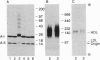
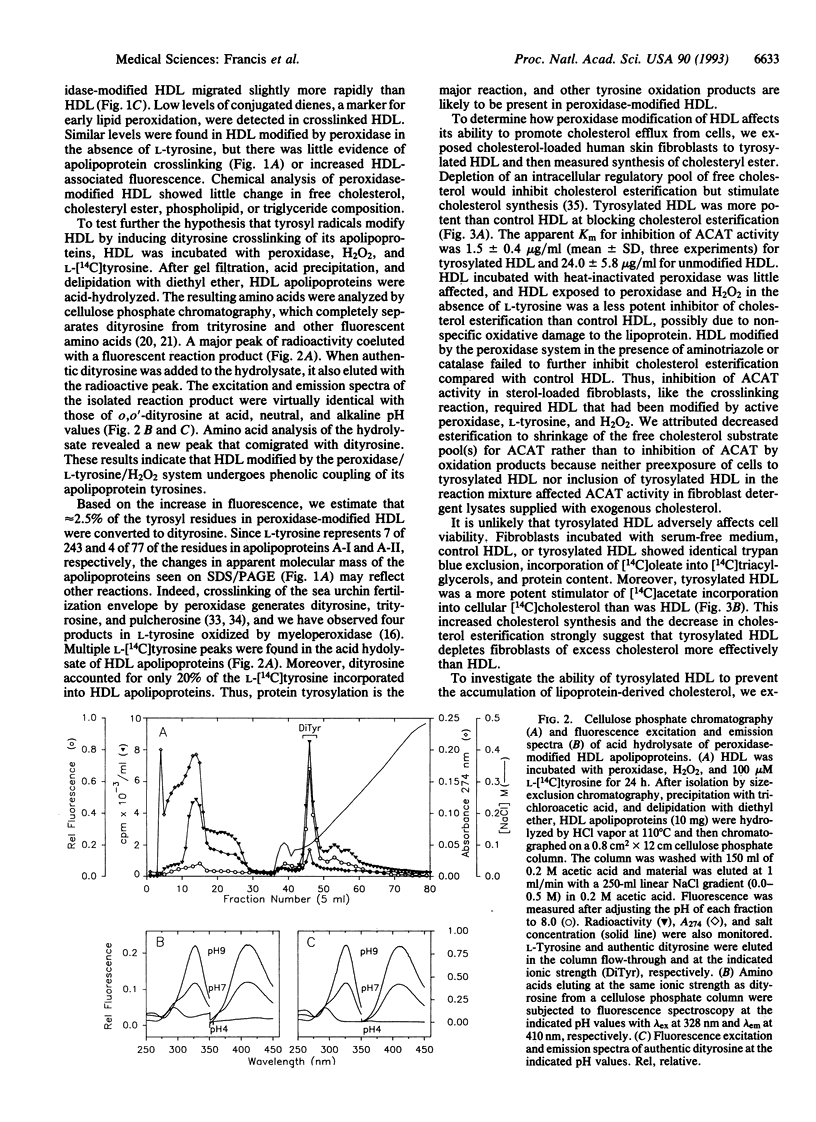
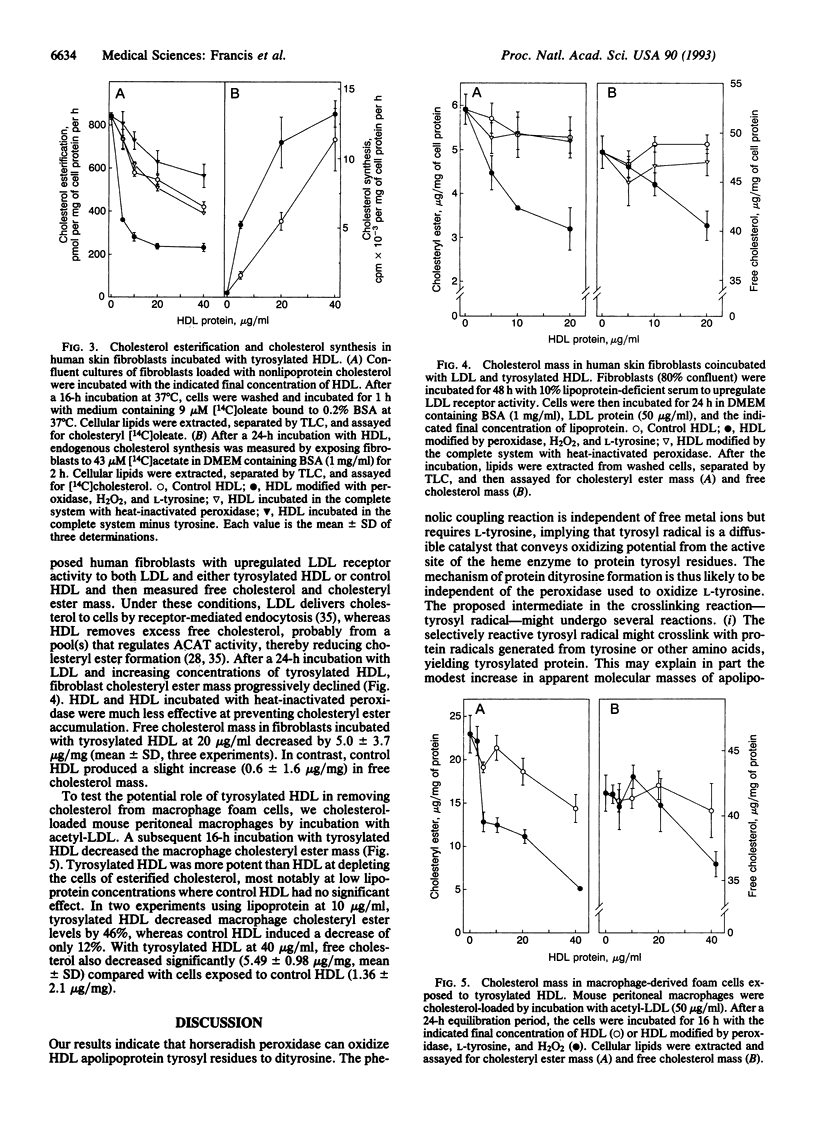
Abstract
Lipoprotein oxidation is thought to play a pivotal role in atherogenesis, yet the underlying reaction mechanisms remain poorly understood. We have explored the possibility that high density lipoprotein (HDL) might be oxidized by peroxidase-generated tyrosyl radical. Exposure of HDL to L-tyrosine, H2O2, and horseradish peroxidase crosslinked its apolipoproteins and strikingly increased protein-associated fluorescence. The reaction required L-tyrosine but was independent of free metal ions; it was blocked by either catalase or the heme poison aminotriazole. Dityrosine and other tyrosine oxidation products were detected in the apolipoproteins of HDL modified by the peroxidase/L-tyrosine/H2O2 system, implicating tyrosyl radical in the reaction pathway. Further evidence suggests that tyrosylated HDL removes cholesterol from cultured cells more effectively than does HDL. Tyrosylated HDL was more potent than HDL at inhibiting cholesterol esterification by the acyl-CoA:cholesterol acyltransferase reaction, stimulating the incorporation of [14C]acetate into [14C]cholesterol, and depleting cholesteryl ester stores in human skin fibroblasts. Moreover, exposure of mouse macrophage foam cells to tyrosylated HDL markedly diminished cholesteryl ester and free cholesterol mass. We have recently found that myeloperoxidase, a heme protein secreted by activated phagocytes, can also convert L-tyrosine to o,o'-dityrosine. This raises the possibility that myeloperoxidase-generated tyrosyl radical may modify HDL, enabling the lipoprotein to protect the artery wall against pathological cholesterol accumulation.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aeschbach R., Amadò R., Neukom H. Formation of dityrosine cross-links in proteins by oxidation of tyrosine residues. Biochim Biophys Acta. 1976 Aug 9;439(2):292–301. doi: 10.1016/0005-2795(76)90064-7. [DOI] [PubMed] [Google Scholar]
- Amadò R., Aeschbach R., Neukom H. Dityrosine: in vitro production and characterization. Methods Enzymol. 1984;107:377–388. doi: 10.1016/0076-6879(84)07026-9. [DOI] [PubMed] [Google Scholar]
- Basu S. K., Goldstein J. L., Anderson G. W., Brown M. S. Degradation of cationized low density lipoprotein and regulation of cholesterol metabolism in homozygous familial hypercholesterolemia fibroblasts. Proc Natl Acad Sci U S A. 1976 Sep;73(9):3178–3182. doi: 10.1073/pnas.73.9.3178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bayse G. S., Michaels A. W., Morrison M. The peroxidase-catalyzed oxidation of tyrosine. Biochim Biophys Acta. 1972 Sep 19;284(1):34–42. doi: 10.1016/0005-2744(72)90043-5. [DOI] [PubMed] [Google Scholar]
- Berliner J. A., Territo M. C., Sevanian A., Ramin S., Kim J. A., Bamshad B., Esterson M., Fogelman A. M. Minimally modified low density lipoprotein stimulates monocyte endothelial interactions. J Clin Invest. 1990 Apr;85(4):1260–1266. doi: 10.1172/JCI114562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brinton E. A., Oram J. F., Chen C. H., Albers J. J., Bierman E. L. Binding of high density lipoprotein to cultured fibroblasts after chemical alteration of apoprotein amino acid residues. J Biol Chem. 1986 Jan 5;261(1):495–503. [PubMed] [Google Scholar]
- Brown M. S., Goldstein J. L. A receptor-mediated pathway for cholesterol homeostasis. Science. 1986 Apr 4;232(4746):34–47. doi: 10.1126/science.3513311. [DOI] [PubMed] [Google Scholar]
- Chung B. H., Wilkinson T., Geer J. C., Segrest J. P. Preparative and quantitative isolation of plasma lipoproteins: rapid, single discontinuous density gradient ultracentrifugation in a vertical rotor. J Lipid Res. 1980 Mar;21(3):284–291. [PubMed] [Google Scholar]
- Foerder C. A., Shapiro B. M. Release of ovoperoxidase from sea urchin eggs hardens the fertilization membrane with tyrosine crosslinks. Proc Natl Acad Sci U S A. 1977 Oct;74(10):4214–4218. doi: 10.1073/pnas.74.10.4214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GROSS A. J., SIZER I. W. The oxidation of tyramine, tyrosine, and related compounds by peroxidase. J Biol Chem. 1959 Jun;234(6):1611–1614. [PubMed] [Google Scholar]
- Glomset J. A. The plasma lecithins:cholesterol acyltransferase reaction. J Lipid Res. 1968 Mar;9(2):155–167. [PubMed] [Google Scholar]
- Heider J. G., Boyett R. L. The picomole determination of free and total cholesterol in cells in culture. J Lipid Res. 1978 May;19(4):514–518. [PubMed] [Google Scholar]
- Heinecke J. W. Free radical modification of low-density lipoprotein: mechanisms and biological consequences. Free Radic Biol Med. 1987;3(1):65–73. doi: 10.1016/0891-5849(87)90040-2. [DOI] [PubMed] [Google Scholar]
- Heinecke J. W., Li W., Daehnke H. L., 3rd, Goldstein J. A. Dityrosine, a specific marker of oxidation, is synthesized by the myeloperoxidase-hydrogen peroxide system of human neutrophils and macrophages. J Biol Chem. 1993 Feb 25;268(6):4069–4077. [PubMed] [Google Scholar]
- Heinecke J. W., Li W., Francis G. A., Goldstein J. A. Tyrosyl radical generated by myeloperoxidase catalyzes the oxidative cross-linking of proteins. J Clin Invest. 1993 Jun;91(6):2866–2872. doi: 10.1172/JCI116531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henriksen T., Evensen S. A., Carlander B. Injury to cultured endothelial cells induced by low density lipoproteins: protection by high density lipoproteins. Scand J Clin Lab Invest. 1979 Jun;39(4):369–375. doi: 10.3109/00365517909106121. [DOI] [PubMed] [Google Scholar]
- Hessler J. R., Robertson A. L., Jr, Chisolm G. M., 3rd LDL-induced cytotoxicity and its inhibition by HDL in human vascular smooth muscle and endothelial cells in culture. Atherosclerosis. 1979 Mar;32(3):213–229. doi: 10.1016/0021-9150(79)90166-7. [DOI] [PubMed] [Google Scholar]
- Ho Y. K., Brown M. S., Goldstein J. L. Hydrolysis and excretion of cytoplasmic cholesteryl esters by macrophages: stimulation by high density lipoprotein and other agents. J Lipid Res. 1980 May;21(4):391–398. [PubMed] [Google Scholar]
- Hurst J. K., Barrette W. C., Jr Leukocytic oxygen activation and microbicidal oxidative toxins. Crit Rev Biochem Mol Biol. 1989;24(4):271–328. doi: 10.3109/10409238909082555. [DOI] [PubMed] [Google Scholar]
- Joschek H. I., Miller S. I. Photooxidation of phenol, cresols, and dihydroxybenzenes. J Am Chem Soc. 1966 Jul 20;88(14):3273–3281. doi: 10.1021/ja00966a019. [DOI] [PubMed] [Google Scholar]
- Jürgens G., Hoff H. F., Chisolm G. M., 3rd, Esterbauer H. Modification of human serum low density lipoprotein by oxidation--characterization and pathophysiological implications. Chem Phys Lipids. 1987 Nov-Dec;45(2-4):315–336. doi: 10.1016/0009-3084(87)90070-3. [DOI] [PubMed] [Google Scholar]
- Klebanoff S. J. Oxygen metabolism and the toxic properties of phagocytes. Ann Intern Med. 1980 Sep;93(3):480–489. doi: 10.7326/0003-4819-93-3-480. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Mackness M. I., Arrol S., Durrington P. N. Paraoxonase prevents accumulation of lipoperoxides in low-density lipoprotein. FEBS Lett. 1991 Jul 29;286(1-2):152–154. doi: 10.1016/0014-5793(91)80962-3. [DOI] [PubMed] [Google Scholar]
- Nagano Y., Arai H., Kita T. High density lipoprotein loses its effect to stimulate efflux of cholesterol from foam cells after oxidative modification. Proc Natl Acad Sci U S A. 1991 Aug 1;88(15):6457–6461. doi: 10.1073/pnas.88.15.6457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nomura K., Suzuki N., Matsumoto S. Pulcherosine, a novel tyrosine-derived, trivalent cross-linking amino acid from the fertilization envelope of sea urchin embryo. Biochemistry. 1990 May 15;29(19):4525–4534. doi: 10.1021/bi00471a004. [DOI] [PubMed] [Google Scholar]
- Oram J. F. Effects of high density lipoprotein subfractions on cholesterol homeostasis in human fibroblasts and arterial smooth muscle cells. Arteriosclerosis. 1983 Sep-Oct;3(5):420–432. doi: 10.1161/01.atv.3.5.420. [DOI] [PubMed] [Google Scholar]
- Oram J. F., Mendez A. J., Slotte J. P., Johnson T. F. High density lipoprotein apolipoproteins mediate removal of sterol from intracellular pools but not from plasma membranes of cholesterol-loaded fibroblasts. Arterioscler Thromb. 1991 Mar-Apr;11(2):403–414. doi: 10.1161/01.atv.11.2.403. [DOI] [PubMed] [Google Scholar]
- Oram J. F. Receptor-mediated transport of cholesterol between cultured cells and high-density lipoproteins. Methods Enzymol. 1986;129:645–659. doi: 10.1016/0076-6879(86)29096-5. [DOI] [PubMed] [Google Scholar]
- Parthasarathy S., Barnett J., Fong L. G. High-density lipoprotein inhibits the oxidative modification of low-density lipoprotein. Biochim Biophys Acta. 1990 May 22;1044(2):275–283. doi: 10.1016/0005-2760(90)90314-n. [DOI] [PubMed] [Google Scholar]
- Phillips M. C., Johnson W. J., Rothblat G. H. Mechanisms and consequences of cellular cholesterol exchange and transfer. Biochim Biophys Acta. 1987 Jun 24;906(2):223–276. doi: 10.1016/0304-4157(87)90013-x. [DOI] [PubMed] [Google Scholar]
- Rothblat G. H., Mahlberg F. H., Johnson W. J., Phillips M. C. Apolipoproteins, membrane cholesterol domains, and the regulation of cholesterol efflux. J Lipid Res. 1992 Aug;33(8):1091–1097. [PubMed] [Google Scholar]
- Steinberg D., Parthasarathy S., Carew T. E., Khoo J. C., Witztum J. L. Beyond cholesterol. Modifications of low-density lipoprotein that increase its atherogenicity. N Engl J Med. 1989 Apr 6;320(14):915–924. doi: 10.1056/NEJM198904063201407. [DOI] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warnick G. R. Enzymatic methods for quantification of lipoprotein lipids. Methods Enzymol. 1986;129:101–123. doi: 10.1016/0076-6879(86)29064-3. [DOI] [PubMed] [Google Scholar]
- Weisgraber K. H., Mahley R. W. Subfractionation of human high density lipoproteins by heparin-Sepharose affinity chromatography. J Lipid Res. 1980 Mar;21(3):316–325. [PubMed] [Google Scholar]