Abstract
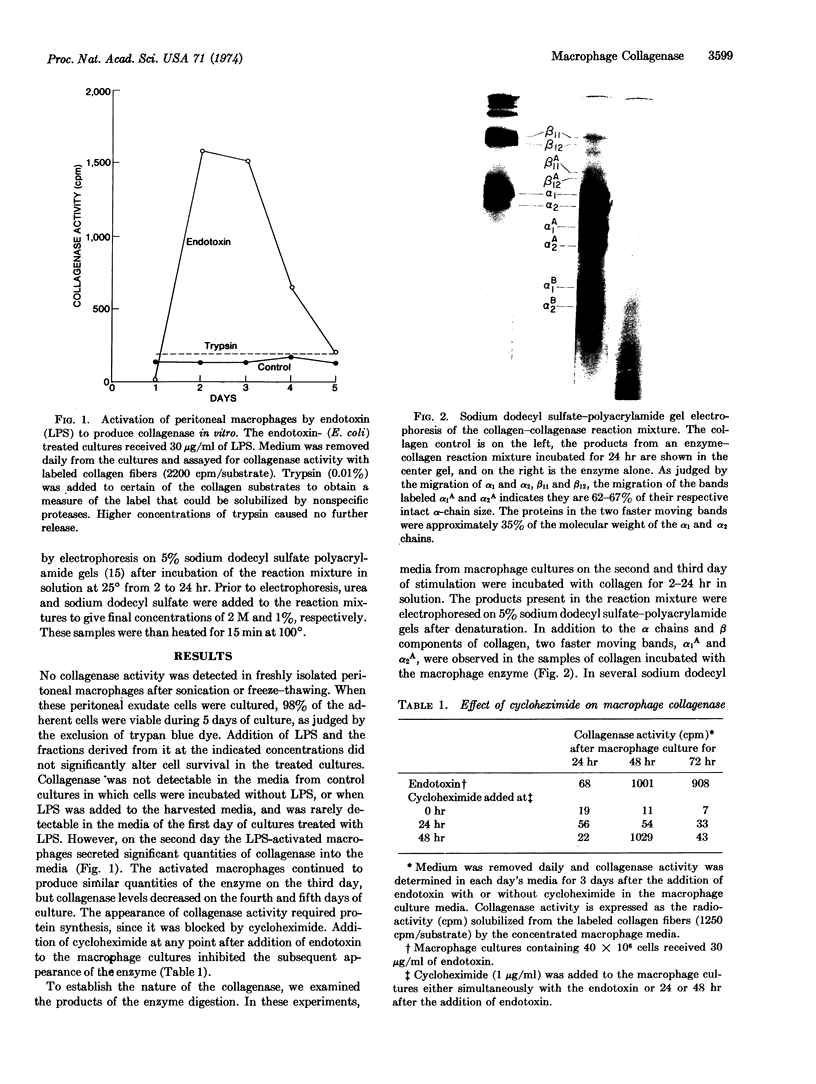
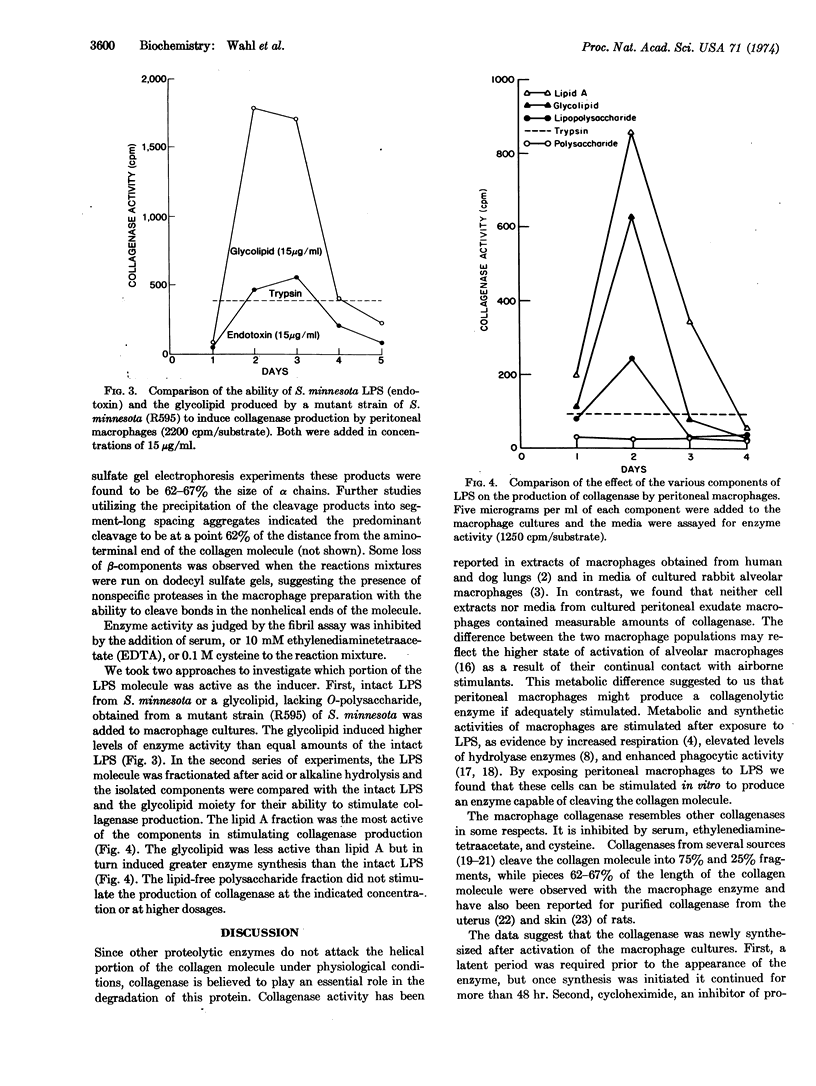
Peritoneal exudate macrophages, when exposed to bacterial lipopolysaccharide in culture, were found to produce collagenase (EC 3.4.24.3). This enzyme was not detected in extracts of the macrophages or in media from nonstimulated macrophage cultures. Lipidcontaining fractions of the lipopolysaccharide, including a glycolipid from the rough mutant of Salmonella minnesota (R595) and lipid A, were potent stimulators of collagenase production. The lipid-free polysaccharide fraction had no effect. Cycloheximide prevented the production of collagenase by endotoxin-treated macrophages, suggesting that it was newly synthesized.
Keywords: lipid A, lipopolysaccharide, connective tissue
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Andersson J., Möller G., Sjöberg O. Selective induction of DNA synthesis in T and B lymphocytes. Cell Immunol. 1972 Aug;4(4):381–393. doi: 10.1016/0008-8749(72)90040-8. [DOI] [PubMed] [Google Scholar]
- Bennett W. E., Cohn Z. A. The isolation and selected properties of blood monocytes. J Exp Med. 1966 Jan 1;123(1):145–160. doi: 10.1084/jem.123.1.145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chiller J. M., Skidmore B. J., Morrison D. C., Weigle W. O. Relationship of the structure of bacterial lipopolysaccharides to its function in mitogenesis and adjuvanticity. Proc Natl Acad Sci U S A. 1973 Jul;70(7):2129–2133. doi: 10.1073/pnas.70.7.2129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eisen A. Z., Jeffrey J. J., Gross J. Human skin collagenase. Isolation and mechanism of attack on the collagen molecule. Biochim Biophys Acta. 1968 Mar 25;151(3):637–645. doi: 10.1016/0005-2744(68)90010-7. [DOI] [PubMed] [Google Scholar]
- Furthmayr H., Timpl R. Characterization of collagen peptides by sodium dodecylsulfate-polyacrylamide electrophoresis. Anal Biochem. 1971 Jun;41(2):510–516. doi: 10.1016/0003-2697(71)90173-4. [DOI] [PubMed] [Google Scholar]
- Gery I., Krüger J., Spiesel S. Z. Stimulation of B-lymphocytes by endotoxin. Reactions of thymus-deprived mice and karyotypic analysis of dividing cells in mice bearing T 6 T 6 thymus grafts. J Immunol. 1972 Apr;108(4):1088–1091. [PubMed] [Google Scholar]
- Graham R. C., Jr, Karnovsky M. J., Shafer A. W., Glass E. A., Karnovsky M. L. Metabolic and morphological observations on the effect of surface-active agents of leukocytes. J Cell Biol. 1967 Mar;32(3):629–647. doi: 10.1083/jcb.32.3.629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gross J., Nagai Y. Specific degradation of the collagen molecule by tadpole collagenolytic enzyme. Proc Natl Acad Sci U S A. 1965 Oct;54(4):1197–1204. doi: 10.1073/pnas.54.4.1197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harper E., Bloch K. J., Gross J. The zymogen of tadpole collagenase. Biochemistry. 1971 Aug 3;10(16):3035–3041. doi: 10.1021/bi00792a008. [DOI] [PubMed] [Google Scholar]
- Jeffrey J. J., Gross J. Collagenase from rat uterus. Isolation and partial characterization. Biochemistry. 1970 Jan 20;9(2):268–273. doi: 10.1021/bi00804a012. [DOI] [PubMed] [Google Scholar]
- Lazarus G. S., Daniels J. R., Brown R. S., Bladen H. A., Fullmer H. M. Degradation of collagen by a human granulocyte collagenolytic system. J Clin Invest. 1968 Dec;47(12):2622–2629. doi: 10.1172/JCI105945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- NOWOTNY A. Relation of structure to function in bacterial O antigens. II. Fractionation of lipids present in Boivin-type endotoxin of Serratia marcescens. J Bacteriol. 1963 Feb;85:427–435. doi: 10.1128/jb.85.2.427-435.1963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagai Y., Lapiere C. M., Gross J. Tadpole collagenase. Preparation and purification. Biochemistry. 1966 Oct;5(10):3123–3130. doi: 10.1021/bi00874a007. [DOI] [PubMed] [Google Scholar]
- Nath I., Poulter L. W., Turk J. L. Effect of lymphocyte mediators on macrophages in vitro. A correlation of morphological and cytochemical changes. Clin Exp Immunol. 1973 Mar;13(3):455–466. [PMC free article] [PubMed] [Google Scholar]
- Nathan C. F., Karnovsky M. L., David J. R. Alterations of macrophage functions by mediators from lymphocytes. J Exp Med. 1971 Jun 1;133(6):1356–1376. doi: 10.1084/jem.133.6.1356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- OREN R., FARNHAM A. E., SAITO K., MILOFSKY E., KARNOVSKY M. L. Metabolic patterns in three types of phagocytizing cells. J Cell Biol. 1963 Jun;17:487–501. doi: 10.1083/jcb.17.3.487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosenstreich D. L., Nowotny A., Chused T., Mergenhagen S. E. In vitro transformation of mouse bone-marrow-derived (B) lymphocytes induced by the lipid component of endotoxin. Infect Immun. 1973 Sep;8(3):406–411. doi: 10.1128/iai.8.3.406-411.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tokoro Y., Eisen A. Z., Jeffrey J. J. Characterization of a collagenase from rat skin. Biochim Biophys Acta. 1972 Jan 20;258(1):289–302. doi: 10.1016/0005-2744(72)90986-2. [DOI] [PubMed] [Google Scholar]
- Tripodi D., Nowotny A. Relation of structure to function in bacterial O-antigens. V. Nature of active sites in endotoxic lipopolysaccharides of Serratia marcescens. Ann N Y Acad Sci. 1966 Jun 30;133(2):604–621. doi: 10.1111/j.1749-6632.1966.tb52392.x. [DOI] [PubMed] [Google Scholar]
- WESTPHAL O., NOWOTNY A., LUDERITZ O., HURNI H., EICHENBERGER E., SCHONHOLZER G. Die Bedeutung der Lipoid-Komponente (Lipoid A) für die biologischen Wirkungen bakterieller Endotoxine (Lipopolysaccharide). Pharm Acta Helv. 1958 Aug-Oct;33(8-10):401–411. [PubMed] [Google Scholar]
- Wiener E., Levanon D. The in vitro interaction between bacterial lipopolysaccharide and differentiating monocytes. Lab Invest. 1968 Dec;19(6):584–590. [PubMed] [Google Scholar]



