Abstract
B-1 cells can be differentiated from B-2 cells because they are predominantly located in the peritoneal and pleural cavities and have distinct phenotypic patterns and activation properties. A mononuclear phagocyte derived from B-1 cells (B-1CDP) has been described. As the B-1CDP cells migrate to inflammatory/infectious sites and exhibit phagocytic capacity, the microbicidal ability of these cells was investigated using the Leishmania major infection model in vitro. The data obtained in this study demonstrate that B-1CDP cells are more susceptible to infection than peritoneal macrophages, since B-1CDP cells have a higher number of intracellular amastigotes forms and consequently release a larger number of promastigotes. Exacerbated infection by L. major required lipid bodies/PGE2 and IL-10 by B-1CDP cells. Both infection and the production of IL-10 were decreased when PGE2 production was blocked by NSAIDs. The involvement of IL-10 in this mechanism was confirmed, since B-1CDP cells from IL-10 KO mice are more competent to control L. major infection than cells from wild type mice. These findings further characterize the B-1CDP cells as an important mononuclear phagocyte that plays a previously unrecognized role in host responses to L. major infection, most likely via PGE2-driven production of IL-10.
Introduction
Macrophages represent distinct cells with phagocytic activity distributed through all tissues. Originally tissue-resident macrophages were assumed to be derived from circulating monocytes [1] differentiated from bone marrow progenitors. Recently, a change in this dogma was provided with conclusive evidences for the existence of a monocyte-independent differentiation pathway of resident macrophages, leading to shift in the paradigm of this model [2,3].
Recently, other studies have suggested that other cell lines could originate phagocytic macrophages [4,5]. These studies are based on previous experiments that demonstrated that B-1 cells present in mice and humans could differentiate into cells with characteristics similar to macrophages. Borrello and Phipps demonstrated that B-1 cells from the peritoneal cavity of mice differentiate into a phagocytic cell similar to macrophage-like cells [6]. Differentiation decreases immunoglobulin M expression but the expression of rearranged VH11 or VH12, heavy chain genes persist [7]. Graf et al demonstrated that B/macrophage cells express COX-1, and up-regulate COX-2 expression and prostaglandin E2 production in response to pro-inflammatory signals [8].
Several studies investigated the origin [9–12], immunological properties [9,13–18] and the involvement these cells in inflammatory reactions [15,19–28]. Despite the great interest on this cell type, little is known about B-1 cells and mainly on B-1 cell derived phagocytes (B-1CDP) in models of infections by microorganisms [7,21,29,30].
L. major is a protozoan parasite transmitted by sandflies of the genus Lutzomyia that inject the promastigote form into the dermis of the host. Once injected, the parasite is rapidly enclosed by phagocytic cells and transforms into the replicative intracellular amastigote form [31]. In susceptible hosts, such as BALB/c mice, L. major elicits a Th2 immune response and induces a progressive infection. In susceptible hosts, macrophages produce anti-inflammatory factors, such IL-10, TGF-β and PGE2, which act in favor of the protozoan [32].
Based on these data, we decided to investigate the interaction of B-1CDP cells from BALB/c mice with L. major to elucidate the possible influence of these cells on the progression of infection in vitro.
Material and Methods
Ethics statement
This study was carried out in strict accordance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Institutes of Health (USA). The protocol was approved by the Committee on the Ethics of Animal Experiments of the Health Science Center of the Federal University of Rio de Janeiro (CEUA-CCS, Permit Number: IMPPG 038-05/16) and all efforts were made to minimize suffering.
Mice and parasite
BALB/c mice of both sexes, aging 6–8 wk, were from Oswaldo Cruz Institute Animal Care Facility (Fiocruz, Rio de Janeiro, Brazil) C57BL/6 IL-10 Knock-out (KO) and C57BL/6 wild-type mice were kindly donated by Professor João Santana Silva from the Department of Pharmacology, School of Medicine, USP, Ribeirão Preto. L. major strain LV39 (MRHO/Sv/59/P) was isolated monthly from footpads of infected BALB/c mice and maintained in vitro as proliferating promastigotes. Parasites were maintained in Schneider medium (Life Technologies) supplemented with 10% FCS, 1% glutamine and 2% human urine.
Cell culture
B-1CDP cells obtained as previously described [33]. Briefly, resident peritoneal cells were collected from peritoneal washouts of BALB/c mice. Cells (2 X 106) were dispensed on 10 cm diameter plastic plates and the cultures incubated ay 37°C in 7% CO2 for 1h. After incubation, the culture supernatants were aspirated to remove non-adherent cells. Adherent monolayers were rinsed with antibiotic-free RPMI-1640 medium (Sigma), contained 15 mM HEPES, 2g of sodium bicarbonate/liter, 1mM L-glutamine and kept in 0,5 ml of antibiotic-free RPMI medium plus 10% fetal bovine serum for 6 days. B1 cells present in the supernatant of these cultures were aspirated, centrifuged, re-suspended in RPMI medium plus 1 0% fetal bovine serum and dispensed on cover slips in the bottom of 6 well plates. After 3 days in culture B-1CDP, adherent to the glass surface were removed from the substrate by ice-cold phosphate-buffered saline. Cells were counted, added (2 X 105) to glass cover slips inserted in 24-well tissue culture plates. Peritoneal macrophages cultures were made as above described using adherent cells from the peritoneal cavity of BALB/c. Peritoneal macrophages were counted, added (2 X 105) to glass cover slips inserted in 24-well tissue culture plates.
Infection
B-1CDP cells and peritoneal macrophages were plated in 24 wells tissue culture plates (Nunc, Roskilde, Denmark) at 2 X 105 cells/well in RPMI medium plus 10% fetal bovine serum. Cells immediately received 1X106 stationary phase L. major promastigote, and were incubated in medium 10% FCS at 37°C. After 4 hours, monolayers were extensively washed with warm HBSS, to remove extracellular parasites. All cultures were done in medium 1% Nutridoma-SP, instead of FCS.
Antibodies, cytokines and inhibitors
B-1CDP cells or peritoneal macrophage monolayers were treated with 1 μg/mL indomethacin, 10 μg/mL Aspirin (Sigma-Aldrich) or 1 μM/mL NS-398 (CaymanChem), or equivalent dosage of solvent (DMSO). Neutralizing anti-TGF-β and normal chicken IgY (R&D System), anti-IL-10 and rat IgG1 isotype control (BioSource Europe, Nivelles), were used at 10 μg/mL.
Assessment of intracellular load of L. major
After 3 days, infected B-1CDP cells and macrophages monolayers were extensively washed, and medium was replaced by 0,5 ml of Schneider medium (Life Technologies), supplemented with 20% FCS and 2% human urine [34]. Monolayers were cultured at 26°C for additional 3 days. Intracellular load of L. major amastigote was estimated by production of proliferating extracellular motile promastigote in Schneider medium [34]. Alternatively, infected B-1CDP cells and peritoneal macrophages were cultured on glass coveslips place inside culture vessels (Corning) at 37°C in 7% CO2. After 4 days, coverslips were washed and stained with May-Grunwald Giemsa (Sigma-Aldrich), and intracellular amastigotes were counted in 100 B-1CDP cells or macrophages. Results are shown as amastigote number per phagocyte, and as percentage of infected macrophages. All results are mean and SE of triplicated cultures.
Lactate Dehydrogenase Assay
In order to evaluate the integrity infected B-1CDP cells and peritoneal macrophages, an aliquot of the supernatant was withdrawn and frozen for determination of LDH release. LDH activity was evaluated by using an assay kit (Doles Reagents, Brazil), which measure the amount of a colored complex derived from the NADH formed by the enzymatic reaction, using a spectrophotometric method (A 490).
Immunofluorescence Microscopy
Infected B-1CDP cells and macrophages were obtained as described above. After different time in culture, cells were fixed for 1 h in 3.5% formalin. After fixation, cells were washed in PBS containing 1% BSA (Sigma) to block free sites and permeabilized by treatment with 0.05% saponin (Sigma) for 30 min. Polyclonal goat antibody anti-mouse IgM-FITC (μ chain specific) (Sigma) (1:1000) and biotin rat antibody anti-mouse F4/80 was diluted in PBS/BSA/saponin and incubated for 1 h at room temperature. After three washes in the same solution, glass cover slips were incubated with anti-rat IgG-FITC (F4/80). After incubation, glass cover slips were washed again and incubated in 1 μg /mL of DAPI. After 10 min of incubation, glass cover slips were washed, mounted in 0.1 M glycerol and sealed with nail varnish. Images were acquired using a Zeiss Observer Z.1 microscope in a 63x objectives. The images were processed with a deconvolution module using Zen software.
Determination of mediators
The concentrations of cytokines in the supernatant obtained from cultures of infected B-1CDP cells or peritoneal macrophages were quantified after 24 hours of incubation by the method of sandwich immunoassay (ELISA) according to methodology recommended by the manufacturer (R&D). The optical density was obtained by reading in a plate spectrophotometer (Versamax Microplates Reader Molecular Devices, USA), with filter of 405 nm. The concentrations of cytokines were calculated from a standard curve of recombinant cytokines. Quantification of PGE2 was obtained by a PGE2 specific EIA kit, according to methodology recommended by the manufacturer (Cayman Chemical, Ann Arbor, MZ).
Lipid bodies
To observe LBs, B-1CDP cells and macrophages were first fixed in 3,5% formalin in PBS for 30 min at room temperature, then stained with Nile red (Sigma) for 10 minutes (RT) at a final concentration of 10 mg/ml [35]. Following brief washes with PBS, and the slides were, stained with DAPI (Sigma). The morphology of fixed cells was observed, and Nile Red LBs were counted by light microscopy with a 100X objective lens in 50 consecutively scanned leukocytes.
Statistical analysis
Statistical analysis was performed in the program GraphPad InStat version 3.01 (San Diego, USA). Data were analyzed by the method of Student’t test. Differences with a p value 0.05 or lower were considered significant.
Results
B-1CDP cells are more susceptible to infection with L. major in comparison with peritoneal macrophages
Macrophages correspond to the main reservoir of Leishmania infection in vivo. We evaluated the susceptibility/resistance to L. major infection of B-1CDP cells in comparison with peritoneal macrophages.
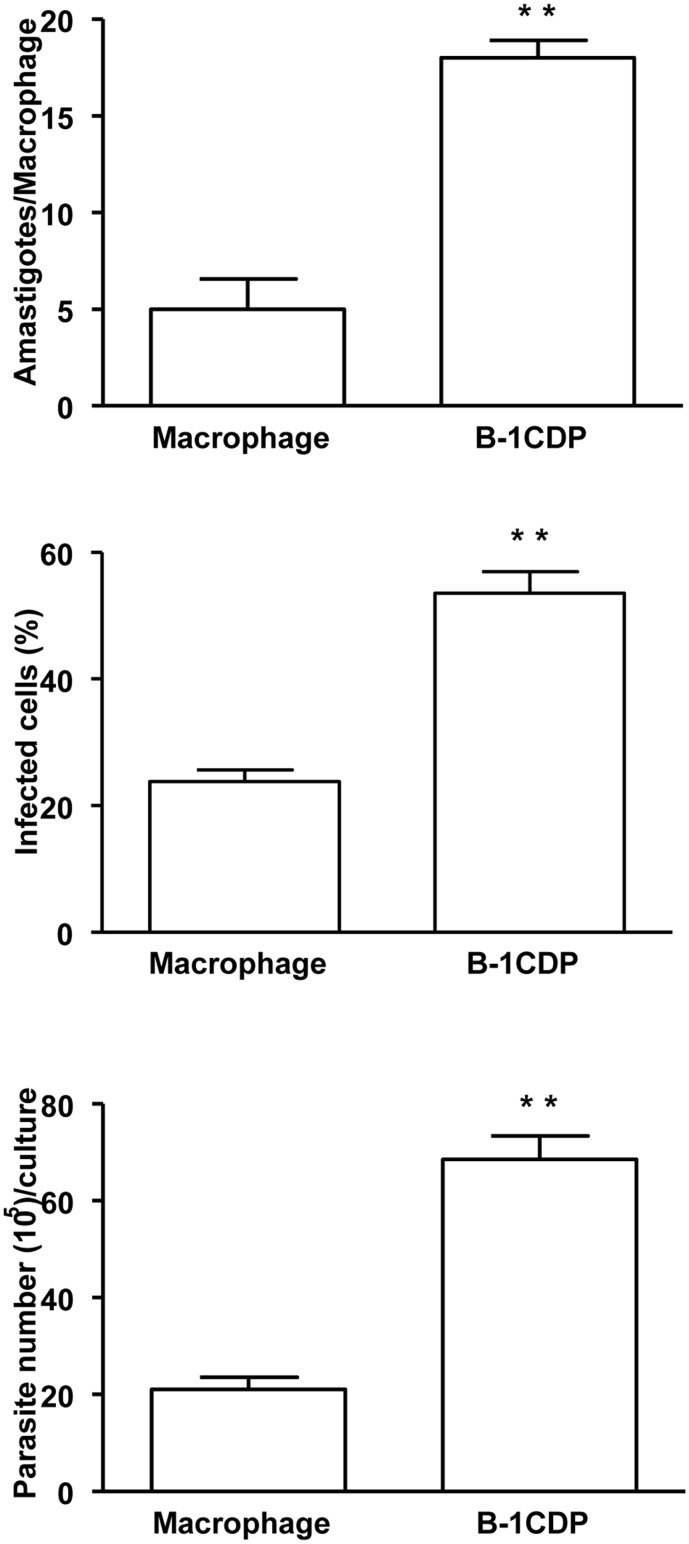
B-1CDP cells and peritoneal macrophages were infected with L. major promastigotes and amastigotes were counted. Our results demonstrated that B-1CDP cells exhibit a greater number of intracellular amastigotes when compared with peritoneal macrophages (Fig 1A and S1 Fig) and were more permissive to infection, since they showed a higher percentage of infected cells (Fig 1B).
Fig 1. B-1CDP cells exhibit a susceptible phenotype to infection with L. major in vitro when compared to peritoneal macrophages.
B-1CDP cells and peritoneal macrophages were cultured (105/ml) and infected with metacyclic promastigotes of L. major. After 24 hours, the cell culture was washed and phagocytes were cultured for another 3 days with DMEM supplemented with 10% FBS at 37°C. After this period, cells were stained and amastigotes inside the phagocytes were counted under the light microscope (A) and set the percentage of infected cells (B). To quantify promastigotes forms in the supernatants, the cells were infected with L. major. After 24 hours of infection, cells were washed and cultured in Schneider medium for 5 days at a temperature of 27°C. After this period, the promastigotes were quantified in the supernatant of the cultures of infected phagocytes (C). All cultures were performed in triplicate and bars show the mean + SD. Statistical analysis were performed by T-Test from representative results of three similar experiments. **p ≤ 0.05.
Furthermore, after 5 days of culture we observed a greater release of motile promastigotes forms of L. major in the supernatant of infected B-1CDP cells when compared to promastigotes present in the supernatant of peritoneal macrophages (Fig 1C). Despite of these differences, the parasite burden at 4 hours post-infection was identical for both cell types (S2 Fig).
Detection of IL-10 in culture supernatants from infected B-1CDP cells controls L. major growth
It is known that B-1 cells produce IL-10 and use this cytokine as an autocrine growth factor [36]. Also this cytokine is an important regulator of macrophages [37]. Since B-1CDP cells are potent sources of IL-10, we decided to investigate whether IL-10 could be involved in infectivity of B-1CDP cells to L. major in vitro.
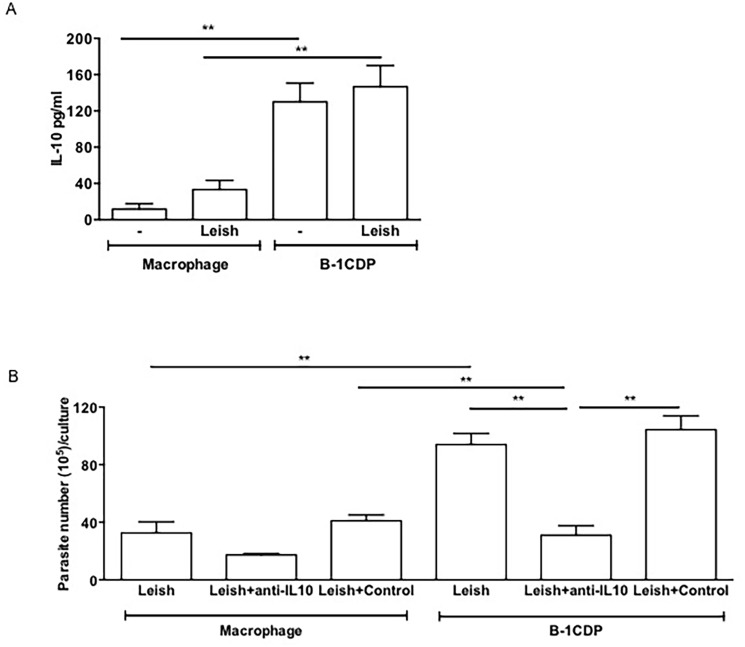
The results shown in Fig 2A confirm that B-1CDP cells produce more IL-10 than peritoneal macrophages. We also evaluated the production of TGF-β, which is another important immunoregulatory factor produced by phagocytes [38–40]. However, our results did not show a remarkable production of TGF-β by B-1CDP cells (data not shown).
Fig 2. IL-10 is determinant for the increased parasite load in B-1CDP.
B-1CDP cells and peritoneal macrophages were cultured in the presence or absence of L. major (MOI 10:1). After 24 hours, the supernatant was collected and IL-10 (A) was measured by ELISA. All cultures were performed in triplicate and bars show the mean ± SD. Representative result of three similar experiments **p <0.05. B-1CDP cells and peritoneal macrophages were treated or not with doses of monoclonal neutralizing anti-IL-10 or control isotype. Once were infected with L. major, after 24 hours, the cell cultures were washed with DMEM and incubated 3 days and then passed to Schneider medium. After 5 days in medium Schneider, promastigotes were counted in the supernatant (B). Statistical analysis were performed by Student’ t test from representative results of three similar experiments and bars show the mean +SD. **p ≤ 0.05.
Next we evaluated the possible immunomodulatory role of IL-10 in the infection of B-1CDP cells in comparison with peritoneal macrophages. Our results showed that the use of a neutralizing anti-IL-10 antibody induced a remarkable reduction in parasitic load both in peritoneal macrophages and B-1CDP cells (Fig 2B). It is important to emphasize that the negative modulation induced by neutralization of IL-10 was more pronounced in cultures of infected B-1CDP cells. This result strongly suggests that there is indeed a close relationship between the susceptibility of B-1CDP cells to infection by L. major and the increased production of the cytokine IL-10.
B-1CDP cells produce high amounts of lipid bodies
Lipid bodies are organelles consisting of a core of rich neutral lipids and wrapped with a monolayer of phospholipids, cholesterol, lipids, which contain a variety of proteins associated with different functions in cell metabolism, signaling, and inflammation [41]. Several groups have reported that host cells have their lipid metabolism altered and increase their number of lipid bodies following infection with intracellular pathogens [42–46]. Furthermore, our group has shown an increase of lipid bodies in macrophages infected with Trypanosoma cruzi, and their role in disease development [40].
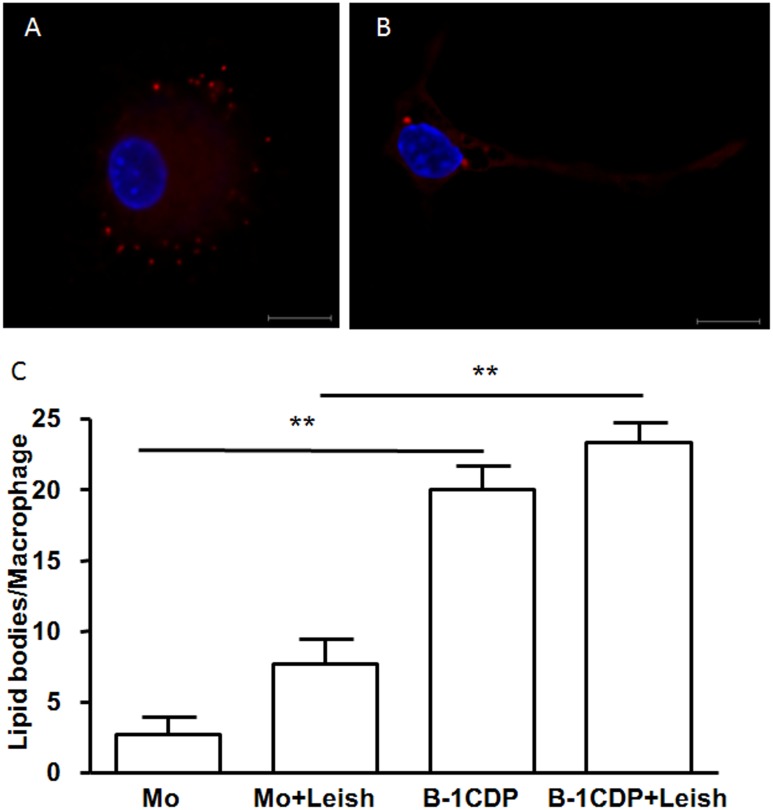
Based on this information, we investigated the presence of cytoplasmic lipid body organelles in B-1CDP cells and compared their number to peritoneal macrophages. We also investigated the generation of these organelles when peritoneal macrophages or B-1CDP cells were infected with L. major. Our data demonstrate that B-1CDP cells express a large number of lipid bodies in the cytoplasm even in the absence of stimulus (Fig 3A). After 24 hours of infection, we found that B-1CDP cells had a larger amount of lipid bodies compared to peritoneal macrophages (Fig 3B). However, infection per se did not increase statistically the number of lipid bodies in B-1CDP cells (Fig 3C).
Fig 3. B-1CDP cells naturally express increased numbers of lipid bodies as compared to peritoneal macrophages.
B-1CDP cells (A) and peritoneal macrophages (B) were incubated with glass coverslips, some cultures were infected with L. major (3C). Stained with Nile red (Sigma), the slides were washed and stained with DAPI (Sigma). The morphology of fixed cells was observed, and Nile red LBs were counted by light microscopy with a 100× objective lens in 50 consecutively scanned leukocytes. Statistical analysis were performed by Student’ t test from representative results of three similar experiments and bars show the mean +SD. **p ≤ 0.05. Bar, 10 μm. Representative of two experiments with identical results.
B-1CDP cells secrete high levels of PGE2
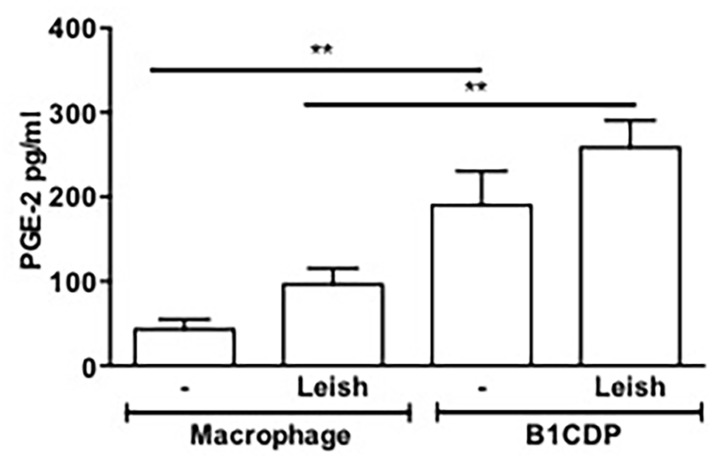
Lipid bodies are cellular sites devoted to the synthesis of PGE2 [40]. Previous studies reported that Leishmania infection induces PGE2 production and that this production could favor the persistence and/or progression of disease [47]. Furthermore, B-1CDP cells constitutively express COX-1 and COX-2 enzymes, and produce considerable quantities of PGE2 in response to inflammatory signals [8]. Based on these data, we evaluated the involvement of lipid mediator PGE2 in infection of B-1CDP cells with L. major. For this purpose, we measured this lipid mediator in the culture supernatant after 24 hours of infection. As demonstrated in Fig 4, B-1CDP cells produce considerable amounts of PGE2 even in the absence of stimulus, and infection did not changing it significantly.
Fig 4. B-1CDP cells secrete high levels of PGE2 regardless the infection.
B-1CDP cells and macrophages were incubated in the presence or absence of L. major (MOI 10:1). After 24 hours of infection the supernatant was collected, and PGE2 was measured by EIA. All cultures were performed in triplicate and bars show the mean ± SD. Statistical analysis were performed by Student’ t test from representative results of three similar experiments and bars show the mean +SD. **p ≤ 0.05.
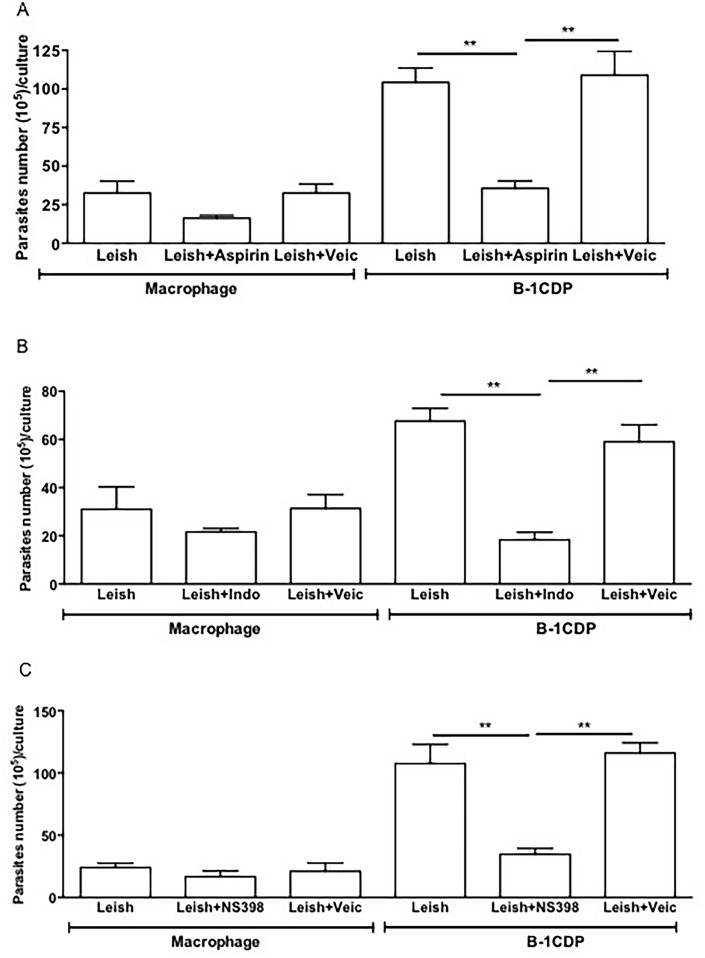
In order to determine whether PGE2 synthesis regulates parasite load, we used three different non-steroidal anti-inflammatory drugs (NSAIDs): Aspirin (Fig 5A), indomethacin (Fig 5B) and NS-398 (Fig 5C). The three drugs act as inhibitors of PGE2 production [38,48]. Our results demonstrate that inhibition of PGE2 production was accompanied by diminished parasite burden. It is important to emphasize that the inhibition was more pronounced in cultures of B-1CDP phagocytes treated with inhibitors (Fig 5A–5C). Treatment with NSAIDs drugs were not toxic to phagocytes, as measured by LDH release (S3 Fig).
Fig 5. Blockage of the cyclooxygenase pathway shift the B-1CDP cells to protective phenotype in the L major infection.
Macrophages and B-1CDP were incubated in the presence or absence of L. major treated or not with (A) aspirin (10 mg/mL), (B) indomethacin (1 mg/mL) or (C) NS-398 (1 mM). After 24 hours of incubation the cells were washed and incubated again for 5 days. After this time, the promastigotes were counted in the culture supernatant. All cultures were performed in triplicate and bars show the mean ± SD. Statistical analysis were performed by Student’ t test from representative results of three similar experiments and bars show the mean +SD. **p ≤ 0.05.
Effect of aspirin on IL-10 production and parasite release
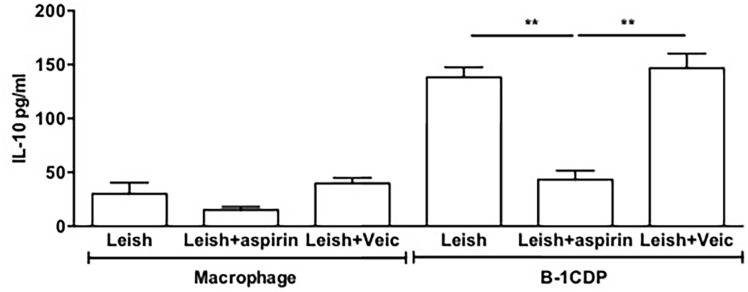
After verifying the effect of cyclooxygenase inhibition on the parasite load, we investigated an effect on the production of IL-10 by B-1CDP. Our data demonstrate a significant decrease in the levels of IL-10 in the supernatant when we used NSAIDs in our system (Fig 6).
Fig 6. Effect of inhibition of PGE2 on IL-10 releases.
Macrophages and B-1CDP cells were incubated with or without aspirin (10μg/mL) for 1 hour. They were then incubated in the presence or absence of L. major (MOI 10:1). After 24 hours of incubation, the supernatant was collected and IL-10 measured by ELISA. All cultures were performed in triplicate and bars show the mean ± SD. Statistical analysis were performed by Student’ t test from representative results of three similar experiments and bars show the mean +SD. **p ≤ 0.05.
B-1CDP cells from IL-10 KO mice are more competent to control L. major infection
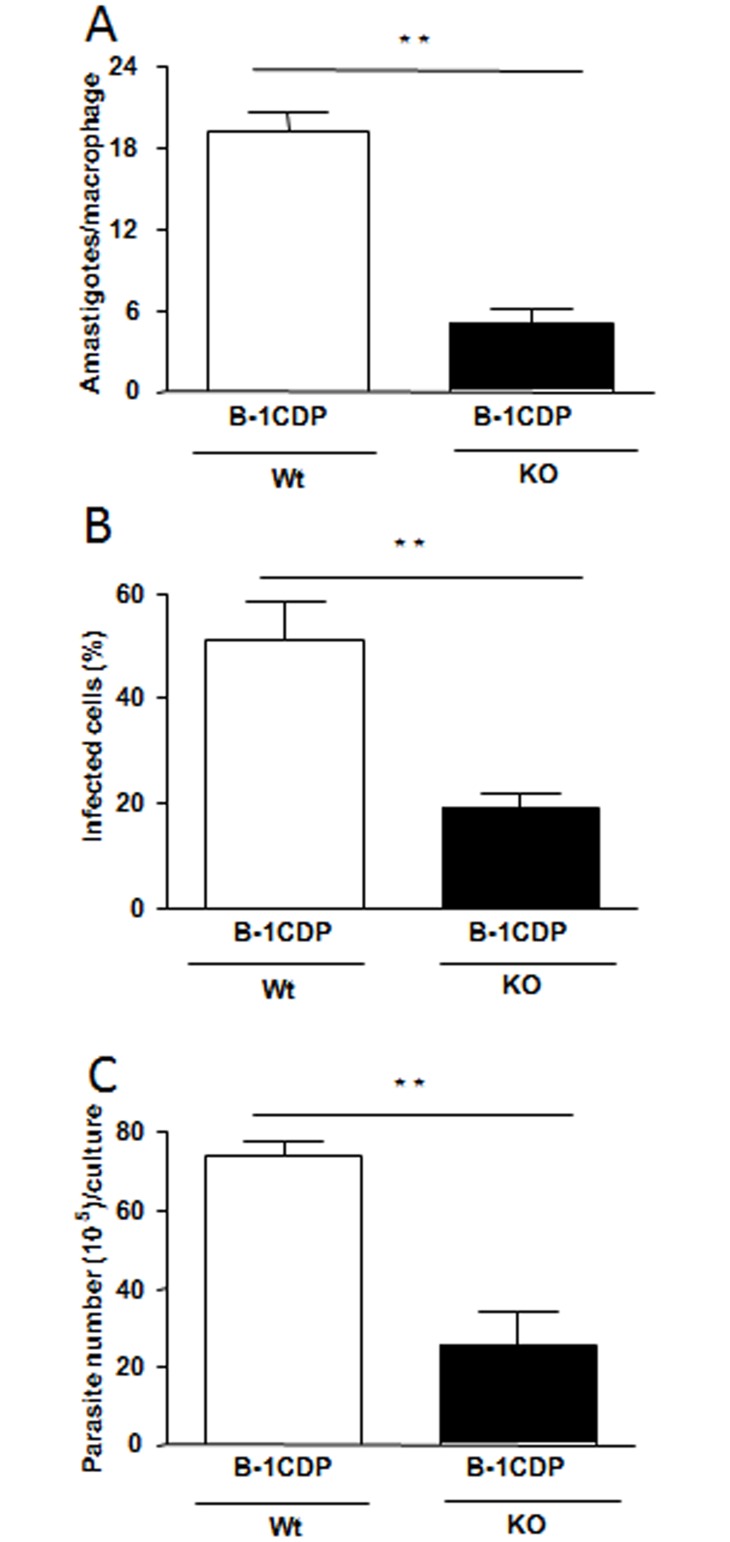
To confirm that the production of IL-10 is involved in the susceptibility to infection by L. major, we used B-1CDP cells from IL-10 KO mice. When compared to B-1CDP from wild type mice, the cells from IL-10 KO mice were more resistant to infection. Our data demonstrated a decrease in the number of intracellular amastigotes (Fig 7A) and percentage of infected cells (Fig 7B). Also, we observed the significant decrease in the amount of promastigote forms released by B-1CDP cells from IL-10 KO mice (Fig 7C). However, we did not observe any differences in parasitic capture by B-1CDP cells 4 hours after infection (data not shown).
Fig 7. B-1CDP cells from IL-10 deficient mice are more competent to control L. major infection.
To confirm the production of IL-10 is involved in the susceptibility to infection by L. major, we used B-1CDP cells from IL-10 KO mice. When compared to B-1CDP from wild type mice, the cells from IL-10 KO mice are more resistant to infection. Our data demonstrate decreased in the number of intracellular amastigotes (A) and percentage of infected cells (B). We also observed the significant decrease in the liberated promastigotes forms by B-1CDP from IL-10 KO mice (C). Statistical analysis were performed by Student’ t test from representative results of three similar experiments and bars show the mean +SD. **p ≤ 0.05.
Discussion
Phagocytosis is predominantly performed by professional phagocytes, such as polymorphonuclear cells, monocytes and macrophages [49]. Other types of cells such as fibroblasts and epithelial cells are also capable of phagocytizing particles, but in a much smaller degree compared to professional phagocytes.
Unlike professional phagocytes, most B cell subpopulations cannot perform phagocytosis [50]. However, several studies have discussed the mechanisms by which B-1 cells differentiate into phagocytes and the ontogenetic and physiological implications of this phenomenon. Reports in the literature have shown that pre-CD5+ B cells can differentiate into cells similar to macrophages [43,51].
Moreover, a link between the development of B-1 cells and macrophages was established by identifying bi-phenotypic B cell/macrophage progenitors in fetal liver [17] and adult bone marrow [51]. The presence of bi-phenotypic B cell/macrophage precursor in mammals suggests a close evolutionary relationship between B cells and macrophages derived from monocytes, which indicates the possibility of a common phylogenetic ancestor with attributes of both cell types, which has already been demonstrated in teleost fish [52].
Borrello and Phipps [6] demonstrated the differentiation of splenocytes simultaneously expressing B-cell and macrophage characteristics, when co-cultured with fibroblasts. Almeida and colleagues [33] showed that peritoneal B-1 cells proliferate and differentiate into new mononuclear phagocytic cells with no relation to blood derived monocytes. A further evidence for this B-1 cell/phagocyte link are discussed elsewhere [4].
In the present study, we demonstrate that phagocytes derived from B-1 cells (called B-1CDP) have the ability to phagocytose the pathogenic protozoan parasite L. major. Moreover, B-1CDP cells are more susceptible to infection with this parasite when compared with murine peritoneal macrophages; the intracellular load of amastigote can also be indirectly evaluated by the ability of host cells to release promastigotes following culture in Schneider medium [34]. The amount of released promastigotes in the supernatants after 5 days of infection was higher in cultures of the B-1CDP cells, as compared to cultures of murine peritoneal macrophages. We investigated which cytokines could be involved in this phenomenon. The cytokine IL-10 is produced by different cell types, such as macrophages, dendritic cells (DC), B cells and different subtypes of T lymphocytes [53,54]. Many of the effects of IL-10 on the functions of T cells and NK cells are now known to be mediated by a direct effect of IL -10 on macrophages. In addition, IL-10 may act directly on CD4+ T cells by inhibiting the production and proliferation of IL-2, IFN-γ, IL-4, IL-5 and TNF-α [53,55,56]. It is well characterized that IL-10 plays an important role in susceptibility to leishmaniasis. It suppresses the synthesis of the inflammatory cytokine IFN-γ and can inhibit the production of NO by activated macrophages and also down-regulates the expression of MHC class I and class II and co-stimulatory molecules (B7.1 and B7.2) on the surface of macrophages [57]. Furthermore, BALB/c mice deficient in IL-10 could control the L. major infection, suggesting that this cytokine might play an important role in mediating susceptibility to and pathogenesis of experimental cutaneous leishmaniasis [58].
The TGF-β is an important cytokine with anti-inflammatory activity, which was characterized as an important factor related susceptibility to experimental infection by Leishmania in vivo [59]. Our results indicated that the susceptibility to L. major infection presented by B-1CDP cells is related to the production of IL-10. Surprisingly, our results also indicated that the cytokine TGF-β is not involved in the increased infection of B-1CDP cells (data not shown).
B-1CDP cells produce a large quantity of IL-10 even without stimulation, in agreement with a marked anti-inflammatory profile [13,21], and with a propensity for infection by Leishmania. The role of IL-10 was confirmed by showing that a neutralizing antibody specific for IL-10 induced significant reduction in the amount of promastigotes released in culture supernatants.
Another aspect that could lead to increased susceptibility of B-1CDP cells to L. major infection, were the lipid bodies presented in that cell population. Lipid bodies are key organelles involved in cholesterol metabolism and the synthesis of fatty acids indicating that both anabolic and catabolic phases of lipid metabolism occur in these organelles [60,61]. Leukocytes have few lipid bodies, but these cells can be stimulated to form new lipid bodies quickly [62]. Accumulation of lipid bodies was also identified within peritoneal macrophages during the acute phase of experimental infection with the parasite T. cruzi and L. amazonensis [40,63]. Consistent with the role of inflammation on the leucocytes, the lipid bodies formed by these cells are important sites for the production of inflammatory mediators. Lipid bodies contain arachidonic acid, which serves as a precursor for the synthesis of eicosanoids, and all the enzymes necessary for this synthesis, including cyclooxygenase (COX), PGE2 synthase, 5/15-lipoxygenase (5-LO and 15-LO), and leukotriene C4 synthase [64].
As demonstrated in our study, the B-1CDP cells contain a large amount of cytoplasmatic lipid bodies compared with murine peritoneal macrophages. As the increased numbers of cytoplasmatic lipid bodies is associated to increase PGE2 production, the result on the L. major infection was expected. Our data shows that the inhibition of the PGE2 modulated the infection causing the decreased release of promastigotes forms by B-1CDP cells. PGE2 is known to be an important inducer of cytokines as TGF-β [38,48] and IL-10 [65].
The formation of lipid bodies within inflammatory macrophages positively correlates with increased generation of PGE2, indicating a role for these organelles in enhanced production of eicosanoids observed in Chagas´ disease [66]. The increased capacity of macrophages to generate PGE2 in the course of a pathogenic infection due to the increased formation of lipid bodies may contribute to mechanism by which intracellular pathogens survive in the host cells. The high concentration of PGE2 is a potent inhibitor of the T cell response and NO production by macrophages, favoring the growth of the intracellular parasite [38,39,67]. In addition, PGE2 is essential for the increased growth of parasites in macrophages that have ingested of apoptotic cells [38,48].
IL-10 produced by B-1CDP cells is the key to understanding their increased susceptibility to infection. Using B-1CDP cells from IL-10 KO mice the infection with L. major was less effective. These results strongly suggest that IL-10 secreted by B-1CDP cells, and probably B-1 cells as well, are involved in the susceptibility to infection. Popi and colleagues [68], using the Coxiella burnetti cell infection model in vitro, obtained a similar result. They found that peritoneal macrophages from BALB/c XID mice, which are deficient in B-1 cells, mice are more resistant to intracellular infection by C. burnetti when compared with peritoneal macrophages from wild-type BALB/c mice. The involvement of B-1 cells in experimental infection with Leishmania remains unclear, only two independent studies have reported the involvement of these cells in the infection, however the results were not conclusive [69,70].
Altogether, our data unveil that PGE2-driven production of IL-10 determines susceptibility of B-1CDP cells to L. major infection. Little is known about the physiology of B-1CDP cells, and their immune functions. The implications of our present findings in the pathophysiology of immunity remain open for further investigation. The “promiscuous" expression of both myeloid and lymphoid characteristics of a single cell type, and the factors governing differentiation of B-1 cells in B-1CDP cells will certainly open new avenues for the understanding of lymphoid and myeloid cells and their role in parasitic diseases.
Supporting Information
B-1CDP cells (A) and peritoneal macrophages (B) were cultured (105/ml) and infected with promastigotes of L. major. After 24 hours, the cell culture was washed and phagocytes were cultured for another 3 days with DMEM supplemented with 10% FBS at 37°C. After this period, cells were fixed and permeabilized. Following incubation with Polyclonal goat antibody anti-mouse IgM-FITC (B-1CDP cells) and rat anti-mouse F4/80-FITC (macrophages). After incubation, glass cover slips were washed again and incubated in 1 microgram/mL of DAPI. After 10 min of incubation, glass cover slips were washed and mounted. Images were acquired using a Zeiss Observer Z.1 microscope in a 63x objectives. After the acquisition, images were processed with a deconvolution module using Zen software. Bar, 10 μm. Representative of two experiments with identical results.
(TIF)
B-1CDP cells and peritoneal macrophages were cultured (105/ml) and infected with metacyclic promastigotes of L. major. After 4 hours, the cell culture was washed and cells were stained and parasites inside the phagocytes were counted under the light microscope. All cultures were performed in triplicate and bars show the mean +SD.
(TIF)
B-1CDP cells and peritoneal macrophages were cultured (105/ml) and infected with promastigotes of L. major were treated or not with aspirin (10 mg/mL), indomethacin (1 mg/mL) or NS-398 (1 mM). After 72 hours of incubation the supernatant was withdrawn and frozen for determination of LDH release.
(TIF)
Acknowledgments
We thank Jorgete Logullo e Lindomar Miranda for helpful technical assistance. CGFD, AM, CBM, and GADR are senior investigators from CNPq.
Data Availability
All relevant data are within the paper and its Supporting Information files.
Funding Statement
This work was supported by Brazilian National Research Council (CNPq) and Rio de Janeiro State Science Foundation (FAPERJ). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. van Furth R, Cohn ZA. The origin and kinetics of mononuclear phagocytes. J Exp Med. 1968;128(3):415–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Okabe Y, Medzhitov R. Tissue-specific signals control reversible program of localization and functional polarization of macrophages. Cell. 2014;157(4):832–44. 10.1016/j.cell.2014.04.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Ginhoux F, Jung S. Monocytes and macrophages: developmental pathways and tissue homeostasis. Nat Rev Immunol. 2014;14(6):392–404. 10.1038/nri3671 [DOI] [PubMed] [Google Scholar]
- 4. Lopes JD, Mariano M. B-1 cell: the precursor of a novel mononuclear phagocyte with immuno-regulatory properties. An Acad Bras Cienc. 2009;81(3):489–96. [DOI] [PubMed] [Google Scholar]
- 5. Popi AF, Osugui L, Perez KR, Longo-Maugeri IM, Mariano M. Could a B-1 cell derived phagocyte "be one" of the peritoneal macrophages during LPS-driven inflammation? PLoS One. 2012;7(3):e34570 10.1371/journal.pone.0034570 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Borrello MA, Phipps RP. Fibroblast-secreted macrophage colony-stimulating factor is responsible for generation of biphenotypic B/macrophage cells from a subset of mouse B lymphocytes. J Immunol. 1999;163(7):3605–11. [PubMed] [Google Scholar]
- 7. Popi AF, Motta FL, Mortara RA, Schenkman S, Lopes JD, Mariano M. Co-ordinated expression of lymphoid and myeloid specific transcription factors during B-1b cell differentiation into mononuclear phagocytes in vitro. Immunology. 2009;126(1):114–22. 10.1111/j.1365-2567.2008.02883.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Graf BA, Nazarenko DA, Borrello MA, Roberts LJ, Morrow JD, Palis J, et al. Biphenotypic B/macrophage cells express COX-1 and up-regulate COX-2 expression and prostaglandin E(2) production in response to pro-inflammatory signals. Eur J Immunol. 1999;29(11):3793–803. [DOI] [PubMed] [Google Scholar]
- 9. Hayakawa K, Hardy RR, Stall AM, Herzenberg LA, Herzenberg LA. Immunoglobulin-bearing B cells reconstitute and maintain the murine Ly-1 B cell lineage. Eur J Immunol. 1986;16(10):1313–6. [DOI] [PubMed] [Google Scholar]
- 10. Herzenberg LA, Stall AM, Lalor PA, Sidman C, Moore WA, Parks DR, et al. The Ly-1 B cell lineage. Immunol Rev. 1986;93:81–102. [DOI] [PubMed] [Google Scholar]
- 11. Kipps TJ. The CD5 B cell. Adv Immunol. 1989;47:117–85. [DOI] [PubMed] [Google Scholar]
- 12. Cumano A, Paige CJ, Iscove NN, Brady G. Bipotential precursors of B cells and macrophages in murine fetal liver. Nature. 1992;356(6370):612–5. [DOI] [PubMed] [Google Scholar]
- 13. Cruz-Leal Y, Machado Y, Lopez-Requena A, Canet L, Laborde R, Alvares AM, et al. Role of B-1 cells in the immune response against an antigen encapsulated into phosphatidylcholine-containing liposomes. Int Immunol. 2014;26(8):427–37. 10.1093/intimm/dxu042 [DOI] [PubMed] [Google Scholar]
- 14. Thies FG, Laurindo MF, Perez EC, Novaes e Brito RR, Mariano M, Popi AF. Cross talk between peritoneal macrophages and B-1 cells in vitro. PLoS One. 2013;8(5):e62805 10.1371/journal.pone.0062805 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Bonamin LV, Sato C, Zalla Neto R, Morante G, Cardoso TN, de Santana FR, et al. Immunomodulation of Homeopathic Thymulin 5CH in a BCG-Induced Granuloma Model. Evid Based Complement Alternat Med. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Pugliese LS, Goncalves TO, Popi AF, Mariano M, Pesquero JB, Lopes JD. B-1 lymphocytes differentiate into functional osteoclast-like cells. Immunobiology. 2012;217(3):336–44. 10.1016/j.imbio.2011.07.014 [DOI] [PubMed] [Google Scholar]
- 17. Katoh S, Tominaga A, Migita M, Kudo A, Takatsu K. Conversion of normal Ly-1-positive B-lineage cells into Ly-1-positive macrophages in long-term bone marrow cultures. Dev Immunol. 1990;1(2):113–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Masmoudi H, Mota-Santos T, Huetz F, Coutinho A, Cazenave PA. All T15 Id-positive antibodies (but not the majority of VHT15+ antibodies) are produced by peritoneal CD5+ B lymphocytes. Int Immunol. 1990;2(6):515–20. [DOI] [PubMed] [Google Scholar]
- 19. Laurindo MF, Thies FG, Perez EC, Novaes e Brito RR, Mariano M, Popi AF. B16 melanoma cells increase B-1 cell survival, IL-10 production and radioresistance in vitro. Immunobiology. 2013;218(4):609–19. 10.1016/j.imbio.2012.07.032 [DOI] [PubMed] [Google Scholar]
- 20. Palos MC, Azevedo MC, Thies FG, Osugui L, Alvares AM, Laurindo MF, et al. Different inflammatory stimuli in the footpad of mice influence the kinetics of resident peritoneal cells. Inflamm Res. 2012;61(11):1187–94. 10.1007/s00011-012-0514-y [DOI] [PubMed] [Google Scholar]
- 21. Barbeiro DF, Barbeiro HV, Faintuch J, Ariga SK, Mariano M, Popi AF, et al. B-1 cells temper endotoxemic inflammatory responses. Immunobiology. 2011;216(3):302–8. 10.1016/j.imbio.2010.08.002 [DOI] [PubMed] [Google Scholar]
- 22. Pennell CA, Arnold LW, Haughton G, Clarke SH. Restricted Ig variable region gene expression among Ly-1+ B cell lymphomas. J Immunol. 1988;141(8):2788–96. [PubMed] [Google Scholar]
- 23. Hardy RR, Kishimoto T, Hayakawa K. Differentiation of B cell progenitors in vitro: generation of surface IgM+ B cells, including Ly-1 B cells, from Thy-1- asialoGM1+ cells in newborn liver. Eur J Immunol. 1987;17(12):1769–74. [DOI] [PubMed] [Google Scholar]
- 24. Hayakawa K, Hardy RR, Parks DR, Herzenberg LA. The "Ly-1 B" cell subpopulation in normal immunodefective, and autoimmune mice. J Exp Med. 1983;157(1):202–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Takeshita H, Taniuchi I, Kato J, Watanabe T. Abrogation of autoimmune disease in Lyn-deficient mice by the mutation of the Btk gene. Int Immunol. 1998;10(4):435–44. [DOI] [PubMed] [Google Scholar]
- 26. Askenase PW, Kawikova I, Paliwal V, Akahira-Azuma M, Gerard C, Hugli T, et al. A new paradigm of T cell allergy: requirement for the B-1 cell subset. Int Arch Allergy Immunol. 1999;118(2–4):145–9. [DOI] [PubMed] [Google Scholar]
- 27. Murakami K, Okada K, Ikawa Y, Aida Y. Bovine leukemia virus induces CD5- B cell lymphoma in sheep despite temporarily increasing CD5+ B cells in asymptomatic stage. Virology. 1994;202(1):458–65. [DOI] [PubMed] [Google Scholar]
- 28. Murakami Y, Kong YY, Nishimura Y, Nomoto K, Umesue M, Omoto K, et al. Prevention of anti-T-cell receptor alpha beta monoclonal antibody-induced side-effects by treatment with cyclosporin A without interference of monoclonal antibody-induced immunosuppression in mice. Immunology. 1995;86(2):238–43. [PMC free article] [PubMed] [Google Scholar]
- 29. Ghosn EE, Russo M, Almeida SR. Nitric oxide-dependent killing of Cryptococcus neoformans by B-1-derived mononuclear phagocyte. J Leukoc Biol. 2006;80(1):36–44. [DOI] [PubMed] [Google Scholar]
- 30. Santos-Lima EC, Vasconcellos R, Reina-San-Martin B, Fesel C, Cordeiro-Da-Silva A, Berneman A, et al. Significant association between the skewed natural antibody repertoire of Xid mice and resistance to Trypanosoma cruzi infection. Eur J Immunol. 2001;31(2):634–45. [DOI] [PubMed] [Google Scholar]
- 31. Cunningham AC. Parasitic adaptive mechanisms in infection by leishmania. Exp Mol Pathol. 2002;72(2):132–41. [DOI] [PubMed] [Google Scholar]
- 32. Liese J, Schleicher U, Bogdan C. The innate immune response against Leishmania parasites. Immunobiology. 2008;213(3–4):377–87. 10.1016/j.imbio.2008.02.001 [DOI] [PubMed] [Google Scholar]
- 33. Almeida SR, Aroeira LS, Frymuller E, Dias MA, Bogsan CS, Lopes JD, et al. Mouse B-1 cell-derived mononuclear phagocyte, a novel cellular component of acute non-specific inflammatory exudate. Int Immunol. 2001;13(9):1193–201. [DOI] [PubMed] [Google Scholar]
- 34. Gomes NA, Barreto-de-Souza V, DosReis GA. Early in vitro priming of distinct T(h) cell subsets determines polarized growth of visceralizing Leishmania in macrophages. Int Immunol. 2000;12(9):1227–33. [DOI] [PubMed] [Google Scholar]
- 35. Greenspan P, Fowler SD. Spectrofluorometric studies of the lipid probe, nile red. J Lipid Res. 1985;26(7):781–9. [PubMed] [Google Scholar]
- 36. O'Garra A, Chang R, Go N, Hastings R, Haughton G, Howard M. Ly-1 B (B-1) cells are the main source of B cell-derived interleukin 10. Eur J Immunol. 1992;22(3):711–7. [DOI] [PubMed] [Google Scholar]
- 37. Moore KW, O'Garra A, de Waal Malefyt R, Vieira P, Mosmann TR. Interleukin-10. Annu Rev Immunol. 1993;11:165–90. [DOI] [PubMed] [Google Scholar]
- 38. Freire-de-Lima CG, Nascimento DO, Soares MB, Bozza PT, Castro-Faria-Neto HC, de Mello FG, et al. Uptake of apoptotic cells drives the growth of a pathogenic trypanosome in macrophages. Nature. 2000;403(6766):199–203. [DOI] [PubMed] [Google Scholar]
- 39. Luna-Gomes T, Filardy AA, Rocha JD, Decote-Ricardo D, LaRocque-de-Freitas IF, Morrot A, et al. Neutrophils increase or reduce parasite burden in Trypanosoma cruzi-infected macrophages, depending on host strain: role of neutrophil elastase. PLoS One. 2014;9(3):e90582 10.1371/journal.pone.0090582 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. D'Avila H, Freire-de-Lima CG, Roque NR, Teixeira L, Barja-Fidalgo C, Silva AR, et al. Host cell lipid bodies triggered by Trypanosoma cruzi infection and enhanced by the uptake of apoptotic cells are associated with prostaglandin E(2) generation and increased parasite growth. J Infect Dis. 2011;204(6):951–61. 10.1093/infdis/jir432 [DOI] [PubMed] [Google Scholar]
- 41. Bozza PT, Magalhaes KG, Weller PF. Leukocyte lipid bodies—Biogenesis and functions in inflammation. Biochim Biophys Acta. 2009;1791(6):540–51. 10.1016/j.bbalip.2009.01.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Charron AJ, Sibley LD. Host cells: mobilizable lipid resources for the intracellular parasite Toxoplasma gondii. J Cell Sci. 2002;115(Pt 15):3049–59. . [DOI] [PubMed] [Google Scholar]
- 43. Jackson KE, Klonis N, Ferguson DJ, Adisa A, Dogovski C, Tilley L. Food vacuole-associated lipid bodies and heterogeneous lipid environments in the malaria parasite, Plasmodium falciparum. Mol Microbiol. 2004;54(1):109–22. [DOI] [PubMed] [Google Scholar]
- 44. Cocchiaro JL, Kumar Y, Fischer ER, Hackstadt T, Valdivia RH. Cytoplasmic lipid droplets are translocated into the lumen of the Chlamydia trachomatis parasitophorous vacuole. Proc Natl Acad Sci U S A. 2008;105(27):9379–84. 10.1073/pnas.0712241105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Peyron P, Vaubourgeix J, Poquet Y, Levillain F, Botanch C, Bardou F, et al. Foamy macrophages from tuberculous patients' granulomas constitute a nutrient-rich reservoir for M. tuberculosis persistence. PLoS Pathog. 2008;4(11):e1000204 10.1371/journal.ppat.1000204 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Mattos KA, Lara FA, Oliveira VG, Rodrigues LS, D'Avila H, Melo RC, et al. Modulation of lipid droplets by Mycobacterium leprae in Schwann cells: a putative mechanism for host lipid acquisition and bacterial survival in phagosomes. Cell Microbiol. 2011;13(2):259–73. 10.1111/j.1462-5822.2010.01533.x [DOI] [PubMed] [Google Scholar]
- 47. Arias-Negrete S, Keller K, Chadee K. Proinflammatory cytokines regulate cyclooxygenase-2 mRNA expression in human macrophages. Biochem Biophys Res Commun. 1995;208(2):582–9. [DOI] [PubMed] [Google Scholar]
- 48. Freire-de-Lima CG, Xiao YQ, Gardai SJ, Bratton DL, Schiemann WP, Henson PM. Apoptotic cells, through transforming growth factor-beta, coordinately induce anti-inflammatory and suppress pro-inflammatory eicosanoid and NO synthesis in murine macrophages. J Biol Chem. 2006;281(50):38376–84. [DOI] [PubMed] [Google Scholar]
- 49. Rabinovitch M. Professional and non-professional phagocytes: an introduction. Trends Cell Biol. 1995;5(3):85–7. [DOI] [PubMed] [Google Scholar]
- 50. Aderem A, Underhill DM. Mechanisms of phagocytosis in macrophages. Annu Rev Immunol. 1999;17:593–623. [DOI] [PubMed] [Google Scholar]
- 51. Spencker T, Neumann D, Strasser A, Resch K, Martin M. Lineage switch of a mouse pre-B cell line (SPGM-1) to macrophage-like cells after incubation with phorbol ester and calcium ionophore. Biochem Biophys Res Commun. 1995;216(2):540–8. [DOI] [PubMed] [Google Scholar]
- 52. Li J, Barreda DR, Zhang YA, Boshra H, Gelman AE, Lapatra S, et al. B lymphocytes from early vertebrates have potent phagocytic and microbicidal abilities. Nat Immunol. 2006;7(10):1116–24. [DOI] [PubMed] [Google Scholar]
- 53. Moore KW, de Waal Malefyt R, Coffman RL, O'Garra A. Interleukin-10 and the interleukin-10 receptor. Annu Rev Immunol. 2001;19:683–765. [DOI] [PubMed] [Google Scholar]
- 54. Kamanaka M, Kim ST, Wan YY, Sutterwala FS, Lara-Tejero M, Galan JE, et al. Expression of interleukin-10 in intestinal lymphocytes detected by an interleukin-10 reporter knockin tiger mouse. Immunity. 2006;25(6):941–52. 10.1016/j.immuni.2006.09.013 . [DOI] [PubMed] [Google Scholar]
- 55. Schandene L, Alonso-Vega C, Willems F, Gerard C, Delvaux A, Velu T, et al. B7/CD28-dependent IL-5 production by human resting T cells is inhibited by IL-10. J Immunol. 1994;152(9):4368–74. [PubMed] [Google Scholar]
- 56. Joss A, Akdis M, Faith A, Blaser K, Akdis CA. IL-10 directly acts on T cells by specifically altering the CD28 co-stimulation pathway. Eur J Immunol. 2000;30(6):1683–90. [DOI] [PubMed] [Google Scholar]
- 57. Padigel UM, Alexander J, Farrell JP. The role of interleukin-10 in susceptibility of BALB/c mice to infection with Leishmania mexicana and Leishmania amazonensis. J Immunol. 2003;171(7):3705–10. [DOI] [PubMed] [Google Scholar]
- 58. Kane MM, Mosser DM. The role of IL-10 in promoting disease progression in leishmaniasis. J Immunol. 2001;166(2):1141–7. [DOI] [PubMed] [Google Scholar]
- 59. Barral-Netto M, Barral A, Brownell CE, Skeiky YA, Ellingsworth LR, Twardzik DR, et al. Transforming growth factor-beta in leishmanial infection: a parasite escape mechanism. Science. 1992;257(5069):545–8. [DOI] [PubMed] [Google Scholar]
- 60. Brasaemle DL, Dolios G, Shapiro L, Wang R. Proteomic analysis of proteins associated with lipid droplets of basal and lipolytically stimulated 3T3-L1 adipocytes. J Biol Chem. 2004;279(45):46835–42. [DOI] [PubMed] [Google Scholar]
- 61. Fujimoto T. [Lipid droplet as an independent organelle]. Seikagaku. 2004;76(6):578–84. [PubMed] [Google Scholar]
- 62. Bozza PT, Melo RC, Bandeira-Melo C. Leukocyte lipid bodies regulation and function: contribution to allergy and host defense. Pharmacol Ther. 2007;113(1):30–49. [DOI] [PubMed] [Google Scholar]
- 63. Pinheiro RO, Nunes MP, Pinheiro CS, D'Avila H, Bozza PT, Takiya CM, et al. Induction of autophagy correlates with increased parasite load of Leishmania amazonensis in BALB/c but not C57BL/6 macrophages. Microbes Infect. 2009;11(2):181–90. 10.1016/j.micinf.2008.11.006 [DOI] [PubMed] [Google Scholar]
- 64. Bandeira-Melo C, Weller PF, Bozza PT. Identifying intracellular sites of eicosanoid lipid mediator synthesis with EicosaCell assays. Methods Mol Biol. 2011;717:277–89. 10.1007/978-1-61779-024-9_16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Stolina M, Sharma S, Lin Y, Dohadwala M, Gardner B, Luo J, et al. Specific inhibition of cyclooxygenase 2 restores antitumor reactivity by altering the balance of IL-10 and IL-12 synthesis. J Immunol. 2000;164(1):361–70. [DOI] [PubMed] [Google Scholar]
- 66. Melo RC, Fabrino DL, D'Avila H, Teixeira HC, Ferreira AP. Production of hydrogen peroxide by peripheral blood monocytes and specific macrophages during experimental infection with Trypanosoma cruzi in vivo. Cell Biol Int. 2003;27(10):853–61. . [DOI] [PubMed] [Google Scholar]
- 67. Renz H, Gong JH, Schmidt A, Nain M, Gemsa D. Release of tumor necrosis factor-alpha from macrophages. Enhancement and suppression are dose-dependently regulated by prostaglandin E2 and cyclic nucleotides. J Immunol. 1988;141(7):2388–93. [PubMed] [Google Scholar]
- 68. Popi AF, Zamboni DS, Mortara RA, Mariano M. Microbicidal property of B1 cell derived mononuclear phagocyte. Immunobiology. 2009;214(8):664–73. 10.1016/j.imbio.2008.12.007 [DOI] [PubMed] [Google Scholar]
- 69. Hoerauf A, Solbach W, Lohoff M, Rollinghoff M. The Xid defect determines an improved clinical course of murine leishmaniasis in susceptible mice. Int Immunol. 1994;6(8):1117–24. [DOI] [PubMed] [Google Scholar]
- 70. Babai B, Louzir H, Cazenave PA, Dellagi K. Depletion of peritoneal CD5+ B cells has no effect on the course of Leishmania major infection in susceptible and resistant mice. Clin Exp Immunol. 1999;117(1):123–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
B-1CDP cells (A) and peritoneal macrophages (B) were cultured (105/ml) and infected with promastigotes of L. major. After 24 hours, the cell culture was washed and phagocytes were cultured for another 3 days with DMEM supplemented with 10% FBS at 37°C. After this period, cells were fixed and permeabilized. Following incubation with Polyclonal goat antibody anti-mouse IgM-FITC (B-1CDP cells) and rat anti-mouse F4/80-FITC (macrophages). After incubation, glass cover slips were washed again and incubated in 1 microgram/mL of DAPI. After 10 min of incubation, glass cover slips were washed and mounted. Images were acquired using a Zeiss Observer Z.1 microscope in a 63x objectives. After the acquisition, images were processed with a deconvolution module using Zen software. Bar, 10 μm. Representative of two experiments with identical results.
(TIF)
B-1CDP cells and peritoneal macrophages were cultured (105/ml) and infected with metacyclic promastigotes of L. major. After 4 hours, the cell culture was washed and cells were stained and parasites inside the phagocytes were counted under the light microscope. All cultures were performed in triplicate and bars show the mean +SD.
(TIF)
B-1CDP cells and peritoneal macrophages were cultured (105/ml) and infected with promastigotes of L. major were treated or not with aspirin (10 mg/mL), indomethacin (1 mg/mL) or NS-398 (1 mM). After 72 hours of incubation the supernatant was withdrawn and frozen for determination of LDH release.
(TIF)
Data Availability Statement
All relevant data are within the paper and its Supporting Information files.