Abstract
Recent results from laboratory investigations and clinical trials indicate important roles for estrogen receptor (ER) agonists in protecting the central nervous system (CNS) from noxious consequences of neuroinflammation and neurodegeneration. Neurodegenerative processes in several CNS disorders including spinal cord injury (SCI), multiple sclerosis (MS), Parkinson's disease (PD), and Alzheimer's disease (AD) are associated with activation of microglia and astrocytes, which drive the resident neuroinflammatory response. During neurodegenerative processes, activated microglia and astrocytes cause deleterious effects on surrounding neurons. The inhibitory activity of ER agonists on microglia activation might be a beneficial therapeutic option for delaying the onset or progression of neurodegenerative injuries and diseases. Recent studies suggest that ER agonists can provide neuroprotection by modulation of cell survival mechanisms, synaptic reorganization, regenerative responses to axonal injury, and neurogenesis process. The anti-inflammatory and neuroprotective actions of ER agonists are mediated mainly via two ERs known as ERα and ERβ. Although some studies have suggested that ER agonists may be deleterious to some neuronal populations, the potential clinical benefits of ER agonists for augmenting cognitive function may triumph over the associated side effects. Also, understanding the modulatory activities of ER agonists on inflammatory pathways will possibly lead to the development of selective anti-inflammatory molecules with neuroprotective roles in different CNS disorders such as SCI, MS, PD, and AD in humans. Future studies should be concentrated on finding the most plausible molecular pathways for enhancing protective functions of ER agonists in treating neuroinflammatory and neurodegenerative injuries and diseases in the CNS.
Keywords: estrogen receptor agonists, inflammation, neurodisorders, neuroprotection
1. Introduction
Estrogens are involved in the development and maintenance of normal reproductive functions. They also play very important roles in the immune system as well as in the central nervous system (CNS) in human body (Warner and Gustafsson, 2014). Especially, 17β-estradiol (E2) is the most potent estrogen produced in the human body. Estrone and estriol, the other two active metabolites of E2, are found to be less potent than E2 on estrogen receptors (ERs). Recent studies indicated the organ specific roles of these two estrogen metabolites (Watson et al., 2008).
Elwood Jensen and co-workers first discovered the estrogen binding protein known as ERα (Jensen et al., 1962). The first ERα knockout mouse was created in 1993 (Lubahn et al., 1993) but the knockout mouse showed normal functions of life. Following characterization of ERβ, researchers speculated that ERβ would imitate the action of ERα and support the survival of the ERα knockout mouse. Then, ERβ and double ERαβ knockout mice were created to solve the question (Krege et al., 1998). All single and double knockout studies involving ERα and ERβ showed the drastic impairment of reproductive function without much alteration in normal functions life (Couse et al., 1999). Recently, ER agonists have clearly been shown to possess neuroprotective effects in spinal cord injury (SCI) in rats (Sribnick et al., 2009a). Reduced levels of estrogen are associated with the development of neurodegenerative disorders such as Alzheimer's disease (AD) (Launer et al., 1999; Zandi et al., 2002) and Parkinson's disease (PD) (Currie et al., 2004; Ragonese et al., 2004). Recent clinical trials in post-menopausal women demonstrated deleterious effects of estrogen-based hormone therapy (Lai et al., 2013). So, development of synthetic estrogenic molecules that selectively mimic estrogen can greatly improve the outcomes in the hormone-based therapy (McDonnell et al., 2000). Most synthetic estrogens have been evaluated for their binding affinities to the ERα or ERβ and their ability to regulate ER-dependent transcription in reporter systems (Sun et al., 1999) but their neuroprotective potentials remain to be fully elucidated.
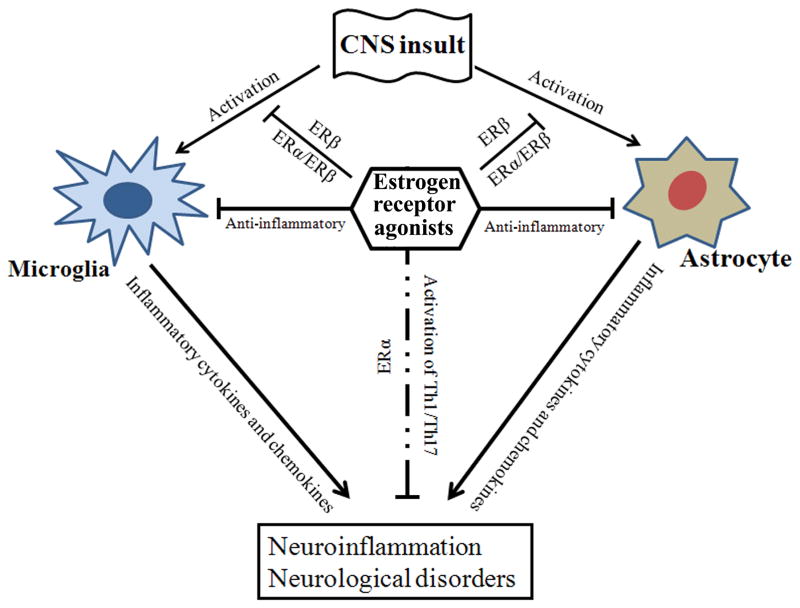
The innate immune responses are regulated by the complex signaling pathways between the immune system and the CNS in the brain (Rivest 2009). Microglia are involved in activation of astrocytes and migration of peripheral immune cells (Voskuhl et al., 2009; Sofroniew and Vinters, 2010) to respond to infection or injury in the brain. Estrogens and ER agonists could modulate the activation of many different cell types of the immune system (Straub, 2007) and the CNS (Spencer et al., 2008; Dumitriu et al., 2010). Recent investigation suggests that estrogens can suppress the activation of microglia and recruit the blood-derived monocytes in rat brain after intracerebroventricular injection of bacterial lipopolysaccharide (LPS) (Vegeto et al., 2003). This investigation also showed an increase in expression of C3 receptor and matrix metalloproteinase-9 (MMP-9) following LPS exposure (Vegeto et al., 2003). Estrogens can also inhibit expression of pro-inflammatory cytokines such as IL-1β and TNF-α in primary astrocytes following LPS exposure (Lewis et al., 2008). These studies suggest that depending on the signaling mechanisms, estrogens can play dual roles for attenuation of neuroinflammation and neurodegeneration by inhibiting activation of microglia and astrocytes (Fig. 1).
Fig. 1.
A schematic representation of anti-inflammatory roles of estrogen and ER agonists in neurodisorders. Insults to the CNS lead to overactivation of microglia and astrocytes. Activated microglia and astrocytes can release pro-inflammatory cytokines and chemokines to induce neuroinflammation to promote pathogenesis in different neurodisorders. Treatment with ER agonists can be useful to inhibit the activation of microglia and astrocytes after the CNS insults. Estrogen driven Th1/Th17 cell response is thought to be carried out via involvement of ERα.
The effects of estrogen and ER agonists are mainly mediated by two genetically distinct receptors, ERα and ERβ, of the nuclear receptor superfamily (Gronemeyer et al., 2004). ERα and ERβ regulate gene expression upon binding to estrogen-responsive elements in target gene promoters, by interfering with other transcription factors, or by modulating a variety of signaling pathways (Schultz et al., 2005). Estrogen and ER agonists are capable of altering the transcription of a large number of genes (Madak-Erdogan et al., 2013) that are known to participate in neuroinflammatory responses in astroglia (Barreto et al., 2009), interneurons (Kritzer 2002), and microglia in frontal cortex (Sierra et al., 2008). Still the involvement of estrogen and ER agonists in regulation of many neuroinflammatory genes in the cerebral cortex remain to be evaluated.
2. Estrogen receptors (ERs) and their subtypes
Estrogen and ER agonists modulate cell signaling pathways mainly through binding to ERα and ERβ, which belong to the nuclear receptor family of transcription factors. It is well established that ERα and ERβ harbor evolutionarily preserved and functionally dissimilar domains as well as high degree of specific sequence homology. The DNA-binding domain at the center is the most preserved part, which participates in binding to specific DNA sequence in the promoter region of the target gene. The C-terminal part is used for ligand-binding. The N-terminal part appears to be variable in length as well as in sequence. Otherwise, ERα and ERβ have substantial sequence homology and comparable affinities for binding to estrogen and ER agonists. Both ERα and ERβ are capable of binding to the same DNA sequence in the promoter of the target gene.
The binding of ER agonists to the ERs triggers estrogen signaling pathways in the target cells. ER agonists can also activate different signaling pathways such as PI3K/Akt and MAPK/ERK to provide neuroprotection (Bourque et al., 2012). Upon activation, ERs act as transcription factors and modulate the expression of many estrogen responsive target genes and this process depends on the presence of other signaling molecules in the cells (Nilsson et al., 2001; Katzenellenbogen and Katzenellenbogen, 2002).
The ERα and ERβ genes are located on different chromosomes and code for 66 and 59 kDa proteins, respectively (Enmark et al., 1997). Mutiple splice variants of both ERα and ERβ have been characterized. Different ERs and other receptor isoforms contribute to the complexity of estrogen signaling. Although various splice variants of ERs have been discovered, their biological functions are not yet clearly determined. A recent investigation indicates that determination of functions of different ERα splice variants and their specificity in the cells may be helpful in understanding the estrogen-based therapy in CNS diseases (Ishunina et al., 2013). Majority of ERα variants have variation in 5′-untranslated region (UTR) but their coding sequence is same. Although some alternative ERβ isoforms are produced in different cells and tissues at different stages (Saji et al., 2002), only the ERβ isoform with 530 amino acids is recognized as the wild-type ERβ in humans (Leygue et al., 1998). Recent investigations indicate that different ERβ isoforms can differently modulate estrogen signaling for regulation of expression of the target gene (Leung et al., 2006; Ramsey et al., 2004). The existence of some common isoforms and species-specific isoforms of ERβ has been reported (Lewandowski et al., 2002). Interactions of different isoforms of ERβ and ERα have not yet been investigated in details. The human ERβ2 isoform is not capable of binding to ligand and does not possess transcriptional ability. It is possible that abolition of transcriptional activity occurs due to dimerization of ERβ2 with ERα (Ogawa et al., 1998).
Unlike other nuclear receptors such as the retinoic acid receptor (RAR) and thyroid hormone receptor (TR), ER ligand cavity can vary in size for estrogen and ER agonists (Brzozowski et al., 1997). Thus, many compounds with diverse molecular structures can bind to ERs. Not only synthetic compounds but also environmental pollutants such as polynuclear aromatic hydrocarbons, phthalate esters, xenohormones, and many pesticides have high affinity for binding to ERs (Bolger et al., 1998). Phytoestrogens, which are estrogen-like compounds derived from plants, have estrogenic properties when used in humans and ranch animals (Oostenbrink et al., 2000). Some of the phytoestrogens can modulate the activities of endocrine signaling pathways and thus they are described as the endocrine disruptors. Exposure to endocrine disruptors may be related to induction of breast cancer and impairment of reproductive function (Witorsch, 2002a). In contrast, many other studies suggest that dietary phytoestrogens in soy and grain products can reduce the risk of some hormone-associated cancers (Witorsch, 2002b). Genistein, which is an abundant phytoestrogen in soy, has much higher selectivity for ERβ than for ERα and can inhibit cancer growth (Barkhem et al., 1998). Based on various exciting results from recent investigations, it appears that genistein is on its way to be an important replacement for estrogen in the treatment of cancer, cardiovascular incidences, diabetes, inflammatory diseases, and metabolic diseases (Heldring et al., 2007; Rimbach et al., 2008; Węgrzyn et al., 2010). These studies suggest that careful use of ER agonists including phytoestrogens can engage specific ERs for providing therapeutic benefits in a number of challenging diseases in humans.
3. Potential therapeutic effects of ER agonists in CNS disorders
Many recent investigations indicated that ER agonists play crucial roles in enhancing memory and cognition and ameliorating neuroinflammation and neurodegenerative diseases. But the beneficial role of ER agonists in acute injury has only recently been a focus of intense investigation. Previous acute injury experiments were performed using males to determine the initiation, progression, and pathophysiological mechanisms with the assumption that results obtained from these studies using males might be applicable to females as well. But recent results suggest that both sex and ER agonists are equally important in producing specific outcomes from the treatment of neurodisorders. Various investigations further showed that females are less affected due to acute insults such as brain ischemia (Green and Simpkins, 2000; Hurn and Macrae, 2000), traumatic CNS injury (Roof and Hall, 2000), hypoxia (Saiyed and Riker, 1993), and toxicity induced by a drug (Cadet et al., 1994). Some of the recent advances in molecular mechanisms of estrogen and ER agonists mediated attenuation of neuroinflammation and neurodegeneration are being described below.
3.1. Traumatic brain injury
Traumatic brain injury (TBI), which is an injury in the brain, causes serious disability and even death. Various studies clearly show that significant gender differences in the occurrence and pathophysiology of TBI do exist. Males have been reported to encounter TBI more frequently than females due to sporting disasters, motor vehicle accidents, combat operations, and street violences. Similarly, occurrence of cerebrovascular stroke (CS) is more frequent in males than the pre-menopausal females (Barrett-Connor and Bush, 1991). The occurrence of CS in older post-menopausal females is almost same when compared with the age-matched males (Wolf, 1990). Following ischemia, hypoxia, or TBI, young female rodents could survive longer than their male counterparts, as shown by number of studies (Zhang et al., 1998; Carswell et al., 1999; Hall et al., 1998). Administrations of ER agonists can provide neuroprotection against different type of neuronal injury in vitro and in vivo and the treatments reduce the extent of injury and, in some cases, decrease mortality and behavioral deficiency (Shao et al., 2012; Schreihofer and Ma, 2013). Various investigators have examined many ER agonists with different doses. ER agonists have been administered at physiological and pharmacological levels at different time points before or after TBI. Results indicated that lower physiological concentrations of ER agonists should be administered before the injury to exert protective actions, while pharmacological doses of ER agonists may be protective even when administered at 3 h following induction of injury (Yang et al., 2000). Although ER agonists provide neuroprotection against neural injury, the precise molecular signaling pathways by which they achieve neuroprotection still remain mostly unclear. Studies in numerous cell culture models have indicated inhibition of the neuronal death after treatment with the ER agonists following toxic insults. Similarly, many animal model studies demonstrated that different doses of ER agonists could produce different outcomes in toxic insults such as oxidative stress, glucose deprivation, hypoxia, and physical injury (Wise et al., 2001). More investigations in animal models will be needed to establish the optimum doses of ER agonists in the treatment of TBI and other injuries in the brain.
3.2. Spinal cord injury
Spinal cord injury (SCI) is a highly complex CNS injury that can be associated with different levels of contusion, axonal damage, oxygen depletion, hemorrhage, and pathophysiological mechanisms. Primary injury to the spinal cord causes the immediate insults to the neurons, axons, glial cells, and blood vessels at the site of injury (Ray et al., 2003; Samantaray et al., 2010; Ray et al., 2011). Secondary injury involves devastating pathophysiological mechanisms including increase in reactive oxygen species (ROS), reperfusion, glutamate concentration, and mitochondrial damage (Carlson et al., 1998; Mills et al., 2000). Mitochondrial damage in SCI alters Na+/K+-ATPase activity, increases intracellular Ca2+ level, and activates glutamate receptors (Agrawal et al., 2000; Li et al., 2000; Wingrave et al., 2004). Increase in intracellular free Ca2+ level following SCI leads to activation of the Ca2+-activated protease calpain and phospholipases (Dhillon et al., 1999; Ray et al., 2003). Upon activation, calpain can degrade a number of cytoskeletal proteins such as neurofilament proteins, α-spectrin, and myelin basic protein (Ray et al., 2003). A sustained activation of calpain can cleave calpastatin, which is an endogenous calpain inhibitor, resulting in an uncontrolled calpain mediated proteolysis in tissue (Pang et al., 2003). Altogether these effects cause biochemical and metabolic changes in the spinal cord leading to neural cell death and progressive tissue damage (Sribnick et al., 2009a). Both primary and secondary injury mechanisms of SCI can result in significant neurological deficits, permanent paralysis, or even death of the SCI victim.
Recent studies suggest that estrogen and ER agonists can protect neurons and inhibit axonal degeneration during early phase (48 h) following SCI in rats (Sribnick et al., 2005; Sribnick et al., 2009a). Similar outcomes have been observed in ischemia and TBI (Dubal et al., 2001; Jover et al., 2002). Experimental TBI in animal models indicate that TBI females recover better than TBI males after treatment with ER agonists (Bayir et al., 2004). A number of cell culture studies established the impressive neuroprotective potential of ER agonists in glial cells as well as in neurons following exposure to oxygen free radicals or glutamate toxicity (Sribnick et al., 2004; Das et al., 2005; Sribnick et al., 2009b). Other studies also confirm that ER agonists can be used as potent anti-oxidant (Moosmann and Behl, 1999) and anti-inflammatory agents (Dimayuga et al., 2005) to provide functional neuroprotection. We recently reported that miR-7-1 potentiated ER agonists for functional neuroprotection in VSC4.1 motoneurons (Chakrabarti et al., 2014). Indeed, the multi-action characteristics of ER agonists make them very attractive therapeutic agents for treatment of SCI.
3.3. Cerebral ischemia
Cerebral ischemia or stroke is the sudden loss of CNS function due to inhibition in the blood supply to the brain. Low physiological levels of estrogen can dramatically reduce overall infarct size in the middle cerebral artery with permanent occlusion (Dubal et al., 1998), indicating the therapeutic potentials of estrogen and ER agonists in cerebral ischemia. The risk of stroke treatment outcomes clearly depend on the gender of the stroke victims (Hurn and Macrae, 2000; McCullough et al., 2001). Young women with normal endogenous levels of estrogen have significantly less risk and severity of stroke than age-matched men. Both the vasoprotective and neuroprotective roles of estrogen have been documented in experimental cerebral ischemia (McCullough et al., 2001). It is interesting to note that ER agonists through activation of ERα and ERβ can exert their vasodilatory and neuroprotective effects in systemic circulation (Luksha et al., 2009). Recently, it has been reported that another G protein-coupled ER, called GPR30, is involved in acute vasodilatory effect of ER agonists in arteries and veins in humans (Haas et al., 2007; Murata et al., 2013). Another recent study suggests that GPR30 agonists have the potential to reduce neuronal injury following global cerebral ischemia (Kosaka et al., 2012). Hyper-activation of N-methyl-d-aspartate receptors (NMDARs) has been observed in different neurodegenerative conditions (Vizi et al., 2013). NMDARs consist of three different subtypes: NR1, NR2, and NR3. The NR2B subunit is observed in the extra-synaptic sites and it plays a significant role in glutamate-mediated neuronal cytotoxicity in both cell culture and animal models (Liu and Zhao, 2013). Neuroprotective potential of estrogen has been reported to be partially mediated by GPR30 and the subsequent down regulation of NR2B-containing NMDARs (Liu et al., 2012). But, the details about GPR30 mediated vasoactive effects in cerebral microcirculation still remain unclear (Murata et al., 2013). The molecular pathways leading to vessel dysfunction during cerebral ischemia and reperfusion include inhibition of K+ channels (Bari et al., 1996), enhanced oxidative stress (Hossmann et al., 2006), and reduced level of nitric oxide (NO) (Cipolla et al., 2008). Studies suggest that ER agonists are capable of improving microvascular dysfunction by preserving the process of vasodilation (Watanabe et al., 2001) or by reducing oxidative stress (Stirone et al., 2005) in experimental cerebral ischemia.
3.4. Multiple sclerosis
Multiple sclerosis (MS) is a heterogeneous neuroinflammatory demyelinating autoimmune disease triggered by T helper 1 (Th1) and Th17 cells. Current studies also clearly indicate MS as a neurodegenerative disease. Studies performed in many different laboratories have shown that the clinical severity of both active and adoptive experimental autoimmune encephalomyelitis (EAE), which is an animal model of MS, is reduced by treatment with ER agonists in several types of mouse models (Bebo et al., 2001; Ito et al., 2001; Liu et al., 2002; Liu et al., 2003; Subramanian et al., 2003). Estriol can effectively reduce the severity of the EAE in animals when administered after disease onset (Kim et al., 1999). Estriol has been shown to inhibit a number of inflammatory cytokines such as IFN-γ, TNF-α, IL-2, and IL-6 in the 6 to 8 weeks old C57BL/6 mice (Palaszynski et al., 2004). The immunomodulatory effects of ER agonists influence T cell differentiation and effector functions including the expansion of the CD4+CD25+ T regulatory (Treg) cells in EAE animals (Polanczyk et al., 2004). Estrogen and ER agonists can also modulate the functions of many different organ systems including the immune system. ER agonists have also been shown to induce apoptosis in T cells through activation of Fas-Fas ligand pathway, thereby causing immunosuppression (Do et al., 2002; Singh et al., 2012). Estrogen can influence development of CD4+ T cell subpopulations and function through regulation of cytokine profiles (Pernis et al., 2007). It also regulates the expression of adhesion and accessory molecules on endothelial cells and alters leukocyte migration. Low doses of estrogen have been shown to enhance antigen-specific Th1 and Th17 cell responses as well as several other IFN-γ-producing cells via differential activation of MAPK, NF-κB, and AP-1 signaling pathways (Kassi and Moutsatsou, 2010). It is believed that ERα, but not ERβ, is necessary for the enhanced estrogen-driven Th1/Th17 cell responsiveness (Fig. 1). Estrogen may also influence CD8+ T cells and multiple signaling cascades through cytosolic Ca2+ influx. Although estrogen has multiple roles in inflammatory diseases, other ER agonists also exert their anti-inflammatory and neuroprotective roles to prevent inflammation and autoimmunity in the CNS. Antigen presenting cells (APCs) such as dendritic cells (DCs) and macrophages may play important roles in connecting the innate immune system with the adaptive immune system. A recent study indicated that endogenous estrogen level can modulate the number of APCs (Nalbandian and Kovats, 2005). ER agonists can regulate activities of T cells through direct involvement of APC functions. Studies have shown that splenic DCs isolated from estrogen-treated animals produce lower levels of TNF-α, IFN-γ, and IL-12 upon LPS exposure, while macrophages produce decreased levels of TNF-α (Flohé et al., 2008). One of the major roles for ERα signaling in T lymphocytes is the induction of anti-inflammatory effects of estrogen and protection against CNS inflammation. Because estrogen treatment provides protective effects in EAE animals, efficacy of estrogen and ER agonists are currently being evaluated in clinical trials for treatment of MS patients.
3.5. Parkinson's disease
Parkinson's disease (PD) is a debilitating movement disorder that is mainly characterized by the irreversible and selective degeneration of the dopaminergic neurons in the substantia nigra pars compacta (Hornykiewicz, 1989). There is no effective treatment for PD although L-dopa is an impressive choice in the treatment of some PD patients. All other current medications for PD are symptomatic treatments that hardly prevent the progression of PD. An important observation is highly notable that ER agonists modulate the dopaminergic neurotransmission and may be used to alleviate major symptoms of PD (Session et al., 1994; Giladi et al., 1995). Another study reported that estrogen therapy could lower the severity of initial phase of PD at least prior to administration of L-dopa in PD women (Saunders-Pullman et al., 1999), indicating that estrogen and ER agonists could be useful therapy in PD patients. Inhibition of neuroinflammatory signaling molecules and blockage of neurodegenerative pathways by estrogen and ER agonists may be promising therapeutic approach against PD.
So far, several clinical trials have been reported showing no significant effect (Strijks et al., 1999) or a moderate anti-parkinsonian effect (Blanchet et al., 1999) of estrogen therapy in PD. Hence, the exact role of estrogen on the survival of dopaminergic neurons in humans still remains a mystery. However, some PD animal model studies with newer selective ER modulators (SERMs) like raloxifene supported both pro-dopaminergic and anti-dopaminergic activities of ER agonists in parkinsonism. Many recent reports indicate that incidence of PD is higher in men than in women (Marder et al., 1996; Bower et al., 1999; Baldereschi et al., 2000; Bower et al., 2000), while another study shows existence of no such sex difference in occurrence of PD (de Rijk et al., 1995). Obviously, more studies are needed to resolve this controversy. It should be noted that during progression of PD, men manifest more parkinsonian motor features or dyskinesia than women do (Lyons et al., 1998). Treatment with ER agonists can inhibit development of dementia (Marder et al., 1996), indicating efficacy of ER agonists in brain disorder. While no proven mechanism yet exists for neuroprotective action of estrogen, it is highly plausible that estrogen may reduce oxidative stress and inhibit mitochondrial dysfunction so as to prevent progression of pathophysiology in PD (Numakawa et al., 2011). Estrogen can prevent degeneration of dopaminergic neurons in the 1-methyl-4-phenyl-1,2,3,6-tetrapyridine (MPTP)-induced PD animal brain (Numakawa et al., 2011). While the mechanism responsible for degeneration of dopaminergic neurons in MPTP animal model of PD is unclear, oxidative stress and neuroinflammation are thought to play key roles in ultimate demise of the nigrostriatal dopaminergic neurons. Calpain is a proteolytic enzyme that is activated in response to increases in intracellular free Ca2+ and oxidatative stress, both of which are presumed to be present at high levels in dying dopaminergic neurons in MPTP animal model of PD. In fact, recent results indicate a significant role of calpain mediated proteolysis in degeneration and loss of dopaminergic neurons in an animal model of PD (Crocker et al., 2003). It will be interesting to examine whether ER agonists can provide neuroprotective functions through regulation of expression of the Ca2+-binding protein and calpain so as to prevent loss of dopaminergic neurons in the animal model of PD.
3.6. Alzheimer's disease
Alzheimer's disease (AD) is characterized by deficiencies in memory and cognition due to degeneneration of mostly hippocampal neurons in the old people. It is the most common cause of dementia, which impairs daily life and activities due to abnormal brain functioning and behavioral difficulties in the AD patients. Currently available symtomatic treatments do not prevent pathogenesis in AD patients. So, innovative and novel therapeutic agents are urgently needed for proper treatment and inhibition of pathogenic mechanisms in AD patients. The neuroprotective actions of ER agonists in AD are supposed to be regulated by activation of ERs in different neurons in different areas of the brain. Both ERα and ERβ subtypes are widely distributed in the hippocampus, frontal cortex, and amygdala regions (Shughrue et al., 1997; Shughrue and Merchenthaler, 2000; Hart et al., 2001). A very recent study identified alteration in normal distribution of ERs in hippocampal neurons in AD (Liu et al., 2008). A shift of ERα localization from the nucleus to the cytoplasm has been shown to inhibit the development of AD pathology in transgenic mice (Jorm et al., 1987) as well as in humans (Henderson et al., 1994). Folstein test scores in women with terminal stage AD showed an increase in ERα level in the frontal cortex (Kawas et al., 1997). Also, allele differences in ERα appear to correlate well with a high-risk for development of AD in women who are already suffering from Down's syndrome (Schupf et al., 2008). When compared with the same age control groups, immunoreactivity of ERβ was found to be increased in the hippocampal cells in AD (Savaskan et al., 2001). Upregulation of ERs in hippocampus of AD patients should provide an advantage to the therapeutic approach using ER agonists. Collectively, recent investigations suggest that the levels of expression of the ERα and ERβ can play important roles for ER agonists for inhibition of neuroinflammation and achieving neuroprotective functions in the AD brain. Several interesting studies have already described these phenomena in details (Lee et al., 2014).
4. Mechanisms of action of ER agonists for neuroprotection
As mentioned previously, both ERα and ERβ are involved in estrogen mediated neuroprotection. Selective expression of ERα could restore protective action of estrogen against amyloid-β (Aβ) peptide in HT22 cell line, suggesting a prominent role for ERα in estrogen mediated neuroprotection (Kim et al., 2001). The selective ERα agonist (propylpyrazole triol, PPT) and ERβ agonist (diarylpropionitrile; DPN) are supposed to show similar neuroprotective actions in culture studies. But a previous report showed higher neuroprotective potential of the ERα agonist PPT than the ERβ agonist DPN (Behl et al., 1995). In contrast, another study with primary neuron culture showed comparable levels of neuroprotection by estrogen and selective ER agonists against Aβ peptide mediated neurodegeneration, indicating the contribution of both ERα and ERβ in neuroprotection (Corder et al., 2004). Some other investigations showed that both PPT and DPN can provide neuroprotection against glutamate toxicity by increasing the expression of the anti-apoptotic Bcl-2 protein and also modulating the stress kinase signaling pathways (Zhao et al., 2007; Zhao and Brinton, 2007). These results suggest that DPN has a higher Ca2+ dependency for its activity than other ER agonists (Zhao and Brinton, 2007). Altogether, recent reports strongly imply that both ERα and ERβ are involved in achieving significant neuroprotection by the ER agonists against different neurodisorders through activation of various cell survival signaling pathways.
5. Benefits and limitations of selective ER modulators (SERMs)
The mode of action of ER agonists in different neurodisorders depends mostly on the presence of ERs. Therapeutic strategy with SERMs can be another promising option for the treatment of neurodegenerative disorders. Some synthetic and natural SERMs like tamoxifen, raloxifene, or bazedoxifene (Mickley and Dluzen 2004; Zhao et al., 2005; Kokiko et al., 2006; Zhao et al., 2006) and genistein (Azcoitia et al., 2006) are neuroprotective (Table I). New neuroprotective SERMs (Neuro-SERMs) may be developed to avoid feminizing effects and specifically target the nervous system to promote cognitive function and to reduce the risk of neurodegenerative diseases (Brinton., 2004, Zhao et al., 2005).
Table 1. Estrogen and SERM derivatives examined for potential neuroprotective functions.
| Estrogen and SERM derivatives | Estrogen or SERM | Functions | References |
|---|---|---|---|
| Estrogen derivatives | 17β-Estradiol | Antioxidant, neuroprotective | Chakrabarti et al., 2014; Mosquera et al., 2014 |
| 17α-Estradiol | Inhibits GABA receptor-induced cell loss | McClean and Nuñez, 2008; Rakkestad et al., 2014 | |
| Estriol | Reduces the disease symptoms in multiple sclerosis (MS) | Sicotte et al., 2002; Ziehn et al., 2012 | |
| Equilin | Used in hormone replacement therapy | Sawicki et al., 1999; Okamoto et al., 2010 | |
| Triphenylethylene derivatives | Tamoxifen | Neuroprotection to Aβ-induced toxicity | O'Neill et al. 2004; Herrera et al., 2011 |
| 4-Hydroxy tamoxifen | Neuroprotection to Aβ-induced toxicity | O'Neill et al., 2004; Arevalo et al., 2011 | |
| Droloxifene | Inhibits estrogen mediated neuroprotection | Christian, 2001; Zhao et al., 2005 | |
| Ospemifene | Induces production and release of pro-inflammatory molecules by glial cells | Cerciat et al., 2010; Arevalo et al., 2011 | |
| Phytoestrogens | Coumestrol | Protects hippocampal neurons in cerebral ischemia | Castro et al., 2012; Castro et al., 2014 |
| Daidzein | Decreases cell death and improves synaptic function in oxygen–glucose deprivation (OGD) | Schreihofer and Redmond, 2009; Hurtado et al., 2012 | |
| Equol | Vasorelaxant, anti-oxidant, and neuroprotective in transient focal cerebral ischemia | Jackman et al., 2007; Ma et al., 2010. | |
| Formononetin | Protects neurons from NMDA-induced excitotoxic injury and neurodegenerative disorders in central nervous system | Occhiuto et al., 2008; Occhiuto et al., 2009; Tian et al., 2013 | |
| Genistein | Reduces oxidative stress, hippocampal neuron death, and cognitive defects in neurodegenerative disorders | Azcoitia et al., 2006; Malinowska et al., 2010; Wang et al., 2013 | |
| Benzothiophene derivatives | Arzoxifene | GPR30-dependent and ER-independent neuroprotection | Littleton-Kearney et al., 2002; Abdelhamid et al., 2011 |
| Bazedoxifene | Anti-inflammatory effects in astrocytes, inhibition of IL-6, IFN-ɣ, and NF-κB p65 transactivation | Cerciat et al., 2010; Arevalo et al., 2011 | |
| Raloxifene | Poor bioavailability, ERα dependent but ERβ and GPR30 independent hypoxia induced neuroprotection | Kushwaha et al., 2013; Rzemieniec et al., 2014 |
Most of the investigations have so far been focused on tamoxifen and raloxifene for developing effective SERMs against neurodegenerative diseases (Table I). Further studies are needed in animal models with new SERMs like lasofoxifene, bazedoxifene, arzoxifene, and ospemifene to identify an effective agent against neurodegenerative diseases. Bazedoxifene can provide neuroprotection against kainic acid toxicity in rat hippocampal neurons (Kulkarni et al., 2013). Also, combination therapy with ospemifene and bazedoxifene can inhibit inflammatory response in astrocytes (Cerciat et al., 2010). Further studies are needed to uncover the neuroprotective efficacy of SERMs (Azcoitia et al., 2006; Schreihofer and Redmond, 2009).
Clinical application of SERMs as neuroprotective agents will require better permeability of the drug molecules so as to cross the blood-brain-barrier in the CNS. Many investigators are actively working to overcome the current limitations of SERMs to increase their clinical applicability. SERMs with higher affinity to the ERs are also under intense investigation to identify an effective neuroprotective agent (Brinton, 2004; Zhao et al., 2005). One such SERM is 7α-[(4R,8R)-4,8,12-trimethyltridecyl]estra-1,3,5-trien-3,17β-diol that contains the combined structures of vitamin E and estrogen. This molecule has been neuroprotective in rat primary hippocampal neurons and it is capable of binding to both ERα and ERβ (Zhao et al., 2007). Non feminizing analogs of estrogen are other interesting molecules, which are also under intense investigation (Petrone et al., 2014). Other molecules such as ent-17-desoxyestradiol, 17α estrogen, ent-estrogen, and 2-adamantylestrone also show neuroprotective effects in animals with neurodegenerative conditions (Jung et al., 2006; Wang et al., 2006).
Although recent investigations have identified several important molecular targets of ER agonists and SERMs in the CNS in humans, their precise molecular mechanisms of action remain mostly unknown. It has been suggested that ER agonists modulate different signaling pathways involing MAPK, PI3K/Akt, CREB, and NF-κB. In general, ER agonists and SERMs can exert their cell specific neuroprotective efficacy by modulating neuronal death, remyelination, and inhibiting chronic pro-inflammatory responses.
6. Conclusion and future direction
Estrogen and ER agonists work mainly via ERα and ERβ for mediation of their antiinflammatory and neuroprotective effects in many CNS injuries and diseases. Currently, cell culture and animal model studies suggest that ER agonists hold great promise for amelioration of the devastating consequences of several neurodisorders in humans. In addition to ER agonists, vigorous research is focused on SERMs for identification of the most appropriate therapeutic agents for treatment CNS injuries and diseases in humans in the future. Further studies are needed to establish the exact molecular mechanisms of ER agonists and SERMs for inhibition of neuroinflammation and neurodegeneration in diverse neurodisoders in the human brain and spinal cord.
Highlights.
Estrogen receptor (ER) agonists play crucial roles in the immune system and the CNS
Cell signaling pathways are modulated by ER agonists via binding to ERα and ERβ
ER agonists can promote transcription of a large number of neuroprotective genes
Neuroinflammation and neurodegeneration in the CNS are prevented by ER agonists
ER agonists play significant roles in enhancing memory and cognition
Acknowledgments
This work was supported in part by a grant (SC SCIRF-11-002) from the South Carolina Spinal Cord Injury Research Fund (SC SCIRF, Columbia, SC, USA), an incentive award from the United Soybean Board (USB, Chesterfield, MO, USA), an internal award from the Research Development Fund (RDF, University of SC School of Medicine, Columbia, SC, USA), grants (R01 NS65456, R01 AT006888, R01 ES019313, P01 AT003961, and P20 GM103641) from the National Institutes of Health (NIH, Bethesda, MD, USA), and merit awards (I0 BX001262 and I0 BX001357) from the Veterans Health Administration (VHA, Baltimore, MD, USA).
Footnotes
Conflict of interest: Authors have no conflict of interest to declare.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Abdelhamid R, Luo J, Vandevrede L, Kundu I, Michalsen B, Litosh VA, Schiefer IT, Gherezghiher T, Yao P, Qin Z, Thatcher GR. Benzothiophene selective estrogen receptor modulators provide neuroprotection by a novel GPR30-dependent mechanism. ACS Chem Neurosci. 2011;2:256–268. doi: 10.1021/cn100106a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Agrawal SK, Nashmi R, Fehlings MG. Role of L- and N-type calcium channels in the pathophysiology of traumatic spinal cord white matter injury. Neuroscience. 2000;99:179–188. doi: 10.1016/s0306-4522(00)00165-2. [DOI] [PubMed] [Google Scholar]
- Arevalo MA, Diz-Chaves Y, Santos-Galindo M, Bellini MJ, Garcia-Segura LM. Selective oestrogen receptor modulators decrease the inflammatory response of glial cells. J Neuroendocrinol. 2012;24:183–190. doi: 10.1111/j.1365-2826.2011.02156.x. [DOI] [PubMed] [Google Scholar]
- Azcoitia I, Moreno A, Carrero P, Palacios S, Garcia-Segura LM. Neuroprotective effects of soy phytoestrogens in the rat brain. Gynecol Endocrinol. 2006;22:63–69. doi: 10.1080/09513590500519161. [DOI] [PubMed] [Google Scholar]
- Baldereschi M, Di Carlo A, Rocca WA, Vanni P, Maggi S, Perissinotto E, Grigoletto F, Amaducci L, Inzitari D. Parkinson's disease and parkinsonism in a longitudinal study: two-fold higher incidence in men. ILSA Working Group. Italian Longitudinal Study on Aging. Neurology. 2000;55:1358–1363. doi: 10.1212/wnl.55.9.1358. [DOI] [PubMed] [Google Scholar]
- Bari F, Louis TM, Meng W, Busija DW. Global ischemia impairs ATP-sensitive K+ channel function in cerebral arterioles in piglets. Stroke. 1996;27:1874–1880. doi: 10.1161/01.str.27.10.1874. [DOI] [PubMed] [Google Scholar]
- Barkhem T, Carlsson B, Nilsson Y, Enmark E, Gustafsson J, Nilsson S. Differential response of estrogen receptor alpha and estrogen receptor beta to partial estrogen agonists/antagonists. Mol Pharmacol. 1998;54:105–112. doi: 10.1124/mol.54.1.105. [DOI] [PubMed] [Google Scholar]
- Barreto G, Santos-Galindo M, Diz-Chavez Y, Pernia O, Carrero P, Azcoitia I, Garcia-Segura LM. Selective estrogen receptor modulators decrease reactive astrogliosis in the injured brain: Effects of aging and prolonged depletion of ovarian hormones. Endocrinology. 2009;150:5010–5015. doi: 10.1210/en.2009-0352. [DOI] [PubMed] [Google Scholar]
- Barrett-Connor E, Bush TL. Estrogen and coronary heart disease in women. JAMA. 1991;265:1861–1867. [PubMed] [Google Scholar]
- Bayir H, Marion DW, Puccio AM, Wisniewski SR, Janesko KL, Clark RS, Kochanek PM. Marked gender effect on lipid peroxidation after severe traumatic brain injury in adult patients. J Neurotrauma. 2004;21:1–8. doi: 10.1089/089771504772695896. [DOI] [PubMed] [Google Scholar]
- Bebo BF, Fyfe-Johnson A, Jr, Adlard K, Beam AG, Vandenbark AA, Offner H. Low-dose estrogen therapy ameliorates experimental autoimmune encephalomyelitis in two different inbred mouse strains. J Immunol. 2001;166:2080–2089. doi: 10.4049/jimmunol.166.3.2080. [DOI] [PubMed] [Google Scholar]
- Behl C, Widmann M, Trapp T, Holsboer F. 17-β estradiol protects neurons from oxidative stress-induced cell death in vitro. Biochem Biophys Res Commun. 1995;216:473–482. doi: 10.1006/bbrc.1995.2647. [DOI] [PubMed] [Google Scholar]
- Blanchet PJ, Fang J, Hyland K, Arnold LA, Mouradian MM, Chase TN. Short-term effects of high-dose 17β-estradiol in postmenopausal PD patients: a crossover study. Neurology. 1999;53:91–95. doi: 10.1212/wnl.53.1.91. [DOI] [PubMed] [Google Scholar]
- Bolger R, Wiese TE, Ervin K, Nestich S, Checovich W. Rapid screening of environmental chemicals for estrogen receptor binding capacity. Environ Health Perspect. 1998;106:551–555. doi: 10.1289/ehp.98106551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bourque M, Dluzen DE, Di Paolo T. Signaling pathways mediating the neuroprotective effects of sex steroids and SERMs in Parkinson's disease. Front Neuroendocrinol. 2012;33:169–178. doi: 10.1016/j.yfrne.2012.02.003. [DOI] [PubMed] [Google Scholar]
- Bower JH, Maraganore DM, McDonnell SK, Rocca WA. Incidence and distribution of parkinsonism in Olmsted County, Minnesota, 1976-1990. Neurology. 1999;52:1214–1220. doi: 10.1212/wnl.52.6.1214. [DOI] [PubMed] [Google Scholar]
- Bower JH, Maraganore DM, McDonnell SK, Rocca WA. Influence of strict, intermediate, and broad diagnostic criteria on the age- and sex-specific incidence of Parkinson's disease. Mov Disord. 2000;15:819–825. doi: 10.1002/1531-8257(200009)15:5<819::aid-mds1009>3.0.co;2-p. [DOI] [PubMed] [Google Scholar]
- Brinton RD. Requirements of a brain selective estrogen: advances and remaining challenges for developing a NeuroSERM. J Alzheimers Dis. 2004;6:S27–S35. doi: 10.3233/jad-2004-6s607. [DOI] [PubMed] [Google Scholar]
- Brzozowski AM, Pike AC, Dauter Z, Hubbard RE, Bonn T, Engstrom O, Ohman L, Greene GL, Gustafsson JA, Carlquist M. Molecular basis of agonism and antagonism in the oestrogen receptor. Nature. 1997;389:753–758. doi: 10.1038/39645. [DOI] [PubMed] [Google Scholar]
- Cadet K, Ladenheim B, Baum I, Carlson E, Epstein C. Cu,Zn-superoxide dismutase (CuZnSOD) transgenic mice show resistance to the lethal effects of methylenedioxyamphetamine (MDA) and of methylenedioxymethamphetamine (MDMA) Brain Res. 1994;655:259–262. doi: 10.1016/0006-8993(94)91624-1. [DOI] [PubMed] [Google Scholar]
- Carlson SL, Parrish ME, Springer JE, Doty K, Dossett L. Acute inflammatory response in spinal cord following impact injury. Exp Neurol. 1998;151:77–88. doi: 10.1006/exnr.1998.6785. [DOI] [PubMed] [Google Scholar]
- Carswell HVO, Anderson NH, Clark JS, Graham D, Jeffs B, Dominizak AF, Macrae IM. Genetic and gender influences on sensitivity to focal cerebral ischemia in the stroke-prone SHR. Hypertension. 1999;33:681–685. doi: 10.1161/01.hyp.33.2.681. [DOI] [PubMed] [Google Scholar]
- Castro CC, Pagnussat AS, Moura N, da Cunha MJ, Machado FR, Wyse AT, Netto CA. Coumestrol treatment prevents Na+, K+ -ATPase inhibition and affords histological neuroprotection to male rats receiving cerebral global ischemia. Neurol Res. 2014;36:198–206. doi: 10.1179/1743132813Y.0000000286. [DOI] [PubMed] [Google Scholar]
- Castro SB, Junior CO, Alves CC, Dias AT, Alves LL, Mazzoccoli L, Mesquita FP, Figueiredo NS, Juliano MA, Castañon MC, Gameiro J, Almeida MV, Teixeira HC, Ferreira AP. Immunomodulatory effects and improved prognosis of experimental autoimmune encephalomyelitis after O-tetradecanoyl-genistein treatment. Int Immunopharmacol. 2012;12:465–470. doi: 10.1016/j.intimp.2011.12.025. [DOI] [PubMed] [Google Scholar]
- Cerciat M, Unkila M, Garcia-Segura LM, Arevalo MA. Selective estrogen receptor modulators decrease the production of interleukin-6 and interferon-γ-inducible protein-10 by astrocytes exposed to inflammatory challenge in vitro. Glia. 2010;58:93–102. doi: 10.1002/glia.20904. [DOI] [PubMed] [Google Scholar]
- Chakrabarti M, Banik NL, Ray SK. miR-7-1 potentiated estrogen receptor agonists for functional neuroprotection in VSC4.1 motoneurons. Neuroscience. 2014;256:322–333. doi: 10.1016/j.neuroscience.2013.10.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Christian MS. Introduction/overview: gender-based differences in pharmacologic and toxicologic responses. Int J Toxicol. 2001;20:145–148. doi: 10.1080/109158101317097719. [DOI] [PubMed] [Google Scholar]
- Cipolla MJ, Bullinger LV. Reactivity of brain parenchymal arterioles after ischemia and reperfusion. Microcirculation. 2008;15:495–501. doi: 10.1080/10739680801986742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corder EH, Ghebremedhin E, Taylor MG, Thal DR, Ohm TG, Braak H. The biphasic relationship between regional brain senile plaque and neurofibrillary tangle distributions: modification by age, sex, and APOE polymorphism. Ann N Y Acad Sci. 2004;1019:24–28. doi: 10.1196/annals.1297.005. [DOI] [PubMed] [Google Scholar]
- Couse JF, Korach KS. Estrogen receptor null mice: what have we learned and where will they lead us. Endocr Rev. 1999;20:358–417. doi: 10.1210/edrv.20.3.0370. [DOI] [PubMed] [Google Scholar]
- Crocker SJ, Smith PD, Jackson-Lewis V, Lamba WR, Hayley SP, Grimm E, Callaghan SM, Slack RS, Melloni E, Przedborski S, Robertson GS, Anisman H, Merali Z, Park DS. Inhibition of calpains prevents neuronal and behavioral deficits in an MPTP mouse model of Parkinson's disease. J Neurosci. 2003;23:4081–4091. doi: 10.1523/JNEUROSCI.23-10-04081.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Currie LJ, Harrison MB, Trugman JM, Bennett JP, Wooten GF. Postmenopausal estrogen use affects risk for Parkinson disease. Arch Neurol. 2004;61:886–888. doi: 10.1001/archneur.61.6.886. [DOI] [PubMed] [Google Scholar]
- Das A, Sribnick EA, Wingrave JM, Del Re AM, Woodward JJ, Appel SH, Banik NL, Ray SK. Calpain activation in apoptosis of ventral spinal cord 4.1 (VSC4.1) motoneurons exposed to glutamate: calpain inhibition provides functional neuroprotection. J Neurosci Res. 2005;81:551–562. doi: 10.1002/jnr.20581. [DOI] [PubMed] [Google Scholar]
- de Rijk MC, Breteler MM, Graveland GA, Ott A, Grobbee DE, van der Meche FG, Hofman A. Prevalence of Parkinson's disease in the elderly: the Rotterdam Study. Neurology. 1995;45:2143–2146. doi: 10.1212/wnl.45.12.2143. [DOI] [PubMed] [Google Scholar]
- Dhillon HS, Carman HM, Prasad RM. Regional activities of phospholipase C after experimental brain injury in the rat. Neurochem Res. 1999;24:751–755. doi: 10.1023/a:1020779413122. [DOI] [PubMed] [Google Scholar]
- Dimayuga FO, Reed JL, Carnero GA, Wang C, Dimayuga ER, Dimayuga VM, Perger A, Wilson ME, Keller JN, Bruce-Keller AJ. Estrogen and brain inflammation: effects on microglial expression of MHC, co-stimulatory molecules and cytokines. J Neuroimmunol. 2005;161:123–136. doi: 10.1016/j.jneuroim.2004.12.016. [DOI] [PubMed] [Google Scholar]
- Do Y, Ryu S, Nagarkatti M, Nagarkatti PS. Role of death receptor pathway in estradiol- induced T-cell apoptosis in vivo. Toxicol Sci. 2002;70:63–72. doi: 10.1093/toxsci/70.1.63. [DOI] [PubMed] [Google Scholar]
- Dubal DB, Kashon ML, Pettigrew LC, Ren JM, Finklestein SP, Rau SW, Wise PM. Estradiol protects against ischemic injury. JCBFM. 1998;18:1253–1258. doi: 10.1097/00004647-199811000-00012. [DOI] [PubMed] [Google Scholar]
- Dubal DB, Zhu H, Yu J, Rau SW, Shughrue PJ, Merchenthaler I, Kindy MS, Wise PM. Estrogen receptor α, not β, is a critical link in estradiol-mediated protection against brain injury. Proc Natl Acad Sci U S A. 2001;98:1952–1957. doi: 10.1073/pnas.041483198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dumitriu D, Rapp PR, McEwen BS, Morrison JH. Estrogen and the aging brain: an elixir for the weary cortical network. Ann N Y Acad Sci. 2010;1204:104–112. doi: 10.1111/j.1749-6632.2010.05529.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Enmark E, Pelto-Huikko M, Grandien K, Lagercrantz S, Lagercrantz J, Fried G, Nordenskjold M, Gustafsson JA. Human estrogen receptor beta-gene structure, chromosomal localization, expression pattern. J Clin Endocrinol Metab. 1997;82:4258–4265. doi: 10.1210/jcem.82.12.4470. [DOI] [PubMed] [Google Scholar]
- Flohé SB, Agrawal H, Flohé S, Rani M, Bangen JM, Schade FU. Diversity of interferon gamma and granulocyte-macrophage colony-stimulating factor in restoring immune dysfunction of dendritic cells and macrophages during polymicrobial sepsis. Mol Med. 2008;14:247–256. doi: 10.2119/2007-00120.Flohe. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giladi N, Honigman S. Hormones and Parkinson's disease. Neurology. 1995;45:1028–1029. doi: 10.1212/wnl.45.5.1028-a. [DOI] [PubMed] [Google Scholar]
- Green PS, Simpkins JW. Neuroprotective effects of estrogens: potential mechanisms of action. Int J Dev Neurosci. 2000;18:347–358. doi: 10.1016/s0736-5748(00)00017-4. [DOI] [PubMed] [Google Scholar]
- Gronemeyer H, Gustafsson JA, Laudet V. Principles for modulation of the nuclear receptor superfamily. Nat Rev Drug Discov. 2004;3:950–964. doi: 10.1038/nrd1551. [DOI] [PubMed] [Google Scholar]
- Haas E, Meyer MR, Schurr U, Bhattacharya I, Minotti R, Nguyen HH, Heigl A, Lachat M, Genoni M, Barton M. Differential effects of 17β-estradiol on function and expression of estrogen receptor alpha, estrogen receptor beta, and GPR30 in arteries and veins of patients with atherosclerosis. Hypertension. 2007;49:1358–1363. doi: 10.1161/HYPERTENSIONAHA.107.089995. [DOI] [PubMed] [Google Scholar]
- Hall ED, Sutter DM. Gender differences in infarct size in mice after permanent focal ischemia and in the protective effects of Cu-Zn superoxide dismutase over expression; Annual meeting of the Society of Neuroscience.1998. [Google Scholar]
- Hart SA, Patton JD, Woolley CS. Quantitative analysis of ERα and GAD colocalization in the hippocampus of the adult female rat. J Comp Neurol. 2001;440:144–155. doi: 10.1002/cne.1376. [DOI] [PubMed] [Google Scholar]
- Heldring N, Pike A, Andersson S, Matthews J, Cheng G, Hartman J, Tujague M, Ström A, Treuter E, Warner M, Gustafsson JA. Estrogen receptors: how do they signal and what are their targets. Physiol Rev. 2007;87:905–931. doi: 10.1152/physrev.00026.2006. [DOI] [PubMed] [Google Scholar]
- Henderson VW, Paganini-Hill A, Emanuel CK, Dunn ME, Buckwalter JG. Estrogen replacement therapy in older women. Comparisons between Alzheimer's disease cases and nondemented control subjects. Arch Neurol. 1994;51:896–900. doi: 10.1001/archneur.1994.00540210068014. [DOI] [PubMed] [Google Scholar]
- Herrera JL, Fernandez C, Diaz M, Cury D, Marin R. Estradiol and tamoxifen differentially regulate a plasmalemmal voltage-dependent anion channel involved in amyloid-beta induced neurotoxicity. Steroids. 2011;76:840–844. doi: 10.1016/j.steroids.2011.02.014. [DOI] [PubMed] [Google Scholar]
- Hornykiewicz O. The neurochemical basis of the pharmacology of Parkinson's disease. In: Calne DB, editor. Handbook of Experimental Pharmacology. New York: Springer Verlag; 1989. pp. 185–204. [Google Scholar]
- Hossmann KA. Pathophysiology and therapy of experimental stroke. Cellular and Mol Neurobiol. 2006;26:1057–1083. doi: 10.1007/s10571-006-9008-1. [DOI] [PubMed] [Google Scholar]
- Hurn PD, Macrae IM. Estrogen as a neuroprotectant in stroke. JCBFM. 2000;20:631–652. doi: 10.1097/00004647-200004000-00001. [DOI] [PubMed] [Google Scholar]
- Hurtado O, Ballesteros I, Cuartero MI, Moraga A, Pradillo JM, Ramírez-Franco J, Bartolomé- Martín D, Pascual D, Torres M, Sánchez-Prieto J, Salom JB, Lizasoain I, Moro MA. Daidzein has neuroprotective effects through ligand-binding-independent PPARγ activation. Neurochem Int. 2012;61:119–127. doi: 10.1016/j.neuint.2012.04.007. [DOI] [PubMed] [Google Scholar]
- Ishunina TA, Sluiter AA, Swaab DF, Verwer RW. Transcriptional activity of human brain estrogen receptor-α splice variants: evidence for cell type-specific regulation. Brain Res. 2013;1500:1–9. doi: 10.1016/j.brainres.2012.12.050. [DOI] [PubMed] [Google Scholar]
- Ito A, Bebo BF, Jr, Matejuk A, Zamora A, Silverman M, Fyfe-Johnson A, Offner H. Estrogen treatment down-regulates TNF-alpha production and reduces the severity of experimental autoimmune encephalomyelitis in cytokine knockout mice. J Immunol. 2001;167:542–552. doi: 10.4049/jimmunol.167.1.542. [DOI] [PubMed] [Google Scholar]
- Jackman KA, Woodman OL, Chrissobolis S, Sobey CG. Vasorelaxant and antioxidant activity of the isoflavone metabolite equol in carotid and cerebral arteries. Brain Res. 2007;1141:99–107. doi: 10.1016/j.brainres.2007.01.007. [DOI] [PubMed] [Google Scholar]
- Jensen EV. On the mechanism of estrogen action. Perspect Biol Med. 1962;6:47–59. doi: 10.1353/pbm.1963.0005. [DOI] [PubMed] [Google Scholar]
- Jorm AF, Korten AE, Henderson AS. The prevalence of dementia: a quantitative integration of the literature. Acta Psychiatr Scand. 1987;76:465–479. doi: 10.1111/j.1600-0447.1987.tb02906.x. [DOI] [PubMed] [Google Scholar]
- Jover T, Tanaka H, Calderone A, Oguro K, Bennett MV, Etgen AM, Zukin RS. Estrogen protects against global ischemia-induced neuronal death and prevents activation of apoptotic signaling cascades in the hippocampal CA1. J Neurosci. 2002;22:2115–2124. doi: 10.1523/JNEUROSCI.22-06-02115.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jung ME, Wilson AM, Simpkins JW. A nonfeminizing estrogen analog protects against ethanol withdrawal toxicity in immortalized hippocampal cells. J Pharmacol Exp Ther. 2006;319:543–550. doi: 10.1124/jpet.106.103630. [DOI] [PubMed] [Google Scholar]
- Kassi E, Moutsatsou P. Estrogen receptor signaling and its relationship to cytokines in systemic lupus erythematosus. J Biomed Biotechnol. 2010;2010:317452. doi: 10.1155/2010/317452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katzenellenbogen BS, Katzenellenbogen JA. Biomedicine. Defining the “S” in SERMs. Science. 2002;295:2380–2381. doi: 10.1126/science.1070442. [DOI] [PubMed] [Google Scholar]
- Kawas C, Resnick S, Morrison A, Brookmeyer R, Corrada M, Zonderman A, Bacal C, Donnell Lingle D, Metter E. A prospective study of estrogen replacement therapy and the risk of developing Alzheimer's disease: The Baltimore Longitudinal Study of Aging. Neurology. 1997;48:1517–1521. doi: 10.1212/wnl.48.6.1517. [DOI] [PubMed] [Google Scholar]
- Kim H, Bang OY, Jung MW, Ha SD, Hong HS, Huh K, Kim SU, Mook-Jung I. Neuroprotective effects of estrogen against beta-amyloid toxicity are mediated by estrogen receptors in cultured neuronal cells. Neurosci Lett. 2001;302:58–62. doi: 10.1016/s0304-3940(01)01659-7. [DOI] [PubMed] [Google Scholar]
- Kim S, Liva SM, Dalal MA, Verity MA, Voskuhl RR. Estriol ameliorates autoimmune demyelinating disease: implications for multiple sclerosis. Neurology. 1999;52:1230–1280. doi: 10.1212/wnl.52.6.1230. [DOI] [PubMed] [Google Scholar]
- Kokiko ON, Murashov AK, Hoane MR. Administration of raloxifene reduces sensorimotor and working memory deficits following traumatic brain injury. Behav Brain Res. 2006;170:233–240. doi: 10.1016/j.bbr.2006.02.026. [DOI] [PubMed] [Google Scholar]
- Kosaka Y, Quillinan N, Bond C, Traystman R, Hurn P, Herson P. GPER1/GPR30 activation improves neuronal survival following global cerebral ischemia induced by cardiac arrest in mice. Transl Stroke Res. 2012;3:500–507. doi: 10.1007/s12975-012-0211-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krege JH, Hodgin JB, Couse JF, Enmark E, Warner M, Mahler JF, Sar M, Korach KS, Gustafsson JA, Smithies O. Generation and reproductive phenotypes of mice lacking estrogen receptor beta. Proc Natl Acad Sci U S A. 1998;95:15677–15682. doi: 10.1073/pnas.95.26.15677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kritzer MF. Regional, laminar, and cellular distribution of immunoreactivity for ER alpha and ER beta in the cerebral cortex of hormonally intact, adult male and female rats. Cereb Cortex. 2002;12:116–128. doi: 10.1093/cercor/12.2.116. [DOI] [PubMed] [Google Scholar]
- Kulkarni J, Gavrilidis E, Worsley R, Van Rheenen T, Hayes E. The role of estrogen in the treatment of men with schizophrenia. Int J Endocrinol Metab. 2013;11:129–136. doi: 10.5812/ijem.6615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kushwaha AK, Vuddanda PR, Karunanidhi P, Singh SK, Singh S. Development and evaluation of solid lipid nanoparticles of raloxifene hydrochloride for enhanced bioavailability. Biomed Res Int. 2013;2013:584549. doi: 10.1155/2013/584549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lai K, Cui J, Ni S, Zhang Y, He J, Yao K. The effects of postmenopausal hormone use on cataract: a meta-analysis. PLoS One. 2013;8:e78647. doi: 10.1371/journal.pone.0078647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Launer LJ, Andersen K, Dewey ME, Letenneur L, Ott A, Amaducci LA, Brayne C, Copeland JR, Dartigues JF, Kragh- Sorensen P, Lobo A, Martinez-Lage JM, Stijnen T, Hofman A. Rates and risk factors for dementia and Alzheimer's disease: results from EURODEM pooled analyses. EURODEM Incidence Research Group and Work Groups. European Studies of Dementia. Neurology. 1999;52:78–84. doi: 10.1212/wnl.52.1.78. [DOI] [PubMed] [Google Scholar]
- Lee JH, Jiang Y, Han DH, Shin SK, Choi WH, Lee MJ. Targeting estrogen receptors for the treatment of Alzheimer's disease. Mol Neurobiol. 2014;49:39–49. doi: 10.1007/s12035-013-8484-9. [DOI] [PubMed] [Google Scholar]
- Leung YK, Mak P, Hassan S, Ho SM. Estrogen receptor (ER)-beta isoforms: a key to understanding ER-beta signaling. Proc Natl Acad Sci U S A. 2006;103:13162–13167. doi: 10.1073/pnas.0605676103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lewandowski S, Kalita K, Kaczmarek L. Estrogen receptor beta. Potential functional significance of a variety of mRNA isoforms. FEBS Lett. 2002;524:1–5. doi: 10.1016/s0014-5793(02)03015-6. [DOI] [PubMed] [Google Scholar]
- Lewis DK, Johnson AB, Stohlgren S, Harms A, Sohrabji F. Effects of estrogen receptor agonists on the regulation of the inflammatory response in astrocytes from young adult and middle-aged female rats. J Neuroimmunol. 2008;195:47–59. doi: 10.1016/j.jneuroim.2008.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leygue E, Dotzlaw H, Lu B, Glor C, Watson PH, Murphy LC. Estrogen receptor beta: mine is longer than yours? J Clinic Endocrinol Metab. 1998;83:3754–3755. doi: 10.1210/jcem.83.10.5187-1. [DOI] [PubMed] [Google Scholar]
- Li S, Jiang Q, Stys PK. Important role of reverse Na+-Ca2+ exchange in spinal cord white matter injury at physiological temperature. J Neurophysiol. 2000;84:1116–1119. doi: 10.1152/jn.2000.84.2.1116. [DOI] [PubMed] [Google Scholar]
- Littleton-Kearney MT, Ostrowski NL, Cox DA, Rossberg MI, Hurn PD. Selective estrogen receptor modulators: tissue actions and potential for CNS protection. CNS Drug Rev. 2002;8:309–330. doi: 10.1111/j.1527-3458.2002.tb00230.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu HB, Loo KK, Palaszynski K, Ashouri J, Lubahn DB, Voskuhl RR. Estrogen receptor alpha mediates estrogen's immune protection in autoimmune disease. J Immunol. 2003;171:6936–6940. doi: 10.4049/jimmunol.171.12.6936. [DOI] [PubMed] [Google Scholar]
- Liu HY, Buenafe AC, Matejuk A, Ito A, Zamora A, Dwyer J, Vandenbark AA, Offner H. Estrogen inhibition of EAE involves effects on dendritic cell function. J Neurosci Res. 2002;70:238–248. doi: 10.1002/jnr.10409. [DOI] [PubMed] [Google Scholar]
- Liu SB, Zhang N, Guo YY, Zhao R, Shi TY, Feng SF, Wang SQ, Yang Q, Li XQ, Wu YM, Ma L, Hou Y, Xiong LZ, Zhang W, Zhao MG. G-protein-coupled receptor 30 mediates rapid neuroprotective effects of estrogen via depression of NR2B-containing NMDA receptors. J Neurosci. 2012;32:4887–4900. doi: 10.1523/JNEUROSCI.5828-11.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu SB, Zhao MG. Neuroprotective effect of estrogen: role of nonsynaptic NR2B-containing NMDA receptors. Brain Res Bull. 2013;93:27–31. doi: 10.1016/j.brainresbull.2012.10.004. [DOI] [PubMed] [Google Scholar]
- Liu XA, Zhu LQ, Zhang Q, Shi HR, Wang SH, Wang Q, Wang JZ. Estradiol attenuates tau hyperphosphorylation induced by upregulation of protein kinase-A. Neurochem Res. 2008;33:1811–1820. doi: 10.1007/s11064-008-9638-4. [DOI] [PubMed] [Google Scholar]
- Lubahn DB, Moyer JS, Golding TS, Couse JF, Korach KS, Smithies O. Alteration of reproductive function but not prenatal sexual development after insertional disruption of the mouse estrogen receptor gene. Proc Natl Acad Sci U S A. 1993;90:11162–11166. doi: 10.1073/pnas.90.23.11162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luksha L, Kublickiene K. The role of estrogen receptor subtypes for vascular maintenance. Gynecol Endocrinol. 2009;25:82–95. doi: 10.1080/09513590802485038. [DOI] [PubMed] [Google Scholar]
- Lyons KE, Hubble JP, Troster AI, Pahwa R, Koller WC. Gender differences in Parkinson's disease. Clin Neuropharmacol. 1998;21:118–121. [PubMed] [Google Scholar]
- Ma Y, Sullivan JC, Schreihofer DA. Dietary genistein and equol (4', 7 isoflavandiol) reduce oxidative stress and protect rats against focal cerebral ischemia. Am J Physiol Regul Integr Comp Physiol. 2010;299:R871–R877. doi: 10.1152/ajpregu.00031.2010. [DOI] [PubMed] [Google Scholar]
- Madak-Erdogan Z, Charn TH, Jiang Y, Liu ET, Katzenellenbogen JA, Katzenellenbogen BS. Integrative genomics of gene and metabolic regulation by estrogen receptors α and β, and their coregulators. Mol Syst Biol. 2013;9:676. doi: 10.1038/msb.2013.28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malinowska M, Wilkinson FL, Langford-Smith KJ, Langford-Smith A, Brown JR, Crawford BE, Vanier MT, Grynkiewicz G, Wynn RF, Wraith JE, Wegrzyn G, Bigger BW. Genistein improves neuropathology and corrects behaviour in a mouse model of neurodegenerative metabolic disease. PLoS One. 2010;5:e14192. doi: 10.1371/journal.pone.0014192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marder K, Tang MX, Mejia H, Alfaro B, Cote L, Louis E, Groves J, Mayeux R. Risk of Parkinson's disease among first-degree relatives: A community-based study. Neurology. 1996;47:155–160. doi: 10.1212/wnl.47.1.155. [DOI] [PubMed] [Google Scholar]
- McClean J, Nuñez JL. 17alpha-Estradiol is neuroprotective in male and female rats in a model of early brain injury. Exp Neurol. 2008;210:41–50. doi: 10.1016/j.expneurol.2007.09.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCullough LD, Alkayed NJ, Traystman RJ, Williams MJ, Hurn PD. Postischemic estrogen reduces hypoperfusion and secondary ischemia after experimental stroke. Stroke. 2001;32:796–802. doi: 10.1161/01.str.32.3.796. [DOI] [PubMed] [Google Scholar]
- McDonnell DP. Selective estrogen receptor modulators (SERMs): a first step in the development of perfect hormone replacement therapy regimen. J Soc Gynecol Investig. 2000;7:S10–S15. doi: 10.1016/s1071-5576(99)00055-6. [DOI] [PubMed] [Google Scholar]
- Mickley KR, Dluzen DE. Dose-response effects of estrogen and tamoxifen upon methamphetamine-induced behavioral responses and neurotoxicity of the nigrostriatal dopaminergic system in female mice. Neuroendocrinology. 2004;79:305–316. doi: 10.1159/000079710. [DOI] [PubMed] [Google Scholar]
- Mills CD, Xu GY, Johnson KM, McAdoo DJ, Hulsebosch CE. AIDA reduces glutamate release and attenuates mechanical allodynia after spinal cord injury. Neuroreport. 2000;11:3067–3070. doi: 10.1097/00001756-200009280-00007. [DOI] [PubMed] [Google Scholar]
- Moosmann B, Behl C. The antioxidant neuroprotective effects of estrogens and phenolic compounds are independent from their estrogenic properties. Proc Natl Acad Sci U S A. 1999;96:8867–8872. doi: 10.1073/pnas.96.16.8867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mosquera L, Colón JM, Santiago JM, Torrado AI, Meléndez M, Segarra AC, Rodríguez-Orengo JF, Miranda JD. Tamoxifen and estradiol improved locomotor function and increased spared tissue in rats after spinal cord injury: their antioxidant effect and role of estrogen receptor alpha. Brain Res. 2014;1561:11–22. doi: 10.1016/j.brainres.2014.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murata T, Dietrich HH, Xiang C, Dacey RG., Jr G protein-coupled estrogen receptor agonist improves cerebral microvascular function after hypoxia/reoxygenation injury in male and female rats. Stroke. 2013;44:779–785. doi: 10.1161/STROKEAHA.112.678177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nalbandian G, Kovats S. Understanding sex biases in immunity: effects of estrogen on the differentiation and function of antigen-presenting cells. Immunol Res. 2005;31:91–106. doi: 10.1385/IR:31:2:091. [DOI] [PubMed] [Google Scholar]
- Nilsson S, Makela S, Treuter E, Tujague M, Thomsen J, Andersson G, Enmark E, Pettersson K, Warner M, Gustafsson JA. Mechanisms of estrogen action. Physiol Rev. 2001;81:1535–1565. doi: 10.1152/physrev.2001.81.4.1535. [DOI] [PubMed] [Google Scholar]
- Numakawa T, Matsumoto T, Numakawa Y, Richards M, Yamawaki S, Kunugi H. Protective action of neurotrophic factors and estrogen against oxidative stress-mediated neurodegeneration. J Toxicol. 2011;2011:405194. doi: 10.1155/2011/405194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Occhiuto F, Palumbo DR, Samperi S, Zangla G, Pino A, De Pasquale R, Circosta C. The isoflavones mixture from Trifolium pratense L. protects HCN 1-A neurons from oxidative stress. Phytother Res. 2009;23:192–196. doi: 10.1002/ptr.2584. [DOI] [PubMed] [Google Scholar]
- Occhiuto F, Zangla G, Samperi S, Palumbo DR, Pino A, De Pasquale R, Circosta C. The phytoestrogenic isoflavones from Trifolium pratense L. (Red clover) protects human cortical neurons from glutamate toxicity. Phytomedicine. 2008;15:676–682. doi: 10.1016/j.phymed.2008.04.007. [DOI] [PubMed] [Google Scholar]
- Ogawa S, Inoue S, Watanabe T, Orimo A, Hosoi T, Ouchi Y, Muramatsu M. Molecular cloning and characterization of human estrogen receptor betacx: a potential inhibitor of estrogen action in human. Nucleic Acids Res. 1998;26:3505–3512. doi: 10.1093/nar/26.15.3505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okamoto Y, Liu X, Suzuki N, Okamoto K, Kim HJ, Laxmi YR, Sayama K, Shibutani S. Equine estrogen-induced mammary tumors in rats. Toxicol Lett. 2010;193:224–228. doi: 10.1016/j.toxlet.2010.01.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Neill K, Chen S, Diaz Brinton R. Impact of the selective estrogen receptor modulator, tamoxifen, on neuronal outgrowth and survival following toxic insults associated with aging and Alzheimer's disease. Exp Neurol. 2004;188:268–278. doi: 10.1016/j.expneurol.2004.01.014. [DOI] [PubMed] [Google Scholar]
- Oostenbrink BC, Pitera JW, van Lipzig MM, Meerman JH, van Gunsteren WF. Simulations of the estrogen receptor ligand-binding domain: affinity of natural ligands and xenoestrogens. J Med Chem. 2000;43:4594–4605. doi: 10.1021/jm001045d. [DOI] [PubMed] [Google Scholar]
- Palaszynski KM, Liu H, Loo KK, Voskuhl RR. Estriol treatment ameliorates disease in males with experimental autoimmune encephalomyelitis: implications for multiple sclerosis. J Neuroimmunol. 2004;149:84–89. doi: 10.1016/j.jneuroim.2003.12.015. [DOI] [PubMed] [Google Scholar]
- Pang Z, Bondada V, Sengoku T, Siman R, Geddes JW. Calpain facilitates the neuron death induced by 3-nitropropionic acid and contributes to the necrotic morphology. J Neuropathol Exp Neurol. 2003;62:633–643. doi: 10.1093/jnen/62.6.633. [DOI] [PubMed] [Google Scholar]
- Pernis AB. Estrogen and CD4+ T cells. Curr Opin Rheumatol. 2007;19:414–420. doi: 10.1097/BOR.0b013e328277ef2a. [DOI] [PubMed] [Google Scholar]
- Petrone AB, Gatson JW, Simpkins JW, Reed MN. Non-feminizing estrogens: a novel neuroprotective therapy. Mol Cell Endocrinol. 2014;389:40–47. doi: 10.1016/j.mce.2013.12.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Polanczyk MJ, Carson BD, Subramanian S, Afentoulis M, Vandenbark AA, Ziegler SF, Offner H. Cutting edge: estrogen drives expansion of the CD4+CD25+ regulatory T cell compartment. J Immunol. 2004;173:2227–2230. doi: 10.4049/jimmunol.173.4.2227. [DOI] [PubMed] [Google Scholar]
- Ragonese P, D’Amelio M, Salemi G, Aridon P, Gammino M, Epifanio A, Morgante L, Savettieri G. Risk of Parkinson disease in women: effect of reproductive characteristics. Neurology. 2004;62:2010–2014. doi: 10.1212/wnl.62.11.2010. [DOI] [PubMed] [Google Scholar]
- Rakkestad KE, Sørvik IB, Overby GR, Debernard KA, Mathisen GH, Paulsen RE. 17α- Estradiol down-regulates glutathione synthesis in serum deprived PC-12 cells. Free Radic Res. 2014;48:1170–1178. doi: 10.3109/10715762.2014.930455. [DOI] [PubMed] [Google Scholar]
- Ramsey TL, Risinger KE, Jernigan SC, Mattingly KA, Klinge CM. Estrogen receptor beta isoforms exhibit differences in ligand-activated transcriptional activity in an estrogen response element sequence-dependent manner. Endocrinology. 2004;145:149–160. doi: 10.1210/en.2003-1043. [DOI] [PubMed] [Google Scholar]
- Ray SK, Hogan EL, Banik NL. Calpain in the pathophysiology of spinal cord injury: neuroprotection with calpain inhibitors. Brain Res Rev. 2003;42:169–185. doi: 10.1016/s0165-0173(03)00152-8. [DOI] [PubMed] [Google Scholar]
- Ray SK, Samantaray S, Smith JA, Matzelle DD, Das A, Banik NL. Inhibition of cysteine proteases in acute and chronic spinal cord injury. Neurotherapeutics. 2011;8:180–186. doi: 10.1007/s13311-011-0037-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rimbach G, Boesch-Saadatmandi C, Frank J, Fuchs D, Wenzel U, Daniel H, Hall WL, Weinberg PD. Dietary isoflavones in the prevention of cardiovascular disease-a molecular perspective. Food Chem Toxicol. 2008;46:1308–1319. doi: 10.1016/j.fct.2007.06.029. [DOI] [PubMed] [Google Scholar]
- Rivest S. Regulation of innate immune responses in the brain. Nature Rev Immunol. 2009;9:429–439. doi: 10.1038/nri2565. [DOI] [PubMed] [Google Scholar]
- Roof RL, Hall ED. Gender differences in acute CNS trauma and stroke: neuroprotective effects of estrogen and progesterone. J Neurotrauma. 2000;17:367–388. doi: 10.1089/neu.2000.17.367. [DOI] [PubMed] [Google Scholar]
- Rzemieniec J, Litwa E, Wnuk A, Lason W, Gołas A, Krzeptowski W, Kajta M. Neuroprotective action of raloxifene against hypoxia-induced damage in mouse hippocampal cells depends on ERα but not ERβ or GPR30 signalling. J Steroid Biochem Mol Biol. 2014 doi: 10.1016/j.jsbmb.2014.05.005. http://dx.doi.org/10.1016/j.jsbmb.2014.05.005. [DOI] [PubMed]
- Saiyed M, Riker WK. Cholinergic and anticholinergic drug effects on survival during hypoxia: significant gender differences. J Pharmacol Exp Ther. 1993;264:1146–1153. [PubMed] [Google Scholar]
- Saji S, Omoto Y, Shimizu C, Warner M, Hayashi Y, Horiguchi S, Watanabe T, Hayashi S, Gustafsson JA, Toi M. Expression of estrogen receptor (ER) (beta)cx protein in ER(alpha)-positive breast cancer: specific correlation with progesterone receptor. Cancer Res. 2002;62:4849–4853. [PubMed] [Google Scholar]
- Samantaray S, Sribnick EA, Das A, Thakore NP, Matzelle D, Yu SP, Ray SK, Wei L, Banik NL. Neuroprotective efficacy of estrogen in experimental spinal cord injury in rats. Ann N Y Acad Sci. 2010;1199:90–94. doi: 10.1111/j.1749-6632.2009.05357.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saunders-Pullman R, Gordon-Elliott J, Parides M, Fahn S, Saunders HR, Bressman S. The effect of estrogen replacement on early Parkinson's disease. Neurology. 1999;52:1417–1421. doi: 10.1212/wnl.52.7.1417. [DOI] [PubMed] [Google Scholar]
- Savaskan E, Olivieri G, Meier F, Ravid R, Muller-Spahn F. Hippocampal estrogen beta-receptor immunoreactivity is increased in Alzheimer's disease. Brain Res. 2001;908:113–119. doi: 10.1016/s0006-8993(01)02610-5. [DOI] [PubMed] [Google Scholar]
- Sawicki MW, Erman M, Puranen T, Vihko P, Ghosh D. Structure of the ternary complex of human 17β-hydroxysteroid dehydrogenase type 1 with 3-hydroxyestra-1,3,5,7-tetraen-17-one (equilin) and NADP+ Proc Natl Acad Sci U S A. 1999;96:840–845. doi: 10.1073/pnas.96.3.840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schreihofer DA, Ma Y. Estrogen receptors and ischemic neuroprotection: who, what, where, and when? Brain Res. 2013;1514:107–122. doi: 10.1016/j.brainres.2013.02.051. [DOI] [PubMed] [Google Scholar]
- Schreihofer DA, Redmond L. Soy phytoestrogens are neuroprotective against stroke-like injury in vitro. Neuroscience. 2009;158:602–609. doi: 10.1016/j.neuroscience.2008.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schultz JR, Petz LN, Nardulli AM. Cell- and ligand-specific regulation of promoters containing activator protein-1 and Sp1 sites by estrogen receptors α and β. J Biol Chem. 2005;280:347–354. doi: 10.1074/jbc.M407879200. [DOI] [PubMed] [Google Scholar]
- Schupf N, Lee JH, Wei M, Pang D, Chace C, Cheng R, Zigman WB, Tycko B, Silverman W. Estrogen receptor-alpha variants increase risk of Alzheimer's disease in women with Down syndrome. Dement Geriatr Cogn Disord. 2008;25:476–482. doi: 10.1159/000126495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Session DR, Pearlstone MM, Jewelewicz R, Kelly AC. Estrogens and Parkinson's disease. Med Hypotheses. 1994;42:280–282. doi: 10.1016/0306-9877(94)90133-3. [DOI] [PubMed] [Google Scholar]
- Shao B, Cheng Y, Jin K. Estrogen, neuroprotection and neurogenesis after ischemic stroke. Curr Drug Targets. 2012;13:188–198. doi: 10.2174/138945012799201702. [DOI] [PubMed] [Google Scholar]
- Shughrue PJ, Lane MV, Merchenthaler I. Comparative distribution of estrogen receptor-alpha and -beta mRNA in the rat central nervous system. J Comp Neurol. 1997;388:507–525. doi: 10.1002/(sici)1096-9861(19971201)388:4<507::aid-cne1>3.0.co;2-6. [DOI] [PubMed] [Google Scholar]
- Shughrue PJ, Merchenthaler I. Estrogen is more than just a “sex hormone”: Novel sites for estrogen action in the hippocampus and cerebral cortex. Front Neuroendocrinol. 2000;21:95–101. doi: 10.1006/frne.1999.0190. [DOI] [PubMed] [Google Scholar]
- Sicotte NL, Liva SM, Klutch R, Pfeiffer P, Bouvier S, Odesa S, Wu TC, Voskuhl RR. Treatment of multiple sclerosis with the pregnancy hormone estriol. Ann Neurol. 2002;52:421–428. doi: 10.1002/ana.10301. [DOI] [PubMed] [Google Scholar]
- Sierra A, Gottfried-Blackmore A, Milner TA, McEwen BS, Bulloch K. Steroid hormone receptor expression and function in microglia. Glia. 2008;56:659–674. doi: 10.1002/glia.20644. [DOI] [PubMed] [Google Scholar]
- Singh NP, Singh UP, Nagarkatti PS, Nagarkatti M. Prenatal exposure of mice to diethylstilbestrol disrupts T-cell differentiation by regulating Fas/Fas ligand expression through estrogen receptor element and nuclear factor-κB motifs. J Pharmacol Exp Ther. 2012;343:351–361. doi: 10.1124/jpet.112.196121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sofroniew MV, Vinters HV. Astrocytes: biology and pathology. Acta Neuropathol. 2010;119:7–35. doi: 10.1007/s00401-009-0619-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spencer JL, Waters EM, Romeo RD, Wood GE, Millner TA, McEwen BS. Uncovering the mechanism of estrogen effects on hippocampal function. Front Neuroendocrinol. 2008;29:219–237. doi: 10.1016/j.yfrne.2007.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sribnick EA, Del Re AM, Ray SK, Woodward JJ, Banik NL. Estrogen attenuates glutamate-induced cell death by inhibiting Ca2+ influx through L-type voltage-gated Ca2+ channels. Brain Res. 2009a;1276:159–170. doi: 10.1016/j.brainres.2009.04.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sribnick EA, Matzelle DD, Ray SK, Banik NL. Brain and Spinal Cord Trauma. In: Estrogen as a prospective multi-active agent for the treatment of spinal cord injury. Banik NL, Ray SK, editors. New York: Springer; 2009b. pp. 581–598. [Google Scholar]
- Sribnick EA, Ray SK, Banik NL. Estrogen as a multi-active neuroprotective agent in traumatic injuries. Neurochem Res. 2004;29:2007–2014. doi: 10.1007/s11064-004-6874-0. [DOI] [PubMed] [Google Scholar]
- Sribnick EA, Wingrave JM, Matzelle DD, Wilford GG, Ray SK, Banik NL. Estrogen attenuated markers of inflammation and decreased lesion volume in acute spinal cord injury in rats. J Neurosci Res. 2005;82:283–293. doi: 10.1002/jnr.20622. [DOI] [PubMed] [Google Scholar]
- Stirone C, Duckles SP, Krause DN, Procaccio V. Estrogen increases mitochondrial efficiency and reduces oxidative stress in cerebral blood vessels. Mol Pharmacol. 2005;68:959–965. doi: 10.1124/mol.105.014662. [DOI] [PubMed] [Google Scholar]
- Straub RH. The complex role of estrogens in inflammation. Endocr Rev. 2007;28:521–574. doi: 10.1210/er.2007-0001. [DOI] [PubMed] [Google Scholar]
- Strijks E, Kremer JA, Horstink MW. Effects of female sex steroids on Parkinson's disease in postmenopausal women. Clin Neuropharmacol. 1999;22:93–97. doi: 10.1097/00002826-199903000-00005. [DOI] [PubMed] [Google Scholar]
- Subramanian S, Matejuk A, Zamora A, Vandenbark AA, Offner H. Oral feeding with ethinyl estradiol suppresses and treats experimental autoimmune encephalomyelitis in SJL mice and inhibits the recruitment of inflammatory cells into the central nervous system. J Immunol. 2003;170:1548–1555. doi: 10.4049/jimmunol.170.3.1548. [DOI] [PubMed] [Google Scholar]
- Sun J, Meyers MJ, Fink BE, Rajendran R, Katzenellenbogen A, Katzenellenbogen BS. Novel ligands that function as selective estrogens or antiestrogens for estrogen receptor-α or estrogen receptor-β. Endocrinology. 1999;140:800–804. doi: 10.1210/endo.140.2.6480. [DOI] [PubMed] [Google Scholar]
- Tian Z, Liu SB, Wang YC, Li XQ, Zheng LH, Zhao MG. Neuroprotective effects of formononetin against NMDA-induced apoptosis in cortical neurons. Phytother Res. 2013 doi: 10.1002/ptr.4928. [DOI] [PubMed] [Google Scholar]
- Vegeto E, Belcredito S, Etteri S, Ghisletti S, Brusadelli A, Meda C, Krust A, Dupont S, Ciana P, Chambon P, Maggi A. Estrogen receptor-α mediates the brain anti-inflammatory activity of estradiol. Proc Natl Acad Sci U S A. 2003;100:9614–9619. doi: 10.1073/pnas.1531957100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vizi ES, Kisfali M, Lőrincz T. Role of nonsynaptic GluN2B-containing NMDA receptors in excitotoxicity: evidence that fluoxetine selectively inhibits these receptors and may have neuroprotective effects. Brain Res Bull. 2013;93:32–38. doi: 10.1016/j.brainresbull.2012.10.005. [DOI] [PubMed] [Google Scholar]
- Voskuhl RR, Peterson RS, Song B, Ao Y, Morales LBJ, Tiwari-Woodruff S, Sofroniew MV. Reactive astrocytes form scar-like perivascular barriers to leukocytes during adaptive immune inflammation of the CNS. J Neurosci. 2009;29:11511–11522. doi: 10.1523/JNEUROSCI.1514-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang R, Tu J, Zhang Q, Zhang X, Zhu Y, Ma W, Cheng C, Brann DW, Yang F. Genistein attenuates ischemic oxidative damage and behavioral deficits via eNOS/Nrf2/HO-1 signaling. Hippocampus. 2013;23:634–647. doi: 10.1002/hipo.22126. [DOI] [PubMed] [Google Scholar]
- Wang X, Dykens JA, Perez E, Liu R, Yang S, Covey DF, Simpkins JW. Neuroprotective effects of 17β-estradiol and nonfeminizing estrogens against H2O2 toxicity in human neuroblastoma SK-N-SH cells. Mol Pharmacol. 2006;70:395–404. doi: 10.1124/mol.106.022384. [DOI] [PubMed] [Google Scholar]
- Warner M, Gustafsson JA. Estrogen receptor β and liver X receptor β: biology and therapeutic potential in CNS diseases. Mol Psychiatry. 2014 doi: 10.1038/mp.2014.23. [DOI] [PubMed] [Google Scholar]
- Watanabe Y, Littleton-Kearney MT, Traystman RJ, Hurn PD. Estrogen restores postischemic pial microvascular dilation. Am J Physiol Heart Circ Physiol. 2001;281:H155–H160. doi: 10.1152/ajpheart.2001.281.1.H155. [DOI] [PubMed] [Google Scholar]
- Watson CS, Jeng YJ, Kochukov MY. Nongenomic actions of estradiol compared with estrone and estriol in pituitary tumor cell signaling and proliferation. FASEB J. 2008;22:3328–3336. doi: 10.1096/fj.08-107672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wegrzyn G, Jakóbkiewicz-Banecka J, Gabig-Cimińska M, Piotrowska E, Narajczyk M, Kloska A, Malinowska M, Dziedzic D, Gołebiewska I, Moskot M, Wegrzyn A. Genistein: a natural isoflavone with a potential for treatment of genetic diseases. Biochem Soc Trans. 2010;38:695–701. doi: 10.1042/BST0380695. [DOI] [PubMed] [Google Scholar]
- Wingrave JM, Sribnick EA, Wilford GG, Matzelle DD, Mou JA, Ray SK, Hogan EL, Banik NL. Relatively low levels of calpain expression in juvenile rat correlate with less neuronal apoptosis after spinal cord injury. Exp Neurol. 2004;187:529–532. doi: 10.1016/j.expneurol.2004.02.001. [DOI] [PubMed] [Google Scholar]
- Wise PM, Dubal DB, Wilson ME, Rau SW, Liu Y. Estrogens: trophic and protective factors in the adult brain. Front Neuroendocrinol. 2001;22:33–66. doi: 10.1006/frne.2000.0207. [DOI] [PubMed] [Google Scholar]
- Witorsch RJ. Low-dose in utero effects of xenoestrogens in mice and their relevance to humans: an analytical review of the literature. Food Chem Toxicol. 2002a;40:905–912. doi: 10.1016/s0278-6915(02)00069-8. [DOI] [PubMed] [Google Scholar]
- Witorsch RJ. Endocrine disruptors: can biological effects and environmental risks be predicted? Regul Toxicol Pharmacol. 2002b;36:118–130. doi: 10.1006/rtph.2002.1564. [DOI] [PubMed] [Google Scholar]
- Wolf PA. An overview of the epidemiology of stroke. Stroke. 1990;21:4–6. [PubMed] [Google Scholar]
- Yang SH, Shi J, Day AL, Simpkins JW. Estradiol exerts neuroprotective effects when administered after ischemic insult. Stroke. 2000;31:745–750. doi: 10.1161/01.str.31.3.745. [DOI] [PubMed] [Google Scholar]
- Zandi PP, Carlson MC, Plassman BL, Welsh-Bohmer KA, Mayer LS, Steffens DC, Breitner JC. Hormone replacement therapy and incidence of Alzheimer disease in older women: the Cache County Study. JAMA. 2002;288:2123–2129. doi: 10.1001/jama.288.17.2123. [DOI] [PubMed] [Google Scholar]
- Zhang YQ, Shi J, Rajakumar G, Day AL, Simpkins JW. Effects of gender and estradiol treatment on focal brain ischemia. Brain Res. 1998;784:321–324. doi: 10.1016/s0006-8993(97)00502-7. [DOI] [PubMed] [Google Scholar]
- Zhao L, Brinton RD. Estrogen receptor alpha and beta differentially regulate intracellular Ca2+ dynamics leading to ERK phosphorylation and estrogen neuroprotection in hippocampal neurons. Brain Res. 2007;1172:48–59. doi: 10.1016/j.brainres.2007.06.092. [DOI] [PubMed] [Google Scholar]
- Zhao L, Jin C, Mao Z, Gopinathan MB, Rehder K, Brinton RD. Design, synthesis, and estrogenic activity of a novel estrogen receptor modulator - a hybrid structure of 17β- estradiol and vitamin E in hippocampal neurons. J Med Chem. 2007;50:4471–4481. doi: 10.1021/jm070546x. [DOI] [PubMed] [Google Scholar]
- Zhao L, O’Neill K, Brinton RD. Selective estrogen receptor modulators (SERMs) for the brain: current status and remaining challenges for developing NeuroSERMs. Brain Res Rev. 2005;49:472–493. doi: 10.1016/j.brainresrev.2005.01.009. [DOI] [PubMed] [Google Scholar]
- Zhao L, O’Neill K, Brinton RD. Estrogenic agonist activity of ICI 182,780 (Faslodex) in hippocampal neurons: implications for basic science understanding of estrogen signaling and development of estrogen modulators with a dual therapeutic profile. J Pharmacol Exp Thera. 2006;319:1124–1132. doi: 10.1124/jpet.106.109504. [DOI] [PubMed] [Google Scholar]
- Ziehn MO, Avedisian AA, Dervin SM, O'Dell TJ, Voskuhl RR. Estriol preserves synaptic transmission in the hippocampus during autoimmune demyelinating disease. Lab Invest. 2012;92:1234–1245. doi: 10.1038/labinvest.2012.76. [DOI] [PMC free article] [PubMed] [Google Scholar]