Abstract
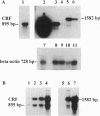
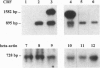
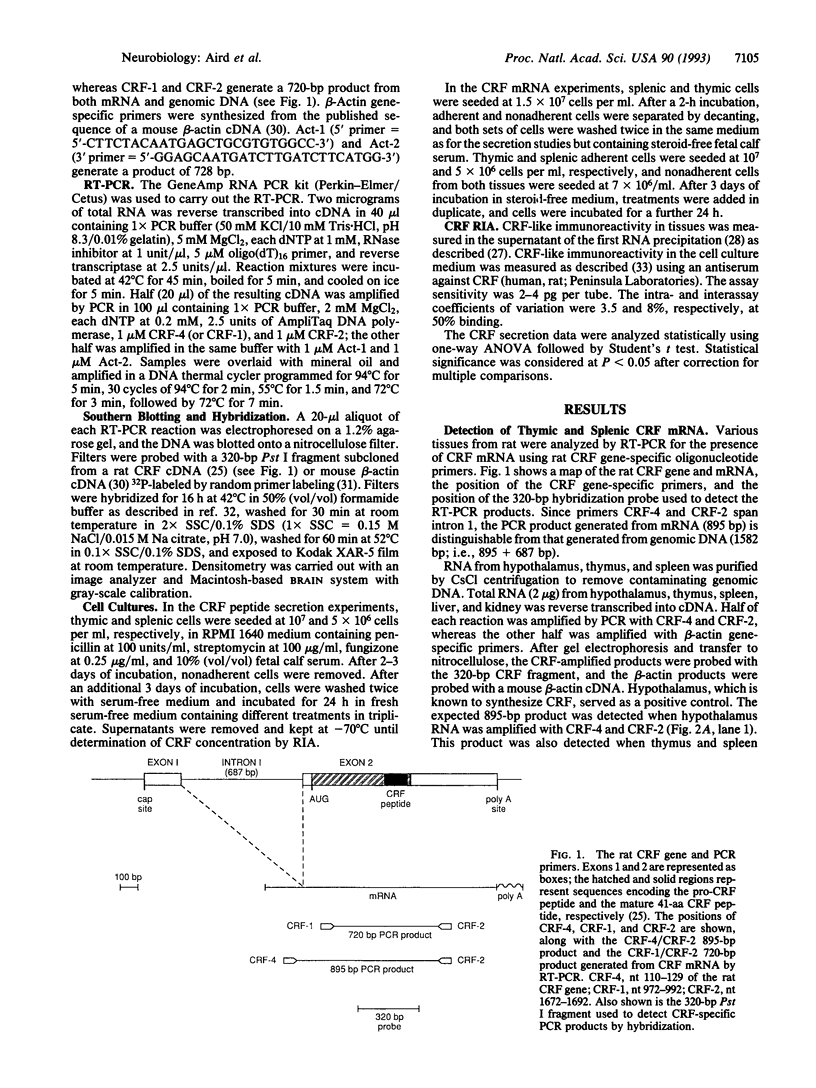
Corticotropin-releasing factor (CRF) initiates stress-induced immunosuppression via the hypothalamic-pituitary-adrenal axis. CRF has also been shown to have direct stimulatory and suppressive effects on immune cells. We have previously detected immunoreactive and bioactive CRF in the rat spleen and thymus. To determine if CRF is synthesized in these tissues, we analyzed rat spleen and thymus for the presence of CRF mRNA. RNA was reverse transcribed, and the resulting cDNA was amplified by the polymerase chain reaction with CRF gene-specific oligonucleotide primers. After Southern blotting and hybridization with an internal CRF gene probe, a product of the expected size was detected in the spleen, thymus, and hypothalamus (positive control) but not in liver or kidney (negative controls), indicating that CRF is synthesized in the spleen and thymus. Furthermore, CRF could be secreted from splenic and thymic adherent cells in culture. Secretion increased severalfold in response to nordihydroguaiaretic acid (NDGA), a lipoxygenase pathway inhibitor, whereas interleukin 1 had no effect, suggesting that regulation of CRF secretion may differ from that in the hypothalamus. CRF mRNA was detected in NDGA-stimulated thymic adherent cells and in both control and NDGA-stimulated splenic nonadherent cells. The finding that CRF is synthesized in the spleen and thymus suggests that locally synthesized "immune" CRF, acting as an autocrine or paracrine cytokine, may have direct regulatory effects on immune function.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abood M. E., Eberwine J. H., Erdelyi E., Evans C. J. Regulation of both preproenkephalin mRNA and its derived opioids by haloperidol--a method for measurement of peptides and mRNA in the same tissue extract. Brain Res Mol Brain Res. 1990 Aug;8(3):243–248. doi: 10.1016/0169-328x(90)90023-7. [DOI] [PubMed] [Google Scholar]
- Bateman A., Singh A., Kral T., Solomon S. The immune-hypothalamic-pituitary-adrenal axis. Endocr Rev. 1989 Feb;10(1):92–112. doi: 10.1210/edrv-10-1-92. [DOI] [PubMed] [Google Scholar]
- Cannon J. G., Tatro J. B., Reichlin S., Dinarello C. A. Alpha melanocyte stimulating hormone inhibits immunostimulatory and inflammatory actions of interleukin 1. J Immunol. 1986 Oct 1;137(7):2232–2236. [PubMed] [Google Scholar]
- Chirgwin J. M., Przybyla A. E., MacDonald R. J., Rutter W. J. Isolation of biologically active ribonucleic acid from sources enriched in ribonuclease. Biochemistry. 1979 Nov 27;18(24):5294–5299. doi: 10.1021/bi00591a005. [DOI] [PubMed] [Google Scholar]
- Dave J. R., Eiden L. E., Eskay R. L. Corticotropin-releasing factor binding to peripheral tissue and activation of the adenylate cyclase-adenosine 3',5'-monophosphate system. Endocrinology. 1985 Jun;116(6):2152–2159. doi: 10.1210/endo-116-6-2152. [DOI] [PubMed] [Google Scholar]
- Daynes R. A., Robertson B. A., Cho B. H., Burnham D. K., Newton R. Alpha-melanocyte-stimulating hormone exhibits target cell selectivity in its capacity to affect interleukin 1-inducible responses in vivo and in vitro. J Immunol. 1987 Jul 1;139(1):103–109. [PubMed] [Google Scholar]
- Feinberg A. P., Vogelstein B. A technique for radiolabeling DNA restriction endonuclease fragments to high specific activity. Anal Biochem. 1983 Jul 1;132(1):6–13. doi: 10.1016/0003-2697(83)90418-9. [DOI] [PubMed] [Google Scholar]
- Geenen V., Legros J. J., Franchimont P., Defresne M. P., Boniver J., Ivell R., Richter D. The thymus as a neuroendocrine organ. Synthesis of vasopressin and oxytocin in human thymic epithelium. Ann N Y Acad Sci. 1987;496:56–66. doi: 10.1111/j.1749-6632.1987.tb35746.x. [DOI] [PubMed] [Google Scholar]
- Grino M., Chrousos G. P., Margioris A. N. The corticotropin releasing hormone gene is expressed in human placenta. Biochem Biophys Res Commun. 1987 Nov 13;148(3):1208–1214. doi: 10.1016/s0006-291x(87)80261-9. [DOI] [PubMed] [Google Scholar]
- Handagama P., Rappolee D. A., Werb Z., Levin J., Bainton D. F. Platelet alpha-granule fibrinogen, albumin, and immunoglobulin G are not synthesized by rat and mouse megakaryocytes. J Clin Invest. 1990 Oct;86(4):1364–1368. doi: 10.1172/JCI114848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hargreaves K. M., Costello A. H., Joris J. L. Release from inflamed tissue of a substance with properties similar to corticotropin-releasing factor. Neuroendocrinology. 1989 May;49(5):476–482. doi: 10.1159/000125155. [DOI] [PubMed] [Google Scholar]
- Jain R., Zwickler D., Hollander C. S., Brand H., Saperstein A., Hutchinson B., Brown C., Audhya T. Corticotropin-releasing factor modulates the immune response to stress in the rat. Endocrinology. 1991 Mar;128(3):1329–1336. doi: 10.1210/endo-128-3-1329. [DOI] [PubMed] [Google Scholar]
- Karalis K., Sano H., Redwine J., Listwak S., Wilder R. L., Chrousos G. P. Autocrine or paracrine inflammatory actions of corticotropin-releasing hormone in vivo. Science. 1991 Oct 18;254(5030):421–423. doi: 10.1126/science.1925600. [DOI] [PubMed] [Google Scholar]
- Kavelaars A., Ballieux R. E., Heijnen C. J. Beta-endorphin secretion by human peripheral blood mononuclear cells: regulation by glucocorticoids. Life Sci. 1990;46(17):1233–1240. doi: 10.1016/0024-3205(90)90498-g. [DOI] [PubMed] [Google Scholar]
- Kavelaars A., Ballieux R. E., Heijnen C. J. The role of IL-1 in the corticotropin-releasing factor and arginine- vasopressin-induced secretion of immunoreactive beta-endorphin by human peripheral blood mononuclear cells. J Immunol. 1989 Apr 1;142(7):2338–2342. [PubMed] [Google Scholar]
- Kavelaars A., Berkenbosch F., Croiset G., Ballieux R. E., Heijnen C. J. Induction of beta-endorphin secretion by lymphocytes after subcutaneous administration of corticotropin-releasing factor. Endocrinology. 1990 Feb;126(2):759–764. doi: 10.1210/endo-126-2-759. [DOI] [PubMed] [Google Scholar]
- Keller S. E., Weiss J. M., Schleifer S. J., Miller N. E., Stein M. Stress-induced suppression of immunity in adrenalectomized rats. Science. 1983 Sep 23;221(4617):1301–1304. doi: 10.1126/science.6612346. [DOI] [PubMed] [Google Scholar]
- Linton E. A., McLean C., Nieuwenhuyzen Kruseman A. C., Tilders F. J., Van der Veen E. A., Lowry P. J. Direct measurement of human plasma corticotropin-releasing hormone by "two-site" immunoradiometric assay. J Clin Endocrinol Metab. 1987 May;64(5):1047–1053. doi: 10.1210/jcem-64-5-1047. [DOI] [PubMed] [Google Scholar]
- McGillis J. P., Park A., Rubin-Fletter P., Turck C., Dallman M. F., Payan D. G. Stimulation of rat B-lymphocyte proliferation by corticotropin-releasing factor. J Neurosci Res. 1989 Jul;23(3):346–352. doi: 10.1002/jnr.490230316. [DOI] [PubMed] [Google Scholar]
- Moll U. M., Lane B. L., Robert F., Geenen V., Legros J. J. The neuroendocrine thymus. Abundant occurrence of oxytocin-, vasopressin-, and neurophysin-like peptides in epithelial cells. Histochemistry. 1988;89(4):385–390. doi: 10.1007/BF00500641. [DOI] [PubMed] [Google Scholar]
- Munck A., Guyre P. M., Holbrook N. J. Physiological functions of glucocorticoids in stress and their relation to pharmacological actions. Endocr Rev. 1984 Winter;5(1):25–44. doi: 10.1210/edrv-5-1-25. [DOI] [PubMed] [Google Scholar]
- Petraglia F., Sawchenko P. E., Rivier J., Vale W. Evidence for local stimulation of ACTH secretion by corticotropin-releasing factor in human placenta. Nature. 1987 Aug 20;328(6132):717–719. doi: 10.1038/328717a0. [DOI] [PubMed] [Google Scholar]
- Potter E., Behan D. P., Fischer W. H., Linton E. A., Lowry P. J., Vale W. W. Cloning and characterization of the cDNAs for human and rat corticotropin releasing factor-binding proteins. Nature. 1991 Jan 31;349(6308):423–426. doi: 10.1038/349423a0. [DOI] [PubMed] [Google Scholar]
- Redei E., Branch B. J., Gholami S., Lin E. Y., Taylor A. N. Effects of ethanol on CRF release in vitro. Endocrinology. 1988 Dec;123(6):2736–2743. doi: 10.1210/endo-123-6-2736. [DOI] [PubMed] [Google Scholar]
- Singh V. K. Stimulatory effect of corticotropin-releasing neurohormone on human lymphocyte proliferation and interleukin-2 receptor expression. J Neuroimmunol. 1989 Aug;23(3):257–262. doi: 10.1016/0165-5728(89)90058-1. [DOI] [PubMed] [Google Scholar]
- Smith E. M., Hughes T. K., Jr, Hashemi F., Stefano G. B. Immunosuppressive effects of corticotropin and melanotropin and their possible significance in human immunodeficiency virus infection. Proc Natl Acad Sci U S A. 1992 Jan 15;89(2):782–786. doi: 10.1073/pnas.89.2.782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith E. M., Morrill A. C., Meyer W. J., 3rd, Blalock J. E. Corticotropin releasing factor induction of leukocyte-derived immunoreactive ACTH and endorphins. 1986 Jun 26-Jul 2Nature. 321(6073):881–882. doi: 10.1038/321881a0. [DOI] [PubMed] [Google Scholar]
- Stephanou A., Jessop D. S., Knight R. A., Lightman S. L. Corticotrophin-releasing factor-like immunoreactivity and mRNA in human leukocytes. Brain Behav Immun. 1990 Mar;4(1):67–73. doi: 10.1016/0889-1591(90)90007-d. [DOI] [PubMed] [Google Scholar]
- Sundar S. K., Becker K. J., Cierpial M. A., Carpenter M. D., Rankin L. A., Fleener S. L., Ritchie J. C., Simson P. E., Weiss J. M. Intracerebroventricular infusion of interleukin 1 rapidly decreases peripheral cellular immune responses. Proc Natl Acad Sci U S A. 1989 Aug;86(16):6398–6402. doi: 10.1073/pnas.86.16.6398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sundar S. K., Cierpial M. A., Kilts C., Ritchie J. C., Weiss J. M. Brain IL-1-induced immunosuppression occurs through activation of both pituitary-adrenal axis and sympathetic nervous system by corticotropin-releasing factor. J Neurosci. 1990 Nov;10(11):3701–3706. doi: 10.1523/JNEUROSCI.10-11-03701.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson R. C., Seasholtz A. F., Herbert E. Rat corticotropin-releasing hormone gene: sequence and tissue-specific expression. Mol Endocrinol. 1987 May;1(5):363–370. doi: 10.1210/mend-1-5-363. [DOI] [PubMed] [Google Scholar]
- Tokunaga K., Taniguchi H., Yoda K., Shimizu M., Sakiyama S. Nucleotide sequence of a full-length cDNA for mouse cytoskeletal beta-actin mRNA. Nucleic Acids Res. 1986 Mar 25;14(6):2829–2829. doi: 10.1093/nar/14.6.2829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsagarakis S., Gillies G., Rees L. H., Besser M., Grossman A. Interleukin-1 directly stimulates the release of corticotrophin releasing factor from rat hypothalamus. Neuroendocrinology. 1989 Jan;49(1):98–101. doi: 10.1159/000125096. [DOI] [PubMed] [Google Scholar]
- Ulisse S., Fabbri A., Dufau M. L. Corticotropin-releasing factor receptors and actions in rat Leydig cells. J Biol Chem. 1989 Feb 5;264(4):2156–2163. [PubMed] [Google Scholar]
- Vale W., Spiess J., Rivier C., Rivier J. Characterization of a 41-residue ovine hypothalamic peptide that stimulates secretion of corticotropin and beta-endorphin. Science. 1981 Sep 18;213(4514):1394–1397. doi: 10.1126/science.6267699. [DOI] [PubMed] [Google Scholar]
- Webster E. L., Tracey D. E., Jutila M. A., Wolfe S. A., Jr, De Souza E. B. Corticotropin-releasing factor receptors in mouse spleen: identification of receptor-bearing cells as resident macrophages. Endocrinology. 1990 Jul;127(1):440–452. doi: 10.1210/endo-127-1-440. [DOI] [PubMed] [Google Scholar]