Abstract
Mutations in the disco (disconnected) gene prevent the establishment of stable connections between the larval optic nerves, the Bolwig's nerves, and their target cells in the brain during embryonic development. The failure of this initial connection is associated with aberrant development of the optic lobes which are largely degenerate in the mutant adult fly. In order to understand the role of disco in establishing this connection, we isolated and characterized the disco gene. A 22 kb DNA fragment can completely rescue the mutant phenotype. A single transcript, 2.9 kb in length, is found in this region and is expressed throughout development of the fly. We determined the nucleotide sequence of the disco gene to be unique when compared with sequences in a number of databases. The predicted amino acid sequence contains a region with similarity to the consensus established for the zinc finger motif. Mobilization of a P-element inserted near the gene resulted in the deletion of the 5' end of the gene and produced flies indistinguishable from those carrying the disco allele.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aronson B. D., Somerville R. L., Epperly B. R., Dekker E. E. The primary structure of Escherichia coli L-threonine dehydrogenase. J Biol Chem. 1989 Mar 25;264(9):5226–5232. [PubMed] [Google Scholar]
- Bate C. M. Pioneer neurones in an insect embryo. Nature. 1976 Mar 4;260(5546):54–56. doi: 10.1038/260054a0. [DOI] [PubMed] [Google Scholar]
- Bentley D., Keshishian H. Pathfinding by peripheral pioneer neurons in grasshoppers. Science. 1982 Dec 10;218(4577):1082–1088. doi: 10.1126/science.218.4577.1082. [DOI] [PubMed] [Google Scholar]
- Berg J. M. Zinc finger domains: hypotheses and current knowledge. Annu Rev Biophys Biophys Chem. 1990;19:405–421. doi: 10.1146/annurev.bb.19.060190.002201. [DOI] [PubMed] [Google Scholar]
- Bier E., Vaessin H., Shepherd S., Lee K., McCall K., Barbel S., Ackerman L., Carretto R., Uemura T., Grell E. Searching for pattern and mutation in the Drosophila genome with a P-lacZ vector. Genes Dev. 1989 Sep;3(9):1273–1287. doi: 10.1101/gad.3.9.1273. [DOI] [PubMed] [Google Scholar]
- Chang T. H., Clark M. W., Lustig A. J., Cusick M. E., Abelson J. RNA11 protein is associated with the yeast spliceosome and is localized in the periphery of the cell nucleus. Mol Cell Biol. 1988 Jun;8(6):2379–2393. doi: 10.1128/mcb.8.6.2379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Falk D. R., Roselli L., Curtiss S., Halladay D., Klufas C. The characterization of chromosome breaks in Drosophila melanogaster. I. Mass isolation of deficiencies which have an end point in the 14A-15A region. Mutat Res. 1984 Mar;126(1):25–34. doi: 10.1016/0027-5107(84)90166-0. [DOI] [PubMed] [Google Scholar]
- Frankel A. D., Bredt D. S., Pabo C. O. Tat protein from human immunodeficiency virus forms a metal-linked dimer. Science. 1988 Apr 1;240(4848):70–73. doi: 10.1126/science.2832944. [DOI] [PubMed] [Google Scholar]
- Frischauf A. M., Lehrach H., Poustka A., Murray N. Lambda replacement vectors carrying polylinker sequences. J Mol Biol. 1983 Nov 15;170(4):827–842. doi: 10.1016/s0022-2836(83)80190-9. [DOI] [PubMed] [Google Scholar]
- Fu Y. H., Marzluf G. A. nit-2, the major positive-acting nitrogen regulatory gene of Neurospora crassa, encodes a sequence-specific DNA-binding protein. Proc Natl Acad Sci U S A. 1990 Jul;87(14):5331–5335. doi: 10.1073/pnas.87.14.5331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujita S. C., Zipursky S. L., Benzer S., Ferrús A., Shotwell S. L. Monoclonal antibodies against the Drosophila nervous system. Proc Natl Acad Sci U S A. 1982 Dec;79(24):7929–7933. doi: 10.1073/pnas.79.24.7929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gibson T. J., Postma J. P., Brown R. S., Argos P. A model for the tertiary structure of the 28 residue DNA-binding motif ('zinc finger') common to many eukaryotic transcriptional regulatory proteins. Protein Eng. 1988 Sep;2(3):209–218. doi: 10.1093/protein/2.3.209. [DOI] [PubMed] [Google Scholar]
- Goodman C. S., Bastiani M. J., Doe C. Q., du Lac S., Helfand S. L., Kuwada J. Y., Thomas J. B. Cell recognition during neuronal development. Science. 1984 Sep 21;225(4668):1271–1279. doi: 10.1126/science.6474176. [DOI] [PubMed] [Google Scholar]
- Hope I. A., Struhl K. Functional dissection of a eukaryotic transcriptional activator protein, GCN4 of yeast. Cell. 1986 Sep 12;46(6):885–894. doi: 10.1016/0092-8674(86)90070-x. [DOI] [PubMed] [Google Scholar]
- Lee K. J., Freeman M., Steller H. Expression of the disconnected gene during development of Drosophila melanogaster. EMBO J. 1991 Apr;10(4):817–826. doi: 10.1002/j.1460-2075.1991.tb08014.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
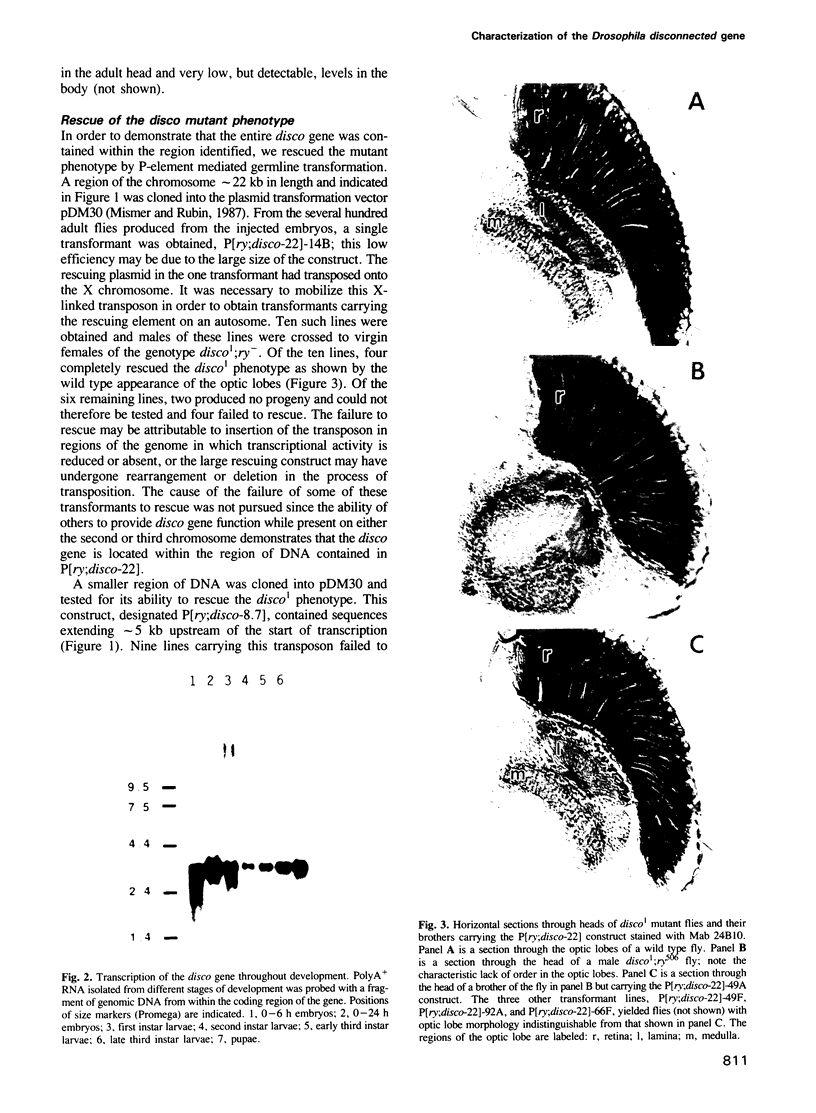
- Levy L. S., Ganguly R., Ganguly N., Manning J. E. The selection, expression, and organization of a set of head-specific genes in Drosophila. Dev Biol. 1982 Dec;94(2):451–464. doi: 10.1016/0012-1606(82)90362-1. [DOI] [PubMed] [Google Scholar]
- Ma J., Ptashne M. Deletion analysis of GAL4 defines two transcriptional activating segments. Cell. 1987 Mar 13;48(5):847–853. doi: 10.1016/0092-8674(87)90081-x. [DOI] [PubMed] [Google Scholar]
- Melamed J., Trujillo-Cenóz O. The fine structure of the eye imagina disks in muscoid flies. J Ultrastruct Res. 1975 Apr;51(1):79–93. doi: 10.1016/s0022-5320(75)80010-4. [DOI] [PubMed] [Google Scholar]
- Milbrandt J. A nerve growth factor-induced gene encodes a possible transcriptional regulatory factor. Science. 1987 Nov 6;238(4828):797–799. doi: 10.1126/science.3672127. [DOI] [PubMed] [Google Scholar]
- Mismer D., Rubin G. M. Analysis of the promoter of the ninaE opsin gene in Drosophila melanogaster. Genetics. 1987 Aug;116(4):565–578. doi: 10.1093/genetics/116.4.565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ptashne M. How eukaryotic transcriptional activators work. Nature. 1988 Oct 20;335(6192):683–689. doi: 10.1038/335683a0. [DOI] [PubMed] [Google Scholar]
- Robertson H. M., Preston C. R., Phillis R. W., Johnson-Schlitz D. M., Benz W. K., Engels W. R. A stable genomic source of P element transposase in Drosophila melanogaster. Genetics. 1988 Mar;118(3):461–470. doi: 10.1093/genetics/118.3.461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scazzocchio C. Of moulds and men, or two fingers are not better than one. Trends Genet. 1990 Oct;6(10):311–313. doi: 10.1016/0168-9525(90)90238-2. [DOI] [PubMed] [Google Scholar]
- Schuh R., Aicher W., Gaul U., Côté S., Preiss A., Maier D., Seifert E., Nauber U., Schröder C., Kemler R. A conserved family of nuclear proteins containing structural elements of the finger protein encoded by Krüppel, a Drosophila segmentation gene. Cell. 1986 Dec 26;47(6):1025–1032. doi: 10.1016/0092-8674(86)90817-2. [DOI] [PubMed] [Google Scholar]
- Seecof R. L., Kaplan W. D., Futch D. G. Dosage compensation for enzyme activities in Drosophila melanogaster. Proc Natl Acad Sci U S A. 1969 Feb;62(2):528–535. doi: 10.1073/pnas.62.2.528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spradling A. C., Rubin G. M. Transposition of cloned P elements into Drosophila germ line chromosomes. Science. 1982 Oct 22;218(4570):341–347. doi: 10.1126/science.6289435. [DOI] [PubMed] [Google Scholar]
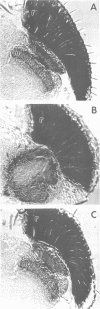
- Steller H., Fischbach K. F., Rubin G. M. Disconnected: a locus required for neuronal pathway formation in the visual system of Drosophila. Cell. 1987 Sep 25;50(7):1139–1153. doi: 10.1016/0092-8674(87)90180-2. [DOI] [PubMed] [Google Scholar]
- Tix S., Minden J. S., Technau G. M. Pre-existing neuronal pathways in the developing optic lobes of Drosophila. Development. 1989 Apr;105(4):739–746. doi: 10.1242/dev.105.4.739. [DOI] [PubMed] [Google Scholar]
- Trujillo-Cenóz O., Melamed J. The development of the retina-lamina complex in muscoid flies. J Ultrastruct Res. 1973 Mar;42(5):554–581. doi: 10.1016/s0022-5320(73)80027-9. [DOI] [PubMed] [Google Scholar]
- Wharton K. A., Yedvobnick B., Finnerty V. G., Artavanis-Tsakonas S. opa: a novel family of transcribed repeats shared by the Notch locus and other developmentally regulated loci in D. melanogaster. Cell. 1985 Jan;40(1):55–62. doi: 10.1016/0092-8674(85)90308-3. [DOI] [PubMed] [Google Scholar]
- Wilson C., Pearson R. K., Bellen H. J., O'Kane C. J., Grossniklaus U., Gehring W. J. P-element-mediated enhancer detection: an efficient method for isolating and characterizing developmentally regulated genes in Drosophila. Genes Dev. 1989 Sep;3(9):1301–1313. doi: 10.1101/gad.3.9.1301. [DOI] [PubMed] [Google Scholar]
- Zinn K., McAllister L., Goodman C. S. Sequence analysis and neuronal expression of fasciclin I in grasshopper and Drosophila. Cell. 1988 May 20;53(4):577–587. doi: 10.1016/0092-8674(88)90574-0. [DOI] [PubMed] [Google Scholar]