Abstract
Hypothalamic neurons expressing agouti-related peptide (AgRP) regulate eating and glucose metabolism. Ablation of FOXO1 in AgRP neurons of mice results in reduced food intake, leanness, improved glucose homeostasis, and increased sensitivity to insulin and leptin. We tentatively identified G-protein–coupled receptor Gpr17 as an effector of FOXO1 orexigenic signals in AgRP neurons. In this study, we generated and characterized AgRP neuron–specific Gpr17 knockout mice (Agrp-Gpr17−/−) to test the hypothesis that Gpr17 regulates appetite, energy expenditure, and metabolism. Agrp-Gpr17−/− mice show reduced food intake, increased relative energy expenditure, and increased satiety, resulting in leanness and reduced body fat. They also show increased central nervous system sensitivity to insulin and leptin and reduced plasma glucose excursions following the administration of glucose or pyruvate. In summary, AgRP neuron–specific Gpr17 knockouts phenocopy FOXO1 knockouts in the same cell type, thus supporting our original hypothesis and providing further impetus to develop Gpr17 antagonists for the treatment of obesity.
Introduction
Obesity and diabetes affect an increasingly large swath of the population of the world (1). Finding novel and effective biochemical pathways to reverse the pathophysiology of these conditions is an important goal of biomedical research. The central nervous system (CNS) plays an integral role in maintaining glucose homeostasis and energy balance (2). It exerts systemic effects by sensing and integrating hormonal and metabolite cues to regulate nutrient turnover and storage through neural efferents and neuroendocrine mechanisms (3). A variety of effector neurons, such as agouti-related peptide (AgRP) and proopiomelanocortin (POMC) neurons, has been identified as potential targets for treatment of metabolic disease (4,5). Identifying biochemical pathways in these neurons that are amenable to pharmacological manipulation holds promise for developing novel therapies.
AgRP/neuropeptide Y (Npy)–expressing neurons promote feeding and regulate glucose metabolism (6–8). AgRP neurons express insulin and leptin receptors, through which they regulate energy balance and peripheral glucose homeostasis (9). FOXO1 is a shared element in insulin and leptin signaling in the CNS (10) and functions in regulating food intake and energy efficiency (10–13). In healthy and disease states, a decrease in food intake is associated with decreased energy expenditure or increased energy efficiency (14). This tight linkage between energy intake and energy expenditure is thought to be the primary cause of recidivism of obesity after weight loss (15) and is mediated in part by FOXO1 activation in neuropeptidergic cells of the arcuate nucleus of the hypothalamus (ARH). In fact, mice lacking FOXO1 in either POMC or AgRP neurons have a favorable metabolic profile, which is characterized by leanness and increased sensitivity to insulin and leptin. In POMC neuron–specific FOXO1 knockouts (KOs) (12), we have proposed that the uncoupling of food intake from energy expenditure is mediated by carboxypeptidase E, a prohormone-converting enzyme required for the generation of α-melanocyte–stimulating hormone (α-MSH). In AgRP neurons, we proposed that the orphan G-protein–coupled receptor Gpr17 mediates the metabolic effects of FOXO1 (13).
Gpr17 has been deorphanized as a dual receptor for purinergic and cysteinyl-leukotriene ligands (16). However, this view has been disputed (17), and its endogenous ligands remain unclear (18). We hypothesized that ablating Gpr17 or reducing its expression in AgRP neurons will produce the beneficial effects on energy balance and glucose homeostasis. To test this hypothesis, we generated conditional KOs as well as inducible knockdowns of Gpr17 in AgRP neurons. We report that constitutive or induced Gpr17 loss of function results in reduced feeding, increased relative energy expenditure, increased CNS sensitivity to insulin and leptin, and improved glucose tolerance. Agrp-Gpr17−/− phenocopy Agrp-Foxo1−/− mice, thus supporting our original contention that Gpr17 mediates key aspects of FOXO1 function in this cell type.
Research Design and Methods
Experimental Animals
C57BL/6 mice were from The Jackson Laboratory. Embryonic stem cells with a conditional allele of Gpr17 obtained from the KOMP Program at The Jackson Laboratory were used to generate Gpr17loxP/loxP mice in the Columbia University transgenic mouse core facility. Agrp-ires-Cre transgenic mice have been described previously (19). The Columbia University Animal Care and Utilization Committee approved all procedures. Normal chow diet had 62.1% of calories from carbohydrates, 24.6% from protein, and 13.2% from fat (PicoLab Rodent Diet 20, catalog #5053; Purina Mills). We measured body composition by nuclear magnetic resonance (Bruker Optics). The results of genotyping with primers are presented in Supplementary Fig. 1. We excluded from analyses Agrp-Gpr17−/− mice that showed somatic recombination owing to stochastic embryonic expression of Agrp-ires-Cre. Therefore, Agrp-Gpr17−/− mice in these studies have Gpr17 ablated specifically in AgRP neurons, but not in non-AgRP neurons or elsewhere. We used male 16-week-old mice for metabolic experiments, with the exception of leptin sensitivity tests. We used bilateral cannula implanted in the skull for adenoviral injection. Adenovirus (1 × 1012 viral particles/mL) was injected into the ARH bilaterally via a cannula and injector in a volume of 0.5 μL/side.
Metabolic Analyses
We measured food intake as described, using a TSE Labmaster Platform (TSE Systems) for indirect calorimetry and activity measurements (20). For refeeding experiments, we killed mice and collected blood for hormone measurements 4 h after refeeding, unless otherwise noted. For measurements of food intake during the 24-h refeeding time course, we removed food at lights out on the previous day, refed mice the next morning, and recorded refeeding for the following 24 h. Recording starts at the time of refeeding. For fasting experiments, unless noted in the figure legend, “short fasting” means fasting for 5–6 h during the light phase; overnight fasting was performed by removing food before the dark phase (∼15 h). We examined leptin sensitivity in 18-week-old, weight-matched male mice. Mice were habituated to hopper feeding first and then to twice-daily PBS injection for 3 days. Leptin (1 mg/kg body wt) was injected intraperitoneally twice daily for 3 days at 7:30 a.m. and 6:30 p.m. We used ELISA for leptin and insulin measurements (Millipore) and colorimetric assays for plasma free fatty acid measurements (Wako). GTT was performed as described previously (20) using 2 g/kg glucose by intraperitoneal injection after overnight fast. For the pyruvate tolerance test (PTT), mice were fasted for 6 h during the daytime followed by intraperitoneal injection of sodium pyruvate (2 g/kg). Blood glucose concentrations were measured at the indicated time points using a glucometer (OneTouch Ultra; LifeScan). We subcutaneously implanted an IPTT-300 transponder (Bio Medic Data Systems) to collect body temperature data. For the cold challenge experiment, mice were swiftly switched from ambient room temperature to a cold housing cabinet (10°C). Mouse body temperature was collected at the time interval indicated.
Immunostaining
We processed mouse brains and cut 10-μm-thick coronal sections for immunohistochemistry as described previously (13), using phospho (p)-Akt (catalog #4060; Cell Signaling Technology), pStat3 (catalog #9131; Cell Signaling Technology), protein S6 (pS6) antibodies (catalog #4858; Cell Signaling Technology), and cFos antibody (catalog #2250; Cell Signaling Technology).
RNA and Protein Procedures
We extracted RNA with Trizol (Invitrogen) and reverse transcribed with Superscript II reverse transcriptase. We performed quantitative PCR using primers spanning introns. Primer sequences are available upon request. We used a radioimmunoprecipitation assay buffer to extract protein and loaded an equal amount of protein for gel electrophoresis. Western blotting (with antibody information) and quantification have been described previously (21).
Statistical Analyses
We analyzed data with a t test or one-way or two-way ANOVA using GraphPad Prism software for statistical analyses and graphics generation. Statistical tests are specified in the corresponding figure legend.
Results
Generation of AgRP-Specific Gpr17 Knockout Mice
To investigate the metabolic function of Gpr17 in AgRP neurons, we generated a mutant Gpr17 allele using homologous recombination to introduce loxP sequences around the Gpr17 coding sequence in exon 2 (Supplementary Fig. 1A). We performed Southern blots to verify the correct integration of the 5′ and 3′ arms in the genomic DNA (Supplementary Fig. 1B). In addition, we verified the genomic insertion of the loxP sequences by PCR (Supplementary Fig. 1C). To ablate Gpr17 in AgRP neurons, we crossed Gpr17lox/lox with Agrp-ires-Cre mice. Cre-mediated deletion of the loxP-flanked Gpr17 DNA resulted in null Gpr17 alleles in AgRP neurons (hereafter called Agrp-Gpr17−/− mice).
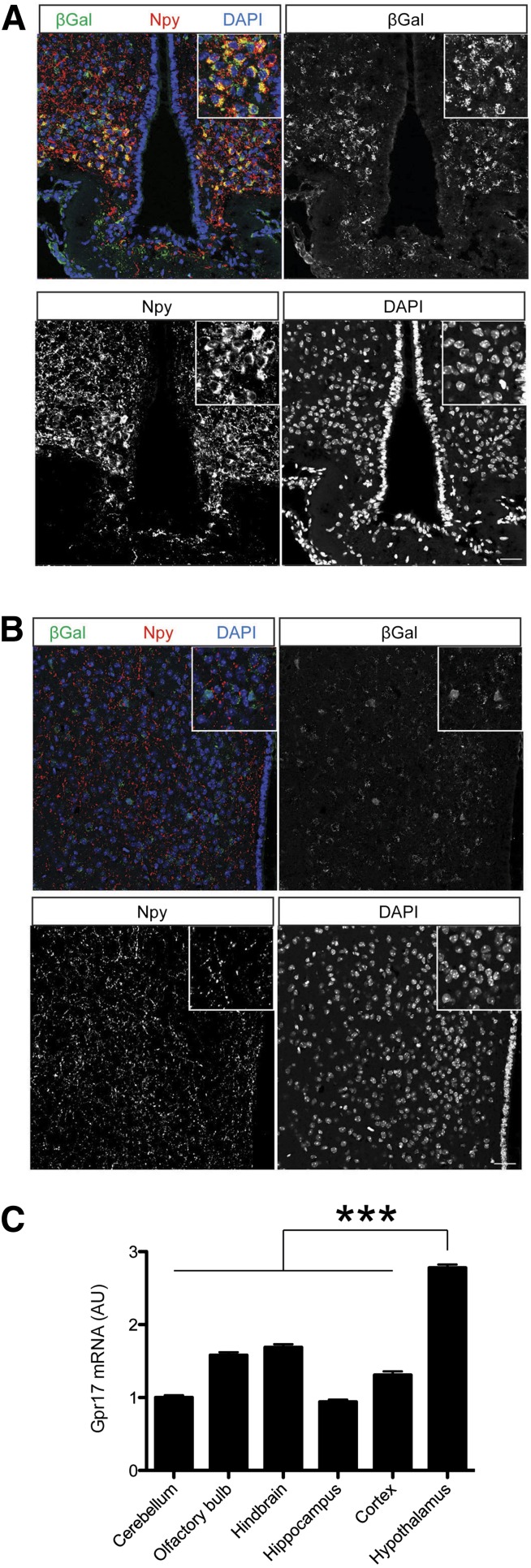
First, we sought independent confirmation of Gpr17 localization in the CNS. To this end, we introduced a reporter cDNA encoding β-galactosidase (β-Gal) after the first (noncoding) exon of Gpr17 (Supplementary Fig. 1A). AgRP neurons express Npy. Therefore, immunostaining with Npy antibody labels AgRP neuron soma as well as processes in the ARH. We performed double-labeling immunohistochemistry to detect β-Gal and Npy expression in the hypothalamus. We found high levels of β-Gal expression in the mediobasal hypothalamus. Confocal microscopy demonstrated that Npy neuron soma colocalized with β-Gal staining (Fig. 1A) in the ARH. This is in agreement with our previous report that Gpr17 is highly expressed in AgRP neurons (13). We also detected β-Gal expression outside the arcuate nucleus, in agreement with previous reports of widespread Gpr17 localization in the CNS (22,23). In the dorsomedial hypothalamus, β-Gal expression showed no colocalization with Npy-labeled processes (Fig. 1B). To corroborate these findings, we measured Gpr17 mRNA in various parts of the brains of C57BL6/J mice and found that the highest levels of Gpr17 are indeed in the hypothalamus (Fig. 1C).
Figure 1.
Colocalization of Gpr17-β-Gal and Npy/AgRP neurons. Immunohistochemistry with β-Gal (green) and Npy (red) of ARH (A) and dorsomedial hypothalamus (B) in mice bearing a Gpr17-β-Gal knockin allele, used as a reporter to localize Gpr17-expressing neurons. We used DAPI (blue) to counterstain nuclei. Scale bar: 50 μm. C: Gpr17 mRNA expression is enriched in the hypothalamus (n = 5 each). We present data as the mean ± SEM. ***P < 0.001 (one-way ANOVA with Tukey post-test). AU, arbitrary units.
Agrp-Gpr17−/− Mice Are Leaner Than Controls
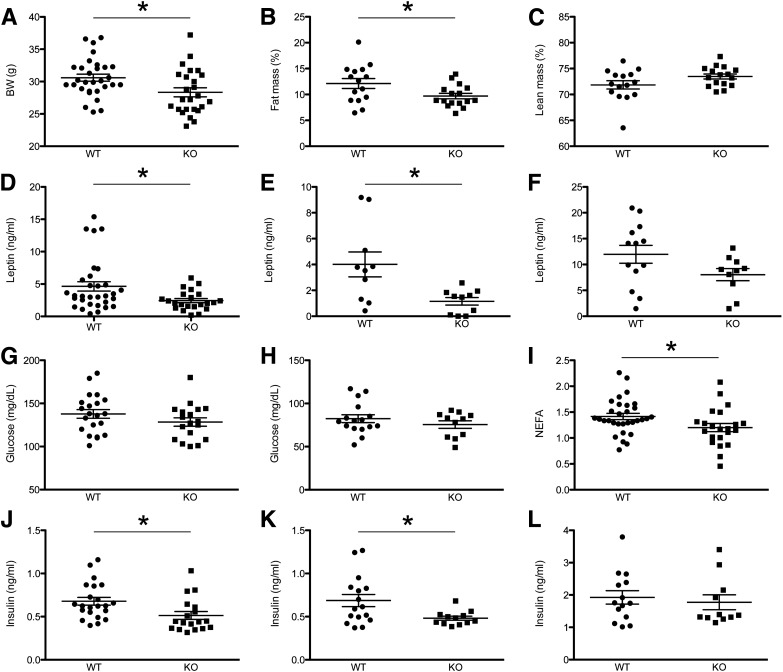
Body weight of male Agrp-Gpr17−/− mice showed a modest, yet statistically significant, decrease compared with wild-type (WT) controls (Fig. 2A). Moreover, male Agrp-Gpr17−/− mice showed changes in body composition, with decreased fat mass (Fig. 2B) and a trend toward an increase in lean mass (Fig. 2C). Plasma leptin levels correlate with adiposity and energy balance and represent an indicator of overall leptin sensitivity. For this reason, we measured leptin under ad libitum (Fig. 2D), fasting (Fig. 2E), and refed conditions (Fig. 2F). Leptin levels were lower in Agrp-Gpr17−/− mice under all conditions, which is consistent with decreased adiposity and is suggestive of improved energy balance. There were no differences in glucose levels between Agrp-Gpr17−/− and WT animals during ad libitum feeding (Fig. 2G) and fasting (Fig. 2H), despite a modest trend toward a decrease in Agrp-Gpr17−/−. The main source of circulating free fatty acids is adipocyte lipolysis, which is inhibited by insulin. Interestingly, free fatty acids were lower in ad libitum–fed Agrp-Gpr17−/− than WT animals (Fig. 2I). We measured insulin under various feeding conditions and found that Agrp-Gpr17−/− animals have significantly lower insulin after a short fast (Fig. 2J) or an overnight fast (Fig. 2K), but not after refeeding (Fig. 2L). In sum, these data show that male Agrp-Gpr17−/− mice have decreased body weight and adiposity and are more sensitive to insulin and leptin.
Figure 2.
Metabolic features of Agrp-Gpr17−/− mice. A: Body weight (BW) of adult male WT and KO mice on chow diet (n = 25–30 for each genotype). The y-axis is limited to the range 20–40 g to better visualize individual differences. Body fat (B) and lean mass (C) in male mice analyzed by MRI (n = 15–16 for each genotype). In C, the y-axis is limited to the range 60–85% to better visualize individual differences. Serum leptin in ad libitum–fed (D), fasted (E), and refed (F) mice (n = 10–16 for each genotype). Serum glucose level after a short fast (G) (n = 18–21 for each genotype) or after overnight fasting (H) (n = 11–16 for each genotype). I: Serum fatty acids in ad libitum–fed mice (n = 22–31 for each genotype). Serum insulin after a short fast (J) (n = 18–22 for each genotype), an overnight fast (n = 12–16 for each genotype) (K), or refeeding (L) (n = 11–14 for each genotype). We present data as the mean ± SEM. *P < 0.05 (unpaired t test). NEFA, nonesterified fatty acid.
Agrp-Gpr17−/− Mice Eat Less but Have Increased Relative Energy Expenditure
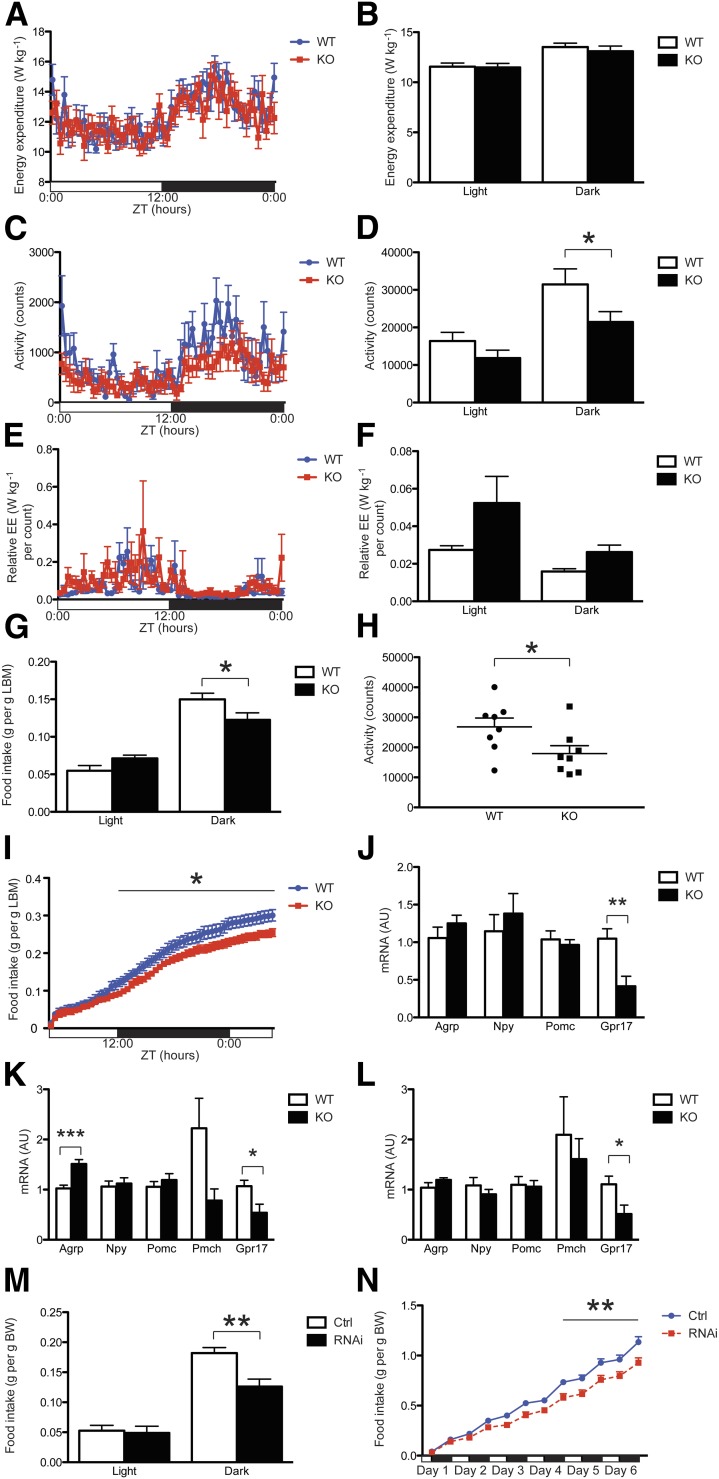
Next, we measured respiratory exchanges, energy expenditure, and food intake in adult male mice that were matched for body weight and composition. When fed ad libitum, Agrp-Gpr17−/− mice were comparable to WT controls with regard to VO2 and respiratory quotient (Supplementary Fig. 2A and B). Knockout mice showed the same energy expenditure (Fig. 3A and B) but reduced locomotor activity during the dark phase of the 24-h light-dark cycle (Fig. 3C and D). Thus, relative energy expenditure, defined as the ratio of locomotor activity to total energy expenditure, was increased in Agrp-Gpr17−/− mice throughout the light cycle (Fig. 3E and F). Food intake was also reduced during the dark phase of the 24-h light cycle (Fig. 3G).
Figure 3.
Altered feeding and energy balance in Agrp-Gpr17−/− mice. Energy expenditure (A and B), locomotor activity (counts/15 min [C] and total counts during 12-h light or dark phase [D]), and relative energy expenditure (E and F) in mice during the light and dark phases of the light cycle (n = 8 for each genotype). G: Food intake during the light and dark phases of the light cycle (n = 8 for each genotype). H: Locomotor activity during overnight fasting (n = 8 for each genotype). I: Rebound food intake (g/g lean body mass) after an overnight fast (n = 8 for each genotype). J: Neuropeptide mRNA expression in the ARH of ad libitum–fed mice after an overnight fast (n = 7 for each genotype). mRNA levels in basal hypothalamus after an overnight fast (K) (n = 10–12) or refeeding (L) (n = 8–9 for each genotype). Food intake during the light and dark phases 4 days after viral injection (M), and the time course of cumulative food intake (g/weight/24 h) (N) after viral injection (n = 5 for each type of viral injection). In panels A, C, E, and I, black bars represent the dark phase, and white bars represent the light phase. Scale: hours. We present data as the mean ± SEM. *P < 0.05, **P < 0.01, ***P < 0.001 (two-way ANOVA with Bonferroni post-tests or unpaired t test). Act, locomotor activity; AU, arbitrary units; BW, body weight; Ctrl, control; EE, energy expenditure; LBM, lean body mass; RNAi, RNA interference; W, weight; ZT, Zeitgeber time.
To determine whether reduced food intake was due to increased satiety, we examined mice during fasting and refeeding. Male Agrp-Gpr17−/− mice showed significantly reduced locomotor activity during an overnight fast, suggesting reduced food-foraging behavior (Fig. 3H). Moreover, they showed significantly reduced rebound food intake after an overnight fast (Fig. 3I). Both findings are consistent with the conclusion that they have increased satiety.
Next, we profiled orexigenic and anorexigenic neuropeptide expression using quantitative RT-PCR in ARH biopsy samples from mice fed ad libitum (Fig. 3J). Agrp, Npy, and Pomc levels were comparable in samples from WT and KO mice, while Gpr17 showed a significant decrease in KO samples. We further analyzed hypothalamic samples from overnight-fasted (Fig. 3K) and refed (Fig. 3L) mice. Of note, Agrp showed a modest, statistically significant increase in KO samples after fasting, which is consistent with a possible compensation to defend energy balance. These data indicate that Gpr17 acts on satiety and energy homeostasis through mechanisms that are largely independent of neuropeptide expression.
Cre-Mediated Gpr17 Knockdown in AgRP Neurons of Adult Mice
Yet, these changes may reflect developmental effects on AgRP neurons from the genetic KO. To address this possibility, we performed a Gpr17 knockdown in adult animals using adenovirus-mediated, Cre-dependent activation of RNA interference expression. Mice bearing Agrp-ires-Cre were injected intracerebroventricularly either with control adenovirus expressing Cre-dependent green fluorescence protein (Fig. 3M) or with adenovirus expressing Gpr17 small interfering RNA (siRNA) after Cre-mediated recombination. The specificity and efficiency of this approach are demonstrated by the Cre-dependent green fluorescence protein expression in AgRP neurons (Supplementary Fig. 2C). We further validated the siRNA-dependent Gpr17 knockdown by immunohistochemistry (Supplementary Fig. 2D). Prior to the injection, both groups of mice showed similar food intake (Supplementary Fig. 2E). Four days after the injection, the RNA interference group significantly decreased food intake during the dark phase (Fig. 3M). The cumulative food intake of animals that received the Gpr17 siRNA was significantly lower compared with that of controls (Fig. 3N). The average food intake per day of the siRNA group after injection was also lower compared with that of controls (Supplementary Fig. 2F).
Gpr17 Ablation Increases Leptin Sensitivity
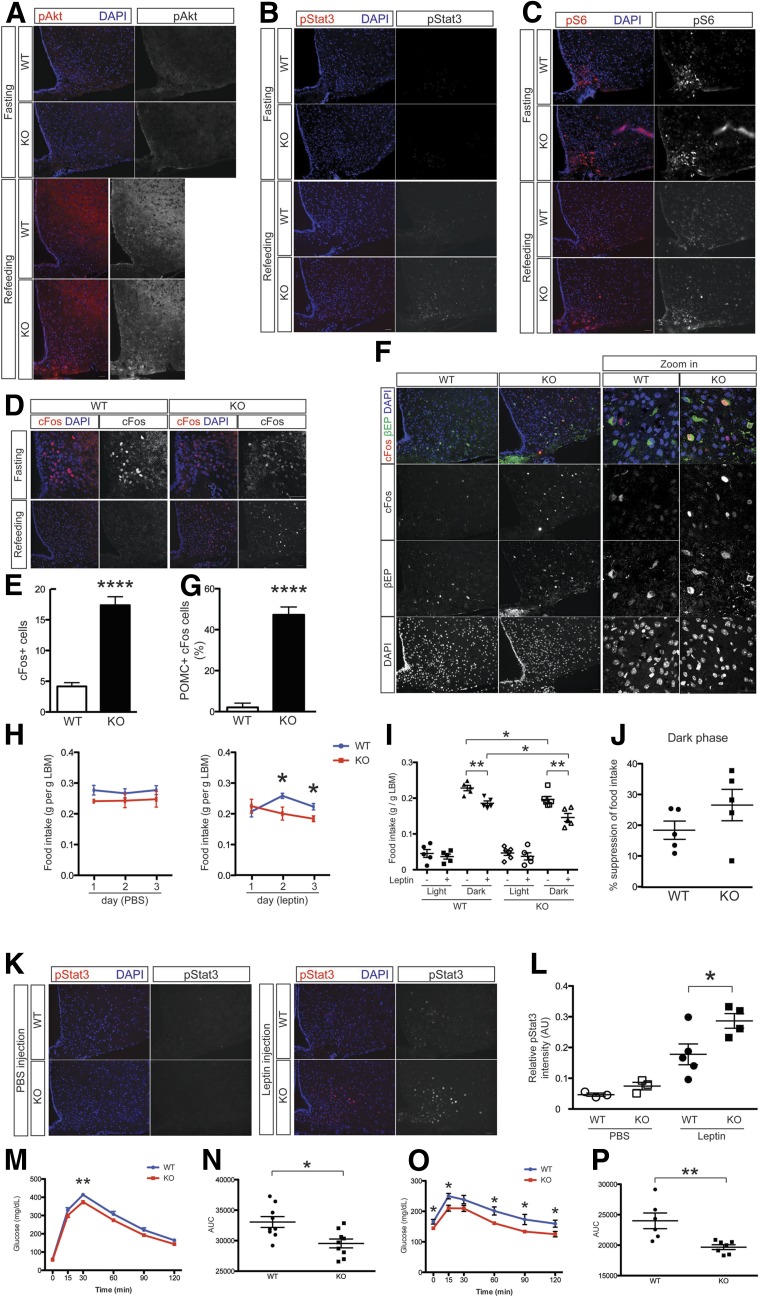
Agrp-Gpr17−/− mice have improved energy homeostasis, resulting from an unusual combination of decreased food intake and increased relative energy expenditure that uncouples these two variables of the energy expenditure equation (24). Insulin and leptin inhibit food intake (25,26). We postulated that this is because of the increased sensitivity of AgRP neurons to hormones and nutrients. To test this possibility, we examined hormone- and nutrient-induced signaling by a fasting-refeeding experiment. We obtained hypothalamic samples from WT and Agrp-Gpr17−/− mice killed after an overnight fast or after a 4-h refeeding. During fasting, due to the low insulin and leptin levels, pAkt and pStat3 staining in the ARH is low (Fig. 4A and B, top panels). After refeeding (Fig. 4A and B, bottom panels), despite decreased serum insulin and leptin levels (Fig. 2), we detected increased pAkt and pStat3 staining in the hypothalamus of KO mice, especially in the medial basal part of the ARH with a localization consistent with AgRP neurons.
Figure 4.
CNS signaling and peripheral metabolism in mutant mice. Representative images of pAkt (A), pStat3 (B), and pS6 (C) immunohistochemistry in the ARH of mice fasted overnight or refed for 4 h. D: Representative confocal images of hypothalamic cFos immunohistochemistry in mice fasted overnight or refed for 4 h. Scale bar: 50 μm. E: Quantified cFos-positive cell number per hypothalamic hemisphere in refed WT and KO mice (n = 8–12). F: Immunohistochemistry with βEP (green) and cFos (red) of ARH with representative areas magnified by confocal microscopy. Scale bar: 50 μm. G: Quantification of the percentage of POMC-positive cFos cells (n = 8–12). H: Normalized, 24-h food intake in 18-week-old, body weight–matched male mice after intraperitoneal PBS or leptin injection (n = 5). I: Food intake in WT and KO mice during the light and dark phases after PBS or leptin injection (n = 5). J: Quantification of the percentage of the suppression of 24-h food intake by leptin in WT and KO mice (n = 5). K: Representative images of pStat3 immunohistochemistry in the ARHs of mice in response to PBS or leptin injection. L: Quantified pStat3 immunofluorescence shown in K (n = 3–5). Time course (M) and AUC (N) of glucose tolerance test results (n = 9 for each genotype). Time course (O) and AUC (P) of PTTs performed after a short fast (n = 6–7 for each genotype). In H and J, the range of the y-axis is limited to better visualize individual differences. We present data as the mean ± SEM. *P < 0.05, **P < 0.01, ****P < 0.0001 (two-way ANOVA with Bonferroni post-tests or unpaired t test). AU, arbitrary units; LBM, lean body mass.
Amino acid sensing by mammalian target of rapamycin (mTOR) promotes satiety (27). We monitored mTOR activity by immunohistochemistry with antibodies against its substrate, ribosomal pS6, in fasted and refed mice. During fasting, samples from WT and KO mice show comparable amounts of pS6 staining in the ARH (Fig. 4C, top panels). After refeeding, Agrp-Gpr17−/− mice showed stronger pS6 staining in the mediobasal ARH (Fig. 4C, bottom panels), consistent with the localization of AgRP neurons. Therefore, knocking out Gpr17 in AgRP neurons renders mice more sensitive to leptin, insulin, and amino acid signaling in the ARH, potentially providing an explanation for their increased satiety.
AgRP neurons are activated during fasting to promote food intake and reduce energy expenditure. In contrast, POMC neurons, expressing anorexigenic neuropeptide α-MSH, are activated during refeeding to functionally antagonize the physiological activity of AgRP neurons (28). Fasting and refeeding induce distinct temporal activation of different neuron populations in the ARH (29). We used cFos staining as a marker of neuronal activation. As demonstrated in Fig. 4D, during fasting (Fig. 4D, top panels), we detected cFos in the mediobasal part of the ARH, immediately adjacent to the third ventricle, representing orexigenic AgRP neurons. In contrast, during refeeding (Fig. 4D, bottom panels) we detected cFos in other areas of the ARH. Confocal microscopy images demonstrate that the intensity of cFos staining is lower in the ARH of conditional KO mice during fasting but is higher in other neuronal populations after refeeding. The reduced AgRP neuron activation during fasting is consistent with the reduced feeding of KO mice.
We noticed more cFos staining in the hypothalamic samples from KO mice, especially the ARH, after refeeding (Fig. 4D, bottom panels). We quantified the results and found that KO mice have substantially more cFos-positive cells in the ARH (Fig. 4E), suggesting more activation of anorexigenic neurons after refeeding. POMC neurons are activated in response to refeeding to produce precursor polypeptide POMC, the cleavage products of which include α-MSH, ACTH, and β-endorphin (βEP). Using double labeling with βEP and cFos antibodies to quantify the percentage of POMC neurons among refeeding-activated neurons (Fig. 4F), we saw that nearly 50% of cFos-positive neurons are POMC neurons in the KO hypothalamus sample (Fig. 4G). In contrast, a much lower percentage is detected in WT samples. These data suggest that the activation of POMC neurons is increased in Agrp-Gpr17−/− mice, providing an additional mechanism to explain their metabolic phenotype.
As Agrp-Gpr17−/− mice are more sensitive to hormones and nutrients (Fig. 4A–C), we assessed the contribution of this increased sensitivity to the feeding phenotype. To address this question, we measured the leptin response by challenging mice with a submaximal dose of leptin. Leptin, but not PBS, curtailed food intake more markedly in Agrp-Gpr17−/− mice than in WT mice (Fig. 4H). Moreover, leptin effectively reduced food intake in both groups of animals during the dark phase of the 24 h light cycle (Fig. 4I), but the reduction of food intake tended to be more marked in KO mice (Fig. 4J). To confirm this finding, we examined Stat3 activation in the ARH (Fig. 4K) and found increased pStat3 immunoreactivity in Agrp-Gpr17−/− mice after leptin injection (Fig. 4L). Therefore, we conclude that Agrp-Gpr17−/− mice are more sensitive to hormone and nutrient signaling, which could contribute to their energy balance phenotype.
Peripheral Metabolism in Agrp-Gpr17−/− Mice
AgRP neurons and POMC neurons are integral components of the CNS melanocortin signaling pathway. POMC neurons produce α-MSH that activates melanocortin 4 receptor, while AgRP neurons release Agrp, which antagonizes α-MSH. Central melanocortin also regulates thermogenesis in brown adipose tissue (30). Specifically, the intracerebroventricular injection of α-MSH increases sympathetic activity and brown adipose tissue temperature, while injection of Agrp has the opposite effect (31). To test the effect of Gpr17 in AgRP neurons on thermogenesis, we measured the body temperatures of WT and Agrp-Gpr17−/− mice (Supplementary Fig. 3). Ad libitum–fed WT and KO mice showed comparable body temperatures (Supplementary Fig. 3A). Fasting and refeeding caused similar changes in the body temperatures of WT and Agrp-Gpr17−/− mice (Supplementary Fig. 3B). However, during a cold challenge in which mice were kept at 10°C, KO mice maintained a higher body temperature for a longer time (Supplementary Fig. 3C). A potential explanation of these data is that Agrp-Gpr17−/− mice support increased thermogenesis, possibly through increased release of free fatty acids from adipocytes. This phenotype is also reminiscent of findings in AgRP-specific Foxo1 KOs (13).
AgRP neurons also regulate hepatic glucose production through mechanisms that are dependent on insulin receptor and FOXO1 signaling (13,32). We wished to test whether this effect could also be ascribed to Gpr17 signaling. To that end, we subjected overnight-fasted mice to intraperitoneal glucose tolerance tests. Agrp-Gpr17−/− mice showed lower plasma glucose excursions after a bolus glucose injection that resulted in a statistically significant decrease of the glucose area under the curve (AUC) (Fig. 4M and N). Second, we used PTT results as an indirect measure of hepatic gluconeogenesis after a short fast. Agrp-Gpr17−/− mice showed significantly lower glucose excursions after the PTT, with a decrease of the AUC (Fig. 4O and P). We conclude that Agrp-Gpr17−/− mice have improved glucose metabolism and, likely, decreased hepatic glucose production.
We measured the mRNA expression of critical genes involved in gluconeogenic and lipogenic pathways (Supplementary Fig. 4). Most genes showed no difference between WT and KO mice (Supplementary Fig. 4A), but we found increased expression of genes required for lipid use during fasting (i.e., Mttp) and for lipid synthesis after refeeding (i.e., Pklr, Acc1, Fas, and Elovl6) (Supplementary Fig. 4B–F). Hepatic triglyceride content was comparable between WT and KO mice (Supplementary Fig. 4G).
To test whether changes to hepatic glucose metabolism are cell autonomous or nonautonomous, we isolated primary hepatocytes from WT and KO mice and assessed insulin signaling by Western blotting with antibodies to phosphorylated (activated) components of the insulin signaling pathway (Supplementary Fig. 4H). Hepatocytes isolated from KO mice show increased basal and reduced insulin-dependent phosphorylation of mTOR (Supplementary Fig. 4I). Quantification of the data showed that Akt (Ser and Thr) and GSK are phosphorylated to the same extent (Supplementary Fig. 4J–L). We also performed glucose production assays in WT and KO hepatocytes and found similar induction by cAMP and similar suppression by insulin (Supplementary Fig. 4M and N). These data indicate that Gpr17 ablation in AgRP neurons affects hepatic glucose metabolism in a cell-nonautonomous manner (i.e., Gpr17 ablation in AgRP neurons alters AgRP neuronal activity, which in turn may regulate hepatic glucose production via vagal innervation).
Discussion
The goal of this work was to test the hypothesis, as set forth in a previous study (13), that orphan receptor Gpr17 mediates orexigenic and metabolic signals in AgRP neurons. We found that constitutive germline ablation or induced knockdown of Gpr17 in AgRP neurons of adult animals reduces eating without increasing energy efficiency and results in a lean, metabolically fit phenotype. This unusual dissociation of energy intake from energy expenditure phenocopies observations in AgRP-specific FOXO1 KOs (13), lending further support to the notion that Gpr17 mediates some of the effects of this transcription factor.
Gpr17 was deorphanized by sequence homology with purinergic and cysteinyl-leukotriene receptors (16). However, whether these ligands activate Gpr17 in AgRP neurons remains unclear (18). This is consistent with our prior observation (13) that purinergic agonists, when used alone, do not affect eating. With the development of new Gpr17-specific agonists that are devoid of purinergic activity (33), we are now poised to interrogate the mechanism of action of this interesting receptor in more detail. We do not know whether there exists an endogenous ligand for Gpr17. However, it is interesting that Gpr17 expression is regulated at the transcriptional level (13), suggesting that the receptor itself might be a sensor of nutritional status, rather than its putative ligands. It could be envisioned, for example, that Gpr17 signals by interacting with insulin, leptin, or melanocortin receptors.
In fact, Gpr17 expression is not restricted to neurons or to the CNS (16,17,22,23). This had led to some confusion about its overall function. For example, in the CNS Gpr17 overexpression has been linked to myelination (22) as well as to the response to ischemic damage (23). The underlying theme appears to be that Gpr17 conveys a stress response that is then translated into a cell type–specific event. In this regard, fasting could be viewed as the “stress” that activates AgRP neurons, wherein Gpr17 mediated an eating response.
Gpr17 is present in other cell types outside the CNS, including endocrine cells in the gastrointestinal system (H.R., D.A., unpublished observations). The heterogeneity of Gpr17 expression and function might explain the failure to find a food intake or glucose phenotype reported by another group (34) in global Gpr17 KOs. While this difference may depend on the gene-targeting approach, genetic background, or environmental factors (husbandry, diet, microbiota, experimental design, and calorimetry systems), we think that this is unlikely, based on the fact that in our hands a global Gpr17 KO also causes a distinct metabolic phenotype, which is characterized by a modest reduction of food intake and a substantial increase in energy expenditure, as well as resistance to the effects of high-fat feeding to impair melanocortin signaling (H.R., D.A., unpublished observations). These differences between tissue-specific conditional KOs and whole-body KOs are not unprecedented (35), and certainly will not deter us from further studies of this interesting receptor. For example, global KOs of Agrp or Npy have few, if any, metabolic effects, and yet few would doubt that these are important orexigenic signals (8,36). Based on our findings, we propose that Gpr17-mediated regulation of feeding is independent of neuropeptide-mediated mechanisms. Interestingly, AgRP-specific Gpr17 KOs respond to fasting with a modest yet significant increase of Agrp expression in the ARH, which is consistent with a compensatory neuropeptide response. Also consistent with an adaptive compensatory response is the observed stronger activation of anorexigenic POMC neurons in response to refeeding after Gpr17 ablation in AgRP neurons.
While the effects of Gpr17 ablation in AgRP neurons on body weight and food intake are modest, it should be remembered that the weight loss required to obtain lasting medical benefits regarding avoidance of the major comorbidities of obesity is also modest (1). Moreover, it is important to note that the reduction in food intake in Gpr17 KO mice is dissociated from the reduction in energy expenditure that commonly causes weight regain (14). The dissociation of food intake from energy expenditure is indeed the recurring theme of FOXO CNS function and may offer a safer and more reliable alternative to maintaining long-term energy homeostasis.
In sum, we remain committed to the idea that the antagonism of Gpr17 can be leveraged to therapeutic ends. In fact, the dual effect of Gpr17 ablation to increase both insulin and leptin sensitivity would place a putative Gpr17 antagonist in a drug class of its own to promote metabolic fitness.
Supplementary Material
Article Information
Acknowledgments. The authors thank Dr. Lin of the Columbia University Transgenic Core Facility for technical help. The authors also thank Ana Flete-Castro and Travis J. Morgenstern for excellent technical help. In addition, the authors thank the members of the Accili laboratory for critical discussion of the data.
Funding. H.R. is the recipient of a mentor-based postdoctoral fellowship from the American Diabetes Association and the Naomi Berrie Diabetes Research Fellowship. This work is partly supported by National Institutes of Health-National Institute of Diabetes and Digestive and Kidney Diseases grants K99-DK-098294 (H.R.), 5R01-DK-057539 (D.A.), and DK-63608 (Columbia University Diabetes Research Center).
Duality of Interest. This work is also supported by an unrestricted grant from Merck Research Laboratories. No other potential conflicts of interest relevant to this article were reported.
Author Contributions. H.R. designed and conducted the experiments, analyzed the data, and wrote the manuscript. J.R.C. conducted the experiments and analyzed the data. N.K. analyzed embryonic stem cell clones by Southern blotting. D.A. designed the experiments, analyzed the data, and wrote the manuscript. D.A. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Footnotes
This article contains Supplementary Data online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db15-0390/-/DC1.
References
- 1.Wang YC, McPherson K, Marsh T, Gortmaker SL, Brown M. Health and economic burden of the projected obesity trends in the USA and the UK. Lancet 2011;378:815–825 [DOI] [PubMed] [Google Scholar]
- 2.Kaiyala KJ, Schwartz MW. Toward a more complete (and less controversial) understanding of energy expenditure and its role in obesity pathogenesis. Diabetes 2011;60:17–23 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Morton GJ, Cummings DE, Baskin DG, Barsh GS, Schwartz MW. Central nervous system control of food intake and body weight. Nature 2006;443:289–295 [DOI] [PubMed] [Google Scholar]
- 4.Dietrich MO, Horvath TL. Hypothalamic control of energy balance: insights into the role of synaptic plasticity. Trends Neurosci 2013;36:65–73 [DOI] [PubMed] [Google Scholar]
- 5.Adan RA. Mechanisms underlying current and future anti-obesity drugs. Trends Neurosci 2013;36:133–140 [DOI] [PubMed] [Google Scholar]
- 6.Aponte Y, Atasoy D, Sternson SM. AGRP neurons are sufficient to orchestrate feeding behavior rapidly and without training. Nat Neurosci 2011;14:351–355 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Krashes MJ, Koda S, Ye C, et al. Rapid, reversible activation of AgRP neurons drives feeding behavior in mice. J Clin Invest 2011;121:1424–1428 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Luquet S, Perez FA, Hnasko TS, Palmiter RD. NPY/AgRP neurons are essential for feeding in adult mice but can be ablated in neonates. Science 2005;310:683–685 [DOI] [PubMed] [Google Scholar]
- 9.Varela L, Horvath TL. Leptin and insulin pathways in POMC and AgRP neurons that modulate energy balance and glucose homeostasis. EMBO Rep 2012;13:1079–1086 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kim KW, Donato J Jr, Berglund ED, et al. FOXO1 in the ventromedial hypothalamus regulates energy balance. J Clin Invest 2012;122:2578–2589 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kitamura T, Feng Y, Kitamura YI, et al. Forkhead protein FoxO1 mediates Agrp-dependent effects of leptin on food intake. Nat Med 2006;12:534–540 [DOI] [PubMed] [Google Scholar]
- 12.Plum L, Lin HV, Dutia R, et al. The obesity susceptibility gene Cpe links FoxO1 signaling in hypothalamic pro-opiomelanocortin neurons with regulation of food intake. Nat Med 2009;15:1195–1201 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ren H, Orozco IJ, Su Y, et al. FoxO1 target Gpr17 activates AgRP neurons to regulate food intake. Cell 2012;149:1314–1326 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Rosenbaum M, Goldsmith R, Bloomfield D, et al. Low-dose leptin reverses skeletal muscle, autonomic, and neuroendocrine adaptations to maintenance of reduced weight. J Clin Invest 2005;115:3579–3586 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Schwartz MW, Woods SC, Seeley RJ, Barsh GS, Baskin DG, Leibel RL. Is the energy homeostasis system inherently biased toward weight gain? Diabetes 2003;52:232–238 [DOI] [PubMed] [Google Scholar]
- 16.Ciana P, Fumagalli M, Trincavelli ML, et al. The orphan receptor GPR17 identified as a new dual uracil nucleotides/cysteinyl-leukotrienes receptor. EMBO J 2006;25:4615–4627 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Qi AD, Harden TK, Nicholas RA. Is GPR17 a P2Y/leukotriene receptor? Examination of uracil nucleotides, nucleotide sugars, and cysteinyl leukotrienes as agonists of GPR17. J Pharmacol Exp Ther 2013;347:38–46 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bäck M, Powell WS, Dahlén SE, et al. Update on leukotriene, lipoxin and oxoeicosanoid receptors: IUPHAR Review 7. Br J Pharmacol 2014;171:3551–3574 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tong Q, Ye CP, Jones JE, Elmquist JK, Lowell BB. Synaptic release of GABA by AgRP neurons is required for normal regulation of energy balance. Nat Neurosci 2008;11:998–1000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Banks AS, Kon N, Knight C, et al. SirT1 gain of function increases energy efficiency and prevents diabetes in mice. Cell Metab 2008;8:333–341 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Cook JR, Matsumoto M, Banks AS, Kitamura T, Tsuchiya K, Accili D. A mutant allele encoding DNA binding-deficient FoxO1 differentially regulates hepatic glucose and lipid metabolism. Diabetes 2015;64:1951–1965 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Chen Y, Wu H, Wang S, et al. The oligodendrocyte-specific G protein-coupled receptor GPR17 is a cell-intrinsic timer of myelination. Nat Neurosci 2009;12:1398–1406 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lecca D, Trincavelli ML, Gelosa P, et al. The recently identified P2Y-like receptor GPR17 is a sensor of brain damage and a new target for brain repair. PLoS One 2008;3:e3579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Leibel RL, Rosenbaum M, Hirsch J. Changes in energy expenditure resulting from altered body weight. N Engl J Med 1995;332:621–628 [DOI] [PubMed] [Google Scholar]
- 25.Halaas JL, Gajiwala KS, Maffei M, et al. Weight-reducing effects of the plasma protein encoded by the obese gene. Science 1995;269:543–546 [DOI] [PubMed] [Google Scholar]
- 26.Woods SC, Lotter EC, McKay LD, Porte D Jr. Chronic intracerebroventricular infusion of insulin reduces food intake and body weight of baboons. Nature 1979;282:503–505 [DOI] [PubMed] [Google Scholar]
- 27.Cota D, Proulx K, Smith KA, et al. Hypothalamic mTOR signaling regulates food intake. Science 2006;312:927–930 [DOI] [PubMed] [Google Scholar]
- 28.Guyenet SJ, Schwartz MW. Clinical review: regulation of food intake, energy balance, and body fat mass: implications for the pathogenesis and treatment of obesity. J Clin Endocrinol Metab 2012;97:745–755 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wu Q, Lemus MB, Stark R, et al. The temporal pattern of cfos activation in hypothalamic, cortical, and brainstem nuclei in response to fasting and refeeding in male mice. Endocrinology 2014;155:840–853 [DOI] [PubMed] [Google Scholar]
- 30.Voss-Andreae A, Murphy JG, Ellacott KL, et al. Role of the central melanocortin circuitry in adaptive thermogenesis of brown adipose tissue. Endocrinology 2007;148:1550–1560 [DOI] [PubMed] [Google Scholar]
- 31.Yasuda T, Masaki T, Kakuma T, Yoshimatsu H. Hypothalamic melanocortin system regulates sympathetic nerve activity in brown adipose tissue. Exp Biol Med (Maywood) 2004;229:235–239 [DOI] [PubMed] [Google Scholar]
- 32.Könner AC, Janoschek R, Plum L, et al. Insulin action in AgRP-expressing neurons is required for suppression of hepatic glucose production. Cell Metab 2007;5:438–449 [DOI] [PubMed] [Google Scholar]
- 33.Hennen S, Wang H, Peters L, et al. Decoding signaling and function of the orphan G protein-coupled receptor GPR17 with a small-molecule agonist. Sci Signal 2013;6:ra93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Mastaitis J, Min S, Elvert R, et al. GPR17 gene disruption does not alter food intake or glucose homeostasis in mice. Proc Natl Acad Sci USA 2015;112:1845–1849 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Accili D. A note of caution on the Knockout Mouse Project. Nat Genet 2004;36:1132. [DOI] [PubMed] [Google Scholar]
- 36.Qian S, Chen H, Weingarth D, et al. Neither agouti-related protein nor neuropeptide Y is critically required for the regulation of energy homeostasis in mice. Mol Cell Biol 2002;22:5027–5035 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.