Abstract
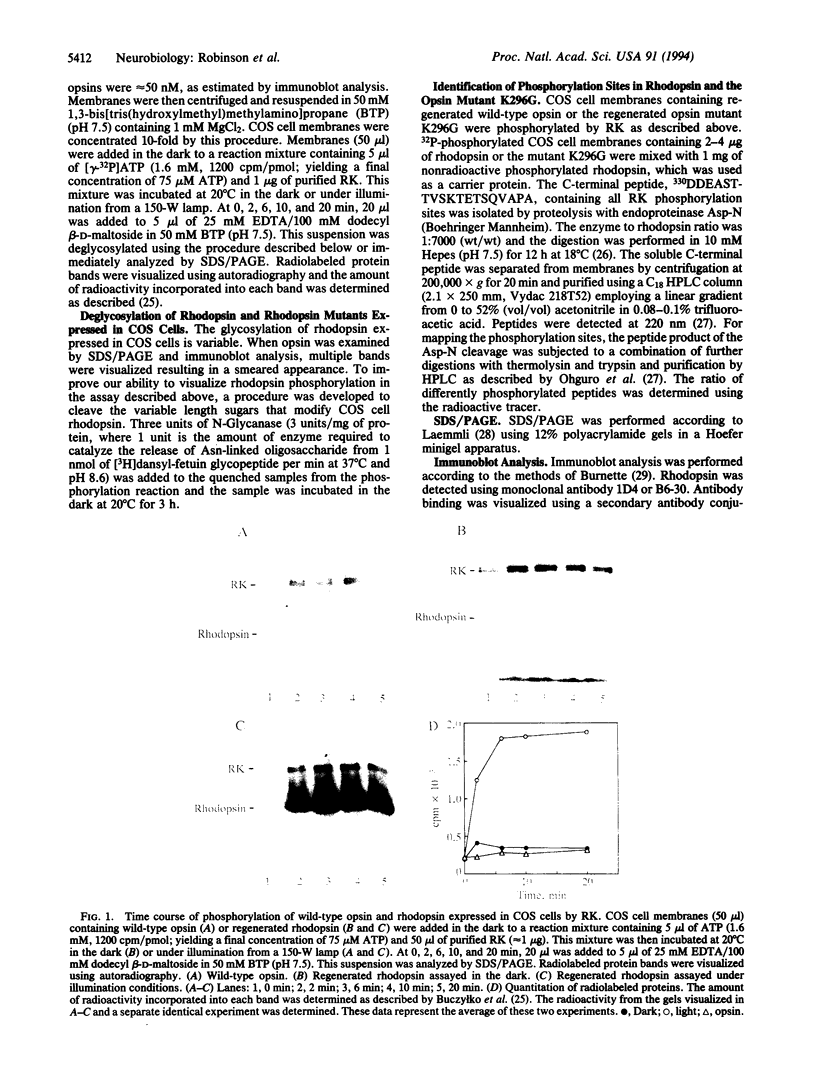
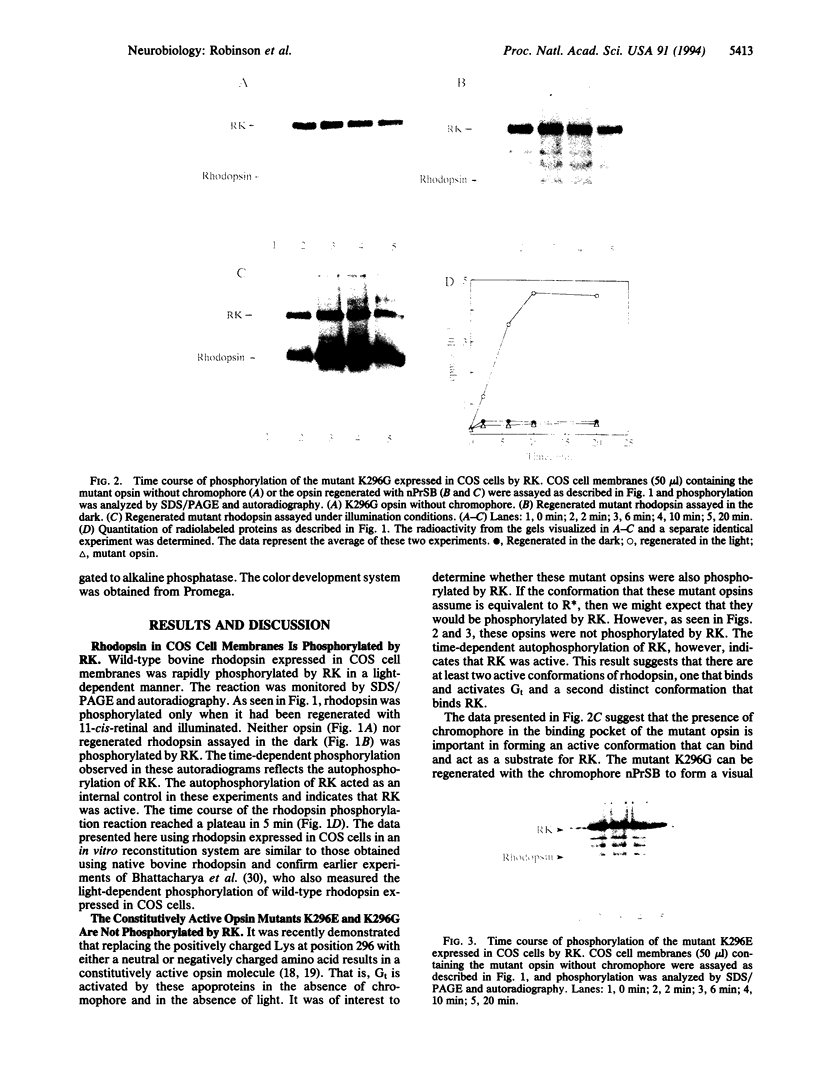
More than 70 mutations in the gene encoding the visual pigment rhodopsin have been identified in patients with autosomal dominant retinitis pigmentosa. Most of these mutations are thought to interfere with proper folding of the membrane protein. However, families with a severe phenotype of retinitis pigmentosa have been identified and shown to carry a mutation at the site of chromophore attachment, Lys-296. This mutation disrupts the inactive conformation of opsin and results in a constitutively active protein that can activate the rod-specific GTP-binding protein, transducin, in the absence of light and in the absence of the chromophore 11-cis-retinal. It has been suggested that this mutant opsin molecule may cause rod degeneration by depletion of the components used to inactivate rhodopsin, such as rhodopsin kinase. In this work we test this idea by determining whether two constitutively active opsin mutants are phosphorylated by rhodopsin kinase. We found that opsin mutants where Lys-296 is replaced either by Glu (K296E) or by Gly (K296G) are not substrates of rhodopsin kinase in the absence of chromophore. However, when K296G is regenerated with a Schiff base complex of 11-cis-retinal and n-propylamine and exposed to illumination, phosphorylation of opsin occurs. These experiments suggest that in the rod photoreceptors of patients with retinitis pigmentosa carrying a mutation at Lys-296, there is persistent activation of the GTP-binding protein-mediated cascade. This may result in a situation that mimics long-term exposure to continuous illumination and results in the degeneration of photoreceptors.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baldwin J. M. The probable arrangement of the helices in G protein-coupled receptors. EMBO J. 1993 Apr;12(4):1693–1703. doi: 10.1002/j.1460-2075.1993.tb05814.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhattacharya S., Ridge K. D., Knox B. E., Khorana H. G. Light-stable rhodopsin. I. A rhodopsin analog reconstituted with a nonisomerizable 11-cis retinal derivative. J Biol Chem. 1992 Apr 5;267(10):6763–6769. [PubMed] [Google Scholar]
- Birnbaumer L., Abramowitz J., Brown A. M. Receptor-effector coupling by G proteins. Biochim Biophys Acta. 1990 May 7;1031(2):163–224. doi: 10.1016/0304-4157(90)90007-y. [DOI] [PubMed] [Google Scholar]
- Bownds D. Site of attachment of retinal in rhodopsin. Nature. 1967 Dec 23;216(5121):1178–1181. doi: 10.1038/2161178a0. [DOI] [PubMed] [Google Scholar]
- Buczyłko J., Gutmann C., Palczewski K. Regulation of rhodopsin kinase by autophosphorylation. Proc Natl Acad Sci U S A. 1991 Mar 15;88(6):2568–2572. doi: 10.1073/pnas.88.6.2568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burnette W. N. "Western blotting": electrophoretic transfer of proteins from sodium dodecyl sulfate--polyacrylamide gels to unmodified nitrocellulose and radiographic detection with antibody and radioiodinated protein A. Anal Biochem. 1981 Apr;112(2):195–203. doi: 10.1016/0003-2697(81)90281-5. [DOI] [PubMed] [Google Scholar]
- Cohen G. B., Yang T., Robinson P. R., Oprian D. D. Constitutive activation of opsin: influence of charge at position 134 and size at position 296. Biochemistry. 1993 Jun 15;32(23):6111–6115. doi: 10.1021/bi00074a024. [DOI] [PubMed] [Google Scholar]
- Dohlman H. G., Thorner J., Caron M. G., Lefkowitz R. J. Model systems for the study of seven-transmembrane-segment receptors. Annu Rev Biochem. 1991;60:653–688. doi: 10.1146/annurev.bi.60.070191.003253. [DOI] [PubMed] [Google Scholar]
- Dryja T. P., Berson E. L., Rao V. R., Oprian D. D. Heterozygous missense mutation in the rhodopsin gene as a cause of congenital stationary night blindness. Nat Genet. 1993 Jul;4(3):280–283. doi: 10.1038/ng0793-280. [DOI] [PubMed] [Google Scholar]
- Fahmy K., Jäger F., Beck M., Zvyaga T. A., Sakmar T. P., Siebert F. Protonation states of membrane-embedded carboxylic acid groups in rhodopsin and metarhodopsin II: a Fourier-transform infrared spectroscopy study of site-directed mutants. Proc Natl Acad Sci U S A. 1993 Nov 1;90(21):10206–10210. doi: 10.1073/pnas.90.21.10206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fain G. L., Lisman J. E. Photoreceptor degeneration in vitamin A deprivation and retinitis pigmentosa: the equivalent light hypothesis. Exp Eye Res. 1993 Sep;57(3):335–340. doi: 10.1006/exer.1993.1132. [DOI] [PubMed] [Google Scholar]
- Farrar G. J., Kenna P., Redmond R., McWilliam P., Bradley D. G., Humphries M. M., Sharp E. M., Inglehearn C. F., Bashir R., Jay M. Autosomal dominant retinitis pigmentosa: absence of the rhodopsin proline----histidine substitution (codon 23) in pedigrees from Europe. Am J Hum Genet. 1990 Dec;47(6):941–945. [PMC free article] [PubMed] [Google Scholar]
- Ferretti L., Karnik S. S., Khorana H. G., Nassal M., Oprian D. D. Total synthesis of a gene for bovine rhodopsin. Proc Natl Acad Sci U S A. 1986 Feb;83(3):599–603. doi: 10.1073/pnas.83.3.599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ganter U. M., Longstaff C., Pajares M. A., Rando R. R., Siebert F. Fourier transform infrared studies of active-site-methylated rhodopsin. Implications for chromophore-protein interaction, transducin activation, and the reaction pathway. Biophys J. 1991 Mar;59(3):640–644. doi: 10.1016/S0006-3495(91)82279-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hargrave P. A., McDowell J. H. Rhodopsin and phototransduction: a model system for G protein-linked receptors. FASEB J. 1992 Mar;6(6):2323–2331. doi: 10.1096/fasebj.6.6.1544542. [DOI] [PubMed] [Google Scholar]
- Hofmann K. P., Pulvermüller A., Buczyłko J., Van Hooser P., Palczewski K. The role of arrestin and retinoids in the regeneration pathway of rhodopsin. J Biol Chem. 1992 Aug 5;267(22):15701–15706. [PubMed] [Google Scholar]
- Hubbard R., Kropf A. THE ACTION OF LIGHT ON RHODOPSIN. Proc Natl Acad Sci U S A. 1958 Feb;44(2):130–139. doi: 10.1073/pnas.44.2.130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khorana H. G. Rhodopsin, photoreceptor of the rod cell. An emerging pattern for structure and function. J Biol Chem. 1992 Jan 5;267(1):1–4. [PubMed] [Google Scholar]
- Kühn H. Light-regulated binding of rhodopsin kinase and other proteins to cattle photoreceptor membranes. Biochemistry. 1978 Oct 17;17(21):4389–4395. doi: 10.1021/bi00614a006. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lefkowitz R. J. G-protein-coupled receptors. Turned on to ill effect. Nature. 1993 Oct 14;365(6447):603–604. doi: 10.1038/365603a0. [DOI] [PubMed] [Google Scholar]
- Longstaff C., Calhoon R. D., Rando R. R. Deprotonation of the Schiff base of rhodopsin is obligate in the activation of the G protein. Proc Natl Acad Sci U S A. 1986 Jun;83(12):4209–4213. doi: 10.1073/pnas.83.12.4209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nathans J. Rhodopsin: structure, function, and genetics. Biochemistry. 1992 Jun 2;31(21):4923–4931. doi: 10.1021/bi00136a001. [DOI] [PubMed] [Google Scholar]
- Ohguro H., Palczewski K., Ericsson L. H., Walsh K. A., Johnson R. S. Sequential phosphorylation of rhodopsin at multiple sites. Biochemistry. 1993 Jun 1;32(21):5718–5724. doi: 10.1021/bi00072a030. [DOI] [PubMed] [Google Scholar]
- Oprian D. D., Molday R. S., Kaufman R. J., Khorana H. G. Expression of a synthetic bovine rhodopsin gene in monkey kidney cells. Proc Natl Acad Sci U S A. 1987 Dec;84(24):8874–8878. doi: 10.1073/pnas.84.24.8874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oprian D. D. The ligand-binding domain of rhodopsin and other G protein-linked receptors. J Bioenerg Biomembr. 1992 Apr;24(2):211–217. doi: 10.1007/BF00762679. [DOI] [PubMed] [Google Scholar]
- Palczewski K., Buczyłko J., Kaplan M. W., Polans A. S., Crabb J. W. Mechanism of rhodopsin kinase activation. J Biol Chem. 1991 Jul 15;266(20):12949–12955. [PubMed] [Google Scholar]
- Palczewski K., Pulvermüller A., Buczyłko J., Hofmann K. P. Phosphorylated rhodopsin and heparin induce similar conformational changes in arrestin. J Biol Chem. 1991 Oct 5;266(28):18649–18654. [PubMed] [Google Scholar]
- Pulvermüller A., Palczewski K., Hofmann K. P. Interaction between photoactivated rhodopsin and its kinase: stability and kinetics of complex formation. Biochemistry. 1993 Dec 28;32(51):14082–14088. doi: 10.1021/bi00214a002. [DOI] [PubMed] [Google Scholar]
- Rao V. R., Cohen G. B., Oprian D. D. Rhodopsin mutation G90D and a molecular mechanism for congenital night blindness. Nature. 1994 Feb 17;367(6464):639–642. doi: 10.1038/367639a0. [DOI] [PubMed] [Google Scholar]
- Ren Q., Kurose H., Lefkowitz R. J., Cotecchia S. Constitutively active mutants of the alpha 2-adrenergic receptor. J Biol Chem. 1993 Aug 5;268(22):16483–16487. [PubMed] [Google Scholar]
- Robinson P. R., Cohen G. B., Zhukovsky E. A., Oprian D. D. Constitutively active mutants of rhodopsin. Neuron. 1992 Oct;9(4):719–725. doi: 10.1016/0896-6273(92)90034-b. [DOI] [PubMed] [Google Scholar]
- Strosberg A. D. Structure/function relationship of proteins belonging to the family of receptors coupled to GTP-binding proteins. Eur J Biochem. 1991 Feb 26;196(1):1–10. doi: 10.1111/j.1432-1033.1991.tb15778.x. [DOI] [PubMed] [Google Scholar]
- Zhukovsky E. A., Robinson P. R., Oprian D. D. Transducin activation by rhodopsin without a covalent bond to the 11-cis-retinal chromophore. Science. 1991 Feb 1;251(4993):558–560. doi: 10.1126/science.1990431. [DOI] [PubMed] [Google Scholar]
- Zhukovsky E. A., Robinson P. R., Oprian D. D. Transducin activation by rhodopsin without a covalent bond to the 11-cis-retinal chromophore. Science. 1991 Feb 1;251(4993):558–560. doi: 10.1126/science.1990431. [DOI] [PubMed] [Google Scholar]