Abstract
Objective
Diet-induced thermogenesis (DIT) is lower in evening and at night than in the morning. This may help explain why meal-timing affects body weight regulation and why shift work is a risk factor for obesity. The separate effects of the endogenous circadian system—independent of behavioral cycles—and of circadian misalignment on DIT are unknown.
Methods
Thirteen healthy adults undertook a randomized, crossover study with two 8-day laboratory visits: three baseline days followed either by repeated simulated night shifts including 12-h inverted behavioral cycles (circadian misalignment) or by recurring simulated day shifts (circadian alignment). DIT was determined for up to 114 min (hereafter referred to as “early DIT”) following identical meals given at 8AM and 8PM in both protocols.
Results
During baseline days, early DIT was 44% lower in the evening than morning. This was primarily explained by a circadian influence rather than any behavioral cycle effect; early DIT was 50% lower in the biological evening than biological morning, independent of behavioral cycle influences. Circadian misalignment had no overall effect on early DIT.
Conclusions
The circadian system plays a dominating role in the morning/evening difference in early DIT and may contribute to the effects of meal-timing on body weight regulation.
Keywords: meal timing, specific dynamic action, thermic effect of food, time-of-day, obesity
Introduction
Diet-induced thermogenesis (DIT) is lower in the evening and at night than in the morning (1). This may help explain why meal timing is associated with body weight and why shift work is a risk factor for obesity (2-8). This 24-h rhythm in DIT could be caused by the behavioral cycle (including the sleep/wake and fasting/feeding cycles) and/or the endogenous circadian system. In mammals, the circadian system is composed of the central pacemaker in the suprachiasmatic nucleus of the hypothalamus along with circadian oscillators in virtually all tissues and organs and it is closely tied to metabolic regulation (9-11). Misalignment between the endogenous circadian system and 24-h environmental/behavioral cycles (i.e., circadian misalignment) may decrease DIT and consequently contribute to the increased risk for obesity in shift workers. To examine the separate and relative impact of the aforementioned three influences on DIT, we assessed DIT for up to 114 min following identical mixed meals (hereafter referred to as “early DIT”) given at 8AM and 8PM when the behavioral cycle of participants was aligned or misaligned with their circadian system using a 12-h rapid shift of the behavioral cycle (Fig. 1).
Fig. 1. Circadian alignment protocol (top panel) and circadian misalignment protocol (bottom panel).
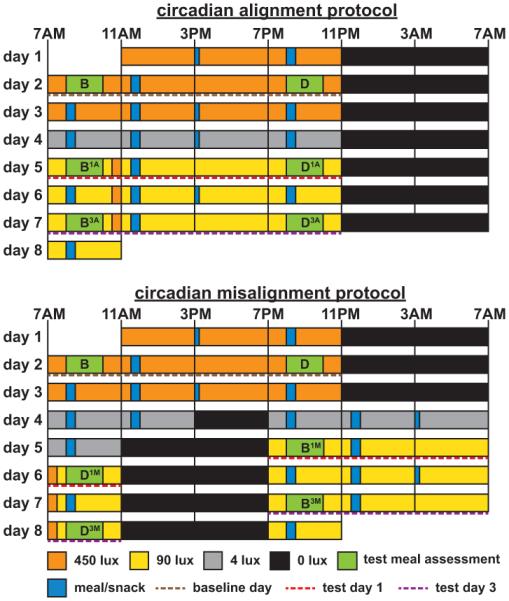
On day 1 in both protocols, participants received an ad libitum lunch at approximately 12PM. Caloric intake was prorated for the 12-h behavioral cycle on day 4 of the circadian misalignment protocol (i.e., they received 50% of the caloric content as compared to the other 24-h days). Light levels indicated are in the horizontal angle of gaze: ~90 lux, to simulate typical room light intensity, ~450 lux during the first three baseline wake episodes to enhance circadian entrainment, 30-min periods of ~450 lux to simulate the morning commute both preceding the day work shift (circadian alignment protocol) and following the night work shift (circadian misalignment protocol)—this was expected to oppose the central circadian pacemaker from delaying its phase during the circadian misalignment protocol (31), ~4 lux to permit assessment of the dim-light melatonin onset; 0 lux during scheduled sleep episodes. Light levels during test meal assessments were 450 lux on baseline days and 90 lux on experimental days. Light blue bars represent meals and snacks (narrow bar). Green bars represent test meal assessments, with the test meals consumed within the first 20 min. The letters B and D indicate breakfast and dinner, respectively. Numbers following B or D indicate test days (first or third) and letters following these numbers indicate whether the test meals were consumed during circadian alignment (A) or circadian misalignment (M). To graphically represent the independent effects of the behavioral cycle, circadian phase and circadian misalignment in subsequent figures, we (1) averaged breakfast time (BA and BM) and dinner time (DA and DM) test meal values separately across both protocols for each test day (behavioral cycle effect); (2) averaged 8AM (BA and DM) and 8PM (DA and BM) test meal values separately across both protocols for each test day (circadian phase effect); and (3) averaged alignment (BA and DA) and misalignment (BM and DM) test meal values within each protocol for each test day (circadian misalignment effect).
Methods
Participants
Other aspects of this study—which was designed to test different hypotheses—have been published before (12). Indirect calorimetry data were obtained from 13 healthy, non-smoking, drug and medication free (excepting oral contraceptives) adults (mean±SD [range] age, 29±10 y [20-49 y]; BMI, 25.0±3.0 kg/m2 [20.4-29.5 kg/m2]; 6 men). Females were admitted to the laboratory on either days 3 or 4 (n=2) or days 12-18 (n=5) of their menstrual cycle. For their second laboratory visit, females were admitted at a similar menstrual cycle phase to that of their first admission to the laboratory (mean±SD for the difference between visits, −1±1 menstrual cycle day). Participants had no shift work experience in the past three years and less than six months cumulative lifetime shift work exposure and had not crossed more than one time zone in the prior three months. Participants provided written informed consent and the Partners Human Research Committee approved this study.
Pre-inpatient study conditions
Participants selected and maintained a normal sleep/wake schedule, with an 8-h sleep opportunity, for ≥11 days (mean±SD 17±4 days) before each laboratory visit (mean±SD bedtime, 23:11±0:48 hh:mm; wake time, 7:12±0:47 hh:mm; data from 7 sleep periods preceding the final ambulatory sleep period before inpatient admissions). On the night immediately preceding each admission, participants had a sleep opportunity between 11PM and 7AM to aid the adaptation of their endogenous circadian system to the initial laboratory sleep/wake schedule.
Experimental design and inpatient study conditions
We employed two intricate laboratory protocols to test the separate impact of the behavioral cycle, circadian phase (biological morning [defined here as the endogenous circadian phase equivalent to approximately 8AM] vs. biological evening [~8PM]) and circadian misalignment on DIT. Participants were studied twice, once when the timing of their behavioral cycle was normal such that they slept and fasted at night and were awake and eating in the day (circadian alignment protocol; Fig. 1, top panel) and once when their behavioral cycle was rapidly inverted by 12 h such that they slept and fasted during the day and were awake and eating at night (circadian misalignment protocol; Fig 1, bottom panel). Together, the two protocols allowed the separate assessment of behavioral and circadian influences by evenly scheduling behavioral factors (e.g., the sleep/wake and fasting/feeding cycle) relative to two circadian phases separated by approximately 12 h. In addition, the protocols allowed the separate assessment of the impact of circadian misalignment by having assessments occur when the behavioral cycle was aligned and misaligned with the circadian system. Also, by comparing the impact of the behavioral cycle, circadian phase and circadian misalignment following acute and repeated exposure to misalignment (test day 1 [day 5 of the alignment protocol and day 5/6 of the misalignment protocol] vs. test day 3 [day 7 of the alignment protocol and day 7/8 of the misalignment protocol]; Fig. 1), we could assess if the three aforementioned factors depended on circadian misalignment exposure duration. Participants remained in a personal laboratory room throughout each laboratory protocol, which were randomly ordered and the washout period between laboratory visits was two to eight weeks (mean±SD 4±2 wks; Fig. 1). Light levels—in the horizontal angle of gaze—during the protocols are shown in Fig. 1. Eleven participants completed the two 8-day laboratory visits. Two additional participants contributed to the data from baseline day 2 (they completed baseline day 2 and thereafter discontinued the study). Thus, we assessed the separate effect of the behavioral cycle, circadian phase and circadian misalignment on DIT in 11 participants and the morning/evening difference (under normal behavioral cycle conditions) in DIT in 13 participants.
Diet
The timing of all meals and snacks is shown in Fig. 1. On day 1 of both laboratory protocols, an ad libitum lunch was provided after which participants were maintained on an isocaloric diet to meet the participant’s “daily” calorie requirements calculated according to the Harris-Benedict equation (activity factor 1.4; 45-50% carbohydrate, 30-35% fat, 15-20% protein, 150 mEq Na+ [±20%], 100 mEq K+ [±20%], and ≥2.5 L water/24 h). Diet was identical within each participant between laboratory visits, except for the required and prorated additional food and water given during the 12-h behavioral cycle (day 4) in the circadian misalignment protocol (i.e., 50% of a 24-h day’s worth of energy and fluid were provided). We assessed each participant’s metabolic response to identical test meals (each 33.3% of calculated daily calorie intake and consumed within 20 min) given both 1 h (“breakfast”) and 13 h (“dinner”) following scheduled wake time (day 2 of the circadian alignment and misalignment protocols [baseline days], day 5 and 7 in the alignment protocol and on day 5/6 and 7/8 in the misalignment protocol [experimental days]). Participants chose one of two test meals: (1) Glucola (0.45 g/kg), bagel with butter, cereal with milk and sugar, egg, and peanuts; (2) Glucola (0.45 g/kg), bagel with butter, cereal with milk and sugar, turkey sausage, and almonds. Glucola was consumed within the first minute and other food items were consumed subsequently, in the order listed above. The test meal given 1 h following scheduled wake was preceded by a dinner meal (30% of calculated daily calorie intake) consumed in the prior wake period (8PM when aligned and 8AM when misaligned). The test meal given 13 h following scheduled wake was preceded by a lunch meal (33.3% of calculated daily calorie intake), consumed at 11:30AM when aligned and 11:30PM when misaligned. For the dinner meal that preceded the “breakfast” test meal, participants preselected one of two meals which was identical within each participant across both protocols. For the lunch meal that preceded the “dinner” test meal, participants again preselected one of two meals which was identical within each participant across both protocols. The abovementioned dinner and lunch meals had the same macronutrient ratios as the test meals. On baseline day 2 and experimental days, when no snack was given, the calorie content of the breakfast (test meal), lunch and dinner (test meal) meals was 33.3% of calculated daily energy intake, with all meals totaling 100% of calculated daily energy intake. On other 24-h study days, the calorie content of breakfast, lunch, snack and dinner was 30%, 30%, 10% and 30% of calculated daily energy intake, respectively. For the 12-h behavioral cycle (day 4 of the circadian misalignment protocol), subjects consumed breakfast and lunch. The breakfast contained 60% and the lunch contained 40% of calculated energy intake required for the 12-h behavioral cycle.
Indirect calorimetry
Energy expenditure (EE) was derived from indirect calorimetry data (Vmax Encore 29N, VIASYS Healthcare Inc., Yorba Linda, CA). Measurements lasted for 24 min. Fasted assessments began 30 min before the start of each test meal and postprandial measurements began 30 and 90 min following the start of each test meal—thus we only assessed “early” postprandial EE (i.e., up to 114 min after meal start). Oxygen uptake (V̇O2) and carbon dioxide (V̇CO2) production data were recorded every min and data from the first 5 min of each recording were discarded. The remaining data were used to calculate energy expenditure (EE) using Weir’s formula (13):
| (1) |
Data analysis and statistics
Early DIT was calculated by subtracting resting EE from the average of the two postprandial EE assessments. For the baseline days, we employed linear mixed models with participant included as random factor to test the effect of meal time (morning vs. evening) on fasting and postprandial energy expenditure, and early DIT data (n=13). During experimental days, we used linear mixed models (again participant included as random factor), to test the independent effects of the behavioral cycle (subjective breakfast test meal [1 h after lights on] vs. subjective dinner test meal [13 h after lights on]), circadian phase (8AM test meal [biological morning] vs. 8PM test meal [biological evening]), and alignment condition (circadian alignment vs. circadian misalignment), and their interaction with test day (first vs. third) on fasting and early postprandial EE, and early DIT (n=11). To graphically represent these independent effects we: (1) averaged breakfast time and dinner time test meal values separately across both protocols for each test day (behavioral cycle effect); (2) averaged 8AM and 8PM test meal values separately across both protocols for each test day (circadian phase effect); and (3) averaged breakfast time and dinner time test meal values within each protocol for each test day (circadian misalignment effect). Where necessary, Bonferroni-adjusted multiple comparisons were conducted. Statistical significance was accepted as P<0.05. Data are presented as mean±SEM, unless otherwise indicated.
Results
Baseline days: early DIT and early postprandial EE were lower in the evening than morning
Early DIT was 44% lower in the evening (0.13±0.01 kcal/min) than morning (0.24±0.02 kcal/min; P<0.0001; Fig. 2). Early postprandial EE was 4% lower in the evening (1.18±0.03 kcal/min) than morning (1.24±0.04 kcal/min; P=0.0001; Fig. 2). Resting EE (REE) was not significantly different between the morning (1.00±0.04 kcal/min) and evening (1.05±0.04 kcal/min; P=0.084; Fig. 2).
Fig. 2. Baseline days (n=13); Early diet-induced thermogenesis (DIT, top panel), absolute early postprandial energy expenditure (postprandial EE, middle panel) and resting energy expenditure (REE, bottom panel) during the morning and evening test sessions during a normal sleep/wake cycle.
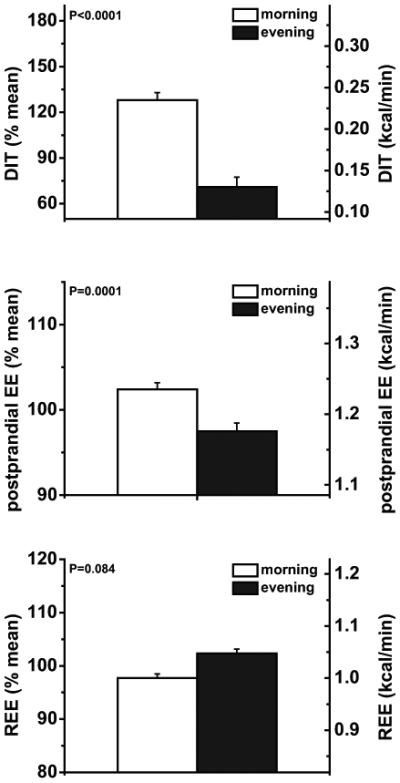
Data are represented as mean±SEM.
Experimental days: early DIT, early postprandial EE, and REE were similar at breakfast time and dinner time, independent of circadian effects
Early DIT, early postprandial EE, and REE were not affected by the behavioral cycle, comparing responses to identical test meals given at breakfast time vs. dinner time whilst controlling for circadian phase effects (all P≥0.087; Fig. 3 to 5, left panels). These findings were not influenced by exposure duration to circadian misalignment (first test day vs. third test day; all P≥0.38; Fig. 3 to 5, left panels).
Fig. 3. Experimental days (n=11); Effects of the behavioral cycle (left panel), circadian phase (middle panel) and circadian misalignment (right panel) on early diet-induced thermogenesis (DIT).
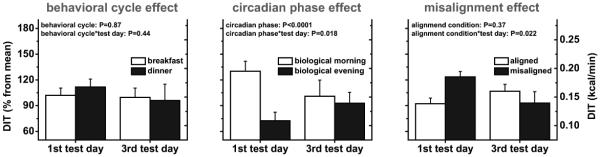
Data are derived as described in the legend of Fig. 1. Data are represented as mean±SEM.
Experimental days: early DIT and early postprandial EE were lower in the biological evening than in the biological morning, a circadian phase effect, independent of behavioral cycle effects
Early DIT was 34% lower in the biological evening (0.13±0.01 kcal/min) than in the biological morning (0.19±0.02 kcal/min; P<0.0001; Fig. 3, middle panel). This circadian phase effect was dependent on exposure duration to circadian misalignment (first vs. third test day; P=0.018), with early DIT being 50% lower in the biological evening (0.12±0.02 kcal/min) than in the biological morning (0.23±0.02 kcal/min) on the first test day (P<0.0001), while by the third test day there was no difference (P=0.20; Fig. 3, middle panel). This reflected both an increase in early DIT in the biological evening from the first to third test day (+16%) and a reduction in early DIT in the biological morning across test days (−30%). Early postprandial EE was 3% lower in the biological evening (1.14±0.02 kcal/min) than in the biological morning (1.17±0.03 kcal/min; P=0.025), irrespective of exposure duration to circadian misalignment (P=0.19; Fig. 4, middle panel). REE was not significantly different between the biological morning (0.98±0.03 kcal/min) and biological evening (1.01±0.03 kcal/min), regardless of exposure duration to circadian misalignment (both P≥0.091; Fig. 5, middle panel).
Fig. 4. Experimental days (n=11); Effects of the behavioral cycle (left panel), circadian phase (middle panel) and circadian misalignment (right panel) on absolute early postprandial energy expenditure (postprandial EE).
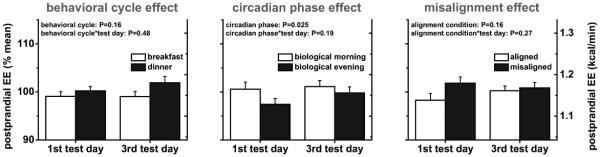
Data are derived as described in the legend of Fig. 1. Data are represented as mean±SEM.
Fig. 5. Experimental days (n=11); Effects of the behavioral cycle (left panel), circadian phase (middle panel) and circadian misalignment (right panel) on resting energy expenditure (REE).
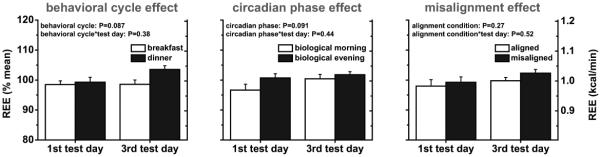
Data are derived as described in the legend of Fig. 1. Data are represented as mean±SEM.
Experimental days: no overall effect of circadian misalignment on early DIT
Overall early DIT was not significantly different between circadian alignment and misalignment conditions (P=0.37; Fig. 3, right panel). This effect depended on circadian misalignment exposure duration (P=0.022), with early DIT being 23% higher during circadian misalignment (0.20 ± 0.02 kcal/min) than circadian alignment (0.16 ± 0.01 kcal/min) on test day 1 (P=0.027), whereas there was no difference on test day 3 (P=0.31; Fig. 3, right panel). Early postprandial EE and REE were not affected by circadian misalignment, regardless of misalignment exposure duration (all P≥0.16; Fig. 4 and 5, right panels).
Discussion
We show that early DIT is lower in the evening than in the morning during a normal sleep/wake cycle, consistent with previous observations (1). For the first time, our results reveal a strong endogenous circadian effect on early DIT, with early DIT twice as large during the biological morning as compared to the biological evening on test day 1 (when the central circadian clock was expected to be minimally blunted and/or shifted). Early DIT was not significantly influenced by the behavioral cycle, i.e., comparing breakfast time with dinner time, independent of endogenous circadian phase. These findings suggest that the morning/evening difference in early DIT is caused primarily by the endogenous circadian system and not by the behavioral cycle.
We found that early DIT was 44% lower (mean difference, 0.11 kcal/min) in the evening than in the morning on baseline days. However, early postprandial EE was only 4% lower (mean difference, −0.06 kcal/min) in the evening than in the morning on baseline days. It is unclear if the morning/evening difference in postprandial EE would importantly influence total energy expenditure, and consequently energy balance. Future studies are needed to determine whether changes in meal timing influence energy balance and body weight regulation (3, 5), through this morning/evening difference in DIT. Repeated exposure to circadian misalignment removed the effect of the endogenous circadian system on early DIT. This likely reflects a blunting and/or phase shift of the central circadian pacemaker caused by repeated exposure to circadian misalignment (14-17).
DIT is a result of “obligatory” (energy cost of absorbing, processing and storing nutrients) and “facultative” (primarily sympathetic nervous system activity) mechanisms (18). Absorption rate has been shown to be lower in the evening than morning (19) and circadian clock genes are rhythmically expressed in the gastrointestinal system in rodents (20, 21). However, it is unknown whether the day/night rhythm in absorption rate is driven by the behavioral cycle and/or endogenous circadian system. If glucose gastrointestinal absorption rate is under endogenous circadian control and would have been slower in the biological evening than in the biological morning this could have resulted in reduced glycogen synthesis, a process which is a major determinant of glucose-induced thermogenesis, and thus could have been an mechanism underlying the endogenous circadian phase effect on early DIT (22, 23). Considering we only measured DIT until 114 min following the start of test meal consumption, a circadian phase effect on the temporal pattern of nutrient absorption from the gastrointestinal system could also help explain our DIT results. Future studies are required to verify the magnitude of the difference between the biological morning and evening in DIT when assessed over the full postprandial period, while addressing confounding effects of either sleep or sleep deprivation if the measurement duration would require measurements beyond habitual bedtime.
Impaired glucose uptake/tolerance decreases DIT, mainly by reducing the conversion of exogenous glucose to glycogen (24-26). In healthy humans, glucose tolerance is reduced in the biological evening vs. the biological morning (27, 28) which may help explain the lower early DIT in the biological evening vs. the biological morning.
The sympathetic nervous system stimulates DIT (18). However, the endogenous circadian rhythm in circulating epinephrine and/or norepinephrine levels is unlikely to contribute to the lower early DIT in the biological evening than biological morning, because epinephrine and norepinephrine levels are higher in the biological evening than in the biological morning (29).
We found that circadian misalignment increased early DIT on test day 1, but there was no effect of alignment condition by test day 3, which did not support our original hypothesis of a decrease of DIT with circadian misalignment. When assessing the impact of circadian misalignment across both test days, we found no effect (i.e., no net impact of circadian misalignment on early DIT). Thus, short-term circadian misalignment is unlikely to increase body weight via changes in early DIT. DIT has been reported to be transiently lower during simulated night work (30). However, it is unknown if that resulted from food consumption at different circadian phases and/or circadian misalignment. In our study, by having assessments at two distinctive circadian phases in both the circadian alignment and misalignment protocols, we could separate the circadian phase affect (averaged 8AM and 8PM test meal values separately across both protocols) from the circadian misalignment affect (averaged breakfast time and dinner time test meal values within each protocol).
Strengths of our study include conducting measurements under highly-controlled conditions. Study limitations include that we only assessed early DIT at two times in the wake episode and at two circadian phases. Thus, we may have missed any differences between conditions that occur a few hours after a meal and may not have captured the maximal effect of the behavioral cycle and/or the circadian system if the peak and/or trough of their effects were missed. Early DIT assessments occurred at 8AM and 8PM for several reasons: (a) these times are compatible with a typical meal schedule without interrupting sleep; (b) they are targeting the maximum and minimum DIT previously reported under regular sleep/wake cycles; (c) DIT can only be assessed after at least ~8 h of fasting, limiting the number of assessments to two per day; and (d) by scheduling them 12 h apart, they occurred at the same clock time in the aligned and 12-h misaligned conditions. A shorter fast occurred before dinner than breakfast. We purposely designed our study such that there was a shorter fasting period before dinner than breakfast because this is typically the case for most meal schedules. Our sample size was relativity small, thus we may have been statistically underpowered to detect some differences. We studied healthy, non-obese people. Future studies are required to test whether similar effects are observed in other populations such as shift workers or obese individuals.
The decrease in early DIT from the morning to the evening is primarily due to the influence of the endogenous circadian system, and not the behavioral cycle. These results provide a possible mechanism to help explain why the timing of food intake per se is associated with body weight.
What is already known about this subject?
Diet-induced thermogenesis is lower in evening and at night than in the morning and this may help explain why meal timing is associated with body weight and why shift work is a risk factor for obesity.
The mechanisms underlying the 24-h rhythm in diet-induced thermogenesis are not well understood.
What this study adds?
The decrease in diet-induced thermogenesis from the morning to the evening may be primarily due to the influence of the endogenous circadian system, and not the behavioral cycle (including the sleep/wake and fasting/feeding cycle).
The impact of the endogenous circadian system on diet-induced thermogenesis appears to be abolished following repeated days of circadian misalignment.
Short-term circadian misalignment may have no overall impact on diet-induced thermogenesis.
Acknowledgements
We thank the research volunteers and the technical staff of the Center for Clinical Investigation at Brigham and Women’s Hospital. We also thank Janis F. Swain, R.D., Karen Yee, R.D., and Leigh K. Keating, R.D., for their expert assistance with diet preparation. Funding: This study was supported by National Heart, Lung, and Blood Institute Grant R01 HL094806 to F.A.J.L.S. C.J.M. was partly supported by the National Space Biomedical Research Institute through National Aeronautics and Space Administration Grant NCC 9-58, and by National Heart, Lung, and Blood Institute Grant R01 HL094806, National Institute of Diabetes and Digestive and Kidney Diseases Grant R01 DK099512, and National Heart, Lung, and Blood Institute Grant R01 HL118601. F.A.J.L.S. was supported in part by National Heart, Lung, and Blood Institute Grant R01 HL094806, National Institute of Diabetes and Digestive and Kidney Diseases Grant R01 DK099512, and National Heart, Lung, and Blood Institute Grant R01 HL118601. The project described was supported by Clinical Translational Science Award UL1RR025758 to Harvard University and Brigham and Women’s Hospital from the National Center for Research Resources. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
Footnotes
Conflict of interest statement
The authors have nothing to disclose.
Author contributions: F.A.J.L.S. and C.J.M. designed the study and supervised data collection by J.I.G., S.M., J.N.Y., N.T. and Center for Clinical Investigation technical staff. J.I.G., S.M., J.N.Y. and N.T. assisted C.J.M. and F.A.J.L.S with data management and analysis. C.J.M. and F.A.L.J.S. formulated the statistical analysis plan. C.J.M. performed statistical analyses. C.J.M. and F.A.L.J.S. drafted the manuscript and all authors contributed to and approved the final version.
References
- 1.Romon M, Edme J, Boulenguez C, Lescroart J, Frimat P. Circadian variation of diet-induced thermogenesis. Am J Clin Nutr. 1993;57:476–480. doi: 10.1093/ajcn/57.4.476. [DOI] [PubMed] [Google Scholar]
- 2.Arble DM, Bass J, Laposky AD, Vitaterna MH, Turek FW. Circadian Timing of Food Intake Contributes to Weight Gain. Obesity. 2009;17:2100–2102. doi: 10.1038/oby.2009.264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Jakubowicz D, Barnea M, Wainstein J, Froy O. High caloric intake at breakfast vs. dinner differentially influences weight loss of overweight and obese women. Obesity. 2013;21:2504–2512. doi: 10.1002/oby.20460. [DOI] [PubMed] [Google Scholar]
- 4.Pan A, Schernhammer ES, Sun Q, Hu FB. Rotating Night Shift Work and Risk of Type 2 Diabetes: Two Prospective Cohort Studies in Women. PLoS Med. 2011;8:e1001141. doi: 10.1371/journal.pmed.1001141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Garaulet M, Gómez-Abellán P, Alburquerque-Béjar JJ, Lee Y-C, Ordovás JM, Scheer FA. Timing of food intake predicts weight loss effectiveness. International Journal of Obesity. 2013;37:604–611. doi: 10.1038/ijo.2012.229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Baron KG, Reid KJ, Kern AS, Zee PC. Role of Sleep Timing in Caloric Intake and BMI. Obesity. 2011;19:1374–1381. doi: 10.1038/oby.2011.100. [DOI] [PubMed] [Google Scholar]
- 7.Di Lorenzo L, De Pergola G, Zocchetti C, L'Abbate N, Basso A, Pannacciulli N, et al. Effect of shift work on body mass index: results of a study performed in 319 glucose-tolerant men working in a Southern Italian industry. Int J ObesRelat Metab Disord. 2003;27:1353–1358. doi: 10.1038/sj.ijo.0802419. [DOI] [PubMed] [Google Scholar]
- 8.Suwazono Y, Dochi M, Sakata K, Okubo Y, Oishi M, Tanaka K, et al. A Longitudinal Study on the Effect of Shift Work on Weight Gain in Male Japanese Workers. Obesity. 2008;16:1887–1893. doi: 10.1038/oby.2008.298. [DOI] [PubMed] [Google Scholar]
- 9.Morris CJ, Aeschbach D, Scheer FA. Circadian system, sleep and endocrinology. Mol Cell Endocrinol. 2012;349:91–104. doi: 10.1016/j.mce.2011.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Mohawk JA, Green CB, Takahashi JS. Central and Peripheral Circadian Clocks in Mammals. Annual Review of Neuroscience. 2012;35:445–462. doi: 10.1146/annurev-neuro-060909-153128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Morris CJ, Yang JN, Scheer FA. The impact of the circadian timing system on cardiovascular and metabolic function. Prog Brain Res. 2012;199:337–358. doi: 10.1016/B978-0-444-59427-3.00019-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Morris CJ, Yang JN, Garcia JI, Myers S, Bozzi I, Wang W, et al. Endogenous circadian system and circadian misalignment impact glucose tolerance via separate mechanisms in humans. Proceedings of the National Academy of Sciences. 2015 doi: 10.1073/pnas.1418955112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Weir JB. New methods for calculating metabolic rate with special reference to protein metabolism. J Physiol (Lond) 1949;109:1–9. doi: 10.1113/jphysiol.1949.sp004363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fonken LK, Workman JL, Walton JC, Weil ZM, Morris JS, Haim A, et al. Light at night increases body mass by shifting the time of food intake. Proceedings of the National Academy of Sciences. 2010;107:18664–18669. doi: 10.1073/pnas.1008734107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Salgado-Delgado RC, Saderi N, Basualdo MdC, Guerrero-Vargas NN, Escobar C, Buijs RM. Shift Work or Food Intake during the Rest Phase Promotes Metabolic Disruption and Desynchrony of Liver Genes in Male Rats. PLoS ONE. 2013;8:e60052. doi: 10.1371/journal.pone.0060052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Qian J, Block GD, Colwell CS, Matveyenko AV. Consequences of Exposure to Light at Night on the Pancreatic Islet Circadian Clock and Function in Rats. Diabetes. 2013;62:3469–3478. doi: 10.2337/db12-1543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ohta H, Yamazaki S, McMahon DG. Constant light desynchronizes mammalian clock neurons. Nature Neuroscience. 2005;8:267–269. doi: 10.1038/nn1395. [DOI] [PubMed] [Google Scholar]
- 18.Acheson KJ, Ravussin E, Wahren J, Jequier E. Thermic effect of glucose in man. Obligatory and facultative thermogenesis. J Clin Invest. 1984;74:1572–1580. doi: 10.1172/JCI111573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Clench J, Reinberg A, Dziewanowska Z, Ghata J, Smolensky M. Circadian changes in the bioavailability and effects of indomethacin in healthy subjects. Eur J Clin Pharmacol. 1981;20:359–369. doi: 10.1007/BF00615406. [DOI] [PubMed] [Google Scholar]
- 20.Hoogerwerf WA. Role of clock genes in gastrointestinal motility. Am J Physiol Gastrointest Liver Physiol. 2010;299:G549–555. doi: 10.1152/ajpgi.00147.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hoogerwerf WA, Hellmich HL, Cornelissen G, Halberg F, Shahinian VB, Bostwick J, et al. Clock gene expression in the murine gastrointestinal tract: endogenous rhythmicity and effects of a feeding regimen. Gastroenterology. 2007;133:1250–1260. doi: 10.1053/j.gastro.2007.07.009. [DOI] [PubMed] [Google Scholar]
- 22.Schneeberger D, Tappy L, Temler E, Jeanpretre N, Jequier E. Effects of muscarinic blockade on the thermic effect of oral or intravenous carbohydrate. Eur J Appl Physiol Occup Physiol. 1991;63:242–249. doi: 10.1007/BF00233855. [DOI] [PubMed] [Google Scholar]
- 23.Flatt JP. The biochemistry of energy expenditure. In: Bray G, editor. Recent Advances in Obesity Research II: Proceedings of the Second International Congress on Obesity. Newman Publishing Ltd; London: 1978. pp. 211–228. [Google Scholar]
- 24.Ravussin E, Acheson KJ, Vernet O, Danforth E, Jequier E. Evidence that insulin resistance is responsible for the decreased thermic effect of glucose in human obesity. J Clin Invest. 1985;76:1268–1273. doi: 10.1172/JCI112083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ravussin E, Bogardus C, Schwartz RS, Robbins DC, Wolfe RR, Horton ES, et al. Thermic effect of infused glucose and insulin in man. Decreased response with increased insulin resistance in obesity and noninsulin-dependent diabetes mellitus. J Clin Invest. 1983;72:893–902. doi: 10.1172/JCI111060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Segal KR, Albu J, Chun A, Edano A, Legaspi B, Pi-Sunyer FX. Independent effects of obesity and insulin resistance on postprandial thermogenesis in men. J Clin Invest. 1992;89:824–833. doi: 10.1172/JCI115661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Van Cauter E, Shapiro ET, Tillil H, Polonsky KS. Circadian modulation of glucose and insulin responses to meals: relationship to cortisol rhythm. The American journal of physiology. 1992;262:E467–E475. doi: 10.1152/ajpendo.1992.262.4.E467. [DOI] [PubMed] [Google Scholar]
- 28.Frank SA, Roland DC, Sturis J, Byrne MM, Refetoff S, Polonsky KS, et al. Effects of aging on glucose regulation during wakefulness and sleep. American Journal of Physiology. 1995;269:E1006–E1016. doi: 10.1152/ajpendo.1995.269.6.E1006. [DOI] [PubMed] [Google Scholar]
- 29.Scheer FA, Hu K, Evoniuk H, Kelly EE, Malhotra A, Hilton MF, et al. Impact of the human circadian system, exercise, and their interaction on cardiovascular function. Proc Natl Acad Sci USA. 2010;107:20541–20546. doi: 10.1073/pnas.1006749107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.McHill AW, Melanson EL, Higgins J, Connick E, Moehlman TM, Stothard ER, et al. Impact of circadian misalignment on energy metabolism during simulated nightshift work. Proceedings of the National Academy of Sciences. 2014;111:17302–17307. doi: 10.1073/pnas.1412021111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Roden M, Koller M, Pirich K, Vierhapper H, Walhauser F. The circadian melatonin and cortisol secretion pattern in permanent night shift workers. American Journal of Physiology. 1993;34:R261–R267. doi: 10.1152/ajpregu.1993.265.1.R261. [DOI] [PubMed] [Google Scholar]


