Abstract
Traditional leukocyte adhesion assays have provided significant insight into the mechanisms of leukocyte rolling in part through the use of homogeneously coated surfaces. These assays typically involve protein coating of glass coverslips or plastic petri dishes applied via a static drop of protein solution. With this approach, it is difficult to spatially control the location of proteins to fabricate surface-bound protein gradients that mimic in vivo situations. Microfluidic patterning of proteins with microfluidic devices has become a popular technique due to the ability to spatially pattern proteins on a cellular scale. Despite the advantages of microfluidic patterning, few studies have systematically investigated the effects of perfusion time, protein concentration, and perfusion shear stress on protein deposition. Herein, we demonstrated the fabrication of both line and step gradients of P-selectin on glass substrates that support cell rolling and adhesion assays. Investigation of the flow conditions during the microfluidic patterning led to several significant findings. We observed that the protein deposition time of 5 min was sufficient to deposit adequate P-selectin to support neutrophil rolling. We demonstrated that the amount of membrane P-selectin (mP-selectin) or recombinant P-selectin (rP-selectin) deposited showed a dependence on the perfusion shear stress between 4.0 and 32.0 dyn/cm2, while similar studies with fibronectin or fibrinogen showed no shear stress dependence. Finally, we also created step changes in surface adherent protein concentration of P-selectin to characterize leukocyte-rolling behavior in response to sudden changes in ligand density.
I. INTRODUCTION
The recruitment of leukocytes to the blood vessel wall is a critical process in a number of acute and chronic physiological pathologies including thrombosis,1,2 inflammation,3 atherosclerosis,4,5 and cancer metastasis.6,7 Interaction of leukocytes with the cells of the vessel wall leads to leukocyte rolling, which enables leukocytes to survey the endothelium for inflammatory signals that can activate them and cause firm adhesion and transendothelial migration. The initial adhesion and subsequent rolling of leukocytes on activated endothelial cells, activated platelets, or other leukocytes is mediated by a family of transmembrane glycoproteins called the selectins: P-selectin, E-selectin, and L-selectin.3 Studies have suggested that surface expression of either P-selectin or E-selectin on the blood vessel wall is not homogeneous,8,9 but rather these molecules are often clustered into clathrin-coated pits.10,11
Conventional leukocyte adhesion assays such as static drop, which create a homogeneously coated surface through the incubation of a drop of protein for several hours/overnight, have provided a significant understanding of the mechanisms of the receptor-ligand interactions between leukocytes and the endothelium, as well as with other leukocytes. However, these methods lack the complexity present on the endothelium surface. Therefore, to spatially control the protein locations and fabricate more realistic surface bound protein gradients and combinations in a time and cost effective matter, a new technique is needed.
Micro- and nanofabrication patterning techniques have emerged as tools for fabricating protein patterns that mimic the in vivo microenvironment and regulate leukocyte behavior. For example, microcontact printing has been utilized to pattern P-selectin to study the rate of neutrophil recruitment under venous shear flow conditions,12 or to create asymmetric P-selectin patterns to examine the rolling of human leukemic myeloid (HL-60) cells and develop a label-free cell-separation technique.13 Alternatively, photolithography based patterning methods have been developed to fabricate micron-sized islands of T cell receptor (TCR) ligands (anti-CD3) surrounded by intercellular adhesion molecule-1 (ICAM-1) to investigate T-cell immunological synapse formation,14 to pattern surface-immobilized gradients of P-selectin and ICAM-1 to study HL-60 promyelocytes and Jurkat T lymphocytes rolling and adhesion,15 and to determine the critical patch lengths of P- and L-selectin for the initiation of HL-60 cell rolling under shear flow.16 Block copolymer micellar nanolithography was utilized to pattern platelet receptor glycoprotein Ibα (GpIbα) and study the role of ligand spacing on neutrophil adhesion under flow via the neutrophil integrin Mac-1 (CD11b/CD18),17 while particle lithography was employed to fabricate dots of an anti-human monoclonal antibody (PL1) to P-selectin glycoprotein ligand-1 (PSGL-1) and investigate the role of ligand spacing on neutrophil activation and spreading.18
Microfluidic patterning (μFP) of proteins for studies of leukocyte biology is particularly advantageous due to the fact that both diffusible or substrate bound protein gradients can be generated. For example, Jeon et al. implemented a network of microfluidic channels to generate a soluble gradient of interleukin-8 (IL-8) to study neutrophil chemotaxis,19 while Kim and Haynes investigated the dynamics of neutrophil chemotaxis under competing chemoattractant gradients.20 Schmidt et al. employed microfluidic patterning to fabricate lines of substrate-bound P-selectin to measure bond kinetics through a catch strip assay,21 while our group reported the fabrication of surface-bound lines of P-selectin and E-selectin to investigate the effect of ligand density on neutrophil rolling velocities.22
Although μFP of leukocyte ligands has become more frequently employed, the effects of different flow conditions on ligand deposition have not been well examined. In the present study, we describe the use of μFP to fabricate micron scale line patterns of P-selectin on glass substrates (Figure 1). We investigated the effects of perfusion time, shear stress, and protein concentration on the amount of P-selectin deposited through the use of immunofluorescence microscopy and in vitro leukocyte rolling assays. We determined that optimization of the (μFP) flow conditions reduces the time required to deposit sufficient amounts of P-selectin to support neutrophil rolling to as little as 5 min. This is a significant advantage over traditional approaches that require several hours to deposit similar levels of P-selectin. We also demonstrate that the amount of P-selectin deposited exhibited a shear stress dependence which was in contrast to microfluidic patterning of fibrinogen, fibronectin, or L-selectin. Finally, we show that microfluidic patterning could be utilized to fabricate step gradients in P-selectin to characterize the rolling velocity of leukocytes in response to sudden changes in P-selectin site density.
FIG. 1.
Schematic diagram of the methods of microfluidic patterning of protein, and leukocyte rolling assays: (a) Following attachment of the PDMS stamp to a glass coverslip, a solution of mP-selectin was perfused through the channel to deposit protein on the surface; the amount of protein deposited was optimized by varying the protein concentration, perfusion time, and shear stresses; the PDMS stamp is removed and the glass slide is blocked with 0.5% HSA. (b) Solutions of neutrophils were perfused over the surface perpendicular to the protein patterns at well defined shear rates and the rolling interactions were imaged and recorded.
II. METHODS
A. Neutrophil isolation from whole blood
Blood was obtained via venipuncture from healthy adult volunteers and collected into heparin-coated vacutainers (BD Biosciences, Franklin Lakes, NJ) after informed consent was obtained, according to methods approved by the University of Oklahoma Institutional Review Board. Neutrophils were isolated from whole blood by centrifugation using Lympholyte® - poly (Cedarlane, Burlingtone, NC) following manufacturer's protocol. After isolation, neutrophils were re-suspended at 106 cells/ml in 0.5% human serum albumin (HSA; Gemini Bioproducts, West Sacramento, CA) in Hanks balanced salt solution (HBSS; Walkersville, MD).
B. Microfluidic chamber fabrication and assembly
Microfluidic chambers were prepared from polydimethylsiloxane (PDMS; Sylgard® 184 Silicone Elastomer Kit; Dow Corning, Midland, MI) as previously described.23 Briefly, the PDMS prepolymer and crosslinker were mixed in a 10:1 weight ratio and poured over photoresist structures fabricated by negative photolithography using KMPR 1050 photoresist (MicroChem, Westborough, MA).23 Straight channel photoresist patterns had dimensions (width × height) of 1000 μm × 50 μm, 1000 μm × 100 μm, or 300 μm × 50 μm, while bifurcated channels had dimensions of 500 × 50 μm. After the PDMS was degassed for an hour, an elbow outlet port was placed at one end of the template prior to curing PDMS solution. The PDMS solution was then cured at 80 °C for an hour. The PDMS stamps were sealed to glass cover slips that were treated with a 28% solution of nitric acid for 1 h and then with a 1% AquaSil siliconizing solution for 15 s. PDMS stamps were reversibly sealed to the glass coverslips by first treating the surface of the PDMS with a high voltage BD-20AC laboratory corona treater (Electro-Technic Products; Chicago, IL) for a few seconds prior to attaching to the glass coverslip.22
C. Microfluidic patterning of mP-selectin
μFP was performed using two general types of microfluidic channel designs: straight microchannels and bifurcated (Y-shaped) microchannels. The outlet elbow of the microfluidic channel was connected to a 1 ml syringe via silastic® tubing (1.57 mm ID; 3.18 mm O.D.; Dow Corning, Midland, MI), and HBSS was placed into the reservoir(s). The microfluidic system was then charged with HBSS, and any air bubbles were removed. Membrane P-selectin (mP-selectin) was generously donated by Dr. Roger McEver (Oklahoma Medical Rsearch Foundation, Oklahoma City, OK) and patterned on glass coverslips by varying the deposition shear rate and time. After the protein was patterned, the channels were perfused with HBSS at 24.0 dyn/cm2 to remove any non-adherent or loosely bound protein. The PDMS stamps were then removed and the glass coverslips were blocked with 0.5% HSA in HBSS solution for 30 min at room temperature (Figure 1(a)). Following HSA incubation, the glass coverslips were gently rinsed several times with HBSS buffer via a transfer pipette before attaching a glycotech flow chamber (height = 0.254 mm, width = 5.0 mm) onto the glass coverslips.
To determine the optimum mP-selectin concentration that would support neutrophil rolling, solutions of different mP-selectin concentrations (0.05, 0.075, 0.1, 0.15, and 0.75 μg/ml) were perfused through the microfluidic chambers at a wall shear rate of 16.0 dyn/cm2 for 15 min. The optimum μFP perfusion time was determined by perfusing a 0.1 μg/ml mP-selectin solution at 16.0 dyn/cm2 for 5, 10, or 15 min through the 1000 μm × 50 μm microfluidic channels. To elucidate the optimum μFP flow rate, we perfused a 0.1 μg/ml mP-selectin solution for 15 min at various shear rates (0, 4.0, 8.0, 16.0, and 32.0 dyn/cm2) through the 1000 μm × 50 μm microfluidic channels. Control experiments consisted of placing 20 μl drops of mP-selectin solutions (0.1 and 0.75 μg/ml) on the glass coverslips, incubating for 3 h at room temperature, rinsing the coverslip with HBSS using a transfer pipette to remove any unadsorbed P-selectin, blocking with a 0.5% HSA/HBSS solution, rinsing with HBSS using a transfer pipette, and attaching the glycotech flow chamber. In some experiments, μFP was used to fabricate step gradients of mP-selectin on glass coverslips by perfusing a 0.2 μg/ml mP-selectin solution through one inlet port of a Y-shaped microfluidic channel and a 0.5 μg/ml mP-selectin solution through the other inlet port for 15 min at a perfusion shear rate of 16.0 dyn/cm2. Due to the geometry of the patterning channels, the shear stress was calculated using an equation for rectangular channels with height to width ratios between 0.1 and 1, based on the paper by Son24
where
D. Fluorescence microscopy of protein patterns
To characterize the relative surface densities of mP-selectin deposited on the glass coverslips by the μFP technique or the static drop method, indirect immunofluorescence labeling was performed and the fluorescence intensities were recorded. In addition, recombinant P-selectin (0.5 μg/ml; R&D Systems, Inc. Minneapolis, MN) was used in some microfluidic patterning experiments. Briefly, surfaces coated with mP-selectin or recombinant p-selectin were incubated with 0.5% HSA for one hour, followed by incubation with mouse anti P-selectin mAb G1 (20 μg/ml; generously donated by Rodger McEver, OMRF, Oklahoma City, OK) for one hour. After extensive washes with HBSS, the surfaces were incubated with Alexa Fluor 488 conjugated chicken anti mouse IgG (20 μg/ml; Life Tech, Carlsbad, CA) for one hour at room temperature and washed again with HBSS. In some experiments, fibrinogen conjugated–Alexa fluor647 (Molecular Probes, Inc., Eugene, OR) or fibronectin conjugated-HiLyte fluor 488 (Cytoskeleton, Inc. Denver, CO) was perfused at 5 μg/ml for 15 min at various shear stresses (4.0, 8.0, 16.0, and 32.0 dyn/cm2) through the 300 μm × 50 μm microfluidic channels to determine whether protein deposition is dependent on shear stress.
Fluorescence images of the protein patterns were acquired using a Zeiss microscope equipped with a 20× (NA 0.8) objective and a Hamamatsu ORCA-Flash 4.0 scientific CMOS camera (Figures 4, 5, 6, & 7) or an AxioCam ERC 5 camera (Figures 2 & 8). The images were then analyzed using ImagePro (Mediacy, Rockville, MD) imaging software.
FIG. 4.
Effect of microfluidic patterning time on mP-selectin deposition and neutrophil rolling. Immunofluorescence images of glass coverslips coated with mP-selectin utilizing μFP perfusion times of (a) 5 min, (b) 10 min, or (c) 15 min. The protein line patterns were formed by perfusing a 0.5 μg/ml mP-selectin solution through microfluidic channels (300 μm × 50 μm cross-section) at a perfusion shear stress of 16.0 dyn/cm2. (d) Representative fluorescence intensity line profiles for the different μFP perfusion times. (e) Mean neutrophil rolling velocities on P-selectin coated substrates fabricated with μFP patterning times of 5, 10, and 15 min. The protein patterns for the rolling experiments were formed by perfusing a 0.1 μg/ml mP-selectin solution through microfluidic channels (1000 μm × 50 μm cross-section) at a perfusion shear stress of 16.0 dyn/cm2. No rolling was observed for static patterns at 15 min of patterning time. Data are shown as the average value ± SEM and were derived from three to four experiments with blood from at least three different donors. The number of cells analyzed for each point ranged from 32 to 104 cells. Statistical significance was assessed by comparing each concentration curve to the 15 min curve using a two-way ANOVA with a post-hoc Sidak's multiple comparison test: * P < 0.05 and *** P < 0.0001.
FIG. 5.
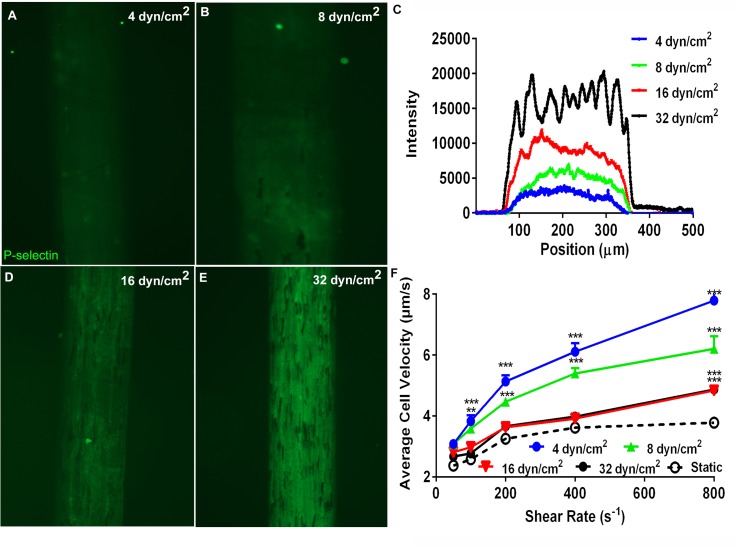
Effect of microfluidic patterning perfusion shear stresses on P-selectin deposition and neutrophil rolling. Immunofluorescence images of glass coverslips coated with mP-selectin utilizing μFP perfusion shear stresses of (a) 4.0 dyn/cm2, (b) 8.0 dyn/cm2, (d) 16.0 dyn/cm2, or (e) 32.0 dyn/cm2. The protein line patterns were formed by perfusing a 0.5 μg/ml mP-selectin solution through microfluidic channels (300 μm × 50 μm cross-section) at the indicated perfusion shear stress level for 15 min. (c) Representative fluorescence intensity line profiles for the different perfusion conditions. (f) Mean neutrophil rolling velocities on mP-selectin coated substrates fabricated with different μFP perfusion shear stresses. The protein patterns for the rolling experiments were formed by perfusing a 0.1 μg/ml mP-selectin solution through microfluidic channels (1000 μm × 50 μm cross-section) at different perfusion shear stresses (4.0–32.0 dyn/cm2) for 15 min. Data are shown as the average value ± SEM and were derived from two to four experiments. The number of cells analyzed for each point ranged from 22 to 154 cells. Statistical significance was assessed by comparing each concentration curve to the static drop curve using a two-way ANOVA with a post-hoc Sidak's multiple comparison test: * P < 0.05, ** P < 0.001, and *** P < 0.0001.
FIG. 6.
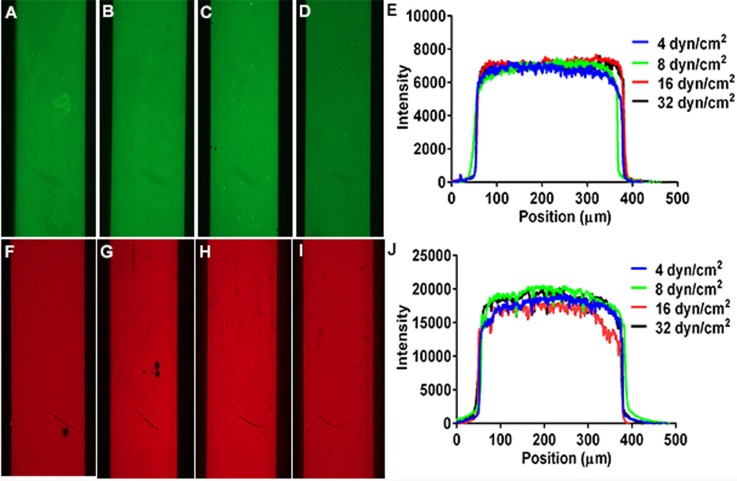
Effect of perfusion shear stress on fibronectin and fibrinogen deposition. Fluorescence images of glass coverslips coated with HiLyte Fluor488-fibronectin or Alexa647-fibrinogen utilizing different μFP perfusion shear stresses. Solutions of 5 μg/ml HiLyte Fluor488-fibronectin were perfused through microfluidic channels (300 μm × 50 μm cross-section) for 15 min at shear stresses of (a) 4.0 dyn/cm2, (b) 8.0 dyn/cm2, (c) 16.0 dyn/cm2, or (d) 32.0 dyn/cm2. (e) Representative fluorescence intensity line profiles of Alexa488-fibronectin deposited at the indicated shear stresses. Solutions of 5 μg/ml Alexa647-fibrinogen were perfused through microfluidic channels (300 μm × 50 μm cross-section) for 15 min at shear stresses of (f) 4.0 dyn/cm2, (g) 8.0 dyn/cm2, (h) 16.0 dyn/cm2, or (i) 32.0 dyn/cm2. (j) Representative fluorescence intensity line profiles of Alexa488-fibronectin deposited at the indicated shear stresses.
FIG. 7.
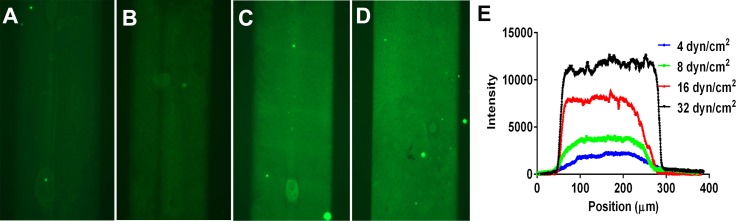
Effect of perfusion shear stress on recombinant P-selectin deposition. Immunofluorescence images of glass coverslips coated with recombinant P-selectin (rP-slectin) utilizing μFP perfusion shear stresses of (a) 4.0 dyn/cm2, (b) 8.0 dyn/cm2, (c) 16.0 dyn/cm2, or (d) 32.0 dyn/cm2. Solutions of 0.5 μg/ml of rP-selectin were perfused through microfluidic channels (300 μm × 50 μm cross-section) for 15 min at the indicated shear stresses. (e) Representative fluorescence intensity line profiles for the different perfusion conditions.
FIG. 2.
Immunofluorescence images of glass coverslips coated with mP-selectin via static drop and μFP. Immunofluorescent images of glass coverslips coated with different concentrations of mP-selectin using either a standard static drop procedure ((a)–(c)) or the μFP technique ((e)–(g)). Fluorescence intensity line scans of the static drop (d) and the μFP (h) coated substrates revealed a 5-fold higher amount of protein deposited by the μFP method. The protein line patterns were formed by perfusing the different mP-selectin solutions through microfluidic channels (300 μm × 50 μm cross-section) at a perfusion shear stress of 16.0 dyn/cm2 for 15 min.
FIG. 8.
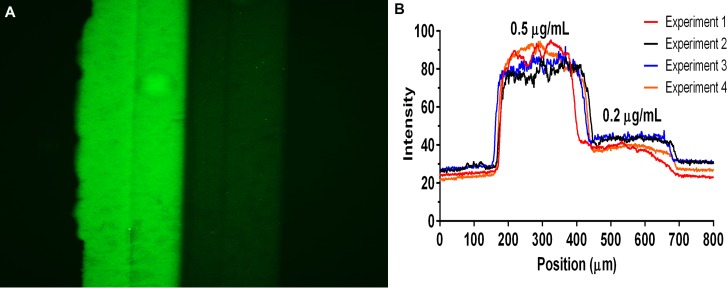
Immunofluorescence image of P-selectin Step Patterns. (a) A representative fluorescence image of a step pattern of mP-selectin formed by perfusing two different concentrations of mP-selectin (0.2 and 0.5 μg/ml) through a Y-shaped microfluidic device (main channel cross-section 500 μm × 50 μm) for 15 min at 16.0 dyn/cm2. (b) Fluorescence intensity line profiles of 4 different step patterns.
E. Neutrophil rolling experiments
The ability of the mP-selectin coated glass coverslips to support neutrophil rolling was tested and characterized by mounting a GlycoTech parallel flow chamber (GlycoTech, Inc. Gaitherburg, MD) on top of the coverslips, such that the direction of flow was perpendicular to the protein patterns and initially filling the chambers with HBSS (Figure 1(b)). Neutrophil solutions were then initially perfused perpendicular to the patterned protein lines at a rate of 0.5 dyn/cm2. Neutrophils were allowed to accumulate for one minute, before the cell suspension was switched to cell-free HBSS. The shear rate was subsequently increased to 1.0 dyn/cm2, and thereafter increased every 45 s to 2.0, 4.0, and 8.0 dyn/cm2 or until all the cells had rolled off the patterns.
In some experiments, neutrophils were perfused over surfaces patterned with a step change in mP-selectin surface density. In these experiments, neutrophils were allowed to accumulate at 0.5 dyn/cm2 for 1 min, the perfusion solution changed to cell-free HBSS, and the shear rate changed to either 2.0 dyn/cm2 or 8.0 dyn/cm2. To determine whether the sequence of exposure to different mP-selectin surface densities affected neutrophil rolling, neutrophils were perfused over the surface in one direction (i.e., 0.2 μg/ml → 0.5 μg/ml) and then the flow direction was reversed and a fresh neutrophil solution was perfused over the surface (i.e., 0.5 μg/ml → 0.2 μg/ml).
F. Data acquisition and cell tracking analysis
Neutrophil rolling on mP-selectin coated surfaces was observed with an inverted Zeiss microscope equipped with a 10X objective and a CCD camera (DAGE-MTI CCD-300) and recorded on video tapes or DVDs at 30 frames per second. Instantaneous rolling velocities were measured using frame-by-frame analysis using MetaMorph image analysis software.23 Each experiment was repeated at least three times with at least three separate donors to verify consistency of results.
G. Statistical analysis
All data values were expressed as mean ± standard error of the mean unless otherwise specified. When appropriate, statistical significance was assessed by a two way ANOVA with a post-hoc Sidak's multiple comparisons test, with P < 0.05 considered as statistically significant.
III. RESULTS AND DISCUSSION
A. Microfluidic flow patterning deposits more mP-selectin than static drop incubation
We previously used microfluidic channels to fabricate lines of mPselectin on glass coverslips for use in neutrophil rolling assays.22 However, in this previous study, mP-selectin was allowed adsorb to the glass substrates under static conditions for 3 h. To ascertain if the amount of time required for the protein patterning procedure could be reduced by increasing the transport of mP-selectin to the surface by convection we perfused solutions of 0.75 and 1.5 μg/ml of mP-selectin through 1000 μm × 100 μm microfluidic devices at 16.0 dyn/cm2 for 15 min. These solutions represent typical concentrations of mP-selectin utilized in a standard static drop coating method for preparing glass coverslips for neutrophil rolling studies. When neutrophils were perfused over the microfluidic patterned mP-selectin surfaces at venous levels of shear stress (0.5–2.0 dyn/cm2), we observed immediate arrest of the neutrophils rather than the normal rolling behavior seen in static drop patterning.
To determine whether this immediate arrest of neutrophils upon contact with the surface was due to a high mP-selectin site density, we performed immunofluorescence staining of both statically deposited and laminar flow deposited mP-selectin (Figure 2). When glass slides were coated by static drops of mP-selectin, there was a significant increase in the fluorescence intensity when the mP-selectin concentration was increased from 0.15 (Figure 2(a)) to 0.75 μg/ml (Figure 2(b)). However, a further increase of the mP-selectin concentration to 1.5 μg/ml (Figure 2(c)) had virtually no effect on the fluorescence intensity. Similarly when glass slides were coated with lines of mP-selectin by μFP, the fluorescence intensity also increased with mP-selectin concentration (Figures 2(e)–2(h)). It is important to note that the fluorescence intensities of the mP-selectin deposited on the glass slides by the μFP method were approximately 5 times higher than those deposited by the static drops for the same mP-selectin solution concentration. These results suggest that the μFP method was capable of either (i) depositing more total mP-selectin or (ii) depositing more functional mP-selectin than the static drop method and may explain why immediate arrest of neutrophils rather than rolling was observed.
B. Adsorption of mP-selectin during microfluidic pattering is dependent upon the solution concentration, perfusion time, and perfusion shear stress
Having observed that μFP deposits more mP-selectin than static drops, we then proceeded to determine the range of mP-selectin solution concentrations, perfusion times, and perfusion rates (i.e., shear stress) that can be utilized with μFP to support leukocyte rolling. Previous studies have demonstrated that E-, L-, and P-selectin-mediated leukocyte rolling velocities are dependent upon the selectin ligand density on the surface.15,25,26 Thus we perfused solutions of isolated neutrophils perpendicularly across lines of mP-selectin formed by μFP and utilized the neutrophil rolling velocities as an indirect measure of mP-selectin ligand density.
1. mP-selectin solution concentration effects
To form mP-selectin lines with different site densities, the solution concentration of mP-selectin was varied (0.05–0.15 μg/ml) while the shear rate (16.0 dyn/cm2) and μFP time (15 min) were kept constant. When neutrophils were perfused over the mP-selectin lines, neutrophils would rapidly attach at the upstream region of the line, roll across the line, and then detach from the surface when they reached the downstream region of the line. To analyze rolling velocities over a wide range of wall shear stresses, we allowed neutrophils to initially accumulate on the lines at a low wall shear stress 0.5 dyn/cm2 and then systematically increased the wall shear stress every 60 s until a shear stress of 8.0 dyn/cm2 was reached.
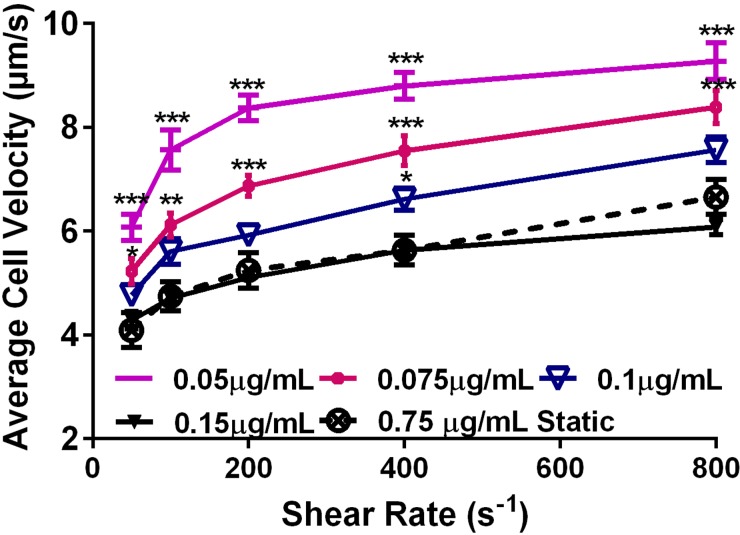
As shown in Figure 3, the rolling velocity of neutrophils on the μFP mP-selectin lines steadily decreased as the solution concentration of mP-selectin perfused during μFP was increased from 0.05 to 0.15 μg/ml. A two way ANOVA was used to assess the difference in neutrophil rolling velocities with varying shear rate and patterning concentration. Figure 3 shows that at mP-selectin concentrations of 0.05 and 0.075 μg/ml there was a significant increase over the range of shear rates used in the rolling velocity as compared to the control substrate coated via a 0.75 μg/ml mP-selectin static drop (P < 0.05). However, for concentrations of 0.1 and 0.15 μg/ml, there was no statistically significant difference in the rolling velocity of neutrophils except at 400 s–1 for 0.1 μg/ml concentration. This was somewhat surprising given the greater than 5-fold difference between the coating concentrations and the 12-fold (15 min vs 3 h) difference in coating time. As an additional control, substrates were coated with 0.1 μg/ml of P-selectin via the static drop method for 3 h; however, no rolling was observed indicating insufficient protein deposition. These results provide additional evidence that the μFP technique is an efficient method to deposit mP-selectin site densities that support neutrophil rolling in a short amount of time. In addition, these results demonstrate that the amount of mP-selectin deposited by μFP depends on the solution concentration of mP-selectin.
FIG. 3.
Effect of the P-selectin patterning concentration on neutrophil rolling velocities. Mean velocities of neutrophils rolling on mP-selectin lines of different coating concentrations. Protein line patterns were formed via μFP at 16.0 dyn/cm2 for 15 min. Data are shown as the average value ±SEM and were derived from three to four experiments with blood from at least three different donors. The number of cells analyzed for each point ranged from 19 to 102 cells. Statistical significance was assessed by comparing each concentration curve to the static 0.75 μg/ml curve using a two-way ANOVA with a post-hoc Sidak's multiple comparison test: * P < 0.05, ** P < 0.001, and *** P < 0.0001.
2. μFP perfusion time effects
To determine how the amount of mP-selectin deposited on glass coverslips was dependent on the duration of the μFP perfusion, we perfused solutions of 0.1 μg/ml mP-selectin through microfluidic channels at a wall shear stress of 16.0 dyn/cm2 for different time intervals. Figures 4(a)–4(d) show that the fluorescence intensity of the mP-selectin patterned lines increased when the time of patterning was increased from 5 min to 10 min. However, a further increase in the time to 15 min did not significantly increase the fluorescence intensity. Similar to the concentration studies described previously, we also measured the rolling velocities of neutrophils that were perfused perpendicularly across the mP-selectin lines. As illustrated in Figure 4(e), the rolling velocity of neutrophils was inversely related to the μFP duration. Deposition of mP-selectin lines by five minutes of μFP produced average neutrophil rolling velocities of 7.43 ± 0.97 μm/s when neutrophils were perfused at a wall shear stress of 2.0 dyn/cm2. Doubling of the μFP time to 10 min reduced the rolling velocities to 6.15 ± 0.43 μm/s, however, there was no significant change in rolling velocities (6.15 ± 0.40 μm/s, P-value 0.993) when the μFP time was further increased to 15 min. As a control, we also prepared substrates by coating glass coverslips with a 0.75 μg/ml mP-selectin static drop for 15 min. However, no neutrophil rolling was observed on these substrates. Overall these results demonstrate that at a shear condition of 16.0 dyn/cm2, 5 min of μFP is sufficient to fabricate surfaces which support neutrophil rolling, and that 10 min of μFP is sufficient to reach the equilibrium amount of mP-selectin deposited. We expect that the perfusion time required to deposit sufficient amounts of P-selectin will vary depending upon the magnitude of the shear condition.
3. Effect of μFP perfusion shear stress on deposition of mP-selectin
To investigate the role of perfusion rate on the amount of mP-selectin deposited during μFP, we perfused solutions of 0.1 μg/ml mP-selectin through microfluidic channels for 15 min at various wall shear stress (4.0–32.0 dyn/cm2). As shown in Figures 5(a)–5(e), the fluorescence intensity of the mP-selectin patterned lines was highly dependent upon the wall shear stress generated during the μFP process (i.e., patterning shear stress). There was a steady increase in the fluorescence as the shear stress was increased from 4.0 to 16.0 dyn/cm2 (Figure 5(e)). Further increasing the shear rate to 32.0 dyn/cm2 had little effect on the fluorescence intensity. Similar to the fluorescence results, Figure 5(f) shows that the rolling velocity of neutrophils on the mP-selectin lines was also highly dependent upon the wall shear stress generated during μFP process. A patterning shear stress of 4.0 dyn/cm2 produced the highest rolling velocities and is indicative of a low mP-selectin ligand density. Subsequent increases of the patterning shear rate to 8.0 dyn/cm2 and 16.0 dyn/cm2 led to an increase in the mP-selectin site densities as indicated by the reduced neutrophil rolling velocities. Further increasing the patterning shear rate to 32.0 dyn/cm2 led to no change in the rolling velocities suggesting that a similar amount of mP-selectin was deposited as to the patterning shear stress of 16.0 dyn/cm2. It should be noted that the rolling velocities obtained with a patterning shear stress of 16.0 or 32.0 dyn/cm2 were very similar to the rolling velocity of neutrophils on glass coverslips coated by a 3 h incubation with a static drop of 0.75 μg/ml. As a control, we also coated some glass slides with a static drop (i.e., 0 dyn/cm2) of 0.1 μg/ml mP-selectin for 15 min; however, no neutrophil adhesion or rolling was observed on these surfaces. Taken together, these results demonstrate that the amount of mP-selectin deposited by μFP depends upon the flow conditions and suggest that mP-selectin adsorption is transport-limited for patterning shear stresses between 4.0 and 32.0 dyn/cm2.
4. Effect of μFP perfusion shear stress on deposition of fibrinogen and fibronectin
The observation that the amount of mP-selectin deposited by microfluidic patterning was dependent upon the shear stress was surprising given the fact that previous studies on the effect of shear rate on protein adsorption to a surface have reported that adsorption is independent of shear rate.27 To determine whether this observation was unique to mP-selectin, we performed similar perfusion shear stress studies with recombinant P-selectin and fluorescently labeled Alexa 647-fibrinogen and HiLyte Fluor 488 labeled fibronectin. When HiLyte Fluor 488 labeled fibronectin was perfused through the microfluidic chambers at a concentration of 5 μg/ml for 15 min at various wall shear stresses (4.0–32.0 dyn/cm2), virtually no change in the fluorescence intensity of the patterned lines was observed (Figures 6(a)–6(e)). Similar results were obtained when Alexa-647 fibrinogen was perfused at a concentration of 5 μg/ml for 15 min at various wall shear stresses (4.0–32.0 dyn/cm2) (Figures 6(f)–6(j)).
The exact cause for this shear stress dependency is unknown at this time but could be related to the unique shape of mP-selectin. It has been reported that mP-selectin forms rosettes of 2–6 molecules that are joined by their hydrophobic membrane-spanning domains.28 To determine whether the rosette shape of mP-selectin was the cause of the shear dependency, we performed a set of experiments in which we perfused a commercially available recombinant human P-selectin (rP-selectin) through our microfluidic devices at a concentration of 0.5 μg/ml and shear stresses of 4.0, 8.0, 16.0, and 32.0 dyn/cm2 for 15 min. We then performed immunohistochemistry to fluorescently quantify the amount of P-selectin deposited. As shown in Figure 7, we observed that the amount of rP-selectin deposited was also dependent upon the perfusion shear stress level. These results suggest that the shear dependency of mP-selectin was not dependent upon its rosette shape but may be related to the general structure of P-selectin.
5. Neutrophil rolling on step gradients of mP-selectin
It is known that concentration gradients play an important role in regulating neutrophil chemotaxis, however, very little is known about how neutrophils respond to spatial changes in the concentration of surface-adsorbed adhesion molecules during rolling. To investigate the effects of concentration changes during neutrophil rolling, we fabricated surfaces with step changes in mP-selectin concentrations using “Y-shaped” microfluidic channels and measured the changes in neutrophil rolling velocities at the concentration interface. Figure 8(a) shows a representative fluorescence image of a glass slide that was patterned with two different mP-selectin coating concentrations (0.2 and 0.5 μg/ml). Figure 8(b) shows the lines profiles for four substrates that were prepared in an identical fashion and demonstrate the reproducibility of the technique and the abrupt change in the amount of mP-selectin deposited.
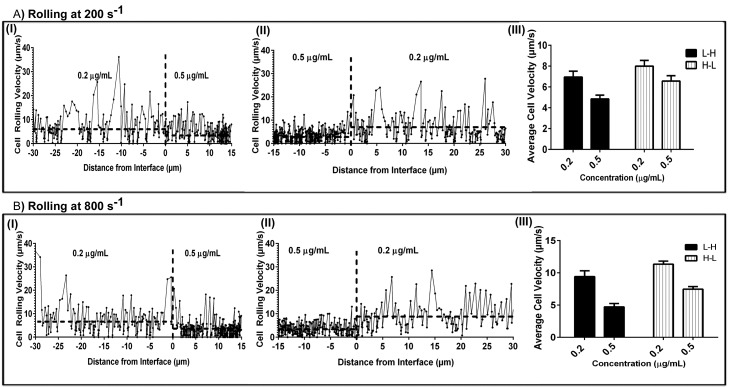
To directly examine changes in neutrophil rolling velocities in response to a step change in mP-selectin concentration, we performed frame-by-frame image analysis to measure the instantaneous rolling velocity for a population of cells. Cells were tracked for 15 μm on the high concentration (0.5 μg/ml) side of the interface and 30 μm on the low concentration (0.2 μg/ml) side. These lengths were chosen to give ample room for velocity to be tracked, greater than one cell length, while maintaining a similar number of data points on both sides of the interface. The mean cell velocity was plotted versus distance from the interface (Figure 9).
FIG. 9.
Neutrophil Rolling Behavior on P-selectin Step Patterns Instantaneous and average rolling velocities of neutrophils rolling on increasing (0.2 → 0.5 μg/ml) and decreasing (0.5 → 0.2 μg/ml) step patterns of mP-selectin. Frame by frame velocities of representative neutrophils rolling on an increasing (A.I, B.I) and decreasing (A.II, B.II) P-selectin step patterns at 200 s−1 and 800 s−1. The horizontal dotted lines represent the average rolling velocity for the representative cell on each side of the interface (indicated by vertical dotted line). The mean velocities for a population of neutrophils rolling on the step gradients of P-selectin are presented as average ± SEM at 200 s−1 (A.III) and 800 s−1 (B.III). mP-selectin step patterns were fabricated by perfusing two different concentrations of mP-selectin (0.2 and 0.5 μg/ml) through Y-shaped microfluidic channels (main channel cross-section 500 μm × 50 μm) for 15 min at 16.0 dyn/cm2.
When neutrophils rolled across a surface that had a mP-selectin step change from 0.2 → 0.5 μg/ml at 200 s–1, there was an immediate drop in neutrophil rolling velocity at the interface. Figure 9A shows a representative example of a single cell rolling across the interface, as well as the average velocity change for a population of cells (6.96 → 4.86 = 2.1 μm/sec). To determine whether the direction of the concentration change (i.e., low to high or high to low) influenced the change in neutrophil rolling velocities, we changed the direction of flow and perfused neutrophils over the same surface. As expected, when neutrophils rolled over the interface that changed from 0.5 → 0.2 μg/ml mP-selectin there was an immediate increase in neutrophil rolling velocity (6.57 → 8.00 = 1.43 μm/sec). It is interesting to note that despite using the same substrate and shear rate there were differences in the absolute change of the rolling velocities on a given substrate concentration depending on the direction of rolling.
To further probe this observation, we repeated the above step change experiments but at a higher shear rate (800 s–1). Similar to the results above, regardless of the direction of flow, there was an immediate change in the neutrophil rolling velocities at the interface (Figure 9B). In addition, we observed that changes in rolling velocities were lower when rolled over a decreasing concentration step change (0.5 → 0.2 μg/ml) (7.47 → 11.35 = 3.88 μm/s) than over an increasing step change (0.2 → 0.5 μg/ml) (9.17 → 4.77 = −4.40 μm/s). The cause for this difference is not known, but these results suggest that a neutrophil's response to a step change in ligand density may be dependent upon its rolling history.
IV. CONCLUSIONS
In this study, we describe a simple and straight-forward method to fabricate line patterns of mP-selectin via microfluidic patterning. This technique not only has the advantage of providing precise control over the location of a protein, such as mP-selectin, on a surface, but also reduces the preparation time. For example, we demonstrate that optimization of the flow patterning process can reduce the time to deposit sufficient levels of mP-selectin to support leukocyte rolling to as little as 5 min, which is a 30 fold decrease in time compared to static coating methods. Another important and novel aspect of our work is the observation that the amount of mP-selectin deposited was dependent upon the perfusion flow or shear stress. Most studies of protein adsorption to a surface have reported that adsorption is independent of fluid flow. However, the rate or amount of protein that adsorbs onto a surface can be kinetically limited or transport limited. Our results that the deposition of fibronectin and fibrinogen were independent of shear stress agree with this normal behavior. Conversely our results with both mP-selectin and rP-selectin suggest that deposition in our system was dependent upon the shear stresses between 4.0 and 32.0 dyn/cm2. Currently, we do not know the exact cause for these observations. However, one possibility is that the shear is affecting the binding kinetics of P-selectin to the surface. In support of this possibility is the recent work by Roy et al., who showed using microfluidic channels that the binding kinetics of Concanavalin A and Abrus Agglutinin were a function of shear rates.29 Below a critical wall shear rate of ∼4000 s–1, the binding efficiency was enhanced with shear rate, however, above this shear rate, the binding rate decreased with shear. The authors suggest that Langmuir association and dissociation constants were dependent on the fluid forces. Although our system does not involve protein-protein interactions, the adsorption of P-selectin to the glass surface could be viewed as a binding reaction.
Finally, we demonstrate that the use of Y-shaped microfluidic channels allows one to easily fabricate surfaces with step changes in adsorbed mP-selectin, which can be used to investigate the behavior of rolling neutrophils to abrupt changes in ligand densities. Our results suggest that a neutrophil's response to a step change in ligand density may be dependent upon its rolling history. This intriguing result provides additional support to the importance of utilizing substrates with well-defined and spatially controlled protein patterns in probing the biology of rolling leukocytes. We anticipate that this technique can be applied with other leukocyte ligands (i.e., E-selectin, L-selectin, ICAM-1) as well as and to fabricate multi-ligand surfaces (e.g., P-selectin/ICAM) relevant to leukocyte rolling.
ACKNOWLEDGMENTS
This work was supported by the National Institutes of Health Grant Nos. R21HL115601 and R44HL114246. The authors would like to thank Dr. Trevor Snyder for critically reading the manuscript.
References
- 1. Afshar-Kharghan V. and Thiagarajan P., “ Leukocyte adhesion and thrombosis,” Curr. Opin. Hematol. 13(1), 34–39 (2006). 10.1097/01.moh.0000190107.54790.de [DOI] [PubMed] [Google Scholar]
- 2. Saha P., Humphries J., Modarai B., Mattock K., Waltham M., Evans C. E., Ahmad A., Patel A. S., Premaratne S., Lyons O. T. A. et al. , “ Leukocytes and the natural history of deep vein thrombosis: Current concepts and future directions,” Arterioscler. Thromb. Vasc. Biol. 31(3), 506–512 (2011). 10.1161/ATVBAHA.110.213405 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. McEver R. P., “ Selectins: Initiators of leucocyte adhesion and signalling at the vascular wall,” Cardiovasc. Res. 2015, 1–9. 10.1093/cvr/cvv154 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Gerszten R. E., Lim Y. C., Ding H. T., Snapp K., Kansas G., Dichek D. A., Cabañas C., Sánchez-Madrid F., Gimbrone M. A., Rosenzweig A. et al. , “ Adhesion of monocytes to vascular cell adhesion molecule-1-transduced human endothelial cells: Implications for atherogenesis,” Circ. Res. 82(8), 871–878 (1998). 10.1161/01.RES.82.8.871 [DOI] [PubMed] [Google Scholar]
- 5. Weber C., Zernecke A., and Libby P., “ The multifaceted contributions of leukocyte subsets to atherosclerosis: Lessons from mouse models,” Nat. Rev. Immunol. 8(10), 802–815 (2008). 10.1038/nri2415 [DOI] [PubMed] [Google Scholar]
- 6. Kim Y. J., Borsig L., Han H. L., Varki N. M., and Varki A., “ Distinct selectin ligands on colon carcinoma mucins can mediate pathological interactions among platelets, leukocytes, and endothelium,” Am. J. Pathol. 155(2), 461–472 (1999). 10.1016/S0002-9440(10)65142-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Borsig L., Wong R., Hynes R. O., Varki N. M., and Varki A., “ Synergistic effects of L- and P-Selectin in facilitating tumor metastasis can involve non-mucin ligands and implicate leukocytes as enhancers of metastasis,” Proc. Natl. Acad. Sci. U.S.A. 99(4), 2193–2198 (2002). 10.1073/pnas.261704098 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Burns A. R., Bowden R. A., Abe Y., Walker D. C., Simon S. I., Entman M. L., and Smith C. W., “ P-selectin mediates neutrophil adhesion to endothelial cell borders,” J. Leukoc. Biol. 65(3), 299–306 (1999). [DOI] [PubMed] [Google Scholar]
- 9. Kim M. B. and Sarelius I. H., “ Role of shear forces and adhesion molecule distribution on P-selectin-mediated leukocyte rolling in postcapillary venules,” Am. J. Physiol. Heart Circ. Physiol. 287(6), H2705–H2711 (2004). 10.1152/ajpheart.00448.2004 [DOI] [PubMed] [Google Scholar]
- 10. Setiadi H. and McEver R. P., “ Clustering endothelial E-selectin in clathrin-coated pits and lipid rafts enhances leukocyte adhesion under flow,” Blood 111(4), 1989–1998 (2008). 10.1182/blood-2007-09-113423 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Setiadi H. and McEver R. P., “ Signal-dependent distribution of cell surface P-selectin in clathrin-coated pits affects leukocyte rolling under flow,” J. Cell Biol. 163(6), 1385–1395 (2003). 10.1083/jcb.200307178 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Lee D. and King M. R., “ Microcontact printing of P-selectin increases the rate of neutrophil recruitment under shear flow,” Biotechnol. Prog. 24(5), 1052–1059 (2008). 10.1002/btpr.35 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Lee C. H., Bose S., Van Vliet K. J., Karp J. M., and Karnik R., “ Examining the lateral displacement of HL60 cells rolling on asymmetric P-selectin patterns,” Langmuir 27(1), 240–249 (2011). 10.1021/la102871m [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Doh J. and Irvine D. J., “ Immunological synapse arrays: Patterned protein surfaces that modulate immunological synapse structure formation in T cells,” Proc. Natl. Acad. Sci. U.S.A. 103(15), 5700–5705 (2006). 10.1073/pnas.0509404103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Herman C. T., Potts G. K., Michael M. C., Tolan N. V., and Bailey R. C., “ Probing dynamic cell-substrate interactions using photochemically generated surface-immobilized gradients: application to selectin-mediated leukocyte rolling,” Integr. Biol. (Camb). 3, 779–791 (2011). 10.1039/c0ib00151a [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Tong Z., Cheung L. S.-L., Stebe K. J., and Konstantopoulos K., “ Selectin-mediated adhesion in shear flow using micropatterned substrates: Multiple-bond interactions govern the critical length for cell binding,” Integr. Biol. 4(8), 847 (2012). 10.1039/c2ib20036h [DOI] [PubMed] [Google Scholar]
- 17. Kruss S., Erpenbeck L., Amschler K., Mundinger T. A., Boehm H., Helms H. J., Friede T., Andrews R. K., Schön M. P., and Spatz J. P., “ Adhesion maturation of neutrophils on nanoscopically presented platelet glycoprotein Ibα,” ACS Nano 7(11), 9984–9996 (2013). 10.1021/nn403923h [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Taylor Z. R., Keay J. C., Sanchez E. S., Johnson M. B., and Schmidtke D. W., “ Independently controlling protein dot size and spacing in particle lithography,” Langmuir 28, 9656–9663 (2012). 10.1021/la300806m [DOI] [PubMed] [Google Scholar]
- 19. Li Jeon N., Baskaran H., Dertinger S. K. W., Whitesides G. M., Van de Water L., and Toner M., “ Neutrophil chemotaxis in linear and complex gradients of interleukin-8 formed in a microfabricated device,” Nat. Biotechnol. 20(8), 826–830 (2002). 10.1038/nbt712 [DOI] [PubMed] [Google Scholar]
- 20. Kim D. and Haynes C. L., “ Neutrophil chemotaxis within a competing gradient of chemoattractants,” Anal. Chem. 84(14), 6070–6078 (2012). 10.1021/ac3009548 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Schmidt B. J., Huang P., Breuer K. S., and Lawrence M. B., “ Catch strip assay for the relative assessment of two-dimensional protein association kinetics,” Anal. Chem. 80(4), 944–950 (2008). 10.1021/ac071529i [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Nalayanda D. D., Kalukanimuttam M., and Schmidtke D. W., “ Micropatterned surfaces for controlling cell adhesion and rolling under flow,” Biomed. Microdevices 9(2), 207–214 (2007). 10.1007/s10544-006-9022-6 [DOI] [PubMed] [Google Scholar]
- 23. Coghill P. A., Kesselhuth E. K., Shimp E. A., Khismatullin D. B., and Schmidtke D. W., “ Effects of microfluidic channel geometry on leukocyte rolling assays,” Biomed. Microdevices 15(1), 183–193 (2013). 10.1007/s10544-012-9715-y [DOI] [PubMed] [Google Scholar]
- 24. Son Y., “ Determination of shear viscosity and shear rate from pressure drop and flow rate relationship in a rectangular channel,” Polymer (Guildf). 48(2), 632–637 (2007). 10.1016/j.polymer.2006.11.048 [DOI] [Google Scholar]
- 25. McEver R. P. and Zhu C., “ Rolling cell adhesion,” Annu. Rev. Cell Dev. Biol. 26, 363–396 (2010). 10.1146/annurev.cellbio.042308.113238 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Alon R., Rossiter H., Wang X., Springer T. A., and Kupper T. S., “ Distinct cell surface ligands mediate t lymphocyte attachment and rolling on p and e selectin under physiological flow,” J. Cell Biol. 127(5), 1485–1495 (1994). 10.1083/jcb.127.5.1485 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Ortega-Vinuesa J. L., Tengvall P., Wälivaara B., and Lundström I., “ Stagnant versus dynamic conditions: A comparative adsorption study of blood proteins,” Biomaterials 19(1-3), 251–262 (1998). 10.1016/S0142-9612(97)00206-8 [DOI] [PubMed] [Google Scholar]
- 28. Ushiyama S., Laue T. M., Moore K. L., Erickson H. P., and McEver R. P., “ Structural and functional characterization of monomeric soluble p-selectin and comparison with membrane p-selectin,” J. Biol. Chem. 268(20), 15229–15237 (1993). [PubMed] [Google Scholar]
- 29. Roy B., Das T., Maiti T. K., and Chakraborty S., “ Effect of fluidic transport on the reaction kinetics in lectin microarrays,” Anal. Chim. Acta 701(1), 6–14 (2011). 10.1016/j.aca.2011.05.049 [DOI] [PubMed] [Google Scholar]