Abstract
The membrane bounding the vacuole of plant cells contains two electrogenic proton pumps. These are the vacuolar H(+)-ATPase (EC 3.6.1.3), an enzyme common to all eukaryotes, and a vacuolar H(+)-translocating pyrophosphatase (EC 3.6.1.1), which is ubiquitous in plants but otherwise known in only a few phototrophic bacteria. Although the substrate-binding subunit of the vacuolar H(+)-pyrophosphatase has been identified and purified and cDNAs encoding it have been isolated and characterized, the minimal unit competent in pyrophosphate (PPi)-energized H+ translocation is not known. Here we address this question and show that heterologous expression of the cDNA (AVP) encoding the substrate-binding subunit of the vacuolar H(+)-pyrophosphatase from the vascular plant Arabidopsis thaliana in the yeast Saccharomyces cerevisiae results in the production of vacuolarly localized functional enzyme active in PPi-dependent H+ translocation. Since the heterologously expressed pump is indistinguishable from the native plant enzyme with respect to PPi hydrolysis, H+ translocation, activation by potassium, and selective inhibition by calcium and 1,1-diphosphonates, it is concluded that all of the known catalytic functions of the enzyme map to the one subunit encoded by AVP.
Full text
PDF


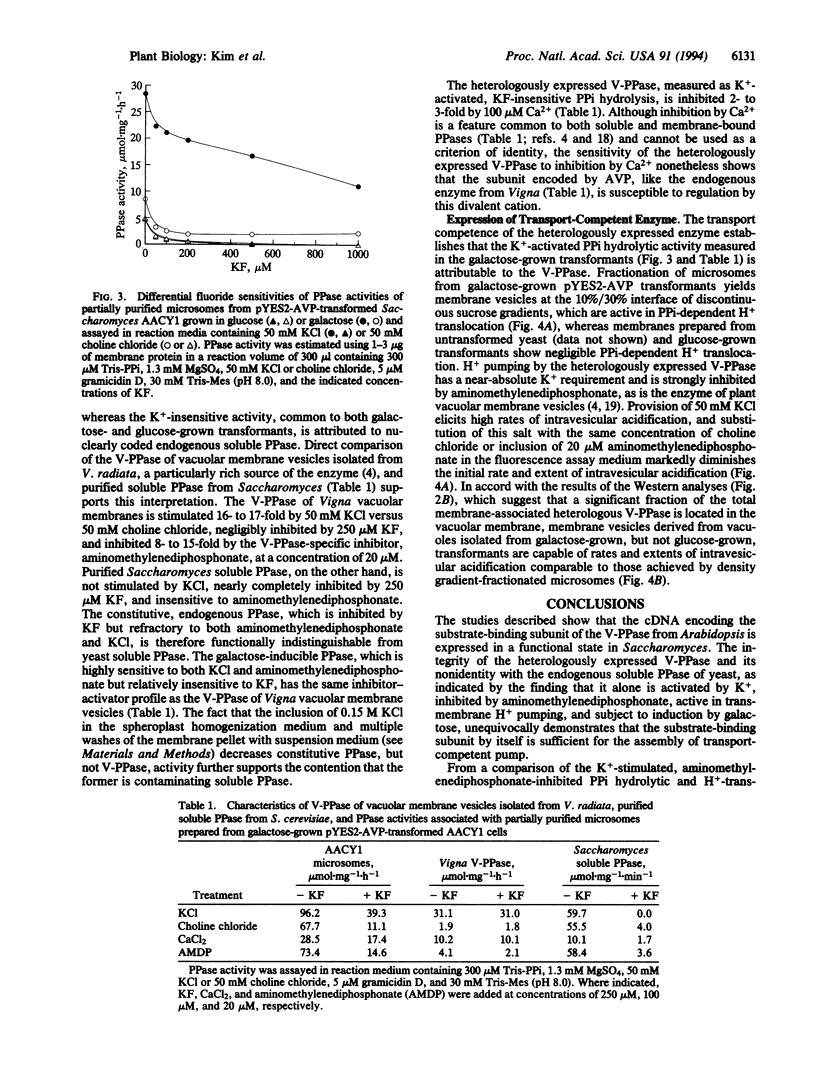
Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Cooper A., Bussey H. Yeast Kex1p is a Golgi-associated membrane protein: deletions in a cytoplasmic targeting domain result in mislocalization to the vacuolar membrane. J Cell Biol. 1992 Dec;119(6):1459–1468. doi: 10.1083/jcb.119.6.1459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kane P. M., Kuehn M. C., Howald-Stevenson I., Stevens T. H. Assembly and targeting of peripheral and integral membrane subunits of the yeast vacuolar H(+)-ATPase. J Biol Chem. 1992 Jan 5;267(1):447–454. [PubMed] [Google Scholar]
- Nelson N. Evolution of organellar proton-ATPases. Biochim Biophys Acta. 1992 May 20;1100(2):109–124. doi: 10.1016/0005-2728(92)90072-a. [DOI] [PubMed] [Google Scholar]
- Parry R. V., Turner J. C., Rea P. A. High purity preparations of higher plant vacuolar H+-ATPase reveal additional subunits. Revised subunit composition. J Biol Chem. 1989 Nov 25;264(33):20025–20032. [PubMed] [Google Scholar]
- Peterson G. L. A simplification of the protein assay method of Lowry et al. which is more generally applicable. Anal Biochem. 1977 Dec;83(2):346–356. doi: 10.1016/0003-2697(77)90043-4. [DOI] [PubMed] [Google Scholar]
- Rea P. A., Britten C. J., Jennings I. R., Calvert C. M., Skiera L. A., Leigh R. A., Sanders D. Regulation of vacuolar h-pyrophosphatase by free calcium : a reaction kinetic analysis. Plant Physiol. 1992 Dec;100(4):1706–1715. doi: 10.1104/pp.100.4.1706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rea P. A., Britten C. J., Sarafian V. Common identity of substrate binding subunit of vacuolar h-translocating inorganic pyrophosphatase of higher plant cells. Plant Physiol. 1992 Oct;100(2):723–732. doi: 10.1104/pp.100.2.723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rea P. A., Kim Y., Sarafian V., Poole R. J., Davies J. M., Sanders D. Vacuolar H(+)-translocating pyrophosphatases: a new category of ion translocase. Trends Biochem Sci. 1992 Sep;17(9):348–353. doi: 10.1016/0968-0004(92)90313-x. [DOI] [PubMed] [Google Scholar]
- Roberts C. J., Nothwehr S. F., Stevens T. H. Membrane protein sorting in the yeast secretory pathway: evidence that the vacuole may be the default compartment. J Cell Biol. 1992 Oct;119(1):69–83. doi: 10.1083/jcb.119.1.69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts C. J., Raymond C. K., Yamashiro C. T., Stevens T. H. Methods for studying the yeast vacuole. Methods Enzymol. 1991;194:644–661. doi: 10.1016/0076-6879(91)94047-g. [DOI] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sarafian V., Kim Y., Poole R. J., Rea P. A. Molecular cloning and sequence of cDNA encoding the pyrophosphate-energized vacuolar membrane proton pump of Arabidopsis thaliana. Proc Natl Acad Sci U S A. 1992 Mar 1;89(5):1775–1779. doi: 10.1073/pnas.89.5.1775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schiestl R. H., Gietz R. D. High efficiency transformation of intact yeast cells using single stranded nucleic acids as a carrier. Curr Genet. 1989 Dec;16(5-6):339–346. doi: 10.1007/BF00340712. [DOI] [PubMed] [Google Scholar]
- Sze H., Ward J. M., Lai S. Vacuolar H(+)-translocating ATPases from plants: structure, function, and isoforms. J Bioenerg Biomembr. 1992 Aug;24(4):371–381. doi: 10.1007/BF00762530. [DOI] [PubMed] [Google Scholar]
- Wilcox C. A., Redding K., Wright R., Fuller R. S. Mutation of a tyrosine localization signal in the cytosolic tail of yeast Kex2 protease disrupts Golgi retention and results in default transport to the vacuole. Mol Biol Cell. 1992 Dec;3(12):1353–1371. doi: 10.1091/mbc.3.12.1353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhen R. G., Baykov A. A., Bakuleva N. P., Rea P. A. Aminomethylenediphosphonate: A Potent Type-Specific Inhibitor of Both Plant and Phototrophic Bacterial H+-Pyrophosphatases. Plant Physiol. 1994 Jan;104(1):153–159. doi: 10.1104/pp.104.1.153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zitomer R. S., Montgomery D. L., Nichols D. L., Hall B. D. Transcriptional regulation of the yeast cytochrome c gene. Proc Natl Acad Sci U S A. 1979 Aug;76(8):3627–3631. doi: 10.1073/pnas.76.8.3627. [DOI] [PMC free article] [PubMed] [Google Scholar]