Abstract
The diagnosis, investigation, and management of ovarian cancer are in a state of flux—balancing ever rapid advances in our understanding of its biology with 3 decades of clinical trials. Clinical trials that started with empirically driven selections have evolved in an evidence‐informed manner to gradually improve outcome. Has this improved understanding of the biology and associated calls to action led to appropriate changes in therapy? In this review, the authors discuss incorporating emerging data on biology, combinations, dose, and scheduling of new and existing agents with patient preferences in the management of women with ovarian cancer. Cancer 2015;121:3203–3211. © 2015 American Cancer Society.
Keywords: ovarian cancer, treatment, strategy, biology, biomarkers
Short abstract
The authors discuss the current understanding of the biology of ovarian cancer, diagnostic and treatment strategies, and high‐priority directions for investigation.
Changing Concepts for Systemic Therapy: The Challenge
The established standard strategy for treatment of advanced ovarian cancer (OC), up until recently, has been debulking surgery and 6 to 8 cycles of platinum/taxane‐based chemotherapy followed by surveillance for potential recurrence (Table 1).1, 2, 3 The success of this strategy depends on the skill of the surgeon and the extent of debulking, and perhaps on the intensity and delivery of chemotherapy.1 Unfortunately, all too often, initial therapy fails, and 3 of 4 women who initially present with advanced disease relapse.4 Subsequent sequential treatment strategies maximize quality and length of life5 but are not curative. Second‐line chemotherapy is still based on a crude but simple and effective algorithm, duration of the platinum‐free interval (PFI),6 whereby platinum sensitivity is defined as relapse occurring ≥6 months from the time of initial chemotherapy completion. Rechallenge with platinum doublet chemotherapy at this disease‐sensitive time is the standard of care worldwide, with 6 to 8 cycles of therapy prescribed followed in complete responders by a “watch‐and‐wait” period.7, 8, 9 The PFI has been used as a predictive measure of the likely response to subsequent platinum and nonplatinum therapy. The defined number of cycles was established initially, like most other indications for chemotherapy, on the concept of log‐cell kill from the Skipper‐Schobel‐Wilcox and Goldie‐Coldman models of cell growth kinetics10, 11 and was validated by a lack of improvement in overall survival (OS) from increasing the number of cycles.12 Chemotherapy primarily damages rapidly proliferating cells13 and, thus, is effective only in that proportion of tumor cells within the cell growth cycle. The best response to chemotherapy is often a reduction in tumor volume; however, not all clonal cells may be eradicated, and mutations that allow cancer cells to survive, accumulated during therapy, may induce subsequent drug resistance. Recurrence may also be the result of reactivating tumor‐initiating cells or tumor cells with stem‐like properties that are not targeted by standard chemotherapy.
Table 1.
Prioritization Questions in the Management of Ovarian Cancer and Current Proposed Strategies
| Question | Current Practice | Remaining Challenges | Current Proposed Strategy |
|---|---|---|---|
| Should we continue adjuvant chemotherapy in type 1 OC? | Carboplatin/paclitaxel for OC, all histologic subtypes | To determine the benefit of chemotherapy in low‐grade serous, low‐grade endometrioid, mucinous, and clear cell carcinomas | • Meta‐analyses from phase 2/3 clinical trials • Clinical database for patient follow‐up • Expert pathology for histology diagnosis • Trials need to be designed to take into consideration the different histology subtypes • Control arms would potentially incorporate a no‐treatment arm |
| What is the importance of “maintenance” therapy in HGSOC as part of the treatment plan? | Adjuvant maintenance therapy is for a predefined period | Treatment duration in maintenance may be arbitrary and needs precision, balancing safety, efficacy with cost and effectiveness | • Stratification on BRCA1/2 status • Clinical trials investigating the timing of treatment strategy |
| Choice of maintenance between antiangiogenics and PARP inhibitors needs further definition | • Better characterization of the dominant tumor feature that needs to be targeted at the time of recurrence with imaging, ctDNA, and tumor tissue profiling | ||
| • Integrated biomarkers: scheduled and defined in the laboratory manual as part of the clinical trial | |||
| • Clinical database for patient follow‐up | |||
| How we should decide on sequence therapy? | The platinum‐free interval (PFI) is used as an algorithm to define subsequent therapy | PFI is subject to variability based on follow‐up and imaging and is not based on clear biologic principles | • Validate algorithms in a consistent manner, particularly after maintenance therapy |
| PFI may be modulated by therapy, making interpretation of PFI for subsequent therapy more challenging | • Correlate effect of PFI with tumor biology using tissue assessment and functional imaging |
Abbreviations: BRCA1/2, breast cancer susceptibility genes 1 and 2; CT, computed tomography; ctDNA, circulating tumor deoxyribonucleic acid; HGSOC, high‐grade serous ovarian cancer; OC, ovarian cancer; PARP, poly (ADP‐ribose) polymerase.
Recurrent disease follows a frequent relapse‐response pattern for a modest time before becoming resistant to treatment. Clinically, this escalating resistance manifests as successively shorter disease‐free intervals with each subsequent line of therapy. At the time of platinum‐resistance, different monochemotherapies are used until patients develop disease progression or unacceptable toxicity.1, 14 Continuing monochemotherapy in this population when there is clinical benefit may be important for sustained response15 despite the high likelihood of unavoidable relapse or progression. In effect, the current paradox is to stop treatment during initial response and to resume/continue the same treatment while disease becomes increasingly more resistant. Unfortunately, the patient will ultimately die of her disease with a median OS of approximately 12 months after the development of platinum resistance.
This existing treatment strategy in OC has been largely driven by activity in high‐grade serous (high‐grade serous OC [HGSOC]) and endometrioid (high‐grade serous endometrial cancer [HGSEC]) histologies, but all epithelial types have been treated uniformly for “want of something better” (Fig. 1, Table 1). There are 5 disparate histologic types of OC that have a diverse genomic landscape with different natural histories and patterns of response to therapy.16, 17
Figure 1.
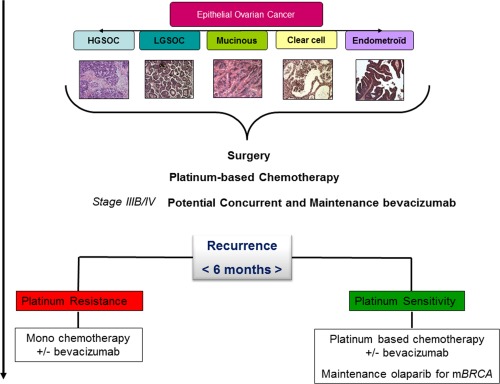
Current treatment strategy in ovarian cancer is illustrated. HGSOC, high‐grade serous ovarian cancer; LGSOC, low‐grade serous ovarian cancer; mBRCA, breast cancer gene mutation.
It is now time to demand evidence of benefit in each of the OC subtypes and to challenge the dogma of cytotoxic chemotherapy for all types of OC.16, 18 Chemoresistance is consistently reported in type I OC, admittedly mostly based on smaller, retrospective, single‐institution studies. Women with clear cell, mucinous, and low‐grade serous tumors are routinely offered standard chemotherapy,19 more for want of an alternative strategy or for fear of under‐treating than belief that the treatment is effective. Perhaps we should abandon current adjuvant chemotherapy for specific OC subtypes in which the evidence is inadequate. What is the benefit of standard adjuvant chemotherapy that we should expect in confirmed clear cell20, 21, 22 or low‐grade serous23, 24 cancer after complete debulking surgery, either at an early stage or even at an advanced stage? In this setting, in which there is insufficient level 1 evidence of true benefit from cytotoxic chemotherapy, perhaps well designed clinical trials should be the new standard, potentially with a control no‐treatment arm. This would allow the detection of an improvement in efficacy, or not. The paradox and potentially ethical dilemma remains that patients often have to satisfy prior “standards” of chemotherapy to be eligible for clinical trials, standards that apply because all epithelial subtypes were grouped together; standards that perhaps today would not meet the rigorous level 1 evidence base required by patients, clinicians, and regulators.
Platinum‐based adjuvant chemotherapy remains the mainstay of systemic treatment for HGSOC/HGSEC. Germline and somatic breast cancer gene (BRCA) status appears to be prognostic and increasingly predictive and has an impact at least on short‐term to moderate‐term clinical outcomes. Future HGSOC trials should stratify for this biologically relevant characteristic as a baseline requirement. Similarly, the option of radiation therapy for selected histologic types needs to be revisited.25, 26 Radiotherapy typically is used for palliation in patients with HGSOC but may be beneficial in those with clear cell OC, as suggested by recent retrospective studies.21, 27 Hormone therapy, which is rarely used in HGSOC, may be a useful and safe strategy for some specific histology subgroups. such as endometrioid or low‐grade serous.28, 29 Previous clinical trials of hormones have produced modest response rates overall; however, they have consistently identified a small subset of patients who respond very well to endocrine therapy,30 and we need to understand what is unique about these responders. Targeted therapy may also be appropriate in specific histologic subtypes, such as mitogen‐activated protein kinase kinase (MEK) inhibitors in low grade serous OC. On the basis of activity observed with selimetinib in a phase 2 trial dedicated to low‐grade serous OC,31 phase 3 studies are ongoing to assess MEK inhibitors in this histologic subgroup (the MEK Inhibitor in Low‐Grade Serous Ovarian Cancer [MILO] trial, national clinical trial [NCT] 01849874; and Gynecologic Oncology Group Trial 281 [GOG281], NCT02101788).
Incorporating Maintenance and Sequential Therapy
Current adjuvant treatment in HGSOC is discontinued after 6 to 8 cycles, although it is clear that cancer cells persist, judging by recurrence patterns. This is based on modeling effects of chemotherapy and tumor cell kill and on toxicity, which is often cumulative and sometimes irreversible. Maintenance therapy was initially proposed to delay subsequent progression through the continuation of treatment with the expectation that delaying progression could translate into an improved overall duration of survival.32 Maintenance treatment has been successfully validated in other malignancies with a high‐risk of recurrence, such as breast cancer (trastuzumab and hormone therapy). Maintaining a lower level of disease burden may be a way to improve disease control and, thus, the time to progression. Residual disease after primary debulking surgery in OC remains one of the most powerful prognostic and predictive factors for OS.33 Gompertzian modeling demonstrates increased percentages of necrotic and hypoxic tissues within the tumor with tumor growth, both of which are major factors in drug resistance.34 This aligns with the drive to understand the tumor microenvironment and cancer cell biology to improve disease control. HGSOC is characterized by genomic instability,35 with genomic and transcriptomic heterogeneity in time and space that result in a broad variety of—and potentially functional—lesion‐specific deregulation of cellular pathways.36 Attempting to lower tumor burden with maintenance therapy may limit the number of clones and acquisition of additional mutations that engender therapeutic resistance. The tumor‐initiating cell theory further supports a maintenance strategy by attempting to silence tumorigenic potential.37
This closely mirrors the use of human immunodeficiency virus (HIV) antiretroviral treatments in an attempt to maintain low viral loads, thus avoiding viral resistance.38 The short life cycle of HIV and the high error rate cause the virus to mutate very rapidly, resulting in high genetic variability.39 Thus, the more active copies of the virus, the greater the possibility that one resistant to antiretroviral drugs will arise,40 accentuating the importance of maximal viral load suppression at treatment initiation. After virus genotyping and HIV drug‐resistance testing, a 3‐drug combination is introduced without a “drug holiday.”
However, in patients with HGSOC, continuing platinum‐based chemotherapy has produced no evidence of long‐term benefit, may contribute to chemoresistance, and leads to cumulative toxicity.41 The effect of maintenance paclitaxel remains controversial,42, 43, 44 and several modest sized trials have demonstrated some potential PFS benefit without improving OS45, 46 but with significant cumulative toxicity. Results from the completed GOG212 trial (NCT00108745), which compared paclitaxel with polyglutamate paclitaxel or observation after standard treatment, may resolve this question. Maintenance treatment with erlotinib, an epidermal growth factor receptor‐targeted therapeutic,47 or anticancer antigen 125 (anti‐CA 125) antibodies, such as abagovomab,48 failed to have an impact on the outcomes of women who had no clinical evidence of disease after standard therapy.49, 50 New expectations have surfaced for maintenance therapy in HGSOC that incorporates complementary mechanisms of action beside standalone chemotherapy, including targeting deficiencies in the homologous recombination repair (HRR) pathway and angiogenesis. Deoxyribonucleic acid (DNA) repair defects consistent with HRR dysfunction are referred to as the “BRCAness phenotype,” a potential HGSOC Achilles heel.51 Two randomized phase 2 trials testing the poly (ADP‐ribose) polymerase (PARP) inhibitor olaparib as maintenance therapy after platinum‐based chemotherapy (prescribed as long as patients were benefiting from therapy) have demonstrated a marked improvement in PFS for patients with recurrent, platinum‐sensitive HGSOC, particularly for those with BRCA mutations.52, 53
Targeting the tumor microenvironment through the inhibition of tumor‐associated angiogenesis has been also an effective strategy supporting the maintenance‐treatment hypothesis. Continuation of bevacizumab alone in the adjuvant setting for 12 or 15 months after chemotherapy first‐line chemotherapy in combination with bevacizumab produced a modest but significant PFS benefit in 2 phase 3 studies (GOG218 and ICON7 [a Gynecologic Cancer Intergroup trial]).54, 55 It is noteworthy that, based on a planned survival analysis of the ICON7 trial, in the high‐risk subgroup (patients with suboptimally debulked stage III/IV disease and nonsurgical patients), a significant 9.4‐month improvement in median OS was observed.56 Two phase 3 trials with bevacizumab prescribed until disease progression or intolerance—one in the platinum‐sensitive setting (the OCEANS [Ovarian Cancer Comparing Efficacy and Safety of Chemotherapy and Antiangiogenic Therapy in Platinum‐Sensitive Recurrent Disease] trial), in which bevacizumab was combined with carboplatin/gemcitabine followed by bevacizumab alone, and one in the platinum‐resistant setting (the AURELIA [A Study of Avastin Added to Chemotherapy in Patients With Platinum‐Resistant Ovarian Cancer] trial), in which bevacizumab was combined with monochemotherapy—demonstrated a significant benefit for maintenance on PFS, but no OS benefit was observed in either study.57, 58 The benefit of adding and continuing an antiangiogenic agent was further confirmed with pazopanib, nintedanib, and cediranib.59, 60, 61 Data from the German Gynecologic Oncology Study Group‐Ovarian Cancer 16 (AGO‐OVAR16) phase 3 trials in women with advanced OC who had not progressed after front‐line chemotherapy indicated a statistically significant PFS benefit for patients who received pazopanib versus placebo for up to 24 months59 (P = .0021). AGO‐OVAR12 is another phase 3 study of antiangiogenic therapy evaluating nintedanib as front‐line therapy for women with advanced OC.60 The nintedanib arm exhibited a mild improvement in PFS. This PFS benefit was higher among patients from the low‐risk group of women who had small residual tumors after surgery. Cediranib given concurrently with platinum‐based chemotherapy in ICON6 and continued as maintenance for women in first platinum‐sensitive recurrence significantly improved PFS by 3.2 months and improved OS by 2.7 months.61 However, the sample size in that trial had to be reconfigured midway through the study, because the development of cedarinib had been discontinued. Despite this growing body of evidence, there remains a discrepancy in antiangiogenic use across the world. Defining the optimal therapy, schedule, and sequence of antiangiogenics is a priority for OC therapy standardization.
When Should Effective Treatment be Introduced, Interrupted, or Changed?
Sequential therapeutic interventions may maintain and consolidate the initial treatment response. This can be accomplished using a multifaceted clinical strategy based on tumor biology (Fig. 2).
Figure 2.
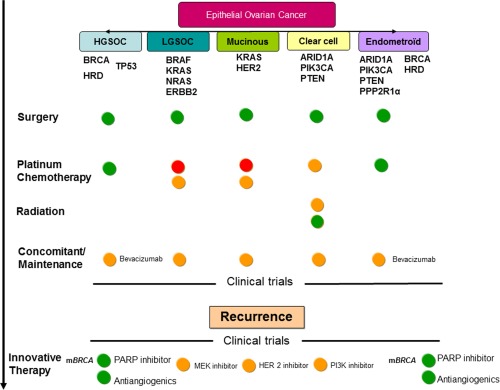
Treatment evolution based on biology and evidence is illustrated. Green circles indicate data evidence for treatment use; orange circles, lack of validation to support treatment use; red circles, no evidence for treatment use. ARID1A indicates AT‐rich interactive domain 1A; BRAF, v‐raf murine sarcoma viral oncogene homolog B1; HER2, human epidermal growth factor receptor 2; HGSOC, high‐grade serous ovarian cancer; HRD, homologous repair deficiency; KRAS, Kirsten rat sarcoma viral oncogene homolog; LGSOC, low‐grade serous ovarian cancer; mBRCA, breast cancer susceptibility gene mutation; MEK, mitogen extra cellular signal‐regulated kinase; NRAS, neuroblastoma RAS viral oncogene homolog; PARP, poly (ADP‐ribose) polymerase; PIK3CA, phosphoinositide‐3‐kinase, catalytic, α polypeptide; PPP2R1α, protein phosphatase 2, subunit A, α isoform; PTEN, phosphatase and tensin homolog; TP53, tumor protein p53.
Therapeutic strategies
Adding a third chemotherapy drug to standard chemotherapy or high‐dose, sequential chemotherapy increases toxicity but not disease control.62, 63, 64 Therefore, adding cytotoxic chemotherapy is not the answer. Combination treatment strategies should be based on biologic rationale and validated in rigorous clinical trials: for example, a test of combination therapy that directly or indirectly silences driver targets, such as HRR or tumor protein 53 (TP53).65 Targeting BRCA dysfunction is a validated approach for HGSOC, as is targeting TP53. Further investigations are also needed to dissect the clinical impact of diverse mutations in these driver genes, because there is probably variation in the functional consequences from different mutations in candidate genes.
Microenvironment plasticity is central to success and persistence of benefit from therapeutic intervention, as shown by the data available on bevacizumab. Antiangiogenics have demonstrated benefit at the time of persistent/recurrent disease, perhaps of greater benefit than when used in combination with chemotherapy. The microenvironment also includes stromal and immune events. There is growing evidence that OC is immunogenic and immunotrophic. These findings argue for close examination of the new immunotherapy options, with important consideration focused on clearly defining biomarkers that are reliably and reproducibly predictive of response to treatment.66 Immune targeting strategies in combination with therapy or sequentially as consolidation would be rational and are being evaluated.
Treatment timing
The combination/sequential approach used in HIV may not be clinically meaningful in OC and could lead to increased toxicity. Time and sequence specificity of treatment, based on the pharmacokinetics and pharmacodynamics of each drug, should be considered (Table 1). Preclinical data and drug pharmacology may allow sequential dosing to improve efficacy and reduce toxicity. For example, a phase 2 randomized clinical trials incorporating olaparib with carboplatin and paclitaxel (in combination and sequentially) have demonstrated increased toxicity during combination therapy, which required reductions in the dose intensity of carboplatin and olaparib, without improved effectiveness during the combination phase.53 In contrast, sequential therapy was simpler, well tolerated, and improved PFS.52 However, combined platinum‐taxane chemotherapy is probably better than sequential therapy and may be associated with a PFS advantage67. This emphasizes the importance exploring synergy caused by mechanisms of drug action or pharmacokinetics in determining treatment timing.
Treatment timing in oncology remains an important issue. Upfront maintenance therapy would appear to be the most favorable period during which to invest and may have the greatest potential, because the fewest resistant or dormant tumor cells are present. Tools are needed to identify persistent subclonal populations after the completion of initial therapy to better identify those women who are more likely relapse and to differentiate them from the women who may remain “free from relapse” without need for further treatment. Monitoring techniques for circulating cancer cells68 or circulating tumor DNA69 may impact our definitions of response and guide subsequent therapies used in the maintenance setting. Circulating tumor DNA is being assessed in different disease types,70 not just as a marker of recurrence but also as a marker of resistant subclones.71, 72
Imaging and CA 125 monitoring are the diagnostic modalities that currently determine the timing of new therapeutic interventions. The question of when to stop treatment in the maintenance setting—when a tumor progresses according Response Evaluation Criteria in Solid Tumors, at the time of clinical progression, or after an arbitrary period—remains to be defined. Several issues endure with the use of CA 125 monitoring. It may not track response or progression, optimally depending on the treatment used73; lead‐time bias is a confounding variable; and, finally, the attrition or attenuation of CA 125 rise over time limits its accuracy as a quantitative predictor and, consequently, the ability to make therapeutic choices based on the maker alone.74 Dynamic functional imaging should be explored in OC subtypes to guide treatment, provided there is sufficient sensitivity and specificity. Dynamic imaging can be expensive; therefore, validation with tissue biomarkers of early recurrence should be prioritized. The evaluation of techniques like positron emission tomography‐magnetic resonance imaging is critical, along with objective assessment of their potential for predicting early treatment failure75 to allow the discontinuation of ineffective, potentially toxic, and almost certainly expensive therapies.
Treatment selection
Comprehensive clinical assessment that incorporates performance status, previous treatments, and type of treatments is essential for making treatment decisions. The PFI remains an accepted predictive algorithm for the choice of next treatment in practice and for clinical trials (Table 1).76 However, the emergence of a maintenance approach scrambles this initial definition in terms of the time to progression by prolonging the PFI through maintenance therapy. What is the difference between a relapse within 5 months after standard adjuvant carboplatin plus paclitaxel chemotherapy and a relapse within 7 months by adding bevacizumab to the same chemotherapy? There is no clear answer, but the patient will be treated differently according to PFI criteria, because as she will be considered platinum‐resistant in the former case, but not in the latter. Thus, biologic precision is needed to replace the currently used algorithm of PFI, which has the advantage of simplicity and ease of use but remains arbitrary and subject to variability based on intervals for follow‐up or imaging (Table 1). The potential for measuring drug‐induced, dynamic rewiring of signaling pathways provides the opportunity for novel sequential and time‐dependent approaches.77 Consequently, there is a growing practice of performing repeat biopsies. Molecular profiling to help match patients to specific targeted therapeutics is an under‐evaluated resource in OC.78 A retrospective, but as yet unvalidated strategy has identified a predictive response signature to the antiangiogenic bevacizumab, which has resulted in the classification of a novel type of immune signature.79 Molecular subclassifications of HGSOC obtained from gene expression signatures16, 80 suggest that patients with the mesenchymal subtype may have a sustained benefit from bevacizumab.81 Those retrospective, yet to be validated analyses have generated hypotheses that have to be evaluated prospectively to be proven clinically useful. Currently, profiling objectives are toward the identification and validation of biomarkers in prospective clinical trials.82 With advancing technology, costs should become less prohibitive and may be augmented by the development of customized, subtype‐specific panels for defined populations. This will require integration with access to targeted agents and clinical trials. This also will require clinical trials to develop and incorporate outcomes and endpoints that are relevant and meaningful for patients.83, 84
Integration of Therapeutic Approaches
The therapeutic approach in OC includes surgical intervention, which remains the mainstay of treatment alongside appropriate systemic therapy. Ongoing efforts around the world are investigating the relevance of timing of surgical intervention at initial diagnosis and the value of surgery at recurrence. Furthermore, to optimize treatment timing and strategy, bioinformatics and mathematical modeling approaches are exploring research data on the relation and communication between cellular and extracellular signaling pathways.77 These models can be context‐dependent and must integrate tumor type specificity to determine pathway relevance.85, 86
Conclusion
The treatment of ovarian cancer is on the verge of a major change that will propel improvement in outcomes.87 Recent developments emphasize that a multipronged approach is warranted, integrating genomics, subtype‐specific maintenance therapy, and other directions that will be used alongside increasingly sensitive disease‐detection tools. Better definition of treatment timing may further allow exploitation of additional benefits of surgery as the role of surgery remains central to OC treatment. Taken together, this will allow us to build on our understanding of the heterogeneity of OC to tailor treatment. Future therapeutic strategy should target genetics, microenvironment, and sequence schedule to keep cancer cells from developing resistance. To move forward, we have to take some bold steps based on biology, require objective evidence of benefit, and reflect on experience from other cancers and diseases. It is important to constantly re‐evaluate data and expect that, for therapy to be considered “standard,” it has to demonstrate objective, clinically meaningful, as well as statistically significant benefit. The timing is right for a paradigm shift that will require clinicians to have the courage and confidence to not prescribe treatments without evidence of benefit. This will necessitate optimizing surgery and appropriate evidence‐based recommendations for each subtype of ovarian cancer. This therapeutic direction has to be supported by excellence and precision in pathology, molecular profiling, and imaging. In equal measure, it will require open dialogue with patients and being honest about treatments that have evidence of benefit and, especially, those that do not.
FUNDING SUPPORT
No specific funding was disclosed.
CONFLICT OF INTEREST DISCLOSURES
The authors made no disclosures.
REFERENCES
- 1. Jayson GC, Kohn EC, Kitchener HC, Ledermann JA. Ovarian cancer. Lancet. 2014;384:1376‐1388. [DOI] [PubMed] [Google Scholar]
- 2. Muggia FM, Braly PS, Brady MF, et al. Phase III randomized study of cisplatin versus paclitaxel versus cisplatin and paclitaxel in patients with suboptimal stage III or IV ovarian cancer: a Gynecologic Oncology Group study. J Clin Oncol. 2000;18:106‐115. [DOI] [PubMed] [Google Scholar]
- 3. Piccart MJ, Bertelsen K, James K, et al. Randomized intergroup trial of cisplatin‐paclitaxel versus cisplatin‐cyclophosphamide in women with advanced epithelial ovarian cancer: 3‐year results. J Natl Cancer Inst. 2000;92:699‐708. [DOI] [PubMed] [Google Scholar]
- 4. Coleman RL, Monk BJ, Sood AK, Herzog TJ. Latest research and treatment of advanced‐stage epithelial ovarian cancer. Nat Rev Clin Oncol. 2013;10:211‐224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Friedlander ML, Stockler MR, Butow P, et al. Clinical trials of palliative chemotherapy in platinum‐resistant or ‐refractory ovarian cancer: time to think differently [letter]? J Clin Oncol. 2013;31:2362. [DOI] [PubMed] [Google Scholar]
- 6. Colombo N, Gore M. Treatment of recurrent ovarian cancer relapsing 6–12 months post platinum‐based chemotherapy. Crit Rev Oncol Hematol. 2007;64:129‐138. [DOI] [PubMed] [Google Scholar]
- 7. Parmar MK, Ledermann JA, Colombo N, et al. Paclitaxel plus platinum‐based chemotherapy versus conventional platinum‐based chemotherapy in women with relapsed ovarian cancer: the ICON4/AGO‐OVAR‐2.2 trial. Lancet. 2003;361:2099‐2106. [DOI] [PubMed] [Google Scholar]
- 8. Pfisterer J, Plante M, Vergote I, et al. Gemcitabine plus carboplatin compared with carboplatin in patients with platinum‐sensitive recurrent ovarian cancer: an intergroup trial of the AGO‐OVAR, the NCIC CTG, and the EORTC GCG. J Clin Oncol. 2006;24:4699‐4707. [DOI] [PubMed] [Google Scholar]
- 9. Pujade‐Lauraine E, Wagner U, Aavall‐Lundqvist E, et al. Pegylated liposomal doxorubicin and carboplatin compared with paclitaxel and carboplatin for patients with platinum‐sensitive ovarian cancer in late relapse. J Clin Oncol. 2010;28:3323‐3329. [DOI] [PubMed] [Google Scholar]
- 10. Skipper HE. Kinetics of mammary tumor cell growth and implications for therapy. Cancer. 1971;28:1479‐1499. [DOI] [PubMed] [Google Scholar]
- 11. Goldie JH, Coldman AJ. A mathematic model for relating the drug sensitivity of tumors to their spontaneous mutation rate. Cancer Treat Rep. 1979;63:1727‐1733. [PubMed] [Google Scholar]
- 12. Bertelsen K, Jakobsen A, Stroyer I, et al. A prospective randomized comparison of 6 and 12 cycles of cyclophosphamide, Adriamycin and cisplatin in advanced epithelial ovarian cancer: a Danish Ovarian Study Group trial (DACOVA). Gynecol Oncol. 1993;49:30‐36. [DOI] [PubMed] [Google Scholar]
- 13. Corrie PG, Pippa G. Cytotoxic chemotherapy: clinical aspects. Medicine. 2008;36:24‐28. [Google Scholar]
- 14. Gordon AN, Fleagle JT, Guthrie D, Parkin DE, Gore ME, Lacave AJ. Recurrent epithelial ovarian carcinoma: a randomized phase III study of pegylated liposomal doxorubicin versus topotecan. J Clin Oncol. 2001;19:3312‐3322. [DOI] [PubMed] [Google Scholar]
- 15. Andreopoulou E, Gaiotti D, Kim E, et al. Pegylated liposomal doxorubicin HCL (PLD; Caelyx/Doxil): experience with long‐term maintenance in responding patients with recurrent epithelial ovarian cancer. Ann Oncol. 2007;18:716‐721. [DOI] [PubMed] [Google Scholar]
- 16. Cancer Genome Atlas Research Network . Integrated genomic analyses of ovarian carcinoma. Nature. 2011;474:609‐615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Sung PL, Chang YH, Chao KC, Chuang CM; Task Force on Systematic Review and Meta‐Analysis of Ovarian Cancer . Global distribution pattern of histological subtypes of epithelial ovarian cancer: a database analysis and systematic review. Gynecol Oncol. 2014;133:147‐154. [DOI] [PubMed] [Google Scholar]
- 18. Bookman MA, Gilks CB, Kohn EC, et al. Better therapeutic trials in ovarian cancer [serial online]. J Natl Cancer Inst. 2014;106:dju029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Despierre E, Yesilyurt BT, Lambrechts S, et al. Epithelial ovarian cancer: rationale for changing the 1‐fits‐all standard treatment regimen to subtype‐specific treatment. Int J Gynecol Cancer. 2014;24:468‐477. [DOI] [PubMed] [Google Scholar]
- 20. Friedlander M, Russell K, Millis SZ, et al. Molecular profiling of clear cell ovarian carcinoma [abstract]. J Clin Oncol. 2014;32(15 suppl). Abstract 5516. [Google Scholar]
- 21. Tan D, Rye T, Barrie C, et al. Analysis of outcomes in patients (pts) with recurrent ovarian clear cell carcinoma (ROCCC): time to rethink our approach to treatment [abstract]. J Clin Oncol. 2014;32(15 suppl). Abstract 5548. [Google Scholar]
- 22. Hoskins PJ, Le N, Gilks B, et al. Low‐stage ovarian clear cell carcinoma: population‐based outcomes in British Columbia, Canada, with evidence for a survival benefit as a result of irradiation. J Clin Oncol. 2012;30:1656‐1662. [DOI] [PubMed] [Google Scholar]
- 23. Gourley C, Farley J, Provencher DM, et al. Gynecologic Cancer Intergroup (GCIG) consensus review for ovarian and primary peritoneal low‐grade serous carcinomas. Int J Gynecol Cancer. 2014;24(9 suppl 3):S9‐S13. [DOI] [PubMed] [Google Scholar]
- 24. Gershenson DM, Sun CC, Bodurka D, et al. Recurrent low‐grade serous ovarian carcinoma is relatively chemoresistant. Gynecol Oncol. 2009;114:48‐52. [DOI] [PubMed] [Google Scholar]
- 25. Thomas G. Revisiting the role of radiation treatment for non‐serous subtypes of epithelial ovarian cancer [serial online]. Am Soc Clin Oncol Educ Book. 2013. doi: 10.1200/EdBook_AM.2013.3.e205. [DOI] [PubMed] [Google Scholar]
- 26. Macrie BD, Strauss JB, Helenowski IB, et al. Patterns of recurrence and role of pelvic radiotherapy in ovarian clear cell adenocarcinoma. Int J Gynecol Cancer. 2014;24:1597‐1602. [DOI] [PubMed] [Google Scholar]
- 27. Tan DS, Rye T, Barrie C, et al. Beneficial effect of adjuvant chemotherapy and whole abdominal or pelvic radiotherapy (WAPRT) on progression free and overall survival [abstract]. Ann Oncol. 2014;25(suppl 4):iv306. Abstract 878PD. [Google Scholar]
- 28. Pan Y, Kao MS. Endometrioid ovarian carcinoma benefits from aromatase inhibitors: case report and literature review. Curr Oncol. 2010;17:82‐85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Gershenson DM, Sun CC, Iyer RB, et al. Hormonal therapy for recurrent low‐grade serous carcinoma of the ovary or peritoneum. Gynecol Oncol. 2012;125:661‐666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Modugno F, Laskey R, Smith AL, Andersen CL, Haluska P, Oesterreich S. Hormone response in ovarian cancer: time to reconsider as a clinical target? Endocr Relat Cancer. 2012;19:R255‐R279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Farley J, Brady WE, Vathipadiekal V, et al. Selumetinib in women with recurrent low‐grade serous carcinoma of the ovary or peritoneum: an open‐label, single‐arm, phase 2 study. Lancet Oncol. 2013;14:134‐140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Markman M. Maintenance chemotherapy: an evolving and increasingly acceptable strategy in cancer management. Curr Oncol Rep. 2010;12:349‐351. [DOI] [PubMed] [Google Scholar]
- 33. du Bois A, Reuss A, Pujade‐Lauraine E, Harter P, Ray‐Coquard I, Pfisterer J. Role of surgical outcome as prognostic factor in advanced epithelial ovarian cancer: a combined exploratory analysis of 3 prospectively randomized phase 3 multicenter trials: by the Arbeitsgemeinschaft Gynaekologische Onkologie Studiengruppe Ovarialkarzinom (AGO‐OVAR) and the Groupe d'Investigateurs Nationaux Pour les Etudes des Cancers de l'Ovaire (GINECO). Cancer. 2009;115:1234‐1244. [DOI] [PubMed] [Google Scholar]
- 34. Shannon AM, Bouchier‐Hayes DJ, Condron CM, Toomey D. Tumor hypoxia, chemotherapeutic resistance and hypoxia‐related therapies. Cancer Treat Rev. 2003;29:297‐307. [DOI] [PubMed] [Google Scholar]
- 35. Chene G, Tchirkov A, Pierre‐Eymard E, et al. Early telomere shortening and genomic instability in tubo‐ovarian preneoplastic lesions. Clin Cancer Res. 2013;19:2873‐2882. [DOI] [PubMed] [Google Scholar]
- 36. Hoogstraat M, de Pagter MS, Cirkel GA, et al. Genomic and transcriptomic plasticity in treatment‐naive ovarian cancer. Genome Res. 2014;24:200‐211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Zhang S, Balch C, Chan MW, et al. Identification and characterization of ovarian cancer‐initiating cells from primary human tumors. Cancer Res. 2008;68:4311‐4320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Glickman MS, Sawyers CL. Converting cancer therapies into cures: lessons from infectious diseases. Cell. 2012;148:1089‐1098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Perelson AS, Neumann AU, Markowitz M, Leonard JM, Ho DD. HIV‐1 dynamics in vivo: virion clearance rate, infected cell life‐span, and viral generation time. Science. 1996;271:1582‐1586. [DOI] [PubMed] [Google Scholar]
- 40. Schmit JC, Cogniaux J, Hermans P, et al. Multiple drug resistance to nucleoside analogues and nonnucleoside reverse transcriptase inhibitors in an efficiently replicating human immunodeficiency virus type 1 patient strain. J Infect Dis. 1996;174:962‐968. [DOI] [PubMed] [Google Scholar]
- 41. Mei L, Chen H, Wei DM, et al. Maintenance chemotherapy for ovarian cancer [serial online]. Cochrane Database Syst Rev. 2013;6:CD007414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Pecorelli S, Favalli G, Gadducci A, et al. Phase III trial of observation versus 6 courses of paclitaxel in patients with advanced epithelial ovarian cancer in complete response after 6 courses of paclitaxel/platinum‐based chemotherapy: final results of the After‐6 Protocol 1. J Clin Oncol. 2009;27:4642‐4648. [DOI] [PubMed] [Google Scholar]
- 43. Markman M, Liu PY, Wilczynski S, et al. Phase III randomized trial of 12 versus 3 months of maintenance paclitaxel in patients with advanced ovarian cancer after complete response to platinum and paclitaxel‐based chemotherapy: a Southwest Oncology Group and Gynecologic Oncology Group trial. J Clin Oncol. 2003;21:2460‐2465. [DOI] [PubMed] [Google Scholar]
- 44. Markman M, Liu PY, Moon J, et al. Impact on survival of 12 versus 3 monthly cycles of paclitaxel (175 mg/m2) administered to patients with advanced ovarian cancer who attained a complete response to primary platinum‐paclitaxel: follow‐up of a Southwest Oncology Group and Gynecologic Oncology Group phase 3 trial. Gynecol Oncol. 2009;114:195‐198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Hess LM, Rong N, Monahan PO, Gupta P, Thomaskutty C, Matei D. Continued chemotherapy after complete response to primary therapy among women with advanced ovarian cancer: a meta‐analysis. Cancer. 2010;116:5251‐5260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Gadducci A, Katsaros D, Zola P, et al. Weekly low‐dose paclitaxel as maintenance treatment in patients with advanced ovarian cancer who had microscopic residual disease at second‐look surgery after 6 cycles of paclitaxel/platinum‐based chemotherapy: results of an open noncomparative phase 2 multicenter Italian study (After‐6 Protocol 2). Int J Gynecol Cancer. 2009;19:615‐619. [DOI] [PubMed] [Google Scholar]
- 47. Vergote IB, Jimeno A, Joly F, et al. Randomized phase III study of erlotinib versus observation in patients with no evidence of disease progression after first‐line platin‐based chemotherapy for ovarian carcinoma: a European Organisation for Research and Treatment of Cancer‐Gynaecological Cancer Group, and Gynecologic Cancer Intergroup study. J Clin Oncol. 2014;32:320‐326. [DOI] [PubMed] [Google Scholar]
- 48. Sabbatini P, Harter P, Scambia G, et al. Abagovomab as maintenance therapy in patients with epithelial ovarian cancer: a phase III trial of the AGO OVAR, COGI, GINECO, and GEICO—the MIMOSA study. J Clin Oncol. 2013;31:1554‐1561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. McGuire WP. Maintenance therapy for ovarian cancer: of Helsinki and Hippocrates. J Clin Oncol. 2009;27:4633‐4634. [DOI] [PubMed] [Google Scholar]
- 50. Bookman MA. Should studies of maintenance therapy be maintained in women with ovarian cancer? J Gynecol Oncol. 2013;24:105‐107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Rigakos G, Razis E. BRCAness: finding the Achilles heel in ovarian cancer. Oncologist. 2012;17:956‐962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Ledermann J, Harter P, Gourley C, et al. Olaparib maintenance therapy in patients with platinum‐sensitive relapsed serous ovarian cancer: a preplanned retrospective analysis of outcomes by BRCA status in a randomised phase 2 trial. Lancet Oncol. 2014;15:852‐861. [DOI] [PubMed] [Google Scholar]
- 53. Oza AM, Cibula D, Benzaquen AO, et al. Olaparib combined with chemotherapy for recurrent platinum‐sensitive ovarian cancer: a randomized phase 2 trial. Lancet Oncol. 2015;16:87‐97. [DOI] [PubMed] [Google Scholar]
- 54. Burger RA, Brady MF, Bookman MA, et al. Incorporation of bevacizumab in the primary treatment of ovarian cancer. N Engl J Med. 2011;365:2473‐2483. [DOI] [PubMed] [Google Scholar]
- 55. Perren TJ, Swart AM, Pfisterer J, et al. A phase 3 trial of bevacizumab in ovarian cancer. N Engl J Med. 2011;365:2484‐2496. [DOI] [PubMed] [Google Scholar]
- 56. Oza AM, Perren TJ, Swart AM, et al. ICON7: final overall survival results in the GCIG phase III randomized trial of bevacizumab in women with newly diagnosed ovarian cancer (not Ann Oncol. 2013) [abstract]. Eur J Cancer. 2013;49(suppl 3). Abstract LBA6. [Google Scholar]
- 57. Aghajanian C, Blank SV, Goff BA, et al. OCEANS: a randomized, double‐blind, placebo‐controlled phase III trial of chemotherapy with or without bevacizumab in patients with platinum‐sensitive recurrent epithelial ovarian, primary peritoneal, or fallopian tube cancer. J Clin Oncol. 2012;30:2039‐2045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Pujade‐Lauraine E, Hilpert F, Weber B, et al. Bevacizumab combined with chemotherapy for platinum‐resistant recurrent ovarian cancer: the AURELIA open‐label randomized phase III trial. J Clin Oncol. 2014;32:1302‐1308. [DOI] [PubMed] [Google Scholar]
- 59. du Bois A, Floquet A, Kim JW, et al. Randomized, double‐blind, phase III trial of pazopanib versus placebo in women who have not progressed after first‐line chemotherapy for advanced epithelial ovarian, fallopian tube, or primary peritoneal cancer (AEOC): results of an International Intergroup trial (AGO‐OVAR16). J Clin Oncol. 2014;32:3374‐3382. 25225436 [Google Scholar]
- 60. du Bois A, Kristensen G, et al. AGO‐OVAR 12: a randomized placebo‐controlled GCIG/ENGOT‐Intergroup phase III trial of standard frontline chemotherapy +/‐ nintedanib for advanced ovarian cancer. Int J Gynecol Cancer. 2013;23(8 suppl 1):7‐8. [Google Scholar]
- 61. Ledermann JA, Perren TJ, Raja FA, et al. Randomised double‐blind phase III trial of cediranib (AZD 2171) in relapsed platinum sensitive ovarian cancer: results of the ICON6 trial [abstract]. Eur J Cancer. 2013;49(suppl 2). Abstract LBA10. [Google Scholar]
- 62. du Bois A, Weber B, Rochon J, et al. Addition of epirubicin as a third drug to carboplatin‐paclitaxel in first‐line treatment of advanced ovarian cancer: a prospectively randomized gynecologic cancer intergroup trial by the Arbeitsgemeinschaft Gynaekologische Onkologie Ovarian Cancer Study Group and the Groupe d'Investigateurs Nationaux pour l'Etude des Cancers Ovariens. J Clin Oncol. 2006;24:1127‐1135. [DOI] [PubMed] [Google Scholar]
- 63. Möbus V, Wandt H, Frickhofen N, et al. Phase III trial of high‐dose sequential chemotherapy with peripheral blood stem cell support compared with standard dose chemotherapy for first‐line treatment of advanced ovarian cancer: intergroup trial of the AGO‐Ovar/AIO and EBMT. J Clin Oncol. 2007;25:4187‐4193. [DOI] [PubMed] [Google Scholar]
- 64. Bookman MA, Brady MF, McGuire WP, et al. Evaluation of new platinum‐based treatment regimens in advanced‐stage ovarian cancer: a phase III trial of the Gynecologic Cancer Intergroup. J Clin Oncol. 2009;27:1419‐1425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Muller PA, Vousden KH. Mutant p53 in cancer: new functions and therapeutic opportunities. Cancer Cell. 2014;25:304‐317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Zsiros E, Tanyi J, Balint K, Kandalaft LE. Immunotherapy for ovarian cancer: recent advances and perspectives. Curr Opin Oncol. 2014;26:492‐500. [DOI] [PubMed] [Google Scholar]
- 67. Alvarez Secord A, Berchuck A, Higgins RV, et al. A multicenter, randomized, phase 2 clinical trial to evaluate the efficacy and safety of combination docetaxel and carboplatin and sequential therapy with docetaxel then carboplatin in patients with recurrent platinum‐sensitive ovarian cancer. Cancer. 2012;118:3283‐3293. [DOI] [PubMed] [Google Scholar]
- 68. Tomao F, Papa A, Strudel M, et al. Investigating molecular profiles of ovarian cancer: an update on cancer stem cells. J Cancer. 2014;5:301‐310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Esposito A, Bardelli A, Criscitiello C, et al. Monitoring tumor‐derived cell‐free DNA in patients with solid tumors: clinical perspectives and research opportunities. Cancer Treat Rev. 2014;40:648‐655. [DOI] [PubMed] [Google Scholar]
- 70. Lipson EJ, Velculescu VE, Pritchard TS, et al. Circulating tumor DNA analysis as a real‐time method for monitoring tumor burden in melanoma patients undergoing treatment with immune checkpoint blockade [serial online]. J Immunother Cancer. 2014;2:42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Morelli MP, Overman MJ, Dasari A, et al. Characterizing the patterns of clonal selection in circulating tumor DNA from patients with colorectal cancer refractory to anti‐EGFR treatment. Ann Oncol. 2015;26:731‐736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Lebofsky R, Decraene C, Bernard V, et al. Circulating tumor DNA as a non‐invasive substitute to metastasis biopsy for tumor genotyping and personalized medicine in a prospective trial across all tumor types. Mol Oncol. 2015;9:783‐790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Azad NS, Annunziata CM, Steinberg SM, et al. Lack of reliability of CA125 response criteria with anti‐VEGF molecularly targeted therapy. Cancer. 2008;112:1726‐1732. [DOI] [PubMed] [Google Scholar]
- 74. Rustin G, van der Burg M, Griffin C, Qian W, Swart AM. Early versus delayed treatment of relapsed ovarian cancer. Lancet. 2011;377:380‐381. [DOI] [PubMed] [Google Scholar]
- 75. Lee SI, Catalano OA, Dehdashti F. Gynecologic cancer imaging with MRI, FDGPET‐CT and PET‐MR. J Nucl Med. 2015;56:436‐443. [DOI] [PubMed] [Google Scholar]
- 76. Friedlander M, Trimble E, Tinker A, et al. Gynecologic Cancer Intergroup: clinical trials in recurrent ovarian cancer. Int J Gynecol Cancer. 2011;21:771‐775. [DOI] [PubMed] [Google Scholar]
- 77. Lee MJ, Ye AS, Gardino AK, et al. Sequential application of anticancer drugs enhances cell death by rewiring apoptotic signaling networks. Cell. 2012;149:780‐794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Kohn EC, Romano S, Lee JM. Clinical implications of using molecular diagnostics for ovarian cancers [serial online]. Ann Oncol. 2013;24(suppl 10):x22‐x26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Gourley C, McCavigan A, Perren T, et al. Molecular subgroup of high‐grade serous ovarian cancer (HGSOC) as a predictor of outcome following bevacizumab [abstract]. J Clin Oncol. 2014;32(15 suppl). Abstract 5502. [Google Scholar]
- 80. Tothill RW, Tinker AV, George J, et al. Novel molecular subtypes of serous and endometrioid ovarian cancer linked to clinical outcome. Clin Cancer Res. 2008;14:5198‐5208. [DOI] [PubMed] [Google Scholar]
- 81. Winterhoff BJN, Kommoss S, Oberg AL, et al. Bevacizumab and improvement of progression‐free survival (PFS) for patients with the mesenchymal molecular subtype of ovarian cancer [abstract]. J Clin Oncol. 2014;32(15 suppl). Abstract 5509. [Google Scholar]
- 82. Spreafico A, Oza AM, Clarke B, et al. Genotype matched treatment for patients with advanced type I epithelial ovarian cancer (EOC) [abstract]. J Clin Oncol. 2014;32(15 suppl). Abstract 5506. [DOI] [PubMed] [Google Scholar]
- 83. Wilson MK, Karakasis K, Oza AM. Outcomes and endpoints in trials of cancer treatment: the past, present, and future [serial online]. Lancet Oncol. 2015;16:e32‐e42. [DOI] [PubMed] [Google Scholar]
- 84. Wilson MK, Collyar D, Chingos DT, et al. Outcomes and endpoints in cancer trials: bridging the divide [serial online]. Lancet Oncol. 2015;16:e43‐e52. [DOI] [PubMed] [Google Scholar]
- 85. Dion V. Tissue specificity in DNA repair: lessons from trinucleotide repeat instability. Trends Genet. 2014;30:220‐229. [DOI] [PubMed] [Google Scholar]
- 86. Sieber OM, Tomlinson SR, Tomlinson IP. Tissue, cell and stage specificity of (epi)mutations in cancers. Nat Rev Cancer. 2005;5:649‐655. [DOI] [PubMed] [Google Scholar]
- 87. Vaughan S, Coward JI, Bast RC Jr, et al. Rethinking ovarian cancer: recommendations for improving outcomes. Nat Rev Cancer. 2011;11:719‐725. [DOI] [PMC free article] [PubMed] [Google Scholar]


