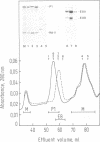
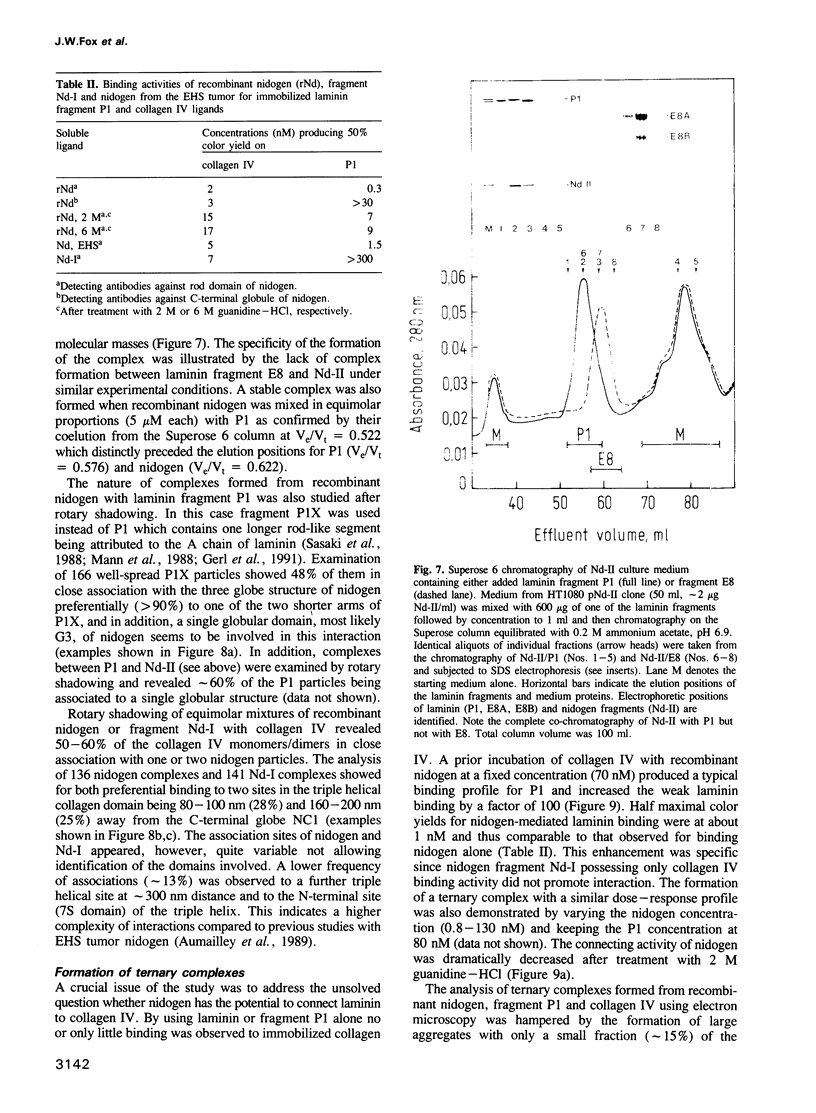
Abstract
Recombinant mouse nidogen and two fragments were produced in mammalian cells and purified from culture medium without resorting to denaturing conditions. The truncated products were fragments Nd-I (positions 1-905) comprising the N-terminal globule and rod-like domain and Nd-II corresponding mainly to the C-terminal globule (position 906-1217). Recombinant nidogen was indistinguishable from authentic nidogen obtained by guanidine dissociation from tumor tissue with respect to size, N-terminal sequence, CD spectra and immunochemical properties. They differed in protease stability and shape indicating that the N-terminal domain of the more native, recombinant protein consists of two globules connected by a flexible segment. This established a new model for the shape of nidogen consisting of three globes of variable mass (31-56 kDa) connected by either a rod-like or a thin segment. Recombinant nidogen formed stable complexes (Kd less than or equal to 1 nM) with laminin and collagen IV in binding assays with soluble and immobilized ligands and as shown by electron microscopy. Inhibition assays demonstrated different binding sites on nidogen for both ligands with different specificities. This was confirmed in studies with fragment Nd-I binding to collagen IV and fragment Nd-II binding to laminin fragment P1. In addition, recombinant nidogen but not Nd-I was able to bridge between laminin or P1 and collagen IV. Formation of such ternary complexes implicates a similar role for nidogen in the supramolecular organization of basement membranes.
Full text
PDF








Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alitalo K., Vaheri A., Krieg T., Timpl R. Biosynthesis of two subunits of type IV procollagen and of other basement membrane proteins by a human tumor cell line. Eur J Biochem. 1980 Aug;109(1):247–255. doi: 10.1111/j.1432-1033.1980.tb04790.x. [DOI] [PubMed] [Google Scholar]
- Aumailley M., Wiedemann H., Mann K., Timpl R. Binding of nidogen and the laminin-nidogen complex to basement membrane collagen type IV. Eur J Biochem. 1989 Sep 1;184(1):241–248. doi: 10.1111/j.1432-1033.1989.tb15013.x. [DOI] [PubMed] [Google Scholar]
- Beck K., Hunter I., Engel J. Structure and function of laminin: anatomy of a multidomain glycoprotein. FASEB J. 1990 Feb 1;4(2):148–160. doi: 10.1096/fasebj.4.2.2404817. [DOI] [PubMed] [Google Scholar]
- Boshart M., Weber F., Jahn G., Dorsch-Häsler K., Fleckenstein B., Schaffner W. A very strong enhancer is located upstream of an immediate early gene of human cytomegalovirus. Cell. 1985 Jun;41(2):521–530. doi: 10.1016/s0092-8674(85)80025-8. [DOI] [PubMed] [Google Scholar]
- Carlin B., Jaffe R., Bender B., Chung A. E. Entactin, a novel basal lamina-associated sulfated glycoprotein. J Biol Chem. 1981 May 25;256(10):5209–5214. [PubMed] [Google Scholar]
- Chen C., Okayama H. High-efficiency transformation of mammalian cells by plasmid DNA. Mol Cell Biol. 1987 Aug;7(8):2745–2752. doi: 10.1128/mcb.7.8.2745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Desjardins M., Bendayan M. Heterogenous distribution of type IV collagen, entactin, heparan sulfate proteoglycan, and laminin among renal basement membranes as demonstrated by quantitative immunocytochemistry. J Histochem Cytochem. 1989 Jun;37(6):885–897. doi: 10.1177/37.6.2723404. [DOI] [PubMed] [Google Scholar]
- Desjardins M., Bendayan M. Ontogenesis of glomerular basement membrane: structural and functional properties. J Cell Biol. 1991 May;113(3):689–700. doi: 10.1083/jcb.113.3.689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Durkin M. E., Chakravarti S., Bartos B. B., Liu S. H., Friedman R. L., Chung A. E. Amino acid sequence and domain structure of entactin. Homology with epidermal growth factor precursor and low density lipoprotein receptor. J Cell Biol. 1988 Dec;107(6 Pt 2):2749–2756. doi: 10.1083/jcb.107.6.2749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dziadek M., Paulsson M., Timpl R. Identification and interaction repertoire of large forms of the basement membrane protein nidogen. EMBO J. 1985 Oct;4(10):2513–2518. doi: 10.1002/j.1460-2075.1985.tb03964.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dziadek M., Timpl R. Expression of nidogen and laminin in basement membranes during mouse embryogenesis and in teratocarcinoma cells. Dev Biol. 1985 Oct;111(2):372–382. doi: 10.1016/0012-1606(85)90491-9. [DOI] [PubMed] [Google Scholar]
- Eaton D. L., Wood W. I., Eaton D., Hass P. E., Hollingshead P., Wion K., Mather J., Lawn R. M., Vehar G. A., Gorman C. Construction and characterization of an active factor VIII variant lacking the central one-third of the molecule. Biochemistry. 1986 Dec 30;25(26):8343–8347. doi: 10.1021/bi00374a001. [DOI] [PubMed] [Google Scholar]
- Engel J., Furthmayr H. Electron microscopy and other physical methods for the characterization of extracellular matrix components: laminin, fibronectin, collagen IV, collagen VI, and proteoglycans. Methods Enzymol. 1987;145:3–78. doi: 10.1016/0076-6879(87)45003-9. [DOI] [PubMed] [Google Scholar]
- Engel J., Schalch W. Antibody binding constants from Farr test and other radioimmunoassays. A theoretical and experimental analysis. Mol Immunol. 1980 May;17(5):675–680. doi: 10.1016/0161-5890(80)90166-2. [DOI] [PubMed] [Google Scholar]
- Fiers W., Contreras R., Haegemann G., Rogiers R., Van de Voorde A., Van Heuverswyn H., Van Herreweghe J., Volckaert G., Ysebaert M. Complete nucleotide sequence of SV40 DNA. Nature. 1978 May 11;273(5658):113–120. doi: 10.1038/273113a0. [DOI] [PubMed] [Google Scholar]
- Gough N. M. Rapid and quantitative preparation of cytoplasmic RNA from small numbers of cells. Anal Biochem. 1988 Aug 15;173(1):93–95. doi: 10.1016/0003-2697(88)90164-9. [DOI] [PubMed] [Google Scholar]
- Klein G., Langegger M., Timpl R., Ekblom P. Role of laminin A chain in the development of epithelial cell polarity. Cell. 1988 Oct 21;55(2):331–341. doi: 10.1016/0092-8674(88)90056-6. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Mann K., Deutzmann R., Aumailley M., Timpl R., Raimondi L., Yamada Y., Pan T. C., Conway D., Chu M. L. Amino acid sequence of mouse nidogen, a multidomain basement membrane protein with binding activity for laminin, collagen IV and cells. EMBO J. 1989 Jan;8(1):65–72. doi: 10.1002/j.1460-2075.1989.tb03349.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mann K., Deutzmann R., Timpl R. Characterization of proteolytic fragments of the laminin-nidogen complex and their activity in ligand-binding assays. Eur J Biochem. 1988 Dec 1;178(1):71–80. doi: 10.1111/j.1432-1033.1988.tb14430.x. [DOI] [PubMed] [Google Scholar]
- Nagayoshi T., Sanborn D., Hickok N. J., Olsen D. R., Fazio M. J., Chu M. L., Knowlton R., Mann K., Deutzmann R., Timpl R. Human nidogen: complete amino acid sequence and structural domains deduced from cDNAs, and evidence for polymorphism of the gene. DNA. 1989 Oct;8(8):581–594. doi: 10.1089/dna.1989.8.581. [DOI] [PubMed] [Google Scholar]
- Olsen D. R., Nagayoshi T., Fazio M., Mattei M. G., Passage E., Weil D., Timpl R., Chu M. L., Uitto J. Human nidogen: cDNA cloning, cellular expression, and mapping of the gene to chromosome Iq43. Am J Hum Genet. 1989 Jun;44(6):876–885. [PMC free article] [PubMed] [Google Scholar]
- Paulsson M., Aumailley M., Deutzmann R., Timpl R., Beck K., Engel J. Laminin-nidogen complex. Extraction with chelating agents and structural characterization. Eur J Biochem. 1987 Jul 1;166(1):11–19. doi: 10.1111/j.1432-1033.1987.tb13476.x. [DOI] [PubMed] [Google Scholar]
- Paulsson M., Deutzmann R., Dziadek M., Nowack H., Timpl R., Weber S., Engel J. Purification and structural characterization of intact and fragmented nidogen obtained from a tumor basement membrane. Eur J Biochem. 1986 May 2;156(3):467–478. doi: 10.1111/j.1432-1033.1986.tb09605.x. [DOI] [PubMed] [Google Scholar]
- Paulsson M., Dziadek M., Suchanek C., Huttner W. B., Timpl R. Nature of sulphated macromolecules in mouse Reichert's membrane. Evidence for tyrosine O-sulphate in basement-membrane proteins. Biochem J. 1985 Nov 1;231(3):571–579. doi: 10.1042/bj2310571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sasaki M., Kleinman H. K., Huber H., Deutzmann R., Yamada Y. Laminin, a multidomain protein. The A chain has a unique globular domain and homology with the basement membrane proteoglycan and the laminin B chains. J Biol Chem. 1988 Nov 15;263(32):16536–16544. [PubMed] [Google Scholar]
- Schittny J. C., Timpl R., Engel J. High resolution immunoelectron microscopic localization of functional domains of laminin, nidogen, and heparan sulfate proteoglycan in epithelial basement membrane of mouse cornea reveals different topological orientations. J Cell Biol. 1988 Oct;107(4):1599–1610. doi: 10.1083/jcb.107.4.1599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sorokin L., Sonnenberg A., Aumailley M., Timpl R., Ekblom P. Recognition of the laminin E8 cell-binding site by an integrin possessing the alpha 6 subunit is essential for epithelial polarization in developing kidney tubules. J Cell Biol. 1990 Sep;111(3):1265–1273. doi: 10.1083/jcb.111.3.1265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Timpl R., Paulsson M., Dziadek M., Fujiwara S. Basement membranes. Methods Enzymol. 1987;145:363–391. doi: 10.1016/0076-6879(87)45021-0. [DOI] [PubMed] [Google Scholar]
- Timpl R. Structure and biological activity of basement membrane proteins. Eur J Biochem. 1989 Apr 1;180(3):487–502. doi: 10.1111/j.1432-1033.1989.tb14673.x. [DOI] [PubMed] [Google Scholar]
- Tsao T., Hsieh J. C., Durkin M. E., Wu C. Y., Chakravarti S., Dong L. J., Lewis M., Chung A. E. Characterization of the basement membrane glycoprotein entactin synthesized in a baculovirus expression system. J Biol Chem. 1990 Mar 25;265(9):5188–5191. [PubMed] [Google Scholar]
- Vara J. A., Portela A., Ortín J., Jiménez A. Expression in mammalian cells of a gene from Streptomyces alboniger conferring puromycin resistance. Nucleic Acids Res. 1986 Jun 11;14(11):4617–4624. doi: 10.1093/nar/14.11.4617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yurchenco P. D., Furthmayr H. Self-assembly of basement membrane collagen. Biochemistry. 1984 Apr 10;23(8):1839–1850. doi: 10.1021/bi00303a040. [DOI] [PubMed] [Google Scholar]
- Yurchenco P. D., Schittny J. C. Molecular architecture of basement membranes. FASEB J. 1990 Apr 1;4(6):1577–1590. doi: 10.1096/fasebj.4.6.2180767. [DOI] [PubMed] [Google Scholar]