Abstract
These guidelines provide an evidence-based framework for the management of patients with large non-pedunculated colorectal polyps (LNPCPs), in addition to identifying key performance indicators (KPIs) that permit the audit of quality outcomes. These are areas not previously covered by British Society of Gastroenterology (BSG) Guidelines.
A National Institute of Health and Care Excellence (NICE) compliant BSG guideline development process was used throughout and the Appraisal of Guidelines for Research and Evaluation (AGREE II) tool was used to structure the guideline development process. A systematic review of literature was conducted for English language articles up to May 2014 concerning the assessment and management of LNPCPs. Quality of evaluated studies was assessed using the Scottish Intercollegiate Guidelines Network (SIGN) Methodology Checklist System. Proposed recommendation statements were evaluated by each member of the Guideline Development Group (GDG) on a scale from 1 (strongly agree) to 5 (strongly disagree) with >80% agreement required for consensus to be reached. Where consensus was not reached a modified Delphi process was used to re-evaluate and modify proposed statements until consensus was reached or the statement discarded. A round table meeting was subsequently held to finalise recommendations and to evaluate the strength of evidence discussed. The GRADE tool was used to assess the strength of evidence and strength of recommendation for finalised statements.
KPIs, a training framework and potential research questions for the management of LNPCPs were also developed. It is hoped that these guidelines will improve the assessment and management of LNPCPs.
Keywords: COLONIC POLYPS, COLORECTAL ADENOMAS, SURGICAL RESECTION, ENDOSCOPIC POLYPECTOMY, ENDOSCOPY
Objective
To provide a structured framework for the management of large non-pedunculated colorectal polyps (LNPCPs).
Aims and methods
The purpose of the guideline is to provide an evidence-based framework for the optimal management of LNPCPs for clinicians involved in their management, including gastroenterologists, nurse practitioners, physicians, colorectal surgeons, radiologists and pathologists. These guidelines refer specifically to lesions considered benign at the time of assessment and/or lesions without biopsy-proven malignancy. The management of malignant lesions is detailed in a recent position statement by the Association of Coloproctologists of Great Britain and Ireland (ACPGBI) and updated National Institute of Health and Care Excellence (NICE) guidelines for colorectal carcinoma.1–3
LNPCPs carry an increased risk of colorectal cancer, can be challenging lesions to resect endoscopically and are associated with an increased risk of incomplete excision and complications. The UK incidence of LNPCPs is unknown and no previous framework exists for the management of these lesions.
Key questions we sought to cover included:
What are the key definitions and terms associated with LNPCPs?
What are the available management options?
What are the key principles for optimal management, including both assessment and therapy?
Which are the most complex lesions and how should they be managed?
What histopathological considerations are important in the management of LNPCPs?
When is surgical or conservative management more appropriate than endoscopic therapy?
Can multidisciplinary input into assessment and therapy improve management?
What information should patients be given about their management?
How should anticoagulant and antiplatelet drugs be managed before and after procedure?
How should patients be followed up after endoscopic removal of LNPCPs?
What are the most appropriate key performance indicators for monitoring the quality of management of LNPCPs?
What can be done to improve formal training in the management of LNPCPs?
What aspects of LNPCP management have the weakest evidence base and what are the key research questions which will help address these?
The Appraisal of Guidelines for Research and Evaluation (AGREE II) instrument provided a methodological framework for the development of the guidelines. In accordance with the British Society of Gastroenterology (BSG) NICE-compliant guideline process, a Guideline Development Group (GDG) including gastroenterologists, endoscopists, colorectal surgeons, gastrointestinal pathologists and a patient representative was selected to ensure wide-ranging expertise across all relevant disciplines. The surgical and histopathological representatives were nominated by the ACPGBI and the Royal College of Pathologists, respectively. A writing subcommittee was formed to identify key search terms for a comprehensive literature review of the management of LNPCPs and to develop draft recommendation statements.
A literature search for English language articles published up to the present was performed using PubMed. The term ‘colonic polypectomy’ was entered into the PubMed MeSH database. A total of 5989 articles were returned. The terms ‘therapy’ and ‘surgery’ were used to filter the results based on relevance, after which, 2230 articles were returned and scrutinised for relevant articles. Additional PubMed searches were performed using additional search terms agreed by the writing subcommittee. The search terms used were ‘colorectal laterally spreading type polyps’, ‘endoscopic mucosal resection’, ‘complex colonic polyps’, ‘difficult colonic polyps’, ‘surgical management of colorectal laterally spreading type polyps’, ‘endoscopic polypectomy’, ‘anticoagulation in endoscopic polypectomy’, ‘obtaining informed consent for endoscopic procedures’, ‘diathermy in polypectomy’, ‘argon plasma coagulation for polypectomy’, ‘submucosal injection for endoscopic mucosal resection’, ‘malignant colonic polyps’, ‘piecemeal endoscopic mucosal resection’, ‘colorectal endoscopic submucosal dissection’, ‘surgical management of colonic polyps’, ‘laparoscopic surgery of colonic polyps’, ‘training in endoscopic polypectomy’ and ‘transanal endoscopic microsurgery’
Returned abstracts were reviewed for relevance. Additional references were obtained by cross-referencing and by recommendation from the GDG. Relevant published national and international guidelines were also scrutinised. The ‘Scottish Intercollegiate Guidelines Network (SIGN) Methodology Checklist System’ was used to evaluate the quality of studies and studies considered of suboptimal quality were excluded.4
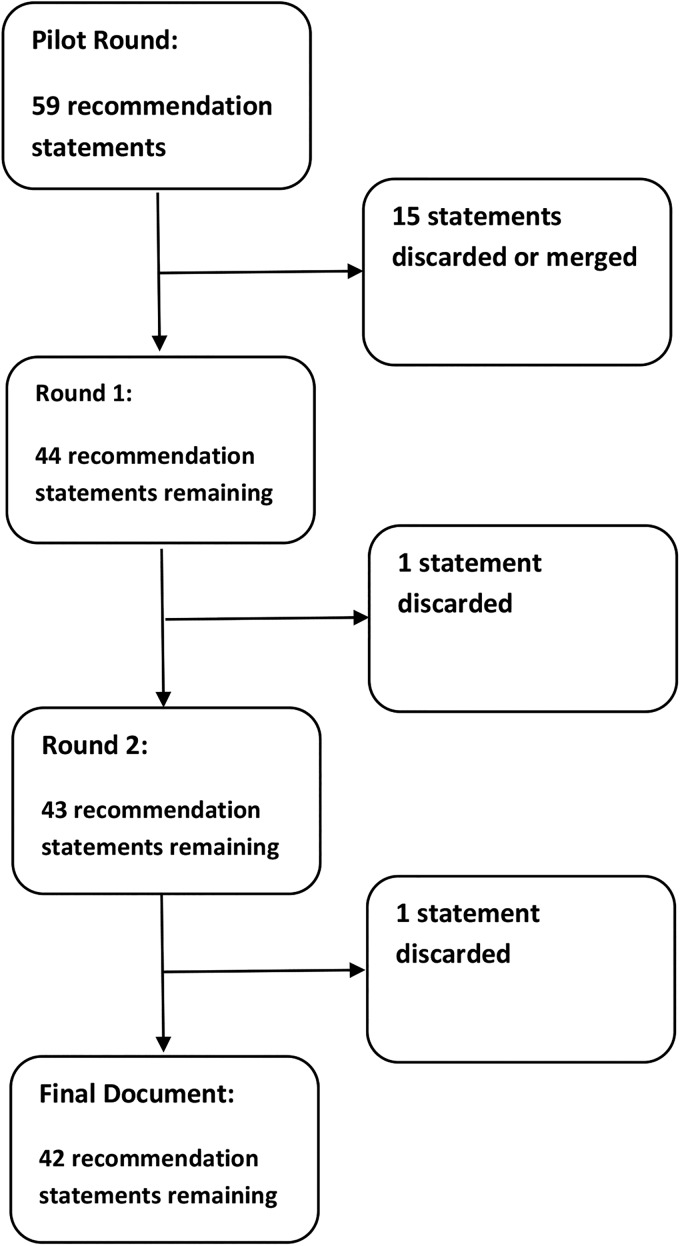
Initial draft statements formulated by the writing committee were reviewed by the GDG to allow for modification and to identify additional references. After a preliminary discussion, formal anonymous voting rounds were undertaken. Each statement was scored by each member of the GDG using a five-point scale. Consensus required at least 80% agreement. Where consensus was not reached, feedback from the GDG members was disseminated after each round to allow members to reconsider their original position. Where appropriate, revisions to statements were made and a further voting round was undertaken. A final round of voting for statements where consensus had not been reached took place at a round table meeting at the BSG offices on 26 March 2014 (figure 1). Voting was anonymous throughout, with the final round of voting made using an electronic keypad system.
Figure 1.
Diagram of statements used/discarded at each round.
The GRADE tool was used to evaluate the strength of evidence and the strength of recommendations made (see below). The GRADE system specifically separates the strength of evidence from the strength of a recommendation. While the strength of a recommendation may often reflect the evidence base, the GRADE system allows for occasions where this is not the case—for example, where it seems good sense to make a recommendation despite the absence of high-quality scientific evidence such as a large randomised controlled trial (RCT) (table 1).
Table 1.
An overview of the GRADE system5
| GRADE—strength of evidence | GRADE—strength of recommendation |
|---|---|
|
High quality: Further research is very unlikely to change our confidence in the estimate of effect |
The trade-offs: Taking into account the estimated size of the effect for main outcomes, the confidence limits around those estimates and the relative value placed on each outcome |
|
Moderate quality: Further research is likely to have an important impact on our confidence in the estimate of effect and may change the estimate |
The quality of the evidence |
|
Low quality: Further research is very likely to have an important impact on our confidence in the estimate of effect and is likely to change the estimate |
Translation of the evidence into practice in a particular setting: Taking into consideration important factors that could be expected to modify the size of expected effects |
|
Very low quality: Any estimate of effect is very uncertain |
Uncertainty about the baseline risk for the population of interest |
Executive summary of key recommendations
Guideline recommendations:
- Definitions
- We suggest that the term ‘non-pedunculated colorectal polyp’ (NPCP) is the most appropriate term to define sessile and flat colonic lesions, whereas the Paris classification and the term ‘laterally spreading type polyp’ (LST) may be used to subclassify lesions further.
- We suggest that the term ‘large NPCP’ (LNPCP) may be used to describe NPCPs >2 cm in size.
- We recommend that lesions displaying the following characteristics are identified as those with an increased risk of malignancy: lesions exhibiting: pit pattern type V, Paris 0–IIc or 0–IIa+IIc morphology, non-granular LST (laterally spreading type polyp, LST-NG), granular LSTs (LST-G) with a dominant nodule, distorted surface pattern, colour and vessels (NICE NBI type III), thick and irregular microvessels (Sano capillary pattern type III) (GRADE of evidence: moderate; Strength of recommendation: strong).
- We recommend that the following lesions with the following characteristics are identified as having an increased risk of incomplete excision/recurrence: size >40 mm, location involving ileocaecal valve, appendix, diverticulum or dentate line; within an inflamed segment of colitis; prior failed attempt at resection or recurrence at site of previous resection (excluding unifocal, diminutive and easily resected/ablated residual adenoma on first site check); non-lifting sign after submucosal injection; endoscopist concern about difficult location (eg, behind flexure or fold, in stenotic diverticular disease) (GRADE of evidence: low; Strength of recommendation: strong).
- We recommend that endoscopic factors associated with an increased risk of adverse events include: caecal location, size >40 mm and endoscopist inexperience (GRADE of evidence: low; Strength of recommendation: strong).
- Complex NPCP: we suggest this term to describe NPCPs with any of the following features: (a) increased risk of malignancy; (b) increased risk of incomplete resection/recurrence; (c) increased risk of adverse event; (d) size, morphology, size, access (SMSA) level 4 (GRADE of evidence: low; Strength of recommendation: weak).
- Service provision and management principles
- We recommend that hospitals that detect or manage LNPCPs should develop a referral pathway to facilitate their management and processes to monitor the quality of the service. The pathway should ensure that patients have access to, and information about, a full range of therapeutic options, including laparoscopic surgery, a provision for the management of complex rectal lesions and endoscopists capable of performing endotherapy on complex NPCPs (GRADE of evidence: very low; Strength of recommendation: strong).
- We suggest that clinicians involved in the management of LNPCPs should have access to a multidisciplinary network such as a multidisciplinary meeting (MDM) to discuss complex cases (complex as defined in these guidelines). Membership should include at least one complex NPCP endoscopist, at least one colorectal laparoscopic surgeon and a gastrointestinal histopathologist (GRADE of evidence: very low; Strength of recommendation: weak).
- We recommend that all endoscopists performing endotherapy on LNPCPs should be highly experienced in standard polypectomy, should have endoscopy service approval for this work and should be subject to regular audit to ensure their key performance indicators are above minimum quality standards (GRADE of evidence: low; Strength of recommendation: strong).
- We suggest that patients with benign NPCPs should not undergo surgery without prior complex polyp MDM discussion (GRADE of evidence: very low; Strength of recommendation: weak).
- We suggest that primary therapeutic management of LNPCPs should be undertaken within 8 weeks of receipt of referral (GRADE of evidence: very low; Strength of recommendation: weak).
- We recommend that endoscopic resection should be first-line therapy for the removal of LNPCPs where there is no suspicion of malignancy (suspicion of malignancy as defined in these guidelines) (GRADE of evidence: moderate; Strength of recommendation: strong).
- We recommend that piecemeal resection (either endoscopic or surgical) should be avoided if malignancy is suspected (GRADE of evidence: low; Strength of recommendation: strong).
- We suggest that in the context of significant comorbidity, conservative management may sometimes be appropriate after detailed patient discussion and documentation (GRADE of evidence: very low; Strength of recommendation: weak).
- Lesion assessment
- We recommend that all LNPCPs should be photographed or videoed before removal (GRADE of evidence: very low; Strength of recommendation: strong).
- We suggest that a size estimate of LNPCPs should be made, ideally by measuring against an open snare (GRADE of evidence: low; Strength of recommendation: weak).
- We recommend that the Paris classification should be used wherever possible to describe polyp morphology (GRADE of evidence: low; Strength of recommendation: strong).
- We recommend that the surface characteristics of a polyp should be described using a classification system such as the NICE NBI or Kudo Pit Pattern classification. The use of image enhancement techniques (digital or chromoendoscopic) can improve diagnostic accuracy in lesion assessment (GRADE of evidence: moderate; Strength of recommendation: strong).
- We suggest that if a lesion may be amenable to endoscopic removal, biopsies should be used with caution, as there is a risk of submucosal tethering due to scarring, rendering the lesion unresectable. Where biopsies are required because of concern about cancer, they should be targeted to the area exhibiting features indicative of cancer, avoiding flat areas and the lesion periphery. Tunnelling biopsies (biopsy through biopsy) should not be used (GRADE of evidence: low; Strength of recommendation: weak).
- Endoscopic management: pre-procedure
- We recommend that adequate planning should be undertaken (including length of time booked for procedure, endoscopist and nursing staff skills and endoscopic equipment) so that before an attempt at advanced polypectomy, the endoscopist has a high level of confidence that complete resection can be achieved in a single procedure (GRADE of evidence: very low; Strength of recommendation: strong).
- We recommend that antiplatelet drugs such as clopidogrel and prasugrel, and newer antiplatelet agents such as ticagrelor should be stopped at least 7 days before resection in accordance with BSG Antiplatelet Guidelines (GRADE of evidence: moderate; Strength of recommendation: strong).
- We recommend that warfarin should be stopped at least 5 days before resection of LNPCPs, and the international normalised ratio (INR) should be confirmed as <1.5 before the procedure, in accordance with BSG Anticoagulation Guidelines (GRADE of evidence: moderate; Strength of recommendation: strong).
- We suggest that general recommendations about the management of newer anticoagulants which have differing properties, such as rivaroxaban and dabigatran, cannot be made owing to a lack of evidence. Appropriate specialist advice should be sought in this situation (GRADE of evidence: very low; Strength of recommendation: weak).
- We suggest that patients should consent to the risk of thromboembolic events such as stroke and venous thromboembolism when stopping anticoagulants before endoscopic resection (GRADE of evidence: very low; Strength of recommendation: strong).
- Advice given should be tailored to a patient's individual risk with a ‘bridging regimen’ of low molecular weight heparin given to high-risk individuals in accordance with BSG guidelines. The risk of bleeding versus risk of thromboembolic episode should also be explained (GRADE of evidence: low; Strength of recommendation: weak).
- We suggest that where cessation of anticoagulants or antiplatelet drugs is contraindicated owing to comorbidity, or where there is uncertainty, appropriate specialist advice should be sought. If the anticoagulation/antiplatelet medication is temporary and the lesion has been adequately assessed as being of low risk for cancer, deferral of resection until after this medication can be discontinued may be appropriate (Grade of evidence: very low; Strength of recommendation: weak).
- We suggest that evidence for the cessation/continuation of low-dose aspirin in the context of LNPCPs is weak and the decision should be individualised according to patient risk (GRADE of evidence: low; Strength of recommendation: weak).
- We recommend that when obtaining consent for the endoscopic resection of LNPCPs, written information in plain English should be given. Management options including endoscopic therapy, surgery and conservative management should be discussed. For endoscopic therapy, patients should be informed of the potential need for subsequent check procedures and surveillance endoscopy. The risks of post-procedure bleeding (both immediate and delayed), perforation and residual polyp/recurrence should be explained (GRADE of evidence: very low; Strength of recommendation: strong).
- Endoscopic management: peri-procedure
- We recommend that carbon dioxide should be used in preference to air insufflation during colonoscopy to improve patient comfort and safety (GRADE of evidence: high; Strength of recommendation: strong).
- We recommend that the use of contrast agents such as indigo carmine or methylene blue in the submucosal injection solution may be considered to help demarcate a lesion, its resection margins, and to outline a clear submucosal plane (GRADE of evidence: low; Strength of recommendation: strong).
- We suggest that the addition of low-concentration adrenaline to the submucosal injection solution may be considered to keep the resection field clear during endoscopic resection (GRADE of evidence: low; Strength of recommendation: weak).
- We suggest consideration of the use of colloidal-type submucosal injection solutions in preference to normal saline lifting solution for LNPCPs (GRADE of evidence: low; Strength of recommendation: weak).
- We suggest that endoscopists should be familiar with the range of snares available, although a single optimal snare cannot be recommended (GRADE of evidence: very low; Strength of recommendation: weak).
- We suggest that a prolonged pure coagulation current should be avoided owing to an increased risk of delayed post-polypectomy bleeding and thermal tissue injury (GRADE of evidence: low; Strength of recommendation: weak).
- We suggest that although en bloc endoscopic snare resection of lesions <20 mm is recommended to reduce the risk of recurrence and to enable more accurate histopathological interpretation, this practice should be used with caution in LNPCPs owing to an increased risk of diathermy-associated thermal injury and perforation (GRADE of evidence: low; Strength of recommendation: weak).
- We recommend that therapy-naïve lesions that fail to lift after adequate submucosal injection should not be subject to attempted resection with conventional snare polypectomy technique (GRADE of evidence: low; Strength of recommendation: strong).
- We recommend that during endoscopic piecemeal resection, the snare should be used to resect a lesion completely wherever possible. Thermal coagulation techniques, such as argon plasma coagulation (APC) and soft coagulation, may be used as adjuncts when snare resection of small residual fragments of polyp is not possible (GRADE of evidence: low; Strength of recommendation: strong).
- We recommend careful post-procedure inspection of the resection site and photographic documentation of completeness of resection (GRADE of evidence: low; Strength of recommendation: strong).
- We recommend that with the exception of the rectum or caecum, a tattoo should be applied in accordance with local policy to aid endoscopic follow-up or subsequent surgical resection. As tattooing may cause submucosal fibrosis, the tattoo should be placed at least 3 cm from the lesion (GRADE of evidence: very low; Strength of recommendation: strong).
- Endoscopic management: post-procedure
- We recommend that written information about the risk of post-procedure complications (including bleeding risk for up to 2 weeks), together with recommended actions and an emergency phone number should be provided for patients (GRADE of evidence: very low; Strength of recommendation: strong).
- We suggest that recommencement of anticoagulant and antiplatelet treatment after polypectomy should be considered on an individual basis, weighing up the risks of post-procedure bleeding with the risks of a thromboembolic event. Further specialist advice (ideally sought before the procedure) may be appropriate (GRADE of evidence: low; Strength of recommendation: weak).
- We recommend that in the case of piecemeal endoscopic mucosal resection (EMR), the initial follow-up should take place within 2–6 months (GRADE of evidence: low; Strength of recommendation: strong).
- We recommend that on follow-up, the scar site should be positively identified, scrutinised and photographed. Image enhancement with techniques such as dye spray and digital enhancement may aid detection of residual neoplasia on a polypectomy scar. Areas of possible residual polyp require tissue diagnosis and definitive treatment (GRADE of evidence: low; Strength of recommendation: strong).
- We suggest that the management of residual/recurrent polyp tissue can be challenging and should be performed by an endoscopist with complex NPCP experience (GRADE: low; Strength of recommendation: weak).
- We suggest that the management of ongoing recurrence should be discussed in a complex polyp MDM (GRADE of evidence: low; Strength of recommendation: weak).
- Surgical management of LNPCPs
- We recommend that surgical therapy should be considered where malignancy is suspected or concerns about the likelihood of incomplete endoscopic resection arise after complex polyp MDM discussion (GRADE of evidence: moderate; Strength of recommendation: strong).
- We recommend that laparoscopic therapy should be used in preference to open surgery in the surgical management of LNPCPs (GRADE of evidence: high; Strength of recommendation: strong).
Definitions and terminology
The term ‘non-pedunculated colorectal polyp’ (NPCP) was considered the clearest and most appropriate term to define sessile and flat colonic lesions (table 2). In accordance with other international series, it was agreed that the Paris classification and the term ‘laterally spreading type polyp’ (LST) may be used to subclassify lesions further. It was also agreed that these guidelines should focus primarily on polyps at least 2 cm in size, given the increased complexity associated with their removal and the increased risk of malignancy in this group.6 7 These lesions are referred to as LNPCPs unless specified otherwise. However, much of the guidance in this document may be applicable to smaller polyps.
-
We recommend that lesions with the following characteristics should be identified as those with as increased risk of malignancy: lesions exhibiting; pit pattern type V, Paris 0–IIc or 0–IIa+IIc morphology, non-granular LST (LST-NG), granular LSTs (LST-G) with a dominant nodule, distorted surface pattern, colour and vessels (NICE NBI type III), thick and irregular microvessels (Sano capillary pattern type III) (GRADE of evidence: moderate; Strength of recommendation: strong).
Consensus reached: 100% agreement
Table 2.
Summary of key performance indicators (KPIs) for the management of LNPCPs
| Domain | KPI | Minimum standard | Aspirational standard |
|---|---|---|---|
| Optimal decision-making | Surgery rate for LNPCPs | No current standard defined | |
| Endoscopic skill | Recurrence/residual polyp at 12 months in endoscopically managed LNPCPs | <10% | <5% |
| Safety | Perforation rate—EMR of LNPCPs | <2% | <0.5% |
| Perforation rate—ESD of LNPCPs | No current standard defined | ||
| Post-procedure bleeding rate—EMR of LNPCPs | <5% | No current standard defined | |
| Post-procedure bleeding rate—ESD of LNPCPs | No current standard defined | ||
| Timeliness | Time from diagnosis to referral for definitive therapy | <4 Weeks (28 days) (record % compliance with this timeline; no current standard defined) | |
| Time from referral to definitive therapy | <8 Weeks (56 days) (record % compliance with this timeline; no current standard defined) | ||
| Volume of procedures | Number of LNPCP procedures per endoscopist per year | No current standard defined |
EMR, endoscopic mucosal resection; ESD, endoscopic submucosal dissection; LNPCPs, large non-pedunculated colorectal polyps.
NPCPs with morphological features of depression (Paris 0–IIc/IIa+c) appear to correlate strongly with malignancy. A 2002 Paris workshop quoted an unpublished study of 3680 lesions where 61% of 0–IIc lesions displayed submucosal invasion, markedly higher than the morphological group with the next highest incidence of submucosal invasion (Paris Is: 34%).8 Lesions displaying surface characteristics of pit pattern type V are strongly associated with deep submucosal invasion. Specific analysis of lesions with type V pit pattern found a vastly higher incidence of malignancy than with other pit pattern types (56% vs 4.4% (pit pattern III) vs 5% (pit pattern IV) vs 0% (pit patterns I+II), n=479, p<0.001).9 10
LSTs may be divided into granular (LST-G) and non-granular (LST-NG) types.11 In a study of 511 LSTs, the frequency of submucosal invasion with LST-NG type lesions was twice that of LST-G type lesions (14% vs 7%, p<0.01).12 Closer scrutiny of LST-NG type lesions suggests that pseudo-depressed LST-NG lesions are associated with the highest risk of submucosal invasion: a Japanese study of 1363 LSTs of at least 10 mm in size demonstrated submucosal invasion in 42.1% of pseudo-depressed LST-NG lesions compared with 6.1% flat elevated LST-NGs (p<0.01).13 LST-G lesions with a nodule >10 mm were also strongly associated with submucosal invasion (>10 mm nodule: (29.8%) vs <10 mm nodule: (2%), OR=71.01, p<0.001).12 In view of these results, it appears that both LST-G type lesions with a large dominant nodule and LST-NGs warrant greatest concern.13
The identification of irregular and thickened microvessels using narrow-band imaging (NBI) (Sano capillary pattern classification) has been identified as an accurate method of determining depth of submucosal invasion.14 A study of 130 NPCPs reported that the Sano CP type III pattern was associated with 84.8% sensitivity, 88.7% specificity and 87.7% diagnostic accuracy for differentiating deep submucosal invasion (sm2/3) from more superficial involvement (sm1).15
Another recently validated method of identifying deep submucosal invasion, the NICE NBI classification, allows examination of the surface characteristic of a polyp based on surface appearance, colour and vessel pattern without the aid of magnifying colonoscopy.16 17 A 2013 Japanese study demonstrated an overall sensitivity and negative predictive value for high confidence prediction of deep malignant submucosal invasion of 92% in a tertiary centre setting.16
We recommend that lesions with the following characteristics are identified as having an increased risk of incomplete excision/recurrence: size >40 mm, location involving ileocaecal valve, appendix, diverticulum or dentate line; within an inflamed segment of colitis; prior failed attempt at resection or recurrence at site of previous resection (excluding unifocal, diminutive and easily resected/ablated residual adenoma on first site check); non-lifting sign after submucosal injection; endoscopist concern about difficult location (eg, behind flexure or fold, in stenotic diverticular disease); (GRADE of evidence: low; Strength of recommendation: strong).
Consensus reached: 92.3% agreement
Various features of NPCPs have been identified that may predict the difficulty of achieving complete resection.18 19
Very large lesions are more technically challenging and time-consuming to remove as they are associated with a higher likelihood of needing eventual surgical management.7 11 20 A study of LNPCPs managed within the UK Bowel Cancer Screening Programme (BCSP) identified lesions >40 mm as more likely to require surgery (20–29 mm (7.8%) vs 30–39 mm (23.9%) vs >40 mm (27.5%), p<0.001) and requiring an increased number of endoscopic procedures to achieve clearance (20–29 mm (1.84) vs 30–39 mm (2.31) vs >40 mm (2.33), p<0.001).7
Polyps that cross two haustral folds and polyps behind a fold or that have a ‘clamshell’ distribution around a fold are recognised as challenging to remove endoscopically.20
NPCPs that fail to lift in response to an accurately placed submucosal fluid injection (non-lifting sign) without prior intervention have an increased risk of deep submucosal invasion indicating a reduced likelihood of successful removal with snare polypectomy (see later).9 21 NPCPs subject to a previously failed endoscopic attempt, that have occurred in the context of IBD or are located in a site of previous endoscopic resection site are likely to be subject to scarring and submucosal fibrosis and may not lift adequately after submucosal fluid injection. An analysis of cases of failed endotherapy highlighted non-lifting lesions as a major risk factor (relative risk (RR)=4.96, 95% CI 3.51 to 7.01, p<0.001).9
Peri-diverticular polyps may also pose a problem with endoscopic access as this portion of the colon may be narrower and less amenable to a stable endoscopic position. Moreover, polyp tissue may encroach into a diverticulum. Lesions involving the ileocaecal valve have also been associated with a higher failure rate (RR=2.61, 95% CI 1.28 to 5.32, p=0.020).9 These lesions may be difficult to access and visualise (especially in distinguishing ileal mucosa from adenomatous tissue), while ileal involvement adds further complexity (table 3).11 20
We recommend that endoscopic factors associated with an increased risk of adverse events include: caecal location, size >40 mm and endoscopist inexperience (GRADE of evidence: low; Strength of recommendation: strong).
Consensus reached: 84.6% agreement
Table 3.
Independent risk factors for failed endotherapy9
| Feature | Statistical association (n=479) |
|---|---|
| Previous intervention | OR: 3.75; 95% CI 1.77 to 7.94; p=0.001 |
| Ileocaecal valve involvement | OR=3.38; 95% CI 1.20 to 9.52; p=0.021 |
| Difficult position | OR=2.17; 95% CI 1.14 to 4.12; p=0.019 |
| Lesion size >40 mm | OR=4.37; 95% CI 2.43 to 7.88; p<0.001 |
| Previous APC use | OR=3.51; 95% CI 1.69 to 7.27; p=0.001 |
APC, argon plasma coagulation.
NPCPs located in the right colon, especially in the caecal pole, and lesions >40 mm appear to be linked to an increased risk of adverse events following advanced polypectomy. Right-sided lesions are associated with an increased risk of perforation due to thermal tissue injury with polypectomy in the thinner right-sided colon.22 Lesions involving the caecal pole, including those that affect the appendiceal orifice, are considered to carry the highest risk as this is where the colonic wall is at its thinnest, while the front-on access angle increases the potential for the entire colonic wall to be ensnared during polypectomy.11 An Australian study identified right-sided location as an important risk factor for post-procedure bleeding (PPB) (adjusted OR=4.4, 95% CI 1.3 to 14.1, p=0.014), with the highest incidence found in the caecum.22 These findings were similar to that of a retrospective analysis of 146 lesions where an almost fivefold increased risk of delayed haemorrhage was seen with right-sided polyps (OR=4.67, 95% CI 1.88 to 11.61, p=0.001), while univariate analysis suggested that caecal polyps conferred the highest risk (OR=13.82, 95% CI 2.66 to 71.73). Multivariate analysis also reported an increase in bleeding risk by 13% for every 1 mm increase in polyp diameter (OR=1.13, 95% CI 1.05 to 1.20, p<0.001).23
A polyp size of >40 mm was identified as a major risk factor for PPB in a study of 493 LNPCPs compared with resection of lesions <40 mm (OR=43.043, 95% CI 4.306 to 430.314, p=0.001).24
Further evidence of caecal location and lesion size >40 mm as risk factors for adverse events was reported in a study of adverse events from 167 208 polypectomies performed within the English Bowel Cancer Screening Programme. Caecal location (OR=2.13, 95% CI 1.36 to 3.34, p<0.01) and polyp size of >40 mm (OR=3.90, 95% CI 3.35 to 4.94, p<0.001) were both identified as strong risk factors for adverse events in endoscopic polypectomy. The risk of adverse events increased further with combination of both these factors with a predicted risk of bleeding of one in eight.25
Endoscopist inexperience also appears to be a clear risk factor for adverse outcomes. An almost threefold increase in the risk of heavy bleeding and perforation with inexperienced endoscopists was seen in a 2008 study (OR=2.96, 95% CI 1.57 to 5.61, p=0.0008).26 A trend of increased adverse events after therapeutic colonoscopy by less experienced endoscopists has also been shown in large-volume studies by Singh et al27 ((n=24 509, RR=5.4, 95% CI 3.0 to 9.0, p=0.02) and Chukmaitov et al28 (n=2 315 126, OR=1.18, 95% CI 1.07 to 1.30).
Complex NPCP. We suggest this term to describe NPCPs with any of the following features: (a) increased risk of malignancy; (b) increased risk of incomplete resection/recurrence; (c) increased risk of adverse event; (d) SMSA level 4 (GRADE of evidence: low; Strength of recommendation: weak).
Consensus reached: 92.3% agreement
The GDG considered it important to use the term ‘complex NPCP’ to describe lesions with a greater than average risk of malignancy, incomplete resection/recurrence or complications that may be best suited to management by clinicians with the relevant skills and experience within a multidisciplinary environment. An additional method of stratifying lesion complexity has also been devised. The SMSA scoring system predicts the difficulty of achieving successful endoscopic polypectomy based on the size, morphology, site and access of a polyp (see below). A study stratifying lesions (n=220) using the SMSA scoring system reported a lower level of endoscopic clearance with lesions felt to be the most complex (SMSA level 4) than with less complex lesions (SMSA level 2 and 3) (87.5% vs 97.5%, p=0.009). This system may aid in service planning and stratifying lesions that require referral to an expert centre (tables 4 and 5).19 29
Table 4.
Scoring system to assess polyp difficulty19
| Parameter | Range | Score |
|---|---|---|
| Size | <1 cm | 1 |
| 1–1.9 cm | 3 | |
| 2–2.9 cm | 5 | |
| 3–3.9 cm | 7 | |
| >4 cm | 9 | |
| Morphology | Pedunculated | 1 |
| Sessile | 2 | |
| Flat | 3 | |
| Site | Left | 1 |
| Right | 2 | |
| Access | Easy | 1 |
| Difficult | 3 |
Table 5.
SMSA scores with corresponding difficulty levels19
| Polyp level | Range of scores |
|---|---|
| I | 4–5 |
| II | 6–8 |
| III | 9–12 |
| IV | >12 |
SMSA, size, morphology, size, access.
Service provision and management principles
We recommend that hospitals that detect or manage LNPCPs should develop a referral pathway to facilitate their management and processes to monitor the quality of the service. The pathway should ensure that patients have access to, and information about, a full range of therapeutic options, including laparoscopic surgery, a provision for the management of complex rectal lesions and endoscopists capable of performing endotherapy on complex NPCPs (GRADE of evidence: very low; Strength of recommendation: strong).
Consensus reached: 100% agreement
A structured referral pathway may ensure better interspecialty communication and timely and efficient management of LNPCPs.30 A pathway enables the creation of an audit trail and subsequent monitoring of performance. Patients, irrespective of their location, should have access to a full range of management options that minimise the risk of morbidity and mortality. This includes access to endoscopists capable of performing advanced therapy on LNPCPs. In expert hands, over 90% of selected lesions may be successfully removed, and surgery avoided, including lesions previously felt to be endoscopically unresectable.9 31 32
The management of rectal lesions also requires special consideration given the complexity and morbidity associated with resectional surgery in this area and possible need for a permanent stoma.33 In this context it is important to differentiate between complex benign polyps (the main subject of this document) and early rectal cancer.
The management of rectal NPCPs is discussed in greater detail in ‘Surgical management of LNPCPs’.
The provision of advanced endoscopy services is also likely to be more cost-effective for hospital trusts and so a referral network to another centre is appropriate if the necessary expertise is not available locally. A 2013 UK analysis estimated a cost saving of £726 288 in a study of 220 patients (£3301.31 per patient) managed with endoscopy as opposed to surgery.29
For lesions where surgery is required, laparoscopic surgery should be available as a minimally invasive option with an equivalent lesion resection rate and accelerated post-operative recovery34 (see ‘Surgical management of LNPCPs’).
We suggest that clinicians involved in the management of LNPCPs should have access to a multidisciplinary network such as a MDM to discuss complex cases (complex as defined in these guidelines). Membership should include at least one complex NPCP endoscopist, at least one colorectal laparoscopic surgeon and a gastrointestinal histopathologist (GRADE of evidence: very low; Strength of recommendation: weak).
Consensus reached: 92% agreement
We recommend that all endoscopists performing endotherapy on LNPCPs should be highly experienced in standard polypectomy, should have endoscopy service approval for this work and should be subject to regular audit to ensure their key performance indicators are above minimum quality standards (GRADE of evidence: low; Strength of recommendation: strong).
Consensus reached: 92.3% agreement
Although advanced polypectomy is an effective modality, the technical demands mean that the potential for serious complications such as haemorrhage and perforation are higher than for standard snare polypectomy. Patient safety is paramount and the ability to accurately identify underperformance will allow prompt remedial action.35 36 In addition, failure to achieve complete resection complicates further management and means the risk of subsequent malignancy is suboptimally managed.37 Increased endoscopist experience is associated with superior outcomes. A 2002 study reported significantly increased successful LNPCP clearance by the expert group compared with a non-expert group (76% vs 40%, p=0.01).38 Endoscopist inexperience conclusively appears to directly affect patient safety. An almost threefold increase in the risk of heavy bleeding and perforation with the least experienced endoscopists and significantly increased adverse events for therapeutic colonoscopy with less experienced endoscopists in large-volume trials strongly highlights the importance of endoscopists who manage LNPCPs independently gaining sufficient experience beforehand.26–28 Technical endotherapy skill appears to vary widely even amongst experienced endoscopists. The CARE Study (n=418) found outcomes of incomplete resection varied widely between experienced endoscopists. The incomplete resection rate (IRR) for polyps thought to have been completely resected was higher than expected (IRR: 10.1% (95% CI 6.9% to 13.3%)), and increased significantly with larger polyp size (IRR 10–20 mm vs <10 mm: 17.3% vs 6.8%, p=0.003).39
These findings suggest that advanced endoscopic polypectomy capabilities are not universal. Auditing outcomes using identified key performance indicators (KPIs) may enable endoscopists managing LNPCPs independently to demonstrate competency with consistent high-quality outcomes, resulting in improved outcomes and safety.32 40
We suggest that patients with benign NPCPs should not undergo surgery without prior complex polyp MDM discussion (GRADE of evidence: very low; Strength of recommendation: weak).
Consensus reached: 84.6% agreement
There is increasing support for the view that multidisciplinary management can improve the management of LNPCPs, ideally via a dedicated complex polyp MDM or within an existing colorectal multidisciplinary team meeting where endoscopists capable of performing endotherapy on complex NPCPs are available. Key multidisciplinary team stakeholders should include a complex NPCP endoscopist, a laparoscopic colorectal surgeon and a gastrointestinal histopathologist. It is recognised that radiological input may be warranted in certain cases—for example, where there is difficulty in determining whether a lesion is benign or malignant. However, the GDG felt that the proportion of cases where radiological investigation changes the management of NPCPs was low. Radiological input was therefore not considered mandatory for a complex polyp MDM but suggested for consideration in selected cases.
Reports from specialised MDMs within the fields of gastroenterology and endoscopy have commented that increased, more rounded, clinician input contributes to a more robust decision-making process and closer analysis of the full range of management options.41 42 A prospective study (n=1909) reported that a benign hepatopancreatobiliary MDM before endoscopic retrograde cholangiopancreatography was associated with improved safety and decreased overall complications compared with control cases (6.9% vs 12.0%, p<0.001) and lower severe complication rates (0.4% vs 2.5%, p=0.035).43 Increased interaction between endoscopists and colorectal surgeons should encourage consideration of all possible management options. The availability of a multidisciplinary network with access to an expert centre may result in enhanced treatment options and avoidance of surgery.9 29 32 The therapeutic capabilities of different endoscopists are not uniform, and increasing evidence suggests that many LNPCPs initially felt to be endoscopically unresectable and therefore referred for surgery can be removed endoscopically in an expert setting. This is preferable given the increased cost, mortality and morbidity associated with surgery.39 44 45 A 2014 study of 38 LNPCPs initially referred for surgery without biopsy-proven cancer32 reported successful endotherapy in 71% of cases including 26% of lesions for which previous endotherapy was unsuccessful, whereas a 2011 Australian study and a 2013 UK study were able to achieve complete endoscopic resection in 74.5% of previously attempted lesions and 87.5% of the most complex LNPCPS, respectively.9 29 40 Close interaction with histopathology is also important to establish comprehensive information about the adequacy of histopathology specimens, the possibility of malignant features, and establishing whether complete resection after endotherapy can be determined.
We suggest that primary therapeutic management of LNPCPs should be undertaken within 8 weeks of receipt of referral (GRADE of evidence: very low; Strength of recommendation: weak).
Consensus reached: 100% agreement
Previous reports suggest that 7–15% of LNPCPs may already harbour malignancy.46 The risk of malignancy in this patient group indicates a need for timely treatment. However, this needs to be balanced with ensuring that patients are managed by clinicians with the appropriate expertise. There is also a need to ensure that a lesion has been adequately assessed either at the referring or receiving centre before treatment, which may necessitate additional diagnostic endoscopy and assessment time to ensure optimal management. An 8-week target was suggested as feasible and aligned with the National Health Service (NHS) 62-day target from referral to treatment for suspected cancers.47 Although there is no evidence for 8 weeks specifically, a drive towards ensuring that management is timely is desirable. The exact time sequence for adenoma to carcinoma transformation with NPCPs is unclear, but growth model studies have sought to estimate progression times. A 2001 polyp growth model study reported a transformation rate of 3% a year for lesions >1 cm and 20% a year for lesions with carcinoma in situ.48 A pre-colonoscopy barium enema study of polyps >1 cm left untreated between 12 and 229 months estimated a cumulative risk of cancer at the polyp site at 5, 10 and 20 years as 2.5%, 8% and 24%, respectively.49 It appears unlikely that a projected time frame of 8 weeks will compromise patient safety, while more time is available to ensure that an appropriate endoscopist is available.
We recommend that endoscopic resection is first-line therapy for the removal of LNPCPs where there is no suspicion of malignancy (suspicion of malignancy as defined in these guidelines) (GRADE of evidence: moderate; Strength of recommendation: strong).
Consensus reached: 92.3% agreement
While surgical therapy has historically been used to remove some colorectal LSTs, endoscopic removal is now recognised as first-line therapy internationally. While endoscopic mucosal resection (EMR) and endoscopic submucosal dissection (ESD) are both management options, the limited availability of ESD in Western countries, together with technical considerations such as procedure time and a higher level of perforation (up to 10%), means that EMR appears the most viable option for lesions with no features indicative of malignancy.11 50 The availability of EMR is high and international studies, including those of complex lesions, have shown that EMR is effective, with reported curative rates of approximately 90%.9 31
The ACE study demonstrated treatment success in 91% of treatment-naive lesions and 74.5% of previously attempted lesions, with 89.2% of LNPCPS successfully removed in a single session.9 A 2012 study of 315 ‘defiant’ polyps referred to an expert centre reported successful endoscopic removal in 91% of cases, all in a single session.31 A 2013 UK study of the endoscopic management of 220 colorectal lesions using EMR in an expert centre, demonstrated successful endoscopic treatment in 96% of cases with 87.5% of LNPCPs felt to be the most complex (SMSA level 4) successfully removed.29 The economic argument for endoscopic management as first-line treatment is strong with a cost saving of $5108.45 per patient compared with surgery in a UK setting, and a $6990 saving per patient estimated in an Australian study (186 LNPCPs).44 Surgical resection appears less safe, with reported rates of morbidity and mortality of 20% and 1%, respectively.45
We recommend that piecemeal resection (either endoscopic or surgical) should be avoided if malignancy is suspected (GRADE of evidence: low; Strength of recommendation: strong).
Consensus reached: 84.6% agreement
An important oncological principle is that suspected malignant lesions are removed en bloc. En bloc lesion removal is associated with a lower level of lesion recurrence and a higher early cure rate than piecemeal resection.51 In addition, en bloc resection allows precise histological analysis such as definitive evaluation of lateral and vertical resection margins and depth of invasion and thus is essential to ascertain the presence of favourable or unfavourable histological criteria.52 Although, en bloc resection of LNPCPs using EMR is often not possible, the likelihood of achieving this is higher with ESD, with various studies demonstrating en bloc resection with this technique at a rate of approximately 90%.53 A Japanese retrospective analysis comparing lesions managed by ESD (n=145, 66% containing malignancy) with piecemeal EMR (pEMR) (n=228, 69% containing malignancy) demonstrated only 2% recurrence with ESD compared with 14% recurrence with EMR (p<0.0001), reporting a markedly higher cure rate with no significant difference in complications between the two groups.54 Another comparison study between en bloc endoscopic removal (ESD/EMR) and pEMR of benign lesions reported a similar trend for recurrence (n=269, ESD: 0%, EMR: 1.4%, pEMR: 12.1%, p<0.001) with similar complication rates.55
Unlike piecemeal resection, en bloc removal may be effective as both a diagnostic and therapeutic tool where a suspicion of malignancy exists. A 2012 Japanese retrospective series (n=589) assessing ESD outcomes for lesions with suspected but not proven malignancy at endoscopic assessment demonstrated en bloc and curative resection in 87% and 80% of cases, respectively.56 A 2013 multicentre Japanese study reported outcomes from a series of lesions removed by ESD that were retrospectively found to contain submucosal malignancy. Five-year recurrence-free survival was reported in 98% of ‘low-risk’ cases managed with ESD (negative vertical margins, were reported as well as moderately differentiated adenocarcinoma, absence of lymphovascular invasion, and invasion depth <1000 µm), whereas figures of 87% and 97% were reported in ‘high-risk’ lesions (presence of any of the earlier described features) for lesions managed with ESD and ESD + surgery, respectively.57 However, while the potential efficacy of ESD is clear, there are significant challenges with achieving appropriate training, access and standardisation in a non-Japanese setting. The use of multidisciplinary networks appears important in ensuring increased access to ESD for UK patients.
Aside from surgical resectional therapy, the use of minimally invasive surgical therapy such as transanal endoscopic microsurgery (TEMS) resection in the management of rectal polyps can be used to achieve en bloc resection of rectal lesions where malignancy requires exclusion. TEMS for the management of rectal LNPCPs has been associated with lower rates of early recurrence than with pEMR, in addition to allowing more robust histological examination. It should be noted, however, that late recurrence rates appear equivalent when allowing for repeat EMR and that TEMS has been associated with longer hospitalisation. In a few specialist centres, TEMS can involve an overnight stay only or be performed as a day-case procedure.58 TEMS is discussed in greater detail in ‘Surgical management of LNPCPs’.
pEMR is already established as resulting in a higher level of recurrence, but the risk appears even larger with malignancy. One study found a 10-fold increase in recurrence compared with benign lesions (n=50, 33.3% vs 3.1%, p<0.05) while a 2009 Japanese study (n=572) also reported higher recurrence (25% vs 17.1%, p<0.01).59 60 Piecemeal removal of malignancy has also been identified as an independent risk factor for incomplete resection by a 2011 Korean study (n=236, OR=3.365, 95% CI 1.295 to 8.744, p=0.013).61 Unlike en bloc retrieval, piecemeal removal results in the retrieval of poorer quality histological samples and it is often not possible to evaluate the completeness of resection, depth of invasion, lateral resection margins and other prognostic features. Surgery is often required in this situation owing to inadequate staging.29
The ability to evaluate resection margins is vital as it helps to ascertain the completeness of resection and can predict the likelihood of residual disease. A meta-analysis of 31 studies (n=1900) identified positive resection margins (<1 mm) as a strong risk factor for residual disease (OR=22, p<0.0001).62 A finding of indeterminate resection margin status, a common problem with piecemeal removal, may also predict an increased risk of residual/recurrent disease as demonstrated by Butte et al63 (n=143, resection margins <1 mm: 16%, indeterminate margins: 21%, negative resection margins (>1 mm): 0%, p=0.009) (figure 2). Evaluation of the depth of submucosal invasion is also important as its depth has been shown to correspond to the risk of lymph node metastases. A large analysis of T1 colorectal carcinomas in 2002 (n=7543) found that lesions with deep submucosal invasion (sm3) were associated with a highly significant risk of lymph node metastases (p<0.001).64 This histological information is therefore vital in establishing whether a patient has been cured, the risk of recurrence and planning of subsequent treatment.
Figure 2.
Poor prognostic histological features.66
Although piecemeal endotherapy is effective for the management of benign lesions, for probable malignancy (eg, non-lifting sign in treatment-naïve lesions, pit pattern V, Paris 0–IIc, LST-NG, NICE NBI type III, Sano capillary pattern type 3), a higher level of recurrence and incomplete resection, an inability to sample or remove the lymph node basin and an inability to confirm eradication owing to the retrieval of suboptimal histological specimens highlight its inadequacy.18
Another note of caution is that several reports indicate a high level of residual malignancy on surgical resection specimens where complete polypectomy had been considered to have taken place. A study of 143 malignant lesions managed endoscopically reported residual malignancy in 19% of cases, while another analysis of 63 lesions resected endoscopically with a retrospective finding of early malignancy found residual malignancy in the colon wall and/or lymph nodes in almost 50% of cases managed surgically.63 65 However, this situation may be minimised through detailed assessment for complexity, advanced morphology and risk of complications as mentioned above.
We suggest that in the context of significant comorbidity, conservative management may sometimes be appropriate after detailed patient discussion and documentation (GRADE of evidence: very low; Strength of recommendation: weak).
Consensus reached: 85.7% agreement
While LNPCPs are associated with a risk of malignant transformation and may sometimes already harbour malignancy, the risk of symptomatic malignancy and cancer-related mortality from these lesions may be outweighed by patient factors that may more imminently reduce life expectancy. In this context, subjecting a patient to the additional immediate risks of endoscopic or surgical resection may not be in their best interests.
As previously discussed, adenoma to carcinoma transformation to a point where a lesion becomes symptomatic may take years.48 Patient factors requiring consideration include advanced age, frailty, comorbidities such as chronic cardiorespiratory conditions and other established malignancy. The use of mortality index models such as the Schonberg Index may help to stratify individual patient risk before attempting invasive treatment.66 For patients with increased age or severe comorbidity, both endoscopic and surgical therapy may prove hazardous, with the use of sedation and general anaesthetic posing significant cardiopulmonary safety concern. An Australian study reported an increased risk in 30-day mortality in non-cardiac surgery in patients over 70 (OR=1.09 per year over 70 years, 95% CI 1.04 to 1.13, p<0.001).67 The risks of increased surgical mortality and morbidity are important factors, as is consideration of whether a patient might survive a serious endoscopic complication and subsequent treatment. Conservative management may therefore prove appropriate where life expectancy is already greatly reduced.
Lesion assessment
We recommend that all LNPCPs should be photographed or videoed before removal (GRADE of evidence: very low; Strength of recommendation: strong).
Consensus reached: 100% agreement
Comprehensive documentation with photos or video is considered good practice. A study comparing lesion assessment between US and Japanese expert endoscopists demonstrated a significant difference in the interpretation of flat lesions, including the identification of lesion depression.68 Misclassification may have implications for subsequent management (eg, endotherapy vs surgery). The use of imaging before therapy may allow for more accurate lesion assessment by additional multidisciplinary specialists without the need for repeat endoscopy.42
We suggest that a size estimate of LNPCPs should be made, ideally by measuring against an open snare (GRADE of evidence: low; Strength of recommendation: weak).
Consensus reached: 100% agreement
Pathological estimation appears to be the most accurate method of assessing lesion size, but size estimation during endoscopy is important for deciding upon surveillance intervals and important also when considering the malignant potential of an NPCP and technical considerations such as deciding on en bloc or piecemeal resection or the resection plane.69 There is extensive evidence that visual size estimation during endoscopy continues to be inaccurate. A 1997 study including 61 LNPCPs, using pathological size estimation as a reference, reported that 20% of lesions were inaccurately estimated.70 A 2013 study (n=230) found that 62.6% of lesions were mis-sized by >33%, with 47.8% of lesions undergoing inappropriate surveillance because of this.71 The use of measurement tools has been shown to improve the accuracy of endoscopic size estimates.72 A readily available modality is the use of an open snare and their use as a size reference may improve accuracy.
We recommend that the Paris classification should be used wherever possible to describe polyp morphology (GRADE of evidence: low; Strength of recommendation: strong).
Consensus reached: 100% agreement
A Paris classification model for the description of polyps based on morphology was described in 2002.8 This was further revised in 2003 to enable the evaluation of superficial lesions with respect to the depth of submucosal invasion. Lesions were classified as protruding (0–I; incorporating pedunculated and sessile polyps), non-protruding and non-excavated (0–II; flat—further divided as elevated (IIa), flat (IIb) and depressed (IIc)) and excavated (0–III).73 Lesion morphology appears to accurately predict the risk of malignancy. Non-protruding depressed lesions were highlighted as having an increased risk of malignancy.8 The initial finding of increased risk of submucosal invasion with Paris 0–IIc lesions compared with sessile lesions (n=3680, 61% vs 3%) has been repeated (n=479) (IIc or IIa+c: 31.8% vs IIb: 11.1% vs Is: 7.5% (p=0.001)).8 9 Furthermore, these lesions also correlate with Kudo Pit Pattern type V, a more established indicator of likely malignancy.74 This demonstrates the reliability of the Paris classification in predicting malignancy and its use in guiding optimal management.8 73
We recommend that the surface characteristics of a polyp should be described using a classification system such as the NICE NBI or Kudo Pit Pattern classification. The use of image enhancement techniques (digital or chromoendoscopic) can improve diagnostic accuracy in lesion assessment (GRADE of evidence: Moderate; Strength of recommendation: strong).
Consensus reached: 91.7% agreement
The use of pit pattern classification has been well described and is a robust method of delineating between hyperplastic and adenomatous polyps, and also accurate in predicting deep malignant submucosal invasion based on polyp surface characteristics.74–76 A finding of a ‘type V’ pit pattern is strongly associated with a risk of deep submucosal malignancy compared with other pit pattern types.9 10 Subclassification of type V pit pattern to VI (irregular arrangement) and VN (amorphous structure) can further stratify malignancy risk. The increased association of type VN pattern with malignancy was confirmed by a finding of malignancy in 100% of these lesions in data from a 2008 Japanese analysis, compared with a reported rate of malignancy of approximately 30% in type VI lesions.77 78 Further subclassification of the type VI pattern to mildly irregular and severely irregular has been proposed owing to a marked difference in malignancy incidence between the two groups (7–17% and 56–85%, respectively).77 78 While a learning curve is required to interpret pit patterns, and the potential for interobserver variation exists, the use of training modules suggests that pit pattern recognition can be achieved even by inexperienced endoscopists.79
Enhanced imaging techniques may help to improve diagnostic accuracy when assessing NPCPs.
NBI is a form of digital image enhancement that uses narrow-band filters and high-intensity blue light to enhance surface mucosal and vascular pattern visualisation. A multicentre RCT (n=667) found that NBI had greater accuracy than both standard and high definition white light endoscopy at correctly predicting polyp histology with a sensitivity of 90% (95% CI 85.3% to 93.4%, p<0.001) and accuracy of 82% (95% CI 77.4% to 85.4%, p<0.001).80 The importance of NBI is also reflected in its inclusion in the NICE classification system, which has demonstrated accuracy in identifying deep submucosal invasion. In addition, it has high availability and it appears that it can be used by inexperienced endoscopists with appropriate training. A Japanese study demonstrated 90% accuracy (95% CI 85.1% to 93.3%) by a student group using the system.11 16
Both NBI and magnifying chromoendoscopy seem to be accurate in delineating between neoplastic and non-neoplastic polyps. A study comparing both modalities with white light endoscopy reported a diagnostic accuracy of >90% compared with white light endoscopy (59%).81 The utility of magnifying chromoendoscopy has also been confirmed by a large prospective study (n=4215), which demonstrated the accuracy of magnifying chromoendoscopy at estimating the depth of invasion of early colorectal neoplasms using combined mucosal and morphological patterns. The sensitivity, specificity and diagnostic accuracy of the invasive pattern to differentiate mucosal cancer or superficial invasion (sm1) (<1000 µm) from deeper invasion (sm2–3) (≥1000 µm) was reported as 85.6%, 99.4% and 98.8%, respectively.82
Recent European Society of Gastrointestinal Endoscopy (ESGE) guidelines adopt a similar position by recommending the use of conventional or virtual (NBI) magnified chromoendoscopy to predict the risk of invasive cancer and deep submucosal invasion.83
We suggest that if a lesion may be amenable to endoscopic removal, biopsies should be used with caution, as there is a risk of submucosal tethering due to scarring, rendering the lesion unresectable. Where biopsies are required because of concern about cancer, they should be targeted to the area exhibiting features indicative of cancer, avoiding flat areas and the lesion periphery. Tunnelling biopsies (biopsy through biopsy) should not be used (GRADE of evidence: low; Strength of recommendation: weak).
Consensus reached: 92.3% agreement
Taking biopsy specimens of the colonic mucosa can result in fibrosis and subsequent non-lifting, also associated with malignancy and previous endoscopic resection attempts, making successful endoscopic removal more difficult to achieve.9 Multiple studies have reported that taking biopsy specimens can complicate the removal of colorectal lesions by compromising the submucosal lift from a fluid injection owing to submucosal fibrosis from a post-biopsy scar. A Korean study demonstrated a significantly reduced rate of submucosal elevation in a biopsy group compared with a non-biopsy group (n=42, 77% vs 45%, p=0.03).84 A delay between carrying out biopsies and subsequent endotherapy may also increase the difficulty in achieving successful resection. A 2008 study reported that previous biopsies significantly increased the incidence of the non-lifting sign, especially over 21 days after the biopsy (n=76, OR=16.208, 95% CI 1.024 to 256.442, p=0.048).85 All lesions assessed less than 21 days after biopsy did lift, however, suggesting an attempt at resection should be made as soon as possible after biopsy. These factors suggest that caution is required with biopsy use, especially when malignancy is not suspected and prompt repeat endoscopy cannot be guaranteed.85
Obtaining biopsies of a polyp may not contribute towards obtaining an accurate diagnosis. A 2005 study of 532 polyps asserted that colorectal biopsies were inadequate for grading of colorectal neoplasia, finding that the histopathological diagnosis was underestimated in up to 10% of cases while advanced neoplasia was underestimated in up to 60% of cases.86–88
Although important, histopathological assessment appears less significant in the management of benign polyps than with malignancy, in which the pathological assessment, including depth of invasion (by Haggitt level, Kikuchi level and quantitative measures), differentiation, lymphovascular invasion, tumour budding etc, are all important in the consideration of subsequent management. The GDG considered the major histopathological considerations for LNPCPs as described below (figure 3).
Figure 3.
Major histopathological considerations in the management of large non-pedunculated colorectal polyps (LNPCPs).201
Where malignancy is suspected, careful targeting should be used to improve diagnostic accuracy and minimise submucosal fibrosis in the event of subsequent endotherapy.89
Endoscopic management: pre-procedure
We recommend that adequate planning should be undertaken (including length of time booked for procedure, endoscopist and nursing staff skills and endoscopic equipment) so that before an attempt at advanced polypectomy, the endoscopist has a high level of confidence that complete resection can be achieved in a single procedure (GRADE of evidence: very low; Strength of recommendation: strong).
Consensus reached: 100% agreement
Given the potential complexity of advanced polypectomy, adequate planning is required. In addition to the exclusion of malignancy and potential complications related to endotherapy, an important aim, where possible, is to attempt complete endoscopic resection in a single session.90 The significance of single session completion is reflected by its regular reporting as an important outcome in large volume trials while the ACE study demonstrated significantly lower treatment success with previously attempted lesions (75.4%) than with treatment-naïve lesions (91%) (OR=3.75, 95% CI 1.77 to 7.94, p=0.01).9 31 Key to achieving this aim is ensuring that adequate time is allocated for the procedure, an appropriate endoscopist is selected, optimal assessment has been undertaken (such as within a complex polyp MDM) and that all relevant professionals and equipment are readily available, which may not be the case at the time of detection.35
We recommend that antiplatelet drugs such as clopidogrel and prasugrel, and newer antiplatelet agents such as ticagrelor should be stopped at least 7 days before resection in accordance with BSG Antiplatelet Guidelines (GRADE of evidence: moderate; Strength of recommendation: strong).
Consensus reached: 92.3% agreement
Clopidogrel and prasugrel are classified as thienopyridines and have a different antiplatelet mechanism than aspirin. The BSG, ESGE and American Society of Gastrointestinal Endoscopy (ASGE) advise their cessation based on an increased haemorrhage risk.91–93 A meta-analysis of five observational studies concerning clopidogrel use with polypectomy compared 574 patients who continued clopidogrel therapy before polypectomy with 6169 control patients. A significantly increased risk of delayed post- polypectomy bleeding (RR=4.66, 95% CI 2.37 to 9.17, p<0.00001) was demonstrated.94 This concurred with another study where the incidence of delayed bleeding after polypectomy was over three times higher in the clopidogrel group (n=375, 3.5% vs 1%, p=0.02) but immediate bleeding incidence was similar in both groups.95 Prasugrel and newer antiplatelet agents such as ticagrelor appear to be more potent than clopidogrel and also require cessation. An RCT comparing prasugrel with clopidogrel (n=13 608) found that prasugrel was associated with a significantly higher rate of major bleeding (2.4% vs 1.8%, HR=1.32, 95% CI 1.03 to 1.68; p=0.03).96 Pharmacological studies have shown that clopidogrel, prasugrel and newer agents such as ticagrelor may affect platelet aggregation for up to 7 days and so cessation at around 7 days before LNPCP endotherapy appears appropriate.92 93
-
We recommend that warfarin should be stopped at least 5 days before resection of LNPCPs and the INR should be confirmed as <1.5 before the procedure, in accordance with BSG Anticoagulation Guidelines (GRADE of evidence: moderate; Strength of recommendation: strong).
We suggest that general recommendations about the management of newer anticoagulants which have differing properties, such as rivaroxaban and dabigatran, cannot currently be made owing to a lack of evidence. Appropriate specialist advice should be sought in this situation (GRADE of evidence: very low; Strength of recommendation: weak).
Consensus reached: 92.3% agreement
Cessation of warfarin before endotherapy is advocated by both the BSG and ASGE.91 93 A study of 1657 patients undergoing colonoscopic polypectomy showed that warfarin was strongly associated with PPB (OR=13.37, 95% CI 4.10 to 43.65, p<0.001).97 A single dose of warfarin can be detectable up to 120 h after ingestion and therefore cessation 5 days before endoscopy has been recommended with an INR established as near normal (<1.5).93
Newer anticoagulants such as dabigatran, rivaroxaban and apixaban are being used increasingly instead of warfarin as they do not require regular monitoring. In addition they have a much shorter half-life (dabigatran: 14–17 h, rivaroxaban: 4–9 h) meaning that they may be stopped closer to the time of endoscopy than warfarin. As they are renally excreted, caution is required with their use in the context of renal impairment, especially before endoscopic polypectomy, with earlier cessation likely to be needed to achieve normal patient clotting function.98 In the absence of evidence-based recommendations, obtaining specialist input about the management of these drugs before and after endoscopy is advised.
-
We recommend that patients should consent to the risk of thromboembolic events such as stroke and venous thromboembolism when stopping anticoagulants before endoscopic resection (GRADE of evidence: very low; Strength of recommendation: strong).
We suggest that advice given should be tailored to a patient's individual risk with a ‘bridging regimen’ of low molecular weight heparin given to high-risk individuals in accordance with BSG guidelines. The risk of bleeding versus risk of thromboembolic episode should also be explained (GRADE of evidence: low; Strength of recommendation: weak).
Consensus reached: 85.7% agreement
In certain ‘high-risk’ situations, temporary antithrombotic cessation may not be possible. The risk of embolism in patients with mechanical cardiac valves causing major morbidity, such as peripheral ischaemia, neurological deficit and mortality, is reduced from 4 per 100 patient years to 2.2 per 100 patient years and 1 per 100 patient years with antiplatelet and anticoagulant therapy, respectively.91 Bridging therapy with low molecular weight (LMW) heparin is advocated in this scenario owing to a reduction in risk of major embolism with temporary antithrombotic withdrawal. A prospective study of 224 high-risk patients with LMW heparin bridging therapy reported only two cases of thromboembolism due to warfarin cessation (0.9%, 95% CI 0.2% to 3.2%).99 In patients taking antiplatelet therapy such as clopidogrel for a drug-eluting cardiac stent, withdrawal of this also poses an increased risk of stent occlusion, major embolism and death.91
Endoscopic therapy may be delayed until a safer time is possible for antithrombotic withdrawal, but this may vary on an individual basis.92 In patients taking temporary anticoagulant therapy for venous thromboembolism, endotherapy may need to be delayed until treatment is completed or until antithrombotic therapy has been established for at least 1 month and temporary withdrawal does not appear to pose a significantly increased thromboembolic risk. In the event of permanent anticoagulant therapy (eg, for recurrent venous thromboembolism), bridging therapy will be required and specialist input may also be of use in this case.93 In patients with ‘low-risk’ conditions for thromboembolic events, such as uncomplicated atrial fibrillation or bioprosthetic cardiac valves, the practice of temporary antithrombotic therapy cessation for up to 5 days before endoscopy appears safe. A study of 1024 patients in which warfarin was stopped before endotherapy reported an incidence of thromboembolism of 0.4% if warfarin was stopped for <5 days compared with 2.2% in patients whose warfarin was stopped for >7 days.100 However, individual patient risk should be assessed. An analysis of 987 patients undergoing endoscopic procedures with anticoagulant cessation reported an incidence of stoke of approximately 1%, increasing to 2.93% in the presence of multiple comorbidities (p=0.004–0.04).101
We suggest that where cessation of anticoagulants or antiplatelet medications is contraindicated owing to comorbidity, or where there is uncertainty, appropriate specialist advice should be sought. If the anticoagulation/antiplatelet medication is temporary and the lesion has been adequately assessed as being of low risk for cancer, deferral of resection until after this medication can be discontinued may be appropriate (GRADE of evidence: very low; Strength of recommendation: weak).
Consensus reached: 100% agreement
In complex situations, such as patients requiring advanced polypectomy who have metallic cardiac valves or atrial fibrillation with a cardiomyopathy, cessation of drugs such as warfarin or clopidogrel may be necessary and bridging therapy with aspirin or LMW heparin may be appropriate.93 The timing of medication cessation or change may vary and in these situations cardiology and/or haematology input is appropriate. If antithrombotic drugs are being given for a finite period—for instance, clopidogrel with cardiac drug-eluting stent insertion within 12 months, or warfarin for a recently diagnosed pulmonary embolism, it may be more appropriate to defer endotherapy to a time when antithrombotic therapy has finished or where temporary cessation is less likely to result in complications.91 Evidence to support this view are the results of a study (n=2223) of patients receiving antiplatelet therapy after cardiac stent insertion reporting a HR for stent thrombosis of 89.78 (95% CI 29.90 to 269.60, p<0.001) with premature withdrawal of antiplatelet medication.102 As previously discussed, with reported malignancy transformation rates of 3% a year for lesions >1 cm this approach appears safe.48 The increasing use of newer anticoagulant and antiplatelet drugs may result in an endoscopist being unfamiliar with a particular drug. In this case, or if a problem with antithrombotic medication is anticipated, it should be considered good practice to ensure that appropriate specialist advice has been obtained.91 92
We suggest that the evidence for the cessation/continuation of low-dose aspirin in the context of LNPCPs is weak and the decision should be individualised according to patient risk (GRADE of evidence: low; Strength of recommendation: weak).
Consensus reached: 100% agreement
Conflicting reports about the safety of continuing aspirin before advanced polypectomy have been published. While it appears that many endoscopists stop aspirin before polypectomy, UK and US guidelines advise that it can be continued.91 93 Multiple case–control studies have suggested that aspirin does not increase haemorrhage risk in colonoscopy and polypectomy.92 An example includes a case–control study of 20 636 patients undergoing colonoscopy with polypectomy, which showed no significant difference with aspirin use in bleeding (40%) and non-bleeding groups (33%) (n=20 636, OR=1.41, 95% CI 0.68 to 3.04, p=0.32).103 Another example is a 2008 study demonstrating a similar frequency of PPB in aspirin and control groups (41% vs 39%; n=4592; p=0.80).104 Although specific LNPCP data are limited, a Japanese study examining the risk of bleeding with aspirin with ESD (n=582) showed similar levels of PPB with both aspirin interruption (15.4%) and cessation groups (16.1%), suggesting that aspirin continuation is safe.105 Given conflicting data and opinion, it does appear appropriate to manage aspirin use according to individualised patient risk, such as a scenario that an LNPCP presents a high risk of PPB.
We recommend that when obtaining consent for the endoscopic resection of LNPCPs, written information in plain English should be given. Management options including endoscopic therapy, surgery and conservative management should be discussed. For endoscopic therapy, patients should be informed of the potential need for subsequent check procedures and surveillance endoscopy. The risks of post-procedure bleeding (both immediate and delayed), perforation and residual polyp/recurrence should be explained (GRADE of evidence: very low; Strength of recommendation: strong).
Consensus reached: 92.9% agreement
Consent should adhere to the standards outlined by the Department of Health and the General Medical Council for obtaining valid informed consent.36 106 107 Principles of obtaining valid informed consent for any procedure include that a patient should understand and retain the information given to them, acknowledge the potential ramifications of a treatment and be aware of all management options for a condition, including conservative management.108 Discussions should include the potential risk of complications, the possibility of having malignancy within the polyp despite previous benign histology and radiology, and the benefits of a day-case procedure as opposed to a procedure involving a hospital admission.109 Patients may decide to have no therapeutic management, despite the risk of subsequent malignancy. This may be appropriate in elderly patients or those with comorbidities that reduce life expectancy more imminently than a malignant colonic polyp.110
The most serious complications related to advanced polypectomy procedure such as EMR and ESD are bleeding, perforation and incomplete resection. Reported figures for EMR are far higher than with standard polypectomy where rates of up to 1 in 100 and 1 in 500 have been reported for delayed bleeding and perforation respectively.36 The incidence of perforation with EMR appears to range between 0.5% and 1.3% while severe PPB has been reported in approximately 3–10% of cases in large-volume studies.7 9 31 111 Information pertaining to the risk of serious complications and alternative treatment may be given in a written form and this practice appears to be in place across various centres.108 Early recurrence with the need for additional treatment is also a prominent concern with the use of piecemeal endotherapy, with a 2014 meta-analysis examining piecemeal endoscopic resection suggesting that early recurrence occurs in up to 20% of cases.112 It would be appropriate to advise patients that early recurrence does not represent treatment failure as lesion clearance has been achieved in the vast majority of cases with follow-up endotherapy in almost all reported studies.9 31 112 The potential for late recurrence after 12 months, which may suggest treatment failure, should also be mentioned. Recent estimates from studies with large follow-up numbers after 12 months suggest a figure of between 4% and 7%.29 111 Data from LNPCP management within the BCSP (n=436) 5 mm reported 6% recurrence at 12 months, similar to figures of up to 6.9% which have been reported in other case series (figures 4–9).7 29
Figure 4.
Suggested management algorithm after large non-pedunculated colorectal polyps (LNPCP) identification. Box A (see figure 5); box B (see figure 6); box C (see figure 7). MDM, multidisciplinary meeting; MDT, multidisciplinary team.
Figure 5.
Box A: Lesion assessment. NBI, narrow band imaging.
Figure 6.
Box B: Definitions. NBI, narrow band imaging; NPCP, non-pedunculated colorectal polyps; SMSA, size, morphology, size, access.
Figure 7.
Box C: Endoscopic management. APC, argon plasma coagulation.
Figure 8.
Key performance indicators (KPIs) for the management of large non-pedunculated colorectal polyps (LNPCPs). EMR, endoscopic mucosal resection; ESD, endoscopic submucosal dissection.
Figure 9.
Research questions and potential studies to improve large non-pedunculated colorectal polyps (LNPCPs) evidence base. MDT, multidisciplinary team.
Endoscopic management: peri-procedure
We recommend that carbon dioxide should be used in preference to air insufflation during colonoscopy to improve patient comfort and safety (GRADE of evidence: high; Strength of recommendation: strong).
Consensus reached: 100% agreement
There is evidence that carbon dioxide (CO2) insufflation improves patient comfort during colonoscopy in comparison with air insufflation, especially with longer procedures such as advanced polypectomy. A trial of 219 patients found that CO2 was associated with significantly reduced pain (p=0.014) and bloating (p<0.001) in comparison with air insufflation as well as increased patient satisfaction (p=0.04).113 Another study showed that CO2 use was associated with significantly reduced post-procedure admission after endoscopic polypectomy (OR=0.39, 95% CI 0.16 to 0.95, p=0.04).114 CO2 insufflation has also been associated with increased patient safety. As CO2 is non-inflammable, the risk of combustibility with the use of diathermy and APC (both of which are important components of advanced polypectomy) is eliminated as oxygen is required for an explosion.115 The improved experience for the patient may allow longer procedure times, previously limited by patient discomfort, enabling single session resection.
We recommend that the use of contrast agents such as indigo carmine or methylene blue in the submucosal injection solution is considered to help demarcate a lesion, its resection margins, and to outline a clear submucosal plane (GRADE of evidence: low; Strength of recommendation: strong).
Consensus reached: 100% agreement
The use of contrast agents, to enhance lesion demarcation and a visualisation of post-resection margins in order to aid ascertainment of complete resection is well established.35 46 50 114 116–118 This appears especially relevant with serrated lesions where the resection margins appear more difficult to delineate. For example, almost half of serrated lesions where complete resection was considered to have occurred were found to have residual tissue in the CARE study.39
A study of 445 patients described how the use of indigo carmine facilitated the recognition of deeper planes of resection and identification of tissue deep to the submucosa, enabling the identification and management of all cases of post-resection perforation at the earliest opportunity.118
An association between methylene blue and potential DNA damage to colonocytes, reported in laboratory-based work, has no supporting clinical evidence.119
We suggest that the addition of low-concentration adrenaline to the submucosal injection solution may be considered to keep the resection field clear during endoscopic resection (GRADE of evidence: low; Strength of recommendation: weak).
Consensus reached: 100% agreement
The use of adrenaline in submucosal injection solutions has been advocated to reduce the risk of immediate PPB. A 2001 study demonstrated reduced immediate PPB with a 1:10 000 adrenaline-containing solution compared with saline (1/75 vs 7/76, p=0.03) and a 2004 study also reported the same result (1/50 vs 8/50; p<0.05). No improvement has been demonstrated with delayed PPB.120 121 A 2007 Korean study showed that this finding applied to LNPCPs (1/75 vs 7/76, p=0.03).92 122 Although the above evidence cites the use of a1:10 000 concentration of adrenaline, the consensus of the GDG is that concentrations less than 1:100 000 are also effective and potentially decrease the risk of cardiovascular ischaemia where large injection volumes are required.
We suggest consideration of the use of colloidal-type submucosal injection solutions in preference to normal saline lifting solution for LNPCPs (GRADE of evidence: low; Strength of recommendation: weak).
Consensus reached: 92.3% agreement
Available evidence suggests colloidal-type solutions to be optimal for submucosal injection in view of technical and safety factors, with findings of a longer lasting lift facilitating easier resection than saline.32 123 Animal model studies show that colloidal-type solutions such as succinylated gelatin (gelofusin) enable a longer lasting lift (mean of 36 min) and increased en bloc resection.124 125
Sodium hyaluronate commonly used for ESD, has also demonstrated superiority over normal saline in porcine models.126 A 2004 Japanese study group found that sodium hyaluronate produced a longer lasting lift than both normal saline and hypertonic solutions, with later reports citing reduced tissue injury.127 128
These findings appear to have been replicated in human studies. In a 2005 study (n=223), glycerol demonstrated a higher en bloc resection rate (63.6% vs 48.9%, p<0.05) and complete resection rate (45.5% vs 24.6%, p<0.01) than saline.129 A double blind RCT reported gelofusin solution (GS) as better than saline with significantly reduced procedure time (GS: 12.0 min (IQR=8.0–28.0) vs normal saline: 24.5 min (IQR=15.0–36.0), p=0.006) and reduced the number of piecemeal resections made (GS; resections=3.0 (1.0–6.0) vs normal saline; resections=5.5 (3.0–10.0), p=0.028).125 130
We suggest that endoscopists should be familiar with the range of snares available, although a single optimal snare cannot be recommended (GRADE of evidence: very low; Strength of recommendation: weak).
Consensus reached: 100% agreement
There is no evidence available to suggest an optimal snare for use in advanced polypectomy. Various sizes, shapes and textures of snares are available. Larger snares (>2 cm) are preferred by some operators with the intention of en bloc resection or wide-field resection of larger polyps, though operators should be aware that this carries a potentially increased risk of perforation if a large volume and depth of tissue is within the snare. Smaller, thinner (monofilament) snares are often preferred where increased precision is required and spiral or stiffer snares are often used by some operators where gripping of a flat elevated lesion is thought to be optimised.131 Reports favouring the use of a particular snare such as spiral or crescentic snares appear subjective, limiting the ability to recommend a particular snare.46 132
We suggest that a prolonged pure coagulation current should be avoided owing to an increased risk of delayed post-polypectomy bleeding and thermal tissue injury (GRADE of evidence: low; Strength of recommendation: weak).
Consensus reached: 92.3% agreement
The choice of polypectomy diathermy settings seems to vary extensively. A North American survey of endoscopic practice in 2004 (n=198) found that blended current (46%) and coagulation current (46%) were more commonly used, with lower reported use of varied (4%) and pure cutting current (3%).133 A 2013 Israeli survey (n=100) found that 42% used a pure coagulation current, with 38% using blended current and 20% using pure cutting current.134 A pure cutting current is likely to be related to higher rates of immediate PPB owing to poor haemostasis properties, and the avoidance of its use in endoscopic polypectomy has been advocated by some groups including the ESGE.92 135 A cutting current does have good incisional properties, however, enabling high-quality resection specimens and inducing less thermal tissue injury.136 137 A 1992 study comparing the use of coagulation current with a form of cutting current in snare polypectomy (n=1485) found the latter to be associated with immediate haemorrhage (p=0.03).138 A subsequent multicentre study (n=5152) identified pure cutting current as one of the greatest risk factors for immediate PPB (OR=6.95, 95% CI 4.42 to 10.04).92 139
Pure coagulation current appears to be commonly used and has good haemostasis properties. However, higher settings and prolonged use induce higher levels of thermal tissue injury. Porcine models have demonstrated a greater depth of tissue injury with coagulation current than both blended (p=0.0157) and pure cutting current, (p=0.0461).136 The increased risk of tissue injury is of particular concern in the thinner right colon, which is more susceptible to diathermy-induced perforation.35 137 140 141 The use of blended current or automated current that regulates coagulation and cutting current (such as Endocut) has been advocated as a safer diathermy option with the rationale that they provide adequate incision properties combined with effective haemostasis. A trial comparing blended and microprocessor-controlled automated current (n=148) found that automated current produced less tissue damage than blended current with a conventional electrosurgical generator (p<0.02) while also producing higher-quality resection specimens (p=0.024) allowing for more accurate histological evaluation (p=0.046).142 These findings suggest that the rationale for the use of an automated current appears sound.92 136
We suggest that while en bloc endoscopic snare resection of lesions <20 mm is recommended to reduce the risk of recurrence and to enable more accurate histopathological interpretation, this practice should be used with caution in LNPCPs owing to an increased risk of diathermy-associated thermal injury and perforation (GRADE of evidence: low; Strength of recommendation: weak).
Consensus reached: 84.6% agreement
En bloc snare resection is desirable owing to reduced recurrence and the ability to obtain more accurate histological analysis. In addition, a study by Kim et al24 suggests that the risk of incomplete resection in piecemeal resection is significantly higher with lesions >30 mm (n=497, OR=2.688, 95% CI 1.036 to 6.993, p=0.042).
A 2014 meta-analysis of 33 studies examining snare removal of NPCPs unequivocally demonstrates lower recurrence with en bloc resection than with piecemeal removal (3% (95% CI 2% to 5%) vs 20% (95% CI 16% to 25%), p<0.0001).112
Although it is possible to remove some LNPCPs en bloc with snare resection, it may be technically difficult to achieve owing to reduced snare stiffness while uncertainty about the resection plane may lead to concerns about perforation due to lack of control of tissue volume and from thermal injury due to an inability to control the cutting plane.143 144 A 2012 Korean study also showed that where EMR was carried out for NPCPs >30 mm, the likelihood of using piecemeal resection increased significantly for technical reasons (OR=7.246, 95% CI 4.672 to 11.235, p<0.001).24 Where en bloc specimen retrieval is required, such as with suspected malignancy, techniques such as ESD and surgery may be required. However, for benign lesions, piecemeal EMR has been shown to have comparable efficacy, especially when allowing for repeat treatment of recurrence, and with less morbidity. The high complete eradication rates reported by various studies such as 90% by Buchner et al31 and 96% quoted by Longcroft-Wheaton et al, 29 including 87.5% of SMSA level 4 lesions support this. In addition, while a 2009 study reported lower rates of early recurrence with en bloc TEMS for rectal lesions in comparison with pEMR, it should be noted that late recurrence was similar in both groups when allowing for repeat endoscopic therapy (TEMS: 9.6% vs EMR: 13.8%, p=0.386).58
We recommend that treatment-naïve lesions which fail to lift after adequate submucosal injection should not be subject to attempted resection with conventional snare polypectomy technique (GRADE of evidence: low; Strength of recommendation: strong).
Consensus reached: 92.3% agreement
Uno et al first described an association between non-lifting lesions in response to a submucosal injection and malignancy in 1994.145 All cases defined as non-lifting were found to contain malignancy. A 1999 Japanese study also demonstrated an association between the non-lifting sign and deep submucosal invasion (n=60). All lesions with deep submucosal invasion (sm3), lesions associated with a higher rate of lymph node metastases and so requiring surgery, displayed the non-lifting sign, whereas 93.5% of lesions with more superficial submucosal invasion (sm1) were successfully lifted.21 A later study repeated these findings with only 20% of sm3 lesions lifting as opposed to 82.4% of sm1 lesions (p<0.05),85 and a 2007 study reported that the non-lifting sign displayed an accuracy rate of 94.8% (n=271, p<0.05).146 Correlation between the non-lifting sign in treatment-naïve lesions and deep submucosa invasion with lymph node involvement appears strong.40 85 In view of this, while en bloc removal may be possible, the mucosectomy action of snare polypectomy is less likely to be effective in treatment-naïve non-lifting lesions owing to irregularity of the submucosal plane.18
We recommend that during endoscopic piecemeal resection, the snare should be used to resect a lesion completely wherever possible. Thermal coagulation techniques such as APC and soft coagulation may be used as adjuncts when snare resection of small residual fragments of polyp is not possible (GRADE of evidence: low; Strength of recommendation: strong).
Consensus reached: 100% agreement
APC is a non-contact method of thermal coagulation considered safe for use in therapeutic endoscopy. The use of APC as an adjunct to endoscopic snare resection has been supported by various studies. Zlatanic et al147 reported a 50% reduction of residual adenoma on follow-up endoscopy compared with no APC use (n=77, 100% reduced to 50%). A 2003 study also demonstrated successful endoscopic clearance with the additional APC use in 90% of lesions with incomplete endoscopic snare resection (n=77).148 A larger study in 2011 commented that the use of APC on visible residual adenoma after piecemeal polypectomy did not reduce lesion recurrence (n=105; OR=0.46, p=0.29). This finding may be due to the application of APC to larger areas of tissue but also highlights the fact that APC should not be relied upon as the sole treatment of residual adenoma.149 150
A 2002 study examining APC use on post-resection margins reported a reduced rate of adenoma recurrence after piecemeal EMR in lesions where complete resection was thought to have been achieved (1/10 APC, 7/11 no APC; p=0.02). This effect might be due to the treatment of microscopic residual foci at the resection margins not visible to the endoscopist.151
Thermal coagulation may also be provided by the use of soft coagulation from diathermy applied to tissue via the snare tip. However, no data to definitively support its use have yet been obtained.
The use of hot biopsy avulsion has also been described as an ablative technique for flat polyp tissue considered unsnarable, with a small 2014 study (n=20) reporting no residual tissue on surveillance in 85% of cases.152
We recommend careful post-procedure inspection of the resection site and photographic documentation of completeness of resection (GRADE of evidence: low; Strength of recommendation: strong).
Consensus reached: 100% agreement
Imaging of a resection site is important to document and confirm whether complete resection has taken place, and also to confirm exclusion of a perforation. Taking steps to assess for complete resection appears important as incomplete resection seems to be far more prevalent than first thought, even among experienced endoscopists. The CARE study demonstrated increasing rates of incomplete resection with larger lesions. Of 10–20 mm lesions felt to be completely resected at endoscopy, 23.3% were found to be incompletely resected, despite the endoscopist considering complete resection to have taken place, significantly higher than with smaller lesions (17.3% vs 6.8%, p=0.003).39 A 2014 study demonstrated histological evidence of recurrence in 7% of LNPCPs where complete resection was felt to have occurred both on initial resection and follow-up (n=252).170
The ASGE recommend photo documentation in the area of a tattoo post-endoscopic resection as it may enable identification of a scar site where no residual tissue is present.153 154
We recommend that with the exception of the rectum or caecum, a tattoo should be applied in accordance with local policy to aid endoscopic follow-up or subsequent surgical resection. As tattooing may cause submucosal fibrosis, the tattoo should be placed at least 30 mm from the lesion (GRADE of evidence: very low; Strength of recommendation: strong).
Consensus reached: 92.3% agreement
The use of tattoo application with an indelible marker such as Indian ink has been highlighted as an important practice in post-endoscopic removal to enable identification of the resection site on follow-up and lesion identification in cases requiring surgical resection. Caution has been advised with the tattoo practice to avoid complicating endoscopic resection.155 156 Various case series have reported sublesional submucosal fibrosis resulting from tattoo application, thus compromising subsequent endoscopic resection by both EMR and ESD.157 158 In view of this, a tattoo should be placed away from a lesion, with a distance of at least 3 cm recommended in one case series.157
Endoscopic management: post-procedure
We recommend that written information about the risk of post-procedure complications (including bleeding risk for up to 2 weeks), together with recommended actions and an emergency phone number should be provided to patients (GRADE of evidence: very low; Strength of recommendation: strong).
Consensus reached: 100% agreement
Patients with serious complications related to polypectomy, such as haemorrhage and perforation, may not present with symptoms until several days after the procedure. An analysis of PPB from 14 575 cases reported a mean presentation time of 5 days after the procedure, with cases occurring up to 17 days after polypectomy, while there have been reports of PPB occurring up to 30 days after the procedure.159 160 An analysis of post-colonoscopy perforations found that 24% of cases presented over 48 h after colonoscopy, with 9% presenting after over 15 days.161 In view of this, the provision of a clear post-procedure plan is important and may expedite appropriate management and improve patient safety.
We suggest that recommencement of anticoagulant and antiplatelet therapy after polypectomy should be considered on an individual basis, weighing up the risks of post-procedure bleeding with the risks of a thromboembolic event. Further specialist advice (ideally sought before the procedure) may be appropriate (GRADE of evidence: low; Strength of recommendation: weak).
Consensus reached: 100% agreement
While it is ideal that these drugs are restarted as soon as possible after endotherapy to reduce thromboembolism risk, the lack of evidence for restarting antiplatelet agents after advanced polypectomy is recognised, while conflicting evidence exists about the safety of restarting anticoagulants such as warfarin.91 A study of 94 patients restarting warfarin on the same day of endotherapy reported a delayed bleeding rate of <1%.162 However, a later study of 173 patients where warfarin was restarted within 7 days after endotherapy had an increased risk of bleeding (OR=5.2, 95% CI 2.2 to 12.5, p<0.001)).104 Events during endotherapy such as peri-procedural bleeding, in addition to patient factors, may also raise concerns about significant post-polypectomy haemorrhage. In light of this, management, should be considered on an individual basis along with specialist input.91 93
We recommend that in the case of piecemeal EMR, initial follow-up should take place within 2–6 months (GRADE of evidence: low; Strength of recommendation: strong).
Consensus reached: 92.9% agreement
Early follow-up endoscopy after piecemeal resection is advocated owing to potentially high rates of incomplete resection and early lesion recurrence, with histological evaluation often unable to assess for completeness of resection as with en bloc removal.163 Recurrence levels appear to increase the longer the period of time left before follow-up after the initial treatment and early intervention of recurrent/residual tissue allows for prompt eradication.164 A Japanese study reported recurrence rates of 18.4%, 23.1% and 30.7% with follow-up at 6, 12 and 24 months, respectively, while a US study reported a similar trend with recurrence almost three times higher after 24 months than at 12 months.60 165 Initial follow-up at a later point such as 6 months also appears safe with similar levels of recurrence between 3 and 6 months. However, follow-up may also be appropriate sooner—for example, if malignancy or high-grade dysplasia on histology is found.53 60 Follow-up within 6 months is in line with the position of the US Multi-Society Task Force for Colorectal Cancer and the American Cancer Society who recommend that lesions removed piecemeal should be considered for follow-up endoscopy between 2 and 6 month intervals until complete excision.166 After lesion-specific follow-up has established clearance, further endoscopic surveillance should be consistent with existing BSG polyp surveillance guidelines.167
We recommend that on follow-up, the scar site should be positively identified, scrutinised and photographed. Image enhancement with techniques such as dye spray and digital enhancement may aid detection of residual neoplasia on a polypectomy scar. Areas of possible residual polyp require tissue diagnosis and definitive treatment (GRADE of evidence: low; Strength of recommendation: strong).
Consensus reached: 84.6% agreement
Evidence suggests that incomplete resection occurs at a higher rate than previously thought. The CARE study found a high incidence of incomplete resection (10.1%) in cases where complete resection was considered to have been achieved, including incomplete resection in almost half (47.6%) of sessile serrated adenomas, with a wide variation of incomplete polyp resection between endoscopists.39 Further justification for careful analysis of the scar site on follow-up endoscopy are reports of ‘late recurrence’ in an area where complete resection was believed to have occurred, first described in a 1992 series where almost half of the cases of recurrence occurred where no recurrence had been identified on earlier examination.168 A 2009 study reported late recurrence of residual adenoma in 4.4% of cases at 12 months’ follow-up (n=138). In 7.2% of cases, evidence of residual adenoma was present in biopsy specimens from scar sites where no visible adenoma was seen. Negative biopsy results at early follow-up appeared to be predictive of continued eradication on late follow-up in 97.9% of cases in comparison with the remaining lesions (RR=0.15, 95% CI 0.035 to 0.618, p=0.005).169 The practice of biopsy retrieval as part of a follow-up resection site examination was supported by an analysis of 252 LNPCPs in which biopsy evidence of residual/recurrent adenoma was found in 7% of cases where no visible adenoma was present, while late recurrence was seen in 10.47% of cases where no adenoma was identified at initial follow-up.170 Taking biopsy specimens from an apparently clear polypectomy scar site appears justified as it may identify residual tissue that might otherwise remain undetected.
Image enhancement may also improve diagnostic accuracy.171 Magnification endoscopy seems to accurately identify residual tissue. A study of 77 LNPCPs reported similar accuracy to histological evaluation. The sensitivity of magnification endoscopy for predicting residual tissue at resection margins was 98% (95% CI 90% to 100%); specificity was 90% (95% CI 79% to 100%) with an overall accuracy of 94.5% (95% CI 87.2% to 98.6%).172 Chromoendoscopy was found to accurately predict completeness of endoscopic resection in a 2004 study of 684 lesions (sensitivity 80%; specificity 97%; accuracy 94%).173 A 2011 study comparing the accuracy of NBI with white light examination for the detection of residual neoplasia found that NBI increased detection of residual neoplasia at the resection site, with 63% of identified lesions found to be more extensive with NBI than initially thought with white light examination.174
-
We suggest that the management of residual/recurrent polyp tissue can be challenging and should be performed by an endoscopist with complex NPCP experience (GRADE: low; Strength of recommendation: weak).
We suggest that the management of ongoing recurrence should be discussed in a complex polyp MDM (GRADE of evidence: low; Strength of recommendation: weak).
Consensus reached: 100% agreement
While a proportion of recurrent/residual polyp tissue can be successfully treated with repeat snare resection, complete eradication at repeat therapy may be much more difficult to achieve, such as with larger areas of recurrence. Repeat treatment with EMR may not be achievable owing to submucosal fibrosis.32
ESD appears to be a less invasive management option in cases of complex recurrence, with various reports of its efficacy in scar-embedded polyps and subsequent avoidance of surgical resection. A 2009 study reported successful clearance of lesion recurrence with ESD in 15 cases after failed EMR.175 A Japanese study reported curative resection with ESD in large areas of recurrence (>2 cm) where EMR was not possible,176 while a UK study also reported successful salvage ESD in 11 of 12 cases.177 However, it should be noted that these studies are small and ESD availability in the West remains limited. Surgical resection remains an effective treatment while conservative management appears appropriate if patient comorbidities suggest that this will not significantly alter life expectancy. Various factors such as a patient's wishes and comorbidity and availability of treatment modalities may affect management, and access to a multidisciplinary network may optimise management.42 117 178
Surgical Management of LNPCPs
We recommend that surgical therapy should be considered where malignancy is suspected or concerns about the likelihood of incomplete endoscopic resection arise after complex polyp MDM discussion (GRADE of evidence: moderate; Strength of recommendation: strong).
Consensus reached: 92.9% agreement
While expert endoscopic management is the preferred first-line management in LNPCPs due to superior patient safety, surgical resection still has an important role.44 45 Surgery (or ESD where expertise is available) may be preferred for lesions that have a higher risk of malignancy, or where there is a high risk of residual polyp after endoscopic resection. Although morbidity and mortality rates are higher with surgical resection, the results of complete resection are better and there is a reduced need for endoscopic follow-up.34
Surgical resection is also an effective option where recurrence cannot be managed endoscopically.7 9 53 111 Even with the most advanced polypectomy techniques such as ESD, deeper submucosal invasion cannot be managed, with surgery often required when it is found at endoscopy. Surgery offers the highest chance of oncologically complete resection for these malignant lesions. ESD compares less favourably in this situation, with a large case series (n=1111) featuring both benign and malignant lesions reporting an en bloc resection rate of 88% and curative rate of 89%.179 180 Surgical resection is the only treatment where deep submucosal infiltration and lymph node infiltration may be managed effectively. Reported curative rates for surgical resection are 100% for stage 1 disease with a rate >91% for stage IIIa disease, indicating its efficacy.181
A proportion of cases with malignancy are found after surgery in lesions previously thought to be benign, although this varies considerably depending on patient selection and operator expertise at assessment. Studies analysing histopathology post-surgical resection in polyps considered benign have an estimated invasive malignancy in up to 22% of lesions.45 109 In view of this, where there is diagnostic uncertainty at assessment, surgical resection may be an appropriate management option.2 18 109
In addition, in cases where endoscopic access is considered difficult by an expert advanced endoscopist, with concern about causing complications or achieving a successful resection, surgical therapy may provide a more effective primary option rather than as an additional invasive procedure as secondary treatment.182
Rectal lesions require special consideration due to the complexity and morbidity associated with both open and laparoscopic resectional surgery in this area and the availability of endotherapy and minimally invasive local resectional surgery such as TEMS. A 1998 study reported (n=591) patients with 3.2% mortality and 30% postoperative morbidity at 30 days with open proctectomy,183 while a 1999 study (n=681) cited a 0.6% perioperative mortality and 22% postoperative morbidity.184 A 2010 laparoscopic low anterior resection series (n=132) reported similar morbidity (20.5%).185 In addition, with low rectal lesions where non-sphincter saving surgery such as an abdominoperineal resection is often used, a permanent stoma will be required.186
Where suspicion about malignancy exists and en bloc resection is considered desirable to ensure adequate histological analysis, the use of either ESD or minimally invasive local resectional surgery such as TEMS is preferable to conventional resectional surgery; however, ESD availability, as previously discussed, is currently limited. A 2014 meta-analysis of 111 ESD and 10 TEMS series (n=2077) comparing LNPCP management outcomes found en bloc resection to be higher with TEMS (TEMS: 98.7% (95% CI 97.4% to 99.3%) vs ESD: 87.8% (95% CI 84.3% to 90.6%), p<0.001) while the curative resection rate was also superior (TEMS: 88.5% (95% CI 85.9% to 90.6%) vs ESD: 74.6% (95% CI 70.4% to 78.4%), p<0.001).187 A 2010 meta-analysis of TEMS also demonstrated a significantly reduced postoperative complication rate compared with resectional surgery (n=629, OR=0.16 (95% CI 0.06 to 0.38), p<0.003), while a 2012 study showed significantly reduced morbidity (n=78, 14.6% (TEMS) vs 37.1% (resectional surgery), p=0.046).33 188
The available evidence suggests that pEMR is preferable to TEMS for the management of benign rectal NPCPs. A retrospective comparison of TEMS and piecemeal EMR for the management of large rectal NPCPs (n=292) found that while early recurrence rates were lower in TEMS (10.2% vs 31.0%, p<0.001) when allowing for repeat endoscopic therapy on follow-up, late recurrence after 12 months was similar (9.6% vs 13.8%, p=0.386). TEMS was also associated with greater morbidity (postoperative complications: 24% (TEMS) vs 13% (EMR), p=0.038) and a longer hospital stay (median hospitalisation after the procedure: 3 days (TEMS) vs 0 days (EMR), p<0.001).58 Another consideration is evidence that pEMR appears to be more cost-effective. International cost analysis suggests that the cost of EMR is around $2000, with subsequent follow-up roughly half this figure. In comparison, the cost of TEMS is estimated to be around $7800.29 44 189 However, TEMS may be indicated as first-line treatment for selected benign rectal NPCPs that occupy significant rectal circumference and are technically difficult to remove with snare retrieval owing to its soft texture and the risk of significant bleeding.190 191
Equivalent curative efficacy, reduced morbidity and reduced associated cost justify the preference for pEMR in the management of most benign rectal NPCPs. However, optimal management of complex rectal NPCPs appears ideally suited to complex polyp MDM discussion where all available modalities are potentially available.
We recommend that laparoscopic therapy should be used in preference to open surgery in the surgical management of LNPCPs (GRADE of evidence: high; Strength of recommendation: strong).
Consensus reached: 92.9% agreement
Laparoscopic surgery (LS) has largely replaced open surgical resection (OS) for the removal of LNPCPs where endoscopic resection is deemed unsuitable.192 A meta-analysis demonstrated comparable therapeutic efficacy with similar 3-year recurrence rates, including in the management of colorectal cancer. While being less invasive, LS is considered oncologically safe. This is particularly relevant when a malignancy is subsequently found (tumour recurrence at 3 years for LS: 16% vs OS: 18%; 95% CI 0.63% to 1.17%; p=0.32).193–195 A 10-year UK analysis (n=192 620, 3709 laparoscopic procedures) reported that laparoscopic surgery was associated with a reduced 30-day (OR=0.57; 95% CI 0.44 to 0.74; p<0.001) and 365-day mortality (OR=0.53; 95% CI 0.42 to 0.67; p<0.001).196 While mortality and postoperative complications appears similar between open and laparoscopic groups, LS has a clear association with accelerated postoperative recovery with reduced pain and the earlier return of bowel function (Salimath et al, n=261; OS: 4.4 days; 95% CI 4.2 to 4.6 vs LS: 3.7 days; 95% CI 3.5 to 3.9; p<0.001) and reduced hospital stay (Vlug et al, n=427; p<0.001 and Salimath et al; OS: 8.01 days; 95% CI 7.1 to 8.9 vs 4.38 days; 95% CI 4.0 to 4.8; p<0.001).194 197
Key performance indicators for the management of LNPCPs
Ensuring high quality LNPCP management is essential to reduce complications and future cancer risk. The development of KPIs, with defined minimum and aspirational standards, allows a standardised way of monitoring and auditing clinical quality outcomes.
Widely varying outcomes have been reported in colonic polyp management, particularly with larger lesions and even between experienced endoscopists.39 There is evidence of varying management of LNPCPs between different UK centres, which has resulted in outcomes that may be considered suboptimal.7 Recent data from the BCSP (n=557) demonstrated high levels of piecemeal endoscopic management of malignant polyps where secondary surgical management was subsequently required (16.1%), while almost 80% of NPCPs managed with primary surgery were benign.7 Given evidence of the efficacy of endotherapy, including for the most complex polyps, it seems likely that many of these lesions might have been managed effectively endoscopically, with lower associated morbidity and mortality risks and reduced cost.29
The application and monitoring of KPIs, not previously described in LNPCP management, should help to improve quality by identifying potentially suboptimal performance at an earlier stage, reducing patient risk and permitting support and remedial action to be taken. The use of KPIs is intended as a monitoring system with outliers warranting further investigation/analysis. For example, if an outcome can be clearly explained, such as a skilled endoscopist tackling more complex lesions having a higher recurrence/residual rate than a less skilled endoscopist tackling more simple lesions, then this may be acceptable. If, however, endoscopists tackling similarly complex lesions have widely differing outcomes with one endoscopist producing clearly inferior outcomes, the KPI may then call into question the appropriateness for that individual to be managing certain lesions.
Various domains for evaluating LNPCP management performance were considered by the writing committee.
Potential KPIs within each domain were subsequently formulated. A preliminary round with anonymous voting was then used to assess the suitability of 16 potential KPIs. Seven KPIs were identified within the agreed domains and voted on in accordance with the guideline development process (see table 6).
Table 6.
A comparison of outcomes from trials of endoscopic management of LNPCPs
| Moss et al9 | BCSP7 | Buchner et al31 | Longcroft- Wheaton et al29 | |
|---|---|---|---|---|
| Number of LNCPCs | 479 | 436 | 308 | 187 |
| Mean size (mm) | 35.6 | 29.5 | 23 | 41.5 |
| Cases with complete resection considered achieved after single session (%) | 89.2 | NA | 91 | 90% |
| Malignancy in resection specimen (%) | 6.9 | 6 | 4.4 | 5.9 |
| Need for surgery (%) | 16.3 | 16.1 | 10 | 9 |
| 3-Month recurrence (%) | 20.4 | 16.5 | 27 | 14.5 |
| 12-Month recurrence (%) | 2 | 6 | 16.3 | 3.9 |
| Delayed bleeding | 2.9 | 3 | 7.2 | 2.7 |
| Perforation | 1.3 | 0.5 | 0.4 | 0.45 |
BCSP, Bowel Cancer Screening Programme; LNPCPs, large non-pedunculated colorectal polyps.
In setting quantitative minimum and aspirational standards, where available, the results from various national and international studies felt to be of suitable design and quality were reviewed as possible reference points. Where it was not felt possible to set a defined standard for a KPI, the phrase “no current standard defined” was used with a view to monitoring outcomes and identifying acceptable minimum standards in the future. As with the guideline statements, LNPCPs refer to non-pedunculated colorectal polyps at least 20 mm in size (tables 7 and 8).
Table 7.
Summary of domains and KPIs for LNPCPs
| Domain | Proposed KPIs |
|---|---|
| Optimal decision-making | Surgery rate for LNPCPs |
| Endoscopic skill | Recurrence/residual polyp at 12 months in endoscopically managed LNPCPs |
| Safety | ▸ Perforation rate ▸ Post-procedure bleeding rate |
| Timeliness | ▸ Time from diagnosis to referral for definitive therapy ▸ Time from referral to definitive therapy |
| Volume of procedures | ▸ Number of procedure per endoscopist per year |
KPIs, key performance indicators; LNPCPs, large non-pedunculated colorectal polyps.
Table 8.
Optimal decision-making domain summary
| Domain | Optimal decision-making |
|---|---|
| Objective | Assessment of the appropriateness of decision-making in the management of LNPCPs |
| KPI | Surgery rate for LNPCPs |
| Denominator | Include all patients with NPCPs, including lesions that prove to be cancers Exclude patients with LNPCPs undergoing primary surgery for cancer (where no endoscopic resection has been attempted). Do not exclude patients with cancers who undergo endoscopic therapy |
| Numerator | Patients with LNPCPs undergoing surgery for that lesion |
| Analysis | Calculate at patient level (not LNPCP level) Report as percentage (proportion of patients) |
| Frequency | Calculate annually |
| Level of analysis | Service level |
| Minimum standard | No current standard defined |
| Aspirational standard | No current standard defined |
| Action | Qualitative review of each case |
| Evidence | Swan et al44 Longcroft-Wheaton et al29 Bertelson et al45 Lee et al7 Moss et al9 Buchner et al31 |
KPI, key performance indicators; LNPCPs, large non-pedunculated colorectal polyps.
Key performance indicators and standards for the management of LNPCPs
In view of the increased cost, morbidity and mortality with surgical management, endoscopic removal is considered first-line management for LNPCPs, as detailed earlier in these guidelines.9 29 44 45 There is, however, wide variation in the UK between difference centres for management of LNPCPs with regards to cases managed primarily with endotherapy and surgery.7 The GDG agreed that evaluation of the proportion of patients with LNPCPs managed surgically (excluding primary surgical management for cancer) would identify:
patients with benign LNPCPs undergoing surgery where endoscopic management might have been possible;
patients with LNPCPs which ultimately proved to be cancers where primary endoscopic therapy was attempted, but who later required surgery.
Although the GDG recognised that both these situations may occur even in expert hands, it was felt that determining and monitoring this proportion would provide useful additional information on decision-making in the management of LNPCPs. The GDG felt that this KPI could be analysed at both an individual and overall service level:
Level of agreement for KPI: 91.7%
Level of agreement for standard: 92%
The presence of recurrence and/residual tissue is a marker for assessing success of endotherapy in keeping with international literature reporting outcomes of LNPCP management (table 9). The GDG considered the measurement of 12-month (late) outcomes to be more appropriate than measurement of 3-month (early) outcomes, as the former relates more directly to health outcomes and is consistent with the standardised use of 12 months as an outcome of treatment success internationally. In addition, 12-month surveillance is commonly undertaken with lesions removed both en bloc and piecemeal. Residual polyp tissue seen on early endoscopic follow-up may be treated on repeat endoscopy giving eradication rates comparable to en bloc resection techniques.9 58 113 In addition, analysis at 12 months allows measurement of ‘late recurrence’ which has been reported in cases where no recurrence was found at initial endoscopic follow-up in multiple studies.169 170
Level of agreement for KPI: 100%
Level of agreement for standard: 100%
Table 9.
Endoscopic skill domain summary
| Domain | Endoscopic skill |
|---|---|
| Objective | Assessment of endotherapy success |
| KPI | Recurrence/residual polyp at 12 months in endoscopically managed LNPCPs |
| Denominator | Include all patients undergoing 12-month surveillance after resection of LNPCPs. Recurrent/residual polyp identified and cleared before 12-month surveillance does not warrant inclusion within this parameter. As not all surveillance will occur on schedule, allow up to 15 months for surveillance to occur. Only count the first surveillance at or after 12 months. Exclude patients who do not have surveillance within 12–15 months (owing to surgical resection, death, comorbidity, emigration, etc). NB—monitor this figure to ensure surveillance recall process is robust |
| Numerator | Patients undergoing 12–15-month surveillance with endoscopic or histological evidence of polyp recurrence at the site of resected LNPCP |
| Analysis | Calculate at LNPCP level Report as percentage (proportion of LNPCPs with recurrence on 12-month surveillance) |
| Frequency | Calculate annually |
| Level of analysis | Service and individual colonoscopist level |
| Minimum standard | <10% |
| Aspirational standard | <5% |
| Action | Qualitative review of each case |
| Evidence |
Supporting evidence: Belderbos et al112 Moss et al9 Barendse et al58 Khashab et al169 Knabe et al170 Supporting evidence for quality standard Lee et al7 Longcroft-Wheaton et al29 |
KPI, key performance indicators; LNPCPs, large non-pedunculated colorectal polyps.
It is essential that patients receive high-quality management and the risk of harm is minimised (table 10). Perforation and PPB are more commonly associated with advanced polypectomy than with diagnostic colonoscopy and polypectomy of smaller polyps.198 The reported incidence of perforation after LNPCP resection is 0.5–1.3%, whereas for diagnostic polyps and standard polypectomy the risk or perforation is quoted at 1:1000 and 1:500, respectively.7 9 36 PPB following resection of LNPCPs is the commonest complication with a reported incidence of 2.9–7.2%.9 31 As potentially life-threatening complications that may warrant emergency treatment, perforation and PPB seem appropriate markers of patient safety.
Table 10.
Safety domain summary
| Domain | Safety |
|---|---|
| Objective | To maximise the safety of endoscopic therapy |
| KPI |
|
| Denominator | Include all patients with LNPCPs undergoing endotherapy |
| Numerator | Patients with LNPCPs undergoing endotherapy who present with a perforation (definite or probable) within 30 days of endotherapy |
| Analysis | Calculate at patient level (not at LNPCP level)Report as percentage (proportion of patients) |
| Frequency | Calculate annually. As these are rare events, calculating the rates over longer periods of time may be useful |
| Level of analysis | Service and individual colonoscopist level |
| Minimum standard |
EMR Perforation: <2%; PPB: <5% ESD Perforation and PPB: no current standard defined |
| Aspirational standard |
EMR Perforation: <0.5%; PPB: no current standard defined ESD Perforation and PPB: no current standard defined |
| Action | Qualitative review of each case |
| Evidence |
Supporting evidence—perforation: Rutter et al25 Rabeneck et al26 Nivatongs198 NHS BCSP Publication36 Supporting evidence for standards—perforation: Lee et al7 Moss et al9 Buchner et al31 Longcroft-Wheaton et al29 Supporting evidence—post-procedure bleeding: Metz et al22 Sawhney et al104 NHS BCSP Publication36 Supporting evidence—post-procedure bleeding standards: Lee et al7 Moss et al9 Buchner et al31 |
BCSP, Bowel Cancer Screening Programme; EMR, endoscopic mucosal resection; ESD, endoscopic submucosal dissection; KPI, key performance indicators; LNPCPs, large non-pedunculated colorectal polyps; NHS, National Health Service; PPB, post-procedure bleeding.
ESD practice in the UK is felt to be too limited to set minimum standards while separate NICE guidance covers this modality. Thus, the GDG agreed that both safety KPIs for ESD (perforation and PPB) should be considered as auditable outcomes.
Perforation is defined as: ‘air, bowel contents or instrumentation outside the bowel lumen’36 199
PPB is defined as: rectal bleeding within 30 days of the procedure resulting in any of the following:
- Minor
- procedure aborted
- unplanned post-procedure medical consultation
- unplanned hospital admission, or prolongation of hospital stay, for ≤3 nights
- Intermediate
- haemoglobin drop of ≥2g
- transfusion
- unplanned admission or prolongation for 4–10 nights
- intensive therapy unit admission for 1 night
- interventional procedure (endoscopic or radiological)
- Major
- surgery
- unplanned admission or prolongation for >10 nights
- ITU admission >1 night
- Fatal
- death36
Endotherapy perforation rate
Level of agreement for KPI: 100%
Level of agreement for standard: 92%
Post-polypectomy bleeding rate
Level of agreement for KPI: 92.3%
Level of agreement for standard: 85%
Given the potentially high rate of malignancy in LNPCPs, an aim to manage lesions within the NHS 62-day target was considered desirable by the GDG (table 11). However, it was recognised that the need for prompt treatment needed to be balanced with ensuring any treatment was performed by an appropriately skilled clinician. It was also recognised that delays might occur both in assessment/referral as well as in providing treatment.6 47 200
Level of agreement for KPI: 100%
Level of agreement for standards: 84%
Table 11.
Timeliness domain summary
| Domain | Timeliness |
|---|---|
| Objective | Provide a timely service and minimise delay in cancer diagnosis and therapy |
| KPI |
|
| Denominator |
Inclusions: Include all patients with LNPCPs Exclusions: Exclude LNPCPs removed at the time of detection |
| Analysis | Record date of LNPCP detection Record date of referral to clinician who will perform therapy. Note this is not the date patient is referred to the colorectal MDT Record date of definitive therapy Calculate mean time from referral to therapy Calculate mean time from detection to referral for therapy Calculate interval in days |
| Frequency | Calculate annually |
| Level of analysis | Service level |
| Minimum standard | Time from diagnosis to referral: <4 weeks (28 days)—no current standard defined for proportion meeting this standard Time from referral to definitive management: <8 weeks (56 days)—no current standard defined for proportion meeting this standard |
| Aspirational standard | No current standard defined |
| Action | Review cases where time from diagnosis to referral is >4 weeks (28 days) Review cases where time from diagnosis to referral is >8 weeks (56 days) |
| Evidence | Muto et al6 http://www.scotland.gov.uk/About/Performance/scotPerforms/partnerstories/NHSScotlandperformance/Cancerwaitingtimes47 |
KPI, key performance indicators; LNPCPs, large non-pedunculated colorectal polyps; MDT, multidisciplinary team.
There is evidence that increased procedure numbers and experience are associated with better outcomes and reduced adverse events (table 12). In addition, it is common practice in other disciplines to consider undertaking a minimum number of procedures to maintain acceptable standards as it allows meaningful measurement of other KPIs. For example, the BCSP mandates a minimum number of 150 colonoscopies per year.26–28 36 The GDG acknowledged there was no clear evidence of the annual incidence of LNPCPs and therefore felt they could not propose a minimum number of procedures per year per endoscopist. Nevertheless, the GDG felt it important to monitor procedural volume per endoscopist.
Level of agreement for KPI: 92.3%
Level of agreement for standard: 92%
Table 12.
Volume of procedures per endoscopist domain
| Domain | Volume of procedures per endoscopist |
|---|---|
| Objective | Safeguard to ensure that endoscopists undertake a sufficient number of procedures a year to maintain acceptable standards |
| KPI | Number of NPCPs of ≥20 mm in size removed per endoscopist per year |
| Inclusions | All NPCPs of ≥20 mm in size removed per endoscopist per year |
| Analysis | Count of number of NPCPs (not patients) removed per endoscopist per year |
| Frequency | Annual analysis |
| Level of analysis | Service and individual endoscopist level |
| Minimum standard | No current standard defined |
| Aspirational standard | No current standard defined |
| Action | Review in conjunction with other KPIs. Consider focusing NPCP therapy on fewer clinicians to maintain and improve skills |
| Evidence | Rabeneck et al26 Singh et al27 Chukmaitov et al28 NHS BCSP Publication36 |
BCSP, Bowel Cancer Screening Programme; KPI, key performance indicators; NHS, National Health Service; NPCP, non-pedunculated colorectal polyps.
Advanced polypectomy training and accreditation
The GDG discussed ways to improve training in the management of LNPCPs during a round table meeting. Owing to the lack of an evidence base it was not felt to be possible to create guidelines for training and the aim was therefore to formulate a reference model for training in advanced polypectomy techniques.
Entry requirements for training
There was agreement that reaching a minimum number of diagnostic colonoscopy procedures was required to allow development of essential basic colonoscopy and therapeutic skills before entering advanced polypectomy training. There was broad opinion that handling and decision-making skills develop after around 250–350 colonoscopies, with further development after an extensive period of independent practice. Evidence that increased endoscopic experience is associated with improved performance and a reduced rate of adverse events reinforced this view. A minimum number of 500 independent (post-certification) colonoscopies was felt to be a suitable number to ensure that adequate experience has been achieved in both observed and independent practice. There was unanimous opinion that snare polypectomy experience and skill were the key identifiers of endoscopists suitable for advanced training and that competency in snare polypectomy of smaller lesions (up to 2 cm) needed to be established. This may be assessed with a formal assessment tool such as the ‘DOPyS’ assessment tool.154 In addition to formal assessment, evidence of regular snare polypectomy experience with lesions >1 cm in the preceding year was considered desirable as well as performance data for all colonoscopy practice in that period.
Training programme
An apprenticeship programme such as a dedicated fellowship in a recognised advanced endoscopy centre was considered to be the preferred model for delivering advanced polypectomy training to trainees, whereas non-trainees such as consultants wishing to develop advanced polypectomy skills would require a period of mentorship. The availability of fellowships in specific regions may be linked to population demands. An agreed appropriate learning curve starts with a trainee continuing to develop individual colonoscopy skills while watching and assisting their mentor resect large lesions. During this period, trainees may gain significant experience and develop their technique on colonic lesions between 10 and 19 mm in size before progressing to larger lesions and piecemeal resection. At this point trainees would be encouraged to bring cases to dedicated training lists. LNPCP location and accessibility also confers increased lesion complexity in addition to size, and rectal lesions where the bowel wall is thicker and access is easier may be an ideal starting point for obtaining hands-on experience. Trainers and mentors would be required to ensure that their performance data (KPIs) met minimum standards before supervising fellows.
Certification
Dividing certification into provisional and full certification was strongly supported. Achieving provisional certification would be based on outcomes data and mentor opinion and would be the start of independent practice—that is, the trainer not in the room. Full certification would be obtained based on achieving satisfactory KPIs while provisionally certified in addition to mentor opinion, and maintenance of full certification status would be dependent on achieving satisfactory KPIs.
Other potential training modalities
Training workshops were suggested as a modality for reinforcing technical and decision-making skills obtained during a fellowship programme while simulator and tissue simulator models allow hands-on exposure in a safe setting. There is also growing support for the use of live animal training models. In the UK, the BSG have indicated their support for this modality.
Footnotes
Contributors: Chairman and endoscopy lead: MDR. Lead author: AC. Endoscopy representatives: JAB, ST-G, PB, BPS, AMV, JA, BJR, SD. Surgical leads: RP and WVG. Histopathological lead: MBL. Patient lead: GL. Writing, review and collation of evidence group: AC, MDR, JAB and SD.
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
References
- 1.The AGREE Next Steps Consortium. Appraisal of Guidelines for Research and Evaluation II. AGREE 2 Instrument; 2009:1–56. http://www.agreetrust.org/wp-content/uploads/2013/10/AGREE-II-Users-Manual-and-23-item-Instrument_2009_UPDATE_2013.pdf [Google Scholar]
- 2.Williams JG, Pullan RD, Hill J, et al. . Management of the malignant colorectal polyp: ACPGBI position statement. Colorectal Dis 2013;15(Suppl 2):1–38. 10.1111/codi.12262 [DOI] [PubMed] [Google Scholar]
- 3. http://pathways.nice.org.uk/pathways/colorectal-cancer.
- 4. http://www.sign.ac.uk/methodology/checklists.html.
- 5.Guyatt GH, Oxman AD, Vist GE, et al. . GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ 2008;336:924–6. 10.1136/bmj.39489.470347.AD [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Muto T, Bussey HJR, Morson BC. The evolution of cancer of the colon and rectum. Cancer 1975;36:2251–70. 10.1002/cncr.2820360944 [DOI] [PubMed] [Google Scholar]
- 7.Lee TJW, Rees C, Nickerson C, et al. . Management of large sessile or flat colonic polyps in the English bowel cancer screening programme. Br J Surg 2013;100:1633–9. 10.1002/bjs.9282 [DOI] [PubMed] [Google Scholar]
- 8.Participants in the Paris Workshop. The Paris endoscopic classification of superficial neoplastic lesions: esophagus, stomach, and colon. Gastrointest Endosc 2003;58:S2–43. 10.1016/S0016-5107(03)02169-2 [DOI] [PubMed] [Google Scholar]
- 9.Moss A, Bourke MJ, Williams SJ, et al. . Endoscopic mucosal resection outcomes and prediction of submucosal cancer from advanced colonic mucosal neoplasia. Gastroenterology 2011;140:1909–18. 10.1053/j.gastro.2011.02.062 [DOI] [PubMed] [Google Scholar]
- 10.Nagata S, Tanaka S, Haruma K, et al. . Pit pattern diagnosis of early colorectal carcinoma by magnifying colonoscopy: clinical and histological implications. Int J Oncol 2000;16:927–34. [DOI] [PubMed] [Google Scholar]
- 11.Sanchez-Yague A, Kaltenbach T, Raju G, et al. . Advanced endoscopic resection of colorectal lesions. Gastro Clin N Am 2013;42:459–77. 10.1016/j.gtc.2013.05.012 [DOI] [PubMed] [Google Scholar]
- 12.Uraoka T, Saito Y, Matsuda T, et al. . Endoscopic indications for endoscopic mucosal resection of laterally spreading tumours in the colorectum. Gut 2006;55:1592–7. 10.1136/gut.2005.087452 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Oka S, Tanaka S, Kanao H, et al. . Therapeutic strategy for colorectal laterally spreading tumor. Dig Endosc 2009;21(Suppl 1):S43–6. 10.1111/j.1443-1661.2009.00869.x [DOI] [PubMed] [Google Scholar]
- 14.Uraoka T, Saito Y, Ikematsu H, et al. . Sano's capillary pattern classification for narrow-band imaging of early colorectal lesions. Dig Endosc 2011;23(Suppl 1): 112–15. 10.1111/j.1443-1661.2011.01118.x [DOI] [PubMed] [Google Scholar]
- 15.Ikematsu H, Matsuda T, Emura F, et al. . Efficacy of capillary pattern type IIIA/IIIB by magnifying narrow band imaging for estimating depth of invasion of early colorectal neoplasms. BMC Gastroenterol 2010;10:33 10.1186/1471-230X-10-33 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hayashi N, Tanaka S, Hewett DG, et al. . Endoscopic prediction of deep submucosal invasive carcinoma: validation of the narrow-band imaging international colorectal endoscopic (NICE) classification. Gastrointest Endosc 2013;78:625–32. 10.1016/j.gie.2013.04.185 [DOI] [PubMed] [Google Scholar]
- 17.Tanaka S, Sano Y. Aim to unify the narrow band imaging (NBI) magnifying classification for colorectal tumors: current status in Japan from a summary of the consensus symposium in the 79th Annual Meeting of the Japan Gastroenterological Endoscopy Society. Dig Endosc 2011;23(Suppl 1):131–9. 10.1111/j.1443-1661.2011.01106.x [DOI] [PubMed] [Google Scholar]
- 18.Ramirez M, Schierling S, Papaconstantinou HT, et al. . Management of the malignant polyp. Clin Colon Rectal Surg 2008;21:286–90. 10.1055/s-0028-1089944 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gupta S, Miskovic D, Rembacken B, et al. . A novel method for determining the difficulty of colonoscopic polypectomy. Frontline Gastroenterol 2013;4:244–8. 10.1136/flgastro-2013-100331 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Tholoor S, Tsakgournis O, Basford P, et al. . Managing difficult polyps: techniques and pitfalls. Ann Gastroenterol 2013;26:1–8. [PMC free article] [PubMed] [Google Scholar]
- 21.Ishiguro A, Uno Y, Ishiguro Y, et al. . Correlation of lifting versus non-lifting and microscopic depth of invasion in early colorectal cancer. Gastrointest Endosc 1999;50:329–33. 10.1053/ge.1999.v50.98591 [DOI] [PubMed] [Google Scholar]
- 22.Metz AJ, Bourke MJ, Moss A, et al. . Factors that predict bleeding following endoscopic mucosal resection of large colonic lesions. Endoscopy 2011;43:506–11. 10.1055/s-0030-1256346 [DOI] [PubMed] [Google Scholar]
- 23.Buddingh KT, Herngreen T, Haringsma J, et al. . Location in the right hemi-colon is an independent risk factor for delayed post-polypectomy hemorrhage: a multi-center case-control study. Am J Gastroenterol 2011;106:1119–24. 10.1038/ajg.2010.507 [DOI] [PubMed] [Google Scholar]
- 24.Kim HH, Kim JH, Park SJ, et al. . Risk factors for incomplete resection and complications in endoscopic mucosal resection for lateral spreading tumors. Dig Endosc 2012;24:259–66. 10.1111/j.1443-1661.2011.01232.x [DOI] [PubMed] [Google Scholar]
- 25.Rutter MD, Nickerson C, Rees CJ, et al. . Risk factors for adverse events related to polypectomy in the English Bowel Cancer Screening Programme. Endoscopy 2014;46:90–7. 10.1055/s-0033-1344987 [DOI] [PubMed] [Google Scholar]
- 26.Rabeneck L, Paszat LF, Hilsden RJ, et al. . Bleeding and perforation after outpatient colonoscopy and their risk factors in usual clinical practice. Gastroenterology 2008;135:1899–906, 906.e1 10.1053/j.gastro.2008.08.058 [DOI] [PubMed] [Google Scholar]
- 27.Singh H, Penfold RB, DeCoster C, et al. . Colonoscopy and its complications across a Canadian regional health authority. Gastrointest Endosc 2009;69:665–71. 10.1016/j.gie.2008.09.046 [DOI] [PubMed] [Google Scholar]
- 28.Chukmaitov A, Bradley CJ, Dahman B, et al. . Association of polypectomy techniques, endoscopist volume, and facility type with colonoscopy complications. Gastrointest Endosc 2013;77:436–46. 10.1016/j.gie.2012.11.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Longcroft-Wheaton G, Duku M, Mead R, et al. . Risk stratification system for evaluation of complex polyps can predict outcomes of endoscopic mucosal resection. Dis Colon Rectum 2013;56:960–6. 10.1097/DCR.0b013e31829193e0 [DOI] [PubMed] [Google Scholar]
- 30.Moss A. From gastroenterologist to surgeon to gastroenterologist for management of large sessile colonic polyps: something new under the sun? Gastrointest Endosc 2014;79:108–10. 10.1016/j.gie.2013.08.037 [DOI] [PubMed] [Google Scholar]
- 31.Buchner AM, Guarner-Argente C, Ginsberg GG. Outcomes of EMR of defiant colorectal lesions directed to an endoscopy referral center. Gastrointest Endosc 2012;76:255–63. 10.1016/j.gie.2012.02.060 [DOI] [PubMed] [Google Scholar]
- 32.Friedland S, Banerjee S, Kochar R, et al. . Outcomes of repeat colonoscopy in patients with polyps referred for surgery without biopsy-proven cancer. Gastrointest Endosc 2014;79:101–7. 10.1016/j.gie.2013.06.034 [DOI] [PubMed] [Google Scholar]
- 33.Allaix ME, Arezzo A, Giraudo G, et al. . Transanal endoscopic microsurgery vs. laparoscopic total mesorectal excision for T2N0 rectal cancer. J Gastrointest Surg 2012;16:2280–7. 10.1007/s11605-012-2046-8 [DOI] [PubMed] [Google Scholar]
- 34.Hauenschild L, Bader FG, Laubert T, et al. . Laparoscopic colorectal resection for benign polyps not suitable for endoscopic polypectomy. Int J Colorectal Dis 2009;24:755–9. 10.1007/s00384-009-0688-0 [DOI] [PubMed] [Google Scholar]
- 35.Vormbrock K, Monkemuller K. Difficult colon polypectomy. World J Gastrointest Endosc 2012;4:269–80. 10.4253/wjge.v4.i7.269 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.NHS Bowel Cancer Screening Programme. Quality Assurance Guidelines for Colonoscopy. NHS BCSP Publication No.6, 2011. http://www.cancerscreening.nhs.uk/bowel/publications/nhsbcsp06.pdf [Google Scholar]
- 37.Pabby A, Schoen RE, Weissfeld JL, et al. . Analysis of colorectal cancer occurrence during surveillance colonoscopy in the dietary Polyp Prevention Trial. Gastrointest Endosc 2005;61:385–91. 10.1016/S0016-5107(04)02765-8 [DOI] [PubMed] [Google Scholar]
- 38.Brooker JC, Saunders BP, Shah SG, et al. . Endoscopic resection of large sessile colonic polyps by specialist and non-specialist endoscopists. Br J Surg 2002;89:1020–4. 10.1046/j.1365-2168.2002.02157.x [DOI] [PubMed] [Google Scholar]
- 39.Pohl H, Srivastava A, Bensen SP, et al. . Incomplete polyp resection during colonoscopy-results of the complete adenoma resection (CARE) study. Gastroenterology 2013;144:74–80.e1. 10.1053/j.gastro.2012.09.043 [DOI] [PubMed] [Google Scholar]
- 40.Friedland S, Shelton A, Kothari S, et al. . Endoscopic management of nonlifting colon polyps. Diag Ther Endosc 2013;2013:412936 10.1155/2013/412936 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Vaughan-Shaw PG, Wheeler JM, Borley NR. The impact of a dedicated Mdt on the management of early rectal cancer (abstract). Gut 2013;62:A216. [DOI] [PubMed] [Google Scholar]
- 42.Lamb CA, Barbour JA. Developing an endoscopic mucosal resection service in a district general hospital. Frontline Gastroenterol 2012;3:272–7. 10.1136/flgastro-2012-100212 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Liao ZH, Liang-Hao H, Zhao-Shen L, et al. . Multidisciplinary team meeting before therapeutic ERCP: a prospective study with 1,909 cases. J Interv Gastroenterol 2011;1:64–9. 10.4161/jig.1.2.15047 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Swan MP, Bourke MJ, Alexander S, et al. . Large refractory colonic polyps: is it time to change our practice? A prospective study of the clinical and economic impact of a tertiary referral colonic mucosal resection and polypectomy service (with videos). Gastrointest Endosc 2009;70:1128–36. 10.1016/j.gie.2009.05.039 [DOI] [PubMed] [Google Scholar]
- 45.Bertelson NL, Kalkbrenner KA, Merchea A, et al. . Colectomy for endoscopically unresectable polyps: how often is it cancer? Dis Colon Rectum 2012;55:1111–16. 10.1097/DCR.0b013e3182695115 [DOI] [PubMed] [Google Scholar]
- 46.Gallegos-Orozco JF, Gurudu SR. Complex colon polypectomy. Gastroenterol Hepatol 2010;6:375–82. [PMC free article] [PubMed] [Google Scholar]
- 47. http://www.scotland.gov.uk/About/Performance/scotPerforms/partnerstories/NHSScotlandperformance/Cancerwaitingtimes.
- 48.Wilson LS, Lightwood J. Model of estimated rates of colorectal cancer from polyp growth by year of surveillance. J Med Screen 2001;8:187–96. 10.1136/jms.8.4.187 [DOI] [PubMed] [Google Scholar]
- 49.Stryker SJ, Wolff BG, Culp CE, et al. . Natural history of untreated colonic polyps. Gastroenterology 1987;93:1009–13. [DOI] [PubMed] [Google Scholar]
- 50.Kantsevoy SV, Adler DG, Conway JD, et al. . Endoscopic mucosal resection and endoscopic submucosal dissection. Gastrointest Endosc 2008;68:11–18. 10.1016/j.gie.2008.01.037 [DOI] [PubMed] [Google Scholar]
- 51.Weusten BL. En bloc resection: does it really matter? Endoscopy 2014;46:279–81. 10.1055/s-0034-1365404 [DOI] [PubMed] [Google Scholar]
- 52.Bujanda L. Malignant colorectal polyps. World J Gastroenterol 2010;16:3103 10.3748/wjg.v16.i25.3103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Hotta K, Saito Y, Matsuda T, et al. . Local recurrence and surveillance after endoscopic resection of large colorectal tumors. Dig Endosc 2010;22(Suppl 1):S63–8. 10.1111/j.1443-1661.2010.00965.x [DOI] [PubMed] [Google Scholar]
- 54.Saito Y, Fukuzawa M, Matsuda T, et al. . Clinical outcome of endoscopic submucosal dissection versus endoscopic mucosal resection of large colorectal tumors as determined by curative resection. Surg Endosc 2010;24:343–52. 10.1007/s00464-009-0562-8 [DOI] [PubMed] [Google Scholar]
- 55.Terasaki M, Tanaka S, Oka S, et al. . Clinical outcomes of endoscopic submucosal dissection and endoscopic mucosal resection for laterally spreading tumors larger than 20 mm. J Gastroenterol Hepatol 2012;27:734–40. 10.1111/j.1440-1746.2011.06977.x [DOI] [PubMed] [Google Scholar]
- 56.Kiriyama S, Saito Y, Yamamoto S, et al. . Comparison of endoscopic submucosal dissection with laparoscopic-assisted colorectal surgery for early-stage colorectal cancer: a retrospective analysis. Endoscopy 2012;44:1024–30. 10.1055/s-0032-1310259 [DOI] [PubMed] [Google Scholar]
- 57.Yoda Y, Ikematsu H, Matsuda T, et al. . A large-scale multicenter study of long-term outcomes after endoscopic resection for submucosal invasive colorectal cancer. Endoscopy 2013;45:718–24. 10.1055/s-0033-1344234 [DOI] [PubMed] [Google Scholar]
- 58.Barendse RM, van den Broek FJ, van Schooten J, et al. . Endoscopic mucosal resection vs transanal endoscopic microsurgery for the treatment of large rectal adenomas. Colorectal Dis 2012;14:e191–6. 10.1111/j.1463-1318.2011.02863.x [DOI] [PubMed] [Google Scholar]
- 59.Seo GJ, Sohn DK, Han KS, et al. . Recurrence after endoscopic piecemeal mucosal resection for large sessile colorectal polyps. World J Gastroenterol 2010;16:2806–11. 10.3748/wjg.v16.i22.2806 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Hotta K, Fujii T, Saito Y, et al. . Local recurrence after endoscopic resection of colorectal tumors. Int J Colorectal Dis 2009;24:225–30. 10.1007/s00384-008-0596-8 [DOI] [PubMed] [Google Scholar]
- 61.Park JJ, Cheon JH, Kwon JE, et al. . Clinical outcomes and factors related to resectability and curability of EMR for early colorectal cancer. Gastrointest Endosc 2011;74:1337–46. 10.1016/j.gie.2011.07.069 [DOI] [PubMed] [Google Scholar]
- 62.Hassan C, Zullo A, Risio M, et al. . Histologic risk factors and clinical outcome in colorectal malignant polyp: a pooled-data analysis. Dis Colon Rectum 2005;48:1588–96. 10.1007/s10350-005-0063-3 [DOI] [PubMed] [Google Scholar]
- 63.Butte JM, Tang P, Gonen M, et al. . Rate of residual disease after complete endoscopic resection of malignant colonic polyp. Dis Colon Rectum 2012;55:122–7. 10.1097/DCR.0b013e3182336c38 [DOI] [PubMed] [Google Scholar]
- 64.Nascimbeni R, Burgar LJ, Nivatongs S, et al. . Risk of lymph node metastasis in T1 carcinoma of the colon and rectum. Dis Colon Rectum 2002;45:200–6. 10.1007/s10350-004-6147-7 [DOI] [PubMed] [Google Scholar]
- 65.Gonçalves BM, Fontainhas V, Caetano AC, et al. . Oncological outcomes after endoscopic removal of malignant colorectal polyps. Rev Esp Enferm Dig 2013;105:454–61. 10.4321/S1130-01082013000800003 [DOI] [PubMed] [Google Scholar]
- 66.Schonberg MA, Davis RB, McCarthy EP, et al. . Index to predict 5-year mortality of community-dwelling adults aged 65 and older using data from the National Health Interview Survey. J Gen Intern Med 2009;24:1115–22. 10.1007/s11606-009-1073-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.McNicol L, Story DA, Leslie K, et al. . Postoperative complications and mortality in older patients having non-cardiac surgery at three Melbourne teaching hospitals. Med J Aust 2007;186:447–52. [DOI] [PubMed] [Google Scholar]
- 68.Church JM, Muto T, Appau K. Flat lesions of the colorectal mucosa: differences in recognition between Japanese and American endoscopists. Dis Colon Rectum 2004;47:1462–6. 10.1007/s10350-004-0608-x [DOI] [PubMed] [Google Scholar]
- 69.Rubio CA, Hoog CM, Brostrom O, et al. . Assessing the size of polyp phantoms in tandem colonoscopies. Anti Cancer Research 2009;29:1539–46. [PubMed] [Google Scholar]
- 70.Schoen RE, Gerber LD, Margulies C. The pathologic measurement of polyp size is preferable to the endoscopic estimate. Gastrointest Endosc 1997;46:492–6. 10.1016/S0016-5107(97)70002-6 [DOI] [PubMed] [Google Scholar]
- 71.Eichenseer PJ, Dhanekula R, Jakate S, et al. . Endoscopic mis-sizing of polyps changes colorectal cancer surveillance recommendations. Dis Colon Rectum 2013;56:315–21. 10.1097/DCR.0b013e31826dd138 [DOI] [PubMed] [Google Scholar]
- 72.Gopalswamy N, Shenoy V, Choudhry M, et al. . Is in vivo measurement of size of polyps during colonoscopy accurate? Gastrointest Endosc 1997;46:497–502. 10.1016/S0016-5107(97)70003-8 [DOI] [PubMed] [Google Scholar]
- 73.The Endoscopic Classification Review Group. Update on the Paris classification of superficial neoplastic lesions in the digestive tract. Endoscopy 2005;37:570 10.1055/s-2005-861352 [DOI] [PubMed] [Google Scholar]
- 74.Kudo S, Hirota S, Nakajima T, et al. . Colorectal tumours and pit pattern. J Clin Pathol 1994;47:880–5. 10.1136/jcp.47.10.880 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Helbig CD, Rex DK. Narrow band imaging (NBI) versus white-light (WL) for colon polyp detection using high definition (HD) colonoscopes. Gastrointest Endosc 2006;63:AB213 10.1016/j.gie.2006.03.525 [DOI] [Google Scholar]
- 76.East JE, Suzuki N, Ignjatovic A, et al. . A Randomised trial of NBI for adenoma detection in high risk patients: flat adenomas, bowel preparation and endoscopist sub-group analysis. Gastrointest Endosc 2011;73:AB376 10.1016/j.gie.2010.10.015 [DOI] [Google Scholar]
- 77.Kanao H. Clinical significance of type VI pit pattern subclassification in determining the depth of invasion of colorectal neoplasms. World J Gastroenterol 2008;14:211 10.3748/wjg.14.211 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Kudo S, Lambert R, Allen JI, et al. . Nonpolypoid neoplastic lesions of the colorectal mucosa. Gastrointest Endosc 2008;68:S3–47. 10.1016/j.gie.2008.07.052 [DOI] [PubMed] [Google Scholar]
- 79.Ignjatovic A, Thomas-Gibson S, East JE, et al. . Development and validation of a training module on the use of narrow-band imaging in differentiation of small adenomas from hyperplastic colorectal polyps. Gastrointest Endosc 2011;73:128–33. 10.1016/j.gie.2010.09.021 [DOI] [PubMed] [Google Scholar]
- 80.Rastogi A, Early DS, Gupta N, et al. . Randomized, controlled trial of standard-definition white-light, high-definition white-light, and narrow-band imaging colonoscopy for the detection of colon polyps and prediction of polyp histology. Gastrointest Endosc 2011;74:593–602. 10.1016/j.gie.2011.04.050 [DOI] [PubMed] [Google Scholar]
- 81.Tischendorf JJ, Wasmuth HE, Koch A, et al. . Value of magnifying chromoendoscopy and narrow band imaging (NBI) in classifying colorectal polyps: a prospective controlled study. Endoscopy 2007;39:1092–6. 10.1055/s-2007-966781 [DOI] [PubMed] [Google Scholar]
- 82.Matsuda T, Fujii T, Saito Y, et al. . Efficacy of the invasive/non-invasive pattern by magnifying chromoendoscopy to estimate the depth of invasion of early colorectal neoplasms. Am J Gastroenterol 2008;103:2700–6. 10.1111/j.1572-0241.2008.02190.x [DOI] [PubMed] [Google Scholar]
- 83.Kaminski MF, Hassan C, Bisschops R, et al. . Advanced imaging for detection and differentiation of colorectal neoplasia: European Society of Gastrointestinal Endoscopy (ESGE) Guideline. Endoscopy 2014;46:435–57. 10.1055/s-0034-1365348 [DOI] [PubMed] [Google Scholar]
- 84.Cho SB, Park SY, Yoon KW, et al. . The effect of post-biopsy scar on the submucosal elevation for endoscopic resection of rectal carcinoids. Korean J Gastroenterol 2009;53:36–42. [PubMed] [Google Scholar]
- 85.Han KS, Sohn DK, Choi DH, et al. . Prolongation of the period between biopsy and EMR can influence the nonlifting sign in endoscopically resectable colorectal cancers. Gastrointest Endosc 2008;67:97–102. 10.1016/j.gie.2007.05.057 [DOI] [PubMed] [Google Scholar]
- 86.Gondal G, Grotmol T, Hofstad B, et al. . Biopsy of colorectal polyps is not adequate for grading of neoplasia. Endoscopy 2005;37:1193–7. 10.1055/s-2005-921031 [DOI] [PubMed] [Google Scholar]
- 87.Absar MS, Haboubi NY. Colonic neoplastic polyps: biopsy is not efficient to exclude malignancy. The Trafford experience. Tech Coloproctol 2004;8(Suppl 2):s257–60. 10.1007/s10151-004-0172-3 [DOI] [PubMed] [Google Scholar]
- 88.Chen CH, Wu KL, Hu ML, et al. . Is a biopsy necessary for colon polyps suitable for polypectomy when performing a colonoscopy? Chang Gung Med J 2011;34:506–11. [PubMed] [Google Scholar]
- 89.Kim EK, Han DS, Kim HS. A biopsy before the colorectal ESD—is it useful? Gastrointest Endosc 2012;75:AB407. [Google Scholar]
- 90.Lohnert MS, Wittmer A, Doniec JM. [Endoscopic removal of large colorectal polyps]. Zentralbl Chir 2004;129:291–5. 10.1055/s-2004-822832 [DOI] [PubMed] [Google Scholar]
- 91.ASGE Standards of Practice Committee. Management of antithrombotic agents for endoscopic procedures. Gastrointest Endosc 2009;70:1060–70. 10.1016/j.gie.2009.09.040 [DOI] [PubMed] [Google Scholar]
- 92.Boustiere C, Veitch A, Vanbiervliet G, et al. . Endoscopy and antiplatelet agents. European Society of Gastrointestinal Endoscopy (ESGE) Guideline. Endoscopy 2011;43:445–61. 10.1055/s-0030-1256317 [DOI] [PubMed] [Google Scholar]
- 93.Veitch AM, Baglin TP, Gershlick AH, et al. . Guidelines for the management of anticoagulant and antiplatelet therapy in patients undergoing endoscopic procedures. Gut 2008;57:1322–9. 10.1136/gut.2007.142497 [DOI] [PubMed] [Google Scholar]
- 94.Gandhi S, Narula N, Mosleh W, et al. . Meta-analysis: colonoscopic post-polypectomy bleeding in patients on continued clopidogrel therapy. Aliment Pharmacol Ther 2013;37:947–52. 10.1111/apt.12292 [DOI] [PubMed] [Google Scholar]
- 95.Singh M, Mehta N, Murthy UK, et al. . Postpolypectomy bleeding in patients undergoing colonoscopy on uninterrupted clopidogrel therapy. Gastrointest Endosc 2010;71:998–1005. 10.1016/j.gie.2009.11.022 [DOI] [PubMed] [Google Scholar]
- 96.Wiviott SD, Braunwald E, McCabe CH, et al. . Prasugrel versus clopidogrel in patients with acute coronary syndromes. N Engl J Med 2007;357:2001–15. 10.1056/NEJMoa0706482 [DOI] [PubMed] [Google Scholar]
- 97.Hui AJ, Wong RM, Ching JY, et al. . Risk of colonoscopic polypectomy bleeding with anticoagulants and antiplatelet agents: analysis of 1657 cases. Gastrointest Endosc 2004;59:44–8. 10.1016/S0016-5107(03)02307-1 [DOI] [PubMed] [Google Scholar]
- 98.Woodhouse C, Evans G, Muller AF. The new oral anticoagulants: practical management for patients attending for endoscopic procedures. Frontline Gastroenterol 2013;4:213–18. 10.1136/flgastro-2013-100325 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Kovacs MJ, Kearon C, Rodger M, et al. . Single-arm study of bridging therapy with low-molecular-weight heparin for patients at risk of arterial embolism who require temporary interruption of warfarin. Circulation 2004;110:1658–63. 10.1161/01.CIR.0000142859.77578.C9 [DOI] [PubMed] [Google Scholar]
- 100.Garcia DA, Regan S, Henault LE, et al. . Risk of thromboembolism with short-term interruption of warfarin therapy. Arch Intern Med 2008;168:63–9. 10.1001/archinternmed.2007.23 [DOI] [PubMed] [Google Scholar]
- 101.Blacker DJ, Wijdicks EF, McClelland RL. Stroke risk in anticoagulated patients with atrial fibrillation undergoing endoscopy. Neurology 2003;61:964–8. 10.1212/01.WNL.0000086817.54076.EB [DOI] [PubMed] [Google Scholar]
- 102.Iakovou I, Schmidt T, Bonizzoni E, et al. . Incidence, predictors, and outcome of thrombosis after successful implantation of drug-eluting stents. JAMA 2005;293:2126–30. 10.1001/jama.293.17.2126 [DOI] [PubMed] [Google Scholar]
- 103.Yousfi M, Gostout CJ, Baron TH, et al. . Postpolypectomy lower gastrointestinal bleeding: potential role of aspirin. Am J Gastroenterol 2004;99:1785–9. 10.1111/j.1572-0241.2004.30368.x [DOI] [PubMed] [Google Scholar]
- 104.Sawhney MS, Salfiti N, Nelson DB, et al. . Risk factors for severe delayed postpolypectomy bleeding. Endoscopy 2008;40:115–19. 10.1055/s-2007-966959 [DOI] [PubMed] [Google Scholar]
- 105.Ninomiya Y, Oka S, Tanaka S, et al. . Risk of bleeding after endoscopic submucosal dissection for colorectal tumors in patients with continued use of low-dose aspirin. J Gastroenterol 2015;50:1–6. [DOI] [PubMed] [Google Scholar]
- 106. https://www.gov.uk/government/uploads/system/uploads/attachment_data/file/138296/dh_103653__1_.pdf.
- 107. http://www.gmc-uk.org/guidance/ethical_guidance/consent_guidance_index.asp.
- 108.del Carmen MG, Joffe S. Informed consent for medical treatment and research: a review. Oncologist 2005;10:636–41. 10.1634/theoncologist.10-8-636 [DOI] [PubMed] [Google Scholar]
- 109.Brozovich M, Read TE, Salgado J, et al. . Laparoscopic colectomy for apparently benign colorectal neoplasia: a word of caution. Surg Endosc 2008;22:506–9. 10.1007/s00464-007-9497-0 [DOI] [PubMed] [Google Scholar]
- 110.Carson RC, Juszczak M, Davenport A, et al. . Is maximum conservative management an equivalent treatment option to dialysis for elderly patients with significant comorbid disease? Clin J Am Soc Nephrol 2009;4:1611–19. 10.2215/CJN.00510109 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Ferrara F, Luigiano C, Ghersi S, et al. . Efficacy, safety and outcomes of ‘inject and cut’ endoscopic mucosal resection for large sessile and flat colorectal polyps. Digestion 2010;82:213–20. 10.1159/000284397 [DOI] [PubMed] [Google Scholar]
- 112.Belderbos TDG, Leenders M, Moons LMG, et al. . Local recurrence after endoscopic mucosal resection of nonpedunculated colorectal lesions: systematic review and meta-analysis. Endoscopy 2014;46:388–400. 10.1055/s-0034-1364970 [DOI] [PubMed] [Google Scholar]
- 113.Geyer M, Guller U, Beglinger C. Carbon dioxide insufflation in routine colonoscopy is safe and more comfortable: results of a randomized controlled double-blinded trial. Diag Ther Endosc 2011;2011:378906 10.1155/2011/378906 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Bassan MS, Holt B, Moss A, et al. . Carbon dioxide insufflation reduces number of postprocedure admissions after endoscopic resection of large colonic lesions: a prospective cohort study. Gastrointest Endosc 2013;77:90–5. 10.1016/j.gie.2012.06.004 [DOI] [PubMed] [Google Scholar]
- 115.Maple JT, Banerjee S, Barth BA, et al. . Methods of luminal distention for colonoscopy. Gastrointest Endosc 2013;77:519–25. 10.1016/j.gie.2012.09.025 [DOI] [PubMed] [Google Scholar]
- 116.Ahmad NA, Kochman ML, Long WB, et al. . Efficacy, safety, and clinical outcomes of endoscopic mucosal resection: a study of 101 cases. Gastrointest Endosc 2002;55:390–6. 10.1067/mge.2002.121881 [DOI] [PubMed] [Google Scholar]
- 117.Christodoulou D, Kandel G, Tsianos EV, et al. . Endoscopic resection of colonic polyps—a review. Ann Gastroenterol 2007;20:180–94. [Google Scholar]
- 118.Swan MP, Bourke MJ, Moss A, et al. . The target sign: an endoscopic marker for the resection of the muscularis propria and potential perforation during colonic endoscopic mucosal resection. Gastrointest Endosc 2011;73:79–85. 10.1016/j.gie.2010.07.003 [DOI] [PubMed] [Google Scholar]
- 119.Davies J, Burke D, Olliver JR, et al. . Methylene blue but not indigo carmine causes DNA damage to colonocytes in vitro and in vivo at concentrations used in clinical chromoendoscopy. Gut 2007;56:155–6. 10.1136/gut.2006.107300 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Dobrowolski S, Dobosz M, Babicki A, et al. . Prophylactic submucosal saline-adrenaline injection in colonoscopic polypectomy: prospective randomized study. Surg Endosc 2004;18:990–3. 10.1007/s00464-003-9214-6 [DOI] [PubMed] [Google Scholar]
- 121.Hsieh YH, Lin HJ, Tseng GY, et al. . Is submucosal epinephrine injection necessary before polypectomy? A prospective, comparative study. Hepatogastroenterology 2001;48:1379–82. [PubMed] [Google Scholar]
- 122.Lee SH, Chung IK, Kim SJ, et al. . Comparison of polypectomy bleeding between epinephrine and saline submucosal injection for large colon polyps by conventional polypectomy: a prospective randomized, multicenter study. World J Gastroenterol 2007;13:2973–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Turner J, Green J, Dolwani S. Use of gelofusine for endoscopic mucosal resection. Gut 2010;59:1446–7; author reply 7 10.1136/gut.2009.198648 [DOI] [PubMed] [Google Scholar]
- 124.Feitoza AB, Gostout CJ, Burgart LJ, et al. . Hydroxypropyl methylcellulose: a better submucosal fluid cushion for endoscopic mucosal resection. Gastrointest Endosc 2003;57:41–7. 10.1067/mge.2003.25 [DOI] [PubMed] [Google Scholar]
- 125.Moss A, Bourke MJ, Kwan V, et al. . Succinylated gelatin substantially increases en bloc resection size in colonic EMR: a randomized, blinded trial in a porcine model. Gastrointest Endosc 2010;71:589–95. 10.1016/j.gie.2009.10.033 [DOI] [PubMed] [Google Scholar]
- 126.Fujishiro M, Yahagi N, Kakushima N, et al. . Outcomes of endoscopic submucosal dissection for colorectal epithelial neoplasms in 200 consecutive cases. Clin Gastroenterol Hepatol 2007;5:678–83; quiz 45 10.1016/j.cgh.2007.01.006 [DOI] [PubMed] [Google Scholar]
- 127.Fujishiro M, Yahagi N, Kashimura K, et al. . Comparison of various submucosal injection solutions for maintaining mucosal elevation during endoscopic mucosal resection. Endoscopy 2004;36:579–83. 10.1055/s-2004-814517 [DOI] [PubMed] [Google Scholar]
- 128.Fujishiro M, Yahagi N, Kashimura K, et al. . Tissue damage of different submucosal injection solutions for EMR. Gastrointest Endosc 2005;62:933–42. 10.1016/j.gie.2005.07.052 [DOI] [PubMed] [Google Scholar]
- 129.Uraoka T, Takahiro F, Saito Y, et al. . Effectiveness of glycerol as a submucosal injection for EMR. Gastrointest Endosc 2005;61:736–40. 10.1016/S0016-5107(05)00321-4 [DOI] [PubMed] [Google Scholar]
- 130.Moss A, Bourke MJ, Metz AJ. A randomized, double-blind trial of succinylated gelatin submucosal injection for endoscopic resection of large sessile polyps of the colon. Am J Gastroenterol 2010;105:2375–82. 10.1038/ajg.2010.319 [DOI] [PubMed] [Google Scholar]
- 131.Holt BA, Bourke MJ. Wide field endoscopic resection for advanced colonic mucosal neoplasia: current status and future directions. Clin Gastroenterol Hepatol 2012;10:969–79. 10.1016/j.cgh.2012.05.020 [DOI] [PubMed] [Google Scholar]
- 132.Yamano HO, Matsushita HO, Yamanaka K, et al. . A study of physical efficacy of different snares for endoscopic mucosal resection. Dig Endosc 2004;16:S85–8. 10.1111/j.1443-1661.2004.00374.x [DOI] [Google Scholar]
- 133.Singh N, Harrison M, Rex DK. A survey of colonoscopic polypectomy practices among clinical gastroenterologists. Gastrointest Endosc 2004;99:414–18. 10.1016/S0016-5107(04)01808-5 [DOI] [PubMed] [Google Scholar]
- 134.Cartera D, Beer-Gabel M, Zbar A, et al. . A survey of colonoscopic polypectomy practice amongst Israeli gastroenterologists. Ann Gastroenterol 2013;26:135–140. [PMC free article] [PubMed] [Google Scholar]
- 135.Rey JF, Beilenhoff U, Neumann CS, et al. . European Society of Gastrointestinal Endoscopy (ESGE). European Society of Gastrointestinal Endoscopy (ESGE) guideline: the use of electrosurgical units. Endoscopy 2010;42:764–72. 10.1055/s-0030-1255594 [DOI] [PubMed] [Google Scholar]
- 136.Chino A, Karasawa T, Uragami N, et al. . A comparison of depth of tissue injury caused by different modes of electrosurgical current in a pig colon model. Gastrointest Endosc 2004;59:374–9. 10.1016/S0016-5107(03)02712-3 [DOI] [PubMed] [Google Scholar]
- 137.Parra-Blanco A, Kaminaga N, Kojima T, et al. . Colonoscopic polypectomy with cutting current: is it safe? Gastrointest Endosc 2000;51:676–81. 10.1067/mge.2000.105203 [DOI] [PubMed] [Google Scholar]
- 138.Van Gossum A, Cozzoli A, Adler M, et al. . Colonoscopic snare polypectomy: analysis of 1485 resections comparing two types of current. Gastrointest Endosc 1992;38:472–5. 10.1016/S0016-5107(92)70479-9 [DOI] [PubMed] [Google Scholar]
- 139.Kim HS, Kim TI, Kim WH, et al. . Risk factors for immediate postpolypectomy bleeding of the colon: a multicenter study. Am J Gastroenterol 2006;101:1333–41. 10.1111/j.1572-0241.2006.00638.x [DOI] [PubMed] [Google Scholar]
- 140.ASGE Technology Committee. Electrosurgical generators. Gastrointest Endosc 2013;78:197–208. 10.1016/j.gie.2013.04.164 [DOI] [PubMed] [Google Scholar]
- 141. http://www.bsg.org.uk/pdf_word_docs/polypectomy_08.pdf.
- 142.Fry LC, Lazenby AJ, Mikolaenko I, et al. . Diagnostic quality of: polyps resected by snare polypectomy: does the type of electrosurgical current used matter? Am J Gastroenterol 2006;101:2123–7. 10.1111/j.1572-0241.2006.00696.x [DOI] [PubMed] [Google Scholar]
- 143.Mannath J, Ragunath K. Endoscopic mucosal resection: who and how? Therap Adv Gastroenterol 2011;4:275– 82 10.1177/1756283X10388683 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Woodward TA, Heckman MG, Cleveland P, et al. . Predictors of complete endoscopic mucosal resection of flat and depressed neoplasia of the colon. Am J Gastroenterol 2012;107:650–4. 10.1038/ajg.2011.473 [DOI] [PubMed] [Google Scholar]
- 145.Uno Y, Munakata A, Uno Y, et al. . The non-lifting sign of invasive colon cancer. Gastrointest Endosc 1994;40:485–9. 10.1016/S0016-5107(94)70216-0 [DOI] [PubMed] [Google Scholar]
- 146.Kobayashi N, Saito Y, Sano Y, et al. . Determining the treatment strategy for colorectal neoplastic lesions: endoscopic assessment or the non-lifting sign for diagnosing invasion depth? Endoscopy 2007;39:701–5. 10.1055/s-2007-966587 [DOI] [PubMed] [Google Scholar]
- 147.Zlatanic J, Waye JD, Kim PS, et al. . Large sessile colonic adenomas: use of argon plasma coagulator to supplement piecemeal snare polypectomy. Gastrointest Endosc 1999;49:731–5. 10.1016/S0016-5107(99)70291-9 [DOI] [PubMed] [Google Scholar]
- 148.Regula J, Wronska E, Polkowski M, et al. . Argon plasma coagulation after piecemeal polypectomy of sessile colorectal adenomas: long-term follow-up study. Endoscopy 2003;35:212–8. 10.1055/s-2003-37254 [DOI] [PubMed] [Google Scholar]
- 149.Mannath J, Subramanian V, Singh R, et al. . Polyp recurrence after endoscopic mucosal resection of sessile and flat colonic adenomas. Dig Dis Sci 2011;56:2389–95. 10.1007/s10620-011-1609-y [DOI] [PubMed] [Google Scholar]
- 150.Conio M, Repici A, Demarquay JF, et al. . EMR of large sessile colorectal polyps. Gastrointest Endosc 2004;60:234–41. 10.1016/S0016-5107(04)01567-6 [DOI] [PubMed] [Google Scholar]
- 151.Brooker JC, Saunders BP, Shah SG, et al. . Treatment with argon plasma coagulation reduces recurrence after piecemeal resection of large sessile colonic polyps: a randomized trial and recommendations. Gastrointest Endosc 2002;55:371–5. 10.1067/mge.2002.121597 [DOI] [PubMed] [Google Scholar]
- 152.Veerappan SG, Ormonde D, Yusoff IF, et al. . Hot avulsion: a modification of an existing technique for management of nonlifting areas of a polyp (with video). Gastrointest Endosc 2014;80:884–8. 10.1016/j.gie.2014.05.333 [DOI] [PubMed] [Google Scholar]
- 153.ASGE Standards of Practice Committee. Role of endoscopy in the staging and management of colorectal cancer. Gastrointest Endosc 2013;78:8–12. 10.1016/j.gie.2013.04.163 [DOI] [PubMed] [Google Scholar]
- 154.Gupta S, Bassett P, Man R, et al. . Validation of a novel method for assessing competency in polypectomy. Gastrointest Endosc 2012;75:568–75. 10.1016/j.gie.2011.09.028 [DOI] [PubMed] [Google Scholar]
- 155.Kethu SR, Banerjee S, Desilets D, et al. . Endoscopic tattooing. Gastrointest Endosc 2010;72:681–5. 10.1016/j.gie.2010.06.020 [DOI] [PubMed] [Google Scholar]
- 156.Salomon P, Berner JS, Waye JD. Endoscopic India ink injection: a method for preparation, sterilization, and administration. Gastrointest Endosc 1993;39:803–5. 10.1016/S0016-5107(93)70269-2 [DOI] [PubMed] [Google Scholar]
- 157.Moss A, Bourke MJ, Pathmanathan N. Safety of colonic tattoo with sterile carbon particle suspension: a proposed guideline with illustrative cases. Gastrointest Endosc 2011;74:214–18. 10.1016/j.gie.2011.01.056 [DOI] [PubMed] [Google Scholar]
- 158.Ono S, Fujishiro M, Goto O, et al. . Endoscopic submucosal dissection for colonic laterally spreading tumours is difficult after target tattooing. Gastrointest Endosc 2009;69(3 Pt 2):763–6. 10.1016/j.gie.2008.08.024 [DOI] [PubMed] [Google Scholar]
- 159.Sorbi D, Norton I, Conio M, et al. . Postpolypectomy lower GI bleeding: descriptive analysis. Gastrointest Endosc 2000;51:690–6. 10.1067/mge.2000.105773 [DOI] [PubMed] [Google Scholar]
- 160.Singharam C, Torbey CF, Jacoby RF. Delayed post polypectomy bleeding. Am J Gastroenterol 1995;90:146–7. [PubMed] [Google Scholar]
- 161.Anderson ML, Pasha TM, Leighton JA. Endoscopic perforation of the colon: lessons from a 10-year study. Am J Gastroenterol 2000;95:3418–22. 10.1111/j.1572-0241.2000.03356.x [DOI] [PubMed] [Google Scholar]
- 162.Timothy SK, Hicks TC, Opelka FG, et al. . Colonoscopy in the patient requiring anticoagulation. Dis Colon Rectum 2001;44:1845–8; discussion, 8–9 10.1007/BF02234465 [DOI] [PubMed] [Google Scholar]
- 163.Higaki S, Hashimoto S, Harada K, et al. . Long-term follow-up of large flat colorectal tumors resected endoscopically. Endoscopy 2003;35:845–9. 10.1055/s-2003-42622 [DOI] [PubMed] [Google Scholar]
- 164.Zauber AG, Winawer SJ, O'Brien MJ, et al. . Colonoscopic polypectomy and long-term prevention of colorectal-cancer deaths. N Engl J Med 2012;366:687–96. 10.1056/NEJMoa1100370 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Gandhi S, Reynolds MW, Boyer G, et al. . Recurrence and malignancy rates in a benign colorectal neoplasm patient cohort: results of a 5-year analysis in a managed care environment. Am J Gastroenterol 2001;96:2761–7. 10.1111/j.1572-0241.2001.04137.x [DOI] [PubMed] [Google Scholar]
- 166.Winawer SJ, Zauber AG, Fletcher RH, et al. . Guidelines for colonoscopy surveillance after polypectomy: a consensus update by the US Multi-Society Task Force on Colorectal Cancer and the American Cancer Society. Gastroenterology 2006;130:1872–85. 10.1053/j.gastro.2006.03.012 [DOI] [PubMed] [Google Scholar]
- 167.Atkin WS, Saunders BP. Surveillance guidelines after removal of colorectal adenomatous polyps. Gut 2002;51:v6–9. 10.1136/gut.51.suppl_5.v6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Walsh RM, Ackroyd FW, Shellito PC. Endoscopic resection of large sessile colorectal polyps. Gastrointest Endosc 1992;38:303–9. 10.1016/S0016-5107(92)70421-0 [DOI] [PubMed] [Google Scholar]
- 169.Khashab M, Eid E, Rusche M, et al. . Incidence and predictors of “late” recurrences after endoscopic piecemeal resection of large sessile adenomas. Gastrointest Endosc 2009;70:344–9. 10.1016/j.gie.2008.10.037 [DOI] [PubMed] [Google Scholar]
- 170.Knabe M, Pohl J, Gerges C, et al. . Standardised long term follow-up after endoscopic resection of large, non-pedunculated colorectal lesions: a prospective two-centre study. Am J Gastroenterol 2014;109:183–9. 10.1038/ajg.2013.419 [DOI] [PubMed] [Google Scholar]
- 171.Ji R, Zuo X-L, Li C-Q, et al. . Confocal endomicroscopy for in vivo prediction of completeness after endoscopic mucosal resection. Surg Endosc 2011;25: 1933–8. 10.1007/s00464-010-1490-3 [DOI] [PubMed] [Google Scholar]
- 172.Cipolletta L, Bianco MA, Garofano ML, et al. . Can magnification endoscopy detect residual adenoma after piecemeal resection of large sessile colorectal lesions to guide subsequent treatment? A prospective single-center study. Dis Colon Rectum 2009;52:1774–9. 10.1007/DCR.0b013e3181b5539c [DOI] [PubMed] [Google Scholar]
- 173.Hurlstone DP, Cross SS, Brown S, et al. . A prospective evaluation of high-magnification chromoscopic colonoscopy in predicting completeness of EMR. Gastrointest Endosc 2004;59:642–50. 10.1016/S0016-5107(04)00156-7 [DOI] [PubMed] [Google Scholar]
- 174.Rogart JN, Aslanian HR, Siddiqui UD. Narrow band imaging to detect residual or recurrent neoplastic tissue during surveillance endoscopy. Dig Dis Sci 2011;56:472–8. 10.1007/s10620-010-1289-z [DOI] [PubMed] [Google Scholar]
- 175.Zhou P, Yao L, Qin X, et al. . Endoscopic Submucosal Dissection for Locally Recurrent Colorectal Lesions After Previous Endoscopic Mucosal Resection. Dis Colon Rectum 2009;52:305–10. [DOI] [PubMed] [Google Scholar]
- 176.Sakamoto T, Saito Y, Matsuda T, et al. . Treatment strategy for recurrent or residual colorectal tumors after endoscopic resection. Surg Endosc 2011;25:255–60. 10.1007/s00464-010-1169-9 [DOI] [PubMed] [Google Scholar]
- 177.Hurlstone DP, Said E. Salvage endoscopic submucosal dissection for refractory post polypectomy fibrosis and recurrence intraepithelial neoplasia: expanding the technological envelope in the UK. Gut 2013;62:A225 10.1136/gutjnl-2013-304907.522 [DOI] [Google Scholar]
- 178.Cohen LB, Waye JD. Treatment of colonic polyps—practical considerations. Clin Gastroenterol 1986;15:359–76. [PubMed] [Google Scholar]
- 179.Repici A, Hassan C, Pagano N, et al. . High efficacy of endoscopic submucosal dissection for rectal laterally spreading tumors larger than 3 cm. Gastrointest Endosc 2013;77:96–101. 10.1016/j.gie.2012.08.036 [DOI] [PubMed] [Google Scholar]
- 180.Saito Y, Uraoka T, Yamaguchi Y, et al. . A prospective, multicenter study of 1111 colorectal endoscopic submucosal dissections (with video). Gastrointest Endosc 2010;72:1217–25. 10.1016/j.gie.2010.08.004 [DOI] [PubMed] [Google Scholar]
- 181.Watanabe T, Itabashi M, Shimada Y, et al. . Japanese Society for Cancer of the Colon and Rectum (JSCCR) guidelines 2010 for the treatment of colorectal cancer. Intl J Clin Oncol 2012;17:1–29. 10.1007/s10147-011-0315-2 [DOI] [PubMed] [Google Scholar]
- 182.Chen HH, Wexner SD, Weiss EG, et al. . Laparoscopic colectomy for benign colorectal disease is associated with a significant reduction in disability as compared with laparotomy. Surg Endosc 1998;12:1397–400. 10.1007/s004649900867 [DOI] [PubMed] [Google Scholar]
- 183.Longo WE, Virgo KS, Johnson FE, et al. . Outcome after proctectomy for rectal cancer in Department of Veterans Affairs Hospitals: a report from the National Surgical Quality Improvement Program. Ann Surg 1998;228:64–70. 10.1097/00000658-199807000-00010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Enker WE, Merchant N, Cohen AM, et al. . Safety and efficacy of low anterior resection for rectal cancer: 681 consecutive cases from a specialty service. Ann Surg 1999;230:544–54. 10.1097/00000658-199910000-00010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Zhu Q-L. Laparoscopic low anterior resection for rectal carcinoma: complications and management in 132 consecutive patients. World J Gastroenterol 2010;16:4605 10.3748/wjg.v16.i36.4605 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Madbouly KM, Remzi FH, Erkek BA, et al. . Recurrence after transanal excision of T1 rectal cancer: should we be concerned? Dis Colon Rectum 2005;48:711–19; discussion 9–21 10.1007/s10350-004-0666-0 [DOI] [PubMed] [Google Scholar]
- 187.Arezzo A, Passera R, Saito Y, et al. . Systematic review and meta-analysis of endoscopic submucosal dissection versus transanal endoscopic microsurgery for large noninvasive rectal lesions. Surg Endosc 2014;28:427–38. 10.1007/s00464-013-3238-3 [DOI] [PubMed] [Google Scholar]
- 188.Sgourakis G, Lanitis S, Kontovounisios C, et al. . Outcomes of transanal endoscopic microsurgery for T1 and T2 rectal cancer. Hell J Surg 2010;82:161–70. 10.1007/s13126-010-0028-4 [DOI] [PubMed] [Google Scholar]
- 189.Cocilovo C, Smith LE, Stahl T, et al. . Transanal endoscopic excision of rectal adenomas. Surg Endosc 2003;17:1461–3. 10.1007/s00464-002-8929-0 [DOI] [PubMed] [Google Scholar]
- 190.Middleton PF, Sutherland LM, Maddern GJ. Transanal endoscopic microsurgery: a systematic review. Dis Colon Rectum 2005;48:270–84. 10.1007/s10350-004-0804-8 [DOI] [PubMed] [Google Scholar]
- 191.Papagrigoriadis S. Transanal endoscopic micro-surgery (TEMS) for the management of large or sessile rectal adenomas: a review of the technique and indications. Int Semin Surg Oncol 2006;3:13 10.1186/1477-7800-3-13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Benedix F, Köckerling F, Lippert H, et al. . Laparoscopic resection for endoscopically unresectable colorectal polyps: analysis of 525 patients. Surg Endosc 2008;22:2576–82. 10.1007/s00464-008-0059-x [DOI] [PubMed] [Google Scholar]
- 193.Bonjer HJ. Laparoscopically assisted vs open colectomy for colon cancer. Arch Surg 2007;142:6 10.1001/archsurg.142.1.6 [DOI] [PubMed] [Google Scholar]
- 194.Vlug MS, Wind J, Hollmann MW, et al. . Laparoscopy in combination with fast track multimodal management is the best perioperative strategy in patients undergoing colonic surgery: a randomized clinical trial (LAFA-study). Ann Surg 2011;254:868–75. 10.1097/SLA.0b013e31821fd1ce [DOI] [PubMed] [Google Scholar]
- 195.The Clinical Outcomes of Surgical Therapy Study Group. A comparison of laparoscopically assisted and open colectomy for colon cancer. N Engl J Med 2004;350:2050–9. 10.1056/NEJMoa032651 [DOI] [PubMed] [Google Scholar]
- 196.Faiz O, Warusavitarne J, Bottle A, et al. . Laparoscopically assisted vs. open elective colonic and rectal resection: a comparison of outcomes in English National Health Service Trusts between 1996 and 2006. Dis Colon Rectum 2009;52:1695–704. 10.1007/DCR.0b013e3181b55254 [DOI] [PubMed] [Google Scholar]
- 197.Salimath J, Jones MW, Hunt DL, et al. . Comparison of return of bowel function and length of stay in patients undergoing laparoscopic versus open colectomy. JSLS 2007;11:72–5. [PMC free article] [PubMed] [Google Scholar]
- 198.Nivatongs S. Complications in colonoscopic polypectomy. An experience with 1555 polypectomies. Dis Colon Rectum 1986;29:825–30. [DOI] [PubMed] [Google Scholar]
- 199.ASGE Standards of Practice Committee. Complications of colonoscopy. Gastrointest Endosc 2011;74:745–52. 10.1016/j.gie.2011.07.025 [DOI] [PubMed] [Google Scholar]
- 200.Atkin WS, Morson BC, Cuzick J. Long-term risk of colorectal cancer after excision of rectosigmoid adenomas. N Engl J Med 1992;326:658–62. 10.1056/NEJM199203053261002 [DOI] [PubMed] [Google Scholar]
- 201.Quirke P, Risio M, Lambert R, et al. . Quality assurance in pathology in colorectal cancer screening and diagnosis—European recommendations. Virchows Arch 2011;458:1–19. 10.1007/s00428-010-0977-6 [DOI] [PMC free article] [PubMed] [Google Scholar]