 Male 11 Female |
| Age (SD) | 60.00(6.93) | 58.21(9.71) | 60.83(9.98) |
| Education (SD) | 13.92(2.87) | 13.57(2.14) | 15.00 (3.79) |
| Handedness* | 11R;1L;1Mix | 12R;1L;1Mix | 15R; 3Mix |
| Chronicityˆ | 14.38 (8.56) | 9.79 (9.23) | 10.56 (8.36) |
| FSIQ | 111.62(18.62) | 97.07(13.07) | 107.78(15.73) |
| BDI-II | 7.23(6.89) | 10.14(9.68) | 7.39(6.20) |
R=Right-handed; L=Left-handed; Mix=Mixed-handed
Years since lesion onset
The vmPFC, dlPFC, and NF groups did not differ with regard to age [F (2,42) = .33, p = .72], education [F (2,42) = .94, p = .40], lesion chronicity [F (2,42) =1.09, p = .36], FSIQ [F (2,42) = 2.63, p = .08], Verbal Comprehension Index (VCI) [F = (2,42) = 1.20, p = .31], Perceptual Reasoning Index (PRI) [F (2,42) = 1.46, p = .24], Working Memory Index (WMI) [F (2,42) = 2.92, p = .07], Processing Speed Index (PSI) [F (2,42) = 3.06, p = .06], or WRAT Word Reading [F (2,42) = .79, p = .46] .
D-KEFS Performances
ANOVAs comparing group performances revealed significant group differences on the following tests, at p ≤ .05 uncorrected (Table 3): Category Fluency, Category Fluency Switching, Design Fluency Composite, Design Fluency Switching, Color-Word Inhibition, Color-Word Inhibition/Switching, and three variables on the Sorting Test (Confirmed Correct Sorts, Sort Description, and Sort Recognition). Follow-up tests revealed that on all of these variables, there was no significant difference between performances of the vmPFC and NF groups. For Category Fluency, Category Fluency Switching, Design Fluency Composite, Design Fluency Switching, Color-Word Inhibition, and Sort Recognition, the dlPFC group performed significantly worse than the vmPFC and the NF groups. On Color-Word Inhibition/Switching, Confirmed Correct Sorts, and Sort Description, performances of participants with dlPFC and vmPFC lesions did not significantly differ, but the dlPFC group performed significantly worse than the NF group.
Qualitatively, it is notable that all group mean scaled scores in the vmPFC and NF lesion groups were nine or above. However, mean scaled scores for the dlPFC group were below a scaled score of nine on nine indices. In general, on almost every measure, performances of participants with dlPFC lesions were lower than performances of the other two groups. This is illustrated in Figure 3. Furthermore, several group differences approached or exceeded one standard deviation. When examining partial eta squared for the analysis, many of the values would be considered large, indicating that a relatively large proportion of score variability can be attributed to group differences.
Figure 3.
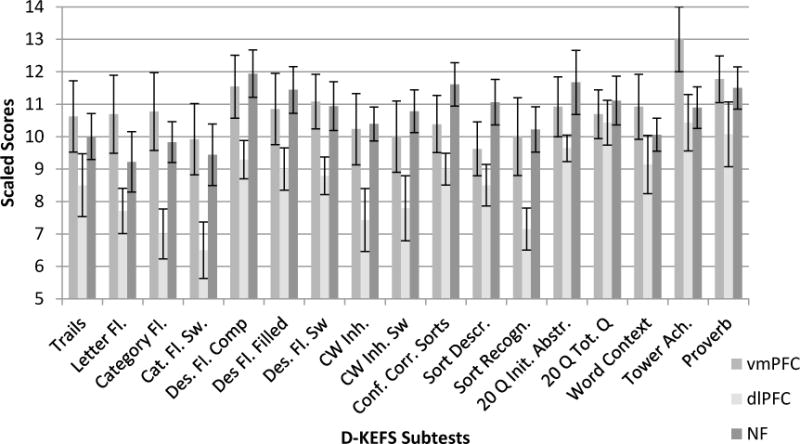
Mean D-KEFS performances in Scaled Scores (error bars are SEMs)
D-KEFS performances controlling for FSIQ
FSIQ was not significantly different among the groups but to increase the specificity of findings to executive functions, ANCOVAs were performed with FSIQ as a covariate. This analysis is also important in light of recent findings that dlPFC contributes to overall intellectual function (Barbey et al., 2012). FSIQ-corrected group means and results of the analysis are presented in Table 4. There were no significant differences in D-KEFS scores between the groups when the effects of FSIQ were removed. The adjusted means of the vmPFC and NF groups declined slightly while the adjusted mean of the dlPFC lesion group increased slightly, thus eliminating significant differences. However, several of the effect size values remained in the medium-large range, suggesting that lesion location accounted for a notable amount of variance in the D-KEFS scores, even when controlling for FSIQ. The observed power values suggest that this analysis did not have sufficient power to detect significant effects if they exist, which is likely the result of a small sample size. The results are promising, however, that with a larger sample size and sufficient power, group differences may be detected.
Since FSIQ is a broad construct consisting of a variety of skills, we considered the effects of individual WAIS Index scores on D-KEFS performances. When controlling for both VCI and PRI, only one D-KEFS group difference remained significant (Letter Fluency). When controlling for the WMI alone, three D-KEFS group differences remained significant (Color-Word Inhibition, Confirmed Correct Sorts, and Free Sorting Description score). When controlling for the PSI alone, all significant differences on the D-KEFS disappeared. These analyses suggest that all of the elements underlying FSIQ play a role in D-KEFS performances in the current sample. This is particularly true for processing speed since it was the only individual index score that eliminated all significant differences in the D-KEFS group performances. Based on this, it can be concluded that the broad concept of FSIQ, and processing speed in particular, can explain the significant D-KEFS group differences. We also controlled for WRAT Word Reading score and found that six D-KEFS group differences remained significant (Category Fluency, Design Fluency Composite, Color-Word Inhibition, Color-Word Inhibition/Switching, Confirmed Correct Sorts in the Free Sorting condition, Sort Description, and Sort Recognition. This suggests that WRAT Word Reading scores played a much smaller role in explaining group D-KEFS differences compared to IQ indices.
Measures with Extra Processing Demands
The D-KEFS includes several measures with increased processing demands. Examples include the switching conditions on Category Fluency, Design Fluency, and Color-Word Interference tests. These measures are theoretically more challenging and more sensitive to subtle dysfunction (D-KEFS Technical Manual; Delis, Kaplan, & Kramer, 2001). We evaluated whether these measures provide additional information beyond their more traditional counterparts by conducting paired samples t-tests for the following measures: Category Fluency and Category Fluency Switching, Design Fluency Filled Dots and Design Fluency Switching, and Color-Word Inhibition and Color-Word Inhibition/Switching conditions. Results are presented in Table 5. There were no significant differences in performances on measures with extra processing demands and more traditional measures among the entire sample or each individual group.
Table 5.
Measures with extra processing demands
| Cat. Fluency/ Cat. Fluency Switch |
Des. Fluency-Filled/ Des. Fluency Switch |
C-W Inhibition/ C-W Switching |
||||
|---|---|---|---|---|---|---|
| All Pts | t = 1.13 | p = .27 | t = .57 | p = .57 | t = −.72 | p = .48 |
| vmPFC | t = 1.06 | p = .31 | t = −.31 | p = .76 | t = .46 | p = .66 |
| dlPFC | t = .48 | p = .64 | t = .46 | p = .66 | t = −.81 | p = .43 |
| NF | t = .50 | p = .62 | t = .83 | p = .42 | t = −.79 | p = .44 |
DISCUSSION
We investigated performances of patients with vmPFC, dlPFC, and NF lesions on select primary measures from all nine of the D-KEFS subtests. Participants in each group did not significantly differ with regard to age, education, lesion chronicity, FSIQ and more specific WAIS index scores, or WRAT Word Reading scores. Without controlling for FSIQ, results revealed significant group differences on nine variables, including Category Fluency, Category Fluency Switching, Design Fluency Composite, Design Fluency Switching, Color-Word Inhibition, Color-Word Inhibition/Switching, and three variables on the Sorting Test (Confirmed Correct Sorts, Sort Description, and Sort Recognition). On most of these measures, with the exception of Color-Word Inhibition/Switching, Confirmed Correct Sorts, and Sort Description, participants with dlPFC lesions performed significantly worse than the other two groups. On Color-Word Inhibition/Switching, Confirmed Correct Sorts, and Sort Description, participants with dlPFC lesions performed lower only compared to the NF group. However, performances of participants with vmPFC and NF lesions were not significantly different on any of the considered measures, again highlighting the elusive nature of deficits associated with vmPFC lesions. In the current study, there were several patients, including the famous patient EVR (Eslinger & Damasio, 1985), who had clear difficulties with social behavior and emotional regulation in their everyday life, but performed well on the D-KEFS.
In a subsequent analysis, we controlled for FSIQ to increase the specificity of the findings to executive functions and account for recent findings about the contribution of the dlPFC to overall intelligence (Barbey et al., 2012). Unlike the Barbey et al. (2012) study, we did not extract the general intelligence factor (g) but used the psychometric intelligence estimate of FSIQ. With the effects of FSIQ removed, there were no significant differences in group performances on any of the D-KEFS subtests, suggesting that FSIQ explains a significant amount of variance in D-KEFS performances. In addition, we considered the effects of WAIS index scores since FSIQ is a multi-faceted concept. The results of these analyses revealed that, in addition to the broad concept of FSIQ, processing speed is the strongest underlying factor explaining D-KEFS group differences. Our results are generally consistent with Barbey et al.’s (2012) findings, where performance differences on the D-KEFS were negated when the effects of overall intellectual function (g) were removed. The authors concluded that damage to the dlPFC “leads to disproportionate defects in intelligence” and “does not lead to deficits in executive function” (p. 7). We cannot confirm that assertion based on our study as the differences in FSIQ were not statistically significant among the groups post-lesion, although FSIQ was qualitatively lower in the dlPFC group. In addition, we looked at a premorbid estimate of intellectual function (WRAT Word Reading scores), which were also not significantly different among the groups. In fact, the mean group WRAT Word Reading scores and the post-lesion FSIQ scores were similar in the dlPFC group (97.57 and 95.71, respectively). In addition, previous research and reported cases of patients with frontal lobe damage suggest that the reduction in psychometric IQ after frontal lobe lesions is relatively small (Tranel, Anderson, & Manzel, 2008). It remains possible that lesions in certain locations result in disproportionate decreases in the psychometrically-derived concept of general intelligence (g). It is interesting to contemplate what we would know about the association between specific functions and brain regions if all neuropsychological studies controlled for comprehensively-assessed FSIQ or general intelligence (g).
However, there may be other explanations to consider regarding the role of dlPFC in intelligence and specific executive functions. Both Barbey et al. (2012) and our study used the D-KEFS to assess executive functions. It is possible the D-KEFS is not a particularly sensitive measure of frontal lobe damage. In our sample, the range of scores in all groups was limited with few scores falling in the impaired range and in the Barbey et al. (2012) sample, the combined executive function variable based on the D-KEFS was a standard score of 91.31. By lowering floor effects and raising ceiling effects, the D-KEFS may not have an adequate range of items of medium difficulty that would increase the discrimination power of its measures. It is possible that increasing task difficulty has also made the D-KEFS, or at least some subtests on the D-KEFS, mainly a measure of aptitude rather than impairment. As a result, most individuals achieve scores in the average range even after completing few items of the test correctly. Further examination of this issue is critical to determine the clinical utility of the D-KEFS. A way to partially resolve this would be to compare the performance of the same participants on the D-KEFS and on the more frequently used older measures, from which the D-KEFS was derived.
From a clinical perspective, another important finding was the lack of significant differences in performances on measures with additional processing demands (Category Switching, Design Fluency Switching, and Color-Word Inhibition/Switching) and their more traditional counterparts (Category Fluency, Design Fluency-Filled Dots, and Color-Word Inhibition). This finding suggests that the D-KEFS measures with additional processing demands may not provide a significant improvement in detecting subtle dysfunction over more traditional measures, which raises concerns about their clinical utility. This is consistent with Lippa and Davis’ (2010) findings showing that the majority of their patients (57.1 percent) performed better on the D-KEFS Color-Word Inhibition/Switching trial compared to the Inhibition only trial with regard to time or errors. In our study, 21.7 percent of all participants obtained the same of faster time on the Inhibition/Switching subtest compared to the Inhibition only subtest, suggesting that for a relatively large proportion of patients, the Switching portion of the D-KEFS Color-Word does not function as a more demanding test, as originally intended.
The current investigation showed that combining individuals with frontal lobe lesions in one group regardless of the specific lesion location may result in a loss of valuable information. For example, if scores of the vmPFC and dlPFC lesion groups for the D-KEFS Category Fluency test (10.77 and 7.00, respectively) were combined, they would create an average of 8.89, which is less meaningful. This observation highlights the importance of considering lesions in different areas of the prefrontal cortex separately.
Several limitations need to be considered when interpreting the results of the current investigation. The most serious limitation is a relatively small sample size. Most lesion studies face this limitation due to the sparse number of available participants with well-characterized lesions restricted to a particular brain region. In this regard, the sample size of the current study is comparable to sample sizes used in other lesion studies. However, due to the small sample size, the study did not have sufficient power to detect true group differences if they are present. There were relatively large effect sizes for many of the measures, even after controlling for FSIQ, which is a promising finding for future investigations with larger sample sizes and adequate power. Therefore, results of this study should be interpreted cautiously and considered as preliminary. However, they provide valuable guidance for future studies. Other limitations included the lack of racial diversity in the sample and the consideration of the effects of age and sex. A limited sample of the D-KEFS measures was considered. It is possible that other indices (e.g., error rates, etc.) would be more sensitive/specific to damage in different regions of the prefrontal cortex. The value and clinical utility of these variables remains to be examined.
It is important to highlight that this investigation was guided primarily by the lesion method. There are other avenues for studying brain-behavior relationships, such as the associationist theories (for a review, please refer to Catani et al., 2012), which rely more heavily on the analysis of network disruption. While Map-3 lesion mapping considers white matter damage (as shown in figures 1 and 2), disruption of particular white matter tracts was not explicitly addressed in this study. This may be a fruitful direction for future research.
Finally, individuals included in the Patient Registry commonly participate in research projects and it is possible that practice effects played a role in performances on some of the D-KEFS measures derived from other commonly-used measures of executive functioning. There is limited literature on the magnitude of practice effects in patients with different brain lesions. It is possible that patients with lesions in particular locations benefit less from repeated test administration. For example, Waters-Wood, Xiao, Denburg, Hernandez and Bechara (2012) found that participants with vmPFC lesions did not show significant practice effects on the IGT compared to a non-neurological control group. There is also preliminary evidence to suggest that patients with frontal lobe lesions show more variability on re-testing (Murtha, Cismaru, Waechter, & Chertkow, 2002; Stuss, Murphy, Binns, & Alexander, 2003) but more research is needed to investigate this issue. Previous research indicates that there are multiple factors influencing the magnitude of practice effects, some of which include IQ, age, education, type of test used, patient population, and time elapsed since the previous administration (Calamia, Markon, & Tranel, 2012). Age, IQ, and level of education were not significantly different among the groups in this study, which would help minimize differences in practice effects among the groups. Also, the participants were exposed to the D-KEFS for the first time in this study, which also likely helped minimize practice effects on at least some of the tests with similar underlying constructs but different assessment methods (e.g., WCST and D-KEFS Sorting Test). However, a more in-depth investigation of possible practice effects was not conducted as a part of this study and, generally, more research is needed to investigate practice effects in different populations.
With regard to clinical implications, the current study provides preliminary information about performances of patients with vmPFC, dlPFC, and NF lesions on the primary measures of the D-KEFS. Qualitatively, there were sizeable and clinically-meaningful differences between the groups, some approaching and exceeding one standard deviation. These differences were generally consistent with previous research, suggesting that patients with dlPFC lesions perform worse on common executive function measures compared to patients with vmPFC and NF lesions. However, significant group differences disappeared when controlling for FSIQ with processing speed emerging as the most significant underlying factor eliminating all significant differences. It is important for neuropsychologists to be aware of emerging findings about the non-specific nature of reduced executive function performances (at least on the D-KEFS) with dlPFC lesions. While some of the D-KEFS tests are showing promise in measuring executive dysfunction associated with frontal lobe damage, for many subtests, the range of scores was somewhat limited, raising questions about the sensitivity of the measures. This continues to call into question whether the D-KEFS is a worthy investment of time and provides superior information compared to more traditional and often simpler counterparts (see Lezak et al., 2012). Finally, the current study also highlights the continued difficulty of detecting vmPFC dysfunction with traditional executive function measures and the importance of a careful clinical interview, collateral information, and observation to detect potential changes in aspects of executive functioning that are not easily captured by existing tests.
Acknowledgments
The study was supported by NINDS P01 NS19632 to D.T. We thank Joel Bruss for his help with figures.
Contributor Information
Ekaterina Keifer, Email: katya.keifer@gmail.com, Neurobehavioral Associates, P.C., 10730 W 143rd Avenue, Suite 37, Orland Park, IL 60462, (319) 321-5794.
Daniel Tranel, Email: daniel-tranel@uiowa.edu, Departments of Neurology and Psychology, 2206 RCP, University of Iowa, Iowa City, IA 52242, (319) 384-6050.
References
- Baldo JV, Delis DC, Wilkins DP, Shimamura AP. Is it bigger than a breadbox? Performance of patients with prefrontal lesions on a new executive function test. Archives of Clinical Neuropsychology. 2004;19:407–419. doi: 10.1016/S0887-6177(03)00074-X. [DOI] [PubMed] [Google Scholar]
- Baldo JV, Shimamura AP, Delis DC. Verbal and design fluency in patients with frontal lobe lesions. Journal of the International Neuropsychological Society. 2001;7:586–596. doi: 10.1017/s1355617701755063. [DOI] [PubMed] [Google Scholar]
- Barbey AK, Colom R, Grafman J. Dorsolateral prefrontal contributions to human intelligence. Neuropsychologia. 2012 doi: 10.1016/j.neuropsychologia.2012.05.017. http://dx.doi.org/10.1016/j.neuropsychologia.2012.05.017. [DOI] [PMC free article] [PubMed]
- Barbey AK, Colom R, Solomon J, Krueger F, Forbes C, Grafman J. An integrative architecture for general intelligence and executive functions revealed by lesion mapping. Brain. 2012;135 doi: 10.1093/brain/aws021. Epub, March 6th, 2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calamia M, Markon K, Tranel D. Scoring higher the second time around: Meta-analysis of practice effects in neuropsychological assessment. The Clinical Neuropsychologist. 2012;26:543–570. doi: 10.1080/13854046.2012.680913. [DOI] [PubMed] [Google Scholar]
- Catani M, Dell’Acqua F, Bizzi A, Forkel SJ, Williams SC, Simmons A, Murphy DG, Thiebaut de Schotten M. Beyond cortical localization in clinic- anatomical correlation. Cortex. 2012;48:1262–1287. doi: 10.1016/j.cortex.2012.07.001. [DOI] [PubMed] [Google Scholar]
- Cato MA, Delis DC, Abildskov TJ, Bigler E. Assessing the elusive cognitive deficits associated with ventromedial prefrontal damage: A case of modern-day Phineas Gage. Journal of the International Neuropsychological Society. 2004;10:453–465. doi: 10.1017/S1355617704103123. [DOI] [PubMed] [Google Scholar]
- Chan RCK, Shum D, Toulopoulou T, Chen EYH. Assessment of executive functions: Review of instruments and identification of critical issues. Archives of Clinical Neuropsychology. 2008;23:201–216. doi: 10.1016/j.acn.2007.08.010. [DOI] [PubMed] [Google Scholar]
- Crawford JR, Sutherland DM, Garthwaite PH. On the reliability and standard errors of measurement of contrast measures from the D-KEFS. Journal of the International Neuropsychological Society. 2008;14:1069–1073. doi: 10.1017/S1355617708081228. [DOI] [PubMed] [Google Scholar]
- Damasio AR, Anderson SW, Tranel D. The frontal lobes. In: Heilman KM, Valenstein E, editors. Clinical Neuropsychology. 5. New York: Oxford University Press Inc; 2012. [Google Scholar]
- Damasio H, Frank R. Three-dimensional in vivo mapping of brain lesions in humans. Archives of Neurology. 1992;49:137–143. doi: 10.1001/archneur.1992.00530260037016. [DOI] [PubMed] [Google Scholar]
- Delis DC, Kaplan E, Kramer JH. Delis-Kaplan Executive Function System:Technical Manual. San Antonio, TX: Harcourt Assessment Company; 2001. [Google Scholar]
- Eslinger PJ, Damasio AR. Severe disturbance in higher cognition after bilateral frontal lobe ablation: Patient EVR. Neurology. 1985;35:1731–1741. doi: 10.1212/wnl.35.12.1731. [DOI] [PubMed] [Google Scholar]
- Frank R, Damasio H, Grabowski TJ. Brainvox: An interactive, multimodal visualization and analysis system for neuroanatomical imaging. NeuroImage. 1997;5:13–30. doi: 10.1006/nimg.1996.0250. [DOI] [PubMed] [Google Scholar]
- Gläscher J, Adolphs R, Damasio H, Bechara A, Rudrauf D, Calamia M, Paul LK, Tranel D. Lesion mapping of cognitive control and value-based decision making in the prefrontal cortex. Proceedings of the National Academy of Sciences. 2012;109:14681–14686. doi: 10.1073/pnas.1206608109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gläscher J, Tranel D, Paul LK, Rudrauf D, Rorden C, Hornaday A, Grabowski, Adolphs R. Lesion mapping of cognitive abilities linked to intelligence. Neuron. 2009;61:681–691. doi: 10.1016/j.neuron.2009.01.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keil K, Baldo J, Kaplan E, Kramer J, Delis DC. Role of frontal cortex in inferential reasoning: Evidence from the Word Context Test. Journal of International Neuropsychological Society. 2005;11:426–433. [PubMed] [Google Scholar]
- Lezak MD, Howieson D, Bigler E, Tranel D. Neuropsychological assessment. 5. New York: Oxford University Press; 2012. [Google Scholar]
- Lippa SM, Davis RN. Inhibition/switching is not necessarily harder than inhibition: An analysis of the D-KEFS color-word interference test. Archives of Clinical Neuropsychology. 2010;25:146–152. doi: 10.1093/arclin/acq001. [DOI] [PubMed] [Google Scholar]
- Murtha S, Cismaru R, Waechter R, Chertkow H. Increased variability accompanies frontal lobe damage in dementia. Journal of the International Neuropsychological Society. 2002;8:360–372. doi: 10.1017/s1355617702813170. [DOI] [PubMed] [Google Scholar]
- Strauss E, Sherman EMS, Spreen O. A Compendium of Neuropsychological Tests: Administration, Norms, and Commentary. 3. Oxford University Press Inc; New York: 2006. [Google Scholar]
- Stuss DT, Murphy KJ, Binns MA, Alexander MP. Staying on the job: the frontal lobes control individual performance variability. Brain. 2003;126:2363–23800. doi: 10.1093/brain/awg237. [DOI] [PubMed] [Google Scholar]
- Tranel D, Anderson SW, Benton A. Development of the concept of “executive function” and its relationship to the frontal lobes. In: Boller F, Grafman J, editors. Handbook of Neuropsychology. New York, NY: Elsevier Science; 1994. pp. 125–148. [Google Scholar]
- Tranel D, Anderson SW, Manzel K. Is the prefrontal cortex important for fluid intelligence? A neuropsychological study using Matrix Reasoning. The Clinical Neuropsychologist. 2008;22:242–261. doi: 10.1080/13854040701218410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waters-Wood SM, Xiao L, Denburg NL, Hernandez M, Bechara A. Failure to learn from repeated mistakes: Persistent decision-making impairment as measured by the IGT in patients with ventromedial prefrontal cortex lesions. Journal of the International Neuropsychological Society. 2012;18:927–930. doi: 10.1017/S135561771200063X. [DOI] [PubMed] [Google Scholar]
- Yochim B, Baldo J, Kane K, Delis DC. D-KEFS Tower Test performance in patients with lateral prefrontal cortex lesions: The importance of error monitoring. Journal of Clinical and Experimental Neuropsychology. 2008:1–6. doi: 10.1080/13803390802448669. iFirst. [DOI] [PubMed] [Google Scholar]
- Yochim B, Baldo J, Nelson A, Delis DC. D-KEFS Trail Making Test performance in patients with lateral prefrontal cortex lesions. Journal of International Neuropsychological Society. 2007;13:704–709. doi: 10.1017/S1355617707070907. [DOI] [PubMed] [Google Scholar]
- Zald DH, Andreotti C. Neuropsychological assessment of the orbital and ventromedial prefrontal cortex. Neuropsychologia. 2010;48:3377–91. doi: 10.1016/j.neuropsychologia.2010.08.012. [DOI] [PubMed] [Google Scholar]


