Abstract
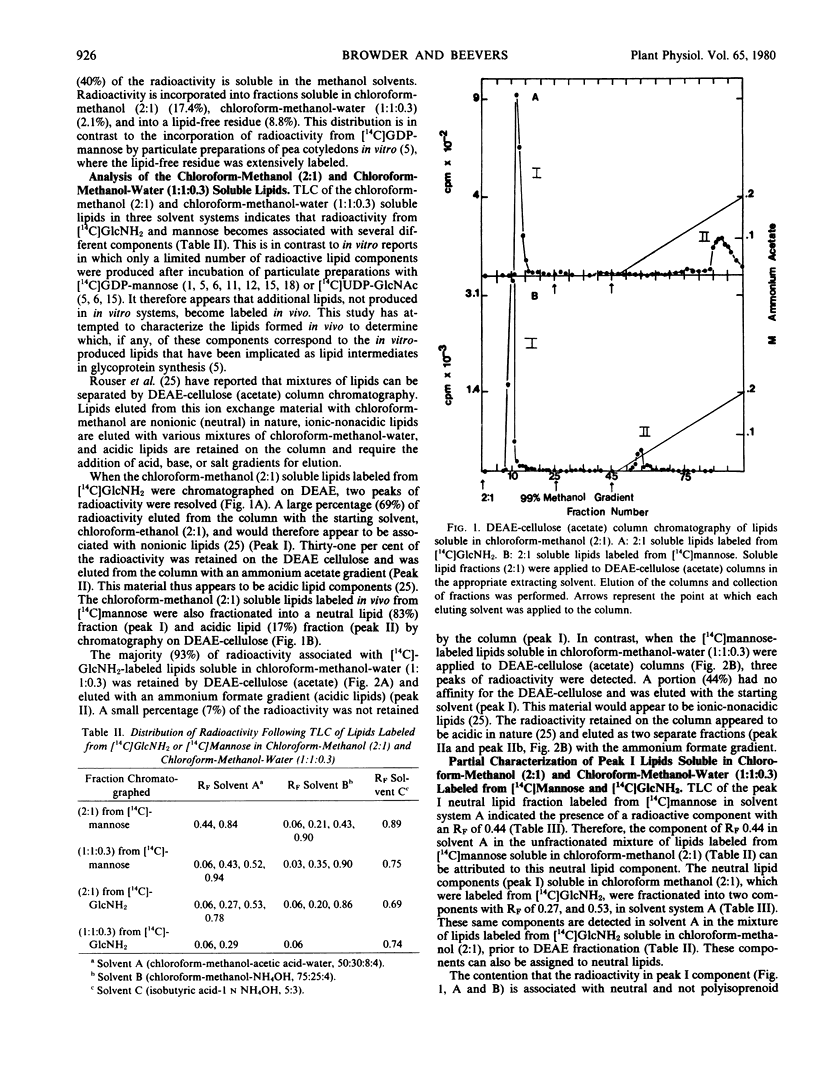
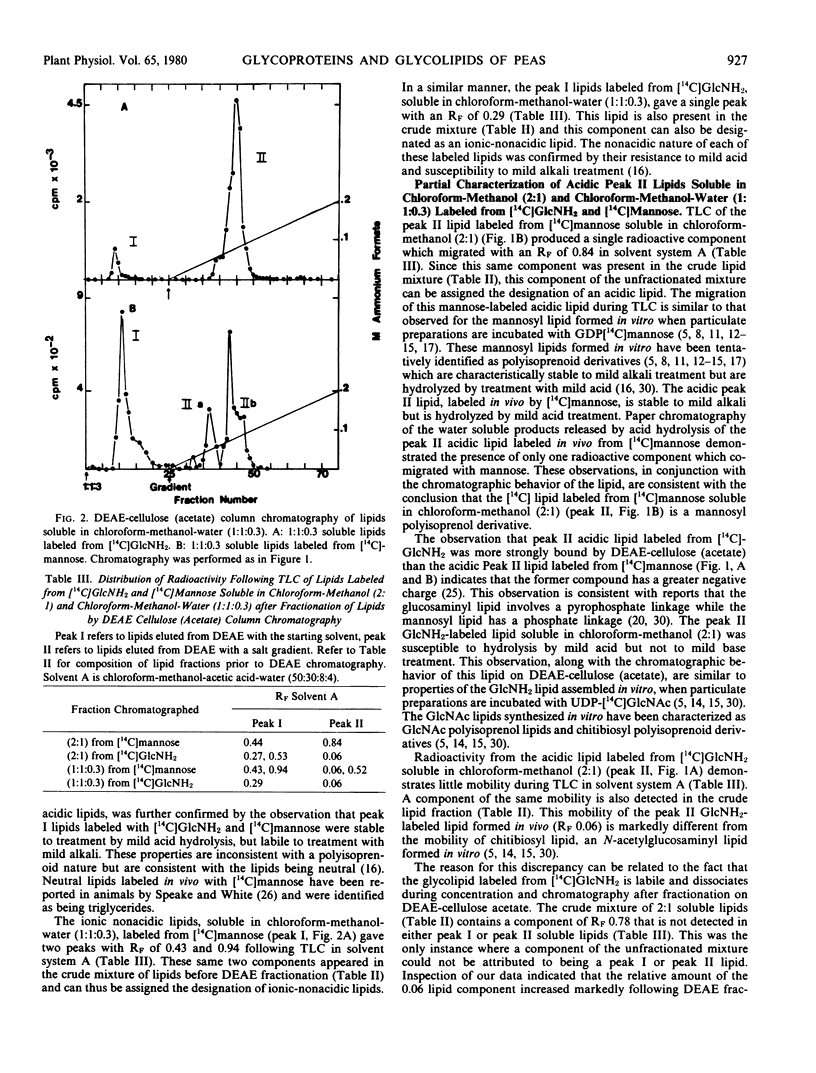
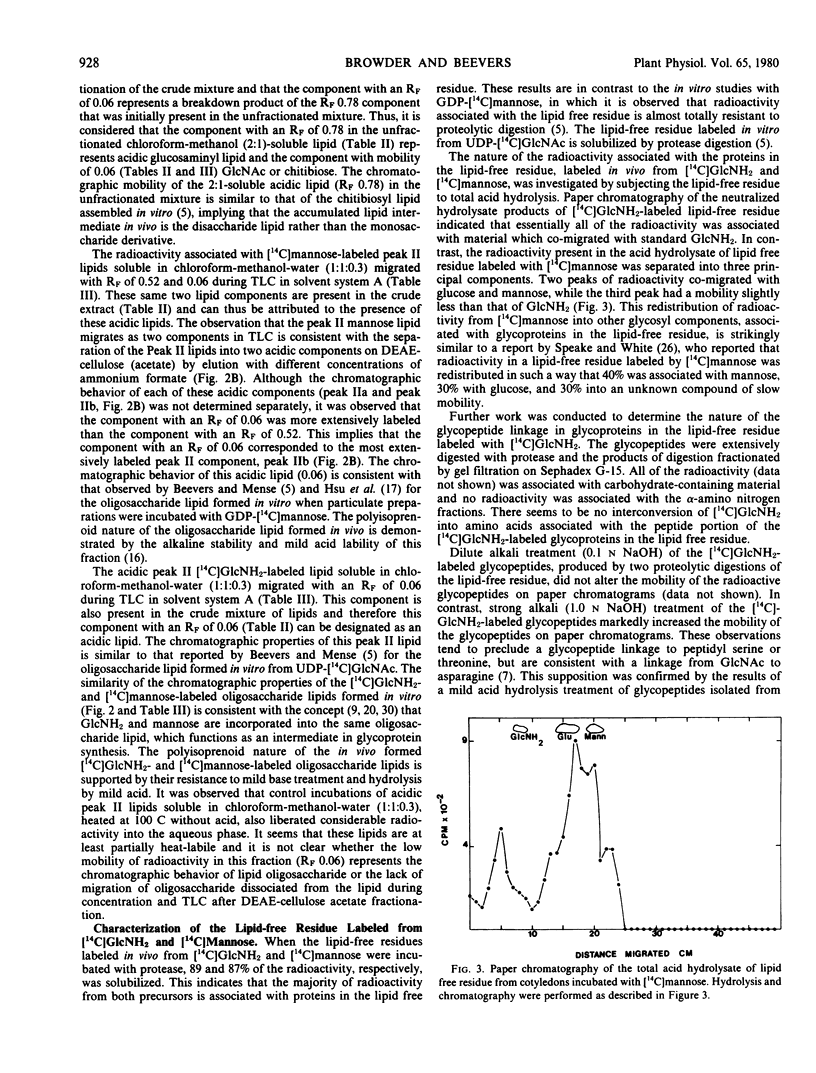
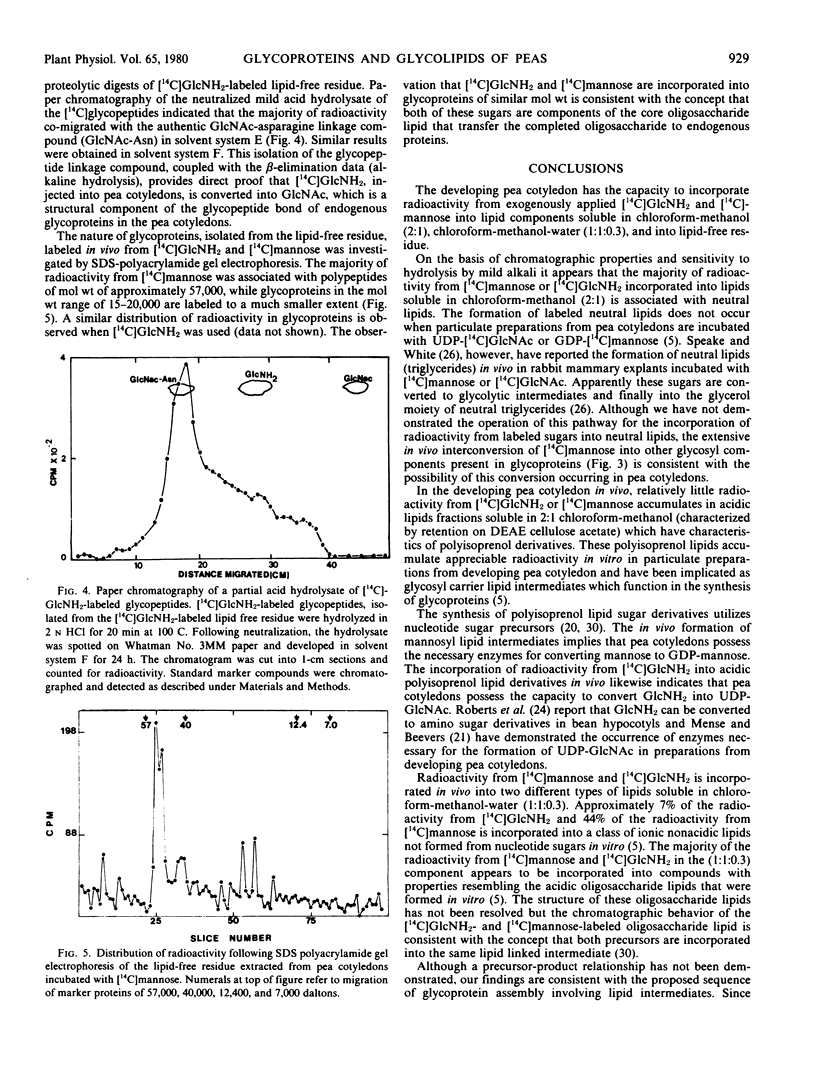
Developing pea cotyledons incorporate radioactivity in vivo from [14C]glucosamine and [14C]mannose into glycolipids and glycoproteins. Several different lipid components are labeled including neutral, ionicnonacidic, and acidic lipids. The acidic lipids labeled in vivo appear similar to the polyisoprenoid lipid intermediates formed in vitro in pea cotyledons. Radioactivity from [14C]glucosamine and [14C]mannose is also incorporated into glycopeptides. Considerable redistribution of [14C]mannose into other glycosyl components found in endogenous glycoproteins is observed. An N-acetylglucosamine to asparagine glycopeptide linkage has been isolated from [14C]glucosamine-labeled glycoproteins.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Barr R. M., De Luca L. M. The in vivo incorporation of mannose, retinol and mevalonic acid into phospholipids of hamster liver. Biochem Biophys Res Commun. 1974 Sep 9;60(1):355–363. doi: 10.1016/0006-291x(74)90212-5. [DOI] [PubMed] [Google Scholar]
- Basha S. M., Beevers L. Glycoprotein Metabolism in the Cotyledons of Pisum sativum during Development and Germination. Plant Physiol. 1976 Jan;57(1):93–97. doi: 10.1104/pp.57.1.93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beevers L., Mense R. M. Glycoprotein Biosynthesis in Cotyledons of Pisum sativum L: Involvement of Lipid-linked Intermediates. Plant Physiol. 1977 Nov;60(5):703–708. doi: 10.1104/pp.60.5.703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brett C. T., Leloir L. F. Dolichyl monophosphate and its sugar derivatives in plants. Biochem J. 1977 Jan 1;161(1):93–101. doi: 10.1042/bj1610093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Browder S. K., Beevers L. Characterization of the glycopeptide bond in legumin from Pisum sativum L. FEBS Lett. 1978 May 1;89(1):145–148. doi: 10.1016/0014-5793(78)80541-9. [DOI] [PubMed] [Google Scholar]
- Chambers J., Elbein A. D. Biosynthesis and characterization of lipid-linked sugars and glycoproteins in aorta. J Biol Chem. 1975 Sep 10;250(17):6904–6915. [PubMed] [Google Scholar]
- Chambers J., Forsee W. T., Elbein A. D. Enzymatic transfer of mannose from mannosyl-phosphoryl-polyprenol to lipid-linked oligosaccharides by pig aorta. J Biol Chem. 1977 Apr 25;252(8):2498–2506. [PubMed] [Google Scholar]
- Delmer D. P., Kulow C., Ericson M. C. Glycoprotein Synthesis in Plants: II. Structure of the Mannolipid Intermediate. Plant Physiol. 1978 Jan;61(1):25–29. doi: 10.1104/pp.61.1.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ericson M. C., Delmer D. P. Glycoprotein synthesis in plants: I. Role of lipid intermediates. Plant Physiol. 1977 Mar;59(3):341–347. doi: 10.1104/pp.59.3.341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forsee W. T., Elbein A. D. Biosynthesis of acidic glycolipids in cotton fibers. Possible intermediates in cell wall synthesis. Biochem Biophys Res Commun. 1972 Nov 15;49(4):930–939. doi: 10.1016/0006-291x(72)90301-4. [DOI] [PubMed] [Google Scholar]
- Forsee W. T., Elbein A. D. Biosynthesis of mannosyl- and glucosyl-phosphoryl-polyprenols in cotton fibers. J Biol Chem. 1973 Apr 25;248(8):2858–2867. [PubMed] [Google Scholar]
- Forsee W. T., Elbein A. D. Glycoprotein biosynthesis in plants. Demonstration of lipid-linked oligosaccharides of mannose and N-acetylglucosamine. J Biol Chem. 1975 Dec 25;250(24):9283–9293. [PubMed] [Google Scholar]
- Forsee W. T., Valkovich G., Elbein A. D. Glycoprotein biosynthesis in plants. Formation of lipid-linked oligosaccharides of mannose and N-acetylglucosamine by mung bean seedlings. Arch Biochem Biophys. 1976 Jun;174(2):469–479. doi: 10.1016/0003-9861(76)90375-1. [DOI] [PubMed] [Google Scholar]
- Hsu A. F., Baynes J. W., Heath E. C. The role of a dolichol-oligosaccharide as an intermediate in glycoprotein biosynthesis. Proc Natl Acad Sci U S A. 1974 Jun;71(6):2391–2395. doi: 10.1073/pnas.71.6.2391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Lehle L., Fartaczek F., Tanner W., Kauss H. Formation of polyprenol-linked mono- and oligosaccharides in Phaseolus aureus. Arch Biochem Biophys. 1976 Aug;175(2):419–426. doi: 10.1016/0003-9861(76)90529-4. [DOI] [PubMed] [Google Scholar]
- Lucas J. J., Waechter C. J. Polyisoprenoid glycolipids involved in glycoprotein biosynthesis. Mol Cell Biochem. 1976 Apr 28;11(2):67–78. doi: 10.1007/BF01792788. [DOI] [PubMed] [Google Scholar]
- Nagahashi J., Beevers L. Subcellular Localization of Glycosyl Transferases Involved in Glycoprotein Biosynthesis in the Cotyledons of Pisum sativum L. Plant Physiol. 1978 Mar;61(3):451–459. doi: 10.1104/pp.61.3.451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagahashi J., Browder S. K., Beevers L. Glycosylation of pea cotyledon membranes. Plant Physiol. 1980 Apr;65(4):648–657. doi: 10.1104/pp.65.4.648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts R. M., Pollard W. E. The Incorporation of d-Glucosamine into Glycolipids and Glycoproteins of Membrane Preparations from Phaseolus aureus Hypocotyls. Plant Physiol. 1975 Mar;55(3):431–436. doi: 10.1104/pp.55.3.431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Speake B. K., White D. A. The formation of lipid-linked sugars as intermediates in glycoprotein synthesis in rabbit mammary gland. Biochem J. 1978 Feb 15;170(2):273–283. doi: 10.1042/bj1700273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spiro M. J., Spiro R. G., Bhoyroo V. D. Lipid-saccharide intermediates in glycoprotein biosynthesis. I. Formation of an oligosaccharide-lipid by thyroid slices and evaluation of its role in protein glycosylation. J Biol Chem. 1976 Oct 25;251(20):6400–6408. [PubMed] [Google Scholar]
- Struck D. K., Lennarz W. J. Utilization of exogenous GDP-mannose for the synthesis of mannose-containing lipids and glycoproteins by oviduct cells. J Biol Chem. 1976 Apr 25;251(8):2511–2519. [PubMed] [Google Scholar]
- Studier F. W. Analysis of bacteriophage T7 early RNAs and proteins on slab gels. J Mol Biol. 1973 Sep 15;79(2):237–248. doi: 10.1016/0022-2836(73)90003-x. [DOI] [PubMed] [Google Scholar]
- Waechter C. J., Lennarz W. J. The role of polyprenol-linked sugars in glycoprotein synthesis. Annu Rev Biochem. 1976;45:95–112. doi: 10.1146/annurev.bi.45.070176.000523. [DOI] [PubMed] [Google Scholar]


