Abstract
Aims
It is common to advise that analgesics, and especially non-steroidal anti-inflammatory drugs (NSAIDs), be taken with food to reduce unwanted gastrointestinal adverse effects. The efficacy of single dose analgesics depends on producing high, early, plasma concentrations; food may interfere with this. This review sought evidence from single dose pharmacokinetic studies on the extent and timing of peak plasma concentrations of analgesic drugs in the fed and fasting states.
Methods
A systematic review of comparisons of oral analgesics in fed and fasting states published to October 2014 reporting kinetic parameters of bioavailability, time to maximum plasma concentration (tmax), and its extent (Cmax) was conducted. Delayed-release formulations were not included.
Results
Bioavailability was not different between fasted and fed states. Food typically delayed absorption for all drugs where the fasting tmax was less than 4 h. For the common analgesics (aspirin, diclofenac, ibuprofen, paracetamol) fed tmax was 1.30 to 2.80 times longer than fasted tmax. Cmax was typically reduced, with greater reduction seen with more rapid absorption (fed Cmax only 44–85% of the fasted Cmax for aspirin, diclofenac, ibuprofen and paracetamol).
Conclusion
There is evidence that high, early plasma concentrations produces better early pain relief, better overall pain relief, longer lasting pain relief and lower rates of remedication. Taking analgesics with food may make them less effective, resulting in greater population exposure. It may be time to rethink research priorities and advice to professionals, patients and the public.
Keywords: analgesic, bioavailability, food, maximum plasma concentration, oral, pharmacokinetic
Introduction
There is a consensus in worldwide recommendations that prescription and non-prescription (over-the-counter, OTC) non-steroidal anti-inflammatory drugs (NSAIDs) should be taken with food or with milk, and that this advice applies also to aspirin, and perhaps to paracetamol. For example, the NICE Clinical Knowledge Summary on NSAIDs advises ‘Avoiding taking the NSAID on an empty stomach’ and to ‘Take aspirin after food (to reduce the risk of gastrointestinal adverse effects)’, though it does not give this advice for paracetamol [1]. The UK National Health Service NHS Choices website advises that ‘There are no known interactions between NSAIDs and food. Ideally, take NSAIDs after eating and avoid taking them on an empty stomach. This will help minimize the risk of the medicine upsetting your stomach’ [2]. German guidelines for family physicians advise that in elderly patients NSAIDs should be taken with food and never on an empty stomach [3]. A consumer's guide on arthritis medications endorsed by the Canadian Rheumatology Association suggests that ‘Taking your NSAIDs with food may help to reduce stomach upset’ [4]. A number of Australian and New Zealand sources echo this general advice, and it appears to be almost universal advice, whatever the circumstance.
The theoretical rationale for taking analgesics with food is clearly to reduce or prevent adverse events. It is unclear whether this means common but mild and reversible adverse events like dyspepsia, or rare but serious adverse events like upper or lower gastrointestinal bleeding. The evidence that taking oral analgesics with food actually accomplishes this desired effect is non-existent. In single dose studies, only aspirin of the commonly used non-opioid analgesics is associated with any increased adverse event reporting over placebo (number needed to harm 44, 95% CI 23, 345) [5]. Rainsford & Bjarnason found no evidence of benefit from taking NSAIDs with food [6]. They suggested that it would be more appropriate to take OTC NSAIDs on an empty stomach because rapid onset of action is beneficial.
More rapid absorption of ibuprofen produces earlier and higher maximum plasma concentrations, producing earlier onset of analgesia combined with better overall and longer lasting analgesia in dental pain models [7–9]. The effect of fast acting formulations was pronounced, with 200 mg of a fast acting formulation producing the same or better analgesia as 400 mg ibuprofen acid, and with a reduced requirement for additional analgesic use [8].
An understanding of the effects of food on the pharmacokinetics of analgesics has therefore become a priority, particularly its effect on occasional use of OTC drugs. We have therefore conducted a systematic review of studies reporting kinetic results of oral analgesics in the fasted and fed states in humans. We considered any formulation where the drug was immediately available for absorption following ingestion, but excluded those specifically formulated for delayed or controlled release.
Methods
There is no published protocol for this review. Two authors independently carried out searches for potentially relevant studies, assessed studies for inclusion and extracted data using a piloted spreadsheet. Any disagreements were settled by discussion with a third author.
We used a number of methods to search for comparisons of oral analgesics in the fed and fasted states to 31 October 2014. We searched PubMed using a series of free text terms: ‘fed AND fasted AND analgesic’, and ‘fed AND fasted AND drug name’, using ‘food’, ‘meal’ or ‘milk’ as alternatives to fed. We also examined reviews for aspirin, paracetamol, and individual NSAIDs where they mentioned pharmacokinetic results, and the reference lists of any retrieved studies. For each identified study we used the ‘related citations’ function of PubMed to identify reports citing the trial in case the citing reference was also a study reporting on oral analgesics used in the fed and fasted state. Finally, we entered each included trial into Google Scholar and used the citations function to identify reports that cited the trials. Methods like this have been used with good effect to identify non-randomized studies where electronic searching is insensitive [10,11]. We also searched http://Clinicaltrials.gov to identify completed or ongoing studies.
To be included, studies were required to have data on kinetic parameters in the fasted and fed state for single doses of aspirin, dipyrone, paracetamol, or any NSAID, or for a principal metabolite in blood following oral administration in humans. There was no restriction by date or language of publication. We accepted any definition of fed, meal or milk. We accepted any formulation other than delayed release formulations. Fast-acting formulations of ibuprofen were analyzed separately from ibuprofen acid, and diclofenac potassium separately from diclofenac sodium.
From each study we noted the study design (whether it was a randomized, crossover design, for example), the number and demographics of the individuals involved in the testing, the duration of fasting, the components of the meals used, the time of drug administration in relation to the meal, and the dose and formulation of the analgesic drug. The kinetic parameters noted were bioavailability (using the longest duration provided), the maximum plasma concentration (Cmax) and the time at which Cmax occurred (tmax).
No statistical analyses were planned. Drug concentrations were converted to mg l–1 and area under the plasma concentration–time curve (AUC) to mg l–1 h. We calculated a mean tmax for individual drugs weighted by the number of participants in each study. For Cmax we calculated a dose-related mean, weighted by the number of participants in each study, with units of mg l–1 mg–1 dose. All calculations were carried out by one author and checked by another.
We wanted to compare bioavailability (AUC), tmax and Cmax in the fed and fasted state.
Results
Studies identified
Searches identified 63 publications which were obtained and read. Twenty-five were excluded because they collected urine samples only, were reviews without relevant data, investigated delayed release preparations, had no fasting or fed results, or were duplicate publications (Figure1). Six potentially relevant studies identified in http://Clinicaltrials.gov did not provide any results and could not be used (Supplementary Table 1). We included 38 publications [12–49] with 656 unique individuals, reporting 46 fed/fasted comparisons involving 874 participants in the comparisons. Papers were published between 1972 and 2012. Because of the crossover nature of many of the studies, the number in the comparisons is more than the number of individuals. Included studies measured the pharmacokinetics of aspirin, dipyrone, paracetamol and 16 different NSAIDs. Fast acting formulations were represented by diclofenac potassium (four studies, the only diclofenac formulation with data), ibuprofen lysine (two studies), naproxen sodium (one study) and paracetamol with bicarbonate (one study).
Figure 1.
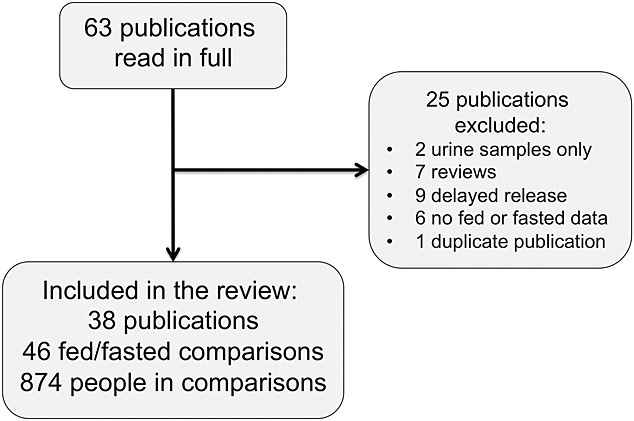
Flow chart of search results
Details of the comparisons are given in Supplementary Table 2. Of the 46 comparisons, 33 had a randomized crossover design, 12 had a crossover design but did not mention randomization and one was opportunistic. Study participants were predominantly healthy volunteers generally aged 18–50 years (20 comparisons were male only, 22 were mixed male and female), three studies were in adult patients (post-operative, inpatients, sciatica), and one was in children with cancer. The duration of fasting was typically overnight, usually with a minimum of 10 h before ingesting the test medication. Fasting patients were sometimes given a light meal about 4 h after ingestion. Analgesics were taken with these meals or a few minutes after the meal.
The types of meal were variously described (Supplementary Table 2). Twenty-eight comparisons gave details of the meals for volunteers in the fed part of the studies. These typically consisted of tea or coffee, bread or toast with jam, and often with cheese, eggs or bacon. Other meals were described only as standardized, without details (12), as high fat (4), or had some other description (2).
Drug analysis was for the parent compound except for:
aspirin, for which salicylic acid concentrations were also reported, and used;
dipyrone, where methylaminoantipyrine, the first metabolite of dipyrone which is enzymatically hydrolyzed in the intestine was measured;
nabumetone, where 6-methoxy-2-naphthlacetic acid was measured.
Effects of food on bioavailability
There was no difference in bioavailability between fasting and fed states as measured by the area under the concentration–time curve (Figure2).
Figure 2.
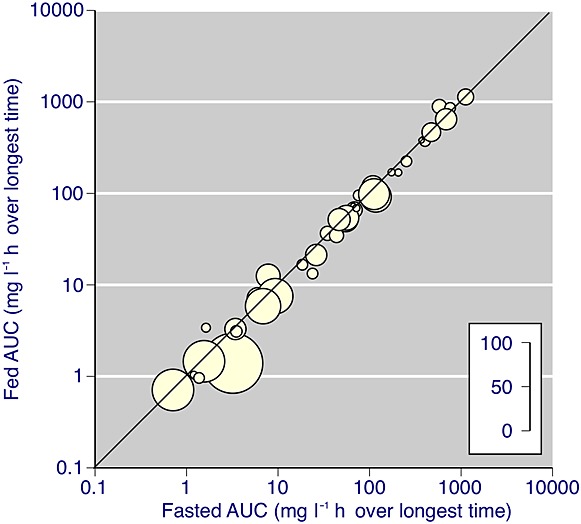
AUC for fasting and fed states assessed over longest time reported (all drugs). A logarithmic scale is used because of the wide range of AUC values. The inset scale indicates the number of participants in each comparison
Effects of food on tmax
Food typically delayed absorption for all drugs with a fasting tmax of under 4 h (Figure3, Table 1). The degree of delay varied between drugs, from little or no delay in absorption for celecoxib, diflunisal, dipyrone, meloxicam and timegadine, to an increase of fed tmax over fasted tmax of 250% or more for bromfenac, diclofenac, etoricoxib, ketoprofen and tenoxicam. Ibuprofen, paracetamol and naproxen were intermediate between these extremes (Table 1).
Figure 3.
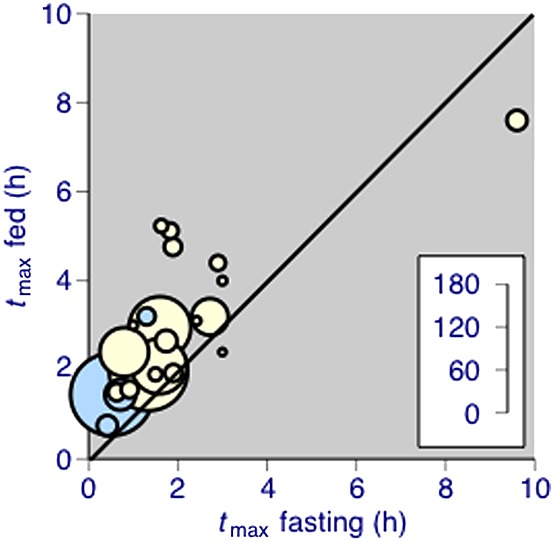
tmax in fed and fasted state for individual drugs. The inset scale indicates the number of participants in each comparison. Blue symbols are fast-acting formulations of diclofenac potassium, ibuprofen lysine, naproxen sodium and paracetamol plus bicarbonate
Table 1.
Mean results by drug, weighted by number of participants in pooled analyses
| Number of | Mean tmax (h) (weighted by n) | Mean Cmax (mg l–1 mg–1 dose ) (weighted by n) | ||||||
|---|---|---|---|---|---|---|---|---|
| Drug | Comparison | Participant | Fasted | Fed | Fed/Fasting (%) | Fasted | Fed | Fed/Fasting g(%) |
| Aspirin (as salicylic acid) | 3 | 30 | 1.74 | 2.65 | 152 | 0.093 | 0.079 | 85 |
| Bromfenac | 1 | 68 | 0.80 | 2.40 | 300 | 0.078 | 0.022 | 28 |
| Celecoxib | 2 | 51 | 2.72 | 3.19 | 117 | 0.0041 | 0.0065 | 159 |
| Dexketoprofen trometamol | 1 | 21 | 0.63 | 1.50 | 238 | 0.095 | 0.053 | 56 |
| Diclofenac potassium | 4 | 115 | 0.52 | 1.45 | 280 | 0.041 | 0.022 | 54 |
| Diclofenac sodium | 3 | 22 | 1.83 | 5.12 | 280 | 0.025 | 0.011 | 44 |
| Diflunisal | 1 | 12 | 2.42 | 3.10 | 128 | 0.144 | 0.120 | 83 |
| Dipyrone (as methylaminoantipyrine) | 1 | 18 | 1.50 | 1.90 | 127 | 0.010 | 0.0097 | 97 |
| Etoricoxib | 1 | 12 | 1.00 | 3.00 | 300 | 0.020 | 0.013 | 65 |
| Ibuprofen acid | 6 | 109 | 1.34 | 1.96 | 147 | 0.076 | 0.059 | 78 |
| Ibuprofen fast-acting | 2 | 43 | 0.71 | 1.44 | 203 | 0.096 | 0.070 | 73 |
| Indomethacin | 3 | 86 | 1.60 | 2.95 | 184 | 0.065 | 0.030 | 46 |
| Ketoprofen | 2 | 24 | 1.89 | 4.76 | 252 | 0.153 | 0.094 | 61 |
| Meloxicam | 2 | 22 | 9.30 | 7.10 | 76 | 0.059 | 0.068 | 115 |
| Nabumetone (as 6-methoxy-2-naphthlacetic acid) | 1 | 12 | 3.00 | 4.00 | 133 | 0.023 | 0.036 | 157 |
| Naproxen | 2 | 24 | 1.90 | 1.93 | 102 | 0.146 | 0.136 | 93 |
| Naproxen sodium | 1 | 24 | 1.30 | 3.20 | 246 | 0.291 | 0.229 | 79 |
| Paracetamol | 4 | 77 | 1.58 | 2.08 | 132 | 0.019 | 0.011 | 58 |
| Paracetamol + bicarbonate | 1 | 28 | 0.42 | 0.75 | 179 | 0.024 | 0.013 | 54 |
| Surprofen | 1 | 24 | 0.92 | 1.56 | 170 | 0.095 | 0.042 | 44 |
| Tenidap | 1 | 21 | 2.90 | 4.40 | 152 | 0.150 | 0.153 | 102 |
| Tenoxicam | 2 | 18 | 1.63 | 5.23 | 320 | 0.143 | 0.118 | 83 |
| Timegadine | 1 | 10 | 3.00 | 2.40 | 80 | 0.0013 | 0.0028 | 215 |
Effects of food on Cmax
Food reduced the Cmax for all drugs with a fasting tmax of under 2 h (Table 1). The greatest degree of reduction in Cmax was in analgesics with the shortest fasting tmax, with reductions by about 50% or more for bromfenac, dexketoprofen, diclofenac, indomethacin, paracetamol and surprofen. For celecoxib, nabumetone and timegadine Cmax increased in the fed state, while meloxicam was unaffected.
Discussion
The results of the review are clear. Taking analgesics with food does not affect the overall bioavailability, but for many drugs food substantially prolongs absorption and tmax, while substantially reducing Cmax. This is particularly the case for those drugs whose fasting tmax is below 2 h, which includes analgesics typically available without prescription in many parts of the world, namely aspirin, diclofenac potassium, ibuprofen (as acid and salt) and paracetamol (Figure4). Naproxen is affected to a much lesser extent.
Figure 4.
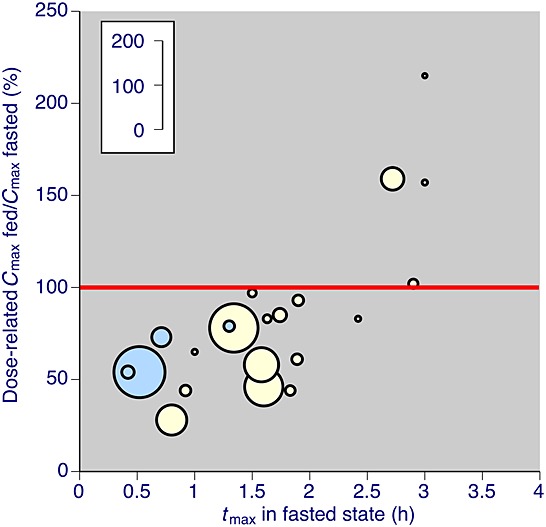
Cmax in fed and fasted state for individual drugs. The inset scale indicates the number of participants in each comparison. Blue symbols are fast-acting formulations of diclofenac potassium, ibuprofen lysine, naproxen sodium and paracetamol plus bicarbonate. Note that only data with tmax when fasting of less than 4 h are shown
The strength of these findings is that the systematic review covered all simple analgesics and NSAIDs, using a search strategy likely to be comprehensive, and which was unrestricted by date or language of publication. Included studies covered a 40 year period from 1972 to 2012. The effects of food on delayed tmax appeared to be similar in all the drugs examined with a fasting tmax of 4 h or less. The effects of food on reduced Cmax appeared to be similar in all the drugs examined with a fasting tmax of less than 2 h. Results were not compromised by the inclusion of delayed or controlled formulations specifically designed to delay absorption.
There are several potential weaknesses. This review covers only a part of the potentially available data, since many bioavailability studies have been performed for regulatory or other purposes, but not published. We are aware that this may be the case from discussions with pharmaceutical companies. Effects of food are often explored to demonstrate bioavailability equivalence following small changes in formulation. This review shows that bioavailability was unaffected by food (Figure2), and the results of such studies would therefore have been seen as unremarkable and not worthy of publication. Effects on Cmax and tmax would probably have been overlooked as having no importance. Recent research has shown that Cmax and tmax are more important than bioavailability, and this study may provoke companies with unpublished studies to revisit them, with a view to confirming the results presented here. We do not consider lack of publication of the reports to be a form of publication bias, since there were no grounds for bias at the time.
Other weaknesses include the small size of many of the individual studies, so some imprecision in the estimates of the magnitude of food effects is therefore likely. There were also insufficient details to determine whether the type of meal had any major consequences for any particular drug, or for drugs in general, and to explore links between physicochemical properties of individual drugs, type of meal and the effects of food. Finally, these data, while relevant to occasional use of analgesics, for headache, for example, cannot necessarily be extrapolated to long term repeat dosing.
The implications of the results are significant, and stem from the increasing understanding of the importance of early, high, plasma drug concentrations for pain relief, particularly in the treatment of acute pain. An analysis of a randomized trial in acute pain demonstrated a direct link between the concentration of ibuprofen and the absence of patients with moderate or severe pain at 1 h after dosing [7]. There was also a direct link between dose, plasma ibuprofen concentration and the changes in Western Ontario and McMaster Universities Osteoarthritis Index and visual analogue scale pain scores between baseline and after 7 days of treatment in osteoarthritis [50,51], with a weak correlation in rheumatoid arthritis [52].
Moreover, two recent patient level analyses of data from different acute pain studies have demonstrated that speed of onset of analgesia is an important determinant of good and possibly long lasting pain relief [8,9]. For fast-acting ibuprofen formulations with a median tmax of 30–40 min, 200 mg provided the same pain relief after third molar extraction (number needed to treat [NNT] for at least 50% maximum pain relief over 6 h 2.9, 95% confidence interval (CI) 2.3, 4.1) as 400 mg ibuprofen acid with a median tmax of 100 min (NNT 2.7, 95% CI 2.5, 3.1) in a Cochrane review [53] and in an updated analysis [8]. Poor analgesia leads to more frequent recourse to additional analgesic consumption [8,9].
At least for ibuprofen, where we have most information, early, high plasma concentrations produce better analgesia in acute pain, and result in reduced need for remedication (Figure5). There is no obvious reason why this should not be the case for other analgesics. If taking analgesics with food delays absorption and reduces peak plasma concentrations, poorer analgesia is likely to result. This indeed is what happened in the only test of taking food with analgesics we could find, with bromfenac [22], though the effects of food on paracetamol and naproxen were minimal. The impact on efficacy of taking analgesics with food is largely unexplored in acute and chronic pain, and this makes the case for new studies examining the effects of food on analgesic effects, as well as pharmacokinetics.
Figure 5.
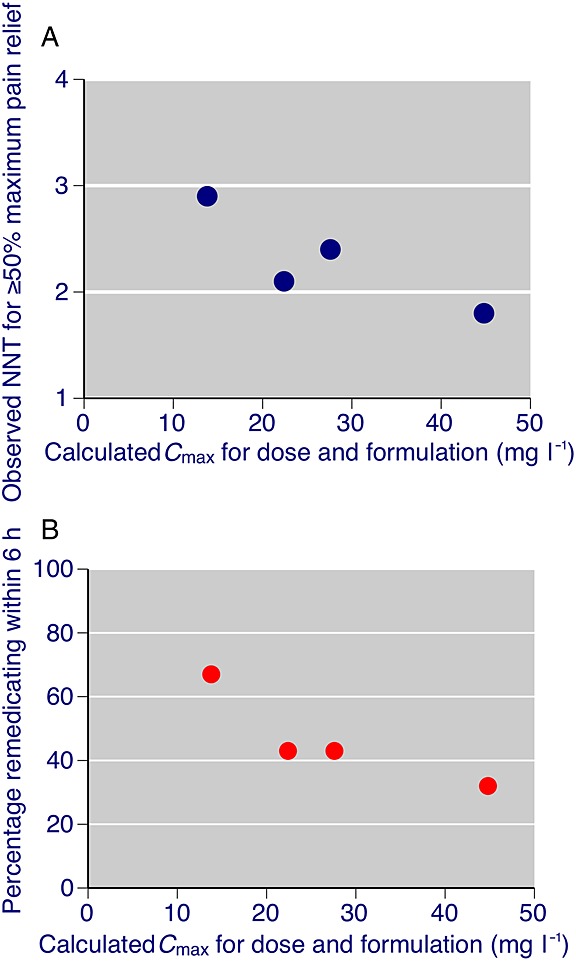
Relationship between A) Cmax for doses of 200 mg and 400 mg ibuprofen in standard acid and fast acting formulations and observed NNT (equation to regression line –0.031 × + 3.1 (r2 = 0.74)) and B) observed remedication rate (equation to regression line –1.0 × + 74 (r2 = 0.79)). Calculated using data from ss. Weighted mean Cmax values were multiplied by dose to obtain Cmax values; NNT and percentage remedicating values were taken from tables or text
The advice to take aspirin and NSAIDs with food stems from a desire to protect people from gastrointestinal adverse events; common events like dyspepsia, and rare, serious events like gastrointestinal bleeding. Aspirin is associated with increased rates of adverse events and gastric irritation [54], but there is no evidence that taking occasional doses of NSAIDs or most other analgesics produces higher adverse event rates than placebo [5]. Nor is there any convincing evidence that food prevents any other adverse events [6].
Conclusion
This leaves us with a conundrum. Current advice to take aspirin and NSAIDs with food is of no known benefit, but has a major effect on the most commonly used analgesics, including paracetamol, in a way that probably reduces analgesic efficacy for a given dose and increases the likelihood of additional doses or different analgesics being taken. There is a clear link between higher dose and greater risk of harm for NSAIDs in chronic pain [55]. Analgesics are taken to reduce pain. The evidence we have is strongly suggestive that taking them with food reduces their efficacy, especially those available to the public without prescription. It may be time to rethink research priorities and advice to patients and public.
Competing Interests
All authors have completed the Unified Competing Interest form at http://www.icmje.org/coi_disclosure.pdf (available on request from the corresponding author). The work was supported by an investigator-initiated unrestricted educational grant from Reckitt Benckiser. Reckitt Benckiser had no role in the study design, data collection analysis and interpretation, or writing of the paper. RAM is the owner of Oxford Medical Knowledge (OMK), which received the unrestricted educational grant from Reckitt Benckiser. Dr Moore reports grants from Reckitt Benckiser, during the conduct of the study, personal fees from Reckitt Benckiser, personal fees from Novartis, grants and personal fees from Grunenthal, personal fees from Orion Pharma, personal fees from Futura Pharma, personal fees from Astellas, personal fees from Eli Lilly, personal fees from Pfizer and personal fees from Menarini, outside the submitted work. Ms Derry has nothing to disclose. Professor Wiffen has nothing to disclose. Dr Straube reports personal fees from Oxford Medical Knowledge, outside the submitted work.
The Oxford Pain Relief Trust provided institutional support for this review. We are grateful to various library services for helping us obtain copies of papers from some rather difficult to find journals.
Supporting Information
Additional Supporting Information may be found in the online version of this article at the publisher's web-site:
Table S1 Details of potentially useful studies without results
Table S2 Details of individual studies
References
- 1.Clinical Knowledge Summary. Analgesia – mild to moderate pain. Available at http://cks.nice.org.uk/analgesia-mild-to-moderate-pain#!topicsummary (last accessed 1 December 2014)
- 2. NHS Choices. Available at http://www.nhs.uk/Conditions/Anti-inflammatories-non-steroidal/Pages/Interactions-othermedicines.aspx (last accessed 1 December 2014)
- 3.Schubert I, Fessler J, editors. Hausärztliche Leitlinien: herausgeben von der Leitliniengruppe Hessen und der PMV forschungsgruppe. Deutscher: Ärzte-Verlag; 2009. [Google Scholar]
- 4. Arthritis Medications: a consumer's guide. Available at http://www.arthritis.ca/document.doc?id=341 (last accessed 1 December 2014)
- 5.Moore RA, Derry S, McQuay HJ, Wiffen PJ. Single dose oral analgesics for acute postoperative pain in adults. Cochrane Database Syst Rev. 2011;9:CD008659. doi: 10.1002/14651858.CD008659.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rainsford KD, Bjarnason I. NSAIDs: take with food or after fasting? J Pharm Pharmacol. 2012;64:65–9. doi: 10.1111/j.2042-7158.2011.01406.x. [DOI] [PubMed] [Google Scholar]
- 7.Laska EM, Sunshine A, Marrero I, Olson N, Siegel C, McCormick N. The correlation between blood levels of ibuprofen and clinical analgesic response. Clin Pharmacol Ther. 1986;40:1–7. doi: 10.1038/clpt.1986.129. [DOI] [PubMed] [Google Scholar]
- 8.Moore RA, Derry S, Straube S, Ireson-Paine J, Wiffen PJ. Faster, higher, stronger? Evidence for formulation and efficacy for ibuprofen in acute pain. Pain. 2014;155:14–21. doi: 10.1016/j.pain.2013.08.013. [DOI] [PubMed] [Google Scholar]
- 9.Moore RA, Derry S, Straube S, Ireson-Paine J, Wiffen PJ. Validating speed of onset as a key component of good analgesic response in acute pain. Eur J Pain. 2014 doi: 10.1002/ejp.536. May 22. doi: 10.1002/ejp.536. [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lemeshow AR, Blum RE, Berlin JA, Stoto MA, Colditz GA. Searching one or two databases was insufficient for meta-analysis of observational studies. J Clin Epidemiol. 2005;58:58867–73. doi: 10.1016/j.jclinepi.2005.03.004. [DOI] [PubMed] [Google Scholar]
- 11.Ruppen W, Derry S, McQuay H, Moore RA. Incidence of epidural hematoma, infection, and neurologic injury in obstetric patients with epidural analgesia/anesthesia. Anesthesiology. 2006;105:394–9. doi: 10.1097/00000542-200608000-00023. [DOI] [PubMed] [Google Scholar]
- 12.Agrawal NG, Porras AG, Matthews CZ, Rose MJ, Woolf EJ, Musser BJ, Dynder AL, Mazina KE, Lasseter KC, Hunt TL, Schwartz JI, McCrea JB, Gottesdiener KM. Single- and multiple-dose pharmacokinetics of etoricoxib, a selective inhibitor of cyclooxygenase-2, in man. J Clin Pharmacol. 2003;43:268–76. doi: 10.1177/0091270003251122. [DOI] [PubMed] [Google Scholar]
- 13.Bannwarth B, Lapicque F, Netter P, Monot C, Tamisier JN, Thomas P, Royer RJ. The effect of food on the systemic availability of ketoprofen. Eur J Clin Pharmacol. 1988;33:643–5. doi: 10.1007/BF00542503. [DOI] [PubMed] [Google Scholar]
- 14.Bogentoft C, Carlsson I, Ekenved G, Magnusson A. Influence of food on the absorption of acetylsalicylic acid from enteric-coated dosage forms. Eur J Clin Pharmacol. 1978;14:351–5. doi: 10.1007/BF00611905. [DOI] [PubMed] [Google Scholar]
- 15.Busch U, Heinzel G, Narjes H. Effect of food on pharmacokinetics of meloxicam, a new non steroidal anti-inflammatory drug (NSAID) Agents Actions. 1991;32:52–3. doi: 10.1007/BF01983308. [DOI] [PubMed] [Google Scholar]
- 16.Caillé G, du Souich P, Besner JG, Gervais P, Vézina M. Effects of food and sucralfate on the pharmacokinetics of naproxen and ketoprofen in humans. Am J Med. 1989;86:38–44. doi: 10.1016/0002-9343(89)90155-1. [DOI] [PubMed] [Google Scholar]
- 17.Chaikin P, Marriott TB, Simon D, Weintraub HS. Comparative bioavailability of suprofen after coadministration with food or milk. J Clin Pharmacol. 1988;28:1132–5. doi: 10.1002/j.1552-4604.1988.tb05730.x. [DOI] [PubMed] [Google Scholar]
- 18.Coates PE, Mesure R. Pharmacokinetics of tenidap sodium administered with food or antacid in healthy volunteers. Br J Clin Pharmacol. 1995;39(Suppl 1):17S–9. doi: 10.1111/j.1365-2125.1995.tb04496.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Day RO, Lam S, Paull P, Wade D. Effect of food and various antacids on the absorption of tenoxicam. Br J Clin Pharmacol. 1987;24:323–8. doi: 10.1111/j.1365-2125.1987.tb03176.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Emori HW, Paulus H, Bluestone R, Champion GD, Pearson C. Indomethacin serum concentrations in man. Effects of dosage, food, and antacid. Ann Rheum Dis. 1976;35:333–8. doi: 10.1136/ard.35.4.333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Flusser D, Zylber-Katz E, Granit L, Levy M. Influence of food on the pharmacokinetics of dipyrone. Eur J Clin Pharmacol. 1988;34:105–7. doi: 10.1007/BF01061429. [DOI] [PubMed] [Google Scholar]
- 22.Forbes JA, Sandberg RA, Bood-Björklund L. The effect of food on bromfenac, naproxen sodium, and acetaminophen in postoperative pain after orthopedic surgery. Pharmacotherapy. 1998;18:492–503. [PubMed] [Google Scholar]
- 23.Francis RJ, Dixon JS, Lowe JR, Harris PA. The effects of food and of antacid on the single oral dose pharmacokinetics of tenoxicam. Eur J Drug Metab Pharmacokinet. 1985;10:309–14. doi: 10.1007/BF03189758. [DOI] [PubMed] [Google Scholar]
- 24.Geisslinger G, Dietzel K, Bezler H, Nuernberg B, Brune K. Therapeutically relevant differences in the pharmacokinetical and pharmaceutical behavior of ibuprofen lysinate as compared to ibuprofen acid. Int J Clin Pharmacol Ther Toxicol. 1989;27:324–8. [PubMed] [Google Scholar]
- 25.George S, Dauwe K, McBurney A, Ward J. The influence of food intake on the bioavailability of timegadine, a novel non-steroidal anti-inflammatory drug. Br J Clin Pharmacol. 1983;15:495–8. doi: 10.1111/j.1365-2125.1983.tb01537.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hasan SM, Ahmed T, Talib N, Hasan F. Pharmacokinetics of diclofenac sodium in normal man. Pak J Pharm Sci. 2005;18:18–24. [PubMed] [Google Scholar]
- 27.Kapil R, Nolting A, Roy P, Fiske W, Benedek I, Abramowitz W. Pharmacokinetic properties of combination oxycodone plus racemic ibuprofen: two randomized, open-label, crossover studies in healthy adult volunteers. Clin Ther. 2004;26:2015–25. doi: 10.1016/j.clinthera.2004.12.013. [DOI] [PubMed] [Google Scholar]
- 28.Klueglich M, Ring A, Scheuerer S, Trommeshauser D, Schuijt C, Liepold B, Berndl G. Ibuprofen extrudate, a novel, rapidly dissolving ibuprofen formulation: relative bioavailability compared to ibuprofen lysinate and regular ibuprofen, and food effect on all formulations. J Clin Pharmacol. 2005;45:1055–61. doi: 10.1177/0091270005279579. [DOI] [PubMed] [Google Scholar]
- 29.Koch PA, Schultz CA, Wills RJ, Hallquist SL, Welling PG. Influence of food and fluid ingestion on aspirin bioavailability. J Pharm Sci. 1978;67:1533–5. doi: 10.1002/jps.2600671110. [DOI] [PubMed] [Google Scholar]
- 30.Lafontaine D, Mailhot C, Vermeulen M, Bissonnette B, Lambert C. Influence of chewable sucralfate or a standard meal on the bioavailability of naproxen. Clin Pharm. 1990;9:773–7. [PubMed] [Google Scholar]
- 31.Levine MA, Walker SE, Paton TW. The effect of food or sucralfate on the bioavailability of S(+) and R(-) enantiomers of ibuprofen. J Clin Pharmacol. 1992;32:1110–4. [PubMed] [Google Scholar]
- 32.Manvelian G, Daniels S, Altman R. A phase I study evaluating the pharmacokinetic profile of a novel, proprietary, nano-formulated, lower-dose oral indomethacin. Postgrad Med. 2012;124:197–205. doi: 10.3810/pgm.2012.07.2580. [DOI] [PubMed] [Google Scholar]
- 33.Marzo A, Dal Bo L, Wool C, Cerutti R. Bioavailability, food effect and tolerability of S-naproxen betainate sodium salt monohydrate in steady state. Arzneimittelforschung. 1998;48:935–40. [PubMed] [Google Scholar]
- 34.McEwen J, De Luca M, Casini A, Gich I, Barbanoj MJ, Tost D, Artigas R, Mauleón D. The effect of food and an antacid on the bioavailability of dexketoprofen trometamol. J Clin Pharmacol. 1998;38(12 Suppl):41S–5. [PubMed] [Google Scholar]
- 35.Paulson SK, Vaughn MB, Jessen SM, Lawal Y, Gresk CJ, Yan B, Maziasz TJ, Cook CS, Karim A. Pharmacokinetics of celecoxib after oral administration in dogs and humans: effect of food and site of absorption. J Pharmacol Exp Ther. 2001;297:638–45. [PubMed] [Google Scholar]
- 36.Poli A, Moreno RA, Ribeiro W, Dias HB, Moreno H, Jr, Muscara MN, De Nucci G. Influence of gastric acid secretion blockade and food intake on the bioavailability of a potassium diclofenac suspension in healthy male volunteers. Int J Clin Pharmacol Ther. 1996;34:76–9. [PubMed] [Google Scholar]
- 37.Rostami-Hodjegan A, Shiran MR, Ayesh R, Grattan TJ, Burnett I, Darby-Dowman A, Tucker GT. A new rapidly absorbed paracetamol tablet containing sodium bicarbonate. I. A four-way crossover study to compare the concentration-time profile of paracetamol from the new paracetamol/sodium bicarbonate tablet and a conventional paracetamol tablet in fed and fasted volunteers. Drug Dev Ind Pharm. 2002;28:523–31. doi: 10.1081/ddc-120003448. [DOI] [PubMed] [Google Scholar]
- 38.Runkel RA, Kraft KS, Boost G, Sevelius H, Forchielli E, Hill R, Magoun R, Szakacs JB, Segre E. Naproxen oral absorption characteristics. Chem Pharm Bull (Tokyo) 1972;20:1457–66. doi: 10.1248/cpb.20.1457. [DOI] [PubMed] [Google Scholar]
- 39.Scallion R, Moore KA. Effects of food intake on the pharmacokinetics of diclofenac potassium soft gelatin capsules: a single-dose, randomized, two-way crossover study. Clin Ther. 2009;31:2233–41. doi: 10.1016/j.clinthera.2009.10.001. [DOI] [PubMed] [Google Scholar]
- 40.Scheidel B, Blume H, Walter K, Stanislaus F, Babej-Dölle RM. The bioavailability of enteric coated diclofenac formulations. 2. Bioavailability following single administration of a multiple-unit formulation in comparison to a single-unit formulation under fasting and non-fasting conditions].[Article in German. Arzneimittelforschung. 1994;44:544–50. [PubMed] [Google Scholar]
- 41.Siemon D, de Vries JX, Stötzer F, Walter-Sack I, Dietl R. Fasting and postprandial disposition of R(-)- and S(+)-ibuprofen following oral administration of racemic drug in healthy individuals. Eur J Med Res. 1997;2:215–9. [PubMed] [Google Scholar]
- 42.Stempak D, Gammon J, Halton J, Champagne M, Koren G, Baruchel S. Modulation of celecoxib pharmacokinetics by food in pediatric patients. Clin Pharmacol Ther. 2005;77:226–8. doi: 10.1016/j.clpt.2004.10.015. [DOI] [PubMed] [Google Scholar]
- 43.Stillings M, Havlik I, Chetty M, Clinton C, Schall R, Moodley I, Muir N, Little S. Comparison of the pharmacokinetic profiles of soluble aspirin and solid paracetamol tablets in fed and fasted volunteers. Curr Med Res Opin. 2000;16:115–24. [PubMed] [Google Scholar]
- 44.Tanner T, Aspley S, Munn A, Thomas T. The pharmacokinetic profile of a novel fixed-dose combination tablet of ibuprofen and paracetamol. BMC Clin Pharmacol. 2010;10:10. doi: 10.1186/1472-6904-10-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Terhaag B, Gramatte T, Hrdlcka P, Richter K, Feller K. The influence of food on the absorption of diclofenac as a pure substance. Int J Clin Pharmacol Ther Toxicol. 1991;29:418–21. [PubMed] [Google Scholar]
- 46.Tobert JA, DeSchepper P, Tjandramaga TB, Mullie A, Buntinx AP, Meisinger MA, Huber PB, Hall TL, Yeh KC. Effect of antacids on the bioavailability of diflunisal in the fasting and postprandial states. Clin Pharmacol Ther. 1981;30:385–9. doi: 10.1038/clpt.1981.177. [DOI] [PubMed] [Google Scholar]
- 47.Türck D, Busch U, Heinzel G, Narjes H, Nehmiz G. Effect of food on the pharmacokinetics of meloxicam after oral administration. Clin Drug Invest. 1995;9:270–6. [Google Scholar]
- 48.Vidon N, Pfeiffer A, Godbillon J, Rongier M, Gauron S, Hirtz J, Bernier JJ, Dubois JP. Evaluation of the gastric absorption and emptying of drugs under various pH conditions using a simple intubation method: application to diclofenac. Br J Clin Pharmacol. 1989;28:121–4. doi: 10.1111/j.1365-2125.1989.tb03515.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.von Schrader HW, Buscher G, Dierdorf D, Mügge H, Wolf D. Nabumetone - a novel anti-inflammatory drug: the influence of food, milk, antacids, and analgesics on bioavailability of single oral doses. Int J Clin Pharmacol Ther Toxicol. 1983;21:311–21. [PubMed] [Google Scholar]
- 50.Gallelli L, Galasso O, Urzino A, Saccà S, Falcone D, Palleria C, Longo P, Corigliano A, Terracciano R, Savino R, Gasparini G, De Sarro G, Southworth SR. Characteristics and clinical implications of the pharmacokinetic profile of ibuprofen in patients with knee osteoarthritis. Clin Drug Investig. 2012;32:827–33. doi: 10.1007/s40261-012-0011-7. [DOI] [PubMed] [Google Scholar]
- 51.Bradley JD, Rudy AC, Katz BP, Ryan SI, Kalasinski LA, Brater DC, Hall SD, Brandt KD. Correlation of serum concentrations of ibuprofen stereoisomers with clinical response in the treatment of hip and knee osteoarthritis. J Rheumatol. 1992;19:130–4. [PubMed] [Google Scholar]
- 52.Grennan DM, Aarons L, Siddiqui M, Richards M, Thompson R, Higham C. Dose–response study with ibuprofen in rheumatoid arthritis: clinical and pharmacokinetic findings. Br J Clin Pharmacol. 1983;15:311–6. doi: 10.1111/j.1365-2125.1983.tb01504.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Derry C, Derry S, Moore RA, McQuay HJ. Single dose oral ibuprofen for acute postoperative pain in adults. Cochrane Database Syst Rev. 2009;3:CD001548. doi: 10.1002/14651858.CD001548.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Edwards JE, Oldman AD, Smith LA, Carroll D, Wiffen PJ, McQuay HJ, Moore RA. Oral aspirin in postoperative pain: a quantitative systematic review. Pain. 1999;81:289–97. doi: 10.1016/S0304-3959(99)00022-6. [DOI] [PubMed] [Google Scholar]
- 55.Hernandez-Diaz S, Rodriguez LA. Association between nonsteroidal anti-inflammatory drugs and upper gastrointestinal tract bleeding/perforation: an overview of epidemiologic studies published in the 1990s. Arch Intern Med. 2000;160:2093–9. doi: 10.1001/archinte.160.14.2093. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1 Details of potentially useful studies without results
Table S2 Details of individual studies


