Abstract
Background
Specialised sexually transmitted infection (STI) clinics in the Netherlands provide STI care for high-risk groups, including female sex workers (FSW), at the clinic and by outreach visiting commercial sex workplaces with a permit. The objective was to investigate the STI positivity rate and determinants of an STI diagnosis among FSW tested by STI clinics in the Netherlands.
Methods
Sexually transmitted infection clinics report demographic, behavioural and diagnostic information of every consultation to the National Institute for Public Health and the Environment. We analysed all consultations of FSW between 2006 and 2013. Trends in STI positivity rate (chlamydia, gonorrhoea, infectious syphilis, HIV and hepatitis B) were analysed using χ2 for trend and logistic regression was used to analyse determinants associated with an STI diagnosis. Differences between consultations at the STI clinic and consultations during outreach were analysed using χ2 tests.
Results
The positivity rate for any STI (overall 9.5 %) was stable from 2006 to 2013. Chlamydia positivity rate (overall 7.1 %) decreased (p < 0.001) and gonorrhoea positivity rate (overall 2.6 %) increased (p < 0.001). For gonorrhoea, the highest positivity rate was found oropharyngeal (2.0 %). Characteristics associated with STI were a younger age [adjusted odds ratio (aOR) 0.96, 95 % confidence interval (CI) 0.95–0.97 per year], a previous STI diagnosis (aOR 1.63, 95 % CI 1.38–1.92) and being notified for an STI by partner notification (aOR 2.61, 95 % CI 2.0–3.40). The STI positivity rate was significantly lower among FSW tested at outreach locations (8.6 %) compared to FSW tested at the STI clinic (11.7 %, p < 0.001).
Conclusions
The STI positivity rate among FSW remained stable, but underlying this was a decreasing chlamydia trend and an increasing gonorrhoea trend, suggesting a shift in STI risks among FSW over time. Condom use during oral sex should be promoted since oropharyngeal gonorrhoea was frequently diagnosed and because of the potential spread of antimicrobial resistant gonococci.
Electronic supplementary material
The online version of this article (doi:10.1186/s12982-015-0034-7) contains supplementary material, which is available to authorized users.
Keywords: Sexually transmitted infections (STI), HIV, STD, Female sex workers, Commercial sex
Background
The policy concerning commercial sex work in the Netherlands is quite different compared to other countries. In many countries, selling of sex is illegal (Belgium [1], South-Africa [2], Canada [3] and most states in the United States of America [4]) or the purchase of sex is considered a legal offence (Sweden [5]). In the Netherlands, commercial sex work is legal since the brothel prohibition was lifted in 2000 [6]. In order to sell sex, employers must obtain a permit from the municipalities. The goal of the legalisation was to regulate voluntary prostitution, to decrease the amount of involuntary prostitution and to improve the social position of sex workers. However, the legislation may have led to an increase in illegal prostitution and some argue that the quality of work circumstances of sex workers (legal and illegal) did not improve [7].
Female sex workers (FSW) are considered a high-risk group for acquisition of sexually transmitted infection (STI) [8, 9], due to their social vulnerability and factors associated with their work like a history of multiple sex partners, inconsistent condom use or co-infection with other STI [10, 11]. Even though in the Netherlands there were lower STI rates among FSW compared to other high-risk groups [12], FSW remain a high-risk group for STI transmission to their numerous clients. There is a potential for some of these clients to act as a bridging population for further spread of STI into the general population [13–15]. Because FSW are at high-risk for STI acquisition and transmission, they are one of the target groups for free STI care at specialised STI clinics located at public health services throughout the Netherlands. These STI clinics provide STI care additional to the regular national health services (like general practitioners), to reach people who might otherwise not seek STI care timely [16]. The STI clinics are funded by the government based on the subsidy regulation Sexual Health [17]. High-risk groups, including FSW, can visit these clinics for STI testing. Some STI clinics also perform outreach activities for FSW, where they routinely visit known commercial sex workplaces such as brothels, sex clubs and window-based prostitution in order to provide STI counselling, testing and hepatitis B vaccination. There is wide variation between STI clinics in the frequency they perform outreach activities, varying from weekly to yearly. All workplaces with a permit for commercial sex work are obligated to provide access to staff of the STI clinic and public health services [18]. Although testing is voluntarily, FSW are advised to go for STI testing four times a year [19].
Although FSW are an important population for STI prevention and control, information about the prevalence and trends of STI among FSW is limited in the Netherlands, compared to other risk groups like young people and men having sex with men [20]. In addition, little is known about the associated determinants for different STI. More insight into the characteristics of this population and the determinants associated with STI occurrence might help target STI control. Therefore, the objective of our study was to investigate the STI positivity rate, trends in STI and associated determinants among FSW who received STI care by the STI clinics (through a visit or by outreach) between 2006 and 2013 in the Netherlands. Secondary aim was to investigate differences between FSW tested through a visit or by outreach.
Methods
For this study, we used surveillance data from STI clinics who all routinely submit an anonymised predefined set of variables (demographics, sexual behaviour, and diagnostic information) of the medical records of each consultation to the Dutch National Institute of Public Health and the Environment (RIVM). Data of the consultations are extracted from the medical records and transferred to the RIVM using a secured web-based application named SOAP [12].
Study population
We included all consultations reported by all STI clinics in the Netherlands between 2006 and 2013 of FSW of 18 years or older, the legal age to perform sex work in the Netherlands. A FSW was defined as a woman who self-reported to have exchanged sex (vaginal, anal or oral) for money or other valuable goods in the 6 months prior to the consultation at an STI clinic. During every consultation, the staff of the STI clinic asks whether the visitor had exchanged sex for money or other valuable goods. Due to the anonymous character of the available data, we cannot retrieve whether the data contains repeated consultations of an individual FSW. Therefore, the unit of analysis is a consultation.
Data
Variables included in this study were age; year of consultation; ethnicity; previous STI diagnosis (chlamydia, gonorrhoea or syphilis) in the last 2 years; ever HIV tested; STI symptoms; current STI diagnosis (chlamydia, gonorrhoea, syphilis, HIV, hepatitis B); whether the FSW was notified for an STI through partner notification and condom use at last sexual contact and whether this was a casual (paying or non-paying) or steady partner. Except for current STI diagnoses, all variables were self-reported. The categories of the variables are presented in Table 1. Due to a policy chance in the registration, ethnicity was a combination variable that consisted of self-defined ethnicity (from 2006 until 2010) and ethnicity based on (parental) country of birth (from 2011 until 2013) [21]. The variables STI symptoms and partner notification were reported since 2007 and the variable condom use at last sexual contact, and whether this contact was a steady or a casual partner were reported since 2011. Degree of urbanisation of the attendees’ residence (five categories based on population density of the postal code area, see Table 1), was added to the database and analysed for the years 2006–2013 [22]. Because we used data that was routinely collected for surveillance purposes and data was obtained anonymously, no ethical approval was needed.
Table 1.
Characteristics of female sex workers tested by STI clinics in the Netherlands, 2006–2013
| 2006 | 2007 | 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | Total | |
|---|---|---|---|---|---|---|---|---|---|
| % | % | % | % | % | % | % | % | % | |
| Number of consultations (n) | 2854 | 3367 | 3810 | 4214 | 4971 | 5301 | 5877 | 5770 | 36,164 |
| Age (median) | 29 | 28 | 29 | 30 | 30 | 30 | 30 | 31 | 30 |
| Ethnicitya | |||||||||
| Native Dutch | 51.7 | 45.1 | 43.9 | 44.3 | 42.6 | 25.8 | 29.6 | 31.6 | 37.6 |
| Turkish | 0.2 | 0.2 | 0.3 | 0.3 | 0.2 | 0.3 | 0.3 | 0.3 | 0.3 |
| North-African | 1.1 | 1.2 | 0.8 | 0.5 | 0.5 | 2.5 | 2.2 | 2.3 | 1.5 |
| Surinamese | 1.4 | 1.3 | 1.1 | 1.0 | 0.6 | 2.9 | 3.7 | 3.6 | 2.2 |
| Dutch Antillean | 0.7 | 0.4 | 0.5 | 0.2 | 0.3 | 1.3 | 1.8 | 1.8 | 1.0 |
| East-European | 15.3 | 16.4 | 17.5 | 21.5 | 23.2 | 34.4 | 33.0 | 31.0 | 25.6 |
| Sub-Saharan African | 2.3 | 2.0 | 1.8 | 1.8 | 2.0 | 2.6 | 2.8 | 2.9 | 2.3 |
| Mid-South American | 8.5 | 8.0 | 9.2 | 9.3 | 9.1 | 10.1 | 11.8 | 11.6 | 10.0 |
| Other European | 13.7 | 19.5 | 19.7 | 16.4 | 16.6 | 4.8 | 5.0 | 4.9 | 11.5 |
| Asian | 2.3 | 2.4 | 2.9 | 2.9 | 3.5 | 5.8 | 5.2 | 6.4 | 4.3 |
| Unknown | 0.2 | 0.2 | 0.3 | 0.2 | 0.1 | 9.5 | 4.5 | 3.4 | 2.8 |
| Rest | 2.5 | 3.2 | 2.1 | 1.7 | 1.4 | 0.1 | 0.1 | 0.2 | 1.2 |
| Ever HIV tested | |||||||||
| No | 27.5 | 20.0 | 16.6 | 14.4 | 14.3 | 12.6 | 10.7 | 9.3 | 14.5 |
| Yes, positive | 0.1 | 0.2 | 0.1 | 0.1 | 0.1 | 0.2 | 0.2 | 0.3 | 0.2 |
| Yes, negative | 63.6 | 72.6 | 76.1 | 79.3 | 80.9 | 81.7 | 83.3 | 85.5 | 79.3 |
| Unknown | 8.8 | 7.2 | 7.2 | 6.2 | 4.7 | 5.5 | 5.8 | 4.9 | 6.1 |
| STI in last 2 yearsb | |||||||||
| No | 50.2 | 55.2 | 82.9 | 81.2 | 82.0 | 84.2 | 83.4 | 81.8 | 77.5 |
| Yes | 18.3 | 13.5 | 11.1 | 10.5 | 10.3 | 7.6 | 8.4 | 8.8 | 10.4 |
| Unknown | 31.6 | 31.3 | 6.0 | 8.3 | 7.6 | 8.2 | 8.2 | 9.4 | 12.0 |
| Symptomsc | |||||||||
| No | 23.8 | 71.2 | 76.5 | 77.5 | 76.0 | 79.6 | 82.5 | 66.5 | |
| Yes | 15.9 | 25.2 | 21.6 | 21.7 | 23.5 | 19.7 | 17.0 | 19.0 | |
| Unknown | 60.3 | 3.6 | 1.9 | 0.8 | 0.5 | 0.7 | 0.5 | 15.4 | |
| Notified for an STI by partner notificationc | |||||||||
| No | 31.6 | 94.5 | 97.9 | 97.9 | 97.7 | 96.2 | 96.5 | 83.1 | |
| Yes | 0.7 | 1.9 | 1.3 | 1.4 | 2.0 | 2.1 | 2.2 | 1.6 | |
| Unknown | 67.7 | 3.6 | 0.8 | 0.6 | 0.3 | 1.7 | 1.3 | 14.5 | |
| Degree of urbanisation | |||||||||
| Very high | 33.6 | 37.5 | 38.8 | 36.7 | 32.4 | 31.2 | 29.8 | 29.7 | 33.1 |
| High | 15.0 | 14.6 | 16.7 | 15.7 | 14.4 | 14.5 | 13.9 | 13.9 | 14.7 |
| Medium | 8.4 | 7.8 | 8.6 | 8.5 | 7.4 | 6.3 | 7.1 | 7.9 | 7.6 |
| Low | 3.0 | 3.6 | 4.3 | 4.2 | 4.1 | 4.0 | 4.6 | 4.0 | 4.1 |
| Very low | 3.3 | 2.8 | 3.4 | 4.1 | 4.1 | 3.8 | 3.9 | 3.2 | 3.6 |
| Unknown | 36.6 | 33.7 | 28.1 | 30.7 | 37.5 | 40.1 | 40.7 | 41.2 | 36.8 |
| Condom use at last sexual contact by type of partnerc | |||||||||
| No condom use with steady partner | 29.1 | 15.7 | 16.8 | 20.3 | |||||
| Yes condom use with steady partner | 2.8 | 5.2 | 2.6 | 3.6 | |||||
| No condom use with casual partner | 7.6 | 10.8 | 9.2 | 9.2 | |||||
| Yes condom use with casual partner | 51.6 | 60.4 | 64.1 | 58.9 | |||||
| Unknown | 8.9 | 8.0 | 7.3 | 8.1 | |||||
STI sexually transmitted infections
aEthnicity was based on self-defined ethnicity for 2006–2010 and on (parental) country of birth from 2011–2013
bDiagnosed with chlamydia, gonorrhoea or syphilis in the last 2 years
cNot reported for all years
Each STI consultation involved laboratory testing and, if indicated, medical examination by a physician or specialised nurse. FSW were offered standard testing for chlamydia, gonorrhoea, syphilis and HIV (nation-wide opt-out policy since 2007, meaning that FSW are standard tested for HIV unless they object [23, 24]). As FSW are indicated for vaccination against hepatitis B, they are screened for hepatitis B. Chlamydia and gonorrhoea were tested vaginally, oropharyngeal and, if indicated, anorectal. All laboratories used nucleic acid amplified tests (NAAT) to diagnose chlamydia and gonorrhoea. Culture of Neisseria Gonorrhoeae was sometimes performed in symptomatic cases or in positive NAAT tests. Syphilis screening was done using Treponema pallidum haemagglutination or Treponema pallidum particle agglutination (TPHA/TPPA) tests followed by a venereal disease research laboratory (VDRL) test and/or a fluorescent treponemal antibody (FTA) test. Infectious syphilis included diagnoses of primary and secondary syphilis as well as Lues latens recens. HIV was tested by enzyme-linked immunosorbent assay (ELISA) combotest (antibody/p24 antigen) confirmed by Western Blot test. Hepatitis B core antibody was used as screening-test for hepatitis B, and if positive hepatitis B surface antigen and anti-HBs were tested [25].
Outcome measures and statistical analyses
We calculated the STI positivity rate as the number of consultations in which at least one STI (chlamydia, gonorrhoea, infectious syphilis, HIV, hepatitis B) was diagnosed per 100 consultations. STI specific positivity rates were calculated out of those consultations where a laboratory test was performed for these specific STI. To increase the readability, we will report the STI positivity rate among FSW as a percentage when we mean the STI positivity rate per 100 consultations. χ2 test for trend was performed to assess trends in STI positivity rates over time. Positivity rates by anatomic location were calculated for the years 2008–2013.
Uni- and multivariable logistic regression analyses for any STI, chlamydia and gonorrhoea were done for the years 2011–2013, because this represents the most recent situation and this avoids the break in trend concerning the definition of ethnicity. Since it is an explorative study, all variables described above were included in the univariable analysis as well as in the multivariable analysis. For the outcomes infectious syphilis, HIV and hepatitis B, univariable analyses were done for the years 2006–2013, due to the small amount of positive diagnoses.
Most STI clinics (22 of 26) performed outreach activities. Six of those clinics used different coding for the consultations at the clinic and outreach consultations. In order to investigate whether there were differences between FSW tested at the STI clinic and FSW tested during outreach, we analysed the characteristics and STI positivity rate separately according to location of testing using a χ2 test. All analyses were done using SPSS version 18 with a significance level of p < 0.05.
Results
Characteristics of study population
Between 2006 and 2013, 801,864 consultations were registered at the STI clinics of which 36,296 (4.5 %) were among FSW. Of all consultations among FSW 36,164 (99.6 %) were 18 years or older. The number of consultations among FSW aged 18 years or older increased from 2854 (4.1 % of all consultations) in 2006 to 5877 (4.8 % of all consultations) in 2012. In 2013, there were 5770 consultations among FSW (4.3 % of all consultations).
The median age of all FSW was 30 years (interquartile range 24–38 years). In the period 2006–2010 using self-defined ethnicity, the greatest number of consultations were among native Dutch FSW (45.0 %), while in the period 2011–2013 using (parental) country of birth, the greatest number of consultations were among East-European FSW (32.8 %). The percentage of FSW ever tested for HIV increased from 63.7 % in 2006 to 85.8 % in 2013. Of the consultations in 2011–2013 where the last sexual contact was a steady partner, a condom was used in 15.1 %, while a condom was used in 58.9 % of the consultations where the last sexual contact was a casual (paying or non-paying) partner (Table 1).
STI positivity rate
The overall STI positivity rate among FSW was 9.5 %, and was stable throughout the study period (χ2 for trend, p = 0.5). The positivity rate for chlamydia decreased over the years from 8.0 % in 2006 to 6.5 % in 2013 (χ2 for trend, p = 0.03). For gonorrhoea, the positivity rate increased from 2.1 % in 2006 to 3.1 % in 2013 (χ2 for trend, p < 0.001). The positivity rate for infectious syphilis decreased from 0.6 % in 2006 to 0.1 % in 2013 (χ2 for trend, p < 0.001). Positivity rates for HIV and hepatitis B remained stable [overall 0.1 % (p = 0.27) and 1.0 % (p = 0.12) respectively] (Fig. 1).
Fig. 1.
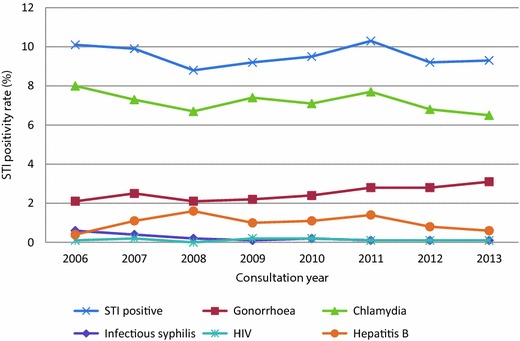
STI positivity rate among FSW tested by STI clinics in the Netherlands, 2006–2013
Table 2 shows the positivity rate by anatomic location for chlamydia and gonorrhoea. For chlamydia, the highest positivity rate was found anorectal (6.2 %), while for gonorrhoea the highest positivity rate was found oropharyngeal (2.0 %). Of the 780 gonorrhoea diagnoses between 2008 and 2013, 418 (53.6 %) were diagnosed in the oropharynx. This percentage increased from 35.8 % in 2008 to 59.0 % in 2013. There was also an increase in the percentage of consultations where gonorrhoea was tested oropharyngeal; of the 3810 consultations in 2008, there were 1870 consultations (49.1 %) where gonorrhoea was tested oropharyngeal, in 2013 this was in 4725 of the 5770 consultations (81.9 %).
Table 2.
Number of tests and positivity rate for chlamydia and gonorrhoea by anatomic location
| 2008 | 2009 | 2010 | 2011 | 2012 | 2013 | Total | |
|---|---|---|---|---|---|---|---|
| Number of consultations | 3810 | 4214 | 4971 | 5301 | 5877 | 5770 | 29,943 |
| Chlamydia diagnosesa | 254 | 311 | 352 | 408 | 397 | 373 | 2095 |
| Chlamydia urogenital | |||||||
| n tested | 3762 | 4177 | 4932 | 5287 | 5861 | 5751 | 29,770 |
| n positive (% positive) | 237 (6.30) | 275 (6.58) | 312 (6.33) | 345 (6.53) | 327 (5.58) | 303 (5.27) | 1799 (6.04) |
| Chlamydia oropharyngeal | |||||||
| n tested | 933 | 1264 | 1925 | 3761 | 4407 | 4511 | 16,801 |
| n positive (% positive) | 18 (1.93) | 31 (2.45) | 51 (2.65) | 89 (2.37) | 86 (1.95) | 67 (1.49) | 342 (2.04) |
| Chlamydia anorectal | |||||||
| n tested | 515 | 599 | 697 | 1163 | 1754 | 2343 | 7071 |
| n positive (% positive) | 38 (7.38) | 47 (7.85) | 42 (6.03) | 80 (6.88) | 101 (5.76) | 129 (5.51) | 437 (6.18) |
| Gonorrhoea diagnosesa | 81 | 93 | 120 | 146 | 162 | 178 | 780 |
| Gonorrhoea urogenital | |||||||
| n tested | 3762 | 4173 | 4933 | 5286 | 5805 | 5749 | 29,708 |
| n positive (% positive) | 57 (1.52) | 55 (1.32) | 84 (1.70) | 94 (1.78) | 104 (1.79) | 103 (1.79) | 497 (1.67) |
| Gonorrhoea oropharyngeal | |||||||
| n tested | 1870 | 2339 | 3305 | 3869 | 4519 | 4725 | 20,627 |
| n positive (% positive) | 29 (1.55) | 37 (1.59) | 70 (2.12) | 84 (2.17) | 93 (2.06) | 105 (2.22) | 418 (2.03) |
| Gonorrhoea anorectal | |||||||
| n tested | 1134 | 1345 | 1657 | 2124 | 2658 | 2822 | 11,740 |
| n positive (% positive) | 22 (1.94) | 19 (1.41) | 18 (1.09) | 27 (1.27) | 30 (1.13) | 29 (1.03) | 145 (1.24) |
aAll infections diagnosed, including those based on pooled samples and infection with an unknown anatomic location. Infections do not add to the total number of diagnoses since infection can occur in multiple locations
Determinants of STI
The multivariable analysis showed that a younger age, previous STI diagnosis, and being notified for an STI were significantly associated with an STI (chlamydia, gonorrhoea, infectious syphilis, HIV and/or hepatitis B), chlamydia and gonorrhoea diagnoses (Table 3). FSW previously tested HIV negative were significantly less likely to be diagnosed with chlamydia compared to FSW who were never HIV tested [adjusted odds ratio (aOR) 0.45, 95 % confidence interval (CI) 0.39–0.53]. An East-European ethnicity was associated with a higher risk for gonorrhoea compared to native Dutch (aOR 1.44, 95 % CI 1.19–1.84), but with a lower risk for chlamydia (aOR 0.79, 95 % CI 0.67–0.93). FSW with a Sub-Saharan African, a Mid-South American or a North-African ethnicity were also less likely to be diagnosed with chlamydia than native Dutch FSW (Table 3).
Table 3.
Uni- and multivariable logistic regression for determinants of STI among FSW, 2011–2013
| Any STIa, N = 16,948 | Chlamydia, N = 16,933b | Gonorrhoea, N = 16,932b | |||||||
|---|---|---|---|---|---|---|---|---|---|
| N positive | Crude OR (95 % CI) | aOR (95 % CI) | N positive | Crude OR (95 % CI) | aOR (95 % CI) | N positive | Crude OR (95 % CI) | aOR (95 % CI) | |
| Age | 1624 | 0.95 (0.95–0.96) | 0.96 (0.95–0.97) | 1178 | 0.95 (0.94–0.95) | 0.95 (0.95–0.96) | 486 | 0.95 (0.94–0.96) | 0.97 (0.96–0.98) |
| Consultation year | 1624 | 0.94 (0.88–1.0) | 0.97 (0.91–1.04) | 1178 | 0.91 (0.85–0.98) | 0.93 (0.86–1.01) | 486 | 1.06 (0.95–1.19) | 1.11 (0.99–1.25) |
| Ethnicityc | |||||||||
| Native Dutch | 483 | Reference | Reference | 385 | Reference | Reference | 124 | Reference | Reference |
| Turkish | 5 | 0.90 (0.36–2.27) | 0.66 (0.26–1.68) | 4 | 0.91 (0.33–2.52) | 0.61 (0.21–1.73) | 2 | 1.43 (0.35–5.95) | 1.37 (0.32–2.55) |
| North African | 30 | 0.77 (0.52–1.13) | 0.59 (0.40–0.87) | 22 | 0.71 (0.46–1.10) | 0.52 (0.33–0.82) | 10 | 1.02 (0.53–1.97) | 0.93 (0.48–1.80) |
| Surinamese | 53 | 0.92 (0.68–1.24) | 0.86 (0.63–1.16) | 41 | 0.89 (0.64–1.24) | 0.82 (0.58–1.15) | 16 | 1.09 (0.64–1.85) | 1.07 (0.63–1.84) |
| Dutch Antilleans | 41 | 1.61 (1.14–2.28) | 1.30 (0.91–1.85) | 31 | 1.50 (1.02–2.21) | 1.15 (0.77–1.72) | 12 | 1.77 (0.97–3.24) | 1.36 (0.73–2.54) |
| Eastern European | 625 | 1.17 (1.03–1.32) | 1.01 (0.88–1.16) | 427 | 0.98 (0.85–1.14) | 0.79 (0.67–0.93) | 214 | 1.55 (1.24–1.95) | 1.44 (1.19–1.84) |
| Sub-Saharan African | 39 | 0.84 (0.60–1.19) | 0.65 (0.46–0.92) | 13 | 0.34 (0.19–0.60) | 0.25 (0.14–0.44) | 9 | 0.76 (0.39–1.51) | 0.78 (0.39–1.56) |
| Mid-South-American | 128 | 0.67 (0.55–0.82) | 0.84 (0.68–1.04) | 85 | 0.56 (0.44–0.71) | 0.72 (0.56–0.93) | 37 | 0.77 (0.53–1.12) | 0.99 (0.67–1.46) |
| Other European | 75 | 0.91 (0.71–1.18) | 0.98 (0.76–1.28) | 54 | 0.82 (0.61–1.10) | 0.86 (0.64–1.17) | 25 | 1.20 (0.78–1.86) | 1.34 (0.86–2.09) |
| Asian | 91 | 0.94 (0.74–1.19) | 1.07 (0.84–1.37) | 74 | 0.96 (0.74–1.25) | 1.10 (0.84–1.44) | 20 | 0.81 (0.50–1.30) | 0.89 (0.55–1.45) |
| Unknown | 91 | 0.52 (0.38–0.69) | 0.57 (0.42–0.77) | 40 | 0.51 (0.37–0.71) | 0.58 (0.41–0.82) | 16 | 0.65 (0.39–1.11) | 0.77 (0.45–1.32) |
| Rest | 3 | 1.38 (0.41–4.67) | 0.98 (0.29–3.36) | 2 | 1.12 (0.26–4.81) | 0.80 (0.18–3.48) | 1 | 1.76 (0.24–13.16) | 1.24 (0.16–9.53) |
| STI in last 2 years | |||||||||
| No | 1257 | Reference | Reference | 915 | Reference | Reference | 369 | Reference | Reference |
| Yes | 206 | 1.75 (1.49–2.05) | 1.63 (1.38–1.92) | 154 | 1.77 (1.48–2.12) | 1.58 (1.30–1.91) | 62 | 1.71 (1.30–2.25) | 1.41 (1.06–1.89) |
| Unknown | 161 | 1.26 (1.06–1.50) | 1.13 (0.91–1.39) | 109 | 1.16 (0.95–1.43) | 1.05 (0.82–1.35) | 55 | 1.46 (1.09–1.94) | 1.11 (0.77–1.59) |
| Ever HIV tested | |||||||||
| No | 325 | Reference | Reference | 256 | Reference | Reference | 79 | Reference | Reference |
| Yes, positive | 3 | 0.39 (0.12–1.26) | 0.66 (0.20–2.21) | 2 | 0.33 (0.08–1.39) | 0.57 (0.13–2.48) | 1 | 0.58 (0.08–4.30) | 1.12 (0.15–8.48) |
| Yes, negative | 1181 | 0.42 (0.37–0.48) | 0.48 (0.42–0.55) | 842 | 0.39 (0.34–0.45) | 0.45 (0.39–0.53) | 362 | 0.58 (0.45–0.75) | 0.77 (0.59–1.01) |
| Unknown | 115 | 0.67 (0.53–0.84) | 0.80 (0.61–1.04) | 78 | 0.57 (0.44–0.75) | 0.71 (0.52–0.96) | 44 | 1.12 (0.77–1.63) | 1.36 (0.87–2.11) |
| Symptoms | |||||||||
| No | 1174 | Reference | Reference | 854 | Reference | Reference | 341 | Reference | Reference |
| Yes | 437 | 0.55 (0.38–1.75) | 1.24 (0.10–1.41) | 312 | 1.50 (1.31–1.72) | 1.12 (0.97–1.30) | 143 | 1.70 (1.39–2.07) | 1.38 (1.12–1.70) |
| Unknown | 13 | 1.64 (0.91–2.95) | 1.37 (0.73–2.56) | 12 | 2.13 (1.16–3.92) | 2.01 (1.04–3.88) | 2 | 0.83 (0.20–3.37) | 0.44 (0.10–1.95) |
| Notified for an STI by partner notification | |||||||||
| No | 1522 | Reference | Reference | 1105 | Reference | Reference | 450 | Reference | Reference |
| Yes | 83 | 2.95 (2.30–3.79) | 2.61 (2.0–3.40) | 61 | 2.84 (2.14–3.77) | 2.19 (1.61–3.00) | 29 | 3.12 (2.11–4.62) | 2.59 (1.70–3.93) |
| Unknown | 19 | 1.20 (0.68–1.77) | 0.92 (0.54–1.54) | 12 | 0.95 (0.53–1.71) | 0.84 (0.44–1.61) | 7 | 1.38 (0.64–2.95) | 1.01 (0.44–2.30) |
| Degree of urbanisation | |||||||||
| Very high | 485 | Reference | Reference | 344 | Reference | Reference | 154 | Reference | Reference |
| High | 220 | 0.97 (0.82–1.15) | 1.06 (0.89–1.26) | 166 | 1.04 (0.86–1.26) | 1.09 (0.89–1.33) | 61 | 0.85 (0.63–1.14) | 0.99 (0.73–1.35) |
| Medium | 113 | 0.99 (0.80–1.23) | 1.10 (0.88–1.38) | 85 | 1.05 (0.82–1.35) | 1.12 (0.86–1.45) | 31 | 0.85 (0.58–1.26) | 1.02 (0.68–1.54) |
| Low | 63 | 0.93 (0.71–1.22) | 1.06 (0.80–1.41) | 42 | 0.87 (0.63–1.21) | 0.92 (0.65–1.30) | 22 | 1.03 (0.65–1.62) | 1.37 (0.86–2.19) |
| Very low | 49 | 0.82 (0.60–1.12) | 0.96 (0.70–1.31) | 39 | 0.93 (0.66–1.31) | 1.07 (0.75–1.52) | 9 | 0.48 (0.24–0.94) | 0.61 (0.31–1.21) |
| Unknown | 694 | 1.07 (0.95–1.21) | 1.07 (0.94–1.21) | 502 | 1.09 (0.95–1.26) | 1.10 (0.95–1.28) | 209 | 1.01 (0.82–1.25) | 1.01 (0.81–1.27) |
| Gonorrhoea | 486 | 121 | 4.83 (3.89–5.98) | 3.77 (3.01–4.72) | |||||
| Chlamydia | 1178 | 121 | 4.83 (3.89–5.98) | 3.76 (3.00–4.72) | |||||
| Condom use at last sexual contact by type of partner | |||||||||
| No condom use steady partner | 368 | Reference | Reference | 279 | Reference | Reference | 95 | Reference | Reference |
| Yes condom use steady partner | 56 | 0.85 (0.64–1.15) | 0.86 (0.64–1.17) | 44 | 0.89 (0.64–1.24) | 0.93 (0.66–1.30) | 12 | 0.72 (0.39–1.31) | 0.73 (0.40–1.35) |
| No condom use casual partner | 200 | 1.23 (1.02–1.47) | 1.19 (0.98–1.44) | 153 | 1.23 (1.00–1.51) | 1.21 (0.98–1.50) | 53 | 1.24 (0.88–1.74) | 1.14 (0.81–1.63) |
| Yes condom use casual partner | 862 | 0.79 (0.69–0.90) | 0.89 (0.78–1.02) | 611 | 0.74 (0.64–0.86) | 0.86 (0.73–1.00) | 276 | 1.00 (0.79–1.27) | 1.11 (0.86–1.42) |
| Unknown | 138 | 0.94 (0.76–1.15) | 1.06 (0.85–1.33) | 91 | 0.81 (0.63–1.04) | 0.91 (0.70–1.19) | 50 | 1.34 (0.95–1.90) | 1.52 (1.04–2.22) |
In italics: OR is statistically significant (p < 0.05)
STI sexually transmitted infections, FSW female sex worker, OR odds ratio, aOR adjusted odds ratio, CI confidence interval
aAt least one positive diagnose for chlamydia, gonorrhoea, infectious syphilis, HIV or hepatitis B
bOnly consultations were included where a related clinical test was done
cEthnicity was based (parental) country of birth from 2011–2013
Infectious syphilis was significantly associated with a previous STI diagnosis and several ethnicities (such as North-African, Dutch Antilleans and East-European). HIV and hepatitis B were more frequently diagnosed among Sub-Saharan African and Mid-South American ethnicities than among native Dutch. Hepatitis B was significantly associated with STI related symptoms. A previous negative HIV test was associated with a lower risk for infectious syphilis, hepatitis B and HIV. Results can be found in Additional file 1.
Outreach versus STI clinic
Table 4 shows the results of the comparison between outreach consultations and consultations performed at the STI clinics. The location of the consultation was known for 8606 consultations (24 % of all consultations) of which 34.2 % were outreach consultations. The STI positivity rate was significantly lower for outreach consultations compared to consultations at the STI clinics (8.6 and 11.7 % respectively, p < 0.001). FSW tested during outreach were older than FSW tested at the STI clinics (median age 32 versus 28 years, p < 0.001) and had more frequently a non-Dutch ethnicity (35.5 versus 30.9 %, p < 0.001).
Table 4.
Characteristics and STI prevalence of FSW tested at STI clinic or tested during outreach activities
| STI clinic | Outreach | P-value | |
|---|---|---|---|
| % | % | ||
| Number of consultations (n) | 5560 | 2946 | |
| Age (median) | 28 | 32 | <0.001 |
| Ethnicity | |||
| Native Dutch | 35.5 | 30.9 | <0.001 |
| Turkish | 0.4 | 0.0 | 0.002 |
| North African | 1.7 | 1.2 | 0.079 |
| Surinamese | 2.1 | 1.1 | <0.001 |
| Dutch Antillean | 1.3 | 1.0 | 0.304 |
| Eastern European | 22.2 | 26.2 | <0.001 |
| Sub-Saharan African | 2.5 | 1.8 | 0.028 |
| Mid-South American | 13.5 | 12.3 | 0.136 |
| Other European | 14.8 | 13.7 | 0.193 |
| Asian | 4.7 | 10.0 | <0.001 |
| Unknown | 1.3 | 1.6 | 0.180 |
| Rest | 0.1 | 0.0 | 0.077 |
| Previous STI | |||
| No | 72.6 | 80.5 | <0.001 |
| Yes | 13.3 | 3.9 | <0.001 |
| Unknown | 14.1 | 15.6 | 0.057 |
| Ever HIV tested | |||
| No | 16.4 | 11.2 | <0.001 |
| Yes, positive | 0.1 | 0.0 | 0.195 |
| Yes, negative | 72.3 | 74.5 | 0.027 |
| Unknown | 11.1 | 14.2 | <0.001 |
| Symptoms | |||
| No | 59.1 | 85.1 | <0.001 |
| Yes | 22.8 | 12.4 | <0.001 |
| Unknown | 18.1 | 2.5 | <0.001 |
| STI positivity rate | |||
| STI overall | 11.7 | 8.6 | <0.001 |
| Chlamydia | 8.4 | 6.2 | <0.001 |
| Gonorrhoea | 3.6 | 2.5 | 0.004 |
| Infectious syphilis | 0.4 | 0.1 | 0.009 |
| HIV | 0.2 | 0.1 | 0.078 |
| Hepatitis B | 1.6 | 1.5 | 0.877 |
Unknown for 27,558 consultations
STI sexually transmitted infections, FSW female sex worker
Discussion
The number of consultations of FSW as well as the proportion FSW of all STI clinic consultations increased during the study period 2006–2013. The overall STI positivity rate among FSW who received care by the STI clinic remained stable, but there was a decrease in the chlamydia positivity rate and an increase in the gonorrhoea positivity rate. Determinants significantly associated with STI positivity were a younger age, a previous STI diagnosis and being notified by partner notification.
STI clinics in the Netherlands provide STI care to specific high-risk groups such as FSW, people below the age of 25 years and people with STI-related symptoms [16]. Compared to all female STI clinic attendees in 2013 [12], FSW were older, less often native Dutch, more often ever tested for HIV, more frequently using a condom in consultations where the last sexual contact was a casual partner and less often diagnosed with an STI (STI positivity rate of 9.5 % compared to 13.2 %) [12]. The chlamydia positivity rate among FSW in 2013 was relatively low compared to the chlamydia positivity rate among all women who attended these STI clinics (6.5 and 12.2 % respectively) [12]. The younger age, which appeared to be a risk factor for STI acquisition, and less frequent condom use with a casual partner among all female STI clinic attendees compared to FSW could explain this difference. Although among FSW the highest chlamydia positivity rate was found anorectal (5.5 % in 2013), this was still lower compared to all women who visited the STI clinic (10.2 % in 2013) [12]. The positivity rate of chlamydia that we found among FSW was comparable to the chlamydia prevalence among FSW in other West-European countries [26–29]. Countries outside Europe showed higher chlamydia prevalence [30, 31]. This could be explained by a lack of epidemiological surveillance systems and FSW-targeted health services in these countries leading to insufficient knowledge about FSW and their STI risk and how to target appropriate prevention. Indeed, there is a lower condom use with clients among FSW in countries outside Europe, compared to countries in Western Europe [30, 32]. The positivity rate of gonorrhoea in our study was comparable to the prevalence among FSW in other European countries (around 5 % or less [27, 28, 33]).
While the positivity rate for chlamydia was lower among FSW than among all women who attended the STI clinic, the positivity rate for gonorrhoea in our study was higher (3.1 % compared to 1.8 %) [12]. This may be explained by the high proportion of East-European FSW who have a higher gonorrhoea prevalence. Another explanation is the higher positivity rate for oropharyngeal gonorrhoea among FSW (2.2 % in 2013) than among all female STI clinic attendees (1.5 % in 2013), probably due to more unsafe oral sex contacts [12, 32]. FSW often do not use a condom when performing oral sex, because they can charge a higher fee for this service (personal communication with health care workers). We did find a relative high percentage of condom use in consultations where the last sexual contact was a casual partner, but perhaps this is reported for vaginal/anal intercourse only since oral sex might not be considered as ‘sex’ [8, 15, 34]. Increasing detection of oropharyngeal infections may partially explain the increase in gonorrhoea positivity. Literature shows a decreased susceptibility of gonorrhoea for ceftriaxone and cefotaxime among FSW in the Netherlands [35]. The threat of emerging resistance of gonorrhoea against the current first line treatment, in combination with the increasing gonorrhoea positivity rate among FSW raises concerns. Apart from condom advice for oral sex, FSW practicing oral sex should always be tested oropharyngeal to actively detect oropharyngeal infections present.
Female sex workers who were younger, previously diagnosed with an STI, or who were notified through partner notification, were at higher risk for chlamydia and gonorrhoea, which is in line with the literature [20, 36]. While East-European FSW were at higher risk for gonorrhoea compared with native Dutch FSW, they were at lower risk for chlamydia. We found no literature to support this. Perhaps this could be explained by the high chlamydia prevalence among heterosexual men and women in the Netherlands, and/or a higher prevalence of gonorrhoea among East-European FSW [12, 32].
The higher STI positivity rate among consultations at the clinic compared to consultations performed during outreach activities is notable. The Prostitution & Health Centre in Amsterdam, which provides care for commercial sex workers, reported comparable results in their annual report [37]. There are several possible explanations for the difference in STI positivity rate between STI clinic and outreach consultations. First, FSW with STI related symptoms or FSW who experienced condom failure actively visit the STI clinic for STI testing. Second, during outreach nurses will redirect FSW with symptoms to the STI clinic for STI testing and physical examination. Third, since only commercial sex workplaces with a permit are obligated to provide the STI clinic access, the outreach consultations will mainly be among legal FSW. Workplaces with a permit are stimulated by the public health services to promote a safe-sex policy and they often have guidelines for condom use and STI testing (personal communication with health care workers). We expect that FSW without a permit will mainly be tested at the STI clinic, since their workplaces are not visited by the STI clinic. We cannot exclude that higher STI rates would be found among these FSW.
Not all FSW (with or without a permit) will be tested at the STI clinic. Reaching FSW currently not seen by STI clinics and FSW outside workplaces with a permit remains a challenge for public health services. Some STI clinics already do internet searches to detect individuals who sell sex and some STI clinics try to visit workplaces without a permit where selling of sex is suspected in order to reach FSW who are currently not seen.
Strengths and limitations
A strength of our study is that we used data from all STI clinics in the Netherlands, which means we covered the whole FSW population tested by STI clinics. Furthermore, many consultations were included in the database and a wide time span was analysed.
A limitation of this study is that most variables (like being a FSW, testing history and condom use) were self-reported which could have led to recall bias and social desirability bias. For example, women reporting not to have exchanged sex for money or other valuable goods are not categorised as sex workers and thus are not included in this study.
Furthermore, we only have information on consultations and we cannot identify repeated consultations for the same FSW. Therefore, odds ratios of determinants associated with STI occurrence could be over- or underestimated since some consultations referred to the same FSW.
Finally, we cannot extrapolate these results to all FSW in the Netherlands. This data only contains FSW tested by STI clinics, but FSW can also visit their general practitioner for STI testing, conduct a self-test, or visit a clinic in their country of origin. This data also cannot be extrapolated to FSW who are not tested at all. We would expect to find a higher STI positivity rate among these FSW, since they are probably not reached by preventive public health services. We suggest further research into STI prevalence among FSW who are not currently tested by STI clinics and how to reach them for counselling and primary prevention.
Conclusions
The positivity rate of any STI among FSW remained stable, but underlying this was a decreasing trend in the chlamydia positivity rate and an increasing trend in the gonorrhoea positivity rate, suggesting a shift in STI risks among FSW over time. Because of the potential transmission of STI, especially gonorrhoea, syphilis and HIV, to the general population and because of the increasing gonorrhoea positivity rate, preventive counselling and increasing awareness of STI risks among FWS remains important as well as regular STI testing of FSW. Condom use during oral sex should be promoted since gonorrhoea was frequently diagnosed in the oropharynx and because of the potential spread of antimicrobial resistant gonococci.
Authors’ contributions
MV analysed and interpreted the data and drafted the manuscript. PW helped interpreting the data and revised the manuscript. HG, MvV and FK revised and corrected the draft. BB helped interpreting the data and revised the manuscript. All authors read and approved the final manuscript.
Acknowledgements
The authors would like to thank all public health nurses and physicians of the STI clinics for their contribution in the data collection and for their input during the interviews.
Compliance with ethical guidelines
Competing interests The authors declare that they have no competing interests.
Abbreviations
- AOR
adjusted odds ratio
- CI
confidence interval
- ELISA
enzyme-linked immunosorbent assay
- FSW
female sex worker
- FTA
fluorescent treponemal antibody
- NAAT
nucleic acid amplified tests
- OR
odds ratio
- RIVM
National Institute of Public Health and the Environment
- TPHA
Treponema pallidum haemagglutination
- TPPA
Treponema pallidum particle agglutination
- VDRL
venereal disease research laboratory
- STI
sexually transmitted infections
Additional file
Additional file 1: Univariable logistic regression for determinants of infectious syphilis, HIV and hepatitis B among female sex workers, 2006 to 2013. Additional file 1 shows the crude odds ratios for the association between determinants included in the study and the outcome of having infectious syphilis, HIV or hepatitis B. Results are presented for female sex workers who were tested by the STI clinic in the Netherlands in the study period 2006 to 2013.
Footnotes
Maud M. A. Verscheijden and Petra J. Woestenberg contributed equally to this work.
Contributor Information
Maud M. A. Verscheijden, Email: m.m.a.verscheijden@student.ru.nl
Petra J. Woestenberg, Email: Petra.Woestenberg@rivm.nl
Hannelore M. Götz, Email: hm.gotz@Rotterdam.nl
Maaike G. van Veen, Email: MvVeen@ggd.amsterdam.nl
Femke D. H. Koedijk, Email: f.koedijk@ggdtwente.nl
Birgit H. B. van Benthem, Email: Birgit.van.Benthem@rivm.nl
References
- 1.Wetboek van Strafrecht België (Penal Code Belgium) Art. 380. 2014.
- 2.Arnott J, Crago AL. Rights not rescue: a report on female, male, and trans sex workers’ human rights in Botswana, Namibia, and South Africa. New York: Open Society Initiative for Southern Afrika; 2009. [Google Scholar]
- 3.Canadian HIV/AIDS Legal Network . Sex, work, rights. Reforming Canadian criminal laws on prostitution. Toronto: Canadian HIV/AIDS Legal Network; 2005. [Google Scholar]
- 4.Legal Prostitution. US Federal and State Prostitution Laws and Related Punishments. 2010. http://prostitution.procon.org/view.resource.php?resourceID=119#3. Accessed 15 Aug 2014.
- 5.Niemi J. What we talk about when we talk about buying sex. Violence Against Women. 2010;16(2):159–172. doi: 10.1177/1077801209355239. [DOI] [PubMed] [Google Scholar]
- 6.Dutch Ministry of Foreign Affairs . FAQ about Dutch Policy on Prostitution 2010. Den Haag: Dutch Ministry of Foreign Affairs; 2010. [Google Scholar]
- 7.Dekker H, Tap R, Homburg G. Evaluatie opheffing bordeelverbod. De sociale positie van prostituees 2006 (Evaluation of the lifting of the brothel prohibition. The social position of prostitutes). Amsterdam: WODC; 2006.
- 8.van Veen MG, Gotz HM, van Leeuwen PA, Prins M, van de Laar MJ. HIV and sexual risk behavior among commercial sex workers in the Netherlands. Arch Sex Behav. 2010;39(3):714–723. doi: 10.1007/s10508-008-9396-z. [DOI] [PubMed] [Google Scholar]
- 9.Wang H, Chen RY, Ding G, Ma Y, Ma J, Jiao JH, et al. Prevalence and predictors of HIV infection among female sex workers in Kaiyuan City, Yunnan Province, China. Int J Infect Dis. 2009;13(2):162–169. doi: 10.1016/j.ijid.2008.05.1229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fernandes FR, Mousquer GJ, Castro LS, Puga MA, Tanaka TS, Rezende GR, et al. HIV seroprevalence and high-risk sexual behavior among female sex workers in Central Brazil. AIDS Care. 2014 doi: 10.1080/09540121.2014.894609. [DOI] [PubMed] [Google Scholar]
- 11.Brussa L. Sex work in Europe. A mapping of the prostitution scene in 25 European countries. Amsterdam: TAMPEP International Foundation; 2009. [Google Scholar]
- 12.Van Aar F, Koedijk FD, Van den Broek IV, Op de Coul ELM, Soetens LC, Woestenberg PJ, et al. Sexually transmitted infections, including HIV, in the Netherlands in 2013. Bilthoven: National Institute for Public Health and the Environment; 2014. [Google Scholar]
- 13.Ghys PD, Jenkins C, Pisani E. HIV surveillance among female sex workers. AIDS. 2001;15(Suppl 3):S33–S40. doi: 10.1097/00002030-200104003-00005. [DOI] [PubMed] [Google Scholar]
- 14.Talbott JR. Size matters: the number of prostitutes and the global HIV/AIDS pandemic. PLoS One. 2007;2(6):e543. doi: 10.1371/journal.pone.0000543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kinsler JJ, Blas MM, Cabral A, Carcamo C, Halsey N, Brown B. Understanding STI risk and condom use patterns by partner type among female sex workers in Peru. Open AIDS J. 2014;8:17–20. doi: 10.2174/1874613601408010017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Van Veen MG, Van Der Sande MA, David S, Koedijk FD. Aanvullende regeling Seksuele Gezondheidszorg (ASG). Betere aansluiting preventie en behandeling (Additional regulations Sexual Health Care. Better joining prevention and treatment) Seksoa. 2012.
- 17.Subsidieregeling publieke gezondheid (Subsidy regulation Public Health). 2015.
- 18.Nadere Regels Seksinrichtingen (Additional Regulations Sex Establishments) Art. 15. 2014.
- 19.Soa Aids Nederland. Soa-test voor prostituees (Soa tests for sex workers). 2013. http://www.soaaids.nl/nl/informatie-voor/prostitutie/seks-soas/soa-test. Accessed 5 June 2015.
- 20.de Coul EL, Warning TD, Koedijk FD, Dutch STIC. Sexual behaviour and sexually transmitted infections in sexually transmitted infection clinic attendees in the Netherlands, 2007–2011. Int J STD AIDS. 2014;25(1):40–51. doi: 10.1177/0956462413491736. [DOI] [PubMed] [Google Scholar]
- 21.Stronks K, Kulu-Glasgow I, Agyemang C. The utility of ‘country of birth’ for the classification of ethnic groups in health research: the Dutch experience. Ethn Health. 2009;14(3):255–269. doi: 10.1080/13557850802509206. [DOI] [PubMed] [Google Scholar]
- 22.Statline. Gebieden in Nederland 2012 (areas in the Netherlands). CBS. 2012. http://statline.cbs.nl/StatWeb/publication/?DM=SLNL&PA=81498ned&D1=0-1,47-48&D2=a&HDR=T&STB=G1&VW=T. Accessed 15 Aug 2014.
- 23.Dukers-Muijrers NH, Niekamp AM, Vergoossen MM, Hoebe CJ. Effectiveness of an opting-out strategy for HIV testing: evaluation of 4 years of standard HIV testing in a STI clinic. Sex Transm Infect. 2009;85(3):226–230. doi: 10.1136/sti.2008.033191. [DOI] [PubMed] [Google Scholar]
- 24.Heijman RL, Stolte IG, Thiesbrummel HF, van Leent E, Coutinho RA, Fennema JS, et al. Opting out increases HIV testing in a large sexually transmitted infections outpatient clinic. Sex Transm Infect. 2009;85(4):249–255. doi: 10.1136/sti.2008.033258. [DOI] [PubMed] [Google Scholar]
- 25.Nederlandse Vereniging voor Dermatologie en Venerologie. Diagnostiek en Behandeling van Seksueel Overdraagbare Aandoeningen (SOA) (Diagnostics and Treatment of STIs). 2012. http://www.nvdv.nl/wp-content/uploads/2014/08/Samenvattingskaart-SOA-2014-2015.pdf. Accessed Aug 2014.
- 26.Mak RP, Van Renterghem L, Traen A. Chlamydia trachomatis in female sex workers in Belgium: 1998–2003. Sex Transm Infect. 2005;81(1):89–90. doi: 10.1136/sti.2004.010272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Vall-Mayans M, Villa M, Saravanya M, Loureiro E, Merono M, Arellano E, et al. Sexually transmitted Chlamydia trachomatis, Neisseria gonorrhoeae, and HIV-1 infections in two at-risk populations in Barcelona: female street prostitutes and STI clinic attendees. Int J Infect Dis. 2007;11(2):115–122. doi: 10.1016/j.ijid.2005.11.005. [DOI] [PubMed] [Google Scholar]
- 28.Platt L, Grenfell P, Bonell C, Creighton S, Wellings K, Parry J, et al. Risk of sexually transmitted infections and violence among indoor-working female sex workers in London: the effect of migration from Eastern Europe. Sex Transm Infect. 2011;87(5):377–384. doi: 10.1136/sti.2011.049544. [DOI] [PubMed] [Google Scholar]
- 29.Folch C, Esteve A, Sanclemente C, Martro E, Lugo R, Molinos S, et al. Prevalence of human immunodeficiency virus, Chlamydia trachomatis, and Neisseria gonorrhoeae and risk factors for sexually transmitted infections among immigrant female sex workers in Catalonia, Spain. Sex Transm Dis. 2008;35(2):178–183. doi: 10.1097/OLQ.0b013e31815a848d. [DOI] [PubMed] [Google Scholar]
- 30.van Gemert C, Stoove M, Kwarteng T, Bulu S, Bergeri I, Wanyeki I, et al. Chlamydia prevalence and associated behaviours among female sex workers in vanuatu: results from an integrated bio-behavioural survey, 2011. AIDS Behav. 2014 doi: 10.1007/s10461-014-0791-2. [DOI] [PubMed] [Google Scholar]
- 31.Esquivel CA, Briones Ezcarzaga ML, Castruita Limones DE, Lazalde Ramos BP, Salas EV, Gutierrez AA, et al. Prevalence of Chlamydia trachomatis infection in registered female sex workers in northern Mexico. Sex Transm Dis. 2003;30(3):195–198. doi: 10.1097/00007435-200303000-00003. [DOI] [PubMed] [Google Scholar]
- 32.Platt L, Jolley E, Rhodes T, Hope V, Latypov A, Reynolds L, et al. Factors mediating HIV risk among female sex workers in Europe: a systematic review and ecological analysis. BMJ Open. 2013 doi: 10.1136/bmjopen-2013-002836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.D’Antuono A, Andalo F, Carla EM, De Tommaso S. Prevalence of STDs and HIV infection among immigrant sex workers attending an STD centre in Bologna, Italy. Sex Transm Infect. 2001;77(3):220. doi: 10.1136/sti.77.3.220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Deering KN, Bhattacharjee P, Bradley J, Moses SS, Shannon K, Shaw SY, et al. Condom use within non-commercial partnerships of female sex workers in southern India. BMC Public Health. 2011;11(Suppl 6):S11. doi: 10.1186/1471-2458-11-S6-S11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Koedijk FD, van Veen MG, de Neeling AJ, Linde GB, van der Sande MA. Increasing trend in gonococcal resistance to ciprofloxacin in the Netherlands, 2006–8. Sex Transm Infect. 2010;86(1):41–45. doi: 10.1136/sti.2009.037135. [DOI] [PubMed] [Google Scholar]
- 36.Sarkar K, Bal B, Mukherjee R, Saha MK, Chakraborty S, Niyogi SK, et al. Young age is a risk factor for HIV among female sex workers—an experience from India. J Infect. 2006;53(4):255–259. doi: 10.1016/j.jinf.2005.11.009. [DOI] [PubMed] [Google Scholar]
- 37.Prostitutie & Gezondheidscentrum P&G292. Jaarverslag 2013 (Annual report 2013 Prostitution and Health Centre). Amsterdam: Prostitutie & Gezondheidscentrum P&G292. 2014.


