Abstract
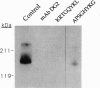
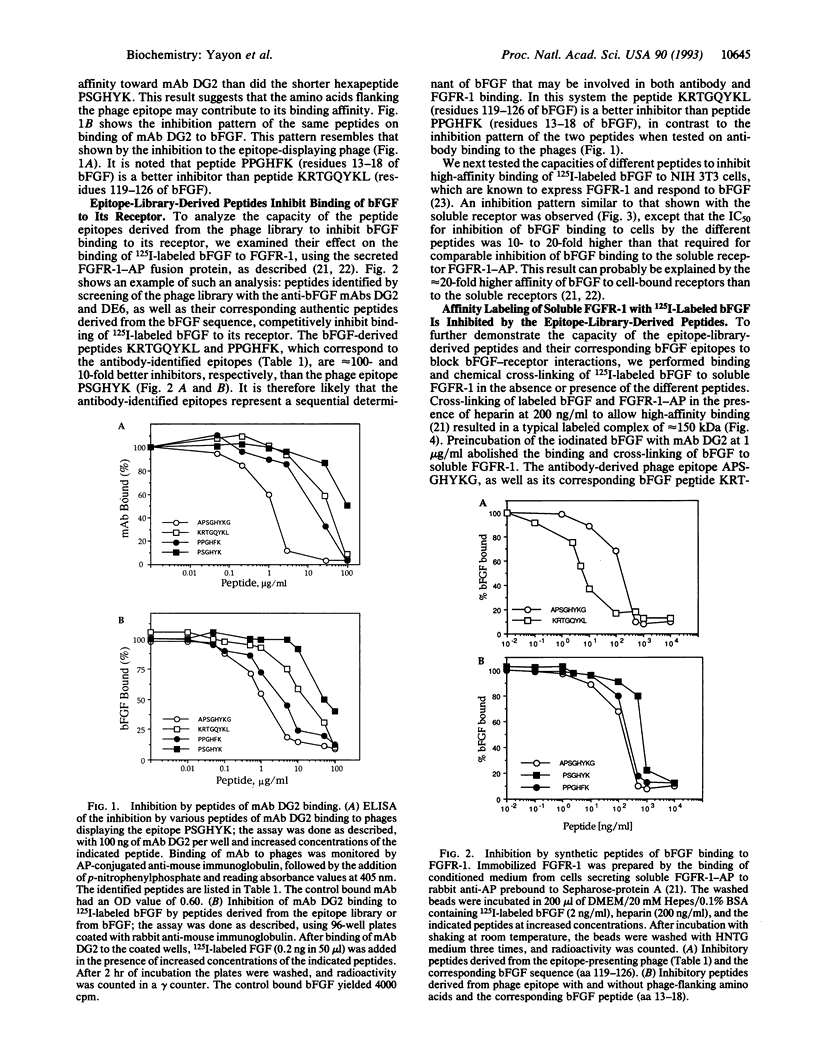
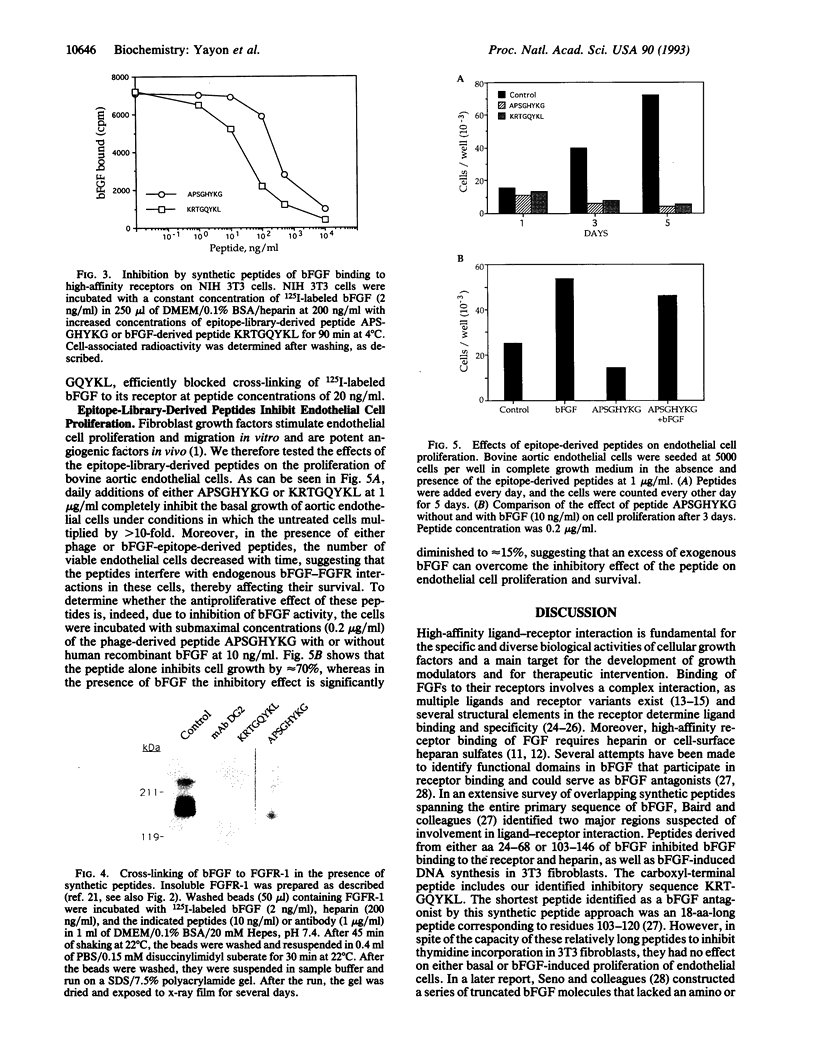
Basic fibroblast growth factor (bFGF) is known to bind to its cell-surface receptors with high affinity and in a heparin-dependent manner. In an attempt to predict the receptor recognition site on bFGF we screened phage-epitope libraries with monoclonal antibodies DG2 and DE6, which inhibit bFGF binding to its receptor. On the affinity-isolated phages, we identified several peptide sequences as the putative antibody-binding epitopes on bFGF. The identified library epitopes shared the consensus sequence Pro-(Pro/Ser)-Gly-His-(Tyr/Phe)-Lys, corresponding to two continuous protein sequences of bFGF: Pro-Pro-Gly-His-Phe-Lys and Arg-Thr-Gly-Gln-Tyr-Lys at amino acids 13-18 and 120-125 of bFGF, respectively. Synthetic peptides of the corresponding phage epitopes or of the above bFGF sequences specifically inhibited binding of the antibodies to bFGF, blocked binding of bFGF to its high-affinity receptor, and inhibited basal and bFGF-induced proliferation of vascular endothelial cells at submicromolar peptide concentrations. The potent inhibition of bFGF binding and biological activity by peptides recognized by the antibodies suggests that these sequences are functionally involved in receptor binding and may constitute part of the receptor-binding determinants on bFGF.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baird A., Schubert D., Ling N., Guillemin R. Receptor- and heparin-binding domains of basic fibroblast growth factor. Proc Natl Acad Sci U S A. 1988 Apr;85(7):2324–2328. doi: 10.1073/pnas.85.7.2324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Basilico C., Moscatelli D. The FGF family of growth factors and oncogenes. Adv Cancer Res. 1992;59:115–165. doi: 10.1016/s0065-230x(08)60305-x. [DOI] [PubMed] [Google Scholar]
- Becker D., Lee P. L., Rodeck U., Herlyn M. Inhibition of the fibroblast growth factor receptor 1 (FGFR-1) gene in human melanocytes and malignant melanomas leads to inhibition of proliferation and signs indicative of differentiation. Oncogene. 1992 Nov;7(11):2303–2313. [PubMed] [Google Scholar]
- Burgess W. H., Maciag T. The heparin-binding (fibroblast) growth factor family of proteins. Annu Rev Biochem. 1989;58:575–606. doi: 10.1146/annurev.bi.58.070189.003043. [DOI] [PubMed] [Google Scholar]
- Burgess W. H., Shaheen A. M., Ravera M., Jaye M., Donohue P. J., Winkles J. A. Possible dissociation of the heparin-binding and mitogenic activities of heparin-binding (acidic fibroblast) growth factor-1 from its receptor-binding activities by site-directed mutagenesis of a single lysine residue. J Cell Biol. 1990 Nov;111(5 Pt 1):2129–2138. doi: 10.1083/jcb.111.5.2129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casscells W., Lappi D. A., Olwin B. B., Wai C., Siegman M., Speir E. H., Sasse J., Baird A. Elimination of smooth muscle cells in experimental restenosis: targeting of fibroblast growth factor receptors. Proc Natl Acad Sci U S A. 1992 Aug 1;89(15):7159–7163. doi: 10.1073/pnas.89.15.7159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eriksson A. E., Cousens L. S., Weaver L. H., Matthews B. W. Three-dimensional structure of human basic fibroblast growth factor. Proc Natl Acad Sci U S A. 1991 Apr 15;88(8):3441–3445. doi: 10.1073/pnas.88.8.3441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Folkman J., Klagsbrun M. Angiogenic factors. Science. 1987 Jan 23;235(4787):442–447. doi: 10.1126/science.2432664. [DOI] [PubMed] [Google Scholar]
- Givol D., Yayon A. Complexity of FGF receptors: genetic basis for structural diversity and functional specificity. FASEB J. 1992 Dec;6(15):3362–3369. [PubMed] [Google Scholar]
- Gross J. L., Herblin W. F., Dusak B. A., Czerniak P., Diamond M. D., Sun T., Eidsvoog K., Dexter D. L., Yayon A. Effects of modulation of basic fibroblast growth factor on tumor growth in vivo. J Natl Cancer Inst. 1993 Jan 20;85(2):121–131. doi: 10.1093/jnci/85.2.121. [DOI] [PubMed] [Google Scholar]
- Gross J. L., Herblin W. F., Eidsvoog K., Horlick R., Brem S. S. Tumor growth regulation by modulation of basic fibroblast growth factor. EXS. 1992;61:421–427. doi: 10.1007/978-3-0348-7001-6_70. [DOI] [PubMed] [Google Scholar]
- Halaban R., Langdon R., Birchall N., Cuono C., Baird A., Scott G., Moellmann G., McGuire J. Basic fibroblast growth factor from human keratinocytes is a natural mitogen for melanocytes. J Cell Biol. 1988 Oct;107(4):1611–1619. doi: 10.1083/jcb.107.4.1611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harper J. W., Lobb R. R. Reductive methylation of lysine residues in acidic fibroblast growth factor: effect on mitogenic activity and heparin affinity. Biochemistry. 1988 Jan 26;27(2):671–678. doi: 10.1021/bi00402a027. [DOI] [PubMed] [Google Scholar]
- Jaye M., Schlessinger J., Dionne C. A. Fibroblast growth factor receptor tyrosine kinases: molecular analysis and signal transduction. Biochim Biophys Acta. 1992 Jun 10;1135(2):185–199. doi: 10.1016/0167-4889(92)90136-y. [DOI] [PubMed] [Google Scholar]
- Johnson D. E., Williams L. T. Structural and functional diversity in the FGF receptor multigene family. Adv Cancer Res. 1993;60:1–41. doi: 10.1016/s0065-230x(08)60821-0. [DOI] [PubMed] [Google Scholar]
- Klagsbrun M. The fibroblast growth factor family: structural and biological properties. Prog Growth Factor Res. 1989;1(4):207–235. doi: 10.1016/0955-2235(89)90012-4. [DOI] [PubMed] [Google Scholar]
- Miki T., Bottaro D. P., Fleming T. P., Smith C. L., Burgess W. H., Chan A. M., Aaronson S. A. Determination of ligand-binding specificity by alternative splicing: two distinct growth factor receptors encoded by a single gene. Proc Natl Acad Sci U S A. 1992 Jan 1;89(1):246–250. doi: 10.1073/pnas.89.1.246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nabel E. G., Yang Z. Y., Plautz G., Forough R., Zhan X., Haudenschild C. C., Maciag T., Nabel G. J. Recombinant fibroblast growth factor-1 promotes intimal hyperplasia and angiogenesis in arteries in vivo. Nature. 1993 Apr 29;362(6423):844–846. doi: 10.1038/362844a0. [DOI] [PubMed] [Google Scholar]
- Oldenburg K. R., Loganathan D., Goldstein I. J., Schultz P. G., Gallop M. A. Peptide ligands for a sugar-binding protein isolated from a random peptide library. Proc Natl Acad Sci U S A. 1992 Jun 15;89(12):5393–5397. doi: 10.1073/pnas.89.12.5393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ornitz D. M., Yayon A., Flanagan J. G., Svahn C. M., Levi E., Leder P. Heparin is required for cell-free binding of basic fibroblast growth factor to a soluble receptor and for mitogenesis in whole cells. Mol Cell Biol. 1992 Jan;12(1):240–247. doi: 10.1128/mcb.12.1.240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rapraeger A. C., Krufka A., Olwin B. B. Requirement of heparan sulfate for bFGF-mediated fibroblast growth and myoblast differentiation. Science. 1991 Jun 21;252(5013):1705–1708. doi: 10.1126/science.1646484. [DOI] [PubMed] [Google Scholar]
- Reilly T. M., Taylor D. S., Herblin W. F., Thoolen M. J., Chiu A. T., Watson D. W., Timmermans P. B. Monoclonal antibodies directed against basic fibroblast growth factor which inhibit its biological activity in vitro and in vivo. Biochem Biophys Res Commun. 1989 Oct 31;164(2):736–743. doi: 10.1016/0006-291x(89)91521-0. [DOI] [PubMed] [Google Scholar]
- Scott J. K. Discovering peptide ligands using epitope libraries. Trends Biochem Sci. 1992 Jul;17(7):241–245. doi: 10.1016/0968-0004(92)90401-t. [DOI] [PubMed] [Google Scholar]
- Scott J. K., Loganathan D., Easley R. B., Gong X., Goldstein I. J. A family of concanavalin A-binding peptides from a hexapeptide epitope library. Proc Natl Acad Sci U S A. 1992 Jun 15;89(12):5398–5402. doi: 10.1073/pnas.89.12.5398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scott J. K., Smith G. P. Searching for peptide ligands with an epitope library. Science. 1990 Jul 27;249(4967):386–390. doi: 10.1126/science.1696028. [DOI] [PubMed] [Google Scholar]
- Seno M., Sasada R., Kurokawa T., Igarashi K. Carboxyl-terminal structure of basic fibroblast growth factor significantly contributes to its affinity for heparin. Eur J Biochem. 1990 Mar 10;188(2):239–245. doi: 10.1111/j.1432-1033.1990.tb15395.x. [DOI] [PubMed] [Google Scholar]
- Werner S., Duan D. S., de Vries C., Peters K. G., Johnson D. E., Williams L. T. Differential splicing in the extracellular region of fibroblast growth factor receptor 1 generates receptor variants with different ligand-binding specificities. Mol Cell Biol. 1992 Jan;12(1):82–88. doi: 10.1128/mcb.12.1.82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yayon A., Klagsbrun M. Autocrine regulation of cell growth and transformation by basic fibroblast growth factor. Cancer Metastasis Rev. 1990 Nov;9(3):191–202. doi: 10.1007/BF00046360. [DOI] [PubMed] [Google Scholar]
- Yayon A., Klagsbrun M. Autocrine transformation by chimeric signal peptide-basic fibroblast growth factor: reversal by suramin. Proc Natl Acad Sci U S A. 1990 Jul;87(14):5346–5350. doi: 10.1073/pnas.87.14.5346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yayon A., Klagsbrun M., Esko J. D., Leder P., Ornitz D. M. Cell surface, heparin-like molecules are required for binding of basic fibroblast growth factor to its high affinity receptor. Cell. 1991 Feb 22;64(4):841–848. doi: 10.1016/0092-8674(91)90512-w. [DOI] [PubMed] [Google Scholar]
- Yayon A., Zimmer Y., Shen G. H., Avivi A., Yarden Y., Givol D. A confined variable region confers ligand specificity on fibroblast growth factor receptors: implications for the origin of the immunoglobulin fold. EMBO J. 1992 May;11(5):1885–1890. doi: 10.1002/j.1460-2075.1992.tb05240.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang J. D., Cousens L. S., Barr P. J., Sprang S. R. Three-dimensional structure of human basic fibroblast growth factor, a structural homolog of interleukin 1 beta. Proc Natl Acad Sci U S A. 1991 Apr 15;88(8):3446–3450. doi: 10.1073/pnas.88.8.3446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu X., Komiya H., Chirino A., Faham S., Fox G. M., Arakawa T., Hsu B. T., Rees D. C. Three-dimensional structures of acidic and basic fibroblast growth factors. Science. 1991 Jan 4;251(4989):90–93. doi: 10.1126/science.1702556. [DOI] [PubMed] [Google Scholar]
- Zimmer Y., Givol D., Yayon A. Multiple structural elements determine ligand binding of fibroblast growth factor receptors. Evidence that both Ig domain 2 and 3 define receptor specificity. J Biol Chem. 1993 Apr 15;268(11):7899–7903. [PubMed] [Google Scholar]