Abstract
Demographic changes are associated with a steady increase of older patients with end-stage organ failure in need for transplantation. As a result, the majority of transplant recipients are currently older >50 years and organs from elderly donors are more frequently utilized. Nevertheless, the benefit of transplantation in older patients is well recognized whereas the most frequent causes of death among older recipients are potentially linked to side effects of their immunosuppressants.
Immunosenescence is a physiological part of aging linked to higher rates of diabetes, bacterial infections and malignancies representing the major causes of death in older patients. These age-related changes impact older transplant candidates and may have significant implications for an age-adapted immunosuppression. For instance, immunosenescence is linked to lower rates of acute rejections in older recipients while the engraftment of older organs has been associated with higher rejection rates. Moreover, new-onset diabetes mellitus following transplantation is more frequent in the elderly, potentially related to corticosteroids, calcineurin inhibitors and mTOR inhibitors.
This review presents current knowledge for an age-adapted immunosuppression based on both, experimental and clinical studies in and beyond transplantation. Recommendations of maintenance and induction therapy may help to improve graft function and to design future clinical trials in the elderly.
Introduction
Increasing numbers of elderly patients with irreversible end organ damage are currently on the waitlist for organ transplantation. Indeed, the majority of transplant recipients and organ donors are >50 years, mainly as a consequence of demographic changes.1–3
The most frequent causes of death in older transplant recipients are linked to immunosuppressive therapies. At the same time, aging aspects are in general not integrated into clinical immunosuppressive trials. Bacterial infections and malignancies are more frequent in the elderly.4,5 Moreover, rates of pre-transplant diabetes mellitus (PDM) and new-onset diabetes mellitus after transplantation (NODAT) are increasing with age. Of note, the use immunosuppressive drugs has been shown to induce hyperglycemia and diabetes, both linked to inferior transplant outcomes, higher rates of acute rejections and infections. Hence, older transplant recipients are more likely to suffer from adverse drug effects of their immunosuppression as reflected by higher rates of diabetes and de novo malignancies. Finally, older recipients are dying more frequently due to bacterial infections compared to younger transplant recipients and those patients remaining on the waitlist.6
In addition, compromised functional capacities of older livers are impacting first pass metabolism and consecutive blood concentrations of administered drugs. A recent prospective study demonstrated a twofold increase in serum troughs levels of calcineurin inhibitors (CNI) in older kidney transplant recipients (65–84 years) compared to young controls, even when adjusted for weight and dose.7 Aging is not only shaping drug metabolism but also impacting immune responses. In a large-scale study, we have recently shown that acute rejection rates decline in parallel to recipient age, a correlation which has also been confirmed for liver and heart transplant recipients.8–10 Thus, the selection of the immunosuppressive drug regime in the elderly is complex and not supported by broad clinical evidence thus far, but rather by few anecdotal observations. Here, we will highlight the critical importance of aging for immunosuppressive therapies and dissect the current literature of experimental science and clinical trials considering the aged patient.
Infections and malignancies in transplant recipients
Major infections in transplant recipients are caused by bacteria and viruses. Of note, bacterial infection rates increase in older transplant recipients5 while viral infections are decreasing with advanced age.11 The individual mortality risk caused by bacterial infections is multi-factorial and relies on several contributing factors such as donor and recipient demographics, incidence of diabetes and advanced age.12 For instance, more than 20% of kidney transplant recipients (60-69 years) are dying due to severe infections. The incidence of bacterial infections with septic shock is twofold increased in graft recipients >50 years.13 In contrast, a comprehensive database analysis of >60,000 renal transplant recipients revealed that the incidence for active viral infection with varicella zoster is decreasing dramatically with advanced age.14 Patients <18 years showed an infection rate of 14% while patients >65 years presented an infection rate of less than 4%. When analyzing the serostatus, the median age of kidney transplant recipients being seropositive for cytomegalovirus and Epstein-Barr virus disease is significantly higher.15 Taken together, the prevalence of seropositivity is increasing with age while the rate of active viral infection is decreasing. However, active viral infections in older patients are associated with inferior outcomes. The incidence of invasive fungal infection is in general very low in organ transplantation with a paucity of data from age-matched studies. In detail, Candida spp. and Aspergillus spp. count for most of the fungal infections16 and might be more frequent in the elderly.17,18
The incidence of cancer is known to be steadily increasing with age, reaching its highest numbers in graft recipients >50 years.19 Skin related cancers and lymphoproliferative disorders are the most common malignancies among transplant recipients. In addition, de novo malignancies are one of the major causes of death, e.g. accounting for one-third of non-hepatic deaths of liver transplant recipients.4
Diabetes mellitus in transplant recipients
The relevance of metabolic disorders increases with aging. Indeed, NODAT is more frequent in the elderly and linked to the immunosuppressive drug regime applied. For instance, the risk of NODAT after kidney transplantation increases 1.5 fold throughout every decade of life.20 The presence of NODAT is associated with increased rates of acute rejections, infections, late cardiovascular events and inferior outcomes. Of note, frequencies of NODAT have been reported with a range of 4 – 25% in renal transplant recipients, 2 – 25% in liver transplant recipients and 4 – 40% in heart transplant recipients.21 PDM is increasing with age as well. The rate of PDM increased consecutively from 7% (18-29 age) to 31% (60-69 age) in a study of >12,000 liver transplant recipients.22
Overall, the likelihood of PDM and NODAT is increased in the elderly and of significant clinical relevance as diabetes is linked to higher rates of graft failure and inferior outcomes.
Immunology of aging
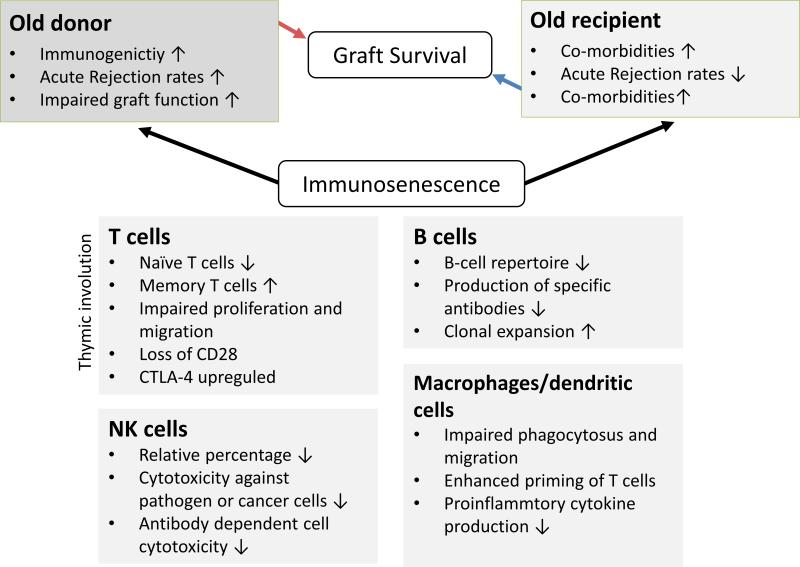
Immunosenescence is characterized by an impaired function of both, adaptive and innate immunity (Figure 1), clinically evident by a compromised response to vaccination and an augmented risk of malignancies in the elderly, mostly linked to a compromised tumour surveillance.23,24 Thymic involution appears to play a critical role for compromised adaptive immune responses, although precise mechanisms remain unclear. Differentiation and emigration of T-cells, as well as the total number of naïve T-cells decline with age.25 Hence, T-cell diversity is compromised and the ability to recognize and eliminate pathogens declines remarkably in the elderly.26 While the overall number of naïve T-cells decreases due to an involuted thymus, a shift towards CD8+ and CD4+ cells and a significant increase in memory and effector T-cells is observed in the elderly.27,28 T-cell senescence is characterized by dysregulated immune functions accompanied with a loss of the co-stimulatory molecule, CD28, shorter telomeres and altered production of pro-inflammatory cytokines.29,30 Of note, CTLA4 is upregulated, particularly on conventional and regulatory T-cells.31 Clearly, the aging immune system may not only require less, but also specific immunosuppression as senescence affects some immune compartments more than others.
Figure 1.
Aging effects graft outcome. Immunosenescence is characterized by changes in both, innate and adaptive immunity. Elderly organ transplant recipients have less acute rejections while the engraftment of old organs is linked to higher rejection rates associated to an augmented immunogenicity. Thus, both donor and recipient age need to be considered for a tailored immunosuppression.
Humoral immune responses are also impacted by aging as evidenced by a decline in the number of progenitor B-cells.32 Moreover, T- and B-cell interactions are affected as CD4+ T-cells, required for B-cell activation, show functional defects in the elderly.33 Humoral immune responses in the elderly are also characterized by a contracted B-cell repertoire. Clinically, it remains unclear if sensitization is age-dependent and if humoral rejections require a different therapeutic approach in the elderly.
In addition to changes of adaptive and humoral immune responses, the innate immune compartment undergoes age-associated changes. While some antigen presenting cells including macrophages express less MHC class II,34 older dendritic cells (DC) appear to have a more pronounced capacity to present antigens. In our own experimental study, we were able to show that organs containing older DCs elicit a more potent IL-17 driven immune response in young recipient animals.
Acute rejections in older recipients
While rejection rates appear less frequent in older recipients, the engraftment of older organs is linked to a higher frequency of acute rejections, potentially caused by an augmented immunogenicity and compromised repair mechanisms.8–10,35 Of note, acute rejections are most frequent when older organs are transplanted into younger recipients while this effect appears blunted when older organs are transplanted into older recipients.8,36 The Eurotransplant Senior Program (ESP) is applying principles of immunosenescence by allocating kidneys from old donors (≥65 years) to old recipients (≥65 years) while keeping ischemic times brief through a loco-regional organ allocation.37 Clearly, immunosuppressive regimens must consider both, donor and recipient age.
Immunosuppression in elderly transplant recipients
In general, aging is associated with impaired organ function and impaired homeostasis affecting liberation, absorption, distribution, metabolism and excretion of immunosuppressants (Table 1).38–40 In contrast, drugs approved for clinical use are mostly not studied in elderly patients who frequently have complex co-morbidities while receiving multi-drug treatment.41 Recognizing complex medical conditions and addressing this issue, the Food and Drug Administration (FDA) has endorsed clinical trials in the elderly.41,42 Of additional clinical relevance, if included into clinical studies, elderly patients had been picked selectively, thus not necessarily representing a general age-matched population.43 A recent meta-analysis revealed that kidney transplant recipients participating in clinical trials in the US were significantly younger.44 Older age as an exclusion criterion was evident in 30% of studies. Obviously, clinical trials in transplant medicine do not consider the clinical reality of a steadily increasing volume of older transplant recipients.
Table 1.
Pharmacokinetic changes in the elderly.
| Pharmacokinetics | Changes in the elderly | Impact on immunosuppression |
|---|---|---|
| Absorption | • GI motility↓ • Splanchnic blood flow↓ • Gastric emptying↓ • Gastric pH Level↑ • Surface area of the small intestine↓ |
• Cmax ↓ and Tmax ↑, delayed drug activity • Dissolvent↓, reduced absorption |
| Distribution | • Lean body mass↓ • Body water↓ • Relative body fat↑ • P-gp expression/activity↓↑ |
• Lipophilic drugs have lower trough levels but longer half-life (e.g. CNI, mTOR inhibitors) • Hydrophilic drugs have a smaller distribution and higher troughs • Changes in absorption in intestine |
| Metabolism | • Serum protein concentration↓ • Liver volume↓ • Liver blood flow↓ • Expression of CYP450 enzyme (liver)↓ |
• Changes in drug clearance and fraction of free drug • Reduced first-pass metabolism and hepatic clearance |
| Excretion | • Renal function ↓ • Biliary excretion ↓ |
• Impaired renal clearance • Impaired hepatic clearance |
Peak of drug concentration (Cmax); time of Cmax (Tmax); p-glycoprotein (P-gp); gastrointestinal (GI); cytochrome P450 (CYP450)
Calcineurin inhibitors
In a recent prospective study that included >2,500 patients, tacrolimus and cyclosporine trough levels were 50% higher in older kidney transplant recipients (≥ 65 years) when normalized for dose and weight,7 linked to an altered first-pass metabolism of CNIs, facilitated by intestinal and hepatic enzymes. The metabolism of tacrolimus is mediated almost exclusively by the cytochrome P450 (CYP450) 3A subfamily in the liver and partly via the CYP3A subfamily and p-glycoprotein (P-gp) in the intestinal mucosa impacting both, oral bioavailability and clearance.45–47 Noticeably, clinical liver samples have shown an 8% decline of CYP3A with every decade of life.48 These findings are also supported by experimental data showing a remarkable age-related decline (up to 70%) of CYP3A activity and expression in the liver of old rats.49 Likewise, cyclosporine is mainly metabolized by CYP3A4 and to a minor extent by CYP3A5.50 Individuals expressing CYP3A5*1 have an approximately twofold higher clearance and need higher doses of CNIs to achieve comparable trough levels, 7,51 but probably not in patients >60 years old.52 CYP3A5 is predominantly expressed by Caucasians but also present in other ethnic populations.53
Expression and activity of P-gp is important for the metabolism of CNIs, however, age-related changes are discussed controversially and their influence on CNIs remain unclear.54,55 The transmembrane protein P-gp, encoded by the ABCB1 gene, is also present in lymphocytes. Cyclosporine has been shown to be both a substrate and inhibitor of P-gp, while tacrolimus inhibits P-gp.54 A study of cyclosporine in the elderly (>65 years) demonstrated higher cyclosporine concentrations within T-cells.56 Moreover, CNI elimination was compromised with increasing age.
In liver transplant recipients, more than 80% of tacrolimus binds to erythrocytes with smaller amounts binding to plasma proteins (approx. 16%) and leukocytes (approx. 0.6%).57 This unbalanced distribution is the result of high concentrations of FKBP-12 in erythrocytes attracting tacrolimus. Interestingly, a study in renal transplant recipients correlated haematocrit with whole blood concentrations of tacrolimus.58 Indeed, haematocrit levels predicted the variability in tacrolimus whole blood concentrations but did not impact the unbound and therapeutically active fraction of tacrolimus. Thus, a higher haematocrit binds more tacrolimus and may overestimate the therapeutic dose of tacrolimus, while a low haematocrit may lead to an underestimation of the dosage. Those effects are of clinical relevance as haematocrit levels change with age, especially in the elderly and renal transplant recipients.
The pharmacokinetics of the CNIs in the elderly population may also be negatively impacted by the formulation of these agents (i.e., brand versus generic). In 2015, the only generic tacrolimus study specifically evaluating generic tacrolimus in the elderly was published.59 This prospective, single-center, randomized, crossover trial evaluated the pharmacokinetic parameters of a generic tacrolimus (Tacni; Teva Pharmaceutical Industries, Petah Tikva, Israel) in 25 renal transplant recipients older than 60 years. Patients were randomized to receive either brand or generic tacrolimus at the time of transplantation. Pharmacokinetic parameters were assessed six and eight weeks after transplantation. After the first assessment, the two groups crossed over; both the AUC (90% confidence interval = 1.10 – 1.23) and Cmax (90% confidence interval = 1.35 – 1.65) of generic tacrolimus did not meet bioequivalence standards. The authors urged caution in using this particular generic formulation in the elderly population due to the possibility for the higher drug exposure potentially increasing the risk of adverse events.
The diabetogenic effects of CNIs are linked to an impaired insulin secretion negatively regulating pancreatic beta-cell growth and function.60 Several studies demonstrated higher incidences of NODAT in renal transplant recipients using tacrolimus compared to controls on cyclosporine.61,62 Thus, tacrolimus treatment is associated with an increased risk for the development of hyperglycaemia and diabetes and cyclosporine might be favourable in patients in a pre-diabetic stage or with existing diabetes mellitus. However, long term follow up of graft recipients maintained on tacrolimus showed superior graft function compared to patients on cyclosporine.63,64 Thus, the higher incidence of diabetes resulting from tacrolimus therapy might be blunted by an improved long-term patient survival and graft function.
T-cell aging may also affect pharmacodynamics. In an experimental study, calcineurin phosphatase activity had declined by more than 50% in old T-cells.65 Moreover, IL-2 production of older T-cells had been impaired with aging while inducible nuclear factor of activated T-cells (NFAT) had been reduced or absent in the elderly.66 Thus, while the impact of T-cell immunosenescence is recognized, CNI applications and dosages require more detailed assessment. Moreover, as significant portions of tacrolimus bind to plasma proteins; age-related changes of hepatic proteins will impact the distribution of the drug.67
mTOR inhibitors
Anti-tumour capacities of mammalian target of rapamycin (mTOR) inhibitors seem of particular relevance in the design of immunosuppressive regimens in the elderly. In fact, de novo post-transplant malignancies were significant lower with sirolimus/everolimus compared to CNI-based maintenance immunosuppression in a multivariate analysis of 30,000 primary renal transplant recipients.68 Additional prospective trials in non-renal transplant recipients have been confirmatory.69
The oral bioavailability of both, sirolimus and everolimus is low (<15%), related to their gastrointestinal and hepatic metabolism. Both drugs are metabolized by CYP450 (e.g. CYP3A4 and CYP3A5) and transported by P-gp. Aging per-se may alter both, enzymatic metabolization of mTOR inhibitors and the capacity of biliary transporters in the liver.70 Hence, age-related functional impairment of liver metabolism is linked to a compromised clearance of mTOR inhibitors that may require reduced dosages.71,72
The serine-threonine kinase mTOR plays a pivotal role in the insulin cascade and inhibitors can alter glucose metabolism, insulin secretion and sensitivity. Consequently, the risk to develop NODAT is increased in the transplant recipient under mTOR treatment. Numerous studies indicated an incidence of NODAT of 15–30%, while the exact mechanism remain vague.73 Moreover, the diabetogenic effect is enhanced by the combination mTOR inhibitors and CNIs.74
Antimetabolites
With a high percentage of mycophenolic acid (MPA) binding reversibly to serum albumin, age-related changes of albumin concentrations gain importance as only the unbound MPA is pharmacological active. Low levels of albumin and impaired renal function are associated with an increased clearance of total MPA. During aging, levels of albumin decline, linked to liver impairment.75,76 For instance, liver transplant recipients with low serum albumin (35g/L) required twofold higher mycophenolate mofetil (MMF) doses than patients with normal albumin levels due to an increase clearance of free drug.77 Those results were confirmed in a meta-analysis of more than 450 renal transplant patients.78 MPA clearance decreases with higher albumin levels due to a smaller fraction of free unbound MPA. In conclusion, albumin levels and changes of renal function necessitate regular measurements of MPA trough levels and dose adaptations in particular in the elderly.
MPA is administered as the pro-drug MMF or enteric-coated mycophenolate sodium (ECMPS). Those aspects might be of age-related clinical relevance as higher ph-levels are present in the proximal GI tract. MMF is hydrolysed to MPA in the GI tract, blood, liver and tissues while ECMPS, the salt of MPA is not getting hydrolysed. Following administration of MMF, higher ph-levels reduce peak concentrations (Cmax) and the AUCs in healthy volunteers diminished significantly.79 In contrast, Cmax and AUC were not impacted by the acid-resistant ECMPS formulation.80
Corticosteroids
Prednisolone and prednisone are primarily metabolized in the liver and, to a smaller degree, in the kidneys.81 Adult liver transplant recipients have shown a broad intra- and interindividual variability in medication pharmacokinetics.82 At the same time, hepatic impairment has shown conflicting data on prednisolone and prednisone metabolism in non-transplant patients.83 Interestingly, CYP3A4 inhibitors decrease the clearance and increase the bioavailability of prednisolone and methylprednisolone.84,85 At the same time, high- doses of steroids were able to increase P-gp and CYP3A concentrations in liver and intestine of rats leading to a decline in tacrolimus concentrations.86 In the plasma, prednisolone binds mainly to albumin, transcortin and partly to α1-acid glycoprotein. Pharmacokinetic characteristics are furthermore complicated by a dose-dependent non-linear plasma protein binding. When higher doses are applied, prednisolone protein binding capacity decreases from 95% to 60-70%.87 Compromised hepatic function in older patients is linked to changes in plasma protein levels, thus affecting plasma protein binding of glucocorticoids. Nevertheless clinical trials in the elderly are lacking.
The clearance of prednisolone88 and methylprednisolone89 declines in the elderly linked to an increased exposure and an augmented adrenal suppression. A decreased elimination of prednisolone is caused by a compromised renal clearance.83 The clearance of lipophilic corticosteroids is furthermore determined by body composition. As body weight is linked to aging, with a peak during the 5th and 6th decade of life and a decline thereafter,87 dose adjustment of corticosteroids should also be based on body weight.
Furthermore, glucocorticoids are also associated with a higher risk of developing NODAT in a dose-dependent manner. A dose of 0.01 mg/kg/day has been associated with a 5% risk for the development of NODAT.90 In contrast, steroid withdrawal has been shown to result in improved insulin sensitivity. A dose reduction of one-third resulted in 24% increase of insulin sensitivity index.91
Belatacept
The receptor fusion protein belatacept is composed of the modified Fc domain of the human immunoglobulin IgG1 linked to the extracellular-binding domain CTLA4. Belatacept has shown a low variability (<30%) of pharmacokinetic parameters from phase I, II, and III trials. Drug exposure was not significantly affected by age or age related parameters such as renal function, albumin level (hepatic function) or diabetes.92
Differential effects of DC aging might influence effects of belatacept. Although, the expression of CD80/CD86, the binding site of belatacept, appears age-independent, the ability of DCs to phagocytize antigens and to migrate to the antigen site declines with age.93 Moreover, CTLA4 is upregulated on old CD4+ T-cells.94 The altered capacity of DCs to present antigen and the enhanced expression of CTLA4 might increase the suppressive mechanism of belatacept in the elderly. Nevertheless, clinical trials that elucidate age-related mechanism of belatacept are lacking.
Of note, a comprehensive meta-analysis of randomized controlled trials showed a better metabolic profile of belatacept with lower incidences of NODAT, hypertension and lower serum lipids levels compared to treatment with CNI.95 This is of particular interest as diabetes is a common co-morbidity in the elderly associated with an increasing risk for infections, acute rejections and graft failure.
Antibody-based Therapies
The pharmacokinetics of antibodies are complex, as the tissue distribution is slow and the volume of distribution is low. Antibodies are metabolized to peptides and amino acids and can be recycled for de novo protein synthesis or used as an energy resource.
Antibodies are mainly catabolized through two major pathways. Nonspecific clearance is mediated by the interaction between the Fc region of the antibody and the Fc receptor. Alternatively, the Fab region of the antibody is binding specifically to its antigenic target. The specific clearance can be saturable linked to the amount of antigens; the nonspecific clearance, in contrast has large capacities. Subsequent to internalization antibodies into the cytoplasm, they are degraded by lysosomes.96,97 Interestingly, aging is linked to impaired macrophage polarization, making the elderly more susceptible to infections while slowing metabolism of monoclonal antibodies.98 In addition, the neonatal Fc receptor for IgG protects from degradation, thus explaining the long elimination half-life of antibodies (e.g. basiliximab is 7.2 days).99 Possible changes in antibody pharmacokinetics seen with aging remain unknown, but may impact half-life and change their exposure.
Biomarkers and the development of new immunosuppressive protocols
Shortcomings of current immunosuppressive drug therapies in the elderly are based on the complexity of aging and the use of trough levels as current “gold standard” to monitor immunosuppressive therapy. However, monitoring blood concentrations might not appropriately reflect effects of immunosenescence or age-related compromised organ function. Therefore, the diagnostic use of biomarkers may be a helpful tool to adjust drug therapies for age-specific changes. A number of promising candidates is available to serve as pharmacodynamic, pharmacogenetic, or immunological markers.100
Several pharmacodynamic assays are currently available assessing enzyme activities that may help to detect inter-individual and age-dependent differences in pharmacokinetics (Table 2). Assessment of calcineurin phosphatase activity and its downstream product NFAT could serve as a diagnostic tools to assess the intracellular effectivity of CNIs in the elderly.101 Along the same lines, the mTOR-dependent kinase p70S6102 and the inosinmonophosphate dehydrogenase for MPA103 can be utilized to assess the effectivity of corresponding target enzymes. Detailed assessment of T-cell subtypes and longitudinal tracking of T-cell depletion could help to avoid over-immunosuppression.104 Moreover, thymic function prior to transplantation is correlated to the rate of post-transplant malignancies. Thus, assessment of rearrangement excision circles (TREC) may be a helpful in predicting post-transplant malignancies subsequent to ATG treatment.105
Table 2.
The use of biomarkers can overcome age-related changes in pharmacokinetics and immunosenescence that may not be sufficiently recognized when using trough levels for drug monitoring.
In addition, several soluble biomarkers detectable in urine and blood have been evaluated to monitor functional immune responses after transplantation.106,107 A meta-analysis concluded that the soluble CD30 marker showed only poor accuracy to predict acute rejection rates in the context of renal transplantation.108 Another approach to assess cell-mediated immune responses is the measurement of intracellular adenosine-tri-phosphate production (iATP). Interestingly, individual levels of iATP correlated with rates of infections and cellular rejections in a meta-analysis of >500 solid organ transplant recipients,109 independently of drug trough levels.110 Taken together, additional information about the individual immune response and monitoring the activity of the target enzymes may be of future relevance to adjust and maintain accurate levels of immunosuppression in the elderly.
Age adapted immunosuppressive protocols
Experiences with immunosuppressive protocols in the elderly are limited as clinical trials have in general excluded elderly recipients or recipients of marginal organs, thus warranting prospective randomized immunosuppressive trials.
Induction therapy
Randomized trials and meta-analyses have clearly demonstrated the superiority of induction therapy compared to conventional maintenance immunosuppression alone in the general renal transplant population. For example, the Kidney Disease: Improving Global Outcomes (KDIGO) guidelines for kidney transplantation recommend IL2-RA as first line induction therapy. However, age-specific recommendations for organ transplantation are missing.111
A large retrospective registry analysis has evaluated effects of induction therapies in >14,000 patients >60 years.112 Interestingly, patients with higher immunological risk profile (peak panel reactive antibodies >20%, prior transplants or black race) receiving high-risk donors (expanded criteria donors [ECD], donation after cardiac death [DCD] or prolonged ischemic time >24h) showed higher rates of rejections when treated with interleukin-2 receptor antibodies (IL2-RA) as compared to treatment with rabbit antithymocyte globulin (rATG). Of note, acute rejections in low risk recipients that received low-risk organs (non ECD, non DCD, living donor, short ischemic time <24h) were comparable after either IL2-RA or rATG induction. Thus, elderly high-risk recipients that receive high-risk donors (peak panel reactive antibodies >20% or prior transplant or black race) and possibly low -risk recipients with high-risk donors may benefit from an induction treatment with rATG, potentially with a dose reduction. A retrospective study evaluating age-dependent risk profiles linked to rATG therapy in elderly renal transplant recipients (>65 years) found no differences for death-censored graft survival, graft function, rejection rates, infections, malignancies and hematologic adverse reactions when compared to non-elderly patients.113 Of note, another retrospective analysis of >300 older renal transplant recipients (≥60 years) treated with reduced cumulative rATG doses showed comparable renal graft function but lower rates of rejection when compared to younger patients (<60 years).114
These results are in part supported by a prospective multicentre trial non-adjusted for age in renal transplant recipients that had received deceased donor kidney transplants with high immunological risk.115 Patients with high risks for acute rejection or delayed graft function treated with rATG (1.5 mg/kg on day 0; 1; 4) showed a lower incidence and less severity of acute rejections when compared to those that had received an induction treatment with basiliximab (20mg on day 0 and 4). Both groups had similar incidences of delayed graft function, serious adverse events, cancer and death. However, rATG treated patients had more frequent infections. A Cochrane analysis of >10,000 kidney transplant recipients, not adjusted for age, evaluated the use of IL2-RA as induction therapy.116 While acute rejection rates had been comparable in patients that received IL2-RA or rATG, less cytomegalovirus infections and malignancies were observed when IL2-RA was used. Taken together, rATG might be favourable in patients with high immunological risk due to a reduced lower incidence of acute rejections while IL2-RA seems to be superior in patients with a low immunological risk profile.
The use of alemtuzumab for induction therapy was associated with a lower rates of NODAT in a large-scale clinical study of renal transplant recipients.61 Moreover, a randomised trial in kidney transplant recipients showed that an induction therapy with alemtuzumab followed by reduced CNI and MMF exposure had been superior to a standard basiliximab based treatment (basiliximab followed by standard-dose tacrolimus, MPA and prednisolone).117 However, a retrospective analysis showed that these superior effects might be blunted in older renal transplant recipients (>60 years) as reflected by a higher risk of acute rejection, graft loss and death.112 These findings were not consistent in a stratified analysis and the risk of acute rejections had been higher in low-risk recipients who had received high-risk donor organs. Taken together, the use of alemtuzumab in the elderly patient remains controversial due to a paucity of data and low evidence.
Maintenance immunosuppression
At this time, only underpowered analyses and few studies of older recipients are available for evaluation. mTOR inhibitors may be attractive as immunosuppressants in the elderly since these agents have been linked to anti-tumour capacities68,118,119 and an accumulation of regulatory T-cells (Tregs).120,121 At the same time, management of wound healing issues linked to mTOR inhibitors need to be addressed. Belatacept has recently been introduced as a maintenance immunosuppression. Meaningful clinical trials in older patients are not available. The BENEFIT-EXT study evaluated transplant recipients (mean recipient age 55 years) that had received suboptimal kidneys (defined as DCD, cold ischemia time >24 h, donors >60 years, or donors >50 years plus two of the following factors – serum creatinine >1.5 mg/dl, cerebrovascular disease, or hypertension). Belatacept-based maintenance immunosuppression sustained improvement in long-term renal function compared to a cyclosporine-based protocol.122 In addition belatacept is associated with improved blood pressure, lipid profile and a lower incidence of diabetes versus treatment with a CNI.95
CNI free protocols may be of interest in the elderly. A randomized trial of elderly renal transplant recipients (>65 years) evaluated, if basiliximab induction and delayed tacrolimus combined with MMF and early steroid discontinuation could preserve renal function compared to standard tacrolimus, MMF and steroids.123 Interestingly, delayed tacrolimus in combination with basiliximab induction did neither improve renal function nor reduce the incidence of delayed graft function.
UNOS data in patients >60 years revealed lower rates of acute rejections when tacrolimus had been used in high-immunological risk recipients.112 Likewise, tacrolimus maintenance immunosuppression reduced the risk of patient death independent of immunological risk while there was no association between tacrolimus use and death-censored graft loss. In elderly patients (>60 years), MPA showed a significant decrease of graft loss and death in both, high- and low-immunological risk recipients. Interestingly, there was no clear effect of steroids in older patients on either graft loss or patient survival.112 Early steroid withdrawal or avoidance is of high relevance in older patients reducing age-prevalent side effects including diabetes, osteoporosis, osteonecrosis, cognitive impairment or impaired wound healing.124 Lower doses of MMF and lower tacrolimus levels in patients >60 years have been associated with improved graft and patient survival while rates of acute rejection were not impacted.125
Acute rejection therapy
Clinical trials evaluating the treatment of acute cellular rejection (ACR) or antibody-mediated rejection (AMR) in the elderly are lacking. Pulse steroids, usually 500 mg for three days followed by tapering for 6 days are the usual treatment for acute cellular rejections, although a number of protocols that use different doses ranging from 125 mg to 1000 mg per day have been reported. Recurrence of ACR warrants augmentation of immunosuppression with repeating pulse steroids or rATG with a subsequent switch to CNIs, or belatacept. The use of rATG might be favourable in steroid resistant rejection as it has shown its efficacy in the aged high immunological risk patient for induction therapy.112
The development of de novo donor specific antibodies (dnDNS) is associated with higher frequencies of graft failure and graft loss.126,127 Of note, non-adherence is one of the most important factors in the development of dnDNS128 with an incidence peak in younger patients.129 Although these antibodies are linked to inferior outcomes, no randomized controlled trial has shown clinical efficacy of desensitisation.130 Current therapeutic options include plasmapheresis, application of intravenous immunoglobulins and rituximab in combination or as monotherapy. Taken together, the development of dnDNS is of critical relevance for both, elderly and younger recipients, even though older patients have a lower risk for dnDNS development and acute rejection. Hence, fostering drug adherence and meticulous drug monitoring are critical to prevent to the development of dnDNS and antibody mediated rejection.131 Although there is no reliable data available thus far demonstrating that elderly graft recipients need different treatments in case of acute rejections, there is clear evidence that acute rejections are less frequent in the elderly.8–10
Recommendations for immunosuppression in elderly transplant recipients
Immunosuppression and immune function in the elderly is in general characterized by less effective immune responses with lower acute rejection rates in addition to more frequent co-morbidities. It is unclear, at this point, if recall mechanisms in sensitized patients will change with aging. Thus, not only an overall reduction of immunosuppression as currently practiced but age-specific immunosuppressive regimens may be beneficial for elderly transplant recipients. This approach needs to consider an optimal protection of the graft with age-specific changes of metabolization linked to adverse effects such as infections, de novo malignancies or nephrotoxicity. When conceptualizing age-dependent immunosuppression, effects of immunosenescence and graft immunogenicity need to be assessed. Those aspects are of importance as older recipients are frequently transplanted with older or marginal organs that have been associated with more frequent acute rejections.
Recommendations for age-specific immunosuppression
Non-sensitized older patients receiving high quality organs
Older recipients receiving organs of reasonable quality from deceased donors (in the US currently assessed with a KDPI <85%) or living donor may be candidates for an induction therapy with basiliximab. IL2-RA are linked to lower rates of infections and malignancies, aspects of importance in the elderly recipient population.
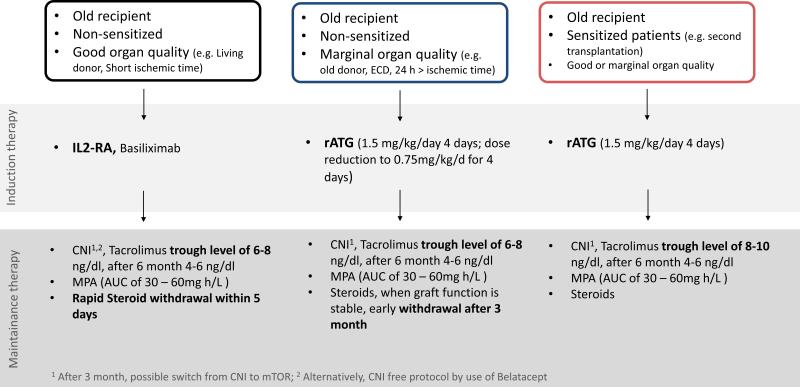
In general, a dose minimization of existing immunosuppressive protocols appears reasonable. Triple immunosuppression with CNIs, MPA and steroids could be applied with some modifications (Figure 2): A CNI dose reduction is supported by a highly reduced first-pass metabolism in the elderly and lower rejection rates reflected by initial trough level of 6 – 8 ng/dl, and levels of 4 – 6 ng/dl by month six. The AUC of 30 – 60mg h/L is the therapeutic level of MPA and can be assured by drug monitoring. The dose can be adjusted based on linear and non-linear regression models or maximum Bayesian estimation.132 Independent of the model is used, the need of dose adjustment is more likely in older patients with renal failure (creatinine clearance <20ml/min/1.73m2) and albumin changes in parallel to compromised hepatic function. A rapid steroid withdrawal within 5 days is recommended particularly given a higher risk of NODAT and infections in the elderly. Although not based on evidence, a switch from CNIs to mTOR inhibitors after three to six months may help maintaining renal function while reducing risk for de novo malignancies. Moreover, implementing belatacept as a CNI replacement appears of interest with a good safety profile of the agent. Those latter approaches will hopefully be tested in future prospective clinical trials.
Figure 2.
Recommendations for an age-specific immunosuppression. Of note, clinical trials in the elderly remain rare and the proposed regimes are rather optional recommendations, not representing guidelines. The AUC of 30 – 60mg h/L is the therapeutic level of MPA and can be assured by drug monitoring. The dose should be based on linear and non-linear regression models or maximum Bayesian estimation.132
Non-sensitized older patients receiving marginal organs
In older non-sensitized patients receiving an organ of marginal quality the use of rATG as induction appears preferable (Figure 2). At the same time, the cumulative dose of rATG may be reduced in the elderly (normal dose of 1.5 mg/kg/day for 4 days may be reduced to 0.75mg/kg/day for 4 days), although further clinical trials are warranted to support this approach. A triple maintenance immunosuppression with CNIs, MPA, and steroids appears reasonable with an MPA (AUC of 30 – 60mg h/L) dose adjustment based on linear and nonlinear regression models or maximum Bayesian estimation.132 Rapid steroid withdrawal within 3 month is recommended to prevent NODAT while keeping CNIs on a lower dose (trough level of 6 – 8 ng/dl, after six months 4 – 6 ng/dl). Again, CNI free regimens including mTOR inhibitors or belatacept appear attractive in theory but warrant prospective trials.
Sensitized older recipients
Sensitized elderly patients have higher rates of rejections,133 although the immunobiology of sensitization and recall mechanisms in the elderly remain unclear. rATG may be a favourable option for induction therapy (normal doses of 1.5 mg/kg/day for 4 days) considering the high immunological risk of the patients. Triple therapy (CNIs, MPA and steroids) appears appropriate without an age-adapted dose reduction (Figure 2). Steroid withdrawal, CNI dose reduction and a switch to CNI free immunosuppression in this population remains to be studied prospectively. MPA (AUC of 30 – 60mg h/L) dose adjustment based on linear and non-linear regression models or maximum Bayesian estimation may be relevant in this patient group with high immunological risk.132 In general, sensitized older patients may need to undergo separate trials to assure that maintenance immunosuppression can be safely reduced or switched as it remains unclear if sensitization in the elderly represents an effective allospecific or rather an unspecifically activated immune response.
Conclusion
Age is broadly impacting pharmacodynamics, pharmacokinetics and immune responses. Yet, older patients have thus far been largely excluded from clinical immunosuppressive trials. In general, older recipients have less frequent acute rejections while older organs have been linked to more potent immune responses and higher acute rejection rates.
Immunosuppressive protocols require an age-adaption that goes beyond the current clinical practice of ‘as much as necessary and as little as possible`. Future clinical trials and the use of new biomarkers will need to include the elderly to define, beyond the current limit evidence, what agents are preferable in the elderly and if a minimization of immunosuppression is safe in the elderly.
Acknowledgments
This work has been supported by grants from the National Institutes of Health (RO1AG039449 to SGT) and the German Research Foundation (KR 4362/1-1 to FK).
Abbreviations
- PDM
pre-transplant diabetes
- NODAT
new-onset diabetes after transplantation
- CNI
calcineurin inhibitors
- DC
dendritic cells
- ESP
Eurotransplant Senior Program
- FDA
Food and Drug Administration
- CYP450
cytochrome P450
- P-gp
P-glycoprotein
- NFAT
nuclear factor of activated T-cells
- mTOR
mammalian target of rapamycin
- MPA
mycophenolic acid
- MMF
mycophenolate mofetil
- ECMPS
enteric-coated mycophenolate sodium
- iATP
intracellular adenosine-tri-phosphate
- KDIGO
Kidney Disease: Improving Global Outcomes
- IL2-RA
interleukin-2 receptor antibodies
- rATG
rabbit antithymocyte globulin
- ECD
expanded-criteria donors
- DCD
donation after cardiac death
- Tregs
regulatory T-cells
- ACR
acute cellular rejection
- AMR
antibody-mediated rejection
- dnDNS
de novo donor specific antibodies
- GI
gastrointestinal
Footnotes
The authors declare no conflicts of interest.
F.K., A.E. and M.Q. performed the literature research and wrote the manuscript. F.K. wrote the article. S.G.T. and S.G. developed the concept, supervised the work and edited the manuscript. H.R.C.B. and U.H. helped editing figures and manuscript.
References
- 1.Matas AJ, Smith JM, Skeans MA, et al. OPTN/SRTR 2012 Annual Data Report: Kidney. American Journal of Transplantation. 2014;14(S1):11–44. doi: 10.1111/ajt.12579. doi:10.1111/ajt.12579. [DOI] [PubMed] [Google Scholar]
- 2.Eurotransplant International Foundation . Annual Report 2013. 2013. [Google Scholar]
- 3.Danovitch GM, Cohen DJ, Weir MR, et al. Current status of kidney and pancreas transplantation in the United States, 1994-2003. Am. J. Transplant. 2005;5(4 Pt 2):904–915. doi: 10.1111/j.1600-6135.2005.00835.x. doi:10.1111/j.1600-6135.2005.00835.x. [DOI] [PubMed] [Google Scholar]
- 4.Watt, Kymberly DS, Pedersen RA, Kremers WK, Heimbach JK, Sanchez W, Gores GJ. Long-term probability of and mortality from de novo malignancy after liver transplantation. Gastroenterology. 2009;137(6):2010–2017. doi: 10.1053/j.gastro.2009.08.070. doi:10.1053/j.gastro.2009.08.070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Karim A, Farrugia D, Cheshire J, et al. Recipient age and risk for mortality after kidney transplantation in England. Transplantation. 2014;97(8):832–838. doi: 10.1097/01.TP.0000438026.03958.7b. doi:10.1097/01.TP.0000438026.03958.7b. [DOI] [PubMed] [Google Scholar]
- 6.Meier-Kriesche HU, Ojo AO, Hanson JA, Kaplan B. Exponentially increased risk of infectious death in older renal transplant recipients. Kidney Int. 2001;59(4):1539–1543. doi: 10.1046/j.1523-1755.2001.0590041539.x. doi:10.1046/j.1523-1755.2001.0590041539.x. [DOI] [PubMed] [Google Scholar]
- 7.Jacobson PA, Schladt D, Oetting WS, et al. Lower calcineurin inhibitor doses in older compared to younger kidney transplant recipients yield similar troughs. Am. J. Transplant. 2012;12(12):3326–3336. doi: 10.1111/j.1600-6143.2012.04232.x. doi:10.1111/j.1600-6143.2012.04232.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Tullius SG, Milford E. Kidney allocation and the aging immune response. N. Engl. J. Med. 2011;364(14):1369–1370. doi: 10.1056/NEJMc1103007. doi:10.1056/NEJMc1103007. [DOI] [PubMed] [Google Scholar]
- 9.Zetterman RK, Belle SH, Hoofnagle JH, et al. Age and liver transplantation: a report of the Liver Transplantation Database. Transplantation. 1998;66(4):500–506. doi: 10.1097/00007890-199808270-00015. [DOI] [PubMed] [Google Scholar]
- 10.Kaczmarek I, Sadoni S, Schmoeckel M, et al. The need for a tailored immunosuppression in older heart transplant recipients. J. Heart Lung Transplant. 2005;24(11):1965–1968. doi: 10.1016/j.healun.2005.04.008. doi:10.1016/j.healun.2005.04.008. [DOI] [PubMed] [Google Scholar]
- 11.Dharnidharka VR, Agodoa LY, Abbott KC. Risk factors for hospitalization for bacterial or viral infection in renal transplant recipients--an analysis of USRDS data. American journal of transplantation : official journal of the American Society of Transplantation and the American Society of Transplant Surgeons. 2007;7(3):653–661. doi: 10.1111/j.1600-6143.2006.01674.x. doi:10.1111/j.1600-6143.2006.01674.x. [DOI] [PubMed] [Google Scholar]
- 12.Shao M, Wan Q, Xie W, Ye Q. Bloodstream infections among solid organ transplant recipients: epidemiology, microbiology, associated risk factors for morbility and mortality. Transplantation reviews (Orlando, Fla.) 2014;28(4):176–181. doi: 10.1016/j.trre.2014.02.001. doi:10.1016/j.trre.2014.02.001. [DOI] [PubMed] [Google Scholar]
- 13.Candel FJ, Grima E, Matesanz M, et al. Bacteremia and septic shock after solid-organ transplantation. Transplantation proceedings. 2005;37(9):4097–4099. doi: 10.1016/j.transproceed.2005.09.181. doi:10.1016/j.transproceed.2005.09.181. [DOI] [PubMed] [Google Scholar]
- 14.Dharnidharka VR, Caillard S, Agodoa LY, Abbott KC. Infection frequency and profile in different age groups of kidney transplant recipients. Transplantation. 2006;81(12):1662–1667. doi: 10.1097/01.tp.0000226068.66819.37. doi:10.1097/01.tp.0000226068.66819.37. [DOI] [PubMed] [Google Scholar]
- 15.Le Page, Amelia K, Mackie FE, McTaggart SJ, Kennedy SE. Cytomegalovirus & Epstein Barr Virus serostatus as a predictor of the long-term outcome of kidney transplantation. Nephrology (Carlton, Vic.) 2013;18(12):813–819. doi: 10.1111/nep.12149. doi:10.1111/nep.12149. [DOI] [PubMed] [Google Scholar]
- 16.Silveira FP, Husain S. Fungal infections in solid organ transplantation. Medical mycology. 2007;45(4):305–320. doi: 10.1080/13693780701200372. doi:10.1080/13693780701200372. [DOI] [PubMed] [Google Scholar]
- 17.Ok Atılgan A, Özdemir BH, Kırnap M, et al. Invasive fungal infections in liver transplant recipients. Experimental and clinical transplantation : official journal of the Middle East Society for Organ Transplantation. 2014;12(Suppl 1):110–116. [PubMed] [Google Scholar]
- 18.Kauffman CA. Fungal infections in older adults. Clinical infectious diseases : an official publication of the Infectious Diseases Society of America. 2001;33(4):550–555. doi: 10.1086/322685. doi:10.1086/322685. [DOI] [PubMed] [Google Scholar]
- 19.AlBugami M, Kiberd B. Malignancies: pre and post transplantation strategies. Transplantation reviews (Orlando, Fla.) 2014;28(2):76–83. doi: 10.1016/j.trre.2013.12.002. doi:10.1016/j.trre.2013.12.002. [DOI] [PubMed] [Google Scholar]
- 20.Peev V, Reiser J, Alachkar N. Diabetes mellitus in the transplanted kidney. Frontiers in endocrinology. 2014;5:141. doi: 10.3389/fendo.2014.00141. doi:10.3389/fendo.2014.00141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pham PT, Pham PT, Pham SV, Pham PT, Pham PT. New onset diabetes after transplantation (NODAT): an overview. Diabetes, metabolic syndrome and obesity : targets and therapy. 2011;4:175–186. doi: 10.2147/DMSO.S19027. doi:10.2147/DMSO.S19027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hoehn RS, Singhal A, Wima K, et al. Effect of pretransplant diabetes on short-term outcomes after liver transplantation: A National cohort study. Liver international : official journal of the International Association for the Study of the Liver. 2014 doi: 10.1111/liv.12770. doi:10.1111/liv.12770. [DOI] [PubMed] [Google Scholar]
- 23.Fulop T, Larbi A, Witkowski JM, Kotb R, Hirokawa K, Pawelec G. Immunosenescence and cancer. Crit Rev Oncog. 2013;18(6):489–513. doi: 10.1615/critrevoncog.2013010597. [DOI] [PubMed] [Google Scholar]
- 24.Weinberger B, Grubeck-Loebenstein B. Vaccines for the elderly. Clin. Microbiol. Infect. 2012;18(Suppl 5):100–108. doi: 10.1111/j.1469-0691.2012.03944.x. doi:10.1111/j.1469-0691.2012.03944.x. [DOI] [PubMed] [Google Scholar]
- 25.Lynch HE, Goldberg GL, Chidgey A. 2009;30(7):366–373. doi: 10.1016/j.it.2009.04.003. doi:10.1016/j.it.2009.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Naylor K, Li G, Vallejo AN, et al. The influence of age on T cell generation and TCR diversity. J. Immunol. 2005;174(11):7446–7452. doi: 10.4049/jimmunol.174.11.7446. [DOI] [PubMed] [Google Scholar]
- 27.Hong MS, Dan JM, Choi J, Kang I. Age-associated changes in the frequency of naïve, memory and effector CD8+ T cells. Mechanisms of ageing and development. 2004;125(9):615–618. doi: 10.1016/j.mad.2004.07.001. doi:10.1016/j.mad.2004.07.001. [DOI] [PubMed] [Google Scholar]
- 28.Saule P, Trauet J, Dutriez V, Lekeux V, Dessaint J, Labalette M. Accumulation of memory T cells from childhood to old age: central and effector memory cells in CD4(+) versus effector memory and terminally differentiated memory cells in CD8(+) compartment. Mechanisms of ageing and development. 2006;127(3):274–281. doi: 10.1016/j.mad.2005.11.001. doi:10.1016/j.mad.2005.11.001. [DOI] [PubMed] [Google Scholar]
- 29.Brzezińska A, Magalska A, Szybińska A, Sikora E. Proliferation and apoptosis of human CD8(+)CD28(+) and CD8(+)CD28(−) lymphocytes during aging. Exp. Gerontol. 2004;39(4):539–544. doi: 10.1016/j.exger.2003.09.026. doi:10.1016/j.exger.2003.09.026. [DOI] [PubMed] [Google Scholar]
- 30.Aubert G, Baerlocher GM, Vulto I, Poon SS, Lansdorp PM. Collapse of telomere homeostasis in hematopoietic cells caused by heterozygous mutations in telomerase genes. PLoS Genet. 2012;8(5):e1002696. doi: 10.1371/journal.pgen.1002696. doi:10.1371/journal.pgen.1002696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Chou JP, Effros RB. T cell replicative senescence in human aging. Curr. Pharm. Des. 2013;19(9):1680–1698. doi: 10.2174/138161213805219711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Larbi A, Cabreiro F, Zelba H, et al. Reduced oxygen tension results in reduced human T cell proliferation and increased intracellular oxidative damage and susceptibility to apoptosis upon activation. Free Radic. Biol. Med. 2010;48(1):26–34. doi: 10.1016/j.freeradbiomed.2009.09.025. doi:10.1016/j.freeradbiomed.2009.09.025. [DOI] [PubMed] [Google Scholar]
- 33.Frasca D, Diaz A, Romero M, Landin AM, Blomberg BB. Age effects on B cells and humoral immunity in humans. Ageing Research Reviews. 2011;10(3):330–335. doi: 10.1016/j.arr.2010.08.004. doi:10.1016/j.arr.2010.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Herrero C, Marqués L, Lloberas J, Celada A. IFN-gamma-dependent transcription of MHC class II IA is impaired in macrophages from aged mice. J. Clin. Invest. 2001;107(4):485–493. doi: 10.1172/JCI11696. doi:10.1172/JCI11696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Demers P, Moffatt S, Oyer PE, Hunt SA, Reitz BA, Robbins RC. Long-term results of heart transplantation in patients older than 60 years. J. Thorac. Cardiovasc. Surg. 2003;126(1):224–231. doi: 10.1016/s0022-5223(03)00055-2. [DOI] [PubMed] [Google Scholar]
- 36.Tullius SG, Tran H, Guleria I, Malek SK, Tilney NL, Milford E. The combination of donor and recipient age is critical in determining host immunoresponsiveness and renal transplant outcome. Ann. Surg. 2010;252(4):662–674. doi: 10.1097/SLA.0b013e3181f65c7d. doi:10.1097/SLA.0b013e3181f65c7d. [DOI] [PubMed] [Google Scholar]
- 37.Frei U, Noeldeke J, Machold-Fabrizii V, et al. Prospective age-matching in elderly kidney transplant recipients--a 5-year analysis of the Eurotransplant Senior Program. Am. J. Transplant. 2008;8(1):50–57. doi: 10.1111/j.1600-6143.2007.02014.x. doi:10.1111/j.1600-6143.2007.02014.x. [DOI] [PubMed] [Google Scholar]
- 38.Aalami OO, Fang TD, Song HM, Nacamuli RP. Physiological features of aging persons. Arch Surg. 2003;138(10):1068–1076. doi: 10.1001/archsurg.138.10.1068. doi:10.1001/archsurg.138.10.1068. [DOI] [PubMed] [Google Scholar]
- 39.Corsonello A, Pedone C, Incalzi RA. Age-related pharmacokinetic and pharmacodynamic changes and related risk of adverse drug reactions. Curr. Med. Chem. 2010;17(6):571–584. doi: 10.2174/092986710790416326. [DOI] [PubMed] [Google Scholar]
- 40.Wooten JM. Pharmacotherapy considerations in elderly adults. South. Med. J. 2012;105(8):437–445. doi: 10.1097/SMJ.0b013e31825fed90. doi:10.1097/SMJ.0b013e31825fed90. [DOI] [PubMed] [Google Scholar]
- 41.Zulman DM, Sussman JB, Chen X, Cigolle CT, Blaum CS, Hayward RA. Examining the evidence: a systematic review of the inclusion and analysis of older adults in randomized controlled trials. J Gen Intern Med. 2011;26(7):783–790. doi: 10.1007/s11606-010-1629-x. doi:10.1007/s11606-010-1629-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.US Government Accountability Office . Prescription Drugs: FDA Guidance and Regulations Related to Data on Elderly Persons in Clinical Drug Trials: GAO-07-47R. Washington, D.C: 2007. [Google Scholar]
- 43.Scott IA, Guyatt GH. Cautionary tales in the interpretation of clinical studies involving older persons. Arch. Intern. Med. 2010;170(7):587–595. doi: 10.1001/archinternmed.2010.18. doi:10.1001/archinternmed.2010.18. [DOI] [PubMed] [Google Scholar]
- 44.Blosser CD, Huverserian A, Bloom RD, et al. Age, exclusion criteria, and generalizability of randomized trials enrolling kidney transplant recipients. Transplantation. 2011;91(8):858–863. doi: 10.1097/TP.0b013e31820f42d9. doi:10.1097/TP.0b013e31820f42d9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Saeki T, Ueda K, Tanigawara Y, Hori R, Komano T. Human P-glycoprotein transports cyclosporin A and FK506. J. Biol. Chem. 1993;268(9):6077–6080. [PubMed] [Google Scholar]
- 46.Kaplan B, Lown K, Craig R, et al. Low bioavailability of cyclosporine microemulsion and tacrolimus in a small bowel transplant recipient: possible relationship to intestinal P-glycoprotein activity. Transplantation. 1999;67(2):333–335. doi: 10.1097/00007890-199901270-00026. [DOI] [PubMed] [Google Scholar]
- 47.Christians U. Transport proteins and intestinal metabolism: P-glycoprotein and cytochrome P4503A. Ther Drug Monit. 2004;26(2):104–106. doi: 10.1097/00007691-200404000-00002. [DOI] [PubMed] [Google Scholar]
- 48.George J, Byth K, Farrell GC. Age but not gender selectively affects expression of individual cytochrome P450 proteins in human liver. Biochem. Pharmacol. 1995;50(5):727–730. doi: 10.1016/0006-2952(95)00192-3. [DOI] [PubMed] [Google Scholar]
- 49.Warrington JS, Greenblatt DJ, von Moltke, Lisa L. Age-related differences in CYP3A expression and activity in the rat liver, intestine, and kidney. J. Pharmacol. Exp. Ther. 2004;309(2):720–729. doi: 10.1124/jpet.103.061077. doi:10.1124/jpet.103.061077. [DOI] [PubMed] [Google Scholar]
- 50.Dai Y, Iwanaga K, Lin YS, et al. In vitro metabolism of cyclosporine A by human kidney CYP3A5. Biochem. Pharmacol. 2004;68(9):1889–1902. doi: 10.1016/j.bcp.2004.07.012. doi:10.1016/j.bcp.2004.07.012. [DOI] [PubMed] [Google Scholar]
- 51.Passey C, Birnbaum AK, Brundage RC, Oetting WS, Israni AK, Jacobson PA. Dosing equation for tacrolimus using genetic variants and clinical factors. Br J Clin Pharmacol. 2011;72(6):948–957. doi: 10.1111/j.1365-2125.2011.04039.x. doi:10.1111/j.1365-2125.2011.04039.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Miura M, Satoh S, Kagaya H, et al. No impact of age on dose-adjusted pharmacokinetics of tacrolimus, mycophenolic acid and prednisolone 1 month after renal transplantation. Eur. J. Clin. Pharmacol. 2009;65(10):1047–1053. doi: 10.1007/s00228-009-0721-9. doi:10.1007/s00228-009-0721-9. [DOI] [PubMed] [Google Scholar]
- 53.Roy J, Lajoie J, Zijenah LS, et al. CYP3A5 genetic polymorphisms in different ethnic populations. Drug Metab. Dispos. 2005;33(7):884–887. doi: 10.1124/dmd.105.003822. doi:10.1124/dmd.105.003822. [DOI] [PubMed] [Google Scholar]
- 54.Vilas-Boas V, Silva R, Gaio AR, et al. P-glycoprotein activity in human Caucasian male lymphocytes does not follow its increased expression during aging. Cytometry A. 2011;79(11):912–919. doi: 10.1002/cyto.a.21135. doi:10.1002/cyto.a.21135. [DOI] [PubMed] [Google Scholar]
- 55.Brenner SS, Klotz U. P-glycoprotein function in the elderly. Eur. J. Clin. Pharmacol. 2004;60(2):97–102. doi: 10.1007/s00228-004-0733-4. doi:10.1007/s00228-004-0733-4. [DOI] [PubMed] [Google Scholar]
- 56.Falck P, Asberg A, Byberg K, et al. Reduced elimination of cyclosporine A in elderly (65 years) kidney transplant recipients. Transplantation. 2008;86(10):1379–1383. doi: 10.1097/TP.0b013e31818aa4b6. doi:10.1097/TP.0b013e31818aa4b6. [DOI] [PubMed] [Google Scholar]
- 57.Zahir H, McCaughan G, Gleeson M, Nand RA, McLachlan AJ. Factors affecting variability in distribution of tacrolimus in liver transplant recipients. Br J Clin Pharmacol. 2004;57(3):298–309. doi: 10.1046/j.1365-2125.2003.02008.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Størset E, Holford N, Midtvedt K, Bremer S, Bergan S, Åsberg A. Importance of hematocrit for a tacrolimus target concentration strategy. Eur. J. Clin. Pharmacol. 2014;70(1):65–77. doi: 10.1007/s00228-013-1584-7. doi:10.1007/s00228-013-1584-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Robertsen I, Åsberg A, Ingerø AO, et al. Use of generic tacrolimus in elderly renal transplant recipients: precaution is needed. Transplantation. 2015;99(3):528–532. doi: 10.1097/TP.0000000000000384. doi:10.1097/TP.0000000000000384. [DOI] [PubMed] [Google Scholar]
- 60.Heit JJ, Apelqvist AA, Gu X, et al. Calcineurin/NFAT signalling regulates pancreatic beta-cell growth and function. Nature. 2006;443(7109):345–349. doi: 10.1038/nature05097. doi:10.1038/nature05097. [DOI] [PubMed] [Google Scholar]
- 61.Shah T, Kasravi A, Huang E, et al. Risk factors for development of new-onset diabetes mellitus after kidney transplantation. Transplantation. 2006;82(12):1673–1676. doi: 10.1097/01.tp.0000250756.66348.9a. doi:10.1097/01.tp.0000250756.66348.9a. [DOI] [PubMed] [Google Scholar]
- 62.Heisel O, Heisel R, Balshaw R, Keown P. New onset diabetes mellitus in patients receiving calcineurin inhibitors: a systematic review and meta-analysis. American journal of transplantation : official journal of the American Society of Transplantation and the American Society of Transplant Surgeons. 2004;4(4):583–595. doi: 10.1046/j.1600-6143.2003.00372.x. doi:10.1046/j.1600-6143.2003.00372.x. [DOI] [PubMed] [Google Scholar]
- 63.Silva HT, Yang HC, Meier-Kriesche H, et al. Long-term follow-up of a phase III clinical trial comparing tacrolimus extended-release/MMF, tacrolimus/MMF, and cyclosporine/MMF in de novo kidney transplant recipients. Transplantation. 2014;97(6):636–641. doi: 10.1097/01.TP.0000437669.93963.8E. doi:10.1097/01.TP.0000437669.93963.8E. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Haddad EM, McAlister VC, Renouf E, Malthaner R, Kjaer MS, Gluud LL. Cyclosporin versus tacrolimus for liver transplanted patients. The Cochrane database of systematic reviews. 2006;(4):CD005161. doi: 10.1002/14651858.CD005161.pub2. doi:10.1002/14651858.CD005161.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Pahlavani MA, Vargas DM. Influence of aging and caloric restriction on activation of Ras/MAPK, calcineurin, and CaMK-IV activities in rat T cells. Proc. Soc. Exp. Biol. Med. 2000;223(2):163–169. doi: 10.1046/j.1525-1373.2000.22322.x. [DOI] [PubMed] [Google Scholar]
- 66.Whisler RL, Beiqing L, Chen M. Age-related decreases in IL-2 production by human T cells are associated with impaired activation of nuclear transcriptional factors AP-1 and NF-AT. Cell. Immunol. 1996;169(2):185–195. doi: 10.1006/cimm.1996.0109. doi:10.1006/cimm.1996.0109. [DOI] [PubMed] [Google Scholar]
- 67.Lindholm A. Factors influencing the pharmacokinetics of cyclosporine in man. Ther Drug Monit. 1991;13(6):465–477. doi: 10.1097/00007691-199111000-00001. [DOI] [PubMed] [Google Scholar]
- 68.Kauffman HM, Cherikh WS, Cheng Y, Hanto DW, Kahan BD. Maintenance immunosuppression with target-of-rapamycin inhibitors is associated with a reduced incidence of de novo malignancies. Transplantation. 2005;80(7):883–889. doi: 10.1097/01.tp.0000184006.43152.8d. [DOI] [PubMed] [Google Scholar]
- 69.Fischer L, Klempnauer J, Beckebaum S, et al. A randomized, controlled study to assess the conversion from calcineurin-inhibitors to everolimus after liver transplantation--PROTECT. Am. J. Transplant. 2012;12(7):1855–1865. doi: 10.1111/j.1600-6143.2012.04049.x. doi:10.1111/j.1600-6143.2012.04049.x. [DOI] [PubMed] [Google Scholar]
- 70.Tateishi T, Nakura H, Asoh M, et al. A comparison of hepatic cytochrome P450 protein expression between infancy and postinfancy. Life Sci. 1997;61(26):2567–2574. doi: 10.1016/s0024-3205(97)01011-4. [DOI] [PubMed] [Google Scholar]
- 71.Zimmerman JJ, Lasseter KC, Lim H, et al. Pharmacokinetics of sirolimus (rapamycin) in subjects with mild to moderate hepatic impairment. J Clin Pharmacol. 2005;45(12):1368–1372. doi: 10.1177/0091270005281350. doi:10.1177/0091270005281350. [DOI] [PubMed] [Google Scholar]
- 72.Kovarik JM, Sabia HD, Figueiredo J, et al. Influence of hepatic impairment on everolimus pharmacokinetics: implications for dose adjustment. Clin. Pharmacol. Ther. 2001;70(5):425–430. [PubMed] [Google Scholar]
- 73.Johnston O, Rose CL, Webster AC, Gill JS. Sirolimus is associated with new-onset diabetes in kidney transplant recipients. Journal of the American Society of Nephrology : JASN. 2008;19(7):1411–1418. doi: 10.1681/ASN.2007111202. doi:10.1681/ASN.2007111202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Flechner SM, Glyda M, Cockfield S, et al. The ORION study: comparison of two sirolimus-based regimens versus tacrolimus and mycophenolate mofetil in renal allograft recipients. American journal of transplantation : official journal of the American Society of Transplantation and the American Society of Transplant Surgeons. 2011;11(8):1633–1644. doi: 10.1111/j.1600-6143.2011.03573.x. doi:10.1111/j.1600-6143.2011.03573.x. [DOI] [PubMed] [Google Scholar]
- 75.McLean AJ, Le Couteur, David G. Aging biology and geriatric clinical pharmacology. Pharmacol. Rev. 2004;56(2):163–184. doi: 10.1124/pr.56.2.4. doi:10.1124/pr.56.2.4. [DOI] [PubMed] [Google Scholar]
- 76.Woo J, Chan HS, Or KH, Arumanayagam M. Effect of age and disease on two drug binding proteins: albumin and alpha-1- acid glycoprotein. Clin. Biochem. 1994;27(4):289–292. doi: 10.1016/0009-9120(94)90032-9. [DOI] [PubMed] [Google Scholar]
- 77.Tredger JM, Brown NW, Adams J, et al. Monitoring mycophenolate in liver transplant recipients: toward a therapeutic range. Liver Transpl. 2004;10(4):492–502. doi: 10.1002/lt.20124. doi:10.1002/lt.20124. [DOI] [PubMed] [Google Scholar]
- 78.van Hest Reinier M, Mathot Ron A A, Pescovitz MD, Gordon R, Mamelok RD, van Gelder T. Explaining variability in mycophenolic acid exposure to optimize mycophenolate mofetil dosing: a population pharmacokinetic meta-analysis of mycophenolic acid in renal transplant recipients. J. Am. Soc. Nephrol. 2006;17(3):871–880. doi: 10.1681/ASN.2005101070. doi:10.1681/ASN.2005101070. [DOI] [PubMed] [Google Scholar]
- 79.Kees MG, Steinke T, Moritz S, et al. Omeprazole impairs the absorption of mycophenolate mofetil but not of enteric-coated mycophenolate sodium in healthy volunteers. J Clin Pharmacol. 2012;52(8):1265–1272. doi: 10.1177/0091270011412968. doi:10.1177/0091270011412968. [DOI] [PubMed] [Google Scholar]
- 80.Staatz CE, Tett SE. Clinical pharmacokinetics and pharmacodynamics of mycophenolate in solid organ transplant recipients. Clin Pharmacokinet. 2007;46(1):13–58. doi: 10.2165/00003088-200746010-00002. doi:10.2165/00003088-200746010-00002. [DOI] [PubMed] [Google Scholar]
- 81.Czock D, Keller F, Rasche FM, Häussler U. Pharmacokinetics and pharmacodynamics of systemically administered glucocorticoids. Clin Pharmacokinet. 2005;44(1):61–98. doi: 10.2165/00003088-200544010-00003. doi:10.2165/00003088-200544010-00003. [DOI] [PubMed] [Google Scholar]
- 82.Sæves I, Line P, Bergan S. The pharmacokinetics of prednisolone and prednisone in adult liver transplant recipients early after transplantation. Ther Drug Monit. 2012;34(4):452–459. doi: 10.1097/FTD.0b013e31825ee3f8. doi:10.1097/FTD.0b013e31825ee3f8. [DOI] [PubMed] [Google Scholar]
- 83.Kawai S, Ichikawa Y, Homma M. Differences in metabolic properties among cortisol, prednisolone, and dexamethasone in liver and renal diseases: accelerated metabolism of dexamethasone in renal failure. J. Clin. Endocrinol. Metab. 1985;60(5):848–854. doi: 10.1210/jcem-60-5-848. doi:10.1210/jcem-60-5-848. [DOI] [PubMed] [Google Scholar]
- 84.Booker BM, Magee MH, Blum RA, Lates CD, Jusko WJ. Pharmacokinetic and pharmacodynamic interactions between diltiazem and methylprednisolone in healthy volunteers. Clin. Pharmacol. Ther. 2002;72(4):370–382. doi: 10.1067/mcp.2002.127944. doi:10.1067/mcp.2002.127944. [DOI] [PubMed] [Google Scholar]
- 85.Lebrun-Vignes B, Archer VC, Diquet B, et al. Effect of itraconazole on the pharmacokinetics of prednisolone and methylprednisolone and cortisol secretion in healthy subjects. Br J Clin Pharmacol. 2001;51(5):443–450. doi: 10.1046/j.1365-2125.2001.01372.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Shimada T, Terada A, Yokogawa K, et al. Lowered blood concentration of tacrolimus and its recovery with changes in expression of CYP3A and P-glycoprotein after high-dose steroid therapy. Transplantation. 2002;74(10):1419–1424. doi: 10.1097/00007890-200211270-00014. doi:10.1097/01.TP.0000038287.39271.8F. [DOI] [PubMed] [Google Scholar]
- 87.Bergmann TK, Barraclough KA, Lee KJ, Staatz CE. Clinical pharmacokinetics and pharmacodynamics of prednisolone and prednisone in solid organ transplantation. Clinical pharmacokinetics. 2012;51(11):711–741. doi: 10.1007/s40262-012-0007-8. doi:10.1007/s40262-012-0007-8. [DOI] [PubMed] [Google Scholar]
- 88.Stuck AE, Frey BM, Frey FJ. Kinetics of prednisolone and endogenous cortisol suppression in the elderly. Clin. Pharmacol. Ther. 1988;43(4):354–362. doi: 10.1038/clpt.1988.43. [DOI] [PubMed] [Google Scholar]
- 89.Tornatore KM, Logue G, Venuto RC, Davis PJ. Cortisol pharmacodynamics after methylprednisolone administration in young and elderly males. J Clin Pharmacol. 1997;37(4):304–311. doi: 10.1002/j.1552-4604.1997.tb04307.x. [DOI] [PubMed] [Google Scholar]
- 90.Penfornis A, Kury-Paulin S. Immunosuppressive drug-induced diabetes. Diabetes & metabolism. 2006;32(5 Pt 2):539–546. doi: 10.1016/s1262-3636(06)72809-9. [DOI] [PubMed] [Google Scholar]
- 91.Midtvedt K, Hjelmesaeth J, Hartmann A, et al. Insulin resistance after renal transplantation: the effect of steroid dose reduction and withdrawal. Journal of the American Society of Nephrology : JASN. 2004;15(12):3233–3239. doi: 10.1097/01.ASN.0000145435.80005.1E. doi:10.1097/01.ASN.0000145435.80005.1E. [DOI] [PubMed] [Google Scholar]
- 92.Zhou Z, Shen J, Hong Y, Kaul S, Pfister M, Roy A. Time-varying belatacept exposure and its relationship to efficacy/safety responses in kidney-transplant recipients. Clinical pharmacology and therapeutics. 2012;92(2):251–257. doi: 10.1038/clpt.2012.84. doi:10.1038/clpt.2012.84. [DOI] [PubMed] [Google Scholar]
- 93.Agrawal A, Agrawal S, Cao J, Su H, Osann K, Gupta S. Altered innate immune functioning of dendritic cells in elderly humans: a role of phosphoinositide 3-kinase-signaling pathway. J. Immunol. 2007;178(11):6912–6922. doi: 10.4049/jimmunol.178.11.6912. [DOI] [PubMed] [Google Scholar]
- 94.Leng Q, Bentwich Z, Borkow G. CTLA-4 upregulation during aging. Mech. Ageing Dev. 2002;123(10):1419–1421. doi: 10.1016/s0047-6374(02)00077-5. [DOI] [PubMed] [Google Scholar]
- 95.Masson P, Henderson L, Chapman JR, Craig JC, Webster AC. Belatacept for kidney transplant recipients. The Cochrane database of systematic reviews. 2014;11:CD010699. doi: 10.1002/14651858.CD010699.pub2. doi:10.1002/14651858.CD010699.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Coffey GP, Stefanich E, Palmieri S, et al. In vitro internalization, intracellular transport, and clearance of an anti-CD11a antibody (Raptiva) by human T-cells. J. Pharmacol. Exp. Ther. 2004;310(3):896–904. doi: 10.1124/jpet.104.067611. doi:10.1124/jpet.104.067611. [DOI] [PubMed] [Google Scholar]
- 97.van Bueren Lammerts, Jeroen J, Bleeker WK, Bøgh HO, et al. Effect of target dynamics on pharmacokinetics of a novel therapeutic antibody against the epidermal growth factor receptor: implications for the mechanisms of action. Cancer Res. 2006;66(15):7630–7638. doi: 10.1158/0008-5472.CAN-05-4010. doi:10.1158/0008-5472.CAN-05-4010. [DOI] [PubMed] [Google Scholar]
- 98.Mahbub S, Deburghgraeve CR, Kovacs EJ. Advanced age impairs macrophage polarization. J. Interferon Cytokine Res. 2012;32(1):18–26. doi: 10.1089/jir.2011.0058. doi:10.1089/jir.2011.0058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Kovarik JM, Kahan BD, Rajagopalan PR, et al. Population pharmacokinetics and exposure-response relationships for basiliximab in kidney transplantation. The U.S. Simulect Renal Transplant Study Group. Transplantation. 1999;68(9):1288–1294. doi: 10.1097/00007890-199911150-00012. [DOI] [PubMed] [Google Scholar]
- 100.Budde K, Matz M, Dürr M, Glander P. Biomarkers of over-immunosuppression. Clin. Pharmacol. Ther. 2011;90(2):316–322. doi: 10.1038/clpt.2011.111. doi:10.1038/clpt.2011.111. [DOI] [PubMed] [Google Scholar]
- 101.Steinebrunner N, Sandig C, Sommerer C, et al. Pharmacodynamic monitoring of nuclear factor of activated T cell-regulated gene expression in liver allograft recipients on immunosuppressive therapy with calcineurin inhibitors in the course of time and correlation with acute rejection episodes--a prospective study. Ann. Transplant. 2014;19:32–40. doi: 10.12659/AOT.889809. doi:10.12659/AOT.889809. [DOI] [PubMed] [Google Scholar]
- 102.Leogrande D, Teutonico A, Ranieri E, et al. Monitoring biological action of rapamycin in renal transplantation. Am. J. Kidney Dis. 2007;50(2):314–325. doi: 10.1053/j.ajkd.2007.05.002. doi:10.1053/j.ajkd.2007.05.002. [DOI] [PubMed] [Google Scholar]
- 103.Vethe NT, Ali AM, Reine PA, et al. Simultaneous quantification of IMPDH activity and purine bases in lymphocytes using LC-MS/MS: assessment of biomarker responses to mycophenolic acid. Ther Drug Monit. 2014;36(1):108–118. doi: 10.1097/FTD.0b013e3182a13900. doi:10.1097/FTD.0b013e3182a13900. [DOI] [PubMed] [Google Scholar]
- 104.Ducloux D, Courivaud C, Bamoulid J, et al. Prolonged CD4 T cell lymphopenia increases morbidity and mortality after renal transplantation. J. Am. Soc. Nephrol. 2010;21(5):868–875. doi: 10.1681/ASN.2009090976. doi:10.1681/ASN.2009090976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Ducloux D, Bamoulid J, Courivaud C, et al. Thymic function, anti-thymocytes globulins, and cancer after renal transplantation. Transpl. Immunol. 2011;25(1):56–60. doi: 10.1016/j.trim.2011.05.003. doi:10.1016/j.trim.2011.05.003. [DOI] [PubMed] [Google Scholar]
- 106.Hricik DE, Nickerson P, Formica RN, et al. Multicenter validation of urinary CXCL9 as a risk-stratifying biomarker for kidney transplant injury. Am. J. Transplant. 2013;13(10):2634–2644. doi: 10.1111/ajt.12426. doi:10.1111/ajt.12426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Jackson JA, Kim EJ, Begley B, et al. Urinary chemokines CXCL9 and CXCL10 are noninvasive markers of renal allograft rejection and BK viral infection. Am. J. Transplant. 2011;11(10):2228–2234. doi: 10.1111/j.1600-6143.2011.03680.x. doi:10.1111/j.1600-6143.2011.03680.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Chen Y, Tai Q, Hong S, et al. Pretransplantation soluble CD30 level as a predictor of acute rejection in kidney transplantation: a meta-analysis. Transplantation. 2012;94(9):911–918. doi: 10.1097/TP.0b013e31826784ad. doi:10.1097/TP.0b013e31826784ad. [DOI] [PubMed] [Google Scholar]
- 109.Kowalski RJ, Post DR, Mannon RB, et al. Assessing relative risks of infection and rejection: a meta-analysis using an immune function assay. Transplantation. 2006;82(5):663–668. doi: 10.1097/01.tp.0000234837.02126.70. doi:10.1097/01.tp.0000234837.02126.70. [DOI] [PubMed] [Google Scholar]
- 110.Schulz-Juergensen S, Burdelski MM, Oellerich M, Brandhorst G. Intracellular ATP production in CD4+ T cells as a predictor for infection and allograft rejection in trough-level guided pediatric liver transplant recipients under calcineurin-inhibitor therapy. Ther Drug Monit. 2012;34(1):4–10. doi: 10.1097/FTD.0b013e31823c5668. doi:10.1097/FTD.0b013e31823c5668. [DOI] [PubMed] [Google Scholar]
- 111.KDIGO clinical practice guideline for the care of kidney transplant recipients. Am. J. Transplant. 2009;9(Suppl 3):S1–155. doi: 10.1111/j.1600-6143.2009.02834.x. doi:10.1111/j.1600-6143.2009.02834.x. [DOI] [PubMed] [Google Scholar]
- 112.Gill J, Sampaio M, Gill JS, et al. Induction immunosuppressive therapy in the elderly kidney transplant recipient in the United States. Clin J Am Soc Nephrol. 2011;6(5):1168–1178. doi: 10.2215/CJN.07540810. doi:10.2215/CJN.07540810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Khanmoradi K, Knorr JP, Feyssa EL, et al. Evaluating safety and efficacy of rabbit antithymocyte globulin induction in elderly kidney transplant recipients. Exp Clin Transplant. 2013;11(3):222–228. doi: 10.6002/ect.2012.0211. doi:10.6002/ect.2012.0211. [DOI] [PubMed] [Google Scholar]
- 114.Patel SJ, Knight RJ, Suki WN, et al. Rabbit antithymocyte induction and dosing in deceased donor renal transplant recipients over 60 yr of age. Clin Transplant. 2011;25(3):E250–6. doi: 10.1111/j.1399-0012.2010.01393.x. doi:10.1111/j.1399-0012.2010.01393.x. [DOI] [PubMed] [Google Scholar]
- 115.Brennan DC, Daller JA, Lake KD, Cibrik D, Del Castillo D. Rabbit antithymocyte globulin versus basiliximab in renal transplantation. N. Engl. J. Med. 2006;355(19):1967–1977. doi: 10.1056/NEJMoa060068. doi:10.1056/NEJMoa060068. [DOI] [PubMed] [Google Scholar]
- 116.Webster AC, Ruster LP, McGee R, et al. Interleukin 2 receptor antagonists for kidney transplant recipients. Cochrane Database Syst Rev. 2010;(1):CD003897. doi: 10.1002/14651858.CD003897.pub3. doi:10.1002/14651858.CD003897.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Haynes R, Harden P, Judge P, et al. Alemtuzumab-based induction treatment versus basiliximab-based induction treatment in kidney transplantation (the 3C Study): a randomised trial. Lancet. 2014;384(9955):1684–1690. doi: 10.1016/S0140-6736(14)61095-3. doi:10.1016/S0140-6736(14)61095-3. [DOI] [PubMed] [Google Scholar]
- 118.Boffa DJ, Luan F, Thomas D, et al. Rapamycin inhibits the growth and metastatic progression of non-small cell lung cancer. Clin. Cancer Res. 2004;10(1 Pt 1):293–300. doi: 10.1158/1078-0432.ccr-0629-3. [DOI] [PubMed] [Google Scholar]
- 119.Luan FL, Ding R, Sharma VK, Chon WJ, Lagman M, Suthanthiran M. Rapamycin is an effective inhibitor of human renal cancer metastasis. Kidney Int. 2003;63(3):917–926. doi: 10.1046/j.1523-1755.2003.00805.x. doi:10.1046/j.1523-1755.2003.00805.x. [DOI] [PubMed] [Google Scholar]
- 120.Gabryšová L, Christensen JR, Wu X, Kissenpfennig A, Malissen B, O'Garra A. Integrated T-cell receptor and costimulatory signals determine TGF-β-dependent differentiation and maintenance of Foxp3+ regulatory T cells. Eur. J. Immunol. 2011;41(5):1242–1248. doi: 10.1002/eji.201041073. doi:10.1002/eji.201041073. [DOI] [PubMed] [Google Scholar]
- 121.Wang Y, Sparwasser T, Figlin R, Kim HL. Foxp3+ T cells inhibit antitumor immune memory modulated by mTOR inhibition. Cancer Res. 2014;74(8):2217–2228. doi: 10.1158/0008-5472.CAN-13-2928. doi:10.1158/0008-5472.CAN-13-2928. [DOI] [PubMed] [Google Scholar]
- 122.Charpentier B, Medina Pestana JO, Del C Rial M, et al. Long-term exposure to belatacept in recipients of extended criteria donor kidneys. Am. J. Transplant. 2013;13(11):2884–2891. doi: 10.1111/ajt.12459. doi:10.1111/ajt.12459. [DOI] [PubMed] [Google Scholar]
- 123.Andrés A, Budde K, Clavien P, et al. A randomized trial comparing renal function in older kidney transplant patients following delayed versus immediate tacrolimus administration. Transplantation. 2009;88(9):1101–1108. doi: 10.1097/TP.0b013e3181ba06ee. doi:10.1097/TP.0b013e3181ba06ee. [DOI] [PubMed] [Google Scholar]
- 124.Woodle ES, First MR, Pirsch J, Shihab F, Gaber AO, van Veldhuisen P. A prospective, randomized, double-blind, placebo-controlled multicenter trial comparing early (7 day) corticosteroid cessation versus long-term, low-dose corticosteroid therapy. Ann. Surg. 2008;248(4):564–577. doi: 10.1097/SLA.0b013e318187d1da. doi:10.1097/SLA.0b013e318187d1da. [DOI] [PubMed] [Google Scholar]
- 125.Badowski M, Gurk-Turner C, Cangro C, et al. The impact of reduced immunosuppression on graft outcomes in elderly renal transplant recipients. Clin Transplant. 2009;23(6):930–937. doi: 10.1111/j.1399-0012.2009.01028.x. doi:10.1111/j.1399-0012.2009.01028.x. [DOI] [PubMed] [Google Scholar]
- 126.Hourmant M, Cesbron-Gautier A, Terasaki PI, et al. Frequency and clinical implications of development of donor-specific and non-donor-specific HLA antibodies after kidney transplantation. Journal of the American Society of Nephrology : JASN. 2005;16(9):2804–2812. doi: 10.1681/ASN.2004121130. doi:10.1681/ASN.2004121130. [DOI] [PubMed] [Google Scholar]
- 127.Seveso M, Bosio E, Ancona E, Cozzi E. De novo anti-HLA antibody responses after renal transplantation: detection and clinical impact. Contributions to nephrology. 2009;162:87–98. doi: 10.1159/000170841. doi:10.1159/000170841. [DOI] [PubMed] [Google Scholar]
- 128.Wiebe C, Gibson IW, Blydt-Hansen TD, et al. Evolution and clinical pathologic correlations of de novo donor-specific HLA antibody post kidney transplant. American journal of transplantation : official journal of the American Society of Transplantation and the American Society of Transplant Surgeons. 2012;12(5):1157–1167. doi: 10.1111/j.1600-6143.2012.04013.x. doi:10.1111/j.1600-6143.2012.04013.x. [DOI] [PubMed] [Google Scholar]
- 129.Everly MJ, Rebellato LM, Haisch CE, et al. Incidence and impact of de novo donor-specific alloantibody in primary renal allografts. Transplantation. 2013;95(3):410–417. doi: 10.1097/TP.0b013e31827d62e3. doi:10.1097/TP.0b013e31827d62e3. [DOI] [PubMed] [Google Scholar]
- 130.Roberts DM, Jiang SH, Chadban SJ. The treatment of acute antibody-mediated rejection in kidney transplant recipients-a systematic review. Transplantation. 2012;94(8):775–783. doi: 10.1097/TP.0b013e31825d1587. doi:10.1097/TP.0b013e31825d1587. [DOI] [PubMed] [Google Scholar]
- 131.Tait BD, Süsal C, Gebel HM, et al. Consensus guidelines on the testing and clinical management issues associated with HLA and non-HLA antibodies in transplantation. Transplantation. 2013;95(1):19–47. doi: 10.1097/TP.0b013e31827a19cc. doi:10.1097/TP.0b013e31827a19cc. [DOI] [PubMed] [Google Scholar]
- 132.Chen H, Chen B. Clinical mycophenolic acid monitoring in liver transplant recipients. World journal of gastroenterology : WJG. 2014;20(31):10715–10728. doi: 10.3748/wjg.v20.i31.10715. doi:10.3748/wjg.v20.i31.10715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Süsal C, Döhler B, Opelz G. Presensitized kidney graft recipients with HLA class I and II antibodies are at increased risk for graft failure: a Collaborative Transplant Study report. Human immunology. 2009;70(8):569–573. doi: 10.1016/j.humimm.2009.04.013. doi:10.1016/j.humimm.2009.04.013. [DOI] [PubMed] [Google Scholar]
- 134.Koefoed-Nielsen PB, Gesualdo MB, Poulsen JH, Jørgensen KA. Blood tacrolimus levels and calcineurin phosphatase activity early after renal transplantation. Am. J. Transplant. 2002;2(2):173–178. doi: 10.1034/j.1600-6143.2002.020209.x. [DOI] [PubMed] [Google Scholar]
- 135.Millán O, Brunet M, Campistol JM, et al. Pharmacodynamic approach to immunosuppressive therapies using calcineurin inhibitors and mycophenolate mofetil. Clinical chemistry. 2003;49(11):1891–1899. doi: 10.1373/clinchem.2003.021725. [DOI] [PubMed] [Google Scholar]
- 136.Hoerning A, Wilde B, Wang J, et al. Pharmacodynamic Monitoring of Mammalian Target of Rapamycin Inhibition by Phosphoflow Cytometric Determination of p70S6 Kinase Activity. Transplantation. 2014 doi: 10.1097/TP.0000000000000273. doi:10.1097/TP.0000000000000273. [DOI] [PubMed] [Google Scholar]
- 137.Sanquer S, Breil M, Baron C, Dhamane D, Astier A, Lang P. Induction of inosine monophosphate dehydrogenase activity after long-term treatment with mycophenolate mofetil. Clinical pharmacology and therapeutics. 1999;65(6):640–648. doi: 10.1016/S0009-9236(99)90085-1. doi:10.1016/S0009-9236(99)90085-1. [DOI] [PubMed] [Google Scholar]
- 138.Vafadari R, Quaedackers ME, Kho MM, et al. Pharmacodynamic analysis of tofacitinib and basiliximab in kidney allograft recipients. Transplantation. 2012;94(5):465–472. doi: 10.1097/TP.0b013e3182626b5a. doi:10.1097/TP.0b013e3182626b5a. [DOI] [PubMed] [Google Scholar]