Abstract
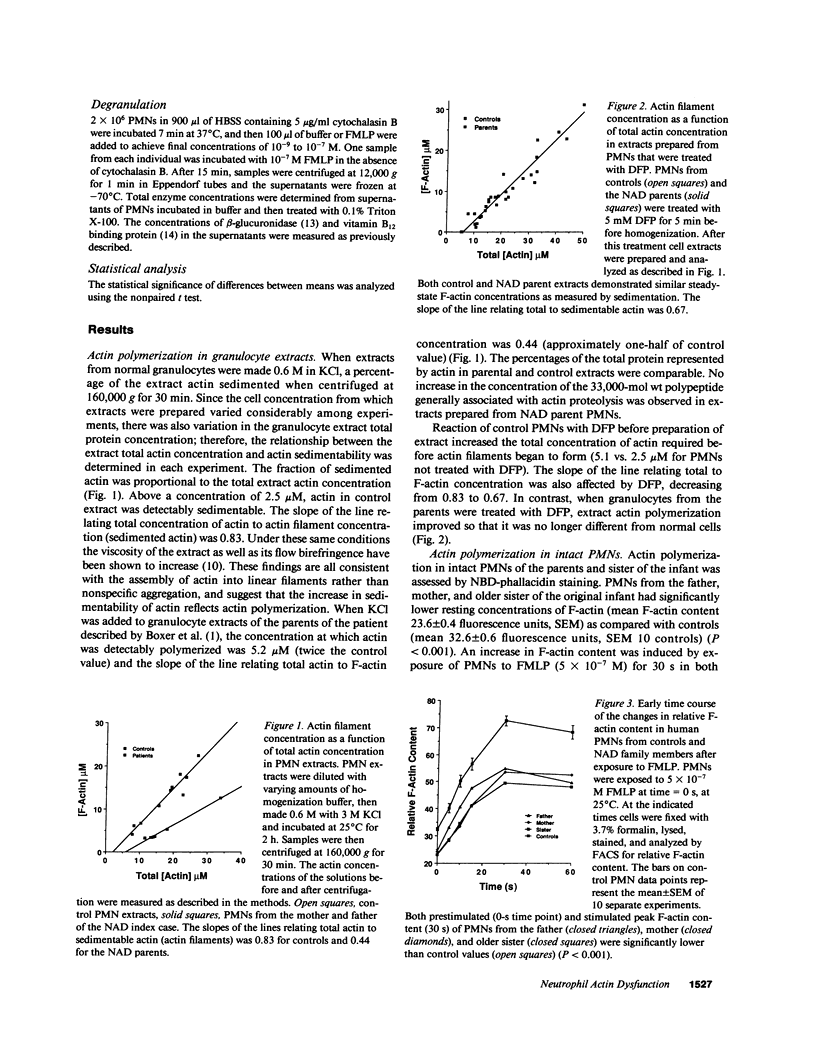
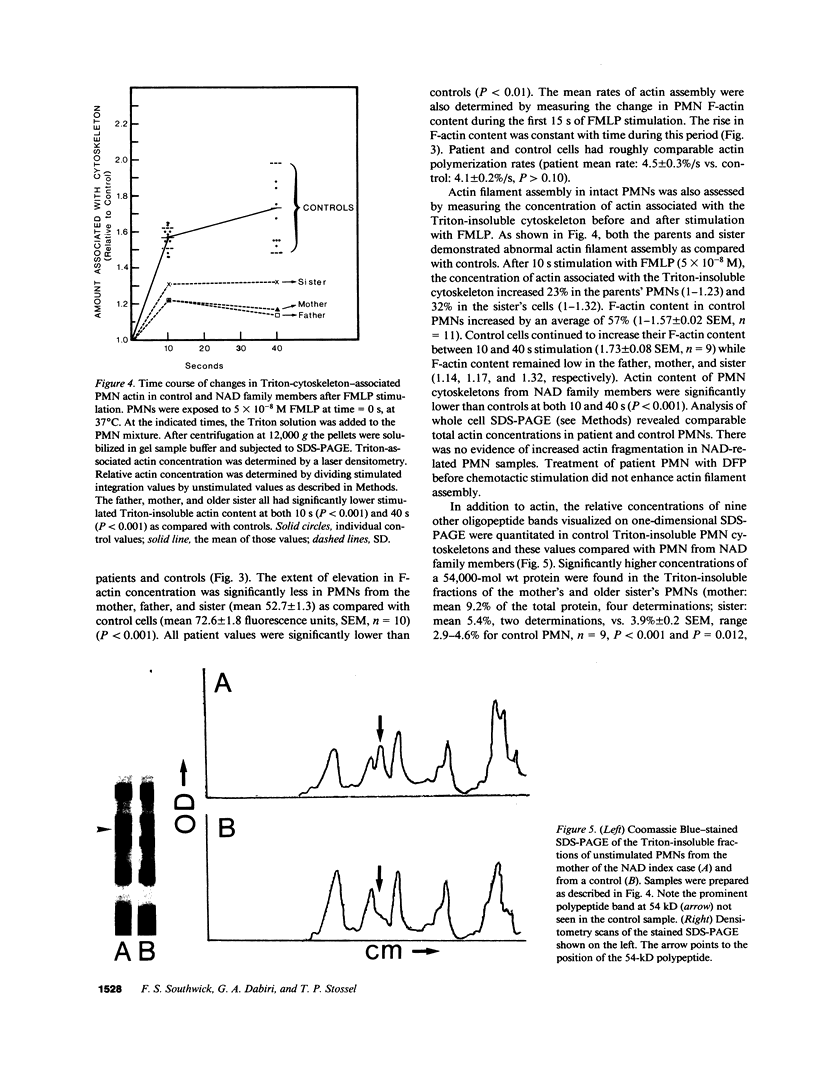
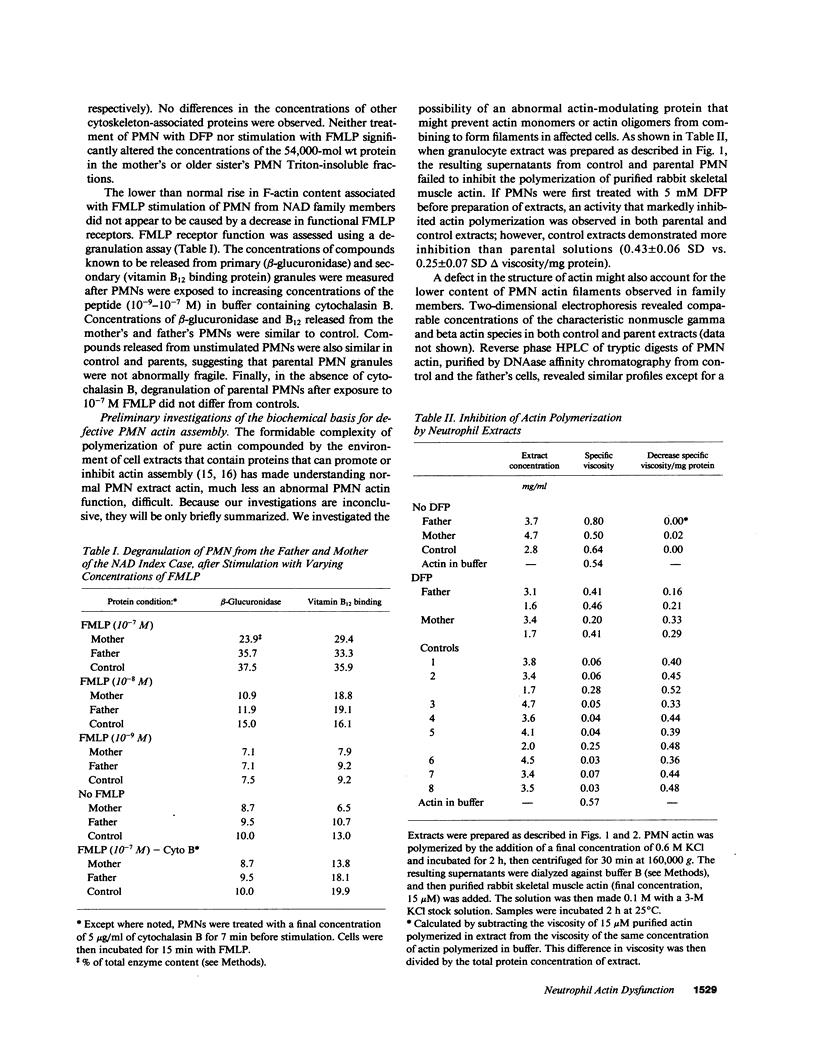
A male infant with a severe neutrophil motility disorder and poorly polymerizable actin in PMN extracts was reported over a decade ago to have neutrophil actin dysfunction (NAD) (1974. N. Engl. J. Med. 291:1093-1099). Polymerized actin (F-actin) content of fixed and permeabilized intact neutrophils from the father, mother, and sister of the NAD index case have been measured using nitrobenzoxadiazole-phallacidin, a fluorescent compound which binds specifically to actin filaments. F-actin content of unstimulated PMN from all three family members was significantly lower than unstimulated control PMN (mean 23.6 +/- 0.4 SEM fluorescent units vs. 32.6 +/- 0.6 for controls). After stimulation with the chemotactic peptide FMLP, maximal F-actin content of NAD family member PMN was below that of controls (52.7 +/- 1.3 vs. 72.6 +/- 1.8). F-actin content of detergent insoluble cytoskeletons after stimulation with FMLP was also significantly lower in PMN from NAD family members as compared with controls (21 +/- 6% vs. 73 +/- 8%). PMN extracts from the father and mother, when treated with 0.6 M KCl, polymerized half as much actin as controls. Whereas diisopropylfluorophosphate treatment of normal PMN decreased actin polymerizability in cell extracts, this treatment increased the assembly of actin in parental PMN extract. Addition of purified actin to NAD extracts failed to reveal an abnormal actin polymerization inhibitory activity, and no obvious structural defect in actin purified from the father's PMNs was noted by HPLC and two dimensional thin layer chromatography of tryptic digests. The present studies of actin assembly in intact PMNs confirm that NAD is associated with a true defect in PMN actin assembly and is a genetic disorder that is recessively inherited.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Amrein P. C., Stossel T. P. Prevention of degradation of human polymorphonuclear leukocyte proteins by diisopropylfluorophosphate. Blood. 1980 Sep;56(3):442–447. [PubMed] [Google Scholar]
- Boxer L. A., Hedley-Whyte E. T., Stossel T. P. Neutrophil action dysfunction and abnormal neutrophil behavior. N Engl J Med. 1974 Nov 21;291(21):1093–1099. doi: 10.1056/NEJM197411212912101. [DOI] [PubMed] [Google Scholar]
- Böyum A. Isolation of mononuclear cells and granulocytes from human blood. Isolation of monuclear cells by one centrifugation, and of granulocytes by combining centrifugation and sedimentation at 1 g. Scand J Clin Lab Invest Suppl. 1968;97:77–89. [PubMed] [Google Scholar]
- Carson M., Weber A., Zigmond S. H. An actin-nucleating activity in polymorphonuclear leukocytes is modulated by chemotactic peptides. J Cell Biol. 1986 Dec;103(6 Pt 2):2707–2714. doi: 10.1083/jcb.103.6.2707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elder J. H., Pickett R. A., 2nd, Hampton J., Lerner R. A. Radioiodination of proteins in single polyacrylamide gel slices. Tryptic peptide analysis of all the major members of complex multicomponent systems using microgram quantities of total protein. J Biol Chem. 1977 Sep 25;252(18):6510–6515. [PubMed] [Google Scholar]
- GOTTLIEBLAU K. S., WASSERMAN L. R., HERBERT V. RAPID CHARCOAL ASSAY FOR INTRINSIC FACTOR (IF), GASTRIC JUICE UNSATURATED B12 BINDING CAPACITY, ANTIBODY TO IF, AND SERUM UNSATURATED B12 BINDING CAPACITY. Blood. 1965 Jun;25:875–884. [PubMed] [Google Scholar]
- HIRS C. H. The oxidation of ribonuclease with performic acid. J Biol Chem. 1956 Apr;219(2):611–621. [PubMed] [Google Scholar]
- Harris H. Microfilament dynamics: few answers but many questions. 1987 Nov 26-Dec 2Nature. 330(6146):310–311. doi: 10.1038/330310a0. [DOI] [PubMed] [Google Scholar]
- Howard T. H., Oresajo C. O. The kinetics of chemotactic peptide-induced change in F-actin content, F-actin distribution, and the shape of neutrophils. J Cell Biol. 1985 Sep;101(3):1078–1085. doi: 10.1083/jcb.101.3.1078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Korn E. D. Actin polymerization and its regulation by proteins from nonmuscle cells. Physiol Rev. 1982 Apr;62(2):672–737. doi: 10.1152/physrev.1982.62.2.672. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Lazarides E., Lindberg U. Actin is the naturally occurring inhibitor of deoxyribonuclease I. Proc Natl Acad Sci U S A. 1974 Dec;71(12):4742–4746. doi: 10.1073/pnas.71.12.4742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mandell B. F., Ohliger D., Rella J. Rapid simultaneous assessment of neutrophil superoxide generation and lysosomal enzyme release. J Immunol Methods. 1987 Jun 26;100(1-2):211–214. doi: 10.1016/0022-1759(87)90191-8. [DOI] [PubMed] [Google Scholar]
- O'Farrell P. H. High resolution two-dimensional electrophoresis of proteins. J Biol Chem. 1975 May 25;250(10):4007–4021. [PMC free article] [PubMed] [Google Scholar]
- Shalit M., Dabiri G. A., Southwick F. S. Platelet-activating factor both stimulates and "primes" human polymorphonuclear leukocyte actin filament assembly. Blood. 1987 Dec;70(6):1921–1927. [PubMed] [Google Scholar]
- Southwick F. S., Stossel T. P. Isolation of an inhibitor of actin polymerization from human polymorphonuclear leukocytes. J Biol Chem. 1981 Mar 25;256(6):3030–3036. [PubMed] [Google Scholar]
- Stossel T. P., Chaponnier C., Ezzell R. M., Hartwig J. H., Janmey P. A., Kwiatkowski D. J., Lind S. E., Smith D. B., Southwick F. S., Yin H. L. Nonmuscle actin-binding proteins. Annu Rev Cell Biol. 1985;1:353–402. doi: 10.1146/annurev.cb.01.110185.002033. [DOI] [PubMed] [Google Scholar]
- White J. R., Naccache P. H., Sha'afi R. I. Stimulation by chemotactic factor of actin association with the cytoskeleton in rabbit neutrophils. Effects of calcium and cytochalasin B. J Biol Chem. 1983 Nov 25;258(22):14041–14047. [PubMed] [Google Scholar]