Abstract
The in vivo ganglionic environment directs the latent herpes simplex virus transcriptional program. Since stress-driven perturbations in sensory neurons are thought to play a critical role in the transition from latency to reactivation, a primary concern in the selection of a valid model of the molecular interactions leading to reactivation is the faithful recapitulation of these environments. In this study reactivation of latently infected ganglia excised and cultured in vitro (explanted) is compared to reactivation occurring in latently infected ganglia in vivo following hyperthermic stress. Three notable points emerged. (i) Neurons in explanted ganglia exhibited marked morphological changes within 2 to 3 h postexplant. DNA fragmentation in neuronal nuclei was detected at 3 h, and atypical expression of cell cycle- and stress-regulated proteins such as geminin, cdk2, cdk4, and cytochrome c became apparent at 2 to 48 h. These changes were associated with axotomy and explant and not with the initiation or progression of reactivation and were not observed in ganglia following in vivo hyperthermic stress. (ii) Despite these differences, during the first 22 h primary reactivation events were restricted to a very small number of neurons in vivo and in explanted ganglia. This suggests that at any given time only a few latently infected neurons are competent to reactivate or that the probability of reactivation occurring in any particular neuron is very low. Importantly, the marked changes detected in explanted ganglia were not correlated with increased reactivation, demonstrating that these changes were not associated with the reactivation process per se. (iii) Secondary spread of virus was evident in explanted ganglia within 36 h, an event not observed in vivo. We conclude that explant reactivation may provide an ancillary system for selected studies of the early events in reactivation. However, clear signs of neuronal degeneration within 2 to 3 h postexplant indicate that these ganglia are undergoing major physiological changes not associated with the reactivation process. This ongoing neurodegeneration could alter even the early virus-host interactions in reactivation, and thus caution in the extrapolation of results obtained in explants to the in vivo interactions initiating reactivation is warranted.
Herpes simplex virus (HSV) invades the host nervous system through the innervating sensory nerve terminals at the site of primary infection. In the sensory ganglion, acute viral replication is resolved within about 10 days postinoculation (p.i.). Despite complete clearance of infectious virus from the ganglion, the viral genome remains in a transcriptionally repressed (latent) state within the nucleus of a population of neurons. Periodically, stimuli including fever, exposure to UV radiation, and social stress trigger the transition from transcriptional repression to activation of the viral genome in a minor fraction of latently infected neurons. The result of this reinitiation of lytic viral gene transcription is the rapid production of a small amount of infectious virus in the ganglion, which can be transported to the surface and amplified through subsequent rounds of replication in permissive cells (for a review see reference 43). The molecular mechanisms controlling latency and reactivation remain poorly understood and a focus of active investigation.
The study of HSV pathogenesis has been greatly facilitated by small animal models that recapitulate most aspects of the viral life cycle observed in humans. The first definitive experimental evidence of viral latency in peripheral and central nervous tissue was obtained by explant cocultivation, a process in which the latently infected ganglion is removed from the animal and cultivated in vitro (9, 17, 32). This and related in vitro methods continue as commonly used procedures to confirm the presence of reactivation-competent virus in ganglia. Easy access to pharmacological manipulation has favored explant cocultivation in investigations of the molecular mechanisms underlying HSV latency and reactivation. More recently, techniques have been developed to induce viral reactivation in the intact animal. Induction triggers of in vivo reactivation in humans, such as UV irradiation and hyperthermic stress, have proved similarly effective in experimental animals, and several well-characterized models are currently in use (7, 25, 29; reviewed in reference 42).
Reactivation is the result of the disruption in latent-phase transcriptional repression, presumably by stress-induced signal transduction pathways that drive perturbations in the neuronal molecular environment. The ganglionic milieu is thus central to the latency-reactivation paradigm. Since the long-term objective is to understand and control HSV reactivation in the living human host, a primary concern in the selection of a model of the early molecular interactions leading to reactivation is the faithful recapitulation of the neuronal environment in vivo. In this study our goal was to gain a greater understanding of how viral reactivation in explanted ganglia resembles and differs from reactivation occurring in the intact host.
Presented here are the results of a direct comparison of the neuronal environment and characteristics of reactivation in latently infected trigeminal ganglia (TG) in vivo following hyperthermic stress and in ganglia removed from the animal and cultivated in medium (explant). Latently infected mice were divided into two groups. Identical analytical procedures were used to evaluate ganglia from mice induced to reactivate virus in vivo by hyperthermic stress and ganglia at various times post-explant into culture in vitro. The rate of reactivation, the cell type(s) and number(s) undergoing reactivation, and the amount of virus produced from 22 to 96 h after induction were determined. Additionally, neuronal morphology and the expression of selected cell cycle- and stress-related proteins were compared from 2 to 72 h postinduction in vivo and in vitro. We demonstrate that explanting ganglia into culture induces rapid morphological changes in neurons accompanied by DNA fragmentation and altered expression and/or intracellular distribution of cell cycle-related proteins. These changes did not correlate with the induction of reactivation from latency and were not observed in ganglia following hyperthermic stress in vivo.
MATERIALS AND METHODS
Viral strains and stock production.
Virus stocks of laboratory strains 17syn+ and KOS/M were generated by routine propagation on rabbit skin cell (RSC) monolayers. Infected cells were harvested and sonicated, and the titer of each stock was determined by serial-dilution plaque assay on RSC monolayers. The wild-type HSV type 1 (HSV-1) strain 17syn+ was originally obtained from John H. Subak-Sharpe at the Medical Research Council Virology Unit in Glasgow, Scotland. Strain KOS, originally obtained from M. Levine, University of Michigan, Ann Arbor, was plaque purified and designated KOS/M (36).
Inoculation of mice.
All procedures involving animals were approved by the Children's Hospital Institutional Animal Care and Use Committee and were in compliance with the Guide for the Care and Use of Laboratory Animals. Animals were housed in American Association for Laboratory Animal Care-approved quarters. Male, outbred, Swiss Webster mice (Harlan Laboratories), 5 to 6 weeks of age, were used throughout these studies. Prior to inoculation, mice were anesthetized by intraperitoneal injection of sodium pentobarbital (50 mg/kg of body weight). A 10-μl drop containing 105 PFU was placed onto each scarified corneal surface. This inoculum titer results in ∼80 and 100% survival of 17syn+- and KOS/M-infected mice, respectively, and 100% of ganglia were latently infected (23, 25). As indicated in one experiment, inoculum titer was reduced by serial 10-fold dilutions with medium, and inoculation was performed as described above.
Replication in vivo.
In order to confirm the efficiency of inoculation and progression of acute infection, three mice from each inoculation group were sacrificed on day 4 p.i., the day of peak replication in the TG. Eyes and TG were removed and assayed for infectious virus titer on RSC monolayers.
In vivo reactivation.
Latent HSV was induced to reactivate in the ganglia of mice in vivo by the use of hyperthermic stress. This procedure has been described in detail previously (25). In brief, latently infected mice were subjected to 43°C for 10 min in a water bath to induce reactivation. At various times postinduction tissues were examined for the presence of infectious virus or expression of select viral or host cell proteins as detailed in the text.
Explant reactivation.
Latent HSV was induced to reactivate in the ganglia of mice in vitro by aseptically removing the ganglia and placing them in minimal essential medium with 5% newborn calf serum in a 5% CO2 incubator at 37°C for the time indicated. At the indicated times postexplant, ganglia were homogenized and plated exactly as for reactivation in vivo. Additional medium and serum conditions were tested in conjunction with the morphological and immunohistochemical studies including the conditions employed by several different groups (11, 32, 35), as well as the addition of nerve growth factor (44).
Antibodies and immunohistochemistry.
Various groups of mice were perfusion fixed with 4% paraformaldehyde, and TG were dissected and fixed overnight at 4°C. Detection of cellular proteins was carried out on 8-μm sections of 4% paraformaldehyde-fixed paraffin-embedded tissues. Previously described dehydration and embedding procedures were utilized (20, 22, 25). Anti-cdk2 (M2-G, sc-163-G), anti-cdk4 (C-22-G, sc-260-G), antigeminin (M-16, sc-8450), and anti-cytochrome c (C-20, sc-8385) were all obtained from Santa Cruz Biotechnology, Inc. These primary antibodies were raised in goats and used at a 1:100 dilution in a three-step biotin-avidin-peroxidase or alkaline phosphatase assay. Antigen unmasking was required for the detection of the cdk2 and cdk4 epitopes and was performed using Vector's unmasking solution and protocol. The biotinylated anti-goat antibody (Vector) was used at a 1:200 dilution, followed by a 1:200 dilution of the peroxidase or alkaline phosphatase avidin conjugate (Vector). Localization of complexes was detected using a solution of diaminobenzidine (Aldrich) or Fast Red (Sigma) as described previously (20, 22, 25).
HSV proteins were detected in whole ganglia processed as described previously (12, 21). In brief, ganglia were fixed for 2 h in 0.5% paraformaldehyde, rinsed in phosphate-buffered saline (PBS), and postfixed overnight in methanol containing 20% dimethyl sulfoxide (DMSO). Ganglia were then incubated for 1 h in a solution of methanol containing 20% DMSO and 10% H2O2. Following two rinses in 100% methanol, ganglia were stored overnight in methanol at −70°C. Ganglia were allowed to equilibrate at room temperature for 15 min, rinsed twice in PBS, and incubated for 2 h at 37°C in PBS containing 3.6 mg of β-d(+)glucose (Sigma)/ml, 100 μg of glucose oxidase (Sigma)/ml, and 130 μg of sodium azide (Sigma)/ml. Ganglia were rinsed twice in PBS and incubated overnight in PBS containing 2% bovine serum albumin, 5% DMSO, 5% normal horse serum, and 0.5 μl of the rabbit anti-HSV antibody (AXL237; Accurate)/ml. Following rinsing for 5 h with multiple changes of PBS, ganglia were incubated overnight in horseradish peroxidase conjugate, rinsed for 5 h, and developed in 0.1 M Tris (pH 8.2) containing 250 μg of diaminobenzidine and 0.004% H2O2/ml.
Terminal deoxynucleotidyltransferase-mediated dUTP-biotin nick end labeling (TUNEL) assay.
DNA fragmentation was measured on 4% paraformaldehyde-fixed sectioned ganglia with the DeadEND colorimetric apoptosis detection system (Promega) according to the manufacturer's instructions as previously detailed (38). Tissues were viewed and photographed using an Olympus BX40 photomicroscope outfitted with an Olympus DP10 digital camera. Statistical analyses were performed using Graphpad Prism software.
RESULTS
Replication in vivo.
Viral titers in eyes and TG were determined on day 4 p.i. and ranged among individual animals from 3 × 104 to 4 × 105 and 8 × 103 to 1 × 105 in eyes and TG, respectively. This range is consistent with many previous experiments, indicating that no significant alterations in the inoculation or infection process had occurred.
Changes in host cell morphology and protein expression.
We first compared the basic morphology of neurons in ganglia at various times postexplant or post-hyperthermic stress with that of neurons in untreated ganglia. Cresyl violet-stained sections of TG from uninfected and latently infected mice before and at 2 to 3, 22, 48, and 72 h postexplant or post-hyperthermic stress in vivo were examined. For each treatment group, 8 or 10 ganglia (from four or five mice) per time point were embedded in a single paraffin block. Six sections (two groups of three consecutive sections) containing a minimum total of 48 ganglionic profiles were examined for each treatment group and time point, a total of 864 TG profiles. The average number of neurons per profile was ∼100; thus, on the order of 86,000 neurons were evaluated for each time point and treatment group. The same numbers of sections from these same groups of ganglia were analyzed by immunohistochemistry for the expression of geminin, cdk2, and cdk4, as well as the cytoplasmic release of cytochrome c. These proteins were selected because their expression in neurons reflects a major shift in the physiological state of the neuron. Because these analyses of explanted ganglia revealed morphological changes and protein expression indicative of neuronal degeneration and/or apoptosis, the TUNEL assay was also performed on six sections, two groups of three consecutive sections from each group and time point, to evaluate the extent of DNA fragmentation.
Cresyl violet staining.
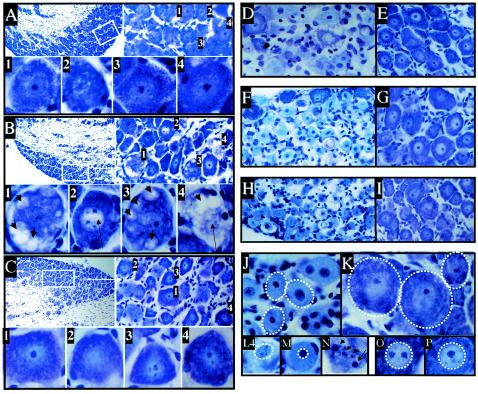
Compared to untreated ganglia (Fig. 1A), striking changes indicative of neuronal degeneration, including cytoplasmic vacuolization and nuclear clearing, were observed in explanted ganglia within 2 to 3 h postexplant (Fig. 1B), changes not present in ganglia subjected to hyperthermic stress in vivo (Fig. 1C). These results were the same in both uninfected and latently infected ganglia. At this early time postexplant, many neurons appeared normal but 22% of neurons exhibited the spectrum of morphological changes shown in Fig. 1B.
FIG. 1.
Morphological changes induced in TG following explant or in vivo hyperthermic stress. Shown are photomicrographs of cresyl violet-stained sections of TG from normal untreated ganglia (A) and ganglia 3 h postexplant (B) and 3 h post-hyperthermic stress (C). At the top left of each panel is a low-magnification micrograph. The boxed region is shown at higher magnification in the top right micrograph. Individual neurons labeled 1 to 4 are shown at high magnification at the bottom of each panel. Also shown are cytoplasmic vacuoles (short arrows) and reduced nuclear staining (long arrows). Sections of ganglia following explant (D, F, and H) or hyperthermic stress (E, G, and I) at 22 (D and E), 48 (F and G), and 72 (H and I) h are shown at the same magnification. Loss of Nissl substance is indicated by reduced staining in the explanted ganglia (D, F, and H) compared to normal ganglia (E, G, and I). In panels J (48 h postexplant) and K (48 h post-hyperthermic stress) the boundaries of the neurons are indicated with dashed lines. These panels are at the same magnification. Note the reduction in size of neurons in panel J compared to those in panel K. In panels L, M, and N (48 h postexplant) and O and P (48 h post-hyperthermic stress) the boundaries of neuronal nuclei are indicated by dashed lines. Note the change in size and staining pattern of the nuclei in explanted ganglia. An example of apoptotic bodies observed in explanted ganglia at 48 and 72 h is shown in panel N (arrows).
Morphological changes in neurons in explanted ganglia became increasingly widespread over the next 70 h (Fig. 1D, F, and H). Nuclear condensation, loss of Nissl substance (evidenced by reduction in the intensity of cresyl violet staining), reduction in average neuronal size (Fig. 1J and K) and nuclear size (Fig. 1L, M, O, and P), and appearance of apoptotic bodies (Fig. 1N) were the predominant changes observed. Within 48 h, fewer than 10% of neurons were morphologically normal compared directly to untreated neurons. In contrast, neurons in ganglia at 22, 48, and 72 h post-hyperthermic stress (Fig. 1E, G, and I) exhibited none of these changes and were indistinguishable from untreated neurons (Fig. 1A). These results were the same in both uninfected and latently infected ganglia. Similar results were obtained when several alternative explant culture conditions employed previously by others were tested (32, 35, 37). No conditions tested prevented the morphological changes observed (data not shown).
Protein expression.
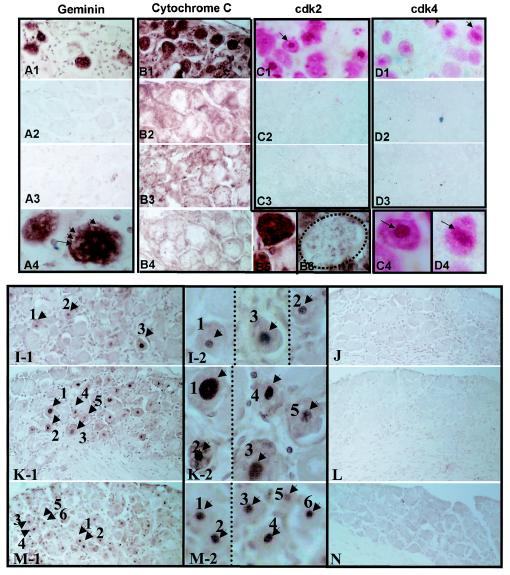
Neurons in explanted ganglia exhibited alterations in the expression and/or localization of the proteins examined compared to that of either untreated ganglia or ganglia following hyperthermic stress (Fig. 2). These changes occurred in both uninfected and latently infected ganglia, indicating that the changes observed resulted from axotomy and explant and not viral latency or reactivation. There was a direct correlation between the time postexplant and the number of neurons exhibiting changes in protein expression. Geminin, a protein involved in blocking the initiation of inappropriate DNA replication in dividing cells (13, 14, 45), is not detected in untreated uninfected or untreated latently infected TG neurons (Fig. 2A3 and data not shown). This protein was expressed in ∼10% of neurons within 2 h (Fig. 2A1), which increased to ∼50% by 22 h postexplant (data not shown). Geminin-expressing neurons (Fig. 2A4) exhibited morphological changes similar to those observed in cresyl violet-stained sections (Fig. 1B1 and B3). In contrast, geminin was not observed in neurons in uninfected or latently infected ganglia at any time following hyperthermic stress (data not shown and Fig. 2A2). Intense cytoplasmic cytochrome c staining was observed in some neurons within 2 to 3 h postexplant (data not shown). By 22 h postexplant, most neurons contained elevated levels of cytochrome c in the cytoplasm (Fig. 2B1 and B5). In contrast, the cytochrome c staining in neurons post-hyperthermic stress (Fig. 2B2 and B6) was not different at any time from that observed in untreated uninfected or latently infected ganglia (Fig. 2B3 and B4, respectively). Schang et al. reported the upregulation and nuclear localization of cdk2 and cdk4 in neurons in explanted ganglia (26). Our findings were consistent with the exception that these investigators reported cdk4 expression in the cytoplasm of neurons prior to explant, which we did not observe (Fig. 2D3). By 72 h postexplant, ∼70% of the neurons were positive for cdk2 and ∼40% were positive for cdk4 and many of the positive neurons exhibited strong nuclear staining (Fig. 2C1 and D1, respectively). However, as in untreated ganglia (Fig. 2C3 and D3), cdk2 and cdk4 remained at undetectable levels following hyperthermic stress in vivo (0 positive of ∼90,000 neurons tested) (Fig. 2C2 and D2). Thus, none of the changes occurring in the expression of the cell cycle-regulatory proteins in neurons following explant were observed in neurons following hyperthermic stress in vivo.
FIG. 2.
Expression of cell cycle- and stress-related proteins and fragmented DNA in TG neurons following explant or in vivo hyperthermic stress. (Top) Immunohistochemical detection of neuronal protein expression. (A1 to A4) Detection of geminin at 2 h postexplant (A1) or post-hyperthermic stress (A2) or in untreated ganglia (A3). The brown precipitate indicates a positive reaction. Higher magnification of geminin-expressing neuron in explanted ganglia shows that cytoplasmic vacuoles (arrows) are abundant in this neuron (A4). (B1 to B6) Immunohistochemical staining for cytochrome c in latently infected ganglia at 22 h postexplant (B1) and post-hyperthermic stress (B2). Staining of untreated uninfected ganglia (B3) and untreated latently infected ganglia (B4) is indistinguishable from that of latently infected ganglia post-hyperthermic stress, light positive punctate staining (brown precipitate). Latently infected ganglia 22 h postexplant display strong diffuse cytoplasmic staining (B1) shown at a higher magnification in B5. For comparison, cytochrome c staining of a typical neuron from ganglia subjected to hyperthermic stress 22 h previously is shown in B6. The boundary of the neuronal cell body is indicated by a dashed line, emphasizing the difference in neuronal size after explantation (B5). Panels C1 through C4 and D1 through D4 show staining for cdk2 and cdk4, respectively. Compare the absence of staining in untreated ganglia (C3 and D3) or ganglia at 72 h post-hyperthermic stress (C2 and D2) to the cytoplasmic and nuclear staining for both cdk2 and cdk4 at 72 h postexplant (red precipitate). Neurons indicated by arrows in panels C1 and D1 are shown at higher magnification in panels C4 and D4. (Bottom) TUNEL staining. TUNEL reactions in ganglia at 3, 24, and 48 h postexplant (I to M) are shown. Arrows and numbers indicate examples of positive reactions in nuclei (brown precipitate), which are shown at higher magnification in the center panels (I-2 to M-2). Most neurons were positive by 48 h postexplant (M-1, arrows, and M-2 at a higher magnification). Shrinkage of neuronal cell bodies and nuclei is evident (M-2). In contrast, no TUNEL-positive neurons were detected in ganglia at 3, 24, or 48 h post-hyperthermic stress (J, L, and N, respectively).
TUNEL assay.
Axotomy induces apoptosis in sensory neurons in vivo (1, 5), and it is reasonable to assume that apoptosis of neurons would also occur in the setting of explant in vitro. Sections from paraffin-embedded uninfected and latently infected ganglia harvested at various times postexplant or post-hyperthermic stress were analyzed for DNA fragmentation by the TUNEL assay to determine the extent and rate of apoptosis occurring in neurons. TUNEL-positive neuronal nuclei were present in explanted ganglia by 3 h postexplant in both uninfected and latently infected ganglia (Fig. 2I-1 and I-2 and data not shown). After 24 h in culture, a significant number (∼50%) of neuronal nuclei showed intense TUNEL-positive staining regardless of whether the mice were previously infected (Fig. 2K-1 and K-2 and data not shown). The number of neurons exhibiting DNA fragmentation increased in parallel with the amount of time postexplant, and by 48 h postexplant most neuronal nuclei were positive (Fig. 2M-1 and M-2). In contrast, no TUNEL-positive neurons were detected in untreated groups (data not shown) and likewise no TUNEL-positive neurons were detected in vivo following hyperthermic stress at any time point tested regardless of infection status (0 positive of ∼270,000; Fig. 2J, L, and N).
Changes in stress-related protein expression and neuron morphology in explanted ganglia are not correlated with changes in the timing, magnitude, or outcome of reactivation.
Viral reactivation is defined as the production of infectious virus from a previously nonproductive (latent) viral genome (2). Thus, the timing and quantity of infectious virus production are considered important diagnostic features of the reactivation process. If the extensive changes seen in explanted ganglia were important for initiating viral reactivation, one would predict that the number of neurons reactivating and/or the rapidity in which reactivation was initiated would differ from that seen in vivo. To test this, groups of mice inoculated with strain 17syn+ 40 to 50 days previously were either reactivated in vivo by the use of hyperthermic stress or ganglia were explanted. The time of reactivation stimulus was considered the time of hyperthermic stress or explant. At various times post-reactivation stimulation, individual ganglia were assayed for infectious virus as described in Materials and Methods.
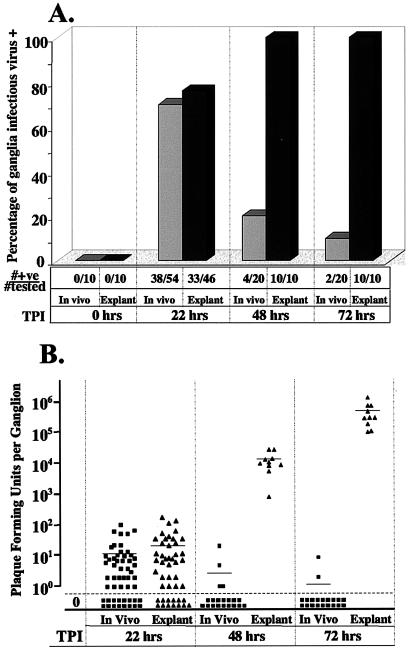
Infectious virus was detected in equivalent percentages of ganglia in vivo (70%, 38 of 54) and in explant (72%, 33 of 46) at 22 h postinduction (P = 1.0, Fisher's exact test) (Fig. 3A). In order to confirm that infectious virus was absent prior to hyperthermic stress or explant, five mice from the same inoculation group were sacrificed and ganglia were removed, homogenized, and immediately plated on indicator monolayers. There was no evidence of infectious virus in these ganglia (0 positive of 10 tested, P < 0.0001, Fisher's exact test). Thus, the infectious virus detected in heat-stressed or explanted ganglia was not preexisting but was generated during the 22 h postinduction. There was also no difference in the amount of virus detected in ganglia reactivated in vivo or in explanted ganglia, which averaged 11 and 21 PFU/ganglion, respectively (P = 0.14, Student's unpaired t test) (Fig. 3B).
FIG. 3.
Frequencies of ganglia positive for virus and virus titers present in ganglia at various times post-reactivation stress. At the indicated times poststress, ganglia were homogenized and plated onto RSC monolayers to detect reactivated virus. (A) The bar graph illustrates the percentage of ganglia positive at various times poststress. Light gray bars, hyperthermic stress; dark gray bars, explant into culture. The numbers of ganglia positive for virus and the numbers tested are indicated below the bar graph. (B) Scattergram in which each point represents the titer obtained from a single reactivated ganglion. Filled squares, post-hyperthermic stress; filled triangles, postexplant. A horizontal line indicates the mean titer of each group. TPI, time postinduction.
However, the difference between the frequency of reactivation following hyperthermic stress in vivo and explant was significant at both 48 h (P < 0.0001, Fisher's exact test) and 72 h (P < 0.0001, Fisher's exact test) postinduction (Fig. 3). The number of infectious-virus-positive ganglia detected in the hyperthermic stress group had declined from the 70% detected at 22 h to 20% (4 of 20) at 48 h post-hyperthermic stress (P = 0.0001, Fisher's exact test) and 10% (2 of 20) at 72 h (P < 0.0001, Fisher's exact test). In contrast, 100% (10 of 10) of the explanted ganglia were positive at 48 and 72 h. The amounts of infectious virus recovered from ganglia in vivo and in explanted ganglia were also different. As shown in Fig. 3B, ganglia cultivated in vitro contained >104 PFU at 48 h and >105 PFU at 72 h. Most ganglia maintained within the animal during the 48 or 72 h post-hyperthermic stress contained no detectable infectious virus. Those that were positive contained an average of <5 PFU Since the ganglia from both groups were assayed identically, the difference in frequency of virus detection and titer could not be attributed to differences in the sensitivity of the procedure utilized to detect infectious virus.
Effect of hyperthermic stress on virus production in explanted ganglia.
It was possible that the differences in virus production observed at 48 h resulted from changes due to hyperthermic stress and not explantation, i.e., hyperthermic stress could theoretically result in reduced permissiveness for viral replication in the ganglia. In order to test this, groups of mice latently infected with strain 17syn+ were subjected to hyperthermic stress or left untreated. The animals were sacrificed 22 h after hyperthermic stress, and the ganglia were explanted for 0, 22, 48, or 72 h. Untreated latently infected mice were also sacrificed, and the ganglia were explanted for 22, 48, and 72 h. The number of virus-positive ganglia and the amount of virus in each ganglion were determined. At 22 h postexplant, a greater percentage of ganglia from those mice that were subjected to hyperthermia 22 h before explant than from the hyperthermia or explant-alone control groups was positive for virus (Table 1). This difference achieves statistical significance if the control groups are grouped together, 17 of 18 versus 13 of 20 (P = 0.04, Fisher's exact test). This is not surprising since the hyperthermic stress treatment 22 h before explant would have induced reactivation in many of the ganglia. It is also clear that the mean titer of virus in these ganglia (88.3 PFU; range, 0 to 508 PFU) is greater than that in the explant (16.5 PFU; range, 0 to 69 PFU) or hyperthermia-alone (6.3 PFU; range, 0 to 25 PFU) group. Again, this difference achieves statistical significance if the control groups are combined (P = 0.01, Student's unpaired t test). Within 48 h, explanted ganglia were uniformly positive for infectious virus and viral titers were equivalent, regardless of hyperthermic stress pretreatment (Table 1). Thus, hyperthermic stress treatment of ganglia in vivo did not result in reduced permissiveness when ganglia were explanted.
TABLE 1.
Effect of hyperthermic stress on virus production in explanted ganglia
| Treatment | Result after time of explant (h):
|
|||||
|---|---|---|---|---|---|---|
| 22
|
48
|
72
|
||||
| % Virus positive (no. positive/no. tested) | Mean viral titera | % Virus positive (no. positive/no. tested) | Mean viral titera | % Virus positive (no. positive/no. tested) | Mean viral titera | |
| Explant | 70 (7/10) | 16.5 | 100 (10/10) | 1.4 × 104 | 100 (10/10) | 5.5 × 105 |
| Hyperthermia | 60 (6/10) | 6.3 | 10 (1/10) | 0.2 | 0 (0/10) | 0 |
| Hyperthermia + explantb | 94 (17/18) | 88.3 | 100 (10/10) | 1.5 × 104 | 100 (10/10) | 2.3 × 105 |
Mean viral titers are the averages of the numbers of PFU recovered from each pair of ganglia.
Mice were subjected to hyperthermic stress, and 22 h later ganglia were explanted for an additional 22, 48, or 72 h.
The preceding experiments demonstrated that, with the use of identical assays to detect infectious virus production in the ganglia, viral replication associated with reactivation was similar in vivo and in vitro during the first 22 h postinduction. Therefore, the physiological changes detected in explanted ganglia during this timeframe did not correlate directly with increased reactivation from latency. In an effort to further compare reactivation in vivo and in vitro, we asked if inoculum titer and viral strain, factors previously demonstrated to influence reactivation in vivo (20, 39, 40), also influenced reactivation in explanted ganglia.
Effect of inoculum titer.
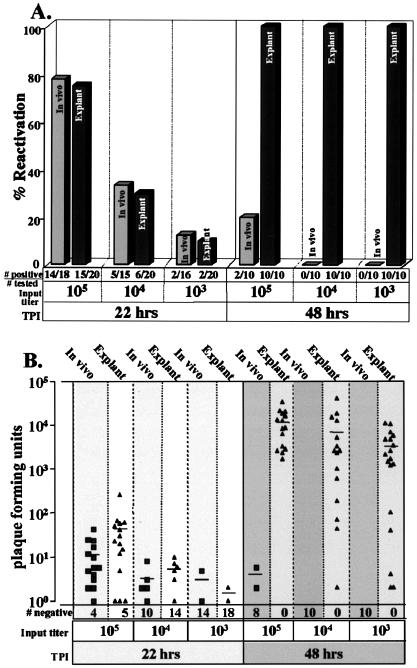
The frequency of reactivation in vivo has been shown elsewhere to correlate directly with the inoculum titer (20). Therefore, groups of mice were inoculated with either 105, 104, or 103 PFU. At >35 days p.i., animals were either subjected to hyperthermic stress or sacrificed and the ganglia were explanted. At 22 and 48 h postinduction, ganglion pairs were homogenized and assayed for infectious virus as described above. The results are shown in Fig. 4. At 22 h postinduction, 78 and 75% of the ganglia from mice inoculated with 105 PFU reactivated following hyperthermic stress or explant, respectively (P = 1.0, Fisher's exact test). At this same time, only 33 and 30% of ganglia from mice inoculated with 104 PFU and 13 and 10% of ganglia from mice inoculated with 103 PFU were positive for infectious virus following hyperthermic stress or explant, respectively (P = 1.0, Fisher's exact test). Thus, there was no difference between the frequency of reactivation in vivo following hyperthermic stress and that in explant during the first 22 h postinduction, and as previously shown for reactivation in vivo, there is a direct correlation between the input titer and the percentage of ganglia that reactivated in explanted ganglia at 22 h (Fig. 4A). The frequency of reactivation in ganglia from mice receiving 105 PFU was significantly different from that in ganglia from mice inoculated with 104 PFU (P = 0.0002, Fisher's exact test) and 103 PFU (P < 0.0001, Fisher's exact test). Likewise, the reactivation frequency in ganglia from mice inoculated with 104 PFU was different from that in ganglia from mice inoculated with 103 PFU (P = 0.045, Fisher's exact test). By 48 h, 100% of explanted ganglia from all input titer groups were positive for infectious virus. In striking contrast, among ganglia undergoing reactivation in vivo, even at the highest input titer the percentage of positive animals decreased from 78 to 20% at 48 h postinduction, and no positive ganglia were detected in the lower-input titer groups at this time (Fig. 4A). Thus, by 48 h, the significant differences in reactivation frequencies resulting from input titer were overwhelmed by differences in ganglionic environment, the in vivo reactivation frequency of 20% being significantly different from the 100% observed in explanted ganglia (P = 0.0007, Fisher's exact test). The amount of virus recovered from the reactivating ganglia was not significantly different at 22 h between ganglia reactivating post-hyperthermic stress and those reactivating postexplant (105 PFU, P = 0.11; 104 PFU, P = 0.56; 103 PFU, P = 0.47) and significantly decreased in both groups with decreasing input titer (105 versus 104, P = 0.010; 104 versus 103, P = 0.019; 105 versus 103, P = 0.006) (Fig. 4B).
FIG. 4.
Analysis of the effect of the inoculum titer on the frequency of reactivation and virus titer within ganglia. (A) Percentage of ganglia positive for virus at 22 or 48 h post-hyperthermic stress in vivo (light gray bars) or explant into culture (dark gray bars). Beneath each bar graph the number of ganglia positive and the number tested are indicated. (B) Scattergram showing the titers of virus recovered from individual ganglia. Filled squares, reactivation following hyperthermic stress; filled triangles, reactivation in culture. A horizontal bar indicates the mean value for each group, and the number of ganglia negative for virus is shown beneath each group. TPI, time postinduction.
One additional point of interest is that, although input titer did not influence the number of explanted ganglia positive for virus at 48 h (100% for all explanted groups), the range of PFU detected in the ganglia within the inoculation group broadened as the inoculum titer was reduced. Among the 15 mice receiving 105 PFU, the titers at 48 h postexplant ranged 20-fold (1.7 × 103 to 3.4 × 104 PFU). In contrast, the range of viral titers in ganglia from the mice receiving an input titer of 104 or 103 PFU spanned over 10,000-fold, 2 × 100 to 4 × 104 and 2 × 100 to 1.1 × 104, respectively. These results suggest that variability in reactivation among animals can be reduced by using an optimum inoculation titer.
Effect of viral strain.
Viral genetic factors influence the frequency of reactivation. HSV-1 strain KOS reactivates in ∼3-fold-fewer mice than does strain 17syn+ in vivo when equivalent levels of latent infections are established (23). To determine whether this difference would also be observed in explanted ganglia, groups of mice were inoculated with strain KOS or 17syn+. At >35 days p.i., mice were induced to reactivate in vivo or in vitro as described above. At 22 h postinduction, ganglia were homogenized and assayed for infectious virus. As anticipated, reactivation occurred in equivalent numbers of 17syn+-infected ganglia at 22 h post-hyperthermic stress or explant, 80% (8 of 10) and 75% (9 of 12), respectively (P = 1.00, Fisher's exact test). Although the frequency of reactivation was reduced in KOS-infected ganglia (an expected outcome), the number of ganglia reactivating at 22 h postexplant, 40% (4 of 10), was not different from the number reactivating at 22 h post-hyperthermic stress, 38% (5 of 13) (P = 1.00, Fisher's exact test). Thus, the effect of viral strain observed in vivo was recapitulated in the first 22 h postexplant. With strain KOS-infected animals, by 48 h postinduction none (0 of 16) of the hyperthermic stress-treated mice were positive whereas all (10 of 10) of the explanted ganglia were positive (P < 0.0001, Fisher's exact test). This finding was similar to that with strain 17syn+ in vivo, where only 20% (4 of 20) were positive at 48 h versus 100% (10 of 10) in vitro (P < 0.0001, Fisher's exact test). Taken together with the preceding section, these results show that the impact of inoculum titer and viral strain on reactivation shown previously in vivo (20, 21, 23, 25, 39) is also observed in explanted ganglia, but not beyond 22 h postinduction. In contrast, at 48 h many ganglia that did not reactivate in vivo did reactivate in the explant setting. Whether this difference reflects the effects of continued stress and/or relaxed requirements for reactivation as the physiological state of the explanted neurons degrades is not yet known.
Type and number of cells expressing lytic viral proteins.
The type and number of cells in the ganglia expressing lytic viral proteins associated with reactivation in vivo have been reported by several groups, including our own. There is agreement that during reactivation viral proteins are expressed in neurons, as opposed to other cell types, and that these neurons are extremely rare (15, 21, 23, 25, 28, 30). In order to determine whether reactivation in explanted ganglia is similar to reactivation in vivo in this regard, ganglia were examined at 22, 48, and 72 h post-hyperthermic stress or postexplant by the use of whole-ganglion immunohistochemistry for HSV lytic proteins. Rare HSV antigen-positive neurons were detected in ganglia at 22 h postinduction. At this time we observed neither significant quantitative nor qualitative differences in staining in ganglia undergoing reactivation in vivo and in vitro. In vivo 67% (16 of 24) of the ganglia contained one or more positive neurons and the average number of positive neurons per ganglion was 2.3 (range, 0 to 13). Likewise in explanted ganglia at 22 h 73% (16 of 22) of the ganglia were positive, and the average number of positive neurons was 2.8 (range, 0 to 13). These percentages were not significantly different (P = 0.75, Fisher's exact test).
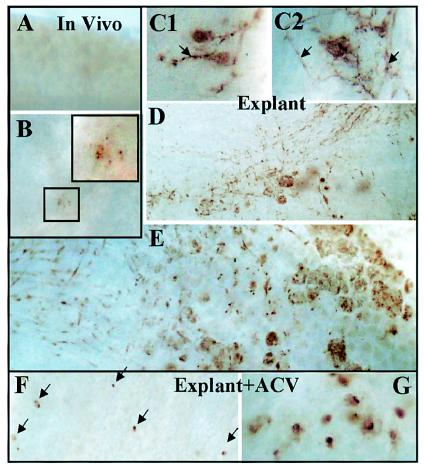
Consistent with our analysis of infectious virus production presented above, by 48 h ganglia reactivated in vivo exhibited a reduced frequency of positive ganglia (30%) and a reduced number of positive neurons per ganglion, which were most often associated with inflammatory infiltrate (Fig. 5A and B). At this time viral antigen was detected in neurons in 100% (10 of 10) of the explanted ganglia. In contrast to involvement of a single neuron as observed in vivo, within 36 h postexplant, areas of positive reaction to HSV proteins involving multiple cells, including satellite and support cells, were apparent (Fig. 5C). These positive regions increased in size through time, and by 72 h postexplant there was extensive neuronal, support cell, and axonal tract staining (Fig. 5D and E). These observations were consistent with the amount of infectious virus found in similarly treated ganglia above and suggest that the major contribution to the infectious virus produced in explanted ganglia beyond 22 h postinduction comes from spread of virus within the ganglion secondary to primary reactivation.
FIG. 5.
Analysis of viral protein expression as a marker for reactivation and virus spread in vivo following hyperthermic stress and in vitro following explant into culture. Shown are micrographs of whole ganglia stained immunohistochemically for viral proteins at 48 h post-in vivo hyperthermic stress (A and B), explanted into culture (C1 and C2, 36 h; D, 48 h; E, 72 h), or explanted into culture in the presence of acyclovir (ACV) (G). Most ganglia subjected to in vivo hyperthermic stress were negative at 48 h poststress (A), but 20% of the ganglia tested contained one or more positive neurons that frequently showed signs of degeneration and fragmentation (B and insets). All of the explanted ganglia at 48 and 72 h postexplant contained immunoreactive regions resembling three-dimensional plaques with many clusters of positive cells (D and E). When acyclovir was added to similar cultures, positive neurons were detected, but viral protein was restricted to individual neurons (F, 72 h postexplant; G, 96 h postexplant).
Spread of virus versus asynchronous reactivation.
While the analysis of lytic viral proteins in explanted ganglia was indicative of the spread of reactivated virus through the ganglia, it was formally possible that some or all of the lytically infected neurons represented recruitment of additional latently infected neurons to undergo primary reactivation. Distinguishing between primary reactivation and spread of infectious virions was possible because we recently determined that viral DNA replication is not required for viral protein expression in neurons entering into the lytic cycle following a reactivation stimulus (N. M. Sawtell, unpublished data). Since the same numbers of neurons initiate lytic-phase transcription and express viral proteins in the presence of viral DNA replication as in its absence, it is possible to block the production of infectious progeny and determine the number of neurons in which reactivation is initiating. Antiviral drugs were employed to determine the relative contributions from primary reactivation or from secondary spread of virus to the observed viral protein expression in the ganglia.
Ganglia were explanted in media with and without 1 μg of acyclovir/ml to inhibit viral DNA replication and examined at 72 and 96 h postexplant. Consistent with our previous experiments, ganglia explanted for 3 days contained foci of viral protein-positive neurons and distribution of viral proteins in axons consistent with lateral spread and distant transport of virus (Fig. 5E). In contrast, ganglia explanted for 3 or 4 days in the presence of acyclovir contained many single discrete viral protein-positive neurons and no evidence of spread to neighboring cells in the ganglion. The number of viral protein-expressing neurons was significantly greater than that observed at 22 h (Fig. 5F and G). These data indicate that the initiation of reactivation in explanted ganglia is not restricted to the first 22 h but is an ongoing process. In contrast, reactivation in vivo is characterized by a transient stress stimulus and a transient reactivation event that resolves within most ganglia between 48 and 72 h postinduction.
DISCUSSION
The molecular mechanisms directing the reactivation of the latent HSV genome remain unknown. A detailed knowledge of the timing and progression of primary reactivation in the ganglia is required to rationally explore these mechanisms. Only by understanding the similarities and differences among models of reactivation will it be possible to develop a cohesive experimental effort among groups working toward understanding this process. In this report, we characterize the reactivation of latent HSV in ganglia in two widely used model systems, comparing the timing of infectious virus production, the quantity of virus produced, and the number of neurons undergoing reactivation. The extent and similarity of physiological changes in the ganglion resulting from the different induction triggers utilized (hyperthermic stress and axotomy followed by explant) were also evaluated. The results of our study provide essential baseline information to facilitate both the interpretation of past studies of HSV reactivation and the design for future studies.
Three major points emerged from this study. First, the physiologic state of explanted ganglionic neurons differs extensively from that of neurons in ganglia induced to reactivate in vivo. In explanted ganglia, neurons exhibited marked morphological changes soon after explant, atypical expression and/or localization of most cell cycle- and stress-related proteins examined, and DNA fragmentation in neuronal nuclei. The number of neurons exhibiting these changes increased as the time postexplant increased. Importantly, these changes were also observed in uninfected explanted ganglia and were absent from ganglia undergoing quantitatively similar reactivation in vivo. Thus, we conclude that these changes resulted from axotomy and explant and not from the initiation or progression of virus reactivation.
The unusual expression in neurons of G1/S-phase protein markers (e.g., cdk2 and cdk4), the cytoplasmic location of cytochrome c, and TUNEL-positive nuclei following explantation indicate that axotomy followed by explant of the ganglia results in the initiation and progression of apoptosis in neurons. Indeed the observed changes are consistent with other reports detailing the neuronal response to axotomy (1, 5). Standard culture conditions for ganglion explant were used (9, 32, 37, 41), and several adjustments to the medium did not significantly alter the outcome.
Second, it is reasonable to anticipate that within a given period of time a greater number of neurons would reactivate in response to the extreme stimulus of axotomy and explant; however, this did not prove to be the case. The profound physiological changes in explanted ganglia did not influence either the number of neurons undergoing reactivation or the number of PFU recovered from ganglia latently infected with wild-type strain 17syn+ or KOS during the first 22 h. In both cases, reactivation occurred only in very rare neurons (one to seven per ganglion with most positive ganglia containing one or two positive neurons). Prior work demonstrates that these ganglia each contained on the order of 6,000 latently infected neurons with a total number of viral genomes in the range of 105 to 106 (19-21, 23, 38-40). Thus, reactivation is an exceedingly rare event occurring in fewer than 1 out of 1,000 latently infected cells.
The rarity of reactivation events that we and others have observed in vivo (15, 21, 23, 25, 28, 30) is consistent with the hypothesis that, at any given time, the latent pool contains a small subset of neurons that are competent to undergo reactivation in response to the appropriate signals. We have found that, despite repeated exposure to hyperthermic stress, the reactivation frequency within a group of mice is maintained through time (unpublished observation). That is, similar numbers of neurons reactivate regardless of whether the animal has been exposed to a reactivation stimulus just once or multiple times over a period of weeks or months. Assuming that neurons that produce infectious virus do not survive (15, 16, 21, 25, 28-31), this observation indicates either that there is a mechanism for some latently infected cells to become reactivation competent at any given time or that all latent genomes are equally competent with a very low probability of reactivating.
Most latently infected neurons contain between 1 and 100 HSV genomes, but very rare neurons contain significantly more than 1,000, suggesting that the latent genome copy number may influence reactivation (19-21, 23, 38-40). It may be that there is a threshold of genome copies required for reactivation. In this model some mechanism, perhaps occult replication of the viral genome in latently infected neurons, (19, 23, 27), results in a few reactivation-competent neurons at any given time. However, our findings do not exclude the alternate hypothesis that every viral genome has an equal, but very low (∼1:105), probability of reentering the lytic pathway. It follows from this hypothesis that 1 out of 10 neurons latently infected with 100 HSV genome copies (1,000 total HSV genomes) would have the same probability of reactivating as would a single neuron containing 1,000 latent viral genomes. In this model there would be no need of a clocking mechanism that would permit only a few neurons to be reactivation competent at any given time. Neither the nature of “competency” nor the mechanism by which this occurs is understood. Although a role for viral genome copy number is an attractive hypothesis (19, 23, 27) and there are data consistent with this hypothesis (23), the relationship between copy number and reactivation competency has yet to be fully delineated.
Regardless of mechanism, the finding that this low number of reactivating neurons is initially unaltered by the physiological changes in explanted neurons indicates that the profound changes occurring within the first 22 h after axotomy and explant were not sufficient to overcome the preexisting barriers to reactivation. Indeed, were these changes precipitating events in reactivation one would predict that more neurons would have initiated reactivation in the explanted ganglia during the first 22 h, and this was not the case. It is therefore clear that, from the aspect of host cell signals, explant is an unnecessarily complex environment in which to dissect the molecular pathway(s) leading to reactivation. Importantly, the complexity of the physiologic changes occurring in explanted ganglionic neurons could obviate the need for certain viral functions required for reactivation in vivo and lead to spurious conclusions about the mechanics of reactivation. We have already identified one example of this type of environmental compensation. A 365-bp region of the viral genome that is required for reactivation in vivo is not required for reactivation in the explant setting (unpublished data).
The third observation of importance was that there was cell-to-cell spread of virus within the ganglia by 36 h postexplant, an event not observed in vivo. Viral proteins were expressed in explanted ganglia in clusters of neurons, satellite cells, and cells along axonal tracts. Consistent with an increased number of infected cells, the infectious virus titer in explanted ganglia increased ∼1,000-fold from 22 to 48 h postexplant. It was not possible to directly determine the relative contributions of primary reactivation and subsequent secondary infection to the infectious virus output. Acyclovir was added to the explant cultures to determine if primary reactivation events continue to occur after 22 h postexplant and to distinguish between those neurons undergoing primary reactivation and those infected as a result of intraganglionic spread of virus. There was a steady increase in the number of neurons expressing viral proteins postexplant (Fig. 5). In the presence of acyclovir, the number of neurons initiating reactivation increased about 10-fold from 22 to 48 h postexplant. However, in the absence of acyclovir, virus titer increased 1,000-fold, indicating that the majority of virus in the explanted ganglion at 48 h is generated through spread within the ganglion. The absence of full immune function is a possible explanation for this increased viral spread (3), but it is also possible that explant-induced changes in neurons and/or supporting cells increase the permissivity for HSV replication. Regardless, it is quite clear that, distinct from reactivation in vivo, neurons continue to reactivate in explanted ganglia. However, at times beyond 22 h postexplant, most transcriptional activity and viral DNA replication detected in explanted ganglia are the result of productive infection of additional cells and not the result of primary reactivation events.
Several groups have studied HSV gene transcription and translation in explanted ganglia as a model of reactivation (4, 6, 10, 18, 24, 25, 33, 34). In some cases it was concluded that the transcriptional program during reactivation differed from that seen during lytic infection or that viral DNA replication was required for immediate-early gene expression. When these data are considered in the context of results presented here, caution should be employed in interpreting these earlier studies. It is likely that the transcriptional events that they were attempting to quantify (occurring after 24 to 48 h in culture) were largely the result of virus spread in the explanted ganglia and not primary reactivation events. It should be emphasized that most investigators monitor reactivation in explanted ganglia by methods requiring release of virus from the explanted tissue, an approach not designed to detect the low levels of virus produced in a very few reactivating neurons within the tissue. The fact that reactivation occurs within 22 h postexplant is not generally appreciated but has been reported previously. Using the more sensitive approach that we have utilized for detecting reactivation in ganglia in vivo, namely, directly grinding the ganglia and plating the homogenate onto indicator cells, Klein detected from 1 to 100 PFU in seven of eight TG latently infected with 17syn+ at 24 h postexplant (8). In addition, we have shown here that strain KOS also reactivates in this time frame, although with reduced frequency.
We conclude that explant reactivation may provide an ancillary system for selected studies of the early events in reactivation. However, clear signs of neuronal degeneration as early as 2 to 3 h postexplant indicate that there are dramatic additional changes in these ganglia that are not associated with the reactivation process. For example, we found that many neurons in ganglia explanted in vitro expressed cdk2 and cdk4, but this did not occur in ganglia induced to reactivate in vitro. On the basis of similar findings in cultivated ganglia in vitro, Schang et al. concluded that neuronal levels of cdk2 were among the factors that determine the outcome of HSV infections of neurons (26). Our results suggest that this conclusion may not be relevant to the in vivo setting, since cdk2 was not upregulated following hyperthermic stress. We did not detect cdk4 expression in TG neurons in vivo before or after hyperthermic stress, although it was upregulated following explant. In contrast, Schang et al. reported expression of cdk4 in the cytoplasm of normal neurons in vivo (26). The reason for this discrepancy is not known, but it might be attributed to the antibodies, fixation, and/or unmasking procedures employed. Regardless, we show here that the nuclear cdk4 expression that we detected in explanted ganglia is not a prerequisite or a precipitating event for reactivation in vivo.
It is also not yet clear in what ways the changes induced in neurons by axotomy and explantation, including neuronal apoptosis demonstrated by cytoplasmic cytochrome c and TUNEL staining, would influence the pathway(s) leading to viral reactivation. Our results suggest that these changes relax the barriers to reactivation and may obviate the need for certain virally encoded functions. While such a system could ultimately provide a fertile experimental field, understanding the basic details of the virus-host interactions resulting in reactivation in the least complex and most relevant setting, i.e., in vivo, appears to be the most direct and best approach to understanding this important question.
Acknowledgments
This work was supported by NIH R01 AI32121 and EY 13168.
REFERENCES
- 1.Berkelaar, M., D. B. Clarke, Y. C. Wang, G. M. Bray, and A. J. Aguayo. 1994. Axotomy results in delayed death and apoptosis of retinal ganglion cells in adult rats. J. Neurosci. 14:4368-4374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Burnet, F. M., and S. W. Williams. 1939. Herpes simplex: a point of view. Med. J. Aust. 1:637-642. [Google Scholar]
- 3.Carr, D. J., S. Noisakran, W. P. Halford, N. Lukacs, V. Asensio, and I. L. Campbell. 1998. Cytokine and chemokine production in HSV-1 latently infected trigeminal ganglion cell cultures: effects of hyperthermic stress. J. Neuroimmunol. 85:111-121. [DOI] [PubMed] [Google Scholar]
- 4.Devi-Rao, G. B., D. C. Bloom, J. G. Stevens, and E. K. Wagner. 1994. Herpes simplex virus type 1 DNA replication and gene expression during explant-induced reactivation of latently infected murine sensory ganglia. J. Virol. 68:1271-1282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Groves, M. J., T. Christopherson, B. Giometto, and F. Scaravilli. 1997. Axotomy-induced apoptosis in adult rat primary sensory neurons. J. Neurocytol. 26:615-624. [DOI] [PubMed] [Google Scholar]
- 6.Halford, W. P., B. M. Gebhardt, and D. J. Carr. 1996. Mechanisms of herpes simplex virus type 1 reactivation. J. Virol. 70:5051-5060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hill, J. M., J. B. Dudley, Y. Shimomura, and H. E. Kaufman. 1986. Quantitation and kinetics of induced HSV-1 ocular shedding. Curr. Eye Res. 5:241-246. [DOI] [PubMed] [Google Scholar]
- 8.Klein, R. J. 1982. Treatment of experimental latent herpes simplex virus infections with acyclovir and other antiviral compounds. Am. J. Med. 73:138-142. [DOI] [PubMed] [Google Scholar]
- 9.Knotts, F. B., M. L. Cook, and J. G. Stevens. 1973. Latent herpes simplex virus in the central nervous system of rabbits and mice. J. Exp. Med. 138:740-744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kosz-Vnenchak, M., J. Jacobson, D. M. Coen, and D. M. Knipe. 1993. Evidence for a novel regulatory pathway for herpes simplex virus gene expression in trigeminal ganglion neurons. J. Virol. 67:5383-5393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Leib, D. A., K. L. Ruffner, C. Hildebrand, P. A. Schaffer, G. E. Wright, and D. M. Coen. 1990. Specific inhibitors of herpes simplex virus thymidine kinase diminish reactivation of latent virus from explanted murine ganglia. Antimicrob. Agents Chemother. 34:1285-1286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Luque, J. M., W. B. Adams, and J. G. Nicholls. 1998. Procedures for whole-mount immunohistochemistry and in situ hybridization of immature mammalian CNS. Brain Res. Protoc. 2:165-173. [DOI] [PubMed] [Google Scholar]
- 13.Lygerou, Z., and P. Nurse. 2000. Cell cycle. License withheld—geminin blocks DNA replication. Science 290:2271-2273. [DOI] [PubMed] [Google Scholar]
- 14.McGarry, T. J., and M. W. Kirschner. 1998. Geminin, an inhibitor of DNA replication, is degraded during mitosis. Cell 93:1043-1053. [DOI] [PubMed] [Google Scholar]
- 15.McLennan, J. L., and G. Darby. 1980. Herpes simplex virus latency: the cellular location of virus in dorsal root ganglia and the fate of the infected cell following virus activation. J. Gen. Virol. 51:233-243. [DOI] [PubMed] [Google Scholar]
- 16.Pereira, R. A., D. C. Tscharke, and A. Simmons. 1994. Upregulation of class I major histocompatibility complex gene expression in primary sensory neurons, satellite cells, and Schwann cells of mice in response to acute but not latent herpes simplex virus infection in vivo. J. Exp. Med. 180:841-850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Plummer, G., C. R. Goodheart, M. Miyagi, G. R. Skinner, M. E. Thouless, and P. Wildy. 1974. Herpes simplex viruses: discrimination of types and correlation between different characteristics. Virology 60:206-216. [DOI] [PubMed] [Google Scholar]
- 18.Rezuchova, I., M. Kudelova, V. Durmanova, A. Vojvodova, J. Kosovsky, and J. Rajcani. 2003. Transcription at early stages of herpes simplex virus 1 infection and during reactivation. Intervirology 46:25-34. [DOI] [PubMed] [Google Scholar]
- 19.Sawtell, N. M. 1997. Comprehensive quantification of herpes simplex virus latency at the single-cell level. J. Virol. 71:5423-5431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sawtell, N. M. 1998. The probability of in vivo reactivation of herpes simplex virus type 1 increases with the number of latently infected neurons in the ganglia. J. Virol. 72:6888-6892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sawtell, N. M. 2003. Quantitative analysis of herpes simplex virus reactivation in vivo demonstrates that reactivation in the nervous system is not inhibited at early times postinoculation. J. Virol. 77:4127-4138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Sawtell, N. M., D. I. Bernstein, and L. R. Stanberry. 1999. A temporal analysis of acyclovir inhibition of induced herpes simplex virus type 1 in vivo reactivation in the mouse trigeminal ganglia. J. Infect. Dis. 180:821-823. [DOI] [PubMed] [Google Scholar]
- 23.Sawtell, N. M., D. K. Poon, C. S. Tansky, and R. L. Thompson. 1998. The latent herpes simplex virus type 1 genome copy number in individual neurons is virus strain specific and correlates with reactivation. J. Virol. 72:5343-5350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sawtell, N. M., and R. L. Thompson. 1992. Herpes simplex virus type 1 latency-associated transcription unit promotes anatomical site-dependent establishment and reactivation from latency. J. Virol. 66:2157-2169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sawtell, N. M., and R. L. Thompson. 1992. Rapid in vivo reactivation of herpes simplex virus in latently infected murine ganglionic neurons after transient hyperthermia. J. Virol. 66:2150-2156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Schang, L. M., A. Bantly, and P. A. Schaffer. 2002. Explant-induced reactivation of herpes simplex virus occurs in neurons expressing nuclear cdk2 and cdk4. J. Virol. 76:7724-7735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sears, A. E., and B. Roizman. 1990. Amplification by host cell factors of a sequence contained within the herpes simplex virus 1 genome. Proc. Natl. Acad. Sci. USA 87:9441-9444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Shimeld, C., D. L. Easty, and T. J. Hill. 1999. Reactivation of herpes simplex virus type 1 in the mouse trigeminal ganglion: an in vivo study of virus antigen and cytokines. J. Virol. 73:1767-1773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Shimeld, C., T. Hill, B. Blyth, and D. Easty. 1989. An improved model of recurrent herpetic eye disease in mice. Curr. Eye Res. 8:1193-1205. [DOI] [PubMed] [Google Scholar]
- 30.Shimeld, C., J. L. Whiteland, N. A. Williams, D. L. Easty, and T. J. Hill. 1996. Reactivation of herpes simplex virus type 1 in the mouse trigeminal ganglion: an in vivo study of virus antigen and immune cell infiltration. J. Gen. Virol. 77:2583-2590. [DOI] [PubMed] [Google Scholar]
- 31.Speck, P. G., and A. Simmons. 1992. Synchronous appearance of antigen-positive and latently infected neurons in spinal ganglia of mice infected with a virulent strain of herpes simplex virus. J. Gen. Virol. 73:1281-1285. [DOI] [PubMed] [Google Scholar]
- 32.Stevens, J. G., and M. L. Cook. 1971. Latent herpes simplex virus in spinal ganglia of mice. Science 173:843-845. [DOI] [PubMed] [Google Scholar]
- 33.Tal-Singer, R., T. M. Lasner, W. Podrzucki, A. Skokotas, J. J. Leary, S. L. Berger, and N. W. Fraser. 1997. Gene expression during reactivation of herpes simplex virus type 1 from latency in the peripheral nervous system is different from that during lytic infection of tissue cultures. J. Virol. 71:5268-5276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tanaka, S., H. Minagawa, Y. Toh, Y. Liu, and R. Mori. 1994. Analysis by RNA-PCR of latency and reactivation of herpes simplex virus in multiple neuronal tissues. J. Gen. Virol. 75:2691-2698. [DOI] [PubMed] [Google Scholar]
- 35.Tenser, R. B., K. A. Hay, and W. A. Edris. 1989. Latency-associated transcript but not reactivatable virus is present in sensory ganglion neurons after inoculation of thymidine kinase-negative mutants of herpes simplex virus type 1. J. Virol. 63:2861-2865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Thompson, R. L., M. L. Cook, G. B. Devi-Rao, E. K. Wagner, and J. G. Stevens. 1986. Functional and molecular analyses of the avirulent wild-type herpes simplex virus type 1 strain KOS. J. Virol. 58:203-211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Thompson, R. L., S. K. Rogers, and M. A. Zerhusen. 1989. Herpes simplex virus neurovirulence and productive infection of neural cells is associated with a function which maps between 0.82 and 0.832 map units on the HSV genome. Virology 172:435-450. [DOI] [PubMed] [Google Scholar]
- 38.Thompson, R. L., and N. M. Sawtell. 2001. Herpes simplex virus type 1 latency-associated transcript gene promotes neuronal survival. J. Virol. 75:6660-6675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Thompson, R. L., and N. M. Sawtell. 1997. The herpes simplex virus type 1 latency-associated transcript gene regulates the establishment of latency. J. Virol. 71:5432-5440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Thompson, R. L., and N. M. Sawtell. 2000. Replication of herpes simplex virus type 1 within trigeminal ganglia is required for high frequency but not high viral genome copy number latency. J. Virol. 74:965-974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Thompson, R. L., and J. G. Stevens. 1983. Biological characterization of a herpes simplex virus intertypic recombinant which is completely and specifically non-neurovirulent. Virology 131:171-179. [DOI] [PubMed] [Google Scholar]
- 42.Wagner, E. K., and D. C. Bloom. 1997. Experimental investigation of herpes simplex virus latency. Clin. Microbiol. Rev. 10:419-443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Whitley, R. J. 2001. Herpes simplex viruses, p. 2461-2510. In D. M. Knipe, P. M. Howley, D. E. Griffin, R. A. Lamb, M. A. Martin, B. Roizman, and S. E. Straus (ed.), Fields virology, vol. 2. Lippincott Williams & Wilkins, Philadelphia, Pa. [Google Scholar]
- 44.Wilcox, C. L., R. L. Smith, C. R. Freed, and E. M. Johnson, Jr. 1990. Nerve growth factor-dependence of herpes simplex virus latency in peripheral sympathetic and sensory neurons in vitro. J. Neurosci. 10:1268-1275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Wohlschlegel, J. A., B. T. Dwyer, S. K. Dhar, C. Cvetic, J. C. Walter, and A. Dutta. 2000. Inhibition of eukaryotic DNA replication by geminin binding to Cdt1. Science 290:2309-2312. [DOI] [PubMed] [Google Scholar]