Abstract
Purpose
To evaluate the association between longitudinal changes in quality of life and rates of progressive visual field loss in glaucoma.
Design
Prospective observational cohort study.
Participants
322 eyes of 161 patients with glaucomatous visual field loss recruited from the Diagnostic Innovations Glaucoma Study (DIGS) followed for an average of 3.5 ± 0.7 years.
Methods
All subjects had NEI VFQ-25 performed annually and standard automated perimetry (SAP) at 6-month intervals. Subjects were included if they had a minimum of 2 NEI VFQ-25 and at least 5 standard automated perimetry (SAP) during follow up. Evaluation of rates of visual field change was performed using the mean sensitivity (MS) of the integrated binocular visual field. Rasch analysis was performed to obtain final scores of disability as measured by the NEI VFQ-25. A joint longitudinal multivariate mixed model was used to investigate the association between change in NEI VFQ-25 Rasch-calibrated scores and change in binocular visual field sensitivity. Potentially confounding socio-economic and clinical variables were also analyzed.
Main outcome measures
The relationship between change in NEI VFQ-25 Rasch-calibrated scores and change in binocular SAP MS.
Results
There was a statistically significant correlation between change in the NEI VFQ-25 Rasch scores during follow-up and change in binocular SAP sensitivity. Each 1db change in binocular SAP MS per year was associated with a change of 2.9 units per year in the NEI VFQ-25 Rasch scores during the follow-up period (R2=26%; P<0.001). Eyes with more severe disease at baseline were also more likely to have a decrease in NEI VFQ-25 scores during follow-up (P<0.001). For subjects with the same amount of change in SAP sensitivity, those with shorter follow-up times had larger changes in NEI VFQ-25 scores (P=0.005). A multivariable model containing baseline and rate of change in binocular MS had an adjusted-R2 of 50% in predicting change in NEI VFQ-25 scores.
Conclusion
Baseline severity, magnitude and rates of change in binocular visual field sensitivity were associated with longitudinal changes in quality of life of glaucoma patients. Assessment of longitudinal visual field changes may help identify patients at higher risk for developing disability from the disease.
Keywords: Rate of visual field loss, quality of life, NEI VFQ-25, Rasch model and glaucoma
INTRODUCTION
Glaucoma is a leading cause of irreversible blindness and visual impairment. It is characterized by progressive optic nerve changes that may lead to loss of visual function and decrease in vision-related quality of life (VRQOL), including reduced vision-dependent mobility, increased risk of motor vehicle collisions, increased incidence of falls, and decreased ability to read or perform other daily life tasks.1–7 Loss of visual function in glaucoma is traditionally measured with standard automated perimetry (SAP); whereas the impact of the disease on VRQOL has frequently been investigated using patient-reported outcomes in the form of questionnaires, such as the National Eye Institute Visual Function Questionnaire (NEI VFQ-25).8–10
Previous studies investigating the relationship between glaucomatous visual field loss and the NEI VFQ-25 have been conducted using cross-sectional data.8, 10–14 Although significant relationships have been reported between SAP and NEI-VFQ-25, the strength of the association has been relatively weak. This can be, at least in part, due to a wide inter-individual variability in subjective perceptions about the impact of visual field loss on quality of life. The study of longitudinal changes in quality of life measures obtained over time may overcome such limitation. Assessing patient-reported outcomes at multiple time points may permit comparisons of quality of life over time in an individual and relate that to the degree of progressive visual field loss. As part of the Los Angeles Latino eye Study, McKean-Cowdin et al15 reported on longitudinal changes in visual acuity and NEI VFQ-25 results in a population-based sample. However, their study did not include a separate analysis of glaucoma patients nor of visual field losses over time as measured by SAP. The Collaborative Initial Glaucoma Treatment Study investigators reported on longitudinal changes in quality of life of glaucoma patients randomized to medical versus surgical treatment, but their analyses did bot include an evaluation of the relationship between changes in quality of life and rates of progressive visual field loss in the disease.16–18
Another reason for the imperfect relationship between the severity of visual field damage and patient-reported VRQOL may be the development of compensatory strategies by affected patients.19 In a previous study, we demonstrated that subjects with a history of fast visual field progression were more likely to report lower quality of life compared to subjects with history of slow field progression.20 It is likely that in subjects with slower field progression there would be more time for development of compensatory strategies that would reduce the impact of field loss on quality of life. However, our previous study only included NEI VFQ-25 data measured at a single point in time at the end of the available follow-up, not allowing the evaluation of the relationship between longitudinal changes in quality of life and rates of visual field loss.
The purpose of this study was to investigate the relationship between longitudinal changes in quality of life and progressive visual field loss in glaucoma. We used Rasch models to analyze data on quality of life of a cohort of glaucoma patients followed over time with NEI VFQ-25 questionnaires and we investigated the relationship between change in quality of life and rates of visual field progression in the disease.
METHODS
This was an observational cohort study. Participants from this study were included in a prospective longitudinal study designed to evaluate optic nerve structure and visual function in glaucoma, the Diagnostic Innovations in Glaucoma Study [DIGS], conducted at the University of California San Diego (UCSD). All DIGS participants who fulfilled the criteria outlined below were included in the present investigation. Methodological details have been described previously.21 The University of California San Diego Human Subjects Committee approved all protocols, and methods adhered to the Declaration of Helsinki.
At each visit during follow-up, subjects underwent a comprehensive ophthalmic examination, including review of medical history, best corrected visual acuity (BCVA), slit-lamp biomicroscopy, intraocular pressure (IOP) measurement using Goldmann applanation tonometry, gonioscopy, dilated fundoscopy examination using a 78-diopter (D) lens, stereoscopic photographs of the optic nerves, and standard automated perimetry (SAP) with 24-2 Swedish Interactive Threshold Algorithm (Carl Zeiss Meditec, Inc., Dublin, CA). To be included subjects had to have spherical refraction less than ± 5.0 D, cylinder correction less than 3.0 D and open angle with gonioscopy. Subjects with coexisting retinal disease, uveitis or non-glaucomatous optic disc neuropathy were excluded from the study.
We included 322 eyes of 161 participants that had a diagnosis of glaucoma at the baseline visit. Eyes were classified as having glaucoma based on repeatable (at least 3 consecutive) abnormal visual field test results at baseline, defined as a pattern standard deviation (PSD) with P < 0.05, and/or glaucoma hemifield test (GHT) results outside normal limits. Glaucomatous eyes also had evidence of glaucomatous optic neuropathy on masked assessment of stereophotographs. A subject was considered to have glaucoma if glaucomatous visual field loss was present in at least one eye. One hundred and five (65%) subjects had bilateral visual field defects.
Subjects had NEI VFQ-25 and SAP visual field tests obtained over time. NEI VFQ-25 questionnaires were obtained annually, whereas SAP testing was obtained at 6-month intervals. For inclusion in the final data analysis, subjects were required to have had a minimum of 2 NEI VFQ-25 questionnaires and a minimum of 5 SAP tests over a minimum duration of 2 years. Data for this study were obtained during the period extending from June 2009 to April 2014.
Monocular and Binocular Visual Fields
Monocular SAP was performed using the 24-2 Swedish Interactive Threshold Algorithm at all visits during the follow-up period. Only reliable tests (≤ 33% fixation losses and false negatives, and ≤ 15% false positives) were included. In addition, all visual fields were evaluated by the UCSD Visual Field Assessment Center (VisFACT).22 Visual fields were reviewed and excluded in the presence of artifacts such as lid or rim artifacts, fatigue effects, inattention or inappropriate fixation. Visual fields were also reviewed for the presence of abnormalities that could indicate diseases other than glaucoma, such as homonymous hemianopia.
In order to evaluate binocular visual field loss, sensitivities of the monocular SAP threshold sensitivities of the right and left eyes were used to calculate an integrated BVF. The sensitivity for each point of the BVF was estimated using the binocular summation model described by Nelson-Quigg et al.23 The 24-2 stimulus presentation pattern consists of 54 points within the central 24° in a 6° grid bracketing the horizontal and vertical meridians. According to this model, the binocular sensitivity can be estimated using the following formula:
where Sr and Sl are the monocular threshold sensitivities for corresponding visual field locations of the right and left eyes, respectively. In order to calculate the binocular sensitivity from the formula above, light sensitivity had to be converted to a linear scale (apostilbs) and then converted back to logarithmic scale (decibels). Evaluation of rates of visual field change was performed using the mean sensitivity (MS) of the BVF. MS was calculated as the average of the BVF threshold sensitivities for the integrated field.
Demographic, Clinical and Socio-economic Variables
Socio-economic and clinical questionnaires were also administered to patients at the time of the baseline NEI VFQ-25. These questionnaires contained a survey about demographics, history of ocular and medical conditions, marital status, health insurance coverage, degree of education and income. As these variables could potentially affect patient perceptions about VRQOL, these variables were included as potentially confounding factors in the analysis of the relationship between change in the NEI VFQ-25 and progressive field loss. These variables were categorized for inclusion in the multivariable models as marital status (married [yes/no]), presence of health insurance (yes/no), degree of education (at least high school degree [yes/no]) and income (less than $25,000/year [yes/no]). For co-morbidities, we investigated the presence or history of the following conditions: diabetes mellitus, arthritis, high blood pressure, heart disease, depression, asthma and cancers. A simple summation score was used to create a comorbidity index.24
Visual acuity was measured during follow-up using an Early Treatment Diabetic Retinopathy (ETDRS) chart and logMAR measurements were used in the analyses. Change in visual acuity during follow-up was calculated as the difference between the logMAR visual acuity at the last follow-up visit and baseline visit for each eye. The eye with better visual acuity at baseline was considered as the “better” eye for the purpose of analysis of change in visual acuity and its relationship with NEI VFQ-25 results. History of glaucoma filtering surgery during follow-up was recorded.
During follow-up patients were treated at the discretion of the attending the ophthalmologist.
Rasch Analysis of NEI VFQ-25 questionnaire
VRQOL was evaluated using the NEI VFQ-25. This questionnaire was designed to assess the dimensions of self-reported vision-target health status that are relevant for subjects with chronic eye diseases.25, 26 The NEI VFQ-25 consists of a set of 25 questions representing 11 subscales plus an additional single-item general health rating question. The subscales are: general vision, near and distance vision activities, ocular pain, vision-related social function, vision-related role function, vision-related mental health, vision-related dependency, driving difficulties, color vision and peripheral vision. Each subscale consists of a minimum of 1 item and a maximum of 4 items.
Rasch analysis was performed to obtain final estimates of “person measures”, which can be used to express where each respondent falls on a linear scale representing the degree of impairment as measured by the NEI VFQ-25.27, 28 These Rasch-calibrated scores were computed and used for subsequent parametric statistical analyses. Rasch analysis was performed using Andrich rating-scale models to obtain the estimates of the required ability of each item, perceived ability of each subject, and the thresholds for each response category.29 Rasch analysis locates item difficulty and person ability on a logit (log odds) scale, which was rescaled to range from 0 to 100 (corresponding to a range from −1.02 to 6.87 in the original logit scale). Person and item measures were examined for fit to the Rasch model using infit and outfit item fit statistics. Rasch analysis was initially conducted using the questionnaire data from 416 patients with glaucoma from the DIGS cohort who had answered at least 1 questionnaire, but had otherwise identical inclusion/exclusion criteria as those outlined above. All patients had repeatable glaucomatous visual field defects with average MD of the worse and better eyes of −6.3dB and −2.5dB, respectively. MD in the better eye ranged from −28.4dB to 2.5dB. This larger dataset was initially used in order to obtain more reliable estimates of the Rasch parameters. The Rasch model was initially fit using only 1 questionnaire from each of the 416 patients. After model parameters were obtained, they were used to score the follow-up questionnaires of those subjects who had follow-up data available.
A criticism of the current version of the NEI VFQ-25 is that the questionnaire does not seem to be unidimensional, i.e., it does not appear measure a single latent construct.30–32 If the unidimensionality assumption is violated, it is not appropriate to report a single summary measure or Rasch-derived score derived from all items from all subscales. To test the hypothesis that the NEI VFQ-25 measures a single underlying construct, we initially conducted a principal components analysis (PCA) of the residuals33 (difference between the observed and expected responses). Data are considered unidimensional if most of the variance is explained by the principal component and there is no significant explanation of the residual variance by the contrasts to the principal component. In general, in order to be considered unidimensional, the variance of the principal component should be greater than 60%.33 Furthermore, the unexplained variance by the contrasts should be less than 2 Eingenvalue units.33 When applied to the questionnaire data from our glaucoma patients, the NEI VFQ-25 revealed evidence of multidimensionality. The variance explained by the principal component was 51%. The unexplained variance by the first contrast accounted for 2.4 Eigenvalue units. The items with high loadings on the first contrast belonged to the dependency (3 items), mental health (3 items) and role limitations (2 items). This finding is similar to those reported in previous studies applying this questionnaire to populations with visual disability. Marella and colleagues31 suggested that the dependency, mental health and role limitaitons items would belong to a separate socioemotional dimension, not directly related to visual functioning. Therefore, we excluded these items from the calculation of final Rasch scores. In addition, the 2 items belonging to the subscale of ocular pain were also excluded according to Marella et al, as they could not fit into the Rasch model. This is also an expected result as ocular pain would likely produce changes in quality of life that are not directly related to those produced by loss of vision from glaucoma. Therefore, the final Rasch model used the items belonging to the general vision, near vision, distance vision, peripheral vision, color vision and social functioning subscales. This final model showed no evidence of multidimensionality, with no contrasts exceeding 2.0 Eigenvalue units.
The fit of the Rasch model was evaluated with the infit and outfit statistics. Values between 0.7 and 1.3 are considerable acceptable for mean square values of infit and outfit.34 For our dataset, infit had a mean value of 0.99 and outfit had a mean value of 0.92, indicating good fit. Person reliability and item reliability indices were 0.88 and 0.98, respectively.
Statistical Analyses
After Rasch scores were calculated for each subject at each point in time, these scores were analyzed along with visual field data using a joint multivariate longitudinal linear mixed model.35 The model was used to evaluate the relationship between the two longitudinal measures obtained over time, i.e., the Rasch scores summarizing NEI VFQ-25 results and the SAP binocular visual field measures. In these models, the average evolution of a specific response is described using a linear function of time, and subject-specific deviations from this average evolution are introduced by random intercepts and random slopes, allowing for different baseline values and different rates of change for each patient. In a joint-modeling approach using mixed models, random-effects are assumed for each response process and the different processes are associated by imposing a joint multivariate distribution on the random effects. This approach has great flexibility and allows joining models for responses of different types and also with different number of observations during follow-up, as in our application. Details of the modeling process have been described before.35, 36 The joint multivariate mixed model allowed the investigation of how the two responses were associated over time, that is, how change in quality of life, as measured by the NEI VFQ-25 Rasch scores was associated with change in SAP.
We also investigated the relationship between change in NEI VFQ-25 scores and change in visual acuity (in logMAR). In addition, we evaluated the effect of potentially confounding socioeconomic and clinical variables on the relationship between change in NEI VFQ-25 scores and change in SAP in a multivariable linear regression model.
Statistical Analyses were performed using Winsteps 3.81.0 (Chicago, IL) and STATA v. 13 (StataCorp, College Station, Texas). The alpha level (type I error) was set at 0.05.
RESULTS
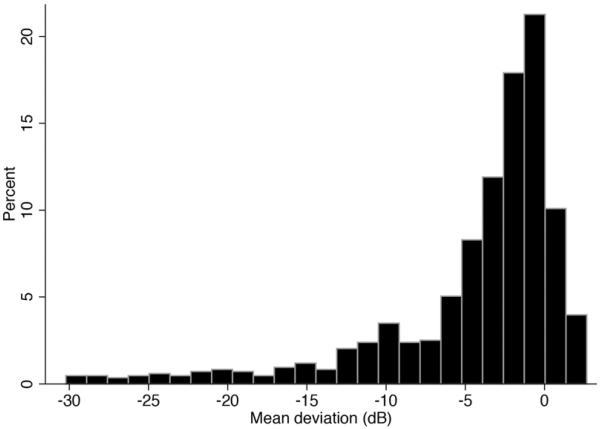
The study included 322 eyes of 161 subjects with glaucomatous visual field loss. Table 1 shows clinical and demographic characteristics of the included subjects. Mean age at baseline was 69.5 ± 10.7 years. There were 85 (53%) males and 76 (47%) females. One hundred fifteen subjects (71%) were White and 46 (29%) were Black. Seven (4%) of the 161 subjects identified themselves as Hispanics. At baseline, average MD of the worse and better eyes of glaucoma patients were −5.2 ± 5.6dB and −2.0 ± 3.6dB, respectively. However, there was a wide range of MD values at baseline for visual fields included in the study (Figure 1). The average binocular MS at baseline was 28.2 ± 3.6dB. Baseline binocular MS values were highly correlated to baseline MD values of the better eye (R2 = 90%).
Table 1.
Baseline clinical and demographic characteristics of subjects included in the study.
| Glaucoma patients (n = 161) | |
|---|---|
| Age, years | 69 ± 11 |
| Gender, % female | 47% |
| Race, % black | 29% |
| LogMAR visual acuity (better eye) | 0.01 ± 0.12 |
| Baseline MD (worse eye), dB | −5.2 ± 5.6 |
| Baseline MD (better eye), dB | −2.0 ± 3.6 |
| Baseline binocular mean sensitivity, dB | 28.2 ± 3.6 |
| Baseline NEI VFQ-25 Rasch-calibrated score | 53 ± 24 |
| History of glaucoma filtering surgery, % yes | 9% |
| Education level, % with at least high school degree | 93% |
| Income, lower than $25,000 | 9% |
| Marital status, % married | 53% |
| Comorbity index | 1.2 ± 1.1 |
| Insurance, % yes | 92% |
Values represent mean ± standard deviation, unless otherwise noted.
MD = mean deviation
dB = decibel
Figure 1.
Distribution of mean deviation values for all visual fields at baseline.
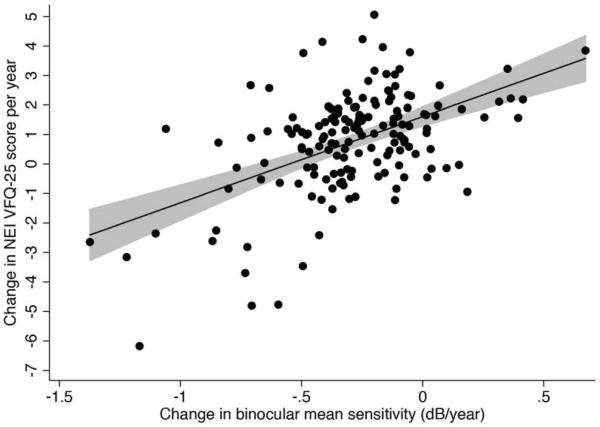
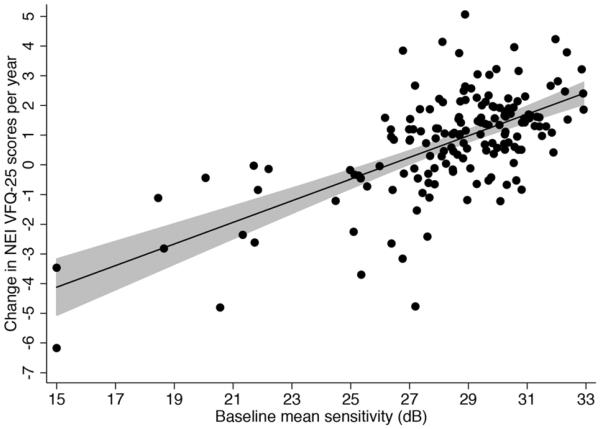
Subjects were followed for an average of 3.5 ± 0.7 years, ranging from 2.0 to 4.8 years, calculated from the date of the first questionnaire to the date of the last questionnaire administration. Subjects answered a median of 3 NEI VFQ-25 questionnaires during follow-up, ranging from 2 to 5. The median number of available SAP visual field tests during follow-up was 7 (IQR: 5 to 9). Table 2 shows results of univariable models investigating factors associated with change in NEI VFQ-25 Rasch scores over time. There was a statistically significant correlation between change in the NEI VFQ-25 Rasch scores during follow-up and change in binocular SAP sensitivity (Figure 2). Each 1db change in binocular SAP MS per year was associated with a change of 2.9 units per year in the NEI VFQ-25 Rasch scores during the follow-up period (R2=26%; P<0.001). Eyes with more severe disease at baseline were also more likely to have a decrease in NEI VFQ-25 scores during follow-up. Each 1dB lower baseline binocular SAP MS was associated with a 0.3 units greater decline per year in NEI VFQ-25 scores during follow-up (P<0.001) (Figure 3). A multivariable model containing baseline binocular MS and change in binocular SAP MS had an adjusted-R2 of 50% in predicting change in NEI VFQ-25 scores.
Table 2.
Results of the univariable and multivariable regression models for prediction of rates of change in NEI VFQ-25 scores.
| Univariable model | Multivariable model | |||
|---|---|---|---|---|
| Coefficient | P-value | Coefficient | P-value | |
| Rate of change in binocular mean sensitivity, per 1dB/year faster loss | −2.9 (−3.7 - −2.2) | <0.001 | −1.8 (−2.5– −1.1) | <0.001 |
| Baseline binocular sensitivity, per 1 dB lower | −0.3 (−0.4 - −0.3) | <0.001 | −0.3 (−0.3 – −0.2) | <0.001 |
| Change in visual acuity, per 0.1 LogMAR | −7.5 (−13.0 - −2.4) | 0.004 | −0.2 (−4.3 – 4.2) | 0.992 |
| History of glaucoma filtering surgey, yes | −1.2 (−2.1 - −0.2) | 0.013 | 0.1 (−0.6 – 0.8) | 0.854 |
| Age, per decade older | −0.5 (−0.7 – −0.2) | <0.001 | −0.1 (−0.3 – 0.1) | 0.450 |
| Gender, female | 0.2 (−0.4 – 0.7) | 0.537 | 0.2 (−0.2 – 0.6) | 0.355 |
| Ancestry, European descent | −0.8 (−1.4 – −0.2) | 0.006 | −0.4 (−0.8 – 0.1) | 0.158 |
| Educational level, at least high school degree | 0.2 (−0.8 – 1.3) | 0.665 | 0.5 (−0.4 – 1.3) | 0.287 |
| Income, lower than $25,000 | −0.5 (−1.5 – 0.4) | 0.258 | −0.3 (−1.0 – 0.4) | 0.454 |
| Marital status, married | 0.9 (−0.3 – 0.8) | 0.305 | 0.3 (−0.1 – 0.7) | 0.179 |
| Comorbity index | 0.0 (−0.2 – 0.2) | 0.994 | 0.1 (−0.1 – 0.3) | 0.302 |
| Insurance, yes | −0.5 (−1.4 - 0.4) | 0.296 | −0.2 (−1.0 – 0.6) | 0.698 |
dB = decibel
Figure 2.
Scatterplot with fitted regression line showing the relationship between change in NEI VFQ-25 scores and change in binocular mean sensitivity. The shaded area corresponds to the 95% confidence interval of the regression slope.
Figure 3.
Scatterplot with fitted regression line showing the relationship between change in NEI VFQ-25 scores and baseline disease severity as measured by binocular mean sensitivity. The shaded area corresponds to the 95% confidence interval of the regression slope.
When the change in NEI VFQ-25 scores was calculated over the entire follow-up period, i.e., the difference between measurements at the baseline and final visit, it showed a significant association with the total change in binocular MS (R2 = 15%; P<0.001). However, for subjects with the same amount of binocular SAP sensitivity loss over time, those with shorter follow-up times had larger changes in NEI VFQ-25 scores compared to those with longer follow-up times (P=0.005) (Figure 4). This indicates that not only the magnitude of SAP losses, but also the rate of SAP change were important in determining the amount of change in NEI VFQ-25 scores.
Figure 4.
The relationship between total change in NEI VFQ-25 scores during follow-up and total change in binocular mean sensitivity according to different durations of follow-up, as predicted by the model. For the same amount of change in binocular standard automated perimetry (SAP) mean sensitivity, subjects with shorter follow-up time had larger changes in NEI VFQ-25 scores.
Change in logMAR visual acuity during follow-up in the eye with better acuity at baseline was also associated with the rate of NEI VFQ-25 score change. Each 0.1 logMAR increase was associated with a decline of 7.5 units in the NEI VFQ-25 scores (R2 =5%; P = 0.004). Subjects with history of filtering surgery during follow-up had 1.2 units per year greater loss in NEI VFQ-25 scores (P=0.013). However, when adjusted for the rate of change in binocular SAP MS and baseline binocular MS, history of glaucoma surgery was not associated with change in quality of life (P=0.757).
Race was also significantly associated with change in NEI VFQ-25 scores over time in the univariable model. White subjects had 0.8 units/year faster decline in NEI VFQ-25 scores than Black subjects during the follow-up period (P=0.006). However, when adjusted for the rate of change in binocular SAP MS and baseline binocular MS, the effect of race was of borderline significance (coefficient = 0.4; P=0.067).
Older age was associated with larger decline in NEI VFQ-25 scores. Each 10 year older difference in baseline age was associated with a 0.5 units greater loss per year in NEI VFQ-25 scores during follow-up (R2 = 8%; P<0.001). There was no significant relationship between the socio-economic variables marital status, education, income and insurance and change in NEI VFQ-25 scores. There was also no significant relationship between the morbidity index and change in NEI VFQ-25 scores.
In the multivariable model adjusting for potentially confounding variables, both the rate of change in binocular MS and baseline binocular MS were still significantly associated with change in NEI VFQ-25 scores (Table 2).
DISCUSSION
In the present study, we showed that changes in binocular visual field sensitivity as assessed by standard perimetry were significantly associated with changes over time in patient-reported quality of life outcomes in glaucoma. Both baseline visual field severity as well as the magnitude of change in binocular sensitivity were associated with the magnitude of change in NEI VFQ-25 scores. In addition, the rate at which the binocular visual field sensitivity occurred was an important factor in determining the magnitude of change in quality of life measures over time. To our knowledge, this is the first study to describe the association between longitudinal changes in quality of life in glaucoma patients and rates of visual field loss. Our findings may have significant implications for predicting disability related to glaucoma.
In the univariable model, each 1dB of change in binocular MS was associated with an average change of 2.9 units in NEI VFQ-25 Rasch calibrated scores. However, the amount of baseline visual field damage was also an important factor influencing the impact of visual field change on quality of life. For subjects with more severe disease at baseline, changes in binocular field sensitivity produced larger changes in NEI VFQ-25 scores compared to subjects with less severe disease at baseline. For example, for a subject with a baseline mean sensitivity of 20dB, a 5 dB loss in binocular sensitivity carried out over a period of 5 years (i.e., at a rate of 1dB/year) would result in a drop of approximately 15 units in NEI VFQ-25 score whereas the same rate and amount of visual field loss for a subject with a baseline MS of 28dB would result in only approximately 4 units of decrease in NEI VFQ scores. To help understand these figures, it is helpful to know that baseline binocular MS values were highly correlated to baseline MD values of the better eye (R2 = 90%). Therefore, a binocular MS of 20dB would correspond approximately to an MD value in the better eye of −10dB (severe defect), whereas a binocular MS of 28dB would correspond to an MD in the better eye of approximately −2dB (mild defect). It is also helpful to know that Rasch scores were scaled to range from 0 to 100, from the worst reported QoL in the sample to the best one. Therefore, a 15-unit change in NEI VFQ-25 Rasch scores corresponded to about 15% change in a scale going from the worst reported quality of life in our sample to the best reported one. This finding is not surprising, as further field losses in subjects with an already compromised binocular visual field are likely to affect areas that are more relevant for performing daily activities and result in decrease in quality of life.
In addition to the magnitude of progressive visual field loss and the amount of baseline damage, the rate of change in binocular SAP sensitivity was an important factor in determining the impact of visual field losses on quality of life. This finding is in agreement with our previous observations.20 In a previous study, we showed that history of fast binocular SAP field loss was associated with worse VRQOL in glaucoma, as measured by the NEI VFQ-25. However, our previous observation was based on only one questionnaire obtained from each patient at the end of follow-up. In the current study, we were able to demonstrate a relationship between rate of visual field progression and decline in quality of life as assessed by longitudinal changes in NEI VFQ-25 scores, providing more robust evidence to our previous findings. For the same amount of field loss and baseline disease severity, subjects who had a faster rate of change had worse decline in NEI VFQ-25 scores. It is possible that patients with slowly progressing disease have more time to adapt to their limited functional status by developing compensatory strategies and, therefore, are less likely to report loss in quality of life. On the other hand, in patients with rapidly progressing disease, the visual field defects may result in substantial impairment in the ability to perform daily activities without time for development of compensatory strategies. This would be more readily recognized by such a patient and more likely to be reported as loss in quality of life over time. This finding underscores the importance of assessing and taking into account rates of change in the management of patients with glaucoma.
A limitation of previous studies evaluating the relationship between SAP and NEI VFQ-25 is derived from the method used to analyze the questionnaire data. Items in the questionnaire require the subject to provide a response based on a Likert scale. For example, to answer the question: “Because of your eyesight, how much difficulty do you have noticing objects off to the side while you are walking along?”, subjects are required to select a response from alternatives such as “No difficulty at all”, “A little difficulty”, “Moderate difficulty”, “Extreme difficulty” or “Stopped doing this because of your eyesight”. Methods for analyzing this type of data used in most previous studies attribute linear scores to each response such as 1, 2, 3, 4 and 5 and then sum up the scores for different items to obtain a composite score. In this case, “Stopped doing this because of your eyesight” is considered as 5 times worse than “little difficulty” and this relationship is assumed to be true for all the other items in the questionnaire that apply the same or corresponding scale. Whenever scores are added in this manner, the interval nature of the data is being presumed, that is, the relative value of each response category across all items is treated as being the same, and the unit increases across the rating scale are given equal value.37 However, there is no reason to believe or assume this to be the case. In fact, it is unlikely that the relative value of each response category will be the same for items that evaluate very different aspects of quality of life, such as driving, reading or walking. Rasch model allows the item difficulty of each question to be based on the way in which subjects responded to the questionnaire. Rasch modeling also establishes the pattern of use of the Likert scale to yield a rating scale structure, without making a priori assumption of linear interval data. Analysis derived from the Rasch model can provide final estimates of “person measures” which can then be used to express where each responded falls on linear scale and be used to compute parametric statistics as performed in our study.
It should be emphasize that although baseline disease severity and changes in visual field sensitivity were able to explain a large proportion of the changes in NEI VFQ-25 scores seen among glaucoma patients in our sample, there was still about 50% of unexplained variation in the outcome. That is, in some patients changes in quality of life did not occur as predicted by the severity or changes in the binocular visual field. In the present study, we did not evaluate the location of field defects as a factor related to change in QoL. It is possible that change in certain types or location of field defects may be more related to changes in quality of life and this should be the subject of future investigation. Patients' perceptions of quality of life may also differ substantially and may be affected by other variables not measured in the current study. In addition, the NEI VFQ-25 questionnaire provides a subjective assessment of quality of life. As a consequence, these questionnaires may not reflect the true impairment experienced by patients in daily activities. 38–40 The use of more objective tests in performance-based assessments could potentially provide a more accurate representation of the impairment experienced by glaucomatous patients on daily activities. 41, 42
We used estimated binocular visual field measures obtained from integrating results of monocular visual fields instead of “true” binocular visual fields. Although “true” binocular fields can be obtained using specific perimetric strategies, they are not routinely performed in clinical practice. Among the several different methods previously proposed for construction of the integrated binocular fields from monocular fields, we used the summation method as it has been shown to have a superior correlation with the “true” binocular field exam compared to other approaches.23 Although the methodology used in our study to obtain binocular mean sensitivity requires some mathematical calculations, it can be easily implemented into the standard software of visual field instruments in order to provide clinicians with information about change in the binocular field, which may help determine risk of disability. Previous cross-sectional studies have suggested that analysis of the better eye provides similar information than that obtained from analysis of binocular data.43 However, although baseline binocular sensitivity values were strongly correlated with MD values of the better eye at baseline, it is important to note that the definition of better eye becomes confusing when one evaluates longitudinal data. For example, the better eye at baseline may not remain as the better eye during follow-up depending on the amount of change that occurs in this eye compared to the other eye. For this reason, we used binocular metrics instead of simply the “better eye” as defined at baseline.
It is also important to note that the population of our study included only subjects with glaucomatous visual field loss in at least one eye. It is known that a significant proportion of glaucomatous subjects may present optic nerve damage despite absence of detectable visual field defects.44,45 Although it is likely that significant changes in quality of life of glaucomatous subjects would occur only in the presence of field defects, future studies are necessary to investigate this hypothesis. In addition, studies should investigate whether measures of structural damage could provide information besides what can be obtained from visual fields in predicting disability in glaucomatous subjects.
Our study has limitations. We had a relatively short follow-up time, considering the long-term duration of a disease such as glaucoma. However, despite the relatively short follow-up, the associations were strongly significant and our results demonstrated the feasibility of measuring change over time in quality of life of glaucoma patients. Our results should motivate future investigations over longer periods of follow-up to help clarify the full time course of changes in quality of life and factors associated with it. Another limitation of our study is that we assumed linear rates of visual field and quality of life change over time. Several studies have suggested that functional changes may not follow a linear course over the natural history of the disease.46,47 Nevertheless, the assumption of linear change is probably a reasonable one for short and medium follow-up periods, as performed in clinical practice. Systematic lens opacity grading was not available during follow-up in order to directly investigate the possible effect of changes in lens opacity in the visual field and quality of life questionnaires. However, we expect that the influence of cataract in our results would be minimized by the analysis of change in visual acuity as a confounding variable, as performed in the study. We also did not investigate change in socio-economic variables as potential predictors of change in disability. We believe that it is unlikely that significant changes in these variables would have occurred in the timeframe of the study. Furthermore, although it is possible that changes in some of the socio-economic variables may be related to change in disability for some subjects, it is expected that this would weaken rather than make stronger the relationship between change in QoL and change in visual field found in our study. Another potentially important confounding variable is cognitive status, which was not investigated in our study. Cognitive decline over time could potentially lead to worse reported QoL and worse performance on visual field tests. Although it is unlikely that a significant proportion of our subjects would have suffered from significant decline to influence the results, future studies should investigate this issue. Finally, it could be argued that some of our patients reported worsening in quality of life simply due to the fact that they became aware of a worsening visual field result as possibly informed by the treating physician. Future investigations using performance-based measures of disability should be able to clarify this issue.
In conclusion, baseline severity, magnitude and rates of change in binocular visual field sensitivity were associated with longitudinal changes in quality of life of glaucoma patients. Assessment of longitudinal visual field changes may help identify patients at higher risk for developing disability from the disease.
Acknowledgments
Financial Disclosure(s): Research support from Carl-Zeiss Meditec (FAM, RNW, LMZ). Research support from Heidelberg Engineering (FAM, RNW, LMZ). Consultant to Carl-Zeiss Meditec, Inc. (FAM, RNW). None: CPBG, EB, PR.
Funding: Supported in part by National Institutes of Health/National Eye Institute grants EY021818 (F.A.M.), EY11008 (L.M.Z.), EY14267 (L.M.Z.), EY019869 (L.M.Z.) and core grant P30EY022589; Unrestricted grant from Research to Prevent Blindness (RPB); Brazilian National Research Council-CAPES grant 12309-13-3 (CPBG); grants for participants' glaucoma medications from Alcon, Allergan, Pfizer, Merck and Santen.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
REFERENCES
- 1.Ramulu P. Glaucoma and disability: which tasks are affected, and at what stage of disease? Curr Opin Ophthalmol. 2009;20:92–8. doi: 10.1097/ICU.0b013e32832401a9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Haymes SA, LeBlanc RP, Nicolela MT, et al. Glaucoma and on-road driving performance. Invest Ophthalmol Vis Sci. 2008;49:3035–41. doi: 10.1167/iovs.07-1609. [DOI] [PubMed] [Google Scholar]
- 3.Owsley C, McGwin G., Jr Vision and driving. Vision Res. 2010;52:2348–61. doi: 10.1016/j.visres.2010.05.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ramulu PY, West SK, Munoz B, et al. Glaucoma and reading speed: the Salisbury Eye Evaluation project. Arch Ophthalmol. 2009;127:82–7. doi: 10.1001/archophthalmol.2008.523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Haymes SA, Leblanc RP, Nicolela MT, et al. Risk of falls and motor vehicle collisions in glaucoma. Invest Ophthalmol Vis Sci. 2007;48:1149–55. doi: 10.1167/iovs.06-0886. [DOI] [PubMed] [Google Scholar]
- 6.Skalicky SE, Goldberg I. Are we ready to assess quality of life routinely in our glaucoma patients? Bull Soc Belge Ophtalmol. 2010;(315):5–7. [PubMed] [Google Scholar]
- 7.Goldberg I, Clement CI, Chiang TH, et al. Assessing quality of life in patients with glaucoma using the Glaucoma Quality of Life-15 (GQL-15) questionnaire. J Glaucoma. 2009;18:6–12. doi: 10.1097/IJG.0b013e3181752c83. [DOI] [PubMed] [Google Scholar]
- 8.McKean-Cowdin R, Wang Y, Wu J, et al. Los Angeles Latino Eye Study Group. Impact of visual field loss on health-related quality of life in glaucoma: the Los Angeles Latino Eye Study. Ophthalmology. 2008;115:941–8. doi: 10.1016/j.ophtha.2007.08.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Spaeth G, Walt J, Keener J. Evaluation of quality of life for patients with glaucoma. Am J Ophthalmol. 2006;141(suppl):S3–14. doi: 10.1016/j.ajo.2005.07.075. [DOI] [PubMed] [Google Scholar]
- 10.Jampel HD. Glaucoma patients' assessment of their visual function and quality of life. Trans Am Ophthalmol Soc. 2001;99:301–17. [PMC free article] [PubMed] [Google Scholar]
- 11.van Gestel A, Webers CA, Beckers HJ, et al. The relationship between visual field loss in glaucoma and health-related quality-of-life. Eye (Lond) 2010;24:1759–69. doi: 10.1038/eye.2010.133. [DOI] [PubMed] [Google Scholar]
- 12.Gutierrez P, Wilson MR, Johnson C, et al. Influence of glaucomatous visual field loss on health-related quality of life. Arch Ophthalmol. 1997;115:777–84. doi: 10.1001/archopht.1997.01100150779014. [DOI] [PubMed] [Google Scholar]
- 13.Parrish RK, II, Gedde SJ, Scott IU, et al. Visual function and quality of life among patients with glaucoma. Arch Ophthalmol. 1997;115:1447–55. doi: 10.1001/archopht.1997.01100160617016. [DOI] [PubMed] [Google Scholar]
- 14.McKean-Cowdin R, Varma R, Wu J, et al. Los Angeles Latino Eye Study Group. Severity of visual field loss and health-related quality of life. Am J Ophthalmol. 2007;143:1013–23. doi: 10.1016/j.ajo.2007.02.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.McKean-Cowdin R, Varma R, Hays RD, et al. Los Angeles Latino Eye Study Group. Longitudinal changes in visual acuity and health-related quality of life: the Los Angeles Latino Eye study. Ophthalmology. 2010;117:1900–7. doi: 10.1016/j.ophtha.2010.01.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mills RP, Janz NK, Wren PA, Guire KE. CIGTS Study Group. Correlation of visual field with quality-of-life measures at diagnosis in the Collaborative Initial Glaucoma Treatment Study (CIGTS) J Glaucoma. 2001;10:192–8. doi: 10.1097/00061198-200106000-00008. [DOI] [PubMed] [Google Scholar]
- 17.Janz NK, Wren PA, Lichter PR, et al. CIGTS Study Group. The Collaborative Initial Glaucoma Treatment Study: interim quality of life findings after initial medical or surgical treatment of glaucoma. Ophthalmology. 2001;108:1954–65. doi: 10.1016/s0161-6420(01)00874-0. [DOI] [PubMed] [Google Scholar]
- 18.Janz NK, Wren PA, Lichter PR, et al. CIGTS Group. Quality of life in newly diagnosed glaucoma patients: the Collaborative Initial Glaucoma Treatment Study. Ophthalmology. 2001;108:887–97. doi: 10.1016/s0161-6420(00)00624-2. discussion 898. [DOI] [PubMed] [Google Scholar]
- 19.Huxlin KR. Perceptual plasticity in damaged adult visual systems. Vision Res. 2008;48:2154–66. doi: 10.1016/j.visres.2008.05.022. [DOI] [PubMed] [Google Scholar]
- 20.Lisboa R, Chun YS, Zangwill LM, et al. Association between rates of binocular visual field loss and vision-related quality of life in patients with glaucoma. JAMA Ophthalmol. 2013;131:486–94. doi: 10.1001/jamaophthalmol.2013.2602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Racette L, Liebmann JM, Girkin CA, et al. ADAGES Group. African Descent and Glaucoma Evaluation Study (ADAGES): III. Ancestry differences in visual function in healthy eyes. Arch Ophthalmol. 2010;128:551–9. doi: 10.1001/archophthalmol.2010.58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Sample PA, Girkin CA, Zangwill LM, et al. ADAGES Study Group. The African Descent and Glaucoma Evaluation Study (ADAGES): design and baseline data. Arch Ophthalmol. 2009;127:1136–45. doi: 10.1001/archophthalmol.2009.187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Nelson-Quigg JM, Cello K, Johnson CA. Predicting binocular visual field sensitivity from monocular visual field results. Invest Ophthalmol Vis Sci. 2000;41:2212–21. [PubMed] [Google Scholar]
- 24.Globe DR, Varma R, Torres M, et al. Los Angeles Latino Eye Study Group. Self-reported comorbidities and visual function in a population-based study: the Los Angeles Latino Eye Study. Arch Ophthalmol. 2005;123:815–21. doi: 10.1001/archopht.123.6.815. [DOI] [PubMed] [Google Scholar]
- 25.Mangione CM, Lee PP, Gutierrez PR, et al. National Eye Institute Visual Function Questionnaire Field Test Investigators. Development of the 25-item National Eye Institute Visual Function Questionnaire. Arch Ophthalmol. 2001;119:1050–8. doi: 10.1001/archopht.119.7.1050. [DOI] [PubMed] [Google Scholar]
- 26.Mangione CM, Lee PP, Pitts J, et al. NEI-VFQ Field Test Investigators. Psychometric properties of the National Eye Institute Visual Function Questionnaire (NEI-VFQ) Arch Ophthalmol. 1998;116:1496–504. doi: 10.1001/archopht.116.11.1496. [DOI] [PubMed] [Google Scholar]
- 27.Boone WJ, Staver JR, Yeale MS. Rasch Analysis in the Human Sciences. Springer; New York: 2014. pp. 69–91. [Google Scholar]
- 28.Bond TG, Fox CM. Applying the Rasch Model: Fundamental Measurement in the Human Sciences. Taylor & Francis; New York: 2007. pp. 29–48. [Google Scholar]
- 29.Andrich D. Rating scales and Rasch measurement. Expert Rev Pharmacoecon Outcomes Res. 2011;11:571–85. doi: 10.1586/erp.11.59. [DOI] [PubMed] [Google Scholar]
- 30.Massof RW, Fletcher DC. Evaluation of the NEI visual functioning questionnaire as an interval measure of visual ability in low vision. Vision Res. 2001;41:397–413. doi: 10.1016/s0042-6989(00)00249-2. [DOI] [PubMed] [Google Scholar]
- 31.Marella M, Pesudovs K, Keeffe JE, et al. The psychometric validity of the NEI VFQ-25 for use in a low-vision population. Invest Ophthalmol Vis Sci. 2010;51:2878–84. doi: 10.1167/iovs.09-4494. [DOI] [PubMed] [Google Scholar]
- 32.Pesudovs K, Gothwal VK, Wright T, Lamoureux EL. Remediating serious flaws in the National Eye Institute Visual Function Questionnaire. J Cataract Refract Surg. 2010;36:718–32. doi: 10.1016/j.jcrs.2009.11.019. [DOI] [PubMed] [Google Scholar]
- 33.Smith EV., Jr Detecting and evaluating the impact of multidimensionality using item fit statistics and principal component analysis of residuals. J Appl Meas. 2002;3:205–31. [PubMed] [Google Scholar]
- 34.Wright BD, Linacre JM, Gustafson JM, Martin-Lof P. [Accessed July 31, 2014];Reasonable mean-square fit values. Rasch Meas Trans. 1994 8(3):370. Available at: http://www.rasch.org/rmt/rmt83b.htm. [Google Scholar]
- 35.Beckett LA, Tancredi DJ, Wilson RS. Multivariate longitudinal models for complex change processes. Stat Med. 2004;23:231–9. doi: 10.1002/sim.1712. [DOI] [PubMed] [Google Scholar]
- 36.Medeiros FA, Zangwill LM, Alencar LM, et al. Rates of progressive retinal nerve fiber layer loss in glaucoma measured by scanning laser polarimetry. Am J Ophthalmol. 2010;149:908–15. doi: 10.1016/j.ajo.2010.01.010. [DOI] [PubMed] [Google Scholar]
- 37.Coste J, Fermanian J, Venot A. Methodological and statistical problems in the construction of composite measurement scales: a survey of six medical and epidemiological journals. Stat Med. 1995;14:331–45. doi: 10.1002/sim.4780140402. [DOI] [PubMed] [Google Scholar]
- 38.Rubenstein LZ, Schairer C, Wieland GD, Kane R. Systematic biases in functional status assessment of elderly adults: effects of different data sources. J Gerontol. 1984;39:686–91. doi: 10.1093/geronj/39.6.686. [DOI] [PubMed] [Google Scholar]
- 39.Kuriansky JB, Gurland BJ, Fleiss JL, Cowan D. The assessment of self-care capacity in geriatric psychiatric patients by objective and subjective methods. J Clin Psychol. 1976;32:95–102. doi: 10.1002/1097-4679(197601)32:1<95::aid-jclp2270320129>3.0.co;2-j. [DOI] [PubMed] [Google Scholar]
- 40.Kiyak HA, Teri L, Borson S. Physical and functional health assessment in normal aging and in Alzheimer's disease: self-reports vs family reports. Gerontologist. 1994;34:324–30. doi: 10.1093/geront/34.3.324. [DOI] [PubMed] [Google Scholar]
- 41.Medeiros FA, Weinreb RN, R Boer E, Rosen PN. Driving simulation as a performance-based test of visual impairment in glaucoma. J Glaucoma. 2012;21:221–7. doi: 10.1097/IJG.0b013e3182071832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lorenzana L, Lankaranian D, Dugar J, et al. A new method of assessing ability to perform activities of daily living: design, methods and baseline data. Ophthalmic Epidemiol. 2009;16:107–14. doi: 10.1080/09286580902738142. [DOI] [PubMed] [Google Scholar]
- 43.Arora KS, Boland MV, Friedman DS, et al. The relationship between better-eye and integrated visual field mean deviation and visual disability. Ophthalmology. 2013;120:2476–84. doi: 10.1016/j.ophtha.2013.07.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Medeiros FA, Alencar LM, Zangwill LM, et al. Prediction of functional loss in glaucoma from progressive optic disc damage. Arch Ophthalmol. 2009;127:1250–6. doi: 10.1001/archophthalmol.2009.276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Alencar LM, Zangwill LM, Weinreb RN, et al. Agreement for detecting glaucoma progression with the GDx guided progression analysis, automated perimetry, and optic disc photography. Ophthalmology. 2010;117:462–70. doi: 10.1016/j.ophtha.2009.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Hood DC, Kardon RH. A framework for comparing structural and functional measures of glaucomatous damage. Prog Retin Eye Res. 2007;26:688–710. doi: 10.1016/j.preteyeres.2007.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Medeiros FA, Zangwill LM, Bowd C, et al. The structure and function relationship in glaucoma: implications for detection of progression and measurement of rates of change. Invest Ophthalmol Vis Sci. 2012;53:6939–46. doi: 10.1167/iovs.12-10345. [DOI] [PMC free article] [PubMed] [Google Scholar]