Abstract
Down syndrome, which arises in individuals carrying an extra copy of chromosome 21, is associated with a greatly increased risk of early-onset Alzheimer disease. It is thought that this risk is conferred by the presence of three copies of the gene encoding amyloid precursor protein (APP) — an Alzheimer disease risk factor — although the possession of extra copies of other chromosome 21 genes may also play a part. Further study of the mechanisms underlying the development of Alzheimer disease in people with Down syndrome could provide insights into the mechanisms that cause dementia in the general population.
Down syndrome (DS) is a complex, highly variable disorder that arises from trisomy of chromosome 21. It was one of the first chromosomal disorders to be identified1 and occurs with an incidence of approximately 1 in 800 births2. Its prevalence within a given population is influenced by infant mortality rates, access to health care, termination rates, average maternal age3 and life expectancy. Indeed, despite the increased availability of prenatal diagnosis and access to the option of termination, the global prevalence of DS is rising because of improvements in life expectancy: the number of adults with DS aged over 40 years has doubled in northern Europe since 1990 and, in the United Kingdom, one-third of the estimated 40,000 people with DS are thought to be over 40 years of age4.
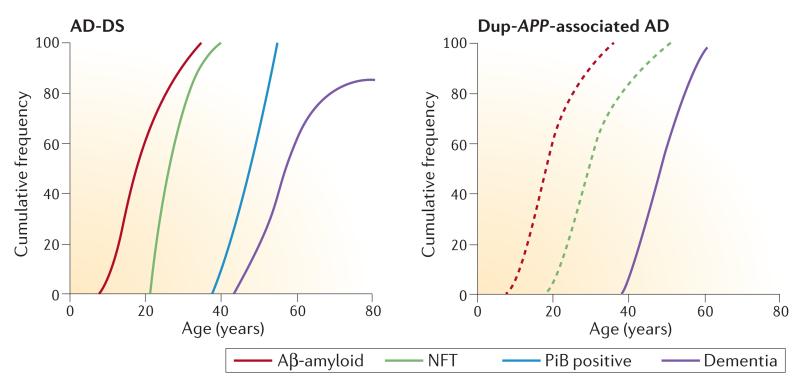
DS is the most common form of intellectual disability. In addition to the features that are found in everyone with the disorder, such as the characteristic facial dysmorphology, there are many DS-associated phenotypes that have variable penetrance and severity. For example, approximately 40% of individuals with DS have heart malformations (usually atrioventricular septal defects)5. A key feature of DS is a striking propensity to develop early-onset Alzheimer disease (EOAD). Complete trisomy of chromosome 21 universally causes the development of amyloid plaques and neurofibrillary tangles (NFTs), which are typical characteristics of AD brain pathology, by the age of 40, and approximately two-thirds of individuals with DS develop dementia by the age of 60 (REFS 6,7). However, rates of dementia do not reach 100%, even in older individuals, suggesting that some individuals with DS are protected from the onset of AD (FIG. 1).
Figure 1. Development of pathology and dementia in AD-DS and Dup-APP.
The graphs show the cumulative frequency of amyloid plaque deposition (measured using histological methods and positron emission tomography with Pittsburgh compound B (PiB), a radioactive analogue of thioflavin that binds to amyloid) and neurofibrillary tangle (NFT) development (measured using histological methods), and the cumulative frequency of dementia in people with Alzheimer disease and Down syndrome (AD-DS)6,33 and in individuals with familial AD induced by duplication of amyloid precursor protein (Dup-APP). As shown, people who have DS can live for many years with substantial amyloid deposition before the development of dementia. Solid lines are based on the data described in Supplementary information S1–S3 (tables). Dashed lines indicate hypothesized development of pathology for which there are currently no data available. Further pathological and clinical studies directly comparing these two populations are required to verify the apparent differences in clinical dementia onset and to determine whether the development of pathology differs from that proposed here. Aβ, amyloid-β.
All of the features of DS arise because of aberrant dosages of coding and/or non-coding sequences present on chromosome 21. Among these sequences, the gene encoding amyloid precursor protein (APP) is thought to have a key role in the pathology of AD. The additional copy of APP may drive the development of AD in individuals with DS (AD-DS) by increasing the levels of amyloid-β (Aβ), a cleavage product of APP that misfolds and accumulates in the brain in people with AD. Consistent with this hypothesis, individuals with small internal chromosome 21 duplications that result in three copies of APP — a rare familial trait known as duplication of APP (Dup-APP) — also develop EOAD8–15. Conversely, partial trisomy of chromosome 21 that does not result in the presence of an extra APP does not lead to AD16,17. Several additional genes on chromosome 21 are proposed to modulate the course of AD-DS, but further work is required to determine their role and relative importance.
The aim of this Opinion article is to present an overview of clinical and pathological features of AD-DS and, by comparing these to other forms of AD (particularly AD induced by Dup-APP), to highlight shared genetic, pathogenic and protective mechanisms and to discuss key future research areas. Similarities in the aetiologies of AD-DS and other forms of AD may highlight common disease mechanisms, whereas differences between these forms of AD may help to identify novel genes and pathways that are important in particular aspects of AD. Recent advances in genetic, cellular and neuroimaging technologies have provided the means to comprehensively explore the link between AD and DS, and recent improvements in the life expectancy of people who have DS mean that more individuals than ever before are developing AD-DS. The growing interest in AD-DS is long overdue, given the high AD burden in the DS population, and it is likely that research into AD-DS may also lead to a better understanding of AD in the general population.
Prevalence of AD-DS
A loss of cognitive function in middle-aged adults with DS was described soon after the identification of the syndrome18, and it was later shown that this loss resulted from the onset of AD dementia. As indicated above, AD is now common in adults with DS who are over the age of 40 years and, like other genetic forms of EOAD, develops two to three decades earlier in individuals with DS than in the general population. Data describing the prevalence of AD-DS vary between studies because of diagnostic issues, such as the presence of variable premorbid deficits, and survey methodology19. However, the prevalence of AD in people who have DS is <5% under the age of 40 (REF. 20) and then roughly doubles with each 5-year interval up to the age of 60. Hence, approximately 5–15% of individuals with DS aged 40–49 years and >30% of those aged 50–59 years experience significant cognitive decline, indicating dementia (FIG. 1). Thus, as with AD in the general population, age is a strong independent risk factor for AD-DS21. By the age of 65, 68–80% of individuals with DS have been shown to have developed dementia6,7 (FIG. 1; Supplementary information S1 (table)), and some studies of institutionalized people with DS suggest that rates are even higher6,20,22. However, not all older individuals with DS develop dementia, with some reaching their 70s without significant symptoms of AD despite having full trisomy of chromosome 21 (REF. 23). After the age of 60, prevalence rates decrease, probably owing to the high mortality rate that is associated with dementia21.
The average age at which menopause begins in women with DS correlates with the age of onset of dementia24–26; however, unlike the incidence of AD in euploid individuals, gender does not affect the incidence of AD-DS20,21. The reasons for this difference between the two populations are unknown, although it is possible that trisomy may cause changes in hormonal or cardiovascular biology that alter AD risk. The influence of gender on dementia development is complex in both the DS and euploid populations, and warrants more-extensive, longitudinal, population-based study.
Although increased levels of triglycerides and total body fat and low rates of exercise are reported in adults with DS27, and higher cholesterol levels have been associated with the risk of developing dementia in this group28, individuals with DS have lower rates of other cardiovascular risk factors — including hypertension, atherosclerosis and smoking29,30 — that are thought to contribute to the development of dementia in the general population31. Further studies are required to understand how trisomy alters the biology of the cardiovascular system and what impact this has on neurodegeneration in people who have DS.
The brain reserve hypothesis is based on the observation that, in the general population, individuals with higher levels of education and/or more-active social and intellectual lifestyles have a lower risk of developing dementia32. The hypothesis predicts that individuals with more-severe premorbid cognitive impairment will have an increased risk of developing dementia. However, no convincing relationship between severity of intellectual disability (or intelligence quotient (IQ) score) and risk of AD has been found in people with DS33, possibly because of diagnostic difficulties in those with severe impairments. Survival time for AD-DS does not differ much from that for late-onset AD (LOAD), with estimates varying between 3.5 years (s.d 2.2)34 and 6.24 years (s.d. 4.1)6. However, individuals with severe intellectual disability and dementia were found to have a longer survival time after diagnosis than those with milder intellectual disability6, further suggesting that reduced brain reserve does not accelerate disease progression in AD-DS.
Thus, people who have DS are at a greatly increased risk of developing dementia, with approximately 70% of individuals developing the condition by the age of 65. However, unlike the situation for LOAD, gender and cognitive reserve do not seem to influence AD-DS onset.
Clinical features of AD-DS
The early symptoms of AD-DS include features that are typical of other forms of AD, such as a decline in memory and language skills that may be present several years before dementia is diagnosed35–37. However, changes in personality and behaviour are more common in the early stages of AD-DS than they are in other forms of AD: individuals typically display either apathy, lack of motivation and stubbornness, or increasing behavioural excesses and impulsivity. These ‘non-cognitive’ changes (also referred to as behavioural and psychological symptoms of dementia (BPSDs))38–42 are associated with deficits in executive functioning and with the frontal atrophy that is visible on MRI scans, which may indicate frontal lobe dysfunction40,43. These changes may be related to pre-existing deficits in the integrity of the frontal tracts that have been observed in individuals with DS44 and that may be worsened by Aβ deposition in the frontal lobes45. Although BPSDs are very prominent in early AD-DS, this presentation is not unique to these individuals — it also occurs, albeit at lower rates, during the early stages of LOAD46 and EOAD47, particularly in cases arising from mutations in the AD risk gene presenilin 1 (PSEN1; which maps to chromosome 14). Further studies are required to determine the earliest changes associated with the development of dementia in people who have DS, and to delineate other clinical differences between AD-DS, LOAD and familial forms of EOAD, such as the frequencies of co-morbidities that may affect the onset and progression of dementia (for example, cardiovascular disease and systemic infections).
Another feature of AD-DS is the more-frequent and earlier appearance of neurological symptoms such as gait disturbance and seizures19 when compared with LOAD. Although heterogeneous, seizures associated with AD-DS often initially present with myoclonic jerks before progressing to tonic–clonic seizures and later to non-epileptic myoclonus with cerebellar signs; electroencephalograms show diffuse slowing and spike-and-wave patterns48–50. In individuals with LOAD, both complex partial and tonic–clonic seizures have been reported to be the predominant type51,52. Although seizures are reported to occur in 0.5–64% of people with LOAD51, more-recent population studies have suggested that seizure incidence in LOAD is relatively low, occurring in <5% of cases of the disease53. By contrast, most people with AD-DS eventually develop seizures, and a sudden onset of seizures in older adults with DS is highly suggestive of AD. Co-morbid seizures are associated with a more-aggressive course of AD-DS54 and a greater dementia-associated mortality rate6. The mechanism underlying this striking clinical feature of AD-DS is not understood, and the study of this may provide significant insights into neurodegeneration, in particular how changes in neuronal structure and organization affect disease progression.
Similarly to other forms of AD, the decline through middle-stage AD-DS dementia progressively affects more cognitive domains and results in symptoms such as dyspraxia, increasing incontinence and pathological grasping and sucking reflexes55,56, as well as symptoms of parkinsonism22. In summary, BPSDs may be an important early feature of AD-DS, and seizures are commonly associated with AD-DS. However, further comparative and mechanistic studies are required to unravel the importance of these clinical observations.
Neuropathological changes in AD-DS
The similarity between the neuropathological changes that occur in AD-DS and those that characterize AD in other individuals was first noted in 1929 (REF. 57) and was important for the widespread recognition of dementia in people who have DS. This discovery also had a key role in the identification of Aβ as the major constituent of amyloid plaques58, the identification of the first AD gene, APP59, and the subsequent development of the amyloid cascade hypothesis60.
The overall distribution and biochemical composition of plaques (largely composed of Aβ) and NFTs (largely composed of tau protein (encoded by microtubule-associated protein tau (MAPT))) in people who have DS, EOAD and LOAD are similar58,61–63. However, a greater deposition of plaques and tangles occurs in the hippocampus in AD-DS than in EOAD64 and, consistent with this, histological studies suggest that the earliest Aβ deposition in AD-DS occurs in the hippocampus65, whereas in LOAD the earliest deposition occurs in the basal cortex66. Furthermore, a lower density of Aβ plaques has been reported in the cortex in AD-DS than in LOAD64,67,68. These differences may relate to amyloid plaques in AD-DS having a more amorphous morphology and a larger average size than those present in LOAD69,70, resulting in a lower density caused by the presence of fewer but larger plaques. In addition, the aggregation kinetics of Aβ may differ in people with DS because of a higher concentration of the peptide resulting from their additional copy of APP. Alternatively, differences in plaque load may result from the neurodevelopmental differences that occur in people who have DS, resulting in changes in synaptic activity, which is known to regulate Aβ production71.
In AD-DS, intracellular accumulation of Aβ precedes extracellular plaque accumulation72–75 but becomes less prominent in older individuals with extensive pathology, as also observed in LOAD76. Additionally, in AD-DS, diffuse plaques composed of non-fibrillary deposits of Aβ develop before those with dense cores that are composed of amyloid (Supplementary information S2(table))34,64,65,73,74,77–88. Diffuse plaques are typically not associated with other forms of neuropathology, such as activated glial cells or synaptic loss, whereas dense-core plaques are often associated with dystrophic neurites and activated astroglia and microglia89. Also, Aβ42 — a form of Aβ that has a high tendency to aggregate — accumulates before deposition of Aβ40 in AD-DS73,74,80, which is consistent with the higher abundance of Aβ42 reported in plaques in other forms of AD89. Cerebral amyloid angiopathy (CAA) — the deposition of Aβ within cerebral blood vessels — is also observed in older individuals with DS74,80,87,90. However, unlike in LOAD, infarcts64 and vascular dementia seem to be rare in AD-DS91, although cases of CAA-associated cerebral haemorrhage have been described92–95.
In contrast to the findings of the histological studies described above, in vivo amyloid imaging by positron emission tomography (PET) indicates that the earliest site of Aβ accumulation in AD-DS, as in EOAD, could be the striatum96 and that enhanced deposition may occur in the frontal and parietal cortex97. This discrepancy may be because amyloid imaging recognizes only a subset of Aβ aggregates, thus not all deposition may be detected98. Nonetheless, most individuals with DS have amyloid-positive PET scans by the age of 50 (REFS 45,96,99,100). Amyloid load, as measured by PET, does not correlate well with cognitive function in adults who have DS in cross-sectional studies45,99, highlighting the importance of factors other than amyloid in the development of dementia. However, longitudinal imaging studies in this population have yet to be undertaken and may be highly informative45,99.
No NFTs have been reported in AD-DS in the absence of dense-core plaque pathology, which is consistent with the predictions of the amyloid cascade hypothesis. The density of NFTs triples between the fourth and fifth decade of life in AD-DS77, mirroring the onset of dementia, and NFT formation rather than amyloid deposition correlates best with cognitive decline34, which is consistent with similar findings in LOAD. Thus, changes in tau may result in neuronal dysfunction in both AD-DS and LOAD. Interestingly, smaller relative changes in nucleolar volume and a trend of reduced cell loss have been reported in the cortex and locus coeruleus in AD-DS compared with LOAD, despite comparable NFT loads, although similar cell loss was observed in other brain areas68. This may reflect a differential response of the trisomic CNS to accumulation of aggregated tau — suggesting, intriguingly, that chromosome 21 could encode a gene (or genes) that is neuroprotective when triplicated. Further study is required to determine whether trisomy 21 may provide protection from neurodegeneration.
As with people in the euploid population, people who have DS may have extensive amyloid deposition but no clinical signs of dementia (FIG. 1). Understanding how pathological changes due to AD relate to cognitive dysfunction is therefore a key research challenge. Identifying the processes that cause an amyloid-laden brain to convert from cognitively intact to impaired is crucial for understanding and successfully treating AD. As people who have DS develop amyloid deposition and NFTs by the age of 40, study of this group of individuals is likely to provide an important insight into the factors that cause dementia. Indeed, observations of AD-DS neuropathology already underpin our mechanistic understanding of AD, providing a detailed sequence of pathological changes and how these may relate to changes in cognition.
Pathological features other than plaques and NFTs also develop in both AD-DS and LOAD. Neuronal accumulation of ubiquitylated and aggregated transactive response DNA-binding protein 43 (TDP43; also known as TARDBP) in the cytoplasm and neurites is similar in AD-DS (7–14% of cases) and familial AD (10–14% of cases), whereas TDP43 neuropathology occurs more frequently in LOAD (29–79% of cases), perhaps because of the later disease onset101,102. Lewy bodies, particularly in the amygdala, occur at a similar frequency in AD-DS and LOAD103, but dementia with Lewy bodies (DLB), which is characterized by cognitive decline with hallucinations and parkinsonism features, is rare in DS104. Granulovacuolar degeneration, the formation of electron-dense granules in double-membrane-bound cytoplasmic vacuoles, associated with plaque and NFT pathology occurs at a similar frequency in AD-DS and AD64. How this pathology relates to the very early endosomal abnormalities that are reported to occur before birth in individuals with DS105 is unclear and warrants further investigation. Recent AD-related genome-wide association studies have highlighted the importance of the endosomal system in LOAD106, indicating that this system may be of particular significance to disease.
AD-DS versus Dup-APP-associated AD
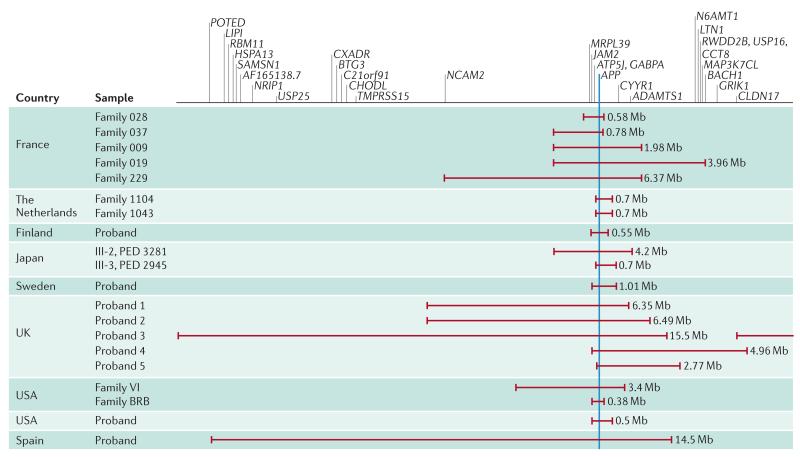
Dup-APP is a rare cause of familial EOAD, and comparison with AD-DS yields pathogenetic insights, as an additional copy of APP is present in both diseases. They therefore differ from other forms of familial AD that are the result of mutations in APP, PSEN1 or PSEN2 that modulate the processing of APP and the generation of Aβ. In Dup-APP, regions of chromosome 21 triplication vary in size8–15,47,107,108 (FIG. 2); the smallest known duplication contains only an additional copy of APP and no other coding genes8. By contrast, in AD-DS, triplication of any chromosome 21 gene in addition to APP may modulate the development of dementia. Studying these genes may therefore provide novel insights into AD mechanisms.
Figure 2. Regions of chromosome 21 duplicated in Dup-APP EOAD and ICH.
Schematic illustrating the genetic regions affected in reported cases of early-onset Alzheimer disease (EOAD) accompanied by duplication of amyloid precursor protein (Dup-APP)8–15,108. The minimal duplicated region is shown in blue: the only gene duplicated in all cases is APP. ADAMTS1, a disintegrin and metalloproteinase with thrombospondin motifs 1; ATP5J, ATP synthase-coupling factor 6; BACH1, BTB and CNC homologue 1; BTG3, BTG family member 3; C21orf91, chromosome 21 open reading frame 91; CCT8, chaperonin containing TCP1 8; CHODL, chondrolectin; CLDN17, claudin 17; CXADR, coxsackie virus and adenovirus receptor homologue; CYYR1, cysteine- and tyrosine-rich 1; GABPA, GA repeat-binding protein-alpha; GRIK1, glutamate receptor ionotropic, kainate 1; HSPA13, heat shock protein 70 kDa 13; ICH, intracerebral haemorrhage; JAM2, junction adhesion molecule 2; LIPI, lipase member I; LTN1, listerin E3 ubiquitin protein ligase 1; MAP3K7CL, MAP3K7 carboxy-terminal like; MRPL39, mitochondrial ribosomal protein L39; N6AMT1, N-6 adenine-specific DNA methyltransferase 1; NCAM2, neural cell adhesion molecule 2; NRIP1, nuclear receptor-interacting protein 1; PED, pedigree; POTED, POTE ankyrin domain family member D; RBM11, RNA-binding motif protein 11; RWDD2B, RWD domain-containing 2B; SAMSN1, SAM domain, SH3 domain and nuclear localization signals 1; TMPRSS15, transmembrane protease serine 15; USP, ubiquitin-specific peptidase.
The age of onset of dementia in individuals with Dup-APP ranges from 39 to 64 years (mean age ~52 years), and dementia shows virtually complete penetrance by the age of 65 years. By contrast, there is a broad variation in age of onset in AD-DS, and many individuals present with significant cognitive decline only after the age of 55 years, or even escape it altogether. This is remarkable given the usual co-morbid health issues and relative lack of brain reserve in individuals with DS. Thus, a possible protective mechanism (or mechanisms) from triplication of an unknown gene (or genes) on chromosome 21 may be important for resistance to dementia in people with DS. Moreover, intracerebral haemorrhage (ICH) is common in individuals with Dup-APP (occurring in 20–50% of cases)9–14,47,108, whereas individuals with DS are generally protected from this pathology, with only occasional reports. Thus, triplication of a chromosome 21 gene (or genes) may protect against some AD co-morbidity, and further comparative study of AD-DS and Dup-APP is required to understand the mechanisms underpinning this observation.
The few histopathological Dup-APP studies that have been carried out report diffuse atrophy with associated neuronal loss, deposition of plaques, CAA and accumulation of intraneuronal Aβ40 and NFTs11,109, and this pathology seems to be similar to AD-DS pathology (Supplementary information S3 (table)). However, further studies are needed75,109. Clinical DLB and cortical Lewy bodies have been observed in a few cases11,13,109, but currently there are insufficient data on these phenotypes to compare Dup-APP with AD-DS or LOAD. As in AD-DS, there is a greatly increased risk of dementia-associated seizures in Dup-APP10–13,47, in contrast to LOAD, in which seizures are relatively rare. This suggests that duplication of APP, and possibly of other genes located nearby, could be epileptogenic; however, as late-onset seizures often follow onset of dementia, they may also be related to synaptic deterioration that results in abnormal synchronization of neuronal networks and hyperexcitability110.
Genes and mechanisms in AD-DS
The presence of three copies of a dosage-sensitive gene (or genes) on chromosome 21 results in greatly enhanced risk of AD. Chromosome 21 carries 233 coding genes, 299 long non-coding genes (Ensembl release 78) and 29 microRNAs (miRBase release 21)111; thus, one or more of these must have a key role in AD. The phenotype resulting from a dosage-sensitive gene depends on the number of copies of the gene in the genome. However, not all genes are dosage sensitive, as homeostasis often prevents a gene from being overexpressed and the regulation of expression is often dependent on environmental context112. Furthermore, trisomy 21 causes widespread transcriptional dysregulation112,113, which may be the result of aneuploidy rather than of triplication of a specific gene. The importance of this in AD-DS remains unclear. Finally, acceleration of the epigenetic changes associated with ageing occurs in the DS brain114 — whether this alters gene expression or modulates the development of AD is an important area for future study.
The development of neuropathology and dementia varies significantly between individuals with DS, and understanding the factors (genetic or environmental) that cause this variation is likely to provide key insights into disease mechanisms. Below, we describe the genes that are currently implicated in the development of AD-DS and highlight the importance of further study of the genetics of AD-DS to understand how variation in the whole genome influences the development of disease.
Triplication of APP
The key dosage-sensitive gene for AD-DS is likely to be APP, as an additional normal copy of this gene is sufficient to cause EOAD in the absence of trisomy of the rest of chromosome 21 (REFS 8–15,47,107). The additional copy of APP in DS does not typically cause substantial Aβ accumulation until the second or third decade of life, although amyloid pathology has been demonstrated in a few childhood post-mortem examinations (BOX 1; FIG. 1). This lack of early Aβ accumulation may be due to APP not becoming dosage sensitive until adulthood, as suggested by both mouse and human studies115–117. However, increased levels of soluble Aβ42 are found in ~50% of trisomy 21 fetal brains118, suggesting that APP may be dosage sensitive during fetal development of individuals with DS but that this change may not be sufficient to cause extensive Aβ deposition in the developing brain — perhaps because of efficient clearance. Consistent with this, overexpression of APP and/or increased levels of Aβ have been reported in trisomy 21 human cell models, including in induced pluripotent stem cells (iPSCs) derived from infants or young adults with DS119–122. Although triplication of APP does not necessarily lead to enhanced expression of APP and subsequent increase in Aβ accumulation in all contexts, overexpression of APP is strongly linked to Aβ deposition in adult life. Thus, elucidating the factors that control the regulation of APP expression will considerably aid our understanding of AD.
Box 1. Identifying risk and protective factors for AD in young children.
It may seem counterintuitive to study infants and young children to understand a disease that presents only in adulthood. However, Alzheimer disease (AD) does not have an abrupt onset but emerges from a lengthy developmental trajectory in which precursors (for example, prodromal changes) surface well before overt dementia symptoms. Several genes involved in neurodevelopment have been suggested to have an important role in AD (including components of the WNT and reelin signalling pathway201,202). Additionally, cultures of cells derived from infants with Down syndrome (DS) show clear overexpression of amyloid precursor protein (APP)119–122, and amyloid-β (Aβ) plaques have been found in the brains of children with DS who are as young as 8 years of age65. Thus, the syndrome offers a longitudinal perspective on the multilevel effects of Aβ and tau pathology during development.
DS is diagnosed prenatally or at birth, and all infants with DS are at a significantly increased risk of subsequently developing AD, although not all will present with dementia, even as ageing adults. It is possible that in adults with DS, patterns of individual differences between those with AD and those without AD are already rooted in their individual differences when they are just infants, at the genetic, cellular, neural, cognitive, behavioural, sleep and/or environmental levels. The challenge is to identify individual differences in childhood that pinpoint risk and protective factors for subsequent AD outcome in adulthood. We can then identify biomarkers and devise early intervention strategies, initially for individuals with DS and subsequently for members of the euploid population, revolutionizing our understanding of the pathways that lead to AD. Thus, a developmental approach is essential, especially as it has already been shown that differences that can be observed in infancy in individuals with DS (for example, in the simple planning of saccadic eye movements) have cascading effects on cognitive outcomes in childhood and adulthood (for example, on numerical processing, language and face processing)203. Therefore, to fully comprehend AD in adults, it is crucial to study its full developmental trajectory, and understanding DS makes this possible.
Interaction of other chromosome 21 genes with APP
Several proteins encoded by other chromosome 21 genes have been suggested to modulate APP processing and Aβ generation (BOX 2; FIG. 3). For example, the transcription factor ETS2 is thought to transactivate the APP promoter, leading to overexpression123. The chromosome 21-encoded proteins small ubiquitin-related modifier 3 (SUMO3) and dual-specificity tyrosinephosphorylation-regulated kinase 1A (DYRK1A) modify APP post-translationally, which may alter Aβ generation124–126. Additionally, the chromosome 21 microRNA miR-155 has been suggested to modulate γ-secretase activity and hence the processing of APP, through its effect on the expression of sorting nexin 27 (REF. 127). Moreover, the β-secretase responsible for processing APP, β-site APP-cleaving enzyme 1 (BACE1), has a homologue, BACE2, encoded on chromosome 21, which may influence the onset of dementia in people with DS128. BACE2 does not have β-secretase activity, and in fact cleaves APP on the carboxy-terminal side of the β-secretase cut site within the Aβ region, preventing generation of the peptide. Thus, enhancing BACE2 expression may be protective against accumulation of Aβ129. However, BACE2 overexpression does not alter Aβ accumulation in a mouse model130, and the protein does not seem to have enhanced expression in the adult DS brain115,131. Whether triplication of any chromosome 21 gene alters APP biology sufficiently to modulate the development of AD remains to be determined.
Box 2. Modelling AD-DS in mice and in human iPSCs.
Amyloid precursor protein (APP) overexpression in mouse models causes dysfunction of basal forebrain cholinergic neurons and synaptic and behavioural changes141,204–206. However, increased expression of wild-type APP, even at levels in excess of those present in Down syndrome (DS), is insufficient to cause extensive Alzheimer disease (AD) neuropathology207. Only mice expressing mutant APP and/or other AD-associated genes recapitulate aspects of AD neuropathology and/ or cognitive change207. Similarly, although altered expression of many chromosome 21 genes modifies mouse models of familial AD, whether a single extra copy of these genes is sufficient to affect pathology and behaviour remains unclear. However, chromosome engineering, which enables the generation of mouse models with large genomic duplications, may help to elucidate the effects of trisomy on neurodegeneration208.
Reprogramming human somatic cells into induced pluripotent stem cells (iPSCs; which are in an embryonic stem cell-like state) is revolutionizing AD modelling, and advances in three-dimensional differentiation now permit the development of extensive amyloid-β (Aβ) and tau pathology in vitro. Comparisons have been made between euploid and trisomy 21 iPSCs derived from multiple sources, including different individuals (non-isogenic)122,209; isogenic lines generated in cell culture, spontaneously or by selection154,210; lines in which one of the three copies of chromosome 21 has been silenced211; monozygotic twins that were discordant for trisomy 21 (REF. 169); and non-integration-reprogrammed isogenic lines from an adult with mosaic DS (a condition in which only a percentage of an individual’s cells carry an extra copy of chromosome 21)121. Neurons derived from iPSCs show cellular phenotypes underpinning AD pathology, such as increased Aβ production, abnormal subcellular distribution of phosphorylated tau, mitochondrial abnormalities and accelerated cellular ageing121,122,154,212. DS iPSC models can be used to dissect the effect of trisomy of individual chromosome 21 genes (for example, by genome editing using clustered regularly interspaced short palindromic repeat–CRISPR-associated protein 9 (CRISPR–Cas9) technology), to develop high-throughput screening assays for phenotype-correcting compounds and to investigate cellular phenotypes in iPSCs generated from individuals with DS with very early versus very late ages of onset of dementia.
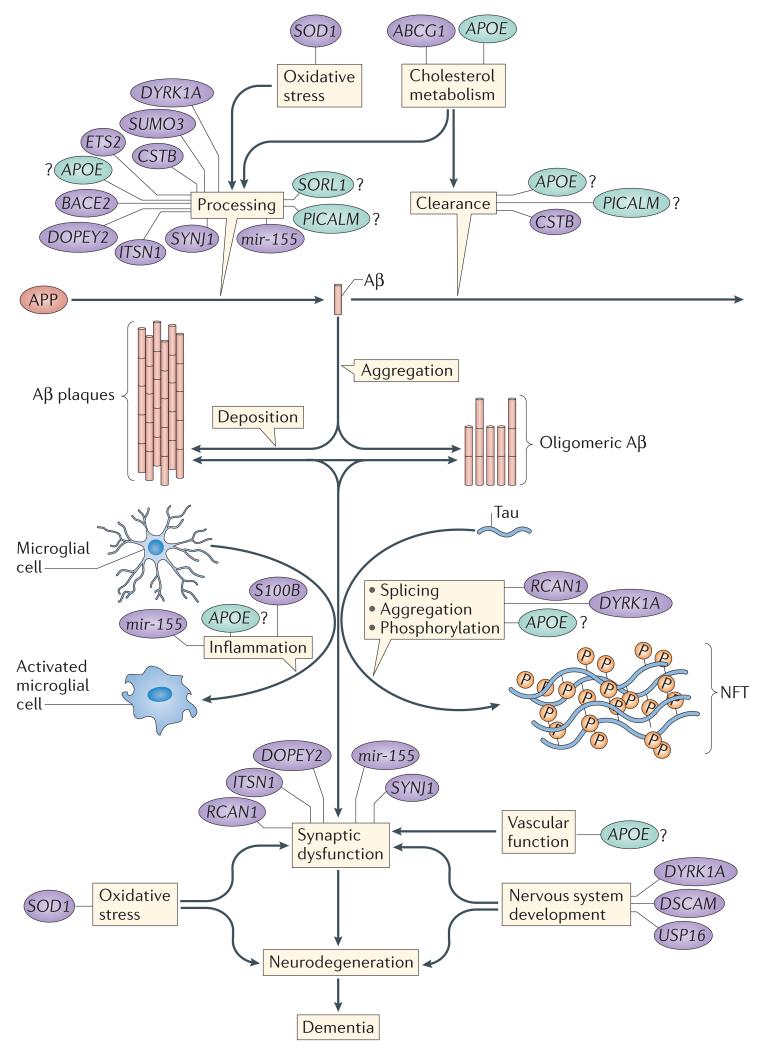
Figure 3. Schematic of suggested mechanisms that are important in AD-DS and their related genes.
Several genes may modulate processes that are relevant to the development of Alzheimer disease in people with Down syndrome (AD-DS); these include non-chromosome 21 genes, such as apolipoprotein E (APOE; which could alter disease by influencing cholesterol metabolism and possibly many other pathways), phosphatidylinositol-binding clathrin assembly protein (PICALM), sortilin-related receptor 1 (SORL1; which may influence disease via the endocytosis system and amyloid precursor protein (APP) processing) and microtubule-associated protein tau (MAPT). Tau aggregates to form neurofibrillary tangles (NFTs). Numerous chromosome 21 genes have also been suggested to influence the development of AD-DS, including genes that may influence APP processing and synaptic function via their role in the secretory–endosome system (including cystatin B (CSTB), DOPEY2, synaptojanin 1 (SYNJ1), intersectin 1 (ITSN1) and the microRNA gene mir-155), APP processing (including small ubiquitin-like modifier 3 (SUMO3), ETS2 and beta-site APP-cleaving enzyme 2 (BACE2)), cholesterol metabolism (including ATP-binding cassette G1 (ABCG1)), cellular signalling and tau phosphorylation (including dual-specificity tyrosine-phosphorylation-regulated kinase 1A (DYRK1A) and regulator of calcineurin 1 (RCAN1)), inflammation (including mir-155 and S100 calcium-binding protein beta (S100B)), synaptic function (including DOPEY2, SYNJ1, ITSN1, RCAN1 and mir-155), neurodevelopment (including ubiquitin-specific peptidase 16 (USP16), DYRK1A and DS cell adhesion molecule (DSCAM)) and oxidative stress (superoxide dismutase 1 (SOD1)). The relative importance of these processes to the development of dementia in AD-DS remains unclear and constitutes an area for future study. Chromosome 21 genes and gene products are shown in purple; non-chromosome 21 genes and gene products are shown in green. Aβ, amyloid-β.
Genes involved in LOAD
Polymorphisms in genes with important functions in LOAD have similar roles in the development of AD-DS; for example, the apolipoprotein E (APOE) ε4 allele is associated with greater Aβ deposition, as well as with earlier onset and increased risk of AD-DS, whereas the APOE ε2 allele leads to reduced Aβ deposition and a lower risk of disease132–138. Similarly, variants of phosphatidylinositol-binding clathrin assembly protein (PICALM) and sortilin-related receptor 1 (SORL1) influence age of onset in AD-DS, as they do in LOAD132,139,140, further supporting the theory that common mechanisms underlie both diseases. Whether variation in other genes with a role in LOAD is also important for AD-DS remains to be determined and is an important area for future study. Large-scale study of the genetic variants that contribute to the onset of dementia in AD-DS will provide an opportunity to gain insights into the mechanisms that underpin variation in the onset of dementia.
Disruption of secretory and endosomal systems
The earliest site of Aβ accumulation in AD-DS is within the neuron72–75, indicating that secretory and endosomal systems are central to Aβ generation. Moreover, an extra copy of APP is sufficient to cause endosomal enlargement and intracellular trafficking defects141,142 via an Aβ-independent mechanism143. Enlargement of endosomes in trisomic neurons may cause axonal trafficking defects that contribute to neuronal degeneration141.
Triplication of chromosome 21 genes other than APP may also affect the secretory–endosome system, thereby affecting synaptic function, Aβ production and Aβ clearance. Small segmental duplications of the chromosome 21 endosome-to-Golgi-trafficking gene DOPEY2 (REF. 144) have been associated with LOAD and mild cognitive impairment14,145, although this was not replicated in an independent study146. A reduction in the dose of the chromosome 21 gene cystatin B (CSTB), which encodes an endogenous inhibitor of lysosomal cathepsins, decreases the accumulation of Aβ and associated cognitive deficits147. Overexpression of another chromosome 21 gene, synaptojanin 1 (SYNJ1), which encodes a phosphoinositide phosphatase that regulates levels of membrane phosphatidylinositol-4,5-bisphosphate, has been associated with endosomal enlargement148, whereas reduced expression of SYNJ1 lowers Aβ accumulation, as well as neuronal dysfunction and cognitive deficits149,150. How endosomal enlargement caused by trisomy contributes to neuronal dysfunction and degeneration is another important area for future research.
Mitochondria and ROS
Mitochondrial dysfunction and enhanced production of reactive oxygen species (ROS) occur in people with DS and in trisomy 21 models151–154, and may contribute to the accelerated ageing reported in people who have DS155. Mitochondrial impairment may directly affect energy-hungry synapses, contributing to cognitive deficits156. Moreover, increased levels of ROS make trisomic neurons more prone to undergoing apoptosis, potentially making them more likely to degenerate151. Trisomy 21-associated increases in ROS levels may alter APP processing, promoting intracellular accumulation of Aβ119,151. Thus, protecting the trisomic brain from ROS may be of therapeutic value, although antioxidant supplementation has failed to show efficacy in preventing dementia in this population157. Interestingly, superoxide dismutase 1 (SOD1), which has a key role in processing ROS, lies on chromosome 21, and upregulation of SOD1 seems to protect against APP and Aβ neurotoxicity158, perhaps by modulating Aβ oligomerization159. Consistent with this, higher SOD1 enzymatic activity correlates with better memory in adults with DS160. However, increased SOD1 activity has also been suggested to cause accelerated cell senescence by increasing the levels of hydrogen peroxide, a form of ROS161.
Neuronal development and function
Several processes are likely to contribute to the intellectual disability associated with DS. These include a reduction in the numbers of neurons and dendritic spines, dendritic arborization, an alteration in the excitatory–inhibitory balance and a global impairment in network connectivity68,162–166. These perturbations in the structure, function and organization of the CNS may profoundly affect its degeneration in AD-DS (BOX 1). Triplication of several chromosome 21 genes contributes to changes in neurodevelopment and/or neuronal function. For example, ubiquitin-specific peptidase 16 (USP16) or DYRK1A upregulation alters stem cell fate167–169, which may in turn alter neuronal differentiation. Additionally, overexpression of several chromosome 21 genes (for example, the microRNA gene mir-155 and the protein-coding genes SYNJ1, regulator of calcineurin 1 (RCAN1), intersectin 1 (ITSN1) and DS cell adhesion molecule (DSCAM)) has been implicated in deficits in synaptic structure and function148,170,171. These genes may also play a part in AD-DS, perhaps via an effect on APP processing or on cognitive reserve. APP overexpression may also affect CNS function independently of the production and accumulation of Aβ, because the expression level of full-length APP influences neurogenesis, neuronal migration, axonal growth and the maintenance of the excitatory–inhibitory balance172,173. How the changes in CNS function caused by trisomy of chromosome 21 affect neurodegeneration in AD-DS is little understood and is a crucial area of future research.
Intracellular signalling and tau
Perturbations in intracellular signalling associated with trisomy 21 (REF. 174) may affect the response of the CNS to pathological changes. For example, overexpression of the chromosome 21 genes RCAN1 and DYRK1A promotes aberrant phosphorylation of tau152,175–177. DYRK1A is dosage sensitive in the adult brain178, and overexpression of this gene modulates tau splicing, altering the relative abundance of tau with three or four microtubule-binding domains (3-repeat (3R) and 4R tau, respectively), which may affect the formation of NFTs179,180. Consistent with this, an increase in the ratio of 3R/4R tau has been reported to occur in AD-DS, as compared with LOAD or age-matched euploid individuals without dementia179,180. Additionally, an increase in the total amount of tau has been reported in the cortex in AD-DS as compared with that in age-matched euploid individuals without dementia, and in DS iPSC-derived neurons122,179; this upregulation may be the result of increased APP levels181. DYRK1A also downregulates the levels of neural restrictive silencing factor (NRSF; also known as REST), a neuroprotective protein168,169, which has reduced expression in people with AD182. Variants in DYRK1A have been associated with risk of LOAD183, further indicating a possible role in disease pathogenesis, although this association was not replicated in an independent study184.
Cholesterol metabolism
Alterations in cholesterol metabolism may contribute to the development of dementia31. Total cholesterol levels have been suggested to predict the onset of dementia in people with DS, particularly in those individuals who have an APOE ε4 allele28. Clinical trials are therefore underway to determine whether statins can prevent decline in older adults with DS, which may provide both clinical and mechanistic insights185. The chromosome 21 lipid transporter ATP-binding cassette G1 (ABCG1) has been suggested to regulate cholesterol efflux and may alter cholesterol metabolism in people with DS186. Whether trisomy of this gene is related to the development of AD-DS remains unclear, as ABCG1 overexpression has been reported both to increase and to decrease Aβ generation in vitro187,188 and does not change Aβ accumulation in vivo189, suggesting that this gene may not be associated with the development of AD-DS. Further study is required to understand the mechanisms that underlie the link between increased cholesterol levels and the onset of dementia in individuals with DS.
Immune system dysfunction
Growing evidence shows that the immune system plays an important part in the development of AD106,190. Individuals with DS are at an increased risk of immune system dysfunction: these individuals have a higher incidence of both autoimmune and infectious disease191, and show upregulation of pro-inflammatory makers, including interleukin-1, in the brain192,193. This dysregulation may contribute to AD-DS through alterations in micro-glial activation190. Microglia in AD-DS have been reported to be associated with both mature Aβ plaques194 and NFTs195, although the contribution of the immune response to AD-DS has yet to be fully explored. The chromosome 21 gene S100 calcium-binding protein beta (S100B) is expressed in astrocytes and is upregulated in both AD196 and AD-DS192, and it may contribute to neurodegeneration by promoting Aβ deposition197 and tau phosphorylation198 and by creating a neurotoxic environment through the release of extracellular signals199.
Translational research
The life expectancy of people with DS is increasing because of better health care and improved social inclusion. However, as with the euploid population, ageing brings new issues; in people with DS, a major ageing-related issue is a vastly increased risk of EOAD. People who have DS develop amyloid plaques and NFTs by the age of 40 years, and many individuals subsequently go on to develop dementia. Despite genetic and Aβ differences between the various forms of EOAD and LOAD, many similarities in disease process are observed such that AD seems to converge on common mechanisms of pathology. Thus, in the AD-DS patient population, it is feasible both to determine the factors (genetic and/or environmental) that cause conversion from pathological disease to cognitive decline and to undertake intervention trials to halt the development of dementia.
As APP gene dosage is the major determinant of AD-DS, it follows that therapies aimed at reducing Aβ (such as BACE inhibition or Aβ immunization) might have a beneficial effect in the DS population. Such approaches are being trialled for people with familial AD arising from APP or PSEN1 mutations200, and similar clinical trials in AD-DS could provide valuable additional insight, given the predictable conversion to AD neuropathology and subsequent dementia in this population. Other treatment options that require further development include DYRK1A inhibitors and ROS modulators. Notably, treatment safety is of particular importance because many individuals with DS are unable to consent to their own participation in clinical trials and because they will probably need to undergo treatment for many years.
Conclusions
Many questions remain to be answered in AD-DS, including, most importantly, the mechanisms underlying the later onset of dementia as compared with Dup-APP, how neurodevelopmental perturbations affect neurodegeneration and the identity of any chromosome 21 gene (or genes) that may protect against dementia. We now have a remarkable set of tools for studying AD-DS, ranging from new model systems to genomics studies. Although there are undoubtedly specific problems in both analysing and treating people who have DS for AD, such as issues of informed consent, trisomy 21 is an extremely important disorder for learning about the development of neurodegeneration and for testing potential therapeutic strategies to the benefit of everyone at risk of AD.
Supplementary Material
Acknowledgements
The authors are funded by a Wellcome Trust Strategic Award (grant number: 098330/Z/12/Z) awarded to The London Down Syndrome (LonDownS) Consortium (J.H., A.K-S., D.N., V.L.J.T. E.M.C.F. and A.S.) and the Medical Research Council (programme number U117527252; awarded to V.L.J.T.), as well as by awards from Alzheimer Research UK (awarded to F.K.W and E.M.C.F), Alzheimer Society (awarded to E.M.C.F and F.K.W.), Bailey Thomas Trust (awarded to A.S.), Epilepsy Research UK (awarded to F.K.W.), Lee Kong Chian School of Medicine, Nanyang Technological University Start-up Grant, and Singapore Ministry of Education Academic Research Fund Tier 1(2014-T1-001-173; awarded to D.N).
Glossary
- Dyspraxia
Disrupted fine or gross motor coordination.
- Early-onset Alzheimer disease
(EOAD). Occurrence of Alzheimer disease before the age of 65 years.
- Euploid
Having a normal chromosome number (46 chromosomes in 23 pairs in humans).
- Executive functioning
Mental processing skills involving the frontal cortex; used for planning, attention focusing, working memory, mental flexibility and self-control.
- Incidence
The rate of new occurrences of a disorder within a specified period of time.
- Lewy bodies
Protein aggregates typically containing α-synuclein.
- Myoclonic jerks
Brief involuntary muscle twitches that are a medical sign of various neurological disorders.
- Parkinsonism
A clinical syndrome including bradykinesia (slow movements), muscle rigidity and tremor, often due to the neurodegenerative condition Parkinson disease but also associated with other neurological conditions, toxins and medications.
- Prevalence
The number of cases of a disorder at one time within a population.
- Tonic–clonic seizures
A common type of epileptic seizure with a tonic phase (stiffening of muscles and loss of consciousness) followed by a clonic phase (rapid, rhythmic jerking of arms and legs).
Footnotes
Competing interests statement
The authors declare competing interests: see Web version for details.
FURTHER INFORMATION
London Down Syndrome (LonDownS) Consortium: http://www.ucl.ac.uk/london-down-syndrome-consortium
See online article: S1 (table) ∣ S2 (table) ∣ S3 (table)
Contributor Information
Frances K. Wiseman, Department of Neurodegenerative Disease, Institute of Neurology, University College London, Queen Square, London WC1N 3BG, UK..
Tamara Al-Janabi, Division of Psychiatry, University College London, Maple House, 149 Tottenham Court Road, London W1T 7NF, UK..
John Hardy, Department of Molecular Neuroscience, Institute of Neurology, University College London, Queen Square, London WC1N 3BG, UK..
Annette Karmiloff-Smith, Centre for Brain and Cognitive Development, Birkbeck, University of London, Malet Street, London WC1E 7HX, UK..
Dean Nizetic, Lee Kong Chian School of Medicine, Nanyang Technological University, Novena Campus, 11 Mandalay Road, Singapore 308232; Blizard Institute, Barts and the London School of Medicine, Queen Mary University of London, 4 Newark Street, London E1 2AT, UK..
Victor L. J. Tybulewicz, Francis Crick Institute, Mill Hill Laboratory, London NW7 1AA, UK..
Elizabeth M. C. Fisher, Department of Neurodegenerative Disease, Institute of Neurology, University College London, Queen Square, London WC1N 3BG, UK..
André Strydom, Division of Psychiatry, University College London, Maple House, 149 Tottenham Court Road, London W1T 7NF, UK..
References
- 1.Lejeune J, Gautier M, Turpin R. Etude des chromosomes somatiques de neuf enfants mongoliens. C. R. Hebd. Seances Acad. Sci. 1959;248:1721–1722. (in French) [PubMed] [Google Scholar]
- 2.de Graaf G, Buckley F, Skotko BG. Estimates of the live births, natural losses, and elective terminations with Down syndrome in the United States. Am. J. Med. Genet. A. 2015;167A:756–767. doi: 10.1002/ajmg.a.37001. [DOI] [PubMed] [Google Scholar]
- 3.Wu JH, Morris JK. Trends in maternal age distribution and the live birth prevalence of Down’s syndrome in England and Wales: 1938–2010. Eur. J. Hum. Genet. 2013;21:943–947. doi: 10.1038/ejhg.2012.288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wu JH, Morris JK. The population prevalence of Down’s syndrome in England and Wales in 2011. Eur. J. Hum. Genet. 2013;21:1016–1019. doi: 10.1038/ejhg.2012.294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wiseman FK, Alford KA, Tybulewicz VLJ, Fisher EMC. Down syndrome — recent progress and future prospects. Hum. Mol. Genet. 2009;18:R75–R83. doi: 10.1093/hmg/ddp010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.McCarron M, McCallion P, Reilly E, Mulryan N. A prospective 14-year longitudinal follow-up of dementia in persons with Down syndrome. J. Intellect. Disabil. Res. 2014;58:61–70. doi: 10.1111/jir.12074. [DOI] [PubMed] [Google Scholar]
- 7.Zigman WB, Schupf N, Urv T, Zigman A, Silverman W. Incidence and temporal patterns of adaptive behavior change in adults with mental retardation. Am. J. Mental Retard. 2002;107:161–174. doi: 10.1352/0895-8017(2002)107<0161:IATPOA>2.0.CO;2. [DOI] [PubMed] [Google Scholar]
- 8.Hooli BV, et al. Role of common and rare APP DNA sequence variants in Alzheimer disease. Neurology. 2012;78:1250–1257. doi: 10.1212/WNL.0b013e3182515972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kasuga K, et al. Identification of independent APP locus duplication in Japanese patients with early-onset Alzheimer disease. J. Neurol. Neurosurg. Psychiatry. 2009;80:1050–1052. doi: 10.1136/jnnp.2008.161703. [DOI] [PubMed] [Google Scholar]
- 10.McNaughton D, et al. Duplication of amyloid precursor protein (APP), but not prion protein (PRNP) gene is a significant cause of early onset dementia in a large UK series. Neurobiol. Aging. 2010;33:426.e13–426.e21. doi: 10.1016/j.neurobiolaging.2010.10.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Rovelet-Lecrux A, et al. APP locus duplication causes autosomal dominant early-onset Alzheimer disease with cerebral amyloid angiopathy. Nat. Genet. 2006;38:24–26. doi: 10.1038/ng1718. [DOI] [PubMed] [Google Scholar]
- 12.Rovelet-Lecrux A, et al. APP locus duplication in a Finnish family with dementia and intracerebral haemorrhage. J. Neurol. Neurosurg. Psychiatry. 2007;78:1158–1159. doi: 10.1136/jnnp.2006.113514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sleegers K, et al. APP duplication is sufficient to cause early onset Alzheimer’s dementia with cerebral amyloid angiopathy. Brain. 2006;129:2977–2983. doi: 10.1093/brain/awl203. [DOI] [PubMed] [Google Scholar]
- 14.Swaminathan S, et al. Analysis of copy number variation in Alzheimer’s disease in a cohort of clinically characterized and neuropathologically verified individuals. PLoS ONE. 2012;7:e50640. doi: 10.1371/journal.pone.0050640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Thonberg H, et al. Mutation screening of patients with Alzheimer disease identifies APP locus duplication in a Swedish patient. BMC Res. Notes. 2011;4:476. doi: 10.1186/1756-0500-4-476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Prasher VP, et al. Molecular mapping of Alzheimer-type dementia in Down’s syndrome. Ann. Neurol. 1998;43:380–383. doi: 10.1002/ana.410430316. [DOI] [PubMed] [Google Scholar]
- 17.Korbel JO, et al. The genetic architecture of Down syndrome phenotypes revealed by high-resolution analysis of human segmental trisomies. Proc. Natl Acad. Sci. USA. 2009;106:12031–12036. doi: 10.1073/pnas.0813248106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Fraser J, Mitchell A. Kalmuc idiocy: report of a case with autopsy with notes on 62 cases. J. Mental Sci. 1876;22:161–169. [Google Scholar]
- 19.Strydom A, et al. Dementia in older adults with intellectual disabilities — epidemiology, presentation, and diagnosis. J. Intellect. Disabil. 2010;7:96–110. [Google Scholar]
- 20.Tyrrell J, et al. Dementia in people with Down’s syndrome. Int. J. Geriatr. Psychiatry. 2001;16:1168–1174. doi: 10.1002/gps.502. [DOI] [PubMed] [Google Scholar]
- 21.Coppus A, et al. Dementia and mortality in persons with Down’s syndrome. J. Intellect. Disabil. Res. 2006;50:768–777. doi: 10.1111/j.1365-2788.2006.00842.x. [DOI] [PubMed] [Google Scholar]
- 22.Visser FE, Aldenkamp AP, vanHuffelen AC, Kuilman M, Overweg J. Prospective study of the prevalence of Alzheimer-type dementia in institutionalized individuals with Down syndrome. Am. J. Mental Retard. 1997;101:400–412. [PubMed] [Google Scholar]
- 23.Krinsky-McHale SJ, et al. Successful aging in a 70-year-old man with Down syndrome: a case study. Intellect. Dev. Disabil. 2008;46:215–228. doi: 10.1352/2008.46:215-228. [DOI] [PubMed] [Google Scholar]
- 24.Coppus AMW, et al. Early age at menopause is associated with increased risk of Dementia and mortality in women with Down syndrome. J. Alzheimers Dis. 2010;19:545–550. doi: 10.3233/JAD-2010-1247. [DOI] [PubMed] [Google Scholar]
- 25.Schupf N, et al. Onset of dementia is associated with age at menopause in women with Down’s syndrome. Ann. Neurol. 2003;54:433–438. doi: 10.1002/ana.10677. [DOI] [PubMed] [Google Scholar]
- 26.Cosgrave MP, Tyrrell J, McCarron M, Gill M, Lawlor BA. Age at onset of dementia and age of menopause in women with Down’s syndrome. J. Intellect. Disabil. Res. 1999;43:461–465. doi: 10.1046/j.1365-2788.1999.00192.x. [DOI] [PubMed] [Google Scholar]
- 27.Draheim CC, Geijer JR, Dengel DR. Comparison of intima-media thickness of the carotid artery and cardiovascular disease risk factors in adults with versus without the Down syndrome. Am. J. Cardiol. 2010;106:1512–1516. doi: 10.1016/j.amjcard.2010.06.079. [DOI] [PubMed] [Google Scholar]
- 28.Zigman WB, et al. Cholesterol level, statin use and Alzheimer’s disease in adults with Down syndrome. Neurosci. Lett. 2007;416:279–284. doi: 10.1016/j.neulet.2007.02.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Ylaherttuala S, Luoma J, Nikkari T, Kivimaki T. Downs-syndrome and atherosclerosis. Atherosclerosis. 1989;76:269–272. doi: 10.1016/0021-9150(89)90110-x. [DOI] [PubMed] [Google Scholar]
- 30.van de Louw J, Vorstenbosch R, Vinck L, Penning C, Evenhuis H. Prevalence of hypertension in adults with intellectual disability in the Netherlands. J. Intellect. Disabil. Res. 2009;53:78–84. doi: 10.1111/j.1365-2788.2008.01130.x. [DOI] [PubMed] [Google Scholar]
- 31.Stampfer MJ. Cardiovascular disease and Alzheimer’s disease: common links. J. Internal Med. 2006;260:211–223. doi: 10.1111/j.1365-2796.2006.01687.x. [DOI] [PubMed] [Google Scholar]
- 32.Stern Y. Cognitive reserve in ageing and Alzheimer’s disease. Lancet Neurol. 2012;11:1006–1012. doi: 10.1016/S1474-4422(12)70191-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Zigman WB, et al. Alzheimer’s disease in adults with Down syndrome. Int. Rev. Res. Ment. Retard. 2008;36:103–145. doi: 10.1016/S0074-7750(08)00004-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Margallo-Lana ML, et al. Fifteen-year follow-up of 92 hospitalized adults with Down’s syndrome: incidence of cognitive decline, its relationship to age and neuropathology. J. Intellect. Disabil. Res. 2007;51:463–477. doi: 10.1111/j.1365-2788.2006.00902.x. [DOI] [PubMed] [Google Scholar]
- 35.Devenny DA, Krinsky-McHale SJ, Sersen G, Silverman WP. Sequence of cognitive decline in dementia in adults with Down’s syndrome. J. Intellect. Disabil. Res. 2000;44:654–665. doi: 10.1046/j.1365-2788.2000.00305.x. [DOI] [PubMed] [Google Scholar]
- 36.Devenny DA, Zimmerli EJ, Kittler P, Krinsky-McHale SJ. Cued recall in early-stage dementia in adults with Down’s syndrome. J. Intellect. Disabil. Res. 2002;46:472–483. doi: 10.1046/j.1365-2788.2002.00417.x. [DOI] [PubMed] [Google Scholar]
- 37.Krinsky-McHale SJ, Devenny DA, Silverman WP. Changes in explicit memory associated with early dementia in adults with Down’s syndrome. J. Intellect. Disabil. Res. 2002;46:198–208. doi: 10.1046/j.1365-2788.2002.00365.x. [DOI] [PubMed] [Google Scholar]
- 38.Adams D, Oliver C. The relationship between acquired impairments of executive function and behaviour change in adults with Down syndrome. J. Intellect. Disabil. Res. 2010;54:393–405. doi: 10.1111/j.1365-2788.2010.01271.x. [DOI] [PubMed] [Google Scholar]
- 39.Ball SL, et al. Personality and behaviour changes mark the early stages of Alzheimer’s disease in adults with Down’s syndrome: findings from a prospective population-based study. Int. J. Geriatr. Psychiatry. 2006;21:661–673. doi: 10.1002/gps.1545. [DOI] [PubMed] [Google Scholar]
- 40.Ball SL, Holland AJ, Treppner P, Watson PC, Huppert FA. Executive dysfunction and its association with personality and behaviour changes in the development of Alzheimer’s disease in adults with Down syndrome and mild to moderate learning disabilities. Br. J. Clin. Psychol. 2008;47:1–29. doi: 10.1348/014466507X230967. [DOI] [PubMed] [Google Scholar]
- 41.Holland AJ, Hon J, Huppert FA, Stevens F. Incidence and course of dementia in people with Down’s syndrome: findings from a population-based study. J. Intellect. Disabil. Res. 2000;44:138–146. doi: 10.1046/j.1365-2788.2000.00263.x. [DOI] [PubMed] [Google Scholar]
- 42.Oliver C, Kalsy S, McQuillan S, Hall S. Behavioural excesses and deficits associated with dementia in adults who have Down syndrome. J. Appl. Res. Intellect. Disabil. 2011;24:208–216. [Google Scholar]
- 43.Nelson LD, Orme D, Osann K, Lott IT. Neurological changes and emotional functioning in adults with Down Syndrome. J. Intellect. Disabil. Res. 2001;45:450–456. doi: 10.1046/j.1365-2788.2001.00379.x. [DOI] [PubMed] [Google Scholar]
- 44.Powell D, et al. Frontal white matter integrity in adults with Down syndrome with and without dementia. Neurobiol. Aging. 2014;35:1562–1569. doi: 10.1016/j.neurobiolaging.2014.01.137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Jennings D, et al. Age dependence of brain ß-amyloid deposition in Down syndrome: an [18F]florbetaben PET study. Neurology. 2015;84:500–507. doi: 10.1212/WNL.0000000000001212. [DOI] [PubMed] [Google Scholar]
- 46.Masters MC, Morris JC, Roe CM. ‘Noncognitive’ symptoms of early Alzheimer disease: a longitudinal analysis. Neurology. 2015;84:617–622. doi: 10.1212/WNL.0000000000001238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Wallon D, et al. The French series of autosomal dominant early onset Alzheimer’s disease cases: mutation spectrum and cerebrospinal fluid biomarkers. J. Alzheimers Dis. 2012;30:847–856. doi: 10.3233/JAD-2012-120172. [DOI] [PubMed] [Google Scholar]
- 48.De Simone R, Puig XS, Gélisse P, Crespel A, Genton P. Senile myoclonic epilepsy: delineation of a common condition associated with Alzheimer’s disease in Down syndrome. Seizure. 2010;19:383–389. doi: 10.1016/j.seizure.2010.04.008. [DOI] [PubMed] [Google Scholar]
- 49.Moller JC, Hamer HM, Oertel WH, Rosenow F. Late-onset myoclonic epilepsy in Down’s syndrome (LOMEDS) Seizure. 2001;10:303–305. [PubMed] [Google Scholar]
- 50.d’Orsi G, Specchio LM. Progressive myoclonus epilepsy in Down syndrome patients with dementia. J. Neurol. 2014;261:1584–1597. doi: 10.1007/s00415-014-7376-x. [DOI] [PubMed] [Google Scholar]
- 51.Friedman D, Honig LS, Scarmeas N. Seizures and epilepsy in Alzheimer’s disease. CNS Neurosci. Ther. 2012;18:285–294. doi: 10.1111/j.1755-5949.2011.00251.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Vossel KA, et al. Seizures and epileptiform activity in the early stages of Alzheimer disease. JAMA Neurol. 2013;70:1158–1166. doi: 10.1001/jamaneurol.2013.136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Irizarry MC, et al. Incidence of new-onset seizures in mild to moderate Alzheimer disease. Arch. Neurol. 2012;69:368–372. doi: 10.1001/archneurol.2011.830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Lott IT, et al. Down syndrome and dementia: seizures and cognitive decline. J. Alzheimers Dis. 2012;29:177–185. doi: 10.3233/JAD-2012-111613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Crayton L, Oliver C, Holland A, Bradbury J, Hall S. The neuropsychological assessment of age related cognitive deficits in adults with Down’s syndrome. J. Appl. Res. Intellect. Disabil. 1998;11:255–272. [Google Scholar]
- 56.Dalton AJ, Mehta PD, Fedor BL, Patti PJ. Cognitive changes in memory precede those in praxis in aging persons with Down syndrome. J. Intellect. Dev. Disabil. 1999;24:169–187. [Google Scholar]
- 57.Struwe F. Histopathologische Untersuchungen über Entstehung und Wesen der senile Plaques. Z. Gesamte Neurol. Psy. 1929;122:291–307. (in German) [Google Scholar]
- 58.Glenner GG, Wong CW. Alzheimer’s disease and Downs syndrome — sharing of a unique cerebrovascular amyloid fibril protein. Biochem. Biophys. Res. Commun. 1984;122:1131–1135. doi: 10.1016/0006-291x(84)91209-9. [DOI] [PubMed] [Google Scholar]
- 59.Goate A, et al. Segregation of a missense mutation in the amyloid precursor protein gene with familial Alzheimer’s disease. Nature. 1991;349:704–706. doi: 10.1038/349704a0. [DOI] [PubMed] [Google Scholar]
- 60.Hardy JA, Higgins GA. Alzheimer’s disease — the amyloid cascade hypothesis. Science. 1992;256:184–185. doi: 10.1126/science.1566067. [DOI] [PubMed] [Google Scholar]
- 61.Goedert M, Spillantini MG, Cairns NJ, Crowther RA. Tau proteins of Alzheimer paired helical filaments — abnormal phosphorylation of all 6 brain isoforms. Neuron. 1992;8:159–168. doi: 10.1016/0896-6273(92)90117-v. [DOI] [PubMed] [Google Scholar]
- 62.Mann DMA. Alzheimer’s disease and Down’s syndrome. Histopathology. 1988;13:125–137. doi: 10.1111/j.1365-2559.1988.tb02018.x. [DOI] [PubMed] [Google Scholar]
- 63.Wisniewski HM, Rabe A. Discrepancy between Alzheimer-type neuropathology and dementia in persons with Down’s syndrome. Ann. NY Acad. Sci. 1986;477:247–260. doi: 10.1111/j.1749-6632.1986.tb40344.x. [DOI] [PubMed] [Google Scholar]
- 64.Mann DMA. The pathological association between Down syndrome and Alzheimer’s disease. Mech. Ageing Dev. 1988;43:99–136. doi: 10.1016/0047-6374(88)90041-3. [DOI] [PubMed] [Google Scholar]
- 65.Leverenz JB, Raskind MA. Early amyloid deposition in the medial temporal lobe of young Down syndrome patients: a regional quantitative analysis. Exp. Neurol. 1998;150:296–304. doi: 10.1006/exnr.1997.6777. [DOI] [PubMed] [Google Scholar]
- 66.Braak H, Braak E. Neuropathological staging of Alzheimer-related changes. Acta Neuropathol. 1991;82:239–259. doi: 10.1007/BF00308809. [DOI] [PubMed] [Google Scholar]
- 67.Egensperger R, et al. Reverse relationship between β-amyloid precursor protein and beta-amyloid peptide plaques in Down’s syndrome versus sporadic/familial Alzheimer’s disease. Acta Neuropathol. 1999;97:113–118. doi: 10.1007/s004010050963. [DOI] [PubMed] [Google Scholar]
- 68.Mann DMA, Yates PO, Marcyniuk B, Ravindra CR. Loss of neurons from cortical and subcortical areas in Down’s syndrome patients at middle-age — quantitative comparisons with younger Down’s patients and patients with Alzheimer’s disease. J. Neurol. Sci. 1987;80:79–89. doi: 10.1016/0022-510x(87)90223-1. [DOI] [PubMed] [Google Scholar]
- 69.Allsop D, Kidd M, Landon M, Tomlinson A. Isolated senile plaque cores in Alzheimer’s disease and Down’s syndrome show differences in morphology. J. Neurol. Neurosurg. Psychiatry. 1986;49:886–892. doi: 10.1136/jnnp.49.8.886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Armstrong RA. Size frequency distributions of β-amyloid (4β) deposits: a comparative study of four neurodegenerative disorders. Folia Neuropathol. 2012;50:240–249. doi: 10.5114/fn.2012.30524. [DOI] [PubMed] [Google Scholar]
- 71.Bero AW, et al. Neuronal activity regulates the regional vulnerability to amyloid-β deposition. Nat. Neurosci. 2011;14:750–756. doi: 10.1038/nn.2801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Gyure KA, Durham R, Stewart WF, Smialek JE, Troncoso JC. Intraneuronal Aβ-amyloid precedes development of amyloid plaques in Down syndrome. Arch. Pathol. Lab. Med. 2001;125:489–492. doi: 10.5858/2001-125-0489-IAAPDO. [DOI] [PubMed] [Google Scholar]
- 73.Hirayama A, Horikoshi Y, Maeda M, Ito M, Takashima S. Characteristic developmental expression of amyloid β40, 42 and 43 in patients with Down syndrome. Brain Dev. 2003;25:180–185. doi: 10.1016/s0387-7604(02)00209-7. [DOI] [PubMed] [Google Scholar]
- 74.Iwatsubo T, Mann DMA, Odaka A, Suzuki N, Ihara Y. Amyloid β protein (Aβ) deposition: Aβ42(43) precedes Aβ40 in Down syndrome. Ann. Neurol. 1995;37:294–299. doi: 10.1002/ana.410370305. [DOI] [PubMed] [Google Scholar]
- 75.Mori C, et al. Intraneuronal Aβ42 accumulation in Down syndrome brain. Amyloid. 2002;9:88–102. [PubMed] [Google Scholar]
- 76.Wegiel J, et al. Intraneuronal Aβ immunoreactivity is not a predictor of brain amyloidosis-β or neurofibrillary degeneration. Acta Neuropathol. 2007;113:389–402. doi: 10.1007/s00401-006-0191-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Wisniewski KE, Wisniewski HM, Wen GY. Occurrence of neuropathological changes and dementia of Alzheimer’s disease in Down’s syndrome. Ann. Neurol. 1985;17:278–282. doi: 10.1002/ana.410170310. [DOI] [PubMed] [Google Scholar]
- 78.Wisniewski KE, Dalton AJ, Mclachlan DRC, Wen GY, Wisniewski HM. Alzheimer’s disease in Down’s syndrome: clinicopathologic studies. Neurology. 1985;35:957–961. doi: 10.1212/wnl.35.7.957. [DOI] [PubMed] [Google Scholar]
- 79.Burger PC, Vogel FS. The development of pathologic changes of Alzheimer’s disease and senile dementia in patients with Down’s syndrome. Am. J. Pathol. 1973;73:457–476. [PMC free article] [PubMed] [Google Scholar]
- 80.Lemere CA, et al. Sequence of deposition of heterogeneous amyloid β-peptides and APO E in Down syndrome: implications for initial events in amyloid plaque formation. Neurobiol. Dis. 1996;3:16–32. doi: 10.1006/nbdi.1996.0003. [DOI] [PubMed] [Google Scholar]
- 81.Ball MJ, Nuttall K. Neurofibrillary tangles, granulovacuolar degeneration, and neuron loss in Down syndrome: quantitative comparison with Alzheimer dementia. Ann. Neurol. 1980;7:462–465. doi: 10.1002/ana.410070512. [DOI] [PubMed] [Google Scholar]
- 82.Mann DMA, Esiri MM. The pattern of acquisition of plaques and tangles in the brains of patients under 50 years of age with Down’s syndrome. J. Neurol. Sci. 1989;89:169–179. doi: 10.1016/0022-510x(89)90019-1. [DOI] [PubMed] [Google Scholar]
- 83.Whalley LJ. The dementia of Down’s syndrome and its relevance to etiological studies of Alzheimer’s disease. Ann. NY Acad. Sci. 1982;396:39–53. doi: 10.1111/j.1749-6632.1982.tb26842.x. [DOI] [PubMed] [Google Scholar]
- 84.Ropper AH, Williams RS. Relationship between plaques, tangles, and dementia in Down syndrome. Neurology. 1980;30:639–644. doi: 10.1212/wnl.30.6.639. [DOI] [PubMed] [Google Scholar]
- 85.Godridge H, Reynolds GP, Czudek C, Calcutt NA, Benton M. Alzheimer-like neurotransmitter deficits in adult Down’s syndrome brain tissue. J. Neurol. Neurosurg. Psychiatry. 1987;50:775–778. doi: 10.1136/jnnp.50.6.775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Murphy GM, et al. Antigenic profile of plaques and neurofibrillary tangles in the amygdala in Down’s syndrome: a comparison with Alzheimer’s disease. Brain Res. 1990;537:102–108. doi: 10.1016/0006-8993(90)90345-c. [DOI] [PubMed] [Google Scholar]
- 87.Motte J, Williams RS. Age-related-changes in the density and morphology of plaques and neurofibrillary tangles in Down syndrome brain. Acta Neuropathol. 1989;77:535–546. doi: 10.1007/BF00687256. [DOI] [PubMed] [Google Scholar]
- 88.Head E, et al. Parallel compensatory and pathological events associated with Tau pathology in middle aged individuals with Down syndrome. J. Neuropathol. Exp. Neurol. 2003;62:917–926. doi: 10.1093/jnen/62.9.917. [DOI] [PubMed] [Google Scholar]
- 89.Serrano-Pozo A, Frosch MP, Masliah E, Hyman BT. Neuropathological alterations in Alzheimer disease. Cold Spring Harb. Perspect. Med. 2011;1:a006189. doi: 10.1101/cshperspect.a006189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Mann DMA. Cerebral amyloidosis, aging and Alzheimer’s disease: a contribution from studies on Down’s syndrome. Neurobiol. Aging. 1989;10:397–399. doi: 10.1016/0197-4580(89)90073-0. [DOI] [PubMed] [Google Scholar]
- 91.Evenhuis HM. The natural history of dementia in Down’s syndrome. Arch. Neurol. 1990;47:263–267. doi: 10.1001/archneur.1990.00530030029011. [DOI] [PubMed] [Google Scholar]
- 92.McCarron MO, Nicoll JAR, Graham DI. A quartet of Down’s syndrome, Alzheimer’s disease, cerebral amyloid angiopathy, and cerebral haemorrhage: interacting genetic risk factors. J. Neurol. Neurosurg. Psychiatry. 1998;65:405–406. doi: 10.1136/jnnp.65.3.405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Mendel T, Bertrand E, Szpak GM, Stepien T, Wierzba-Bobrowicz T. Cerebral amyloid angiopathy as a cause of an extensive brain hemorrhage in adult patient with Down’s syndrome — a case report. Folia Neuropathol. 2010;48:206–211. [PubMed] [Google Scholar]
- 94.Naito KS, Sekijima Y, Ikeda SI. Cerebral amyloid angiopathy-related hemorrhage in a middle-aged patient with Down’s syndrome. Amyloid. 2008;15:275–277. doi: 10.1080/13506120802524981. [DOI] [PubMed] [Google Scholar]
- 95.Donahue JE, Khurana JS, Adelman LS. Intracerebral hemorrhage in two patients with Down’s syndrome and cerebral amyloid angiopathy. Acta Neuropathol. 1998;95:213–216. doi: 10.1007/s004010050789. [DOI] [PubMed] [Google Scholar]
- 96.Handen BL, et al. Imaging brain amyloid in nondemented young adults with Down syndrome using Pittsburgh compound B. Alzheimers Dement. 2012;8:496–501. doi: 10.1016/j.jalz.2011.09.229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Nelson LD, et al. Positron emission tomography of brain β-amyloid and Tau levels in adults with Down syndrome. Arch. Neurol. 2011;68:768–774. doi: 10.1001/archneurol.2011.104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Matveev SV, et al. A distinct subfraction of Aβ is responsible for the high-affinity Pittsburgh compound B-binding site in Alzheimer’s disease brain. J. Neurochem. 2014;131:356–368. doi: 10.1111/jnc.12815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Hartley SL, et al. Cognitive functioning in relation to brain amyloid-β in healthy adults with Down syndrome. Brain. 2014;137:2556–2563. doi: 10.1093/brain/awu173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Landt J, et al. Using positron emission tomography and carbon 11-labeled Pittsburgh compound B to image brain fibrillar β-amyloid in adults with Down syndrome safety, acceptability, and feasibility. Arch. Neurol. 2011;68:890–896. doi: 10.1001/archneurol.2011.36. [DOI] [PubMed] [Google Scholar]
- 101.Lippa CF, et al. Transactive response DNA-binding protein 43 burden in familial Alzheimer disease and Down syndrome. Arch. Neurol. 2009;66:1483–1488. doi: 10.1001/archneurol.2009.277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Davidson YS, et al. TDP-43 pathological changes in early onset familial and sporadic Alzheimer’s disease, late onset Alzheimer’s disease and Down’s syndrome: association with age, hippocampal sclerosis and clinical phenotype. Acta Neuropathol. 2011;122:703–713. doi: 10.1007/s00401-011-0879-y. [DOI] [PubMed] [Google Scholar]
- 103.Gibb WRG, Mountjoy CQ, Mann DMA, Lees AJ. A pathological study of the association between Lewy body disease and Alzheimer’s disease. J. Neurol. Neurosurg. Psychiatry. 1989;52:701–708. doi: 10.1136/jnnp.52.6.701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Prasher VP, Airuehia E, Carey M. The first confirmed case of Down syndrome with dementia with Lewy bodies. J. Appl. Res. Intellect. Disabil. 2010;23:296–300. [Google Scholar]
- 105.Cataldo AM, et al. Aβ localization in abnormal endosomes: association with earliest Aβ elevations in AD and Down syndrome. Neurobiol. Aging. 2004;25:1263–1272. doi: 10.1016/j.neurobiolaging.2004.02.027. [DOI] [PubMed] [Google Scholar]
- 106.Lambert JC, et al. Meta-analysis of 74,046 individuals identifies 11 new susceptibility loci for Alzheimer’s disease. Nat. Genet. 2013;45:1452–1458. doi: 10.1038/ng.2802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Hooli BV, et al. Rare autosomal copy number variations in early-onset familial Alzheimer’s disease. Mol. Psychiatry. 2014;19:676–681. doi: 10.1038/mp.2013.77. [DOI] [PubMed] [Google Scholar]
- 108.Llado A, et al. Large APP locus duplication in a sporadic case of cerebral haemorrhage. Neurogenetics. 2014;15:145–149. doi: 10.1007/s10048-014-0395-z. [DOI] [PubMed] [Google Scholar]
- 109.Cabrejo L, et al. Phenotype associated with APP duplication in five families. Brain. 2006;129:2966–2976. doi: 10.1093/brain/awl237. [DOI] [PubMed] [Google Scholar]
- 110.Noebels J. A perfect storm: converging paths of epilepsy and Alzheimer’s dementia intersect in the hippocampal formation. Epilepsia. 2011;52:39–46. doi: 10.1111/j.1528-1167.2010.02909.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Griffiths-Jones S. The microRNA registry. Nucleic Acids Res. 2004;32:D109–D111. doi: 10.1093/nar/gkh023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Vilardell M, et al. Meta-analysis of heterogeneous Down syndrome data reveals consistent genome-wide dosage effects related to neurological processes. BMC Genomics. 2011;12:229. doi: 10.1186/1471-2164-12-229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Letourneau A, et al. Domains of genome-wide gene expression dysregulation in Down’s syndrome. Nature. 2014;508:345–350. doi: 10.1038/nature13200. [DOI] [PubMed] [Google Scholar]
- 114.Horvath S, et al. Accelerated epigenetic aging in Down syndrome. Aging Cell. 2015;14:491–495. doi: 10.1111/acel.12325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Cheon MS, Dierssen M, Kim SH, Lubec G. Protein expression of BACE1, BACE2 and APP in Down syndrome brains. Amino Acids. 2008;35:339–343. doi: 10.1007/s00726-007-0618-9. [DOI] [PubMed] [Google Scholar]
- 116.Choi JHK, et al. Age-dependent dysregulation of brain amyloid precursor protein in the Ts65Dn Down syndrome mouse model. J. Neurochem. 2009;110:1818–1827. doi: 10.1111/j.1471-4159.2009.06277.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Seo H, Isacson O. Abnormal APP, cholinergic and cognitive function in Ts65Dn Down’s model mice. Exp. Neurol. 2005;193:469–480. doi: 10.1016/j.expneurol.2004.11.017. [DOI] [PubMed] [Google Scholar]
- 118.Teller JK, et al. Presence of soluble amyloid β-peptide precedes amyloid plaque formation in Down’s syndrome. Nat. Med. 1996;2:93–95. doi: 10.1038/nm0196-93. [DOI] [PubMed] [Google Scholar]
- 119.Busciglio J, et al. Altered metabolism of the amyloid β precursor protein is associated with mitochondrial dysfunction in Down’s syndrome. Neuron. 2002;33:677–688. doi: 10.1016/s0896-6273(02)00604-9. [DOI] [PubMed] [Google Scholar]
- 120.Govoni S, et al. Fibroblasts of patients affected by Down’s syndrome oversecrete amyloid precursor protein and are hyporesponsive to protein kinase C stimulation. Neurology. 1996;47:1069–1075. doi: 10.1212/wnl.47.4.1069. [DOI] [PubMed] [Google Scholar]
- 121.Murray A, Letourneau A, Canzonetta C. Isogenic induced pluripotent stem cell lines from an adult with mosaic Down syndrome model accelerated neuronal ageing and neurodegeneration. Stem Cells. 2015;33:2077–2084. doi: 10.1002/stem.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Shi YC, et al. A human stem cell model of early Alzheimer’s disease pathology in Down syndrome. Sci. Transl. Med. 2012;4:124ra29. doi: 10.1126/scitranslmed.3003771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Wolvetang EW, et al. The chromosome 21 transcription factor ETS2 transactivates the β-APP promoter: implications for Down syndrome. Biochim. Biophys. Acta. 2003;1628:105–110. doi: 10.1016/s0167-4781(03)00121-0. [DOI] [PubMed] [Google Scholar]
- 124.Dorval V, Mazzella MJ, Mathews PM, Hay RT, Fraser PE. Modulation of Aβ generation by small ubiquitin-like modifiers does not require conjugation to target proteins. Biochem. J. 2007;404:309–316. doi: 10.1042/BJ20061451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Li YH, et al. Positive and negative regulation of APP amyloidogenesis by sumoylation. Proc. Natl Acad. Sci. USA. 2003;100:259–264. doi: 10.1073/pnas.0235361100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Ryoo SR, et al. Dual-specificity tyrosine(Y)-phosphorylation regulated kinase 1A-mediated phosphorylation of amyloid precursor protein: evidence for a functional link between Down syndrome and Alzheimer’s disease. J. Neurochem. 2008;104:1333–1344. doi: 10.1111/j.1471-4159.2007.05075.x. [DOI] [PubMed] [Google Scholar]
- 127.Wang X, et al. Sorting nexin 27 regulates Aβ production through modulating γ-secretase activity. Cell Rep. 2014;9:1023–1033. doi: 10.1016/j.celrep.2014.09.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Mok KY, et al. Polymorphisms in BACE2 may affect the age of onset Alzheimer’s dementia in Down syndrome. Neurobiol. Aging. 2014;35:1513.e1–1513.e5. doi: 10.1016/j.neurobiolaging.2013.12.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Sun XL, He GQ, Song WH. BACE2, as a novel APP θ-secretase, is not responsible for the pathogenesis of Alzheimer’s disease in Down syndrome. FASEB J. 2006;20:1369–1376. doi: 10.1096/fj.05-5632com. [DOI] [PubMed] [Google Scholar]
- 130.Azkona G, Levannon D, Groner Y, Dierssen M. In vivo effects of APP are not exacerbated by BACE2 co-overexpression: behavioural characterization of a double transgenic mouse model. Amino Acids. 2010;39:1571–1580. doi: 10.1007/s00726-010-0662-8. [DOI] [PubMed] [Google Scholar]
- 131.Holler CJ, et al. BACE2 expression increases in human neurodegenerative disease. Am. J. Pathol. 2012;180:337–350. doi: 10.1016/j.ajpath.2011.09.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Patel A, et al. Association of variants within APOE, SORL1, RUNX1, BACE1 and ALDH18A1 with dementia in Alzheimer’s disease in subjects with Down syndrome. Neurosci. Lett. 2011;487:144–148. doi: 10.1016/j.neulet.2010.10.010. [DOI] [PubMed] [Google Scholar]
- 133.Prasher VP, et al. Significant effect of APOE epsilon 4 genotype on the risk of dementia in Alzheimer’s disease and mortality in persons with Down syndrome. Int. J. Geriatr. Psychiatry. 2008;23:1134–1140. doi: 10.1002/gps.2039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Deb S, et al. APOE epsilon 4 influences the manifestation of Alzheimer’s disease in adults with Down’s syndrome. Br. J. Psychiatry. 2000;176:468–472. doi: 10.1192/bjp.176.5.468. [DOI] [PubMed] [Google Scholar]
- 135.Coppus AMW, et al. The impact of apolipoprotein E on dementia in persons with Down’s syndrome. Neurobiol. Aging. 2008;29:828–835. doi: 10.1016/j.neurobiolaging.2006.12.013. [DOI] [PubMed] [Google Scholar]
- 136.Schupf N, et al. Onset of dementia is associated with apolipoprotein E epsilon 4 in Down’s syndrome. Ann. Neurol. 1996;40:799–801. doi: 10.1002/ana.410400518. [DOI] [PubMed] [Google Scholar]
- 137.Hyman BT, et al. Quantitative analysis of senile plaques in Alzheimer disease: observation of log-normal size distribution and molecular epidemiology of differences associated with apolipoprotein E genotype and trisomy 21 (Down syndrome) Proc. Natl Acad. Sci. USA. 1995;92:3586–3590. doi: 10.1073/pnas.92.8.3586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Royston MC, et al. Apolipoprotein-e epsilon-2 allele promotes longevity and protects patients with Down’s syndrome from dementia. Neuroreport. 1994;5:2583–2585. doi: 10.1097/00001756-199412000-00044. [DOI] [PubMed] [Google Scholar]
- 139.Jones EL, et al. Evidence that PICALM affects age at onset of Alzheimer’s dementia in Down syndrome. Neurobiol. Aging. 2013;34:2441.e1–2441.e5. doi: 10.1016/j.neurobiolaging.2013.03.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Lee JH, et al. Association between genetic variants in sortilin-related receptor 1 (SORL1) and Alzheimer’s disease in adults with Down syndrome. Neurosci. Lett. 2007;425:105–109. doi: 10.1016/j.neulet.2007.08.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Salehi A, et al. Increased App expression in a mouse model of Down’s syndrome disrupts NGF transport and causes cholinergic neuron degeneration. Neuron. 2006;51:29–42. doi: 10.1016/j.neuron.2006.05.022. [DOI] [PubMed] [Google Scholar]
- 142.Torroja L, Chu H, Kotovsky I, White K. Neuronal overexpression of APPL, the Drosophila homologue of the amyloid precursor protein (APP), disrupts axonal transport. Curr. Biol. 1999;9:489–492. doi: 10.1016/s0960-9822(99)80215-2. [DOI] [PubMed] [Google Scholar]
- 143.Jiang Y, et al. Alzheimer’s-related endosome dysfunction in Down syndrome is Aβ-independent but requires APP and is reversed by BACE-1 inhibition. Proc. Natl Acad. Sci. USA. 2010;107:1630–1635. doi: 10.1073/pnas.0908953107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Barbosa S, Pratte D, Schwarz H, Pipkorn R, Singer-Kruger B. Oligomeric Dop1p is part of the endosomal Neo1p–Ysl2p–Arl1p membrane remodeling complex. Traffic. 2010;11:1092–1106. doi: 10.1111/j.1600-0854.2010.01079.x. [DOI] [PubMed] [Google Scholar]
- 145.Swaminathan S, et al. Analysis of copy number variation in Alzheimer’s disease: the NIA-LOAD/NCRAD family study. Curr. Alzheimer Res. 2012;9:801–814. doi: 10.2174/156720512802455331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Chapman J, et al. A genome-wide study shows a limited contribution of rare copy number variants to Alzheimer’s disease risk. Hum. Mol. Genet. 2013;22:816–824. doi: 10.1093/hmg/dds476. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Yang DS, et al. Reversal of autophagy dysfunction in the TgCRND8 mouse model of Alzheimer’s disease ameliorates amyloid pathologies and memory deficits. Brain. 2011;134:258–277. doi: 10.1093/brain/awq341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Cossec JC, et al. Trisomy for synaptojanin1 in Down syndrome is functionally linked to the enlargement of early endosomes. Hum. Mol. Genet. 2012;21:3156–3172. doi: 10.1093/hmg/dds142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.McIntire LBJ, et al. Reduction of synaptojanin 1 ameliorates synaptic and behavioral impairments in a mouse model of Alzheimer’s disease. J. Neurosci. 2012;32:15271–15276. doi: 10.1523/JNEUROSCI.2034-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Zhu L, et al. Reduction of synaptojanin 1 accelerates Aβ clearance and attenuates cognitive deterioration in an Alzheimer mouse model. J. Biol. Chem. 2013;288:32050–32063. doi: 10.1074/jbc.M113.504365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Busciglio J, Yankner BA. Apoptosis and increased generation of reactive oxygen species in Down’s syndrome neurons in vitro. Nature. 1995;378:776–779. doi: 10.1038/378776a0. [DOI] [PubMed] [Google Scholar]
- 152.Shukkur EA, et al. Mitochondrial dysfunction and tau hyperphosphorylation in Ts1Cje, a mouse model for Down syndrome. Hum. Mol. Genet. 2006;15:2752–2762. doi: 10.1093/hmg/ddl211. [DOI] [PubMed] [Google Scholar]
- 153.Phillips AC, et al. Defective mitochondrial function in vivo in skeletal muscle in adults with Down’s syndrome: a 31P-MRS study. PLoS ONE. 2013;8:e84031. doi: 10.1371/journal.pone.0084031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Weick JP, et al. Deficits in human trisomy 21 iPSCs and neurons. Proc. Natl Acad. Sci. USA. 2013;110:9962–9967. doi: 10.1073/pnas.1216575110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Zigman WB. Atypical aging in Down syndrome. Dev. Disabil. Res. Rev. 2013;18:51–67. doi: 10.1002/ddrr.1128. [DOI] [PubMed] [Google Scholar]
- 156.Picard M, Mcewen BS. Mitochondria impact brain function and cognition. Proc. Natl Acad. Sci. USA. 2014;111:7–8. doi: 10.1073/pnas.1321881111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Lott IT, et al. Down syndrome and dementia: a randomized, controlled trial of antioxidant supplementation. Am. J. Med. Genet. A. 2011;155A:1939–1948. doi: 10.1002/ajmg.a.34114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Carlson GA, et al. Genetic modification of the phenotypes produced by amyloid precursor protein overexpression in transgenic mice. Hum. Mol. Genet. 1997;6:1951–1959. doi: 10.1093/hmg/6.11.1951. [DOI] [PubMed] [Google Scholar]
- 159.Murakami K, et al. SOD1 (copper/zinc superoxide dismutase) deficiency drives amyloid β protein oligomerization and memory loss in mouse model of Alzheimer disease. J. Biol. Chem. 2011;286:44557–44568. doi: 10.1074/jbc.M111.279208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Zis P, Dickinson M, Shende S, Walker Z, Strydom A. Oxidative stress and memory decline in adults with Down syndrome: longitudinal study. J. Alzheimers Dis. 2012;31:277–283. doi: 10.3233/JAD-2012-120073. [DOI] [PubMed] [Google Scholar]
- 161.deHaan JB, et al. Elevation in the ratio of Cu/Zn-superoxide dismutase to glutathione peroxidase activity induces features of cellular senescence and this effect is mediated by hydrogen peroxide. Hum. Mol. Genet. 1996;5:283–292. doi: 10.1093/hmg/5.2.283. [DOI] [PubMed] [Google Scholar]
- 162.Anderson JS, et al. Abnormal brain synchrony in Down syndrome. Neuroimage Clin. 2013;2:703–715. doi: 10.1016/j.nicl.2013.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Belichenko PV, et al. Excitatory-inhibitory relationship in the fascia dentata in the Ts65Dn mouse model of down syndrome. J. Comp. Neurol. 2008;512:453–466. doi: 10.1002/cne.21895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Fernandez F, et al. Pharmacotherapy for cognitive impairment in a mouse model of Down syndrome. Nat. Neurosci. 2007;10:411–413. doi: 10.1038/nn1860. [DOI] [PubMed] [Google Scholar]
- 165.Schmidt-Sidor B, Wisniewski KE, Shepard TH, Sersen EA. Brain growth in Down syndrome subjects 15 to 22 weeks of gestational age and birth to 60 months. Clin. Neuropathol. 1990;9:181–190. [PubMed] [Google Scholar]
- 166.Takashima S, Becker LE, Armstrong DL, Chan F. Abnormal neuronal development in the visual cortex of the human fetus and infant with down’s syndrome. A quantitative and qualitative Golgi study. Brain Res. 1981;225:1–21. doi: 10.1016/0006-8993(81)90314-0. [DOI] [PubMed] [Google Scholar]
- 167.Adorno M, et al. Usp16 contributes to somatic stem-cell defects in Down’s syndrome. Nature. 2013;501:380–384. doi: 10.1038/nature12530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Canzonetta C, et al. DYRK1A-dosage imbalance perturbs NRSF/REST levels, deregulating pluripotency and embryonic stem cell fate in Down syndrome. Am. J. Hum. Genet. 2008;83:388–400. doi: 10.1016/j.ajhg.2008.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Hibaoui Y, et al. Modelling and rescuing neurodevelopmental defect of Down syndrome using induced pluripotent stem cells from monozygotic twins discordant for trisomy 21. EMBO Mol. Med. 2014;6:259–277. doi: 10.1002/emmm.201302848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Chang KT, Min KT. Upregulation of three Drosophila homologs of human chromosome 21 genes alters synaptic function: implications for Down syndrome. Proc. Natl Acad. Sci. USA. 2009;106:17117–17122. doi: 10.1073/pnas.0904397106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Cvetkovska V, Hibbert AD, Emran F, Chen BE. Overexpression of Down syndrome cell adhesion molecule impairs precise synaptic targeting. Nat. Neurosci. 2013;16:677–682. doi: 10.1038/nn.3396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Wang BP, et al. The amyloid precursor protein controls adult hippocampal neurogenesis through GABAergic interneurons. J. Neurosci. 2014;34:13314–13325. doi: 10.1523/JNEUROSCI.2848-14.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Nicolas M, Hassan BA. Amyloid precursor protein and neural development. Development. 2014;141:2543–2548. doi: 10.1242/dev.108712. [DOI] [PubMed] [Google Scholar]
- 174.Ma’ayan A, Gardiner KJ, Iyengar R. The cognitive phenotype of Down syndrome: insights from intracellular network analysis. NeuroRx. 2006;3:396–406. doi: 10.1016/j.nurx.2006.05.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Drewes G, et al. Dephosphorylation of Tau-protein and Alzheimer paired helical filaments by calcineurin and phosphatase-2A. Febs Lett. 1993;336:425–432. doi: 10.1016/0014-5793(93)80850-t. [DOI] [PubMed] [Google Scholar]
- 176.Liu F, et al. Overexpression of Dyrk1A contributes to neurofibrillary degeneration in Down syndrome. FASEB J. 2008;22:3224–3233. doi: 10.1096/fj.07-104539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Woods YL, et al. The kinase DYRK phosphorylates protein-synthesis initiation factor eIF2Bepsilon at Ser539 and the microtubule-associated protein tau at Thr212: potential role for DYRK as a glycogen synthase kinase 3-priming kinase. Biochem. J. 2001;355:609–615. doi: 10.1042/bj3550609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Dowjat WK, et al. Trisomy-driven overexpression of DYRK1A kinase in the brain of subjects with Down syndrome. Neurosci. Lett. 2007;413:77–81. doi: 10.1016/j.neulet.2006.11.02. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Shi J, et al. Increased dosage of Dyrk1A alters alternative splicing factor (ASF)-regulated alternative splicing of Tau in Down syndrome. J. Biol. Chem. 2008;283:28660–28669. doi: 10.1074/jbc.M802645200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Wegiel J, et al. Link between DYRK1A overexpression and several-fold enhancement of neurofibrillary degeneration with 3-repeat Tau protein in Down syndrome. J. Neuropathol. Exp. Neurol. 2011;70:36–50. doi: 10.1097/NEN.0b013e318202bfa1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Moore S, et al. APP metabolism regulates Tau protestasis in human cerebral cortex neurons. Cell Rep. 2015;11:689–696. doi: 10.1016/j.celrep.2015.03.068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Lu T, et al. REST and stress resistance in ageing and Alzheimer’s disease. Nature. 2014;507:448–454. doi: 10.1038/nature13163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Kimura R, et al. The DYRK1A gene, encoded in chromosome 21 Down syndrome critical region, bridges between β-amyloid production and tau phosphorylation in Alzheimer disease. Hum. Mol. Genet. 2007;16:15–23. doi: 10.1093/hmg/ddl437. [DOI] [PubMed] [Google Scholar]
- 184.Vazquez-Higuera JL, et al. DYRK1A genetic variants are not linked to Alzheimer’s disease in a Spanish case-control cohort. BMC Med. Genet. 2009;10:129. doi: 10.1186/1471-2350-10-129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Cooper SA, et al. Toward onset prevention of cognitive decline in adults with Down syndrome (the TOP-COG study): study protocol for a randomized controlled trial. Trials. 2014;15:202. doi: 10.1186/1745-6215-15-202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Phillips MC. Molecular mechanisms of cellular cholesterol efflux. J. Biol. Chem. 2014;289:24020–24029. doi: 10.1074/jbc.R114.583658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Kim WS, et al. Role of ABCG1 and ABCA1 in regulation of neuronal cholesterol efflux to apolipoprotein E discs and suppression of amyloid-β peptide generation. J. Biol. Chem. 2007;282:2851–2861. doi: 10.1074/jbc.M607831200. [DOI] [PubMed] [Google Scholar]
- 188.Tansley GH, et al. The cholesterol transporter ABCG1 modulates the subcellular distribution and proteolytic processing of β-amyloid precursor protein. J. Lipid Res. 2007;48:1022–1034. doi: 10.1194/jlr.M600542-JLR200. [DOI] [PubMed] [Google Scholar]
- 189.Burgess BL, et al. ABCG1 influences the brain cholesterol biosynthetic pathway but does not affect amyloid precursor protein or apolipoprotein E metabolism in vivo. J. Lipid Res. 2008;49:1254–1267. doi: 10.1194/jlr.M700481-JLR200. [DOI] [PubMed] [Google Scholar]
- 190.Perry VH, Holmes C. Microglial priming in neurodegenerative disease. Nat. Rev. Neurol. 2014;10:217–224. doi: 10.1038/nrneurol.2014.38. [DOI] [PubMed] [Google Scholar]
- 191.Wilcock DM, Griffin WST. Down’s syndrome, neuroinflammation, and Alzheimer neuropathogenesis. J. Neuroinflammation. 2013;10:84. doi: 10.1186/1742-2094-10-84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Griffin WST, et al. Brain interleukin 1 and S-100 immunoreactivity are elevated in Down syndrome and Alzheimer disease. Proc. Natl Acad. Sci. USA. 1989;86:7611–7615. doi: 10.1073/pnas.86.19.7611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Naude PJW, et al. Serum NGAL is associated with distinct plasma amyloid-β peptides according to the clinical diagnosis of dementia in Down syndrome. J. Alzheimers Dis. 2015;45:733–743. doi: 10.3233/JAD-142514. [DOI] [PubMed] [Google Scholar]
- 194.Mann DMA, et al. Microglial cells and amyloid-β protein (A) deposition — association with Aβ40-containing plaques. Acta Neuropathol. 1995;90:472–477. doi: 10.1007/BF00294808. [DOI] [PubMed] [Google Scholar]
- 195.Xue QS, Streit WJ. Microglial pathology in Down syndrome. Acta Neuropathol. 2011;122:455–466. doi: 10.1007/s00401-011-0864-5. [DOI] [PubMed] [Google Scholar]
- 196.Sheng JG, Mrak RE, Griffin WST. S100β protein expression in Alzheimer disease: potential role in the pathogenesis of neuritic plaques. J. Neurosci. Res. 1994;39:398–404. doi: 10.1002/jnr.490390406. [DOI] [PubMed] [Google Scholar]
- 197.Mori T, et al. Overexpression of human S100B exacerbates cerebral amyloidosis and gliosis in the Tg2576 mouse model of Alzheimer’s disease. Glia. 2010;58:300–314. doi: 10.1002/glia.20924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Esposito G, et al. S100B induces tau protein hyperphosphorylation via Dickopff-1 up-regulation and disrupts the Wnt pathway in human neural stem cells. J. Cell. Mol. Med. 2008;12:914–927. doi: 10.1111/j.1582-4934.2008.00159.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Chen C, et al. Role of astroglia in Down’s syndrome revealed by patient-derived human-induced pluripotent stem cells. Nat. Commun. 2014;5:4430. doi: 10.1038/ncomms5430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Mills SM, et al. Preclinical trials in autosomal dominant AD: implementation of the DIAN-TU trial. Rev. Neurol. 2013;169:737–743. doi: 10.1016/j.neurol.2013.07.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Bothwell M, Giniger E. Alzheimer’s disease: neurodevelopment converges with neurodegeneration. Cell. 2000;102:271–273. doi: 10.1016/s0092-8674(00)00032-5. [DOI] [PubMed] [Google Scholar]
- 202.Purro SA, Galli S, Salinas PC. Dysfunction of Wnt signaling and synaptic disassembly in neurodegenerative diseases. J. Mol. Cell Biol. 2014;6:75–80. doi: 10.1093/jmcb/mjt049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Karmiloff-Smith A, et al. Genetic and environmental vulnerabilities in children with neurodevelopmental disorders. Proc. Natl Acad. Sci. USA. 2012;109:17261–17265. doi: 10.1073/pnas.1121087109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Moran PM, Higgins LS, Cordell B, Moser PC. Age-related learning-deficits in transgenic mice expressing the 751-amino acid isoform of human β-amyloid precursor protein. Proc. Natl Acad. Sci. USA. 1995;92:5341–5345. doi: 10.1073/pnas.92.12.5341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Yamaguchi F, et al. Transgenic mice for the amyloid precursor protein 695 isoform have impaired spatial memory. Neuroreport. 1991;2:781–784. doi: 10.1097/00001756-199112000-00013. [DOI] [PubMed] [Google Scholar]
- 206.Mucke L, et al. High-level neuronal expression of Aβ1–42 in wild-type human amyloid protein precursor transgenic mice: synaptotoxicity without plaque formation. J. Neurosci. 2000;20:4050–4058. doi: 10.1523/JNEUROSCI.20-11-04050.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Balducci C, Forloni GAPP. Transgenic mice: their use and limitations. Neuromolecular Med. 2011;13:117–137. doi: 10.1007/s12017-010-8141-7. [DOI] [PubMed] [Google Scholar]
- 208.Brault V, Pereira P, Duchon A, Herault Y. Modeling chromosomes in mouse to explore the function of genes, genomic disorders, and chromosomal organization. PLoS Genet. 2006;2:e86. doi: 10.1371/journal.pgen.0020086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Park IH, et al. Disease-specific induced pluripotent stem cells. Cell. 2008;134:877–886. doi: 10.1016/j.cell.2008.07.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Li LB, et al. Trisomy correction in Down syndrome induced pluripotent stem cells. Cell Stem Cell. 2012;11:615–619. doi: 10.1016/j.stem.2012.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Jiang J, et al. Translating dosage compensation to trisomy 21. Nature. 2013;500:296–300. doi: 10.1038/nature12394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Chang CY, et al. N-butylidenephthalide attenuates Alzheimer’s disease-like cytopathy in Down syndrome induced pluripotent stem cell-derived neurons. Sci. Rep. 2015;5:8744. doi: 10.1038/srep08744. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.