The effect of cotransplanting mononuclear cells from cord blood (CB-MNCs) and mesenchymal stem cells (MSCs) as treatment for myocardial infarction (MI) was studied in rats. Improvement of left ventricular cardiac function was induced by both cell types, and increased tissue cell proliferation in myocardial tissue and neoangiogenesis was observed. However, combination treatment with CB-MNCs plus MSCs was more effective in reducing infarct size and scarring, and preventing ventricular remodeling.
Keywords: Cord blood mononuclear cells, Mesenchymal stem cells, Cotransplantation, Myocardial infarction
Abstract
The aim of this study was to evaluate the effect of cotransplanting mononuclear cells from cord blood (CB-MNCs) and mesenchymal stem cells (MSCs) as treatment for myocardial infarction (MI). Transplanting CD34+ cells or MSCs separately has been shown effective in treating MI, but the effect of cotransplanting CB-MNCs and MSCs is not clear. In this study, MSCs were separated by their adherence to the tissue culture. The morphology, immunophenotype, and multilineage potential of MSCs were analyzed. CB-MNCs were separated in lymphocyte separation medium 1.077. CD34+ cell count and viability were analyzed by flow cytometry. Infarcted male Sprague-Dawley rats in a specific-pathogen-free grade were divided into four treatment groups randomly: group I, saline; group II, CB-MNCs; group III, MSCs; and group IV, CB-MNCs plus MSCs. The saline, and CB-MNCs and/or MSCs were injected intramyocardially in infarcted rats. Their cardiac function was evaluated by echocardiography. The myocardial capillary density was analyzed by immunohistochemistry. Both cell types induced an improvement in the left ventricular cardiac function and increased tissue cell proliferation in myocardial tissue and neoangiogenesis. However, CB-MNCs plus MSCs were more effective in reducing the infarct size and preventing ventricular remodeling. Scar tissue was reduced significantly in the CB-MNCs plus MSCs group. MSCs facilitate engraftment of CD34+ cells and immunomodulation after allogeneic CD34+ cell transplantation. Cotransplanting MSCs and CB-MNCs might be more effective than transplanting MSCs or CB-MNCs separately for treating MI. This study contributes knowledge toward effective treatment strategies for MI.
Significance
This study assessed cotransplantation of hematopoietic stem cells (CD34+ cells) and mesenchymal stem cells (MSCs) for treatment of myocardial infarction (MI) in a rat model. The results demonstrate that MSCs and mononuclear cells from cord blood may have synergistic effects and cotransplantation might be more effective in the treatment of MI than transplanting MSCs and CD34+ cells separately. This study contributes knowledge toward a more effective treatment protocol for MI.
Introduction
In 1976, Friedenstein et al. [1] first described mesenchymal stem cells (MSCs) from bone marrow. Even more encouraging was the successful use by Gluckman et al. [2] in 1988 of umbilical cord blood (UCB) as a source of stem cells for hematopoietic reconstitution in a 5-year-old boy with Fanconi anemia, an inherited bone marrow failure syndrome, that could only be cured by allogeneic hematopoietic stem cell (HSC) transplantation. In the following decades, stem cell transplantation has been used for treating various diseases, including myocardial infarction (MI). Cardiomyocytes have a small potential for renovation and proliferation in vivo after an acute MI. Consequently, the heart muscle has a limited capacity for self-renewal. Cell treatment can promote tissue repair or revitalize damaged tissues, so it has a great potential to decrease the morbidity and mortality associated with cardiovascular diseases.
UCB contains more abundant CD34+ cells and progenitor cells without mobilization [3, 4]. HSC transplantation improves cardiac function in rat [5] and rabbit [6–8] models of MI and reduces myocardial apoptosis in infarcted hearts of db/db mice [9]. CD34+ cells can promote hematopoietic engraftment and reduce the occurrence of graft-versus-host disease (GHVD) in the treatment of heavily transfused patients with severe aplastic anemia [10]. MSCs also are effective in treating MI [11, 12]. Locally injecting induced pluripotent stem cell-derived MSCs alleviates ventricular remodeling and sustains global and regional myocardial strain in the infarcted myocardium [13].
CD34+ cells and MSCs have their own characteristics and advantages in treating MI. MSCs support hematopoiesis and promote hematopoietic cell engraftment and immune recovery after allogeneic CD34+ cell transplantation. They have suppressive effects on immune cells, including T cells, B cells, natural killer cells, and dendritic cells [14]. However, the effect of cotransplanting CD34+ cells and MSCs for treating MI is not clear.
In this study, infarcted male Sprague-Dawley rats in a specific-pathogen-free (SPF) grade were divided into four treatment groups: group I, saline; group II, CB-MNCs; group III, MSCs; group IV, CB-MNCs plus MSCs. The saline, CB-MNCs, and MSCs were injected intramyocardially in infarcted rats. Their cardiac function was evaluated by echocardiography. The myocardial capillary density (MCD) was analyzed by immunohistochemistry. A better effect after cotransplantation provides a valuable strategy in treating MI.
Materials and Methods
The UCB and human umbilical cord were obtained from healthy, full-term, and naturally delivered newborns after written informed consent was obtained from their mothers and family members. The study protocols were reviewed and approved by the hospital review board and ethics committee of Taizhou People’s Hospital, China.
CB-MNCs
Isolation of CB-MNCs
Cord blood (15 ml) and saline (15 ml) were mixed gently. Lymphocyte separation medium (LSM; TBDscience, Tianjin, China) 1.077 (15 ml) was placed in a 50-ml tube. The 30-ml cell suspension was pipetted on top of the separation medium carefully, without disturbing the interphase. The tube was centrifuged at 440g for 40 minutes. Most of the supernatant was carefully aspirated without disturbing the layer of mononuclear cells in the interphase. The mononuclear cells were then aspirated from the interphase. The mononuclear cells were washed with saline and centrifuged at 360g for 10 minutes. This step was repeated. The mononuclear cell preparation was then ready.
CD34+ Count and Viability
CD 45-fluorescein isothiocyanate (FITC) antibodies (Ab) (10 μl), CD 34-PE Ab reagent (10 μl), and 7-aminoactinomycin D reagent (10 μl) (BD Biosciences, Sparks, MD, http://www.bd.com) were pipetted into a tube. Then 50 μl of the well-mixed sample was pipetted to the bottom of the tube. This tube was vortexed and incubated at room temperature for 15 minutes and protected from light. After incubation, 1 ml of 1× lysing reagent was added to the tube. The tube was again vortexed and incubated at room temperature for 10 minutes. After incubation, the tube was centrifuged at 300g for 5 minutes. The supernatant was discarded, and 1 ml of phosphate-buffered saline (PBS) was added to the tube, mixed, and centrifuged again at 300g for 5 minutes. The supernatant was discarded and 350 µl of PBS was added to the tube and mixed. The test was conducted using the International Society of Hematotherapy and Graft Engineering protocol by BD FACSCanto II (BD Biosciences).
MSCs
Isolation and Culture of MSCs
MSCs were separated and cultured according to previously published reports [15, 16]. The cells were observed under a microscope (IX71; Olympus Corp., Tokyo, Japan, http://www.olympus-global.com) and their morphology recorded.
Immunophenotyping of MSCs
The surface molecules on the MSCs were examined by flow cytometry (BD FACSCanto П; BD Biosciences) with the following antibodies: CD34-PE, CD45-FITC, CD73-PE, CD14-FITC, CD79a-APC, HLA-DR-PE, and CD90-APC (BD, Biosciences), and CD105-PE (eBioscience, San Diego, CA, http://www.ebioscience.com).
Differentiation Potential of MSCs
Osteogenic and adipogenic differentiation was assessed according to the methods of previous reports [15, 16].
Rat Model of MI
Healthy, male Sprague-Dawley rats (250–300 g) were provided by the Animal Center of Second Military Medical University, China. The procedure for the animal experiment was approved by the Institutional Animal Care and Use Committee at the Animal Center of the Second Military Medical University.
Preparation of the MI Model
The rats were anesthetized with 10% chloral hydrate (0.3 ml per 100 g) in the abdomen. They were then fixed on the bench, the hair removed from the abdominal operation area, and the rats were connected to a ventilator. The chest was opened first, then the pericardium, and, finally, the heart was exposed. The left atrial appendage was ligated at a distance of 1 mm (equivalent to the left anterior descending coronary artery) with a no. 5 atraumatic suture. The front wall of the left ventricle became white, and the action of the heart weakened. These are signs of the MI model success. The thoracotomy was then closed layer by layer. The rats were recovered with meperidine analgesia (1.5 mg/kg). The bronchial intubation was extubated after the rats breathed spontaneously and smoothly. Penicillin (400,000 IU/day) was intramuscularly injected to prevent infection.
Identification of the MI Model
Four rats were selected randomly and killed by injecting potassium chloride after the operation (24 hours and 1 week, respectively). Their hearts were removed and the shape, infarct size, and pathological morphology of each heart were observed using hematoxylin-and-eosin (H&E) staining.
CB-MNC and MSC Transplantation
Seven days after inducing MI, the animals were placed under inhalation anesthesia with sevoflurane. The position of the heart was identified using cardiac ultrasound technology. For CD34+ cells, based on a 20-kg recipient, a target dose of ≥2.5× 107 nucleated cells per kilogram was infused [17]. Cells were transplanted as follows: group I, 500 μl saline; group II, human UCB mononuclear cells containing 6.0 × 105 CD34+ cells; group III, 2.0 × 106 viable MSCs; group IV, 1.0 × 106 viable MSCs plus mononuclear cells comprising 3.0 × 105 CD34+ cells. The saline, CB-MNCs, and MSCs were injected intramyocardially at four points in the infarct border zone. Seven days after the first transplantation, the process was repeated.
Detection of the Recovery of Cardiac Function in MI Rats After Transplantation
Assessment of Function by Echocardiography
Transthoracic echocardiography with a 13-MHz linear-array transducer was performed using an echocardiographic system after the second and fourth weeks of the second cell transplantation. Parameters were measured at baseline, after infarction, and after transplantation (second and fourth weeks) when the animals were under inhalation anesthesia with sevoflurane. The left ventricular dimension (LVD) in end diastole (LVDd) and end systole (LVDs), anterior wall thickness (AWT), anterior wall diastole thickness (AWd), anterior wall systole thickness (AWs), posterior wall thickness in diastole and systole, end-diastolic volume (EDV), and end-systolic volume (ESV) were measured. Changes in AW were calculated as follows: [(AWs − AWd)/AWd] ×100. The fractional shortening (FS) was calculated as follows: [(LVDd − LVDs)/LVDd] ×100. The left ventricular ejection fraction (LVEF) was calculated as follows: [(LVD − LVDs)/LVD] ×100.
MCD Analysis by Immunohistochemistry and Mallory Staining
Four weeks after the second transplantation, the rats were killed and the heart tissues were removed. Immunohistochemical detection of vessels was performed with anti-rat CD31 (Merck Millipore, Beijing, China, https://www.merckmillipore.com). Vessels were counted in 10 fields in the peri-infarct zone at a magnification of ×200 with a light microscope and Image Pro-Plus 5.1 software (Media Cybernetics, Inc., MD, http://www.mediacy.com) and reported as the number of vessels per unit area (mm2). The fibrotic area in the left ventricle was calculated by Mallory staining.
Statistical Analysis
Data were expressed as mean ± SEM. The results were analyzed by SPSS software (IBM Corp., Armonk, NY, http://www-01.ibm.com). Pre- and posttransplantion comparisons for each group were performed with a paired Student t test. Comparisons of the control and transplantation groups were performed by analysis of variance. A 5% probability (p < .05) was used as the level of significance.
Results
CB-MNCs
The mononuclear cells were separated from the UCB by density gradient centrifugation. The percentage of CD34+ count and cell viability of mononuclear cells were analyzed by flow cytometry. Mononuclear cells with a viability of >85% were chosen (Fig. 1).
Figure 1.
The percentage of CD34+ cells and viability of the mononuclear cells were analyzed by flow cytometry. Abbreviations: FITC-A, fluorescein isothiocyanate-A; SSC-A, side scatter pulse area.
MSCs
The morphology was observed under a microscope (IX71; Olympus). The surface molecules on the MSCs were then examined by flow cytometry (BD FACSCantoII).The multilineage potential was induced and observed at different magnifications under a microscope. The biological properties of umbilical cord–derived MSCs met the standards set forth by the International Society for Cellular Therapy to define human MSCs (Fig. 2).
Figure 2.
Analysis of mesenchymal stem cells’ morphology (A), immunophenotype (B), and multilineage potential (C).
MI Model
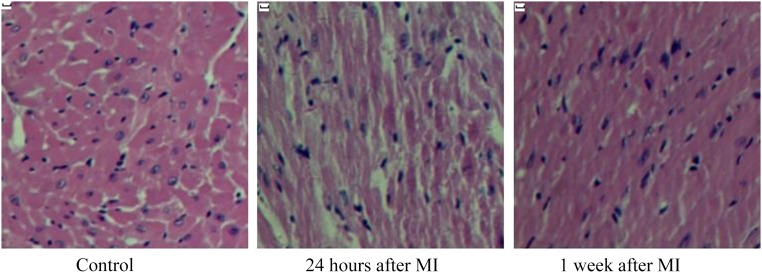
The initial number of rats prepared for the study was 60. Rats with FS higher than 35% after MI were excluded. The overall mortality due to surgical procedures was close to 27%. The infarcted heart tissues were observed by H&E staining. After MI, coagulation necrosis was found in myocardial fibers, and edema was accompanied by hemorrhage. The neutrophils began to infiltrate, and chromatic agglutination was seen in the nuclear envelope under light microscope (Fig. 3).
Figure 3.
The infarcted heart tissue was observed with hematoxylin and eosin staining. Twenty-four hours after MI, myocardial cell degeneration and necrosis was evident. At 1 week after MI, inflammatory cell infiltration was observed. Magnification, ×200. Scale bars = 50 μm. Abbreviation: MI, myocardial infarction.
Recovery of Cardiac Function in MI Rats After Transplantation
The echocardiographic parameters from all groups are listed in Table 1. Changes were monitored at baseline, after MI, and after transplantation (second and fourth weeks). FS, AWT, and LVEF showed significant improvement in cotransplantation group IV 4 weeks after transplantation (p < .01). The LVEF showed some improvement in the cell-treated group, significantly in group IV (Fig. 4, Table 1).
Table 1.
Echocardiographic parameters of all groups at different time points
Figure 4.
FS, AWT, and LVEF showed significant improvement in cotransplantation group IV, 4 weeks after transplantation (p < .01). The LVEF showed some improvement in the cell-treated group, significantly in group IV. Values are the mean ± SEM corresponding to n = 9, 8, 8, and 7 for the saline, CD34, MSCs, and MSCs plus CD 34 groups, respectively. ∗, p < .05; ∗∗, p < .01. Abbreviations: 2W, 2 weeks; 4W, 4 weeks; AWT, anterior wall thickness; FS, fractional shortening; LVEF, left ventricular ejection fraction; MI, myocardial infarction; MSC, mesenchymal stem cell.
Vascular Density Analysis and Mallory Staining
To assess the treatment effect of different groups, ultrathin sections were analyzed by immunohistochemistry. CD31 expression was positive during neoangiogenesis. The cotransplantation group had a larger number of vessels per unit area compared with the other groups. The total number of vessels per mm2 ± SD was 584 ± 59 in the saline group, 809 ± 68 in group II, 792 ± 63 in group III, and 878 ± 74 in group IV. Vascular density was higher in the cell-treated group than in the saline group (p < .05). Neoangiogenesis was more obvious in group IV than in groups II or III.
Cross-sections of the hearts from each group of animals were stained with Mallory stain, and the infarcted area in the left ventricle was calculated. The CD34+ cell group had a smaller proportion of fibrous scar tissue compared with the saline control group (19.6% ± 1.3% vs. 22.3% ± 1.4%; p < .05). The cotransplantation group had a smaller proportion of fibrous scar tissue (13.5% ± 0.9% vs. 22.3% ± 1.4%; p < .01) (Fig. 5).
Figure 5.
Neoangiogenesis and reducing infarct size in infarct area after transplantation. (A): Neovascularization in the infarct area stained with anti-CD31, 4 weeks after transplantation. The cotransplantation group had larger number of vessels per unit area compared with the other groups (p < .05). (B): Representative heart sections from different groups are shown. Fibrotic areas in the left ventricle were calculated in Mallory-stained sections. Values are the mean ± SEM corresponding to n = 9, 8, 8, and 7 for the saline, CD34, MSCs, and MSCs plus CD34 groups, respectively. ∗, p < .05, ∗∗, p < .01. Scale bars = 25 μm. Abbreviation: MSC, mesenchymal stem cell.
Discussion
CD34+ cells have been used for treating malignant diseases such as MI [5–9, 18, 19], hematological malignancies [20–22], Hurler syndrome [23, 24], Krabbe disease [25], primary immunodeficiency diseases [26], bone marrow failure [27], and β-thalassemia [28]. Along with the advantages, CD34+ cells also have limitations. These cells induced intestinal GHVD in an MI rat model [5].
MSCs, but not CD34+ cells, can be mobilized and differentiated into cardiomyocytes after MI in mice [29]. MSCs have suppressive effects on immune cells, besides supporting hematopoiesis [14]. CD34+ cells and MSCs have their respective superiority and inferiority in treating MI. Transplanting CD34+ cells or MSCs separately proved effective in treating MI. However, the more appropriate strategies for treating MI remain to be determined.
In this study, the treatment effect of four groups were compared: saline, CB-MNCs, MSCs, and CB-MNCs plus MSCs. Cells or saline were delivered to the heart by intramyocardial injection at four points in the infarct border zone. The data demonstrated that CD34+ cells and/or MSCs aid cardiac repair. The outcomes were consistent with those reported previously [5, 6, 8]. At 2 weeks after transplantation, no significant improvement was observed in ventricular performance in terms of FS and LVEF in cell-treated groups. Four weeks later, AWT, FS, and LVEF were significantly improved in group IV and showed some improvement in groups II and III. Immunohistochemistry analysis and Mallory staining showed that cotransplantation reduced infarct area and thickened the left ventricle (LV) wall more significantly compared with the results after transplantation of MSCs and CB-MNCs separately. Mallory staining showed the LV anterior wall peri-infarct zones increased obviously in groups III and IV compared with those in groups I and II. This may be because MSCs can maintain their stemness during proliferation. In contrast, HSCs easily differentiated during proliferation and failed to exert their influence on cardiac repair continuously. MSC transplantation was more effective than CB-MNC transplantation, which is consistent with findings reported previously [30]. MSCs and CB-MNCs can repair infarcted myocardium by preventing ventricular remodeling, reducing infarct area, and increasing vascular density.
In treating MI, the therapeutic effect of MSCs is not limited to hematopoiesis support. MSCs were confirmed to facilitate HSC engraftment and immunomodulation after allogeneic HSC transplantation [14]. The repair mechanism of stem cells in MI is controversial. Cell differentiation or fusion cannot fully explain the obvious improvement of the cardiac function [31]. General consensus supports the paracrine hypothesis to explain cardiac repair [30]. Recent studies have shown that MSCs can secrete growth factors and cytokines that can exert their influence on cardiac repair through paracrine modes after MI [31]. Stem cells are said to be cell factories that secrete cytokines. CB-MNCs and MSCs secrete different cytokines that may interact and cascade.
Conclusion
MSCs and CB-MNCs may have synergistic effects, and cotransplantation might be more effective than transplantation of MSCs and CB-MNCs separately for treating MI. This study contributes to knowledge of more effective treatment strategies for MI.
Author Contributions
G.C.: conception and design, provision of study material or patients, data analysis and interpretation, manuscript writing, final approval of manuscript; A.Y.: collection and/or assembly of data, data analysis and interpretation, manuscript writing, final approval of manuscript; H.Y.: collection and/or assembly of data, final approval of manuscript; Z.R., Y.Y., R.W., and Y. R.: data analysis and interpretation, final approval of manuscript; L.Z.: financial support, administrative support, final approval of manuscript.
Disclosure of Potential Conflicts of Interest
The authors indicated no potential conflicts of interest.
References
- 1.Friedenstein AJ, Gorskaja JF, Kulagina NN. Fibroblast precursors in normal and irradiated mouse hematopoietic organs. Exp Hematol. 1976;4:267–274. [PubMed] [Google Scholar]
- 2.Gluckman E, Rocha V, Chastang C, et al. Cord blood banking and transplant in Europe. Eurocord. Bone Marrow Transplant. 1998;22(suppl 1):S68–S74. [PubMed] [Google Scholar]
- 3.McKenna D, Sheth J. Umbilical cord blood: Current status & promise for the future. Indian J Med Res. 2011;134:261–269. [PMC free article] [PubMed] [Google Scholar]
- 4.Lee MW, Jang IK, Yoo KH, et al. Stem and progenitor cells in human umbilical cord blood. Int J Hematol. 2010;92:45–51. doi: 10.1007/s12185-010-0619-4. [DOI] [PubMed] [Google Scholar]
- 5.Xing Y, Wu Y, Wang L, et al. Human umbilical cord blood cell transplantation improves cardiac function in a myocardial infarction rat model but induces intestinal graft versus host disease. Cell Mol Biol (Noisy-le-grand) 2014;60:6–12. [PubMed] [Google Scholar]
- 6.Jun Y, Chunju Y, Qi A, et al. The effects of compound Danshen dripping pills and human umbilical cord blood mononuclear cell transplant after acute myocardial infarction. Exp Clin Transplant. 2014;12:123–128. [PubMed] [Google Scholar]
- 7.Li T, Ma Q, Ning M, et al. Cotransplantation of human umbilical cord-derived mesenchymal stem cells and umbilical cord blood-derived CD34⁺ cells in a rabbit model of myocardial infarction. Mol Cell Biochem. 2014;387:91–100. doi: 10.1007/s11010-013-1874-5. [DOI] [PubMed] [Google Scholar]
- 8.Yuan C, Yu G, Yang T, et al. Enhanced therapeutic effects on acute myocardial infarction with multiple intravenous transplantation of human cord blood mononuclear cells. Int J Cardiol. 2013;168:2767–2773. doi: 10.1016/j.ijcard.2013.03.131. [DOI] [PubMed] [Google Scholar]
- 9.Zeng H, Li L, Chen JX. Overexpression of angiopoietin-1 increases CD133+/c-kit+ cells and reduces myocardial apoptosis in db/db mouse infarcted hearts. PLoS One. 2012;7:e35905. doi: 10.1371/journal.pone.0035905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wang H, Wang Z, Zheng X, et al. Hematopoietic stem cell transplantation with umbilical cord multipotent stromal cell infusion for the treatment of aplastic anemia--a single-center experience. Cytotherapy. 2013;15:1118–1125. doi: 10.1016/j.jcyt.2013.04.007. [DOI] [PubMed] [Google Scholar]
- 11.Williams AR, Hare JM. Mesenchymal stem cells: biology, pathophysiology, translational findings, and therapeutic implications for cardiac disease. Circ Res. 2011;109:923–940. doi: 10.1161/CIRCRESAHA.111.243147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ko IK, Kim BS. Mesenchymal stem cells for treatment of myocardial infarction. Int J Stem Cells. 2008;1:49–54. doi: 10.15283/ijsc.2008.1.1.49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Miao Q, Shim W, Tee N, et al. iPSC-derived human mesenchymal stem cells improve myocardial strain of infarcted myocardium. J Cell Mol Med. 2014;18:1644–1654. doi: 10.1111/jcmm.12351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hao L, Sun H, Wang J, et al. Mesenchymal stromal cells for cell therapy: Besides supporting hematopoiesis. Int J Hematol. 2012;95:34–46. doi: 10.1007/s12185-011-0991-8. [DOI] [PubMed] [Google Scholar]
- 15.Chen G, Yue A, Ruan Z, et al. Monitoring the biology stability of human umbilical cord-derived mesenchymal stem cells during long-term culture in serum-free medium. Cell Tissue Bank. 2014;15:513–521. doi: 10.1007/s10561-014-9420-6. [DOI] [PubMed] [Google Scholar]
- 16.Chen G, Yue A, Ruan Z, et al. Human umbilical cord-derived mesenchymal stem cells do not undergo malignant transformation during long-term culturing in serum-free medium. PLoS One. 2014;9:e98565. doi: 10.1371/journal.pone.0098565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.U.S. Department of Health and Human Services, Food and Drug Administration, Center for Biologics Evaluation and Research Guidance for industry. Minimally manipulated, unrelated allogeneic placental/umbilical cord blood intended for hematopoietic reconstitution for specified indications. Rockville, MD: U.S. Department of Health and Human Services, 2009.
- 18.Acosta SA, Franzese N, Staples M, et al. Human umbilical cord blood for transplantation therapy in myocardial infarction. J Stem Cell Res Ther. 2013;(suppl 4):S4–S005. [PMC free article] [PubMed] [Google Scholar]
- 19.Poliachenko IV, Habriielian AV, Domans’kyĭ TM, et al. [Effects of the umbilical blood stem cells in experimental injury of myocardium] Klin Khir. 2013;(7):56–60. [PubMed] [Google Scholar]
- 20.Kobayashi K, Maeda Y, Hara Y, et al. Clinical outcomes of unrelated donor umbilical cord blood transplantation for 30 adults with hematological malignancies. Anticancer Res. 2009;29:1763–1770. [PubMed] [Google Scholar]
- 21.Kindwall-Keller TL, Hegerfeldt Y, Meyerson HJ, et al. Prospective study of one- vs two-unit umbilical cord blood transplantation following reduced intensity conditioning in adults with hematological malignancies. Bone Marrow Transplant. 2012;47:924–933. doi: 10.1038/bmt.2011.195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wallet HL, Sobh M, Morisset S, et al. Double umbilical cord blood transplantation for hematological malignancies: A long-term analysis from the SFGM-TC registry. Exp Hematol. 2013;41:924–933. doi: 10.1016/j.exphem.2013.05.297. [DOI] [PubMed] [Google Scholar]
- 23.Dusing SC, Thorpe DE, Poe MD, et al. Gross motor development of children with Hurler syndrome after umbilical cord blood transplantation. Phys Ther. 2007;87:1433–1440. doi: 10.2522/ptj.20070017. [DOI] [PubMed] [Google Scholar]
- 24.Boelens JJ, Rocha V, Aldenhoven M, et al. Risk factor analysis of outcomes after unrelated cord blood transplantation in patients with Hurler syndrome. Biol Blood Marrow Transplant. 2009;15:618–625. doi: 10.1016/j.bbmt.2009.01.020. [DOI] [PubMed] [Google Scholar]
- 25.Yagasaki H, Kato M, Ishige M, et al. Successful cord blood transplantation in a 42-day-old boy with infantile Krabbe disease. Int J Hematol. 2011;93:566–568. doi: 10.1007/s12185-011-0835-6. [DOI] [PubMed] [Google Scholar]
- 26.Morio T, Atsuta Y, Tomizawa D, et al. Outcome of unrelated umbilical cord blood transplantation in 88 patients with primary immunodeficiency in Japan. Br J Haematol. 2011;154:363–372. doi: 10.1111/j.1365-2141.2011.08735.x. [DOI] [PubMed] [Google Scholar]
- 27.Bizzetto R, Bonfim C, Rocha V, et al. Outcomes after related and unrelated umbilical cord blood transplantation for hereditary bone marrow failure syndromes other than Fanconi anemia. Haematologica. 2011;96:134–141. doi: 10.3324/haematol.2010.027839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Soni S, Breslin N, Cheerva A. Successful unrelated umbilical cord blood transplantation for class 3 β-thalassemia major using a reduced-toxicity regimen. Pediatr Transplant. 2014;18:E41–E43. doi: 10.1111/petr.12201. [DOI] [PubMed] [Google Scholar]
- 29.Fukuda K, Fujita J. Mesenchymal, but not hematopoietic, stem cells can be mobilized and differentiate into cardiomyocytes after myocardial infarction in mice. Kidney Int. 2005;68:1940–1943. doi: 10.1111/j.1523-1755.2005.00624.x. [DOI] [PubMed] [Google Scholar]
- 30.Armiñán A, Gandía C, García-Verdugo JM, et al. Mesenchymal stem cells provide better results than hematopoietic precursors for the treatment of myocardial infarction. J Am Coll Cardiol. 2010;55:2244–2253. doi: 10.1016/j.jacc.2009.08.092. [DOI] [PubMed] [Google Scholar]
- 31.Wen Z, Zheng S, Zhou C, et al. Repair mechanisms of bone marrow mesenchymal stem cells in myocardial infarction. J Cell Mol Med. 2011;15:1032–1043. doi: 10.1111/j.1582-4934.2010.01255.x. [DOI] [PMC free article] [PubMed] [Google Scholar]