Abstract
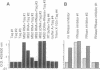
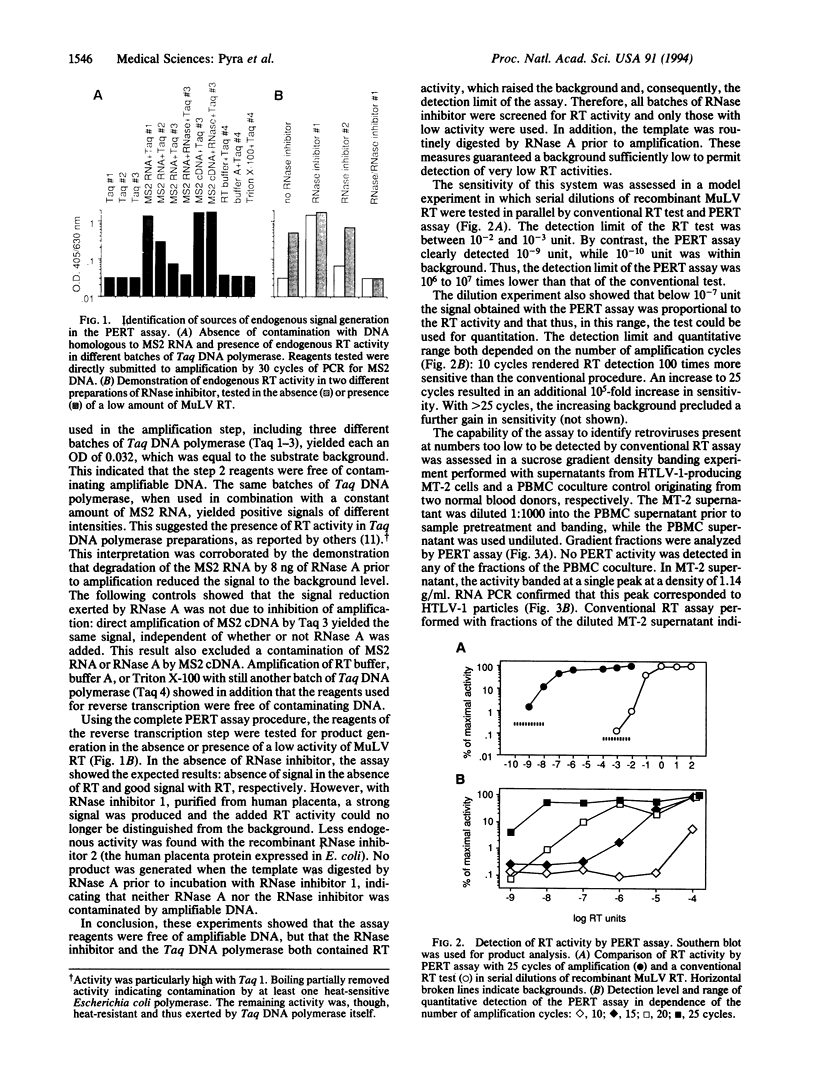
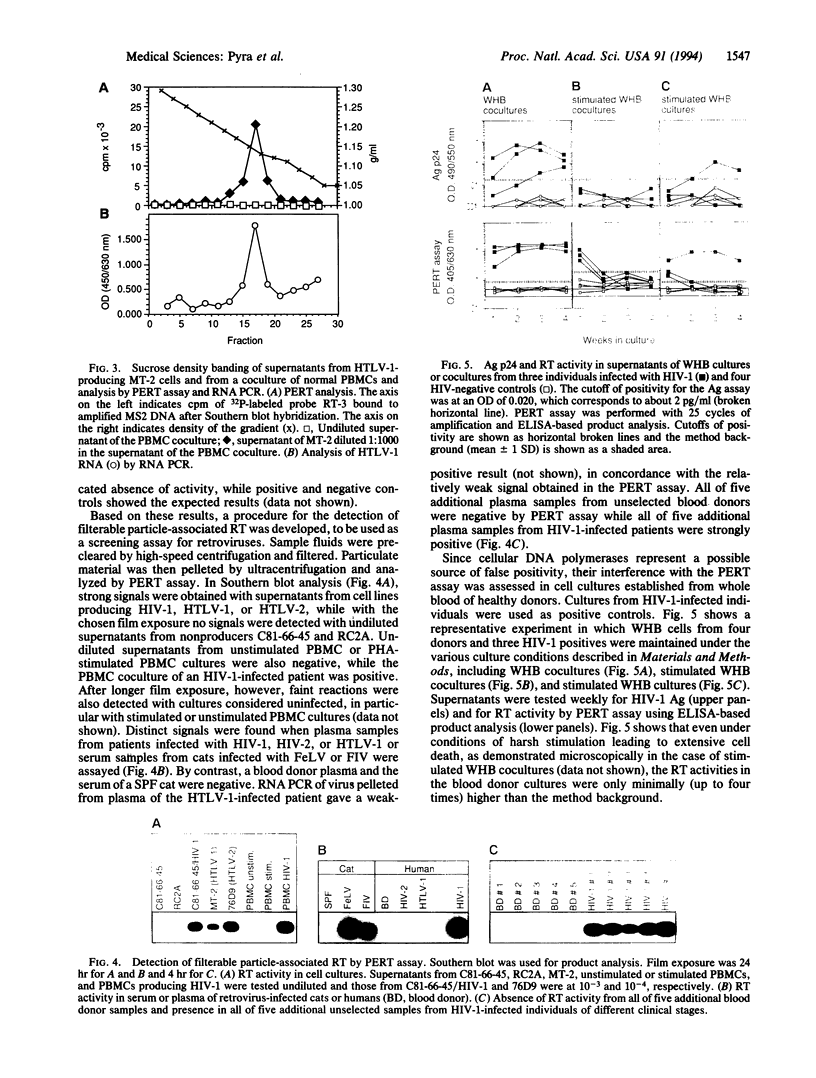
Reverse transcriptase (RT) is an indispensable component of infectious retroviruses. We have developed an ultrasensitive RT test in which RNA of bacteriophage MS2 serves as the template for RT-mediated cDNA synthesis. A fragment of the cDNA is selectively amplified by polymerase chain reaction and the amplification product is analyzed by Southern blot hybridization or enzyme immunoassay. The procedure was 10(6) to 10(7) times more sensitive than a conventional RT test and detected as little as 10(-9) unit of murine leukemia virus RT, which corresponded to 2.1 x 10(2) molecules, a number present in 3-11 virions. As a screening assay for filterable particle-associated RT, it was positive with supernatants from cell cultures producing human immunodeficiency virus (HIV) type 1 or human T-cell leukemia virus (HTLV) type 1 or 2, but was negative with nonproducer cultures. It was positive with plasma samples from all tested individuals infected with HIV-1, HIV-2, or HTLV-1 and sera from cats infected with feline leukemia virus or feline immunodeficiency virus. Control samples from blood donors or uninfected cats were negative. Density banding experiments with culture supernatants showed that the RT activity was associated with virus particles. The assay should detect all replication-competent retroviruses or similar agents. It may be used as a screening assay for such agents, for quantitation of the viral load, drug susceptibility testing of RT, and control of virus inactivation in biological products.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baltimore D. RNA-dependent DNA polymerase in virions of RNA tumour viruses. Nature. 1970 Jun 27;226(5252):1209–1211. doi: 10.1038/2261209a0. [DOI] [PubMed] [Google Scholar]
- Böni J., Schüpbach J. Sensitive and quantitative detection of PCR-amplified HIV-1 DNA products by an enzyme linked immunoassay following solution hybridization with two differently labelled oligonucleotide probes. Mol Cell Probes. 1993 Oct;7(5):361–371. doi: 10.1006/mcpr.1993.1054. [DOI] [PubMed] [Google Scholar]
- Donehower L. A., Bohannon R. C., Ford R. J., Gibbs R. A. The use of primers from highly conserved pol regions to identify uncharacterized retroviruses by the polymerase chain reaction. J Virol Methods. 1990 Apr;28(1):33–46. doi: 10.1016/0166-0934(90)90085-t. [DOI] [PubMed] [Google Scholar]
- Jones M. D., Foulkes N. S. Reverse transcription of mRNA by Thermus aquaticus DNA polymerase. Nucleic Acids Res. 1989 Oct 25;17(20):8387–8388. doi: 10.1093/nar/17.20.8387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krakower J. M., Barbacid M., Aaronson S. A. Radioimmunoassay for mammalian type C viral reverse transcriptase. J Virol. 1977 May;22(2):331–339. doi: 10.1128/jvi.22.2.331-339.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee Y. S. A comparison of reverse transcriptase and antigen capture assays for the detection of HIV. J Virol Methods. 1988 May;20(1):89–93. doi: 10.1016/0166-0934(88)90044-4. [DOI] [PubMed] [Google Scholar]
- Panet A., Baltimore D., Hanafusa T. Quantitation of avian RNA tumor virus reverse transcriptase by radioimmunoassay. J Virol. 1975 Jul;16(1):146–152. doi: 10.1128/jvi.16.1.146-152.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poiesz B. J., Ruscetti F. W., Mier J. W., Woods A. M., Gallo R. C. T-cell lines established from human T-lymphocytic neoplasias by direct response to T-cell growth factor. Proc Natl Acad Sci U S A. 1980 Nov;77(11):6815–6819. doi: 10.1073/pnas.77.11.6815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saiki R. K., Scharf S., Faloona F., Mullis K. B., Horn G. T., Erlich H. A., Arnheim N. Enzymatic amplification of beta-globin genomic sequences and restriction site analysis for diagnosis of sickle cell anemia. Science. 1985 Dec 20;230(4732):1350–1354. doi: 10.1126/science.2999980. [DOI] [PubMed] [Google Scholar]
- Sarngadharan M. G., Robert-Guroff M., Gallo R. C. DNA polymerases of normal and neoplastic mammalian cells. Biochim Biophys Acta. 1978 Dec 11;516(4):419–487. doi: 10.1016/0304-419x(78)90019-7. [DOI] [PubMed] [Google Scholar]
- Stromberg K., Hurley N. E., Davis N. L., Rueckert R. R., Fleissner E. Structural studies of avian myeloblastosis virus: comparison of polypeptides in virion and core component by dodecyl sulfate-polyacrylamide gel electrophoresis. J Virol. 1974 Feb;13(2):513–528. doi: 10.1128/jvi.13.2.513-528.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Temin H. M., Mizutani S. RNA-dependent DNA polymerase in virions of Rous sarcoma virus. Nature. 1970 Jun 27;226(5252):1211–1213. doi: 10.1038/2261211a0. [DOI] [PubMed] [Google Scholar]