Abstract
Mucin overproduction is a hallmark of otitis media (OM). Streptococcus pneumoniae is one of the most common bacterial pathogens causing OM. Mucin MUC5AC plays an important role in mucociliary clearance of bacterial pathogens. However, if uncontrolled, excessive mucus contributes significantly to conductive hearing loss. Currently, there is a lack of effective therapeutic agents that suppress mucus overproduction. In this study, we show that a currently existing antistroke drug, vinpocetine, a derivative of the alkaloid vincamine, inhibited S. pneumoniae–induced mucin MUC5AC upregulation in cultured middle ear epithelial cells and in the middle ear of mice. Moreover, vinpocetine inhibited MUC5AC upregulation by inhibiting the MAPK ERK pathway in an MKP-1–dependent manner. Importantly, ototopical administration of vinpocetine postinfection inhibited MUC5AC expression and middle ear inflammation induced by S. pneumoniae and reduced hearing loss and pneumococcal loads in a well-established mouse model of OM. Thus, these studies identified vinpocetine as a potential therapeutic agent for inhibiting mucus production in the pathogenesis of OM.
Introduction
Mucosal epithelium of the upper respiratory tracts, including the ear, has evolved multiple mechanisms of the host mucosal defense in response to microbial attack. The mucosal epithelial cells form a contiguous lining that acts as a physical barrier to these pathogens. In addition, these epithelial cells secrete a number of defensive molecules into the mucosal fluid, including mucins (1–3). Mucins are a family of high-m.w. glycoproteins, and ∼24 mucin genes have been identified to date. At least 12 mucin genes have been shown to be expressed in the mucosal epithelium of upper respiratory tracts (4, 5). The formation of mucus is important, because it provides a biophysical barrier and a matrix supporting the retention of host antimicrobial molecules. However, uncontrolled, excessive mucin production often results in impaired mucociliary clearance of mucosal epithelia because of increased viscosity of mucus. Indeed, mucus overproduction is a clinical hallmark of otitis media (OM), causing conductive hearing loss (6, 7). Currently, there is lack of effective therapeutic agents for suppressing mucus overproduction.
Streptococcus pneumoniae is one of the most common bacterial pathogens causing middle ear infection, sinusitis, and pneumonia (8, 9). S. pneumoniae is well known to be a potent inducer of mucin glycoprotein (10). Upregulation of mucin MUC5AC has been shown to play an important role in the pathogenesis of OM. We previously showed that S. pneumoniae upregulates MUC5AC in an MAPK ERK-dependent manner and MAPK phosphatase-1 (MKP-1) acts as a negative regulator of S. pneumoniae–induced MUC5AC induction by inhibition of ERK (11, 12). The molecular mechanisms underlying the tight regulation of MUC5AC production by bacterial pathogens still remain largely unknown, and elucidating these mechanisms may help to develop new therapeutic strategies for the inhibition of excessive mucin production (13).
Vinpocetine, an alkaloid extracted from the periwinkle plant, has been used clinically for treating cerebrovascular disorders and improving memory; it is currently used as a dietary supplement worldwide (14, 15). Its side effects were reported rarely in the literature (16). Thus, vinpocetine has long been considered an interesting compound that attracts significant attention from physicians and basic scientists to reposition it as a new therapeutic agent for other clinical disorders (17, 18). Indeed, our previous studies indicate that vinpocetine inhibits TNF-α- and LPS-induced upregulation of proinflammatory mediators, including TNF-α, IL-1β, and MIP-2, by targeting ΙκB kinase/NF-κB–dependent pathway via a direct inhibition of the ΙκB kinase complex, thereby suppressing inflammatory responses in vitro and in vivo (17). However, it remains unclear whether vinpocetine also suppresses mucus overproduction.
In the current study, we showed that vinpocetine suppressed S. pneumoniae–induced upregulation of MUC5AC. Vinpocetine inhibited MUC5AC induction via inhibition of MAPK ERK. Moreover, vinpocetine inhibited ERK via induction of MKP-1. Finally, ototopical and systemic postinfection administration of vinpocetine improved hearing loss and bacterial clearance in the mouse model of S. pneumoniae–induced OM and inhibited S. pneumoniae–induced MUC5AC expression. Thus, our studies identified the novel role of vinpocetine in suppressing bacteria-induced mucin overproduction; this could lead to the development of new therapeutic agents for treating OM.
Materials and Methods
Reagents and Abs
Vinpocetine was purchased from Tocris Bioscience. PD98059 was purchased from Enzo Life Sciences. Recombinant human IL-13 was purchased from R&D Systems. Abs against phospho-ERK1/2 (Thr-202/Tyr-204), total ERK1/2, phospho-STAT6 (Tyr-641), and total STAT6 were purchased from Cell Signaling Technology. Abs against MUC5AC, MKP-1, and β-actin were purchased from Santa Cruz Biotechnology.
Bacterial strains and culture condition
Clinical OM isolates of S. pneumoniae strain 6B, 19F, 23F, and the well-characterized D39 were used in this study (12, 19). Bioluminescent ST556 derivative of serotype 19F S. pneumoniae strain (ST556lux) was provided by Dr. Jing-Ren Zhang (Center for Immunology and Microbial Disease, Tsinghua University, Beijing, China) and has been described previously (20). All the S. pneumoniae strains were grown on chocolate agar plate and in Todd-Hewitt broth supplemented with 0.5% yeast extract at 37°C in 5% CO2 overnight. S. pneumoniae were prepared as described previously (12, 21) for in vitro and in vivo experiments.
Cell culture
All media described below were supplemented with 10% FBS (Sigma-Aldrich) and Pen/Strep (100 U/ml penicillin and 0.1 mg/ml streptomycin; Life Technologies). Human middle ear epithelial cells (HMEEC) were maintained as described previously (22). Air-liquid culture of HMEEC was conducted as described previously (23). All cells were cultured at 37°C in 5% CO2.
Real-time quantitative RT-PCR analysis
Total RNA was isolated with TRIzol reagent (Invitrogen) by following the manufacturer’s instructions. The reverse transcription reaction was performed using TaqMan reverse transcription reagents (Applied Biosystems) (23, 24). Reactions were amplified and quantified using SYBR Green Universal Master Mix reagent and Applied Biosystems StepOnePlus Real-Time PCR System (Applied Biosystems). The relative quantities of mRNAs were obtained using the comparative Ct method and were normalized using human cyclophilin or mouse GAPDH as an endogenous control. The primers for human cyclophilin and mouse GAPDH were described previously (12). The primer sequences for human and mouse MUC5AC and MKP-1 are as follows: human MUC5AC, 5′-TACTCCACAGACTGCACCAACTG-3′ and 5′-CGTGTATTGCTTCCCGTCAA-3′; human MKP-1, 5′-GCTGTGCAGCAAACAGTCGA-3′ and 5′-GCCACCCTGATCGTAGAGTG-3′; mouse MUC5AC, 5′-AAAGACACCAGTAGTCACTCAGCAA-3′ and 5′-CTGGGAAGTCAGTGTCAAACCA-3′; mouse MKP-1, 5′-GCTGTGCAGCAAACAGTCGA-3′ and 5′-CGATTAGTCCTCATAAGGTA-3′.
Plasmids, transfections, and luciferase reporter assay
The luciferase reporter gene construct of MUC5AC was described previously (12, 19). The expression plasmids dominant-negative (DN) mutants of ERK1 and ERK2 have been described previously (19). The constitutively active form of MEK (MEK-CA) was provided by Dr. Alan R. Saltiel (Life Sciences Institute, University of Michigan, Ann Arbor, MI). All transient transfections were completed in triplicate using TransIT-LT1 reagent (Mirus) following the manufacturer’s instructions. For experiments with inhibitors, the transfected HMEEC were pretreated with or without chemical inhibitors for 1 h followed by 5 h of incubation with S. pneumoniae. Transcriptional activity of the reporter gene was measured by luciferase assay and normalized with respect to β-galactosidase activity as described previously (25, 26).
RNA-mediated interference
To generate MKP-1 knockdown construct (shMKP-1), oligonucleotide encoding short hairpin transcript corresponding to MKP-1 was cloned into pSUPER. Knockdown of MKP-1 using shMKP-1 was performed using Lipofectamine 3000 (Invitrogen) following the manufacturer’s instructions. The target sequence for cloning shMKP-1 is 5′-CTGCCTTGATCAACGTCTC-3′.
Western blot analysis
Western blot analysis was performed as described previously and following the manufacturer’s instructions (22, 24). Whole-cell lysate was separated in 8% or 10% SDS-PAGE gel, transferred to PVDF membrane, and incubated with Abs against phospho-ERK1/2, total ERK1/2, phospho-STAT6, total STAT6, MKP-1, or β-actin. Respective proteins were visualized by using secondary HRP-conjugated rabbit or mouse IgG Ab (Cell Signaling Technology) and the ECL detection system (Amersham ECL Prime Western blotting Detection Regent, GE Healthcare).
ELISA
Direct ELISA was used to measure the production of MUC5AC protein as described previously (12, 19). HMEEC were treated with S. pneumoniae for 12 h with vinpocetine or vehicle pretreatment. MUC5AC protein production was measured in the cell culture supernatant as described previously (12, 19).
MKP-1 immunoprecipitation and activity assay
MKP-1 activity was measured by immunoprecipitation and subsequent phosphatase assay as described previously (22, 27). HMEEC cell lysates were incubated with 2 μg MKP-1 Ab or IgG overnight at 4°C, followed by 2 h incubation with protein A/G-agarose beads (Santa Cruz Biotechnology). After centrifugation, the MKP-1 immunoprecipitate was washed four times with lysis buffer, and MKP-1 phosphatase activity was analyzed colorimetrically using the SensoLyte pNPP Protein Phosphatase Assay kit (AnaSpec, Fremont, CA) following the manufacturer’s instructions. Nonspecific hydrolysis of pNPP was assessed in IgG immunoprecipitates as control.
Mice and animal experiments
C57BL/6 mice (7–8 wk old) were purchased from the National Cancer Institute, National Institutes of Health. Anesthetized mice were inoculated transtympanically with S. pneumoniae at a concentration of 1 × 107 CFU per mouse, and saline was inoculated as control (12). The inoculated mice were then sacrificed at 9 h postinoculation. Eardrums of mice were inspected for signs of middle ear inflammation, and photographed for recording pathologic changes of eardrum under the video otoscope (MedRx Deluxe, MedRx). Dissected mouse middle ears were then subjected to total RNA extraction and histologic analysis. For inhibition study, mice were pretreated with vinpocetine i.p. 2 h before S. pneumoniae inoculation. For the ototopical treatment experiments, mice were pretreated or posttreated with vinpocetine through tympanic membrane (3 h before or 3 h after S. pneumoniae inoculation). The inoculated mice were sacrificed at 9 h postinoculation. All animal experiments were approved by the Institutional Animal Care and Use Committee at Georgia State University.
Histology and immunofluorescence assay
For histologic analysis, formalin-fixed paraffin-embedded mouse middle ear tissues were sectioned (4 μm), and then stained with H&E to visualize inflammatory responses and pathological changes in the middle ear. For immunofluorescence (IF) assay, IF detection of MUC5AC and MKP-1 proteins were performed using mouse anti-MUC5AC or rabbit anti–MKP-1, and FITC-conjugated goat anti-mouse or anti-rabbit IgG (Santa Cruz Biotechnology) in HMEEC and the paraffin section of mouse middle ear tissues as described previously (23, 24). Images of stained cells and tissue sections were recorded with light and fluorescence microscopy systems (AxioVert 40 CFL, AxioCam MRC, and AxioVision LE Image system; Carl Zeiss), and analyzed using a quantitative image analysis system with the Visiopharm Integrator System software version 3.0.8.0 (Visiopharm, Horsholm, Denmark).
Auditory brainstem response audiometry
C57BL/6 mice (12–14 wk old) were inoculated transtympanically with S. pneumoniae at a concentration of 2 × 106 CFU per mouse, and saline was inoculated as control. To assess hearing sensitivity on all ears of the inoculated mice, auditory brainstem response (ABR) was evoked by short-duration, pure-tone bursts at 4, 8, 16, and 32 kHz. Thresholds were measured visually based on the appearance of a series of waveform in the ABR obtained at decreasing sound intensities. A computer-aided evoked potential system (Intelligent Hearing Systems, with Smart-EP software, Intelligent Hearing Systems, Miami, FL) was used to test the mouse ABR thresholds as described previously (28, 29). For the posttreatment experiments, mice were posttreated with vinpocetine (10 mg/kg) i.p. 3 h after S. pneumoniae inoculation; this was then repeated with the supplementary half-dose every day during the experiment.
Determination of pneumococcal loads
The bacterial loads in the middle ear cavity of mice were determined using standard plate count (SPC) and in vivo bioluminescence imaging (BLI) methods (30, 31). C57BL/6 mice (10–12 wk old) were inoculated transtympanically with S. pneumoniae or bioluminescent S. pneumoniae (ST556lux) at a concentration of 1 × 107 CFU per mouse, and saline was inoculated as control. For SPC, mice were treated with vinpocetine (10 mg/kg) i.p. or orally 2 h postinfection and sacrificed 24 h postinfection; both bullae were immediately removed. Middle ear effusions were collected with PBS, placed in 10-fold serial dilutions on chocolate agar plates, and incubated at 37°C in 5% CO2 for 24 h for bacterial CFU counts. For noninvasive in vivo BLI in real-time, mice were pretreated i.p. with vinpocetine (10 mg/kg) 2 h before ST556lux inoculation. Bioluminescent signals from the infection site were recorded every 12 h during the experiment using the IVIS Lumina II Imaging System, and quantified as total photon emission within a designed region of interest using Living Image Software Version 3.0 (Caliper Life Sciences, Hopkinton, MA).
Statistical analysis
All experiments were repeated in at least three independent experiments. Data are shown as mean ± SD of n determinations. Statistical analysis was assessed with two-tailed unpaired Student t-test; p < 0.05 was considered statistically significant.
Results
Vinpocetine inhibits S. pneumoniae–induced mucin MUC5AC expression in vitro
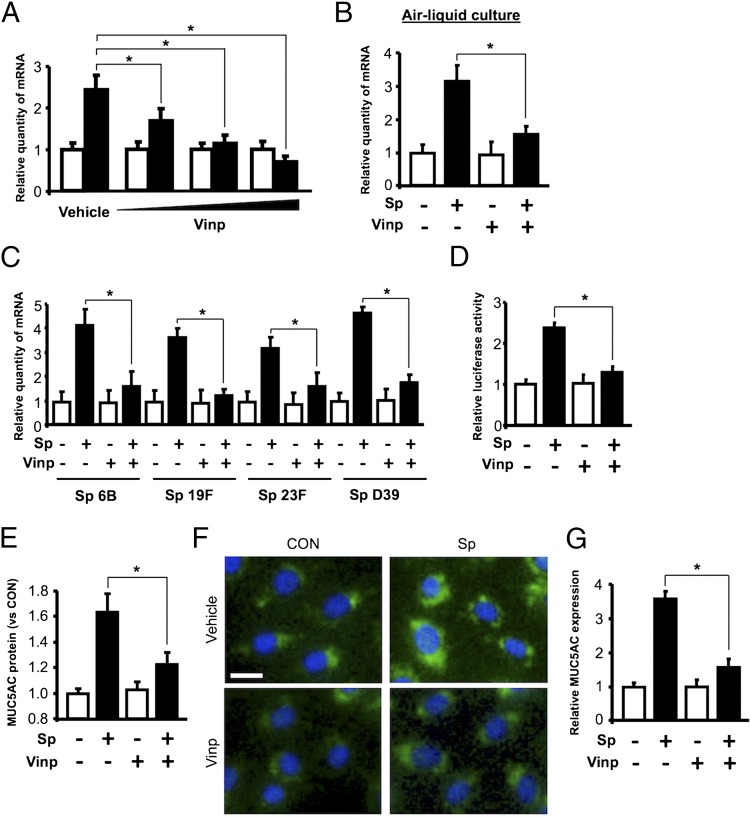
To determine whether vinpocetine inhibits S. pneumoniae–induced upregulation of mucin MUC5AC expression, we first assessed the effect of vinpocetine on MUC5AC induction by S. pneumoniae at the mRNA level using a well-established human middle ear epithelial HMEEC cell culture system in vitro by performing quantitative PCR (qPCR) analysis. As shown in Fig. 1A, vinpocetine inhibited upregulation of MUC5AC mRNA expression induced by S. pneumoniae in a dose-dependent manner. Inhibition of MUC5AC induction by vinpocetine was also observed in HMEEC cultured under air-liquid interface culture conditions (Fig. 1B). Moreover, the inhibitory effects of vinpocetine on upregulation of MUC5AC mRNA expression was also observed in cells treated with a variety of S. pneumoniae strains, including common OM pathogens 6B, 19F, and 23F strains, and the well-characterized strain D39 (Fig. 1C), thereby suggesting that the inhibitory effect of vinpocetine on MUC5AC induction is generalizable to most OM-causing strains of S. pneumoniae. We next determined whether vinpocetine also inhibits S. pneumoniae–induced upregulation of MUC5AC transcription using a MUC5AC promoter-driven luciferase reporter assay. As shown in Fig. 1D, upregulation of MUC5AC transcription by S. pneumoniae was also blocked by vinpocetine. Finally, the inhibitory effect of vinpocetine on MUC5AC induction was also confirmed and quantified at the protein level by performing ELISA and IF staining using MUC5AC-specific Ab (Fig. 1E–1G). In addition, vinpocetine also inhibited upregulation of MUC5AC mRNA expression induced by IL-13 via inhibition of STAT6 phosphorylation (Supplemental Fig. 1). It is well known that IL-13 plays a critical role in mucus-producing cell metaplasia and MUC5AC production by induction of STAT6 phosphorylation, thereby suggesting that the inhibitory effects of vinpocetine on upregulation of MUC5AC mRNA expression are generalizable to induction of MUC5AC by multiple inducers. Taken together, these data demonstrate that vinpocetine blocks S. pneumoniae–induced MUC5AC expression of human middle ear epithelial cells in vitro.
FIGURE 1.
Vinpocetine inhibits S. pneumoniae–induced mucin MUC5AC expression in vitro. HMEEC were pretreated with vinpocetine [5, 10, and 20 μm in (A), 10 μM in (B)–(G)] for 1 h, followed by stimulation of S. pneumoniae [5 h in (A)–(D), or 12 h in (E)–(G)]. Relative quantity of MUC5AC mRNA expression was measured with real-time qPCR analysis in (A) HMEEC, (B) HMEEC cultured under air-liquid interface conditions, or (C) HMEEC stimulated with S. pneumoniae strains 6B, 19F, 23F, or D39. (D) HMEEC transfected with MUC5AC-luciferase reporter gene were pretreated with vinpocetine, followed by S. pneumoniae stimulation, and MUC5AC transcriptional activity was measured with luciferase assay. (E) Expression level of MUC5AC protein was measured in the cell culture supernatant with direct ELISA against MUC5AC. (F) HMEEC were stained with Ab against MUC5AC, probed with FITC-conjugate, and (G) MUC5AC expression in IF-stained cells was analyzed by a relative quantification of immunofluorescence (original magnification ×200; scale bars, 20 μm). Data in (A)–(E) and (G) are mean ± SD (A–E, n = 3; G, n = 5). Statistical analysis was performed using Student t test. Pictures from IF-stained cells of one representative experiment are shown in (F) (n = 5). Data are representative of three or more independent experiments. *p < 0.05. CON, control; Sp, S. pneumoniae; Vinp, vinpocetine.
Vinpocetine inhibits S. pneumoniae–induced mucin MUC5AC expression in the middle ear of mice in vivo
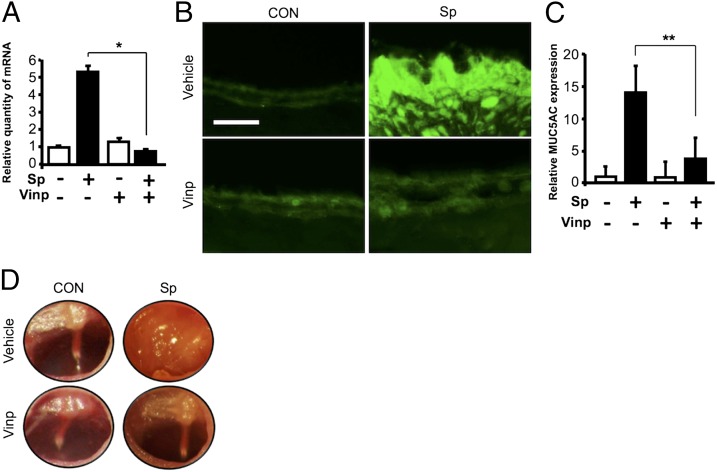
We next determined whether vinpocetine also inhibits S. pneumoniae–induced MUC5AC expression in the middle ear of mice in vivo. We first assessed the effect of vinpocetine on MUC5AC induction by S. pneumoniae at the mRNA level in the middle ear of mice by inoculating S. pneumoniae into the middle ear of wild-type strain C57BL/6 via a transtympanic membrane route. As shown in Fig. 2A, vinpocetine inhibited MUC5AC expression at the mRNA level in the middle ear of mice inoculated with S. pneumoniae as assessed by performing qPCR analysis. Pretreatment of vinpocetine also inhibited MUC5AC upregulation at the protein level in the middle ear tissues of mice as assessed by IF staining using MUC5AC-specific Ab (Fig. 2B, 2C). Consistent with these results, middle ears of mice inoculated with S. pneumoniae showed typical symptoms of OM, including congestion and swelling of tympanic membrane and mucous effusion accumulation inside the bulla at days 1, 2, and 3 after S. pneumoniae inoculation. In addition, vinpocetine inhibited S. pneumoniae–induced pathologic changes of the middle ear, as assessed by performing otoscopic examination in the middle ear of S. pneumoniae–inoculated mice at various time points, including days 1, 2, 3, 5, and 7 (Fig. 2D). These data demonstrate that vinpocetine inhibits S. pneumoniae–induced mucin MUC5AC gene expression in vivo.
FIGURE 2.
Vinpocetine inhibits S. pneumoniae–induced mucin MUC5AC expression in the middle ear of mice in vivo. Mice were pretreated with vinpocetine (10 mg/kg, i.p.) for 2 h and inoculated transtympanically with S. pneumoniae (1 × 107 CFU per mouse) for 9 h. (A) MUC5AC mRNA expression in the middle ear of mice was measured with real-time qPCR analysis. (B) Middle ear tissues were stained with Ab against MUC5AC and probed with FITC-conjugate, and (C) MUC5AC expression in IF-stained tissues was analyzed with a relative quantification of immunofluorescence (original magnification ×400; scale bars, 20 μm). (D) Tympanic cavity of the mouse ear was observed and recorded with the video-otoscope. Data in (A) and (C) are mean ± SD (A, n = 3; C, n = 7). Statistical analysis was performed using Student t test. Pictures of IF-stained middle ear tissues and the video-otoscopy from one representative experiment are shown in (B) and (D) (n = 7). Data are representative of three independent experiments. *p < 0.05; **p < 0.01. CON, control; Sp, S. pneumoniae; Vinp, vinpocetine.
Vinpocetine suppresses S. pneumoniae–induced MUC5AC expression via inhibition of ERK
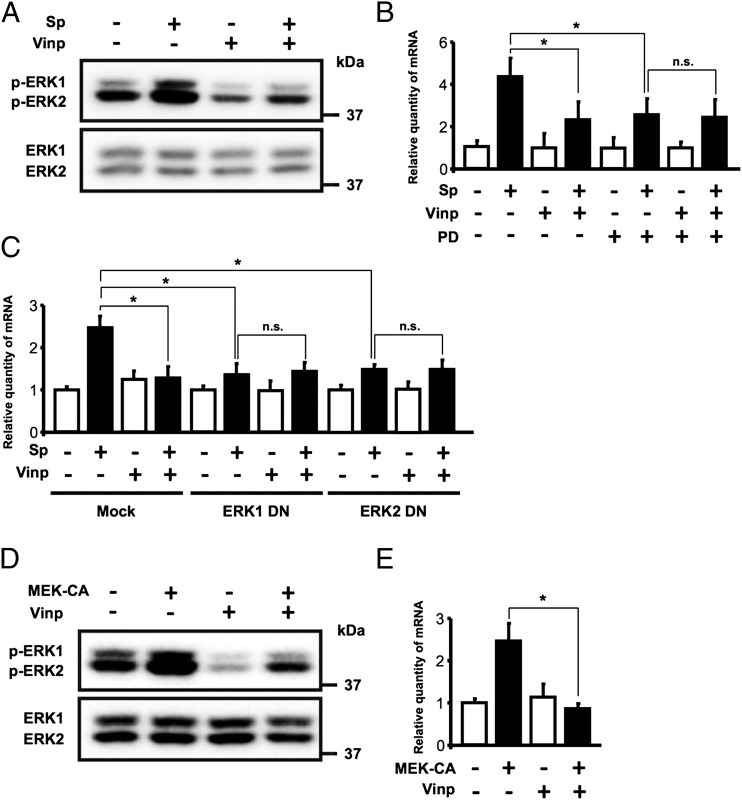
The ERK pathway has an important role in regulating immune and inflammatory responses (32). We previously found that S. pneumoniae upregulates MUC5AC expression via activation of ERK (12, 21). We thus determined whether vinpocetine inhibits MUC5AC via suppression of ERK. As shown in Fig. 3A, vinpocetine blocked S. pneumoniae–induced activation of ERK in HMEEC. Vinpocetine was unable to inhibit MUC5AC induction further in HMEEC in which ERK activation was already blocked by a specific MEK inhibitor, PD98059 (Fig. 3B). Similarly, vinpocetine also failed to attenuate S. pneumoniae–induced MUC5AC expression further in HMEEC transfected with ERK DN mutants (ERK 1 DN and ERK2 DN; Fig. 3C). Because MEK acts as the key upstream activator for ERK, we next sought to determine whether vinpocetine inhibits upregulation of MUC5AC expression induced by expressing MEK-CA. As shown in Fig. 3D, vinpocetine blocked activation of ERK induced by overexpression of MEK-CA. Moreover, vinpocetine suppressed MEK-CA–induced MUC5AC expression in HMEEC (Fig. 3E). Taken together, our data suggest that vinpocetine suppresses S. pneumoniae–induced MUC5AC expression via inhibition of MEK-dependent activation of the ERK pathway.
FIGURE 3.
Vinpocetine suppresses S. pneumoniae–induced MUC5AC expression via inhibition of ERK. (A) HMEEC were pretreated with vinpocetine (10 μM) for 1 h, followed by S. pneumoniae stimulation for 5 min, and cell lysates were analyzed by immunoblotting with the indicated Abs. (B) HMEEC were pretreated with vinpocetine (10 μM) and PD98059 (5 μM) for 1 h, followed by S. pneumoniae stimulation for 5 h, and MUC5AC mRNA expression was measured with real-time qPCR analysis. (C) HMEEC transfected with ERK1 DN or ERK2 DN were pretreated with vinpocetine (10 μM) for 1 h, and MUC5AC mRNA expression was measured with real-time qPCR analysis at 5 h after S. pneumoniae stimulation. (D) HMEEC transfected with MEK-CA were treated with vinpocetine (10 μM) for 2 h, and cell lysates were analyzed by immunoblotting with the indicated Abs. (E) MUC5AC mRNA expression was measured with real-time qPCR analysis in MEK-CA-transfected cells treated with vinpocetine (10 μM) for 6 h. Data in (B), (C), and (E) are mean ± SD (n = 3). Statistical analysis was performed using Student t test. *p < 0.05. Data are representative of three or more independent experiments. n.s., not significant; PD, PD98059; Sp, S. pneumoniae; Vinp, vinpocetine.
Vinpocetine enhances S. pneumoniae–induced upregulation of MKP-1 expression in vitro and in vivo
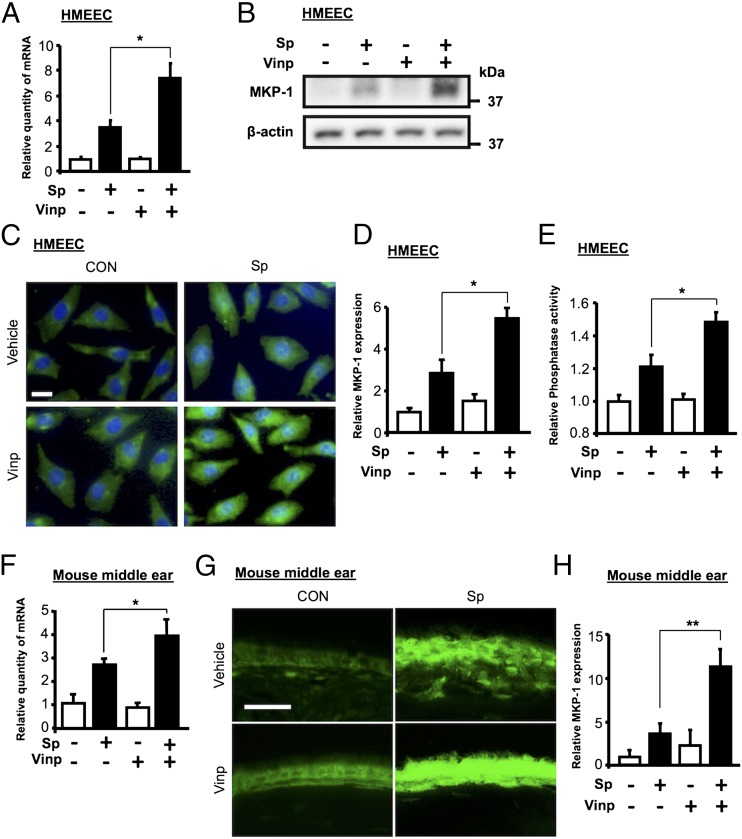
MKP-1 has been shown to be a key negative regulator of inflammatory responses via dephosphorylation of MAPKs (33). We previously showed that MKP-1 is negatively involved in regulating MUC5AC induction by S. pneumoniae (12, 21). We thus sought to determine whether vinpocetine directly upregulates S. pneumoniae–induced MKP-1 expression. Indeed, vinpocetine markedly enhanced S. pneumoniae–induced upregulation of MKP-1 expression at mRNA and protein levels in HMEEC (Fig. 4A, 4B). In addition, vinpocetine-induced upregulation of MKP-1 protein expression in HMEEC was also confirmed and quantified by performing IF staining (Fig. 4C, 4D). Moreover, the phosphatase activity of MKP-1 was increased by S. pneumoniae, and vinpocetine enhanced upregulation of MKP-1 phosphatase activity in HMEEC (Fig. 4E). Consistent with these in vitro findings, vinpocetine also enhanced S. pneumoniae–induced upregulation of MKP-1 expression at mRNA and protein levels in the middle ear of mice inoculated with S. pneumoniae, as assessed by performing qPCR analysis (Fig. 4F) and IF staining using MKP-1–specific Ab (Fig. 4G, 4H). It is evident that vinpocetine enhances S. pneumoniae–induced upregulation of MKP-1 expression and a resultant increase in phosphatase activity of MKP-1 in vitro and in vivo.
FIGURE 4.
Vinpocetine enhances S. pneumoniae–induced upregulation of MKP-1 expression in vitro and in vivo. (A–E) HMEEC were pretreated with vinpocetine (10 μM) for 1 h, followed by the stimulation with S. pneumoniae for 1.5 h. (A) MKP-1 mRNA expression was measured with real-time qPCR analysis in HMEEC, and (B) MKP-1 protein expression was analyzed by immunoblotting in the cell lysates. (C) HMEEC were stained with Ab against MKP-1 and probed with FITC-conjugate, and (D) MKP-1 expression in IF-stained cells was analyzed by a relative quantification of immunofluorescence (original magnification ×100; scale bars, 20 μm). Pictures from IF-stained cells of one representative experiment are shown in (C) (n = 5). (E) HMEEC cell lysates were immunoprecipitated with anti–MKP-1 Ab for phosphatase activity. The absorbance at 405 nm of the reaction product (p-nitrophenol) from the immunoprecipitates was measured by colorimetric mode to determine the phosphatase activity of MKP-1. (F–H) Mice were pretreated with vinpocetine (10 mg/kg, i.p.) for 2 h and inoculated transtympanically with S. pneumoniae (1 × 107 CFU per mouse) for 9 h. (F) MKP-1 mRNA expression was measured with real-time qPCR analysis in the middle ear of mice. (G) Middle ear tissues were stained with Ab against MKP-1 and probed with FITC-conjugate, and (H) MKP-1 expression in IF-stained tissues was analyzed by a relative quantification of immunofluorescence (original magnification ×400; scale bars, 20 μm). Pictures from IF-stained mouse middle ear tissues of one representative experiment are shown in (G) (n = 7). Data in (A), (D), (E), (F), and (H) are mean ± SD (A, E, F, n = 3; D, n = 5; H, n = 7). Statistical analysis was performed using Student t test. Data are representative of three independent experiments. *p < 0.05. **p < 0.01. CON, control; Sp, S. pneumoniae; Vinp, vinpocetine.
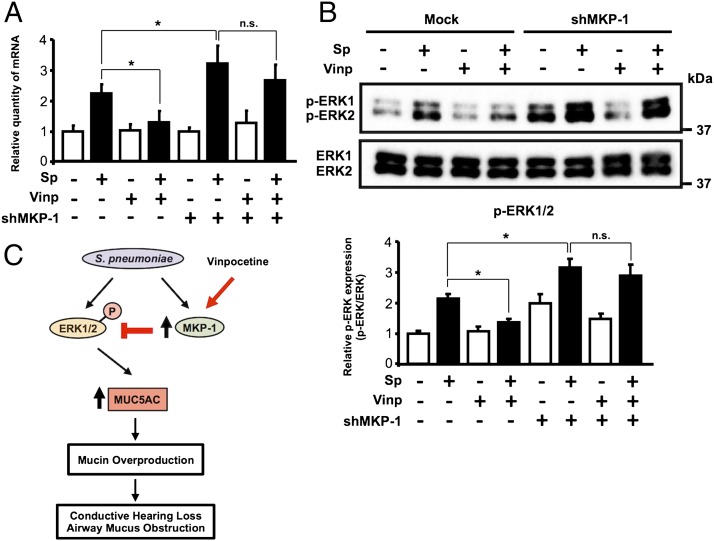
Vinpocetine inhibits S. pneumoniae–induced MUC5AC expression via MKP-1–dependent inhibition of ERK
MKP-1 is known to inactivate ERK by dephosphorylation (34). Having shown that vinpocetine inhibits S. pneumoniae–induced MUC5AC expression via inhibition of ERK and enhances upregulation of MKP-1 induced by S. pneumoniae, we next determined whether vinpocetine inhibits ERK-dependent induction of MUC5AC by S. pneumoniae via upregulation of MKP-1 expression. Interestingly, vinpocetine no longer inhibited S. pneumoniae–induced MUC5AC upregulation in HMEEC in which MKP-1 was already depleted with MKP-1 short hairpin RNA (Fig. 5A, Supplemental Fig. 2). Moreover, vinpocetine failed to inhibit ERK phosphorylation induced by S. pneumoniae in MKP-1–depleted HMEEC (Fig. 5B). Our data suggest that vinpocetine inhibits S. pneumoniae–induced ERK-dependent MUC5AC upregulation via induction of MKP-1 (Fig. 5C).
FIGURE 5.
Vinpocetine inhibits S. pneumoniae–induced MUC5AC expression via MKP-1–dependent inhibition of ERK. (A) HMEEC transfected with shCON or shMKP-1 were pretreated with vinpocetine (10 μM) for 1 h, followed by S. pneumoniae stimulation for 5 h, and MUC5AC mRNA expression was measured with real-time qPCR analysis. (B) HMEEC transfected with shCON, or shMKP-1 were pretreated with vinpocetine (10 μM) for 1 h, followed by S. pneumoniae stimulation for 5 min, and cell lysates were analyzed by immunoblotting with the indicated Abs (upper panel). Phosphorylated ERK (pERK) protein expression was quantified from three independent experiments (lower panel). (C) A schematic model illustrating that vinpocetine inhibits S. pneumoniae–induced mucin MUC5AC expression via MKP-1–dependent inhibition of the ERK pathway. Data in (A) and (B) (lower panel) are mean ± SD (n = 3). Statistical analysis was performed using Student t test. Data are representative of three or more independent experiments. *p < 0.05. CON, control; n.s., not significant; Sp, S. pneumoniae; Vinp, vinpocetine.
Postinfection administration of vinpocetine inhibits upregulation of MUC5AC expression in the mouse model of S. pneumoniae–induced OM
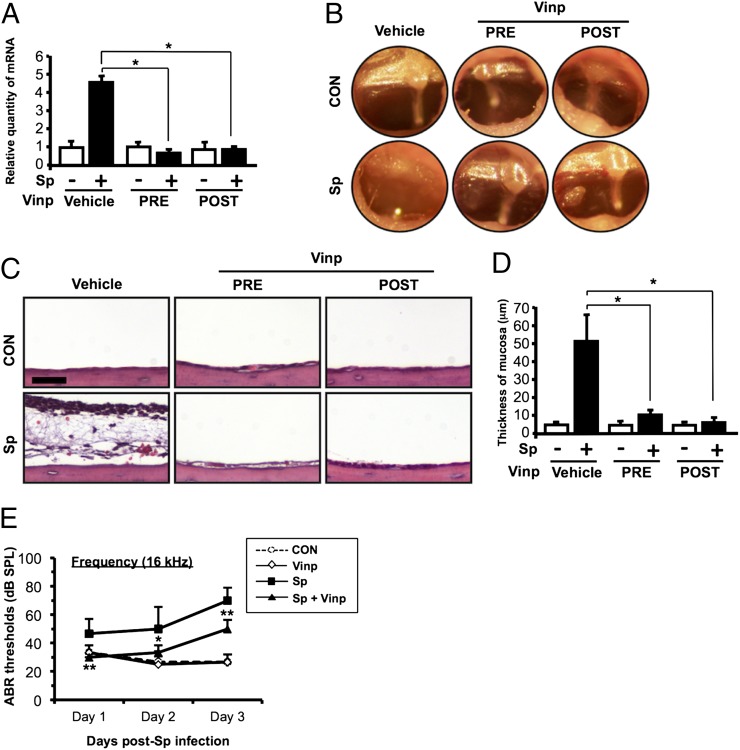
We have shown that vinpocetine, when preinoculated systemically, inhibited S. pneumoniae–induced MUC5AC expression in the mouse models of middle ear infection. However, it is still unclear whether vinpocetine has any therapeutic effect in OM. We thus evaluated whether postinoculation administration of vinpocetine also inhibited induction of MUC5AC in a well-established OM mouse model under more clinically relevant conditions. Interestingly, both ototopical preinoculation and postinoculation administration of vinpocetine suppressed S. pneumoniae–induced MUC5AC upregulation (Fig. 6A), and it inhibited the typical symptoms of OM as assessed by performing otoscopic examination (Fig. 6B). Consistent with these results, vinpocetine also inhibited S. pneumoniae–induced mucosal thickening and polymorphonuclear neutrophil infiltration in the middle ear mucosa as assessed by histologic analysis (Fig. 6C, 6D). ABR is an objective measurement of the evoked potential of auditory activity in the brainstem. It has been commonly used for monitoring the status of middle ear inflammation in animals and children by measuring the threshold shifts because of fluid and mucus accumulation (28, 29). As shown in Fig. 6E, S. pneumoniae indeed induced hearing loss, as reflected by the elevation of ABR thresholds in a mouse model of OM. Importantly, systemic postinoculation administration of vinpocetine markedly attenuated the elevation of the ABR thresholds and improved hearing loss as evaluated by performing ABR analysis of the mouse model of OM (Fig. 6E). Moreover, posttreatment of vinpocetine increased the clearance of S. pneumoniae and decreased the pneumococcal CFU levels in the middle ear effusion at 24 h postinfection in SPC assay (Supplemental Fig. 3A). Consistent with the viable bacterial counts, bioluminescence emission of ST556lux in the middle ear cavity peaked at 12 h postinfection, and vinpocetine significantly attenuated pneumococcal loads in the middle ear cavity at 12, 24, and 36 h postinfection (Supplemental Fig. 3B, 3C). S. pneumoniae adheres to mucous membranes. It is conceivable that upregulation of mucin production further promotes bacterial colonization. These data suggest that vinpocetine decreases pneumococcal loads by inhibiting the expression of mucin MUC5AC and reduces hearing loss, thereby providing strong evidence for the therapeutic potential of vinpocetine in treating OM.
FIGURE 6.
Postinfection administration of vinpocetine inhibits MUC5AC expression in a mouse model of S. pneumoniae–induced OM. (A–D) Mice were inoculated transtympanically with S. pneumoniae (1 × 107 CFU per mouse) for 9 h, and pretreated or posttreated ototopically with vinpocetine (0.5 mg/kg) 3 h before or 3 h after S. pneumoniae inoculation. (A) MUC5AC mRNA expression in the middle ear of mice was measured with real-time qPCR assay. (B) Tympanic cavity of the mouse ear was observed and recorded with the video-otoscope. (C) H&E staining of the middle ear tissues from mice was performed (original magnification ×400; scale bar, 20 μm) for histologic analysis, and (D) the thickness of middle ear mucosa was measured from 15 middle ear sections per experimental group. (E) Hearing thresholds on each ear of the mice were measured with ABR tests across a frequency range of 4–32 kHz for 1–3 d after S. pneumoniae inoculation (2 × 106 CFU per mouse). Mice were posttreated with vinpocetine (10 mg/kg) i.p. 3 h after S. pneumoniae inoculation; this was repeated at a dose of 5 mg/kg/day during the experiment. Pictures of the video otoscopy and H&E-stained middle ear tissues from one representative experiment are shown in (B) and (C) (B, n = 6; C, n = 15). Data in (A), (D), and (E) are mean ± SD (A, n = 3; D, n = 15; E, n = 6). Statistical analysis was performed using Student t test. Data are representative of three independent experiments. *p < 0.05, **p < 0.01. CON, control; PRE, preinoculation; POST, postinoculation; Sp, S. pneumoniae; Vinp, vinpocetine.
Discussion
In the current study, we showed that vinpocetine suppressed S. pneumoniae–induced MUC5AC upregulation in the mucosal epithelia of the middle ear in vitro and in vivo. Interestingly, vinpocetine inhibited S. pneumoniae–induced ERK-dependent upregulation of MUC5AC by upregulating MKP-1, a critical negative feedback regulator for MAPK ERK. Moreover, ototopical or systemic postinfection treatment, as well as the systemic pretreatment, with vinpocetine inhibited S. pneumoniae–induced MUC5AC upregulation in a well-established mouse model of S. pneumoniae–induced OM. Thus, the current study provides novel insights into the molecular mechanisms underlying the tight regulation of mucin overproduction in the pathogenesis of S. pneumoniae–induced OM and may help to repurpose vinpocetine as a new therapeutic agent for OM.
To date, a number of mucoactive agents have been used for the treatment of mucosal inflammatory diseases of the upper respiratory tract. Mucoactive agents can be classified as expectorants (guaifenesin), mucolytics (N-acetylcysteine and peptide enzymes), mucokinetics (β2-adrenoceptor agonist and surfactant), and mucoregulators (corticosteroids and macrolide antibiotics) according to their potential mechanism of action: reducing mucus viscosity, increasing mucociliary clearance, or suppressing mucus hypersecretion (35, 36). Although these medications are useful at certain stages of the disease, treatment with mucoactive agents has not proved to be effective and has shown side effects including vomiting, diarrhea, stomach upset, headache, skin rash, and even bleeding from the gastrointestinal tract (35, 36). In addition, the mechanism of action for most of these drugs remains unclear. Therefore, by identifying the key molecular therapeutic targets, development of a novel therapeutic agent without causing serious side effects is urgently needed for tight control of mucus overproduction in mucosal infections. Interestingly, in the current study, we provided direct in vitro and in vivo evidence that vinpocetine inhibits S. pneumoniae–induced MUC5AC expression by directly upregulating the negative regulator MKP-1 instead of inhibiting the positive regulator ERK. Thus, vinpocetine may have significant therapeutic potential for treating mucosal infectious diseases of the upper respiratory tract without the serious side effects often seen when directly inhibiting positive pathways, such as the MAPK ERK signaling cascade (22).
Another interesting finding in our study is that both ototopical preinfection and postinfection treatment with vinpocetine inhibited not only S. pneumoniae–induced MUC5AC upregulation, but also pathologic changes in a mouse OM model as assessed by otoscopic examination and histologic analysis. Furthermore, systemic postinfection treatment of vinpocetine improved hearing loss as measured by ABR thresholds and reduced pneumococcal loads in the middle ear assessed by SPC and BLI in a mouse OM model, thereby demonstrating its therapeutic potential in OM. Topical applications to the mucous membranes of the upper respiratory tract diseases have the advantage of acting directly on the site of inflammation and producing a higher local drug concentration at the target site without gastrointestinal degradation and hepatic first-pass metabolism. It also allows a rapid onset of therapeutic effects of the drug while minimizing systemic side effects (37, 38). Thus, our findings may have significant therapeutic significance, because topical administration of the currently available drug vinpocetine could lead to the immediate clinical application for the treatment of otitis media.
During the past several years, significant efforts and interests have been focused on drug repositioning for developing new novel therapeutic agents (39). Several drugs, such as sildenafil citrate and thalidomide, have been successfully repositioned to a new indication as a lower-risk and cost-effective repositioning model (40). Vinpocetine was originally discovered and marketed in 1978 under the trade name Cavinton, and it has been used widely in many countries for treatment of cerebrovascular disorder, such as stroke and cognitive impairment, because of its beneficial cerebrovascular effect and neuroprotective profile. To date, there have been no reports of significant side effects and toxicity related to vinpocetine treatment at therapeutic doses, and various clinical studies have demonstrated that vinpocetine is safe for long-term use (41). Thus, vinpocetine has long been considered an interesting compound that constantly attracts scientists and clinicians to investigate its novel therapeutic scheme in the fields of neuropsychiatry, ophthalmology, and otolaryngology. Previously, our studies identified vinpocetine as a novel anti-inflammatory agent that may be repositioned for the treatment of various human inflammatory diseases, including chronic obstructive pulmonary disease, arthritis, and atherosclerosis (17, 42). In the current study, we identified a novel role of vinpocetine in suppressing mucin upregulation. Together, our studies may lead to the development of novel therapeutic strategies for treating OM.
Of additional biological significance in the current study is that vinpocetine inhibits MUC5AC induction by S. pneumoniae via MKP-1–dependent suppression of MEK-mediated ERK activation. We also previously demonstrated that glucocorticoids inhibited p38 MAPK-mediated MUC5AC expression induced by NTHi via upregulation of MKP-1 gene expression (43). Because of the critical role of MAPKs, including ERK, in the regulation of innate immune and inflammatory responses, upregulation of MKP-1 could play an important role in tightly controlling overactive immune and inflammatory response in a negative-feedback manner. In addition, it was demonstrated in the current study that vinpocetine reduced pneumococcal loads in the middle ear infection likely via suppression of MUC5AC induction (44). Thus, vinpocetine may represent an effective therapeutic agent for the tight regulation of overactive host immune response without causing serious adverse effects (45–47).
Supplementary Material
Acknowledgments
We thank Drs. Xi Lin and Qing Chang (Department of Otolaryngology, Emory University School of Medicine) for assistance with ABR analysis and Jing-Ren Zhang (Center for Infectious Disease Research, Tsinghua University School of Medicine) for providing the bioluminescent pneumococcal strain, ST556lux.
This work was supported by National Institutes of Health Grants DC005843, DC004562, and GM107529 (to J.-D.L.) and HL111291 and HL088400 (to C.Y.). J.-D.L. is a Georgia Research Alliance Eminent Scholar in Inflammation and Immunity.
The online version of this article contains supplemental material.
- ABR
- auditory brainstem response
- BLI
- bioluminescence imaging
- DN
- dominant-negative
- HMEEC
- human middle ear epithelial cell
- IF
- immunofluorescence
- MEK-CA
- constitutively active form of MEK
- MKP-1
- MAPK phosphatase-1
- OM
- otitis media
- qPCR
- quantitative PCR
- SPC
- standard plate count.
Disclosures
The authors have no financial conflicts of interest.
References
- 1.Kopp E., Medzhitov R. 2003. Recognition of microbial infection by Toll-like receptors. Curr. Opin. Immunol. 15: 396–401. [DOI] [PubMed] [Google Scholar]
- 2.Rose M. C., Voynow J. A. 2006. Respiratory tract mucin genes and mucin glycoproteins in health and disease. Physiol. Rev. 86: 245–278. [DOI] [PubMed] [Google Scholar]
- 3.Knowles M. R., Boucher R. C. 2002. Mucus clearance as a primary innate defense mechanism for mammalian airways. J. Clin. Invest. 109: 571–577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kerschner J. E. 2007. Mucin gene expression in human middle ear epithelium. Laryngoscope 117: 1666–1676. [DOI] [PubMed] [Google Scholar]
- 5.Gendler S. J., Spicer A. P. 1995. Epithelial mucin genes. Annu. Rev. Physiol. 57: 607–634. [DOI] [PubMed] [Google Scholar]
- 6.Park J. A., Sharif A. S., Shiomi T., Kobzik L., Kasahara D. I., Tschumperlin D. J., Voynow J., Drazen J. M. 2013. Human neutrophil elastase-mediated goblet cell metaplasia is attenuated in TACE-deficient mice. Am. J. Physiol. Lung Cell. Mol. Physiol. 304: L701–L707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Linden S. K., Sutton P., Karlsson N. G., Korolik V., McGuckin M. A. 2008. Mucins in the mucosal barrier to infection. Mucosal Immunol. 1: 183–197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.DeMaria T. F., Bakaletz L. O., Chonmaitree T., Heikkinen T., Hurst D. S., Kawauchi H., Kurono Y., Patel J. A., Sih T. M., Stenfors L. E., Suzuki M. 2002. Recent advances in otitis media. 6. Microbiology and immunology. Ann. Otol. Rhinol. Laryngol. Suppl. 188: 62–81. [PubMed] [Google Scholar]
- 9.Block S. L. 1997. Causative pathogens, antibiotic resistance and therapeutic considerations in acute otitis media. Pediatr. Infect. Dis. J. 16: 449–456. [DOI] [PubMed] [Google Scholar]
- 10.Tsuboi Y., Kim Y., Paparella M. M., Chen N., Schachern P. A., Lin J. 2001. Pattern changes of mucin gene expression with pneumococcal otitis media. Int. J. Pediatr. Otorhinolaryngol. 61: 23–30. [DOI] [PubMed] [Google Scholar]
- 11.Lim J. H., Kim H. J., Komatsu K., Ha U., Huang Y., Jono H., Kweon S. M., Lee J., Xu X., Zhang G. S., et al. 2009. Differential regulation of Streptococcus pneumoniae-induced human MUC5AC mucin expression through distinct MAPK pathways. Am J Transl Res 1: 300–311. [PMC free article] [PubMed] [Google Scholar]
- 12.Lee J., Komatsu K., Lee B. C., Lim J. H., Jono H., Xu H., Kai H., Zhang Z. J., Yan C., Li J. D. 2012. Phosphodiesterase 4B mediates extracellular signal-regulated kinase-dependent up-regulation of mucin MUC5AC protein by Streptococcus pneumoniae by inhibiting cAMP-protein kinase A-dependent MKP-1 phosphatase pathway. J. Biol. Chem. 287: 22799–22811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Song K. S., Lee W. J., Chung K. C., Koo J. S., Yang E. J., Choi J. Y., Yoon J. H. 2003. Interleukin-1 beta and tumor necrosis factor-alpha induce MUC5AC overexpression through a mechanism involving ERK/p38 mitogen-activated protein kinases-MSK1-CREB activation in human airway epithelial cells. J. Biol. Chem. 278: 23243–23250. [DOI] [PubMed] [Google Scholar]
- 14.Bagoly E., Fehér G., Szapáry L. 2007. [The role of vinpocetine in the treatment of cerebrovascular diseases based in human studies]. Orv. Hetil. 148: 1353–1358. [DOI] [PubMed] [Google Scholar]
- 15.Tamaki N., Matsumoto S. 1985. [Agents to improve cerebrovascular circulation and cerebral metabolism—vinpocetine]. Nippon Rinsho 43: 376–378. [PubMed] [Google Scholar]
- 16.Balestreri R., Fontana L., Astengo F. 1987. A double-blind placebo controlled evaluation of the safety and efficacy of vinpocetine in the treatment of patients with chronic vascular senile cerebral dysfunction. J. Am. Geriatr. Soc. 35: 425–430. [DOI] [PubMed] [Google Scholar]
- 17.Jeon K. I., Xu X., Aizawa T., Lim J. H., Jono H., Kwon D. S., Abe J., Berk B. C., Li J. D., Yan C. 2010. Vinpocetine inhibits NF-kappaB-dependent inflammation via an IKK-dependent but PDE-independent mechanism. Proc. Natl. Acad. Sci. USA 107: 9795–9800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Medina A. E. 2010. Vinpocetine as a potent antiinflammatory agent. Proc. Natl. Acad. Sci. USA 107: 9921–9922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ha U., Lim J. H., Jono H., Koga T., Srivastava A., Malley R., Pagès G., Pouysségur J., Li J. D. 2007. A novel role for IkappaB kinase (IKK) alpha and IKKbeta in ERK-dependent up-regulation of MUC5AC mucin transcription by Streptococcus pneumoniae. J. Immunol. 178: 1736–1747. [DOI] [PubMed] [Google Scholar]
- 20.Chen H., Ma Y., Yang J., O’Brien C. J., Lee S. L., Mazurkiewicz J. E., Haataja S., Yan J. H., Gao G. F., Zhang J. R. 2008. Genetic requirement for pneumococcal ear infection. PLoS ONE 3: e2950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ha U. H., Lim J. H., Kim H. J., Wu W., Jin S., Xu H., Li J. D. 2008. MKP1 regulates the induction of MUC5AC mucin by Streptococcus pneumoniae pneumolysin by inhibiting the PAK4-JNK signaling pathway. J. Biol. Chem. 283: 30624–30631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Komatsu K., Lee J. Y., Miyata M., Hyang Lim J., Jono H., Koga T., Xu H., Yan C., Kai H., Li J. D. 2013. Inhibition of PDE4B suppresses inflammation by increasing expression of the deubiquitinase CYLD. Nat. Commun. 4: 1684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sakai A., Koga T., Lim J. H., Jono H., Harada K., Szymanski E., Xu H., Kai H., Li J. D. 2007. The bacterium, nontypeable Haemophilus influenzae, enhances host antiviral response by inducing Toll-like receptor 7 expression: evidence for negative regulation of host anti-viral response by CYLD. FEBS J. 274: 3655–3668. [DOI] [PubMed] [Google Scholar]
- 24.Yoshida H., Jono H., Kai H., Li J. D. 2005. The tumor suppressor cylindromatosis (CYLD) acts as a negative regulator for toll-like receptor 2 signaling via negative cross-talk with TRAF6 AND TRAF7. J. Biol. Chem. 280: 41111–41121. [DOI] [PubMed] [Google Scholar]
- 25.Wang B., Lim D. J., Han J., Kim Y. S., Basbaum C. B., Li J. D. 2002. Novel cytoplasmic proteins of nontypeable Haemophilus influenzae up-regulate human MUC5AC mucin transcription via a positive p38 mitogen-activated protein kinase pathway and a negative phosphoinositide 3-kinase-Akt pathway. J. Biol. Chem. 277: 949–957. [DOI] [PubMed] [Google Scholar]
- 26.Jono H., Lim J. H., Chen L. F., Xu H., Trompouki E., Pan Z. K., Mosialos G., Li J. D. 2004. NF-kappaB is essential for induction of CYLD, the negative regulator of NF-kappaB: evidence for a novel inducible autoregulatory feedback pathway. J. Biol. Chem. 279: 36171–36174. [DOI] [PubMed] [Google Scholar]
- 27.Lin C. J., Ho H. Y., Cheng M. L., You T. H., Yu J. S., Chiu D. T. 2010. Impaired dephosphorylation renders G6PD-knockdown HepG2 cells more susceptible to H(2)O(2)-induced apoptosis. Free Radic. Biol. Med. 49: 361–373. [DOI] [PubMed] [Google Scholar]
- 28.Zheng Q. Y., Johnson K. R., Erway L. C. 1999. Assessment of hearing in 80 inbred strains of mice by ABR threshold analyses. Hear. Res. 130: 94–107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Mitchell C. R., Kempton J. B., Creedon T. A., Trune D. R. 1999. The use of a 56-stimulus train for the rapid acquisition of auditory brainstem responses. Audiol. Neurootol. 4: 80–87. [DOI] [PubMed] [Google Scholar]
- 30.Schachern P. A., Tsuprun V., Ferrieri P., Briles D. E., Goetz S., Cureoglu S., Paparella M. M., Juhn S. 2014. Pneumococcal PspA and PspC proteins: potential vaccine candidates for experimental otitis media. Int. J. Pediatr. Otorhinolaryngol. 78: 1517–1521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Albiger B., Sandgren A., Katsuragi H., Meyer-Hoffert U., Beiter K., Wartha F., Hornef M., Normark S., Normark B. H. 2005. Myeloid differentiation factor 88-dependent signalling controls bacterial growth during colonization and systemic pneumococcal disease in mice. Cell. Microbiol. 7: 1603–1615. [DOI] [PubMed] [Google Scholar]
- 32.Abraham S. M., Clark A. R. 2006. Dual-specificity phosphatase 1: a critical regulator of innate immune responses. Biochem. Soc. Trans. 34: 1018–1023. [DOI] [PubMed] [Google Scholar]
- 33.Wong H. R., Dunsmore K. E., Page K., Shanley T. P. 2005. Heat shock-mediated regulation of MKP-1. Am. J. Physiol. Cell Physiol. 289: C1152–C1158. [DOI] [PubMed] [Google Scholar]
- 34.Murphy L. O., Blenis J. 2006. MAPK signal specificity: the right place at the right time. Trends Biochem. Sci. 31: 268–275. [DOI] [PubMed] [Google Scholar]
- 35.Rogers D. F. 2007. Physiology of airway mucus secretion and pathophysiology of hypersecretion. Respir. Care 52: 1134–1146, discussion 1146–1149. [PubMed] [Google Scholar]
- 36.Balsamo R., Lanata L., Egan C. G. 2010. Mucoactive drugs. Eur. Resp. Rev. 19: 127–133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Howland W. C., 3rd 1996. Fluticasone propionate: topical or systemic effects? Clin. Exp. Allergy 26(Suppl. 3): 18–22. [DOI] [PubMed] [Google Scholar]
- 38.Sawynok J. 2003. Topical and peripherally acting analgesics. Pharmacol. Rev. 55: 1–20. [DOI] [PubMed] [Google Scholar]
- 39.Chong C. R., Sullivan D. J., Jr. 2007. New uses for old drugs. Nature 448: 645–646. [DOI] [PubMed] [Google Scholar]
- 40.Novac N. 2013. Challenges and opportunities of drug repositioning. Trends Pharmacol. Sci. 34: 267–272. [DOI] [PubMed] [Google Scholar]
- 41.Patyar S., Prakash A., Modi M., Medhi B. 2011. Role of vinpocetine in cerebrovascular diseases. Pharmacol. Rep. 63: 618–628. [DOI] [PubMed] [Google Scholar]
- 42.Cai Y., Li J. D., Yan C. 2013. Vinpocetine attenuates lipid accumulation and atherosclerosis formation. Biochem. Biophys. Res. Commun. 434: 439–443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Komatsu K., Jono H., Lim J. H., Imasato A., Xu H., Kai H., Yan C., Li J. D. 2008. Glucocorticoids inhibit nontypeable Haemophilus influenzae-induced MUC5AC mucin expression via MAPK phosphatase-1-dependent inhibition of p38 MAPK. Biochem. Biophys. Res. Commun. 377: 763–768. [DOI] [PubMed] [Google Scholar]
- 44.Sun L., Tang L., Xu Y., Wang S., Li Y., Kang J. 2010. The effect and mechanism of action of carbocysteine on airway bacterial load in rats chronically exposed to cigarette smoke. Respirology 15: 1064–1071. [DOI] [PubMed] [Google Scholar]
- 45.Li L., Chen S. F., Liu Y. 2009. MAP kinase phosphatase-1, a critical negative regulator of the innate immune response. Int J Clin Exp Med 2: 48–67. [PMC free article] [PubMed] [Google Scholar]
- 46.Rogers, D. F. 2007. Mucoactive agents for airway mucus hypersecretory diseases. Respir. Care 52: 1176–1193; discussion 1193-1177. [PubMed]
- 47.Chi H., Barry S. P., Roth R. J., Wu J. J., Jones E. A., Bennett A. M., Flavell R. A. 2006. Dynamic regulation of pro- and anti-inflammatory cytokines by MAPK phosphatase 1 (MKP-1) in innate immune responses. Proc. Natl. Acad. Sci. USA 103: 2274–2279. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.