Abstract
Glioblastoma multiforme (GBM) is an aggressive malignancy associated with profound host immunosuppression mediated in part by FoxP3 expressing regulatory CD4+ T lymphocytes (Tregs) that down-regulate anti-tumor immunity. In order to assess whether FoxP3 was an independent driver differentially expressed in primary versus recurrent GBMs, we stained resected primary and recurrent GBM tumors for CD3, CD4, CD8 and FoxP3 expression using standard immunohistochemistry. Slides were scanned with a high-resolution scanner (ScanScope CS; Aperio), and image analysis software (Aperio ScanScope) was used to enumerate lymphocyte subpopulations allowing for high-throughput analysis and bypassing manual selection bias. As shown in previous studies, enumeration of individual lymphocyte populations did not correlate with clinical outcomes in patients with GBM. However, the CD4+ to regulatory FoxP3+ T cell ratio was diminished in recurrent disease, and increased CD3 and CD8+ to regulatory T cell ratios showed a positive correlation with survival outcomes in primary GBM. These results suggest that while absolute numbers of tumor infiltrating lymphocytes may not be informative for predicting clinical outcomes in patients with GBM, the effective balance of CD3, CD4 and CD8+ T cells to immunosuppressive FoxP3+ regulatory cells may influence clinical outcomes in this patient population.
Electronic supplementary material
The online version of this article (doi:10.1007/s00262-014-1651-7) contains supplementary material, which is available to authorized users.
Keywords: Tumor infiltrating lymphocytes, Regulatory T cells (Tregs), Primary and recurrent GBM, Immunotherapy
Introduction
Glioblastoma multiforme (GBM), the most common adult primary brain tumor, is an aggressive infiltrative astrocytoma, invariably associated with poor patient outcomes necessitating innovative directed therapeutics [1]. Immunotherapy is a promising novel adjuvant treatment modality, with exquisite specificity and demonstrated efficacy in randomized phase III clinical trials that can potentially be harnessed against GBMs [2–6]. GBMs, however, are associated with profound immunosuppression, limiting the utility of immunotherapeutic modalities. We and others have shown that this profound immunosuppression is mediated, at least in part, through elevated proportions of regulatory T lymphocytes (Tregs) expressing FoxP3 in the peripheral blood that can down-regulate immune responses thereby inhibiting antitumor activity [7–13]. Leveraging tumor-specific immune responses in GBMs may hinge on overcoming the milieu of a suppressive microenvironment. As such, a better understanding of type, frequency and function of tumor infiltrating lymphocytes (TILs) is paramount for leveraging immunotherapeutic strategies against GBM. Tipping the balance of TILs in favor of effector T cells mediating tumor lysis, while simultaneously depleting Tregs, may provide robust anti-tumor immunity that bypasses regulatory pathways.
Several studies have attempted to delineate the relationship between effector and regulatory T cells as they relate to prognosis and survival [14–18]. In hepatocellular carcinoma, melanoma, ovarian carcinoma and breast carcinoma, elevated levels of TILs have been associated with favorable prognoses [18–21]. In malignant gliomas, the reported results have been mixed, suggesting that there is not a strong correlation between infiltrating lymphocytes and outcomes in this patient population [22–28]. Consequently, effector TILs that enhance anti-tumor activity may be suppressed by Tregs expressing FoxP3 such that the ratio of these compartments in the intratumoral microenvironment may have stronger prognostic implications than a single T cell subset on the overall anti-tumor immune response [7–9]. We have shown that while patients with GBM have overall depleted lymphocyte numbers in the periphery, the proportion of FoxP3 expressing Tregs is elevated within PBMCs and these cells contribute significantly to the profound systemic cell-mediated immune deficits observed in these patients [13, 29–31]. We therefore examined whether lymphocyte subset populations (CD4+, CD8+, FoxP3+) varied significantly between primary and recurrent GBMs and whether the ratio of CD3, CD4 and CD8+ T cells to regulatory T cell marker expression correlated with clinical outcomes. To avoid evaluator dependent bias in both high power field selection for evaluation and cell counting, we used automated Aperio software analysis that allows for the accurate and expeditious detection of lymphocytic infiltrates from entire tissue sections taken from pathologic tumor specimens. Although FoxP3 expression alone was not differentially expressed, our results demonstrated reduced proportion of CD4+ T cells and lower CD4+/FoxP3+ ratios within TILs in recurrent disease. Within primary GBM tumors, a higher CD3 and CD8+ to FoxP3 regulatory T cell ratio showed a positive correlation with survival, suggesting that effector T cell to regulatory T cell ratios within the tumor microenvironment may be a driver of clinical outcomes in this patient population.
Materials and methods
De-identified archival GBM tissue samples were obtained under IRB approval from the Preston Robert Tisch Brain Tumor Center Biorepository. GBMs were selected from archived resected tumor blocks in cryogenic storage. Serial section slides from each tumor were fixed in formalin, embedded in paraffin and stained for CD3, CD4, CD8 or FoxP3 expression using standard immunohistochemistry (IHC) techniques. Tonsil sections were utilized to determine efficacy of each stain and overall technique.
Study population
The material consisted of representative formalin-fixed, paraffin-embedded glioblastoma sections taken from 39 patients, 21 undergoing primary surgery for untreated GBM (primary group) and an additional 18 patients undergoing surgery for recurrent glioblastoma (recurrent group), all at Duke University Medical Center. The study population consisted of 33 males and 24 females with an age range from 26 to 79 years of age at the time of tumor resection or biopsy. The study population had an average age of 52 and was predominantly made up of individuals of Caucasian descent (n = 55) along with two individuals of African-American descent, one individual of Hispanic descent and one individual of Indian descent. Representative blocks of tissue were chosen by a neuropathologist at Duke University to represent nearly 100 % viable tumor in the selected block and over 1 cm2 of tissue section by light microscopic examination of Hematoxylin and Eosin (H&E) stained sections. Serial unstained sections were cut from these blocks and submitted to IHC staining for FoxP3, CD3, CD4 and CD8 lymphocyte subset analysis.
Immunohistochemistry
After staining for the T cell markers, slides were scanned with a high-resolution scanner (ScanScope CS; Aperio) at 40× magnification and analyzed using image software (Aperio ScanScope). Pixel counts were gated to strongly positive pixel counts using the ScanScope software and the Positive Pixel Count v9 (PPCv9) algorithm embedded in the program. Evaluation of T cell marker density was carried out blinded to clinicopathologic information. Serially stained sections of individual biopsies stained for the T cell markers of interest were examined by one observer using the multiple imaging modality of the software to assure that identical regions of tumor were being examined. In order to determine the possibility that cell size in a histologic section could result in a variation of cell counts via a highly variable pixel count per cell, a manual count of individually stained cells identified on the monitor was performed on a randomly selected subset of three tumor samples. For each stain within these samples, four loci of highest T cell density were identified and manually evaluated for positively stained cells. These same loci were evaluated using the PPCv9 algorithm. The pixel counts and raw cell counts for each loci were entered into an excel file and evaluated for statistical relationship and concordance. We additionally configured the PPCv9 algorithm to produce a hyperpigmented digital color overlay of each tissue area being analyzed to allow clear identification and pathological classification of each positively stained cell.
Once the chosen methodology was successfully evaluated and the stained tumor slides were digitized, whole tissue area analysis was performed on each slide file using the positive pixel count v9 algorithm. Individual data from each slide were recorded and cataloged in an excel file for statistical analysis.
Minor scanning errors were detected and corrected in five slide files out of the total number of 156. The digital slide files were transferred to a high capacity storage volume for transport and convenient analysis.
Statistical analysis
Absolute counts were divided by surface area of each specimen to standardize measurements, and ratio of CD4, CD8 and FoxP3+ cells were measured over CD3+ cells. Analysis of these values was obtained using unpaired t tests with a significant result limited to p values of less than 0.05. Proportions of CD3, CD4 and CD8+ cells over FoxP3 expressing cells were also measured using unpaired t tests with a significant result limited to p values of less than 0.05. Primary GBM survival association with CD3, CD4 and CD8+ to FoxP3 expressing ratios were explored using a linear regression model.
Results
Study population
We analyzed 39 de-identified archival samples—21 from patients with primary GBM and 18 pathology samples from patients with recurrent disease. Only four patients were alive among those with primary disease at the time of this study, and only three were alive among those with recurrent disease.
Strong concordance between manual cell counting and Aperio software analysis of IHC specimens
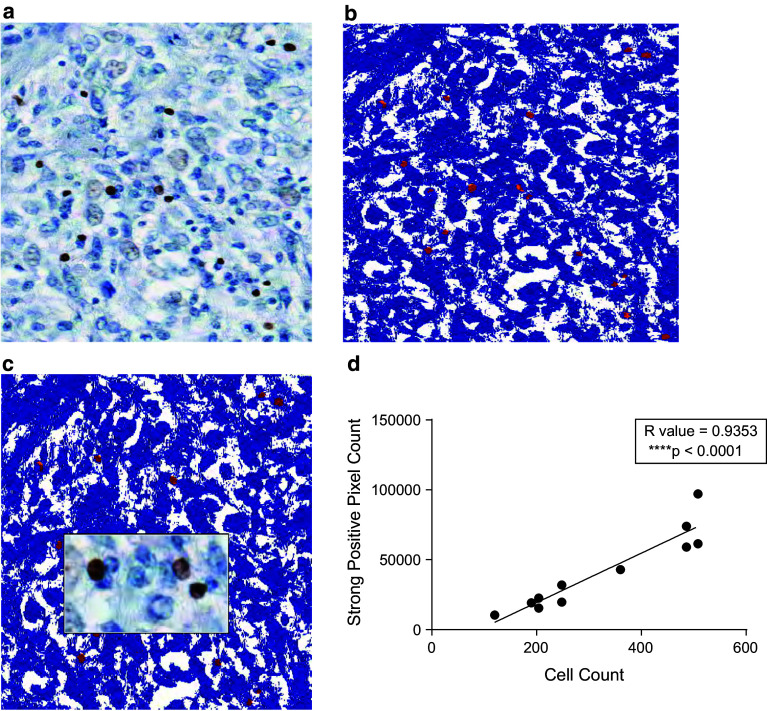
We analyzed CD3, CD4, CD8 and FoxP3 detection from three different intratumoral pathology specimens via conventional cell counting and via the Aperio software analysis. Figure 1 shows a primary GBM IHC specimen stained for FoxP3 next to Aperio software analysis of the same specimen. Strongly positive pixel counts (shown in Fig. 1b as cells colored in red, with background stained in blue) represent the quantitative measure of detecting FoxP3+ cells by software analysis. Manual cell counting by a trained observer correlates well with Aperio software analysis of the identical sections as measured by strongly positive pixel counts with a correlation coefficient of 0.9353 (p < 0.0001, Fig. 1d) on linear regression. Manual counting was limited to selected high-powered fields and subject to observer bias, while automated Aperio slide analysis indiscriminately scanned selected fields.
Fig. 1.
Aperio Analysis of GBM specimens stained for T cell markers by IHC. Primary GBM pathology specimen was stained for FoxP3 using standard IHC (a) and analyzed by Aperio (b) with images superimposed (c). FoxP3 expressing cells by IHC are quantified as strong positive pixels shown in red, while background is shown as blue (b). CD3, CD4, CD8 or FoxP3 expressing cells were manually counted demonstrating strong positive concordance with Aperio quantification of IHC (d)
Strong concordance between FoxP3 expression alone and FoxP3+ CD4+ cells and FoxP3+ CD3+ cells and no concordance between FoxP3 expression and FoxP3+CD8+ cells
FoxP3 is not an exquisitely specific marker for regulatory T cells and can be transiently expressed in activated T effector cells. In order to determine whether FoxP3 expression in TILs within GBM delineated CD4+FoxP3+ T cells or other cell populations, seven separate GBM samples were dual-stained for FoxP3 by CD3, CD4 or CD8. Concordance testing for FoxP3 expression versus FoxP3+CD4+, FoxP3+CD3+ and FoxP3+CD8+ cells demonstrates very strong correlations with FoxP3 expression and FoxP3+CD3+ and FoxP3+CD4+ cells (Supplemental Fig. 1a, b). There were no identified FoxP3+CD8+ lymphocytes and thus no correlation with FoxP3 and FoxP3+CD8+ cells or other FoxP3+ cell subsets (Supplemental Fig. 1c). These data demonstrate strong concordance between FoxP3 expression alone and double staining for FoxP3 by CD4 and CD3 and support the utility of FoxP3 staining as a marker for CD4+FoxP3+ regulatory T cells within GBM tumors.
Primary GBMs have elevated proportions of CD4+/CD3+ cells
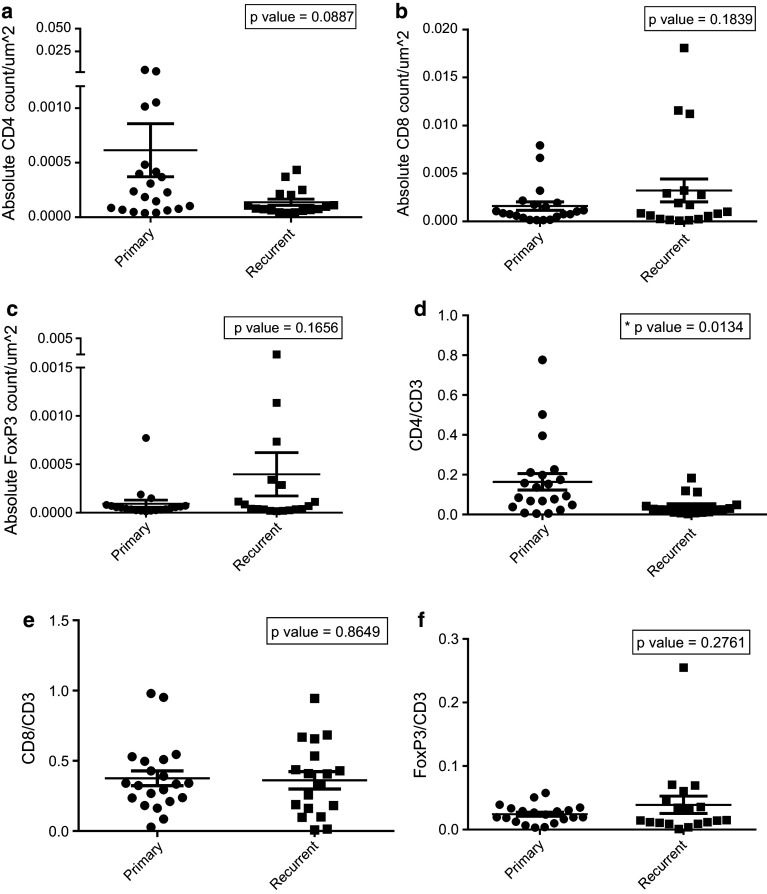
There is no significant difference between primary and recurrent GBMs when comparing absolute cell counts of CD4+ (Fig. 2a), CD8+ (Fig. 2b), and FoxP3+ cells (Fig. 2c) highlighting that the magnitude of TIL infiltration does not vary significantly upon tumor recurrence. However, the composition of the TIL infiltrates differed significantly, with recurrent tumors showing a significant decrease in proportion of CD4+ T cells (expressed as ratio of CD4+/CD3+ cells) (Fig. 2d) (p value = 0.0134). The proportion of CD8+ and FoxP3+ cells (expressed as ratio of CD8+/CD3+ cells and FoxP3+/CD3+ cells) among primary and recurrent GBMs did not significantly differ (Fig. 2e; p value = 0.8649; Fig. 2f; p value = 0.2761).
Fig. 2.
Absolute cell counts and cell count ratios in primary versus recurrent GBM. Primary and recurrent GBM pathology specimens were stained for CD3, CD4, CD8 and FoxP3+ markers and quantified using Aperio software. Absolute counts were divided by surface area of each specimen to standardize measurements, and the ratio of CD4, CD8 and FoxP3+ cells was measured over CD3+ cells. Absolute counts alone were not different between primary and recurrent GBM (a, b, c), but when expressed as percentage of T cell subsets over total CD3+ counts, recurrent GBMs had decreased proportions of CD4+/CD3+ cells (d); however, CD8+ and FoxP3+ cells remained unchanged when expressed as a percentage over total CD3+ cells (e, f)
Recurrent GBMs have higher proportions of FoxP3+/CD4+ cells
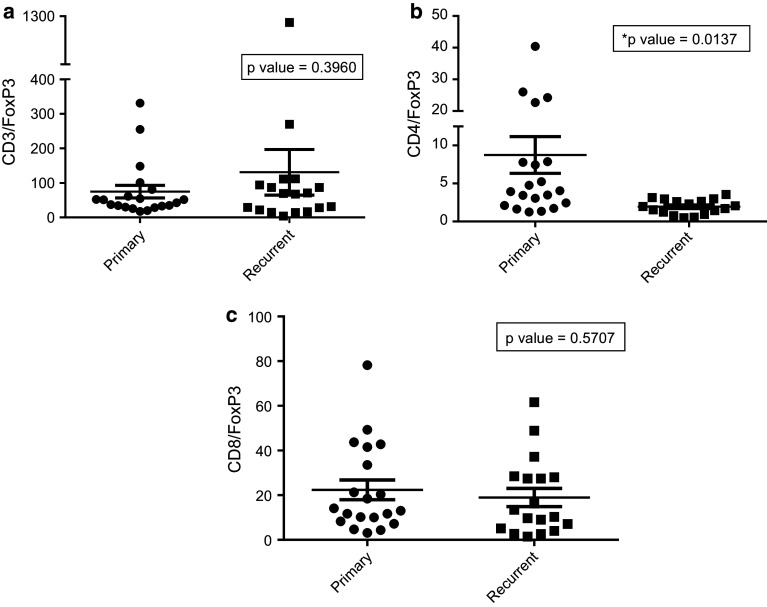
The CD4+ to FoxP3+ ratio is increased in primary GBM versus recurrent GBM (Fig. 3b; p value = 0.0137). There is no difference, however, among ratios of CD3+ and CD8+ cells over FoxP3+ cells (Fig. 3a; p value = 0.3960; Fig. 3c; p value = 0.5707) in primary versus recurrent disease.
Fig. 3.
CD3, CD4 and CD8+ to FoxP3+ ratio in primary versus recurrent GBM. Primary and recurrent GBM pathology specimens were stained for CD3, CD4, CD8 and FoxP3 markers and quantified using Aperio software. Ratios of CD3, CD4 and CD8+ cells were measured over FoxP3+ cells. There is a higher ratio of CD4/FoxP3+ cells in primary versus recurrent GBMs (b) but no difference when comparing CD3/FoxP3+ and CD8/FoxP3+ ratios (a and c)
Positive correlation between survival and CD3+/FoxP3+ and CD8+/FoxP3+ ratios in primary GBMs
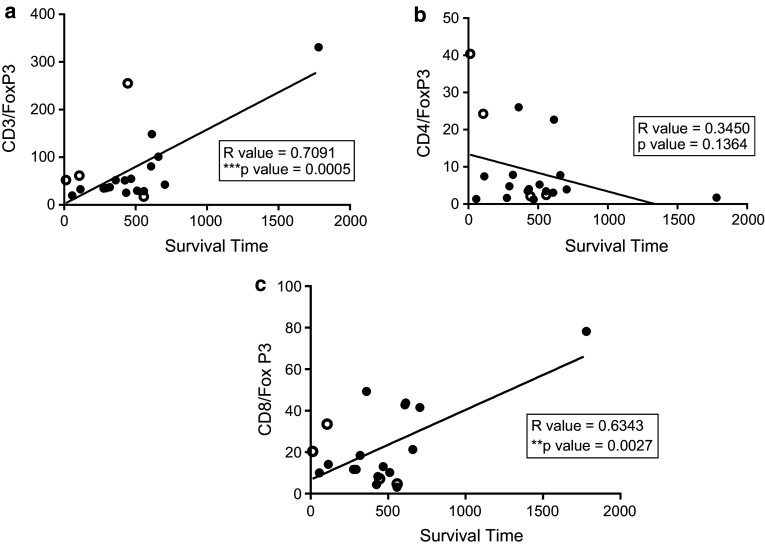
Survival is increased (Fig. 4a) among primary GBM patients who have a higher CD3+ to FoxP3+ ratio (p value = 0.0005, correlation coefficient = 0.7091). Moreover, there is a significant positive correlation between survival and the CD8+ to FoxP3+ ratio in primary GBMs (Fig. 4c; p value = 0.0027, correlation coefficient = 0.6343). Survival is not increased (Fig. 4b) among primary GBM patients who have a higher CD4 to FoxP3+ ratio (p value = 0.1364, correlation coefficient = 0.3450).
Fig. 4.
Correlation of CD3, CD4 and CD8+ over FoxP3+ T cell ratios and survival in primary GBM. Primary GBM pathology specimens were stained for CD3, CD4, CD8 and FoxP3 markers and quantified using Aperio software. Survival (in days) of patients was plotted versus ratios of CD3, CD4 and CD8+ cells over FoxP3+ cells. Patients still alive at time of data analysis are represented by a clear circle. There is a positive correlation between survival and increased CD3+ to FoxP3+ ratios (a; R value = 0.7091) and CD8+ to Foxp3+ ratios (c; R value = 0.6343) but no correlation between survival and CD4+ to FoxP3+ ratios (b; R value = 0.3450)
Discussion
GBM remains invariably aggressive, but tumor-specific immunotherapy may mitigate disease invasion and ameliorate mortality. However, a better understanding of the tumor microenvironment, including the role of Tregs, is requisite to potentiate immunotherapeutic efficacy. CD4+FoxP3+ Tregs are essential for the active suppression of autoimmunity and inhibition of anti-tumor immune responses [10, 26]. Increased FoxP3 expressing cells may potentially confer disease resistance in refractory cases suggesting that as a GBM recurs, there is greater immunotolerance resulting in a greater proportion of FoxP3+ expressing Tregs. We sought to assess, in this pilot study, if FoxP3 expression differed significantly between primary and recurrent GBM tumors, and whether expression has an associated relationship with survival outcomes. While this study did not incorporate treatment history as part of the analysis, a strong relationship would implicate FoxP3 expression levels as a possible independent prognostic indicator in this patient population. Although FoxP3 alone did not demonstrate significant differences nor relationship with outcomes (consistent with previously published data), the CD4+ to FoxP3+ ratio was significantly diminished in recurrent cases. Moreover, survival in primary GBM was adversely affected by decreased proportions of CD3+ and CD8+ cells over Foxp3+ cells, suggesting that immunotolerance from increased proportion of FoxP3+ expressing T cells may exacerbate disease pathogenesis.
The use of the Aperio software to quantify IHC staining allowed for rapid analysis of multiple IHC specimens and correlates well with manual cell counting. Aperio ScanScope scanners are currently used in clinical practice by greater than two-thirds of the top 15 rated US hospitals and two-thirds of the top 15 pharmaceutical companies [32, 33]. This high-throughput approach has several advantages over manual cell counting. Limitations, however, included scanning errors that occurred in roughly one out of 30 slides and can be circumvented through re-scanning and re-analysis. Other limitations were secondary to decreased staining uptake by relevant T cells that went uncounted by Aperio, or conversely, over-counting secondary to increased staining uptake when quantified as strongly positive pixels. These limitations were less common and occurred in roughly one out of 80 slides, but when there was a flagrant discrepancy between the Aperio analysis and manual counting, we removed the slides from the data. However, only two slides out of 156 analyzed slides were removed secondary to software inadequacy. In summary, the Aperio software is an innovative approach to analyzing scanned IHC specimens allowing for rapid real time analysis of multiple samples, and despite minor reading errors from poor IHC staining, remains a valuable, accurate and more practical approach than manual cell counting.
Although the results of this pilot study demonstrate the utility of a high-throughput analysis demonstrating differential expression of T cell regulatory ratios in primary versus recurrent GBM, these results are limited by small sample sizes with limited power for definitive statistical analysis thus requiring further validation to determine whether TIL ratio is independently prognostic. Furthermore, this study did not account for the presence and effect of peripheral Tregs or patient-specific prognostic factors due to the use of de-identified tissues from a biorepository. This further complicates patient survival necessitating further tests to distinguish the relationship between Tregs and outcomes accounting for location, disease burden, partial versus gross total resections and response to chemotherapy. These factors could play a key role in prognosis and are currently being addressed in larger validation studies.
Several studies have attempted to understand the relationship of intratumoral Tregs as they pertain to glioma pathogenesis and patient outcome. These studies have demonstrated that FoxP3 is overexpressed in high grade compared to low-grade gliomas; however, among these high-grade tumors, correlation of FoxP3 expression with survival is nebulous [26, 34]. Heimberger et al. and Lohr et al. demonstrate no correlation with Tregs and survival, while Jacobs et al. and Yue et al. show modest and strong correlations, respectively, [25–28]. The studies by Heimberger et al. and Yue et al. utilized manual counting of selected high-powered fields, while Jacobs et al. utilized four color flow cytometry, and Lohr et al. utilized a novel computational analysis to analyze whole slides, which minimized errors associated with manual cell counting and incomplete slide analyses [25–28]. Similarly, our results using automated analysis of whole tissue sections demonstrate concordance with Heimberger et al. and Lohr et al. since FoxP3 alone was not an independent driver of GBM pathogenesis and patient survival outcomes [25, 26]. Meanwhile, as one of the first studies comparing intratumoral TILs in primary versus recurrent GBM, our results give credence for utilizing regulatory T cell ratios as opposed to the absolute counts in order to stratify differential patient outcomes and disease subsets.
While increased FoxP3 expressing Treg ratios may be a poor predictor for survival, other immunosuppressive populations of cells such as CD8+FoxP3−CD103+ T cells, myeloid-derived suppressor cells and M2 macrophages may contribute to clinical outcomes in patients with GBM and more comprehensive analysis of the ratio of non-regulatory to regulatory populations of leukocytes is requisite [35–37]. Since Foxp3 is not an exhaustive marker of Tregs, and FoxP3 expressing T cells may have a more ubiquitous role than previously understood, additional analysis of Treg phenotype and function may prove informative [38–41]. Additional exhaustion markers such as LAG-3 and TIM-3 alongside functional assessment of regulatory cytokine secretion such as IL-10 and TGF-beta should more clearly delineate Tregs [42]. Thus, the activation state of Tregs, rather than absolute numbers may be more informative in assessing the relative state of immunosuppression. These activation makers juxtaposed with FoxP3 co-staining may allow for further differentiation of tumors in order to discern the extent of Treg immunosuppression.
In summary, our results demonstrated that high-throughput analysis of lymphocyte subsets in GBM using Aperio software analysis is practical and accurate. While FoxP3 expression alone is not informative in distinguishing primary from recurrent GBM, or assessing survival outcomes, increases in T cell regulatory expression ratios are associated with tumor recurrence and decreased survival in primary cases. Although validation studies of our results in larger cohorts are paramount, these ratios may be used to pave the path for disease stratification and novel therapeutics in patients with heavy intratumoral Treg burdens.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgments
This work was supported by the National Institutes of Health (NIH)/National Cancer Institute/National Institute of Neurological Disorders and Stroke Grants [5R01-NS067037 (to D.A. Mitchell), 5R01-CA134844 (to D.A. Mitchell), 5P50-CA108786 (to J.H. Sampson/Bigner)] and a Clinical and Translational Science Awards Grant UL1RR024128 from the National Center for Research Resources, a component of the NIH and NIH Roadmap for Medical Research. Additional support was provided by Accelerate Brain Cancer Cure (ABC2), National Brain Tumor Society, the American Brain Tumor Association, and the Pediatric Brain Tumor Foundation of the United States.
Conflict of interest
The authors declare that they have no conflict of interest with the research presented in this paper.
Abbreviations
- GBM
Glioblastoma multiforme
- H&E
Hematoxylin and eosin
- IHC
Immunohistochemistry
- PBMCs
Peripheral blood mononuclear cells
- PPCv9
Positive pixel count v9
- Tregs
Regulatory T lymphocytes
- TILs
Tumor infiltrating lymphocytes
Footnotes
Elias J. Sayour and Pat McLendon have contributed equally to this work.
References
- 1.Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia T, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO, European Organisation for R, Treatment of Cancer Brain T, Radiotherapy G, National Cancer Institute of Canada Clinical Trials G Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 2005;352(10):987–996. doi: 10.1056/NEJMoa043330. [DOI] [PubMed] [Google Scholar]
- 2.Kantoff PW, Higano CS, Shore ND, Berger ER, Small EJ, Penson DF, Redfern CH, Ferrari AC, Dreicer R, Sims RB, Xu Y, Frohlich MW, Schellhammer PF, Investigators IS. Sipuleucel-T immunotherapy for castration-resistant prostate cancer. N Engl J Med. 2010;363(5):411–422. doi: 10.1056/NEJMoa1001294. [DOI] [PubMed] [Google Scholar]
- 3.Hodi FS, O’Day SJ, McDermott DF, Weber RW, Sosman JA, Haanen JB, Gonzalez R, Robert C, Schadendorf D, Hassel JC, Akerley W, van den Eertwegh AJ, Lutzky J, Lorigan P, Vaubel JM, Linette GP, Hogg D, Ottensmeier CH, Lebbe C, Peschel C, Quirt I, Clark JI, Wolchok JD, Weber JS, Tian J, Yellin MJ, Nichol GM, Hoos A, Urba WJ. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363(8):711–723. doi: 10.1056/NEJMoa1003466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Yuan J, Adamow M, Ginsberg BA, Rasalan TS, Ritter E, Gallardo HF, Xu Y, Pogoriler E, Terzulli SL, Kuk D, Panageas KS, Ritter G, Sznol M, Halaban R, Jungbluth AA, Allison JP, Old LJ, Wolchok JD, Gnjatic S. Integrated NY-ESO-1 antibody and CD8+ T-cell responses correlate with clinical benefit in advanced melanoma patients treated with ipilimumab. Proc Natl Acad Sci USA. 2011;108(40):16723–16728. doi: 10.1073/pnas.1110814108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lee ST, Jiang YF, Park KU, Woo AF, Neelapu SS. BiovaxID: a personalized therapeutic cancer vaccine for non-Hodgkin’s lymphoma. Expert Opin Biol Ther. 2007;7(1):113–122. doi: 10.1517/14712598.7.1.113. [DOI] [PubMed] [Google Scholar]
- 6.Sampson JH, Heimberger AB, Archer GE, Aldape KD, Friedman AH, Friedman HS, Gilbert MR, Herndon JE, 2nd, McLendon RE, Mitchell DA, Reardon DA, Sawaya R, Schmittling RJ, Shi W, Vredenburgh JJ, Bigner DD. Immunologic escape after prolonged progression-free survival with epidermal growth factor receptor variant III peptide vaccination in patients with newly diagnosed glioblastoma. J Clin Oncol. 2010;28(31):4722–4729. doi: 10.1200/JCO.2010.28.6963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bennett CL, Christie J, Ramsdell F, Brunkow ME, Ferguson PJ, Whitesell L, Kelly TE, Saulsbury FT, Chance PF, Ochs HD. The immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome (IPEX) is caused by mutations of FOXP3. Nat Genet. 2001;27(1):20–21. doi: 10.1038/83713. [DOI] [PubMed] [Google Scholar]
- 8.Brunkow ME, Jeffery EW, Hjerrild KA, Paeper B, Clark LB, Yasayko SA, Wilkinson JE, Galas D, Ziegler SF, Ramsdell F. Disruption of a new forkhead/winged-helix protein, scurfin, results in the fatal lymphoproliferative disorder of the scurfy mouse. Nat Genet. 2001;27(1):68–73. doi: 10.1038/83784. [DOI] [PubMed] [Google Scholar]
- 9.Khattri R, Cox T, Yasayko SA, Ramsdell F. An essential role for Scurfin in CD4+CD25+ T regulatory cells. Nat Immunol. 2003;4(4):337–342. doi: 10.1038/ni909. [DOI] [PubMed] [Google Scholar]
- 10.Fontenot JD, Gavin MA, Rudensky AY. Foxp3 programs the development and function of CD4+CD25+ regulatory T cells. Nat Immunol. 2003;4(4):330–336. doi: 10.1038/ni904. [DOI] [PubMed] [Google Scholar]
- 11.Hori S, Nomura T, Sakaguchi S. Control of regulatory T cell development by the transcription factor Foxp3. Science. 2003;299(5609):1057–1061. doi: 10.1126/science.1079490. [DOI] [PubMed] [Google Scholar]
- 12.Walker MR, Kasprowicz DJ, Gersuk VH, Benard A, Van Landeghen M, Buckner JH, Ziegler SF. Induction of FoxP3 and acquisition of T regulatory activity by stimulated human CD4+CD25− T cells. J Clin Invest. 2003;112(9):1437–1443. doi: 10.1172/JCI19441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Learn CA, Fecci PE, Schmittling RJ, Xie W, Karikari I, Mitchell DA, Archer GE, Wei Z, Dressman H, Sampson JH. Profiling of CD4+, CD8+, and CD4+CD25+CD45RO+FoxP3+ T cells in patients with malignant glioma reveals differential expression of the immunologic transcriptome compared with T cells from healthy volunteers. Clin Cancer Res. 2006;12(24):7306–7315. doi: 10.1158/1078-0432.CCR-06-1727. [DOI] [PubMed] [Google Scholar]
- 14.Clark WH, Jr, Elder DE, Guerry Dt, Braitman LE, Trock BJ, Schultz D, Synnestvedt M, Halpern AC. Model predicting survival in stage I melanoma based on tumor progression. J Natl Cancer Inst. 1989;81(24):1893–1904. doi: 10.1093/jnci/81.24.1893. [DOI] [PubMed] [Google Scholar]
- 15.Clemente CG, Mihm MC, Jr, Bufalino R, Zurrida S, Collini P, Cascinelli N. Prognostic value of tumor infiltrating lymphocytes in the vertical growth phase of primary cutaneous melanoma. Cancer. 1996;77(7):1303–1310. doi: 10.1002/(SICI)1097-0142(19960401)77:7<1303::AID-CNCR12>3.0.CO;2-5. [DOI] [PubMed] [Google Scholar]
- 16.Curiel TJ, Coukos G, Zou L, Alvarez X, Cheng P, Mottram P, Evdemon-Hogan M, Conejo-Garcia JR, Zhang L, Burow M, Zhu Y, Wei S, Kryczek I, Daniel B, Gordon A, Myers L, Lackner A, Disis ML, Knutson KL, Chen L, Zou W. Specific recruitment of regulatory T cells in ovarian carcinoma fosters immune privilege and predicts reduced survival. Nat Med. 2004;10(9):942–949. doi: 10.1038/nm1093. [DOI] [PubMed] [Google Scholar]
- 17.Yu P, Fu YX. Tumor-infiltrating T lymphocytes: friends or foes? Lab Invest. 2006;86(3):231–245. doi: 10.1038/labinvest.3700389. [DOI] [PubMed] [Google Scholar]
- 18.Feng X, Li B, Ye H, Long D. Increased frequency of CD4+CD25(high)FoxP3+ regulatory T cells in patients with hepatocellular carcinoma. Arch Immunol Ther Exp (Warsz) 2011;59(4):309–314. doi: 10.1007/s00005-011-0127-0. [DOI] [PubMed] [Google Scholar]
- 19.Oble DA, Loewe R, Yu P, Mihm MC., Jr Focus on TILs: prognostic significance of tumor infiltrating lymphocytes in human melanoma. Cancer Immun. 2009;9:3. [PMC free article] [PubMed] [Google Scholar]
- 20.Polcher M, Braun M, Friedrichs N, Rudlowski C, Bercht E, Fimmers R, Sauerwald A, Keyver-Paik MD, Kubler K, Buttner R, Kuhn WC, Hernando JJ. Foxp3(+) cell infiltration and granzyme B(+)/Foxp3(+) cell ratio are associated with outcome in neoadjuvant chemotherapy-treated ovarian carcinoma. Cancer Immunol Immunother. 2010;59(6):909–919. doi: 10.1007/s00262-010-0817-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Liu MA. Immunologic basis of vaccine vectors. Immunity. 2010;33(4):504–515. doi: 10.1016/j.immuni.2010.10.004. [DOI] [PubMed] [Google Scholar]
- 22.Sonabend AM, Rolle CE, Lesniak MS. The role of regulatory T cells in malignant glioma. Anticancer Res. 2008;28(2B):1143–1150. [PubMed] [Google Scholar]
- 23.El Andaloussi A, Han Y, Lesniak MS. Prolongation of survival following depletion of CD4+CD25+ regulatory T cells in mice with experimental brain tumors. J Neurosurg. 2006;105(3):430–437. doi: 10.3171/jns.2006.105.3.430. [DOI] [PubMed] [Google Scholar]
- 24.Yang I, Tihan T, Han SJ, Wrensch MR, Wiencke J, Sughrue ME, Parsa AT. CD8+ T-cell infiltrate in newly diagnosed glioblastoma is associated with long-term survival. J Clin Neurosci. 2010;17(11):1381–1385. doi: 10.1016/j.jocn.2010.03.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lohr J, Ratliff T, Huppertz A, Ge Y, Dictus C, Ahmadi R, Grau S, Hiraoka N, Eckstein V, Ecker RC, Korff T, von Deimling A, Unterberg A, Beckhove P, Herold-Mende C. Effector T-cell infiltration positively impacts survival of glioblastoma patients and is impaired by tumor-derived TGF-beta. Clin Cancer Res. 2011;17(13):4296–4308. doi: 10.1158/1078-0432.CCR-10-2557. [DOI] [PubMed] [Google Scholar]
- 26.Heimberger AB, Abou-Ghazal M, Reina-Ortiz C, Yang DS, Sun W, Qiao W, Hiraoka N, Fuller GN. Incidence and prognostic impact of FoxP3+ regulatory T cells in human gliomas. Clin Cancer Res. 2008;14(16):5166–5172. doi: 10.1158/1078-0432.CCR-08-0320. [DOI] [PubMed] [Google Scholar]
- 27.Yue Q, Zhang X, Ye HX, Wang Y, Du ZG, Yao Y, Mao Y. The prognostic value of Foxp3+ tumor-infiltrating lymphocytes in patients with glioblastoma. J Neurooncol. 2014;116(2):251–259. doi: 10.1007/s11060-013-1314-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Jacobs JF, Idema AJ, Bol KF, Grotenhuis JA, de Vries IJ, Wesseling P, Adema GJ. Prognostic significance and mechanism of Treg infiltration in human brain tumors. J Neuroimmunol. 2010;225(1–2):195–199. doi: 10.1016/j.jneuroim.2010.05.020. [DOI] [PubMed] [Google Scholar]
- 29.Fecci PE, Ochiai H, Mitchell DA, Grossi PM, Sweeney AE, Archer GE, Cummings T, Allison JP, Bigner DD, Sampson JH. Systemic CTLA-4 blockade ameliorates glioma-induced changes to the CD4+ T cell compartment without affecting regulatory T-cell function. Clin Cancer Res. 2007;13(7):2158–2167. doi: 10.1158/1078-0432.CCR-06-2070. [DOI] [PubMed] [Google Scholar]
- 30.Fecci PE, Sweeney AE, Grossi PM, Nair SK, Learn CA, Mitchell DA, Cui X, Cummings TJ, Bigner DD, Gilboa E, Sampson JH. Systemic anti-CD25 monoclonal antibody administration safely enhances immunity in murine glioma without eliminating regulatory T cells. Clin Cancer Res. 2006;12(14 Pt 1):4294–4305. doi: 10.1158/1078-0432.CCR-06-0053. [DOI] [PubMed] [Google Scholar]
- 31.Mitchell DA, Cui X, Schmittling RJ, Sanchez-Perez L, Snyder DJ, Congdon KL, Archer GE, Desjardins A, Friedman AH, Friedman HS, Herndon JE, 2nd, McLendon RE, Reardon DA, Vredenburgh JJ, Bigner DD, Sampson JH. Monoclonal antibody blockade of IL-2 receptor alpha during lymphopenia selectively depletes regulatory T cells in mice and humans. Blood. 2011;118(11):3003–3012. doi: 10.1182/blood-2011-02-334565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Slodkowska J, Chyczewski L, Wojciechowski M. Virtual slides: application in pulmonary pathology consultations. Folia Histochem Cytobiol. 2008;46(1):121–124. doi: 10.2478/v10042-008-0018-3. [DOI] [PubMed] [Google Scholar]
- 33.Staniszewski W. Virtual microscopy, data management and image analysis in Aperio ScanScope system. Folia Histochem Cytobiol. 2009;47(4):699–701. doi: 10.2478/v10042-009-0115-y. [DOI] [PubMed] [Google Scholar]
- 34.Wang L, Zhang B, Xu X, Zhang S, Yan X, Kong F, Feng X, Wang J. Clinical significance of FOXP3 expression in human gliomas. Clin Transl Oncol. 2014;16(1):36–43. doi: 10.1007/s12094-013-1037-x. [DOI] [PubMed] [Google Scholar]
- 35.Liu Y, Lan Q, Lu L, Chen M, Xia Z, Ma J, Wang J, Fan H, Shen Y, Ryffel B, Brand D, Quismorio F, Liu Z, Horwitz DA, Xu A, Zheng SG. Phenotypic and functional characteristic of a newly identified CD8+Foxp3- CD103+ regulatory T cells. J Mol Cell Biol. 2014;6(1):81–92. doi: 10.1093/jmcb/mjt026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Raychaudhuri B, Rayman P, Ireland J, Ko J, Rini B, Borden EC, Garcia J, Vogelbaum MA, Finke J. Myeloid-derived suppressor cell accumulation and function in patients with newly diagnosed glioblastoma. Neuro Oncol. 2011;13(6):591–599. doi: 10.1093/neuonc/nor042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wu A, Wei J, Kong LY, Wang Y, Priebe W, Qiao W, Sawaya R, Heimberger AB. Glioma cancer stem cells induce immunosuppressive macrophages/microglia. Neuro Oncol. 2010;12(11):1113–1125. doi: 10.1093/neuonc/noq082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hemmi H, Kaisho T, Takeuchi O, Sato S, Sanjo H, Hoshino K, Horiuchi T, Tomizawa H, Takeda K, Akira S. Small anti-viral compounds activate immune cells via the TLR7 MyD88-dependent signaling pathway. Nat Immunol. 2002;3(2):196–200. doi: 10.1038/ni758. [DOI] [PubMed] [Google Scholar]
- 39.deLeeuw RJ, Kost SE, Kakal JA, Nelson BH. The prognostic value of FoxP3+ tumor-infiltrating lymphocytes in cancer: a critical review of the literature. Clin Cancer Res. 2012;18(11):3022–3029. doi: 10.1158/1078-0432.CCR-11-3216. [DOI] [PubMed] [Google Scholar]
- 40.Loebbermann J, Thornton H, Durant L, Sparwasser T, Webster KE, Sprent J, Culley FJ, Johansson C, Openshaw PJ. Regulatory T cells expressing granzyme B play a critical role in controlling lung inflammation during acute viral infection. Mucosal Immunol. 2012;5(2):161–172. doi: 10.1038/mi.2011.62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Correale P, Rotundo MS, Del Vecchio MT, Remondo C, Migali C, Ginanneschi C, Tsang KY, Licchetta A, Mannucci S, Loiacono L, Tassone P, Francini G, Tagliaferri P. Regulatory (FoxP3+) T-cell tumor infiltration is a favorable prognostic factor in advanced colon cancer patients undergoing chemo or chemoimmunotherapy. J Immunother. 2010;33(4):435–441. doi: 10.1097/CJI.0b013e3181d32f01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Goding SR, Wilson KA, Xie Y, Harris KM, Baxi A, Akpinarli A, Fulton A, Tamada K, Strome SE, Antony PA. Restoring immune function of tumor-specific CD4+ T cells during recurrence of melanoma. J Immunol. 2013;190(9):4899–4909. doi: 10.4049/jimmunol.1300271. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.