Abstract
The OPRM1 A118G polymorphism is the most widely studied μ-opioid receptor (MOR) variant. Although its involvement in acute alcohol effects is well characterized, less is known about the extent to which it alters responses to opioids. Prior work has shown that both electrophysiological and analgesic responses to morphine but not to fentanyl are moderated by OPRM1 A118G variation, but the mechanism behind this dissociation is not known. Here we found that humanized mice carrying the 118GG allele (h/mOPRM1-118GG) were less sensitive than h/mOPRM1-118AA littermates to the rewarding effects of morphine and hydrocodone but not those of other opioids measured with intracranial self-stimulation. Reduced morphine reward in 118GG mice was associated with decreased dopamine release in the nucleus accumbens and reduced effects on GABA release in the ventral tegmental area that were not due to changes in drug potency or efficacy in vitro or receptor-binding affinity. Fewer MOR-binding sites were observed in h/mOPRM1-118GG mice, and pharmacological reduction of MOR availability unmasked genotypic differences in fentanyl sensitivity. These findings suggest that the OPRM1 A118G polymorphism decreases sensitivity to low-potency agonists by decreasing receptor reserve without significantly altering receptor function.
INTRODUCTION
Opiate alkaloids isolated from Papaver somniferum, such as morphine and codeine, have been consumed by humans for their analgesic and euphorigenic effects for thousands of years. During the past century, natural and synthetic opioid analgesics of varying potency and intrinsic efficacy have become mainstays of treatment for acute and chronic pain in inpatient and outpatient settings, although these drugs carry significant abuse liability. From 1999 to 2009, the number of prescriptions for opioid drugs in the United States increased fourfold, while admission rates for opioid abuse treatment programs increased sixfold (Paulozzi et al, 2011). In response, drug makers introduced abuse-deterrent formulations of several opioid analgesics, including oxycodone, which inadvertently increased demand for illegal opioids, such as the acetylated morphine derivative, heroin (Cicero et al, 2012).
The psychological and physiological effects of morphine and other opioids are due to their intrinsic activity at μ-opioid receptors (MORs), which are seven-transmembrane G protein-coupled receptors (GPCRs) expressed at presynaptic and postsynaptic sites throughout the mammalian central nervous system, including the ventral tegmental area (VTA), dorsal and ventral striatum, periaquaductal grey area (PAG), and locus coeruleus. Although several brain loci are associated with opioid abuse, the A10 dopaminergic projection from the mesencephalic VTA to the forebrain nucleus accumbens (NAc) is of particular importance, as it is associated with motivated behavior and reward processing (Schultz et al, 1997; Wise, 1998). In the VTA, opioids act via the Gi/o-coupled MOR to disinhibit dopaminergic neurons by attenuating inhibitory GABAergic tone, which promotes burst firing and enhances dopamine release in terminal fields (Di Chiara and Imperato, 1988; Johnson and North, 1992). Thus pharmacogenetic approaches to understanding MOR activity in mesolimbic dopamine circuitry may help elucidate heritable mechanisms altering opioid sensitivity.
One of the most widely studied functional genetic variants of the MOR involves a single-nucleotide polymorphism (rs1799971) in exon 1 of the MOR gene (OPRM1), in which an adenine-to-guanine substitution at position 118 (118A→G or A118G) corresponds to the loss of a putative glycosylation site in the N-terminal extracellular domain (Bond et al, 1998; Huang et al, 2012). Although the involvement of the OPRM1 A118G polymorphism in the acute effects of alcohol and the treatment of alcohol dependence has been widely studied, less is known about how this variant affects opioid reward and its neural substrates. Here we employed behavioral, cellular, and neurochemical assays to probe pharmacogenetic mechanisms of opioid reward in a humanized mouse model of the OPRM1 A118G polymorphism (h/mOPRM1 A118G mice) in which Oprm1 was excised and replaced with OPRM1 containing either A or G at position 118 (Ramchandani et al, 2010).
MATERIALS AND METHODS
See Supplementary Materials for detailed Materials and Methods.
Intracranial Self-Stimulation (ICSS)
h/mOPRM1-118AA and 118GG mice were implanted with stimulating electrodes in the medial forebrain bundle at the level of the lateral hypothalamus and were conditioned to perform ICSS as previously described (Robinson et al, 2013). After baseline testing, mice were removed from the operant chambers, injected with vehicle (i.p. or s.c.), fentanyl (s.c.), oxycodone (i.p.), buprenorphine (i.p.), morphine (i.p.), hydrocodone (i.p.); cocaine (i.p.), or U69,593 (i.p.), and returned to the chambers for 15 min of testing. Maximum response rate (MAX) and brain stimulation reward (BSR) threshold (θ0) were calculated with custom-designed software and normalized to baseline responses.
Microdialysis and Locomotor Activity
Mice were implanted with unilateral microdialysis guide cannulae aimed at the nucleus accumbens as previously described (Chefer et al, 2013). After baseline sampling, mice were challenged with repeated injections of morphine or fentanyl with microdialysis samples collected every 10 min. Locomotor activity was measured using a Coulbourn Instrument TruScan activity monitoring system (Allentown, PA, USA). Dialysate DA concentrations were analyzed using high-pressure liquid chromatography (HPLC) with electrochemical detection as previously described (Chefer et al, 2013). In the fentanyl/β-funaltrexamine experiment, locomotion was measured in MedAssociates locomotor activity chambers (St Albans, VT) as previously described (Robinson et al, 2013).
Electrophysiological Recordings
Acute horizontal midbrain slices were prepared, and whole-cell patch clamp recordings from VTA dopaminergic neurons were obtained as previously described (Robinson et al, 2013). Miniature inhibitory postsynaptic currents (mIPSCs) were recorded in oxygenated aCSF that contained tetrodotoxin (500 nM) and kynurenic acid (1 μM). Recording pipettes with a resistance of 2.5–3.5 MΩ were filled with a potassium chloride-based internal solution (containing the following, in mM: 128 KCl, 20 NaCl, 1 MgCl2, 1 EGTA, 0.3 CaCl2, 10 HEPES, 2 MgATP, 0.3 Na3GTP) containing Alexa Fluor 594 (Life Technologies, Carlsbad, CA). Following a stable baseline, mIPSCs were recorded for 3 min before and after 3-min exposure to morphine (100 nM) or [D-Ala2, N-MePhe4, Gly-ol]-enkephalin (DAMGO; 100 nM) in the presence or absence of naloxone (3 μM), and postdrug mIPSC frequency and amplitude was normalized to baseline. After each recording, slices were stained for tyrosine hydroxylase (TH), and Alexa Fluor 594-filled cells that expressed TH were considered to be dopaminergic.
Preparation of Membranes and Receptor Binding
For binding assays, brainstem membranes from age-matched male mice were prepared as previously described (Roth, 2013). The subcellular fractionation procedure (Kumar et al, 2010) and determination of the number of h/mOPRM1-118A- and 118G-binding sites in each fraction using a radioligand-binding assay were performed as previously described (Roth, 2013). A competition binding assay was used to determine the binding affinity of β-endorphin, MET-enkephalin, DAMGO, fentanyl, morphine, oxycodone, hydrocodone, and buprenorphine for h/mOPRM1-118A and 118G receptors using freshly prepared membranes as previously described (Roth, 2013). The amount of protein per sample was determined using the Bradford Assay.
Functional cAMP Assays
HEK293 cells were co-transfected with plasmids encoding either h/mOPRM1-118A or 118G receptor DNA (10 ng) and the cAMP biosensor GloSensor-22F (Promega), as previously described (Roth, 2013; Vardy et al, 2013). HEK293 cells were treated with 10 μl DAMGO, morphine, fentanyl, buprenorphine, oxycodone, or hydrocodone prepared in drug buffer (0.1 nM–10 μM). After 20 min, cAMP production was stimulated by addition of 10 μl isoproterenol (1.2 μM) in 8% GloSensor reagent. Luminescence was read on a Wallac MicroBeta TriLux plate scintillation counter (PerkinElmer, Waltham, MA). The transduction coefficient (log(T/Ka)) for each agonist was determined for different levels of receptor reserve (ie, transfection with 0.3–10.0 ng receptor DNA) using the operational model of Black and Leff and normalized to the reference ligand, DAMGO, as previously described (Kenakin et al, 2012).
RESULTS
h/mOPRM1-118AA and 118GG Mice Display Differences in the Rewarding Potency of MOR Agonists
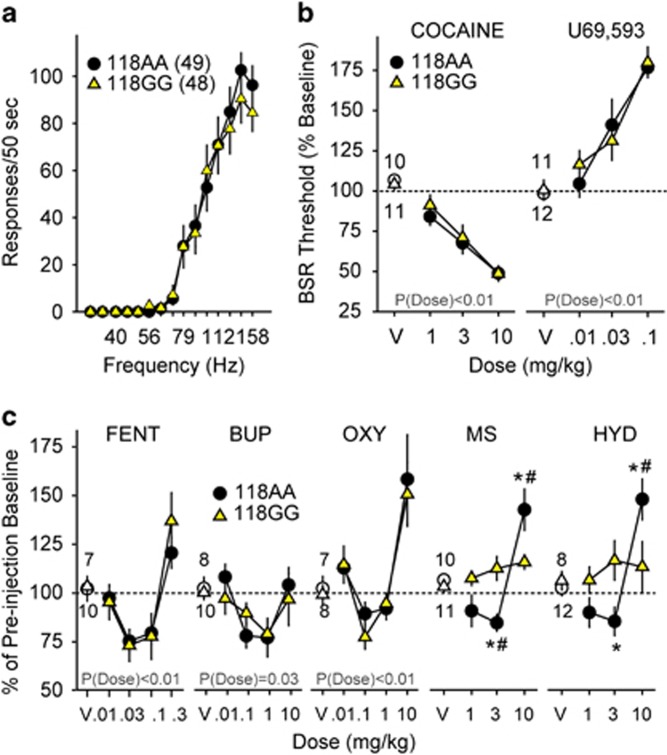
Because the acute rewarding effects of abused substances are critical for the initiation and maintenance of drug taking, we examined the reward-potentiating or -devaluing effects of the MOR agonists morphine, fentanyl, oxycodone, and hydrocodone; the MOR partial agonist buprenorphine; the kappa opioid receptor agonist U69,593; and the psychostimulant cocaine in h/mOPRM1-118AA and 118GG male mice using ICSS. h/mOPRM1-118AA and 118GG littermates displayed no differences in baseline ICSS responding (Figure 1a) measured by average maximum operant response rates (MAX118AA=126.9±11.0 responses/50 s; MAX118GG=107.0±11.5 responses/50 s; t95=1.20, p=0.23) and minimum amount of electrical stimulation (μC) required to sustain responding (θ0 (118AA)=0.42±0.03 μC; θ0 (118GG)=0.41±0.03 μC; t95=0.30, p=0.76). Cocaine (Figure 1b; F3,57=59.1, p<0.001), U69,593 (Figure 1b; F3,63=32.1, p<0.001), fentanyl (Figure 1c; F4,60=15.0, p<0.001), buprenorphine (Figure 1c; F4,64=4.5, p=0.003), and oxycodone (Figure 1c; F4,52=14.7, p<0.001) had dose-dependent effects on BSR threshold in both h/mOPRM1-118AA and GG mice independent of genotype.
Figure 1.
Effects of MOR agonists, U69,593, and cocaine on intracranial self-stimulation (ICSS) in h/mOPRM1-118AA and 118GG mice. (a) Average baseline rate–frequency curves for h/mOPRM1-118AA (black circles; n=49) and 118GG mice (yellow triangles; n=48) performing ICSS. (b and c) Effects of cocaine (1–10 mg/kg i.p.), U69,593 (0.01–0.1 mg/kg i.p.), fentanyl (FENT; 0.01–0.3 mg/kg s.c.), buprenorphine (BUP; 0.01–10 mg/kg i.p.), oxycodone (OXY; 0.01–10 mg/kg i.p.), morphine (MS; 1.0–10 mg/kg i.p.), and hydrocodone (HYD; 1.0–10 mg/kg i.p.) on BSR threshold in h/mOPRM1-118AA (black circles; n=10, 11, 7, 8, 7, 10, and 8, respectively) and 118GG mice (yellow triangles; n=11, 12, 10, 10, 8, 11, and 12, respectively). Values represent mean±SEM. *p<0.05 vs h/mOPRM1-118AA; #p<0.05 vs vehicle.
In contrast, robust interactions between genotype and dose were observed for morphine (Figure 1c; F3,57=8.0, p<0.001) and hydrocodone (Figure 1c; F3,52=6.3, p=0.001). Although cocaine and U69,593 showed expected dose-dependent reward-potentiating and -devaluing effects, respectively, all MOR agonists had biphasic effects on θ0 in h/mOPRM1-118AA mice. In contrast, h/mOPRM1-118GG mice were relatively insensitive to morphine and hydrocodone and showed no significant differences from vehicle across the dose range tested (1.0–10.0 mg/kg i.p.). 118GG mice were also less sensitive to effects of morphine on MAX (Supplementary Table S1; F3,57=7.56, p<0.001), and a main effect of genotype was observed for hydrocodone (F3,52=9.84, p=0.005). These findings indicate that while h/mOPRM1-118GG mice are relatively insensitive to the effects of morphine and hydrocodone on brain reward function, they display no deficits in the rewarding effects of other MOR agonists or cocaine or the anhedonic effects of KOR agonists. For full statistical analyses of ICSS experiments, see Supplementary Table S2.
Mesolimbic Responses to Morphine are Reduced in h/mOPRM1-118GG Mice
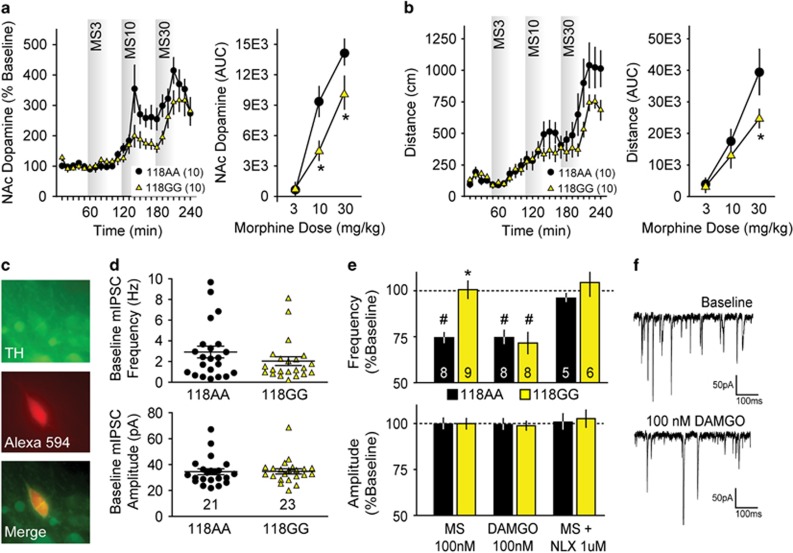
Because ICSS and other motivated behaviors are strongly influenced by dopaminergic neurotransmission, we measured morphine- and fentanyl-stimulated dopamine release in the NAc using in vivo microdialysis in freely moving h/mOPRM1-118AA and 118GG mice. Morphine (Figure 2a; AUC analysis; F2,32=81.1, p<0.001) and fentanyl (Supplementary Figure S1; F3,56=5.97, p=0.002) dose-dependently increased NAc dopamine relative to baseline, and a dose × genotype interaction was observed for morphine (Figure 2a; F2,32=4.57, p=0.02) but not fentanyl (Supplementary Figure S1; F3,42=0.24, p=0.87). Simultaneous measurement of locomotor activity showed that morphine (Figure 2b; F2,36=70.7, p<0.001) and fentanyl (Supplementary Figure S1; F3,42=35.5, p<0.001) dose-dependently increased locomotor activity, and a dose × genotype interaction was observed for morphine (Figure 2b; F2,36=4.50, p=0.02) but not fentanyl (Supplementary Figure S1; F3,42=0.06, p=0.98). These findings indicate that h/mOPRM1-118GG mice exhibit hyposensitivity of dopaminergic responses to morphine.
Figure 2.
Mesolimbic responses to opioids in h/mOPRM1-118AA and 118GG mice. (a and b) Effects of morphine (3–30 mg/kg i.p.) on dopamine release in the nucleus accumbens and locomotor activity in 118AA (n=10) and 118GG (n=10) mice, as determined by area under the curve (AUC) analysis. (c) Alexa Fluor 594 dye-filled neuron stained for tyrosine hydroxylase (TH). (d) Baseline frequency (Hz) and amplitude (pA) of miniature inhibitory postsynaptic currents (mIPSCs) in OPRM1-118AA (black circles; n=21) and 118GG (yellow triangles; n=23) mouse dopaminergic neurons in the ventral tegmental area (VTA). (e) Effects of bath-applied DAMGO (100 nM), morphine (100 nM), and morphine (100 nM) following naloxone (NLX) preapplication (1 μM) on normalized mIPSC frequency and amplitude in 118AA (black bars; n=8, 8, and 5, respectively) and 118GG (yellow bars; n=9, 8, and 6, respectively) dopaminergic neurons. (f) Representative traces showing mIPSCs recorded in the presence of 500 nM tetrodotoxin before and after DAMGO (100 nM) application in 118AA VTA slices. Values represent mean±SEM. *p<0.05 vs h/mOPRM1-118AA; #p<0.05 vs vehicle or baseline.
Opioids disinhibit dopaminergic neurons in the mesencephalic VTA by attenuating GABAergic neurotransmission at presynaptic sites (Johnson and North, 1992). We performed whole-cell patch clamp recordings from dopaminergic neurons in acute midbrain slices to measure the effects of morphine on the frequency and amplitude of mIPSCs compared with effects of the highly selective MOR agonist DAMGO. In VTA dopaminergic neurons (confirmed by post-hoc staining for TH; Figure 2c), no differences were observed in baseline mIPSC frequency (Figure 2d; t42=1.18, p=0.25) or amplitude (Figure 2d; t42=0.10, p=0.92) between h/mOPRM1-118AA and 118GG cells. mIPSCs were completely abolished by bath application of the GABAA receptor antagonist gabazine (SR-95531, data not shown).
Morphine (100 nM) reduced mIPSC frequency in h/mOPRM1-118AA mice (Figure 2e; t7=9.29, p<0.001) but not in 118GG mice, and a significant difference between genotypes was observed (t15=4.59, p<0.001). Morphine did not affect mIPSC amplitudes in either 118AA or 118GG mice (Figure 2e), suggesting that MORs presynaptically inhibit GABAergic transmission. The effects of morphine were blocked by preapplication with the opioid antagonist naloxone (1.0 μM) in both 118AA and 118GG mice (Figure 2e). In contrast to morphine, DAMGO (100 nM) robustly decreased mIPSC frequency in both h/mOPRM1-118AA (Figure 2e and f; t7=6.50, p<0.001) and 118GG (t7=4.91, p=0.002) mice but also had no effect on mIPSC amplitude in cells of either genotype (Figure 2e). These findings indicate that the presynaptic effects of morphine but not DAMGO are blunted in the VTA of 118GG mice, which may reduce disinhibition of dopaminergic neurons by acute morphine exposure. For full results and statistical analysis of electrophysiological experiments, see Supplementary Table S3.
h/mOPRM1-118A and 118G Receptors do not Display Differences in Giα-Coupling
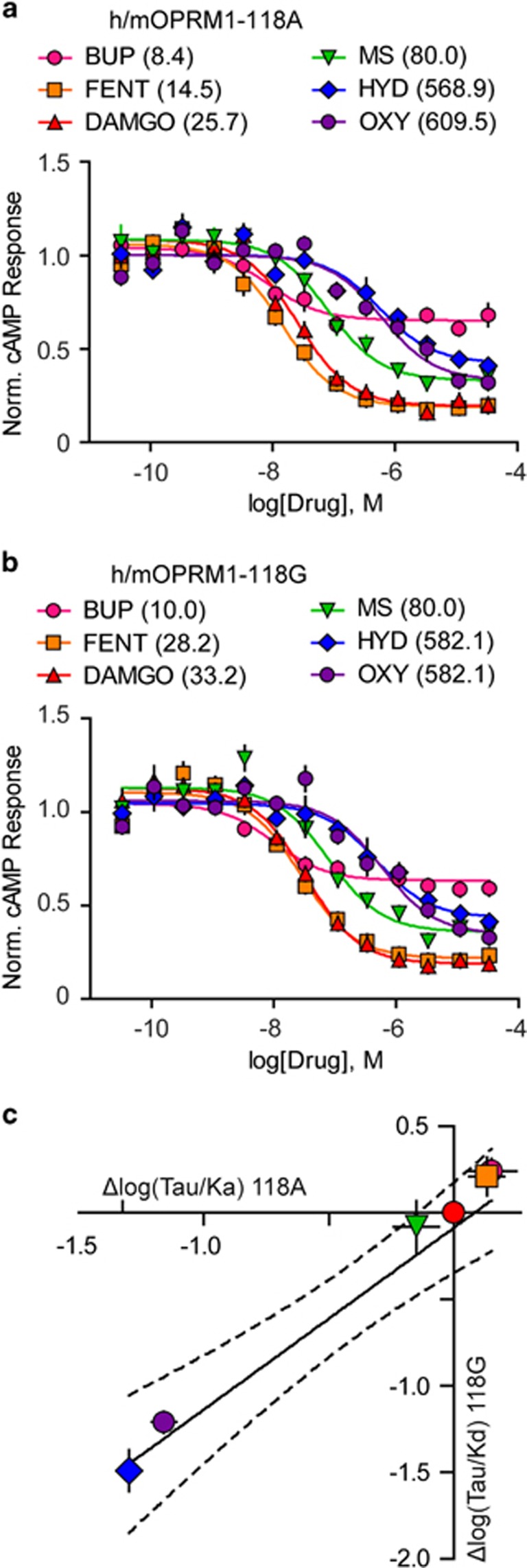
In order to determine why the rewarding potency of morphine and hydrocodone but not oxycodone, fentanyl, or buprenorphine was decreased in h/mOPRM1-118GG mice, we used several pharmacological assays to probe MOR function in vitro. First, we measured Giα-coupling in transfected HEK cells and found no difference in the potency (EC50) or efficacy of DAMGO, fentanyl, morphine, oxycodone, hydrocodone, and buprenorphine to suppress isoproterenol-stimulated cAMP production between h/mOPRM1-118A and 118G receptors (Figure 3a and b). We further examined MOR signal transduction using an application of the Black–Leff operational model (Black et al, 1985) that allowed us to minimize the effects of receptor density on relative agonist activity by comparing relative receptor function given different amounts of receptor reserve (0.3–10 ng transfected DNA/well) (Kenakin et al, 2012). No genotype difference in the normalized transduction coefficient [Δlog(T/KA)] was observed for any agonist tested (Supplementary Table S4). Across agonists, there was a direct, linear relationship between mean Δlog(T/KA) at h/mOPRM1-118A and 118G receptors (Figure 3c; R2=0.94, p=0.001), which suggests that h/mOPRM1-118A and 118G receptor isoforms are equivalent in their ability to couple Giα for these ligands. For full results of cAMP assays and statistical analyses, see Supplementary Table S4.
Figure 3.
Effect of MOR agonists on Giα-coupling in vitro. (a and b) Effects of DAMGO, fentanyl (FENT), buprenorphine (BUP), morphine (MS), oxycodone (OXY), and hydrocodone (HYD) on Giα-coupling in HEK293 cells expressing either h/mOPRM1-118A (a) or 118G (b) receptors (EC50, nM, in parentheses) measured by inhibition of isoproterenol-stimulated cAMP synthesis. (c) Linear regression of transduction coefficients (log(T/KA)) for each agonist normalized to DAMGO (Δlog(T/KA); n=4, 0.3–10.0 ng transfected DNA/experiment). Dashed line indicates 95% confidence interval of regression line. Values represent mean±SEM.
The h/mOPRM1 A118G Polymorphism does not Alter Agonist-Binding Affinity but is Associated with Decreased Tissue Receptor Content
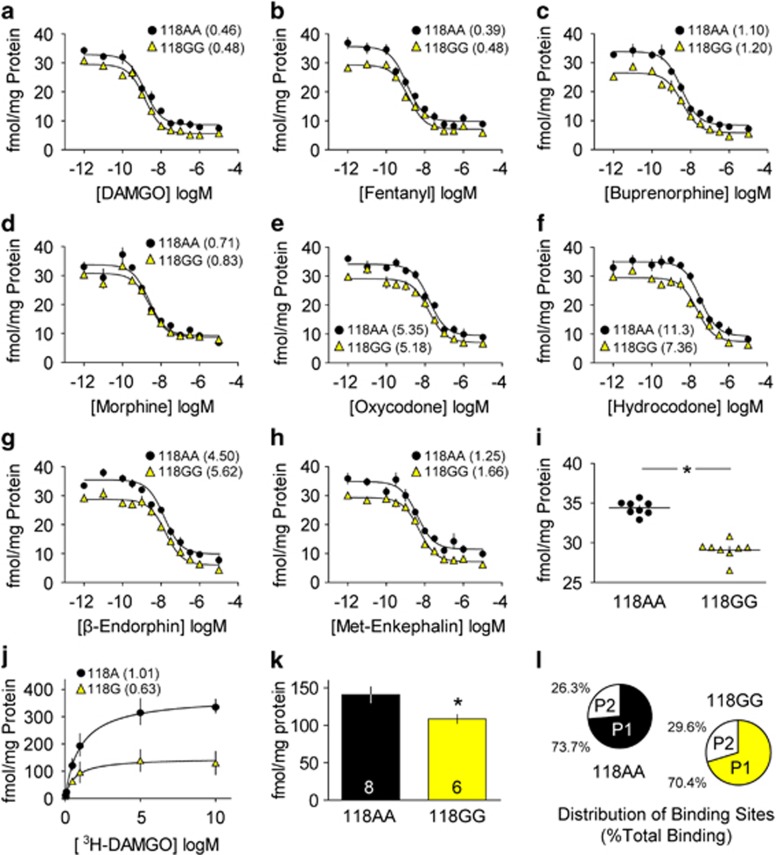
Because it has been reported that the OPRM1 A118G polymorphism increases the potency of the endogenous MOR agonist β-endorphin by increasing ligand-binding affinity (Bond et al, 1998), we performed competition binding assays to determine the affinity of each opioid agonist, as well as endogenous opioid peptides (β-endorphin and MET-enkephalin), using h/mOPRM1-118AA and 118GG mouse brainstem membranes. All drugs tested had nanomolar affinity (Ki) for h/mOPRM1-118A and 118G receptors, and no genotypic differences were observed (Figure 4a–h). A significant reduction in BMAX was observed in 118GG mouse membranes (Figure 4i; t14=10.07; p<0.005). This finding was replicated using CHO-K1 cells stably expressing h/mOPRM1-118A or 118G receptors (Figure 4j): BMAX was 60.5% lower in cells expressing the h/mOPRM1-118G receptor (118A: 363.4±16.3 fmol/mg protein; 118GG: 132.5±10.3 fmol/mg protein). For full results of competition binding assays and statistical analyses, see Supplementary Table S5.
Figure 4.
Radioligand binding using h/mOPRM1-118AA and 118GG mouse brainstem membranes. (a–h) The binding affinity (Ki, nM, in parentheses) of DAMGO (a), fentanyl (b), buprenorphine (c), morphine (d), oxycodone (e), hydrocodone (f), β-endorphin (g), and MET-enkephalin (h) determined by competition binding with [3H]-DAMGO in washed brainstem membranes from h/mOPRM1-118AA (black circles) and 118GG mice (yellow triangles). (i) BMAX (fmol/mg protein) determined by competition binding in panels a–h. (j) Relative [3H]-DAMGO binding in CHO-K1 cells stably expressing OPRM1-118A or 118G receptors (Kd, nM, in parentheses). (k) BMAX (fmol/mg protein) in individual h/mOPRM1-118AA (black bars; n=8) and 118GG mice (yellow bars; n=6) determined by saturation binding with [3H]-DAMGO. (l) Relative distribution (percentage of total specific binding) of MOR-binding sites in brainstem subcellular fractions (P1=nuclear/cytosolic fraction; P2=surface fraction). Values represent mean±SEM. *p<0.05 vs h/mOPRM1-118AA.
In order to address the limitations of interpreting differences in the relative maximum number of binding sites (BMAX) in pooled samples, we conducted single concentration [3H]-DAMGO saturation binding in subcellular membrane fractions prepared from individual h/mOPRM1-118AA and 118GG mice. Across fractions, specific binding (fmol/mg protein) was significantly greater in 118AA mice compared with 118GG littermates (Figure 4k; 118AA: 140.9±10.7 fmol/mg protein; 118GG: 108.5±5.7 fmol/mg protein; t12=2.42; p=0.03), and no significant difference in the percentage of total receptors localized to either the nuclear/cytosolic (P1) or surface (P2) fraction was observed between genotypes (Figure 4l; F1,12=0.98, p=0.34). These data suggest that the total number of MOR-binding sites is lower in h/mOPRM1-118GG mice, and because the relative distribution of receptors in subcellular fractions was not altered, it is unlikely that the h/mOPRM1 A118G polymorphism significantly alters receptor trafficking. For full results and statistical analysis of saturation-binding assays, see Supplementary Table S6.
Reduction of Receptor Reserve with the Irreversible MOR Antagonist β-Funaltrexamine Unmasks Genotypic Differences in Fentanyl Sensitivity
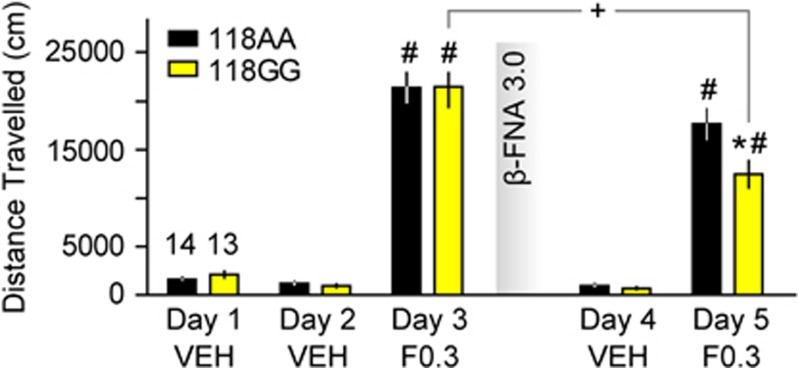
Because h/mOPRM1-118GG mice are less sensitive to MOR agonists with low in vivo potency (eg, hydrocodone and morphine) and display decreased numbers of MOR-binding sites, we hypothesized that reduction of receptor reserve with an irreversible antagonist (β-funaltrexamine or β-FNA) would unmask genotypic differences in drug response to fentanyl. Using a locomotor activity assay, we found that 0.3 mg/kg fentanyl (s.c.) significantly increased locomotor activity in both h/mOPRM1-118AA (Figure 5; t13=11.2, p<0.001) and 118GG mice (Figure 5; t12=10.1, p<0.001) compared with saline. There was no difference between genotypes in response to either saline (F1,25=0.92, p=0.35) or fentanyl (F1,25=0.001, p=0.98), confirming our earlier observations. Forty-eight hours after β-FNA exposure, a robust reduction in the locomotor-stimulant effects of fentanyl was observed in h/mOPRM1-118GG (Figure 5; t12=3.4, p=0.006) but not in h/mOPRM1-118AA mice (t13=1.9, p=0.08), and a significant genotype difference was observed (F1,25=6.1, p=0.02). This effect was specific to fentanyl, as no difference in response to saline was observed following β-FNA exposure (F1,25=1.3, p=0.27). These findings demonstrate that pharmacological reduction of receptor number is sufficient to unmask genotypic differences in drug response, supporting the assertion that the A118G polymorphism alters opioid sensitivity primarily by altering receptor reserve.
Figure 5.
Effects of receptor depletion with β-FNA on the locomotor response to fentanyl. Effects of fentanyl (F0.3; 0.3 mg/kg s.c.) and saline on locomotor activity in h/mOPRM1-118AA (black bars; n=14) and 118GG (yellow bars; n=13) mice before and after administration of β-funaltrexamine (β-FNA; 3.0 mg/kg s.c.). Values represent mean±SEM. *p<0.05 vs h/mOPRM1-118AA; #p<0.05 vs vehicle; +p<0.05 vs previous drug challenge.
DISCUSSION
Using a humanized transgenic mouse model, we investigated whether the OPRM1 A118G polymorphism alters opioid reward and its neural substrates. We found that h/mOPRM1-118GG mice were less sensitive to the reward-potentiating effects of morphine and hydrocodone but not to other opioids tested with higher in vivo potency. Similarly, h/mOPRM1-118GG mice were less sensitive to the locomotor-stimulating effects of morphine but not the highly potent MOR agonist, fentanyl. Attenuated morphine reward and locomotion in h/mOPRM1-118GG mice was associated with decreased morphine- but not fentanyl-induced dopamine release in the NAc in vivo and a reduction in the inhibitory effects of morphine but not the highly potent MOR agonist DAMGO on GABAergic neurotransmission in the VTA ex vivo. These findings were not due to changes in drug potency or efficacy in vitro or changes in ligand-binding affinity. A significant reduction in total MOR-binding sites was observed in 118GG mouse brainstem membranes, which, however, did not affect the relative distribution of receptors in different subcellular fractions. Administration of the irreversible MOR antagonist β-FNA unmasked genotypic differences in the locomotor response to fentanyl, supporting the hypothesis that the OPRM1 A118G polymorphism decreases sensitivity to agonists with low in vivo potency by reducing receptor reserve.
First, we found that h/mOPRM1-118GG mice are less sensitive than 118AA mice to the reward-potentiating effects of morphine but not those of synthetic MOR agonists with higher in vivo potency measured with ICSS. Stimulation of the medial forebrain bundle during ICSS evokes dopamine transients in the NAc, and BSR is attenuated by D1R antagonists (Cheer et al, 2007). Similar to other rewarding drugs, morphine decreases BSR threshold in mice (Elmer et al, 2010; Robinson et al, 2012) in a dopamine-dependent manner (Kornetsky and Duvauchelle, 1994). We also observed reduced effects of morphine but not the highly potent MOR agonist fentanyl on dopamine release in the NAc of h/mOPRM1-118GG mice and reduced inhibition of GABAergic neurotransmission onto dopaminergic VTA neurons by morphine but not the highly potent MOR agonist DAMGO in h/mOPRM1-118GG cells. As presynaptic MOR-mediated control of GABA release at synapses with dopaminergic neurons in the VTA is a critical mechanism regulating opioid reward in mice (Madhavan et al, 2010), reduced MOR function in the VTA likely diminished morphine reward through a dopaminergic mechanism. Given the consistency of diminished locomotor and conditioned rewarding responses to morphine observed both in our mice and in another murine model of the OPRM1 A118G polymorphism (Mague et al, 2009), it is likely that aberrant mesolimbic responses to morphine exist in human OPRM1-118G carriers as well.
We observed that h/mOPRM1-118GG mice are less sensitive to the reward-potentiating effects of hydrocodone, without loss of sensitivity to oxycodone, fentanyl, or buprenorphine. These observations do not appear to be due to direct pharmacodynamic effects per se, as relative in vivo potency rather than in vitro agonist potency or efficacy predicted opioid sensitivity in 118GG mice. As in vivo potency is dependent on both pharmacokinetics and pharmacodynamics, both of these factors must be considered when interpreting behavior associated with the OPRM1 A118G variant. Fentanyl was both highly potent and efficacious in vitro and is approximately 100 times more potent than morphine in humans due to its high lipophilicity and brain penetrance (Eguchi, 2004). Buprenorphine is a weak partial agonist but is 25–50 times more potent than morphine in vivo as a result of its high MOR-binding affinity and lipophilicity. Oxycodone, which is structurally similar to morphine but less potent in vitro, was 30 times more potent than morphine on ICSS, most likely due to superior brain penetrance (Bostrom et al, 2008) and the substantial first pass metabolism of morphine in mice. Hydrocodone, which is equipotent to morphine in humans (Eguchi, 2004), had both low potency and intrinsic efficacy in vitro. Given these observations, it appears that low in vitro potency, bioavailability, and brain penetrance predicts diminished rewarding potency of opioids in h/mOPRM1-118GG mice.
Although low in vivo potency appears to predict decreased opioid sensitivity in h/mOPRM1-118GG mice, we found no evidence that loss of a putative N-terminal glycosylation site affects Giα-coupling, supporting several previous studies (Befort et al, 2001; Beyer et al, 2004; Knapman et al, 2014). This is not surprising given that DAMGO and morphine retain potency and efficacy at the MOR in the absence of the extracellular N-terminus (Surratt et al, 1994). The intracellular C-terminus affects signal transduction, desensitization, and receptor internalization (Koch et al, 2001). Previous investigations using OPRM1-118A and 118G receptors did not find differences in receptor internalization following DAMGO or morphine incubation (Beyer et al, 2004), although direct arrestin recruitment, which is associated with rapid desensitization, internalization, and G protein-independent signaling (Arttamangkul et al, 2006; Dang et al, 2011; Dang and Christie, 2012), has not been measured to our knowledge. Its involvement in OPRM1 A118G phenotypes warrants further investigation, and the inability to measure β-arrestin recruitment is a limitation of our study.
Several in vitro, animal, and human studies suggest that MOR expression is decreased by the OPRM1 A118G polymorphism (Beyer et al, 2004; Kroslak et al, 2007; Mague et al, 2009; Pecina et al, 2015; Ray et al, 2011; Wang et al, 2012; Weerts et al, 2013). These changes may be due to differences in mRNA stability (Zhang et al, 2005), decreased receptor half-life (Huang et al, 2012), and/or receptor trafficking (He et al, 2002). Mice containing a homologous polymorphism at Oprm1 position 112 (A112G) display decreased receptor mRNA in the hypothalamus, PAG, VTA, NAc, and cortex, which was associated with lower MOR protein and specific binding (Mague et al, 2009). Follow-up studies indicate that Oprm1-112G receptors in striatal membranes have lower molecular mass when compared with 112A littermates due to decreased N-terminal glycosylation (Huang et al, 2012). Loss of glycosylation impairs trafficking of several GPCRs to the cell surface, including the KOR (Li et al, 2007) and β2-adrenergic receptor (Rands et al, 1990), but we did not observe any changes in the relative subcellular distribution of MOR-binding sites in h/mOPRM1-118GG mouse brainstem despite a lower total number of binding sites. It has recently been shown that the reinforcing and mesolimbic effects of heroin are enhanced in 112GG mice (Zhang et al, 2014). Given that heroin sensitivity is not dependent on Oprm1 exon 1 (Schuller et al, 1999), it is possible that the A118G polymorphism alters the composition of alternatively spliced receptor populations. Future studies will be required to elucidate molecular mechanisms by which the A118G polymorphism alters receptor availability and determine their functional effects within circuits associated with opioid abuse.
Naive h/mOPRM1-118AA and 118GG mice are equally sensitive to fentanyl, supporting previous findings using this model (Mahmoud et al, 2011). As a highly potent, efficacious, and brain-penetrant agonist, fentanyl has a large receptor reserve in vivo that appears to override differences in tissue receptor content. Total MOR binding is lower in h/mOPRM1-118GG mice, which are more affected by reductions in receptor number following treatment with β-FNA, consistent with fewer ‘spare receptors,' ie, a lower MOR reserve. Receptor depletion reduced the in vivo potency of fentanyl on locomotor stimulation in 118GG mice without significantly affecting 118AA mice. This finding supports the hypotheses that (1) the OPRM1 A118G polymorphism influences opioid sensitivity by altering receptor reserve, and (2) the effect of genotype can be modulated by changes in receptor number. Fentanyl, oxycodone, and buprenorphine differ significantly in their structure and intrinsic efficacy but are highly brain penetrant and have large receptor reserves. Morphine and hydrocodone are less potent and brain penetrant than fentanyl and buprenorphine and have lower receptor reserves. Here we showed different effects of a relatively low-potency (morphine) MOR agonist compared with high-potency (fentanyl, DAMGO) MOR agonists on behavior (ICSS, locomotion) and circuit function (in vivo microdialysis, ex vivo electrophysiology) in h/mOPRM1-118GG and 118AA mice but no difference in the function of 118G and 118A MOR isoforms in vitro. Furthermore, by reducing receptor reserve with an irreversible MOR antagonist we were able to demonstrate a lower MOR reserve in 118GG mice measured with fentanyl-stimulated locomotion. We conclude that agonist-specific differences in opioid sensitivity in 118GG mice are determined by the number of receptor-binding sites and do not involve altered receptor function.
These investigations represent a reverse translational approach to investigating how the OPRM1 A118G polymorphism alters mesolimbic responses to opioids, which may elucidate mechanisms influencing both opioid and alcohol abuse in human populations. Meta-analyses suggest that carrying the OPRM1-118G allele increases postsurgical opioid analgesic requirement (Hwang et al, 2014), although association with opioid dependence is not robust outside of Asian populations (Haerian and Haerian, 2013). It is possible that, given our findings, decreased drug reward is protective against opioid abuse in some individuals, while resistance to negative drug effects promotes intake in others. The OPRM1-118G allele is also associated with greater subjective alcohol reward and improved therapeutic response to naltrexone (Heilig et al, 2011; Ray et al, 2012). We have recently shown that h/mOPRM1-118GG mice are more sensitive to the reward-potentiating effect of alcohol on ICSS and opioid antagonism of alcohol drinking than their 118AA littermates (Bilbao et al, 2014). These findings emphasize the potential of this and other humanized mouse models to parse genetic moderators of disease risk, predict medication efficacy in patient populations, and ultimately inform clinical practice.
FUNDING AND DISCLOSURE
This work was supported by grants from the National Institute on Alcohol Abuse and Alcoholism (F30 AA021312 to JER; R01 AA018335 to CJM), the National Institute on Drug Abuse (R01 DA017204 to BLR), and the Swedish Research Council (Grant no. 2010-3219 to MH); and by the intramural programs of the National Institute on Drug Abuse and National Institute on Alcohol Abuse and Alcoholism. All the work reported here was supported by NIH. Dr Roth has received unrestricted grant support from Dai Nippon Sumitomo, Merck, and Asubio Pharmaceuticals. Dr Roth has consulted for Merck, Novartis, and Pfizer over the past 12 months. The authors declare no conflict of interest.
Footnotes
Supplementary Information accompanies the paper on the Neuropsychopharmacology website (http://www.nature.com/npp)
Supplementary Material
References
- Arttamangkul S, Torrecilla M, Kobayashi K, Okano H, Williams JT. Separation of mu-opioid receptor desensitization and internalization: endogenous receptors in primary neuronal cultures. J Neurosci. 2006;26:4118–4125. doi: 10.1523/JNEUROSCI.0303-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Befort K, Filliol D, Decaillot FM, Gaveriaux-Ruff C, Hoehe MR, Kieffer BL. A single nucleotide polymorphic mutation in the human mu-opioid receptor severely impairs receptor signaling. J Biol Chem. 2001;276:3130–3137. doi: 10.1074/jbc.M006352200. [DOI] [PubMed] [Google Scholar]
- Beyer A, Koch T, Schroder H, Schulz S, Hollt V. Effect of the A118G polymorphism on binding affinity, potency and agonist-mediated endocytosis, desensitization, and resensitization of the human mu-opioid receptor. J Neurochem. 2004;89:553–560. doi: 10.1111/j.1471-4159.2004.02340.x. [DOI] [PubMed] [Google Scholar]
- Bilbao A, Robinson JE, Heilig M, Malanga CJ, Spanagel R, Sommer WH, et al. 2014A pharmacogenetic determinant of mu-opioid receptor antagonist effects on alcohol reward and consumption: evidence from humanized mice Biol Psychiatrypii: S0006-3223(14)00653-2. doi: 10.1016/j.biopsych.2014.08.021(epub ahead of print). [DOI] [PubMed]
- Black JW, Leff P, Shankley NP, Wood J. An operational model of pharmacological agonism: the effect of E/[A] curve shape on agonist dissociation constant estimation. Br J Pharmacol. 1985;84:561–571. doi: 10.1111/j.1476-5381.1985.tb12941.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bond C, LaForge KS, Tian M, Melia D, Zhang S, Borg L, et al. Single-nucleotide polymorphism in the human mu opioid receptor gene alters beta-endorphin binding and activity: possible implications for opiate addiction. Proc Natl Acad Sci USA. 1998;95:9608–9613. doi: 10.1073/pnas.95.16.9608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bostrom E, Hammarlund-Udenaes M, Simonsson US. Blood-brain barrier transport helps to explain discrepancies in in vivo potency between oxycodone and morphine. Anesthesiology. 2008;108:495–505. doi: 10.1097/ALN.0b013e318164cf9e. [DOI] [PubMed] [Google Scholar]
- Cheer JF, Aragona BJ, Heien ML, Seipel AT, Carelli RM, Wightman RM. Coordinated accumbal dopamine release and neural activity drive goal-directed behavior. Neuron. 2007;54:237–244. doi: 10.1016/j.neuron.2007.03.021. [DOI] [PubMed] [Google Scholar]
- Chefer VI, Backman CM, Gigante ED, Shippenberg TS. Kappa opioid receptors on dopaminergic neurons are necessary for kappa-mediated place aversion. Neuropsychopharmacology. 2013;38:2623–2631. doi: 10.1038/npp.2013.171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cicero TJ, Ellis MS, Surratt HL. Effect of abuse-deterrent formulation of OxyContin. N Engl J Med. 2012;367:187–189. doi: 10.1056/NEJMc1204141. [DOI] [PubMed] [Google Scholar]
- Dang VC, Chieng B, Azriel Y, Christie MJ. Cellular morphine tolerance produced by betaarrestin-2-dependent impairment of mu-opioid receptor resensitization. J Neurosci. 2011;31:7122–7130. doi: 10.1523/JNEUROSCI.5999-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dang VC, Christie MJ. Mechanisms of rapid opioid receptor desensitization, resensitization and tolerance in brain neurons. Br J Pharmacol. 2012;165:1704–1716. doi: 10.1111/j.1476-5381.2011.01482.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Di Chiara G, Imperato A. Drugs abused by humans preferentially increase synaptic dopamine concentrations in the mesolimbic system of freely moving rats. Proc Natl Acad Sci USA. 1988;85:5274–5278. doi: 10.1073/pnas.85.14.5274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eguchi M. Recent advances in selective opioid receptor agonists and antagonists. Med Res Rev. 2004;24:182–212. doi: 10.1002/med.10059. [DOI] [PubMed] [Google Scholar]
- Elmer GI, Pieper JO, Hamilton LR, Wise RA. Qualitative differences between C57BL/6J and DBA/2J mice in morphine potentiation of brain stimulation reward and intravenous self-administration. Psychopharmacology. 2010;208:309–321. doi: 10.1007/s00213-009-1732-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haerian BS, Haerian MS. OPRM1 rs1799971 polymorphism and opioid dependence: evidence from a meta-analysis. Pharmacogenomics. 2013;14:813–824. doi: 10.2217/pgs.13.57. [DOI] [PubMed] [Google Scholar]
- He J, Xu J, Castleberry AM, Lau AG, Hall RA. Glycosylation of beta(1)-adrenergic receptors regulates receptor surface expression and dimerization. Biochem Biophys Res Commun. 2002;297:565–572. doi: 10.1016/s0006-291x(02)02259-3. [DOI] [PubMed] [Google Scholar]
- Heilig M, Goldman D, Berrettini W, O'Brien CP. Pharmacogenetic approaches to the treatment of alcohol addiction. Nat Rev Neurosci. 2011;12:670–684. doi: 10.1038/nrn3110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang P, Chen C, Mague SD, Blendy JA, Liu-Chen LY. A common single nucleotide polymorphism A118G of the mu opioid receptor alters its N-glycosylation and protein stability. Biochem J. 2012;441:379–386. doi: 10.1042/BJ20111050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hwang IC, Park JY, Myung SK, Ahn HY, Fukuda K, Liao Q. OPRM1 A118G gene variant and postoperative opioid requirement: a systematic review and meta-analysis. Anesthesiology. 2014;121:825–834. doi: 10.1097/ALN.0000000000000405. [DOI] [PubMed] [Google Scholar]
- Johnson SW, North RA. Opioids excite dopamine neurons by hyperpolarization of local interneurons. J Neurosci. 1992;12:483–488. doi: 10.1523/JNEUROSCI.12-02-00483.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kenakin T, Watson C, Muniz-Medina V, Christopoulos A, Novick S. A simple method for quantifying functional selectivity and agonist bias. ACS Chem Neurosci. 2012;3:193–203. doi: 10.1021/cn200111m. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knapman A, Santiago M, Connor M. Buprenorphine signalling is compromised at the N40D polymorphism of the human mu opioid receptor in vitro. Br J Pharmacol. 2014;171:4273–4288. doi: 10.1111/bph.12785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koch T, Schulz S, Pfeiffer M, Klutzny M, Schroder H, Kahl E, et al. C-terminal splice variants of the mouse mu-opioid receptor differ in morphine-induced internalization and receptor resensitization. J Biol Chem. 2001;276:31408–31414. doi: 10.1074/jbc.M100305200. [DOI] [PubMed] [Google Scholar]
- Kornetsky C, Duvauchelle C. Dopamine, a common substrate for the rewarding effects of brain stimulation reward, cocaine, and morphine. NIDA Res Monogr. 1994;145:19–39. [PubMed] [Google Scholar]
- Kroslak T, Laforge KS, Gianotti RJ, Ho A, Nielsen DA, Kreek MJ. The single nucleotide polymorphism A118G alters functional properties of the human mu opioid receptor. J Neurochem. 2007;103:77–87. doi: 10.1111/j.1471-4159.2007.04738.x. [DOI] [PubMed] [Google Scholar]
- Kumar S, Suryanarayanan A, Boyd KN, Comerford CE, Lai MA, Ren Q, et al. Ethanol reduces GABAA alpha1 subunit receptor surface expression by a protein kinase Cgamma-dependent mechanism in cultured cerebral cortical neurons. Mol Pharmacol. 2010;77:793–803. doi: 10.1124/mol.109.063016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li JG, Chen C, Liu-Chen LY. N-Glycosylation of the human kappa opioid receptor enhances its stability but slows its trafficking along the biosynthesis pathway. Biochemistry. 2007;46:10960–10970. doi: 10.1021/bi700443j. [DOI] [PubMed] [Google Scholar]
- Madhavan A, Bonci A, Whistler JL. Opioid-Induced GABA potentiation after chronic morphine attenuates the rewarding effects of opioids in the ventral tegmental area. J Neurosci. 2010;30:14029–14035. doi: 10.1523/JNEUROSCI.3366-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mague SD, Isiegas C, Huang P, Liu-Chen LY, Lerman C, Blendy JA. Mouse model of OPRM1 (A118G) polymorphism has sex-specific effects on drug-mediated behavior. Proc Natl Acad Sci USA. 2009;106:10847–10852. doi: 10.1073/pnas.0901800106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mahmoud S, Thorsell A, Sommer WH, Heilig M, Holgate JK, Bartlett SE, et al. Pharmacological consequence of the A118G mu opioid receptor polymorphism on morphine- and fentanyl-mediated modulation of Ca(2)(+) channels in humanized mouse sensory neurons. Anesthesiology. 2011;115:1054–1062. doi: 10.1097/ALN.0b013e318231fc11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Paulozzi LJ, Jones CM, Mack KA, Rudd RA. Vital signs: overdoses of prescription opioid pain relievers—United States, 1999—2008. MMWR Morb Mortal Wkly Rep. 2011;60:1487–1492. [PubMed] [Google Scholar]
- Pecina M, Love T, Stohler CS, Goldman D, Zubieta JK. Effects of the mu opioid receptor polymorphism (OPRM1 A118G) on pain regulation, placebo effects and associated personality trait measures. Neuropsychopharmacology. 2015;40:957–965. doi: 10.1038/npp.2014.272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramchandani VA, Umhau J, Pavon FJ, Ruiz-Velasco V, Margas W, Sun H, et al. A genetic determinant of the striatal dopamine response to alcohol in men. Mol Psychiatry. 2010;16:809–817. doi: 10.1038/mp.2010.56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rands E, Candelore MR, Cheung AH, Hill WS, Strader CD, Dixon RA. Mutational analysis of beta-adrenergic receptor glycosylation. J Biol Chem. 1990;265:10759–10764. [PubMed] [Google Scholar]
- Ray LA, Barr CS, Blendy JA, Oslin D, Goldman D, Anton RF. The role of the Asn40Asp polymorphism of the mu opioid receptor gene (OPRM1) on alcoholism etiology and treatment: a critical review. Alcohol Clin Exp Res. 2012;36:385–394. doi: 10.1111/j.1530-0277.2011.01633.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ray R, Ruparel K, Newberg A, Wileyto EP, Loughead JW, Divgi C, et al. Human mu opioid receptor (OPRM1 A118G) polymorphism is associated with brain mu-opioid receptor binding potential in smokers. Proc Natl Acad Sci USA. 2011;108:9268–9273. doi: 10.1073/pnas.1018699108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson JE, Chen M, Stamatakis AM, Krouse MC, Howard EC, Faccidomo S, et al. Levetiracetam has opposite effects on alcohol- and cocaine-related behaviors in C57BL/6J mice. Neuropsychopharmacology. 2013;38:1322–1333. doi: 10.1038/npp.2013.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson JE, Fish EW, Krouse MC, Thorsell A, Heilig M, Malanga CJ. Potentiation of brain stimulation reward by morphine: effects of neurokinin-1 receptor antagonism. Psychopharmacology. 2012;220:215–224. doi: 10.1007/s00213-011-2469-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roth BL.2013. National Institute on Mental Health Psychoactive Drug Screening Program (PDSP) Assay Protocol Book Version IIPDSP Assay Protocol Book Version II National Institute on Mental Health Psychoactive Drug Screening Program (PDSP): Chapel Hill, NC, USA [Google Scholar]
- Schuller AG, King MA, Zhang J, Bolan E, Pan YX, Morgan DJ, et al. Retention of heroin and morphine-6 beta-glucuronide analgesia in a new line of mice lacking exon 1 of MOR-1. Nat Neurosci. 1999;2:151–156. doi: 10.1038/5706. [DOI] [PubMed] [Google Scholar]
- Schultz W, Dayan P, Montague PR. A neural substrate of prediction and reward. Science. 1997;275:1593–1599. doi: 10.1126/science.275.5306.1593. [DOI] [PubMed] [Google Scholar]
- Surratt CK, Johnson PS, Moriwaki A, Seidleck BK, Blaschak CJ, Wang JB, et al. mu opiate receptor. Charged transmembrane domain amino acids are critical for agonist recognition and intrinsic activity. J Biol Chem. 1994;269:20548–20553. [PubMed] [Google Scholar]
- Vardy E, Mosier PD, Frankowski KJ, Wu H, Katritch V, Westkaemper RB, et al. Chemotype-selective modes of action of kappa-opioid receptor agonists. J Biol Chem. 2013;288:34470–34483. doi: 10.1074/jbc.M113.515668. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang YJ, Huang P, Ung A, Blendy JA, Liu-Chen LY. Reduced expression of the mu opioid receptor in some, but not all, brain regions in mice with OPRM1 A112G. Neuroscience. 2012;205:178–184. doi: 10.1016/j.neuroscience.2011.12.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weerts EM, McCaul ME, Kuwabara H, Yang X, Xu X, Dannals RF, et al. Influence of OPRM1 Asn40Asp variant (A118G) on [11C]carfentanil binding potential: preliminary findings in human subjects. Int J Neuropsychopharmacol. 2013;16:47–53. doi: 10.1017/S146114571200017X. [DOI] [PubMed] [Google Scholar]
- Wise RA. Drug-activation of brain reward pathways. Drug Alcohol Depend. 1998;51:13–22. doi: 10.1016/s0376-8716(98)00063-5. [DOI] [PubMed] [Google Scholar]
- Zhang Y, Picetti R, Butelman ER, Ho A, Blendy JA, Kreek MJ. Mouse model of the OPRM1 (A118G) polymorphism: differential heroin self-administration behavior compared to wild type mice. Neuropsychopharmacology. 2014;40:1091–1100. doi: 10.1038/npp.2014.286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Y, Wang D, Johnson AD, Papp AC, Sadee W. Allelic expression imbalance of human mu opioid receptor (OPRM1) caused by variant A118G. J Biol Chem. 2005;280:32618–32624. doi: 10.1074/jbc.M504942200. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.