Abstract
BACKGROUND:
Secretory phospholipases A2 (sPLA2) initiate the biosynthesis of eicosanoids, are increased in the airways of people with severe asthma, and induce mucin hypersecretion. We used IL-13-transformed, highly enriched goblet cells and differentiated (ciliary cell-enriched) human bronchial epithelial cell culture to evaluate the relative contribution of ciliated and goblet cells to airway sPLA2 generation and response. We wished to determine the primary source(s) of sPLA2 and leukotrienes in human airway epithelial cells.
METHODS:
Human bronchial epithelial cells from subjects without lung disease were differentiated to a ciliated-enriched or goblet-enriched cell phenotype. Synthesis of sPLA2, cysteinyl leukotrienes (cysLTs), and airway mucin messenger RNA and protein was measured by real-time-polymerase chain reaction and an enzyme-linked immunosorbent assay, and the localization of mucin and sPLA2 to specific cells types was confirmed by confocal microscopy.
RESULTS:
sPLA2 group IIa, V, and X messenger RNA expression was increased in ciliated-enriched cells (P < .001) but not in goblet-enriched cells. sPLA2 were secreted from the apical (air) side of ciliated-enriched cells but not goblet-enriched cells (P < .001). Immunostaining of sPLA2 V was strongly positive in ciliated-enriched cells but not in goblet-enriched cells. sPLA2 released cysLTs from goblet-enriched cells but not from ciliated-enriched cells, and this result was greatest with sPLA2 V (P < .05). sPLA2 V increased goblet-enriched cell mucin secretion, which was inhibited by inhibitors of lipoxygenase or cyclooxygenase (P < .02).
CONCLUSIONS:
sPLA2 are secreted from ciliated cells and appear to induce mucin and cysLT secretion from goblet cells, strongly suggesting that airway goblet cells are proinflammatory effector cells.
Phospholipases A2 are a group of enzymes that hydrolyze fatty acids, including arachidonic acid, from the sn-2 position of glycerophospholipids, providing the substrate for the synthesis of leukotrienes, prostaglandins, platelet-activating factor, and lysophospholipids.1 More than one-third of the mammalian phospholipases A2 enzymes belong to the secretory phospholipases A2 (sPLA2) family.2 Human genes have been identified for 12 sPLA2 enzymes, which are subdivided into conventional groups (I, II, V, X, and otoconin-90) and two atypical groups (III and XII).3 sPLA2 are released in the plasma and BAL fluid (BALF) of patients with asthma,4 ARDS,5 and pneumonia.6 Of the three sPLA2 expressed in human lungs, group IIa and X enzymes are the major sPLA2 that are increased in the BALF from subjects with asthma.7 Gene silencing of either group V or X sPLA2 in mice with ovalbumin-induced asthma attenuates T-helper cell type 2 inflammation.8‐10 We have reported that sPLA2 stimulate mucus hypersecretion in the ferret trachea through activation of the lipoxygenase (LO) pathway.11
Mucus obstruction of airways and goblet cell hyperplasia are characteristic of severe asthma.12,13 IL-13 is increased in asthma and induces goblet cell hyperplasia with mucus hypersecretion in vivo and in vitro.13‐16 MUC5AC and MUC5B are the predominant gel-forming mucins in the human airway. MUC5AC is expressed primarily by goblet cells of the surface epithelium.17 IL-13 increases MUC5AC-expressing goblet cells in the airways. MUC5AC secretion is also increased during asthmatic inflammation18; even in mild asthma, there are increases in airway goblet cell number and stored and secreted MUC5AC protein.13
Several sPLA2 isoforms are expressed and released by inflammatory cells.19 In addition to inflammatory cells, the epithelial cells lining the respiratory tract may be an important source of sPLA2.6,9,20‐22 It has not been shown which airway epithelial cells produce sPLA2, or which cells types respond to sPLA2 by producing leukotrienes. We hypothesized that airway goblet cells act as immune effector cells and that inflammatory mediators released from goblet cells may act in an autocrine manner, which, in turn, contributes to the severity of asthma. Therefore, we studied the production and secretion of sPLA2 isotypes from differentiated human bronchial epithelial (HBE) cells in ciliated cell-enriched culture or IL-13-differentiated goblet cell-enriched cultures. We also evaluated eicosanoid and mucin synthesis after exposure to specific sPLA2 subtypes in these same HBE cell culture systems.
Materials and Methods
Reagents
Recombinant human IL-13 was obtained from R&D Systems; anti-MUC5AC antibody from Lab Vision Corporation; anti MUC5B antibody from Santa Cruz Biotechnology; anti-sPLA2 group V from the Cayman Chemical Company; acetylated α-tubulin antibody from Cell Signaling Technology; recombinant human sPLA2 group IIa, V, or X from BioVendor LLC; and MK-886 from Calbiochem. Indomethacin, quercetin, and other reagents were purchased from Sigma-Aldrich unless otherwise indicated.
Culture and Differentiation of HBE Cells
The cultivation of HBE cells and differentiation at an air-liquid interface (ALI) have been described previously.12,23‐25 HBE cells (Clonetics; Lonza Group Ltd) were plated at 3,500 cells/cm2 in small-airway epithelial cell growth medium (Clonetics; Lonza Group Ltd) and cultured at 37°C in a 5% CO2 incubator. Second-passage HBE cells were seeded at a density of 2.0 × 105/cm2 onto polyester inserts (6.5-mm diameter, 0.4-μm pore size, and 10-μm thickness; Costar Transwell Clear), coated with type 1 rat tail collagen (Sigma), and then cultured in serum-free DMEM/F12 medium containing ITS-A (1.0%; Invitrogen Ltd), triiodothyronine (10 ng/mL; ICN Biochemicals), epidermal growth factor (recombinant human EGF, 0.5 ng/mL; Invitrogen Ltd), all-trans retinoic acid (10−7 M; Sigma), hydrocortisone (0.5 μg/mL; ICN Biochemicals), bovine serum albumin (2.0 μg/mL; Sigma), bovine pituitary extract (30 μg/mL; Invitrogen Ltd), and antibiotic-antimycotic (1.0%; Invitrogen Ltd). Culture medium was added to both the apical and the basolateral side of the inserts, and the cells were cultured at submerged condition in the medium. The culture medium was changed every 2 days. Confluence was reached within 5 days, and the cells were then cultured at ALI over 14 days after removing the apical medium and feeding from the basolateral side only. The culture medium was changed every 2 days, and the cells were maintained in an incubator under a humidified 5% CO2 atmosphere at 37°C.
IL-13 Exposure
HBE cells were grown from day 0 with IL-13 at an ALI. The culture medium with IL-13 or phosphate-buffered saline (PBS) was changed every 48 h, and HBE cells were cultured for 14 days. To evaluate the effects of IL-13, after withdrawing IL-13 for 24 h on day 15, IL-13-transformed highly enriched goblet cells or ciliated-enriched cells were exposed to IL-13 or PBS for 24 h on day 16 (Fig 1B).
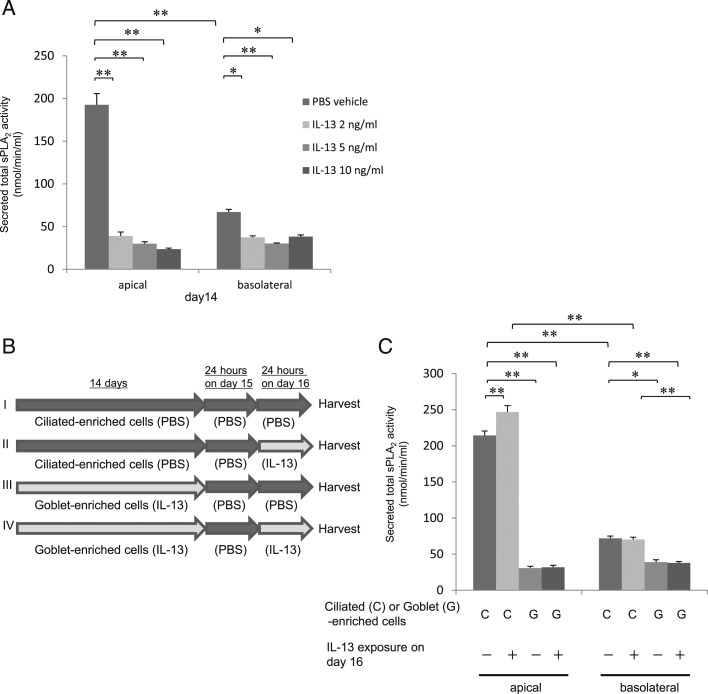
Figure 1 –
A, sPLA2 are secreted primarily from ciliated cells. Secreted total sPLA2 activity from apical supernatant and basolateral culture media in human bronchial epithelial (HBE) cells with basal exposure to IL-13 (0, 2, 5, or 10 ng/mL). B, HBE cells were cultured for 14 d with PBS to produce a ciliated-enriched phenotype (I, II) or with IL-13 to produce goblet-enriched cells (III, IV). After washing the cells and withdrawing IL-13 for 24 h on d 15 (I-IV), the cell cultures were then exposed to IL-13 at 0 (PBS; I, III) or 5 ng/mL (II, IV) for 24 h to evaluate the direct effects of IL-13 on d 16. We measured sPLA2 activity in apical supernatant and basolateral culture media from these cells. C, IL-13 did not inhibit sPLA2 secretion from either cell type. Secreted sPLA2 activity was measured in apical supernatant and basolateral culture media from ciliated cell- or goblet cell-enriched culture after basal exposure to IL-13 at 0 or 5 ng/mL for 24 h. Data are presented as the mean ± SEM from four samples. Significant differences are indicated by **P < .001 and *P < .01. PBS = phosphate-buffered saline; sPLA2 = secretory phospholipases A2.
Real-Time-Polymerase Chain Reaction
To examine the messenger RNA (mRNA) expression of MUC5AC, MUC5B, or sPLA2 group IIa, V, or X isotypes, RNA was extracted from the cells 5, 10, or 14 days after culturing with IL-13 or PBS.12,23‐25 The apical side of the cells was washed three times with PBS, and total RNA from the cells was extracted using the iScript RT-qPCR sample preparation reagent (Bio-Rad). The total RNA was then used to synthesize the first-strand complementary DNA using the iScript cDNA synthesis kit (Bio-Rad). Real-time-polymerase chain reaction (PCR) was performed on the C 1000 thermal cycler equipped with the CFX96 real-time PCR system (Bio-Rad). For the relative quantification of MUC5AC, MUC5B, or sPLA2 group IIa, V, or X mRNA expression, the mRNA expression of glyceraldehyde-3-phosphate dehydrogenase (GAPDH) served as an internal control. EvaGreen was used as a DNA intercalator dye to monitor amplified DNA quantification, and real-time quantitative PCR curves were analyzed by CFX Manager software (Bio-Rad) to obtain threshold cycle values for each sample. The mRNA expression level was calculated based on a generated standard curve.
The following primers were used6,12,23,26:
• MUC5AC forward: 5′-TACTCCACAGACTGCACCAACTG-3′
• MUC5AC reverse: 5′-CGTGTATTGCTTCCCGTCAA-3′
• MUC5B forward: 5′-CACATCCACCCTTCCAAC-3;
• MUC5B reverse: 5′-GGCTCATTGTCGTCTCTG-3′
• sPLA2 subgroup IIa forward: 5′-ATGAAGACCCTCCTACTGTTGGC-3′
• sPLA2 subgroup IIa reverse: 5′-TCAGCAACGAGGGGTGCTCCCTC-3′
• sPLA2 subgroup V forward: 5′-ATGAAAGGCCTCCTCCCACTGGC -3′
• sPLA2 subgroup V reverse: 5′-GGCCTAGGAGCAGAGGATGTTGG -3′
• sPLA2 subgroup X forward: 5′-ATGGGGCCGCTACCTGTGTGCC-3′
• sPLA2 subgroup X reverse: 5′-TCAGTCACACTTGGGCGAGTCC -3′
• GAPDH forward: 5′-TGAACGGGAAGCTCACTGG-3′
• GAPDH reverse: 5′-TCCACCACCCTGTTGCTGTA-3′
Histochemical Analysis
The cells on porous filters were fixed in 10% formalin neutral buffer, embedded in paraffin, and cut into 8-μm slices. To examine morphology, hematoxylin and eosin staining and periodic acid-Schiff (PAS) staining were performed as described previously.12,23,25 Cell morphology was assessed with a light microscope (CKX41; Olympus) and photographed using a digital camera system (AxioCam ICc 1; ZEISS).
Immunohistochemical Analysis
MUC5AC, MUC5B, or sPLA2 V expression was evaluated by immunohistochemistry. The sections were stained with anti-MUC5AC antibody, anti-MUC5B antibody, or anti-sPLA2 group V antibody as the first antibody. EnVisionTM + Dual Link System-HRP (Dako) was added as the second antibody. Antigen-antibody complexes were visualized using the Liquid DAB + Substrate Chromogen System (Dako).12,23,25
Immunofluorescent Confocal Microscopy Analysis
Acetylated α-tubulin, MUC5AC, and sPLA2 group V were identified in HBE cells using immunofluorescence techniques and confocal microscopy.27,28 For the apical surface view, cultured HBE cell membranes were cut from the insert using a scalpel and were fixed in 3% formaldehyde. For the z-stack view, the cell membrane was embedded in Tissue-Tek OCT Compound (Sakura FineTechnical Co Ltd), frozen in liquid nitrogen, and stored at –80°C. The frozen samples were cut into 8-μm slices using a cryostat at –20°C, mounted on slides, and warmed to room temperature before fixation in 3% formaldehyde. The secondary antibodies were goat anti-mouse Alexa Fluor 488 (Invitrogen Ltd) for MUC5AC or sPLA2 group V and goat anti-rabbit Alexa Fluor 568 (Invitrogen Ltd) for acetylated α-tubulin. After incubation with the primary and secondary antibodies, the slides were rinsed with PBS three times, and nuclei were stained with DAPI (Invitrogen Ltd) for 1 min in the dark. The slides were again rinsed three times with PBS and were immersed in VECTASHIELD Mounting Medium for fluorescence (Vector Laboratories). Coverslips were applied, and the slides were stored at 4°C in the dark. Within 24 h of staining, the slides were examined by confocal laser scanning microscopy (LSM 700; ZEISS). Immunostaining was visualized with a blue diode, argon ion, and helium neon laser using 405-, 488-, and 568-nm laser lines, respectively.
Enzyme-Linked Immunosorbent Assay for MUC5AC and MUC5B Mucins in Cell Supernatants
Apically secreted MUC5AC or MUC5B protein was measured using an enzyme-linked immunosorbent assay.12,23,29 The cells were grown at ALI with either PBS or IL-13 for 5, 10, or 14 days. Culture supernatants were collected by adding Hanks’ Balanced Salt Solution (Lonza Group Ltd) to the apical side of the inserts. The 96-well plates (Becton, Dickinson and Company) were coated with cell supernatant samples (50 μL) and bicarbonate-carbonate buffer (50 μL) and were incubated at 37°C overnight until samples dried. After washing, 2% bovine serum albumin/PBS containing 0.05% Tween20 was added to each well and incubated at room temperature for 1 h as blocking. After washing with Tween-PBS, anti-MUC5AC antibody or anti-MUC5B antibody was added and the plate was incubated for 2 h at room temperature. After washing, anti-mouse immunoglobulin, horseradish peroxidase-linked whole antibody (GE Healthcare) for MUC5AC or anti-rabbit immunoglobulin HRP-linked antibody (Cell Signaling Technology) for MUC5B was added, and the plate was incubated for 1 h at room temperature. After washing, 3,3′,5,5′-tetramethylbenzine peroxidase solution was added, and the plate was incubated at room temperature. The reaction was stopped with 2 N H3PO4 and the absorbance was measured at 450 nm with the ELx808 Ultra Microplate Reader (Bio Tek Instruments, Inc). Data were expressed as the percentage above the PBS control.
Assay for sPLA2, Leukotriene B4, Leukotriene C4, and Prostaglandin E2
Differentiated cells were incubated with PBS or IL-13 for 5, 10, or 14 days at ALI. The culture supernatant from each well was collected by adding Hanks’ Balanced Salt Solution to the apical side of the inserts, and the culture media were collected from the basolateral side of the inserts.
The activity of sPLA2 and the concentrations of leukotriene B4 (LTB4), leukotriene C4 (LTC4), and prostaglandin E2 (PGE2) in the apical culture supernatant and the basal culture media were measured with an sPLA2 assay kit and an LTB4, LTC4, or PGE2 EIA kit (Cayman Chemical Company) using high performance liquid chromatography-grade water according to the manufacturer’s instructions. The absorbance was measured with the ELx808 Ultra Microplate Reader (Bio Tek Instruments, Inc). The detection range of the sPLA2 assay is approximately 20 to 200 nmol/min/mL. LTC4, LTB4, and PGE2 EIA have detection limits of approximately 10, 13, and 15 pg/mL, respectively.
Evaluation of Secretory Pathways
Inhibitors were used to evaluate potential mechanisms for sPLA2-induced MUC5AC and cysteinyl leukotriene (cysLT) secretion. Quercetin (a nonspecific LO inhibitor), indomethacin (a cyclooxygenase inhibitor), and MK-886 (a specific 5-LO [5-LO] inhibitor) at 0, 0.1, 1, or 10 μM were used. Cells were cultured for 14 days with 5 ng/mL of IL-13 at ALI, and on day 14 were first exposed to an inhibitor or vehicle for 30 min and then for 60 min or 24 h, adding recombinant sPLA2 group V, 0.5 μg/mL, to the apical side for MUC5AC or cysLT measurement.
Statistical Analysis
Data are expressed as mean ± SEM. Comparisons were made by one-way analysis of variance, except for time-course experiments (Figs 2, 3, 4, 5B), which were analyzed by two-way repeated-measures analysis of variance. Conventionally, adjusted P < .05 was considered significant.
Figure 2 –
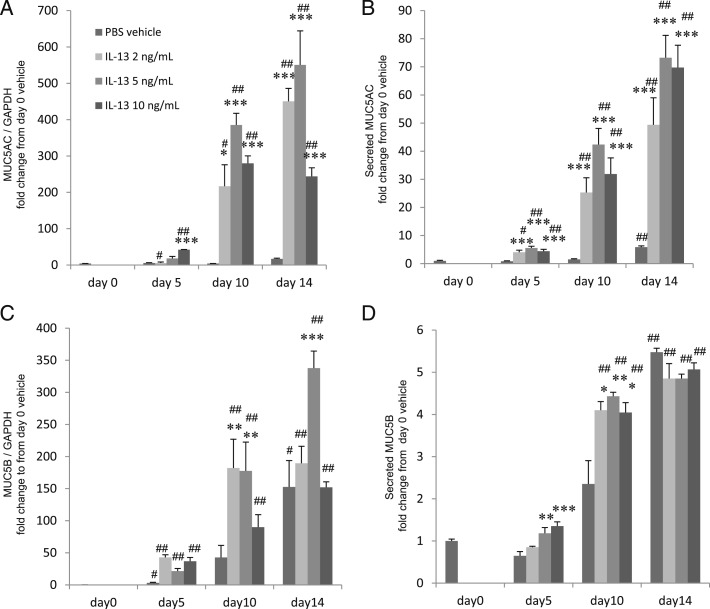
Mucins are secreted from IL-13-transformed goblet cells. MUC5AC or MUC5B messenger RNA (mRNA) expression in cell lysates and MUC5AC or MUC5B protein in supernatants of HBE cells grown with basal exposure to IL-13 (0, 2, 5, or 10 ng/mL) for 0, 5, 10, or 14 d. A, MUC5AC mRNA expression. B, MUC5AC protein. C, MUC5B mRNA expression. D, MUC5B protein. Data are presented as the mean ± SEM from four samples. Significant differences from PBS at each date are indicated by *P < .05, **P < .02, and ***P < .01, and from d 0 are indicated by #P < .05 and ##P < .01. GAPDH = glyceraldehyde-3-phosphate dehydrogenase. See Figure 1 legend for expansion of other abbreviations.
Figure 3 –
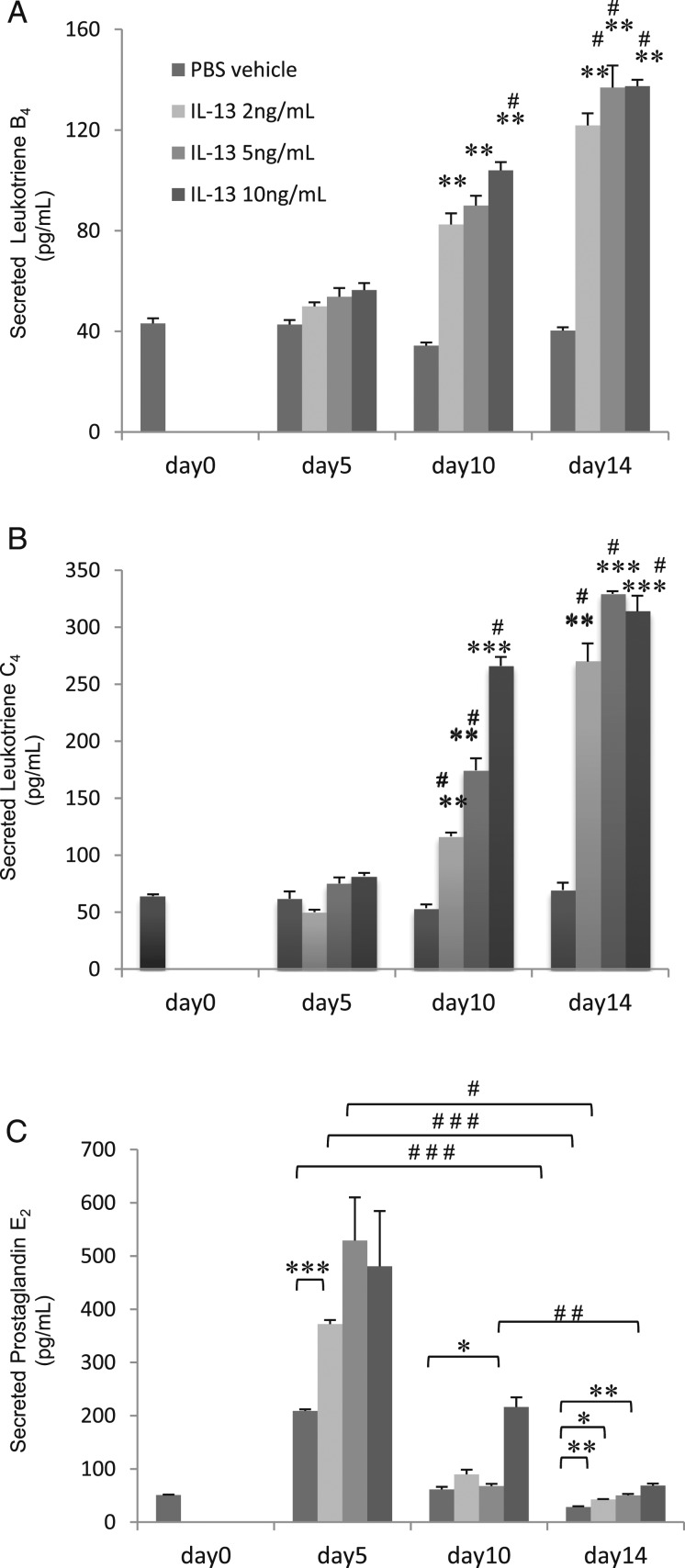
Leukotriene B4 (LTB4), leukotriene C4 (LTC4), and prostaglandin E2 are secreted primarily from goblet cell-enriched culture. TB4, LTC4, or prostaglandin E2 in supernatants of HBE cells grown for 5, 10, or 14 days with basal exposure to IL-13 (0, 2, 5, or 10 ng/mL). Data are presented as the mean ± SEM from four samples. A, LTB4. B, LTC4. Significant differences from control subjects (no IL-13) at each date are indicated by **P < .01, ***P < .001, and from d 0 are indicated by #P < .01. C, Prostaglandin E2. Significant differences are indicated by #P < .05, ##P < .01, and ###P < .001, and from control subjects at each date are indicated by *P < .05, **P < .01, and ***P < .001. See Figure 1 legend for expansion of abbreviations.
Figure 4 –
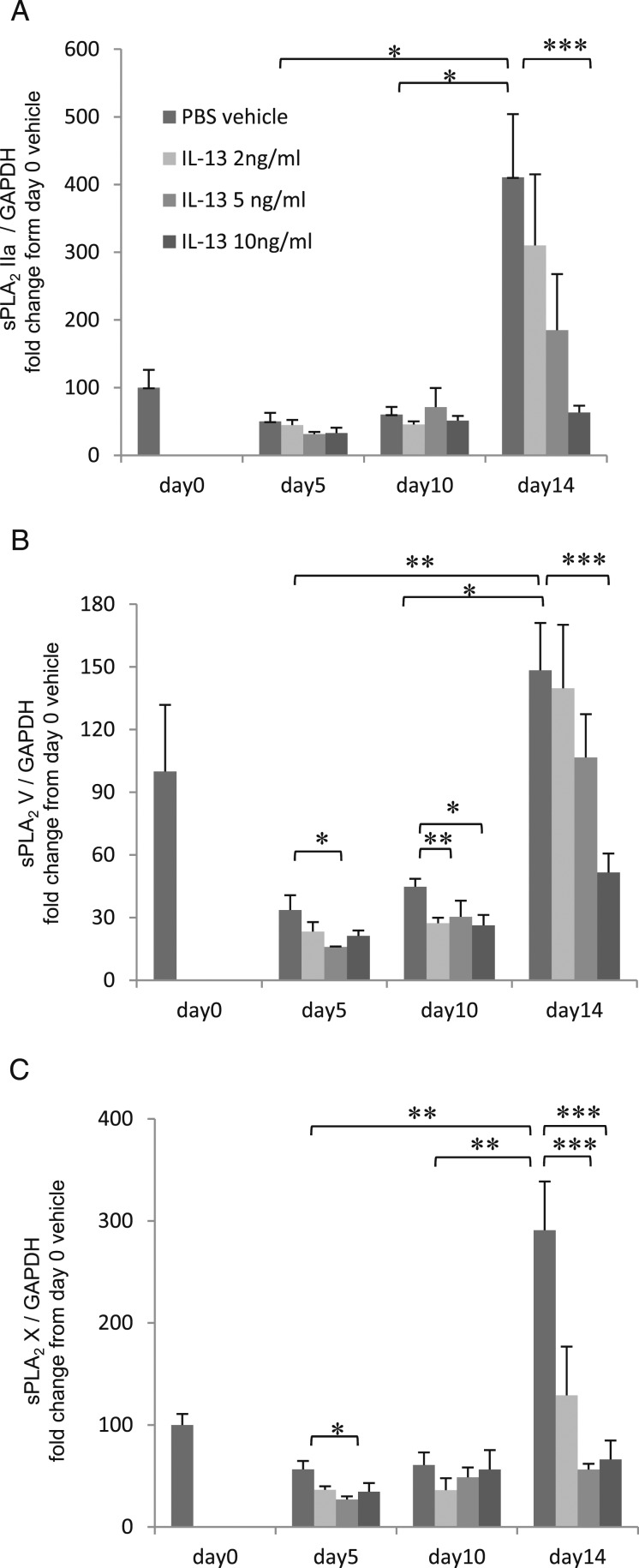
sPLA2 are expressed primarily from ciliated cells. sPLA2 group IIa, V, and X mRNA expression in cell lysates from HBE cells with basal exposure to IL-13 (0, 2, 5, or 10 ng/mL). The results are the mean ± SEM from six samples. A, sPLA2 IIa. B, sPLA2 V. C, sPLA2 X. Significant differences are indicated by *P < .05, **P < .01, and ***P < .001. See Figure 1 and 2 legends for expansion of abbreviations.
Figure 5 –
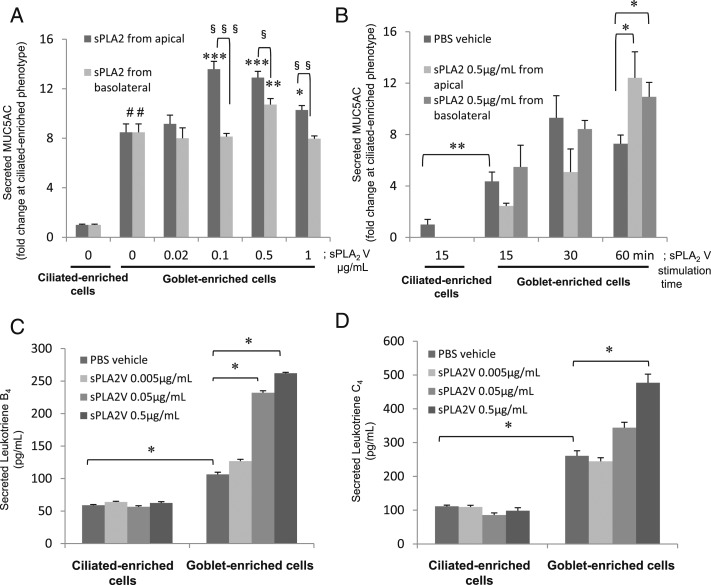
sPLA2 V stimulates MUC5AC, LTB4, and LTC4 secretion from goblet cells, but not ciliated cells. A and B, Apical and basolateral sPLA2 V increased MUC5AC secretion from goblet-enriched cell culture. MUC5AC protein was measured in supernatants from ciliated or goblet cells. A, Apical or basal 60-min exposure to sPLA2 V at 0, 0.02, 0.1, 0.5, or 1 μg/mL. Data are presented as mean ± SEM from five samples. #P < .001 for ciliated cells, *P < .05, **P < .02, ***P < .01 for goblet cells compared with PBS, §P < .02, §§P < .01, §§§P < .001. B, Apical or basal 15-, 30-, or 60-min exposure to sPLA2 V (0 or 0.5 μg/mL). Data are presented as mean ± SEM from five samples. *P < .05, **P < .01. C and D, Apical sPLA2 V stimulates LTB4 and LTC4 secretion from goblet and ciliated-enriched cells. Secreted LTB4 or LTC4 in supernatants of ciliated-enriched and goblet-enriched cells stimulated for 24 h with apical sPLA2 (0, 0.005, 0.05, or 0.5 μg/mL). C, LTB4. D, LTC4. Data are presented as mean ± SEM from four samples. *P < .01. See Figure 1 and 3 legends for expansion of abbreviations.
Results
IL-13-Induced Goblet Cell Differentiation
We have reported previously that IL-13-exposed HBE cells transformed to a highly enriched goblet cell epithelial culture with MUC5AC protein covering the apical surface but that cilia were not seen and that IL-13 increased the number of total cells, goblet cells, PAS-positive cells, and MUC5AC-positive cells.12,29 IL-13 increased the total number of cells (hyperplasia) primarily by increasing the number of goblet cells (from 0.2 to 15.9 cells), PAS-positive cells (from 2.5 to 28.2 cells), and MUC5AC-positive cells (from 0.1 to 25.7 cells/250 μm on slide).30
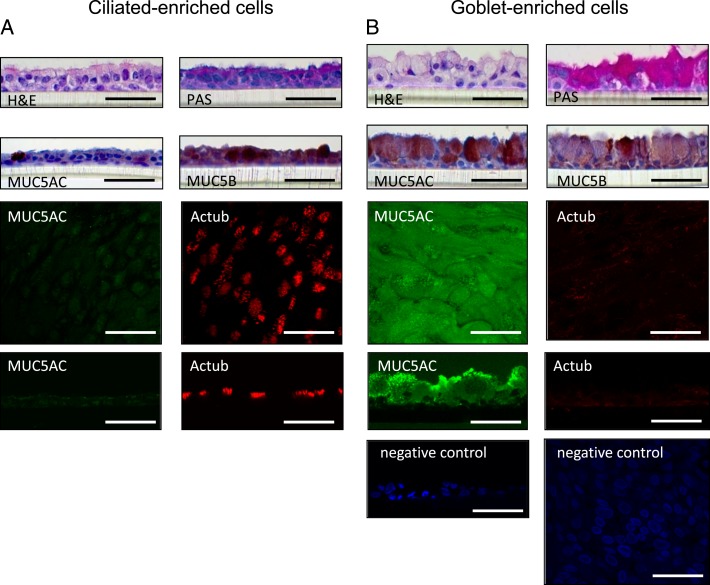
HBE cells cultured at ALI without IL-13 demonstrated a well-differentiated morphology with ciliated cells at the surface of epithelial layers, referred to as ciliated-enriched cells for convenience.30 These cells weakly stain with MUC5B, PAS, and MUC5AC, and strongly stain with acetylated α-tubulin. In the presence of 5 ng/mL IL-13, HBE cells differentiated into a predominantly goblet cell phenotype with secretory granules (similarly referred to as goblet-enriched cells) strongly stain with PAS, MUC5AC, and MUC5B, but not with acetylated α-tubulin. The apical surface of ciliated cells is shown in red immunofluorescence using acetylated α-tubulin and goblet cells in green using MUC5AC. Images show predominantly goblet cells with few or no ciliated cells after growth of 5 ng/mL IL-13. The ciliated cells in culture do not appear to stain for PAS or MUC5B antibodies. Cells grown in the absence of IL-13 supplementation transform predominantly into a ciliated phenotype, with the occasional goblet cell appearing in the epithelial culture. The ciliated cells themselves stain with antibodies to tubulin, but not with PAS or MUC5B. Images taken at the cell surface, where the antibody was placed, show a small amount of nonspecific staining (Fig 6).
Figure 6 –
A, B, HBE cells were differentiated to ciliated cell-enriched or to goblet cell-enriched phenotypes. H&E staining, PAS staining, MUC5AC immunohistochemical staining, MUC5B immunohistochemical staining, and immunofluorescence with laser confocal microscopy of HBE cells grown for 14 d at an air-liquid interface with 0 or 5 ng/mL IL-13. Representative image of apical surface and z-stack immunofluorescence shows MUC5AC (Alexa Fluor 488, green), acetylated Actub (Alexa Fluor 568, red), and nuclei (DAPI, blue). Bar = 50 μm. Actub = α-tubulin; H&E = hematoxylin and eosin; PAS = periodic acid-Schiff. See Figure 1 legend for expansion of other abbreviations.
Mucin mRNA Expression and Protein Secretion From Goblet Cells
As we reported previously,12,23,29,30 mucin (MUC5AC, MUC5B) mRNA expression and protein secretion increased with IL-13 concentration (0, 2, 5, or 10 ng/mL) after 14 days in culture at ALI (Fig 2).
LTB4, LTC4, and PGE2 Secretion From Goblet Cells
LTB4 and LTC4 secretion in apical supernatants was increased with IL-13 (2, 5, or 10 ng/mL) after 14 days in culture compared with ciliated enriched cells grown without IL-13 (Figs 3A, 3B) (LTB4, 137 ± 8.7 pg/mL, and LTC4, 329 ± 2.4 pg/mL, with 5 ng/mL of IL-13; P < .01). PGE2 secretion in apical supernatants was decreased with exposure to IL-13 (2, 5, or 10 ng/mL) at 14 days compared with day 5 at each concentration of IL-13 (Fig 3C) (P < .01).
sPLA2 mRNA Expression and Secretion in Ciliated Cells
After 14 days of culture at ALI, sPLA2 group IIa (Fig 4A), V (Fig 4B), or X (Fig 4C) mRNA expression was greater in ciliated-enriched cells than in goblet-enriched cells (P < .001), and sPLA2 secretion was greater from ciliated-enriched cells than from goblet-enriched cells (Fig 1A) (P < .001). The increased sPLA2 secretion from ciliated-enriched cells was greater in apical supernatant than in basolateral culture medium (Fig 1A) (P < .001). IL-13 (5 ng/mL) exposure for 24 h at the end of 14 days (after withdrawing IL-13 for 24 h) did not inhibit sPLA2 secretion from ciliated-enriched cells or goblet-enriched cells, but rather, modestly increased sPLA2 secretion in apical supernatant from ciliated-enriched cells (P < .001).
sPLA2 Stimulate MUC5AC, LTB4, and LTC4 Secretion in Goblet-Enriched Cells
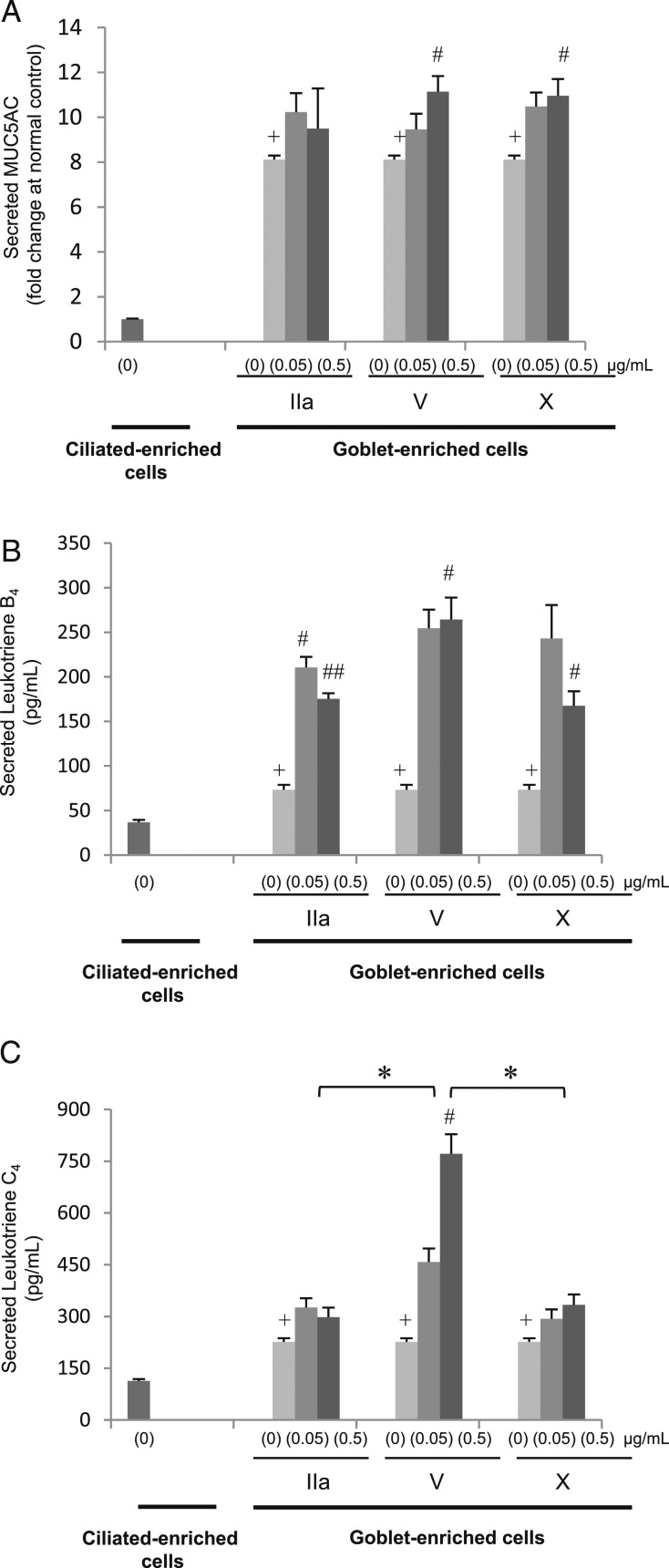
When goblet-enriched cells were apically exposed to sPLA2 group V or X (0.5 μg/mL), this increased MUC5AC secretion compared with the sPLA2 vehicle (PBS) (Fig 7A) (P < .05). LTB4 (Fig 7B) and LTC4 (Fig 7C) secretion was increased in goblet-enriched cells compared with ciliated-enriched cells (P < .01). Apical exposure to sPLA2 group IIa (0.05 or 0.5 μg/mL), V, or X (0.5 μg/mL) increased LTB4 in goblet-enriched cells (Fig 7B) (P < .05), and apical exposure to 0.5 μg/mL sPLA2 group V increased LTC4 release compared with PBS (Fig 7C) (P < .05). Group V produced a greater increase than did group II or X (Fig 7C) (P < .05). sPLA2 group IIa, V, and X, at 0.05 or 0.5 μg/mL had no effect on MUC5AC, LTB4, or LTC4 secretion from ciliated-enriched cells (data not shown).
Figure 7 –
sPLA2 stimulates MUC5AC, LTB4, and LTC4 secretion from goblet cells. Apical sPLA2 subgroups IIa, V, or X with 0 (PBS), 0.05, or 0.5 μg/mL stimulate MUC5AC, LTB4, and LTC4 secretion from goblet cell-enriched culture. Secreted MUC5AC, LTB4, and LTC4 in apical supernatants from goblet cells or ciliated cells that were apically exposed for 60 min or 24 h with sPLA2 IIa, V, X, or PBS. A, MUC5AC; 60 min. B, LTB4; 24 h. C, LTC4; 24 h. Data are presented as the mean ± SEM from four samples. +P < .01 to ciliated cells with PBS; #P < .05, ##P < .01 to goblet cells with PBS; *P < .05. See Figure 1 and 3 legends for expansion of abbreviations.
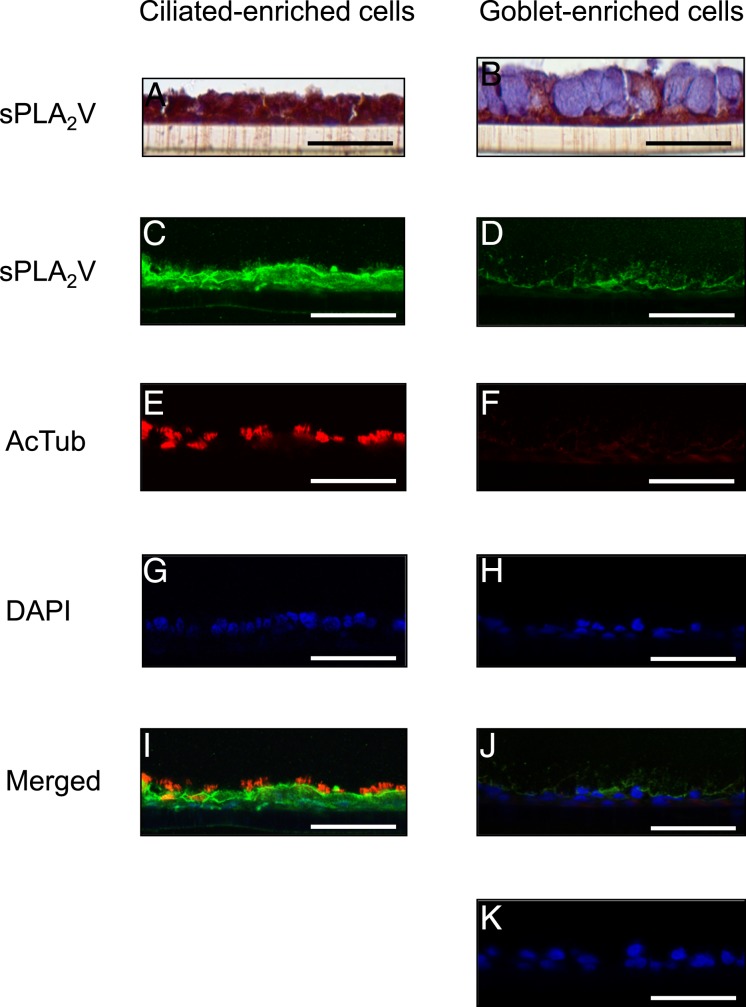
sPLA2 Group V Is Expressed in Ciliated-Enriched Cells and Causes Goblet-Enriched Cells to Secrete MUC5AC, LTB4, and LTC4
HBE cells cultured to a ciliated cell morphology (Figs 8A, 8E, 8I) strongly stain with sPLA2 V antibody (Figs 8A, 8C, 8I). However, with differentiation into goblet cells, there was some sPLA2 V antibody staining in the cytoplasm but not in the mucous granules (Figs 8B, 8D, 8J). In these goblet-enriched cells, a 60-min apical exposure to sPLA2 V (0.1, 0.5, or 1 μg/mL) or 0.5 μg/mL exposure at the basolateral side increased MUC5AC secretion (Fig 5A) (P < .01 except for P < .02 at 1 μg/mL of apical sPLA2 V and P < .05 at 0.5 μg/mL of basolateral sPLA2 V). Apical exposure to sPLA2 V increased LTB4 and LTC4 secretion in goblet-enriched cells but not in ciliated-enriched cells (Figs 5C, 5D) (P < .01 for LTB4 at 0.05 or 0.5 μg/mL of sPLA2 V and LTC4 at 0.5 μg/mL of sPLA2 V).
Figure 8 –
sPLA2 V is expressed in ciliated cells. sPLA2 V immunostaining and immunofluorescence with laser confocal microscopy of ciliated or goblet cell-enriched culture containing mucous granules grown for 14 d with PBS or with 5 ng/mL IL-13. A and B, Immunostaining. C-K, Immunofluorescence. A, C, E, G, I, PBS. B, D, F, H, J, K, IL-13. Immunofluorescence shows sPLA2 V (Alexa Fluor 488, green; C, D), acetylated α-tubulin (AcTub, Alexa Fluor 568, red; E, F), nuclei (DAPI, blue; G, H), and merged image of the double staining and DAPI (I, J). K, Negative control is HBE cells without the primary antibodies. Bar = 50 μm. See Figure 1 and 6 legends for expansion of abbreviations.
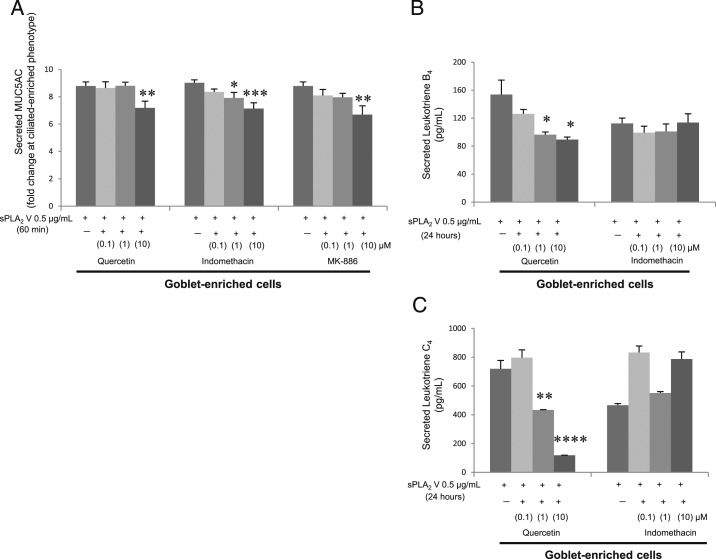
Inhibiting Either Cyclooxygenase or LO Decreases sPLA2 V-Stimulated MUC5AC Secretion
Quercetin 10 μM (an LO inhibitor), indomethacin 1 and 10 μM (a cyclooxygenase inhibitor), and MK-886 10 μM (a specific 5-LO inhibitor) all decreased sPLA2 V-induced MUC5AC secretion from goblet-enriched cells (Fig 9A) (P < .02 at 10 μM of quercetin, P < .05 at 1 μM, P < .01 at 10 μM of indomethacin, P < .02 at 10 μM of MK-886). Although quercetin (0.1, 1, or 10 μM) dose dependently decreased sPLA2 V-induced LTB4 and LTC4 secretion, indomethacin did not affect LTB4 or LTC4 secretion (Figs 9B, 9C).
Figure 9 –
Inhibiting either cyclooxygenase or lipoxygenase decreases sPLA2 V-stimulated MUC5AC secretion from goblet cells. Secreted MUC5AC, LTB4, and LTC4 in apical supernatants of goblet cells apically stimulated for 60 min or 24 h to sPLA2 V with quercetin, indomethacin, or MK-886 (0.1, 1, or 10 μM). A, MUC5AC; 60 min. B, LTB4; 24 h. C, LTC4; 24 h. Data are presented as mean ± SEM from four samples. *P < .05, **P < .02, ***P < .01, or ****P < .001 to quercetin, indomethacin, or MK-886 vehicle (dimethyl sulfoxide). See Figure 1 and 3 legends for expansion of abbreviations.
Discussion
sPLA2 are increased in the plasma and BALF of subjects with asthma,4 and these enzymes increase cysLTs in the asthmatic airway. LTC4, leukotriene D4, and leukotriene E4 cause bronchoconstriction, edema, mucus hypersecretion, and eosinophil chemotaxis, and LTB4 is a potent neutrophil chemoattractant.31 LTB4 and LTC4 are produced in bronchial epithelial cell culture,32 and other epithelial cell types release LTB4 and LTC4 after inflammatory injury.33,34 An epithelial cell source of cysLT may contribute to neutrophil migration in the airway.35
We have shown previously that sPLA2 dramatically increases mucus secretion from the ferret trachea and we called this “secretory hyperresponsiveness.”11 Mucus accumulation and goblet cell hyperplasia in the airway are closely correlated with morbidity and mortality.36,37 The results of the current study suggest that sPLA2 from ciliated cells, perhaps acting as airway sensory receptors, can increase mucus secretion and production of inflammatory mediators from IL-13-transformed goblet cells. A 24-h exposure to IL-13 only modestly increased sPLA2 secretion from the ciliated-enriched cell culture and had no further effect on goblet cell culture.
sPLA2 can act on cellular membranes to release not only arachidonic acid, but also a variety of saturated, monosaturated, and polyunsaturated fatty acids and, in so doing, alter the function of the cell membrane. The physiologic functions of sPLA2 may include a combination of these actions.38
In antigen-challenged mice, sPLA2 V is increased in the bronchial epithelial cells, and the intratracheal application of anti-group V sPLA2 antibody can ameliorate airway inflammation.9 sPLA2 V and X in bronchial cells potentiate arachidonate release and prostaglandin synthesis.6 Group V and X sPLA2 are widely expressed in the airway epithelium of human lungs.6 sPLA2 have the ability to liberate arachidonic acid from cell membranes, leading to the production of eicosanoids, with groups V and X reported to be the most potent.39‐43 Although it remains uncertain how sPLA2 in the medium gains access to the inner leaflet of the plasma membrane, the ability of sPLA2 V and X to release cellular arachidonic acid correlates with their high capacity to bind phosphatidylcholine, a major phospholipid in the outer leaflet of the plasma membrane.41‐43
We showed that in HBE cells, sPLA2 group V stimulated LTC4 secretion significantly more than either group IIa or X. Immunohistochemical staining was positive for sPLA2 V in ciliated-enriched cells, but spared mucous granules. These results suggest that in the airway, group V sPLA2 production and secretion is predominantly from ciliated cells. Group V sPLA2 may contribute to mucus secretion and to cysLT generation in a paracrine fashion, acting on goblet cells and suggesting a role in the pathogenesis of asthma.
Arachidonic acid is a substrate for LO-generating eicosanoids, including leukotrienes.44 Quercetin, a LO inhibitor, decreased sPLA2-induced mucus secretion. Although quercetin has other effects as well, including antihistamine and antiinflammatory effects, we also show that MK-886, a specific 5-LO inhibitor, decreased sPLA2-induced mucus secretion, confirming earlier data that sPLA2 probably stimulates mucus secretion by activating the LO pathway. MK-886 binds a membrane-bound 5-LO-activating protein, thereby inhibiting the translocation and activation of 5-LO, LTC4,45 and leukotriene D4.46,47 These suggest that 5-LO products play a role in sPLA2-induced mucus secretion.
Prostaglandins also stimulate mucus secretion in the airways,48 stomach,49 colon,50 and uterus.51 We show that indomethacin, a cyclooxygenase inhibitor, decreased sPLA2-induced mucus secretion, suggesting that cyclooxygenase pathway products may play a role in sPLA2-induced mucus secretion.
PGE2 is an airway smooth muscle relaxant and may have bronchoprotective and antiinflammatory properties.52‐54 Although it is reported that IL-13 inhibits PGE2 synthetic pathways and decreases PGE2 secretion in HBE cells,55 a clinical study showed that cysLT and PGE2 concentrations in sputum are higher in those with asthma than in control subjects.56 PGE2 also promotes cell growth in bronchial epithelial cells.57 We showed that, consistent with this, PGE2 secretion was increased during the cell growth and differentiation phase of HBE cells. We also showed that IL-13 stimulates PGE2 production and secretion from goblet-enriched cells. Thus, it is possible that PGE2 or cysLT is released from goblet cells and acts in an autocrine manner to enhance sPLA2-stimulated mucin secretion.
It would have been interesting to evaluate the role of sPLA2 in primary cells from patients with asthma. Although the HBE cells used in this study were from subjects without lung disease, it has been reported that bronchial epithelial cell cultures from subjects with asthma contain many more goblet cells and fewer ciliated cells compared with those from healthy subjects.28
Conclusions
The findings in the current study suggest that sPLA2 are released from ciliated airway epithelial cells and act on goblet cells in a paracrine manner to stimulate LTB4, LTC4, and MUC5AC secretion. We reported previously that IL-33 acts directly on goblet-enriched cells, but not ciliated-enriched cells, to stimulate IL-8 production, suggesting that beyond secreting mucus, goblet cells are inflammatory effector cells.30 These data provide additional further support that goblet cells can act as inflammatory effector cells in the airway, and this may contribute to the severity of asthma and other airway diseases.
Acknowledgments
Author contributions: T. T. is the guarantor of the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. T. T. and B. K. R. contributed to the study concept and design; T. T. contributed to the conducting of the experiments and the preparation of the manuscript and the figures; T. S. contributed to the performance of the study; T. T., S. K., and B. K. R. contributed to the data analysis; and T. S., S. K., and B. K. R. contributed to the editing of the manuscript for publication.
Financial/nonfinancial disclosures: The authors have reported to CHEST that no potential conflicts of interest exist with any companies/organizations whose products or services may be discussed in this article.
Role of sponsors: The sponsor had no role in the design of the study, the collection and analysis of the data, or the preparation of the manuscript.
Other contributions: We thank the Lipid Metabolism Project, Tokyo Metropolitan Institute of Medical Science; Makoto Murakami, PhD, for advice regarding immunostaining with sPLA2; and the Anatomic Pathology Research Services at Virginia Commonwealth University Medical Center. We also thank Medical College of Virginia Hospital Director Jorge A. Almenara, PhD, and histotechnologists for technical assistance in the tissue processing, sectioning, and staining. Microscopy was performed at the Virginia Commonwealth University Department of Anatomy and Neurobiology Microscopy Facility, supported, in part, with funding from the National Institutes of Health-National Institute of Neurological Disorders and Stroke Center core grant [5P30NS047463]. We also thank Melissa A. Yopp, BS, MSHA; Jennifer L. Bradley, BS; and Frances K. A. White for technical assistance and Erika Tokita, MD, PhD; Isao Suzaki, MD, PhD; and Shuichi Kawano, MD, for helpful discussions.
ABBREVIATIONS
- 5-LO
5-lipoxygenase
- ALI
air-liquid interface
- BALF
BAL fluid
- cysLT
cysteinyl leukotriene
- GAPDH
glyceraldehyde-3-phosphate dehydrogenase
- HBE
human bronchial epithelial
- LO
lipoxygenase
- LTB4
leukotriene B4
- LTC4
leukotriene C4
- mRNA
messenger RNA
- PAS
periodic acid-Schiff
- PBS
phosphate-buffered saline
- PCR
polymerase chain reaction
- PGE2
prostaglandin E2
- sPLA2
secretory phospholipases A2
Footnotes
FUNDING/SUPPORT: This research was funded in part by a grant from the Denny Hamlin Foundation.
Reproduction of this article is prohibited without written permission from the American College of Chest Physicians. See online for more details.
References
- 1.Dennis EA. Phospholipase A2 in eicosanoid generation. Am J Respir Crit Care Med. 2000;161(2 pt 2):S32-S35. [DOI] [PubMed] [Google Scholar]
- 2.Murakami M, Lambeau G. Emerging roles of secreted phospholipase A(2) enzymes: an update. Biochimie. 2013;95(1):43-50. [DOI] [PubMed] [Google Scholar]
- 3.Lambeau G, Gelb MH. Biochemistry and physiology of mammalian secreted phospholipases A2. Annu Rev Biochem. 2008;77:495-520. [DOI] [PubMed] [Google Scholar]
- 4.Misso NL, Petrovic N, Grove C, Celenza A, Brooks-Wildhaber J, Thompson PJ. Plasma phospholipase A2 activity in patients with asthma: association with body mass index and cholesterol concentration. Thorax. 2008;63(1):21-26. [DOI] [PubMed] [Google Scholar]
- 5.Kim DK, Fukuda T, Thompson BT, Cockrill B, Hales C, Bonventre JV. Bronchoalveolar lavage fluid phospholipase A2 activities are increased in human adult respiratory distress syndrome. Am J Physiol. 1995;269(1 pt 1):L109-L118. [DOI] [PubMed] [Google Scholar]
- 6.Masuda S, Murakami M, Mitsuishi M, et al. Expression of secretory phospholipase A2 enzymes in lungs of humans with pneumonia and their potential prostaglandin-synthetic function in human lung-derived cells. Biochem J. 2005;387(pt 1):27-38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hallstrand TS, Lai Y, Ni Z, et al. Relationship between levels of secreted phospholipase A₂ groups IIA and X in the airways and asthma severity. Clin Exp Allergy. 2011;41(6):801-810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Henderson WR, Jr, Chi EY, Bollinger JG, et al. Importance of group X-secreted phospholipase A2 in allergen-induced airway inflammation and remodeling in a mouse asthma model. J Exp Med. 2007;204(4):865-877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Muñoz NM, Meliton AY, Arm JP, Bonventre JV, Cho W, Leff AR. Deletion of secretory group V phospholipase A2 attenuates cell migration and airway hyperresponsiveness in immunosensitized mice. J Immunol. 2007;179(7):4800-4807. [DOI] [PubMed] [Google Scholar]
- 10.Shoseyov D, Bibi H, Offer S, et al. Treatment of ovalbumin-induced experimental allergic bronchitis in rats by inhaled inhibitor of secretory phospholipase A(2). Thorax. 2005;60(9):747-753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Okamoto K, Kim JS, Rubin BK. Secretory phospholipases A2 stimulate mucus secretion, induce airway inflammation, and produce secretory hyperresponsiveness to neutrophil elastase in ferret trachea. Am J Physiol Lung Cell Mol Physiol. 2007;292(1):L62-L67. [DOI] [PubMed] [Google Scholar]
- 12.Tanabe T, Kanoh S, Tsushima K, Yamazaki Y, Kubo K, Rubin BK. Clarithromycin inhibits interleukin-13-induced goblet cell hyperplasia in human airway cells. Am J Respir Cell Mol Biol. 2011;45(5):1075-1083. [DOI] [PubMed] [Google Scholar]
- 13.Ordoñez CL, Khashayar R, Wong HH, et al. Mild and moderate asthma is associated with airway goblet cell hyperplasia and abnormalities in mucin gene expression. Am J Respir Crit Care Med. 2001;163(2):517-523. [DOI] [PubMed] [Google Scholar]
- 14.Wills-Karp M, Luyimbazi J, Xu X, et al. Interleukin-13: central mediator of allergic asthma. Science. 1998;282(5397):2258-2261. [DOI] [PubMed] [Google Scholar]
- 15.Atherton HC, Jones G, Danahay H. IL-13-induced changes in the goblet cell density of human bronchial epithelial cell cultures: MAP kinase and phosphatidylinositol 3-kinase regulation. Am J Physiol Lung Cell Mol Physiol. 2003;285(3):L730-L739. [DOI] [PubMed] [Google Scholar]
- 16.Young HW, Williams OW, Chandra D, et al. Central role of Muc5ac expression in mucous metaplasia and its regulation by conserved 5′ elements. Am J Respir Cell Mol Biol. 2007;37(3):273-290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Buisine MP, Devisme L, Copin MC, et al. Developmental mucin gene expression in the human respiratory tract. Am J Respir Cell Mol Biol. 1999;20(2):209-218. [DOI] [PubMed] [Google Scholar]
- 18.Evans CM, Kim K, Tuvim MJ, Dickey BF. Mucus hypersecretion in asthma: causes and effects. Curr Opin Pulm Med. 2009;15(1):4-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Triggiani M, Granata F, Giannattasio G, Marone G. Secretory phospholipases A2 in inflammatory and allergic diseases: not just enzymes. J Allergy Clin Immunol. 2005;116(5):1000-1006. [DOI] [PubMed] [Google Scholar]
- 20.Lindbom J, Ljungman AG, Lindahl M, Tagesson C. Expression of members of the phospholipase A2 family of enzymes in human nasal mucosa. Eur Respir J. 2001;18(1):130-138. [DOI] [PubMed] [Google Scholar]
- 21.Lindbom J, Ljungman AG, Lindahl M, Tagesson C. Increased gene expression of novel cytosolic and secretory phospholipase A(2) types in human airway epithelial cells induced by tumor necrosis factor-alpha and IFN-gamma. J Interferon Cytokine Res. 2002;22(9):947-955. [DOI] [PubMed] [Google Scholar]
- 22.Seeds MC, Jones KA, Duncan Hite R, et al. Cell-specific expression of group X and group V secretory phospholipases A(2) in human lung airway epithelial cells. Am J Respir Cell Mol Biol. 2000;23(1):37-44. [DOI] [PubMed] [Google Scholar]
- 23.Kanoh S, Tanabe T, Rubin BK. IL-13-induced MUC5AC production and goblet cell differentiation is steroid resistant in human airway cells. Clin Exp Allergy. 2011;41(12):1747-1756. [DOI] [PubMed] [Google Scholar]
- 24.Kanoh S, Tanabe T, Rubin BK. Dapsone inhibits IL-8 secretion from human bronchial epithelial cells stimulated with lipopolysaccharide and resolves airway inflammation in the ferret. Chest. 2011;140(4):980-990. [DOI] [PubMed] [Google Scholar]
- 25.Tanabe T, Kanoh S, Moskowitz WB, Rubin BK. Cardiac asthma: transforming growth factor-β from the failing heart leads to squamous metaplasia in human airway cells and in the murine lung. Chest. 2012;142(5):1274-1283. [DOI] [PubMed] [Google Scholar]
- 26.Bernacki SH, Nelson AL, Abdullah L, et al. Mucin gene expression during differentiation of human airway epithelia in vitro. Muc4 and muc5b are strongly induced. Am J Respir Cell Mol Biol. 1999;20(4):595-604. [DOI] [PubMed] [Google Scholar]
- 27.Thavagnanam S, Parker JC, McBrien ME, Skibinski G, Heaney LG, Shields MD. Effects of IL-13 on mucociliary differentiation of pediatric asthmatic bronchial epithelial cells. Pediatr Res. 2011;69(2):95-100. [DOI] [PubMed] [Google Scholar]
- 28.Parker J, Sarlang S, Thavagnanam S, et al. A 3-D well-differentiated model of pediatric bronchial epithelium demonstrates unstimulated morphological differences between asthmatic and nonasthmatic cells. Pediatr Res. 2010;67(1):17-22. [DOI] [PubMed] [Google Scholar]
- 29.Tanabe T, Fujimoto K, Yasuo M, et al. Modulation of mucus production by interleukin-13 receptor alpha2 in the human airway epithelium. Clin Exp Allergy. 2008;38(1):122-134. [DOI] [PubMed] [Google Scholar]
- 30.Tanabe T, Shimokawaji T, Kanoh S, Rubin BK. IL-33 stimulates CXCL8/IL-8 secretion in goblet cells but not normally differentiated airway cells. Clin Exp Allergy. 2014;44(4):540-552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hay DW, Torphy TJ, Undem BJ. Cysteinyl leukotrienes in asthma: old mediators up to new tricks. Trends Pharmacol Sci. 1995;16(9):304-309. [DOI] [PubMed] [Google Scholar]
- 32.Jame AJ, Lackie PM, Cazaly AM, et al. Human bronchial epithelial cells express an active and inducible biosynthetic pathway for leukotrienes B4 and C4. Clin Exp Allergy. 2007;37(6):880-892. [DOI] [PubMed] [Google Scholar]
- 33.Hunter JA, Finkbeiner WE, Nadel JA, Goetzl EJ, Holtzman MJ. Predominant generation of 15-lipoxygenase metabolites of arachidonic acid by epithelial cells from human trachea. Proc Natl Acad Sci U S A. 1985;82(14):4633-4637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kowalski ML, Pawliczak R, Wozniak J, et al. Differential metabolism of arachidonic acid in nasal polyp epithelial cells cultured from aspirin-sensitive and aspirin-tolerant patients. Am J Respir Crit Care Med. 2000;161(2 pt 1):391-398. [DOI] [PubMed] [Google Scholar]
- 35.Alcorn JF, Crowe CR, Kolls JK. TH17 cells in asthma and COPD. Annu Rev Physiol. 2010;72:495-516. [DOI] [PubMed] [Google Scholar]
- 36.Sidebotham HJ, Roche WR. Asthma deaths; persistent and preventable mortality. Histopathology. 2003;43(2):105-117. [DOI] [PubMed] [Google Scholar]
- 37.Kuyper LM, Paré PD, Hogg JC, et al. Characterization of airway plugging in fatal asthma. Am J Med. 2003;115(1):6-11. [DOI] [PubMed] [Google Scholar]
- 38.Murakami M, Taketomi Y, Sato H, Yamamoto K. Secreted phospholipase A2 revisited. J Biochem. 2011;150(3):233-255. [DOI] [PubMed] [Google Scholar]
- 39.Bezzine S, Koduri RS, Valentin E, et al. Exogenously added human group X secreted phospholipase A(2) but not the group IB, IIA, and V enzymes efficiently release arachidonic acid from adherent mammalian cells. J Biol Chem. 2000;275(5):3179-3191. [DOI] [PubMed] [Google Scholar]
- 40.Murakami M, Shimbara S, Kambe T, et al. The functions of five distinct mammalian phospholipase A2S in regulating arachidonic acid release. Type IIa and type V secretory phospholipase A2S are functionally redundant and act in concert with cytosolic phospholipase A2. J Biol Chem. 1998;273(23):14411-14423. [DOI] [PubMed] [Google Scholar]
- 41.Murakami M, Kambe T, Shimbara S, et al. Different functional aspects of the group II subfamily (Types IIA and V) and type X secretory phospholipase A(2)s in regulating arachidonic acid release and prostaglandin generation. Implications of cyclooxygenase-2 induction and phospholipid scramblase-mediated cellular membrane perturbation. J Biol Chem. 1999;274(44):31435-31444. [DOI] [PubMed] [Google Scholar]
- 42.Murakami M, Koduri RS, Enomoto A, et al. Distinct arachidonate-releasing functions of mammalian secreted phospholipase A2s in human embryonic kidney 293 and rat mastocytoma RBL-2H3 cells through heparan sulfate shuttling and external plasma membrane mechanisms. J Biol Chem. 2001;276(13):10083-10096. [DOI] [PubMed] [Google Scholar]
- 43.Murakami M, Masuda S, Shimbara S, et al. Cellular arachidonate-releasing function of novel classes of secretory phospholipase A2s (groups III and XII). J Biol Chem. 2003;278(12):10657-10667. [DOI] [PubMed] [Google Scholar]
- 44.Marom Z, Shelhamer JH, Kaliner M. Effects of arachidonic acid, monohydroxyeicosatetraenoic acid and prostaglandins on the release of mucous glycoproteins from human airways in vitro. J Clin Invest. 1981;67(6):1695-1702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Johnson HG, Chinn RA, Morton DR, McNee ML, Miller MD, Nadel JA. Diphenhydramine blocks the leukotriene-C4 enhanced mucus secretion in canine trachea in vivo. Agents Actions. 1983;13(1):1-4. [DOI] [PubMed] [Google Scholar]
- 46.Suzuki S, Takeuchi K, Ishinaga H, Basbaum C, Majima Y. Leukotriene D4 upregulates MUC2 gene transcription in human epithelial cells. Pharmacology. 2008;81(3):221-228. [DOI] [PubMed] [Google Scholar]
- 47.Bai CH, Song SY, Kim YD. The inhibitory effect of the leukotriene receptor antagonist on leukotriene D4-induced MUC2/5AC gene expression and mucin secretion in human airway epithelial cells. Auris Nasus Larynx. 2007;34(2):203-206. [DOI] [PubMed] [Google Scholar]
- 48.Rich B, Peatfield AC, Williams IP, Richardson PS. Effects of prostaglandins E1, E2, and F2 alpha on mucin secretion from human bronchi in vitro. Thorax. 1984;39(6):420-423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Takahashi S, Takeuchi K, Okabe S. EP4 receptor mediation of prostaglandin E2-stimulated mucus secretion by rabbit gastric epithelial cells. Biochem Pharmacol. 1999;58(12):1997-2002. [DOI] [PubMed] [Google Scholar]
- 50.Yagi T, Miyawaki Y, Nishikawa A, Horiyama S, Yamauchi K, Kuwano S. Prostaglandin E2-mediated stimulation of mucus synthesis and secretion by rhein anthrone, the active metabolite of sennosides A and B, in the mouse colon. J Pharm Pharmacol. 1990;42(8):542-545. [DOI] [PubMed] [Google Scholar]
- 51.Muchekehu RW, Quinton PM. A new role for bicarbonate secretion in cervico-uterine mucus release. J Physiol. 2010;588(pt 13):2329-2342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Sastre B, del Pozo V. Role of PGE2 in asthma and nonasthmatic eosinophilic bronchitis [published online ahead of print March 19, 2012]. Mediators Inflamm. doi:10.1155/2012/645383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Pavord ID, Tattersfield AE. Bronchoprotective role for endogenous prostaglandin E2. Lancet. 1995;345(8947):436-438. [DOI] [PubMed] [Google Scholar]
- 54.Gauvreau GM, Watson RM, O’Byrne PM. Protective effects of inhaled PGE2 on allergen-induced airway responses and airway inflammation. Am J Respir Crit Care Med. 1999;159(1):31-36. [DOI] [PubMed] [Google Scholar]
- 55.Trudeau J, Hu H, Chibana K, Chu HW, Westcott JY, Wenzel SE. Selective downregulation of prostaglandin E2-related pathways by the Th2 cytokine IL-13. J Allergy Clin Immunol. 2006;117(6):1446-1454. [DOI] [PubMed] [Google Scholar]
- 56.Aggarwal S, Moodley YP, Thompson PJ, Misso NL. Prostaglandin E2 and cysteinyl leukotriene concentrations in sputum: association with asthma severity and eosinophilic inflammation. Clin Exp Allergy. 2010;40(1):85-93. [DOI] [PubMed] [Google Scholar]
- 57.Skibinski G, Elborn JS, Ennis M. Bronchial epithelial cell growth regulation in fibroblast cocultures: the role of hepatocyte growth factor. Am J Physiol Lung Cell Mol Physiol. 2007;293(1):L69-L76. [DOI] [PubMed] [Google Scholar]