Abstract
Foxp3-expressing regulatory T cells (Tregs) are central regulators of immune homeostasis and tolerance. As it has been suggested that proper Treg function is compromised under inflammatory conditions, seeking for a pathway that enhances or stabilizes Treg function is a subject of considerable interest. We report that IL-27, an IL-12 family cytokine known to have both pro- and anti inflammatory roles in T cells, plays a pivotal role in enhancing Treg function to control T cell-induced colitis, a model for inflammatory bowel disease (IBD) in humans. Unlike wild type (WT) Tregs capable of inhibiting colitogenic T cell expansion and inflammatory cytokine expression, IL-27R-deficient Tregs were unable to downregulate inflammatory T cell responses. Tregs stimulated with IL-27 expressed substantially improved suppressive function in vitro and in vivo. IL-27 stimulation of Tregs induced expression of Lag3, a surface molecule implicated in negatively regulating immune responses. Lag3 expression in Tregs was critical to mediate Treg function in suppressing colitogenic responses. Human Tregs also displayed enhanced suppressive function and Lag3 expression following IL-27 stimulation. Collectively, these results highlight a novel function for the IL-27/Lag3 axis in modulating Treg regulation of inflammatory responses in the intestine.
Keywords: CD4, colitis, IBD, Interleukin-27, Lymphocyte activation gene 3, Treg
Introduction
Foxp3+ regulatory T cells (Tregs) are key regulators of immune homeostasis and tolerance.1 Dysregulated Treg function is linked to the development of inflammatory disorders including inflammatory bowel disease (IBD). There is increasing evidence that inflammation negatively impacts Treg homeostasis and function. Proinflammatory cytokines can directly alter Treg function and confer resistance of inflammatory lymphocytes to Treg-mediated suppression.2-5 Moreover, Treg numbers in patients with inflammatory diseases are often found diminished.2, 6 Therefore, correcting the defects in Treg homeostasis or improving their regulatory function is a subject of considerable interest.
IBD is an idiopathic disease of the intestine mediated by uncontrolled activation of immune cells. CD4 T cells producing IFNγ IL-17, and other inflammatory cytokines in response to commensal antigens (Ag) are major mediators of the disease.7 The importance of Tregs in controlling IBD is well exemplified by the severe intestinal inflammation observed in individuals with the foxp3 gene mutation.8, 9 Experimentally, T cell-induced colitis that shares histopathologic features with IBD is only induced when Tregs are absent and Treg transfer prevents the development of the disease.10, 11 However, greater Treg numbers are often found in the inflamed intestine of IBD patients, suggesting that those Tregs may not properly function to control the inflammation.12 Therefore, finding pathways that promote and enhance Treg function is important to overcome the adverse effects of inflammation.
IL-27 is an IL-12 family cytokine composed of the IL-27p28 and Ebi3 subunits produced by activated antigen presenting cells (APCs).13 IL-27 induces IL-10 production by CD4 effector and Tr1 cells, and plays an important regulatory role in attenuating Th17-mdiated inflammation.14-16 IL-27 also interferes with TGFβ-induced inducible Treg (iTreg) generation and adoptive transfer of Il27ra −/− naïve CD4 T cells into immunodeficient recipients results in attenuated colitis, which is attributed to increased iTreg generation.17, 18 IL-27 also promotes Tbet and CXCR3 expression in Foxp3+ Tregs specialized in controlling Th1-mediated immunity at sites of inflammation.19 Yet, a role for IL-27 in modulating Treg function during Th17 type immunity has not been explored.
Here we identified a novel function for IL-27 in enhancing Treg suppression in inhibiting both T cell expansion and differentiation into Th1/Th17 type colitogenic effector cells during intestinal inflammation. Furthermore, IL-27 stimulation of Tregs significantly improved the Treg function both in vitro and in vivo. Lag3 expression on Tregs induced by IL-27 stimulation played an essential role in inhibiting the early T cell activation and attenuating inflammation in the intestine. Our findings uncover a previously unknown role for IL-27 stimulation in Tregs to modulate Treg suppressive function possibly via Lag3, providing an important basis for the development of novel therapeutic strategies to treat IBD by targeting IL-27/Lag3 in Tregs.
Results
Il27ra−/− Foxp3+ Tregs fail to control T cell-mediated colitis
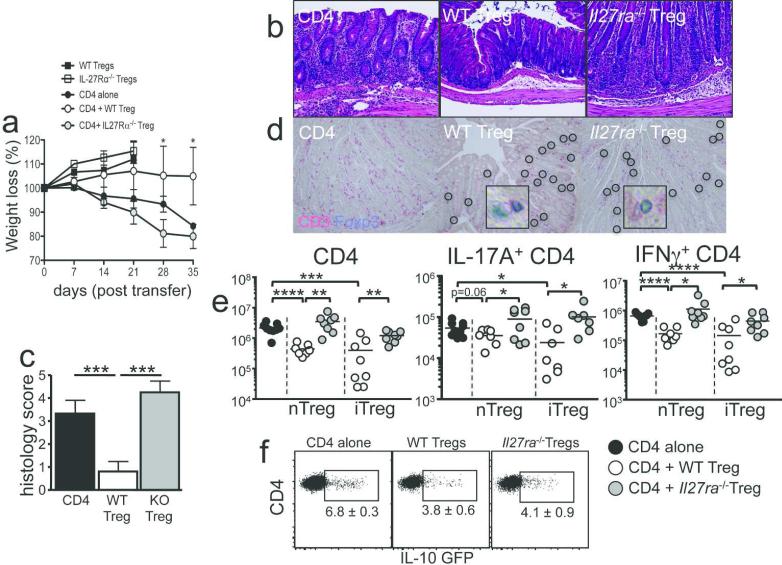
Naïve CD4 T cells transferred into immunodeficient recipients differentiate into Th1/Th17 effector cells and induce colitis, and the inflammation can be prevented by Foxp3+ Treg co-transfer.20, 21 To test the role of IL-27 signaling in Tregs in the context of intestinal inflammation we transferred Rag1 −/− mice with naïve CD4 T cells alone, or together with WT or Il27ra −/− CD25+ thymus-derived tTregs (nTregs), and assessed the development of colitis. Rag1 −/− recipients of CD4 T cells demonstrated a significant weight loss (Figure 1a) and severe inflammation in the colon (Figure 1b and 1c). Severe infiltration of CD3+ T cells in the colon was noted (Figure 1d). WT nTregs cotransferred efficiently prevented the development of inflammation as determined by weight loss (Figure 1a) and histopathology (Figure 1b and 1c). Likewise, CD3+ T cell infiltration was significantly reduced (Figure 1d). Unexpectedly, Il27ra −/− nTregs were unable to suppress the inflammation, and the recipients developed severe disease (Figure 1a-d). IL-27-dependent Treg function was also observed when TGFβ-induced Foxp3+ Tregs (iTregs) were used. WT iTregs cotransferred fully suppressed the inflammation; however, Il27ra −/− iTregs failed to do so (not shown). Treg suppression of colitis was associated with their ability to suppress T cell responses. WT Tregs (both nTregs and iTregs) significantly inhibited CD4 T cell expansion (and infiltration) and T cell expression of Th1/Th17 cytokines in the colon tissues, while such inhibition was not found when Il27ra −/− Tregs were used (Figure 1d-e). IL-10+ conventional CD4 T cells (Tr1 cells) prevent colitis.22 CD4 T cell IL-10 production was measured by transferring Il10GFP naïve CD4 T cells together with WT or Il27ra −/− Tregs. As shown in Figure 1f, GFP expression of CD4 T cells was similar in recipients of WT or Il27ra −/− Tregs. Based on these findings we concluded that IL-27 signaling in Tregs plays an important role in mediating Treg function during T cell-induced colitis. Given that CD4 T cells express functional IL-27Rα, the defect of Il27ra −/− Tregs appears to be cell intrinsic.
Figure 1. IL-27 signaling in Tregs is essential for Treg suppressive function.
Naïve CD45.1+ CD4 T cells were transferred into groups of Rag1 −/− mice (0.25 × 106 cells). CD45.2+ Tregs or CD45.2+ iTregs were transferred together with naïve T cells (0.25 × 106 cells). The recipients were sacrificed 35 days post transfer. (a) Body weight was weekly monitored. (b) Colon tissue was H&E stained (10x). (c) Histology score. (d) Immunohistochemistry analysis for CD3 and Foxp3 expression (20x). Inset, Foxp3+ cells are shown (60x). (e) T cell numbers in the colon LP were enumerated. T cells producing IL-17A or IFNγ were counted. (f) Naïve Il10GFP CD4+ T cells were transferred with WT or Il27ra −/− CD45.1+ Tregs into Rag1−/− mice. GFP expression of CD45.2+ T cells in the mLN was measured. All the experiments were repeated more than twice. Error bar indicated mean ± SD, N=3-7. Each symbol represents individually tested animal. *, p<0.05; **, p<0.01; ***, p<0.001; ****, p<0.0001.
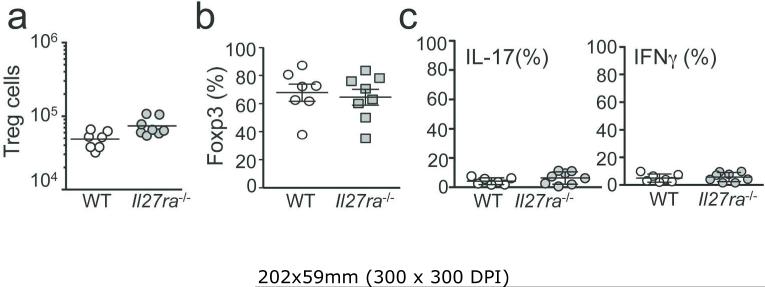
Il27ra−/− Tregs do not display defects in survival or stability
Controversial findings with regard to Il27ra −/− Treg function were reported.18, 23 Cox et al. demonstrated that Il27ra −/− Tregs prevent T cell-induced colitis.18 Kim et al. reported that Il27ra −/− Tregs fail to suppress colitis, which was attributed to impaired survival in the absence of IL-27Rα.23 Unlike the previous studies, the overall Tregs numbers in the colon and lymphoid tissues of the groups shown in Fig 1 remained similar regardless of IL-27Rα expression (Figure 2a), suggesting that the absence of IL-27 signaling does not alter Treg expansion (and/or survival). Immunohistochemistry analysis for Foxp3 expressing cells in the colon confirmed that similar numbers of Foxp3+ cells exist in the colon of WT or Il27ra −/− Treg recipients (Figure 1d). The mechanism underlying the discrepant results is unclear. Different genetic background or commensal bacteria may underlie the different results. Under inflammatory conditions Tregs could lose Foxp3 expression and acquire effector function.24 Yet, Foxp3 expression in WT and Il27ra −/− Tregs was maintained at similar levels (Figure 2b). Moreover, Treg expression of inflammatory cytokines was similarly low regardless of IL-27Rα expression (Figure 2c). Some conventional CD4 T cells acquire Foxp3 expression during proliferation within lymphopenic recipients and play a role in attenuating inflammation.18 Foxp3 expression in conventional CD4 T cells remained similar between all the tested groups (not shown).
Figure 2. Phenotype of WT and Il27ra−/− Tregs.
Naïve CD45.1+ CD4 T cells were transferred into groups of Rag1 −/− mice. CD45.2+ Tregs were cotransferred. The recipients were sacrificed 35 days post transfer. (a) Tregs in the colon LP. (b) Treg Foxp3 expression. (c) IFNγ and IL-17A expression of Tregs. All the experiments were repeated more than twice. Error bar indicated mean ± SD, N=7-8. Each symbol represents individually tested animal.
The lack of IL-27Rα does not affect Treg generation and in vitro suppressive function
Consistent with a previous report,25 the proportion of Foxp3+ nTregs was comparable in WT Foxp3GFP and Il27ra −/− Foxp3GFP mice (Supplementary Figure S1a). TGFβ-induced Foxp3 expression was also comparable in WT and Il27ra −/− cells (not shown). Surface molecules associated with Treg function, such as CTLA 4, CD39, CD73, GITR, and neuropilin-1 (Nrp-1) were expressed at comparable levels on WT and Il27ra −/− Tregs (Supplementary Figure S1b). Finally, suppression measured by in vitro suppression assay was not affected by the lack of IL-27Rα (Supplementary Figure S1c). These results suggest that IL-27 signaling in Tregs appears to be dispensable for Treg homeostasis at steady-state condition and that it may become critical under inflammatory conditions.
Il27ra−/− Tregs fail to inhibit early endogenous proliferation in the gut draining lymph node
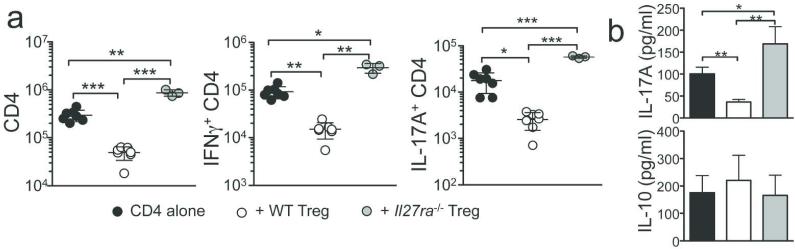
CD4 T cells undergo endogenous proliferation in response to commensal Ag mainly in the gut draining mesenteric LN (mLN) and cotransferred Tregs suppress the proliferation.26 We examined if Il27ra −/− Tregs are defective in suppressing the proliferative responses at the priming site. WT Tregs dramatically inhibited both proliferation and cytokine production, while Il27ra −/− Tregs failed to do so (Figure 3a). IL-17 protein secretion from the colon tissue culture was similarly inhibited by WT but not by Il27ra −/− Tregs (Figure 3b). Of note, IL-10 secretion from the colon was comparable (Figure 3b). Therefore, IL-27 signaling in Foxp3+ Tregs is critical to control endogenous T cell proliferation and concomitant differentiation into Th1/Th17 type effector cells.
Figure 3. Il27ra−/− Tregs fail to inhibit early T cell expansion and cytokine expression.
Groups of Rag1 −/− mice received naïve CD45.1+ CD4 T cells and Tregs. (a) Total numbers of CD45.1+ T cells in the mLN at day 7. (b) IL-17A and IL-10 production from the whole colon tissue culture. Error bar indicated mean ± SD, N=3-7. Each symbol represents individually tested animal. *, p<0.05; **, p<0.01; ***, p<0.001;
IL-27 enhances Treg suppressive function
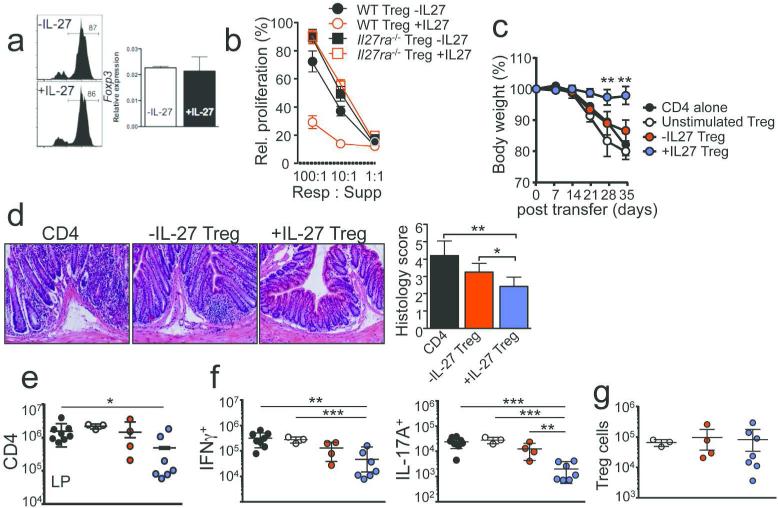
The result that IL-27 signaling in Tregs is essential for suppressive function prompted us to hypothesize that IL-27 enhances Treg function. To test the hypothesis, we activated Tregs with anti CD3/CD28 Abs with IL-2 alone or with IL-2 + IL-27 and then compared the suppressive function. IL-27 stimulation did not affect Foxp3 expression (Figure 4a). Tregs activated without IL-27, expressing >90% suppression when mixed with responder cells at a 1:1 ratio, demonstrated diminished suppression at a 10:1 ratio and lost the suppression at a 100:1 ratio (Figure 4b, filled circle). By contrast, IL-27 pre-stimulated Tregs (+IL-27) maintained ~70% inhibition even at the ratio of 100:1 (Figure 4b, open circle). The IL-27-mediated effect was IL-27-specific since IL-27 stimulated Il27ra −/− Tregs did not display enhanced suppression (Figure 4b, open square). To test whether IL-27-induced improvement of Treg suppression is also seen in vivo, Tregs pre-stimulated with or without IL-27 were administered into Rag1 −/− recipients of CD4 T cells when the recipients expressed the sign of weight loss (day 14 post transfer). It was previously reported that Tregs transferred can cure ongoing colitis.27 However, we found no evidence that freshly isolated unstimulated Tregs transferred after the disease onset reverse the weight loss (Figure 4c). This is probably due to the fact that we only transfer 0.25 × 106 Tregs for treatment compared to 1 × 106 Tregs previously used.27 While Tregs stimulated without IL-27 (-IL-27 Treg) similarly failed to attenuate the disease process (Figure 4c), the recipients of IL-27 stimulated Tregs (+IL-27 Treg) maintained body weight throughout the experiments (Figure 4c). The colon tissues from these groups showed significantly reduced inflammation (Figure 4d). These improved clinical signs were further supported by the suppression of colitogenic T cell responses. Both CD4 T cell expansion (Figure 4e) and cytokine production (Figure 4f) were significantly diminished when IL-27 pre-stimulated Tregs were transferred, while the suppression was lost in recipients of naïve or control Tregs (Figure 4e-4f). Of note, the absolute numbers of Tregs transferred remained similar regardless of Treg stimulation status (Figure 4g), suggesting that the different treatments do not influence Treg expansion and/or survival. These results suggest that IL-27 stimulation enhances Treg suppressive function and that IL-27 stimulated Tregs express significantly improved potential for treating intestinal inflammation.
Figure 4. IL-27 enhances Treg suppressive function.
(a) Tregs were stimulated on anti-CD3/CD28 mAb for 3 days with or without IL-27. Foxp3 expression was measured by flow cytometry and qPCR. (b) WT and Il27ra −/− Tregs were pre-stimulated with or without IL-27. Suppression was determined as described in Methods. (c) Tregs stimulated with or without IL-27 or naïve Tregs were transferred into Rag1 −/− recipients of naïve CD4 T cells at the onset of disease (day 14 post transfer). Body weight was weekly monitored. (d) Histopathology (x20) and histology score (day 35). (e) CD4 T cell expansion in the colon LP was determined. (f) T cell IFNγ and IL-17A expression in the colon. (g) Number of Tregs in the LP. Data shown are representative of two or more independent experiments. Error bar indicated mean ± SD, N=3-7. Each symbol represents individually tested animal. *, p<0.05; **, p<0.01; ***, p<0.001.
Lag3 plays an important role during IL-27-mediated enhanced Treg function
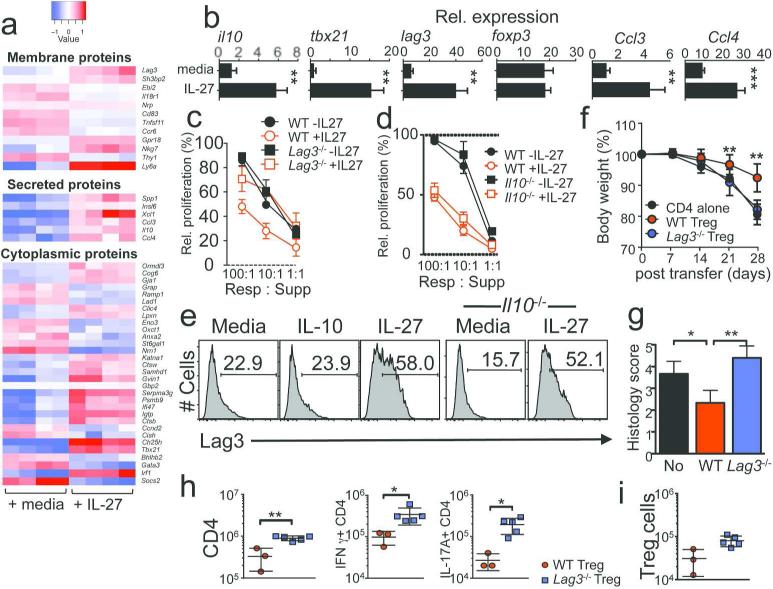
To gain insights into the mechanism underlying IL-27 modulation of Treg function, we compared gene expression profiles of Tregs stimulated with or without IL-27 using an Illumina microarray. We found that 43 and 23 genes were upregulated and downregulated in IL-27-stimulated Tregs, respectively, compared to control Tregs (Figure 5a). Among the genes upregulated by IL-27 included: Lag3, Ccl3, Ccl4, Tbx21, and Il10, while genes downregulated by IL-27 included: Gata3, Socs2, and Tnfsf11 (Figure 5a). qPCR analysis confirmed that IL-27 upregulated Tbx21, Lag3, Ccl3, Ccl4, and Il10 expression (Figure 5b). We were particularly interested in Lag3, a CD4-like molecule expressed in activated T cells or Treg and implicated in negatively regulating immune responses.28 While WT and Lag3−/− Tregs were equally suppressive in vitro (Figure 5c, filled symbols), Lag3 −/− Tregs were defective in preventing colitis (Supplementary Figure S2). Whether IL-27 pre-stimulated Tregs requires Lag3 to express enhanced suppressive function was next examined. Unlike WT Tregs, IL-27-induced enhanced suppression in vitro was significantly diminished in Lag3 −/− Tregs (Figure 5c, open square). IL-10 can be induced by IL-27 stimulation in Tregs (Figure 5b) and Treg IL-10 secretion may mediate suppressive function.36 However, unlike Lag3 −/− Tregs, IL-27 stimulation significantly improved Il10 −/− Treg suppression (Figure 5d), suggesting that IL-27 may enhance Treg function in an IL-10-independent mechanism. IL-10 itself did not upregulate Lag3 (Figure 5e). Moreover, IL-27-induced Lag3 expression in Il10 −/− Tregs (Figure 5e), further demonstrating a direct role of IL-27 in Lag3 upregulation. Rag1 −/− recipients of IL-27-stimulated Lag3 −/− Tregs continuously lost weight similar to CD4 alone group (Figure 5f). Histopathologic examination further supported these findings (Figure 5g). Likewise, Lag3 −/− Tregs were unable to inhibit both CD4 T cell expansion and Th1/Th17 cytokine production in the target tissues (Figure 5h). Lastly, the inability of Lag3 −/− Tregs to suppress colitogenic T cell responses was not due to their defects in survival and/or expansion, since both WT and Lag3 −/− Tregs were found equally in the recipients (Figure 5i). Lag3 −/− Tregs did not acquire effector cytokine production compared to WT Tregs (not shown). These results suggest that Lag3 expressed on Tregs may play a key role in mediating Treg function during colitic inflammation and that Lag3-mediated Treg suppression may occur independent of IL-10.
Figure 5. IL-27-mediated Lag3 expression is critical for IL-27-induced Treg function.
(a) FACS purified Foxp3+ Tregs were stimulated on immobilized anti-CD3/CD28 mAb with or without IL-27. Gene expression was compared using an Illumina microarray. (b) Expression of the listed genes was validated by qPCR. Relative expression was normalized. (c) WT or Lag3 −/− Tregs were prestimulated with or without IL-27, and in vitro suppression was determined. (d) WT or Il10 −/− Tregs were prestimulated with or without IL-27, and in vitro suppression was determined. (e) WT or Il10 −/− Tregs were stimulated in the presence of IL-10 or IL-27 for 3 days. Surface Lag3 expression was determined. (f) IL-27-prestimulated WT or Lag3 −/− Tregs were transferred into Rag1 −/− recipients of CD4 T cells at the onset of the disease (day 14 post T cell transfer). (g) Histology score. (h) CD4 T cell expansion and cytokine expression in the colon LP were determined. (i) Number of Tregs in the colon. Data shown are representative of two independent experiments. Error bar indicated mean ± SD, N=3-5. Each symbol represents individually tested animal. *, p<0.05; **, p<0.01.
Lag3 expression is necessary for Tregs to suppress endogenous proliferation
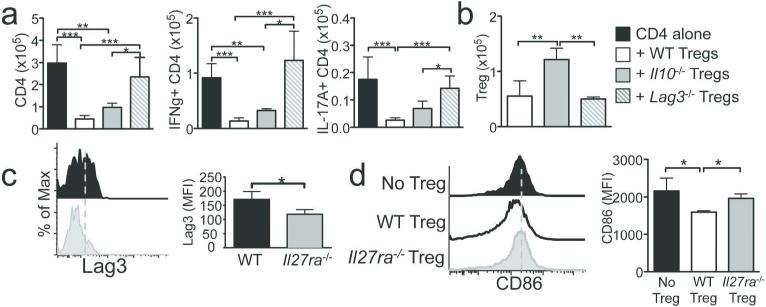
IL-27 signaling in Tregs is essential to suppress early endogenous proliferation (Figure 3a). We next examined whether Lag3 or IL-10 expressed on Tregs plays a key role in suppressing the initial T cell proliferation within the draining mLN. WT Tregs efficiently suppressed both T cell expansion and cytokine production. Likewise, Il10 −/− Tregs were also capable of suppressing T cell activation (Figure 6a). By contrast, Lag3 −/− Tregs failed to do so (Figure 6a). Of note, all tested Tregs equally expanded, although Il10 −/− Tregs seemed to expand greater than the other Tregs (Figure 6b), suggesting that the difference in Treg function is not attributed to differential expansion of Tregs. Therefore, IL-27-mediated Lag3 expression in Tregs may be critical in inhibiting the early T cell activation. Indeed, when Lag3 expression was compared between WT and Il27ra −/− Tregs in Rag1 −/− recipients, we noted that Il27ra −/− Treg expression of Lag3 was substantially lower than that of WT Tregs (Figure 6c), supporting the notion that IL-27 signaling in Tregs induces Lag3 expression in vivo. Lag3 engages MHCII molecules on DCs to limit DC maturation.29 A potent suppression of the initial T cell activation within the draining mLN by WT but not by Lag3 −/− or Il27ra −/− Tregs further supports this possibility. Indeed, mLN DCs of WT Treg recipients expressed significantly lower level of CD86 compared to those of Il27ra −/− Treg recipients (Figure 6d). These results strongly suggest that Tregs inhibit the early T cell activation within the draining lymphoid tissues and that IL-27-mediated Lag3 expression may play a key role in this process in part by inhibiting DC maturation.
Figure 6. Lag3 dependent inhibition of T cell activation in the draining lymphoid tissues.
(a) Rag1 −/− mice received CD45.1+ CD4 T cells alone, or together with WT, Il10 −/− , or Lag3 −/− Tregs. T cell expansion and cytokine expression in the mLN were determined 7 days post transfer. (b) Number of Tregs in the mLN. (c) Lag3 expression on WT and Il27ra −/− Tregs. (d) mLN DC (CD11c+ MHCII+) expression of CD86 was determined at 4 days post T cell transfer. Data shown represent the mean ± SD of 3~8 individually tested recipients. *, p<0.05; ***, p<0.001.
IL-27 stimulation enhances suppressive function and Lag3 expression in human Tregs
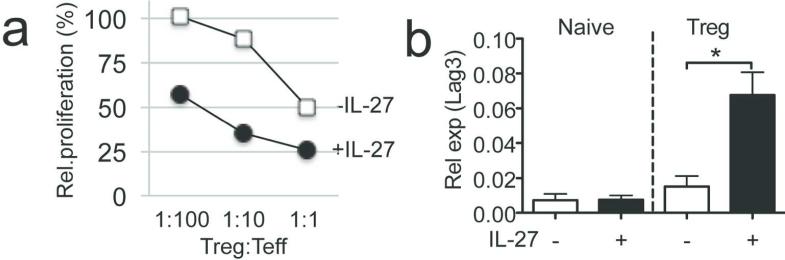
Finally, we tested whether IL-27 enhances suppressive function in human Tregs. PBMC CD25+ CD127− Tregs were isolated from healthy donors, in vitro stimulated with anti-CD3/CD28 Abs in the presence or absence of IL-27, and subsequently tested for in vitro suppression using allogeneic stimulators. IL-27-stimulated Tregs expressed significantly enhanced suppressive function compared to media-stimulated Tregs even at a 100:1 ratio (Figure 7a). Moreover, IL-27 stimulation dramatically increased Lag3 expression in Tregs but not in conventional T cells (Figure 7b). Therefore, IL-27 enhances Treg suppressive function and Lag3 expression in human Tregs.
Figure 7. IL-27 treatment increased human Tregs suppressive functions.
(a) Tregs and naïve CD4 T cells were isolated from healthy donors. Tregs were stimulated with or without IL-27. CFSE labeled naïve CD4 T cell proliferation in response to allogeneic APC stimulation in the presence of varying numbers of Tregs was evaluated. (b) Lag3 expression on conventional and Treg cells with or without IL-27 stimulation was measured by qPCR analysis. Data shown are representative of two independent experiments. Error bars indicate mean ± SD. *, p<0.05.
Discussion
The current study reports a previously uncharacterized role of IL-27 in regulating Treg function during intestinal inflammation. Earlier studies utilizing germline Il27ra −/− mice concluded that IL-27 is dispensable for Treg generation and function.16 Foxp3+ Tregs in WT and Il27ra −/− mice showed similar thymic development and peripheral maintenance. Treg suppressive function determined by a traditional in vitro suppression assay demonstrated that Il27ra −/− Tregs are equally suppressive as WT Tregs.16 Utilizing a T cell-induced colitis model, we now demonstrated that Il27ra −/− Tregs lost regulatory function to suppress colitis, suggesting that IL-27 signaling in Tregs may play a key role for Treg control of commensal Ag reactive T cell mediated colitic inflammation.
Previous studies exploring the role of IL-27 in Foxp3+ Tregs have generated controversial results. Cox et al. reported that Il27ra −/− Tregs efficiently prevent colitic inflammation using the same T cell-induced colitis model.18 Kim et al. reported opposite findings that Il27ra −/− Tregs are unable to protect colitic inflammation and that the inability is derived from poor Treg survival in the absence of IL-27Rα.23 The results reported in this study differ from those previous studies in that Il27ra −/− Tregs are unable to prevent colitis development, yet their survivability is not affected. Under severe inflammatory conditions, Tregs often lose Foxp3 expression and acquire effector function.30 However, defective Il27ra −/− Tregs in the current system maintained WT-level Foxp3 expression and did not express proinflammatory cytokines, an indication of functional defect. Cox et al. used BALB/c Il27ra −/− mice originally developed in Genentech that delete exons 3-8, i.e., most of the extracellular domain of IL-27Rα.31 Kim et al. and the current study used C57BL/6 Il27ra −/− mice developed in the Amgen institute that delete exons 7-8, the extracellular fibronectin type III domain.32 Whether different targeting strategy and/or genetic background account for the different outcome remains to be determined. As colitogenic effector T cells and Tregs would recognize commensal Ag, differences in commensal bacteria may also contribute to the different results.
Our finding that IL-27 stimulation dramatically enhances Treg suppression is worth noting. Examination of IL-27-stimulated Treg function in vitro found that ~70% suppression was still observed at the ratio of 100:1 (responder:Treg), at which ratio, neither naïve nor control stimulated Tregs expressed suppressive capacity. More interesting is that IL-27-stimulated Tregs were capable of attenuating already established intestinal inflammation, which was not observed when control or naïve Tregs were transferred. Although naïve Tregs were previously shown to cure ongoing colitis,33 it is important to note that the same therapeutic effect is achieved using less numbers of Tregs.27 Cell therapy utilizing Tregs is being considered to treat patients with chronic inflammation.34 A clinical trial testing the safety of Treg therapy in IBD patients found that the therapy is safe, although the efficacy needs to be improved.35 Therefore, IL-27-stimulation in Tregs may offer a new strategy to improve the efficacy and hopefully cost effectiveness of the therapy.
What is the cellular mechanism that confers IL-27-stimulated Tregs enhanced suppressive capacity? From an Illumina microarray approach, we identified several genes upregulated by IL-27-stimulation. Amongst them, we focused on Lag3, a surface receptor expressed on activated lymphocytes including Tregs and implicated in negatively regulating T cell activation.28 Our results show that IL-27-induced Lag3 expression in Tregs is key to control colitic inflammation, since Lag3 −/− Tregs are unable to efficiently suppress the inflammation even after IL-27-stimulation. Based on the similarity between Il27ra−/− and Lag3 −/− Tregs both in vitro and in vivo, the IL-27/Lag3 pathway appears to be critical during Treg-mediated immune suppression. It was previously shown that IL-10 production by Tregs is not required for the control of systemic autoimmunity but is essential for regulating immune responses at the intestine.21 Moreover, IL-10 secretion by Tregs within the intestinal lamina propria is believed to be critical for Treg function at effector sites.37 It is possible that Tregs may utilize both IL-10 and Lag3 to achieve the maximal function, although IL-27-induced Lag3 expression in Tregs appears to play a non replaceable role in mediating Treg function especially in attenuating ongoing inflammation in the intestine. Given that Lag3 binds to MHCII molecules to interfere with DC maturation and to suppress T cell activation,29 IL-27 may regulate Treg suppression by both contact-dependent (via Lag3) and -independent (via IL-10) mechanisms. Given that IL-27 is mainly produced by APCs activated by microbial products,13 it is also possible that different amounts of IL-27 produced at the induction vs. effector sites may induce Treg suppression by different mechanisms. Alternatively, an additional factor(s) in addition to IL-27 may be involved in Treg suppressive mechanisms. It was recently reported that Tim-3 is upregulated by IL-27 in conventional T cells and mediates inhibitory function.38 However, we observed that Tim-3 expression remained unchanged following IL-27 stimulation in Tregs (Do and Min, unpublished results). Other Treg molecules associated with suppressive functions (i.e., CTLA4, CD39, and CD73) were similarly expressed on Il27ra −/− Tregs. Therefore, the contribution of Lag3 in Treg function during chronic inflammation in the intestine or other tissues remains to be determined.
STAT3 signaling in Tregs is a key pathway that allows Tregs to specifically suppress Th17 type immunity.39 IL-10 was subsequently found to be the major STAT3 activator as Treg specific Il10ra −/− mice spontaneously develop dysregulated Th17 type inflammation in the intestine, while other STAT3-activating cytokines, IL-6 and IL-23, were unable to exert the same effect.21 Since Il27ra −/− Tregs cannot control both Th1 and Th17 type inflammation and IL-27 utilizes both STAT1 and STAT3,40 we propose that IL-27-STAT3 and IL-27-STAT1 pathways may deliver distinct signals capable of inhibiting Th17 and Th1 type immunity, respectively. The precise molecular mechanism that IL-27 inhibits Th1/Th17 type inflammatory responses requires further examination.
In summary, our results uncover a previously unknown IL-27/Lag3 pathway that controls Treg suppressive function during inflammation in the intestine. Tregs pre-stimulated with IL-27 may be considered as a novel Treg therapy with greater efficacy to treat chronic inflammatory reactions where Treg function could be compromised by inflammatory mediators.
Methods
Mice
C57BL/6, B6 Ly5.1, B6 Thy1.1, B6 Rag1 −/− mice were purchased from the Jackson Laboratory (Bar Harbor, ME). B6 Il27ra −/− mice were obtained from Amgen (Thousand Oaks, CA). B6 Foxp3GFP 41 and B6 Lag3 −/− mice were obtained from Drs. Yasmine Belkaid and Christophe Benoist, respectively. All animal procedures were conducted according to the guidelines of the Institutional Animal Care and Use Committee.
Human studies
PBMC from healthy donors were sorted for Tregs (CD25hi CD127low) using a FACSAria cell sorter (BD Bioscience, San Jose, CA). Cells were stimulated with human T-activator CD3/CD28 Dynabead (Invitrogen, Grand Island, NY) in the presence of IL-27 or media for 3 days. Tregs were harvested, and subsequently used in suppression assay. CFSE labeled naïve CD4 T cells (also obtained from healthy donors) were stimulated for 5 days with allo-APCs in the presence of varying numbers of Tregs. The study was approved by the Institutional Review Board of the Cleveland Clinic.
Cell sorting and adoptive transfer
LN cells (axillary, cervical, inguinal, and mesenteric LN) were pooled and total T cells were purified by negative selection using a magnetic separation. CD25low CD44low naive T cells were further sorted using a FACSAria cell sorter. Foxp3+ Tregs were also purified by FACS based on GFP expression, and transferred together with naïve CD4 T cells. Mice were weekly weighed. In some experiments, Rag1 −/− recipients first received naïve CD4 T cells, and monitored for weight loss. When the initial sign of weight loss was noted (typically ~14 days post transfer), Tregs were adoptively transferred. For H&E staining colon tissues were fixed in 10% Acetic Acid/60% Methanol. Slides were stained with hematoxylin and eosin.
Treg suppression assay
Tregs were stimulated on immobilized anti-CD3/CD28 mAbs for 2 days in the presence of 10 ng/ml career free murine rIL-27 (purchased from R&D system, Minneapolis, MN) and used for suppression assay. CFSE labeled naïve CD4 T cells were plated at a 1:1 ratio with mitomycin C treated T depleted splenocytes and varying concentrations of Tregs. Soluble anti CD3 (1 μg/ml) mAb was added. CFSE dilution was examined. Tregs were transferred into Rag1 −/− recipients of CD4 T cells at T cell transfer or at the onset of disease.
Lamina Propria cell Isolation
Recipients were sacrificed at the indicated time points after T cell transfer. Colons were isolated, cleaned in HBSS, cut into small pieces, resuspended in HBSS supplemented with 0.5 μM EDTA and 15 μg/mL Dithiothreitol (DTT), and shaken for 15 min twice at room temperature. Colons were then resuspended in complete RPMI supplemented with 400 μg/mL DNase and 1 mg/mL collagenase and shaken at 37°C for additional 90 min. Supernatant and colon was passed through a 70 μM cell strainer and washed. Cells were resuspended in a 33% Percoll gradient and centrifuged at room temperature for 20 min. Pellet was collected, washed and used for further experiments.
Ex vivo stimulation
Tissue cells were stimulated with PMA (10 ng/ml) and Ionomycin (1 μM) for 4 hrs in the presence of 2 μM Monensin (Calbiochem, San Diego, CA) during the last 2 hrs of incubation. Cells were immediately fixed with 4% paraformaldehyde, permeabilized, and stained with fluorescence conjugated antibodies. In some experiments, 200 mg of colon tissue was washed in cold PBS supplemented with antibiotics. Tissues were cut into small pieces and cultured in 12-well flat bottom culture plates for 24 hours. Culture supernatant was collected and measured for cytokines by ELISA.
Immunohistochemistry
Sections were fixed in acidic methanol (60% methanol, 10% acetic acid, 30% water), embedded in paraffin and sectioned at 5 micron thickness. Foxp3 and CD3 T cells were demonstrated by sequential double-staining method using rat monoclonal antibody to Foxp3 (eBioscience, clone: FJK-16s) and Rabbit polyclonal antibody to CD3 (Abcam Inc) after heat induced antigen retrieval. As secondary antibodies Rat HRP-Polymer (Biocare Medical) used for Foxp3 and Rabbit AP-Polymer (Biocare Medical) for CD3. Foxp3+ cells were stained with Vina Green chromogen (Biocare Medical) and CD3+ cells with Vulcan Fast Red (Biocare Medical). Hematoxylin was used for nuclear counterstain.
Flow cytometry
Cells were stained with anti-CD4 (RM4-5), anti-IL-17A (eBio17B7), anti-IFN-γ (XMG1.2), anti-Thy1.1 (HIS51), anti-CD45.1 (A20), anti-CD25 (PC61.5), anti-TCRβ (H57-597), anti-phospho-STAT3 (pY705) and anti-FoxP3 (FJK-16a) Abs (all Abs from eBioscience). Cells were acquired using a LSRII (BD Biosciences) flow cytometer and analyzed using a FlowJo software (Treestar, Ashland, OR).
Illumina genechip assay
FACS purified Foxp3eGFP+ Tregs were stimulated on immobilized anti-CD3/CD28 mAbs for 2 days in the presence of 10 ng/ml IL-27 or media. Total RNA was extracted using a QIAGEN RNeasy mini kit followed by clean-up and DNase I treatment in accordance with the protocol provided with the kit. Quality control was performed with Agilent Bioanalyser. Biotinylated cRNA were prepared using the Ambion MessageAmp kit for Illumina arrays. cRNA from each sample was hybridized to arrays on the Mouse Ref-8 expression beadchips for 20 hour hybridization period at 58°C. The beadchips were scanned using an Illumina iScan (Illumina, Inc., San Diego, CA) at a 0.53 Pm resolution. Future extraction was performed using the Illumina GenomeStudio.
Statistical Analysis
The RNA expression data were imported into the R Bioconductor (http://www.bioconductor.org). All raw data were log2 transformed, and the quantile normalization function in the package “lumi” 42. Differential expression was assessed using the Bayesian t statistic in the package LIMMA. P values from comparisons between Tregs treated with media and Tregs treated with IL27 were corrected for multiple testing using the false discovery rate method. Genes with corrected P < 0.05 were considered to be differentially expressed.
Data analysis
Statistical significance was determined by the Student's t-test using the Prism 4 software (GraphPad, La Jolla, CA). A p value of <0.05 was considered statically significant.
Supplementary Material
Acknowledgement
We would like to thank Jennifer Powers for cell sorting.
Grant support: NIH grant AI074932, the Crohn's Colitis Foundation of America Senior Research Award, and the American Asthma Foundation Scholar Award (to B.M.).
Abbreviations
- Ag
antigen
- APC
antigen presenting cell
- Ebi3
EBV-induced gene 3
- IBD
inflammatory bowel disease
- LP
lamina propria
- mLN
mesenteric lymph node
- Th
T helper
- Treg
regulatory T cell
- WT
wild type
Footnotes
Microarray data is available from Gene Expression Omnibus under accession no. 63455.
Disclosures: Authors have no financial disclosures to report.
Author contributions: JD conceived and designed the experiments, performed most of the experiments, and analyzed the data; AV and YOS performed experiments and analyzed the data; ND, SK, EJ, and JJB performed experiments; SAS, BS, WMB, RLF, DAAV critically read manuscript and provided reagents; BM conceived and designed the experiments, analyzed the data, and wrote the manuscript.
Accession nos.
Microarray data is available from Gene Expression Omnibus under accession no. 63455.
References
- 1.Sakaguchi S, Yamaguchi T, Nomura T, et al. Regulatory T cells and immune tolerance. Cell. 2008;133:775–87. doi: 10.1016/j.cell.2008.05.009. [DOI] [PubMed] [Google Scholar]
- 2.Valencia X, Stephens G, Goldbach-Mansky R, et al. TNF downmodulates the function of human CD4+CD25hi T-regulatory cells. Blood. 2006;108:253–61. doi: 10.1182/blood-2005-11-4567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Pasare C, Medzhitov R. Toll pathway-dependent blockade of CD4+CD25+ T cell-mediated suppression by dendritic cells. Science. 2003;299:1033–6. doi: 10.1126/science.1078231. [DOI] [PubMed] [Google Scholar]
- 4.Viglietta V, Baecher-Allan C, Weiner HL, et al. Loss of functional suppression by CD4+CD25+ regulatory T cells in patients with multiple sclerosis. J Exp Med. 2004;199:971–9. doi: 10.1084/jem.20031579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Korn T, Reddy J, Gao W, et al. Myelin-specific regulatory T cells accumulate in the CNS but fail to control autoimmune inflammation. Nat Med. 2007;13:423–31. doi: 10.1038/nm1564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Valencia X, Yarboro C, Illei G, et al. Deficient CD4+CD25high T regulatory cell function in patients with active systemic lupus erythematosus. J Immunol. 2007;178:2579–88. doi: 10.4049/jimmunol.178.4.2579. [DOI] [PubMed] [Google Scholar]
- 7.Khor B, Gardet A, Xavier RJ. Genetics and pathogenesis of inflammatory bowel disease. Nature. 2011;474:307–17. doi: 10.1038/nature10209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.van der Vliet HJ, Nieuwenhuis EE. IPEX as a result of mutations in FOXP3. Clin Dev Immunol. 2007;2007:89017. doi: 10.1155/2007/89017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ramsdell F, Ziegler SF. FOXP3 and scurfy: how it all began. Nat Rev Immunol. 2014;14:343–9. doi: 10.1038/nri3650. [DOI] [PubMed] [Google Scholar]
- 10.Boden EK, Snapper SB. Regulatory T cells in inflammatory bowel disease. Curr Opin Gastroenterol. 2008;24:733–41. doi: 10.1097/mog.0b013e328311f26e. [DOI] [PubMed] [Google Scholar]
- 11.Ostanin DV, Bao J, Koboziev I, et al. T cell transfer model of chronic colitis: concepts, considerations, and tricks of the trade. Am J Physiol Gastrointest Liver Physiol. 2009;296:G135–46. doi: 10.1152/ajpgi.90462.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Holmen N, Lundgren A, Lundin S, et al. Functional CD4+CD25high regulatory T cells are enriched in the colonic mucosa of patients with active ulcerative colitis and increase with disease activity. Inflamm Bowel Dis. 2006;12:447–56. doi: 10.1097/00054725-200606000-00003. [DOI] [PubMed] [Google Scholar]
- 13.Hall AO, Silver JS, Hunter CA. The immunobiology of IL-27. Adv Immunol. 2012;115:1–44. doi: 10.1016/B978-0-12-394299-9.00001-1. [DOI] [PubMed] [Google Scholar]
- 14.Murugaiyan G, Mittal A, Lopez Diego R, et al. IL-27 is a key regulator of IL-10 and IL-17 production by human CD4+ T cells. J Immunol. 2009;183:2435–43. doi: 10.4049/jimmunol.0900568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Fitzgerald DC, Ciric B, Touil T, et al. Suppressive effect of IL-27 on encephalitogenic Th17 cells and the effector phase of experimental autoimmune encephalomyelitis. J Immunol. 2007;179:3268–75. doi: 10.4049/jimmunol.179.5.3268. [DOI] [PubMed] [Google Scholar]
- 16.Batten M, Li J, Yi S, et al. Interleukin 27 limits autoimmune encephalomyelitis by suppressing the development of interleukin 17-producing T cells. Nat Immunol. 2006;7:929–36. doi: 10.1038/ni1375. [DOI] [PubMed] [Google Scholar]
- 17.Huber M, Steinwald V, Guralnik A, et al. IL-27 inhibits the development of regulatory T cells via STAT3. Int Immunol. 2008;20:223–34. doi: 10.1093/intimm/dxm139. [DOI] [PubMed] [Google Scholar]
- 18.Cox JH, Kljavin NM, Ramamoorthi N, et al. IL-27 promotes T cell-dependent colitis through multiple mechanisms. J Exp Med. 2011;208:115–23. doi: 10.1084/jem.20100410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hall AO, Beiting DP, Tato C, et al. The cytokines interleukin 27 and interferon-gamma promote distinct Treg cell populations required to limit infection-induced pathology. Immunity. 2012;37:511–23. doi: 10.1016/j.immuni.2012.06.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Powrie F, Leach MW, Mauze S, et al. Inhibition of Th1 responses prevents inflammatory bowel disease in scid mice reconstituted with CD45RBhi CD4+ T cells. Immunity. 1994;1:553–62. doi: 10.1016/1074-7613(94)90045-0. [DOI] [PubMed] [Google Scholar]
- 21.Chaudhry A, Samstein RM, Treuting P, et al. Interleukin 10 signaling in regulatory T cells is required for suppression of Th17 cell-mediated inflammation. Immunity. 2011;34:566–78. doi: 10.1016/j.immuni.2011.03.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cong Y, Weaver CT, Lazenby A, et al. Bacterial-reactive T regulatory cells inhibit pathogenic immune responses to the enteric flora. J Immunol. 2002;169:6112–9. doi: 10.4049/jimmunol.169.11.6112. [DOI] [PubMed] [Google Scholar]
- 23.Kim G, Shinnakasu R, Saris CJ, et al. A novel role for IL-27 in mediating the survival of activated mouse CD4 T lymphocytes. J Immunol. 2013;190:1510–8. doi: 10.4049/jimmunol.1201017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rubtsov YP, Niec RE, Josefowicz S, et al. Stability of the regulatory T cell lineage in vivo. Science. 2010;329:1667–71. doi: 10.1126/science.1191996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Batten M, Kljavin NM, Li J, et al. Cutting edge: IL-27 is a potent inducer of IL-10 but not FoxP3 in murine T cells. J Immunol. 2008;180:2752–6. doi: 10.4049/jimmunol.180.5.2752. [DOI] [PubMed] [Google Scholar]
- 26.Read S, Malmstrom V, Powrie F. Cytotoxic T lymphocyte-associated antigen 4 plays an essential role in the function of CD25(+)CD4(+) regulatory cells that control intestinal inflammation. J Exp Med. 2000;192:295–302. doi: 10.1084/jem.192.2.295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Mottet C, Uhlig HH, Powrie F. Cutting edge: cure of colitis by CD4+CD25+ regulatory T cells. J Immunol. 2003;170:3939–43. doi: 10.4049/jimmunol.170.8.3939. [DOI] [PubMed] [Google Scholar]
- 28.Huang CT, Workman CJ, Flies D, et al. Role of LAG-3 in regulatory T cells. Immunity. 2004;21:503–13. doi: 10.1016/j.immuni.2004.08.010. [DOI] [PubMed] [Google Scholar]
- 29.Liang B, Workman C, Lee J, et al. Regulatory T cells inhibit dendritic cells by lymphocyte activation gene-3 engagement of MHC class II. J Immunol. 2008;180:5916–26. doi: 10.4049/jimmunol.180.9.5916. [DOI] [PubMed] [Google Scholar]
- 30.Komatsu N, Okamoto K, Sawa S, et al. Pathogenic conversion of Foxp3+ T cells into TH17 cells in autoimmune arthritis. Nat Med. 2014;20:62–8. doi: 10.1038/nm.3432. [DOI] [PubMed] [Google Scholar]
- 31.Chen Q, Ghilardi N, Wang H, et al. Development of Th1-type immune responses requires the type I cytokine receptor TCCR. Nature. 2000;407:916–20. doi: 10.1038/35038103. [DOI] [PubMed] [Google Scholar]
- 32.Yoshida H, Hamano S, Senaldi G, et al. WSX-1 is required for the initiation of Th1 responses and resistance to L. major infection. Immunity. 2001;15:569–78. doi: 10.1016/s1074-7613(01)00206-0. [DOI] [PubMed] [Google Scholar]
- 33.Liu H, Hu B, Xu D, et al. CD4+CD25+ regulatory T cells cure murine colitis: the role of IL-10, TGF-beta, and CTLA4. J Immunol. 2003;171:5012–7. doi: 10.4049/jimmunol.171.10.5012. [DOI] [PubMed] [Google Scholar]
- 34.Gaidot A, Landau DA, Martin GH, et al. Immune reconstitution is preserved in hematopoietic stem cell transplantation coadministered with regulatory T cells for GVHD prevention. Blood. 2011;117:2975–83. doi: 10.1182/blood-2010-08-299974. [DOI] [PubMed] [Google Scholar]
- 35.Himmel ME, Yao Y, Orban PC, et al. Regulatory T-cell therapy for inflammatory bowel disease: more questions than answers. Immunology. 2012;136:115–22. doi: 10.1111/j.1365-2567.2012.03572.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rubtsov YP, Rasmussen JP, Chi EY, et al. Regulatory T cell-derived interleukin-10 limits inflammation at environmental interfaces. Immunity. 2008;28:546–58. doi: 10.1016/j.immuni.2008.02.017. [DOI] [PubMed] [Google Scholar]
- 37.Uhlig HH, Coombes J, Mottet C, et al. Characterization of Foxp3+CD4+CD25+ and IL-10-secreting CD4+CD25+ T cells during cure of colitis. J Immunol. 2006;177:5852–60. doi: 10.4049/jimmunol.177.9.5852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Zhu C, Sakuishi K, Xiao S, et al. An IL-27/NFIL3 signalling axis drives Tim-3 and IL-10 expression and T-cell dysfunction. Nat Commun. 2015;6:6072. doi: 10.1038/ncomms7072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Chaudhry A, Rudra D, Treuting P, et al. CD4+ regulatory T cells control TH17 responses in a Stat3-dependent manner. Science. 2009;326:986–91. doi: 10.1126/science.1172702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Stumhofer JS, Silver JS, Laurence A, et al. Interleukins 27 and 6 induce STAT3-mediated T cell production of interleukin 10. Nat Immunol. 2007;8:1363–71. doi: 10.1038/ni1537. [DOI] [PubMed] [Google Scholar]
- 41.Bettelli E, Carrier Y, Gao W, et al. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature. 2006;441:235–8. doi: 10.1038/nature04753. [DOI] [PubMed] [Google Scholar]
- 42.Du P, Kibbe WA, Lin SM. lumi: a pipeline for processing Illumina microarray. Bioinformatics. 2008;24:1547–8. doi: 10.1093/bioinformatics/btn224. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.