Abstract
The vascular endothelium separates circulating fluid and inflammatory cells from the surrounding tissues. Vascular leak occurs in response to wide-spread inflammatory processes, such as sepsis and Acute Respiratory Distress Syndrome (ARDS), due to the formation of gaps between endothelial cells (EC). Although these disorders are leading causes of mortality in the ICU, no medical therapies exist to restore EC barrier function. Recent evidence highlights a key role for the Abl family of non-receptor tyrosine kinases in regulating vascular barrier integrity. These kinases have well-described roles in cancer progression and neuronal morphogenesis, but their functions in the vasculature have remained enigmatic until recently. The Abl family kinases, c-Abl (Abl1) and Abl related gene (Arg, Abl2), phosphorylate several cytoskeletal effectors that mediate vascular permeability, including myosin light chain kinase, cortactin, vinculin, and β-catenin. They also regulate cell-cell and cell-matrix junction dynamics, and the formation of actin-based cellular protrusions in multiple cell types. Additionally, both c-Abl and Arg are activated by hyperoxia and contribute to oxidant-induced EC injury. These numerous roles of Abl kinases in EC and the current clinical usage of imatinib and other Abl kinase inhibitors have spurred recent interest in repurposing these drugs for the treatment of vascular barrier dysfunction. This review will describe the structure and function of Abl kinases with an emphasis on their roles in mediating vascular barrier integrity. We will also provide a critical evaluation of the potential for exploiting Abl kinase inhibition as a novel therapy for inflammatory vascular leak syndromes.
Keywords: Sepsis, ALI, ARDS, Endothelium, Cytoskeleton, c-Abl, Arg, Imatinib
Inflammation-induced vascular leak underlies the pathophysiology of multiple disorders that affect critically ill patients, including sepsis and Acute Respiratory Distress Syndrome (ARDS). These conditions are major causes of morbidity and mortality in the ICU affecting 750,000 and 200,000 patients/year in the US, respectively1, 2. The lack of medical therapies capable of attenuating vascular leak necessitates additional research into the mechanisms underlying EC barrier dysfunction and clinical trials of novel therapeutic strategies.
Inflammatory vascular leak: Overview
Multiple inflammatory mediator-initiated cellular signaling pathways alter the actin-myosin contractile apparatus of endothelial cells (EC) to regulate vascular permeability. Contractile pathways, which cause disruption of cell-cell junctions, cell rounding, and ultimately vascular leak, are counterbalanced by tethering forces that cause lamellipodia formation, restore cell-cell junctions and resolve paracellular gaps3. Inflammatory disorders, including sepsis and ARDS, create an imbalance in these pathways in favor of vascular leak4. Current work aims to characterize the signaling pathways that determine vascular permeability and identify therapeutic strategies to enhance barrier function. The Abl family kinases, c-Abl (Abl1) and Abl related gene (Arg, Abl2), have recently emerged as key mediators of vascular permeability due to their well characterized roles in the dynamic regulation of the actin cytoskeleton and cell-cell and cell-matrix junctions5–8. Several groups have recently reported that the FDA approved Abl kinase inhibitor imatinib attenuates vascular leak induced by thrombin, histamine, vascular endothelial growth factor (VEGF), lipopolysaccharide (LPS), and oxidative stress9–13. Although the mechanisms are not fully characterized, it is clear that Abl kinase inhibition has potent and pleiotropic effects on vascular barrier function. This is not surprising given that Abl kinases phosphorylate several cytoskeletal effectors that have established roles in vascular permeability, and contribute to NFκB and reactive oxygen species (ROS) signaling pathways14–19. In addition to sepsis and ARDS, inflammatory vascular injury contributes to the pathogenesis of atherosclerosis, ischemia-reperfusion injury, and pulmonary hypertension (PH), thus broadening the potential clinical relevance of this work20. This review will (1) describe the structure and functions of the Abl family kinases (2) discuss the mechanisms by they mediate EC barrier function (3) evaluate the therapeutic potential of Abl kinase inhibition in inflammatory vascular leak syndromes.
1. Abl family kinase structure and function
The cytoplasmic tyrosine kinase c-Abl was originally identified as a virally transduced oncogene that causes murine lymphosarcoma21. Subsequently, the human ortholog of this protein was identified as part of the breakpoint cluster region (BCR)-Abl fusion protein that causes chronic myelogenous leukemia (CML)22. Arg was identified later based on its sequence homology with c-Abl, and together these kinases make up the Abl subfamily of cytoplasmic tyrosine kinases23.
1.1. Structure and regulation of Abl kinases
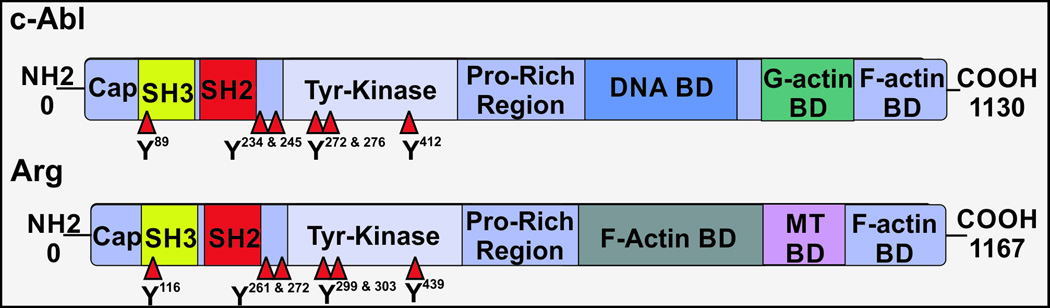
In mammals, both c-Abl and Arg have a myristoylated isoform and a nonmyristolated isoform7. In both isoforms the N-terminal regions of c-Abl and Arg are composed of a highly homologous Src homology (SH)3-SH2-Tyrosine kinase domain cassette and an upstream Cap region14. This region regulates Abl kinase activity via auto-inhibitory interactions that disrupt specific tyrosine phosphorylation events14. In contrast, the C-terminal regions of these kinases, termed the last exon region, are distinct and are composed of multiple cytoskeletal binding elements (Figure 1). Although c-Abl and Arg both contain a calponin homology F-actin binding domain, c-Abl has a unique proline-rich region that binds to G-actin, while Arg has a unique F-actin binding domain that resembles the I/LWEQ domain of talins24, 25. The last exon region of c-Abl also contains a DNA binding domain and nuclear localization and nuclear export sequences, which allow it to move back and forth between the nucleus and the cytoplasm under various conditions26. Due to the fact that the substrates of these kinases are very similar, their functional differences are likely mediated by differential subcellular targeting and diversity in their cytoskeletal binding elements.
Figure 1. Abl Family Kinase Domain Organization.
The Abl Kinase Family is composed of c-Abl and Arg. SH3 - Src Homology 3, SH2 – Src Homology 2, Pro – Proline BD – binding domain, MT – microtubule. Triangles indicate major regulatory phosphorylation sites. Similar figures are availble in comprehensive reviews of Abl family kinase regulation14.
1.2. Overview of Abl family kinase functions
Not surprisingly given their structural properties, the Abl kinases have well-described roles in regulating cytoskeletal structure, including binding and bundling F-actin filaments, phosphorylating cytoskeletal effector proteins, and modulating the activity of MLCK and Rho family GTPases14. Together these functions allow Abl kinases to promote actin-based cellular protrusion formation and alter cell-cell and cell matrix junction dynamics, which underlie their crucial roles in regulating cell shape and migration7, 27. Fibroblasts deficient in these kinases display decreased membrane ruffling and lamellipodia formation28. However, Abl−/−/Arg−/− mouse embryonic fibroblasts (MEF) migrate faster than control cells6. This seemingly paradoxical effect has been attributed Abl kinase inhibition of Rho mediated cellular contractility7, 29. Abl kinases also modulate mechanotransduction pathways to alter the stability of cell-cell and cell-matrix junctions30. The broad implications of these processes are highlighted by the roles of Abl kinases in diverse processes including immune synapse formation, dendrite branching, endocytosis, and epithelial morphogenesis7, 14. Although cytoskeletal rearrangements are critically involved in EC barrier function, the role of Abl kinases in regulating vascular permeability has been recognized only recently.
Given its multitude of cellular functions, it comes as no surprise that c-Abl knockout (KO) mice (c-Abl−/−) die shortly after birth with diverse phenotypes including splenic and thymic atrophy, osteoporosis, and cardiomegaly31–34. In contrast, Arg−/− mice survive to adulthood with behavioral abnormalities as their main phenotype35. Combined KO of c-Abl and Arg (c-Abl−/−;Arg−/−) is embryonic lethal due to impaired neurulation, pericardial edema, and hemorrhage35. Endothelial specific c-Abl KO mice on an Arg−/− background (c-AblECKO;Arg−/−) die during late embryonic development due to focal loss of vasculature and tissue apoptosis and necrosis. However, c-AblECKO mice on an Arg+/− background (c-AblECKO;Arg+/−) are viable to adulthood and display a phenotype with multiple cardiovascular defects including dilation of the left atrium and loss of EC in the left ventricle36. Although these studies demonstrate that Abl kinases are critical to vascular function, additional work is necessary to differentiate the roles of these kinases and determine the extent to which they can compensate for each other.
2. Abl kinase signaling in inflammatory vascular leak
Due to their multitude of substrates, the Abl kinases likely mediate vascular barrier function via several mechanisms. Table 1 describes functions of major Abl kinase substrates that have established roles in EC permeability. The following section will discuss the role of Abl kinases in (1) responding to barrier altering agonists and regulating (2) actin cytoskeletal structure, (3) cell-cell and cell-matrix junctions, (4) NFκB signaling, and (5) ROS signaling.
Table 1. Abl family kinase targets involved in vascular barrier function.
Multiple targets of c-Abl and Arg have established roles in the regulation of vascular permeability, which are briefly described. A complete listing of known Abl kinase substrates is available in a recent comprehensive review14.
| Protein | Function in Vascular Barrier Function |
|---|---|
| c-Abl | Mediates vascular leak downstream of edemagenic agents via effects on AJs and nmMLCK activation11 Attenuates vascular leak downstream of S1P and FTY720 via effects on cortactin and nmMLCK phosphorylation40 |
| Arg | Mediates vascular leak downstream of edemagenic agents via effects on FAs and Rac activity78 |
| β-Catenin | Binds to VE-Cadherin, which structurally and functionally couples AJs to the actin cytoskeleton15 |
| Cav1 | Major component of caveolae, which are responsible for transcellular transport of fluid and albumin80 |
| Crk & CrkL | Adaptor proteins involved in AJ remodeling48 Crk phosphorylation is involved in Rac activation and unphosphorylated Crk promotes AJ disassembly49 |
| Cortactin | Attenuates vascular leak by promoting actin polymerization at lamellipodia81 |
| nmMLCK | Central mediator of permeability via effects on actin-myosin contraction and ultimately cell shape. Involved in formation of both stress fibers and lamellipodia depending on subcellular localization3 |
| Myosin IIB | Component of actin-myosin contractile apparatus3 |
| p190RhoGAP | Associates with p120RhoGEF as part of a complex that inhibits Rho activity, decreases actomyosin contractility, and focal adhesion remodeling6–8, 29 |
| Paxillin | Component of FAs that binds to the integrin cytoplasmic tail, serving as a link to the actin cytoskeleton15 |
| WAVE2/3 & N-WASP | Promote actin polymerization at lamellipodia3 |
2.1. The role of Abl kinases in responding to barrier altering agonists
Multiple groups have reported attenuation of inflammatory vascular leak with the Abl kinase inhibitor imatinib. The first report identified a protective effect of Arg inhibition on vascular leak induced by thrombin, histamine, and VEGF10. Each of these stimuli increase Arg activity and induce a downstream decrease in Rac activity and focal adhesion (FA) number10. However, c-Abl inhibition also attenuates VEGF-induced EC permeability11, suggesting that these kinases may have some overlapping functions in regulating EC barrier integrity. Although the mechanisms are not completely understood, they include increased Rac1/Rap1 activity and decreased Ca2+ mobilization, non-muscle MLCK (nmMLCK) activation, and stress fiber formation11. Additionally, vascular barrier disruption induced by both LPS and oxidative stress are attenuated by imatinib treatment12, 13. These potent barrier protective effects, in response to several barrier disruptive agonists, strongly support the hypothesis that Abl kinases are central mediators of vascular integrity. This work is supported by in vivo studies that demonstrate a protective effect of imatinib in vascular leak induced by injection of VEGF (intradermal), thrombin receptor activating peptide (TRAP) (intravenous, IV), and LPS (intratracheal, IT), and the cecal ligation and puncture (CLP) sepsis model9–12. Additionally, imatinib restores blood-brain-barrier (BBB) integrity and decreases intracerebral hemorrhage in murine models37, 38. Although other kinase targets of imatinib may play a role in these effects, a clear contribution of c-Abl is evidenced by decreased vascular leak in c-AblECKO;Arg+/− mice compared with Arg+/− control mice11.
In contrast with these barrier protective effects, Abl kinase inhibition worsens endothelial barrier disruption in EC challenged with 18% cyclic stretch, and worsens vascular leak in a murine model of ventilator-induced lung injury (VILI) induced by high tidal volume mechanical ventilation (MV)12. These observations indicate differential pathophysiology of VILI and ARDS due to other causes39 and suggest that Abl kinase inhibition in ARDS patients undergoing MV should be approached with caution. Abl kinases also contribute to the barrier protective response to the endogenous bioactive sphingolipid sphingosine 1-phosphate (S1P)40. S1P causes c-Abl activation and formation of a c-Abl/nmMLCK/cortactin complex, which facilitates peripheral actin polymerization40. Similar to S1P, the response to the barrier protective agent activated protein C (APC) is dependent on the S1P Receptor 1 (S1PR1), Rac1 activation, and cortical MLC phosphorylation41. The critical role of S1PR1 in the barrier protective effects induced by APC suggest that c-Abl may also participate in this response. Additionally, the barrier protective response to FTY-720, a pharmaceutical S1P analog with a distinct mechanism of action, involves c-Abl activation42. Furthermore, Abl kinases are necessary for Tie2 expression and EC survival mediated angiopoietin-1, a potent barrier enhancing agonist36. Together this body of work provides strong evidence that Abl kinases play a central role in EC barrier regulation. The published effects of imatinib on permeability are summarized in verbal and schematic form (Table 2 and Figure 2). Although the roles of Abl kinases in other barrier protective pathways remain poorly characterized, Abl kinase inhibition is likely to affect a wide-variety of signaling pathways due to their ability to regulate Rac1/Rap1 activity10, 36. This highly complex picture necessitates additional work to differentiate the roles of c-Abl and Arg and define the pathways by which they alter actin cytoskeletal structure, cell-cell and cell-matrix junctions, and inflammation.
Table 2. Pre-clinical data on the effects of imatinib on EC barrier dysfunction.
Several independent groups have demonstrated that imatinib enhances endothelial barrier function and protects against vascular leak in vitro and in vivo. However, some data demonstrate increased leak after imatinib in other injury models.
| In vitro models | Agonist | Effect on EC barrier |
|---|---|---|
| Rat aortic EC | VEGF | Protective82 |
| HUVEC | Thrombin, Histamine | Protective10 |
| HLMVEC | Thrombin | Protective10 |
| Immortalized EC | VEGF | Protective11 |
| HUVEC | SCF | Protective79 |
| Mouse lung microvascular EC | H2O2 | Protective13 |
| HPAEC | LPS | Protective12 |
| HPAEC | 18% CS | Disruptive12 |
| Murine models | ||
| Bleomycin-induced ALI | Bleomycin | *Anti-inflammatory83 |
| CLP (sepsis) | Fecal peritonitis | Protective10 |
| Isolated perfused lung | TRAP | Protective10 |
| Isolated perfused lung | Ischemia/ reperfusion | *Attenuates cytotoxicity13 |
| IT LPS (neutropenic mice) | LPS | Protective9 |
| IT LPS | LPS | Protective12 |
| Miles assay | VEGF | Protective10 |
| Miles assay | VEGF | Protective79 |
| Miles assay | VEGF | Protective11 |
| VILI | MV (30 mL/kg tidal volume) | Disruptive12 |
CLP – cecal ligation and puncture, LPS - lipopolysaccharide, TRAP - thrombin receptor activating peptide, VEGF - vascular endothelial growth factor, VILI - ventilator-induced lung injury, MV – mechanical ventilation.
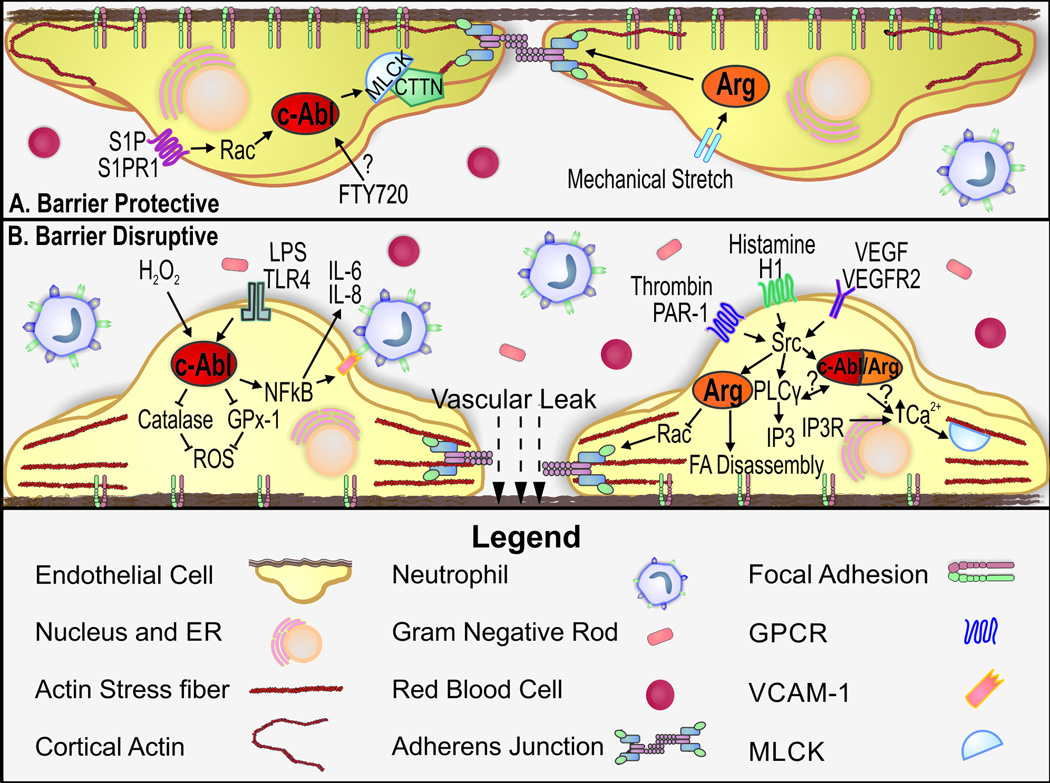
Figure 2. Barrier Disruptive and Barrier Protective Pathways of Abl Family Kinases in Endothelial Cytoskeletal Dynamics.
Published data indicated that Abl family kinases play both barrier disruptive (A) and barrier protective (B) roles in cellular signaling pathways that mediate endothelial permeability (details described in text). For the sake of clarity this figure is not drawn to size. S1P – Sphingosine 1-Phosphate, S1PR1 – S1P Receptor 1, MLCK – Myosin Light Chain Kinase, CTTN – Cortactin, VEGF – Vascular Endothelial Growth Factor, VEGFR2 – VEGF Receptor 2, VE-Cad – Vascular Endothelial Cadherin, Vascular Endothelial Cellular Adhesion Molecule 1 – VCAM-1, Protease Activated Receptor 1 – PAR-1, Histamine Receptor 1 (H1), Inositol 3-Phosphate – IP3, IP3 Receptor – IP3R, PLCγ – Phospholipase C Gamma - Reactive Oxygen Species – ROS, Glutathione peroxidase 1 – GPx1, LPS – Lipopolysaccharide, Toll Like Receptor 4 – TLR, H2O2 – Hydrogen Peroxide, Ca2+ - Calcium, NFκB – Nuclear Factor Kappa B, GPCR – G Protein Coupled Receptor, FA – Focal Adhesion.
2.2. Abl kinase regulation of the actin cytoskeleton
Barrier disruptive agents cause nmMLCK and Rho family kinase mediated cytoskeletal rearrangements, including loss of the stabilizing cortical band of actin microfilaments and the formation of cytoplasmic actin stress fibers3. Abl kinases alter the activity of both nmMLCK and Rho kinases, indicating that they have the potential to regulate EC cytoskeletal dynamics and barrier integrity via multiple mechanisms10, 40. Although the direct effects of Abl kinases on the location of actin-myosin contraction have not been investigated in EC, work in other cell types suggests that these kinases play distinct roles in regulating cytoskeletal structure by this mechanism. In mouse embryonic fibroblasts (MEF), c-Abl−/− causes central contractile apparatus localization, whereas Arg−/− causes peripheral contractile apparatus localization8.
In addition to direct effects on the actin-myosin contractile apparatus, Abl kinases phosphorylate cytoskeletal effectors that promote actin-based cellular protrusion formation, including cortactin, Wiskott-Aldrich syndrome protein (WASp), WASp-family verprolin-homologous proteins (WAVE), and Dok114, 15. Both overlapping and distinct roles of these kinase-mediated phosphorylation events have been identified in the responses to growth factors, including platelet derived growth factor (PDGF) and epidermal growth factor (EGF). In fibroblasts, PDGF induces cortactin phosphorylation, downstream actin polymerization, and cortical wave formation as part of a signaling pathway that is dependent on c-Abl or Arg43. Additionally, the formation of lamellipodia and filopodia in fibroblasts, and the actin comet tail of Shigella, are dependent on c-Abl mediated phosphorylation of WAVE3, Dok1, and N-WASP respectively44–46. In breast cancer cells, EGF-induced formation of invadopodia depends on Arg, but not c-Abl47. These findings need to be confirmed in endothelial cells to determine if barrier altering agonists lead to Abl kinase mediated phosphorylation of these proteins and elucidate the subsequent downstream effects.
2.3. Abl kinase regulation of cell-cell and cell-matrix junction dynamics
Barrier-disruptive agents also disrupt the cell-cell and cell-matrix junctions that maintain barrier integrity15. In human umbilical vein EC (HUVEC), Abl kinase inhibition with imatinib restores the integrity of adherens junctions (AJ) following thrombin stimulation10. The mechanisms underlying these effects have not been described; however, the AJ protein β-catenin is a known target of Abl kinases, and its phosphorylation contributes to AJ internalization15. Although these data suggest that Abl kinases disrupt AJ in EC, work in c-Abl−/−/Arg−/− MEF demonstrates that Abl kinases are also necessary for Rac activation and AJ formation, suggesting that Abl kinases function both upstream and downstream of Rac to promote AJ integrity48, 49.
In addition to cell-cell junctions, cell-matrix junctions (Focal Adhesions, FA) are critical to EC barrier integrity15. In HUVEC, imatinib blocks Arg-mediated barrier disruption by promoting FA junction stability10. The exact mechanism underlying this effect remains unclear, but in fibroblasts, Arg-mediated activation of p190RhoGAP at the periphery decreases Rho activity downstream of integrin-mediated cellular adhesion, which ultimately causes decreases FA dynamics5, 29. However, Abl kinases also phosphorylate the FA proteins Crk/CrkL, and paxillin, which suggests other possible mechanisms for Abl-mediated changes in FA dynamics14, 15. For example, in response to cellular adhesion, c-Abl is recruited to FA, where it phosphorylates paxillin and promotes FA formation50, 51. Studies in mouse neurons have demonstrated that integrin signaling through both c-Abl and Arg promotes cortical dendrite branching in response to cellular adhesive cues52. Abl kinases are therefore critical to both cell-cell and cell-matrix junctional dynamics, providing additional mechanisms by which Abl kinase inhibitors alter vascular integrity.
2.4. Role of Abl kinases in NFκB mediated inflammation
Several barrier disruptive agents activate EC to increase transcriptional activation of NFκB, release of inflammatory cytokines, and upregulation of multiple cellular adhesion molecules (CAMs) that are involved in neutrophil recruitment and extravasation53. Imatinib attenuates LPS-induced VCAM-1 expression and inflammatory cytokine (IL-6, and IL-8) production in human pulmonary artery EC (HPAEC), and these effects are mimicked by silencing of c-Abl, but not Arg12. Our unpublished data indicate that imatinib prevents LPS-induced NFκB phosphorylation and nuclear translocation in HPAEC, suggesting that LPS increases VCAM expression and cytokine production via a c-Abl/NFκB dependent signaling pathway. In a murine model of LPS-induced ALI during recovery from neutropenia, imatinib decreased production of pro-inflammatory cytokines (TNFα, IL-8, IL-1β) and attenuated lung injury9.
Suppression of NFκB activation and inflammatory cytokine production following imatinib treatment has also been reported in peripheral blood mononuclear cells (PBMC), cells of the monocyte-macrophage lineage, and leukocytes isolated from CML patients18. These data are supported by a murine model of TNFα-dependent acute hepatic inflammation, in which imatinib suppressed production of TNFα, IL-6, and IL-8 induced by both LPS and Con-A19. Although these data provide strong evidence for the anti-inflammatory effects of imatinib, the effects of imatinib on NFκB activation appear to increase or decrease depending on cell type and length of imatinib exposure. In pancreatic β cells, imatinib causes an initial increase in NFκB activation, followed by a decrease in activation and cytokine production54, 55. This work provides evidence for a key role of Abl kinase inhibition in NFκB activation. The distinct Abl kinases (c-Abl versus Arg) and the downstream targets that mediate this effect have yet to be determined.
2.5. Abl kinase regulation of ROS signaling pathways
Prolonged MV with high fraction of inspired oxygen during ARDS treatment can lead to hyperoxic lung injury. This involves increased production of ROS, including superoxide (O2−) and hydrogen peroxide (H2O2), which overwhelms antioxidant defenses, leading to cell death and EC barrier dysfunction56. Abl kinases are activated in response to oxidative stress in multiple cell types57. In EC, H2O2 leads to c-Abl activation and nuclear translocation, while increasing protein kinase G (PKG) activity with the cGMP analog 8pCPT-cGMP blocks this effect13. Inhibition of c-Abl, indirectly by increasing PKG activity or directly with imatinib, attenuates the oxidant-induced decline in expression of the antioxidant proteins catalase and glutathione peroxidase (GPx-1), which ultimately decreases EC injury and barrier dysfunction13. Although the role of Arg in oxidant-induced EC injury has not been investigated, work in in MCF-7 breast cancer cells and MEF suggests that both Abl kinases are critical to oxidant-induced injury. In these cell types, H2O2 leads to heterodimerization of c-Abl and Arg, inhibition of antioxidant enzymes, and ultimately oxidant-induced apoptosis16, 17, 57. The growing amount of evidence for a role of Abl kinases in ROS signaling pathways paves the way for future research, in which the role of Arg deserves specific attention.
3. Therapeutic potential of Abl kinase inhibition in inflammatory vascular leak syndromes
The mechanistic studies described above, which were initiated in part due to the serendipitous observation that imatinib quickly reversed pulmonary edema in some patients58, suggest that inhibition of Abl kinases may have therapeutic potential for decreasing vascular permeability. The broad clinical experience with current FDA-approved Abl kinase inhibitors may facilitate repurposing of these drugs for inflammatory vascular leak syndromes. In contrast to some other anti-cancer agents that specifically target oncogenic proteins, the Abl kinase inhibitors have potent effects on non-transformed cells because they block cellular c-Abl/Arg activity. This section will provide a brief review of the FDA-approved Abl kinase inhibitors and describe their known clinical effects on vascular permeability.
3.1. FDA approved Abl family kinase inhibitors
Imatinib (STI-571, Gleevec), the prototype Abl kinase inhibitor, was developed to inhibit the BCR-Abl fusion protein that causes CML59. Resistance and intolerance to imatinib led to the development of second-generation BCR-Abl inhibitors, nilotinib (AMN107, Tasigna) and dasatinib (BMS-354825, Sprycel), and third-generation BCR-Abl inhibitors, ponatinib (AP24534, Iclusig) and bosutinib (SKI606, Bosulif) (Table 3). A common feature of these Abl kinase inhibitors is their mechanism of action involving competitive inhibition by binding to the ATP-binding pocket in the activation loop of the kinase60. High sequence homology in the ATP binding pocket of c-Abl, Arg, and closely related kinases (including Src family kinases) has led to difficulty in development of specific kinase inhibitors. However, off-target kinase inhibition has allowed for imatinib to be repurposed for other conditions including gastrointestinal stromal tumor (c-Kit-dependent) and dermatofibrosarcoma protuberans (PDGFR-dependent)61. Recent work has suggested they may also be beneficial in the treatment of the proliferative lung diseases PH62 and idiopathic pulmonary fibrosis63, as well as systemic inflammatory disorders including rheumatoid arthritis64.
Table 3. Comparison of FDA Appproved Abl Kinase Inhibitors.
A comparison of the pharmacokinetic and pharmacodynamic properties, and the edema-related side effects, of the Abl kinase inhibitors that are currently in clinical usage60, 84
| Drug | Imatinib | Dasatinib | Nilotinib | Ponatinib | Bosutinib |
|---|---|---|---|---|---|
| Off Target Effects | c-KIT, PDGFR, DDR1, NQO2 | c-KIT, PDGFR, DDR1/2, SRC, YES, FYN, LYN, HCK, LCK, FGR, BLK, FRK, CSK, BTK, TEC, BMX, TXK, ACK, BRAF, EGFR, EPHA, MAPK, RAF, SLK, ZAK | c-KIT, PDGFR, DDR1, NQO2 More specific to Abl kinases |
c-KIT, PDGFR, FLT3, FGFR, RET, VEGFR, SRC Inhibits mutant BCR-Abl forms including T315I. |
ALK, CSK, FGR,LYN, PKA, CK1, CK2, SRC, RET,SYK |
| T1/2 (hrs) | 18 | 3–5 | 15–17 | 24 | 22.5 |
| F (%) | 98 | <34 | 30 | 65 | unknown |
| Vd (L/kg) | 2–6 | 36 | 2 | 17.5 | 87 |
| Metabolism | CYP3A4 CYP3A5 CYP2C8 |
CYP3A4 | CYP3A4 CYP2C8 |
CYP3A4 CYP3A5 CYP2C8 CYP2D6 |
CYP3A4 |
| Edema (%) | 53 | 50 | 11 | 13–22 | 14 |
F – Bioavailability, Vd – Volume of Distribution.
Another consequence of the lack of specificity is interference with the physiological function of target kinases, which results in unwanted side-effects (Table 3). Of note, all Abl kinase inhibitors cause varying degrees of subcutaneous edema, as well as pulmonary, peritoneal and pericardial effusions65–67. As these side effects depend on treatment dose and duration68, this paradox may be explained by the chronic kinase inhibition that is present in patients undergoing treatment for CML. Specifically, long-term inhibition of PDGFR on pericytes may impair vascular integrity69. Imatinib has also been occasionally associated with congestive heart failure70, but larger patient series failed to show a relationship between imatinib treatment and cardiac failure71, 72. Additionally, side effects of these drugs may occur due to the wide-spread expression of the Abl family kinases. Although both c-Abl and Arg are broadly expressed throughout the tissues of the body, c-Abl expression is high in smooth muscle cells27, 73, where it plays a critical role in migration and cytokinesis. Neurons express a large amount of Arg, which promotes the development of the neural tube and dendritic spines35, 74. Both c-Abl and Arg are highly expressed in T cells, where they play a role in T-cell function and development75. Inhibition of Abl kinases in these specific cell types may contribute to the side-effects of imatinib, such as lymphopenia. Despite their side-effects, the Abl kinase inhibitors are generally well-tolerated, especially imatinib and nilotinib76.
3.2. Clinical effects of Abl family kinase inhibitors on vascular permeability
Although there are no clinical studies about the effects of Abl kinase inhibitors on inflammatory vascular leak, evidence from a series of case reports indicates that Abl kinase inhibition may decrease permeability in certain patients. The first report indicating the potential of imatinib to reduce EC permeability described a PH patient with peripheral veno-occlusive disease and edema symptoms who experienced improvement of dyspnea and reduction of pulmonary edema (evidenced by computed tomography) within 24 hours after initiation of imatinib therapy58. A second report describes a patient with bleomycin-induced pneumonitis with clinical and radiographic signs of pulmonary edema and fibrosis. Imatinib treatment was followed by a quick improvement in respiratory status and resolution of radiographic findings77. More recently, a patient diagnosed with idiopathic vascular leak experienced almost complete resolution of her symptoms, including dyspnea and cough, and normalization of parameters of vascular leak including serum albumin levels, body weight, and pulmonary leak index after initiation of imatinib treatment78. The rapid responses in these cases suggest a direct effect on the endothelium, supporting the experimental studies described above, but the exact mechanisms remain incompletely understood. Additionally, inhibition of other imatinib-sensitive kinases may have contributed to the observed effects. Among these kinases is PDGFR, a target of all of the FDA approved inhibitors, that is expressed in the pulmonary vasculature and contributes to imatinib’s barrier protective effects in tPA-induced BBB permeability38. Additionally, stem cell factor (SCF) increases vascular permeability via increased VE-Cadherin internalization as well as increased endothelial nitric oxide (NO) synthase (eNOS) phosphorylation and NO production, which can be attenuated by inhibition of the intracellular kinase domain of the SCF receptor (c-Kit) with imatinib79. We anticipate that the array of FDA approved Abl kinase inhibitors with differing specificities will aid in determining the contribution of each of the targets of these inhibitors to vascular leak pathophysiology to maximize potential treatment efficacy.
Concluding Remarks
The recent studies discussed in this review point toward a central role of Abl kinases in endothelial barrier regulation and inflammatory signaling. Abl kinases are activated downstream of several barrier altering agonists and regulate EC cytoskeletal structure and junctional dynamics, NFκB signaling and oxidant-induced EC injury. The protective effects of the Abl kinase inhibitor imatinib observed in most studies not only establishes the relevance of Abl kinases in vascular biology, but also provide a relevant link to clinical conditions associated with vascular leak. Although the majority of this work indicates barrier protective effects of Abl kinase inhibition, Abl kinases play a barrier protective role in the EC response to S1P, FTY720, and VILI. A likely explanation for these paradoxical observations might be the differential role of c-Abl versus Arg, and their downstream targets. This body of work suggests that c-Abl and Arg have distinct functions in the response to barrier altering agonists involved in the pathophysiology of inflammatory vascular leak syndromes. Development of specific c-Abl and Arg inhibitors will help to elucidate the biological roles of c-Abl and Arg, and contribute to tailored therapy. As little is still known from pericytes and smooth muscle cells, the role of Abl kinases in vascular biology may extend well beyond endothelial barrier regulation and inflammation. In support of the experimental work, a series of recent case reports describe protective effects of imatinib in the treatment of inflammatory vascular leak. Because inflammatory vascular leak is prevalent in the ICU population, and data on use of Abl kinase inhibitors in this population is scarce, testing their safety and pharmacokinetics in this population are critical first steps in clinical development of this strategy. To support these initial steps, the European Medicines Agency granted Orphan Drug Designation for application of imatinib in acute respiratory distress syndrome (http://www.orphan-drugs.org/2014/10/20/ema-orphan-drug-designations-october-2014). Although additional work is necessary to differentiate the roles of these two kinases and determine the clinical scenarios and patient populations in which Abl kinase inhibition would be most beneficial, therapeutic manipulation of the Abl kinase family holds great promise as a novel and highly effective intervention for inflammatory vascular leak syndromes.
SIGNIFICANCE.
Inflammatory vascular leak syndromes, such as sepsis and ARDS, are leading causes of mortality in critically ill patients; however, no pharmacologic treatments exist to attenuate vascular leak. Recent work demonstrates that the Abl family kinases are involved in the cellular signaling pathways initiated by both barrier protective and barrier disruptive stimuli. Preclinical studies using both cell culture and animal models indicate that imatinib, an FDA-approved Abl kinase inhibitor, attenuates vascular leak induced by diverse stimuli including thrombin, histamine, VEGF, LPS, and oxidative stress. A series of recent case reports support these findings, further arguing for clinical trials to determine the efficacy of this novel treatment strategy.
ACKNOWLEDGEMENTS
Sources of Funding: This work acknowledges support from NHLBI (F30HL121982 to ANR), AHA (14PRE18860021 to ANR), University of Illinois Institutional Funds, and the Dutch Heart Foundation (grant 2014T064 to JA and grant 2011T072 to GPvNA).
ABBREVIATIONS
- AJ
Adherens Junctions
- ALI
Acute Lung Injury
- APC
Activated Protein C
- ARDS
Acute Respiratory Distress Syndrome
- Arg
Abl Related Gene
- BBB
Blood Brain Barrier
- BCR
Breakpoint Cluster Region
- CLP
Cecal Ligation and Puncture
- CML
Chronic Myelogenous Leukemia
- CS
Cyclic Stretch
- CTTN
Cortactin
- EC
Endothelial Cells
- EGF
Epidermal Growth Factor
- F
Bioavailability
- eNOS
Endothelial Nitric Oxide Synthase
- FA
Focal Adhesions
- GPx-1
Glutathione Peroxidase 1
- H2O2
Hydrogen Peroxide
- HPAEC
Human Pulmonary Artery Endothelial Cells
- HUVEC
Human Umbilical Vein Endothelial Cells
- IP3
Inositol 3-phosphate
- IP3R
IP3 Receptor
- IT
Intratracheal
- IV
Intravenous
- KO
Knockout
- LPS
Lipopolysaccharide
- MEF
Mouse Embryonic Fibroblasts
- nmMLCK
Non Muscle Myosin Light Chain Kinase
- MV
Mechanical Ventilation
- PAR1
Protease-activated Receptor 1
- NO
Nitric oxide
- PBMC
Peripheral Blood Mononuclear Cells
- PDGF
Platelet Derived Growth Factor
- PH
Pulmonary Hypertension
- PKG
Protein Kinase G
- ROS
Reactive Oxygen Species
- S1P
Sphingosine 1-phosphate
- S1PR1
Sphingosine 1-phosphate Receptor 1
- SCF
Stem Cell Factor
- SH
Src Homology
- TLR4
Toll Like Receptor 4
- TRAP
Thrombin Receptor Activating Peptide
- VCAM
Vascular Cellular Adhesion Molecule
- Vd
Volume of distribution
- VEGF
Vascular Endothelial Growth Factor
- VE-Cad
Vascular Endothelial Cadherin
- VEGFR2
VEGF Receptor 2
- VILI
Ventilator-Induced Lung Injury
- WASp
Wiskott-Aldrich Syndrome Protein
- WAVE
Wasp-Family Verprolin-Homologous Protein
REFERENCES
- 1.Angus DC, Linde-Zwirble WT, Lidicker J, Clermont G, Carcillo J, Pinsky MR. Epidemiology of severe sepsis in the united states: Analysis of incidence, outcome, and associated costs of care. Crit Care Med. 2001;29:1303–1310. doi: 10.1097/00003246-200107000-00002. [DOI] [PubMed] [Google Scholar]
- 2.Erickson SE, Martin GS, Davis JL, Matthay MA, Eisner MD, Network NNA. Recent trends in acute lung injury mortality: 1996–2005. Crit Care Med. 2009;37:1574–1579. doi: 10.1097/CCM.0b013e31819fefdf. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Dudek SM, Garcia JG. Cytoskeletal regulation of pulmonary vascular permeability. J Appl Physiol. 2001;91:1487–1500. doi: 10.1152/jappl.2001.91.4.1487. [DOI] [PubMed] [Google Scholar]
- 4.Lee WL, Slutsky AS. Sepsis and endothelial permeability. N Engl J Med. 2010;363:689–691. doi: 10.1056/NEJMcibr1007320. [DOI] [PubMed] [Google Scholar]
- 5.Bradley WD, Hernández SE, Settleman J, Koleske AJ. Integrin signaling through arg activates p190rhogap by promoting its binding to p120rasgap and recruitment to the membrane. Mol Biol Cell. 2006;17:4827–4836. doi: 10.1091/mbc.E06-02-0132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Peacock JG, Miller AL, Bradley WD, Rodriguez OC, Webb DJ, Koleske AJ. The abl-related gene tyrosine kinase acts through p190rhogap to inhibit actomyosin contractility and regulate focal adhesion dynamics upon adhesion to fibronectin. Mol Biol Cell. 2007;18:3860–3872. doi: 10.1091/mbc.E07-01-0075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bradley WD, Koleske AJ. Regulation of cell migration and morphogenesis by abl-family kinases: Emerging mechanisms and physiological contexts. J Cell Sci. 2009;122:3441–3454. doi: 10.1242/jcs.039859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Peacock JG, Couch BA, Koleske AJ. The abl and arg non-receptor tyrosine kinases regulate different zones of stress fiber, focal adhesion, and contractile network localization in spreading fibroblasts. Cytoskeleton (Hoboken) 2010;67:666–675. doi: 10.1002/cm.20479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kim IK, Rhee CK, Yeo CD, Kang HH, Lee DG, Lee SH, Kim JW. Effect of tyrosine kinase inhibitors, imatinib and nilotinib, in murine lipopolysaccharide-induced acute lung injury during neutropenia recovery. Crit Care. 2013;17:R114. doi: 10.1186/cc12786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Aman J, van Bezu J, Damanafshan A, Huveneers S, Eringa EC, Vogel SM, Groeneveld AB, Vonk Noordegraaf A, van Hinsbergh VW, van Nieuw Amerongen GP. Effective treatment of edema and endothelial barrier dysfunction with imatinib. Circulation. 2012;126:2728–2738. doi: 10.1161/CIRCULATIONAHA.112.134304. [DOI] [PubMed] [Google Scholar]
- 11.Chislock EM, Pendergast AM. Abl family kinases regulate endothelial barrier function in vitro and in mice. PLoS One. 2013;8:e85231. doi: 10.1371/journal.pone.0085231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Letsiou E, Rizzo AN, Sammani S, Naureckas P, Jacobson JR, Garcia JG, Dudek SM. Differential and opposing effects of imatinib on lps- and ventilator-induced lung injury. Am J Physiol Lung Cell Mol Physiol. 2014 doi: 10.1152/ajplung.00323.2014. ajplung.00323.02014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Stephens RS, Servinsky LE, Rentsendorj O, Kolb TM, Pfeifer A, Pearse DB. Protein kinase g increases antioxidant function in lung microvascular endothelial cells by inhibiting the c-abl tyrosine kinase. Am J Physiol Cell Physiol. 2014 doi: 10.1152/ajpcell.00375.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Colicelli J. Abl tyrosine kinases: Evolution of function, regulation, and specificity. Sci Signal. 2010;3:re6. doi: 10.1126/scisignal.3139re6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mehta D, Malik AB. Signaling mechanisms regulating endothelial permeability. Physiol Rev. 2006;86:279–367. doi: 10.1152/physrev.00012.2005. [DOI] [PubMed] [Google Scholar]
- 16.Cao C, Leng Y, Kufe D. Catalase activity is regulated by c-abl and arg in the oxidative stress response. J Biol Chem. 2003;278:29667–29675. doi: 10.1074/jbc.M301292200. [DOI] [PubMed] [Google Scholar]
- 17.Cao C, Leng Y, Huang W, Liu X, Kufe D. Glutathione peroxidase 1 is regulated by the c-abl and arg tyrosine kinases. J Biol Chem. 2003;278:39609–39614. doi: 10.1074/jbc.M305770200. [DOI] [PubMed] [Google Scholar]
- 18.Ciarcia R, Vitiello MT, Galdiero M, Pacilio C, Iovane V, d'Angelo D, Pagnini D, Caparrotti G, Conti D, Tomei V, Florio S, Giordano A. Imatinib treatment inhibit il-6, il-8, nf-kb and ap-1 production and modulate intracellular calcium in cml patients. J Cell Physiol. 2012;227:2798–2803. doi: 10.1002/jcp.23029. [DOI] [PubMed] [Google Scholar]
- 19.Wolf AM, Wolf D, Rumpold H, Ludwiczek S, Enrich B, Gastl G, Weiss G, Tilg H. The kinase inhibitor imatinib mesylate inhibits tnf-{alpha} production in vitro and prevents tnf-dependent acute hepatic inflammation. Proc Natl Acad Sci U S A. 2005;102:13622–13627. doi: 10.1073/pnas.0501758102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Weis SM, Cheresh DA. Pathophysiological consequences of vegf-induced vascular permeability. Nature. 2005;437:497–504. doi: 10.1038/nature03987. [DOI] [PubMed] [Google Scholar]
- 21.Abelson HT, Rabstein LS. Lymphosarcoma: Virus-induced thymic-independent disease in mice. Cancer Res. 1970;30:2213–2222. [PubMed] [Google Scholar]
- 22.Ben-Neriah Y, Daley GQ, Mes-Masson AM, Witte ON, Baltimore D. The chronic myelogenous leukemia-specific p210 protein is the product of the bcr/abl hybrid gene. Science. 1986;233:212–214. doi: 10.1126/science.3460176. [DOI] [PubMed] [Google Scholar]
- 23.Kruh GD, Perego R, Miki T, Aaronson SA. The complete coding sequence of arg defines the abelson subfamily of cytoplasmic tyrosine kinases. Proc Natl Acad Sci U S A. 1990;87:5802–5806. doi: 10.1073/pnas.87.15.5802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Van Etten RA, Jackson PK, Baltimore D, Sanders MC, Matsudaira PT, Janmey PA. The cooh terminus of the c-abl tyrosine kinase contains distinct f- and g-actin binding domains with bundling activity. J Cell Biol. 1994;124:325–340. doi: 10.1083/jcb.124.3.325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Galkin VE, Orlova A, Koleske AJ, Egelman EH. The arg non-receptor tyrosine kinase modifies f-actin structure. J Mol Biol. 2005;346:565–575. doi: 10.1016/j.jmb.2004.11.078. [DOI] [PubMed] [Google Scholar]
- 26.Taagepera S, McDonald D, Loeb JE, Whitaker LL, McElroy AK, Wang JY, Hope TJ. Nuclear-cytoplasmic shuttling of c-abl tyrosine kinase. Proc Natl Acad Sci U S A. 1998;95:7457–7462. doi: 10.1073/pnas.95.13.7457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cleary RA, Wang R, Waqar O, Singer HA, Tang DD. Role of c-abl tyrosine kinase in smooth muscle cell migration. Am J Physiol Cell Physiol. 2014;306:C753–C761. doi: 10.1152/ajpcell.00327.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Boyle SN, Michaud GA, Schweitzer B, Predki PF, Koleske AJ. A critical role for cortactin phosphorylation by abl-family kinases in pdgf-induced dorsal-wave formation. Curr Biol. 2007;17:445–451. doi: 10.1016/j.cub.2007.01.057. [DOI] [PubMed] [Google Scholar]
- 29.Hernández SE, Settleman J, Koleske AJ. Adhesion-dependent regulation of p190rhogap in the developing brain by the abl-related gene tyrosine kinase. Curr Biol. 2004;14:691–696. doi: 10.1016/j.cub.2004.03.062. [DOI] [PubMed] [Google Scholar]
- 30.Bays JL, Peng X, Tolbert CE, Guilluy C, Angell AE, Pan Y, Superfine R, Burridge K, DeMali KA. Vinculin phosphorylation differentially regulates mechanotransduction at cell-cell and cell-matrix adhesions. J Cell Biol. 2014;205:251–263. doi: 10.1083/jcb.201309092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Li B, Boast S, de los Santos K, Schieren I, Quiroz M, Teitelbaum SL, Tondravi MM, Goff SP. Mice deficient in abl are osteoporotic and have defects in osteoblast maturation. Nat Genet. 2000;24:304–308. doi: 10.1038/73542. [DOI] [PubMed] [Google Scholar]
- 32.Schwartzberg PL, Stall AM, Hardin JD, Bowdish KS, Humaran T, Boast S, Harbison ML, Robertson EJ, Goff SP. Mice homozygous for the ablm1 mutation show poor viability and depletion of selected b and t cell populations. Cell. 1991;65:1165–1175. doi: 10.1016/0092-8674(91)90012-n. [DOI] [PubMed] [Google Scholar]
- 33.Tybulewicz VL, Crawford CE, Jackson PK, Bronson RT, Mulligan RC. Neonatal lethality and lymphopenia in mice with a homozygous disruption of the c-abl proto-oncogene. Cell. 1991;65:1153–1163. doi: 10.1016/0092-8674(91)90011-m. [DOI] [PubMed] [Google Scholar]
- 34.Qiu Z, Cang Y, Goff SP. C-abl tyrosine kinase regulates cardiac growth and development. Proc Natl Acad Sci U S A. 2010;107:1136–1141. doi: 10.1073/pnas.0913131107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Koleske AJ, Gifford AM, Scott ML, Nee M, Bronson RT, Miczek KA, Baltimore D. Essential roles for the abl and arg tyrosine kinases in neurulation. Neuron. 1998;21:1259–1272. doi: 10.1016/s0896-6273(00)80646-7. [DOI] [PubMed] [Google Scholar]
- 36.Chislock EM, Ring C, Pendergast AM. Abl kinases are required for vascular function, tie2 expression, and angiopoietin-1-mediated survival. Proc Natl Acad Sci U S A. 2013;110:12432–12437. doi: 10.1073/pnas.1304188110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Ma Q, Huang B, Khatibi N, Rolland W, Suzuki H, Zhang JH, Tang J. Pdgfr-α inhibition preserves blood-brain barrier after intracerebral hemorrhage. Ann Neurol. 2011;70:920–931. doi: 10.1002/ana.22549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Su EJ, Fredriksson L, Geyer M, Folestad E, Cale J, Andrae J, Gao Y, Pietras K, Mann K, Yepes M, Strickland DK, Betsholtz C, Eriksson U, Lawrence DA. Activation of pdgf-cc by tissue plasminogen activator impairs blood-brain barrier integrity during ischemic stroke. Nat Med. 2008;14:731–737. doi: 10.1038/nm1787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Slutsky AS, Ranieri VM. Ventilator-induced lung injury. N Engl J Med. 2013;369:2126–2136. doi: 10.1056/NEJMra1208707. [DOI] [PubMed] [Google Scholar]
- 40.Dudek SM, Chiang ET, Camp SM, Guo Y, Zhao J, Brown ME, Singleton PA, Wang L, Desai A, Arce FT, Lal R, Van Eyk JE, Imam SZ, Garcia JG. Abl tyrosine kinase phosphorylates nonmuscle myosin light chain kinase to regulate endothelial barrier function. Mol Biol Cell. 2010;21:4042–4056. doi: 10.1091/mbc.E09-10-0876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Finigan JH, Dudek SM, Singleton PA, Chiang ET, Jacobson JR, Camp SM, Ye SQ, Garcia JG. Activated protein c mediates novel lung endothelial barrier enhancement: Role of sphingosine 1-phosphate receptor transactivation. J Biol Chem. 2005;280:17286–17293. doi: 10.1074/jbc.M412427200. [DOI] [PubMed] [Google Scholar]
- 42.Wang L, Chiang ET, Simmons JT, Garcia JG, Dudek SM. Fty720-induced human pulmonary endothelial barrier enhancement is mediated by c-abl. Eur Respir J. 2011;38:78–88. doi: 10.1183/09031936.00047810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Lapetina S, Mader CC, Machida K, Mayer BJ, Koleske AJ. Arg interacts with cortactin to promote adhesion-dependent cell edge protrusion. J Cell Biol. 2009;185:503–519. doi: 10.1083/jcb.200809085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Sossey-Alaoui K, Li X, Cowell JK. C-abl-mediated phosphorylation of wave3 is required for lamellipodia formation and cell migration. J Biol Chem. 2007;282:26257–26265. doi: 10.1074/jbc.M701484200. [DOI] [PubMed] [Google Scholar]
- 45.Burton EA, Oliver TN, Pendergast AM. Abl kinases regulate actin comet tail elongation via an n-wasp-dependent pathway. Mol Cell Biol. 2005;25:8834–8843. doi: 10.1128/MCB.25.20.8834-8843.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Woodring PJ, Meisenhelder J, Johnson SA, Zhou GL, Field J, Shah K, Bladt F, Pawson T, Niki M, Pandolfi PP, Wang JY, Hunter T. C-abl phosphorylates dok1 to promote filopodia during cell spreading. J Cell Biol. 2004;165:493–503. doi: 10.1083/jcb.200312171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Mader CC, Oser M, Magalhaes MA, Bravo-Cordero JJ, Condeelis J, Koleske AJ, Gil-Henn H. An egfr-src-arg-cortactin pathway mediates functional maturation of invadopodia and breast cancer cell invasion. Cancer Res. 2011;71:1730–1741. doi: 10.1158/0008-5472.CAN-10-1432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Zandy NL, Playford M, Pendergast AM. Abl tyrosine kinases regulate cell-cell adhesion through rho gtpases. Proc Natl Acad Sci U S A. 2007;104:17686–17691. doi: 10.1073/pnas.0703077104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Zandy NL, Pendergast AM. Abl tyrosine kinases modulate cadherin-dependent adhesion upstream and downstream of rho family gtpases. Cell Cycle. 2008;7:444–448. doi: 10.4161/cc.7.4.5452. [DOI] [PubMed] [Google Scholar]
- 50.Lewis JM, Baskaran R, Taagepera S, Schwartz MA, Wang JY. Integrin regulation of c-abl tyrosine kinase activity and cytoplasmic-nuclear transport. Proc Natl Acad Sci U S A. 1996;93:15174–15179. doi: 10.1073/pnas.93.26.15174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lewis JM, Schwartz MA. Integrins regulate the association and phosphorylation of paxillin by c-abl. J Biol Chem. 1998;273:14225–14230. doi: 10.1074/jbc.273.23.14225. [DOI] [PubMed] [Google Scholar]
- 52.Moresco EM, Donaldson S, Williamson A, Koleske AJ. Integrin-mediated dendrite branch maintenance requires abelson (abl) family kinases. J Neurosci. 2005;25:6105–6118. doi: 10.1523/JNEUROSCI.1432-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Collins T, Read MA, Neish AS, Whitley MZ, Thanos D, Maniatis T. Transcriptional regulation of endothelial cell adhesion molecules: Nf-kappa b and cytokine-inducible enhancers. FASEB J. 1995;9:899–909. [PubMed] [Google Scholar]
- 54.Mokhtari D, Li T, Lu T, Welsh N. Effects of imatinib mesylate (gleevec) on human islet nf-kappab activation and chemokine production in vitro. PLoS One. 2011;6:e24831. doi: 10.1371/journal.pone.0024831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Hägerkvist R, Sandler S, Mokhtari D, Welsh N. Amelioration of diabetes by imatinib mesylate (gleevec): Role of beta-cell nf-kappab activation and anti-apoptotic preconditioning. FASEB J. 2007;21:618–628. doi: 10.1096/fj.06-6910com. [DOI] [PubMed] [Google Scholar]
- 56.Kallet RH, Matthay MA. Hyperoxic acute lung injury. Respir Care. 2013;58:123–141. doi: 10.4187/respcare.01963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Cao C, Leng Y, Li C, Kufe D. Functional interaction between the c-abl and arg protein-tyrosine kinases in the oxidative stress response. J Biol Chem. 2003;278:12961–12967. doi: 10.1074/jbc.M300058200. [DOI] [PubMed] [Google Scholar]
- 58.Overbeek MJ, van Nieuw Amerongen GP, Boonstra A, Smit EF, Vonk-Noordegraaf A. Possible role of imatinib in clinical pulmonary veno-occlusive disease. Eur Respir J. 2008;32:232–235. doi: 10.1183/09031936.00054407. [DOI] [PubMed] [Google Scholar]
- 59.Druker BJ, Talpaz M, Resta DJ, Peng B, Buchdunger E, Ford JM, Lydon NB, Kantarjian H, Capdeville R, Ohno-Jones S, Sawyers CL. Efficacy and safety of a specific inhibitor of the bcr-abl tyrosine kinase in chronic myeloid leukemia. N Engl J Med. 2001;344:1031–1037. doi: 10.1056/NEJM200104053441401. [DOI] [PubMed] [Google Scholar]
- 60.Rix U, Hantschel O, Därnberger G, Remsing Rix LL, Planyavsky M, Fernbach NV, Kaupe I, Bennett KL, Valent P, Colinge J, Köcher T, Superti-Furga G. Chemical proteomic profiles of the bcr-abl inhibitors imatinib, nilotinib, and dasatinib reveal novel kinase and nonkinase targets. Blood. 2007;110:4055–4063. doi: 10.1182/blood-2007-07-102061. [DOI] [PubMed] [Google Scholar]
- 61.Schiffer CA. Bcr-abl tyrosine kinase inhibitors for chronic myelogenous leukemia. N Engl J Med. 2007;357:258–265. doi: 10.1056/NEJMct071828. [DOI] [PubMed] [Google Scholar]
- 62.Hoeper MM, Barst RJ, Bourge RC, Feldman J, Frost AE, Galie N, Gomez-Sanchez MA, Grimminger F, Grunig E, Hassoun PM, Morrell NW, Peacock AJ, Satoh T, Simonneau G, Tapson VF, Torres F, Lawrence D, Quinn DA, Ghofrani HA. Imatinib mesylate as add-on therapy for pulmonary arterial hypertension: Results of the randomized impres study. Circulation. 2013;127:1128–1138. doi: 10.1161/CIRCULATIONAHA.112.000765. [DOI] [PubMed] [Google Scholar]
- 63.Daniels CE, Wilkes MC, Edens M, Kottom TJ, Murphy SJ, Limper AH, Leof EB. Imatinib mesylate inhibits the profibrogenic activity of tgf-beta and prevents bleomycin-mediated lung fibrosis. J Clin Invest. 2004;114:1308–1316. doi: 10.1172/JCI19603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Paniagua RT, Sharpe O, Ho PP, Chan SM, Chang A, Higgins JP, Tomooka BH, Thomas FM, Song JJ, Goodman SB, Lee DM, Genovese MC, Utz PJ, Steinman L, Robinson WH. Selective tyrosine kinase inhibition by imatinib mesylate for the treatment of autoimmune arthritis. J Clin Invest. 2006;116:2633–2642. doi: 10.1172/JCI28546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Druker BJ, Guilhot F, O'Brien SG, Gathmann I, Kantarjian H, Gattermann N, Deininger MW, Silver RT, Goldman JM, Stone RM, Cervantes F, Hochhaus A, Powell BL, Gabrilove JL, Rousselot P, Reiffers J, Cornelissen JJ, Hughes T, Agis H, Fischer T, Verhoef G, Shepherd J, Saglio G, Gratwohl A, Nielsen JL, Radich JP, Simonsson B, Taylor K, Baccarani M, So C, Letvak L, Larson RA Investigators I. Five-year follow-up of patients receiving imatinib for chronic myeloid leukemia. N Engl J Med. 2006;355:2408–2417. doi: 10.1056/NEJMoa062867. [DOI] [PubMed] [Google Scholar]
- 66.Saglio G, Kim DW, Issaragrisil S, le Coutre P, Etienne G, Lobo C, Pasquini R, Clark RE, Hochhaus A, Hughes TP, Gallagher N, Hoenekopp A, Dong M, Haque A, Larson RA, Kantarjian HM Investigators E. Nilotinib versus imatinib for newly diagnosed chronic myeloid leukemia. N Engl J Med. 2010;362:2251–2259. doi: 10.1056/NEJMoa0912614. [DOI] [PubMed] [Google Scholar]
- 67.Kantarjian H, Shah NP, Hochhaus A, Cortes J, Shah S, Ayala M, Moiraghi B, Shen Z, Mayer J, Pasquini R, Nakamae H, Huguet F, Boqué C, Chuah C, Bleickardt E, Bradley-Garelik MB, Zhu C, Szatrowski T, Shapiro D, Baccarani M. Dasatinib versus imatinib in newly diagnosed chronic-phase chronic myeloid leukemia. N Engl J Med. 2010;362:2260–2270. doi: 10.1056/NEJMoa1002315. [DOI] [PubMed] [Google Scholar]
- 68.Fraunfelder FW, Solomon J, Druker BJ, Esmaeli B, Kuyl J. Ocular side-effects associated with imatinib mesylate (gleevec) J Ocul Pharmacol Ther. 2003;19:371–375. doi: 10.1089/108076803322279426. [DOI] [PubMed] [Google Scholar]
- 69.Ruan J, Luo M, Wang C, Fan L, Yang SN, Cardenas M, Geng H, Leonard JP, Melnick A, Cerchietti L, Hajjar KA. Imatinib disrupts lymphoma angiogenesis by targeting vascular pericytes. Blood. 2013;121:5192–5202. doi: 10.1182/blood-2013-03-490763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Kerkelä R, Grazette L, Yacobi R, Iliescu C, Patten R, Beahm C, Walters B, Shevtsov S, Pesant S, Clubb FJ, Rosenzweig A, Salomon RN, Van Etten RA, Alroy J, Durand JB, Force T. Cardiotoxicity of the cancer therapeutic agent imatinib mesylate. Nat Med. 2006;12:908–916. doi: 10.1038/nm1446. [DOI] [PubMed] [Google Scholar]
- 71.Tiribelli M, Colatutto A, Marin L, Barbina G, Qualizza U, Damiani D, Toffoletti E, Medeot M, Candoni A, Tonutti E, Sala P, Fanin R. Brain natriuretic peptide level as marker of cardiac function in imatinib--treated chronic myeloid leukemia patients: No evidence of cardiotoxicity of imatinib therapy. Am J Hematol. 2008;83:517–518. doi: 10.1002/ajh.21157. [DOI] [PubMed] [Google Scholar]
- 72.Ribeiro AL, Marcolino MS, Bittencourt HN, Barbosa MM, Nunes MoC, Xavier VF, Clementino NC. An evaluation of the cardiotoxicity of imatinib mesylate. Leuk Res. 2008;32:1809–1814. doi: 10.1016/j.leukres.2008.03.020. [DOI] [PubMed] [Google Scholar]
- 73.Chen S, Tang DD. C-abl tyrosine kinase regulates cytokinesis of human airway smooth muscle cells. Am J Respir Cell Mol Biol. 2014;50:1076–1083. doi: 10.1165/rcmb.2013-0438OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Lin YC, Yeckel MF, Koleske AJ. Abl2/arg controls dendritic spine and dendrite arbor stability via distinct cytoskeletal control pathways. J Neurosci. 2013;33:1846–1857. doi: 10.1523/JNEUROSCI.4284-12.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Gu JJ, Ryu JR, Pendergast AM. Abl tyrosine kinases in t-cell signaling. Immunol Rev. 2009;228:170–183. doi: 10.1111/j.1600-065X.2008.00751.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Waller CF. Imatinib mesylate. Recent Results Cancer Res. 2014;201:1–25. doi: 10.1007/978-3-642-54490-3_1. [DOI] [PubMed] [Google Scholar]
- 77.Carnevale-Schianca F, Gallo S, Rota-Scalabrini D, Sangiolo D, Fizzotti M, Caravelli D, Capaldi A, Anselmetti G, Palesandro E, D'Ambrosio L, Coha V, Obert R, Aglietta M, Grignani G. Complete resolution of life-threatening bleomycin-induced pneumonitis after treatment with imatinib mesylate in a patient with hodgkin's lymphoma: Hope for severe chemotherapy-induced toxicity? J Clin Oncol. 2011;29:e691–e693. doi: 10.1200/JCO.2011.35.6733. [DOI] [PubMed] [Google Scholar]
- 78.Aman J, Peters MJ, Weenink C, van Nieuw Amerongen GP, Vonk Noordegraaf A. Reversal of vascular leak with imatinib. Am J Respir Crit Care Med. 2013;188:1171–1173. doi: 10.1164/rccm.201301-0136LE. [DOI] [PubMed] [Google Scholar]
- 79.Kim JY, Choi JS, Song SH, Im JE, Kim JM, Kim K, Kwon S, Shin HK, Joo CK, Lee BH, Suh W. Stem cell factor is a potent endothelial permeability factor. Arterioscler Thromb Vasc Biol. 2014;34:1459–1467. doi: 10.1161/ATVBAHA.114.303575. [DOI] [PubMed] [Google Scholar]
- 80.Minshall RD, Tiruppathi C, Vogel SM, Niles WD, Gilchrist A, Hamm HE, Malik AB. Endothelial cell-surface gp60 activates vesicle formation and trafficking via g(i)-coupled src kinase signaling pathway. J Cell Biol. 2000;150:1057–1070. doi: 10.1083/jcb.150.5.1057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Dudek SM, Jacobson JR, Chiang ET, Birukov KG, Wang P, Zhan X, Garcia JG. Pulmonary endothelial cell barrier enhancement by sphingosine 1-phosphate: Roles for cortactin and myosin light chain kinase. J Biol Chem. 2004;279:24692–24700. doi: 10.1074/jbc.M313969200. [DOI] [PubMed] [Google Scholar]
- 82.Kurimoto N, Nan YS, Chen ZY, Feng GG, Komatsu T, Kandatsu N, Ko J, Kawai N, Ishikawa N. Effects of specific signal transduction inhibitors on increased permeability across rat endothelial monolayers induced by neuropeptide y or vegf. Am J Physiol Heart Circ Physiol. 2004;287:H100–H106. doi: 10.1152/ajpheart.00922.2003. [DOI] [PubMed] [Google Scholar]
- 83.Rhee CK, Lee SH, Yoon HK, Kim SC, Lee SY, Kwon SS, Kim YK, Kim KH, Kim TJ, Kim JW. Effect of nilotinib on bleomycin-induced acute lung injury and pulmonary fibrosis in mice. Respiration. 2011;82:273–287. doi: 10.1159/000327719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Breccia M, Alimena G. Second-generation tyrosine kinase inhibitors (tki) as salvage therapy for resistant or intolerant patients to prior tkis. Mediterr J Hematol Infect Dis. 2014;6:e2014003. doi: 10.4084/MJHID.2014.003. [DOI] [PMC free article] [PubMed] [Google Scholar]