Abstract
Rationale
Sustained activation of Gq signaling during pressure overload causes cardiac hypertrophy that ultimately progresses to dilated cardiomyopathy. The molecular events that drive hypertrophy decompensation are incompletely understood. Ca2+/calmodulin-dependent protein kinase II delta (CaMKIIδ) is activated downstream of Gq and overexpression of Gq and CaMKIIδ recapitulates hypertrophy decompensation.
Objective
To determine whether CaMKIIδ contributes to hypertrophy decompensation provoked by Gq.
Methods and Results
Compared to Gαq transgenic (Gq) mice, compound Gq/CaMKIIδ knockout (KO) (Gq/KO) mice developed a similar degree of cardiac hypertrophy but exhibited significantly improved left ventricular function, less cardiac fibrosis and cardiomyocyte apoptosis, and fewer ventricular arrhythmias. Markers of oxidative stress were elevated in mitochondria from Gq vs. WT mice and respiratory rates were lower; these changes in mitochondrial function were restored by CaMKIIδ deletion. Gq-mediated increases in mitochondrial oxidative stress, compromised membrane potential and cell death were recapitulated in NRVMs infected with constitutively active Gq and attenuated by CaMKII inhibition. Deep RNA sequencing revealed altered expression of 41 mitochondrial genes in Gq hearts, with normalization of ~40% of these genes by CaMKIIδ deletion. Uncoupling protein 3 (UCP3) was markedly downregulated in Gq or by Gq expression in NRVMs and reversed by CaMKIIδ deletion or inhibition, as was Peroxisome proliferator-activated receptor alpha (PPAR-α). The protective effects of CaMKIIδ inhibition on ROS generation and cell death were abrogated by knock down of UCP3. Conversely, restoration of UCP3 expression attenuated ROS generation and cell death induced by CaMKIIδ. Our in vivo studies further demonstrated that pressure overload induced decreases in PPAR-α and UCP3, increases in mitochondrial protein oxidation, and hypertrophy decompensation which were attenuated by CaMKIIδ deletion.
Conclusions
Mitochondrial gene reprogramming induced by CaMKIIδ emerges as an important mechanism contributing to mitotoxicity in decompensating hypertrophy.
Keywords: Gq, heart failure, oxidative stress, UCP, calcium/calmodulin-dependent protein kinase II, peroxisome proliferator-activated receptor alpha and gamma
INTRODUCTION
Myocardial hypertrophy is the evolutionarily conserved cardiac reaction to hemodynamic overload or injury. This genetically programmed response improves ventricular ejection performance by restoring a more normal ratio of left ventricular wall thickness to intracavitary pressure, two determinants of wall stress.1 While initially compensatory, reactive hypertrophy inevitably “decompensates” and fails. Thus, cardiac hypertrophy is an independent risk factor for both heart failure and death.2, 3
The mechanistic underpinnings of hypertrophy decompensation are poorly understood. The genetic growth program in adult hearts recapitulates growth of the embryonic heart, and includes a reversion to fetal muscle isoforms, alterations in calcium handling proteins, and a lowered threshold for programmed cell death that may ultimately prove detrimental to the adult heart.4 Because it would be therapeutically beneficial to retain the physical features of cardiac hypertrophy that lower wall stress if they could be dissociated from deleterious collateral effects, the molecular dissection of hypertrophy signaling pathways has long been a goal. A central pathway transducing reactive hypertrophy signaling has been linked to the heterotrimeric G protein Gq, which is stimulated by pro-hypertrophic hormones such as angiotensin II, endothelin, and alpha-adrenergic agonists. Accordingly, mice with cardiomyocyte-specific overexpression of the catalytically active α subunit of Gq develop marked cardiac hypertrophy that recapitulates hypertrophy development and subsequent decompensation.5 Conversely, Gαq/Gα11 knockout mice are unable to develop hypertrophy in response to pressure overload.6
Gq signaling has been linked to increases in intracellular calcium, suggesting the calcium/calmodulin dependent kinase II (CaMKII) as a potential downstream signaling effector of Gq-mediated hypertrophy and heart failure.7 Indeed, myocardial overexpression of the predominant cardiac CaMKII isoform (CaMKIIδC), like overexpression of Gαq, is sufficient to induce hypertrophy that transitions to severe cardiac dysfunction.8 Conversely, inhibition or genetic ablation of CaMKIIδ prevents cardiac dysfunction after transverse aortic constriction (TAC), myocardial infarction, or ischemia/reperfusion.9–12 The ability of CaMKII to phosphorylate key calcium handling proteins was initially considered to be the major driver underlying its involvement in cardiac disease but normalization of CaMKII-induced changes in sarcoplasmic reticulum calcium proved insufficient to prevent heart failure induced by CaMKIIδ overexpression.13
Gq induced cardiac decompensation has been associated with mitochondrial dysfunction and increased mitochondrial ROS generation.14–16 Adverse effects of CaMKII have also been shown to be associated with altered mitochondrial calcium handling and oxidative phosphorylation.13, 17, 18 These observations raise the possibility that CaMKIIδ might serve as the downstream mediator of the maladaptive mitochondrial effects of Gq signaling. Were this the case, combined manipulation of Gq and CaMKIIδ would shed light on the specific molecular mechanisms by which Gq stimulates and CaMKIIδ mediates hypertrophy decompensation. We demonstrate here that CaMKIIδ deletion attenuates the maladaptive cardiac effects of Gq, including mitochondrial dysfunction, by modulating the expression of nuclear-encoded mitochondrial genes. We suggest that CaMKIIδ-induced repression of UCP3 is one of the drivers of mitochondrial dysfunction and decompensation to heart failure.
METHODS
A more detailed description of the methods has been included as an online supplement.
Animal models
The generation of Gαq-40 transgenic mice and CaMKIIδ knockout mice has been described previously.5, 9 To study the role of CaMKIIδ in Gαq-induced heart failure, Gαq transgenic mice were crossed into a CaMKIIδ knockout background, to yield Wildtype (WT), CaMKIIδ knockout (KO), Gαq transgenic (Gq) and Gq mice in a CaMKIIδ knockout background (Gq/KO). Studies involving pressure overload used the recently generated cardiac specific CaMKIIδ knockout mice.12
RNA sequencing
Left ventricular RNA was isolated from 4 mice from each group and processed for RNA sequencing studies as previously described.19
Mitochondrial bioenergetics
Mouse heart mitochondria were isolated by differential centrifugation and respiration was measured using a Seahorse XF24 analyzer as previously described.20
Biochemical and histological analysis
Tissue fractionation, RNA isolation and Western blot analysis were performed as described previously.9, 21 Cardiac mitochondrial protein carbonyl was measured using OxiSelect protein carbonyl ELISA kit (Cell Biolabs, San Diego, CA).
Neonatal Ventricular Cardiomyocyte Culture and live cell imaging
Neonatal rat ventricular myocytes (NRVMs) were isolated, cultured, infected and transfected as described previously.21
Statistical analysis
All data are presented as means ± standard error of the mean (SE). Comparisons between groups were performed using the Student t test, the Mann Whitney U test, Kruskal-Wallis test or One-way ANOVA, followed by the Tukey post hoc test, where appropriate. A p value < 0.05 was considered statistically significant.
RESULTS
CaMKIIδ deletion does not affect Gαq-induced cardiac hypertrophy
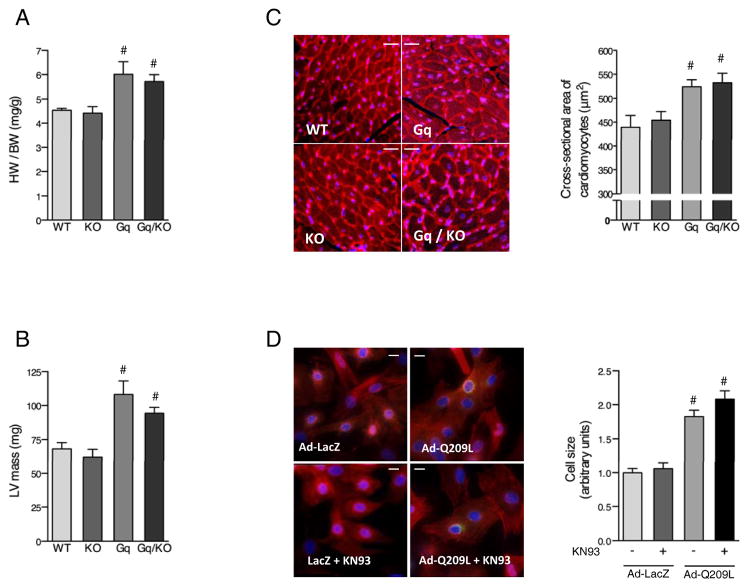
Cardiac hypertrophy and depressed contractile performance induced by Gαq overexpression have been thoroughly described.5 Consistent with previous reports, gravimetric, echocardiographic and histologic analysis revealed significant increases in left ventricular mass and histological cardiomyocyte cross-sectional area in Gq mice compared to WT mice (Fig 1 A–C). We also observed activation of CaMKIIδ in the Gq compared to WT mouse heart, as indicated by increased CaMKII auto-phosphorylation and oxidation as well as by increased phosphorylation of phospholamban at threonine 17, a CaMKII specific site (Supplemental Fig I). Genetic ablation of CaMKIIδ did not diminish Gq-induced cardiac hypertrophy as assessed by comparison of Gq and Gq/KO mice on multiple readouts (Fig 1 A–C). Likewise hypertrophy of NRVMs induced by adenoviral expression of constitutively active Gαq (Ad-Q209L) and demonstrated by increased cardiomyocyte size and ANF immunostaining (Fig 1 D) was unaffected by pharmacological blockade of CaMKII with KN93. Thus the ability of Gq to elicit genetic and morphological changes characteristic of cardiomyocyte hypertrophy does not depend on CaMKII activation.
Figure 1.
CaMKIIδ is not required for Gq induced cardiac hypertrophy. A and B, Gravimetric and echocardiographic indices of left ventricular hypertrophy. Shown are Heart Weight/Body Weight ratio (HW/BW, A, N=6–9) and left ventricular mass (B, N=7 for WT, N=5 for KO, N=9 for Gq and N=8 for Gq/KO). C, Left ventricular sections stained with Rhodamine-labeled wheat germ agglutinin (red) and 4′,6-diamidino-2-phenylindole (DAPI, blue). Scale bar is 20 μM. Bar graph depicts the cross-sectional area of cardiomyocytes expressed in μm2. D, Neonatal rat ventricular myocytes stained with phalloidin (red) atrial natriuretic factor (green) or DAPI (blue) after infection with an adenovirus expressing constitutively activated Gαq (Ad-Q209L) or β-galactosidase (Ad-LacZ) and cultured in the presence or absence of the CaMKII inhibitor KN93. Scale bar is 10 μM. Bar graphs represent average cardiomyocyte circumference 24 hours after infection. All values are expressed as mean ± SEM. #, p < 0.05 versus WT or Ad-LacZ. WT, wildtype; KO, CaMKIIδ-knockout, Gq, Gq-transgenic, Gq/KO, Gq-transgenic in a CaMKIIδ-knockout background.
CaMKIIδ deletion prevents functional decompensation in Gαq-transgenic mice
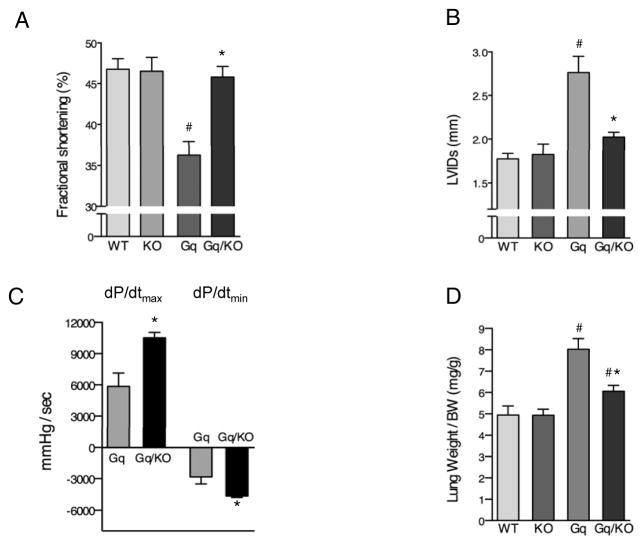
Compared to wild-type littermates, 8 week-old Gq mice had reduced fractional shortening (Fig 2 A), LV systolic dilatation (Fig 2 B), decreased load-independent ventricular contractility and relaxation indices (Fig 2 C) and increased left ventricular filling pressures (Supplemental Fig II A). Another determinant of functional decompensation, lung weight/body weight ratios, was also significantly increased in Gq transgenic mice (Fig 2 D). All of these changes were ameliorated by deletion of CaMKIIδ (Fig 2 A–D, Supplemental Fig II A). Other characteristic heart failure-associated phenotypes of Gq mice including cardiomyocyte apoptosis, fibrosis and ventricular arrhythmias, were likewise improved by CaMKIIδ ablation (Supplemental Fig II B–E). Thus while structural Gq-stimulated cardiac hypertrophy was maintained in the face of CaMKIIδ deficiency (Fig 1), abrogation of CaMKIIδ prevented the associated cardiac decompensation (Fig 2).
Figure 2.
CaMKIIδ knockout prevents Gq-induced cardiac dysfunction. A, B, Echocardiographic indices of left ventricular (LV) function and LV dilatation. Shown are fractional shortening (A), LV Internal Systolic Diameter (LVIDs, B); (N=7 for WT, N=5 for KO, N=9 for Gq and N=8 for Gq/KO). C, Indices of cardiac contractility and relaxation. Shown are maximal rates of LV-pressure increase (dP/dtmax) and decrease (dP/dtmin), N= 4 per group. D, Lung weight/Body Weight ratio, an indicator of the severity of functional decompensation (N= 6–10 per group). All values are expressed as mean ± SEM. #, p < 0.05 versus WT, *; p < 0.05 versus Gq. WT, wildtype; KO, CaMKIIδ-knockout; Gq, Gq-transgenic; Gq/KO, Gq-transgenic in a CaMKIIδ-knockout background.
CaMKIIδ deletion attenuates mitochondrial dysfunction and mitochondrial oxidative stress
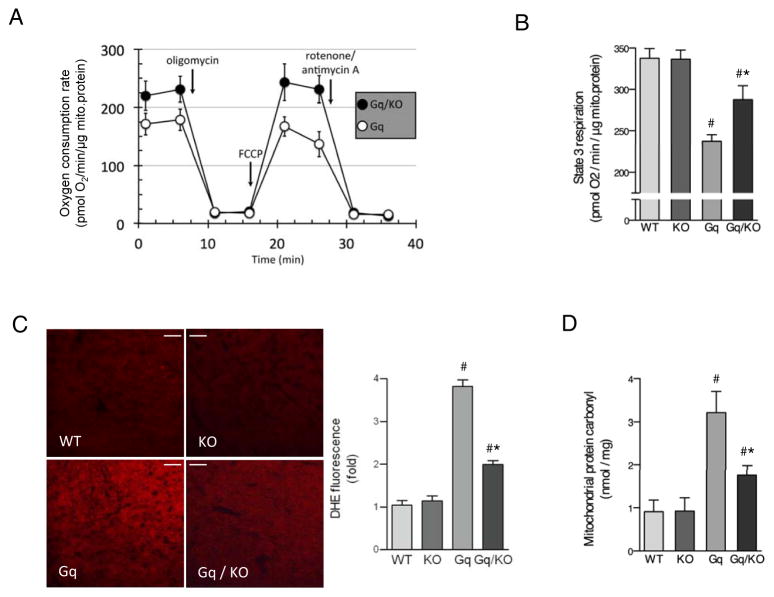
The cardiomyopathy of pressure overload is mediated in part through disruption of the electron transport chain and increased mitochondrial oxidative stress.4 To determine whether Gq signaling affects mitochondrial function in a CaMKII-dependent manner, we isolated mitochondria from the four mouse lines and measured their oxygen consumption rate drive (Fig 3 A). Pyruvate-driven respiration was 40% lower in ventricular mitochondria isolated from Gq than in those isolated from WT mice (Fig 3 B). This impairment was significantly reversed in mitochondria isolated from the Gq/KO mice (Fig 3 B). A similar improvement in mitochondrial respiration in Gq/KO mice was observed with the other complex I substrates palmitoylcarnitine and glutamate, whereas respiration on the complex II substrate succinate was not different in Gq/KO versus Gq mice (data not shown). State 4 respiration (oligomycin) and respiratory control ratios were comparable between all four mouse lines. Together these data indicate that the effect of CaMKIIδ on complex I contribute to impaired mitochondrial respiration induced downstream of Gq.
Figure 3.
CaMKIIδ knockout prevents mitochondrial dysfunction and mitochondrial ROS production in Gq-transgenic mice. A, Representative experiment to depict the approach for measurement of rates of pyruvate/malate-driven oxygen consumption by isolated cardiac mitochondria from GqTg and Gq/KO animals. ADP is present for the first two measurements, followed by measurement of state 4 and then uncoupler-stimulated respiration indicating diminished maximal rates of state 3 and uncoupler-stimulated respiration in the GqTg mitochondria. B, Bar graph depicting differences in maximal state 3 respiration on pyruvate/malate in all experimental groups (N=4 for WT and KO and N=6 for Gq and Gq/KO). C, Cryopreserved myocardial sections were stained with dihydroethidium (DHE), an indicator of total myocardial reactive oxygen species. Scale bar is 60 μM. Bar graph depicts average DHE fluorescence in the different experimental groups (N=5–7 per group). D, Carbonylation of mitochondrial proteins in the different experimental groups as measured by ELISA of cardiac mitochondrial homogenates (N=6–8 per group). All values are expressed as mean ± SEM. #, p < 0.05 versus WT, *; p < 0.05 versus Gq. WT, wildtype, KO, CaMKIIδ-knockout, Gq, Gq-transgenic, Gq/KO, Gq-transgenic in a CaMKIIδ-knockout background.
To more directly link Gq and CaMKII signaling to mitochondrial function we examined changes in mitochondrial oxidative stress. Reactive oxygen species (ROS) were assessed by dihydroethidium (DHE) fluorescence staining in ventricular sections from hearts of all four genotypes. DHE fluorescence was markedly increased in Gq mice and was reduced approximately 50% by concomitant CaMKIIδ deletion (Fig 3 C). To confirm data obtained using DHE fluorescence changes, we evaluated oxidative damage to mitochondrial proteins by quantifying protein carbonyls in isolated mitochondria. In line with previous observations,15 mitochondrial protein carbonyls were increased 3-fold in the Gq mouse heart (Fig 3 D). Remarkably this increase was significantly attenuated in Gq/KO mice (Fig 3 D).
CaMKIIδ activation induces mitochondrial oxidative stress in cardiomyocytes
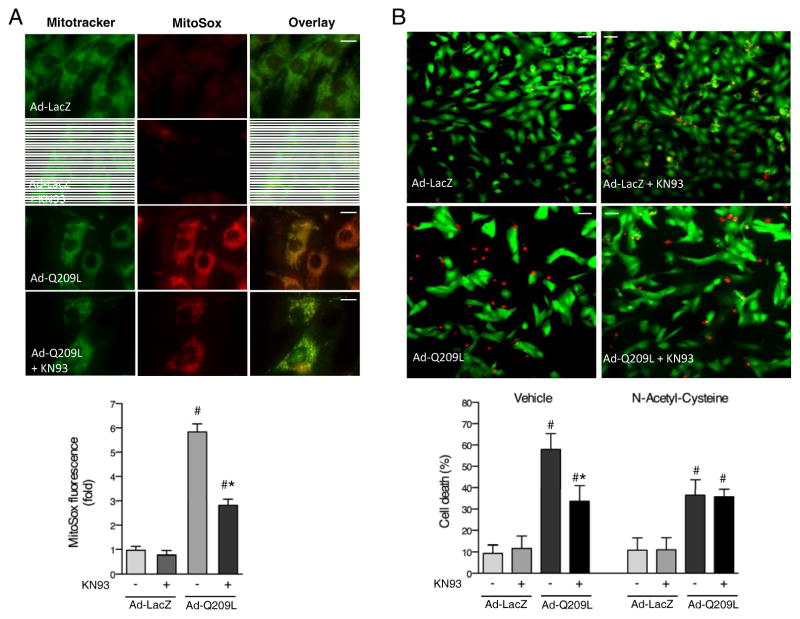
To corroborate and add mechanistic insight to these in vivo findings, neonatal rat ventricular myocytes were infected with an adenovirus expressing Ad-Q209L, with or without the addition of the CaMKII inhibitor KN93 or of an adenovirus expressing catalytically dead CaMKIIδ (CD-CaMKIIδ). Ad-Q209L infection resulted in a 6-fold increase in mitochondrial ROS accumulation assessed by MitoSox fluorescence. The Gq mediated increase in MitoSox fluorescence was reduced by more than 50% in cells treated with KN 93 (Fig 4 A) or co-infected with CD-CaMKIIδ (Supplemental Fig III A). Conversely expression of a constitutively activated CaMKIIδc adenovirus (Ad-CaMKIIδ) significantly increased the MitoSox signal in NRVMs (Supplemental Fig III B).
Figure 4.
CaMKIIδ induces mitochondrial ROS generation and cell death in cardiomyocytes downstream of Gq. A, Live cell imaging of neonatal rat ventricular myocytes (NRVMs) stained with the non-toxic mitochondrial marker, mitotracker-green (green) and the mitochondrial-specific reactive oxygen species indicator MitoSox (red), 12 hours after infection with an adenovirus expressing constitutively activated Gαq (Ad-Q209L) or β-galactosidase (Ad-LacZ) and cultured in the presence or absence of the CaMKII inhibitor KN93. Scale bar is 10 μM. Bar graph depicts average MitoSox intensities in mitochondria from the experimental groups. B, Typical example of NRVMs stained with the live cell indicator calcein (green) and the dead cell indicator propidium iodide (red) after infection with Ad-Q209L or Ad-LacZ and cultured in the presence or absence of KN93. Scale bar is 40 μM. Cells were considered viable when positive for calcein and negative for propidium idodie. Bar graph depicts cell death expressed as the % total cells in the different experimental groups, with or without the addition of the ROS scavenger N-acetyl-cysteine or vehicle. #, p < 0.05 versus Ad-LacZ, * p < 0.05 versus Ad-Q209L.
CaMKIIδ and ROS generation mediate Gαq-induced mitochondria-mediated cell death
Expression of Q209L also led to marked increases in mitochondrial depolarization (assessed using tetramethylrhodamine ethyl ester) and apoptosis (measured by DNA fragmentation), which were prevented by inhibition of CaMKIIδ with KN93 or expression of CD-CaMKIIδ (Supplemental Figs III C, D and Supplemental Fig IV A). We used N-acetyl-cysteine (NAC) as a ROS-scavenger to test the role of ROS in Ad-Q209L-induced cell death. Treatment with NAC decreased Q209L-induced cell death by approximately 60% and did not further reduce cell death in the presence of KN93 (Fig 4 B). The observation that NAC treatment provided no further protection above that afforded by KN93 is consistent with there being a common pathway for the mitochondrial effects of CaMKII and ROS.
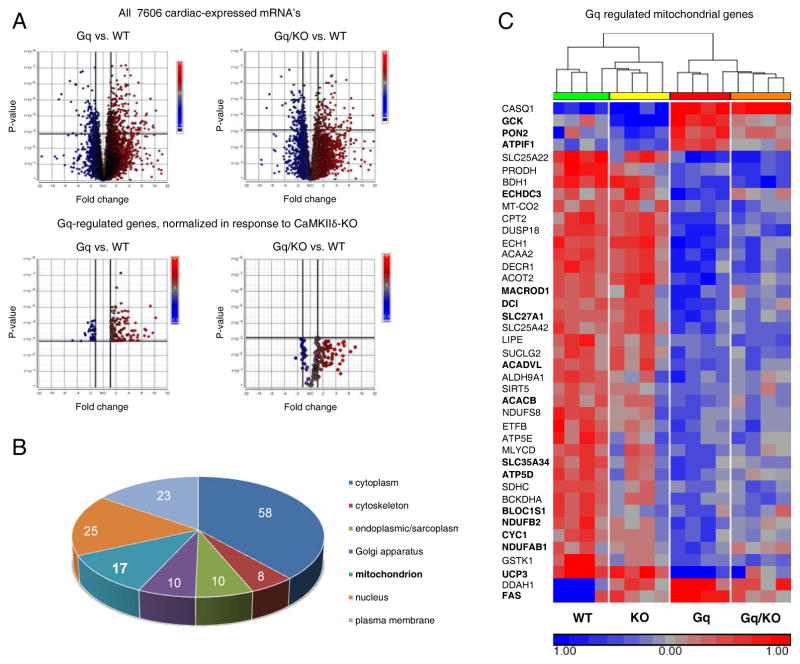
CaMKIIδ deletion prevents mitochondrial gene reprogramming in Gαq-induced heart failure
A plethora of transcriptional responses accompany Gq expression and development of decompensation in the mouse heart, including changes in genes that affect mitochondrial function.19 We applied deep RNA sequencing to discover genes that were differentially regulated in the hearts of Gq mice compared to Gq/KO mice. Of the three hundred and thirty five genes that differed in the hearts of 4 week old Gq versus WT mice, one third (118 genes) were normalized by CaMKIIδ deletion (Fig 5 A). Gene ontology analysis further revealed that 17 genes encoding mitochondrial proteins (41% of the mitochondrial genes that were differentially regulated in Gq vs. WT mice) were normalized in Gq/KO mice (Fig 5 B). The 17 mitochondrial genes normalized by CaMKIIδ deletion (bolded in Fig 5 C) mediate processes such as mitochondrial substrate transport and oxidation, electron transport chain function, and ATP synthesis. Many of these genes, including cytochrome c (CYC1) and NDU subunits (NDUFAB1 and NDUF58), are components of the electron transport chain and changes in these genes may at least partially explain the alterations in mitochondrial respiration demonstrated in Fig 3.
Figure 5.
CaMKIIδ is involved in mitochondrial reprograming downstream of Gq. A, RNA-sequencing of Gq and Gq/KO hearts vs WT hearts. Regulated mRNAs at a fold-change of +/− 50%, false discovery rate (FDR) of 0.02, are shown in the upper panels (thick vertical lines denote fold-change cutoff; thick horizontal lines denote p-value cutoff for FDR 0.02). Lower panels show only mRNAs that are regulated in Gq hearts, and normalized in CaMKIIδ KO hearts. B, Gene Ontology cellular component category analysis of Gq-regulated, CaMKIIδ KO-normalized genes. C, Unbiased hierarchical clustering of Gq regulated mitochondrial genes demonstrates nearly perfect clustering between the groups. Those mRNAs that are normalized by CaMKIIδ KO are shown in Bold. Blue = low expression, red = high expression.
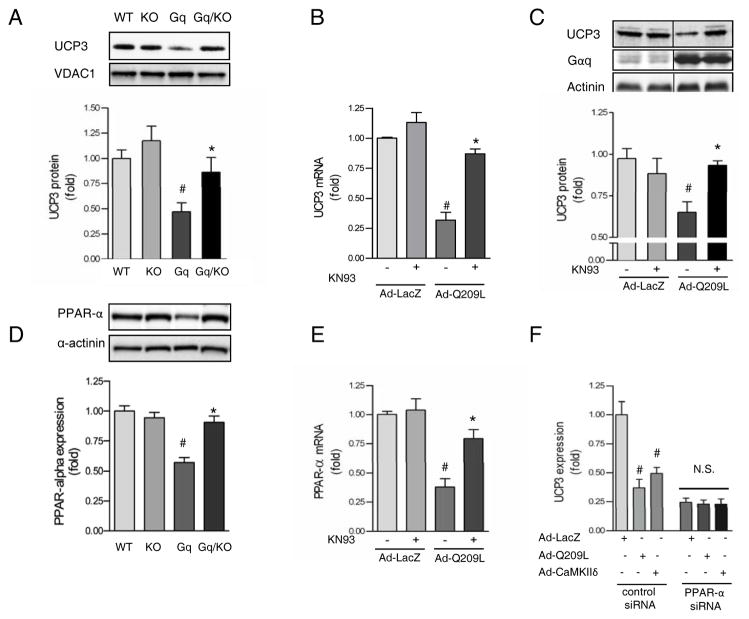
Gq and CaMKIIδ regulate UCP3 and PPAR-α
One of the mitochondrial gene transcripts found to be markedly down regulated in Gq mice and restored in Gq/KO mice (Fig 5 C) was UCP3. The decrease in UCP3 mRNA levels observed through RNA sequencing was confirmed by changes in UCP3 protein expression in mitochondria isolated from Gq and Gq/KO mice (Fig 6 A). To further explore the mechanism and significance of Gq signaling to UCP3 we examined the regulation of UCP3 in NRVMs infected with Ad-Q209L. UCP3 mRNA and protein expression were significantly decreased in cells expressing Q209L and this was prevented by KN93 (Fig 6 B, C) confirming a role for CaMKII in the regulation of UCP3 expression.
Figure 6.
CaMKIIδ down regulates Peroxisome Proliferator-Activated Receptor alpha (PPAR-α) and Uncoupling Protein 3 (UCP3) expression. A, UCP3 protein expression normalized for Voltage-Dependent Anion-selective Channel protein 1 (VDAC1) in lysates of mitochondria from mouse ventricle. B, UCP3 mRNA expression normalized for the internal control 36B4 in neonatal rat ventricular myocytes (NRVMs) infected with constitutively activated Gαq (Ad-Q209L) or β-galactosidase (Ad-LacZ) and cultured in the presence or absence of the CaMKII inhibitor KN93. C, UCP3 protein expression normalized for actinin in whole cell lysates from NRVMs treated as in panel B. The lanes were run on the same gel but were noncontiguous. D, PPAR-α protein expression in whole cell lysates from mouse ventricle. E, PPAR-α mRNA expression normalized for 36B4 from NRVMs treated as in panel B. F, UCP3 mRNA expression normalized for 36B4 in NRVMs which were first transfected with control siRNA or PPAR-α siRNA and subsequently infected with Ad-LacZ, Ad-Q209L, or constitutively activated CaMKIIδc (CaMKIIδ). All values are expressed as mean ± SEM. #, p < 0.05 versus WT or Ad-LacZ, *, p < 0.05 versus Gq or Q209L. WT, wildtype; KO, CaMKIIδ-knockout; Gq, Gq-transgenic; Gq/KO, Gq-transgenic in a CaMKIIδ-knockout background.
Peroxisome proliferator-activated receptor alpha (PPAR-α) has been suggested to regulate the expression of UCP3.22–24 We analyzed the 17 mitochondrial genes normalized by CaMKIIδ deletion for transcription factor binding site enrichment and conservation in their 5kb proximal promoters as previously described.25 This analysis showed significant enrichment (p < 0.0001) for peroxisome proliferator-activated receptor alpha (PPAR-α) binding sites. Moreover, analysis of the RNA sequencing data described above demonstrated that PPAR-α mRNA levels were reduced by one third in Gq mice compared to WT mice (p=0.00032) but not in Gq/KO vs WT mice. Western blot analysis revealed concomitant changes in PPAR-α protein expression; PPAR-α was decreased in the Gq transgenic and this was normalized by CaMKIIδ deletion (Fig 6 D). Expression of Q209L in NRVMs also effectively decreased PPAR-α mRNA through CaMKII signaling (Fig 6 E), consistent with a direct effect of Gq activation on PPAR-α expression. Furthermore the expression of PPAR-α dependent genes such as carnitine palmitoyltransferase 1B (CPT-1b) and pyruvate dehydrogenase kinase 4 (PDK4) was diminished through a KN93 sensitive pathway in cells expressing Q209L and also by expression of CaMKIIδc (Supplemental Fig V).
To demonstrate a functional relationship between PPAR-α and UCP3 expression we down regulated PPAR-α with siRNA. Greater than 85 % downregulation of PPAR-α protein expression was achieved by siRNA treatment (data not shown). Knockdown of PPAR-α significantly decreased basal levels of UCP3 expression (Fig 6 F), directly demonstrating that UCP3 expression is regulated by PPAR-α. Additionally UCP3 levels were not further diminished by Ad-Q209L or Ad-CaMKIIδ when PPAR-α was downregulated (Fig 6 F). Together these findings suggest that CaMKII down regulates UCP3 expression through inhibition of PPAR-α and that this occurs downstream of Gq signaling.
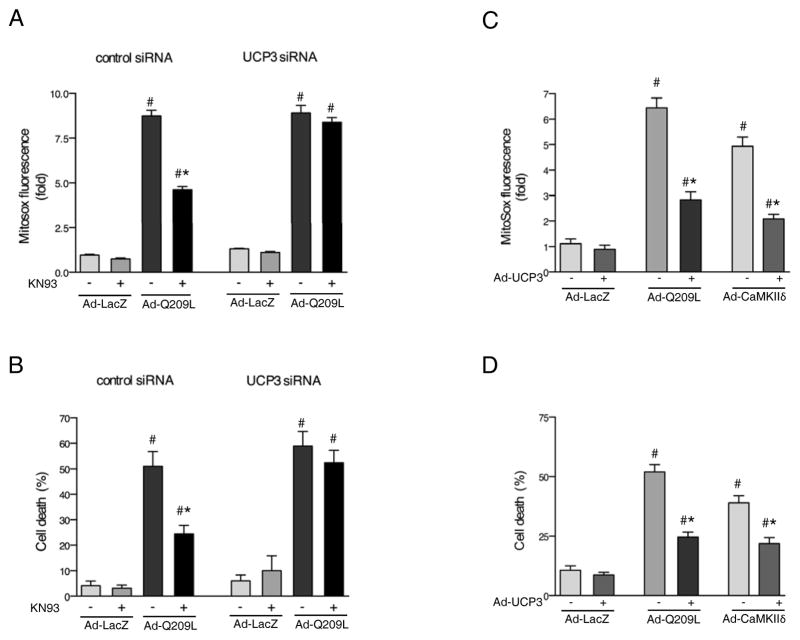
UCP3 contributes to ROS production and cell death
We carried out loss and gain of function experiments using UCP3 siRNA and adenovirus to demonstrate a causal relationship between decreased UCP3 expression and Gαq/CaMKII mediated mitochondrial ROS generation and cell death. Treatment with UCP3 siRNA reduced UCP3 expression in NRVMs by 80% (Supplemental Fig VII A). The ability of KN93 to inhibit Q209L-induced mitochondrial ROS accumulation (Fig VII A) and cell death (Fig 7 B) were both abrogated when UCP3 was depleted. The protective effect of CD-CaMKIIδ on cell death induced by Q209L was also blocked (Supplemental Fig IV B). To further establish the importance of UCP3 downregulation in Gαq- and CaMKII-mediated mitochondrial ROS generation and cell death, we generated an adenovirus expressing UCP3 (Ad-UCP3). Infection with Ad-UCP3 resulted in a 2.5-fold increase in expression of UCP3 and also normalized UCP3 expression in cells co-infected with Ad-Q209L (Supplemental Fig VII B). Importantly the ability of Ad-Q209L and Ad-CaMKIIδ to induce mitochondrial ROS accumulation (Fig 7 C) and cell death (Fig 7 D) were significantly attenuated by co-expression of UCP3.
Figure 7.
CaMKIIδ induced mitochondrial ROS and cell death are dependent on uncoupling protein 3 (UCP3). A, Neonatal Rat Ventricular Myocytes (NRVMs) were first transfected with scrambled siRNA or UCP3 siRNA and subsequently infected with constitutively activated Gαq (Ad-Q209L) or β-galactosidase (Ad-LacZ) and cultured in the presence or absence of the CaMKII inhibitor KN93. A, Average MitoSox intensities in mitochondria from the experimental groups, 8 hours after infection. B, Cell death determined by calcein/propidium iodide staining, 72 hours after infection as in A. C, MitoSox intensities in NRVMs 12 hours after infection with Ad-LacZ, Ad-Q209L or constitutively activated CaMKIIδc (CaMKIIδ) and co-infected with and adenovirus expressing UCP3. D, Cell death determined by calcein/propidium iodide staining, 72 hours after infection as in C. #, p < 0.05 versus or Ad-LacZ, * p < 0.05 versus Ad-Q209L or Ad-CaMKIIδ.
In the experiments above, an adenovirus expressing CaMKIIδc was used. To assess whether the mitochondrial reprograming was CaMKIIδ subtype specific, we compared the response to adenoviral expression of the two major cardiac CaMKIIδ isoforms, CaMKIIδc and CaMKIIδb. CaMKIIδ-mediated downregulation of UCP3, PPAR-α and other PPAR-α dependent genes was observed after infection with constitutively activated CaMKIIδc but these genes remained unchanged after infection with constitutively activated CaMKIIδb (Supplemental Fig V E, F and Supplemental Fig VI).
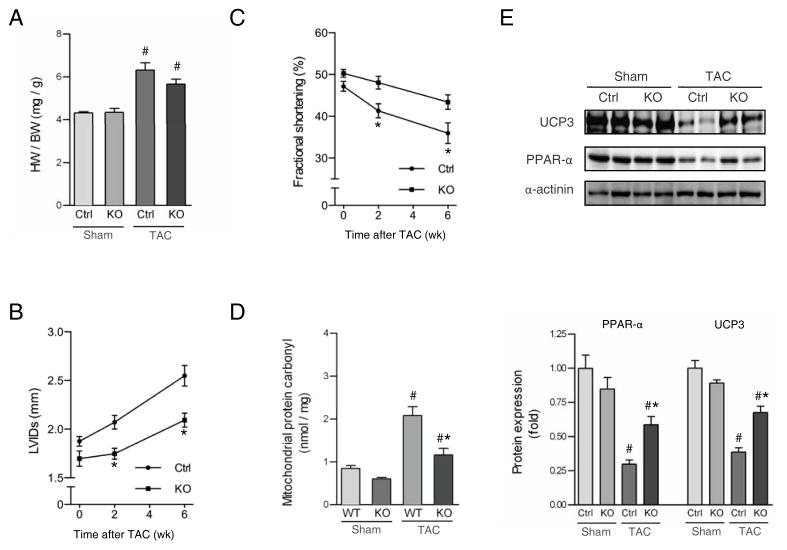
CaMKIIδ deletion prevents mitochondrial stress in pressure overload induced heart failure
We previously demonstrated that CaMKIIδ is required for the transition from hypertrophy to heart failure following TAC-induced pressure overload.9 To prove that the CaMKII signaling pathway responsible for decompensation to failure was cardiomyocyte autonomous, we generated cardiac specific CaMKIIδ KO mice.12 We examined their response to pressure overload induced by TAC and determined that deletion of CaMKIIδ did not prevent TAC-induced increases in hypertrophy (Fig 8 A). Despite development of hypertrophy the loss of CaMKIIδ in cardiomyocytes significantly attenuated the development of ventricular dilation and the decrease in fractional shortening (Fig 8 B, C). In addition, mitochondrial oxidative stress assessed by protein carbonylation was increased after TAC through a CaMKIIδ dependent process (Fig 8 D). Remarkably, as observed for the Gq mice, pressure overload resulted in decreases in PPAR-α and UCP3 expression and these changes were attenuated in the absence of CaMKIIδ (Fig 8 E). Thus the effect of CaMKIIδ on mitochondrial oxidative stress and heart failure development extends beyond the Gq model of cardiomyopathy to also include the response to pressure overload.
Figure 8.
CaMKIIδ deletion prevents mitochondrial oxidative stress in pressure overload induced heart failure. Cardiomyocyte-specific CaMKIIδ knockout mice (KO) or their littermate controls (Ctrl) were subjected to transverse aortic constriction (TAC). A, Heart Weight/Body Weight ratio (HW/BW, N=6–9) 2 weeks after TAC. B, C, Changes in echocardiographic indices of left ventricular (LV) function and LV dilatation after TAC. Shown are Left Ventricular Internal Systolic Diameter (LVIDs, B) and fractional shortening (C), (N=8). D, Carbonylation of mitochondrial proteins in wild type (WT) and global KO mice as measured by ELISA of cardiac mitochondrial homogenates 6 weeks after TAC (N=5–8 per group). E, PPAR-α and UCP3 protein expression normalized for α-actinin measured in lysates from mouse ventricle 6 weeks after TAC. All values are expressed as mean ± SEM. #, p < 0.05 versus Ctrl-sham or WT-sham, *; p < 0.05 versus Ctrl-TAC or WT-TAC.
DISCUSSION
Clinical therapeutics have aimed at preventing or reversing hypertrophy in patients with heart disease since conventional wisdom holds that reactive hypertrophy is maladaptive and perhaps dispensable for functional compensation to hemodynamic overload.26 Hypertrophic growth is, however, an evolutionarily conserved cardiac response to reduce ventricular wall stress in pressure overloaded or dilated ventricles.1 The problem is that this reactive hypertrophy ultimately progresses into to a maladaptive cardiomyopathy with decreased survival. It would therefore seem most therapeutically beneficial to retain the physically advantageous features of cardiac hypertrophy that reduce wall stress, while preventing its subsequent decompensation. Our previous work suggests that CaMKIIδ, while dispensable for hypertrophic growth, plays a key role in the transition from hypertrophy to heart failure.9 How this occurs, and whether CaMKIIδ activation is an essential component of pathological hypertrophy downstream of Gq signaling, is not known. Here, we used combined genetic manipulation of Gq and CaMKIIδ to dissociate growth-promoting and heart failure-inducing hypertrophy signals, in effect beneficially remodeling reactive hypertrophy. In so doing, we uncovered mitochondrial reprogramming as a key mediator of CaMKIIδ induced hypertrophy decompensation, further emphasizing the potential for CaMKII as a therapeutic target.
Mitochondrial reprogramming through CaMKIIδ as a central driver in hypertrophy decompensation
Mitochondrial reprogramming occurs during heart failure development and is characterized by a shift from fatty acid to glucose utilization, reduced ATP availability and increased mitochondrial oxidative stress.27 The relevance of mitochondrial reprogramming is underscored by the ability of mitochondrial-targeted antioxidants to prevent pathological hypertrophy and cardiac aging.15, 28–30. CaMKII is now known to be activated not only by calcium but also by oxidation.31 While it is clear that ROS generation and concomitant CaMKII oxidation contribute to the pathophysiology of heart disease,31–34 the possibility that this maladaptive cascade is further accentuated by reciprocal effects of CaMKII on ROS production has not been previously considered. In the present study we provide evidence that CaMKIIδ induces altered mitochondrial gene expression associated with mitochondrial dysfunction, mitochondrial oxidative stress and ROS-driven cell death. First, we show that CaMKIIδ mediates mitochondrial reprogramming downstream of Gq signaling. Second, we demonstrate that CaMKIIδ-induced mitochondrial reprogramming is associated with impaired respiration and increased mitochondrial oxidative stress. Third, we demonstrate that increased oxidative stress induced by CaMKIIδ contributes to cardiomyocyte cell death and cardiomyopathy. Globally, these findings implicate CaMKIIδ as a central mediator of the mitochondrial reprogramming and cardiomyocyte loss associated with heart failure development.
A critical role for UCP3 in CaMKIIδ-induced mitochondrial ROS in the heart
When activated,23, 35 uncoupling proteins can induce a proton leak by dissipating the proton motive force across the inner mitochondrial membrane, thereby reducing ROS generation at the cost of a reduction in coupling efficiency.36 UCP3 is a major UCP isoform in the heart and it appears to be critical for the cardiac response to multiple stresses.36–42 Downregulation of UCP3 has been associated with increased cardiac ROS levels in a model of doxorubicin-induced heart failure.37 The anti-oxidant effects of hexokinase-II and stanniocalcin-1 in cardiomyocytes have been suggested to involve UCP3.38, 39 Studies using UCP3 knockout mice demonstrate increased angiotensin-induced cardiac ROS levels and greater susceptibility to oxidative stress.39, 42 Furthermore UCP3 knockout mice show impaired myocardial energetics and larger infarcts in response to cardiac ischemia/reperfusion.41 These findings are consistent with and extended by our observation that Gq-induced mitochondrial ROS generation is mediated, at least in part, through transcriptional repression of UCP3. Here we further demonstrate that CaMKII contributes to UCP3 downregulation in response to Gq and that this downregulation is functionally important in mediating Gq and CaMKII induced ROS accumulation and cell death. Our findings suggest that activation of CaMKII, which occurs in response to multiple agonists and stressors, is a nodal point for regulation of mitochondrial ROS generation which in turn sustains a maladaptive feed forward cycle in which ROS generation further activates CaMKII signaling.31
There is a broad precedent for transcriptional regulation of UCP3 by pharmacological interventions and physiological stress.36 The role of CaMKII in transcriptional regulation has also been well documented.7 Here we demonstrate CaMKII-dependent decreases in expression of both UCP3 and the transcription factor PPAR-α in hearts of Gq mice and in NRVMs expressing activated Gq or CaMKIIδ. siRNA-mediated knockdown of PPAR-α reduced UCP3 expression, and no further effects of Gq or CaMKII on UCP3 expression were observed in the absence of PPAR-α, supporting a role for PPAR-α in transcriptional control of UCP3. We also report decreases in both UCP3 and PPAR-α following pressure overload, which are rescued by CaMKIIδ deletion. PPAR-α has been shown to be reduced in experimental models of heart failure and PPAR-α agonists shown to preserve cardiac function.22, 36, 43, 44 The observation that the CaMKII regulated mitochondrial genes are significantly enriched for PPAR-α binding sites suggests a mechanism by which CaMKII activation would induce mitochondrial reprogramming and associated decompensation to heart failure. How CaMKII down regulates PPAR-α and subsequently UCP3 remains to be determined. We used pharmacological inhibitors to test effects of both Class I and Class II histone deacylases (HDACs) on this process in NRVMs but failed to observe any block of CaMKII_c mediated-PPAR-α or UCP3 downregulation. Another possible mechanism for gene repression by CaMKII could be through effects on microRNAs, a topic that is of considerable interest but clearly beyond the scope of the present study.
Clinical implications
Using cellular and murine models of pathological hypertrophy induced by sustained Gq-signaling, we show that deletion of the predominant cardiac isoform (CaMKIIδ) does not block development of hypertrophy but prevents the transition from compensatory cardiac hypertrophy into dilated cardiomyopathy. Our study therefore shows that CaMKIIδ is dispensable for Gq induced hypertrophy, but critical for the subsequent transition from hypertrophy to heart failure. Importantly, similar observations are made when pressure overload rather than Gq is used as a stressor. CaMKII is activated downstream of multiple neurohumoral agonists, including β-adrenergic agonists, angiotensin II and aldosterone and plays a crucial role in their pathophysiological effects in the heart.7, 45, 46 Accordingly pharmacological inhibitors of CaMKII may offer novel therapeutic opportunities for specifically blocking the maladaptive effects of hypertrophy induced by multiple insults.
Limitations
CaMKIIδ can localize to mitochondria and a recent study determined that mitochondrial-localized CaMKII plays a central role in cardiac ischemia/reperfusion injury.18 Our study was focused on CaMKII-induced changes in mitochondrial gene transcription, but we cannot exclude the possibility that salutary effects of deleting CaMKIIδ on mitochondrial function are also mediated, in part, by changes in the activity of CaMKII in mitochondria. In addition, a recent study suggested that CaMKII stimulates NADPH-oxidase-mediated ROS-production, suggesting that CaMKII could also exert detrimental effects by increasing ROS levels through extra-mitochondrial mechanisms.47 The observation that State 4 respiration and respiratory control ratios were comparable between the groups might appear inconsistent with there being a major role for UCP3 downregulation in the mitotoxicity induced by CaMKII. However, our assay conditions were not optimized for the purpose of detecting subtle differences in coupling, thus such changes would not have been observed under our assay conditions. Despite these possible limitations, our study provides compelling evidence for a role of CaMKII and UCP3 in controlling mitochondrial oxidative stress in the heart.
Conclusions
Mitochondrial reprogramming by CaMKIIδ contributes to the maladaptive transition from Gq-mediated hypertrophy to cardiomyopathy.
Novelty and Significance.
What Is Known?
Cardiomyocytes subject to neurohumoral stresses or expressing the signal transducer Gαq undergo a process of hypertrophy which ultimately decompensates into heart failure.
Ca2+/calmodulin-dependent protein kinase II delta (CaMKIIδ) is activated by neurohumoral and oxidative stress and is required for hypertrophy decompensation.
Induction of heart failure by Gαq is mediated through mitochondrial oxidative stress but the link between these events is incompletely understood.
What New Information Does This Article Contribute?
CaMKIIδ mediates heart failure development, mitochondrial dysfunction and mitochondrial oxidative stress in response to pressure overload and Gαq.
CaMKIIδ activation results in changes in expression of nuclear encoded mitochondrial genes that contribute to mitochondrial dysfunction and oxidative stress.
The mitochondrial Uncoupling Protein 3 (UCP3) is downregulated by Gαq/CaMKIIδ and central to the mitochondrial oxidative stress and cell death that contributes to heart failure development.
Studies using CaMKII inhibitors and CaMKIIδ knockout mice demonstrate its key role in the transition from hypertrophy to heart failure induced by pressure overload and other stresses. The underlying mechanisms are unclear. The Gαq transgenic mouse is a genetic model in which heart failure development is linked to mitochondrial oxidative stress. We crossed Gαq TG and CaMKIIδ KO mice and determined that CaMKIIδ deletion prevented Gαq from inducing ventricular dilation and dysfunction, impairing mitochondrial respiration, increasing mitochondrial reactive oxygen species and inducing cardiomyocyte apoptosis. Deep RNA sequencing revealed that there was extensive reprogramming of nuclear encoded mitochondrial genes in response to Gαq which was prevented in the absence of CaMKIIδ. We focused on a mitochondrial uncoupling protein, UCP3, which was markedly downregulated by Gαq signaling through CaMKIIδ. UCP3 was shown by gain and loss of function studies to contribute to mitochondrial ROS production and depolarization as well as to cardiomyocyte cell death. We suggest that CaMKIIδ-mediated suppression of UCP3 is a central driver of the mitochondrial oxidative stress that underlies hypertrophy decompensation. These findings provide novel insights into the mechanistic underpinnings of hypertrophy decompensation and further emphasize the potential for CaMKII as a therapeutic target.
Acknowledgments
We would like to thank Melissa Barlow for assistance with animal breeding and preparation of myocytes and Janny Takens, Sike Oberdorf and Linda van Genne for expert technical assistance and advice.
SOURCES OF FUNDING
This work was supported by the National Institute of Health (NIH HL080101, NS087611 and HL59888) to Drs. JHB, AM and GWD respectively. Dr. Westenbrink is supported by the University of Groningen, ICIN Netherlands Heart Institute and the Dutch Heart Foundation (2012T066). Dr. Ling was supported by an American Heart Association Post-Doctoral Fellowship (0825268F). Charles B Gray was supported by the UCSD Graduate Training Program in Cellular and Molecular Pharmacology (T32 GM007752).
Nonstandard Abbreviations and Acronyms
- Ad-Q209L
Adenoviral expression of constitutively active Gαq
- CaMKII
Calcium, the calcium/calmodulin dependent kinase II
- CD-CaMKIIδ
Adenovirus expressing catalytically dead CaMKIIδ
- Gq
Gαq/Gαq transgenic
- Gq/KO
Gq mice in a CaMKIIδ knockout background
- KO
CaMKIIδ knockout
- NRVMs
Neonatal rat ventricular myocytes
- NAC
N-acetyl-cysteine
- PPAR-α
Peroxisome proliferator-activated receptor alpha
- ROS
Reactive oxygen species
- TAC
Transverse aortic constriction
- TMRE
Tetramethylrhodamine ethyl ester
- UCP3
Uncoupling protein 3
- WT
Wildtype
Footnotes
In December 2014, the average time from submission to first decision for all original research papers submitted to Circulation Research was 14.47 days.
DISCLOSURES
None.
References
- 1.Dorn GW., 2nd The fuzzy logic of physiological cardiac hypertrophy. Hypertension. 2007;49:962–970. doi: 10.1161/HYPERTENSIONAHA.106.079426. [DOI] [PubMed] [Google Scholar]
- 2.Kannel WB, Doyle JT, McNamara PM, Quickenton P, Gordon T. Precursors of sudden coronary death. Factors related to the incidence of sudden death. Circulation. 1975;51:606–613. doi: 10.1161/01.cir.51.4.606. [DOI] [PubMed] [Google Scholar]
- 3.Levy D, Garrison RJ, Savage DD, Kannel WB, Castelli WP. Prognostic implications of echocardiographically determined left ventricular mass in the Framingham Heart Study. N Engl J Med. 1990;322:1561–1566. doi: 10.1056/NEJM199005313222203. [DOI] [PubMed] [Google Scholar]
- 4.Frey N, Olson EN. Cardiac hypertrophy: the good, the bad, and the ugly. Annu Rev Physiol. 2003;65:45–79. doi: 10.1146/annurev.physiol.65.092101.142243. [DOI] [PubMed] [Google Scholar]
- 5.D’Angelo DD, Sakata Y, Lorenz JN, Boivin GP, Walsh RA, Liggett SB, Dorn GW., 2nd Transgenic Galphaq overexpression induces cardiac contractile failure in mice. Proc Natl Acad Sci U S A. 1997;94:8121–8126. doi: 10.1073/pnas.94.15.8121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wettschureck N, Rutten H, Zywietz A, Gehring D, Wilkie TM, Chen J, Chien KR, Offermanns S. Absence of pressure overload induced myocardial hypertrophy after conditional inactivation of Galphaq/Galpha11 in cardiomyocytes. Nat Med. 2001;7:1236–1240. doi: 10.1038/nm1101-1236. [DOI] [PubMed] [Google Scholar]
- 7.Anderson ME, Brown JH, Bers DM. CaMKII in myocardial hypertrophy and heart failure. J Mol Cell Cardiol. 2011;51:468–473. doi: 10.1016/j.yjmcc.2011.01.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zhang T, Maier LS, Dalton ND, Miyamoto S, Ross J, Jr, Bers DM, Brown JH. The deltaC isoform of CaMKII is activated in cardiac hypertrophy and induces dilated cardiomyopathy and heart failure. Circ Res. 2003;92:912–919. doi: 10.1161/01.RES.0000069686.31472.C5. [DOI] [PubMed] [Google Scholar]
- 9.Ling H, Zhang T, Pereira L, Means CK, Cheng H, Gu Y, Dalton ND, Peterson KL, Chen J, Bers D, Brown JH. Requirement for Ca2+/calmodulin-dependent kinase II in the transition from pressure overload-induced cardiac hypertrophy to heart failure in mice. J Clin Invest. 2009;119:1230–1240. doi: 10.1172/JCI38022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Backs J, Backs T, Neef S, Kreusser MM, Lehmann LH, Patrick DM, Grueter CE, Qi X, Richardson JA, Hill JA, Katus HA, Bassel-Duby R, Maier LS, Olson EN. The delta isoform of CaM kinase II is required for pathological cardiac hypertrophy and remodeling after pressure overload. Proc Natl Acad Sci U S A. 2009;106:2342–2347. doi: 10.1073/pnas.0813013106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zhang R, Khoo MS, Wu Y, Yang Y, Grueter CE, Ni G, Price EE, Jr, Thiel W, Guatimosim S, Song LS, Madu EC, Shah AN, Vishnivetskaya TA, Atkinson JB, Gurevich VV, Salama G, Lederer WJ, Colbran RJ, Anderson ME. Calmodulin kinase II inhibition protects against structural heart disease. Nat Med. 2005;11:409–417. doi: 10.1038/nm1215. [DOI] [PubMed] [Google Scholar]
- 12.Ling H, Gray CB, Zambon AC, Grimm M, Gu Y, Dalton N, Purcell NH, Peterson K, Brown JH. Ca2+/Calmodulin-dependent protein kinase II delta mediates myocardial ischemia/reperfusion injury through nuclear factor-kappaB. Circ Res. 2013;112:935–944. doi: 10.1161/CIRCRESAHA.112.276915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zhang T, Guo T, Mishra S, Dalton ND, Kranias EG, Peterson KL, Bers DM, Brown JH. Phospholamban ablation rescues sarcoplasmic reticulum Ca(2+) handling but exacerbates cardiac dysfunction in CaMKIIdelta(C) transgenic mice. Circ Res. 2010;106:354–362. doi: 10.1161/CIRCRESAHA.109.207423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lancel S, Qin F, Lennon SL, Zhang J, Tong X, Mazzini MJ, Kang YJ, Siwik DA, Cohen RA, Colucci WS. Oxidative posttranslational modifications mediate decreased SERCA activity and myocyte dysfunction in Galphaq-overexpressing mice. Circ Res. 2010;107:228–232. doi: 10.1161/CIRCRESAHA.110.217570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dai DF, Johnson SC, Villarin JJ, Chin MT, Nieves-Cintron M, Chen T, Marcinek DJ, Dorn GW, 2nd, Kang YJ, Prolla TA, Santana LF, Rabinovitch PS. Mitochondrial oxidative stress mediates angiotensin II-induced cardiac hypertrophy and Galphaq overexpression-induced heart failure. Circ Res. 2011;108:837–846. doi: 10.1161/CIRCRESAHA.110.232306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Satoh M, Matter CM, Ogita H, Takeshita K, Wang CY, Dorn GW, 2nd, Liao JK. Inhibition of apoptosis-regulated signaling kinase-1 and prevention of congestive heart failure by estrogen. Circulation. 2007;115:3197–3204. doi: 10.1161/CIRCULATIONAHA.106.657981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Elrod JW, Wong R, Mishra S, Vagnozzi RJ, Sakthievel B, Goonasekera SA, Karch J, Gabel S, Farber J, Force T, Brown JH, Murphy E, Molkentin JD. Cyclophilin D controls mitochondrial pore-dependent Ca(2+) exchange, metabolic flexibility, and propensity for heart failure in mice. J Clin Invest. 2010;120:3680–3687. doi: 10.1172/JCI43171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Joiner ML, Koval OM, Li J, He BJ, Allamargot C, Gao Z, Luczak ED, Hall DD, Fink BD, Chen B, Yang J, Moore SA, Scholz TD, Strack S, Mohler PJ, Sivitz WI, Song LS, Anderson ME. CaMKII determines mitochondrial stress responses in heart. Nature. 2012;491:269–273. doi: 10.1038/nature11444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Matkovich SJ, Zhang Y, Van Booven DJ, Dorn GW., 2nd Deep mRNA sequencing for in vivo functional analysis of cardiac transcriptional regulators: application to Galphaq. Circ Res. 2010;106:1459–1467. doi: 10.1161/CIRCRESAHA.110.217513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Dhir A, Zolkowska D, Murphy RB, Rogawski MA. Seizure protection by intrapulmonary delivery of propofol hemisuccinate. J Pharmacol Exp Ther. 2011;336:215–222. doi: 10.1124/jpet.110.173591. [DOI] [PubMed] [Google Scholar]
- 21.Miyamoto S, Purcell NH, Smith JM, Gao T, Whittaker R, Huang K, Castillo R, Glembotski CC, Sussman MA, Newton AC, Brown JH. PHLPP-1 negatively regulates Akt activity and survival in the heart. Circulation research. 2010;107:476–484. doi: 10.1161/CIRCRESAHA.109.215020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Chen R, Liang F, Morimoto S, Li Q, Moriya J, Yamakawa J, Takahashi T, Iwai K, Kanda T. The effects of a PPARalpha agonist on myocardial damage in obese diabetic mice with heart failure. Int Heart J. 2010;51:199–206. doi: 10.1536/ihj.51.199. [DOI] [PubMed] [Google Scholar]
- 23.Azzu V, Jastroch M, Divakaruni AS, Brand MD. The regulation and turnover of mitochondrial uncoupling proteins. Biochim Biophys Acta. 2010;1797:785–791. doi: 10.1016/j.bbabio.2010.02.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yang S, Chen C, Wang H, Rao X, Wang F, Duan Q, Chen F, Long G, Gong W, Zou MH, Wang DW. Protective effects of Acyl-coA thioesterase 1 on diabetic heart via PPARalpha/PGC1alpha signaling. PLoS One. 2012;7:e50376. doi: 10.1371/journal.pone.0050376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dubchak I, Munoz M, Poliakov A, Salomonis N, Minovitsky S, Bodmer R, Zambon AC. Whole-Genome rVISTA: a tool to determine enrichment of transcription factor binding sites in gene promoters from transcriptomic data. Bioinformatics. 2013;29:2059–2061. doi: 10.1093/bioinformatics/btt318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Esposito G, Rapacciuolo A, Naga Prasad SV, Takaoka H, Thomas SA, Koch WJ, Rockman HA. Genetic alterations that inhibit in vivo pressure-overload hypertrophy prevent cardiac dysfunction despite increased wall stress. Circulation. 2002;105:85–92. doi: 10.1161/hc0102.101365. [DOI] [PubMed] [Google Scholar]
- 27.Ardehali H, Sabbah HN, Burke MA, Sarma S, Liu PP, Cleland JG, Maggioni A, Fonarow GC, Abel ED, Campia U, Gheorghiade M. Targeting myocardial substrate metabolism in heart failure: potential for new therapies. Eur J Heart Fail. 2012;14:120–129. doi: 10.1093/eurjhf/hfr173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Treuting PM, Linford NJ, Knoblaugh SE, Emond MJ, Morton JF, Martin GM, Rabinovitch PS, Ladiges WC. Reduction of age-associated pathology in old mice by overexpression of catalase in mitochondria. J Gerontol A Biol Sci Med Sci. 2008;63:813–822. doi: 10.1093/gerona/63.8.813. [DOI] [PubMed] [Google Scholar]
- 29.Dai DF, Santana LF, Vermulst M, Tomazela DM, Emond MJ, MacCoss MJ, Gollahon K, Martin GM, Loeb LA, Ladiges WC, Rabinovitch PS. Overexpression of catalase targeted to mitochondria attenuates murine cardiac aging. Circulation. 2009;119:2789–2797. doi: 10.1161/CIRCULATIONAHA.108.822403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Oka T, Hikoso S, Yamaguchi O, Taneike M, Takeda T, Tamai T, Oyabu J, Murakawa T, Nakayama H, Nishida K, Akira S, Yamamoto A, Komuro I, Otsu K. Mitochondrial DNA that escapes from autophagy causes inflammation and heart failure. Nature. 2012;485:251–255. doi: 10.1038/nature10992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Erickson JR, Joiner ML, Guan X, Kutschke W, Yang J, Oddis CV, Bartlett RK, Lowe JS, O’Donnell SE, Aykin-Burns N, Zimmerman MC, Zimmerman K, Ham AJ, Weiss RM, Spitz DR, Shea MA, Colbran RJ, Mohler PJ, Anderson ME. A dynamic pathway for calcium-independent activation of CaMKII by methionine oxidation. Cell. 2008;133:462–474. doi: 10.1016/j.cell.2008.02.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Swaminathan PD, Purohit A, Soni S, Voigt N, Singh MV, Glukhov AV, Gao Z, He BJ, Luczak ED, Joiner ML, Kutschke W, Yang J, Donahue JK, Weiss RM, Grumbach IM, Ogawa M, Chen PS, Efimov I, Dobrev D, Mohler PJ, Hund TJ, Anderson ME. Oxidized CaMKII causes cardiac sinus node dysfunction in mice. J Clin Invest. 2011;121:3277–3288. doi: 10.1172/JCI57833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.He BJ, Joiner ML, Singh MV, Luczak ED, Swaminathan PD, Koval OM, Kutschke W, Allamargot C, Yang J, Guan X, Zimmerman K, Grumbach IM, Weiss RM, Spitz DR, Sigmund CD, Blankesteijn WM, Heymans S, Mohler PJ, Anderson ME. Oxidation of CaMKII determines the cardiotoxic effects of aldosterone. Nature medicine. 2011;17:1610–1618. doi: 10.1038/nm.2506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Erickson JR, He BJ, Grumbach IM, Anderson ME. CaMKII in the cardiovascular system: sensing redox states. Physiological reviews. 2011;91:889–915. doi: 10.1152/physrev.00018.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Mailloux RJ, Seifert EL, Bouillaud F, Aguer C, Collins S, Harper ME. Glutathionylation acts as a control switch for uncoupling proteins UCP2 and UCP3. J Biol Chem. 2011;286:21865–21875. doi: 10.1074/jbc.M111.240242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Divakaruni AS, Brand MD. The regulation and physiology of mitochondrial proton leak. Physiology (Bethesda) 2011;26:192–205. doi: 10.1152/physiol.00046.2010. [DOI] [PubMed] [Google Scholar]
- 37.Bugger H, Guzman C, Zechner C, Palmeri M, Russell KS, Russell RR., 3rd Uncoupling protein downregulation in doxorubicin-induced heart failure improves mitochondrial coupling but increases reactive oxygen species generation. Cancer Chemother Pharmacol. 2011;67:1381–1388. doi: 10.1007/s00280-010-1441-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Mailloux RJ, Dumouchel T, Aguer C, deKemp R, Beanlands R, Harper ME. Hexokinase II acts through UCP3 to suppress mitochondrial reactive oxygen species production and maintain aerobic respiration. Biochem J. 2011;437:301–311. doi: 10.1042/BJ20110571. [DOI] [PubMed] [Google Scholar]
- 39.Liu D, Huang L, Wang Y, Wang W, Wehrens XH, Belousova T, Abdelrahim M, DiMattia G, Sheikh-Hamad D. Human stanniocalcin-1 suppresses angiotensin II-induced superoxide generation in cardiomyocytes through UCP3-mediated anti-oxidant pathway. PLoS One. 2012;7:e36994. doi: 10.1371/journal.pone.0036994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Boudina S, Han YH, Pei S, Tidwell TJ, Henrie B, Tuinei J, Olsen C, Sena S, Abel ED. UCP3 Regulates Cardiac Efficiency and Mitochondrial Coupling in High Fat-Fed Mice but Not in Leptin-Deficient Mice. Diabetes. 2012 doi: 10.2337/db12-0063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ozcan C, Palmeri M, Horvath TL, Russell KS, Russell RR., 3rd Role of uncoupling protein 3 in ischemia-reperfusion injury, arrhythmias, and preconditioning. Am J Physiol Heart Circ Physiol. 2013;304:H1192–1200. doi: 10.1152/ajpheart.00592.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Perrino C, Schiattarella GG, Sannino A, Pironti G, Petretta MP, Cannavo A, Gargiulo G, Ilardi F, Magliulo F, Franzone A, Carotenuto G, Serino F, Altobelli GG, Cimini V, Cuocolo A, Lombardi A, Goglia F, Indolfi C, Trimarco B, Esposito G. Genetic deletion of uncoupling protein 3 exaggerates apoptotic cell death in the ischemic heart leading to heart failure. J Am Heart Assoc. 2013;2:e000086. doi: 10.1161/JAHA.113.000086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Sarma S, Ardehali H, Gheorghiade M. Enhancing the metabolic substrate: PPAR-alpha agonists in heart failure. Heart Fail Rev. 2012;17:35–43. doi: 10.1007/s10741-010-9208-0. [DOI] [PubMed] [Google Scholar]
- 44.Ichihara S, Obata K, Yamada Y, Nagata K, Noda A, Ichihara G, Yamada A, Kato T, Izawa H, Murohara T, Yokota M. Attenuation of cardiac dysfunction by a PPAR-alpha agonist is associated with down-regulation of redox-regulated transcription factors. J Mol Cell Cardiol. 2006;41:318–329. doi: 10.1016/j.yjmcc.2006.05.013. [DOI] [PubMed] [Google Scholar]
- 45.Swaminathan PD, Purohit A, Hund TJ, Anderson ME. Calmodulin-dependent protein kinase II: linking heart failure and arrhythmias. Circ Res. 2012;110:1661–1677. doi: 10.1161/CIRCRESAHA.111.243956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Grimm M, Ling H, Brown JH. Crossing signals: relationships between beta-adrenergic stimulation and CaMKII activation. Heart Rhythm. 2011;8:1296–1298. doi: 10.1016/j.hrthm.2011.02.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Nishio S, Teshiman Y, Takahashi N, Thuc LC, Saito S, Fukui A, Kume O, Fukunaga N, Hara M, Nakagawa M, Saikawa T. Activation of CaMKII as a key regulator of reactive oxygen species production in diabetic rat heart. J Mol Cell Cardiol. 2012;52:1103–1111. doi: 10.1016/j.yjmcc.2012.02.006. [DOI] [PubMed] [Google Scholar]