Abstract
Background
The prevalence of novel type 1 diabetes mellitus (T1DM) antibodies targeting eukaryote translation elongation factor 1 alpha 1 autoantibody (EEF1A1-AAb) and ubiquitin-conjugating enzyme 2L3 autoantibody (UBE2L3-AAb) has been shown to be negatively correlated with age in T1DM subjects. Therefore, we aimed to investigate whether age affects the levels of these two antibodies in nondiabetic subjects.
Methods
EEF1A1-AAb and UBE2L3-AAb levels in nondiabetic control subjects (n=150) and T1DM subjects (n=101) in various ranges of age (18 to 69 years) were measured using an enzyme-linked immunosorbent assay. The cutoff point for the presence of each autoantibody was determined based on control subjects using the formula: [mean absorbance+3×standard deviation].
Results
In nondiabetic subjects, there were no significant correlations between age and EEF1A1-AAb and UBE2L3-AAb levels. However, there was wide variation in EEF1A1-AAb and UBE2L3-AAb levels among control subjects <40 years old; the prevalence of both EEF1A1-AAb and UBE2L3-AAb in these subjects was 4.4%. When using cutoff points determined from the control subjects <40 years old, the prevalence of both autoantibodies in T1DM subjects was decreased (EEFA1-AAb, 15.8% to 8.9%; UBE2L3-AAb, 10.9% to 7.9%) when compared to the prevalence using the cutoff derived from the totals for control subjects.
Conclusion
There was no association between age and EEF1A1-AAb or UBE2L3-AAb levels in nondiabetic subjects. However, the wide variation in EEF1A1-AAb and UBE2L3-AAb levels apparent among the control subjects <40 years old should be taken into consideration when determining the cutoff reference range for the diagnosis of T1DM.
Keywords: Autoantibodies; Diabetes mellitus, type 1; Eukaryote translation elongation factor 1 alpha 1 autoantibody; Ubiquitin-conjugating enzyme 2L3 autoantibody
INTRODUCTION
Type 1 diabetes mellitus (T1DM) results from immune mediated destruction of pancreatic β-cells. The presence of autoantibodies, such as anti-glutamic acid decarboxylase (GAD65) autoantibody (GADA), precedes clinical disease by months or even years and persists after the disease becomes clinically overt [1,2]. GADA is used not only to diagnose autoimmune-mediated T1DM, but also to predict the course of disease progression [3,4].
We have recently reported on two novel autoantibodies found through exploration of T1DM autoantibody repertoires using protein microarrays [5]. These two autoantibodies, eukaryote translation elongation factor 1 alpha 1 autoantibody (EEF1A1-AAb) and ubiquitin-conjugating enzyme 2L3 autoantibody (UBE2L3-AAb), were detected in 40% of GADA negative T1DM patients as well as in patients with fulminant T1DM [5]. The prevalence of EEF1A1-AAb and UBE2L3-AAb increased as the age of disease onset decreased, whilst EEF1A1-AAb and UBE2L3-AAb were only detected in T1DM patients <40 years old [5]. Given that age may be associated with autoantibody positivity in T1DM subjects, more studies are needed to determine whether the concentration of EEF1A1-AAb and UBE2L3-AAb in control subjects is also affected by age. Cutoff values for antibody positivity are typically set at the 99th percentile of antibody levels obtained from nondiabetic control subjects [6]; therefore, it is important to measure the level of these novel autoantibodies in nondiabetic control subjects over a broad range of ages. However, our previous investigation regarding these antibodies in control subjects across various age ranges was limited because of the unavailability of a sufficient pool of nondiabetic control subjects <40 years old.
In this report, we aimed to investigate whether age affects the level of EEF1A1-AAb and UBE2L3-AAb in nondiabetic subjects and to confirm low levels of EEF1A1-AAb and UBE2L3-AAb in young, nondiabetic control subjects.
METHODS
Sample preparation
The control serum samples were obtained from nondiabetic control subjects ≥18 years old who had a fasting plasma glucose concentration of <6.1 mmol/L and glycosylated hemoglobin value of <5.8% (40.0 mmol/mol). To assure an even age distribution in the control sample, 20 samples were collected in each age group (i.e., 18 to 29, 30 to 39, 40 to 49, 50 to 59, and 60 to 69 years). Age-matched serum samples from T1DM patients were also collected to compare the level of autoantibodies with those of the control samples; T1DM samples were selected as described in a previous report [5]. As we reported previously, EEF1A1-AAb and UBE2L3-AAb were detected only in T1DM patients <40 years old previously [5], and 50 control samples from subjects <40 years old were added to compare the EEF1A1-AAb and UBE2L3-AAb levels between the age-matched control and T1DM groups (Table 1). GADA levels were determined using a commercially available radio-immunoassay kit (RSR Ltd., Cardiff, UK) [7] recommended by the Diabetes Autoantibody Standardization Program [8].
Table 1. Prevalence of EEF1A1-AAb and UBE2L3-AAb by age group.
| Variable | No. (men %) | EEF1A1-AAb, % (number) | UBE2L3-AAb, % (number) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Reference range 1a | Reference range 2b | Reference range 1c | Reference range 2d | |||||||
| Control | T1DM | Control | T1DM | Control | T1DM | Control | T1DM | Control | T1DM | |
| Age, yr | ||||||||||
| 18–29 | 30 (46.7) | 49 (53.1) | 6.7 (2/30) | 30.6 (15/49) | 3.3 (1/30) | 18.4 (9/49) | 6.7 (2/30) | 22.4 (11/49) | 3.3 (1/30) | 16.3 (8/49) |
| 30–39 | 60 (51.7) | 28 (39.3) | 3.3 (2/60) | 3.6 (1/28) | 1.7 (1/60) | 0 | 3.3 (2/60) | 0 | 3.3 (2/60) | 0 |
| 40–49 | 20 (50.0) | 16 (68.8) | 5.0 (1/20) | 0 | 5 (1/20) | 0 | 5.0 (1/20) | 0 | 0 | 0 |
| 50–59 | 20 (50.0) | 6 (66.7) | 0 | 0 | 0 | 0 | 5.0 (1/20) | 0 | 5.0 (1/20) | 0 |
| 60–69 | 20 (50.0) | 2 (0.0) | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| P for trendse | - | - | 0.615 | 0.001 | 0.423 | 0.009 | 0.409 | 0.003 | 0.588 | 0.014 |
EEF1A1-AAb, eukaryote translation elongation factor 1 alpha 1 autoantibody; UBE2L3-AAb, ubiquitin-conjugating enzyme 2L3 autoantibody; T1DM, type 1 diabetes mellitus.
aReference range 1 was estimated from the total control subject sample (upper cutoff value was 1.418 for EEF1A1-AAb), bReference range 2 was estimated from control subjects <40 years old (upper cutoff value was 1.595 for EEF1A1-AAb), cReference range 1 was estimated from the total control subject sample (upper cutoff value was 2.512 for UBE2L3-AAb), dReference range 2 was estimated from control subjects <40 years old (upper cutoff value was 2.643 for UBE2L3-AAb), eDifference in the prevalence of autoantibodies according to age group using linear-by-linear association analysis.
This study was conducted in accordance with the Declaration of Helsinki provisions for the participation of subjects in human research. The Institutional Review Board of the Clinical Research Institute at Seoul National University Hospital approved the study protocol. Written informed consent was obtained from each subject.
Enzyme-linked immunosorbent assay
Immunosorbent 96-well plates coated with EEF1A1 or UBE2L3 (Origene Rockville, MD, USA) were incubated with diluted serum samples (1:1,000 to 1:2,500) in phosphate-buffered saline buffer. All experiments were performed in triplicate for each sample. The recombinant proteins and their corresponding antibodies, anti-EEF1A1 (Merck Millipore, Billerica, MA, USA) and anti-UBE2L3 (Santa Cruz Biotechnology, Santa Cruz, CA, USA) were used as positive controls. Autoantibodies bound to the protein were detected with goat anti-human immunoglobulin G-horseradish peroxidase (Jackson ImmunoResearch, West Grove, PA, USA). The experimental assay coefficient of variability (CV) from enzyme-linked immunosorbent assay (ELISA) was calculated using the standard deviation divided by the mean (×100). The range of intra-assay CV was 8.4% to 10.2% for EEF1A1-AAb and 6.8% to 8.1% for UBE2L3-AAb, while the interassay CV was 24.1% for EEF1A1-AAb and 13.3% for UBE2L3-AAb. The cutoff point for the presence or absence of each autoantibody was determined using control subjects and according to the formula [mean absorbance+3 standard deviation]. This corresponded approximately to 99% of the normal controls.
Statistical analysis
All data were analyzed with SPSS version 16 (SPSS Inc., Chicago, IL, USA). The results are presented as the mean±standard deviation. The differences in EEF1A1-AAb and UBE2L3-AAb levels stratified by age group were assessed using analysis of variance (ANOVA) test with post hoc comparisons using Bonferroni's method. A chi-square test was performed to evaluate differences in the proportion of antibody-positive subjects per group. Given the small number of cases in each group, a Mann-Whitney test was used to compare the level of EEF1A1-AAb and UBE2L3-AAb between T1DM subjects and nondiabetic control subjects in each age group.
RESULTS
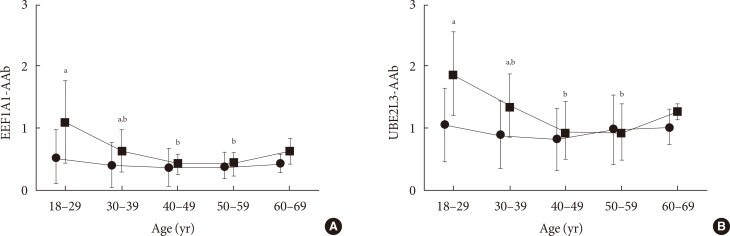
Autoantibodies in the sera of 150 control subjects (mean age, 41±13 years) were profiled. The levels of EEF1A1-AAb and UBE2L3-AAb were calculated using the mean values of two independent ELISA tests and each ELISA test was performed in triplicate. In the nondiabetic control sample, the mean EEF1A1-AAb level was 0.51±0.43 in subjects 18 to 29 years old, 0.39±0.36 in subjects 30 to 39 years old, 0.36±0.31 in subjects 40 to 49 years old, 0.38±0.22 in subjects 50 to 59 years old, and 0.42±0.15 in subjects 60 to 69 years old. There were no significant differences in EEF1A1-AAb levels across age groups (P from ANOVA=0.484 in EEF1A1-AAb) (Fig. 1). The level of UBE2L3-AAb also showed no differences across age groups (1.05±0.58, 0.90±0.55, 0.82±0.49, 0.97±0.56, 1.01±0.27, respectively; P from ANOVA=0.541) (Fig. 1). In addition, there were no differences in EEF1A1-AAb and UBE2L3-AAb levels between control subjects <40 years old and those ≥40 years (P=0.387 and P=0.846, respectively). Significant correlations between age and the level of each autoantibody were not observed among control subjects (r=-0.069, P=0.405 for EEF1A1-AAb; r=-0.005, P=0.951 for UBE2L3-AAb). However, among T1DM subjects, EEF1A1-AAb and UBE2L3-AAb levels were significantly different across age groups (P from ANOVA <0.001 for both antibodies), supporting our previous research [5]. In addition, the levels of both autoantibodies were significantly higher in T1DM patients <40 years old compared to those ≥40 years old (P<0.001 for both autoantibodies). Furthermore, the levels of both autoantibodies in T1DM subjects 18 to 29 years old were significantly higher than all other age groups (P<0.05) except subjects 60 to 69 years old (Fig. 1). We subsequently compared EEF1A1-AAb and UBE2L3-AAb levels between nondiabetic control subjects and T1DM subjects within each age group and found that both autoantibodies were significantly higher in T1DM subjects than control subjects that were 18 to 29 years old and 30 to 39 years old (P<0.001 for both age groups) (Fig. 1).
Fig. 1. The mean levels of (A) eukaryote translation elongation factor 1 alpha 1 autoantibody (EEF1A1-AAb) and (B) ubiquitin-conjugating enzyme 2L3 autoantibody (UBE2L3-AAb) by age group. The mean values with standard deviation in each age group are shown. There were no differences in the levels of EEF1A1-AAb and UBE2L3-AAb across the age groups in control subjects (black circles). However, the levels of both autoantibodies in subjects that were 18 to 29 years old with type 1 diabetes mellitus (T1DM; black squares) were significantly higher than all of the other age groups (P<0.05) except for subjects 60 to 69 years old (black circles, nondiabetic control subjects; black squares, subjects with T1DM). aSignificantly different compared to nondiabetic control subjects in each age group (P<0.05), bSignificantly different compared to 18- to 29-year-old in each disease status (non-diabetic control group and T1DM, respectively; P<0.05).
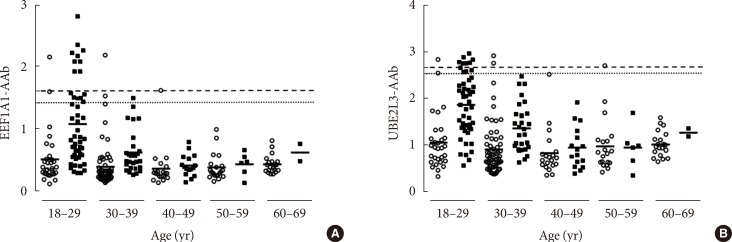
Although there was no variation between age and the mean level of each autoantibody in nondiabetic control subjects, several individual control samples showed significantly high levels of EEF1A1-AAb or UBE2L3-AAb in subjects <40 years old (Fig. 2). Because the reference ranges for both autoantibodies were determined from the standard deviation of the control group, reference range 1 was estimated using the entire control subject sample while the reference range 2 was estimated using only control group subjects <40 years old. Using reference range 1, the prevalence of EEF1A1-AAb was 15.8% (16/101) in T1DM subjects and 3.3% (5/150) in control group subjects (P from chi-square test <0.001). Using reference range 2, the prevalence was lower in both groups: 8.9% (9/101) in the T1DM group and 2.0% (3/150) in the control group (P from chi-square test=0.012). When using reference range 2 among T1DM patients, EEF1A1-AAb was detected only in patients 18 to 29 years old. The prevalence in this age group was 18.4% (9/49) (Table 1). Similar results were found for UBE2L3-AAb. Using reference range 1, the prevalence of UBE2L3-AAb was 10.9% (11/101) in T1DM subjects and 4.0% (6/150) in control subjects (P from chi-square test=0.033), while the prevalence decreased in both groups; i.e., 7.9% (8/101) in the T1DM group and 2.7% (4/150) in the control group, when using reference range 2.
Fig. 2. The level of (A) eukaryote translation elongation factor 1 alpha 1 autoantibody (EEF1A1-AAb) and (B) ubiquitin-conjugating enzyme 2L3 autoantibody (UBE2L3-AAb) in control and type 1 diabetes mellitus (T1DM) samples. Dotted lines represent the [mean absorbance+3×standard deviation] of the total nondiabetic control subject sample; dashed lines represent the [mean absorbance+3×standard deviation] of control subjects <40 years old; simple solid lines in each age group represent the mean level of EEF1A1-AAb and UBE2L3-AAb within the group (white circles, nondiabetic control subjects; black squares, subjects with T1DM).
None of the control subjects with EEF1A1-AAb or UBE2L3-AAb (reference range 1) had GADA. The mean age of control subjects with EEF1A1-AAb or UBE2L3-AAb was 34±10 and 38±13 years, respectively. There was no difference in the mean age between control subjects with these autoantibodies when compared to control subjects without these autoantibodies. All of the control subjects with EEF1A1-AAb (n=5; reference range 1) also had UBE2L3-AAb, a finding consistent with our previous report [5].
DISCUSSION
To examine whether age affects EEF1A1-AAb and UBE2L3-AAb levels in control samples, we compared the levels of both autoantibodies between age-stratified control and T1DM samples. This present study confirmed that there was no direct correlation between the subjects' age and the mean levels of EEF1A1-AAb and UBE2L3-AAb in control subjects. However, we detected the presence of several sporadic nondiabetic subjects with high levels of those autoantibodies in the younger age groups; the prevalence of both EEF1A1-AAb and UBE2L3-AAb in control subjects <40 years old was 4.4%. As there was a wide range of variation in the levels of EEF1A1-AAb and UBE2L3-AAb among young control subjects, reference ranges for those novel autoantibodies were increased for subjects <40 years old. Consequently, the prevalence of EEF1A1-AAb and UBE2L3-AAb decreased in T1DM patients after applying the increased reference range. Nevertheless, the prevalence and the levels of EEF1A1-AAb and UBE2L3-AAb remained higher in the T1DM group compared to the nondiabetic control group. In addition, we confirmed that the levels of EEF1A1-AAb and UBE2L3-AAb were significantly higher in T1DM subjects compared to control subjects even in younger adults <40 years old.
We previously reported that the prevalence of EEF1A1-AAb was 29.5% for T1DM patients and 31.8% in GADA-negative T1DM patients [5], a rate that is comparable to that of ZnT8A in adult T1DM patients in Asian populations [9,10]. However, a wide range of variation in EEF1A1-AAb and UBE2L3-AAb levels in nondiabetic control subjects <40 years old, as observed in this study, suggests a reduced prevalence of these autoantibodies in T1DM patients. In addition, given the very low prevalence of T1DM in the Korean population [11], the observed prevalence of EEF1A1-AAb and UBE2L3-AAb in control subjects <40 years old in this study is relatively high. Thus, to assess the clinical usefulness of these autoantibodies for the detection of T1DM and to determine the reference range cutoff level for nondiabetic control subjects, additional studies with larger control populations <40 years old are needed.
In the case of GADA, which is the most prevalent autoantibody among T1DM subjects, the prevalence in an unaffected population in Scandinavia was 2.4% to 4.7% [12,13,14]. In a similarly unaffected population in the United States, the prevalence was 1.7% to 1.8% [15,16]. Although few studies have examined the association between GADA concentrations and age in the general population, the studies that exist have reported that healthy children had higher GADA levels than healthy adults [15,17], which are findings that are not consistent [18]. The clinical course of unaffected subjects with GADA is also unclear. For example, the Botnia Study reported that subjects with GADA had a significantly higher risk of developing type 2 as well as type 1 diabetes [14]; however, these results are not consistent with the findings of another study [19].
A major limitation of the current study is the absence of follow-up data for control subjects. This limited our ability to define the clinical characteristics of control subjects with EEF1A1-AAb or UBE2L3-AAb. Given that EEF1A1-AAb and UBE2L3-AAb are found in unaffected control subjects <40 years old, long-term follow-up studies on whether these autoantibodies affect glucose metabolism in healthy subjects are needed. Furthermore, EEF1A1 and UBE2L3 have been reported to be associated with other autoimmune diseases such as rheumatoid arthritis [20,21], Crohn disease [22], and systemic lupus erythematosus [23] and both are also expressed in extra-pancreatic tissue [24,25,26,27]. Consequently, additional independent replication studies are needed to further explain the clinical implications of EEF1A1-AAb and UBE2L3-AAb in relation to the diagnosis of T1DM in larger populations with different demographics (e.g., age and ethnicity) as well as the presence of other autoimmune diseases.
In conclusion, there was no association between age and the level of EEF1A1-AAb and UBE2L3-AAb in nondiabetic subjects. However, the level of EEF1A1-AAb and UBE2L3-AAb varied widely among control subjects <40 years old and should be considered an important factor when determining the reference range for these autoantibodies.
ACKNOWLEDGMENTS
This work was supported by NRF (No. 20110025320), the Seoul National University Hospital Research Fund (04-2012-0680 and 30-2011-0100), and 2015 Research Grant from Kangwon National University. We thank Young Min Cho, Hye Seung Jung, Hak C. Jang, Sung Hee Choi, and Young Joo Park for their assistance with the collection of serum samples.
Footnotes
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.
References
- 1.Verge CF, Gianani R, Kawasaki E, Yu L, Pietropaolo M, Jackson RA, Chase HP, Eisenbarth GS. Prediction of type I diabetes in first-degree relatives using a combination of insulin, GAD, and ICA512bdc/IA-2 autoantibodies. Diabetes. 1996;45:926–933. doi: 10.2337/diab.45.7.926. [DOI] [PubMed] [Google Scholar]
- 2.Greenbaum CJ, Sears KL, Kahn SE, Palmer JP. Relationship of beta-cell function and autoantibodies to progression and nonprogression of subclinical type 1 diabetes: follow-up of the Seattle Family Study. Diabetes. 1999;48:170–175. doi: 10.2337/diabetes.48.1.170. [DOI] [PubMed] [Google Scholar]
- 3.Zampetti S, Capizzi M, Spoletini M, Campagna G, Leto G, Cipolloni L, Tiberti C, Bosi E, Falorni A, Buzzetti R NIRAD Study Group. GADA titer-related risk for organ-specific autoimmunity in LADA subjects subdivided according to gender (NIRAD study 6) J Clin Endocrinol Metab. 2012;97:3759–3765. doi: 10.1210/jc.2012-2037. [DOI] [PubMed] [Google Scholar]
- 4.Jin P, Huang G, Lin J, Yang L, Xiang B, Zhou W, Zhou Z. High titre of antiglutamic acid decarboxylase autoantibody is a strong predictor of the development of thyroid autoimmunity in patients with type 1 diabetes and latent autoimmune diabetes in adults. Clin Endocrinol (Oxf) 2011;74:587–592. doi: 10.1111/j.1365-2265.2011.03976.x. [DOI] [PubMed] [Google Scholar]
- 5.Koo BK, Chae S, Kim KM, Kang MJ, Kim EG, Kwak SH, Jung HS, Cho YM, Choi SH, Park YJ, Shin CH, Jang HC, Shin CS, Hwang D, Yi EC, Park KS. Identification of novel autoantibodies in type 1 diabetic patients using a high-density protein microarray. Diabetes. 2014;63:3022–3032. doi: 10.2337/db13-1566. [DOI] [PubMed] [Google Scholar]
- 6.Decochez K, Tits J, Coolens JL, Van Gaal L, Krzentowski G, Winnock F, Anckaert E, Weets I, Pipeleers DG, Gorus FK. High frequency of persisting or increasing islet-specific autoantibody levels after diagnosis of type 1 diabetes presenting before 40 years of age. The Belgian Diabetes Registry. Diabetes Care. 2000;23:838–844. doi: 10.2337/diacare.23.6.838. [DOI] [PubMed] [Google Scholar]
- 7.Powell M, Prentice L, Asawa T, Kato R, Sawicka J, Tanaka H, Petersen V, Munkley A, Morgan S, Rees Smith B, Furmaniak J. Glutamic acid decarboxylase autoantibody assay using 125I-labelled recombinant GAD65 produced in yeast. Clin Chim Acta. 1996;256:175–188. doi: 10.1016/s0009-8981(96)06422-4. [DOI] [PubMed] [Google Scholar]
- 8.Torn C, Mueller PW, Schlosser M, Bonifacio E, Bingley PJ Participating Laboratories. Diabetes Antibody Standardization Program: evaluation of assays for autoantibodies to glutamic acid decarboxylase and islet antigen-2. Diabetologia. 2008;51:846–852. doi: 10.1007/s00125-008-0967-2. [DOI] [PubMed] [Google Scholar]
- 9.Lampasona V, Petrone A, Tiberti C, Capizzi M, Spoletini M, di Pietro S, Songini M, Bonicchio S, Giorgino F, Bonifacio E, Bosi E, Buzzetti R Non Insulin Requiring Autoimmune Diabetes (NIRAD) Study Group. Zinc transporter 8 antibodies complement GAD and IA-2 antibodies in the identification and characterization of adult-onset autoimmune diabetes: Non Insulin Requiring Autoimmune Diabetes (NIRAD) 4. Diabetes Care. 2010;33:104–108. doi: 10.2337/dc08-2305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Yang L, Luo S, Huang G, Peng J, Li X, Yan X, Lin J, Wenzlau JM, Davidson HW, Hutton JC, Zhou Z. The diagnostic value of zinc transporter 8 autoantibody (ZnT8A) for type 1 diabetes in Chinese. Diabetes Metab Res Rev. 2010;26:579–584. doi: 10.1002/dmrr.1128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.DIAMOND Project Group. Incidence and trends of childhood type 1 diabetes worldwide 1990-1999. Diabet Med. 2006;23:857–866. doi: 10.1111/j.1464-5491.2006.01925.x. [DOI] [PubMed] [Google Scholar]
- 12.Kulmala P, Rahko J, Savola K, Vahasalo P, Sjoroos M, Reunanen A, Ilonen J, Knip M. Beta-cell autoimmunity, genetic susceptibility, and progression to type 1 diabetes in unaffected schoolchildren. Diabetes Care. 2001;24:171–173. doi: 10.2337/diacare.24.1.171-a. [DOI] [PubMed] [Google Scholar]
- 13.Samuelsson U, Sundkvist G, Borg H, Fernlund P, Ludvigsson J. Islet autoantibodies in the prediction of diabetes in school children. Diabetes Res Clin Pract. 2001;51:51–57. doi: 10.1016/s0168-8227(00)00199-6. [DOI] [PubMed] [Google Scholar]
- 14.Lundgren VM, Isomaa B, Lyssenko V, Laurila E, Korhonen P, Groop LC, Tuomi T Botnia Study Group. GAD antibody positivity predicts type 2 diabetes in an adult population. Diabetes. 2010;59:416–422. doi: 10.2337/db09-0747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zimmet PZ, Shaten BJ, Kuller LH, Rowley MJ, Knowles WJ, Mackay IR. Antibodies to glutamic acid decarboxylase and diabetes mellitus in the Multiple Risk Factor Intervention Trial. Am J Epidemiol. 1994;140:683–690. doi: 10.1093/oxfordjournals.aje.a117316. [DOI] [PubMed] [Google Scholar]
- 16.Barinas-Mitchell E, Kuller LH, Pietropaolo S, Zhang YJ, Henderson T, Pietropaolo M. The prevalence of the 65-kilodalton isoform of glutamic acid decarboxylase autoantibodies by glucose tolerance status in elderly patients from the cardiovascular health study. J Clin Endocrinol Metab. 2006;91:2871–2877. doi: 10.1210/jc.2005-2667. [DOI] [PubMed] [Google Scholar]
- 17.Kordonouri O, Hartmann R, Gruters-Kieslich A, Knip M, Danne T. Age-specific levels of diabetes-related GAD and IA-2 antibodies in healthy children and adults. J Pediatr Endocrinol Metab. 2002;15:47–52. doi: 10.1515/jpem.2002.15.1.47. [DOI] [PubMed] [Google Scholar]
- 18.Batstra MR, van Driel A, Petersen JS, van Donselaar CA, van Tol MJ, Bruining GJ, Grobbee DE, Dyrberg T, Aanstoot HJ. Glutamic acid decarboxylase antibodies in screening for autoimmune diabetes: influence of comorbidity, age, and sex on specificity and threshold values. Clin Chem. 1999;45:2269–2272. [PubMed] [Google Scholar]
- 19.Dabelea D, Ma Y, Knowler WC, Marcovina S, Saudek CD, Arakaki R, White NH, Kahn SE, Orchard TJ, Goldberg R, Palmer J, Hamman RF Diabetes Prevention Program Research Group. Diabetes autoantibodies do not predict progression to diabetes in adults: the Diabetes Prevention Program. Diabet Med. 2014;31:1064–1068. doi: 10.1111/dme.12437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ditzel HJ, Masaki Y, Nielsen H, Farnaes L, Burton DR. Cloning and expression of a novel human antibody-antigen pair associated with Felty's syndrome. Proc Natl Acad Sci U S A. 2000;97:9234–9239. doi: 10.1073/pnas.97.16.9234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Orozco G, Eyre S, Hinks A, Bowes J, Morgan AW, Wilson AG, Wordsworth P, Steer S, Hocking L, Thomson W, Worthington J, Barton A UKRAG consortium. Study of the common genetic background for rheumatoid arthritis and systemic lupus erythematosus. Ann Rheum Dis. 2011;70:463–468. doi: 10.1136/ard.2010.137174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Fransen K, Visschedijk MC, van Sommeren S, Fu JY, Franke L, Festen EA, Stokkers PC, van Bodegraven AA, Crusius JB, Hommes DW, Zanen P, de Jong DJ, Wijmenga C, van Diemen CC, Weersma RK. Analysis of SNPs with an effect on gene expression identifies UBE2L3 and BCL3 as potential new risk genes for Crohn's disease. Hum Mol Genet. 2010;19:3482–3488. doi: 10.1093/hmg/ddq264. [DOI] [PubMed] [Google Scholar]
- 23.International Consortium for Systemic Lupus Erythematosus Genetics (SLEGEN) Harley JB, Alarcon-Riquelme ME, Criswell LA, Jacob CO, Kimberly RP, Moser KL, Tsao BP, Vyse TJ, Langefeld CD, Nath SK, Guthridge JM, Cobb BL, Mirel DB, Marion MC, Williams AH, Divers J, Wang W, Frank SG, Namjou B, Gabriel SB, Lee AT, Gregersen PK, Behrens TW, Taylor KE, Fernando M, Zidovetzki R, Gaffney PM, Edberg JC, Rioux JD, Ojwang JO, James JA, Merrill JT, Gilkeson GS, Seldin MF, Yin H, Baechler EC, Li QZ, Wakeland EK, Bruner GR, Kaufman KM, Kelly JA. Genome-wide association scan in women with systemic lupus erythematosus identifies susceptibility variants in ITGAM, PXK, KIAA1542 and other loci. Nat Genet. 2008;40:204–210. doi: 10.1038/ng.81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ciechanover A. The ubiquitin-proteasome pathway: on protein death and cell life. EMBO J. 1998;17:7151–7160. doi: 10.1093/emboj/17.24.7151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Knudsen SM, Frydenberg J, Clark BF, Leffers H. Tissue-dependent variation in the expression of elongation factor-1 alpha isoforms: isolation and characterisation of a cDNA encoding a novel variant of human elongation-factor 1 alpha. Eur J Biochem. 1993;215:549–554. doi: 10.1111/j.1432-1033.1993.tb18064.x. [DOI] [PubMed] [Google Scholar]
- 26.Chambers DM, Peters J, Abbott CM. The lethal mutation of the mouse wasted (wst) is a deletion that abolishes expression of a tissue-specific isoform of translation elongation factor 1alpha, encoded by the Eef1a2 gene. Proc Natl Acad Sci U S A. 1998;95:4463–4468. doi: 10.1073/pnas.95.8.4463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lee S, Francoeur AM, Liu S, Wang E. Tissue-specific expression in mammalian brain, heart, and muscle of S1, a member of the elongation factor-1 alpha gene family. J Biol Chem. 1992;267:24064–24068. [PubMed] [Google Scholar]