Abstract
The aim of the present study was to determine the anticancer potential of three species belonging to the Fallopia genus (Polygonaceae): Fallopia convolvulus (F. convolvulus, Fallopia dumetorum (F. dumetorum) and Fallopia aubertii (F. aubertii). For this purpose, crude extracts were obtained and characterized for their phenolic and flavonoid total content and examined for their anticancer activity on three tumor cell lines: breast cancer (MCF7), colon carcinoma (Caco-2) and cervical cancer (HeLa) cells. The cytotoxic potential of the three species was assessed by MTT assay, cell cycle analysis and by the evaluation of mitochondrial membrane potential (MMP). The acute toxicity of the extracts was evaluated using one in vitro cell model (Vero cells, an African Green monkey kidney cell line) and two invertebrate in vivo models (Daphnia magna and Artemia salina). The highest total phenolic and flavonoid content was found in the F. aubertii flower extracts. The cytotoxic effects of the extracts from F. aubertii and F. convolvulus on all three cell lines were examined at concentrations ranging from 3 to 300 µg/ml. G2/M cell cycle arrest was induced by all the extracts, and a significant increase in the subG1 cell population was observed. The hydroethanolic extract from the flowers of F. aubertii induced cell apoptosis more rapidly than the other extracts. The MMP indicates the involvement of the mitochondria in the induction of apoptosis. A positive correlation between the total phenolic content of the extracts and the IC50 values against the HeLa cells was also noted. None of the extracts exhibited significantly toxic effects. Considering the antitumor potential of F. aubertii and F. convolvulus, these two species may represent a good source of plant extracts with anticancer properties.
Keywords: flavonoids, apoptosis, mitochondrial membrane potential, cell cycle analysis, invertebrate bioassays
Introduction
Cancer is the main cause of mortality and morbidity in Europe following cardiovascular diseases and represents the uncontrolled growth and spread of cells that arises from a change in one single cell (1). Each year, millions of individuals are diagnosed with cancer. The disease accounts for the death of approximately 3.5 million individuals annually worldwide (2). It was estimated that only in Europe, in 2012, 3.45 million new cases of cancer were noted, excluding non-melanoma skin cancer, and 1.75 million deaths occurred from cancer (3).
Throughout history, plant extracts and their purified active components have been the backbone of cancer chemotherapeutics (4). Additionally, structural analogues have been obtained by molecular modifications of the natural compounds and have reinforced the anticancer arsenal (5). It is estimated that over 70% of anticancer compounds are either natural products, or natural product-derived substances (6). The rich diversity of the chemical structures provided by natural resources offers valuable templates for exploring novel molecular scaffolds and is the most significant source of new drug developments (7).
Over the past two decades, flavonoid-rich plant extracts and isolated flavonoids have shown anticancer potential (8). Apigenin, baicalein, luteolin, nobiletin and tangeretin have been shown to be the most effective flavonoids against carcinomas of the stomach, whereas luteolin has been shown to be a promising candidate for the treatment of skin cancer (9,10). Hesperidin has been shown to inhibit human pancreatic cancer cell growth and its use has been suggested for the prevention of pancreatic cancer (11). A number of targets and a variety of action mechanisms have been proposed to explain the cytotoxic effects of flavonoids. Genistein, daidzein, luteolin and quercetin are able to inhibit DNA topoisomerase activity and are considered as potential agents for future use in cancer therapeutics (12). Quercetin, luteolin and kaempferol are promising antitumor agents that can block the cell cycle (13), induce apoptosis (14), inhibit angiogenesis (15) and modulate the epxression of several protein kinases (16). Fisetin acts as a dual inhibitor of the phosphatidylinositol 3-kinase (PI3K)/Akt and mammalian target of rapamycin (mTOR) pathways and has been evaluated for its potential inhibitory role against in vitro (17). Myricetin and quercetagetin have been shown to inhibit the activity of PI3Ks (18). Given the high antitumor potential of these compounds, a number of plant extracts rich in flavonoids have been investigated in order to evaluate their anticancer properties (19,20). The low production cost of the plant extracts compared to the pure compounds and the synergistic effects of the natural compounds are the main advantages for using natural extracts (21).
Fallopia Adans is a plant genus which contains approximately 15 species (22–24). The species are widespread over the northern hemisphere (25). With the exception of Fallopia japonica (Houtt.) Ronse Decr. (syn. Polygonum cuspidatum) and Fallopia multiflora (Thunb.) Haraldson, the therapeutic potential of all the other species has not been investigated in detail (26). These plants are invasive and can easily produce biomass and can therefore be introduced in crops (27). Fallopia convolvulus (F. convolvulus) (L.) Á. Löve and Fallopia dumetorum (F. dumetorum) (L.) Holub (syn. Polygonum dumetorum L.) are native to Europe, and Fallopia aubertii (F. aubertii) (L. Henry) Holub [syn. Fallopia baldschuanica (Regel) Holub] is a subspontaneous species introduced from Central Asia as an ornamental plant (25). Chemotaxonomic studies on the genus Fallopia have shown that all species contain flavonoids with a profile relatively uniform for all species and that quercetin glycosides are the major constituents (28,29). The flavonoid fraction of F. convolvulus consist of glycosides of quercetin, kaempferol, myricetin, apigenin, luteolin, rhamnetin and isorhamnetin (28,30). Three characteristic flavonoid structures have also been found in this species: falloconvolin A and B and quercetin-3-O-(2-E-esinapoxyl)-glucopyranoside (31). In F. aubertii, glycosides of quercetin, kaempferol, apigenin, luteolin, myricetin and several chromones structures have been identified (29,32,33). With the exception of rhamnetin, isorhamnetin and characteristic flavonoids, all other flavonoids have also been found in F. dumetorum (28).
The aim of this study was to examine the effects of various plant extracts from three Fallopia species, F. convolvulus, F. dumetorum and F. aubertii, on cancer cell lines in order to further determine their usefulness. The correlations between the polyphenol and flavonoid content and the cytotoxic effects of these extracts were also evaluated.
Materials and methods
Materials
Folin-Ciocalteau reagent, methanol p.a., ethanol p.a., potassium acetate (CH3COOK), quercetin trihydrate, colchicine and dimethyl sulfoxide (DMSO) were purchased from Sigma-Aldrich Chemie GmbH (Steinheim, Germany). Aluminium chloride hexahydrate (AlCl3 × 6H2O), sodium carbonate anhydrous (Na2CO3) and gallic acid were purchased from Scharlau Co. (Barcelona, Spain). Cervical cancer (HeLa) and colon cancer (Caco-2) cells were purchased from Cellonex, Separations (Randburg, South Africa) and breast (MCF7) cancer cells were purchased from Highveld Biological (Johannesburg, South Africa). The Coulter® DNA Prep™ reagents kit was purchased from Beckman Coulter (Fullerton, CA, USA). 3-(4,5-Dimethyl-1,3-thiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) and 5,5′,6,6′tetrachloro-1,1′,3,3′-tetraethylbenzimidazol-carbocyanine iodide (JC-1) were purchased from Sigma (St. Louis, MO, USA). Dulbecco's modified Eagles medium (DMEM) and fetal bovine serum (FBS) was purchased from Thermo Scientific (Logan, UT, USA).
Plant material and preparation of the extracts
F. convolvulus was harvested from Buftea, Ilfov county (July, 2013), F. dumetorum from Zimnicea, Telorman county (June, 2013) and F. aubertii from Bucharest (October, 2013) Romania. The identity was established by comparing with herbarium specimens from ‘Dimitrie Brandza’ Botanical Garden, Bucharest and voucher specimens are available in the herbarium collection of the Department of Botany and Cell Biology, ‘Carol Davila’ University of Medicine and Pharmacy, Bucharest, Romania. F. convolvulus (C) and F. dumetorum (D) consisted of stems, leaves, flowers and fruits and, F. aubertii consisted of flowers (AF), and stems and leaves (AH). A total of 10 g of each material was grounded (mesh 14) and extracted with 3×100 ml solvent (e, ethanol; ha, ethanol 50%; w, water) under reflux, followed by concentration (rotary evaporator, RVO 004; Ingos, Prague, Czech Republic) and lyophilized at −55°C (CoolSafe ScanVac 55; LaboGene, Lynge, Denmark). For the cell culture experiments, plant extracts (AFha, AFe, AFw and AHha), were reconstituted in DMSO at a final concentration of 100 mg/ml and stored at −20°C until use. Serial dilutions were prepared in order to obtain the following concentrations: 3, 30, 100 and 300 µg/ml.
Phytochemical determinations
The total polyphenol content (TPC) was determined according to the Folin-Ciocalteu method described by González et al (34) (at λ=750 nm) and the total flavonoid content (TFC) was determined using the method with AlCl3 as described by Chang et al (35) and Bazylko et al (at λ=429 nm) (36). All determinations were performed in triplicate and were measured using a UV-VIS spectrophotometer (Halo DB-20-220; Dynamica, Salzburg-Mayrwies, Austria).
The results were calculated using linear calibration curves and are expressed as the means ± SEM of the experiments in milligram gallic acid equivalents (GA equiv.) per gram of dry material (DM) and in milligram quercetin equivalents (Q equiv.) per gram of DM.
Assessment of toxicity
In vitro screening of the extracts for their potential cytotoxicity on cancer cell lines
The human cancer cell lines, MCF7 (breast cancer), Caco-2 (colon carcinoma) and HeLa (cervical cancer), were used for the screening process. All cell lines were grown in DMEM supplemented with 10% fetal bovine serum. Each cell line was seeded in 200 µl aliquots at a cell density of 3×104 cells/ml in 96-well plates and left overnight to attach. For the treatment of each cell line, the medium was replaced with fresh medium containing four concentrations (3, 30, 100 and 300 µg/ml) of extract. The treated cells were incubated at 37°C in a humidified 5% CO2 incubator for 48 h. The medium containing the various treatments was removed prior to the addition of MTT solution to the cells and replaced with 200 µl of medium containing 0.5 mg/ml MTT. The cells were incubated for 3 h. Thereafter, the medium was removed and the blue formazan product was solubilized in DMSO. The absorbance was read at 540 nm using a BioTek® PowerWave XS spectrophotometer (BioTek, Winooski, VT, USA).
Optical density (OD) data were analyzed using Excel and the relative cell viability was determined using quadruplicate readings. Untreated cells were considered to have 100% cell viability. Cell viabilities in other test wells were calculated relative to the untreated controls and expressed as a percentage.
Due to the positive correlation between the concentration sued and the biological effects, HeLa cells were used for the determination of the IC50 value of the cytotoxicity of the Fallopia extracts. HeLa cells were seeded in the same manner as described above for the initial screening protocol. The cells were treated with various concentrations of plant extract (12.5–500 µg/ml) and exposed to the extract for 48 h. In the same manner as described above for the initial screening protocol, MTT was used to determine cell viability following incubation.
Evaluation of toxicity using a normal cell model in vitro
Extracts exhibiting cytotoxicity were tested against a normal cell line. Vero cells (an African green monkey kidney cell line) were used and seeded at a density of 1×105 cells/ml. The determination of the cytotoxicity was performed according to the protocol described above.
Assessment of acute toxicity
The assessment of acute toxicity was determined using two different assays as follows:
i). Artemia salina toxicity assay
Brine shrimp (Artemia salina L.) lethality assay was performed using the procedures described in the study by Meyer et al (37) with some modifications. Brine shrimp cysts were obtained from a local aquarium (Bucharest, Romania) and incubated in artificial sea water (40 g/l salinity) for 24 h in a growth chamber (Sanyo MLR-351H; Sanyo, San Diego, CA, USA) at 25±1°C, under continuous aeration, using a 16-h photoperiod and 8 h of darkness. The newly hatched nauplii were separated from the shells, transferred to fresh sea water with a micropipette and incubated for 24 h. Assays were performed in Petri dishes (d=30 mm). Each dish contained 20 larvae in a final volume of 2,000 µl. The plant extract concentrations were in the range of 1,000–3,000 µg/ml (1,000, 1,500, 2,000, 2,500 and 3,000 µg/ml) and the final DMSO concentration was 1% (v/v). A solution of 1% DMSO in artificial seawater was used as a negative control and colchicine in the range of 0.5–10 µg/ml as a positive control. The concentrations were selected after no lethality was registered during a preliminary test using plant extracts at concentrations of 1–1,000 µg/ml. Each sample was performed in duplicate and each test was run twice. Due to the absence of specific information about the stability of the plant extracts in the presence of light, the bioassay was performed in the dark. After 24 h, the number of survivign organisms was counted and recorded. Larvae were considered dead only if they did not move their appendages for 10 sec during observation.
ii). Daphnia magna toxicity assay
Daphnia magna Straus were maintained parthenogenetically at ‘Carol Davila’ University (Department of Pharmaceutical Botany and Cell Biology) from 2012. Prior to the assay, the daphnids were selected according to their size and kept in fresh water under continuous aeration. The bioassay was performed according to the method described in the study by Fan et al (38) with some modifications (39). Ten daphnids were inserted in 10 ml graduated test tubes, and the plant extracts were added in synthetic media in order to obtain solutions of 1,000, 1,500, 2,000, 2,500 and 3,000 µg extract/ml. The final test solutions were 1% DMSO concentration/10 ml final volume. Synthetic medium with 1% DMSO was used as a negative control and colchicine as a positive control. The daphnids were kept under the same conditions as those described above for the Artemia salina assay and the number of surviving daphnids was counted after 24 h. Each sample was performed in duplicate and each test was run twice. The daphnids were considered dead only if they did not move their appendages for 30 sec during observations.
Cell cycle analysis
HeLa cells were seeded at 5×104 cells/ml in 10 ml aliquots in 10 cm culture dishes and treated with IC50 values of AFha, AFe and Cha. The cells were incubated for 16 and 32 h. After the appropriate incubation period, the HeLa cells were trypsinized for 10 min, re-suspended in phosphate-buffered saline (PBS) and transferred to polypropylene tubes. The Coulter DNA Prep reagents kit was used for DNA cell cycle analysis, as per the manufacturer's instructions. Briefly, 100 µl lysis reagent were added to each tube and incubated for 5 min at room temperature. Thereafter, 500 µl PI (50 µg/ml) were added and the tubes were incubated for 15 min at 37°C. Flow cytometric analysis was performed directly following incubation. A Beckman Coulter Cytomics FC500 was used for all flow cytometricanalysis. FlowJo_V10 was used for analysis.
Analysis of mitochondrial membrane potential (MMP)
A total of 300 µl aliquots of trypsinized cells used for cell cycle analysis was removed from its respective culture dish and placed in a separate polypropylene tube for the analysis of MMP. The cells were centrifuged at 500 × g for 5 min at room temperature and washed with PBS to remove the trypsin. Thereafter, a lipophilic cation dye, JC-1, was added to a final concentration of 2 µg/ml. JC-1 was used to determine a change in the MMP. Cells were incubated for 10 min at room temperature in the dark. The cells were washed using 500 µl PBS and centrifuged at 500 × g for 5 min. The wash step was repeated three times prior to flow cytometric analysis.
Statistical analysis
Data are presented as the means ± standard deviation (SD) from at least three independent experiments. Statistical significance was established by the Student's t-test at the level of p<0.05. The statistical significance of the differences between means was assessed by ANOVA with Tukey's post-hoc tests. P-values <0.05 were considered to indicate statistically significant differences.
The lethality percentage (L%) was plotted against the logarithm of concentrations and the lethality, concentration curves were drawn using the least squares fit method and the lethal concentrations that kill 50% of organisms (LC50) were determined using these curves. The upper and lower limits of the 95% confidence interval (CI 95%) and the correlation coefficient (r2) were also calculated.
Cell viability data and the IC50 values were calculated from the concentration-response data using a mathematical Hill function. All calculations were performed using GraphPad Prism version 5.0 software (GraphPad Software, Inc., La Jolla, CA, USA).
Results and Discussion
The present study focused on the cytotoxic effects of some extracts of F. convolvulus, F. dumetorum and F. aubertii on human cancer cell lines (MCF7, Caco-2 and HeLa) in correlation with their content in flavonoids and phenolic compounds. Additionally, the toxicity of the extracts was assessed by alternative toxicity bioassays using an in vitro model with confluent African green monkey kidney (Vero) cells and two in vivo invertebrate models, Artemia salina and Daphnia magna bioassays.
Extraction yield
Several steps such as milling, grinding, homogenization and extraction are required in order to obtain pharmacological active extracts from plant material (40). Extraction efficiency is affected by all these factors in different ways. Under the same conditions (e.g., particle size, temperature, extraction time, solvent:plant material ratio), the solvent and plant material composition are the most important parameters (41). In this study, we obtained six extracts from three plant species of the genus Fallopia. As F. convolvulus and F. dumetorum have a high TFC and TPC (42,43), we prepared only the hydroethanolic 50% extract (Cha and Dha). From F. aubertii, four extracts were obtained: one from stems and leaves extracted with ethanol 50% (AHha) and three extracts from flowers with water (AFw), ethanol 50% (AFha) and ethanol 96% (AFe). The extraction yields are presented in Table I. The extraction yields ranged from 10.2 to 23.5%. AFw exhibited the highest yield compared to all other extracts, possibly due to the mucilage and other compounds soluble in water. The lowest extraction yield was obtained with ethanol 96%. The results are in agreement with the extraction yields obtained for other medicinal plants rich in flavonoids (44).
Table I.
Yield extraction, TFC and TPC for the Fallopia extracts.
| No. | Extract | Yield of crude extract (%) | TFC (mg Q equiv./g DM) | TPC (mg GAE equiv./g DM) |
|---|---|---|---|---|
| 1 | F. convolvulus (hydroethanolic 50% - Cha) | 18.31 | 33.43±0.3510 | 209.24±2.7899 |
| 2 | F. dumetorum (hydroethanolic 50% - Dha) | 10.21 | 22.73±0.3405 | 77.44±0.8382 |
| 3 | F. aubertii herba (hydroethanolic 50% - AHha) | 13.65 | 30.02±0.3214 | 162.33±4.8745 |
| 4 | F. aubertii flores (hydroethanolic 50% - AFha) | 18.28 | 29.57±0.8453 | 252.96±6.4306 |
| 5 | F. aubertii flores (aqueous - AFw) | 23.05 | 23.43±0.3831 | 154.85±4.8467 |
| 6 | F. aubertii flores (ethanol 96% - AFe) | 12.82 | 48.33±0.7122 | 207.04±1.6670 |
Values are the means ± standard deviation of triplicate analyses. GAE, gallic acid; TFC, total flavonoid content; Q, quercetin; TPC, total phenolic content; DM, dry plant material. Superscript letters e, ha and w indicate the solvent used i.e., 96% ethanol, 50% ethanol or w, water, respectively.
Determination of TPC and TFC
Polyphenols are widespread compounds in plant species. Recent studies have reported a positive correlation between TPC/TFC and anticancer properties and have also shown the various mechanisms of action of these compounds in both in in vitro and in vivo models of cancer (45,46). The TPC of the six different extracts was determined from a linear gallic acid (GAE) standard curve (y=0.1053x+0.0320, r2=0.9993) and the TFC was determined from a linear quercetin standard curve (y=0.0765x-0.0084, r2=0.9997). The TPC and TFC of the tested extracts are presented in Table I. The TPC in the tested extracts ranged from 77.44 to 252.96 mg GA equiv./g DM. AFha showed the highest TPC among all the extracts. The decreasing order of the TPC in the extracts was: AFha>Cha>AFe>AHha>AFw>Dha. All results were statistically significant (ANOVA, p<0.0001). However, the Tukey post-hoc (p<0.05) test revealed no differences between the TPC of Cha and AFe. In addition, no signficant difference was observed in the TPC between AHha and AFw (p>0.05).
The TFC in the six extracts ranged from 22.73 to 48.33 mg Q equiv./g DM. The highest TFC was exhibited by AFe. Ethanol 96% was the best solvent for the extraction of flavonoids for the F. aubertii flowers. The result is statistically significant (p<0.05) by comparison with the extraction with water and ethanol 50% of the same plant material. The decreasing order of the TFC in the extracts was: AFe>Cha>AHha>AFha>AFw>Dha. All the results were statistically significant (ANOVA, p<0.0001). According to the Tukey range test (p<0.05), the TFC value was statistically similar for the herba and flores hydroalcoholic extracts of F. aubertii. Both the TPC and TFC were the lowest in Dha. Among the three species, F. aubertii exhibited the highest TPC and TFC.
Assessment of cytotoxicity in vitro
nitial screening of the extracts for their cytotoxic potential
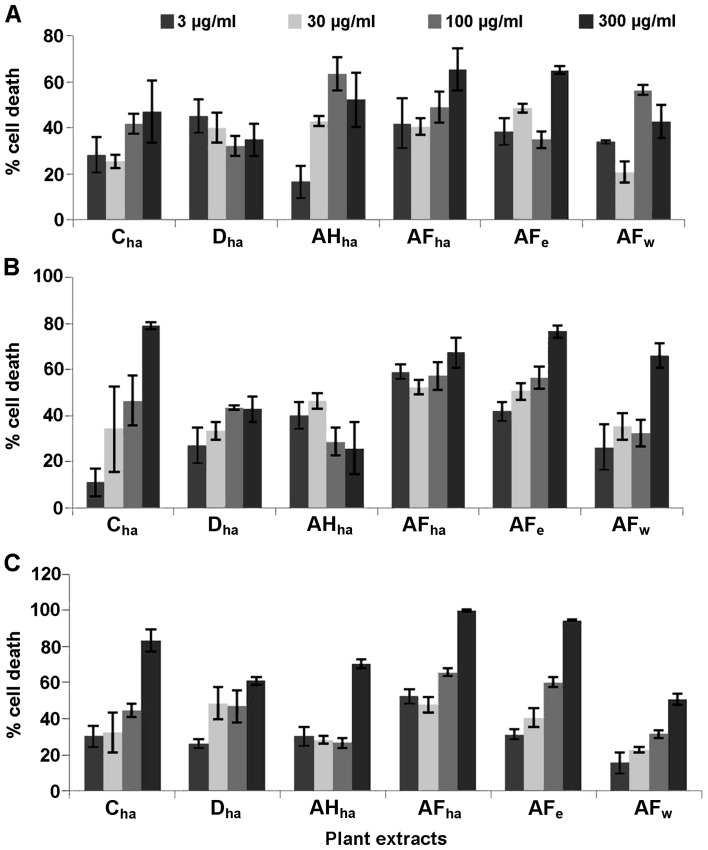
Six Fallopia extracts were screened at 4 different concentrations, namely 3, 30, 100 and 300 µg/ml against the HeLa, Caco-2 and MCF7 cells (Fig. 1) for the determination of their cytotoxic potential. Based on these results, dose-response analysis was performed on the extracts AFha, AFe, Cha and Dha.
Figure 1.
Initial screening of Fallopia extracts against a (A) cervical cancer cell line (MCF7), (B) colon carcinoma cell line (Caco-2) and (C) cervical cancer cell line (HeLa) at the concentrations indicated.
Dose-response analysis and IC50 determination
The cytotoxic effect of the four extracts was determined against the HeLa cells by MTT assay and teh IC50 values were determined. From these results, the concentration of the extracts to be used for further experiments was fixed at 125 µg/ml for Cha and AFe and at 100 µg/ml for AFha (Table II). The IC50 value of Dha was considered too high to pursue its cytotoxic potential. Cytotoxic evaluation was also performed using confluent African green monkey kidney (Vero) cells as a control cell line. All four extracts proved to be non-toxic to the Vero cells (data not shown).
Table II.
IC50 of cytotoxicity to HeLa cells and dose-response curve parameters.
| Extract | IC50 (µg/ml) | IC 95% of IC50 (µg/ml) | Goodness of fit (r2) |
|---|---|---|---|
| F. aubertii flores (hydroethanolic 50% - AFha) | 106.0±5.94 | 96.0–138.2 | 0.9593 |
| F. aubertii flores (ethanol 96% - AFe) | 124.7±8.91 | ND | 0.9453 |
| F. convolvulus (hydroethanolic 50% - Cha) | 122.9±6.98 | 112.9–142.0 | 0.9751 |
| F. dumetorum (hydroethanolic 50% - Dha) | ND | ND | 0.5157 |
ND, not determined. Superscript letters e, ha and w indicate the solvent used i.e., 96% ethanol, 50% ethanol or w, water, respectively.
Cell cycle analysis
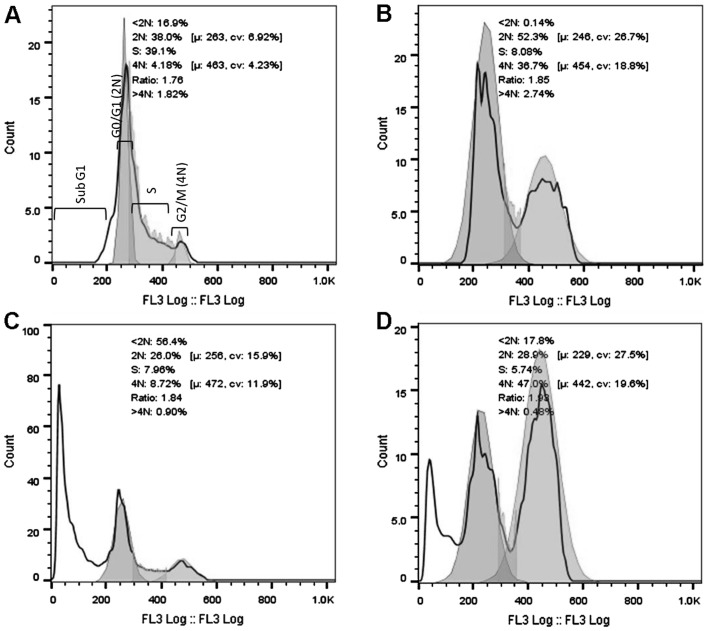
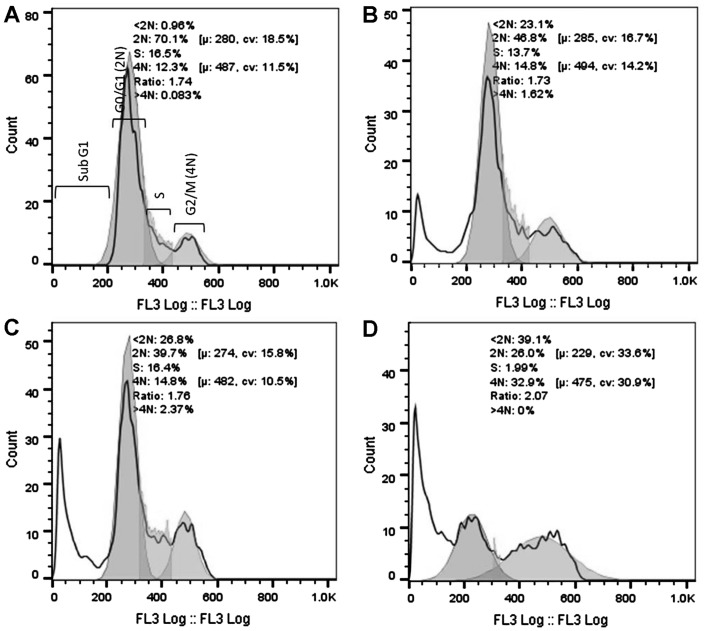
DNA cell cycle analysis was performed using the HeLa cells after 16 and 32 h of exposure to 3 cytotoxic plant extracts. After 16 h of exposure (Fig. 2), a significant increase in the G2/M population was evident for the AFha and Cha extracts. After 16 h, more than half the cell population treated with AFe experienced cell death (subG1). After 32 h of extract exposure (Fig. 3), a significant increase in the subG1 cell population was evident with all extracts.
Figure 2.
Histograms representing DNA cell cycle analysis after 16 h of treatment of the cervical cancer cells (HeLa cells). HeLa cells were treated with (B) 100 µg/ml AFha, (C) 125 µg/ml AFe and (D) 125 µg/ml Cha. (A) Represents untreated control cell population. Cell cycle analysis was performed on a Beckman Coulter Cytomics FC500 flow cytometer following propidium iodide (PI) staining of DNA. FlowJo V10 was used for analysis of results. Ten thousand events were recorded for each sample.
Figure 3.
Histograms representing DNA cell cycle analysis after 32 h of treatment of cervical cancer cells (HeLa cells). HeLa cells were treated with (B) 100 µg/ml AFha, (C) 125 µg/ml AFe and (D) 125 µg/ml Cha. (A) Represents untreated control cell population. Cell cycle analysis was done on a Beckman Coulter Cytomics FC500 flow cytometer following propidium iodide (PI) staining of DNA. FlowJo V10 was used for analysis of results. Ten thousand events were recorded for each sample.
Figs. 2 and 3 show cell cycle analysis used to determine which phase of the cell cycle cells arrest in. It is evident in Fig. 2 that after 16 h of exposure to AFha and Cha, the cells experienced G2/M phase arrest as there was a significant increase in 4N DNA. After 16 h of treatment with AFe, there was a marked increase in the subG1 peak, indicating apoptotic cells. This peak indicates the presence of fragmented DNA, a biochemical hallmark of apoptosis. After 32 h of treatment with the plant extracts, a marked increase in the subG1 cell population was evident, suggesting that the cells were apoptotic.
The mechanism of this G2/M arrest cannot be deduced from propidium iodide (PI) cell cycle analysis and more than one possibility exists. Cdc25B and Cdc25C are phosphatases that regulate the progression of the cell cycle from the G2 phase through to the M phase. They do so by their activity on Cdc2/cyclin A and Cdc2/cyclin B complexes (47). Active Cdc2 complexed to cyclin B1 is required for the progression from the G2 to the M phase. When DNA damage occurs, Cdc25C is deactivated by a cascade process and this results in the phosphorylation and hence, the inactivity of Cdc2/cyclin B and thus arrest of the cell cycle in the G2 phase. G2/M arrest can also occur by problems in the formation of the mitotic spindle and this results in mitotic catastrophe (47). Further studies on the mechanisms of G2/M arrest need to be performed by evaluating the state and levels of Cdc2 and cyclin B proteins, as well as Cdc25C phosphatase. The effects of the plant extracts on tubulin polymerization also need to be determined.
After 16 h of treatment with AFe, the HeLa cells experienced a significant increase in cell death, as indicated by the large subG1 peak. It is thought that cell cycle arrest may have occurred earlier than 16 h and thus was not seen. In order to determine whether cells experience cell cycle arrest, the analysis of the DNA state can be performed at an earlier time interval.
Evaluation of MMP
To determine the onset of the intrinsic pathway of apoptosis, the MMP was evaluated using the lipophilic cationic dye, JC-1. This dye reversibly changes the colour from green to orange as the membrane potential of the mitochondria increases. Thus, an increase in the mean green fluorescence intensity (MFI) would indicate the depolarization of the mitochondrial membranes and hence the involvement of the mitochondria in the induction of apoptosis.
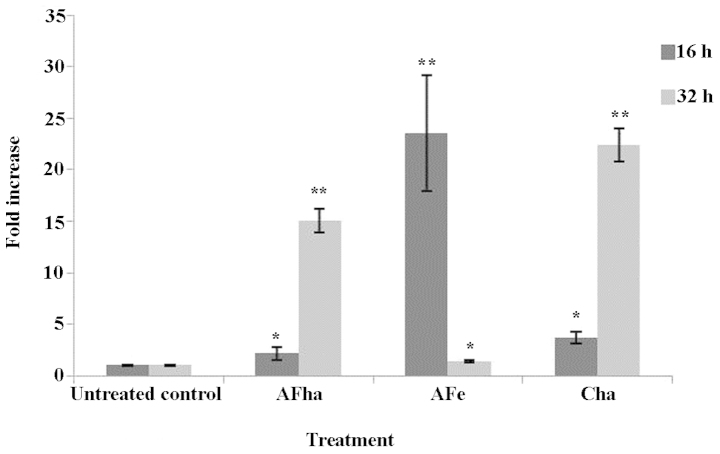
An increase in the MFI in the green channel was evident after 16 h, but more evidently at 32 h of exposure to the AFha and Cha extracts (Fig. 4).
Figure 4.
Fold increase in mean green fluorescence intensity indicating the depolarization of the mitochondria after 16 and 32 h of treatment of cervical cancer cells (HeLa cells). Analysis was performed using a Beckman Coulter Cytomics FC500 flow cytometer following JC-1 staining. Ten thousand events were recorded for each sample. Error bars indicate the SD of triplicate values of a single experiment. Significance was determined using the Students t-test, *p<0.05 and **p<0.005.
Cytotoxic stimuli may induce the permeabilization of cellular membranes and result in the depolarization of the mitochondrial membrane. A method to determine changes in the MMP is by using JC-1. JC-1 forms aggregates when present in high concentrations and the aggregates fluoresce orange (48). If J-aggregates do not form and the dye exists as monomers due to depolarization of the mitochondrial membrane, an increase in green fluorescence will be evident. Fig. 4 shows a significant fold increase in green MFI after 16 h of exposure to AFe and after 32 h of exposure to AFha and Cha. This suggests that the mitochondria are depolarized due to exposure to the extracts and that the intrinsic pathway of apoptosis is activated.
Once the MMP decreases, proteins that are normally found between the inner and outer membrane of the mitochondria are then released and promote the activation of the apoptotic cascades (49).
Assessment of acute toxicity
The assessment of toxicity using alternative methods (e.g., Artemia salina and Daphnia magna bioassays) is widely used due to the many advantages as being inexpensive, time saving and having a high degree of correlation with the acute toxicity (LC50) registered in pharmacotoxicology studies on rodents (mice and rats) mammalian models (50–52).
None of the tested extracts were toxic to both the Artemia salina and Daphnia magna invertebrates. The extracts were first tested in the range of 10 to 1000 µg/ml [10, 50, 100, 250, 500, 750 and 1,000 µg/ml, and no toxicity was observed (L% <0.05) at all tested concentrations]. In order to assess the toxicity at higher concentrations, another experiment was carried out at concentrations between 1,000 and 3,000 µg/ml. LC50 were calculated only at 24 h of exposure due to the lack of information concerning the stability of the extracts and as the extracts tend to precipitate in aqueous DMSO solutions in the second day of the experiments. The brine shrimp lethality test revealed toxic effects only at high concentrations of the extracts from F. dumetorum and F. aubertii. The LC50 exhibited by the five extracts ranged from 1872.16 to 2689.09 µg/ml (Table III). Although the LC50 could be calculated, we consider that the extracts did not present any toxic risk at all. Their toxicity to A. salina was far below the limit of 1,000 µg/ml mentioned by Meyer et al (37). A positive correlation between the concentration and lethality was observed for all six extracts (r2>0.85). With the exception of CEt50, no significant differences were observed (p<0.05). In comparison with the positive control, all LC50 values are at least 1,000-fold higher, thus the toxicity is significantly lower or non-existent. LC50 induced by F. convolvulus extract could not be calculated because of a lethality <35% exhibited at the maximum concentration.
Table III.
Acute toxicity of the extracts to AS and on DM.
| LC50 (µg/ml) | CI 95% of LC50 (µg/ml) | Goodness of fit (r2) | |||||
|---|---|---|---|---|---|---|---|
| No. | Extract | AS | DM | AS | DM | AS | DM |
| 1. | F. convolvulus (hydroethanolic 50% - Cha) | ND | ND | ND | ND | 0.8524 | ND |
| 2. | F. dumetorum (hydroethanolic 50% - Dha) | 2689.09 | 4073.80 | 2664.09–2714.96 | ND | 0.9969 | 0.8769 |
| 3. | F. aubertii herba (hydroethanolic 50% - AHha) | 2576.36 | 2884.03 | 2503.18–2888.15 | ND | 0.9865 | 0.8996 |
| 4. | F. aubertii flores (hydroethanolic 50% - AFha) | 2374.70 | 2398.83 | 2222.08–2522.71 | 2344.22–2454.71 | 0.9471 | 0.9847 |
| 5. | F. aubertii flores (aqueous - AFw) | 2239.55 | 3019.95 | 2186.19–2293.30 | ND | 0.9913 | 0.9399 |
| 6. | F. aubertii flores (ethanol 96% - AFe) | 1872.16 | 2951.20 | 1802.54–1939.97 | ND | 0.9889 | 0.9266 |
| 7 | Colchicine | 1.45 | 4.74 | 1.24–1.69 | 4.50–4.98 | 0.9483 | 0.9880 |
ND, not determined (the value could not be calculated); AS, Artemia salina; DM, Daphnia magna.
The absence of toxicity observed in the brine shrimp lethality test was supported by results of the D. magna bioassay. The LC50 exhibited by the extracts on the daphnids ranged from 2398.83 to 4073.80 µg/ml (Table III). A positive correlation between the concentration and lethality (r2>0.85) and no statistical differences were observed for the determinations performed with the F. dumetorum and F. aubertii extracts (p<0.05). F. convolvulus exhibited no toxicity at all on Daphnia magna, the L% induced by the extract at 3,000 µg/ml being <5%. All results were significantly higher than the positive control (4.74 µg/ml) and the toxicity threshold reported by Guilhermino et al (50) for toxic substances.
In conclusion, of the three species of Fallopia investigated in this study, none was significantly toxic to invertebrate models or to the normal cell model. The highest cytotoxicity to the cancer cells was observed with extracts from the F. convolvulus and F. aubertii flowers. There was a positive correlation between TPC of the extracts and the IC50 values against HeLa cervical cancer cells, with F. aubertii flower hydroethanolic extract (AFha) having the highest TPC content and the lowest IC50. This extract also induced apoptosis at a much earlier time point than the two extracts with the second and third highest TPC values, namely F. convolvulus hydroethanolic extract (Cha) and F. aubertii flower ethanolic extract (AFe), respectively.
Acknowledgements
The authors acknowledge the financial support offered by ‘Carol Davila’ University of Medicine and Pharmacy Bucharest, through research grant no. 33883/11.11.2014. The authors are thankful to PhD Carmen Petronela Comanescu (‘Dimitrie Brandza’ Botanical Garden, Bucharest) for her technical assistance in plants' identification.
Glossary
Abbreviations
- DM
dry material
- DMSO
dimethyl sulfoxide
- JC-1
5,5,6,6tetrachloro-1,1,3,3-tetraethylbenzimidazol-carbocyanine iodide
- MMP
mitochondrial membrane potential
- MTT
3-(4,5-dimethyl- 1,3-thiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide
- PBS
phosphate- buffered saline
- TFC
total flavonoid content
- TPC
total phenolic content
References
- 1.Ridge CA, McErlean AM, Ginsberg MS. Epidemiology of lung cancer. Semin Intervent Radiol. 2013;30:93–98. doi: 10.1055/s-0033-1342949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Baili P, Hoekstra-Weebers J, Van Hoof E, Bartsch HH, Travado L, Garami M, Di Salvo F, Micheli A, Veerus P. (EUROCHIP-3 Working group on Cancer Rehabilitation). Cancer rehabilitation indicators for Europe. Eur J Cancer. 2013;49:1356–1364. doi: 10.1016/j.ejca.2012.10.028. [DOI] [PubMed] [Google Scholar]
- 3.Ferlay J, Steliarova-Foucher E, Lortet-Tieulent J, Rosso S, Coebergh JW, Comber H, Forman D, Bray F. Cancer incidence and mortality patterns in Europe: Estimates for 40 countries in 2012. Eur J Cancer. 2013;49:1374–1403. doi: 10.1016/j.ejca.2012.12.027. [DOI] [PubMed] [Google Scholar]
- 4.Orlikova B, Diederich M. Power from the garden: Plant compounds as inhibitors of the hallmarks of cancer. Curr Med Chem. 2012;19:2061–2087. doi: 10.2174/092986712800228998. [DOI] [PubMed] [Google Scholar]
- 5.Gordaliza M. Natural products as leads to anticancer drugs. Clin Transl Oncol. 2007;9:767–776. doi: 10.1007/s12094-007-0138-9. [DOI] [PubMed] [Google Scholar]
- 6.Karikas GA. Anticancer and chemopreventing natural products: Some biochemical and therapeutic aspects. J BUON. 2010;15:627–638. [PubMed] [Google Scholar]
- 7.Tan G, Gyllenhaal C, Soejarto DD. Biodiversity as a source of anticancer drugs. Curr Drug Targets. 2006;7:265–277. doi: 10.2174/138945006776054942. [DOI] [PubMed] [Google Scholar]
- 8.Singh M, Kaur M, Silakari O. Flavones: An important scaffold for medicinal chemistry. Eur J Med Chem. 2014;84:206–239. doi: 10.1016/j.ejmech.2014.07.013. [DOI] [PubMed] [Google Scholar]
- 9.Wu B, Zhang Q, Shen W, Zhu J. Anti-proliferative and chemosensitizing effects of luteolin on human gastric cancer AGS cell line. Mol Cell Biochem. 2008;313:125–132. doi: 10.1007/s11010-008-9749-x. [DOI] [PubMed] [Google Scholar]
- 10.Sak K. Cytotoxicity of dietary flavonoids on different human cancer types. Pharmacogn Rev. 2014;8:122–146. doi: 10.4103/0973-7847.134247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Patil JR, Chidambara Murthy KN, Jayaprakasha GK, Chetti MB, Patil BS. Bioactive compounds from Mexican lime (Citrus aurantifolia) juice induce apoptosis in human pancreatic cells. J Agric Food Chem. 2009;57:10933–10942. doi: 10.1021/jf901718u. [DOI] [PubMed] [Google Scholar]
- 12.Russo P, Del Bufalo A, Cesario A. Flavonoids acting on DNA topoisomerases: Recent advances and future perspectives in cancer therapy. Curr Med Chem. 2012;19:5287–5293. doi: 10.2174/092986712803833272. [DOI] [PubMed] [Google Scholar]
- 13.Tu SH, Ho CT, Liu MF, Huang CS, Chang HW, Chang CH, Wu CH, Ho YS. Luteolin sensitises drug-resistant human breast cancer cells to tamoxifen via the inhibition of cyclin E2 expression. Food Chem. 2013;141:1553–1561. doi: 10.1016/j.foodchem.2013.04.077. [DOI] [PubMed] [Google Scholar]
- 14.Kuntz S, Wenzel U, Daniel H. Comparative analysis of the effects of flavonoids on proliferation, cytotoxicity, and apoptosis in human colon cancer cell lines. Eur J Nutr. 1999;38:133–142. doi: 10.1007/s003940050054. [DOI] [PubMed] [Google Scholar]
- 15.Mojzis J, Varinska L, Mojzisova G, Kostova I, Mirossay L. Antiangiogenic effects of flavonoids and chalcones. Pharmacol Res. 2008;57:259–265. doi: 10.1016/j.phrs.2008.02.005. [DOI] [PubMed] [Google Scholar]
- 16.Hou D-X, Kumamoto T. Flavonoids as protein kinase inhibitors for cancer chemoprevention: Direct binding and molecular modeling. Antioxid Redox Signal. 2010;13:691–719. doi: 10.1089/ars.2009.2816. [DOI] [PubMed] [Google Scholar]
- 17.Syed DN, Adhami VM, Khan MI, Mukhtar H. Inhibition of Akt/mTOR signaling by the dietary flavonoid fisetin. Anticancer Agents Med Chem. 2013;13:995–1001. doi: 10.2174/18715206113139990129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kong D, Zhang Y, Yamori T, Duan H, Jin M. Inhibitory activity of flavonoids against class I phosphatidylinositol 3-kinase isoforms. Molecules. 2011;16:5159–5167. doi: 10.3390/molecules16065159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Dai J, Mumper RJ. Plant phenolics: Extraction, analysis and their antioxidant and anticancer properties. Molecules. 2010;15:7313–7352. doi: 10.3390/molecules15107313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Dolečková I, Rárová L, Grúz J, Vondrusová M, Strnad M, Kryštof V. Antiproliferative and antiangiogenic effects of flavone eupatorin, an active constituent of chloroform extract of Orthosiphon stamineus leaves. Fitoterapia. 2012;83:1000–1007. doi: 10.1016/j.fitote.2012.06.002. [DOI] [PubMed] [Google Scholar]
- 21.Kitdamrongtham W, Manosroi A, Akazawa H, Gidado A, Stienrut P, Manosroi W, Lohcharoenkal W, Akihisa T, Manosroi J. Potent anti-cervical cancer activity: Synergistic effects of Thai medicinal plants in recipe N040 selected from the MANOSROI III database. J Ethnopharmacol. 2013;149:288–296. doi: 10.1016/j.jep.2013.06.037. [DOI] [PubMed] [Google Scholar]
- 22.Holub J. Fallopia Adans. 1763 instead of Bilderdykia Dum. 1827. Folia Geobot Phytotaxon. 1971;6:171–177. [Google Scholar]
- 23.Haraldson K. Anatomy and taxonomy in Polygonaceae subfam. Polygonoideae Meisn. emend. Jaretzky. Symb. Bot Upsal. 1978;22:1–95. [Google Scholar]
- 24.Decraene LP, Akeroyd JR. Generic limits in Polygonum L. and related genera (Polygonaceae) on the basis of floral characters. J Linn Soc. 1988;98:321–371. doi: 10.1111/j.1095-8339.1988.tb01706.x. [DOI] [Google Scholar]
- 25.Nielsen H, Steinar H. Fallopia Adans. Flora Nordica. Vol. 1: Lycopodiaceae to Polygonaceae. In: Jonsell B, editor. Bergius Foundation; Stockholm: 2000. pp. 273–278. [Google Scholar]
- 26.Song J, Yao H, Li Y, Li X, Lin Y, Liu C, Han J, Xie C, Chen S. Authentication of the family Polygonaceae in Chinese pharmacopoeia by DNA barcoding technique. J Ethnopharmacol. 2009;124:434–439. doi: 10.1016/j.jep.2009.05.042. [DOI] [PubMed] [Google Scholar]
- 27.Tiébré MS, Bizoux JP, Hardy OJ, Bailey JP, Mahy G. Hybridization and morphogenetic variation in the invasive alien Fallopia (Polygonaceae) complex in Belgium. Am J Bot. 2007;94:1900–1910. doi: 10.3732/ajb.94.11.1900. [DOI] [PubMed] [Google Scholar]
- 28.Kim M, Hee Park J, Park CC. Flavonoid chemistry of Fallopia section Fallopia (Polygonaceae) Biochem Syst Ecol. 2000;28:433–441. doi: 10.1016/S0305-1978(99)00084-8. [DOI] [PubMed] [Google Scholar]
- 29.Kim MH, Park JH, Won H, Park CW. Flavonoid chemistry and chromosome numbers of Fallopia section Pleuropterus (Polygonaceae) Can J Bot. 2000;78:1136–1143. doi: 10.1139/b00-079. [DOI] [Google Scholar]
- 30.Smolarz HD. Comparative study on the free flavonoid aglycones in herbs of different species of Polygonum L. Acta Pol Pharm. 2002;59:145–148. [PubMed] [Google Scholar]
- 31.Zhang CF, Chen J, Zhao LQ, Zhang D, Zhang M, Wang ZT. Three new flavonoids from the active extract of Fallopia convolvulus. J Asian Nat Prod Res. 2011;13:136–142. doi: 10.1080/10286020.2010.546357. [DOI] [PubMed] [Google Scholar]
- 32.Olaru OT, Anghel AI, Istudor V, Ancuceanu RV, Dinu M. Contributions to the pharmacognostical and phytobiological study of Fallopia aubertii (L. Henry) Holub. (Polygonaceae) Farmacia. 2013;61:991–999. [Google Scholar]
- 33.Zhao YM, Qi HY, Shi YP. Several chromones from the stems of Polygonum aubertii Henry. J Asian Nat Prod Res. 2010;12:623–628. doi: 10.1080/10286020.2010.485128. [DOI] [PubMed] [Google Scholar]
- 34.González M, Guzmán B, Rudyk R, Romano E, Molina MA. Spectrophotometric determination of phenolic compounds in propolis. Lat Am J Pharm. 2003;22:243–248. [Google Scholar]
- 35.Chang CC, Yang MH, Wen HM, Chern JC. Estimation of total flavonoid content in propolis by two complementary colometric methods. J Food Drug Anal. 2002;10:178–182. [Google Scholar]
- 36.Bazylko A, Parzonko A, Jez W, Osińska E, Kiss AK. Inhibition of ROS production, photoprotection, and total phenolic, flavonoids and ascorbic acid content of fresh herb juice and extracts from the leaves and flowers of Tropaeolum majus. Ind Crops Prod. 2014;55:19–24. doi: 10.1016/j.indcrop.2014.01.056. [DOI] [Google Scholar]
- 37.Meyer BN, Ferrigni NR, Putnam JE, Jacobsen LB, Nichols DE, McLaughlin JL. Brine shrimp: A convenient general bioassay for active plant constituents. Planta Med. 1982;45:31–34. doi: 10.1055/s-2007-971236. [DOI] [PubMed] [Google Scholar]
- 38.Fan W, Cui M, Liu H, Wang C, Shi Z, Tan C, Yang X. Nano-TiO2 enhances the toxicity of copper in natural water to Daphnia magna. Environ Pollut. 2011;159:729–734. doi: 10.1016/j.envpol.2010.11.030. [DOI] [PubMed] [Google Scholar]
- 39.Nitulescu GM, Draghici C, Olaru OT. New potential antitumor pyrazole derivatives: Synthesis and cytotoxic evaluation. Int J Mol Sci. 2013;14:21805–21818. doi: 10.3390/ijms141121805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Gong Y, Liu X, He WH, Xu HG, Yuan F, Gao YX. Investigation into the antioxidant activity and chemical composition of alcoholic extracts from defatted marigold (Tagetes erecta L.) residue. Fitoterapia. 2012;83:481–489. doi: 10.1016/j.fitote.2011.12.013. [DOI] [PubMed] [Google Scholar]
- 41.Ghitescu RE, Volf I, Carausu C, Bühlmann AM, Gilca IA, Popa VI. Optimization of ultrasound-assisted extraction of polyphenols from spruce wood bark. Ultrason Sonochem. 2015;22:535–541. doi: 10.1016/j.ultsonch.2014.07.013. [DOI] [PubMed] [Google Scholar]
- 42.Olaru OT, Ancuceanu RV, Anghel AI, Dinu M. Șeremet OC and Istudor V: Botanical investigation of Fallopia dumetorum (L.) Holub (Polygonaceae) and qualitative and quantitative assessment of its polyphenolic compounds. Acta Med Marisiensis. 2014;60:67–71. [Google Scholar]
- 43.Olaru OT, Anghel AI, Istudor V, Olaru II. The qualitative and quantitative determination of the phenolic compounds in Polygonum convolvulus L. species, Polygonaceae family. Acta Med Marisiensis. 2013;59:162–164. [Google Scholar]
- 44.Sultana B, Anwar F, Ashraf M. Effect of extraction solvent/technique on the antioxidant activity of selected medicinal plant extracts. Molecules. 2009;14:2167–2180. doi: 10.3390/molecules14062167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Sahpazidou D, Geromichalos GD, Stagos D, Apostolou A, Haroutounian SA, Tsatsakis AM, Tzanakakis GN, Hayes AW, Kouretas D. Anticarcinogenic activity of polyphenolic extracts from grape stems against breast, colon, renal and thyroid cancer cells. Toxicol Lett. 2014;230:218–224. doi: 10.1016/j.toxlet.2014.01.042. [DOI] [PubMed] [Google Scholar]
- 46.Sun T, Chen QY, Wu LJ, Yao XM, Sun XJ. Antitumor and antimetastatic activities of grape skin polyphenols in a murine model of breast cancer. Food Chem Toxicol. 2012;50:3462–3467. doi: 10.1016/j.fct.2012.07.037. [DOI] [PubMed] [Google Scholar]
- 47.Busino L, Chiesa M, Draetta GF, Donzelli M. Cdc25A phosphatase: Combinatorial phosphorylation, ubiquitylation and proteolysis. Oncogene. 2004;23:2050–2056. doi: 10.1038/sj.onc.1207394. [DOI] [PubMed] [Google Scholar]
- 48.Smiley ST, Reers M, Mottola-Hartshorn C, Lin M, Chen A, Smith TW, Steele GD, Jr, Chen LB. Intracellular heterogeneity in mitochondrial membrane potentials revealed by a J-aggregate-forming lipophilic cation JC-1. Proc Natl Acad Sci USA. 1991;88:3671–3675. doi: 10.1073/pnas.88.9.3671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Saelens X, Festjens N, Vande Walle L, van Gurp M, van Loo G, Vandenabeele P. Toxic proteins released from mitochondria in cell death. Oncogene. 2004;23:2861–2874. doi: 10.1038/sj.onc.1207523. [DOI] [PubMed] [Google Scholar]
- 50.Guilhermino L, Diamantino T, Silva MC, Soares AM. Acute toxicity test with Daphnia magna: An alternative to mammals in the prescreening of chemical toxicity? Ecotoxicol Environ Saf. 2000;46:357–362. doi: 10.1006/eesa.2000.1916. [DOI] [PubMed] [Google Scholar]
- 51.Hartl M, Humpf HU. Toxicity assessment of fumonisins using the brine shrimp (Artemia salina) bioassay. Food Chem Toxicol. 2000;38:1097–1102. doi: 10.1016/S0278-6915(00)00112-5. [DOI] [PubMed] [Google Scholar]
- 52.Logarto Parra A, Silva Yhebra R, Guerra Sardiñas I, Iglesias Buela L. Comparative study of the assay of Artemia salina L. and the estimate of the medium lethal dose (LD50 value) in mice, to determine oral acute toxicity of plant extracts. Phytomedicine. 2001;8:395–400. doi: 10.1078/0944-7113-00044. [DOI] [PubMed] [Google Scholar]