Abstract
The present study aimed to identify the potential target genes and underlying molecular mechanisms involved in head and neck squamous cell carcinoma (HNSCC) by bioinformatics analysis. Microarray data of a Gene Expression Omnibus series GSE6631 was downloaded from the Gene Expression Omnibus database, which was generated from paired samples of HNSCC and normal tissue from 22 patients, and was used to identify differentially expressed genes (DEGs). Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes enrichment analyses were performed to investigate the functions of the identified DEGs. Furthermore, the protein-protein interaction (PPI) network of these DEGs was constructed using Cytoscape software. Between HNSCC and normal samples there was a difference in 419 DEGs, including 196 upregulated and 223 downregulated genes. The upregulated DEGs were mainly enriched in GO terms of cell adhesion, extracellular matrix (ECM) organization and collagen metabolic process, while the downregulated DEGs were mainly associated with epidermis development and epidermal cell differentiation. The DEGs were enriched in pathways such as ECM-receptor interaction, focal adhesion and drug metabolism. Fibronectin 1 (FN1), epidermal growth factor receptor (EGFR), collagen type I alpha 1 (COL1A1) and matrix metallopeptidase-9 (MMP-9) were hub nodes in the PPI network. These results suggested that cell adhesion and drug metabolism may be associated with HNSCC development, and genes such as FN1, EGFR, COL4A1 and MMP-9 may be potential therapeutic target genes in HNSCC.
Keywords: head and neck squamous cell carcinoma, differentially expressed genes, molecular mechanism, bioinformatics analysis
Introduction
Head and neck squamous cell carcinoma (HNSCC) is the sixth most common type of cancer in the world (1). It is an epithelial cancer arising in the upper aerodigestive tract, including the pharynx, larynx and oral cavity (2). Furthermore, the head and neck region contains several distinct structures, such as the lips, nasopharynx, oropharynx and hypopharynx, which result in the large heterogeneity of HNSCC (2,3). In total, >600,000 novel cases of HNSCC are diagnosed annually (1). Currently, chemotherapy or radiotherapy with locoregional treatment is used for HNSCC patients (4,5). However, the survival rate of this disease is only 40–50% within 5 years after diagnosis and treatment (6).
Numerous studies have explored the pathological mechanism underlying the development of HNSCC (7,8). Several genes have been identified to participate in the progression of HNSCC. For example, Zhang et al (9) reported that fos-related activator-1 could be used as a potential therapeutic target gene in oral squamous cell carcinoma, while transgelin 2 has an oncogenic function and may be regulated by the tumor suppressor microRNA-1 in HNSCC (7). Aberrant promoter methylation of the Nei endonuclease VIII-like 1 gene has a critical role in the progression and development of HNSCC (8). Certain signaling pathways have also been demonstrated to be important in HNSCC. For example, Pedrero et al (10) reported that dysregulation of the phosphatidylinositol-4,5-bisphosphate 3-kinase/AKT/phosphatase and tensin homolog signaling pathway may contribute to early HNSCC tumorigenesis. In addition, cyclooxygenase-2 (COX-2) signaling pathway is closely associated with tumor angiogenesis in HNSCC, and COX-2 overexpression predicts a shorter survival in patients with head and neck cancer (11). The coactivation of the mitogen-activated protein kinase and IκB kinase signaling pathways may suppress the mechanism of signal transduction by regulating the secretion of interleukin-8 and vascular endothelial growth factor in human HNSCC (12). Although numerous factors have been identified to contribute to HNSCC, the pathogenic mechanisms of HNSCC remain to be clearly demonstrated in order to identify potential target genes for the treatment of HNSCC.
In the present study, the differentially expressed genes (DEGs) between HNSCC and normal samples were analyzed to gain a better insight of HNSCC. Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analyses of DEGs were performed, and the protein-protein interaction (PPI) network of these DEGs was constructed. The purpose of the present study was to explore the underlying mechanisms of HNSCC and to identify novel potential target genes for HNSCC therapy.
Materials and methods
Affymetrix microarray data
Gene Expression Omnibus (GEO; www.ncbi.nlm.nih.gov/geo/) is a database repository of high throughput gene expression data, which segregates data into three principle components: Platform (GPL), series (GSE) and sample (GSM). The array data of GSE6631, based on the GPL8300 Affymetrix Human Genome U95 Version 2 Array platform (Affymetrix, Inc., Santa Clara, CA, USA) was downloaded from the GEO database, which was deposited by Kuriakose et al (13). The dataset was generated from paired (from the same patient) samples of tumor and normal tissues from 22 patients with histologically confirmed HNSCC by Kuriakose et al (13).
Data preprocessing and DEGs analysis
The original probe-level data in CEL files (raw probe level data) were converted into gene expression values. Data were normalized using the Bioconductor R package affy version 1.32.0 (Affymetrix, Inc., Santa Clara, CA, USA) (14). Nonspecific probes were filtered. If multiple probes corresponded to the same gene, the average expression value was calculated to represent the expression levels of that gene. The samr package (version 2.0; cran.r-project.org/web/packages/samr/index.html) in R (www.r-project.org/) (15) was applied to identify DEGs between HNSCC and normal samples. ∆=1.3 and fold-change >1.5 were used as the cutoff criteria, based on the experience of the present authors.
Functional enrichment analysis of DEGs
The GO database (geneontology.org/page/go-database) (16) is a collection of numerous gene annotation terms. The knowledge contained in the KEGG database (www.genome.jp/kegg/) (17) was applied to identify functional and metabolic pathways. The Database for Annotation, Visualization and Integrated Discovery (DAVID) version 6.7 (National Cancer Institute at Frederick, Frederick, MD, USA) (18) was used as a gene functional enrichment analysis tool to understand the biological meaning of the results of bioinformatics analysis. GO and KEGG enrichment analyses for the upregulated and downregulated identified DEGs were performed with DAVID. P<0.05 and false discovery rate <0.01 were selected as the cutoff criteria.
Construction of PPI network and disease enrichment analysis
The Search Tool for the Retrieval of Interacting Genes/Proteins (version 9.05; string-db.org) (19) is an online database that contains comprehensive information of proteins. This online tool was applied to analyze the interactions of protein pairs. PPI network of DEGs was constructed using Cytoscape software (version 3.0.1; Cytoscape Consortium San Diego, CA, USA) (20). The degree of connectivity was analyzed and used to obtain the hub proteins in the PPI network.
Results
Identification of DEGs
As represented in Fig. 1, the raw expression data were preprocessed and normalized. A total of 419 DEGs were identified between HNSCC and normal samples, including 196 upregulated genes and 223 downregulated genes.
Figure 1.
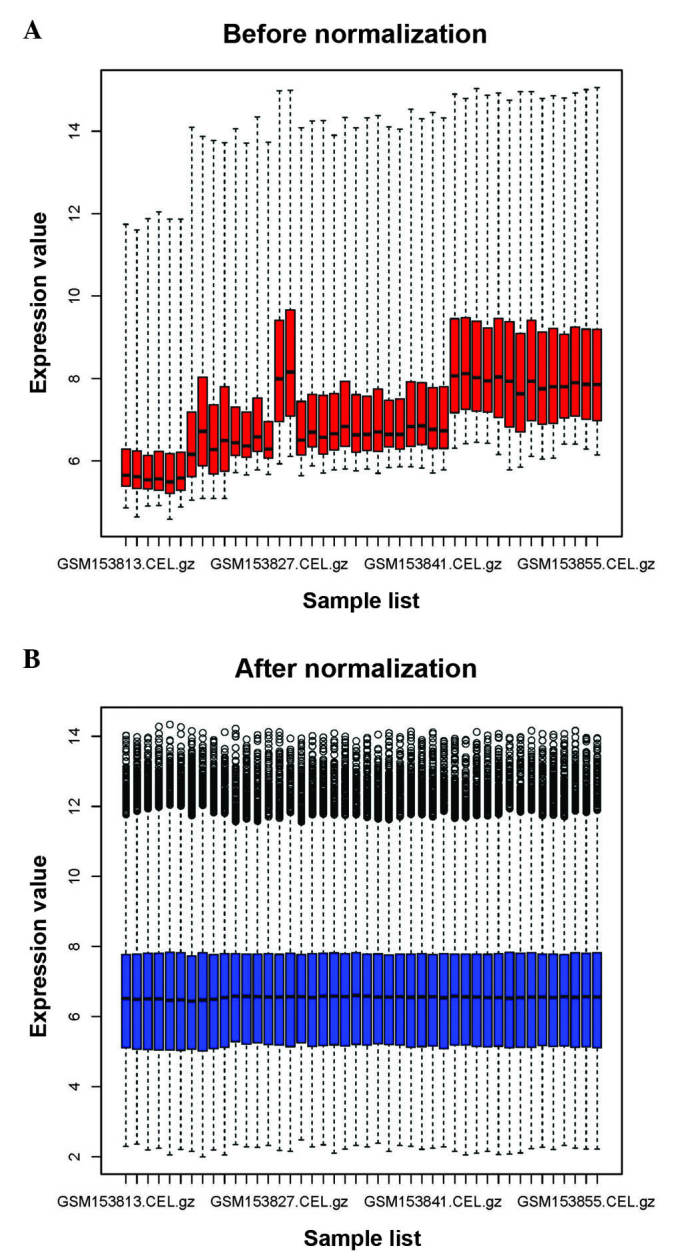
Box plots of data (A) before (red) and (B) after (blue) normalization. The × axis represents the samples from the microarray data, while the y axis represents the gene expression values. The box plot refers to the interquartile range (25–75%), and the median is shown as a black horizontal line.
Functional enrichment analysis of DEGs
A total of 39 GO terms of upregulated and downregulated DEGs were obtained. The top 5 GO terms of upregulated and downregulated genes are indicated in Table I. The upregulated DEGs were significantly associated with cell adhesion, extracellular matrix (ECM) organization, collagen metabolic process and proteinaceous ECM, while the downregulated genes were mainly involved in epidermis development, ectoderm development and epidermal cell differentiation.
Table I.
GO terms most frequently enriched by upregulated and downregulated DEGs in head and neck squamous cell carcinoma.
| Category | Term | Counta | P-value | FDR |
|---|---|---|---|---|
| Upregulated DEGs | ||||
| GOTERM_BP | GO:0007155~cell adhesion | 42 | 4.92E-17 | 8.08E-14 |
| GOTERM_BP | GO:0022610~biological adhesion | 42 | 5.18E-17 | 8.50E-14 |
| GOTERM_BP | GO:0030198~extracellular matrix organization | 17 | 2.12E-13 | 3.48E-10 |
| GOTERM_BP | GO:0043062~extracellular structure organization | 19 | 2.20E-12 | 3.62E-09 |
| GOTERM_BP | GO:0032963~collagen metabolic process | 10 | 3.75E-11 | 6.15E-08 |
| GOTERM_CC | GO:0005578~proteinaceous extracellular matrix | 40 | 1.88E-26 | 2.48E-23 |
| GOTERM_CC | GO:0031012~extracellular matrix | 40 | 3.23E-25 | 4.27E-22 |
| GOTERM_CC | GO:0044420~extracellular matrix part | 25 | 5.83E-22 | 7.70E-19 |
| GOTERM_CC | GO:0044421~extracellular region part | 56 | 4.52E-21 | 5.97E-18 |
| GOTERM_CC | GO:0005581~collagen | 14 | 3.91E-16 | 5.88E-13 |
| GOTERM_MF | GO:0005201~extracellular matrix structural constituent | 15 | 3.11E-12 | 4.32E-09 |
| GOTERM_MF | GO:0050840~extracellular matrix binding | 8 | 3.24E-08 | 4.50E-05 |
| GOTERM_MF | GO:0005198~structural molecule activity | 27 | 9.82E-08 | 1.36E-04 |
| GOTERM_MF | GO:0005509~calcium ion binding | 32 | 4.02E-07 | 5.58E-04 |
| GOTERM_MF | GO:0005518~collagen binding | 7 | 5.28E-06 | 7.33E-03 |
| Downregulated DEGs | ||||
| GOTERM_BP | GO:0008544~epidermis development | 20 | 1.27E-11 | 2.11E-08 |
| GOTERM_BP | GO:0007398~ectoderm development | 20 | 5.04E-11 | 8.39E-08 |
| GOTERM_BP | GO:0009913~epidermal cell differentiation | 13 | 3.26E-10 | 5.42E-07 |
| GOTERM_BP | GO:0030855~epithelial cell differentiation | 15 | 8.05E-09 | 1.34E-05 |
| GOTERM_BP | GO:0030216~keratinocyte differentiation | 11 | 2.64E-08 | 4.40E-05 |
| GOTERM_CC | GO:0001533~cornified envelope | 9 | 3.27E-10 | 4.24E-07 |
| GOTERM_MF | GO:0005198~structural molecule activity | 26 | 5.36E-06 | 7.55E-03 |
Enriched gene number in the GO category. GO, Gene Ontology; DEGs, differentially expressed genes; BP, biological process; CC, cellular component; MF, molecular function; FDR, false discovery rate.
The pathways of these upregulated and downregulated genes are indicated in Table II. The upregulated genes were mainly involved in ECM-receptor interaction, focal adhesion and small cell lung cancer. Genes such as fibronectin 1 (FN1), epidermal growth factor receptor (EGFR) and collagen type I alpha 1 (COL1A1) were identified in the focal adhesion pathway. By contrast, the downregulated DEGs were enriched in drug metabolism. Cytochrome P450 3A5 (CYP3A5) was identified in the drug metabolism pathway.
Table II.
KEGG pathway enrichment analysis of upregulated and downregulated differentially expressed genes.
| KEGG pathway term | Counta | Genes | P-value | FDR |
|---|---|---|---|---|
| Upregulated genes | ||||
| ECM-receptor interaction | 23 | COL1A1, COL4A1, TNC | 1.24E-20 | 1.35E-17 |
| Focal adhesion | 26 | FN1, EGFR, COL1A1 | 4.22E-15 | 4.59E-12 |
| Small cell lung cancer | 11 | FN1, CKS1B, LAMB3, COL4A2 | 2.11E-06 | 0.00229 |
| Downregulated genes | ||||
| Drug metabolism | 9 | CYP3A5, CYP2C18, FMO2, MAOB | 6.49E-06 | 0.0071 |
Enriched gene number in the KEGG pathway term. KEGG, Kyoto Encyclopedia of Genes and Genomes; FDR, false discovery rate; ECM, extracellular matrix; COLA, collagen type alpha; TNC, tenascin C; FN1, fibronectin 1; EGFR, epidermal growth factor receptor; CKS1B, cyclin-dependent kinases regulatory subunit 1; LAMB3, laminin, beta 3; CYP, cytochrome P450; FMO, flavin containing monooxygenase 2; MAOB, monoamine oxidase B.
PPI network construction and disease enrichment analysis
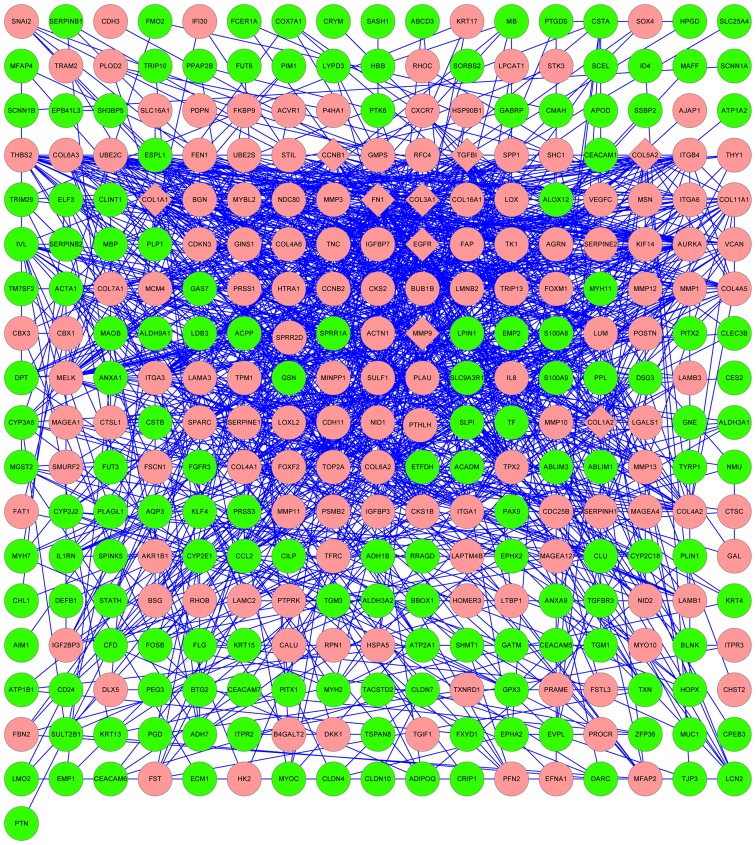
The results of the PPI network analysis are represented in Fig. 2. The upregulated genes FN1, EGFR, COL1A1, matrix metallopeptidase-9 (MMP-9), COL5A2, COL1A2, COL3A1, transforming growth factor, beta-induced and cyclin B1 were selected as hub nodes.
Figure 2.
Protein-protein interaction network for differentially expressed genes. Red nodes correspond to upregulated genes in HNSCC, while green nodes correspond to downregulated genes in HNSCC. Diamonds represent hub nodes (nodes with a degree of connectivity ≥30). HNSCC, head and neck squamous cell carcinoma.
Discussion
In the present study, gene expression profile data were downloaded from the GEO database to identify DEGs in HNSCC using bioinformatics analysis. A total of 419 DEGs between HNSCC and normal samples, including 196 upregulated and 223 downregulated genes, were selected. The results of functional enrichment analysis revealed that the upregulated genes, including FN1, EGFR and COL1A1, were associated with GO term of cell adhesion, while the downregulated DEGs, including CYP3A5, were enriched in drug metabolism pathways. According to the results of the PPI network analysis, FN1, EGFR, COL1A1 and MMP-9 were identified as hub nodes. Therefore, these DEGs and their interacting patners may be involved in HNSCC development.
Cell adhesion is the process of binding of a cell to a surface or substrate, such as the ECM or another cell (21). In the present study, the majority of the upregulated DEGs were enriched in pathways of ECM-receptor interaction and focal adhesion. Previous studies have indicated that ECM-receptor interaction and focal adhesion were associated with cell adhesion (22). Recent evidence suggests that cell adhesion is mediated by several genes, including FN1, EGFR and COL4A1 (23–25). FN1 is an ECM glycoprotein (26) involved in cell adhesion (27), which corresponds to the pathway identified in the present study. It was previously reported that FN1 acts as a tumor suppressor gene, playing a critical role in migration and invasion of laryngeal carcinoma (23), which is the most common type of HNSCC (28). EGFR was also indicated to be associated with HNSCC (29). EGFR is the cell-surface receptor of the EGF family (30). In the present study, EGFR was enriched in GO terms of cell adhesion and pathway of focal adhesion, which was consistent with previous studies that reported that EGFR contributed to transduce extracellular signals to intracellular responses, thus influencing adhesion and proliferation in tumor cells (24,31). Rubin Grandis et al (32) reported that EGFR was overexpressed in HNSCC. High expression levels of EGFR have been associated with reduced survival and increased risk of recurrence in HNSCC (33). COL4A1 is a member of the collagen family, and is also associated with cell adhesion (25). The adhesion of cells to collagen is mediated by fibronectin (25). Tanaka et al (34) indicated that the differential expression of type IV collagen chains was associated with the invasive potential of cell carcinoma. The results of the present study indicated that FN1, EGFR and COL4A1 were upregulated genes in HNSCC and hub nodes in the PPI network, which suggests that FN1, EGFR and COL4A1 may regulate cell adhesion in HNSCC. Thus, cell adhesion may participate in HNSCC through multiple genes, including FN1, EGFR and COL4A1, which may be potential therapeutic target genes in HNSCC.
In the present study, the downregulated DEGs such as CYP3A5, were significantly enriched in the pathway of drug metabolism (P=6.49E-06). CYP3A5 encodes a member of the cytochrome P450 superfamily of enzymes (34). It has been reported that cytochrome P450 proteins catalyze multiple reactions, including drug metabolism (35). Olivieri et al (36) reported that cytochrome P450 gene polymorphisms were important in the tumorigenesis and progression of HNSCC (36). These results suggested that CYP3A5 may regulate HNSCC development through the drug metabolism pathway. Therefore, this pathway may be associated with HNSCC progression.
In addition to FN1, EGFR and COL4A1, MMP-9 was also identified as a hub node in the present PPI network analysis. MMP-9 is an enzyme that belongs to the MMP family (35). It has been reported that MMPs participate in cancer invasion and metastasis (37). In the present study, MMP-9 was an upregulated gene, which was consistent with previous studies (38,39). For example, Riedel et al (38) reported that the expression levels of MMP-9 were significantly higher in HNSCC patients than in healthy individuals. MMP-9 regulates cell proliferation through modulating the nuclear factor-κB signaling pathway in HNSCC (40). Furthermore, MMP-9 was associated with cancer in the present study. Thus, MMP-9 may be a potential target gene for the treatment of HNSCC.
In conclusion, a total of 419 DEGs were identified between HNSCC and normal samples, and the present study indicates that cell adhesion and drug metabolism may be closely associated with HNSCC development. Genes such as FN1, EGFR, COL4A1 and MMP-9 may be potential therapeutic target genes in HNSCC. However, further studies are required to confirm the present results.
References
- 1.Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer. 2010;127:2893–2917. doi: 10.1002/ijc.25516. [DOI] [PubMed] [Google Scholar]
- 2.Rothenberg SM, Ellisen LW. The molecular pathogenesis of head and neck squamous cell carcinoma. J Clin Invest. 2012;122:1951–1957. doi: 10.1172/JCI59889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Hasina R, Whipple ME, Martin LE, Kuo WP, Ohno-Machado L, Lingen MW. Angiogenic heterogeneity in head and neck squamous cell carcinoma: Biological and therapeutic implications. Lab Invest. 2008;88:342–353. doi: 10.1038/labinvest.2008.6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Belcher R, Hayes K, Fedewa S, Chen AY. Current treatment of head and neck squamous cell cancer. J Surg Oncol. 2014;110:551–574. doi: 10.1002/jso.23724. [DOI] [PubMed] [Google Scholar]
- 5.Colevas AD. Chemotherapy options for patients with metastatic or recurrent squamous cell carcinoma of the head and neck. J Clin Oncol. 2006;24:2644–2652. doi: 10.1200/JCO.2005.05.3348. [DOI] [PubMed] [Google Scholar]
- 6.Leemans CR, Braakhuis BJ, Brakenhoff RH. The molecular biology of head and neck cancer. Nat Rev Cancer. 2011;11:9–22. doi: 10.1038/nrc2982. [DOI] [PubMed] [Google Scholar]
- 7.Nohata N, Sone Y, Hanazawa T, Fuse M, Kikkawa N, Yoshino H, Chiyomaru T, Kawakami K, Enokida H, Nakagawa M, et al. miR-1 as a tumor suppressive microRNA targeting TAGLN2 in head and neck squamous cell carcinoma. Oncotarget. 2011;2:29–42. doi: 10.18632/oncotarget.213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chaisaingmongkol J, Popanda O, Warta R, Dyckhoff G, Herpel E, Geiselhart L, Claus R, Lasitschka F, Campos B, Oakes CC, et al. Epigenetic screen of human DNA repair genes identifies aberrant promoter methylation of NEIL1 in head and neck squamous cell carcinoma. Oncogene. 2012;31:5108–5116. doi: 10.1038/onc.2011.660. [DOI] [PubMed] [Google Scholar]
- 9.Zhang L, Pan HY, Zhong LP, Wei KJ, Yang X, Li J, Shen GF, Zhang Z. Fos-related activator-1 is overexpressed in oralsquamous cell carcinoma and associated with tumor lymph node metastasis. J Oral Pathol Med. 2010;39:470–476. doi: 10.1111/j.1600-0714.2009.00869.x. [DOI] [PubMed] [Google Scholar]
- 10.Pedrero JM, Carracedo DG, Pinto CM, Zapatero AH, Rodrigo JP, Nieto CS, Gonzalez MV. Frequent genetic and biochemical alterations of the PI3-K/AKT/PTEN pathway in head and neck squamous cell carcinoma. Int J Cancer. 2005;114:242–248. doi: 10.1002/ijc.20711. [DOI] [PubMed] [Google Scholar]
- 11.Gallo O, Masini E, Bianchi B, Bruschini L, Paglierani M, Franchi A. Prognostic significance of cyclooxygenase-2 pathway and angiogenesis in head and neck squamous cell carcinoma. Hum Pathol. 2002;33:708–714. doi: 10.1053/hupa.2002.125376. [DOI] [PubMed] [Google Scholar]
- 12.Bancroft CC, Chen Z, Dong G, Sunwoo JB, Yeh N, Park C, Van Waes C. Coexpression of proangiogenic factors IL-8 and VEGF by human head and neck squamous cell carcinoma involves coactivation by MEK-MAPK and IKK-NF-kappaB signal pathways. Clin Cancer Res. 2001;7:435–442. [PubMed] [Google Scholar]
- 13.Kuriakose MA, Chen WT, He ZM, Sikora AG, Zhang P, Zhang ZY, Qiu WL, Hsu DF, McMunn-Coffran C, Brown SM, et al. Selection and validation of differentially expressed genes in head and neck cancer. Cell Mol Life Sci. 2004;61:1372–1383. doi: 10.1007/s00018-004-4069-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gautier L, Cope L, Bolstad BM, Irizarry RA. affy - analysis of Affymetrix GeneChip data at the probe level. Bioinformatics. 2004;20:307–315. doi: 10.1093/bioinformatics/btg405. [DOI] [PubMed] [Google Scholar]
- 15.Tusher VG, Tibshirani R, Chu G. Significance analysis of microarrays applied to the ionizing radiation response. Proc Natl Acad Sci USA. 2001;98:5116–5121. doi: 10.1073/pnas.091062498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ashburner M, Ball CA, Blake JA, Botstein D, Butler H, Cherry JM, Davis AP, Dolinski K, Dwight SS, Eppig JT, et al. The Gene Ontology Consortium: Gene Ontology: Tool for the unification of biology. Nat Genet. 2000;25:25–29. doi: 10.1038/75556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Altermann E, Klaenhammer TR. PathwayVoyager: Pathway mapping using the Kyoto Encyclopedia of Genes and Genomes (KEGG) database. BMC Genomics. 2005;6:60. doi: 10.1186/1471-2164-6-60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Huang DW, Sherman BT, Tan Q, Kir J, Liu D, Bryant D, Guo Y, Stephens R, Baseler MW, Lane HC, Lempicki RA. DAVID Bioinformatics Resources: Expanded annotation database and novel algorithms to better extract biology from large gene lists. Nucleic Acids Res. 2007;35(Web Server issue):W169–W175. doi: 10.1093/nar/gkm415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Szklarczyk D, Franceschini A, Kuhn M, Simonovic M, Roth A, Minguez P, Doerks T, Stark M, Muller J, Bork P, et al. The STRING database in 2011: Functional interaction networks of proteins, globally integrated and scored. Nucleic Acids Res. 2011;39(Database issue):D561–D568. doi: 10.1093/nar/gkq973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Smoot ME, Ono K, Ruscheinski J, Wang PL, Ideker T. Cytoscape 2.8: New features for data integration and network visualization. Bioinformatics. 2011;27:431–432. doi: 10.1093/bioinformatics/btq675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gumbiner BM. Cell adhesion: The molecular basis of tissue architecture and morphogenesis. Cell. 1996;84:345–357. doi: 10.1016/S0092-8674(00)81279-9. [DOI] [PubMed] [Google Scholar]
- 22.Albelda SM, Buck CA. Integrins and other cell adhesion molecules. FASEB J. 1990;4:2868–2880. [PubMed] [Google Scholar]
- 23.Wang F, Song G, Liu M, Li X, Tang H. miRNA-1 targets fibronectin 1 and suppresses the migration and invasion of the HEp2 laryngeal squamous carcinoma cell line. FEBS Lett. 2011;585:3263–3269. doi: 10.1016/j.febslet.2011.08.052. [DOI] [PubMed] [Google Scholar]
- 24.Goerner M, Seiwert TY, Sudhoff H. Molecular targeted therapies in head and neck cancer - an update of recent developments. Head Neck Oncol. 2010;2:8. doi: 10.1186/1758-3284-2-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kleinman HK, Martin GR, Fishman PH. Ganglioside inhibition of fibronectin-mediated cell adhesion to collagen. Proc Natl Acad Sci USA. 1979;76:3367–3371. doi: 10.1073/pnas.76.7.3367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Pankov R, Yamada KM. Fibronectin at a glance. J Cell Sci. 2002;115:3861–3863. doi: 10.1242/jcs.00059. [DOI] [PubMed] [Google Scholar]
- 27.Soikkeli J, Podlasz P, Yin M, Nummela P, Jahkola T, Virolainen S, Krogerus L, Heikkilä P, von Smitten K, Saksela O, Hölttä E. Metastatic outgrowth encompasses COL-I, FN1, and POSTN up-regulation and assembly to fibrillar networks regulating cell adhesion, migration, and growth. Am J Pathol. 2010;177:387–403. doi: 10.2353/ajpath.2010.090748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Mao L, Hong WK, Papadimitrakopoulou VA. Focus on head and neck cancer. Cancer Cell. 2004;5:311–316. doi: 10.1016/S1535-6108(04)00090-X. [DOI] [PubMed] [Google Scholar]
- 29.Erjala K, Sundvall M, Junttila TT, Zhang N, Savisalo M, Mali P, Kulmala J, Pulkkinen J, Grenman R, Elenius K. Signaling via ErbB2 and ErbB3 associates with resistance and epidermal growth factor receptor (EGFR) amplification with sensitivity to EGFR inhibitor gefitinib in head and neck squamous cell carcinoma cells. Clin Cancer Res. 2006;12:4103–4111. doi: 10.1158/1078-0432.CCR-05-2404. [DOI] [PubMed] [Google Scholar]
- 30.Herbst RS. Review of epidermal growth factor receptor biology. Int J Radiat Oncol Biol Phys. 2004;59(Suppl 2):S21–S26. doi: 10.1016/j.ijrobp.2003.11.041. [DOI] [PubMed] [Google Scholar]
- 31.Rocha-Lima CM, Soares HP, Raez LE, Singal R. EGFR targeting of solid tumors. Cancer Control. 2007;14:295–304. doi: 10.1177/107327480701400313. [DOI] [PubMed] [Google Scholar]
- 32.Grandis Rubin J, Melhem MF, Gooding WE, Day R, Holst VA, Wagener MM, Drenning SD, Tweardy DJ. Levels of TGF-alpha and EGFR protein in head and neck squamous cell carcinoma and patient survival. J Natl Cancer Inst. 1998;90:824–832. doi: 10.1093/jnci/90.11.824. [DOI] [PubMed] [Google Scholar]
- 33.Bei R, Budillon A, Masuelli L, Cereda V, Vitolo D, Di Gennaro E, Ripavecchia V, Palumbo C, Ionna F, Losito S, et al. Frequent overexpression of multiple ErbB receptors by head and neck squamous cell carcinoma contrasts with rare antibody immunity in patients. J Pathol. 2004;204:317–325. doi: 10.1002/path.1642. [DOI] [PubMed] [Google Scholar]
- 34.Tanaka K, Iyama K, Kitaoka M, Ninomiya Y, Oohashi T, Sado Y, Ono T. Differential expression of·alpha 1(IV), alpha 2(IV), alpha 5(IV) and·alpha 6(IV) collagen chains in the basement membrane of basal cell carcinoma. Histochem J. 1997;29:563–570. doi: 10.1023/A:1026428010104. [DOI] [PubMed] [Google Scholar]
- 35.Bains RK, Kovacevic M, Plaster CA, Tarekegn A, Bekele E, Bradman NN, Thomas MG. Molecular diversity and population structure at the cytochrome P450 3A5 gene in Africa. BMC Genet. 2013;14:34. doi: 10.1186/1471-2156-14-34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Olivieri EH, da Silva SD, Mendonça FF, Urata YN, Vidal DO, Faria MA, Nishimoto IN, Rainho CA, Kowalski LP, Rogatto SR. CYP1A2*1C, CYP2E1*5B, and GSTM1 polymorphisms are predictors of risk and poor outcome in head and neck squamous cell carcinoma patients. Oral Oncol. 2009;45:e73–e79. doi: 10.1016/j.oraloncology.2009.03.004. [DOI] [PubMed] [Google Scholar]
- 37.Curran S, Murray GI. Matrix metalloproteinases: Molecular aspects of their roles in tumour invasion and metastasis. Eur J Cancer. 2000;36:1621–1630. doi: 10.1016/S0959-8049(00)00156-8. [DOI] [PubMed] [Google Scholar]
- 38.Riedel F, Götte K, Schwalb J, Hörmann K. Serum levels of matrix metalloproteinase-2 and −9 in patients with head and neck squamous cell carcinoma. Anticancer Res. 2000;20:3045–3049. [PubMed] [Google Scholar]
- 39.Sinpitaksakul SN, Pimkhaokham A, Sanchavanakit N, Pavasant P. TGF-β1 induced MMP-9 expression in HNSCC cell lines via Smad/MLCK pathway. Biochem Biophys Res Commun. 2008;371:713–718. doi: 10.1016/j.bbrc.2008.04.128. [DOI] [PubMed] [Google Scholar]
- 40.Aggarwal S, Takada Y, Singh S, Myers JN, Aggarwal BB. Inhibition of growth and survival of human head and neck squamous cell carcinoma cells by curcumin via modulation of nuclear factor-kappaB signaling. Int J Cancer. 2004;111:679–692. doi: 10.1002/ijc.20333. [DOI] [PubMed] [Google Scholar]