Abstract
A fast and reproducible microwave-driven protocol has been developed for the synthesis of neridronate-functionalized nanoparticles. Starting from the synthesis of hydrophobic nanoparticles, our method is based on an adaptation from thermal decomposition method to microwave driven synthesis. The new methodology produces a decrease in the reaction times in comparison with traditional procedures. Moreover, the use of the microwave technology increases the reproducibility of the reactions, something important from the point of view of clinical applications. The novelty of this iron oxide nanoparticle is the attachment of Neridronate. The use of this molecule leads a bisphosphonate moiety towards the outside of the nanoparticle that provides Ca2+ binding properties in vitro and selective accumulation in vivo in the atheroma plaque. The protocol allows the synthesis and plaque detection in about 3 hr since the initial synthesis from organic precursors. Their accumulation in the atherosclerotic area in less than 1 hr provides a contrast agent particularly suitable for clinical applications.
Keywords: Bioengineering, Issue 109, Iron oxide nanoparticles, microwave synthesis, bisphosphonates, calcium deposits, magnetic resonance imaging, atheroma plaque.
Introduction
Atherosclerosis is a multifactorial chronic inflammatory disease of the arterial wall resulting from a deregulated lipid metabolism and a defective inflammatory response. Due to the prevalence and the economical and social costs of this and related cardiovascular diseases there is a growing interest in addressing the pathology with new tools, of which nanotechnology is one of the most promising.1-3 However there are very few examples of fast production and characterization of probes which is basic for translation to the clinic.4 In this protocol we use a microwave synthesis of iron oxide nanoparticle for further functionalization with a bisphosphonate and in vivo detection of atherosclerosis in ApoE-/- mice in 1 hr.5 Iron oxide nanoparticles (IONP) are a well-known nanomaterial and its use as a contrast agent for Magnetic Resonance Imaging (MRI) has been established for the detection of different diseases in the last years.6-8
Microwave synthesis (MWS), allows synthesizing nanoparticles in extremely short times with high reproducibility and enhanced yields.9,10 In our protocol we obtain IONP with plaque targeting capabilities in three steps. The final one is the attachment of an aminobisphosphonate, Neridronate, which is key in our strategy due to its calcium-binding properties. Due to their natural analogue pyrophosphate (PPi), Neridronate has been used in the treatment of Osteogenesis Imperfecta (OI) and Paget's disease of bone (PDB) for their high affinity towards bone mineral.11-13
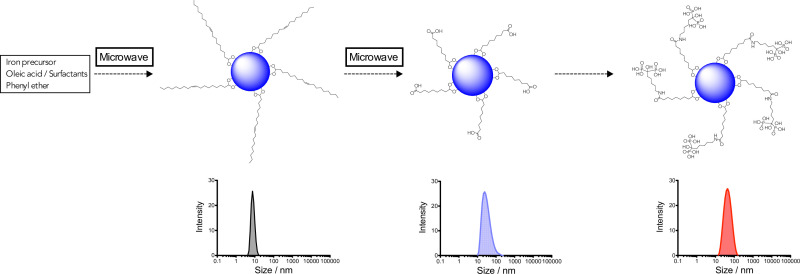
The three steps of the protocol are summarized in scheme 1. Steps one and two are carried out using microwave technology. First step provide oleic acid-coated iron oxide nanoparticles (OA-IONP) by a modification of published methods.14 The protocol is an adaptation to microwave synthesis of the traditional thermal decomposition synthesis.15,16 A mixture containing Fe(acac)3, oleic acid, oleylamine and 1,2-dodecanediol is dissolved in benzyl alcohol and subjected at two heating processes. Purification is carried out washing with EtOH and collecting the particles with a Nd-Fe-B magnet to eliminate the excess of surfactants in the supernatant. Then, OA-IONP are stabilized in CHCl3. As expected, due to the very fast heating, anticipated results showed that the nanoparticles synthesized by microwave are smaller in terms of core (3.7 ± 0.8 nm) and hydrodynamic size (7.5 nm) in comparison with traditional thermal decomposition; however, nanoparticles still present an excellent crystallinity.
The second step consists in a direct chemical modification of the double bond, present in the oleic acid, using a strong oxidant like KMnO4, the original methodology developed in our group was modified for MW conditions.17 A first stage forms the complexes between MnO4- and the double bond. Then, a second stage in acidic conditions, produce the cleavage of the oleic acid molecule giving Azelaic acid-IONP. After these two stages of 9 min each, the sample is purified, first washing with NaHSO3 1% to reduce the excess of MnO4- to MnO2 and then with NaOH 1% to neutralize the acid.
After the purification step, Azelaic-IONP are stabilized in 10 mM phosphate buffer pH = 7.2. This buffer is the best environment for the colloidal stability of the particles similarly to what happened in the original, thermal reaction.18 The use of microwave for the direct oxidation of the double bond contained in OA-IONP is a very good example of the advantages of using this technology in the synthesis of nanoparticles. With the classical method the reaction takes 24 hr, the utilization of microwave decrease the reaction time to 18 min. Moreover, the microwave-driven protocol shows an excellent reproducibility giving nanoparticles with 30 ± 5 nm of hydrodynamic size after 4 repetitions. Apart of the change in the hydrodynamic size, the zeta potential is a good parameter to quickly check the successful of the reaction. Due to the presence of the new carboxylic groups in Azelaic-IONP, the value for the zeta potential is around -44 mV, very similar to the value obtained by the thermal approach.
For the attachment of neridronate to Azelaic-IONP, traditional EDC/sulfo-NHS conjugation is used.19 This synthetic approach is well-established since employing an activated carboxylate with the sulfo-NHS ensures colloidal stability during the reaction. After the elimination of phosphate buffer the reaction with neridronate is carried out in 1 mM HEPES buffer (pH ~7). The reaction renders Neridronate-IONP with a hydrodynamic size of 40 ± 4 nm in a narrow size distribution and -24.1 mV of zeta-potential.
The procedure is described for fast synthesis of IONP for in vivo visualization of atherosclerotic plaque although the feasibility of the method allows the attachment of any peptide/antibody with free amines, using the same conditions, for different purposes within T2-weighted contrast agent MRI field.
Protocol
1. Preparation of Reagents
Prepare 1 mM HEPES Buffer dissolving 23.8 mg of HEPES in 100 ml of distilled water. Adjust the pH to 7.
Prepare 10% NaHSO3 dissolving 10 g of NaHSO3 in 100 ml of distilled water. Stir the mixture for 15 min.
Prepare NaOH solution dissolving 1 g of NaOH in 100 ml of water. Stir for 10 min.
Prepare 10 mM Phosphate buffer dissolving 600 mg of NaH2PO4 in 1 L of water. Add carefully 0.34 ml of phosphoric acid and Stir for 30 min. Adjust the pH to 2.9 (acceptance range 2.7-3.0).
Prepare 10 mM Phosphate buffer dissolving 269 mg of NaH2PO4 and 1.09 g of Na2HPO4 to distilled water to make a volume of 1 L. Adjust the pH to 7.2.
2. Synthesis of Oleic Acid Coated Nanoparticles (OA-IONP)
In a microwave adapted flask add 0.5 g of Fe(acac)3, 1.4 ml of oleic acid, 0.6 ml of oleylamine and 1.19 g of 1,2-hexadodecanediol. Add 10 ml of phenyl ether carefully through the flask wall using a graduated pipette.
Introduce the flask in the microwave reactor and start the microwave protocol. NOTE: Microwave software enables choosing the magnitude for different parameters like temperature, pressure, stirring speed, power and reaction time. Moreover, it has the possibility to load three different stages in the same protocol allowing tunable synthesis. Once the loaded synthetic protocol starts, microwave heats the sample as fast as possible (ramping process) and maintains it during the chosen reaction time (running process). Election of power determines the time of ramping.
- Load a dynamic study in the microwave. The protocol contains three stages:
- Stage 1: set the temperature to 60 °C, time 2 min, pressure 250 psi and 150 W of power. Stir speed has to be in high position and max power in on.
- Stage 2: set the temperature to 200 °C, time 20 min, pressure 250 psi and 300 W of power. Stir speed has to be in high position and max power in on.
- Stage 3: set the temperature to 250 °C, time 10 min, pressure 250 psi and 300 W of power. Stir speed has to be in high position and max power in on.
After finishing the protocol, allow the flask to cool at room temperature. NOTE: Cooling down process can be done with or without gas stream. Both cases provide same results. Aggregates appear in the stir bar and in the wall of the flask, wash it with EtOH and put it onto the Erlenmeyer.
Transfer the reaction mixture to an Erlenmeyer using a glass pipette and add 10 ml of EtOH 98%. Put a Nd-Nb-B magnet below the flask, wait 5 min and remove the supernatant with a glass pipette.
Add 10 ml of EtOH, sonicate the sample at RT for 2 min and 40 kHz, put the sample on the magnet and eliminate the supernatant. Repeat this step at least three times.
Disperse oleic nanoparticles in 30 ml of CHCl3 and sonicate at 40 kHz for 5 min at RT. Check the hydrodynamic size in the zetasizer as per manufacturer's instructions. Put 0.5 ml of OA-IONP in the glass cuvette and add 0.5 ml of CHCl3. Acceptance range 7-10 nm expressed as Z-average size in intensity. NOTE: OA-IONP can be well-dispersed in hexane.
3. Synthesis of Azelaic Acid Nanoparticles (Azelaic Acid-IONP)
Dissolve 44.3 mg of KMnO4 and 150.4 mg of BTACl in a mixture of H2O:CHCl3 (3:2 ml). Add the resultant solution to a 5 ml aliquot of OA-IONP in the microwave adapted flask.
Start the microwave protocol for Azelaic acid-IONP. Set temperature at 105 °C, time 9 min, pressure 250 psi and power at 300 W. Put 10 ml of phosphate buffer pH = 2.9 into the flask and repeat the microwave protocol. After cooling step, recover the nanoparticles using a magnet and eliminate the supernatant.
Add 5 ml of 10% NaHSO3 to an Erlenmeyer flask, sonicate at 40 kHz for 2 min at 25 °C, collect the particles using a magnet and eliminate the supernatant. (The step is repeated 2 times.) Wash the nanoparticles three more times with 1% NaOH and finally re-disperse it in 5 ml of phosphate buffer pH = 7.2.
Check the hydrodynamic size and zeta potential. Put 0.7 ml of Azelaic-IONP into the disposable folded capillary cell and insert it onto the zetasizer. NOTE: Acceptance range for size 25-35 nm expressed as Z-average size in intensity. Acceptance range for Z-Potential -45 ± 5 mV. Larger nanoparticles (~70 nm) can be obtained with phosphate buffer pH >7 instead pH = 2.9 (ref Chem Eur J 2008).
4. Synthesis of Neridronate Nanoparticles (Neridronate-IONP)
Add 12 mg of EDC and 15 mg of Sulfo-NHS in an centrifuge with 2 ml aliquot of Azelaic-IONP. Put the mixture in a vortex at RT for 35 min.
Put a magnet below the centrifuge to destabilize the nanoparticles, aspirate the supernatant and wash the particles with 1.5 ml of HEPES 1 mM pH=7 buffer. (Repeat this step two times.) Afterwards, add 5 mg of neridronate and shake the mixture in a vortex for 2 hr.
Separate nanoparticles with a magnet and wash (3 x 2 ml) with 1 mM HEPES pH=7 buffer. Finally, disperse Neridronate-IONP in 2 ml of 1mM HEPES pH = 7 buffer.
Check the hydrodynamic size and zeta potential. Put 0.7 ml of Neridronate-IONP into the disposable folded capillary cell and insert it onto the zetasizer (see equipment setup). NOTE: Acceptance range for size 40-45 nm expressed as Z-average size in intensity. Acceptance range for Z-Potential -20 ± 5 mV.
5. In Vivo Detection of Atheroma Plaque in ApoE-/- Mice by MRI
- Preparation for the MRI Acquisition NOTE: Several extra systems for animal experimentation are needed. Thus, It will require:
- Use appropriate equipment to anesthetize the animals.
- Obtain a close circuit-circulating warm water system with an external warm air to keep the temperature of the animal stable. NOTE: In this case the MRI compatible monitoring and gating system registers the temperature of the animal inside the MRI magnet.
- Monitor the external temperature in the proximity of the animal, the body (rectal thermometer) temperature of the animal, the respiration sensor located under the body of the animal near the thorax using an integrated interface in the MRI console.
- MRI Experiment
- Anesthetize the animals with vaporized isoflurane (2% for induction during two or three minutes and 1-1.5% for maintenance during the MRI experiment) with an 100% oxygen line.
- Place the animal in the center of the magnet with the help of a profile acquisition.
- After step 5.2.2, tune the RF coil to 300 MHz (7 T) resonance frequency and match the characteristic impedance of the coil to 50 Ohm for optimal signal reception. NOTE: Pay attention to the external wiring and connections going to the measurement system via the adaptor/splitter to each part of the transmitter coil individually (in our case it was a quadrature coil).
- After tuning and matching the coils, plug the coils into the scanner.
- For RF pulse calibration (shape and length) and center frequency adjustment perform both pulse calibration and center of frequency manually. Perform the 90° pulse calibration, coarse shimming (see below), center frequency and receiver gain adjustments manually.
- Perform exact position using a gradient echo (FLASH or GRE) localizer scan (3 plane scout acquisition: axial, coronal and sagittal, also called tripilot.
- Perform the magnet shimming to optimize the magnetic field homogeneity in the center of the magnet. Perform this step manually (see 5.2.3) using a one or single pulse FID sequence and adjust the first and second orders shims or any automatic shimming sequence included in the system NOTE: A well shimmed magnetic field is easily recognized and measured by the T2* (as bigger the better) or narrow FWHM of the spectra.
- MRI Data Acquisition of the Plaque5
- Inject 100 µl (1 mg [Fe] ml-1) of neridronate nanoparticles intravenously into tail vein and acquire images 1 hr post injection. Load the parameters for MRI acquisition of atherosclerotic plaque looking at the abdominal aorta (renal bifurcation).
- Put multi-slice, 10 to 20 slices in interleave mode to minimize artifacts.
- Acquire high-resolution fast spin echo T1-weighted MRI in coronal or axial view with the following parameters: FOV 60 x 30 mm (coronal), 30 x 30 mm (axial), slice thickness 0.8 mm (with the small gradient coil configuration this can be reduced to 0.6 mm), 400 msec TR, 8 msec TE, 256 x 256 acquisition and reconstruction matrix data, 6 (large gradient) to 8 (small gradient) signal averages for 5-8 min average acquisition times. NOTE: TE is especially critical and the presence of blood signal and flow and chemical shift artifacts that can limit the applications. In these cases flow insensitive fast spin echo T2-IR can help to reduce artifacts and the acquisition of complementary data in the exact same location can help to characterize the plaque. Additionally, a presaturation pulse can be used to reduce the fat tissue surrounding the arterial wall for better delineation of the outer boundary of the wall and chemical shift artifact reduction.
- Transfer the images using a standard format such as Dicom and view in appropriate software (e.g. Osirix Imaging Software or AMIDE: Medical Imaging Data Examiner)5. Quantify the contrast effect manually delimiting the vessel area, wall thickness, lumen area and plaque burden5.
Representative Results
In this protocol, the synthesis of three different IONP is described. Starting from hydrophobic OA-IONP, aqueous stable nanoparticles are obtained with the help of microwave-driven synthesis. All nanoparticles presented ultra-small hydrodynamic size (Dh <50 nm) in a very narrow size distribution (Figure 1c). The use of microwave technology renders ultra-small nanoparticles in terms of core sizes. Since microwave produce a fast heating, the rate of the nucleation increase in comparison with others methodologies giving smaller sizes in the core of the nanoparticles. However, the particles still present excellent crystallinity as is shown in the TEM images where the lattice fringes on the Fe3O4 cores can be clearly seen (Figure 1a, b). Other important aspect of the method is the reproducibility. After four repetitions of the synthesis of Azelaic acid-IONP, the same results in the hydrodynamic size and distribution were obtained (Figure 1d).
After functionalization, the Ca2+ binding properties due to bisphosphonates present in neridronate nanoparticles were checked incubating these nanoparticles with different amounts of Ca2+. It was shown that T2 relaxation time increments linearly with the amount of Ca2+ and the time of incubation due to the formation of clusters of nanoparticle whereas nanoparticles without Ca2+ remained stable (Figure 1e), conforming our initial hypothesis.
In vivo MRI experiments were performed in 48 weeks old ApoE-/- mice. Carotids and abdominal aorta basal images were first taken. Lesion due to the formation of the atherosclerotic plaque can be clearly seen. Then, 100 µl (1 mg [Fe] ml-1) of neridronate nanoparticles were intravenously injected into tail vein and the images acquired 1 hr post injection. As it is shown (Figure 2), 1 hr post injection the signal form the plaque is hypointense in comparison to the basal images. Selection of two ROIs (region of interest) allows the quantification of the intensity signal in the lesion area for the comparison between basal and 1 hr post injection images. The plaque to muscle ratio is significantly different between them (p <0.05, Figure 2b).
In addition, signal in the liver was monitored in mice after injection of 100 µl of neridronate nanoparticles to assess if the reduction of the intensity was due to circulation time in the blood and not by selective accumulation. As the graph shows (Figure 2c), nanoparticles were completely cleared from circulation after 20 min confirming the selective accumulation of neridronate nanoparticles towards atherosclerotic plaque. Final ex vivo imaging and histology were performed. Mice were sacrificed and the aortas extracted. Imaging of aortas with and without nanoparticles showed differences in the signal in agreement with in vivo experiments (Figure 2d).
Scheme 1: Synthetic steps followed in the protocol and basic characterization at each point by DLS. Please click here to view a larger version of this figure.
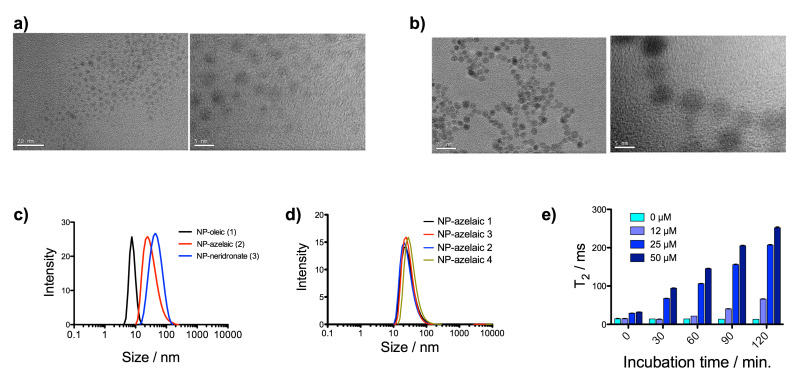 Figure 1: Characterization of nanoparticles. (a) TEM images, at two magnifications, for OA-IONP; (b) TEM images, at two magnifications, for Neridronate-IONP; (c) hydrodynamic size for nanoparticles OA-IONP, Azelaic acid-IONP and Neridronate-IONP; (d) hydrodynamic size for Azelaic acid-IONP in four different synthesis and (e) evolution of T2 relaxation time in a solution of Neridronate-IONP as a function of time and calcium concentration (ref TEM protocol: NIST - NCL Joint Assay Protocol, PCC-X, Measuring the Size of Nanoparticles Using Transmission Electron Microscopy). Please click here to view a larger version of this figure.
Figure 1: Characterization of nanoparticles. (a) TEM images, at two magnifications, for OA-IONP; (b) TEM images, at two magnifications, for Neridronate-IONP; (c) hydrodynamic size for nanoparticles OA-IONP, Azelaic acid-IONP and Neridronate-IONP; (d) hydrodynamic size for Azelaic acid-IONP in four different synthesis and (e) evolution of T2 relaxation time in a solution of Neridronate-IONP as a function of time and calcium concentration (ref TEM protocol: NIST - NCL Joint Assay Protocol, PCC-X, Measuring the Size of Nanoparticles Using Transmission Electron Microscopy). Please click here to view a larger version of this figure.
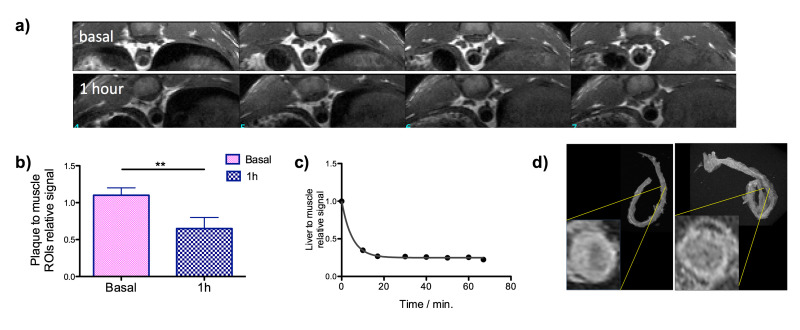 Figure 2: MRI data of the plaque. (a) In vivo MRI of ApoE-/- mouse before (top) and one hour after the i.v. injection of Neridronate-IONP (bottom); (b) plaque to muscle relative signal intensity before (basal) and one hour after the i.v. injection of Neridronate-IONP; (c) liver to muscle relative signal intensity at different time points after the injection of Neridronate-IONP and (d) ex vivo images of the aorta for two mice, with and without the injection of nanoparticles5. Please click here to view a larger version of this figure.
Figure 2: MRI data of the plaque. (a) In vivo MRI of ApoE-/- mouse before (top) and one hour after the i.v. injection of Neridronate-IONP (bottom); (b) plaque to muscle relative signal intensity before (basal) and one hour after the i.v. injection of Neridronate-IONP; (c) liver to muscle relative signal intensity at different time points after the injection of Neridronate-IONP and (d) ex vivo images of the aorta for two mice, with and without the injection of nanoparticles5. Please click here to view a larger version of this figure.
Discussion
Iron oxide nanoparticles (IONP) are one of the most important nanomaterials and it has been used for different applications from long time ago. The use of these materials as contrast agent for magnetic resonance imaging (MRI) is a well-established field. However, the routes of synthesis often take several time and the setting is complicated. Due to dramatically reduce reaction times and enhances reproducibility the use of microwave-driven synthesis seems to be a good alternative for the production of high quality nanoparticles. In the protocol described above, microwave technology has been used for the synthesis of two different nanoparticles. Microwave allow for a fine-tuning of the main parameters that can affect the final characteristics of the particles. It is important to note that the physical properties of the nanoparticles will change if any of the described conditions are changed. Since some chemicals are used in the synthetic procedure, the purification steps are critical to obtain high quality nanoparticles.
In OA-IONP excess of surfactants are used in order to get enough stability in the nanoparticles. After synthesis, three purification steps are mandatory to remove it. For the synthesis of Azelaic acid-IONP, two different microwave stages are required. In the second stage, the final size of the particles can be tuned from ultra-small IONP (Dh <50 nm) using pH = 2.9 to larger hydrodynamic size (Dh>50 nm) using physiological pH. In the purification of the Azelaic acid-IONP, the amount of NaOH used is essential. Enough amount of NaOH has to be added to stabilize the nanoparticles, however too many NaOH can desorb the surfactant from the nanoparticles rendering unstable material.
Typically, IONP possess short circulation time in blood which is one of the main disadvantages. For its use as contrast agent, nanoparticles need to circulate enough time in blood to reach the desired area. To increase the circulation time in blood different approaches are classically carried out. These strategies are mainly based on the attachment of a pegylated moiety that extent the circulation time of the nanoparticle. However, in the case of Neridronate-IONP, the accumulation is produced very fast. The use of an aminobisphosphonate as biomolecule on the nanoparticles to target atheroma plaque is a new concept based on the calcium capabilities of these kinds of compounds. Its accumulation in the lesion area in less than an hr demonstrates the high affinity of Neridronate-IONP towards calcium contained in the atheroma plaque.
For visualization of atheroma plaque, many advanced imaging techniques are usually employed. Among them, positron emission tomography (PET) and MRI are the most standardized techniques. PET provides the best results in terms of functional information due to the high sensitivity and MRI the best results in anatomical information due to the high resolution. Although PET could be the ideal option to follow a synthetic probe, the resolution of this technique in small animals (~1 mm) restricts its use to visualize smaller calcifications in atherosclerotic lesions. MRI is an ideal alternative providing better resolution (~0.1 µm). The lower sensitivity of this technique not avoids the visualization of the contrast agent in the interest region and the better resolution allows identifying small calcifications. In addition, the results show that the combination of the unique fast accumulation of Neridronate-IONP with the high resolution of MRI is an ideal scenario for the detection of atheroma plaque in small animals.
Disclosures
No disclosures
Acknowledgments
This study is supported by a grant from Comunidad de Madrid (S2010/BMD-2326, Inmunothercan-CM), by Fundacio La Marato de TV3 (70/C/2012) and by and by Spanish Economy Ministry (MAT2013-47303 P).
References
- Patel DN, Bailey SR. Nanotechnology in cardiovascular medicine. Catheter. Cardiovasc. Interv. Off. J. Soc. Card. Angiogr. Interv. 2007;69:643–654. doi: 10.1002/ccd.21060. [DOI] [PubMed] [Google Scholar]
- Zhao X, Zhao H, Chen Z, Lan M. Ultrasmall superparamagnetic iron oxide nanoparticles for magnetic resonance imaging contrast agent. J. Nanosci. Nanotechnol. 2014;14:210–220. doi: 10.1166/jnn.2014.9192. [DOI] [PubMed] [Google Scholar]
- Lee DE, et al. Multifunctional nanoparticles for multimodal imaging and theragnosis. Chem. Soc. Rev. 2012;41:2656. doi: 10.1039/c2cs15261d. [DOI] [PubMed] [Google Scholar]
- Osborne EA, et al. Rapid microwave-assisted synthesis of dextran-coated iron oxide nanoparticles for magnetic resonance imaging. Nanotechnology. 2012;23 doi: 10.1088/0957-4484/23/21/215602. [DOI] [PubMed] [Google Scholar]
- Pellico J, et al. Microwave-driven synthesis of bisphosphonate nanoparticles allows in vivo visualisation of atherosclerotic plaque. RSC Adv. 2015;5:1661–1665. [Google Scholar]
- Lin MM, Kim DK, El Haj AJ, Dobson J. Development of superparamagnetic iron oxide nanoparticles (SPIONS) for translation to clinical applications. IEEE Trans. Nanobioscience. 2008;7:298–305. doi: 10.1109/TNB.2008.2011864. [DOI] [PubMed] [Google Scholar]
- Gupta AK, Gupta M. Synthesis and surface engineering of iron oxide nanoparticles for biomedical applications. Biomaterials. 2005;26:3995–4021. doi: 10.1016/j.biomaterials.2004.10.012. [DOI] [PubMed] [Google Scholar]
- Liu F, Laurent S, Fattahi H, Vander Elst L, Muller RN. Superparamagnetic nanosystems based on iron oxide nanoparticles for biomedical imaging. Nanomed. 2011;6:519–528. doi: 10.2217/nnm.11.16. [DOI] [PubMed] [Google Scholar]
- Carenza E, et al. Rapid synthesis of water-dispersible superparamagnetic iron oxide nanoparticles by a microwave-assisted route for safe labeling of endothelial progenitor cells. Acta Biomater. 2014;10:3775–3785. doi: 10.1016/j.actbio.2014.04.010. [DOI] [PubMed] [Google Scholar]
- Osborne EA, et al. Rapid microwave-assisted synthesis of dextran-coated iron oxide nanoparticles for magnetic resonance imaging. Nanotechnology. 2012;23:215602. doi: 10.1088/0957-4484/23/21/215602. [DOI] [PubMed] [Google Scholar]
- Gatti D, Rossini M, Viapiana O, Idolazzi L, Adami S. Clinical development of neridronate: potential for new applications. Ther. Clin. Risk Manag. 2013;9:139–147. doi: 10.2147/TCRM.S35788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drake MT, Clarke BL, Khosla S. Bisphosphonates: mechanism of action and role in clinical practice. Mayo Clin Proc. 2008;83:1032–1045. doi: 10.4065/83.9.1032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Devogelaer JP. Treatment of bone diseases with bisphosphonates, excluding osteoporosis. Curr. Opin. Rheumatol. 2000;12:331–335. doi: 10.1097/00002281-200007000-00017. [DOI] [PubMed] [Google Scholar]
- Pascu O, et al. Surface Reactivity of Iron Oxide Nanoparticles by Microwave-Assisted Synthesis; Comparison with the Thermal Decomposition Route. J. Phys. Chem. C. 2012;116:15108–15116. [Google Scholar]
- Sun S, Zeng H. Size-Controlled Synthesis of Magnetite Nanoparticles. J. Am. Chem. Soc. 2002;124:8204–8205. doi: 10.1021/ja026501x. [DOI] [PubMed] [Google Scholar]
- Hyeon T, Lee SS, Park J, Chung Y, Na HB. Synthesis of highly crystalline and monodisperse maghemite nanocrystallites without a size-selection process. J. Am. Chem. Soc. 2001;123:12798–12801. doi: 10.1021/ja016812s. [DOI] [PubMed] [Google Scholar]
- Herranz F, Morales MP, Roca AG, Vilar R, Ruiz-Cabello J. A new method for the aqueous functionalization of superparamagnetic Fe 2 O 3 nanoparticles. Contrast Media Mol. Imaging. 2008;3:215–222. doi: 10.1002/cmmi.254. [DOI] [PubMed] [Google Scholar]
- Herranz F, Morales MP, Roca AG, Desco M, Ruiz-Cabello J. A new method for the rapid synthesis of water stable superparamagnetic nanoparticles. Chem. Weinh. Bergstr. Ger. 2008;14:9126–9130. doi: 10.1002/chem.200800755. [DOI] [PubMed] [Google Scholar]
- Herranz F, et al. Superparamagnetic iron oxide nanoparticles conjugated to a grass pollen allergen and an optical probe. Contrast Media Mol. Imaging. 2012;7:435–439. doi: 10.1002/cmmi.1466. [DOI] [PubMed] [Google Scholar]