Abstract
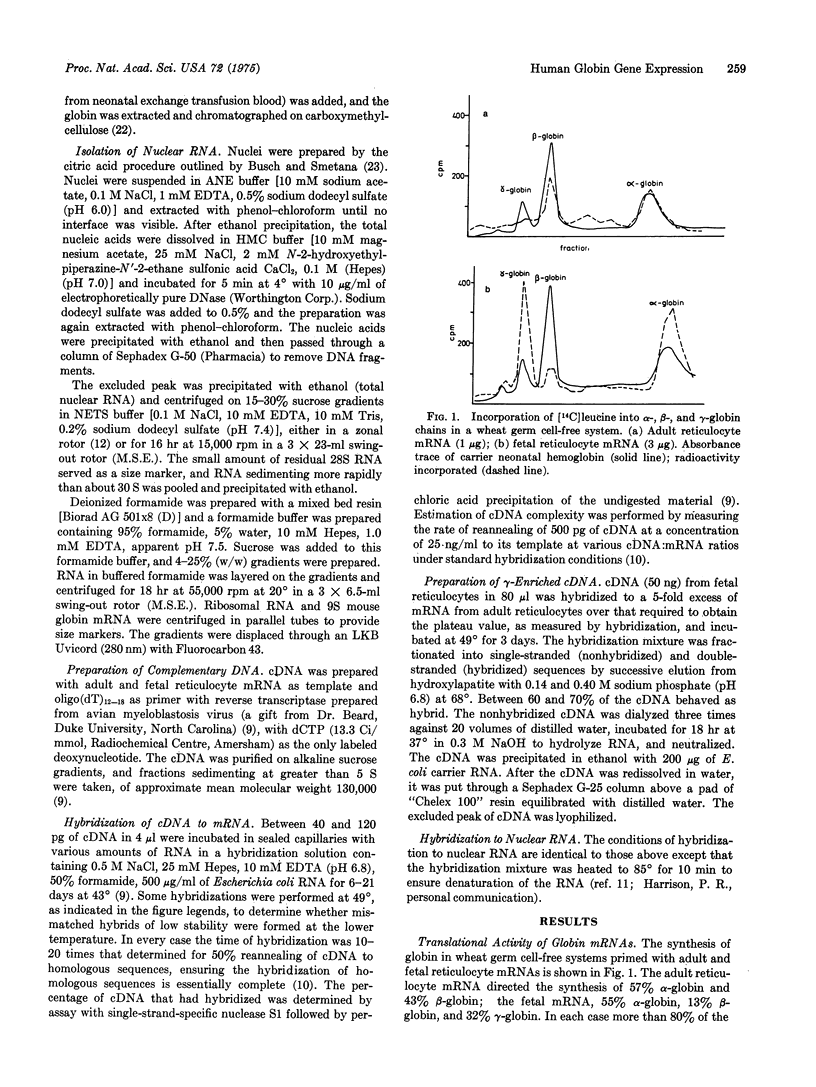
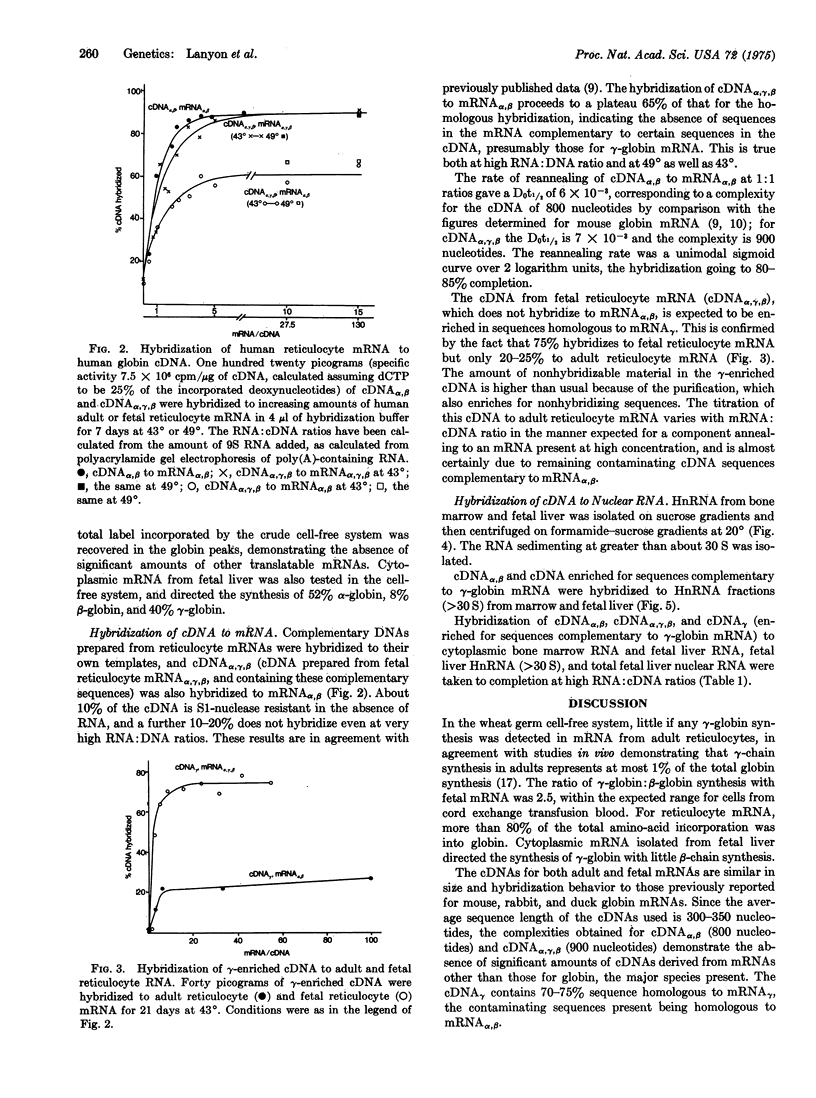
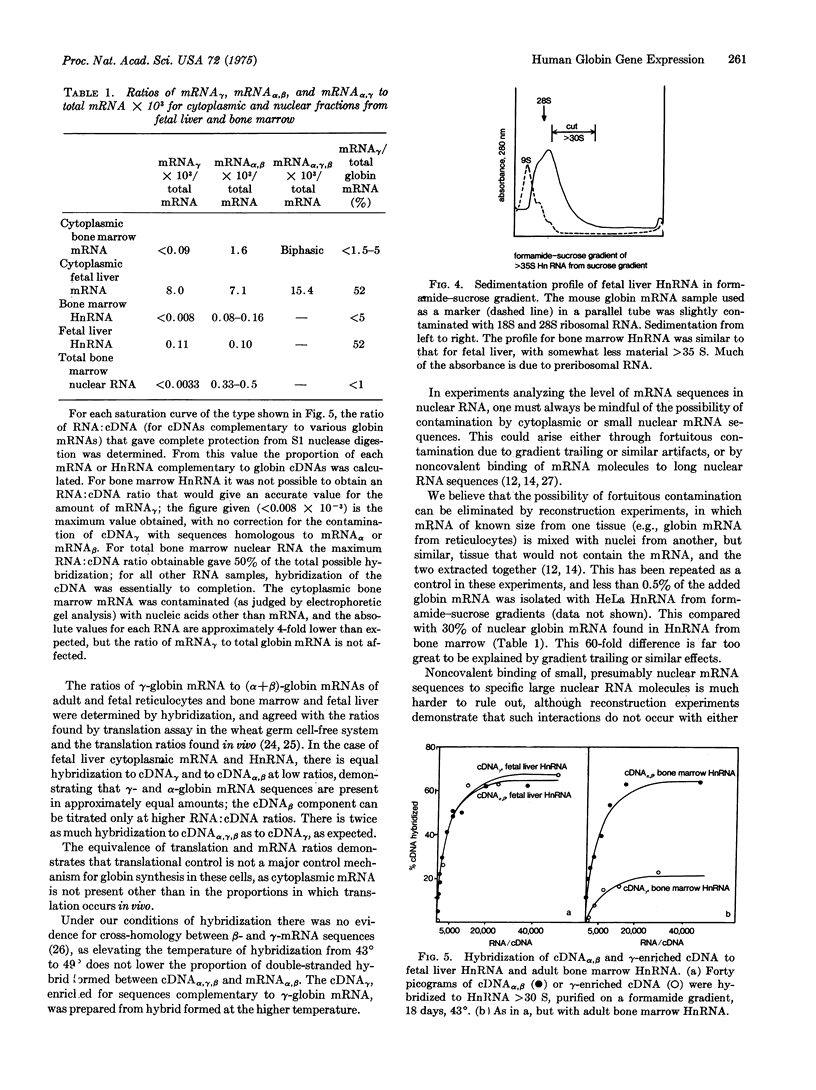
During embryonic development there is a transition from embryonic and fetal to adult beta-type globin chains. The high-molecular-weight RNA found in nuclei from embryonic and adult human erythropoietic tissues, fetal liver, and bone marrow, have been investigated for the presence of gamma(fetal)- and beta(adult)-globin messenger RNA sequences by molecular hybridization. Unlike alpha- and beta-globin mRNA sequences, gamma-globin mRNA sequences are absent from both total and high-molecular-weight nuclear RNA isolated from adult bone marrow. The amount of cytoplasmic gamma-globin mRNA is proportional to the level of gamma-chain synthesis, demonstrating that translational control is not a major control mechanism in the expression of globin genes. Since the gamma-, delta-, and beta-globin genes are known to be closely linked genetically, transcriptional control can discriminate between similar gene sequences that are spatially adjacent to one another.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aviv H., Leder P. Purification of biologically active globin messenger RNA by chromatography on oligothymidylic acid-cellulose. Proc Natl Acad Sci U S A. 1972 Jun;69(6):1408–1412. doi: 10.1073/pnas.69.6.1408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bank A., Terada M., Metafora S., Dow L., Marks P. A. In vitro synthesis of DNA components of human genes for globins. Nat New Biol. 1972 Feb 9;235(58):167–169. doi: 10.1038/newbio235167a0. [DOI] [PubMed] [Google Scholar]
- Bishop J. O., Rosbash M. Reiteration frequency of duck haemoglobin genes. Nat New Biol. 1973 Feb 14;241(111):204–207. doi: 10.1038/newbio241204a0. [DOI] [PubMed] [Google Scholar]
- Brown I. R., Church R. B. Transcription of nonrepeated DNA during mouse and rabbit development. Dev Biol. 1972 Sep;29(1):73–84. doi: 10.1016/0012-1606(72)90045-0. [DOI] [PubMed] [Google Scholar]
- Clegg J. B., Naughton M. A., Weatherball D. J. Abnormal human haemoglobins. Separation and characterization of the alpha and beta chains by chromatography, and the determination of two new variants, hb Chesapeak and hb J (Bangkok). J Mol Biol. 1966 Aug;19(1):91–108. doi: 10.1016/s0022-2836(66)80052-9. [DOI] [PubMed] [Google Scholar]
- Darnell J. E., Jelinek W. R., Molloy G. R. Biogenesis of mRNA: genetic regulation in mammalian cells. Science. 1973 Sep 28;181(4106):1215–1221. doi: 10.1126/science.181.4106.1215. [DOI] [PubMed] [Google Scholar]
- Forget B. G., Benz E. J., Jr, Skoultchi A., Baglioni C., Housman D. Absence of messenger RNA for beta globin chain in beta(0) thalassaemia. Nature. 1974 Feb 8;247(5440):379–381. doi: 10.1038/247379a0. [DOI] [PubMed] [Google Scholar]
- Harrison P. R., Birnie G. D., Hell A., Humphries S., Young B. D., Paul J. Kinetic studies of gene frequency. I. Use of a DNA copy of reticulocyte 9 S RNA to estimate globin gene dosage in mouse tissues. J Mol Biol. 1974 Apr 25;84(4):539–554. doi: 10.1016/0022-2836(74)90115-6. [DOI] [PubMed] [Google Scholar]
- Harrison P. R., Hell A., Birnie G. D., Paul J. Evidence for single copies of globin genes in the mouse genome. Nature. 1972 Sep 22;239(5369):219–221. doi: 10.1038/239219a0. [DOI] [PubMed] [Google Scholar]
- Huisman T. H., Wrightstone R. N., Wilson J. B., Schroeder W. A., Kendall A. G. Hemoglobin Kenya, the product of fusion of amd polypeptide chains. Arch Biochem Biophys. 1972 Dec;153(2):850–853. doi: 10.1016/0003-9861(72)90408-0. [DOI] [PubMed] [Google Scholar]
- Imaizumi T., Diggelmann H., Scherrer K. Demonstration of globin messenger sequences in giant nuclear precursors of messenger RNA of avian erythroblasts. Proc Natl Acad Sci U S A. 1973 Apr;70(4):1122–1126. doi: 10.1073/pnas.70.4.1122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lane C. D., Gregory C. M., Iyazumi T., Scherrer K. The use of the Xenopus oocyte to prove the existence of messenger sequences in high molecular weight RNA. Nat New Biol. 1973 May 16;243(124):78–78. [PubMed] [Google Scholar]
- Nienhuis A. W., Canfield P. H., Anderson W. F. Hemoglobin messenger RNA from human bone marrow. Isolation and translation in homozygous and heterozygous beta-thalassemia. J Clin Invest. 1973 Jul;52(7):1735–1745. doi: 10.1172/JCI107355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Packman S., Aviv H., Ross J., Leder P. A comparison of globin genes in duck reticulocytes and liver cells. Biochem Biophys Res Commun. 1972 Nov 1;49(3):813–819. doi: 10.1016/0006-291x(72)90483-4. [DOI] [PubMed] [Google Scholar]
- Perry R. P., La Torre J., Kelley D. E., Greenberg J. R. On the lability of poly(A) sequences during extraction of messenger RNA from polyribosomes. Biochim Biophys Acta. 1972 Mar 14;262(2):220–226. doi: 10.1016/0005-2787(72)90236-5. [DOI] [PubMed] [Google Scholar]
- Roberts B. E., Paterson B. M. Efficient translation of tobacco mosaic virus RNA and rabbit globin 9S RNA in a cell-free system from commercial wheat germ. Proc Natl Acad Sci U S A. 1973 Aug;70(8):2330–2334. doi: 10.1073/pnas.70.8.2330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ross J., Aviv H., Scolnick E., Leder P. In vitro synthesis of DNA complementary to purified rabbit globin mRNA (RNA-dependent DNA polymerase-reticulocyte-hemoglobin-density gradient centrifugation-oligo(dT) primer). Proc Natl Acad Sci U S A. 1972 Jan;69(1):264–268. doi: 10.1073/pnas.69.1.264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruiz-Carrilo A., Beato M., Schutz G., Feigelson P., Allfrey V. G. Cell-free translation of the globin message within polydisperse high-molecular-weight ribonucleic acid of avian erythrocytes. Proc Natl Acad Sci U S A. 1973 Dec;70(12):3641–3645. doi: 10.1073/pnas.70.12.3641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scherrer K., Marcaud L. Messenger RNA in avian erythroblasts at the transcriptional and translational levels and the problem of regulation in animal cells. J Cell Physiol. 1968 Oct;72(2 Suppl):181+–181+. doi: 10.1002/jcp.1040720413. [DOI] [PubMed] [Google Scholar]
- Sheldon R., Kates J., Kelley D. E., Perry R. P. Polyadenylic acid sequences on 3' termini of vaccinia messenger ribonucleic acid and mammalian nuclear and messenger ribonucleic acid. Biochemistry. 1972 Sep 26;11(20):3829–3834. doi: 10.1021/bi00770a023. [DOI] [PubMed] [Google Scholar]
- Stevens R. H., Williamson A. R. Specific IgG mRNA molecules from myeloma cells in heterogeneous nuclear and cytoplasmic RNA containing poly-A. Nature. 1972 Sep 15;239(5368):143–146. doi: 10.1038/239143a0. [DOI] [PubMed] [Google Scholar]
- Verma I. M., Temple G. F., Fan H., Baltimore D. In vitro synthesis of DNA complementary to rabbit reticulocyte 10S RNA. Nat New Biol. 1972 Feb 9;235(58):163–167. doi: 10.1038/newbio235163a0. [DOI] [PubMed] [Google Scholar]
- Williamson R., Drewienkiewicz C. E., Paul J. Globin messenger sequences in high molecular weight RNA from embryonic mouse liver. Nat New Biol. 1973 Jan 17;241(107):66–68. doi: 10.1038/newbio241066a0. [DOI] [PubMed] [Google Scholar]
- Williamson R., Morrison M., Lanyon G., Eason R., Paul J. Properties of mouse globin messenger ribonucleic acid and its preparation in milligram quantities. Biochemistry. 1971 Aug 3;10(16):3014–3021. doi: 10.1021/bi00792a005. [DOI] [PubMed] [Google Scholar]
- Wood W. G., Weatherall D. J. Haemoglobin synthesis during human foetal development. Nature. 1973 Jul 20;244(5412):162–165. doi: 10.1038/244162a0. [DOI] [PubMed] [Google Scholar]
- Young B. D., Harrison P. R., Gilmour R. S., Birnie G. D., Hell A., Humphries S., Paul J. Kinetic studies of gene frequency. II. Complexity of globin complementary DNA and its hybridization characteristics. J Mol Biol. 1974 Apr 25;84(4):555–568. doi: 10.1016/0022-2836(74)90116-8. [DOI] [PubMed] [Google Scholar]


