Abstract
Obstructive hydrocephalus has been described as a rare complication following foramen magnum decompression for Chiari malformation. However, there are few reports of obstructive hydrocephalus after spinal surgery for other pathologies of the craniovertebral junction (CVJ). The authors herein report a 52-year-old female with achondroplasia presenting with an 8-month history of myelopathy due to spinal cord compression at CVJ. She underwent resection of the C1 posterior arch and part of the edge of the occipital bone. A computed tomography (CT) scan obtained 1-week after the surgery revealed bilateral infratentorial fluid collection. The patient was first managed conservatively; however, on the 17th day, her consciousness level showed sudden deterioration. Emergency CT demonstrated marked hydrocephalus due to obstruction of the cerebral aqueduct. Acute obstructive hydrocephalus can occur late after decompression surgery at the CVJ, and thus should be included in the differential diagnosis of a deteriorating mental status.
Keywords: Achondroplasia, complication, craniovertebral junction, obstructive hydrocephalus, remote cerebellar hemorrhage
Introduction
Obstructive hydrocephalus occurs as a result of anatomic or functional obstruction to the cerebrospinal fluid (CSF) flow due to various pathologies including intracranial infection, tumors, and subarachnoid hemorrhage.[1] Recently, obstructive hydrocephalus has been described as a rare complication following foramen magnum decompression (FMD) for Chiari malformation;[2,3,4,5,6] however, there are few reports of obstructive hydrocephalus after spinal surgery for other pathologies of the craniovertebral junction (CVJ).[7] We herein report the case of a 52-year-old female who developed acute obstructive hydrocephalus after undergoing resection of the C1 posterior arch and part of the occipital bone.
Case Report
History and physical examination
A 52-year-old female with achondroplasia presented with an 8-month history of progressive weakness in the upper extremities with numbness and a gait disturbance. A neurological examination revealed weakness (grade 3/5) in the bilateral intrinsic muscles and hyperreflexia in the bilateral upper extremities with a positive Hoffmann sign; the knee and ankle reflexes were also increased bilaterally. She had decreased sensation in the bilateral upper extremities and a spastic gait with clumsiness of her hands, although she had no bowel or bladder difficulties. Lateral radiographs showed hypoplasia of C1. Computed tomography (CT) disclosed bony canal stenosis caused by the C1 posterior arch and edge of the occipital bone [Figure 1], and magnetic resonance (MR) imaging demonstrated atrophy of the spinal cord and a high-intensity intramedullary lesion at the C1 level [Figure 2].
Figure 1.
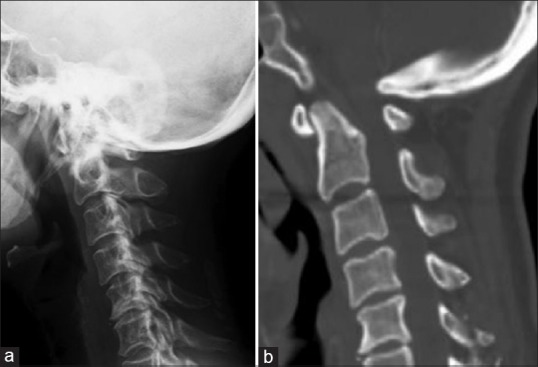
Plain lateral radiographs (a) and sagittal computed tomography (b) of the cervical spine revealing congenital bony canal stenosis at the CVJ caused by the C1 posterior arch and occipital bone
Figure 2.
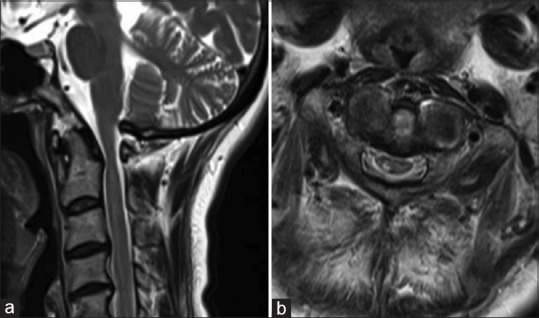
Sagittal magnetic resonance imaging (a) and axial imaging at the C1 level (b) demonstrating an atrophied spinal cord and high-intensity intramedullary lesion
Operation and postoperative course
The patient underwent posterior decompression of the CVJ. We resected the C1 posterior arch and the rim of the occipital bone (approximately, 7 mm from the edge of the foramen magnum). Intraoperative ultrasonography confirmed sufficient resection of the bony structure. At this stage of operation, the dura matter did not expand significantly. The thickened reactive fibrous tissue band was noted between C1 and the occipital bone [Figure 3a]. Because sparing this fibrous tissue band carefully from dural matter did not improve compression at CVJ, the band and the dura mater underneath were carefully dissected longitudinally to achieve decompression, and the dura mater was left open without a patch graft. The arachnoid membrane was preserved [Figure 3b]. The patient's motor weakness and gait disturbance improved immediately after surgery. Although the arachnoid membrane appeared intact during the surgery, the output of suction drainage indicated CSF leakage. One week after the surgery, she complained of a severe headache and nausea. Postoperative MR imaging revealed a moderate collection of CSF at the surgical site. Brain CT demonstrated bilateral infratentorial subdural fluid collection [Figure 4a]. As the neurological condition of the patient was stable, she was managed conservatively with intravenous hydration. On the 17th day, after the surgery, her consciousness level suddenly deteriorated, and she became drowsy and unresponsive to verbal stimuli. In the first place, overdose of hypnotics was suspected. An emergency brain CT scan revealed an increased volume of infratentorial fluid collection and marked hydrocephalus with an obstructed cerebral aqueduct [Figures 4b and 5]. Following rapid deterioration of the patient's neurological status, she underwent emergency external ventricular drainage and subsequent re-exploration of the surgical site. Additional wide craniotomy and Y-shaped durotomy were performed as in FMD. Subdural hemorrhage and hygroma were noted in the subdural space, which was suctioned cautiously. Duraplasty using the occipital periosteum as a patch graft was performed, and the patient's consciousness level improved to normal immediately after the revision surgery. The external ventricular drain was removed 5 days after the revision surgery, and follow-up imaging studies demonstrated the complete resolution of hydrocephalus. At the 12-month follow-up visit, she had regained her full strength and was able to walk without assistance.
Figure 3.
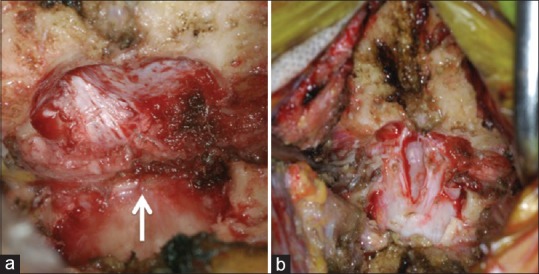
Intraoperative photographs after resection of the posterior arch and part of the occipital bone (a) the thickened reactive fibrous tissue band compressing the dural mater (arrow). Following the removal of the fibrous tissue band with opening of the dura mater, decompression of the spinal cord was achieved (b)
Figure 4.
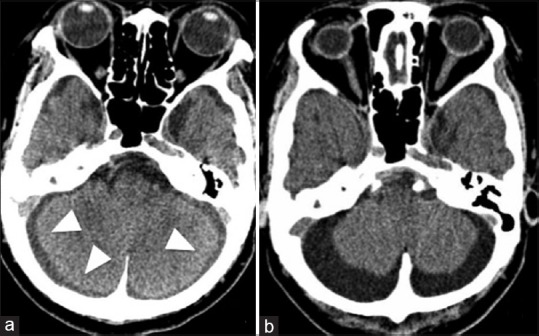
Plain brain computed tomography obtained 7 days after surgery (a) infratentorial fluid collection bilaterally (arrowheads). On the 17th day (b), the infratentorial fluid collection increased
Figure 5.
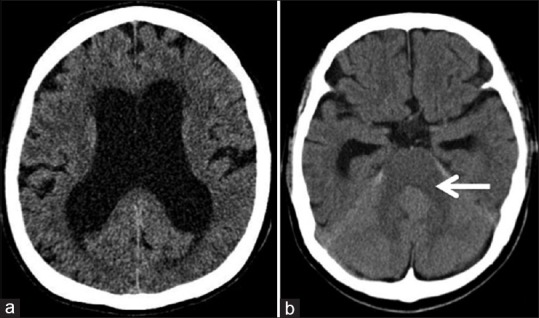
Plain brain computed tomography obtained 17 days after surgery revealing enlargement of the lateral ventricles (a) and an obstructed cerebral aqueduct (b, arrow), which indicated the presence of obstructive hydrocephalus
Discussion
In this paper, we reported a rare case of progressive obstructive hydrocephalus following decompression surgery at the CVJ. Acute obstructive hydrocephalus has recently been recognized to be a rare complication of FMD for Chiari malformation.[2,3,4,5,6] Table 1 summarizes the clinical course of patients who developed hydrocephalus associated with subdural hygroma following FMD in the literature. Time from index surgery to hydrocephalus varied ranging from 3 days to 1 month.
Table 1.
The clinical course of patients who developed hydrocephalus associated with subdural hygroma following foramen magnum decompression in the literature

The underlying mechanism of this life-threatening condition is unclear. Regarding the pathomechanism of postoperative subdural hygroma, a one-way flap valve mechanism through a small defect in the arachnoid membrane is assumed to play a pivotal role in the development of subdural fluid collection accumulating over the tentorial surface of the cerebellum. Subsequently, the infratentorial subdural fluid collection compresses the cerebral aqueduct, presumably causing obstructive hydrocephalus.[3,8] We conjecture that a similar pathomechanism was at work in our case as well. Although the arachnoid membrane in the surgical field appeared intact, the postoperative findings of the drainage fluid and MR imaging suggested the presence of an unnoticed arachnoid defect. To the best of our knowledge, only one case report of obstructive hydrocephalus after spinal surgery at CVJ for pathologies other than Chiari malformation has been reported in which deviation of a C1 lateral mass screw caused iatrogenic subarachnoid hemorrhage.[7] Differently from this case, our patient developed obstructive hydrocephalus in a distinct manner even without the use of a spinal instrument.
Considering its possible pathomechanism, postoperative hydrocephalus may be a potential sequel after surgical manipulation to the dura matter at CVJ. In addition to Chiari malformation, several pathologies at CVJ may require durotomy during their surgical treatment. First, as with our case, patients with achondroplasia patients may require duraplasty in combination with upper cervical decompression, especially when constricting soft tissue band is identified after removal of the bone structure.[9,10,11] Second, durotomy is required in resection of spinal cord tumors. A previous study of 118 cases of spinal dumbbell tumors reported that the C-2 nerve root at CVJ was the most common origin of schwannoma.[12] Finally, incidental durotomy may occur with a reported incidence of 3.5% in elective spinal surgery.[13] Surgeons should recognize that spinal surgery at CVJ has the potential risk of this devastating complication.
In the presence of a progressive headache or declining mental status after spinal surgery, intracranial hemorrhage (ICH), such as remote cerebellar hemorrhage, should also be included in the differential diagnosis of intracranial events following spinal surgery. ICH, a rare but potentially fatal complication after spinal surgery, is known to be associated with excessive CSF loss.[14,15,16] The postulated pathophysiology of ICH is that an increase in the transluminal venous pressure or downward cerebellar sag, caused by intracranial hypotension from CSF loss, may result in the stretching and, therefore, tearing of the cerebral and cerebellar veins.[14,16] The onset of clinical symptoms is notably early as compared with that of obstructive hydrocephalus;[6] according to a review of remote cerebellar hemorrhage, symptoms begin within the first 10 h after surgery in 46% of patients.[17] The clinical management of ICH depends primarily on the location of the lesion and severity of neurological impairment. Patients with limited hemorrhage and mild neurological symptoms can be managed conservatively.[15] In comparison, patients with infratentorial subdural fluid collection may deteriorate suddenly after a while as in our case. Such patients should be closely monitored for the subsequent development of obstructive hydrocephalus and neurological deterioration, even when the initial results of neurological and imaging assessments are favorable.
We report a case of progressive obstructive hydrocephalus following decompression surgery at CVJ. Acute obstructive hydrocephalus can occur after decompression surgery at the CVJ and should be included in the differential diagnosis of a postoperative declining mental status. The detection of infratentorial subdural fluid collection should alert physicians to this rare sequela.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
References
- 1.Carey CM, Tullous MW, Walker ML. Hydrocephalus: Etiology, pathologic effects, diagnosis, and natural history. In: Cheek WR, editor. Pediatric Neurosurgery. 3rd ed. Philadelphia: WB Saunders Company; 1994. pp. 185–201. [Google Scholar]
- 2.Bahl A, Murphy M, Thomas N, Gullan R. Management of infratentorial subdural hygroma complicating foramen magnum decompression: A report of three cases. Acta Neurochir (Wien) 2011;153:1123–8. doi: 10.1007/s00701-010-0920-2. [DOI] [PubMed] [Google Scholar]
- 3.Elton S, Tubbs RS, Wellons JC, 3rd, Blount JP, Grabb PA, Oakes WJ. Acute hydrocephalus following a Chiari I decompression. Pediatr Neurosurg. 2002;36:101–4. doi: 10.1159/000048361. [DOI] [PubMed] [Google Scholar]
- 4.Filis AK, Moon K, Cohen AR. Symptomatic subdural hygroma and hydrocephalus following Chiari I decompression. Pediatr Neurosurg. 2009;45:425–8. doi: 10.1159/000270159. [DOI] [PubMed] [Google Scholar]
- 5.Marshman LA, Benjamin JC, Chawda SJ, David KM. Acute obstructive hydrocephalus associated with infratentorial subdural hygromas complicating Chiari malformation Type I decompression. Report of two cases and literature review. J Neurosurg. 2005;103:752–5. doi: 10.3171/jns.2005.103.4.0752. [DOI] [PubMed] [Google Scholar]
- 6.Perrini P, Rawlinson A, Cowie RA, King AT. Acute external hydrocephalus complicating craniocervical decompression for syringomyelia-Chiari I complex: Case report and review of the literature. Neurosurg Rev. 2008;31:331–5. doi: 10.1007/s10143-008-0129-5. [DOI] [PubMed] [Google Scholar]
- 7.Stovell MG, Pillay R. Subarachnoid hemorrhage and acute hydrocephalus as a complication of C1 lateral mass screws. Spine (Phila Pa 1976) 2013;38:E1162–5. doi: 10.1097/BRS.0b013e31829a7c42. [DOI] [PubMed] [Google Scholar]
- 8.Ranjan A, Cast IP. Symptomatic subdural hygroma as a complication of foramen magnum decompression for hindbrain herniation (Arnold-Chiari deformity) Br J Neurosurg. 1996;10:301–3. doi: 10.1080/02688699650040188. [DOI] [PubMed] [Google Scholar]
- 9.Arishima H, Tsunetoshi K, Kodera T, Kitai R, Takeuchi H, Kikuta K. Intraoperative computed tomography for cervicomedullary decompression of foramen magnum stenosis in achondroplasia: Two case reports. Neurol Med Chir (Tokyo) 2013;53:902–6. doi: 10.2176/nmc.cr2012-0421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bagley CA, Pindrik JA, Bookland MJ, Camara-Quintana JQ, Carson BS. Cervicomedullary decompression for foramen magnum stenosis in achondroplasia. J Neurosurg. 2006;104(3 Suppl):166–72. doi: 10.3171/ped.2006.104.3.166. [DOI] [PubMed] [Google Scholar]
- 11.Jha RM, Klimo P, Smith ER. Foramen magnum stenosis from overgrowth of the opisthion in a child with achondroplasia. J Neurosurg Pediatr. 2008;2:136–8. doi: 10.3171/PED/2008/2/8/136. [DOI] [PubMed] [Google Scholar]
- 12.Ozawa H, Kokubun S, Aizawa T, Hoshikawa T, Kawahara C. Spinal dumbbell tumors: An analysis of a series of 118 cases. J Neurosurg Spine. 2007;7:587–93. doi: 10.3171/SPI-07/12/587. [DOI] [PubMed] [Google Scholar]
- 13.McMahon P, Dididze M, Levi AD. Incidental durotomy after spinal surgery: A prospective study in an academic institution. J Neurosurg Spine. 2012;17:30–6. doi: 10.3171/2012.3.SPINE11939. [DOI] [PubMed] [Google Scholar]
- 14.Andrews RT, Koci TM. Cerebellar herniation and infarction as a complication of an occult postoperative lumbar dural defect. AJNR Am J Neuroradiol. 1995;16:1312–5. [PMC free article] [PubMed] [Google Scholar]
- 15.Kaloostian PE, Kim JE, Bydon A, Sciubba DM, Wolinsky JP, Gokaslan ZL, et al. Intracranial hemorrhage after spine surgery. J Neurosurg Spine. 2013;19:370–80. doi: 10.3171/2013.6.SPINE12863. [DOI] [PubMed] [Google Scholar]
- 16.Khalatbari MR, Khalatbari I, Moharamzad Y. Intracranial hemorrhage following lumbar spine surgery. Eur Spine J. 2012;21:2091–6. doi: 10.1007/s00586-012-2187-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Brockmann MA, Groden C. Remote cerebellar hemorrhage: A review. Cerebellum. 2006;5:64–8. doi: 10.1080/14734220500521032. [DOI] [PubMed] [Google Scholar]


