Abstract
Tuberous sclerosis complex (TSC) is an important cause of epilepsy and autism, as well as renal and pulmonary disease in adults and children. Affected individuals are subject to hamartomas in various organ systems which result from constitutive activation of the protein kinase mTOR (mammalian target of rapamycin). The clinical course, prognosis and appropriate therapy for TSC patients are often different from that for individuals with epilepsy, renal tumors, or inter-stitial lung disease, from other causes. Additionally, TSC serves as a model for other conditions in which the mTOR pathways are also up-regulated. This article reviews the molecular pathophysiology and management of neurological, renal and pulmonary manifestations of the disorder. The use of mTOR inhibitors such as rapamycin and everolimus is discussed and recent clinical trials of these drugs in TSC are reviewed.
Keywords: tuberous sclerosis, angiomyolipoma, epilepsy, lymphangioleiomyomatosis
Introduction
Tuberous sclerosis complex is an autosomal dominant genetic disorder with a birth incidence of 1:6000. An estimated 1 million individuals are affected worldwide, involving all racial and ethnic groups. Classically identified by Vogt's triad of facial angiofibromas, mental retardation, and intractable epilepsy, no more than 30 – 40 % of affected individuals have all 3 of these features [26]. Increased recognition of the disorder has led to a truer picture of its clinical manifestations. Tuberous sclerosis presents most often with neurological symptoms. Up to 90 % of affected individuals experience epileptic seizures. Cognitive impairment, autism, and other behavioral disorders are present in up to 50 – 60 %. Renal manifestations are the next most common, with angiomyolipomas (AMLs) occurring in 80 % of patients. Pulmonary involvement is the third most common cause of TSC-associated morbidity. Lymphangioleiomyomatosis (LAM) occurs in 35 – 40 % of TSC patients, or less often in persons with mosaicism for a TSC genetic mutation (sporadic LAM). TSC can affect virtually any organ system in the body [22, 121]. Renal and pulmonary manifestations are reviewed separately later in this article.
Significant progress has been made in understanding the molecular pathology of TSC. Tuberous sclerosis occurs due to an inactivating mutation in either the TSC-1 gene (protein – hamartin, identified 1997, 9) or the TSC-2 gene (tuberin, 16, identified 1993) [32, 112]. Hamartin and tuberin form a complex which activates the GTPase activating protein Rheb to inhibit the mammalian target of rapamycin (mTOR) [125] (Fig. 1). mTOR is a highly conserved protein kinase which regulates protein synthesis, cellular metabolism, differentiation, growth, and migration (Fig. 1). mTOR in fact exists as 2 complexes with differing functions. mTOR complex 1 (mTORC 1) has a cofactor known as Raptor (regulatory associated protein of mTOR) which activates mTOR's protein kinase domain. This results in increased mRNA transcription and protein synthesis. When a TSC mutation is present a source of inhibition of mTORC1 is absent. This causes constitutive activation of mTOR and, in turn, abnormal cellular proliferation and differentiation, producing the hamartomatous lesions of TSC. mTOR Complex 2 (mTORC 2) has a different co-factor known as Rictor (rapamycin insensitive component of mTOR), is unaffected by Rheb, and regulates protein synthesis in a manner distinct from mTORC1. Activation of mTOR is also important in the propagation of several non- TSC human malignancies [72] and in neurological diseases as diverse as Huntington's chorea [95], Alzheimer's disease [86, 119], and ceroid lipofuscinosis [13]. mTORC 1 and 2 have been shown to regulate the synthesis and turnover of glutamate receptors of neurons and alter the morphology of dendritic spines, thereby affecting processes of long-term potentiation that are crucial for epileptogenesis, learning, memory, and consciousness [44, 93, 108].
Fig. 1.
The mTOR pathway. mTOR1 and mTOR2 are distinguished by their co-factors – raptor and rictor respectively. The tuberous sclerosis gene products function through the GAP protein Rheb (Ras homolog enhanced in brain) to inhibit mTOR, and thereby protein synthesis, cellular growth and division. Rapamycin mimics this by causing the dissociation of raptor from mTOR, resulting in inactivation of the complex. mTOR1 is also inhibited by nutrient deprivation (by courtesy of G. Thomas, Ph.D).
Regulation of mTOR involves multiple intracellular signaling pathways triggered by various membrane receptors and channels, nutrient and energy availability, cellular stress, and feedback mechanisms [22, 49] (Fig. 1). Several pharmacological inhibitors of mTOR exist, including rapamycin (sirolimus), RAD001 (everolimus), and CCI-779 (temsirolimus). These agents work by dissociating mTORC1 from its co-factor Raptor, thereby inactivating it. They have no effect on mTORC2, although agents are under development which inhibit both mTOR complexes. Rapamycin was initially identified in 1975 as an macrocyclic antifungal agent [102] and is used for immunosuppression in transplantation [2] and to limit stenosis of cardiac stents. Everolimus is approved for the treatment of renal cell carcinoma and also for immunosuppression. Temsirolimus is a pro-drug for rapamycin, is administered parenterally, and is indicated for renal cell carcinoma. These agents have obvious potential therapeutic application in TSC which shall be addressed later in this review.
Neurological Manifestations
The hallmark of TSC is the cortical tuber: hamartomas occurring most frequently at the junction of gray and whiter matter. They may involve the entire hemisphere, producing hemimegalencephaly, and often radiate toward the central white matter and ventricular surface. Histologically they consist of bizarre appearing giant neurons and astrocytes, with hypomyelination and disruption of the normal cortical architecture (Fig. 2, 3). Cortical tubers and other TSC hamartomas arise from an inherited or a spontaneous mutation inactivating one of the TSC genes, as well as the occurrence of a second inactivating mutation in one of the other alleles for the TSC genes (“ second hit ”). This means every cell in the brain or body is haploinsufficient for hamartin or tuberin, which causes microanatomic and functional abnormality even in anatomically normal appearing cortex. Seizures are the most common cause of morbidity in individuals with TSC, and poor seizure control is highly associated with developmental delays and autism [9, 54, 55]. Individuals with TSC benefit from aggressive antiepileptic treatment and provision of the highest possible degree of seizure control. Recent work has demonstrated improved development when infantile spasms or partial epilepsy is controlled [42, 50]. Persons with tuberous sclerosis tend to present with either infantile spasms or partial seizures; the latter can appear generalized owing to rapid secondary bilateral synchrony.
Fig. 2.
Large calcified area of cortical dysplasia in the left medial frontal lobe. This area grew in size over 1½ – 2 years, herniating across the mid-line. This was associated with a clinical reduction in seizure frequency. The lesion then regressed spontaneously over the next several years (cranial MRI).
Fig. 3.
Multifocal areas of abnormal T 2 -weighted signal at the gray-white junction represent cortical tubers. Dystrophic calcification is present in the frontal white matter, left greater than right. Prominent calcifications led to consideration of congenital infection and possible child abuse as possible diagnoses, before the child was recognized as having TSC (cranial MRI).
Infantile spasms are present in 30 – 40 % of TSC patients. Earlier onset portends a worse prognosis for developmental delays and subsequent intractable partial epilepsy. They are the presenting sign of the disorder in as many as 15 – 20 % of affected individuals. Identification of infantile spasms in children with tuberous sclerosis may be hampered by the fact that they are frequently not associated with a “ classic ” hypsarrhythmic pattern on EEG. In view of both their frequent association and potentially devastating developmental outcome, infantile spasms can be diagnosed and treated based solely on clinical grounds in TSC patients, even in the absence of classical EEG findings [25, 110]. Vigabatrin has considerable efficacy in the treatment of infantile spasms associated with tuberous sclerosis. A published meta-analysis has identified a 95 % response rate in this clinical setting [45]. Unfortunately, identification of a 30 – 40 % incidence of visual field constriction [68, 75] has limited its use. The exact incidence of visual field constriction in children is unclear, owing to their inability to cooperate with perimetry, but may well be lower than adults. Even normally intelligent adults with visual field loss due to vigabatrin are typically unaware of its presence until detected by routine screening and do not generally complain of impaired functioning in daily life [96, 113]. In children with TSC and infantile spasms, a modest risk of functionally inconsequential visual field constriction contrasts favorably with the high probability of permanent developmental handicap if spasms are not controlled. Vigabatrin is considered the drug of choice for infantile spasms in TSC [48, 110]. In addition to the visual field constriction mentioned above, insomnia, agitation, and constipation can occur in 5 – 10 % [12]. Adrenocorticotrophic hormone (ACTH) as either a gel or aqueous solution administered intramuscularly, and synthetic oral formulations like prednisone, are considered second-line for infantile spasms.
Our own practice is to treat infantile spasms with vigabatrin as a first-line therapy. Valproate and topiramate have efficacy for infantile spasms and may be added to vigabatrin when infantile spasms prove refractory to it as a single agent [12, 38]. Benzodiazepines, such as nitrazepam or clonazepam, are rarely effective as single agents for infantile spasms in TSC and are primarily used as adjunctive therapy or as rescue medications [110].
In contrast to infantile spasms and TSC, only a few relatively small studies to date have specifically looked at anticonvulsant efficacy and tolerability for other types of seizures in TSC. Using topiramate 10 – 60 mg/kg/d as add-on therapy for TSC patients with refractory epilepsy, 3 of 14 became seizure-free and another 6 had seizure frequency reduced by greater than 50 % [70]. Topiramate, zonisamide and acetazolamide inhibit carbonic anhydrase, can aggravate preexisting cognitive impairments, and are inappropriate for individuals with polycystic kidney disease, as nephrolithiasis in this setting could cause acute renal failure [66].
A separate analysis of patients treated with lamotrigine found 24 of 57 (42 %) patients became seizure free and another 21 (37 %) experienced > 50 % reduction in seizures [19, 39]. Lamotrigine is significantly affected by intercurrent anticonvulsant therapy and many individuals had trough serum lamotrigine levels far lower than expected on the basis of their dosage in terms of mg/kg/day, making performance of trough serum levels useful.
Felbamate is a potent antiepileptic agent with primarily anti-glutamatergic properties. In our experience it can be very helpful for patients with tuberous sclerosis, possibly due to increasing evidence suggesting that mTOR may regulate glutamate receptor density of neurons [44, 70, 116]. Use of felbamate is hampered by an approximately 1:10 000 risk of irreversible hepatic failure and aplastic anemia. Nonetheless, felbamate can be a very useful anticonvulsant in individuals with intractable partial seizures, (particularly when combined with vigabatrin [88]). Insomnia and anorexia are more frequent but less serious side-effects.
Sedating anticonvulsants such as barbiturates and benzodiazepines have a limited role in tuberous sclerosis patients and generally should be avoided. One possible exception to this is triple bromide suspension, which can prove effective in medically intractable epilepsy, particularly in those who are not candidates for epilepsy surgery [107, 117].
Growing evidence suggests that mTOR inhibitors may be helpful in the management of epilepsy for patients with TSC. The excita-tory neurotransmitter glutamate as well as subclinical epilepti-form discharges increase the activity of mTOR. This in turn affects the synthesis and density of glutamate receptors and the morphology of neuronal dendritic spines [52, 115] as well as voltage-dependent potassium channels [93, 108, 118]. Additionally, mTOR regulates synaptic long-term potentiation and depression [79, 118], both processes known to be involved with epileptogenesis, as well as learning and memory formation. Several investigators have reported improvement in learning deficits, seizure frequency, motor development, and survival in TSC conditional knock-out mice following treatment with either everolimus or rapamycin [31, 79, 123]. Rapamycin also inhibits epileptogenesis in non-TSC animal models of post-traumatic epilepsy [124]. There have been reports of significant reduction in seizure frequency in TSC patients treated with rapamycin [41, 83]. An open label clinical trial of everolimus for subependymal giant cell astrocytomas in TSC demonstrated an overall 86 % reduction in seizures (p = 0.022) as measured on 24-h video EEG in those patients with concurrent partial epilepsy [64]. Improvements were also noted in normal appearing white matter on diffusion tensor imaging pre- and post-treatment with everolimus [111]. An open label trial of everolimus for epilepsy in TSC is currently underway in the US (www.clinicaltrials.gov, NCT01070316) and a placebo-controlled study is planned.
As many as 20 % of individuals with TSC develop medically intractable partial epilepsy. For these patients, the ketogenic diet, vagus nerve stimulation, and resective epilepsy surgery are increasingly utilized [62]. Recent work suggests that carbohydrate restriction alone (i. e., modified Atkin's ® or low glycemic index diet) may produce benefits equal to that of the classical ketogenic diet. Since carbohydrate depletion serves to inhibit mTOR [89], these diets may have particular relevance for tuberous sclerosis patients. In one series of TSC patients with refractory epilepsy, 11 of 12 patients placed on a ketogenic diet experienced > 50 % reduction in seizures, in spite of the fact that multiple traditional anticonvulsants had failed previously [20]. The diet is often associated with improved cognition, which can relate to improved seizure control as well as elimination of inter-current antiepileptic drugs [61, 90].
Traditionally avoided in patients with tuberous sclerosis, epilepsy surgery has an increasing role in the disorder [51]. Novel techniques such as magnetoencephalography and co-registered magnetic resonance and positron emission tomography scans are increasingly able to localize epileptogenic tubers or adjacent cortex [17]. Many patients have a “ dominant ” tuber or zone of cortical dysplasia causing the bulk of their epileptic activity. These patients are particularly suitable for resective surgery, as are individuals with TSC and intractable epilepsy in the setting of hemimeganencephaly or markedly asymmetric cortical dysplasia. Recent work has also described successful surgical treatment of multifocal partial epilepsy in TSC. Sometimes removal of a primary epileptic focus may cause apparent disruption of an underlying “ epileptic network ” with resulting improvement despite the presence of multiple areas of epileptic cortex. Alternatively some centers employ a “ staged ” technique in which what appears to be the primary focus is resected, cortical grids are replaced, and then further monitoring is undertaken. If appropriate, additional foci are subsequently removed provided that this can be done without unacceptable risk of post-operative deficits. In the largest series of surgical epilepsy patients to date, 37 of 70 (53 %) were seizure-free and another 8 of 70 (11 %) had significantly reduced seizure frequency [73]. These findings were consistent with earlier reports which found 68 – 69 % TSC patients seizure-free following resection [92]. Epilepsy surgery should be considered for all TSC patients with seizures refractory to medical treatment.
Neurocognition
Tuberous sclerosis is frequently associated with cognitive and behavioral impairments. As many as 25 % of TSC patients are autistic and more than half have learning difficulties. Aggressive and obsessive behaviors, as well as other psychological and psychiatric problems are also very common; one recent study of adults with TSC reported a prevalence of 42. Patients with TSC tend to fall into one of 2 groups with regard to their cognitive function. The majority (60 – 70 %) have a normal or nearly normal full scale IQ, with a mean value slightly lower than that of the general population. These individuals nonetheless often have specific cognitive defects relating to shifting attention, working memory, and multi-step reasoning. They also have an increased incidence of obsessive-compulsive behaviors, social and generalized anxiety, and awkwardness in social situations. These attributes can be quite disabling in everyday life despite a normal IQ. The remaining 30 – 40 % of people with TSC have profound mental retardation, with full scale IQs of 40 or less. These persons are more likely to have a history of infantile spasms, intractable epilepsy, and seizure onset before age 1 year [91, 92]. Poorly controlled or sub-clinical seizure activity frequently causes behavioral and learning problems. This is often heralded by a regression in developmental milestones, learning, or behavior. These same symptoms should also raise the suspicion of a growing giant cell astrocytoma with evolving hydrocephalus. Adequate seizure control as well as evaluation for hydrocephalus/tumor are therefore the first consideration in TSC patients with behavioral or cognitive difficulties, particularly in the setting of a recent decline. In many, epileptiform activity is not apparent clinically and is revealed only by video-EEG monitoring.
Subependymal Giant Cell Astrocytomas
Subependymal giant cell astrocytomas (SEGAs) are observed in between 5 and 15 % of patients with tuberous sclerosis, almost exclusively within the first 2 decades of life [24, 43]. Histologically indistinguishable from subependymal nodules, SEGAs typically occur in the region of the foramen of Monro either unilaterally or bilaterally, and are identified by serial growth or association with hydrocephalus on neuroimaging studies [7, 40]. SEGAs only rarely invade the brain parenchyma inferomedially in the region of the thalamus and basal ganglia. More typically, the tumor projects into the ventricle and can produce acute or subacute hydrocephalus by causing obstruction of cerebrospinal fluid through the foramen of Monro (Fig. 4). They are slowly growing and may reach considerable size before producing symptoms, leading to recommendations for periodic neuroim-aging (i. e., every 1 – 3 years) to screen for their occurrence, even in the absence of symptoms [10, 48].
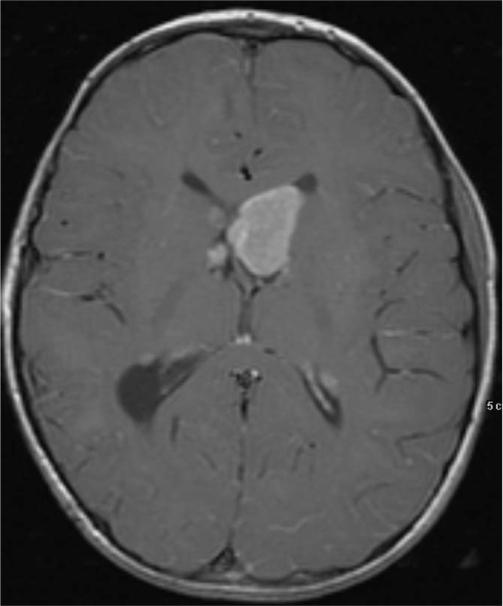
Fig. 4.
Subependymal giant cell astrocytoma in the region of the left foramen of Monro. Once these lesions begin to grow they inexorably continue to do so, eventually producing hydrocephalus (cranial MRI).
The current standard therapy of SEGAs is operative resection. It is important to achieve a gross total resection, as any residual astrocytoma tissue almost certainly will recur [24, 43]. Lesions typically are removed by either a trans-frontal or midline transcallosal approach. While favored to limit damage to the cortex, the trans-callosal method can make gross total resection difficult for larger, bilateral, and more laterally-located SEGAs. We recently described a technique involving creation of an operative corridor by progressive dilation of a balloon catheter placed stereotactically in the region of a subependymal lesion, thereby allowing adequate operative exposure for gross total resection in the vast majority of cases with minimal disruption of surrounding tissues [71].
Although most SEGAs are amenable to surgical resection, some lesions exhibit more aggressive behavior or serial regrowth, are associated with extensive peritumoral edema, or occur in atypical locations such as the pineal or hypothalamic regions. These factors, plus an appreciation that the mTOR inhibitor rapamycin has demonstrated activity against renal angiomyolipomas in both animal models and human clinical trials [7], have led to the clinical use of rapamycin for therapy of subependymal giant cell astrocytomas (Fig. 4). In a report of 5 cases of SEGA in patients with TSC, all tumors reduced significantly in size in response to treatment with rapamycin and one particularly large hypothalamic lesion exhibited central necrosis prior to involution [40]. Overall, an average reduction in astrocytoma volume of 65 % was observed. Surgery was not required in any of the patients and the fairly mild side effects observed included aphthous ulcers, hypercholesterolemia, and extremity edema. Rapamycin was initiated at 3 mg/m 2/day and titrated upward to achieve trough serum levels of 10 – 15 ng/mL. We recently completed an open label trial of everolimus for SEGA in 28 subjects with TSC. 80 % of the 28 patients enrolled also have epilepsy. The median age of study participants was 12.4 years (range 3 – 34 years). Males outnumbered females (17 male vs. 11 female). At interim analysis SEGA volume decreased in every subject. Overall reduction in tumor volume was 34 % at 3 months, 49 % at 6 months, and 55 % at 1 year. One year follow-up data are available on all subjects. As noted patients with epilepsy demonstrated an 87 % reduction (p = 0.022) in partial-onset seizures after 6 months on study medication. 13 of 15 treated patients reported a reduction in the size and erythema of their facial angiofibromas. This is consistent with published reports of TSC patients experiencing improvements in angiofibromas after systemic or topical rapamycin. Everolimus was well tolerated with subjects reporting an average of 0.62 adverse events for each patient-month of treatment. 4 serious adverse events were reported, none of them clearly related to study medication. No patients have exited the study because of an adverse event. No correlation was seen between serum level and reduction in tumor volume, although higher levels were associated with a greater incidence of side effects. A multi-center placebo-controlled trial of everolimus for SEGA recently completed enrollment. Applications have been made to the European Medicines Agency (EMEA) as well as the US Food and Drug Administration (FDA) for approval of everolimus as a therapy for SEGA.
TSC-Associated Renal Disease
Renal TSC phenotypes develop because of abnormal cell growth and proliferation resulting in cysts and solid tumors including angiomyolipomas. Steps that should be taken to slow progression include avoiding nephrotoxic agents such as non-steroidal anti-inflammatory and anticonvulsant drugs, overexposure to imaging contrast, and urological procedures that remove functioning kidney tissue.
Angiomyolipoma
Epidemiological studies involving TSC patients revealed an increase in angiomyolipomas during childhood and adolescence that then stabilized throughout adulthood [78]. A longitudinal study, demonstrated that 55 % of children (mean age 6.9 years) had some type of renal abnormality, and at follow-up 80 % (mean age 10.5 years) had abnormalities [106]. By far the most common form of involvement was angiomyolipomas which demonstrated growth in 56 % of boys and 66 % of girls (Fig. 5). The youngest patient demonstrating abnormal renal lesions in this study was 2 years old, but lesions greater than 4 cm were found only in post-pubertal patients. The authors concluded that renal involvement begins in infancy and increases with age. Studies have also demonstrated that the incidence of angiomyo lipomas in patients with tuberous sclerosis complex increased with age [4, 58], and that rapid growth occurs in childhood and adolescence with slower growth into adulthood [69, 94].
Fig. 5.
Non-contrast computer tomography image demonstrating a large left renal angiomyolipoma and hemorrhage.
Patients with TSC are at risk from hemorrhage of their angiomyolipomas as well as encroachment on normal renal parenchyma leading to chronic kidney disease and eventually end-stage renal disease. Larger angiomyolipomas frequently develop both micro-and macroaneurysms that can rupture and hemorrhage [85, 105]. The risk of hemorrhage from renal angiomyolipomas in patients with TSC is estimated to be between 25 and 50 % [15]. Up to 20 % of such patients with a hemorrhage present with shock [59]. The risk of hemorrhage is proportional to the size of the aneurysm, and aneurysms larger than 5 mm have the greatest risk [120]. Unfortunately, in centers with limited experience in treating TSC renal lesions, patients with suspected retroperitoneal hemorrhage may undergo unnecessary nephrectomy for diagnostic and therapeutic purposes. Such a loss of renal mass often hastens the requirement for renal replacement therapy.
Renal angiomyolipomas also insidiously impinge upon normal renal tissue leading to renal failure [18, 101]. Using death certificates for TSC clinic patients Shepherd et al. found that of their patients at the Mayo clinic, renal failure was the most common cause of death [104]. Given the current world population, the incidence of TSC, and the frequency of renal involvement, approximately 500 000 patients with TSC worldwide have at least chronic kidney disease (CKD) stage 1. Considering the relentless progression of CKD and the inherent increased risk in morbidity and mortality, CKD poses a significant burden on patients with TSC [36].
Angiomyolipomas should be followed by imaging studies on an annual or semi-annual basis. Although ultrasound is adequate for detecting the adipose component of angiomyolipoma, the solid components can be difficult to recognize. MRI is superior to ultrasound or CT scan, especially given the recent concern about the risk of radiation. The purpose of serial imaging is to lower the risk of aneurysm rupture and hemorrhage, for which the treatment modality of choice is embolization. Urological procedures that expose the kidney to ischemia, either warm or cold, have a greater risk of damage to surrounding normal renal parenchyma, and a higher rate of complications in general. These are only infrequently beneficial, due to the risk of progression of chronic kidney disease.
Renal cystic disease
Renal cystic disease occurs in approximately 45 % of patients with TSC and occurs in both TSC1 and TSC2 related disease [4, 11, 27]. Cystic disease varies from microcystic disease that is not detectable by imaging studies, to a polycystic phenotype that can even be severe. This severe, very early onset polycystic phenotype is associated with contiguous deletions involving TSC2 and the adjacent PKD1 gene on chromosome 16p13, occurring in approximately 2 % of TSC patients. Such patients often exhibit mosaicism for the genomic deletions [11, 27] and can develop significant renal insufficiency as teenagers [99]. Reminiscent of the Drosophila gigas eye cells [84] that also lack Tsc2, the epithelial cells lining the TSC cysts are both hypertrophic and hyperplastic with strongly eosinophilic cytoplasm [3]. Blood pressure control is a cornerstone for renal function preservation in the TSC patient. Using older studies to estimate the loss of renal function in cystic disease is flawed because patients are now treated with long-acting angiotensin-converting enzyme inhibitors and angiotensin receptor blockers that slow the loss of renal function [6]. TSC-related renal cystic disease also has a cilial association (Siroky and Bissler, unpublished data) similar to other forms of polycystic kidney disease including other tumor suppressor-associated diseases [1]. Glomerulocystic kidney disease associated with tuberous sclerosis complex is also well documented [5]. In one study among 10 patients with tuberous sclerosis complex, 3 infants and one adolescent exhibited unilateral glomerulocystic kidney disease. Like the tubular derived cysts in TSC, the glomerular spaces exhibited large, bizarre parietal epithelial cells piled up against Bowman's capsule and the glomerular tufts exhibited similarly atypical podocytes [8].
The hallmark of renal cystic disease in TSC is the development of hypertension, and rigorous control of blood pressure using angiotensin-converting enzyme inhibitors and angiotensin receptor blockers is the cornerstone of therapy. Furthermore, patients with significant cystic disease also can exhibit impaired urinary concentration, placing them at risk for significant volume depletion with such conditions as gastroenteritis. Cystic renal disease also can predispose to renal hemorrhage because a cyst can effectively function as a hydraulic wedge and disrupt renal parenchyma following a fall, such as from a seizure.
Fat-poor renal lesions
Solid renal lesions in patients with TSC often prove to be fat-poor angiomyolipomas, but also can be oncocytomas, or renal cell carcinomas. Fat-poor TSC-associated renal angiomyolipomas are typically composed of spindle cells, vascular elements, or epithelioid cells. The composition of the different cellular components can vary from lesion to lesion [57, 74]. Phenotypically epithelioid cells can range from a polygonal cell with a slight degree of nuclear atypia, to an atypical cell of variable size and with significant mitotic activity. Angiomyolipomas often contain patches of spindle cells that exhibit nuclear atypia with mitotic figures, but these tumors typically do not behave in a malignant fashion. Very rarely, renal angiomyolipomas can infiltrate the surrounding tissue [34, 84].
The term “ epithelioid angiomyolipoma ” refers to a tumor that has been reported to exhibit a variety of behaviors, from an aggressive phenotype [80] that recurs after resection and results in death, to a much more benign course [77]. The pathological distinction between epithelioid angiomyolipomas and malignant angiomyolipomas is unclear [30, 53, 57, 74].
There are several case reports of TSC patients with renal oncocytomas [23]. Histologically, oncocytomas contain dense eosinophilic cytoplasm, that may be exclusively or predominantly granular, generally uniform nuclei, and abundant mitochondria. The intercalated cell of the cortical portion of the collecting tubule is often thought to be the cell of origin for oncocytomas [29]. The actual incidence of renal cell carcinoma (RCC) in TSC is not known, but is estimated to be less than 2 % of TSC patients. Some studies suggest that renal carcinomas in TSC patients occur at an average age of 28 years, which is 25 years younger than the average age of RCC in the general population, and there are reports in children and infants with TSC [1]. However, in a series of 209 patients, the median age of diagnosis for renal cell carcinoma in patients with TSC was 50 years of age [67]. The literature for RCC in TSC is confusing for several reasons. First, the typical hematoxylin and eosin appearance of RCC is very similar to that of the epithelioid variant of angiomyolipoma [103], and all the reported cases of RCC have not been studied in sufficient detail to exclude misclassifications of atypical angiomyo lipomas such as the epithelioid variety [21]. HMB-45 and melanin A, diagnostic markers for angiomyolipomas, are extremely useful for differentiating between RCC and the epithelioid variety of angiomyolipoma in TSC. In children where renal cell carcinoma is diagnosed histologically, cytogenetic analysis is required to distinguish the translocation morphology renal cell carcinoma. These tumors have translocations involving the transcription factor EB [t(6;11)], and are also HMB45-positive [87]. This latter RCC occurs in pediatric patients but is not known to be associated with TSC.
Angiomyolipoma classification, cell biology, and clinical trials
Angiomyolipomas are prototypical for a family of tumors that exhibit perivascular epithelioid-cell differentiation, collectively called PEComas. This family of tumors includes lymphangioleiomyomatosis, clear-cell ‘ sugar ’ tumors of the lung, and a group of rare morphologically and immunophenotypically similar lesions that can arise from a variety of soft-tissue and visceral sites. Most PEComas exhibit immunoreactivity for both melanocytic markers (as detected by the HMB-45 and melanin-A antibodies) and smooth-muscle markers (actin and desmin). The origin of the perivascular epithelioid cells is unknown. Because PEComas express melanocytic proteins, and melanocytes are neural crest-derived, it has been postulated that neural crest-lineage cells give rise to angiomyolipomas [35, 98]. Neural crest lineage cells do contribute to renal development. Alternatively, it has been proposed that angiomyolipomas arise from a renal mesenchymal precursor cell [100]. Supporting this latter contention is the fact that rtPCR can detect mRNA for gp100, the antigen target of HMB-45 in both proximal and distal tubules, although the protein product is below the level of detection of immunohisto-chemical analysis with HMB-45 [47].
Recent published reports suggest that mTORC1 inhibition causes a reduction in volume of renal angiomyolipomas, which tends to return to baseline values when treatment is stopped [7, 28]. Current research examining both the natural history of TSC renal lesions (NCT00598455) and optimization of mTORC1 inhibition dosing (NCT00457964) are underway, as is a safety and efficacy of trial aimed at obtaining FDA approval (NCT00790400).
TSC-Associated Pulmonary Manifestations
Lymphangioleiomyomatosis (aka lymphangiomyomatosis or LAM) is the most common pulmonary manifestation of TSC [16] and the third most common cause of TSC-related death [114]. LAM is characterized by diffuse infiltration of the lung with atypical smooth muscle cells and gradual cystic replacement of the pulmonary parenchyma. Tuberous sclerosis associated-LAM (TSC-LAM) occurs almost exclusively in female TSC patients, although a few males with biopsy documented TSC-LAM [76] and clinical diagnoses of TSC-LAM have been reported. TSC-LAM is most frequently diagnosed in women of child-bearing age, but cases have been described prior to puberty and as late as 70 years of age [80]. Radiographic surveys of 3 large TSC clinic populations have revealed cystic changes consistent with LAM in about 26 – 48 % of female patients, most of whom are asymptomatic. Despite the potential to identify the presence of TSC-LAM through screening, most patients are not diagnosed until they become symptomatic, at an average of 30 years [37]. Although periodic screening for patients with negative initial scans may also be advisable, there are no published guidelines. There may be as many as 150 000 patients with TSC-LAM worldwide, based on the estimated total population prevalence of 1/6 000 individuals with TSC, but most remain undiagnosed. The natural history of LAM identified through screening has not been well characterized, but typical manifestations in patients with moderately profuse cystic change in the lung include progressive dyspnea on exertion and recurrent pneumothorax. LAM can also be associated with axial, abdominal and retroperitoneal lymphadenopathy and occasionally with cystic abdominal lymphangiomyomas [37, 97]. Although there is no known effective treatment for LAM, awareness of the diagnosis facilitates informed decisions regarding pregnancy, birth control use, air travel, vaccination, management of pneumothorax and enrollment in clinical trials.
Sporadic lymphangioleiomyomatosis
LAM can also occur in women who do not have TSC. This sporadic form of LAM (S-LAM) is associated with somatic rather than germ-line mutations in TSC genes, and is characterized by onset in the second or third decade of life, a later average age at diagnosis (approximately 35 years), and presentation with shortness of breath, pneumothorax, chylous effusion or incidental discovery on CTs obtained for coronary screening or abdominal or chest pain. S-LAM is often associated with renal angiomyolipomas in about 32 % of patients (vs. 93 % in TSC-LAM), and with lymphangioleiomyomas in about 30 % [77] (vs. 9 % in TSC-LAM), but never with central nervous system or dermato-logical manifestations. Lymphatic complications, such as chylous pleural effusion, are much more common in S-LAM than TSCLAM. Although TSC-LAM is predicted to be 5- to 10-fold more common than S-LAM, patients with S-LAM who present to adult clinics, and enroll in registries and clinical trials, outnumber those with TSC-LAM by more than 5 – 10:1. The 2 leading hypotheses for this paradox are that discovery of TSC-LAM through screening identifies a population of patients with early disease, or, alternatively, that TSC-LAM is a less aggressive disease than S-LAM. Only 1 male with S-LAM has been reported to date [60].
Pathogenesis of LAM
4 lines of evidence suggest that LAM is a metastatic neoplasm. (i) Lymphatic spread is well documented in LAM, both by the frequent occurrence of lymphadenopathy associated with LAM cell infiltration, and the identification of LAM cell clusters in the lymphatic lumen and within pleural effusions [65]. (ii) Hematogenous spread is suggested by the finding of cells containing loss of heterozygosity (LOH) for TSC genes in blood [23]. (iii) LAM recurs in transplanted lungs and genetic studies have demonstrated that the cells comprising the lesions originate from the recipient, the mutational profiles of cells comprising LAM lesions from lymph nodes, kidneys and lungs all show a common origin [56]. The source of LAM cells in the blood, lymphatics and lung is not clear, but renal angiomyolipomas and the uterus have been cited as possible candidates. The role of lymphangiogenesis in the pathogenesis of LAM is an area of intense interest [46].
Radiographic features of LAM
The chest radiograph is useful for the diagnosis of pneumothorax and chylothorax, but is often otherwise uninformative in LAM; it can be quite normal even in patients with moderately severe dyspnea. Later in the disease, bilateral, symmetrical reticular and cystic changes can become visible, sometimes producing a honeycomb or bullous appearance. High resolution CT scan of the chest is much more useful for detecting cystic parenchymal disease in LAM [63]. The CT shows variable numbers of diffuse, bilateral thin walled cysts ranging from 1 to 50 mm in diameter, as well reticular changes, nodular densities, hilar or mediastinal adenopathy or dilated thoracic duct in some patients [82]. Scattered nodular densities may represent multifocal micronodular pneumocyte hyperplasia: color clusters of hyper-plasic type II pneumocytes that have no known physiological or prognostic significance [14, 33, 81]. Other TSC-associated lung disorders, include pulmonary or mediastinal angiomyolipomas, tumor emboli from angiomyolipoma, and clear cell (“ sugar ”) tumor of the lung may also (rarely) be discovered by chest CT in patients with TSC.
Pathology of LAM
Histological changes are similar in S-LAM and TSC-LAM. Microscopic examination of the lung reveals smooth muscle infiltration of the lung parenchyma, airways, lymphatics, and blood vessels, associated with thin-walled cystic change (Fig. 6). The smooth muscle cells have a benign appearance with a low mitotic rate and both spindle-shaped and epithelioid/cuboidal morphologies [14]. Diffuse nodular proliferation of type II cells indicative of MMPH may occur in patients with TSC of both genders, in the presence and absence of LAM. LAM cells stain with actin, vimentin, desmin and HMB-45, an antibody that recognizes the premelanosomal protein gp-100 [14, 33]. The latter is very useful diagnostically, since smooth muscle cell infiltration due to other diseases such as idiopathic pulmonary fibrosis is not associated with HMB-45 positivity. LAM lesions also stain abundantly with lymphangiogenic markers. Serum VEGF-D (VEGF – vascular endothelial growth factor) is elevated 5- to 8-fold in patients with LAM, and is useful for diagnosis and as a biomarker for response to therapy [103, 122].
Fig. 6.
Diffuse thin walled cysts and bilateral chylous effusions (blue vertical arrows posteriorly) in a patient with lymphangioleiomyomatosis (non-contrast CT).
Diagnostic approach, treatment and trials for LAM
Our own practice is to screen female patients for LAM with non-contrast high-resolution CT scans after menarche and every 3 – 5 years thereafter if the initial scan is negative. VEGF-D may prove to be a useful biomarker for decisions regarding the timing of high-resolution CT to screen for LAM. Pulmonary function testing is performed at the time of diagnosis and at intervals varying from every 3 – 6 months in patients with progressive disease and every 1 – 2 years in patients who have very early or very stable disease. Cystic changes in the lung of female patients with TSC, especially non-smoking patients, are presumed to be due to LAM and lung biopsy is not performed. Pneumothorax is common and likely to recur, (70 % of cases) so mechanical or chemical pleurodesis is recommended on the first event. Talc pleurodesis and pleurectomy are avoided in patients who are candidates for lung transplantation in the future [21]. Bronchodilators may be effective at controlling symptoms in the 15 – 20 % of LAM patients who have reversible airflow obstruction [109]. Oximetry at rest and with exertion should be performed routinely in patients with LAM, because occult desaturation is common, and oxygen should be prescribed to maintain saturations greater than 90 %, along with rest, exercise and sleep. Patients should be vaccinated for influenza and Pneumococcus. In the absence of effective treatments for LAM, patients are often empirically treated with drugs that reduce the level of estrogen or antagonize its action, but no controlled studies have been performed. Lung transplantation may be considered for patients with FEV1 < 30 % and predicted or disabling dyspnea. The 11 LAM patients who were enrolled in the Cincinnati angiomyolipoma sirolimus trial (NCT00457808) had a significant improvement in airflow by spirometry, as well as a reduction in gas trapping [7]. A double-blind placebo-controlled trial of rapamycin for LAM (Multi-center International LAM efficacy of sirolimus, or MILES – NCT00414648) recently completed enrollment and will provide further insight into this.
References
- 1.Al-Saleem T, et al. Malignant tumors of the kidney, brain, and soft tissues in children and young adults with the tuberous sclerosis complex. Cancer. 1998;83:2208–2216. [PubMed] [Google Scholar]
- 2.Augustine J, Bodziak K, Hricik D. Use of sirolimus in solid organ transplantation. Drugs. 2007;67:369–391. doi: 10.2165/00003495-200767030-00004. [DOI] [PubMed] [Google Scholar]
- 3.Bernstein J, Meyer R. In: Parenchymal maldevelopment of the kidney in Brennemann-Kelley Practice of Pediatrics. KeHey V, editor. Harper; New York: 1967. pp. 1–30. [Google Scholar]
- 4.Bernstein J, Robbins TO. Renal involvement in tuberous sclerosis. Ann N Y Acad Sci. 1991;615:36–49. doi: 10.1111/j.1749-6632.1991.tb37746.x. [DOI] [PubMed] [Google Scholar]
- 5.Bernstein J. Glomerulocystic kidney disease–nosological considerations. Pediatr Nephrol. 1993;7:464–470. doi: 10.1007/BF00857576. [DOI] [PubMed] [Google Scholar]
- 6.Bernstein J. Renal cystic disease in the tuberous sclerosis complex. Pediatr Nephrol. 1993;7:490–495. doi: 10.1007/BF00857581. [DOI] [PubMed] [Google Scholar]
- 7.Bissler JJ, et al. Sirolimus for angiomyolipoma in tuberous sclerosis complex or lymphangioleiomyomatosis. N Engl J Med. 2008;358:140–151. doi: 10.1056/NEJMoa063564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bissler JJ, Siroky BJ, Yin H. Glomerulocystic kidney disease. Pediatr Nephrol. 2009 doi: 10.1007/s00467-009-1416-2. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bolton P, et al. Neuro-epileptic determinants of autism spectrum disorders in tuberous sclerosis complex. Brain. 2002;125:1247–1255. doi: 10.1093/brain/awf124. [DOI] [PubMed] [Google Scholar]
- 10.Braffman B, et al. MR imaging of tuberous sclerosis: pathogenesis of this phakomatosis, use of gadopentetate dimeglumine, and literature review. Radiology. 1992;183:227–238. doi: 10.1148/radiology.183.1.1549677. [DOI] [PubMed] [Google Scholar]
- 11.Brook-Carter PT, et al. Deletion of the TSC2 and PKD1 genes associated with severe infantile polycystic kidney disease–a contiguous gene syndrome. Nature Genetics. 1994;8:328–332. doi: 10.1038/ng1294-328. [DOI] [PubMed] [Google Scholar]
- 12.Buoni S, et al. Combined treatment with vigabatrin and topiramate in West syndrome. J Child Neurol. 2004;19:385–386. doi: 10.1177/088307380401900512. [DOI] [PubMed] [Google Scholar]
- 13.Cao Y, et al. Autophagy is disrupted in a knock-in mouse model of juvenile neuronal ceroid lipofuscinosis. J Biol Chem. 2006;281:20483–20493. doi: 10.1074/jbc.M602180200. [DOI] [PubMed] [Google Scholar]
- 14.Carrington CB, et al. Lymphangioleiomyomatosis. Physiologic-pathologic-radiologic correlations. Am Rev Respir Dis. 1977;116:977–995. doi: 10.1164/arrd.1977.116.6.977. [DOI] [PubMed] [Google Scholar]
- 15.Casper KA, et al. Tuberous sclerosis complex: renal imaging findings. Radiology. 2002;225:451–456. doi: 10.1148/radiol.2252011584. [DOI] [PubMed] [Google Scholar]
- 16.Castro M, et al. Pulmonary tuberous sclerosis. Chest. 1995;107:189–195. doi: 10.1378/chest.107.1.189. [DOI] [PubMed] [Google Scholar]
- 17.Chandra P, et al. FDG-PET/MRI coregistration and diffusion-tensor imaging distinguish epileptogenic tubers and cortex in patients with tuberous sclerosis complex: a preliminary report. Epilepsia. 2006;47:1543–1549. doi: 10.1111/j.1528-1167.2006.00627.x. [DOI] [PubMed] [Google Scholar]
- 18.Clarke A, et al. End-stage renal failure in adults with the tuberous sclerosis complex. Nephrol Dial Transpl. 1999;14:988–991. doi: 10.1093/ndt/14.4.988. [DOI] [PubMed] [Google Scholar]
- 19.Collins J, et al. Levetiracetam as adjunctive antiepileptic therapy for patients with tuberous sclerosis complex: a retrospective open-label trial. J Child Neurol. 2006;21:53–57. doi: 10.1177/08830738060210011201. [DOI] [PubMed] [Google Scholar]
- 20.Coppola G, et al. The effects of the ketogenic diet in refractory partial seizures with reference to tuberous sclerosis. Eur J Paediatr Neurol. 2006;10:148–1451. doi: 10.1016/j.ejpn.2006.03.001. [DOI] [PubMed] [Google Scholar]
- 21.Corrin B, Liebow AA, Friedman PJ. Pulmonary lymphangiomyomatosis. A review. Am J Pathology. 1975;79:348–382. [PMC free article] [PubMed] [Google Scholar]
- 22.Crino P, Nathanson K, Henske E. The tuberous sclerosis complex. New Engl J Med. 2006;355:1345–1356. doi: 10.1056/NEJMra055323. [DOI] [PubMed] [Google Scholar]
- 23.Crooks DM, et al. Molecular and genetic analysis of disseminated neoplastic cells in lymphangioleiomyomatosis. Proc Natl Acad Sci USA. 2004;101:17462–17467. doi: 10.1073/pnas.0407971101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Cuccia V, et al. Subependymal giant cell astrocytoma in children with tuberous sclerosis. Childs Nerv Sys. 2003;19:232–243. doi: 10.1007/s00381-002-0700-2. [DOI] [PubMed] [Google Scholar]
- 25.Curatolo P, Verdecchia M, Bombardieri R. Tuberous sclerosis complex: a review of neurological aspects. Europ J Paediatr Neurol. 2002;6:15–23. doi: 10.1053/ejpn.2001.0538. [DOI] [PubMed] [Google Scholar]
- 26.Curatolo P. International Review of Child Neurology Series. Mac Keith Press; 2003. Tuberous Sclerosis Complex: From Basic Science to Clinic Phenotypes. [Google Scholar]
- 27.Dabora SL, et al. Mutational analysis in a cohort of 224 tuberous sclerosis patients indicates increased severity of TSC2, compared with TSC1, disease in multiple organs. Am J Hum Genet. 2001;68:64–80. doi: 10.1086/316951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Davies DM, et al. Sirolimus therapy in tuberous sclerosis or sporadic lymphangioleiomyomatosis. N Engl J Med. 2008;358:200–203. doi: 10.1056/NEJMc072500. [DOI] [PubMed] [Google Scholar]
- 29.Eble JN, Hull MT. Morphologic features of renal oncocytoma: a light and electron microscopic study. Hum Pathol. 1984;15:1054–1061. doi: 10.1016/s0046-8177(84)80249-x. [DOI] [PubMed] [Google Scholar]
- 30.Eble JN, Amin MB, Young RH. Epithelioid angiomyolipoma of the kidney: a report of five cases with a prominent and diagnostically confusing epithelioid smooth muscle component. Am J Surg Pathol. 1997;21:1123–1130. doi: 10.1097/00000478-199710000-00001. [DOI] [PubMed] [Google Scholar]
- 31.Ehninger D, et al. Reversal of learning deficits in a Tsc2 ± mouse model of tuberous sclerosis. Nature Medicine. 2008;14:843–848. doi: 10.1038/nm1788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.European Chromosome 16 Tuberous Sclerosis Consortium Identification and characterization of the tuberous sclerosis gene on chromo-some 16. Cell. 1993;75:1305–1315. doi: 10.1016/0092-8674(93)90618-z. [DOI] [PubMed] [Google Scholar]
- 33.Ferrans VJ, et al. Lymphangioleiomyomatosis (LAM): a review of clinical and morphological features. J Nippon Med Sch. 2000;67:311–329. doi: 10.1272/jnms.67.311. [DOI] [PubMed] [Google Scholar]
- 34.Ferry JA, Malt RA, Young RH. Renal angiomyolipoma with sarcomatous transformation and pulmonary metastases. Am J Surg Pathol. 1991;15:1083–1088. doi: 10.1097/00000478-199111000-00008. [DOI] [PubMed] [Google Scholar]
- 35.Folpe AL, et al. Perivascular epithelioid cell neoplasms of soft tissue and gynecologic origin: a clinicopathologic study of 26 cases and review of the literature. Am J Surg Pathol. 2005;29:1558–1575. doi: 10.1097/01.pas.0000173232.22117.37. [DOI] [PubMed] [Google Scholar]
- 36.Fox CH, et al. A quick guide to evidence-based chronic kidney disease care for the primary care physician. Postgrad Med. 2008;120:E01–E06. doi: 10.3810/pgm.2008.07.1798. [DOI] [PubMed] [Google Scholar]
- 37.Franz D, et al. Mutational and radiographic analysis of pulmonary disease consistent with lymphangioleiomyomatosis and micronodular pneumocyte hyperplasia in women with tuberous sclerosis. Am J Respir Crit Care Med. 2001;164:661–668. doi: 10.1164/ajrccm.164.4.2011025. [DOI] [PubMed] [Google Scholar]
- 38.Franz D, Tudor C, Leonard J. Topiramate as therapy for tuberous sclerosis complex-associated seizures. Epilepsia. 2000;41(Suppl 7):87. [Google Scholar]
- 39.Franz DN, et al. Lamotrigine therapy of epilepsy in tuberous sclerosis. Epilepsia. 2001;42:935–940. doi: 10.1046/j.1528-1157.2001.042007935.x. [DOI] [PubMed] [Google Scholar]
- 40.Franz DN, et al. Rapamycin causes regression of astrocytomas in tuberous sclerosis complex. Ann Neurol. 2006;59:490–498. doi: 10.1002/ana.20784. [DOI] [PubMed] [Google Scholar]
- 41.Franz DN. unpublished observations. [Google Scholar]
- 42.Goh S, et al. Infantile spasms and intellectual outcomes in children with tuberous sclerosis complex. Neurology. 2005;65:235–238. doi: 10.1212/01.wnl.0000168908.78118.99. [DOI] [PubMed] [Google Scholar]
- 43.Goh S. Subependymal giant cell tumors in tuberous sclerosis complex. Neurology. 2004;63:1457–1461. doi: 10.1212/01.wnl.0000142039.14522.1a. [DOI] [PubMed] [Google Scholar]
- 44.Gong R, et al. Roles of glutamate receptors and the mammalian target of rapamycin (mTOR) signaling pathway in activity-dependent dendritic protein synthesis in hippocampal neurons. J Biol Chem. 2006;281:18802–18815. doi: 10.1074/jbc.M512524200. [DOI] [PubMed] [Google Scholar]
- 45.Hancock E, Osborne J. Vigabatrin in the treatment of infantile spasms in tuberous sclerosis: literature review. J Child Neurol. 1999;14:71–74. doi: 10.1177/088307389901400201. [DOI] [PubMed] [Google Scholar]
- 46.Henske EP. Metastasis of benign tumor cells in tuberous sclerosis complex. Genes Chromosomes Cancer. 2003;38:376–381. doi: 10.1002/gcc.10252. [DOI] [PubMed] [Google Scholar]
- 47.Henske EP. Tuberous sclerosis and the kidney: from mesenchyme to epithelium, and beyond. Pediatr Nephrol. 2005;20:854–857. doi: 10.1007/s00467-004-1795-3. [DOI] [PubMed] [Google Scholar]
- 48.Hyman M, Whittemore V. National Institutes of Health consensus conference: tuberous sclerosis complex. Arch Neurol. 2000;57:662–665. doi: 10.1001/archneur.57.5.662. [DOI] [PubMed] [Google Scholar]
- 49.Inoki K, Guan K. Complexity of the TOR signaling network. Trends Cell Biol. 2006;16:206–212. doi: 10.1016/j.tcb.2006.02.002. [DOI] [PubMed] [Google Scholar]
- 50.Jambaqué I, et al. Mental and behavioural outcome of infantile epilepsy treated by vigabatrin in tuberous sclerosis patients. Epilepsy Res. 2000;38:151–160. doi: 10.1016/s0920-1211(99)00082-0. [DOI] [PubMed] [Google Scholar]
- 51.Jansen F, et al. Epilepsy surgery in tuberous sclerosis: a systematic review. Epilepsia. 2007;48:1466–1484. doi: 10.1111/j.1528-1167.2007.01117.x. [DOI] [PubMed] [Google Scholar]
- 52.Jaworski J, Sheng M. The growing role of mTOR in neuronal development and plasticity. Mol Neurobiol. 2006;34:205–219. doi: 10.1385/MN:34:3:205. [DOI] [PubMed] [Google Scholar]
- 53.Jimenez RE, et al. Concurrent angiomyolipoma and renal cell neoplasia: a study of 36 cases. Mod Pathol. 2001;14:157–163. doi: 10.1038/modpathol.3880275. [DOI] [PubMed] [Google Scholar]
- 54.Joinson C, et al. Learning disability and epilepsy in an epidemiological sample of individuals with tuberous sclerosis complex. Psychol Med. 2003;33:335–344. doi: 10.1017/s0033291702007092. [DOI] [PubMed] [Google Scholar]
- 55.Jozwiak S, Goodman M, Lamm S. Poor mental development in patients with tuberous sclerosis complex: clinical risk factors. Arch Neurol. 1998;55:379–384. doi: 10.1001/archneur.55.3.379. [DOI] [PubMed] [Google Scholar]
- 56.Karbowniczek M, et al. Recurrent lymphangiomyomatosis after transplantation: genetic analyses reveal a metastatic mechanism. Am J Respir Crit Care Med. 2003;167:976–982. doi: 10.1164/rccm.200208-969OC. [DOI] [PubMed] [Google Scholar]
- 57.Karbowniczek M, Yu MJ, Henske EP. Renal angiomyolipomas from patients with sporadic lymphangiomyomatosis contain both neo-plastic and non-neoplastic vascular structures. Am J Pathol. 2003;162:491–500. doi: 10.1016/S0002-9440(10)63843-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Kennelly MJ, Grossman HB, Cho KJ. Outcome analysis of 42 cases of renal angiomyolipoma [see comments]. J Urol. 1994;152:1988–1991. doi: 10.1016/s0022-5347(17)32286-3. [DOI] [PubMed] [Google Scholar]
- 59.Kessler OJ, et al. Management of renal angiomyoli poma: analysis of 15 cases. Eur Urol. 1998;33:572–575. doi: 10.1159/000019658. [DOI] [PubMed] [Google Scholar]
- 60.Kitaichi M, et al. Pulmonary lymphangioleiomyomatosis: a report of 46 patients including a clinicopathologic study of prognostic factors. Am J Respir Crit Care Med. 1995;151:527–533. doi: 10.1164/ajrccm.151.2.7842216. [DOI] [PubMed] [Google Scholar]
- 61.Kossoff E, et al. A modified Atkins diet is effective for the treatment of intractable pediatric epilepsy. Epilepsia. 2006;47:421–424. doi: 10.1111/j.1528-1167.2006.00438.x. [DOI] [PubMed] [Google Scholar]
- 62.Kossoff E, et al. Tuberous sclerosis complex and the ketogenic diet. Epilepsia. 2005;46:1684–1686. doi: 10.1111/j.1528-1167.2005.00266.x. [DOI] [PubMed] [Google Scholar]
- 63.Koyama M, et al. Chronic cystic lung disease: diagnostic accuracy of high-resolution CT in 92 patients. Am J Roentgenol. 2003;180:827–835. doi: 10.2214/ajr.180.3.1800827. [DOI] [PubMed] [Google Scholar]
- 64.Krueger D, et al. Everolimus for subependymal giant-cell astrocytomas in tuberous sclerosis. New Engl J Med. 2010;363:1801–1811. doi: 10.1056/NEJMoa1001671. [DOI] [PubMed] [Google Scholar]
- 65.Kumasaka T, et al. Lymphangiogenesis-mediated shedd ing of LAM cell clusters as a mechanism for dissemination in lymphangioleiomyomatosis. Am J Surg Pathol. 2005;29:1356–1366. doi: 10.1097/01.pas.0000172192.25295.45. [DOI] [PubMed] [Google Scholar]
- 66.Lamb E, Stevens P, Nashef L. Topiramate increases biochemical risk of nephrolithiasis. Ann Clin Biochem. 2004;41:166–169. doi: 10.1258/000456304322880104. [DOI] [PubMed] [Google Scholar]
- 67.Lane BR, et al. Clinical correlates of renal angiomyolipoma subtypes in 209 patients: classic, fat poor, tuberous sclerosis associated and epithelioid. J Urol. 2008;180:836–843. doi: 10.1016/j.juro.2008.05.041. [DOI] [PubMed] [Google Scholar]
- 68.Lawden M, et al. Visual field defects associated with vigabatrin therapy. J Neurol Neurosurg Psychiatry. 1999;67:716–722. doi: 10.1136/jnnp.67.6.716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Lemaitre L, et al. Renal angiomyolipoma: growth followed up with CT and/or US [see comments]. Radiology. 1995;197:598–602. doi: 10.1148/radiology.197.3.7480725. [DOI] [PubMed] [Google Scholar]
- 70.Lenz G, Avruch J. Glutamatergic regulation of the p70S6 kinase in primary mouse neurons. J Biol Chem. 2005;280:38121–38124. doi: 10.1074/jbc.C500363200. [DOI] [PubMed] [Google Scholar]
- 71.Levine N, et al. Gradual formation of an operative corridor by balloon dilation for resection of subependymal giant cell astrocytomas in children with tuberous sclerosis: specialized minimal access technique of balloon dilation. Minim Invas Neurosurg. 2006;49:317–320. doi: 10.1055/s-2006-950391. [DOI] [PubMed] [Google Scholar]
- 72.Ma L, et al. Identification of S664 TSC2 phosphorylation as a marker for Erk-mediated mTOR activation in tuberous sclerosis and human cancer. Cancer Res. 2007;67:7106–7112. doi: 10.1158/0008-5472.CAN-06-4798. [DOI] [PubMed] [Google Scholar]
- 73.Madhavan D, et al. Surgical outcome in tuberous sclerosis complex: a multicenter survey. Epilepsia. 2007;48:1625–1628. doi: 10.1111/j.1528-1167.2007.01112.x. [DOI] [PubMed] [Google Scholar]
- 74.Mai KT, Perkins DG, Collins JP. Epithelioid cell variant of renal angiomyolipoma [see comments]. Histopathology. 1996;28:277–280. doi: 10.1046/j.1365-2559.1996.d01-421.x. [DOI] [PubMed] [Google Scholar]
- 75.Manuchehri K, et al. A controlled study of vigabatrin and visual abnormalities. Br J Ophthalmol. 2000;84:499–505. doi: 10.1136/bjo.84.5.499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.McCormack FX, Moss J. S-LAM in a man? Am J Respir Crit Care Med. 2007;176:3–5. doi: 10.1164/rccm.200703-514ED. [DOI] [PubMed] [Google Scholar]
- 77.McCormack FX. Lymphangioleiomyomatosis: a clinical update. Chest. 2008;133:507–516. doi: 10.1378/chest.07-0898. [DOI] [PubMed] [Google Scholar]
- 78.McCullough DL, Scott R, Jr, Seybold HM. Renal angiomyolipoma (hamartoma): review of the literature and report of 7 cases. J Urol. 1971;105:32–44. doi: 10.1016/s0022-5347(17)61455-1. [DOI] [PubMed] [Google Scholar]
- 79.Meikle L, et al. Response of a neuronal model of tuberous sclerosis to mammalian target of rapamycin (mTOR) inhibitors: effects on mTORC1 and Akt signaling lead to improved survival and function. J Neurosci. 2008;28:5422–5432. doi: 10.1523/JNEUROSCI.0955-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Moss J, et al. Prevalence and clinical characteristics of lymphangioleiomyomatosis (LAM) in patients with tuberous sclerosis complex. Am J Respir Crit Care Med. 2001;164:669–671. doi: 10.1164/ajrccm.164.4.2101154. [DOI] [PubMed] [Google Scholar]
- 81.Muir TE, et al. Micronodular pneumocyte hyperplasia. Am J Surg Pathol. 1998;22:465–472. doi: 10.1097/00000478-199804000-00012. [DOI] [PubMed] [Google Scholar]
- 82.Muller NL, Chiles C, Kullnig P. Pulmonary lymphangiomyomatosis: correlation of CT with radiographic and functional findings. Radiolo gy. 1990;175:335–339. doi: 10.1148/radiology.175.2.2326457. [DOI] [PubMed] [Google Scholar]
- 83.Muncy J, Butler IJ, Koenig MK. Rapamycin reduces seizure frequency in tuberous sclerosis complex. J Child Neurol. 2009;24:477. doi: 10.1177/0883073808324535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Obuz F, et al. Various radiological appearances of angio myolipomas in the same kidney. Eur Radiol. 2000;10:897–899. doi: 10.1007/s003300051031. [DOI] [PubMed] [Google Scholar]
- 85.Ou YC, et al. Renal angiomyolipoma: experience of 23 patients. Chung Hua i Hsueh Tsa Chih - Chin Med J. 1991;48:217–223. [PubMed] [Google Scholar]
- 86.Paccalin M, et al. Activated mTOR and PKR kinases in lymphocytes correlate with memory and cognitive decline in Alzheimer's disease. Dementia and geriatric cognitive disorders. 2006;22:320–326. doi: 10.1159/000095562. [DOI] [PubMed] [Google Scholar]
- 87.Pea M, et al. Apparent renal cell carcinomas in tuberous sclerosis are heterogeneous: the identification of malignant epithelioid angiomyolipoma. Am J Surg Pathol. 1998;22:180–187. doi: 10.1097/00000478-199802000-00005. [DOI] [PubMed] [Google Scholar]
- 88.Pellock J, et al. Felbamate: consensus of current clinical experience. Epilepsy Res. 2006;71:89–101. doi: 10.1016/j.eplepsyres.2006.06.020. [DOI] [PubMed] [Google Scholar]
- 89.Peng T, Golub TR, Sabatini DM. The immunosuppressant rapamycin mimics a starvation-like signal distinct from amino acid and glucose deprivation. Mol Cell Biol. 2002;22:5575–5584. doi: 10.1128/MCB.22.15.5575-5584.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Pfeifer H, Thiele E. Low-glycemic-index treatment: a liberalized ketogenic diet for treatment of intractable epilepsy. Neurology. 2005;65:1810–1812. doi: 10.1212/01.wnl.0000187071.24292.9e. [DOI] [PubMed] [Google Scholar]
- 91.Prather P, de Vries P. Behavioral and cognitive aspects of tuberous sclerosis complex. J Child Neurol. 2004;19:666–674. doi: 10.1177/08830738040190090601. [DOI] [PubMed] [Google Scholar]
- 92.Pulsifer M, Winterkorn E, Thiele E. Psychological profile of adults with tuberous sclerosis complex. Epilepsy Behav. 2007;10:402–406. doi: 10.1016/j.yebeh.2007.02.004. [DOI] [PubMed] [Google Scholar]
- 93.Raab-Graham K, et al. Activity- and mTOR-dependent suppression of Kv1.1 channel mRNA translation in dendrites. Science. 2006;314:144–148. doi: 10.1126/science.1131693. [DOI] [PubMed] [Google Scholar]
- 94.Rakowski SK, et al. Renal manifestations of tuberous sclerosis complex: Incidence, prognosis, and predictive factors. Kidney Int. 2006;70:1777–1782. doi: 10.1038/sj.ki.5001853. [DOI] [PubMed] [Google Scholar]
- 95.Ravikumar B, et al. Inhibition of mTOR induces autophagy and reduces toxicity of polyglutamine expansions in fly and mouse models of Huntington disease. Nature Genetics. 2004;36:585–595. doi: 10.1038/ng1362. [DOI] [PubMed] [Google Scholar]
- 96.Rowley S, O'Callaghan F, Osborne J. Ophthalmic manifestations of tuberous sclerosis: a population based study. Br J Ophthalmol. 2001;85:420–423. doi: 10.1136/bjo.85.4.420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Ryu JH, et al. The NHLBI lymphangioleiomyomatosis registry: characteristics of 230 patients at enrollment. Am J Respir Crit Care Med. 2006;173:105–111. doi: 10.1164/rccm.200409-1298OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Saito K, et al. Malignant clear cell “ sugar ” tumor of the kidney: clear cell variant of epithelioid angiomyolipoma. J Urol. 2002;168:2533–2534. doi: 10.1016/S0022-5347(05)64188-2. [DOI] [PubMed] [Google Scholar]
- 99.Sampson JR, et al. Renal cystic disease in tuberous sclerosis: role of the polycystic kidney disease 1 gene. Am J Hum Genet. 1997;61:843–851. doi: 10.1086/514888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Sarnat HB, Flores-Sarnat L. Embryology of the neural crest: its inductive role in the neurocutaneous syndromes. J Child Neurol. 2005;20:637–643. doi: 10.1177/08830738050200080101. [DOI] [PubMed] [Google Scholar]
- 101.Schillinger F, Montagnac R. Chronic renal failure and its treatment in tuberous sclerosis. Nephrol Dial Transpl. 1996;11:481–485. [PubMed] [Google Scholar]
- 102.Sehgal S, Baker H, Vézina C. Rapamycin (AY-22,989), a new antifungal antibiotic. II. Fermentation, isolation and characterization. J Antibiot. 1975;28:727–732. doi: 10.7164/antibiotics.28.727. [DOI] [PubMed] [Google Scholar]
- 103.Seyama K, et al. Vascular endothelial growth factor-D is increased in serum of patients with lymphangioleiomyomatosis. Lymphat Res Biol. 2006;4:143–152. doi: 10.1089/lrb.2006.4.143. [DOI] [PubMed] [Google Scholar]
- 104.Shepherd CW, et al. Causes of death in patients with tuberous sclerosis. Mayo Clin Proceed. 1991;66:792–796. doi: 10.1016/s0025-6196(12)61196-3. [DOI] [PubMed] [Google Scholar]
- 105.Steiner MS, et al. The natural history of renal angio myolipoma. J Urol. 1993;150:1782–1786. doi: 10.1016/s0022-5347(17)35895-0. [DOI] [PubMed] [Google Scholar]
- 106.Stillwell TJ, Gomez MR, Kelalis PP. Renal lesions in tuberous sclerosis. J Urol. 1987;138:477–481. doi: 10.1016/s0022-5347(17)43234-4. [DOI] [PubMed] [Google Scholar]
- 107.Takayanagi M, et al. Two successful cases of bromide therapy for refractory symptomatic localization-related epilepsy. Brain Develop. 2002;24:194–196. doi: 10.1016/s0387-7604(02)00023-2. [DOI] [PubMed] [Google Scholar]
- 108.Tavazoie S, et al. Regulation of neuronal morphology and function by the tumor suppressors Tsc1 and Tsc2. Nature Neuroscience. 2005;8:1727–1734. doi: 10.1038/nn1566. [DOI] [PubMed] [Google Scholar]
- 109.Taveira-DaSilva A, et al. Reversible airflow obstruction, proliferation of abnormal smooth muscle cells, and impairment of gas exchange as predictors of outcome in lymphangioleiomyomatosis. Am J Respir Crit Care Med. 2001;164:1072–1076. doi: 10.1164/ajrccm.164.6.2102125. [DOI] [PubMed] [Google Scholar]
- 110.Thiele E. Managing epilepsy in tuberous sclerosis complex. J Child Neurol. 2004;19:680–686. doi: 10.1177/08830738040190090801. [DOI] [PubMed] [Google Scholar]
- 111.Tillema J, Franz D. Data on file in manuscript. in press. [Google Scholar]
- 112.van Slegtenhorst M, et al. Identification of the tuberous sclerosis gene TSC1 on chromosome 9q34. Science. 1997;277:805–808. doi: 10.1126/science.277.5327.805. [DOI] [PubMed] [Google Scholar]
- 113.Verrotti A, et al. Antiepileptic drugs and visual function. J Pediatr Neurol. 2007;36:353–360. doi: 10.1016/j.pediatrneurol.2007.03.001. [DOI] [PubMed] [Google Scholar]
- 114.Vicente MP, Pons M, Medina M. Pulmonary involvement in tuberous sclerosis. Pediatr Pulmonol. 2004;37:178–180. doi: 10.1002/ppul.10414. [DOI] [PubMed] [Google Scholar]
- 115.von der Brelie C, et al. Impaired synaptic plasticity in a rat model of tuberous sclerosis. Eur J Neurosci. 2006;23:686–692. doi: 10.1111/j.1460-9568.2006.04594.x. [DOI] [PubMed] [Google Scholar]
- 116.Wang Y, Barbaro M, Baraban S. A role for the mTOR pathway in surface expression of AMPA receptors. Neurosci Lett. 2006;401:35–39. doi: 10.1016/j.neulet.2006.03.011. [DOI] [PubMed] [Google Scholar]
- 117.Winterkorn EB, Pulsifer M, Thiele E. Cognitive prognosis of patients with tuberous sclerosis complex. Neurology. 2007;68:62–64. doi: 10.1212/01.wnl.0000250330.44291.54. [DOI] [PubMed] [Google Scholar]
- 118.Wong M. Mammalian target of rapamycin (mTOR) inhibition as a potential antiepileptogenic therapy: From tuberous sclerosis to common acquired epilepsies. Epilepsia. 2009;51:27–36. doi: 10.1111/j.1528-1167.2009.02341.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Xu L, et al. Levels of mTOR and its downstream targets 4E-BP1, eEF2, and eEF2 kinase in relationships with tau in Alzheimer’s disease brain. FEBS J. 2005;272:4211–4220. doi: 10.1111/j.1742-4658.2005.04833.x. [DOI] [PubMed] [Google Scholar]
- 120.Yamakado K, et al. Renal angiomyolipoma: relationships between tumor size, aneurysm formation, and rupture. Radiology. 2002;225:78–82. doi: 10.1148/radiol.2251011477. [DOI] [PubMed] [Google Scholar]
- 121.Yates J. Tuberous sclerosis. Eur J Hum Genet. 2006;14:1065–1073. doi: 10.1038/sj.ejhg.5201625. [DOI] [PubMed] [Google Scholar]
- 122.Young LR, Inoue Y, McCormack FX. Diagnostic potential of serum VEGF-D for lymphangioleiomyomatosis. N Engl J Med. 2008;358:199–200. doi: 10.1056/NEJMc0707517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Zeng LH, et al. Rapamycin prevents epilepsy in a mouse model of tuberous sclerosis complex. Ann Neurol. 2008;63:444–453. doi: 10.1002/ana.21331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Zeng LH, Rensing NR, Wong M. The mammalian target of rapamycin signaling pathway mediates epileptogenesis in a model of temporal lobe epilepsy. J Neurosci. 2009;29:6964–6972. doi: 10.1523/JNEUROSCI.0066-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Zhang Y, et al. Rheb is a direct target of the tuberous sclerosis tumour suppressor proteins. Nature Cell. 2003;5:578–581. doi: 10.1038/ncb999. [DOI] [PubMed] [Google Scholar]