Abstract
Apoptosis has been implicated in mediating denervation-induced muscle wasting. In this study we determined the effect of interference of apoptosis on muscle wasting during denervation by using mice genetically deficient in pro-apoptotic Bax. After denervation, muscle wasting was evident in both wild-type and Bax−/− muscles but reduction of muscle weight was attenuated in Bax−/− mice. Apoptotic DNA fragmentation increased in wild-type denervated muscles whereas there was no statistical increase in DNA fragmentation in denervated muscles from Bax−/− mice. Mitochondrial AIF and Smac/DIABLO releases and Bcl-2, p53 and HSP27 increased whereas XIAP and MnSOD decreased to a similar extent in muscles from wild-type and Bax−/− mice following denervation. Mitochondrial cytochrome c release was elevated in denervated muscles from wild-type mice but the increase was suppressed in muscles from Bax−/− mice. Increases in caspase-3 and -9 activities and oxidative stress markers H2O2, MDA/4-HAE and nitrotyrosine were all evident in denervated muscles from wild-type mice but these changes were absent in muscles from Bax−/− mice. Moreover, ARC increased exclusively in denervated Bax−/− muscle. Our data indicate that under conditions of denervation, pro-apoptotic signalling is suppressed and muscle wasting is attenuated when the Bax gene is lacking. These findings suggest that interventions targeting apoptosis may be valuable in ameliorating denervation-associated pathologic muscle wasting in certain neuromuscular disorders that involve partial or full denervation.
Keywords: apoptosis, BCL-2, muscle atrophy, neuromuscular disease
Introduction
Innervation is an essential constituent in the development and survival of mature skeletal muscle. Furthermore, innervation is important for conduction of normal physiologic functions, and in determining and maintaining muscle fibre volume as well as phenotype.1,2 Removal of the nerve source (i.e., denervation) is generally involved in the aetiology and the progression of various neuromuscular diseases. These include spinal muscular atrophy, peripheral neuropathies, and amyotrophic lateral sclerosis.3 Intriguingly, there is a growing body of evidence indicating that apoptosis may have a regulatory role in the denervation-associated pathologic muscle wasting.4–8 Nevertheless, the importance of apoptosis in mediating muscle wasting in response to denervation remains to be fully elucidated.
A cluster of upstream regulators of apoptosis, namely the BCL-2 family, has been proposed as a crucial intracellular checkpoint in the apoptotic signalling pathway.9 In general, mammalian cells possess an entire family of BCL-2 proteins. As defined by the homology shared within four conserved sequence motifs (BH1-4). The BCL-2 family is divided into three subclasses: (a) anti-apoptotic (e.g., Bcl-2, Bcl-XL, Bcl-W, A1, and Mcl-1), (b) multidomain pro-apoptotic (Bax, Bak, and Bok), and (c) BH3-only pro-apoptotic (Bad, Bid, Bim, Bik, Dp5/Hrk, Noxa, and Puma).9–11 The BH3 domain is thought to be fundamentally important for the pro-apoptotic interactions since all pro-apoptotic members contain a BH3 domain, although some anti-apoptotic members also contain the BH3 domain.10 The BH3 sequence motif has a hydrophobic α-helix which is favourable for protein interaction, it has been hypothesized that this is the region for the association among the BCL-2 family proteins via homo- or hetero-oligomerization.10,11 Additionally, there is evidence suggesting that the relative concentrations of pro- and anti-apoptotic BCL-2 members provide tight control over the balance of cell survival and apoptotic cell death.9,10,12
Among the entire BCL-2 family, Bax and Bcl-2 have been designated to be the main protagonists in the regulation of apoptotic signalling as they assume ion channel forming activities. Bax translocates to the mitochondria and exposes its N-terminus via a conformational change upon induction of apoptosis.13–17 This conformational change has been suggested to allow the Bax-Bax-oligomerization and insertion of Bax into the outer mitochondrial membrane,18 which is followed rapidly by the release of the apoptogenic factors (e.g., cytochrome c and AIF) from the mitochondrial intermembrane space. Collectively, Bax oligomerization is critical for mitochondrial membrane permeabilisation whereas Bcl-2 opposes the pro-apoptotic activity of Bax by preventing the process of Bax-Bax-oligomerization.19 Although the precise mechanism of Bax-mediated apoptogenic factor release from the mitochondrial intermembrane space is not fully known, it is indisputable that Bax plays an important role in promoting the activation of apoptotic signalling cascades.
Previously, the activation of mitochondria-associated apoptosis has been shown in skeletal muscle during denervation, which induces a considerable loss of muscle mass.20 Since pro-apoptotic Bax is markedly upregulated by denervation, it could be a candidate in the activation of apoptosis in response to muscle denervation.20–22 Furthermore, oxidative stress may play a role in regulating muscle wasting23 and disuse-associated muscle loss.24–30 The production of reactive oxidants in inactive skeletal muscle (e.g. nitric oxide/peroxynitrite, and/or mitochondrial reactive oxygen species) as well as the potential signalling pathways that could link oxidative stress to the proteolytic processes mediating muscle loss (e.g., signalling of caspase-3, calpain-mediated and proteasome-mediated proteolyses)23,31 could also contribute to denervation-induced muscle wasting.
In this study we tested the hypotheses that deficiency of the pro-apoptotic Bax gene would suppress the pro-apoptotic signalling in the atrophying skeletal muscle during denervation and attenuate the extent of denervation-induced muscle loss. Our data indicate that under conditions of denervation, pro-apoptotic signalling is suppressed and muscle wasting is attenuated when the Bax gene is lacking. The results of this study indicate the future importance of evaluating intervention strategies that target the regulation of apoptosis as a countermeasure for offsetting the denervation-associated muscle wasting pathologies.
Methods
Animals
Experiments were conducted on ~ 3-mo old adult C57BL/6 wild-type mice (N = 8) and homozygous B6.129X1-Baxtm1Sjk/J the Bax gene-deficient mice (N = 6) that were obtained from the Jackson Laboratory JAX® Mice Colony (Bar Harbor, Maine). The deficiency of Bax in the knockout mice was confirmed by immunoblotting on two affinity-purified rabbit polyclonal antibodies raised against peptide mapping at the N-terminus or amino acids 1–171 of Bax (sc-4936 and sc-236, Santa Cruz Biotechnology, Santa Cruz, CA). The mice were housed in pathogen-free conditions at ~ 20°C after arrival. They were exposed to a reverse light condition of 12:12 h of light: darkness each day and were fed chow and water ad libitum for a familiarization period of at least 5 days before the denervation surgery.
Muscle denervation procedures
The mice were placed under a general anaesthesia using 2% isoflurane. After reflex activity had disappeared, an incision was made from the calcaneous to just proximal to the popliteal fossa. The tibial nerve was dissected proximal to the cranial border of the gastrocnemius muscle. Care was taken to avoid any damage to the nerves, blood vessels, and connective tissues. The medial and lateral branches of the tibial nerve that innervate the plantar flexor muscles (i.e., gastrocnemius and soleus) were transected close to their neuromuscular junction32 and ~ 2 mm of the transected nerve was removed. Innervation to the plantaris and the deep toe flexor muscles were left intact so that the animals ambulated normally around the cage after the surgical denervation. Following the surgery, the hamstring muscle layers were closed with reabsorbable suture and the skin incision were closed with wound clips. The incision sites were covered with an antibacterial cream to prevent infection. The denervation procedure was performed in one limb and a sham procedure was performed in the contralateral limb of the mice. The sham surgery was identical to the experimental surgery except that the tibial nerve was not transected. The limb used for the sham surgery served as the intra-animal control limb. The mice recovered quickly and were able to walk around their cage within minutes post-surgery.21,33
Fourteen days after the surgical denervation and sham surgeries the mice were euthanised with an overdose of pentobarbital sodium (5 mg/g body weight, i.p.), while the gastrocnemius muscles from each limb were dissected from the surrounding connective tissues, removed, and stored at −80°C. The institutional animal use and care committee from West Virginia University School of Medicine approved all experiments. The animal care standards were followed by adhering to the recommendations for the care of laboratory animals as advocated by the American Association for Accreditation of Laboratory Animal Care and fully conformed to the Animal Welfare Act of the U.S. Department of Health and Human Services.
Subcellular protein fractionation
Cytosolic and nuclear protein fractions were obtained from gastrocnemius muscle samples as described by Rothermel et al.34 with minor modification. Briefly, after removal of connective tissues, muscles were homogenized on ice in ice-cold lysis buffer (10 mM NaCl, 1.5 mM MgCl2, 20 mM HEPES, 20% glycerol, 0.1% Triton X-100, 1 mM dithiothreitol or DTT, pH 7.4). Following centrifuging at 3,000 rpm to pellet the nuclei and cell debris, the supernatants were collected. The supernatants were then centrifuged three times at 6,000 rpm to remove residual nuclei and stored as nuclei-free total cytosolic protein fraction. A portion of the cytosolic extract (without addition of protease inhibitors) was stored and used for fluorometric caspase protease activity assay while a protease inhibitor cocktail containing 104 mM AEBSF, 0.08 mM aprotinin, 2 mM leupeptin, 4 mM bestatin, 1.5 mM pepstatin A, and 1.4 mM E-64 (Sigma-Aldrich, St Louis, MO) was added to the remaining portion. The cytosolic protein fraction containing protease inhibitors was used in cell death ELISAs, Western immunoblots, H2O2 assays, MDA/4-HAE assays, and nitrotyrosine dot blots. The remaining nuclear pellets were washed 3 times with ice-cold lysis buffer, resuspended in 300 μl of lysis buffer in the presence of 41.5 μl of 5 M NaCl and protease inhibitor cocktail, and rotated for 1 h at 4°C to lyse the nuclei. After centrifugation at 15,000 rpm, the supernatants were collected and stored as a cytosolic-free nuclear protein fraction. We have previously obtained the fractionated cytosolic and nuclear proteins from skeletal and heart muscles using this modified protocol.20,35–38
The release of mitochondria-residing apoptotic factors including cytochrome c, AIF, and Smac/DIABLO into the cytosol, was estimated from a nuclei-free, mitochondria-free cytosolic protein fraction that was prepared as described by Rokhlin et al.39 The protein contents of these mitochondrial apoptotic factors were measured in the mitochondria-free cytosolic fraction as described below. In brief, muscles were dissected from the connective tissues and minced in ice-cold extraction buffer (250 mM sucrose, 20 mM HEPES, 10 mM KCl, 1.5 mM MgCl2, 1 mM EDTA, 1 mM EGTA, 1 mM DTT, and 0.1 mM phenylmethylsulfonyl fluoride, pH 7.4) in the presence of the protease inhibitor cocktail. Following a gentle homogenisation with a Teflon pestle motorized with an electronic stirrer, homogenates were centrifuged at 800 × g for 10 min at 4°C to pellet the nuclei and cell debris. The supernatants were centrifuged at 16,000 × g for 20 min to pellet the mitochondria and the final supernatants were collected as nuclei-free, mitochondria-free cytosolic protein fractions. The subcellular protein fractionation procedures are routinely used in our laboratory to obtain high purity protein fractions as assessed by immunoblotting the fractions with an anti-histone H2B (a nuclear protein) (07371, Upstate, Lake Placid, NY), an anti-CuZnSOD (a cytosolic isoform of superoxide dismutase) (sc-11407, Santa Cruz Biotechnology, Santa Cruz, CA), and an anti-MnSOD (a mitochondrial isoform of superoxide dismutase) antibody (A300449A, Bethyl Lab, Montgomery, TX).20,36,37
The protein contents of the muscle extracts were quantified in duplicate by BCA Protein Assay (Pierce, Rockford, IL) based on the biuret reaction and the bicinchoninic acid detection of cuprous cation.40 As a further means to confirm the protein contents, all the protein samples were measured in duplicate on a different occasion by DC Protein Assay (BioRad, Hercules, CA) based on the reaction of protein with an alkaline copper tartrate solution and Folin reagent, which was similar to the Lowry assay.41
Apoptotic cell death ELISA
A cell death detection ELISA kit (Roche Applied Science, Indianapolis, IN) was used to quantitatively determine the apoptotic DNA fragmentation by measuring the cytosolic histone-associated mono- and oligonucleosomes. Briefly, the nuclei-free cytosolic fraction of gastrocnemius muscle was used as an antigen source in a sandwich ELISA with a primary anti-histone mouse monoclonal antibody coated to the microtitre plate and a second anti-DNA mouse monoclonal antibody coupled to peroxidase. The amount of peroxidase retained in the immunocomplex was determined photometrically by incubating with 2,2′-azino-di-[3-ethylbenzthiazoline sulfonate] (ABTS) as a substrate for 10 min at 20°C. The change in colour was measured at a wavelength of 405 nm by using a Dynex MRX plate reader controlled through PC software (Revelation, Dynatech Laboratories, CA). Measurements were performed in duplicate, with denervated and contralateral control samples analysed on the same microtitre plate in the same setting. The OD405 reading was then normalized to the mg of protein used in the assay.
Fluorometric caspase activity assay
A fluorometric assay was used to examine the protease activities of caspase-3 and -9 in the denervated gastrocnemius muscle. In brief, 50 μl of the total cytosolic protein fraction without protease inhibitor was incubated in 50 μl of assay buffer (50 mM PIPES, 0.1 mM EDTA, 10% glycerol, 10 mM DTT, pH 7.2) with 100 μM of the fluorogenic 7-amino-4-trifluoromethyl coumarin (AFC)-conjugated substrate (Ac-DEVD-AFC for caspase-3, Ac-LEHD-AFC for caspase-9, Alexis Corp., San Diego, CA) at 37°C for 2 h. Caspase specific inhibitor, Z-VAD-FMK (Calbiochem, La Jolla, CA) was used as a control to validate the specificity of caspase. The change in fluorescence was measured on a spectrofluorometer with an excitation wavelength of 390/20 nm and an emission wavelength of 530/25 nm (CytoFluor, Applied Biosystems, Foster City, CA) before and after the 2 h incubation. Caspase activity was estimated as the change in arbitrary fluorescence units normalized to milligram protein. Measurements were performed in duplicate while denervated and intra-animal control samples were run on the same microplate in the same setting.
Western immunoblot analyses
The protein expressions of Bcl-2 associated X protein (Bax), B-cell lymphoma/leukemia-2 (Bcl-2), X-linked inhibitor of apoptosis (XIAP), apoptosis repressor with caspase recruitment domain (ARC), heat shock protein-70 (HSP70), HSP27, HSP60, and superoxide dismutases (CuZn-SOD and Mn-SOD) was determined in the total cytosolic protein fraction while apoptosis inducing factor (AIF) was measured in the nuclear fraction. Both the total cytosolic and nuclear fractions were used to measure the protein content of p53.
Forty micrograms of protein was boiled for 5 min at 95°C in Laemmli buffer and then loaded on each lane of a 12% polyacrylamide gel and separated by SDS-PAGE at room temperature. The gels were blotted to nitrocellulose membranes (VWR, West Chester, PA) and stained with Ponceau S red (Sigma Chemical Co, St Louis, MO) to verify equal loading and transferring of proteins to the membrane in each lane. As another approach to validate similar loading between the lanes, gels were loaded in duplicate with one gel stained with Coomassie blue. The membranes were then blocked in 5% non-fat milk in phosphate buffered saline with 0.05% Tween 20 (PBS-T) at room temperature for 1 h and probed with the following primary antibodies diluted in PBS-T with 2% BSA: anti-Bcl-2 mouse monoclonal antibody (1:100 dilution, sc-7382), anti-Bax rabbit polyclonal antibody (1:200 dilution, sc-493), anti-hILP/XIAP mouse monoclonal antibody (1:250 dilution, 610762, San Jose, CA), anti-ARC rabbit polyclonal antibody (1:200 dilution, sc-11435), anti-AIF mouse monoclonal antibody (1:800 dilution, sc-13116HRP), anti-p53 mouse monoclonal antibody (1:100 dilution, sc-99), anti-HSP70 mouse monoclonal antibody (1:2000 dilution, SPA810B), anti-HSP27 rabbit polyclonal antibody (1:2000 dilution, SPA801), anti-HSP60 rabbit polyclonal antibody (1:2000 dilution, SPA804), anti-SOD-1 rabbit polyclonal antibody (1:500 dilution, sc-11407), or anti-MnSOD goat antibody (1:2000 dilution, A300449A, Bethyl Lab, Montgomery, TX). Bcl-2, Bax, ARC, SOD-1, AIF, and p53 antibodies were purchased from Santa Cruz Biotechnology (Santa Cruz, CA) while HSP70, HSP27, and HSP60 antibodies were purchased from StressGen (Victoria, BC, Canada). All primary antibody incubations were performed overnight at 4°C. Secondary antibodies were conjugated to horseradish peroxidase (Chemicon International, Temecula, CA), and signals were developed by West Pico chemiluminescent substrate (Pierce, Rockford, IL). The signals were then visualized by exposing the membranes to X-ray films (BioMax MS-1, Eastman Kodak, Rochester, NY), and digital records of the films were captured with a Kodak 290 camera. Resulting bands were quantified as optical density (OD) × band area by a one-dimensional image analysis system (Eastman Kodak, Rochester, NY) and recorded in arbitrary units. The molecular sizes of the immunodetected proteins were verified by using pre-stained standard (LC5925, Invitrogen Life Technologies, Bethesda, MD).
Estimation of mitochondrial cytochrome c, Smac/DIABLO, and AIF release
Cytochrome c, AIF, and Smac/DIABLO (second mitochondria-derived activator of caspase) are apoptotic factors normally confined to mitochondria and their release into the cytosolic compartment has been demonstrated during the activation of apoptosis.42 In the present study, the release of Smac/DIABLO and AIF into the cytosolic compartment was estimated by measuring their protein contents in the mitochondria-free cytosolic protein fraction by immunoblotting with an anti-Smac/DIABLO mouse monoclonal antibody (1:500 dilution, 612244, BD Biosciences, San Jose, CA) and an anti-AIF monoclonal mouse antibody. Moreover, a cytochrome c ELISA kit (MBL International, Woburn, MA) was used to assess the protein content of cytochrome c in the mitochondria-free cytosolic fraction to evaluate the release of the mitochondrial cytochrome c into the cytosol. 60 μl the mitochondria-free cytosolic protein fraction was used as an antigen source in a sandwich ELISA with a horseradish peroxidase-conjugated anti-cytochrome c polyclonal antibody in microwell strips coated with an anti-cytochrome c antibody. After washing, the peroxidase retained in the immunocomplex was detected by incubating with a chromogenic substrate, tetramethylbenzidine/hydrogen peroxide (TMB/H2O2) followed by adding an acid solution to terminate the enzyme reaction and to stabilize the developed colour. The change in colour was monitored at a wavelength of 450 nm using a Dynex MRX plate reader. Measurements were performed in duplicate with denervated and contralateral control samples analysed on the same microplate and the cytochrome c content was expressed as OD450 per mg protein.
MDA/4-HAE assay
Polyunsaturated fatty acid peroxides generate malondialdehyde (MDA) and 4-hydroxyalkenals (4-HAE) upon decomposition.43 In the present study, the content of MDA/4-HAE was measured in the cytosolic muscle protein fraction as an indicator of lipid peroxidation using a commercial colorimetric assay kit (21012, Oxis International, Portland, OR). Briefly, 50 μl of the cytosolic protein fraction or standards with known concentrations was incubated with a chromogenic reagent, N-methyl-2-phenylindole, at 45°C for 1 h and the generated chromospheres after incubation were detected by spectrophotometry at 586 nm. The MDA/4-HAE concentration of the samples was determined according to the standard curve generated in the same setting. The results were presented as OD586 normalized to milligram protein used in the assay. Control and experimental samples were run in the same setting to eliminate any assay-to-assay variations.
H2O2 fluorometric assay
The content of hydrogen peroxide (H2O2) in the muscle homogenate was measured in the cytosolic protein fraction using a fluorometric H2O2 detection kit (FLOH 100-3, Cell Technology Inc., Mountain View, CA). 50 μl of the total cytosolic protein fraction of the gastrocnemius muscle was incubated in 50 μl of reaction cocktail containing horseradish peroxidase and 10-acetyl-3,7-dihydroxyphenoxazine (ADHP, a non-fluorescent substrate turns fluorescent after oxidized by H2O2) in sodium phosphate buffer. The fluorescence was measured on a spectrofluorometer with an excitation wavelength of 530/25 nm and an emission wavelength of 590/35 nm (CytoFluor, Applied Biosystems, Foster City, CA) after the incubation. H2O2 content was estimated as the arbitrary fluorescence units normalized to milligram protein used in the assay. Measurements were performed with the denervated and control samples run on the same microplate in the same setting.
Nitrotyrosine dot blot
An immunodot blot was carried out to estimate the total nitrotyrosine content in the cytosolic fraction of the gastrocnemius muscles from experimental and control animals. In brief, 4 μg of cytosolic protein was dotted on a nitrocellulose membrane (VWR, West Chester, PA). After air drying, the membrane was blocked in 5% non-fat milk in PBS-T at room temperature for 1 h and then probed with an anti-nitrotyrosine mouse monoclonal antibody (1:1000 dilution, MAB5404, Chemicon International, Temecula, CA) diluted in PBS-T with 2% BSA incubated at 4°C for overnight. The membrane was incubated with a HRP-conjugated anti-mouse IgG secondary antibody (AP124P, Chemicon International, Temecula, CA) at room temperature for 1 h, and then signals were developed by chemiluminescence (Pierce Biotechnology, Rockford, IL). Negative control experiments were performed by omitting either the anti-nitrotyrosine or the secondary antibody. The signals were then visualized by exposing the membranes to X-ray films (BioMax MS-1, Eastman Kodak, Rochester, NY), and digital records of the films were captured with a Kodak 290 camera. Resulting dot signals were quantified in arbitrary units as optical density (OD) × dot area using Kodak one-dimensional (1-D) image analysis system (Eastman Kodak, Rochester, NY).
Statistical analyses
Statistical analyses were performed using the SPSS 10.0 software package. ANOVA followed by Tukey HSD post hoc analysis was used to examine differences between groups. Independent t-test was performed to examine the differences of the variable percent change between wild-type and Bax−/− animals. All data are given as means ± standard error (SE). Statistical significance was accepted at P < 0.05.
Results
Muscle weight change
The denervation-induced muscle loss was monitored by examining the gastrocnemius muscle wet weight normalized to the animal’s bodyweight between the denervated and intra-animal contralateral control limbs in wild-type and Bax−/− mice. Following 14 days of denervation, the normalized muscle wet weight was decreased by 42 and 33% in wild-type and Bax−/− mice, respectively (wild-type: 4.5 vs. 2.6 mg/g; Bax−/−: 4.2 vs. 2.8 mg/g) (Figure 1). The percent decline in muscle weight of denervated vs. control muscles was lower in the Bax−/− mice (−33%) than the wild-type mice (−42%).
Figure 1.
Muscle weight. The extent of muscle loss following denervation was estimated by examining the gastrocnemius muscle wet weight loss normalized to the animal’s bodyweight between the denervated and the contralateral control sides. The data are expressed as means ± standard error of mean (SE). *P < 0.05, denervated muscle was significantly different from the contralateral control muscle under the same animal strain.
Apoptotic DNA fragmentation
The cell death ELISA analysis indicated that the extent of apoptotic DNA fragmentation in the denervated muscle was 161% greater than the control muscle in the wild-type mice whereas, in the Bax−/− mice, the increase in the apoptotic DNA fragmentation in the denervated muscle relative to the control muscle of was not statistically significant (74%, P > 0.05) (Figure 2). The increase in apoptotic DNA fragmentation was significantly lower in muscles of Bax−/− (74%) as compared to wild-type mice (161%).
Figure 2.
Apoptotic DNA fragmentation. The extent of apoptotic DNA fragmentation was estimated by measuring the cytosolic mono- and oligonucleosomes. The OD405 is normalized to the total milligrams protein content of the sample used in the assay. The normalized data are expressed as means ± SE. *P < 0.05, denervated muscle was significantly different from the contralateral control muscle under the same animal strain;**P < 0.05, Bax−/− mice were significantly different from the wild-type mice under the same experimental condition.
BCL-2 family: Bax and Bcl-2 protein contents
According to our immunoblot analysis, we did not detect the presence of Bax protein in all samples from the Bax−/− animals (Figure 3A). For the wild-type mice, the Bax protein content in the denervated muscle was 415% higher than the contralateral control muscle. The protein content of Bcl-2 in the muscle following denervation was elevated by 353 and 307% relative to the contralateral control muscle in the wild-type and Bax−/− mice, respectively (Figure 3B) while the percent change of Bcl-2 protein content was not different in the wild-type and Bax−/− mice.
Figure 3.
(A) Bax protein content. The Bax protein content was examined by Western immunoblot. The insets show representative blots for the Bax protein. The data are expressed as means ± SE of the arbitrary unit. *P < 0.05, denervated muscle was significant different from the contralateral control muscle under the same animal strain. (B) Bcl-2 protein content. The Bcl-2 protein content was examined by Western immunoblots. The insets show representative blots for the Bcl-2 protein. The data are expressed as means ± SE of the arbitrary unit. *P < 0.05, denervated muscle was significantly different from the contralateral control muscle under the same animal strain.
Mitochondrial cytochrome c release
The protein content of cytochrome c in the cytosolic fraction of denervated muscle increased by 98 and 46% when compared to the control muscle in the wild-type and Bax−/− mice, respectively (Figure 4). As cytochrome c primarily resides in the mitochondria under normal conditions, these results indicate that cytochrome c was released to the cytosolic protein fraction or perhaps accumulated in the cytosol during denervation in both wild-type and Bax−/− muscles. Although the release of mitochondrial cytochrome c increased by 46% in denervated muscles of the Bax−/− mice relative to control muscles, this was significantly lower than the 98% increase in cytosolic cytochrome c muscles from wild-type animals as compared to control muscles.
Figure 4.
Mitochondrial cytochrome c release. The release of mitochondrial cytochrome c was estimated by measuring the protein content of cytochrome c in the mitochondria-free cytosolic fraction using an ELISA. The OD450 is normalized to the total milligrams protein content of the sample used in the assay. The normalized data are expressed as means ± SE. *P < 0.05, denervated muscle was significantly different from the contralateral control muscle under the same animal strain;**P < 0.05, Bax−/− mice were significantly different from the wild-type mice under the same experimental condition.
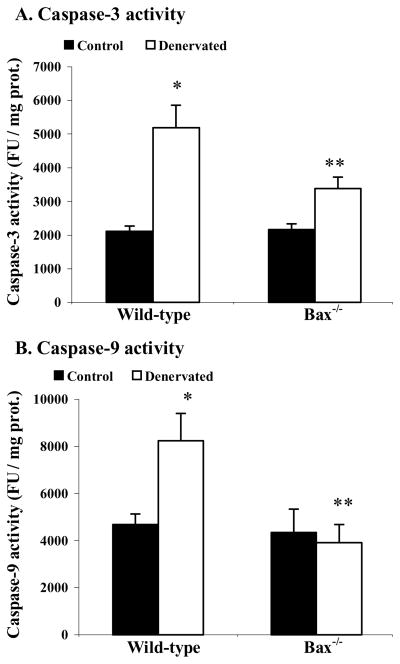
Caspase-3 and -9 protease activities
The activation of caspase-3 and -9 was determined by measuring the specific protease activity in the muscle samples using a fluorometric caspase enzymatic activity assay. There was a 150% increase in the protease activity of caspase-3 in the samples from denervated muscles when compared to the control muscles in the wild-type mice but there was no statistical difference (P > 0.05) in the caspase-3 activity of the denervated Bax−/− muscles relative to the intra-animal control muscles (Figure 5A). The percent increase in caspase-3 activity following denervation was significantly greater in the muscles from wild-type (150%) as compared to muscles from Bax−/− mice (55%). The protease activity of caspase-9 was 81% higher in denervated muscles when compared to the contralateral control muscles in the wild-type animals (Figure 5B). In contrast, no difference in the caspase-9 activity was found between the denervated and control muscles of the Bax−/− mice (Figure 5B). Denervated muscles of the wild-type mice had a significantly greater increase in caspase-9 activity compared to muscles from wild-type animals.
Figure 5.
(A) Caspase-3 protease activity. The caspase-3 activity was assessed by a fluorometric caspase activity assay. The fluorescence unit (FU) is normalized to the total milligrams protein content of the sample used in the assay. The normalized data are expressed as means ± SE. *P < 0.05, denervated muscle was significantly different from the contralateral control muscle under the same animal strain; **P < 0.05, Bax−/− mice were significantly different from the wild-type mice under the same experimental condition. (B) Caspase-9 protease activity. The caspase-9 activity was assessed by a fluorometric caspase activity assay. The fluorescence unit (FU) is normalized to the total milligrams protein content of the sample used in the assay. The normalized data are expressed as means ± SE. *P < 0.05, denervated muscle was significantly different from the contralateral control muscle under the same animal strain; **P < 0.05, Bax−/− mice were significantly different from the wild-type mice under the same experimental condition.
Mitochondrial Smac/DIABLO release, XIAP and ARC protein contents
As indicated by our immunoblot analysis on the mitochondria-free cytosolic fraction, the protein content of Smac/DIABLO, an upstream inhibitor of XIAP, increased by 213% and 252% in the cytosolic fraction (in the absence of mitochondria) of the denervated muscles relative to the control muscles in the wild-type and Bax−/− mice, respectively (Figure 6A). These denoted that mitochondrial Smac/DIABLO was released/accumulated into the cytosol during denervation in both wild-type and Bax−/− mice. No significant difference was found in the percent change of mitochondrial Smac/DIABLO release in the wild-type and Bax−/− mice. The protein content of XIAP, an inhibitor of caspases, as detected by immunoblotting on the total cytosolic fraction was 41 and 39% lower in the muscle following denervation when compared to the contralateral control muscle in the wild-type and Bax−/− mice, respectively (Figure 6B). But, no difference was found in the denervation-induced decrease in the XIAP protein content between the wild-type and Bax−/− mice. ARC, an apoptotic suppressor, protein content was not altered with denervation in the wild-type animals (P > 0.05) whereas the protein content of ARC increased by 37% in the denervated muscle relative to the contralateral control muscle in the Bax−/− mice (Figure 6C).
Figure 6.
(A) Mitochondrial Smac/DIABLO release. The protein content of Smac/DIABLO was measured by Western immunoblot in the mitochondria-free cytosolic fraction in order to estimate the mitochondrial Smac/DIABLO release. The insets show representative blots. The data are expressed as means ± SE of the arbitrary unit.*P < 0.05, denervated muscle was significantly different from the contralateral control muscle under the same animal strain. (B) XIAP protein content. The protein content of XIAP was measured by Western immunoblots in the total cytosolic fraction. The insets show representative blots. The data are expressed as means ± SE of the arbitrary unit.*P < 0.05, denervated muscle was significantly different from the contralateral control muscle under the same animal strain. (C) ARC protein content. The protein content of ARC was measured by Western immunoblots in the total cytosolic fraction. The insets show representative blots. The data are expressed as means ± SE of the arbitrary unit. *P < 0.05, denervated muscle was significantly different from the contralateral control muscle under the same animal strain; **P < 0.05, Bax−/− mice were significantly different from the wild-type mice under the same experimental condition.
Mitochondrial AIF release and nuclear translocation
The mitochondrial release and nuclear translocation of AIF were estimated by immunoblotting the mitochondria-free cytosolic and nuclear muscle protein fractions, respectively. The AIF protein content measured in the mitochondria-free cytosolic fraction of the denervated muscle was 80% and 74% higher than the intra-animal control muscle in the wild-type and Bax−/− mice, respectively (Figure 7A). These results indicate that AIF was released/accumulated from the mitochondria into the cytosolic fraction during denervation in both the wild-type and Bax−/− animals. The percent change of mitochondrial AIF release/accumulation was similar between the wild-type and Bax−/− mice. In contrast, we did not find any difference in the nuclear AIF protein content between the denervated and control muscles from either the wild-type or Bax−/− mice (Figure 7B).
Figure 7.
(A) Mitochondrial AIF release. The protein content of AIF was measured by Western immunoblots in the mitochondria-free cytosolic fraction in order to estimate the mitochondrial AIF release. The insets show representative blots. The data are expressed as means ± SE of the arbitrary unit. *P < 0.05, denervated muscle was significantly different from the contralateral control muscle under the same animal strain. (B) AIF nuclear translocation. The protein content of AIF was measured by Western immunoblots in the nuclear fraction in order to estimate the nuclear translocation of AIF. The insets show representative blots. The data are expressed as means ± SE of the arbitrary unit.
Tumour suppressor p53 protein content
The p53 protein content as estimated from immunoblots analyses increased by 175 and 167% in the nuclear fraction of denervated muscles compared to the contralateral control muscles in wild-type and Bax−/− mice, respectively (Figure 8A). Denervation produced a similar change in nuclear p53 protein content in muscles from wild-type and Bax−/− mice. There was a 353 and 362% increase in the p53 protein content in the cytosolic protein fraction of denervated muscles when compared to the control muscles in wild-type and Bax−/− mice, respectively (Figure 8B). In addition, the change in the cytosolic p53 protein content was similar in denervated vs. control muscles from wild-type and Bax−/− mice.
Figure 8.
(A) Nuclear p53 protein content. The protein content of p53 was measured by Western immunoblots in the nuclear fraction. The insets show representative blots. The data are expressed as means ± SE of the arbitrary unit. *P < 0.05, denervated muscle was significantly different from the contralateral control muscle under the same animal strain. (B) Cytosolic p53 protein content. The protein content of p53 was measured by Western immunoblots in the total cytosolic fraction. The insets show representative blots. The data are expressed as means ± SE of the arbitrary unit.*P < 0.05, denervated muscle was significantly different from the contralateral control muscle under the same animal strain.
Stress proteins: HSP70, HSP27, and HSP60 protein contents
There was a large inter-animal variability in the response of HSP70 protein content to denervation. As a result, we did not find any statistical significant difference in HSP70 between denervated and contralateral control muscles in wild-type or Bax−/− mice (Figure 9A). In contrast, the protein content of HSP27 was elevated by 82 and 106% in denervation muscles relative to the control muscle in wild-type and Bax−/− mice, respectively (Figure 9B). However, the magnitude of the change of HSP27 protein content was not significantly different between the wild-type and Bax−/− mice. Although the protein content of HSP60 tended to decrease (− ~ 20%) in denervated muscles when compared to the control muscles in both the wild-type and Bax−/− mice, the change did not reach statistical significance (Figure 9C).
Figure 9.
(A) HSP70 protein content. The protein content of HSP70 was measured by Western immunoblots in the total cytosolic fraction. The insets show representative blots. The data are presented as means ± SE of the arbitrary unit. (B) HSP27 protein content. The protein content of HSP27 was measured by Western immunoblots in the total cytosolic fraction. The insets show representative blots. The data are presented as means ± SE of the arbitrary unit. *P < 0.05, denervated muscle was significantly different from the contralateral control muscle under the same animal strain. (C) HSP60 protein content. The protein content of HSP60 was measured by Western immunoblots in the total cytosolic fraction. The insets show representative blots. The data are presented as means ± SE of the arbitrary unit.
Oxidative stress markers: H2O2, MDA/4-HAE, and nitrotyrosine contents
In the wild-type animals, the contents of H2O2, MDA/4-HAE, and nitrotyrosine were elevated by 59, 28, and 114%, respectively, in the denervated muscle relative to the contralateral control muscle but these changes were not found in the Bax−/− animals (Figure 10). Moreover, the relative differences in H2O2, MDA/4-HAE, and nitrotyrosine contents were significantly greater in the denervated vs. control muscles of wild-type mice when compared to the Bax−/− mice (wild-type vs. Bax−/− mice: H2O2, + 59% vs. −15%; MDA/4-HAE, + 28% vs. + 1%; nitrotyrosine, + 114% vs. −2%).
Figure 10.
(A) H2O2 content. The H2O2 content was determined by a fluorometric assay. The fluorescence unit (FU) is normalized to the total milligrams protein content of the sample used in the assay. The normalized data are presented as means ± SE. *P < 0.05, denervated muscle was significantly different from the contralateral control muscle under the same animal strain;**P < 0.05, Bax−/− mice were significantly different from the wild-type mice under the same experimental condition. (B) MDA/4-HAE content. The level of lipid peroxidation was estimated by measuring the content of MDA/4-HAE. The OD586 is normalized to the total milligrams protein content of the sample used in the assay. The normalized data are presented as means ± SE. *P < 0.05, denervated muscle was significantly different from the contralateral control muscle under the same animal strain;**P < 0.05, Bax−/− mice were significantly different from the wild-type mice under the same experimental condition. (C) Nitrotyrosine content. The content of nitrotyrosine was measured in the cytosolic fraction by an immunodot blot. The data are expressed as OD x resulting band area, and expressed in arbitrary units. The insets show representative blots for nitrotyrosine. The data are presented as means ± SE. *P < 0.05, denervated muscle was significantly different from the contralateral control muscle under the same animal strain; **P < 0.05, Bax−/− mice were significantly different from the wild-type mice under the same experimental condition.
Antioxidant enzyme: MnSOD and CuZnSOD protein contents
MnSOD protein content as estimated by immunoblot analysis was diminished by 23% and 21% in the denervated muscle when compared to the contralateral control muscle in the wild-type and Bax−/− mice, respectively (Figure 11A). However, no significant difference was found in the percent change of MnSOD protein content between the wild-type and Bax−/− mice. Also, we did not find any difference in the CuZnSOD protein content between the denervated and control muscles from either the wild-type or the Bax−/− mice (Figure 11B).
Figure 11.
(A) MnSOD protein content. The protein content of Mn-SOD was assessed in cytosolic fraction by immunoblot analysis. The data are expressed as OD x resulting band area, and expressed in arbitrary units. The insets show representative blots for MnSOD. The data are presented as means ± SE. *P < 0.05, denervated muscle was significantly different from the contralateral control muscle under the same animal strain. (B) CuZnSOD protein content. The protein content of CuZnSOD was assessed in cytosolic fraction by immunoblot analysis. The data are expressed as OD x resulting band area, and expressed in arbitrary units. The insets show representative blots for MnSOD. The data are presented as means ± SE.
Discussion
Activation of apoptotic signalling in denervated rat muscle has been shown by the augmentation of apoptotic DNA fragmentation, increases in the Bax/Bcl-2 ratio, mitochondrial release of cytochrome c to the cytoplasm, increases in Smac/DIABLO and AIF, increases in mRNA, active protein and proteolytic activity of caspase-3 and -9, decreases in XIAP and MnSOD, and elevations of cleaved PARP protein, p53 and HSP70.20 In addition, increased labelling for DNA breaks as indicated by transferase-mediated dUTP biotin nick-end labelling (TUNEL) and altered BCL-2 expression have been shown in skeletal muscle following denervation.5,21,22,44–49 By using mice that are deficient of the Bax gene, an upstream essential pro-apoptotic mediator, we provide further evidence supporting that apoptotic signalling has a significant role and may be involved in mediating muscle wasting during denervation. We have demonstrated that the extent of muscle loss following denervation is attenuated in the skeletal muscle of Bax−/− mice compared to wild-type animals. The lower level of muscle loss in Bax-deficient muscles could not be due to different levels of stresses sensed by the muscles in wild-type and Bax−/− mice because the relative changes in HSP70, HSP60 and HSP27 stress proteins were similar in muscles of both types of mice.
Increases in apoptotic DNA fragmentation, caspase-3 and -9 protease activities and oxidative stress markers including H2O2, MDA/4-HAE and nitrotyrosine contents were evident in the wild-type muscles after denervation but these changes were not found in muscles from the Bax−/− mice. The magnitude of the mitochondrial cytochrome c release in the denervated muscle of the Bax−/− mice was also lower than the wild-type mice. Moreover, ARC protein content increased exclusively in the Bax−/− muscle but not the wild-type muscle following denervation. Overall, these findings suggest that interference of the pro-apoptotic signalling (i.e., lacking the Bax gene) attenuates the extent of muscle loss during denervation. Assuming that muscle denervation resulting from degeneration and retraction of motoneurons is a general process contributing to the disease progression of some neuromuscular disorders, these findings suggest that interventions that target apoptosis may be a valuable therapeutic strategy in ameliorating denervation-associated pathologic muscle wasting in certain neuromuscular disorders.
The majority of apoptotic investigations have studied mitotic cell populations. However, recent data demonstrate that apoptosis may have an important regulatory role in mediating the process of post-mitotic skeletal muscle wasting under both physiologic and pathophysiologic conditions.3,50 Clinical reports have well documented that apoptosis is involved in a variety of muscle disorders including spinal muscular myopathies, peripheral neuropathies, amyotrophic lateral sclerosis, muscle dystrophies, metabolic/mitochondrial myopathies, inflammatory myopathies, congenital myopathies, distal myopathies, thyroid-associated ophthalmyopathies, chronic heart failure-associated myopathies, critical ill myopathies, and burn injury-associated muscle wasting.3,4,7,8,51–59
A recent study clearly demonstrated that inhibition of apoptosis improves the outcome of skeletal muscle wasting pathology in the genetically knockout mice that were deficient of the laminin-α2 gene, an animal model of congenital muscular dystrophy.60 By adopting genetic intervention by crossbreeding the laminin-α2-deficient mice with pro-apoptotic Bax-deficient mice or with mice carrying the muscle-specific anti-apoptotic Bcl-2 transgene, Girgenrath and colleagues60 have shown that the extent of muscle pathology is ameliorated by either inactivation of the pro-apoptotic protein Bax or overexpression of the anti-apoptotic protein Bcl-2. It is worth noting that by inactivating Bax, both the postnatal growth rate and muscle fibre morphology are improved while the amount of fixed muscular contractures is reduced in the skeletal muscle taken from the laminin-α2/Bax-deficient animals, and these findings denote that pro-apoptotic Bax plays an important role in the progression of muscular pathologies.60 The data from our study are consistent with the idea that Bax-associated pro-apoptotic signalling is an important mediator in muscle wasting pathologies particularly induced by muscle denervation. Our results indicate that in response to the removal of nerve supply, the extent of muscle loss is attenuated and is accompanied by the suppression of pro-apoptotic signalling in skeletal muscle that is deficient of the Bax gene.
Differences in apoptotic signalling in response to muscle denervation were observed in skeletal muscles taken from the mice that were deficient of the Bax gene compared to the muscles from wild-type mice in this study. In the wild-type mouse muscle, we found changes in the apoptotic components including apoptotic DNA fragmentation, Bax, cytochrome c, Smac/DIABLO, AIF, caspases, XIAP, and p53. We also found an increase in Bcl-2 in denervated muscle of both wild-type and Bax−/− mice. The increase in Bcl-2 appears to be a general adaptive response to denervation because we have previously found elevated levels of Bcl-2 in denervated rat muscle.20 We interpret the increase in Bcl-2 as an attempt to counteract the apoptotic response and reduce the extent muscle fibre atrophy. However, the denervation-induced activation of the pro-apoptotic components was generally suppressed in muscles that lacked the pro-apoptotic Bax protein. The significant elevations of apoptotic DNA fragmentation, caspase-3 and -9 activities as found in the wild-type muscle following denervation were absent in the Bax-deficient denervated muscle. The denervation-induced mitochondrial cytochrome c release was also suppressed in the Bax-deficient muscle relative to the wild-type muscle. In addition, the increase in protein abundance of ARC, an endogenous apoptotic suppressor protein, was evident only in Bax-deficient muscle but not the wild-type muscle after denervation. The pro-apoptotic role of Bax is via its ability to facilitate the formation of “channels” in the outer mitochondrial membrane.61–63 This results in the release of apoptogenic factors (e.g., cytochrome c, AIF and Smac/DIABLO) from the mitochondrial inner membrane to the cytosol.64 We predicted that the extent of the releases of all apoptogenic proteins would be suppressed similarly in Bax-deficient denervated muscle if their releases were all directed by the same Bax-mediated mechanism. However, the present data clearly indicate that deficiency of Bax, selectively influences the mitochondrial release of cytochrome c but not AIF and Smac/DIABLO in the experimental model of muscle denervation. We interpret this to indicate that the regulatory control for the mitochondrial release of AIF and Smac/DIABLO differs from the mechanisms of cytochrome c release in skeletal muscle during denervation. We speculate that Bax may regulate a channel selective release of apoptogenic proteins from the mitochondrial inner membrane under conditions that include muscle loss via denervation.
When ARC was first identified as an apoptotic inhibitor it was considered to interact with selective caspases which is expressed in skeletal muscle and heart.65 However, the anti-apoptotic function of ARC may also be mediated by interacting with the activation of pro-apoptotic Bax.66 Therefore, it is not unreasonable to expect that ARC responds to muscle denervation differently in Bax-deficient and wild-type muscles. However, it is unclear if the observed increase in ARC abundance may have contributed to counteract or suppress the activation of the pro-apoptotic signalling during denervation in the Bax-deficient muscle. Nevertheless, the exact reason for the denervation-induced elevation of ARC in muscle that was deficient of the Bax gene is not known and it warrants further investigation.
Another notable finding in this study is that the oxidative stress markers including H2O2, MDA/4-HAE, and nitrotyrosine contents were all elevated following denervation in the wild-type muscle but none of these markers increased in the Bax-deficient denervated muscle. This is not totally unexpected because there is some evidence that oxidative stress is involved at least in part, in regulating muscle wasting.23 Several studies have shown that elevated oxidative stress contributes to the process of muscle loss as induced by muscle disuse.24–30 Furthermore, the proposition for the role of oxidative stress in muscle wasting is strengthened by the documented data showing the existences of possible mechanisms responsible for the production of reactive oxidants in inactive skeletal muscle (xanthine oxidase, reactive iron, NAD(P)H oxidase, nitric oxide/peroxynitrite, and/or mitochondrial reactive oxygen species) as well as the potential signalling pathways that could link oxidative stress to the proteolytic processes mediating muscle loss (e.g., signalling of caspase-3, calpain-mediated and proteasome-mediated proteolyses).23 The involvement of oxidative stress has also been implicated together with the down regulation of NOS/NO in response to denervation.31 It is noted that the present findings in muscle from wild-type mice are in agreement with the proposition that oxidative stress may have a role in mediating the muscle loss. Moreover, the absence of the increases in the markers of oxidative stress in the Bax-deficient denervated muscle raises the possibility that Bax is either directly or indirectly related to the reduction-oxidation or redox events during muscle denervation. There are at least two possible explanations to explain the observation that resistance to oxidative stress was increased in Bax deficient mice. First, given that oxidative stress is known to be an activator of apoptosis, it is commonly thought that increased oxidative stress acts upstream of apoptotic cascades. However, it appears that modulation of the essential apoptotic mediator (e.g., deficiency of Bax) can influence the events of oxidative stress. It is unlikely that the effect of oxidative stress is simply downstream of apoptosis, or in particular, pro-apoptotic Bax, because of the complex connection and interaction between redox regulation and apoptosis. Although the exact mechanism that coordinates oxidative stress and apoptosis is still under investigation, our data suggest that these two cellular events might cooperate in an interactive manner at least in skeletal muscle. It is possible that there is both feedback and/or feed-forward coordination between oxidative stress and apoptosis, however, such information will remain lacking until the underlying mechanisms are fully resolved. A second possibility is that muscle from Bax deficit mice had an improved balance between Bcl-2 and/or other anti-apoptotic proteins and the apoptotic Bax protein and this may have provided a more favourable environment for offsetting oxidant stress. It is well known that Bcl-2, is both an intimate working partner of Bax67 and that it has an antioxidant-promoting ability which provides resistance to oxidative stress-induced apoptosis.68 We speculate that the absence of Bax provided an environment where Bcl-2 could function to reduce oxidative stress during denervation of Bax-deficient muscle. Even though the anti-apoptotic Bcl-2 protein was elevated to a similar extent in response to denervation in Bax-null and wild-type muscles, the absence of Bax in the mutant mice would provide an optimal environment for Bcl-2 to contribute to oxidant stress reduction. In contrast, part of ability for Bcl-2 to respond to oxidant stress would be “buffered” by the presence of Bax in wild-type mice. Nevertheless, additional research is required to more fully examine the relationship between Bax and oxidative stress in denervated muscle.
We did not detect differences in the normalized muscle wet weight of the non-denervated gastrocnemius muscles of Bax-deficient and wild-type mice. This observation suggests that deficiency of the pro-apoptotic Bax has no influence on the normal growth/maturation of the skeletal muscle mass during the early lifespan from the birth to young adulthood (e.g., ~ 3-mo old mice). It appears that the proposed role of Bax in mediating the pro-apoptotic signalling that results in the decline of muscle mass, is restricted to conditions invoking an atrophic stimuli (e.g., denervation-induced muscle loss). It is worth noting that our study was conducted in young adult mice so it is not known if the deficiency of Bax would contribute to age-associated muscle loss (i.e., sarcopenia). The role of Bax in ageing-associated muscle wasting requires further investigation.
In conclusion, the present data provide evidence demonstrating that interference of pro-apoptotic signalling by the knockout of the pro-apoptotic Bax gene attenuates the extent of muscle wasting in response to skeletal muscle denervation. These results indicate that apoptotic regulation may be involved in mediating the process of muscle loss during muscle denervation and suggest that strategies targeting apoptosis may be of clinical importance in offsetting muscle wasting pathologies which are related to muscle denervation. Further investigation is required to evaluate other potential apoptotic interventions (e.g., overexpression of the anti-apoptotic factors including Bcl-2, IAP, and ARC) in inhibiting the denervation-associated muscle wasting, and therefore ameliorating muscle wasting in certain neuromuscular diseases.
Acknowledgments
We are thankful to Dr. William Wonderlin for providing access to the CytoFluor spectrofluorometer. This study was supported by NIH: National Institute on Aging Grant R01AG021530.
References
- 1.Hughes SM. Muscle development: Electrical control of gene expression. Curr Biol. 1998;8:R892–R894. doi: 10.1016/s0960-9822(07)00554-4. [DOI] [PubMed] [Google Scholar]
- 2.Pette D. Historical Perspectives: Plasticity of mammalian skeletal muscle. J Appl Physiol. 2001;90:1119–1124. doi: 10.1152/jappl.2001.90.3.1119. [DOI] [PubMed] [Google Scholar]
- 3.Tews DS. Apoptosis and muscle fibre loss in neuromuscular disorders. Neuromuscul Disord. 2002;12:613–622. doi: 10.1016/s0960-8966(02)00030-5. [DOI] [PubMed] [Google Scholar]
- 4.Fidzianska A, Goebel HH, Warlo I. Acute infantile spinal muscular atrophy. Muscle apoptosis as a proposed pathogenetic mechanism. Brain. 1990;113 (Pt 2):433–445. doi: 10.1093/brain/113.2.433. [DOI] [PubMed] [Google Scholar]
- 5.Migheli A, Mongini T, Doriguzzi C, et al. Muscle apoptosis in humans occurs in normal and denervated muscle, but not in myotonic dystrophy, dystrophinopathies or inflammatory disease. Neurogenetics. 1997;1:81–87. doi: 10.1007/s100480050012. [DOI] [PubMed] [Google Scholar]
- 6.Schoser BG, Wehling S, Blottner D. Cell death and apoptosis-related proteins in muscle biopsies of sporadic amyotrophic lateral sclerosis and polyneuropathy. Muscle Nerve. 2001;24:1083–1089. doi: 10.1002/mus.1114. [DOI] [PubMed] [Google Scholar]
- 7.Tews DS, Goebel HH. DNA fragmentation and BCL-2 expression in infantile spinal muscular atrophy. Neuromuscul Disord. 1996;6:265–273. doi: 10.1016/0960-8966(96)00018-1. [DOI] [PubMed] [Google Scholar]
- 8.Tews DS, Goebel HH. DNA-fragmentation and expression of apoptosis-related proteins in muscular dystrophies. Neuropathol Appl Neurobiol. 1997;23:331–338. [PubMed] [Google Scholar]
- 9.Danial NN, Korsmeyer SJ. Cell death: Critical control points. Cell. 2004;116:205–219. doi: 10.1016/s0092-8674(04)00046-7. [DOI] [PubMed] [Google Scholar]
- 10.Chao DT, Korsmeyer SJ. BCL-2 family: Regulators of cell death. Annu Rev Immunol. 1998;16:395–419. doi: 10.1146/annurev.immunol.16.1.395. [DOI] [PubMed] [Google Scholar]
- 11.Mikhailov V, Mikhailova M, Pulkrabek DJ, Dong Z, Venkatachalam MA, Saikumar P. Bcl-2 prevents Bax oligomerization in the mitochondrial outer membrane. J Biol Chem. 2001;276:18361–18374. doi: 10.1074/jbc.M100655200. [DOI] [PubMed] [Google Scholar]
- 12.Chao DT, Linette GP, Boise LH, White LS, Thompson CB, Korsmeyer SJ. Bcl-XL and Bcl-2 repress a common pathway of cell death. J Exp Med. 1995;182:821–828. doi: 10.1084/jem.182.3.821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Basanez G, Nechushtan A, Drozhinin O, et al. Bax, but not Bcl-xL, decreases the lifetime of planar phospholipid bilayer membranes at subnanomolar concentrations. Proc Natl Acad Sci USA. 1999;96:5492–5497. doi: 10.1073/pnas.96.10.5492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cartron PF, Moreau C, Oliver L, Mayat E, Meflah K, Vallette FM. Involvement of the N-terminus of Bax in its intracellular localization and function. FEBS Lett. 2002;512:95–100. doi: 10.1016/s0014-5793(02)02227-5. [DOI] [PubMed] [Google Scholar]
- 15.Desagher S, Martinou JC. Mitochondria as the central control point of apoptosis. Trends Cell Biol. 2000;10:369–377. doi: 10.1016/s0962-8924(00)01803-1. [DOI] [PubMed] [Google Scholar]
- 16.Hsu YT, Wolter KG, Youle RJ. Cytosol-to-membrane redistribution of Bax and Bcl-X(L) during apoptosis. Proc Natl Acad Sci USA. 1997;94:3668–3672. doi: 10.1073/pnas.94.8.3668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wolter KG, Hsu YT, Smith CL, Nechushtan A, Xi XG, Youle RJ. Movement of Bax from the cytosol to mitochondria during apoptosis. J Cell Biol. 1997;139:1281–1292. doi: 10.1083/jcb.139.5.1281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zha H, Aime-Sempe C, Sato T, Reed JC. Proapoptotic protein Bax heterodimerizes with Bcl-2 and homodimerizes with Bax via a novel domain (BH3) distinct from BH1 and BH2. J Biol Chem. 1996;271:7440–7444. doi: 10.1074/jbc.271.13.7440. [DOI] [PubMed] [Google Scholar]
- 19.Antonsson B, Montessuit S, Lauper S, Eskes R, Martinou JC. Bax oligomerization is required for channel-forming activity in liposomes and to trigger cytochrome c release from mitochondria. Biochem J. 2000;345 (Pt 2):271–278. [PMC free article] [PubMed] [Google Scholar]
- 20.Siu PM, Alway SE. Mitochondria-associated apoptotic signalling in denervated rat skeletal muscle. J Physiol. 2005;565:309–323. doi: 10.1113/jphysiol.2004.081083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Alway SE, Degens H, Krishnamurthy G, Chaudhrai A. Denervation stimulates apoptosis but not Id2 expression in hindlimb muscles of aged rats. J Gerontol A Biol Sci Med Sci. 2003;58:687–697. doi: 10.1093/gerona/58.8.b687. [DOI] [PubMed] [Google Scholar]
- 22.Tews DS, Goebel HH, Schneider I, Gunkel A, Stennert E, Neiss WF. DNA-fragmentation and expression of apoptosis-related proteins in experimentally denervated and reinnervated rat facial muscle. Neuropathol Appl Neurobiol. 1997;23:141–149. [PubMed] [Google Scholar]
- 23.Powers SK, Kavazis AN, DeRuisseau KC. Mechanisms of disuse muscle atrophy: Role of oxidative stress. Am J Physiol Regul Integr Comp Physiol. 2005;288:R337–R344. doi: 10.1152/ajpregu.00469.2004. [DOI] [PubMed] [Google Scholar]
- 24.Koesterer TJ, Dodd SL, Powers S. Increased antioxidant capacity does not attenuate muscle atrophy caused by unweighting. J Appl Physiol. 2002;93:1959–1965. doi: 10.1152/japplphysiol.00511.2002. [DOI] [PubMed] [Google Scholar]
- 25.Kondo H, Miura M, Itokawa Y. Oxidative stress in skeletal muscle atrophied by immobilization. Acta Physiol Scand. 1991;142:527–528. doi: 10.1111/j.1748-1716.1991.tb09191.x. [DOI] [PubMed] [Google Scholar]
- 26.Kondo H, Miura M, Nakagaki I, Sasaki S, Itokawa Y. Trace element movement and oxidative stress in skeletal muscle atrophied by immobilization. Am J Physiol. 1992;262:E583–E590. doi: 10.1152/ajpendo.1992.262.5.E583. [DOI] [PubMed] [Google Scholar]
- 27.Kondo H, Nakagaki I, Sasaki S, Hori S, Itokawa Y. Mechanism of oxidative stress in skeletal muscle atrophied by immobilization. Am J Physiol. 1993;265:E839–E844. doi: 10.1152/ajpendo.1993.265.6.E839. [DOI] [PubMed] [Google Scholar]
- 28.Kondo H, Miura M, Itokawa Y. Antioxidant enzyme systems in skeletal muscle atrophied by immobilization. Pflugers Arch. 1993;422:404–406. doi: 10.1007/BF00374299. [DOI] [PubMed] [Google Scholar]
- 29.Kondo H, Nishino K, Itokawa Y. Hydroxyl radical generation in skeletal muscle atrophied by immobilization. FEBS Lett. 1994;349:169–172. doi: 10.1016/0014-5793(94)00641-5. [DOI] [PubMed] [Google Scholar]
- 30.Lawler JM, Song W, Demaree SR. Hindlimb unloading increases oxidative stress and disrupts antioxidant capacity in skeletal muscle. Free Radic Biol Med. 2003;35:9–16. doi: 10.1016/s0891-5849(03)00186-2. [DOI] [PubMed] [Google Scholar]
- 31.Tews DS, Goebel HH, Schneider I, Gunkel A, Stennert E, Neiss WF. Expression of different isoforms of nitric oxide synthase in experimentally denervated and reinnervated skeletal muscle. J Neuropathol Exp Neurol. 1997;56:1283–1289. doi: 10.1097/00005072-199712000-00003. [DOI] [PubMed] [Google Scholar]
- 32.Degens H, Meessen NE, Wirtz P, Binkhorst RA. The development of compensatory hypertrophy in the plantaris muscle of the rat. Anat Anz. 1995;177:285–289. doi: 10.1016/S0940-9602(11)80203-7. [DOI] [PubMed] [Google Scholar]
- 33.Alway SE, Degens H, Krishnamurthy G, Smith CA. Potential role for Id myogenic repressors in apoptosis and attenuation of hypertrophy in muscles of aged rats. Am J Physiol Cell Physiol. 2002;283:C66–C76. doi: 10.1152/ajpcell.00598.2001. [DOI] [PubMed] [Google Scholar]
- 34.Rothermel B, Vega RB, Yang J, Wu H, Bassel-Duby R, Williams RS. A protein encoded within the Down syndrome critical region is enriched in striated muscles and inhibits calcineurin signaling. J Biol Chem. 2000;275:8719–8725. doi: 10.1074/jbc.275.12.8719. [DOI] [PubMed] [Google Scholar]
- 35.Siu PM, Bryner RW, Martyn JK, Alway SE. Apoptotic adaptations from exercise training in skeletal and cardiac muscles. FASEB J. 2004;18:1150–1152. doi: 10.1096/fj.03-1291fje. [DOI] [PubMed] [Google Scholar]
- 36.Siu PM, Pistilli EE, Butler DC, Alway SE. Aging influences cellular and molecular responses of apoptosis to skeletal muscle unloading. Am J Physiol Cell Physiol. 2005;288:C338–C349. doi: 10.1152/ajpcell.00239.2004. [DOI] [PubMed] [Google Scholar]
- 37.Siu PM, Alway SE. Id2 and p53 participate in apoptosis during unloading-induced muscle atrophy. Am J Physiol Cell Physiol. 2005;288:C1058–C1073. doi: 10.1152/ajpcell.00495.2004. [DOI] [PubMed] [Google Scholar]
- 38.Siu PM, Pistilli EE, Alway SE. Apoptotic responses to hindlimb suspension in gastrocnemius muscles from young adult and aged rats. Am J Physiol Regul Integr Comp Physiol. 2005;289:R1015–R1026. doi: 10.1152/ajpregu.00198.2005. [DOI] [PubMed] [Google Scholar]
- 39.Rokhlin OW, Glover RA, Taghiyev AF, et al. Bisindolylmaleimide IX facilitates tumor necrosis factor receptor family-mediated cell death and acts as an inhibitor of transcription. J Biol Chem. 2002;277:33213–33219. doi: 10.1074/jbc.M204612200. [DOI] [PubMed] [Google Scholar]
- 40.Smith PK, Krohn RI, Hermanson GT, et al. Measurement of protein using bicinchoninic acid. Anal Biochem. 1985;150:76–85. doi: 10.1016/0003-2697(85)90442-7. [DOI] [PubMed] [Google Scholar]
- 41.Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951;193:265–275. [PubMed] [Google Scholar]
- 42.Susin SA, Lorenzo HK, Zamzami N, et al. Molecular characterization of mitochondrial apoptosis-inducing factor. Nature. 1999;397:441–446. doi: 10.1038/17135. [DOI] [PubMed] [Google Scholar]
- 43.Esterbauer H, Schaur RJ, Zollner H. Chemistry and biochemistry of 4-hydroxynonenal, malonaldehyde and related aldehydes. Free Radic Biol Med. 1991;11:81–128. doi: 10.1016/0891-5849(91)90192-6. [DOI] [PubMed] [Google Scholar]
- 44.Borisov AB, Carlson BM. Cell death in denervated skeletal muscle is distinct from classical apoptosis. Anat Rec. 2000;258:305–318. doi: 10.1002/(SICI)1097-0185(20000301)258:3<305::AID-AR10>3.0.CO;2-A. [DOI] [PubMed] [Google Scholar]
- 45.Jejurikar SS, Marcelo CL, Kuzon WM., Jr Skeletal muscle denervation increases satellite cell susceptibility to apoptosis. Plast Reconstr Surg. 2002;110:160–168. doi: 10.1097/00006534-200207000-00027. [DOI] [PubMed] [Google Scholar]
- 46.Jin H, Wu Z, Tian T, Gu Y. Apoptosis in atrophic skeletal muscle induced by brachial plexus injury in rats. J Trauma. 2001;50:31–35. doi: 10.1097/00005373-200101000-00005. [DOI] [PubMed] [Google Scholar]
- 47.Olive M, Ferrer I. Bcl-2 and bax immunohistochemistry in denervation-reinnervation and necrosis-regeneration of rat skeletal muscles. Muscle Nerve. 2000;23:1862–1867. doi: 10.1002/1097-4598(200012)23:12<1862::aid-mus10>3.0.co;2-q. [DOI] [PubMed] [Google Scholar]
- 48.Tang H, Cheung WM, Ip FC, Ip NY. Identification and characterization of differentially expressed genes in denervated muscle. Mol Cell Neurosci. 2000;16:127–140. doi: 10.1006/mcne.2000.0864. [DOI] [PubMed] [Google Scholar]
- 49.Yoshimura K, Harii K. A regenerative change during muscle adaptation to denervation in rats. J Surg Res. 1999;81:139–146. doi: 10.1006/jsre.1998.5504. [DOI] [PubMed] [Google Scholar]
- 50.Primeau AJ, Adhihetty PJ, Hood DA. Apoptosis in heart and skeletal muscle. Can J Appl Physiol. 2002;27:349–395. doi: 10.1139/h02-020. [DOI] [PubMed] [Google Scholar]
- 51.Li M, Dalakas MC. Expression of human IAP-like protein in skeletal muscle: A possible explanation for the rare incidence of muscle fiber apoptosis in T-cell mediated inflammatory myopathies. J Neuroimmunol. 2000;106:1–5. doi: 10.1016/s0165-5728(99)00162-9. [DOI] [PubMed] [Google Scholar]
- 52.Monici MC, Toscano A, Girlanda P, Aguennouz M, Musumeci O, Vita G. Apoptosis in metabolic myopathies. Neuroreport. 1998;9:2431–2435. doi: 10.1097/00001756-199807130-00050. [DOI] [PubMed] [Google Scholar]
- 53.Sandri M, Minetti C, Pedemonte M, Carraro U. Apoptotic myonuclei in human Duchenne muscular dystrophy. Lab Invest. 1998;78:1005–1016. [PubMed] [Google Scholar]
- 54.Serdaroglu A, Gucuyener K, Erdem S, Kose G, Tan E, Okuyaz C. Role of apoptosis in Duchenne’s muscular dystrophy. J Child Neurol. 2002;17:66–68. doi: 10.1177/088307380201700120. [DOI] [PubMed] [Google Scholar]
- 55.Yan C, Ikezoe K, Nonaka I. Apoptotic muscle fiber degeneration in distal myopathy with rimmed vacuoles. Acta Neuropathol (Berl) 2001;101:9–16. doi: 10.1007/s004010000249. [DOI] [PubMed] [Google Scholar]
- 56.Ikezoe K, Yan C, Momoi T, et al. A novel congenital myopathy with apoptotic changes. Ann Neurol. 2000;47:531–536. [PubMed] [Google Scholar]
- 57.Koga M, Hiromatsu Y, Jimi A, Inoue Y, Nonaka K. Possible involvement of Fas-mediated apoptosis in eye muscle tissue from patients with thyroid-associated ophthalmopathy. Thyroid. 1998;8:311–318. doi: 10.1089/thy.1998.8.311. [DOI] [PubMed] [Google Scholar]
- 58.Vescovo G, Volterrani M, Zennaro R, et al. Apoptosis in the skeletal muscle of patients with heart failure: Investigation of clinical and biochemical changes. Heart. 2000;84:431–437. doi: 10.1136/heart.84.4.431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Yasuhara S, Perez ME, Kanakubo E, et al. Skeletal muscle apoptosis after burns is associated with activation of proapoptotic signals. Am J Physiol Endocrinol Metab. 2000;279:E1114–E1121. doi: 10.1152/ajpendo.2000.279.5.E1114. [DOI] [PubMed] [Google Scholar]
- 60.Girgenrath M, Dominov JA, Kostek CA, Miller JB. Inhibition of apoptosis improves outcome in a model of congenital muscular dystrophy. J Clin Invest. 2004;114:1635–1639. doi: 10.1172/JCI22928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Garcia-Saez AJ, Coraiola M, Dalla SM, Mingarro I, Menestrina G, Salgado J. Peptides derived from apoptotic Bax and Bid reproduce the poration activity of the parent full-length proteins. Biophys J. 2005;88:3976–3990. doi: 10.1529/biophysj.104.058008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Oakes SA, Scorrano L, Opferman JT, et al. Proapoptotic BAX and BAK regulate the type 1 inositol trisphosphate receptor and calcium leak from the endoplasmic reticulum. Proc Natl Acad Sci USA. 2005;102:105–110. doi: 10.1073/pnas.0408352102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Scorrano L, Oakes SA, Opferman JT, et al. BAX and BAK regulation of endoplasmic reticulum Ca2+: A control point for apoptosis. Science. 2003;300:135–139. doi: 10.1126/science.1081208. [DOI] [PubMed] [Google Scholar]
- 64.Bouillet P, Strasser A. BH3-only proteins - evolutionarily conserved proapoptotic Bcl-2 family members essential for initiating programmed cell death. J Cell Sci. 2002;115:1567–1574. doi: 10.1242/jcs.115.8.1567. [DOI] [PubMed] [Google Scholar]
- 65.Koseki T, Inohara N, Chen S, Nunez G. ARC, an inhibitor of apoptosis expressed in skeletal muscle and heart that interacts selectively with caspases. Proc Natl Acad Sci USA. 1998;95:5156–5160. doi: 10.1073/pnas.95.9.5156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Gustafsson AB, Tsai JG, Logue SE, Crow MT, Gottlieb RA. Apoptosis repressor with caspase recruitment domain protects against cell death by interfering with Bax activation. J Biol Chem. 2004;279:21233–21238. doi: 10.1074/jbc.M400695200. [DOI] [PubMed] [Google Scholar]
- 67.Sharpe JC, Arnoult D, Youle RJ. Control of mitochondrial permeability by Bcl-2 family members. Biochim Biophys Acta. 2004;1644:107–113. doi: 10.1016/j.bbamcr.2003.10.016. [DOI] [PubMed] [Google Scholar]
- 68.Kowaltowski AJ, Fenton RG, Fiskum G. Bcl-2 family proteins regulate mitochondrial reactive oxygen production and protect against oxidative stress. Free Radic Biol Med. 2004;37:1845–1853. doi: 10.1016/j.freeradbiomed.2004.09.005. [DOI] [PubMed] [Google Scholar]