Abstract
Decapentaplegic (Dpp), a Drosophila morphogen signaling protein, transfers directly at synapes made at sites of contact between cells that produce Dpp and cytonemes that extend from recipient cells. The Dpp that cytonemes receive moves together with activated receptors toward the recipient cell body in motile puncta. Genetic loss-of-function conditions for diaphanous, shibire, neuroglian and capricious perturbed cytonemes by reducing their number or only the synapses they make with cells they target; and reduced cytoneme-mediated transport of Dpp and Dpp signaling. These experiments provide direct evidence that cells use cytonemes to exchange signaling proteins, that cytoneme-based exchange is essential for signaling and normal development, and that morphogen distribution and signaling can be contact-dependent, requiring cytoneme synapses.
Keywords: Drosophila cytonemes, Dpp signaling, morphogen, synapse
In many contexts during development, cell fate is determined by morphogen signaling proteins. The Drosophila wing imaginal disc, for instance, expresses the morphogen Decapentaplegic (Dpp), a transforming growth factor-β family member that regulates the fate, proliferation and patterning of its cells (reviewed in (1, 2). The disc expresses Dpp in a stripe of cells alongside the anterior/posterior (A/P) compartment border, and Dpp disperses across the disc to form exponential concentration gradients to either side that regulate target genes in adjacent cells in a concentration-dependent manner. Whereas the dispersion of Dpp across the disc and the functional importance of its concentration gradients are well established, the mechanism that moves Dpp from producing to target cells is not.
We tested the model that morphogens are transported along specialized signaling filopodia (cytonemes) that receive protein released at sites where producing and receiving cells contact each other (3). Cytonemes are on both the apical and basal surfaces of wing disc cells. Apical cytonemes that orient toward Dpp-producing disc cells contain the Dpp receptor Thickveins (Tkv), and cytoneme shape, orientation and distribution depend on expression of Dpp (3–5). There are basal cytonemes that contain Hedgehog (Hh) and the Interference Hedgehog (Ihog) proteins (6, 7). Hh is also present in short cytonemes that extend from Hh-producing cells in the female germline stem cell niche (8). These correlations are suggestive, but they do not establish that cytonemes mediate transfers of signaling proteins from producing to target cells, or that such transfers, if they occur, are required for signaling.
The wing disc has associated trachea whose development depends in part on signaling from the disc (9). Larval trachea form an interconnected network of oxygen carrying tubes; one, the transverse connective (TC) of Tr2, is bound to the wing disc (Fig. 1A). During the third larval instar (L3), Branchless, (the fly Fibroblast Growth Factor (FGF)) produced by a group of disc cells induces a new branch, the Air Sac Primordium (ASP), to grow from the TC (9). The ASP is juxtaposed to the basal surface of the wing disc columnar epithelium; it is a monolayered epithelial tube. At the late L3 stage, the ASP has many cytonemes that extend toward the disc (Fig. 1B). Cells at the ASP tip extend long (≥30 μm) cytonemes that contain the FGF receptor Breathless (FGFR) and appear to touch FGF-producing disc cells. The presence and orientation of these cytonemes are dependent on FGF (5, 9). The late L3 ASP also has shorter cytonemes that contain Tkv and that extend from its lateral flank toward Dpp-expressing disc cells (5).
Figure 1. Dpp produced in the wing disc signals to disc-associated tracheal cells.
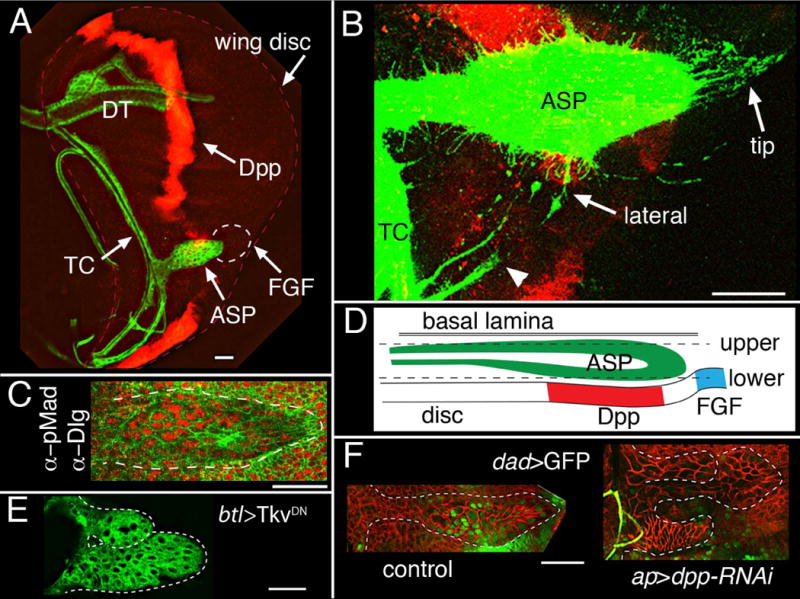
(A) Projection image of a 3rd instar wing disc (outlined with dashed red line) showing disc-associated trachea (marked with green; membrane tethered GFP) and Dpp-expressing disc cells (red, marked by antibody against LacZ that was expressed in the dpp domain). TC (transverse connective), DT (dorsal trunk) and ASP are labeled. Dotted circle indicates area of disc that expresses FGF. (B) Expression of CD8:RFP marks dpp-expressing disc cells (red); expression of CD8:GFP in trachea (lexO-CD8:GFP, UAS-CD8:RFP/+; btl-LexA/+; dpp-Gal4/+) marks cytonemes extending from ASP tip, from the lateral, medial region of the ASP, and from the TC (arrowhead), showing that some ASP and TC cytonemes orient toward Dpp-expressing cells. This plane of focus does not detect all dpp-expressing cells due to folds in the disc near the A/P organizer, but it did detect many dpp-expressing anterior cells that are in the plane of focus as “scattered” in the A compartment region between the ASP and TC. (C) staining an ASP with antibody to phosphorylated MAD (red) to show Dpp signaling in the medial region. Antibody to Dlg (Discs large, green) marks cell outlines in ASP (bounded by white dashed line) and discs. (D) Cartoon of a sagittal ASP section depicting the position of the disc epithelium and basal lamina relative to the ASP in the late L3; dashed lines represent approximate locations of the upper and lower optical sections in all Figures. (E) Over-expression of TkvDN in trachea (btl-Gal4) generated bifurcated, abnormally shaped ASPs. (F) dpp-RNAi expression in the dorsal compartment of the disc (ap-Gal4 tub-Gal80ts) reduced Dad expression (Dad-nlsGFP; green) in disc-associated trachea (right panel) compared to control (ap-Gal4 tub-Gal80ts Dad-nlsGFP) ASP (left panel); abnormal ASP growths are indicated by white dotted lines; cells are marked with α-Dlg staining (red); both panels show lower layer of ASP. Orientation of discs in all figures: anterior, left; dorsal, down. Conditions of Gal80ts inactivation for (E, F) are described in SOM and Table S1. Scale bars, 30 μm.
In the wing disc, Dpp induces several changes in responding cells: induction of Daughters against Dpp (Dad) expression (10), increased phosphorylation of the Mothers against dpp protein (pMad) (11), and decreased tkv expression (11). Dpp signal transduction does not change expression of the other Dpp receptor subunit Punt (Put). Elevated Dad expression, increased pMad and decreased tkv expression were observed in the ASP, presumably due to Dpp signaling, and their abundance indicates that Dpp signal transduction is probably higher in the lower layer cells that face the disc epithelium than in the cells that are further away in the upper layer (Figs. 1C,D, S1A–D; Table S1). Put expression was uniform. Dpp expression was not detected in the TC or ASP (Figs. 1A, S1E). These results show that Dpp signal transduction in the ASP inversely correlates with distance from Dpp-expressing cells in the wing disc.
Over-expressing dominant negative forms of Tkv or Put, or Dad (which negatively regulates Dpp signaling) in the trachea generated abnormally shaped ASPs and reduced Dpp signaling in the ASP (Figs. 1E, S1F–H; Tables S2, S3). Expression of dpp-RNAi in the wing disc generated similar phenotypes and reduced Dpp signaling (Fig. 1F, Table S3), indicating that the wing disc is the source of the Dpp that activates signal transduction in the ASP, and establishing that Dpp signaling from the disc is essential for normal ASP development.
ASP cytonemes receive Dpp from the wing disc
To investigate the basis for disc-dependent Dpp signaling in the ASP, we over-expressed an isoform of Dpp coupled to Green Fluorescent Protein (Dpp:GFP) (12, 13) in the disc dpp-expression domain (see SOM). GFP fluorescence was detected both in Dpp-expressing disc cells and in the ASP. Amounts of Dpp:GFP in the ASP were highest in the medial region of ASP nearest the Dpp-expressing disc cells and in the lower layer (Figs. 1D, 2A, Table S1), showing that Dpp:GFP produced by the wing disc distributed to the ASP in a manner that correlates with amonts of Dpp signal transduction (Figs. 1C, S1A–D). To examine the subcellular localization of marked Dpp in the ASP, we expressed Dpp coupled to mCherry fluorescent protein (Dpp:Cherry) (5) in the disc dpp-expression domain and Dpp signaling was monitored in unfixed, “live” preparations with a transgene that expresses nuclear-localized GFP (nGFP) under Dad control. Dpp:Cherry puncta were observed in multiple optical sections of ASP cells with strongly-marked GFP-positive nuclei (Fig. 2B,B′); the presence of Dpp:Cherry puncta at apical positions (Fig. 2B″) indicated that Dpp:Cherry had likely been taken up from the disc by these ASP cells.
Figure 2. The ASP takes up Dpp; and ASP cytonemes contain activated Tkv receptor.
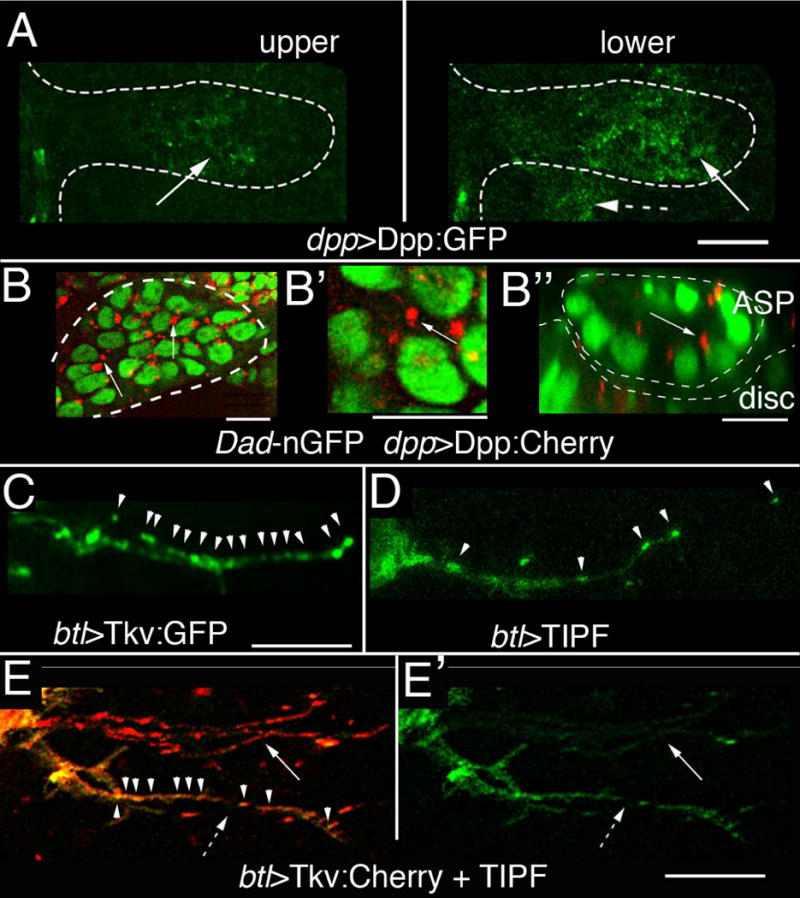
(A) Dpp:GFP expressed in the disc dpp domain (dpp-LexA lexO-Dpp:GFP; dashed arrow) is present (arrows) in the upper and lower ASP layers in this unfixed preparation. ASP is outlined by white dotted lines. (B–B″) Dpp:Cherry expressed in the disc dpp domain (dpp-Gal4/UAS-Dpp:Cherry, Dad-nGFP/tub-Gal80ts) was detected as intracellular puncta (arrows) in ASP cells that also induce Dad expression. ASP outline is marked by white line (B, B′, sagittal sections; B″, transverse section). (C) Expression of Tkv:GFP (btl-Gal4 UAS-Tkv:GFP) marks puncta (arrowheads) in ASP cytoneme. (D) Expression of TIPF (btl-Gal4 UAS-TIPF) marks puncta in ASP cytoneme. (E, E′) TIPF (green) and Tkv:Cherry fluorescence (btl-Gal4/UAS-Tkv:Cherry; tub-Gal80ts/UAS-TIPF) co-localizes (arrowheads) in puncta in some but not all ASP cytonemes. (arrow) cytoneme with Tkv:Cherry only; (dashed arrow) cytoneme with both TIPF and Tkv:Cherry; left panel (merge); right panel (TIPF only). Gal80ts inactivation for (B, E) was for 6–8 hrs in mid L3, followed by incubation at 25°C for 6–12 hrs. Scale bars, 10 μm, except for (A), 30 μm.
Whereas most tip cytonemes extended toward the region of the disc that expresses FGF (5, 9), some TC and lateral cytonemes extended toward Dpp-expressing disc cells (Fig. 1B). Expression of Tkv:GFP marked puncta in these cytonemes (Fig. 2C). To determine if activated Tkv was present in cytonemes, we over-expressed a variant of Tkv (TIPF) that fluoresces only in the phosphorylated state and that has been used to monitor receptor activation for Dpp or BMP signaling (14). ASP cytonemes with bright fluorescent puncta were present under conditions of TIPF over-expression (Fig. 2D). Expression of Tkv:Cherry and TIPF together in the TC and ASP generated puncta with both green (TIPF) and red (Tkv:Cherry) fluorescence, indicating that Tkv in these puncta had been activated (Fig. 2E,E′). We propose that the presence of activated Tkv indicates that these cytonemes had received Dpp. The presence of cytonemes with only red fluorescence suggests that not all the cytonemes had received Dpp.
To further validate and characterize Dpp reception, ASPs were marked with either CD8:Cherry (mCherry fused to the extracellular and transmembrane domains of the mouse lymphocyte protein CD8), Tkv:Cherry or FGFR:Cherry and Dpp:GFP was expressed in the disc dpp domain in a pulse during L3 (see SOM). The ASP grows from the TC on the anterior side of the disc and extends posteriorly across the stripe of Dpp-expressing cells by late L3 (9) (Fig. 3A). At “mid” or “late” stages, animals that expressed CD8:Cherry and Dpp:GFP had long ASP tip cytonemes marked with Cherry fluorescence that oriented toward FGF-expressing disc cells. These cytonemes had no apparent GFP fluorescence (Fig. 3B). Lateral ASP cytonemes that projected toward Dpp-expressing disc cells were also visible. These lateral cytonemes had both Cherry and GFP fluorescence (Fig. 3B, B′), indicating that Dpp:GFP had been received by these cytonemes. Dpp:GFP in puncta “free” from either cells or cytonemes was not detected.
Figure 3. Tkv-containing cytonemes transport Dpp.
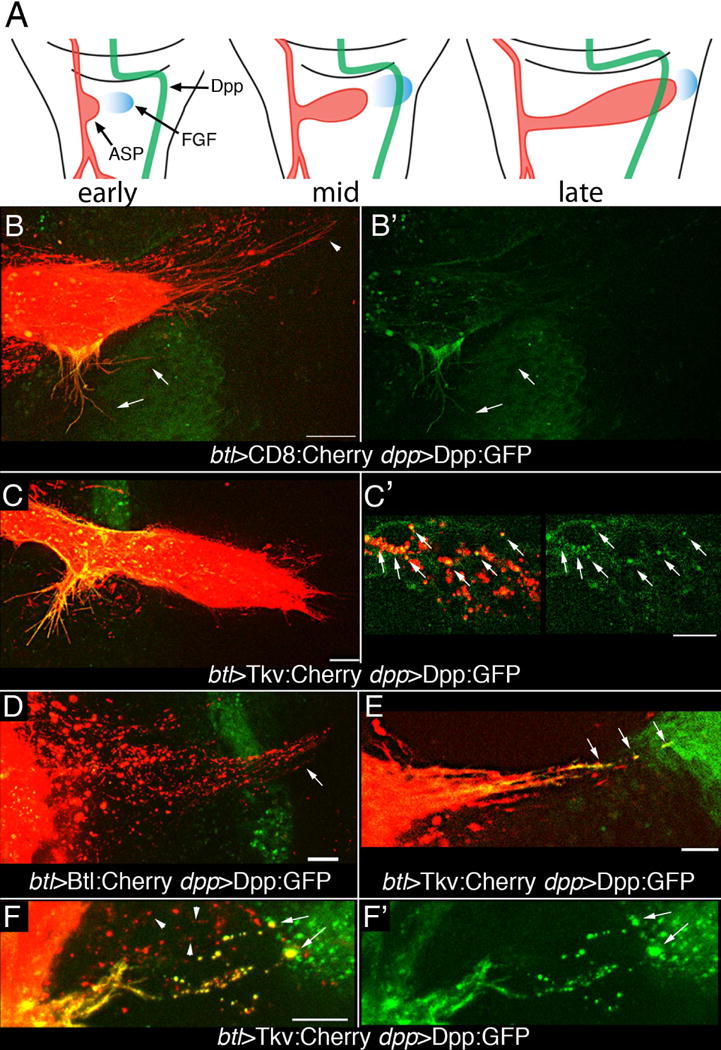
(A) Drawings of three 3rd instar stages depict growth and development of the ASP (red) relative to wing disc cells expressing Dpp (green) and FGF (blue). (B, B′) Expression of CD8:Cherry in the ASP and Dpp:GFP in the dpp domain of the disc (btl-Gal4 UAS-CD8:Cherry dpp-LHG/lexO-Dpp:GFP) marks the ASP and ASP cytonemes (red) and dpp-expressing disc cells (green). GFP fluorescence is in lateral ASP cytonemes (arrows) and in lower medial region of ASP, but not in tip ASP cytonemes (arrowhead). Left panel (merge), right panel (GFP). (C, C′) Expression of Tkv:Cherry in the ASP and Dpp:GFP in the in the dpp domain of the disc (btl-Gal4/UAS-Tkv:Cherry; dpp-LHG/lexO-Dpp:GFP) marks the ASP and lateral ASP cytonemes (red), but few tip cytonemes; lateral Tkv-containing ASP cytonemes and the medial region of the ASP have received Dpp:GFP (green) (C). Dpp:GFP and Tkv:Cherry colocalize in puncta in ASP cells (arrows, C′). (D) FGFR:Cherry expressed in ASP and Dpp:GFP in the dpp domain of the disc (btl-Gal4/UAS-Btl:Cherry dpp-LHG/lexO-Dpp:GFP) marks puncta in ASP tip cytonemes (arrow) that project beyond Dpp-expressing disc cells (green); no localization of FGFR:Cherry with Dpp:GFP was apparent in tip cytonemes. (E, F) Only cytonemes marked with Tkv:Cherry that appear to contact Dpp:GFP expressing disc cells (btl-Gal4 UAS-Tkv:Cherry; dpp-LHG/lexO-Dpp:GFP) have GFP fluorescence in puncta and at their tips (arrows). Cytonemes that do not appear to make contact do not have GFP fluorescence at their tips or in their Tkv-containing puncta (F, arrowheads) lack GFP fluorescence. (F) merge; (F′) Dpp:GFP. Animals were raised at 18°C to minimize transgene expression and were incubated at 22–25°C for 12–16 hrs prior to analysis. Scale bars, 10 μm.
ASPs marked with Tkv:Cherry provided evidence that Dpp transport by cytonemes is associated with its receptor. “Late” stage ASPs that expressed Tkv:Cherry had Dpp:GFP present in their medial region and in lateral cytonemes that extended from these cells, but there were few Tkv:Cherry-marked tip cytonemes and Dpp:GFP was present in much lower amounts in the distal ASP cells (Fig. 3C). Some of the Dpp:GFP present in the medial ASP cells was associated with Tkv puncta (Fig. 3C′). These images show that Dpp:GFP appears to move from the disc and be taken up by tracheal cells.
In “mid” stage ASPs that expressed FGFR:Cherry and whose tip had not grown beyond the Dpp-expressing zone of the disc, FGFR:Cherry-marked tip cytonemes extended over Dpp-expressing disc cells toward the cells that expressed FGF (Fig. 3D). No Dpp:GFP puncta localized with the FGFR:Cherry-marked cytonemes. The absence of Dpp:GFP in the FGFR:Cherry-containing tip cytonemes is consistent with the localization of the FGFR and Tkv receptors to different cytonemes (5), and suggests that FGF and Dpp reception may be receptor-dependent and specific for cytonemes that contain FGFR or Tkv, respectively.
To better understand cytoneme-mediated movement of Dpp, we analyzed “early” and “mid” stage preparations that had Tkv:Cherry expressed in trachea and Dpp:GFP expressed in the disc. Dpp source cells are distal to the ASP at these stages. Long, Tkv:Cherry-marked cytonemes extended toward Dpp-expressing disc cells (Fig. 3E,F). These cytonemes contained motile puncta (movie S1). Some cytonemes had both Tkv:Cherry and Dpp:GFP fluorescence and had brightly fluorescent ends that localized with Dpp:GFP; these images suggest that these cytonemes contact Dpp-expressing disc cells. Not all cytonemes had both Tkv:Cherry and Dpp:GFP, suggesting that some but not all cytonemes had received Dpp:GFP. These images are consistent with the patterns of TIPF fluorescence (Fig. 2E). The presence of Dpp:GFP in tracheal cytonemes and the apparent contacts of cytonemes with Dpp-producing disc cells suggest that the Dpp:GFP may move from the disc to the tracheal cells by direct transfer at sites of cytoneme contact.
Cytonemes synapse with wing disc cells
The cytoneme model of signaling protein dispersion posits that distant cells contact directly despite their physical separation. To probe the apparent contacts at higher resolution, we adapted the GRASP technique (GFP Reconstitution Across Synaptic Partner) that was developed to image membrane contacts at neuronal synapses (15, 16). We expressed CD4:GFP1–10 (a fragment of GFP that includes ten strands of the GFP β-barrel photocenter fused as an extracellular postscript to the transmembrane domain of the mouse lymphocyte protein CD4) and CD4:GFP11 (a fragment that includes the 11th strand of the GFP β-barrel). To image cytoneme contacts, the two parts of GFP were expressed separately in tracheal cells and either FGF- or Dpp-expressing disc cells. These non-fluorescent GFP fragments generated fluorescence that localized specifically at the disc cells that expressed either FGF or Dpp (Fig. 4A–C). Expression of mCherry-CAAX (CAAX is a plasma membrane targeting motif) in the disc dpp domain revealed that GRASP fluorescence correlates with dpp-expressing cells (Fig. 4C). Fluorescence was separated from the ASP cells by up to 40 μm (Fig. 4A,B), the approximate length of the longest cytonemes that projected from the ASP toward disc cells, indicating that ASP and disc cells synapse even when separated. GFP fluorescence was not observed in animals that expressed only one of the fragments.
Figure 4. Tracheal cytonemes contact Dpp- and FGF-expressing disc cells.
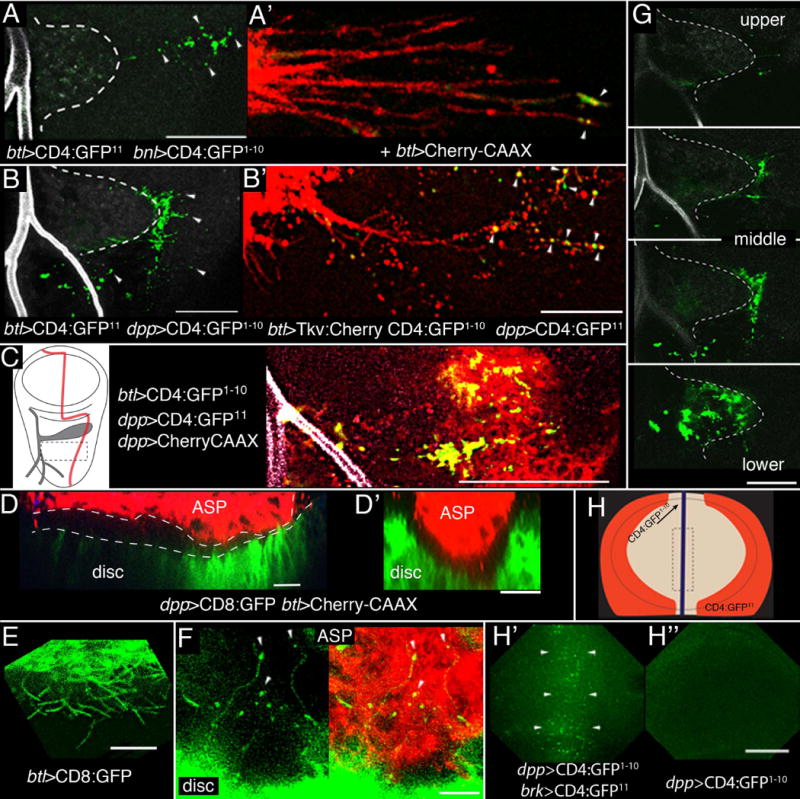
(A,A′,B,B′) Green fluorescence (arrowheads) from reconstituted GFP (GRASP) due to contact between ASP cytonemes and disc shown in projection images composed of several “upper – mid” optical sections. ASP (dashed white line), disc and TC lumen were imaged at 405 nm for background fluorescence (grey). Normal dpp expression includes cells anterior to the stripe at the A/P compartment border (see Fig. 1A, B). Marking cytonemes with Cherry-CAAX (A’) or Tkv:Cherry (B’) showed that GRASP fluorescence was cytoneme-associated (arrowheads). (C) Left panel: drawing of 3rd instar wing disc depicting Dpp-expressing cells (red) and ASP and TC (gray). Right panel: region outlined by dashed lines in left panel for GRASP fluorescence (green) at the basal surface of dpp-expressing disc cells (red). (D,D′) Sagittal (D) and transverse (D′) sections in the mid region of ASP show the spatial relationship of the ASP (red) lower layer to dpp-expressing disc cells (green, dpp-CD8:GFP: red, btl-Cherry-CAAX). (E) CD8:GFP expressed in the ASP marks cytonemes emanating from the lower aspect of the ASP; they orient toward the disc. (F) CD8:GFP expressed in the disc marks cytonemes that extend toward and appear to contact (arrowheads) ASP cells marked with Cherry:CAAX (btl-Cherry:CAAX dpp-CD8:GFP). (G) GFP reconstitution in four optical sections of (B) from upper layer, two middle layers and from the interface between lower layer and disc. (H) Drawing of the wing pouch region of a wing disc showing the stripe of dpp expression at the organizer (purple) and the flanking regions that express brinker (brk, orange). Box with dashed line indicates region imaged in (H′, H”). (H′) Reconstituted GFP (arrowheads) in the organizer region in disc with expression of the GFP fragments in the brk and dpp domains. (H”) Control with CD4:GFP1–10 expression in the dpp domain only. Scale bars: 30 μm except for (A′, B′, E, F), 10 μm.
To show that the GRASP fluorescence was associated with cytoneme contacts, cytonemes were marked independently of the GRASP GFP fragments by expression of mCherry-CAAX or Tkv:Cherry. Fluorescence of reconstituted GFP was mostly at or near cytoneme tips that contacted source cells (Figs. 4A′, B′). Tkv:Cherry fluorescence had a punctal distribution in these cytonemes and was also present at contact sites (Fig. 4B′). An estimate of the size of the CD4 domains (diameter ~65Å; 17, 18) and of the linkers that join CD4 to the GFP fragments suggests that the apposition of a cytoneme tip with a target cell at a synapse is less than 20 nm. This distance is comparable to neuronal and immune synapses, and because GFP photocenter maturation is not instantaneous (19), the GRASP fluorescence indicates that cytonemes can make relatively stable contacts with target cells.
The proximity of the tubular ASP and the disc varies along the ASP proximodistal axis (1.5–10 μm) and the anatomies of the two epithelia are complex (Fig. 4D,D″). The ASP cells that overlie Dpp-expressing disc cells are in close apposition, yet in this region cytonemes emanated from both the ASP (Fig. 4E) and the disc (Fig. 4F). The ASP cytonemes in this region were short (≤10 μm); the disc cytonemes were as long as 30 μm and many had bright bulbous tips at apparent points of contact with ASP cells. GRASP marked the contacts between the lower layer of the ASP and the disc (Fig. 4G), but did not resolve the relative contribution of the ASP and disc cytonemes.
In the wing pouch primordium of the wing disc, Dpp-dependent cytonemes on the apical cell surfaces orient toward the stripe of Dpp-expressing cells at the A/P developmental organizer and may ferry Dpp from the A/P organizer to cells as far away as the disc flanks (3–5). We applied GRASP to image contacts between the wing disc A/P organizer and flank cells by expressing the GFP fragments at the A/P organizer and in flank cells (Fig. 4H). In discs these discs, GFP fluorescence was observed in the region of the organizer (Fig. 4H′), in contrast to discs that expressed only one of the complementing fragments (Fig. 4H″). This pattern of GFP reconstitution suggests that cytonemes may extend from the cells at the disc flanks to synapse with cells of the A/P organizer.
Dpp signaling in the ASP requires cytoneme-mediated transport
We identified four genes that are required for ASP morphogenesis and for cytoneme function: diaphanous (dia), shibire (shi), neuroglian (nrg) and capricious (caps). Mutant loss-of-function conditions were induced selectively in trachea during the L3 stage (see SOM), and mutant ASPs were abnormal or duplicated at variable expressivity and penetrance (Table S2, Fig. S1); we show and describe ASPs that were most normal in appearance. Wing discs in these experiments were not mutant and wing disc development appeared normal.
The formin Dia is an actin nucleation factor (20) whose activated form localizes to the tips of filopodia (21). When Dia:GFP and activated Dia:GFP (22) were expressed in the ASP, Dia:GFP was mostly in the cell body and was present at low levels in cytonemes, but activated Dia:GFP was prominent in most cytonemes and localized to cytonemes tips (Fig. 5A). The distribution of activated Dia indicates that cytoneme tips may be sites of actin nucleation. To examine the role of Dia, we expressed diaRNAi in the ASP during the L3 stage. In >85% of the animals (n=26), growth of the ASP was decreased and ASP morphogenesis was abnormal (e.g., Fig. S1J). The number of cytonemes was also decreased and many of the cytonemes that extended from mutant ASPs were abnormally short and had blunt tips (Fig. 5B–E), and Dpp signal transduction (Dad-GFP expression and pMad abundance) was decreased (Fig. 5F,G; Table S3). We did not detect changes to cell shape, number of dividing cells or number of dying cells in mutant ASPs (Fig. S2A,B). Thus, Dia appears to be required by the ASP to make cytonemes and that the defective cytonemes that are made in the absence of normal Dia function are incapable of mediating Dpp signaling from the disc.
Figure 5. ASP cytonemes require dia and Shi and nrg.
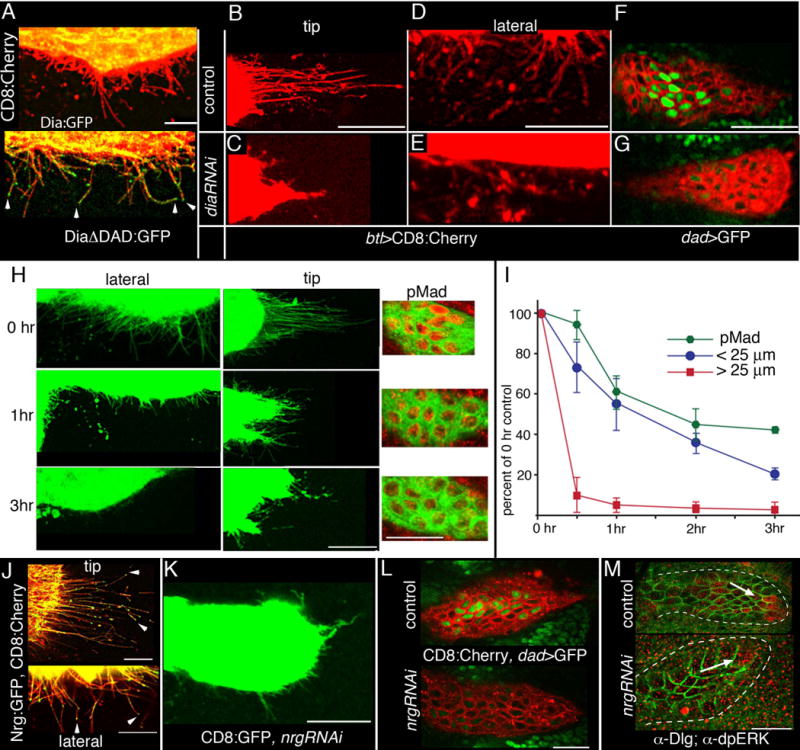
(A) In the ASP, localization of Dia:GFP is predominantly in the cell bodies; activated Dia (DiaΔDad:GFP) localizes to cytoneme tips (btl-Gal4, UAS-CD8:Cherry/+; tub-Gal80ts /UAS-Dia:GFP or UAS-DiaΔDad:GFP). (B–G) Expression of diaRNAi shortened lateral and tip ASP cytonemes (btl-Gal4, UAS-CD8:Cherry/tub-Gal80ts; Dad-GFP/UAS-diaRNAi), and reduced expression of Dad-GFP. Control genotype: btl-Gal4, UAS-CD8:Cherry/tub-Gal80ts; Dad-GFP/+). (H, I) Late 3rd instar larvae that co-expressed shts1 and CD8:GFP (btl-Gal4, UAS-CD8:GFP, UAS-shits1) were incubated at 30°C for the indicated times and after dissection, GFP fluorescence and α-pMad staining (red) were imaged in the lower layer of the ASPs (see Fig. 1C). The perimeter of each of five ASPs was measured, cytonemes were counted (I) around the perimeter in approximately 35–40 optical sections, and the length of each cytoneme was measured. Graph (I) shows the average percentage change to the number of ASP cytonemes/μm perimeter in the length ranges of <25μm (blue) and >25μm (red). Amounts of pMad were determined by measuring the mean fluorescence intensity (555 nm) in four ASPs for each time point for a region of the lower ASP level that contained approximately 11 cells. (J) Nrg:GFP (btl-Gal4, UAS-CD8:Cherry/UAS-Nrg:GFP; tub-Gal80ts) localizes to and concentrates at the tips (arrowheads) of ASP cytonemes. (K) Late 3rd instar larvae that co-expressed nrgRNAi and CD8:GFP (btl-Gal4, UAS-CD8:GFP/UAS-nrgRNAi; tub-Gal80ts/+). Lateral and tip cytonemes were stunted and reduced in number. (L) Expression of Dad-GFP was reduced in a lower ASP layer that expresses nrgRNAi (lower panel; btl-Gal4, UAS-CD8:Cherry/UAS-nrgRNAi; Dad-GFP/tub-Gal80ts) compared to control (upper panel; btl-Gal4, UAS-CD8:Cherry/+; Dad-GFP/tub-Gal80ts). (M) dpERK staining (arrows, red) is partially reduced in ASP that expresses nrgRNAi (lower panel; btl-Gal4 /UAS-nrgRNAi; tub-Gal80ts/+); (upper panel), control btl-Gal4/UAS-nrgRNAi; tub-Gal80ts/+); outline of ASP marked with dashed line and α-Dlg (green) outlines cells. Conditions for conditional inactivation are described in SOM and Table S2. Scale bars, 25 μm.
We expressed a conditional mutant of shibire (fruit fly dynamin; shits1) (23) together with CD8:GFP in trachea and compared α-pMAD staining as well as the number and length of cytonemes in ASPs that were isolated from larvae that had been incubated at either permissive (18°C) or restrictive (30°C) temperature (Figure 5H). Dynamin is a multimer (24, 25), and under non-permissive conditions the Shits1 protein functions as a dominant negative (23). Control larvae subjected to three hrs at 30°C did not change the number of “short” (<25 μm) or “long” (> 25 μm) ASP cytonemes (~4.4% and ~4.9% reduction, respectively) or reduce amounts of pMad (~7%). However, shits1 larvae subjected to 30°C had decreased cytoneme numbers and pMad abundance (Fig. 5I and Table S3). The number of long cytonemes present after 30 min at 30°C was less than 10% of that in control experiments; numbers of short cytonemes also declined after 30 min at 30°C. Reductions in numbers of short cytonemes and amounts of pMad became more severe over time intervals of up to three hours. A two hour heat pulse and one hr incubation at 20°C partially restored both long and short cytonemes (see SOM), but the ASP morphology was not normal. Indeed, duplicated, abnormally shaped ASPs were produced when a 24 hour incubation at 20°C followed a one hour heat pulse (Fig. S1I). Adults that developed at 20°C after a two hr heat pulse appeared to have normal morphology and we did not examine the structure or function of their dorsal air sacs. Thus, Shi inactivation was not lethal in the cells of the ASP, that the consequences of Shi inactivation on ASP development were partially reversible, and the effects on cytonemes preceded the reduction in signaling (as revealed by amounts of pMad).
To distinguish whether ASPs that are deficient for dia or shi expression failed to activate Dpp signal transduction because they did not receive Dpp from the wing disc or were incapable of initiating a response, we expressed Dpp:Cherry directly in ASPs with the btl-Gal4 driver (see SOM). Ectopic Dpp induced pMad in ASPs with reduced dia or Shi function (Figs. S3A, B). Thus, conditions that reduced dia expression or inactivated Shi did not abrogate the ability of ASP cells to respond to Dpp, and blocking cytoneme-mediated uptake of Dpp from the disc appears to be the most likely cause of the signaling deficits.
Cytonemes were also defective in loss-of-function conditions for nrg and caps, both of which encode putative cell adhesion transmembrane proteins. Nrg is an L1-type Cell Adhesion Molecule implicated in the development and stability of neuronal synapses (26). Although fluorescence of an in-frame protein trap Nrg:GFP fusion protein was detected in the ASP, ASP cytonemes could not be resolved because of “background” expression in the wing disc. However, over-expression of Nrg:GFP in the ASP revealed that Nrg distributes in ASP cytonemes and concentrates at cytoneme tips (Fig. 5J). Expression of nrgRNAi reduced the number of both tip and lateral cytonemes (Fig. 5K, Table S4), abrogated Dad-GFP expression and dpERK staining (Fig. 5L,M), and caused growth of abnormal, duplicated ASP lobes (Fig. S1K). Expression of nrgRNAi had no apparent effect on cell shape or the number or distribution of dividing or dying cells (Fig. S2C,D). Expression of Dpp:Cherry together with nrgRNAi in the ASP restored Dpp signaling (Fig. S3C), indicating that nrg-deficient ASP cells can activate Dpp signal transduction.
We identified caps in an enhancer trap screen for genes that are expressed in the ASP (see SOM and Fig. S2E,F). Caps:GFP that was expressed in trachea was detected in ASP cytonemes, and concentrated at the tips (Fig. 6A). Caps and its paralog Tartan (Trn) have extracellular domains containing leucine-rich repeats (LRR) and contribute partially redundant functions to the formation of compartment boundaries of the wing disc (27). caps mutants do not mediate selection of synaptic partners normally (28–31), and Caps protein localizes at filopodia tips during partner recognition and synapse stabilization (29). We observed similar types of effects on ASP cytonemes.
Figure 6. ASP cytonemes require Caps.
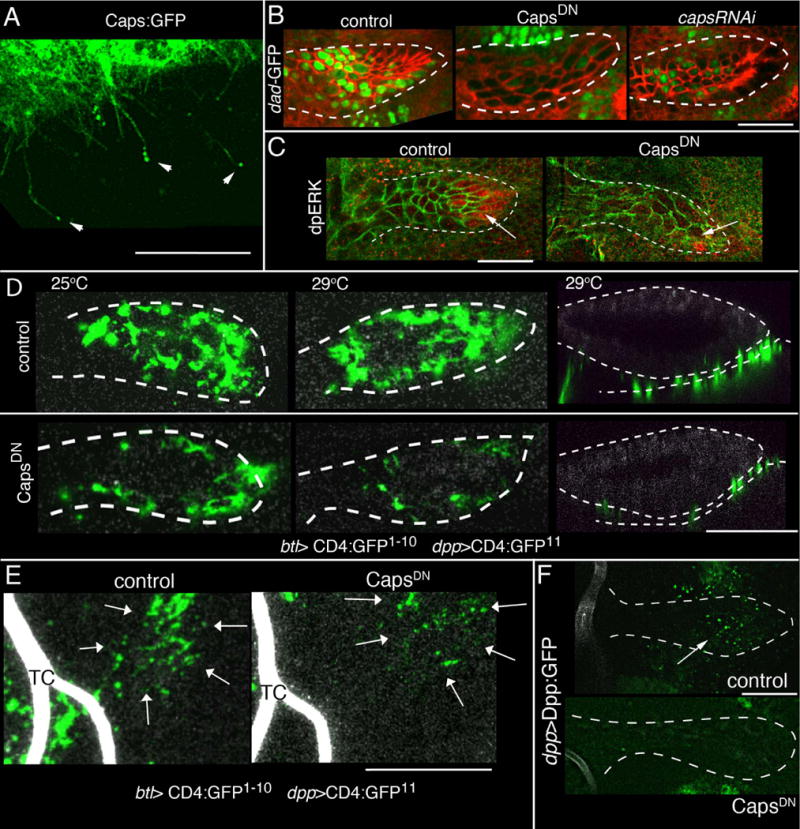
(A) Caps:GFP (btl-Gal4 UAS-Caps:GFP, tub-Gal80ts); localizes to and concentrates at the tips (arrowheads) of cytonemes. Condition for conditional inactivation are described in SOM. (B) ASP expression (btl-Gal4) of CapsDN (middle panel) and capsRNAi (right panel) reduced Dad expression in the ASP (Dad-nlsGFP; green); control (left panel). (C) ASP expression (btl-Gal4) of CapsDN reduced dpERK staining (red) at the tip of ASP. Cells are marked with α-Dlg staining (red, B; green, C). (D) Sagittal optical sections at “low” level of ASP (left and middle panels) and in coronal sections (right panels) showing GRASP fluorescence is reduced by expression of CapsDN (at 29°C); CapsDN genotype includes two copies of UAS-CapsDN. TC indicates the lumen of the transverse connective. (E) CapsDN expression in the TC reduces GRASP fluorescence (arrows) associated with Dpp-expressing disc cells. Genotypes: same as (D). (F) Dpp:GFP uptake in ASP (arrow) in presence (bottom panel) and absence (top panel) of CapsDN. Genotypes: same as (D). (D–F) ASP, disc and TC are imaged for autofluorescence at 405 nm (grey). Scale bar: 30 μm, except for (A), 10 μm.
Lack of caps function also led to abnormal ASP development. Expression of capsRNAi, trnRNAi, or a dominant negative Caps mutant (CapsDN) that localizes similarly to wild type Caps in synapses and decreases synaptic contacts (29, 31) reduced Dpp signaling and yielded abnormal ASPs (SOM, Fig. 6B, S1L–N, Tables S2,3). Phenotypes were more extreme in a heterozygous caps trn double mutant background. Expression of CapsDN did not cause detectable changes to cell polarity, cell morphology, mitotic activity or cell viability (Fig. S2G,H).
CapsDN reduced amounts of dpERK (diphospho-Extracellular signal-regulated kinase; Fig. 6C), indicating that caps function was also required for FGF signaling. Evidence that signal transduction per se was not abrogated in ASP cells that lack caps function was obtained by over-expressing FGF ubiquitously. Heat shock-induced expression of FGF or expression of Dpp:Cherry in the ASP increased amounts of dpERK or pMAD, respectively, throughout the ASP, attenuating the effects of CapsDN (Fig. S3D). These experiments show that Dpp and FGF proteins that are produced by the disc (Fig. 1 and 9) require caps function in the ASP to activate signal transduction in ASP cells, and show that mutant ASP cells that cannot receive FGF and Dpp from the disc are competent for signal transduction.
The presence of Caps:GFP in the tips of cytonemes (Fig. 6A), the role of Caps at neuronal synapses (29), the fact that cytonemes make contact with Dpp-producing cells (Fig. 4B,C,G) and receive Dpp at apparent points of contact (Fig. 3E,F), and the essential role of caps for Dpp signaling, suggest that Caps may be required for cytonemes to establish functional contacts for Dpp exchange. However, the number and distribution of ASP cytonemes did not change under caps loss-of-function conditions (Fig. S2I), indicating that the ASP cells do not require Caps to make cytonemes. In contrast, the contacts that ASP cytonemes made with Dpp-expressing disc cells required caps. We monitored these contacts with GRASP fluorescence: GFP fluorescence at the interface of Dpp-expressing disc cells and the lower layer of the ASP, and at cytoneme contacts of the lateral ASP and TC was reduced when CapsDN was expressed in trachea (Figs. 1D, 4C,D, 6D,E). In addition, CapsDN reduced uptake of Dpp:GFP from the disc (Fig. 6F), suggesting that although ASP cells make cytonemes in the absence of Caps function, Caps-deficient cytonemes that do not make stable synapses do not transfer Dpp from producing to recipient cells.
Discussion
This study revealed an essential role for cytoneme-based transport of signaling proteins in long distance paracrine signaling. This mechanism involves contact-dependent transfer of signaling proteins from producing to responding cells, and although we studied signaling between two epithelial tissues in a Drosophila larva, evidence from other systems supports a general role for cytonemes in paracrine signaling.
Studies of cells in culture indicate that filopodia receive and transport signaling proteins that are taken up from culture media. In experiments with human adenocarcinoma cells, uptake of EGF protein from the culture medium led to retrograde transport by filopodia along with activated EGFR, and was sensitive to cytochalasin D, a disruptor of F-actin (32). Actin-based cytonemes that carry FGFR-rich puncta and that are dependent on the small GTPase RhoD are present in cultured mouse mesenchymal cells (33).
Some characteristics of Dpp signaling in the ASP are consistent with these cell culture experiments. Dpp that was taken up by an ASP cell was present in motile puncta that translocated along the ASP cell’s cytoneme, and some puncta in the ASP cytonemes contained both Dpp and its receptor (Fig. 2C,D, 3C,E,F, 4B′). Drosophila cytonemes are actin-based (3). However, in contrast to cultured cells, signaling in the ASP did not appear to involve uptake of signaling proteins from the extracellular milieu, but was dependent on synaptic contact between the tip of a cytoneme that extended from a responding ASP cell and the cell body of a Dpp-expressing disc cell. This signaling mechanism appears to involve specific dynamic interactions between signaling and responding cells.
ASP cells express both the Tkv Dpp receptor and FGFR, and segregate these receptors to puncta in distinct cytonemes (5). At the early L3 stage, the ASP is small and does not extend across the disc, and both the Dpp- and FGF-expressing disc cells are distal to its tip. Both Tkv- and FGFR-containing cytoneme extended distally from the tip (5). The FGFR-conaining cytonemes extended beyond the Dpp-expressing cells and did not take up Dpp (Fig. 3D). At later L3 stages, the ASP has grown across the disc, and although the FGF-expressing disc cells are distal to it, the Dpp-expressing disc cells lie under its medial region. In these ASPs, the Tkv-containing cytonemes emanated from the medial region and reached as much as 40 μm to pick up Dpp from disc cells (Fig. 3B,C). Thus in the ASP, spatially restricted Dpp signal transduction (Fig. 1C,F) and uptake (Fig. 2A, 3B,C) were associated with cytonemes whose orientation and composition appeared to be specific for Dpp.
The dynamism of this signaling system may be inferred from steady-state images. The distribution and orientation of cytonemes changes if expression of signaling protein is compromised and if signaling protein is over-expressed in ectopic locations (4, 5, 9). These properties suggest that cytonemes are changeable and that their distributions reflect the relative positions of signal-producing and -receiving cells. The different distributions of Tkv-containing cytonemes in the temporal sequence described above are consistent with this idea and with a model of cytoneme impermanence. The observation that some ASP cytonemes contain Tkv, make contact with Dpp-producing cells and take up Dpp whereas other cytonemes contain Tkv but do not make contact with Dpp-producing cells or take up Dpp (Fig. 2E, 3F) may also suggest that cytoneme contacts may be transient.
Plasticity may be an important attribute of cytonemes that function in a developmental system such as the ASP in which relations between producing and receiving cells change as the larva develops. Cytonemes may have are the capacity to regulate release and uptake of signals and to direct signals to a pre-selected target. Regulated release may be implied by the absence of Dpp uptake and Dpp signal transduction in ASP mutant conditions that abolish synaptic contacts by ASP cytonemes. In these experiments, the signal producing cells were not mutant, and the wing discs, which depend on Dpp signaling, developed normally, indicating that the signaling defect was specific to the ASP cells that made defective cytonemes. Because filopodia of cultured cells take up signaling proteins from culture medium and activate signal transduction (32), we may assume that ASP cytonemes are similarly capable of taking up signaling protein that their receptors encounter and that the inability of cytoneme-defective cells to take up Dpp or activate signal transduction may indicate that Dpp was not released in the absence of cytoneme contact.
There may be a functional analogy to neuronal signaling. Neurons make asymmetric extensions that send and receive signals, make specific contacts where signal release and uptake are regulated, and require the diaphanous, neuroglian, shibire and capricious genes for contact-mediated signal exchange and signaling. In the developing Drosophila retina, Hh moves to the termini of retinal axons where it signals to post-synaptic laminal neurons in the brain (34). Perhaps the strongest precedent is Wingless delivery at developing neuromuscular junctions in the Drosophila larva; in this case, Wingless moves to the post-synaptic cell after release in a vesicular form from the pre-synaptic neuron (35). Our studies have been limited to the cytonemes that are made by receiving cells, but in other contexts, cytonemes extend from cells that deliver signaling proteins, such as the Hh-containing cytonemes of the wing disc (7), the cytonemes that extend from cap cells in the female germline stem cell niche (8) and that are associated with Notch and EGF signaling (36–39). Cytonemes that transport Hh across the chick limb bud from Hh producing cells have also been described (40). The widespread presence of cytonemes in many cell types and in many contexts suggests that they may provide a general mechanism to move signaling proteins between non-neuronal cells at sites of direct contact.
Materials and Methods
Drosophila stocks
Transgenes
btl-Gal4 (9), ap-Gal4 (Bloomington Stock Center (BSC)); dpp-Gal4/CyO; HS-Bnl (9); UAS-Tkv:GFP (4); UAS-Dpp:Cherry, UAS-CD8:Cherry, UAS-CD8:GFP (5), Dad-nEGFP(III) (42), UAS-FGFRDN (43); dpp-LHG/TM6 (LexA-Gal4 Activation Domain fusion; III) (44), dpp-LHG (II; this study), lexO-Dpp:GFP/TM6 and brkBM14-LHG (44), btl-LHG (II and III) (this study), lexO-CD4:GFP11(II), UAS-CD4:GFP1–10 (III) (16), UAS-dppRNAi(BSC), UAS-putRNAi (BSC); tub-Gal80ts (II and III, BSC), UAS-Dad (II) (BSC), UAS-TkvQD/TM6B (activated Tkv; 45), UAS-TkvDN, UAS-PutDN (ΔGSK, Dominant negative forms of Tkv and Put lacking GS and Kinase domain; 46), UAS-TIPF(14), UAS-capsRNAi (BSC, VDRC), UAS-trnRNAi (NIG, BSC), UAS-capsDN (29); UAS-CD4:GFP10 (II; this study), UAS-diaRNAi (BSC, NIG), UAS-shits1 (23), lexO-Cherry CAAX (II & III; 44); UAS-Dia:GFP (47) and UAS-DiaΔDad:GFP (22); UAS-nrgRNAi (II & III) (BSC); 10XUAS-IVS-mCD8::RFP; 13XlexO-mCD8::GFP (BSC). UAS-Nrg:GFP (II) (26).
Insertions and mutations
Dadj1E4-LacZ/TM3, tkvk16713-LacZ/CyO, dpp10638-LacZ/CyO, put10460-LacZ/TM3 (BSC), Nrg:GFP protein trap line (flytrap line G00305). Conditional inactivation of Dpp was in dpp4/dpp56 L3 larvae for 18 hours at 29°C as described (4).
Over-expression
tub-Gal80ts was present to limit expression to the L3 stage. Expression drivers were: ap-Gal4 for dppRNAi; btl-Gal4 for Dad, TkvDN, TkvQD, PutDN, put-RNAi, CapsDN, capsRNAi, trnRNAi, diaRNAi, Dia:GFP, DiaΔDad:GFP, nrgRNAi, and Nrg:GFP. Animals were reared at 18°C until L3 and were incubated at 29°C as indicated in Table S2 prior to dissection. For knockdown under heterozygous mutant background (Table S2, Fig. S1M), CapsDN and capsRNAi expression was driven by btl-Gal4 or by dpp>Gal4 in capsC28fs trnδ17 and caps65.2 trnS064117 double mutants. At 25°C, CapsDN and capsRNAi over-expression is embryonic lethal in the Caps mutant background; animals were therefore reared at 20°C to the L3 stage and were incubated at 25°C for one day prior to dissection.
Ectopic expression
For Figure S3 A–C, crosses were, for dia, shi and nrg: btl-Gal4, UAS-CD8:GFP/+; tub-Gal80ts/UAS-dpp:Cherry to either UAS-diaRNAi, UAS-shits or UAS-nrgRNAi. Control larvae expressed either shits, diaRNAi or nrgRNAi, but lacked dpp:Cherry; experimental larvae had UAS-dpp:Cherry. Animals were reared at 18°C to minimize the effects of Dpp over-expression. To express diaRNAi, L3 larvae were incubated at 25°C for 5–6 hrs. Shits larvae were treated similarly and were then shifted to 29°C for 1 hr. ASPs in the Shits larvae did not grow normally due to temperature sensitivity of shits at 25°C. nrgRNAi induction was for 14–18 hr at 29°C. CapsDN larvae (btl-Gal4, UAS-CD8:GFP/UAS-CapsDN; UAS-CapsDN/HS-Bnl) were reared at 20°C until L3; heat-shock induction of Bnl was for 30 min at 37°C followed by 3 hr incubation in 20°C.
Dual expression
LexA and Gal4
10XUAS-IVS-mCD8:RFP, 13XlexO-mCD8:GFPflies (BSC) were crossed to dpp-Gal4/SM5; btl-LHG flies to mark Dpp-producing cells in wing disc with RFP and trachea with GFP. To express either Tkv:Cherry or FGFR:Cherry in trachea simultaneously with Dpp:GFP in the wing disc, UAS-Tkv:Cherry/CyO-Weep; dpp-LHG/TM6 or UAS-FGFR:Cherry/CyO-Weep; dpp-LHG flies were crossed to btl-Gal4; lexO-Dpp:GFP/TM6 flies. To minimize toxic effects, btl-Gal4/UAS-Tkv:Cherry (or FGFR:Cherry); lexO-Dpp:GFP/dpp-LHG animals were grown at 18°C until the L2 stage and were shifted to 20°C.
Enhancer trap screening
Approximately 500 lines with randomly inserted enhancer trap transposons (gift from E. Heberlein) were screened for tracheal expression (UAS-GFP). A line with elevated expression in the ASP was identified; its Gal4 transposon was mapped by ends out sequencing to the first exon of caps. Wing disc GFP expression was similar to the expression of caps as indicated by in-situ hybridization (27).
GFP reconstitution
Genotype for reconstitution between Dpp signaling partners: dpp-Gal4/lexO-CD4:GFP11; btl-LHG/UAS-CD4:GFP1–10. Genotype for reconstitution between FGF signaling partners: btl-LHG/lexO-CD4:GFP11; bnl-Gal4/UAS-CD4:GFP1–10. For reconstitution in the wing disc: dpp-Gal4/lexO-CD4:GFP11; brk-LexA/UAS-CD4:GFP1–10. For reconstitution with marked cytonemes:btl-LHG, lexO-CherryCAAX/lexO-CD4:GFP11; bnl-Gal4, btl-LHG/UAS-CD4:GFP10. For reconstitution with in the presence of marked Tkv: btl-Gal4, dpp-LHG/UAS-Tkv:Cherry; lexO-CD4:GFP11, UAS-CD4:GFP1–10. For reconstitution with marked Dpp source: btl-Gal4, dpp-LHG/+; lexO-Cherry:CAAX/UAS-CD4:GFP1–10, lexO-CD4:GFP11. For reconstitution together with CapsDN over-expression, btl-Gal4, dpp-lexA; UAS-CD4:GFP1–10, lexO-CD4:GFP11 flies were crossed with either w−(control) or UAS-capsDN. Larvae were reared in room temperature and shifted to 25°C or 29°C for 1 day prior to assay.
shibire inactivation
Larvae (btl>Gal4, UAS>CD8:GFP, UAS>shits1 (23)) were raised at 18°C prior to a single heat shock for 1/2 hr, 1 hr, 2 hr or 3 hrat 30°C. Larvae were dissected and imaged for ASP cytonemes or were fixed for pMAD staining. Rescue after heat shock was by returning larvae to 18°C prior to dissection and imaging. Control heat shock was with larvae expressing CD8:GFP in trachea (btl-Gal4 UAS-CD8:GFP) at 30°C for 0 and 3 hr. No significant change in numbers of cytonemes (either <25 μm (4.4%±4.7% reduction); or >25 μm (4.7%±7.6% increase)) was detected. Rescue after 30° at 2 hr was at 20°C for 1 hr, followed by dissection and imaging. Increases in numbers of cytonemes (<25 μm (1.9X ± .4%, P = 0.0471); or >25 μm (11X ± 2.9%, P = 0.0196)) were evaluated by the unpaired t-test.
Quantitation and statistical analyses
Cytonemes were counted and measured in z-section stacks of confocal images from five ASPs for each data point and were binned as <25 μm or > 25 μm. Lengths represent measures from each tip along the connecting shaft to the point of its widening base either at the plasma membrane or lamellipodia-like protrusion. The size variation between ASPs was normalized by measuring the perimeter of each ASP and then calculating the number of cytonemes per unit length. Values in Figure 5I are plotted as percent of the 0 hr time point. pMad levels were quantified by measuring the mean intensity of 555 nm fluorescence in the cells of lower layer of ASP, subtracting background fluorescence, and normalized with respect to pMAD fluorescence at the A/P border of the same wing disc. Values were plotted as percentage of the 0 hr time point. Statistical significance values were calculated with t-test or ANOVA followed by Tukey HSD test.
Molecular cloning
btl>LHG
The P[B123] fragment upstream of the btl gene (48) was amplified from a genomic clone obtained from (49) with 5′ primer GGCTCGAGATAATCGCATTCTGACCTCGGTAAAC and 3′ primer GGTCTAGAGGATCGTACCCGTAATCCG, and the product was cloned in pCASPER4. The LexA:Gal4H-GAD portion was isolated from the pDppattB-LHG plasmid (44) and was inserted at the pCASPER4 NotI site.
Tkv:Cherry
The NotI–HindIII fragment from a Tkv:GFP construct (4) was ligated to a mCherry fragment with 5′ HindIII and 3′ KpnI sites in the presence of pUAST that had been digested with NotI and KpnI. Primers for mCherry amplification: 5′ primer, GCAAGCTTATGGTGAGCAAGGGCGAGGAGG; 3′ primer, AGGTACCTTACTTGTACAGCTCGTCCATGCCGC). Tkv:Cherry and Tkv:GFP are similar in phenotype, activity and localization in cytonemes.
In-situ hybridization
RNA in situ hybridization was performed according to (50). DIG labeled antisense probe was generated by transcription from a T7 promoter joined to a 600 bp fragment of dpp cDNA amplified with PCR primers: CAAGGAGGCGCTCATCAAG and TTGTAATACGACTCACTATAGGGAGACACCAGCAGTCCGTAGTTGC. Alkaline phosphatase conjugated α-DIG antibody (Roche) was used to detect the DIG labeled probe.
Immunohistochemistry
The following antisera were used: α-pMAD (from Ed Laufer and P. ten Dijke, at 1:2000; 51); α-dpERK (Sigma; 1:250), α-Apontic (from R. Schuh,; 52); α-Discs large (4F3; 1:50), α-DE-cadherin (DECAD2; 1:20), and anti-β-galactosidase were from Developmental Studies Hybridoma Bank. dpERK staining was carried out as described (9) with antibody obtained from Cell Signaling. Secondary antibodies were conjugated to Alexa-Fluor 488, 555 or 647. To assay for cell lethality, α-cleaved caspase-3 (Asp175, Cell Signaling) was used as described (53). Cell proliferation was monitored with α-phosphohistone H3 antibody (Ser 10, Cell Signaling).
Imaging techniques
Wing discs were dissected and mounted as described (5) except that the second small coverslip was omitted. Images were made using a Leica TCS SPE or TCSSP2 confocal microscope with either 405, 488, 561 or 635 wavelength lasers and with LAS-AF software; or using a custom built Zeiss spinning disc confocal with EMCCD Hamamatsu Camera (9100-13) and Volocity 5.5 software; or with a standard Zeiss Axioplan II fluorescent microscope with sensicam CCD camera (Cooke Corporation, USA) and Slidebook-4 acquisition software (Intelligent Imaging Innovations). Patterns of cytonemes were consistent in all three types of systems. Brightfield images were made on a Leica DMR microscope equipped with SPOT CCD Camera (Diagnostics Instruments) and SPOT acquisition software. Final images were analyzed and processed with NIH ImageJ.
Supplementary Material
Figure S1. Dpp signaling in the ASP. (A) Transverse section of the medial region of an unfixed ASP (btl-CD8:Cherry Dad-nlsGFP) showing GFP fluorescence in cells of lower layer and in the disc cells below. (B, B′) tkv expression (red, tkv-lacZ detected by anti-β-galactosidase staining) is higher in the upper (B) than lower (B′) level of the ASP (outlined by dashed white line). (C, C′) Expression of put (red, put-lacZ detected by anti-β-galactosidase staining) is similar in upper (C) and lower (C′) levels of the ASP (green, btl-Gal4 UAS-CD8:GFP). (D, D′) Optical sections showing Dad (D) and pMad (D′) assays of Dpp signaling in medial region of the upper and lower layers of ASP: (Dad-lacZ (red), α-pMAD staining (red), α-Dlg (green)). (E) RNA in-situ hybridization detects dpp expression in the disc but not in the trachea. Black dashed line marks position of ASP and TC. (F–N) Functional knockdown conditions in the ASP that induce morphogenetic malformations in the ASP (bifurcations and abnormal shapes) and tracheal duplications (arrows in (G, H). Genotypes: btl-Gal4, UAS-CD8:GFP; tub-Gal80ts X UAS-PutDN (F), X UAS-Dad (G), X UAS-TkvDN (H), X UAS-shits (I), X UAS-diaRNAi (J), X UAS-nrgRNAi (K), and X UAS-trnRNAi (N). shits ASP in (J) was incubated at 30°C for 1 hr followed by 20°C for 24 hrs. (L) Genotype: btl-Gal4 UAS-CD8:GFP / UAS-CapsDN; UAS-CapsDN and (M) btl-Gal4 UAS-CD8:GFP / UAS-CapsDN; capsC28fs trnΔ17 / UAS-CapsDN. Blue fluorescence in (G) and (H) is autofluorescence of lumen at 405 nm. (N) α-Dlg (red). Scale bar, 30 μm.
Figure S2. Characterizations of ASP in normal and mutant conditions. (A–D) Expression of diaRNAi (A, B) or nrgRNAi (C, D) did not change the number of α-phosphohistone-3 or α-Caspase-3 staining (red). (E, F) Expression of caps and trn detected by α-LacZ antibody ((E), caps-LacZ (P{PZ}caps02937; green) and GFP fluorescence ((F), trn-GFP). (G, H) Expression of CapsDN did not change the number of α-phosphohistone-3 or α-Caspase-3 staining (red). (I) Number and distribution of ASP cytonemes were not significantly changed by CapsDN over-expression. (B, G, H), α-Dlg (white); (A–D, G–I) CD8:GFP. Scale bar, 30 μm.
Figure S3: Cells with defective cytonemes do not activate signal transduction but are signaling competent. ASPs (outlined by dashed lines) that expressed under btl-Gal4: GFP (A–D), RNAi directed against dia (A), inactive Shits (B), RNAi directed against nrg (C), or CapsDN (D). α-pMad antibody stained ASP cells that also expressed Dpp:Cherry ((driven by btl-Gal4; upper panels) but did stain ASP cells that did not express Dpp:Cherry (lower panels). Right panels: pMad staining; middle panels Dpp:Cherry fluorescence; left panels: merge with CD8:GFP fluorescence. (E) ASPs that expressed GFP and CapsDN under btl-Gal4 stained for dpERK in the presence of ectopic FGF (driven by heat shock; right panel) but not in the absence of ectopic FGF (left panel). Scale bar: 30 mm.
Supplementary Movie S1: Movement of co-localized Dpp:GFP and Tkv:Cherry puncta in cytonemes. Tkv:Cherry was expressed in trachea (btl-Tkv:Cherry); Dpp:GFP was expressed in the dpp domain of the wing disc (dpp-Dpp:GFP). Each frame of the movie is a maximum Z-projection that was compiled with ImageJ, and is shown at two frames per second. Each Z-stack (0.4 μm steps) was captured at intervals of two time points per minute at 488 nm and 561 nm using an inverted spinning disc confocal microscope. Scale bar, 40 μm.
Acknowledgments
We thank K. Basler, M. Affolter, K. Scott, E. Laufer, P. ten Dijke, T. Kitamoto, J. Pielage and C. Bökel, K. Wharton, A. Nose, L. Luo, M. Milan, B. Shilo, the Bloomington Stock Center and the Developmental Studies Hybridoma Bank for reagents, L. Lin, P. Rao for discussions and M. Barna for access to the spinning disc confocal microscope. Funding from: NIH K99HL114867 to S.R. and GM030637 to T.B.K.
References
- 1.Gelbart WM. The decapentaplegic gene: a TGF-beta homologue controlling pattern formation in Drosophila. Develop. 1989;107(Suppl):65. doi: 10.1242/dev.107.Supplement.65. [DOI] [PubMed] [Google Scholar]
- 2.Wartlick O, et al. Dynamics of Dpp signaling and proliferation control. Science. 2011 Mar 4;331:1154. doi: 10.1126/science.1200037. [DOI] [PubMed] [Google Scholar]
- 3.Ramirez-Weber FA, Kornberg TB. Cytonemes: cellular processes that project to the principal signaling center in Drosophila imaginal discs. Cell. 1999;97:599. doi: 10.1016/s0092-8674(00)80771-0. [DOI] [PubMed] [Google Scholar]
- 4.Hsiung F, Ramirez-Weber FA, Iwaki DD, Kornberg TB. Dependence of Drosophila wing imaginal disc cytonemes on Decapentaplegic. Nature. 2005;437:560. doi: 10.1038/nature03951. [DOI] [PubMed] [Google Scholar]
- 5.Roy S, Hsiung F, Kornberg TB. Specificity of Drosophila cytonemes for distinct signaling pathways. Science. 2011 Apr 15;332:354. doi: 10.1126/science.1198949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bilioni A, et al. Balancing hedgehog, a retention and release equilibrium given by Dally, Ihog, Boi and shifted/dWif. Dev Biol. 2013 Dec 29; doi: 10.1016/j.ydbio.2012.12.013. [DOI] [PubMed] [Google Scholar]
- 7.Callejo A, et al. Dispatched mediates Hedgehog basolateral release to form the long-range morphogenetic gradient in the Drosophila wing disk epithelium. Proc Natl Acad Sci U S A. 2011 doi: 10.1073/pnas.1106881108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rojas-Rios P, Guerrero I, Gonzalez-Reyes A. Cytoneme-mediated delivery of hedgehog regulates the expression of bone morphogenetic proteins to maintain germline stem cells in Drosophila. PLoS Biol. 2012;10:e1001298. doi: 10.1371/journal.pbio.1001298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sato M, Kornberg TB. FGF is an essential mitogen and chemoattractant for the air sacs of the drosophila tracheal system. Develop Cell. 2002;3:195. doi: 10.1016/s1534-5807(02)00202-2. [DOI] [PubMed] [Google Scholar]
- 10.Tsuneizumi K, et al. Daughters against dpp modulates dpp organizing activity in Drosophila wing development. Nature. 1997 Oct 9;389:627. doi: 10.1038/39362. [DOI] [PubMed] [Google Scholar]
- 11.Tanimoto H, Itoh S, ten Dijke P, Tabata T. Hedgehog creates a gradient of DPP activity in Drosophila wing imaginal discs. Mol Cell. 2000;5:59. doi: 10.1016/s1097-2765(00)80403-7. [DOI] [PubMed] [Google Scholar]
- 12.Entchev EV, Schwabedissen A, Gonzalez-Gaitan M. Gradient formation of the TGF-beta homolog Dpp. Cell. 2000;103:981. doi: 10.1016/s0092-8674(00)00200-2. [DOI] [PubMed] [Google Scholar]
- 13.Teleman AA, Cohen SM. Dpp gradient formation in the Drosophila wing imaginal disc. Cell. 2000 Dec 8;103:971. doi: 10.1016/s0092-8674(00)00199-9. [DOI] [PubMed] [Google Scholar]
- 14.Michel M, Raabe I, Kupinski AP, Perez-Palencia R, Bokel C. Local BMP receptor activation at adherens junctions in the Drosophila germline stem cell niche. Nat Commun. 2011;2:415. doi: 10.1038/ncomms1426. [DOI] [PubMed] [Google Scholar]
- 15.Feinberg EH, et al. GFP Reconstitution Across Synaptic Partners (GRASP) defines cell contacts and synapses in living nervous systems. Neuron. 2008;57:353. doi: 10.1016/j.neuron.2007.11.030. [DOI] [PubMed] [Google Scholar]
- 16.Gordon MD, Scott K. Motor control in a Drosophila taste circuit. Neuron. 2009;61:373. doi: 10.1016/j.neuron.2008.12.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ryu SE, et al. Crystal structure of an HIV-binding recombinant fragment of human CD4. Nature. 1990 Nov 29;348:419. doi: 10.1038/348419a0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wang JH, et al. Atomic structure of a fragment of human CD4 containing two immunoglobulin-like domains. Nature. 1990 Nov 29;348:411. doi: 10.1038/348411a0. [DOI] [PubMed] [Google Scholar]
- 19.Cabantous S, Terwilliger TC, Waldo GS. Protein tagging and detection with engineered self-assembling fragments of green fluorescent protein. Nat Biotechnol. 2005 Jan;23:102. doi: 10.1038/nbt1044. [DOI] [PubMed] [Google Scholar]
- 20.Castrillon DH, Wasserman SA. Diaphanous is required for cytokinesis in Drosophila and shares domains of similarity with the products of the limb deformity gene. Development. 1994 Dec;120:3367. doi: 10.1242/dev.120.12.3367. [DOI] [PubMed] [Google Scholar]
- 21.Homem CC, Peifer M. Exploring the roles of diaphanous and enabled activity in shaping the balance between filopodia and lamellipodia. Molecular biology of the cell. 2009 Dec;20:5138. doi: 10.1091/mbc.E09-02-0144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Rousso T, Shewan AM, Mostov KE, Schejter ED, Shilo BZ. Apical targeting of the formin Diaphanous in Drosophila tubular epithelia. eLife. 2013;2:e00666. doi: 10.7554/eLife.00666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kitamoto T. Conditional modification of behavior in Drosophila by targeted expression of a temperature-sensitive shibire allele in defined neurons. J Neurobiol. 2001 May;47:81. doi: 10.1002/neu.1018. [DOI] [PubMed] [Google Scholar]
- 24.Muhlberg AB, Warnock DE, Schmid SL. Domain structure and intramolecular regulation of dynamin GTPase. EMBO J. 1997 Nov 17;16:6676. doi: 10.1093/emboj/16.22.6676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Hinshaw JE. Dynamin spirals. Curr Opin Struct Biol. 1999 Apr;9:260. doi: 10.1016/S0959-440X(99)80036-0. [DOI] [PubMed] [Google Scholar]
- 26.Enneking EM, et al. Transsynaptic coordination of synaptic growth, function, and stability by the L1-type CAM Neuroglian. PLoS Biol. 2013;11:e1001537. doi: 10.1371/journal.pbio.1001537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Milan M, Weihe U, Perez L, Cohen SM. The LRR proteins capricious and Tartan mediate cell interactions during DV boundary formation in the Drosophila wing. Cell. 2001 Sep 21;106:785. doi: 10.1016/s0092-8674(01)00489-5. [DOI] [PubMed] [Google Scholar]
- 28.Hong W, et al. Leucine-rich repeat transmembrane proteins instruct discrete dendrite targeting in an olfactory map. Nature neuroscience. 2009 Dec;12:1542. doi: 10.1038/nn.2442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kohsaka H, Nose A. Target recognition at the tips of postsynaptic filopodia: accumulation and function of Capricious. Development (Cambridge, England) 2009 Apr;136:1127. doi: 10.1242/dev.027920. [DOI] [PubMed] [Google Scholar]
- 30.Shishido E, Takeichi M, Nose A. Drosophila synapse formation: regulation by transmembrane protein with Leu-rich repeats, CAPRICIOUS. Science (New York, N.Y) 1998 Jun 26;280:2118. doi: 10.1126/science.280.5372.2118. [DOI] [PubMed] [Google Scholar]
- 31.Taniguchi H, Shishido E, Takeichi M, Nose A. Functional dissection of drosophila capricious: its novel roles in neuronal pathfinding and selective synapse formation. Journal of neurobiology. 2000 Jan;42:104. doi: 10.1002/(sici)1097-4695(200001)42:1<104::aid-neu10>3.0.co;2-v. [DOI] [PubMed] [Google Scholar]
- 32.Lidke DS, Lidke KA, Rieger B, Jovin TM, Arndt-Jovin DJ. Reaching out for signals: filopodia sense EGF and respond by directed retrograde transport of activated receptors. J Cell Biol. 2005 Aug 15;170:619. doi: 10.1083/jcb.200503140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Koizumi K, et al. RhoD activated by fibroblast growth factor induces cytoneme-like cellular protrusions through mDia3C. Molecular biology of the cell. 2012 Oct 3; doi: 10.1091/mbc.E12-04-0315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Huang Z, Kunes S. Hedgehog, transmitted along retinal axons, triggers neurogenesis in the developing visual centers of the Drosophila brain. Cell. 1996;86:411. doi: 10.1016/s0092-8674(00)80114-2. [DOI] [PubMed] [Google Scholar]
- 35.Korkut C, et al. Trans-synaptic transmission of vesicular Wnt signals through Evi/Wntless. Cell. 2009;139:393. doi: 10.1016/j.cell.2009.07.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Cohen M, Georgiou M, Stevenson NL, Miodownik M, Baum B. Dynamic filopodia transmit intermittent Delta-Notch signaling to drive pattern refinement during lateral inhibition. Dev Cell. 2010 Jul 20;19:78. doi: 10.1016/j.devcel.2010.06.006. [DOI] [PubMed] [Google Scholar]
- 37.De Joussineau C, et al. Delta-promoted filopodia mediate long-range lateral inhibition in Drosophila. Nature. 2003 Dec 4;426:555. doi: 10.1038/nature02157. [DOI] [PubMed] [Google Scholar]
- 38.Renaud O, Simpson P. Scabrous modifies epithelial cell adhesion and extends the range of lateral signalling during development of the spaced bristle pattern in Drosophila. Dev Biol. 2001 Dec 15;240:361. doi: 10.1006/dbio.2001.0482. [DOI] [PubMed] [Google Scholar]
- 39.Peng Y, Han C, Axelrod JD. Planar polarized protrusions break the symmetry of EGFR signaling during Drosophila bract cell fate induction. Dev Cell. 2012 Sep 11;23:507. doi: 10.1016/j.devcel.2012.07.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Sanders TA, Llagostera E, Barna M. Specialized filopodia direct long-range transport of SHH during vertebrate tissue patterning. Nature. 2013 May 30;497:628. doi: 10.1038/nature12157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.see Supplementary Materials in Science online.
- 42.Ninov N, et al. Dpp signaling directs cell motility and invasiveness during epithelial morphogenesis. Curr Biol. 2010;20:513. doi: 10.1016/j.cub.2010.01.063. [DOI] [PubMed] [Google Scholar]
- 43.Reichman-Fried M, Shilo BZ. Breathless, a Drosophila FGF receptor homolog, is required for the onset of tracheal cell migration and tracheole formation. Mech Dev. 1995;52:265. doi: 10.1016/0925-4773(95)00407-r. [DOI] [PubMed] [Google Scholar]
- 44.Yagi R, Mayer F, Basler K. Refined LexA transactivators and their use in combination with the Drosophila Gal4 system. Proc Natl Acad Sci U S A. 2010;107:16166. doi: 10.1073/pnas.1005957107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Nellen D, Burke R, Struhl G, Basler K. Direct and long-range action of a DPP morphogen gradient. Cell. 1996;85:357. doi: 10.1016/s0092-8674(00)81114-9. [DOI] [PubMed] [Google Scholar]
- 46.Haerry TE, Khalsa O, O’Connor MB, Wharton KA. Synergistic signaling by two BMP ligands through the SAX and TKV receptors controls wing growth and patterning in Drosophila. Development (Cambridge, England) 1998;125:3977. doi: 10.1242/dev.125.20.3977. [DOI] [PubMed] [Google Scholar]
- 47.Massarwa R, Schejter ED, Shilo BZ. Apical secretion in epithelial tubes of the Drosophila embryo is directed by the Formin-family protein Diaphanous. Dev Cell. 2009 Jun;16:877. doi: 10.1016/j.devcel.2009.04.010. [DOI] [PubMed] [Google Scholar]
- 48.Ohshiro T, Emori Y, Saigo K. Ligand-dependent activation of breathless FGF receptor gene in Drosophila developing trachea. Mechanisms of development. 2002;114:3. doi: 10.1016/s0925-4773(02)00042-4. [DOI] [PubMed] [Google Scholar]
- 49.Ribeiro C, Ebner A, Affolter M. In vivo imaging reveals different cellular functions for FGF and Dpp signaling in tracheal branching morphogenesis. Developmental cell. 2002;2:677. doi: 10.1016/s1534-5807(02)00171-5. [DOI] [PubMed] [Google Scholar]
- 50.Klebes A, Biehs B, Cifuentes F, Kornberg TB. Expression profiling of Drosophila imaginal discs. Genome biology. 2002;3:RESEARCH0038. doi: 10.1186/gb-2002-3-8-research0038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Persson U, et al. The L45 loop in type I receptors for TGF-beta family members is a critical determinant in specifying Smad isoform activation. FEBS Lett. 1998;434:83. doi: 10.1016/s0014-5793(98)00954-5. [DOI] [PubMed] [Google Scholar]
- 52.Eulenberg KG, Schuh R. The tracheae defective gene encodes a bZIP protein that controls tracheal cell movement during Drosophila embryogenesis. The EMBO journal. 1997;16:7156. doi: 10.1093/emboj/16.23.7156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Guha A, Lin L, Kornberg TB. Organ renewal and cell divisions by differentiated cells in Drosophila. Proceedings of the National Academy of Sciences of the United States of America. 2008 Aug 5;105:10832. doi: 10.1073/pnas.0805111105. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Figure S1. Dpp signaling in the ASP. (A) Transverse section of the medial region of an unfixed ASP (btl-CD8:Cherry Dad-nlsGFP) showing GFP fluorescence in cells of lower layer and in the disc cells below. (B, B′) tkv expression (red, tkv-lacZ detected by anti-β-galactosidase staining) is higher in the upper (B) than lower (B′) level of the ASP (outlined by dashed white line). (C, C′) Expression of put (red, put-lacZ detected by anti-β-galactosidase staining) is similar in upper (C) and lower (C′) levels of the ASP (green, btl-Gal4 UAS-CD8:GFP). (D, D′) Optical sections showing Dad (D) and pMad (D′) assays of Dpp signaling in medial region of the upper and lower layers of ASP: (Dad-lacZ (red), α-pMAD staining (red), α-Dlg (green)). (E) RNA in-situ hybridization detects dpp expression in the disc but not in the trachea. Black dashed line marks position of ASP and TC. (F–N) Functional knockdown conditions in the ASP that induce morphogenetic malformations in the ASP (bifurcations and abnormal shapes) and tracheal duplications (arrows in (G, H). Genotypes: btl-Gal4, UAS-CD8:GFP; tub-Gal80ts X UAS-PutDN (F), X UAS-Dad (G), X UAS-TkvDN (H), X UAS-shits (I), X UAS-diaRNAi (J), X UAS-nrgRNAi (K), and X UAS-trnRNAi (N). shits ASP in (J) was incubated at 30°C for 1 hr followed by 20°C for 24 hrs. (L) Genotype: btl-Gal4 UAS-CD8:GFP / UAS-CapsDN; UAS-CapsDN and (M) btl-Gal4 UAS-CD8:GFP / UAS-CapsDN; capsC28fs trnΔ17 / UAS-CapsDN. Blue fluorescence in (G) and (H) is autofluorescence of lumen at 405 nm. (N) α-Dlg (red). Scale bar, 30 μm.
Figure S2. Characterizations of ASP in normal and mutant conditions. (A–D) Expression of diaRNAi (A, B) or nrgRNAi (C, D) did not change the number of α-phosphohistone-3 or α-Caspase-3 staining (red). (E, F) Expression of caps and trn detected by α-LacZ antibody ((E), caps-LacZ (P{PZ}caps02937; green) and GFP fluorescence ((F), trn-GFP). (G, H) Expression of CapsDN did not change the number of α-phosphohistone-3 or α-Caspase-3 staining (red). (I) Number and distribution of ASP cytonemes were not significantly changed by CapsDN over-expression. (B, G, H), α-Dlg (white); (A–D, G–I) CD8:GFP. Scale bar, 30 μm.
Figure S3: Cells with defective cytonemes do not activate signal transduction but are signaling competent. ASPs (outlined by dashed lines) that expressed under btl-Gal4: GFP (A–D), RNAi directed against dia (A), inactive Shits (B), RNAi directed against nrg (C), or CapsDN (D). α-pMad antibody stained ASP cells that also expressed Dpp:Cherry ((driven by btl-Gal4; upper panels) but did stain ASP cells that did not express Dpp:Cherry (lower panels). Right panels: pMad staining; middle panels Dpp:Cherry fluorescence; left panels: merge with CD8:GFP fluorescence. (E) ASPs that expressed GFP and CapsDN under btl-Gal4 stained for dpERK in the presence of ectopic FGF (driven by heat shock; right panel) but not in the absence of ectopic FGF (left panel). Scale bar: 30 mm.
Supplementary Movie S1: Movement of co-localized Dpp:GFP and Tkv:Cherry puncta in cytonemes. Tkv:Cherry was expressed in trachea (btl-Tkv:Cherry); Dpp:GFP was expressed in the dpp domain of the wing disc (dpp-Dpp:GFP). Each frame of the movie is a maximum Z-projection that was compiled with ImageJ, and is shown at two frames per second. Each Z-stack (0.4 μm steps) was captured at intervals of two time points per minute at 488 nm and 561 nm using an inverted spinning disc confocal microscope. Scale bar, 40 μm.


