Abstract
The autophosphorylation sites of the beta 2 isozyme of protein kinase C (PKC) were recently identified as Ser-16/Thr-17 near the NH2 terminus, Thr-314/Thr-324 in the hinge region, and Thr-634/Thr-641 near the COOH terminus [Flint, A.J., Paladini, R.D. & Koshland, D.E. (1990) Science 294, 408-411]. To define the role of autophosphorylation we constructed three site-directed mutants of PKC beta 1 isozyme in which each pair of phosphorylatable residues is changed to alanine. Wild-type PKC beta 1 and the mutant proteins were transiently overexpressed in COS cells, resulting in at least a 20-fold increase in [3H]phorbol 12,13-dibutyrate binding compared with control transfectants. Enzyme assays of PKC partially purified from transfected cells indicated at least a 5-fold increase in PKC activity upon expression of the wild-type protein or the NH2-terminal and hinge mutants. In contrast, no increased activity was detected upon expression of the COOH-terminal mutant. Immunoblot analysis using a beta isoform-specific antibody showed that wild-type, NH2-terminal mutant, and hinge mutant proteins are similarly distributed between the Triton-soluble and insoluble fractions. In contrast, the COOH-terminal mutant protein is largely Triton-insoluble. Immunoblot analysis also indicated that this mutant is resistant to down-regulation upon chronic exposure of cells to phorbol ester. Moreover, RNA blot analysis showed that overexpression of wild-type PKC but not of the COOH-terminal mutant enhances phorbol ester induction of c-FOS and c-JUN mRNA. Our results indicate that (i) alteration in the NH2-terminal and hinge autophosphorylation sites has no effect on PKC function by the criteria examined and (ii) the COOH-terminal autophosphorylation sites are critical for PKC function and possibly subcellular localization in COS cells.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aderem A. The MARCKS brothers: a family of protein kinase C substrates. Cell. 1992 Nov 27;71(5):713–716. doi: 10.1016/0092-8674(92)90546-o. [DOI] [PubMed] [Google Scholar]
- Bell R. M., Burns D. J. Lipid activation of protein kinase C. J Biol Chem. 1991 Mar 15;266(8):4661–4664. [PubMed] [Google Scholar]
- Chomczynski P., Sacchi N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem. 1987 Apr;162(1):156–159. doi: 10.1006/abio.1987.9999. [DOI] [PubMed] [Google Scholar]
- Flint A. J., Paladini R. D., Koshland D. E., Jr Autophosphorylation of protein kinase C at three separated regions of its primary sequence. Science. 1990 Jul 27;249(4967):408–411. doi: 10.1126/science.2377895. [DOI] [PubMed] [Google Scholar]
- Freisewinkel I., Riethmacher D., Stabel S. Downregulation of protein kinase C-gamma is independent of a functional kinase domain. FEBS Lett. 1991 Mar 25;280(2):262–266. doi: 10.1016/0014-5793(91)80307-o. [DOI] [PubMed] [Google Scholar]
- House C., Robinson P. J., Kemp B. E. A synthetic peptide analog of the putative substrate-binding motif activates protein kinase C. FEBS Lett. 1989 Jun 5;249(2):243–247. doi: 10.1016/0014-5793(89)80632-5. [DOI] [PubMed] [Google Scholar]
- Housey G. M., Johnson M. D., Hsiao W. L., O'Brian C. A., Murphy J. P., Kirschmeier P., Weinstein I. B. Overproduction of protein kinase C causes disordered growth control in rat fibroblasts. Cell. 1988 Feb 12;52(3):343–354. doi: 10.1016/s0092-8674(88)80027-8. [DOI] [PubMed] [Google Scholar]
- Huang K. P., Chan K. F., Singh T. J., Nakabayashi H., Huang F. L. Autophosphorylation of rat brain Ca2+-activated and phospholipid-dependent protein kinase. J Biol Chem. 1986 Sep 15;261(26):12134–12140. [PubMed] [Google Scholar]
- Jaken S., Leach K., Klauck T. Association of type 3 protein kinase C with focal contacts in rat embryo fibroblasts. J Cell Biol. 1989 Aug;109(2):697–704. doi: 10.1083/jcb.109.2.697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karin M., Smeal T. Control of transcription factors by signal transduction pathways: the beginning of the end. Trends Biochem Sci. 1992 Oct;17(10):418–422. doi: 10.1016/0968-0004(92)90012-x. [DOI] [PubMed] [Google Scholar]
- Kikkawa U., Kishimoto A., Nishizuka Y. The protein kinase C family: heterogeneity and its implications. Annu Rev Biochem. 1989;58:31–44. doi: 10.1146/annurev.bi.58.070189.000335. [DOI] [PubMed] [Google Scholar]
- Kikkawa U., Takai Y., Minakuchi R., Inohara S., Nishizuka Y. Calcium-activated, phospholipid-dependent protein kinase from rat brain. Subcellular distribution, purification, and properties. J Biol Chem. 1982 Nov 25;257(22):13341–13348. [PubMed] [Google Scholar]
- Mitchell F. E., Marais R. M., Parker P. J. The phosphorylation of protein kinase C as a potential measure of activation. Biochem J. 1989 Jul 1;261(1):131–136. doi: 10.1042/bj2610131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mochly-Rosen D., Koshland D. E., Jr Domain structure and phosphorylation of protein kinase C. J Biol Chem. 1987 Feb 15;262(5):2291–2297. [PubMed] [Google Scholar]
- Molina C. A., Ashendel C. L. Tumor promoter 12-O-tetradecanoylphorbol-13-acetate and sn-1,2-dioctanoylglycerol increase the phosphorylation of protein kinase C in cells. Cancer Res. 1991 Sep 1;51(17):4624–4630. [PubMed] [Google Scholar]
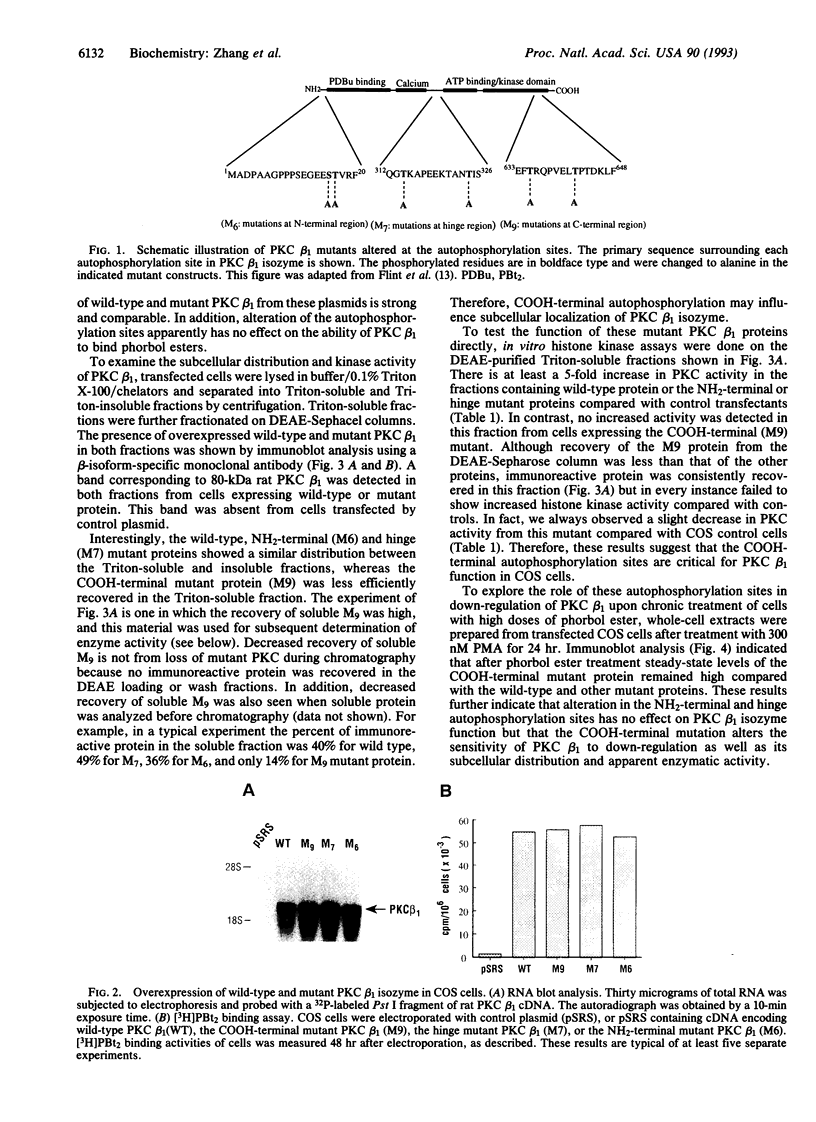
- Muramatsu M., Kaibuchi K., Arai K. Protein kinase C mutants in the auto-inhibitory region exhibit two distinct properties. FEBS Lett. 1992 Oct 12;311(1):75–79. doi: 10.1016/0014-5793(92)81371-r. [DOI] [PubMed] [Google Scholar]
- Newton A. C., Koshland D. E., Jr Protein kinase C autophosphorylates by an intrapeptide reaction. J Biol Chem. 1987 Jul 25;262(21):10185–10188. [PubMed] [Google Scholar]
- Nishizuka Y. Studies and perspectives of protein kinase C. Science. 1986 Jul 18;233(4761):305–312. doi: 10.1126/science.3014651. [DOI] [PubMed] [Google Scholar]
- Nishizuka Y. The role of protein kinase C in cell surface signal transduction and tumour promotion. Nature. 1984 Apr 19;308(5961):693–698. doi: 10.1038/308693a0. [DOI] [PubMed] [Google Scholar]
- Ohno S., Konno Y., Akita Y., Yano A., Suzuki K. A point mutation at the putative ATP-binding site of protein kinase C alpha abolishes the kinase activity and renders it down-regulation-insensitive. A molecular link between autophosphorylation and down-regulation. J Biol Chem. 1990 Apr 15;265(11):6296–6300. [PubMed] [Google Scholar]
- Ono Y., Fujii T., Ogita K., Kikkawa U., Igarashi K., Nishizuka Y. The structure, expression, and properties of additional members of the protein kinase C family. J Biol Chem. 1988 May 15;263(14):6927–6932. [PubMed] [Google Scholar]
- Pears C. J., Kour G., House C., Kemp B. E., Parker P. J. Mutagenesis of the pseudosubstrate site of protein kinase C leads to activation. Eur J Biochem. 1990 Nov 26;194(1):89–94. doi: 10.1111/j.1432-1033.1990.tb19431.x. [DOI] [PubMed] [Google Scholar]
- Pears C., Parker P. J. Down-regulation of a kinase defective PKC-alpha. FEBS Lett. 1991 Jun 17;284(1):120–122. doi: 10.1016/0014-5793(91)80776-y. [DOI] [PubMed] [Google Scholar]
- Spudich A., Meyer T., Stryer L. Association of the beta isoform of protein kinase C with vimentin filaments. Cell Motil Cytoskeleton. 1992;22(4):250–256. doi: 10.1002/cm.970220405. [DOI] [PubMed] [Google Scholar]
- Takebe Y., Seiki M., Fujisawa J., Hoy P., Yokota K., Arai K., Yoshida M., Arai N. SR alpha promoter: an efficient and versatile mammalian cDNA expression system composed of the simian virus 40 early promoter and the R-U5 segment of human T-cell leukemia virus type 1 long terminal repeat. Mol Cell Biol. 1988 Jan;8(1):466–472. doi: 10.1128/mcb.8.1.466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wartmann M., Jans D. A., Parker P. J., Nagamine Y., Hemmings B. A., Jaken S., Eppenberger U., Fabbro D. Overexpression of the alpha-type protein kinase (PK) C in LLC-PK1 cells does not lead to a proportional increase in the induction of two 12-O-tetradecanoylphorbol-13-acetate-inducible genes. Cell Regul. 1991 Jun;2(6):491–502. doi: 10.1091/mbc.2.6.491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolf M., Sahyoun N. Protein kinase C and phosphatidylserine bind to Mr 110,000/115,000 polypeptides enriched in cytoskeletal and postsynaptic density preparations. J Biol Chem. 1986 Oct 5;261(28):13327–13332. [PubMed] [Google Scholar]
- van Straaten F., Müller R., Curran T., Van Beveren C., Verma I. M. Complete nucleotide sequence of a human c-onc gene: deduced amino acid sequence of the human c-fos protein. Proc Natl Acad Sci U S A. 1983 Jun;80(11):3183–3187. doi: 10.1073/pnas.80.11.3183. [DOI] [PMC free article] [PubMed] [Google Scholar]